Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Seems like they have $42million left until they advance in their trials in 2022, then I expect a big offering for funding.
Once GNPX is bankrupt, the Company officials won't be, they will more than likely wind up at new companies and do the very same things?
Taking past failed products, tweaking them and sending them back though trials while obtaining an empty shell and diluting it to nothing?
this is so much bigger than Genprex?
IMO
New O/S 47,808,570 not as many diluted as I suspected but that 72 million dollar deficit is a huge issue?
IMO
They obtain shells, sell millions of shares, file bankruptcy then move on to another shell?
One would have to imagine, a federal agency has to be backing this possible corruption?
Who would allow blatant theft like this could be?
Over and over again?
IMO
Why do these groups of Officials jump from Bio penny stock to Bio penny stock and do nothing but dilute?
They never have a viable product?
They take shelved products, tweak them and send them back through the process, the whole time doing nothing but selling shares?
How does the SEC or the FBI not see the same individuals doing the same things over and over again?
when is jail?
IMO
Stop hiding your current O/S count?
Show people what you are up to?
Transparency?
IMO
There might be an internal group of people who obtain shells get already failed or shelved products, tweak them and create an entire path of dilution based off of re failure?
How many times can they fail over and over at the expense of shareholders?
It's OK to fail, it's not ok to steal?
Look at the history of the people involved in Genprex?
Have they ever had successful in PKs?
GNPX dilution will only grow stronger as the O/S continues to increase?
Right Mike?
IMO
New O/S 47,363,729 Dilution of almost 20 million and that is as of 5/10/21?
No Telling what Genprex has diluted since?
This is madness to allow some of these shells to come in sell hundreds of millions of shares have zero results and just file for bankruptcy?
This should be investigated by a government agency?
IMO
There is a reason they will not report the current O/S as of today?
Just look at the chart?
Why do they allow these pharma bros to fleece the public while diluting hundreds of millions of dollars?
IMO
New O/S dilution up 43,276,764 that was as of 3/15 though?
Probably lots of million more diluted since?
IMO
Genprex's Reqorsa combo shows encouraging activity in lung cancer, shares up 3%
GNPX has not reported the O/S since November 2020?
How many hundreds if millions did they dilute since then?
Anyone belive it's still 39 million?
Not a chance?
IMO
Soon 10 plus
National Securities just raised target to 7
New O/S bumped to 43 million up from 39? It's only 16 million of dilution on the backs of shareholders?
So in just over a month GNPX has bled 16 million out of the O/S?
More to come?
Much more dilution??
Stay Tuned?
IMO
Yea, good luck with that. Finally taking off here. :)
Trying to generate cheap shares!!!
$GNPX
what are you talking about? toxic funding, no way, that was a smart raise
12 million of toxic funding?
this is just the begging, GNPX could mount endless toxic funding and dilution while it possible sinks into oblivion?
Keep an eye on that O/S? They have to report it now, cant hide anymore?
2021 is going to be nausiating for Investors?
IMO
Same BS Oncolix and Redman did with BioVectra? This is the exact same pattern these gentleman use to produce funding through toxic dilution?
There will be drops and rises over the next 2 years but ultimately GNPX seems to be following the same pattern as the past shell scams?
Did they take a shelved product tweak it and send it back through the process just to sell shares and make millions rich off the backs of investors?
Don't know, but to say some of the members of this team have not done this in the past, knowing or not knowing they were scamming, would be idiocy?
IMO
O/S up yet again, dilution really starting to thrive here at the possible bogus bio shell?
Shares Out
39,618,722
Show em what you're good at GNPX?
IMO
O/S up another million? Whats a little dilution among friends?
ONCX like a dilution machine just vomiting loses all over Investors?
IMO
Oh look..... our local short is back.
3 months late filings and millions in dilution?
Great plan?
IMO
JUST IN: $GNPX Genprex Receives Conditional FDA Acceptance of Proprietary Name REQORSA(TM) for Lead Drug Candidate for Treatment of Non-Small Cell Lung Cancer
Approval of new name marks important branding milestone and aligns with the program’s overall progress REQORSA immunogene therapy uses the company’s unique, proprietary ONCOPREX™ nanoparticle delivery system to deliver cancer-fighting genes FDA gra...
Read the whole news GNPX - Genprex Receives Conditional FDA Acceptance of Proprietary Name REQORSA(TM) for Lead Drug Candidate for Treatment of Non-Small Cell Lung Cancer
* * $GNPX Video Chart 10-29-2020 * *
Link to Video - click here to watch the technical chart video
Glad I own this one. Making me $$$
$GNPX
This group GNPX BOD are stock destroyers look at the shells they have been involved with?
They take shelved products recirculate them through the system with a failed tweek, obtain a shell and dikute every last penny?
John Varner aka Rodney Varner aka J R Varner aka John R Varner thinks its clever to change names on multiple SEC related docs?
Taking half a million dollars a year from where? Who is paying for Varner and the Tulip Ryan Confer?
IMO
What's disgusting is the short scare tactics being used here to gain discounted shares. Bankruptcy is shorts future.
IMO
Lets remind the Board J Rodney Varner aka Rodney Varner aka Rodney J Varner aka whatever has increased his salary by a BOD vote?
I'd like to see the vote results with all the board members and how they voted?
I know redman probably voted yea, that guy did the same crap with Oncolix before it went belly up?
This is disgusting GNPX?
IMO
480,000 grand!! J Rodney Varner steals from shareholders and calls it a salary?
What a trashy thing to do?
Wheres the 480k salary come from in a business that doesn't produce a dime?
Scam?
Fraud?
Theft?
Disgraceful?
Produce somthing besides dilution chief?
IMO
Oh look. Our local short is back. Hopefully he's worried and very short. Bankruptcy is in his future
only about 20 plus million diluted in the last few months?
O/S bumped a modest 6 million shares?
Wonder what GNPX is doing with ther newly acquired 20 million dollar of stolen shareholder value?
Enjoy is J Ruddy, we know there is more to come?
Keep the Diltuion coming?
Outstanding Shares
38,870,704
08/07/2020
IMO
Nope that was last week he posted !!
Amigo Zride is amateur and a little pocket investor !
Guess you were WRONG
News: $GNPX 4 Penny Stocks To Buy Right Now According To Analysts
These Names Are On Analysts’ Lists Of Penny Stocks Right Now; Are They On Yours? Penny stocks aren’t just “short-term” movers or “lotto trades” as some might assume. If you’ve been a reader of our articles this year, you have come to learn tha...
Find out more GNPX - 4 Penny Stocks To Buy Right Now According To Analysts
* * $GNPX Video Chart 08-14-2020 * *
Link to Video - click here to watch the technical chart video
Classic bullshit from our amigo Zride fake
Pushing luck with four days of green . Correction on deck. Might be a good idea this time to take some profit before exits get too crowded. When this thing goes up whether or not on news or no (FNMA) news it will always pullback .
Back below $2 by Wednesday.
News: $GNPX How Africa Plans to Create Its Own Medical Technology
Some technological advancements, such as biomedical engineering, can be used in saving lives because the biomedical engineering field draws knowledge from different disciplines such as computer science, public health, clinical practice and biomedical sciences. The information from these four...
Find out more GNPX - How Africa Plans to Create Its Own Medical Technology
|
Followers
|
72
|
Posters
|
|
|
Posts (Today)
|
2
|
Posts (Total)
|
3909
|
|
Created
|
04/27/18
|
Type
|
Free
|
| Moderators | |||

Our technologies are designed to administer disease-fighting genes to provide new therapies for large patient populations with cancer and diabetes who currently have limited treatment options.
We are developing gene therapies for patients with cancer and diabetes to bring new therapies to large patient populations. Our lead product candidate, Reqorsa® Immunogene Therapy, is designed to be administered in combination with other therapies, including targeted therapies and immunotherapies, for non-small cell lung cancer and small cell lung cancer.
We have assembled a multidisciplinary team of executives and advisors with broad business experience in the biotech and pharmaceutical industries, and research and clinical experience at preeminent medical and academic institutions around the world. Our management team pairs an unmatched expertise with a knack for innovation in research.
At Genprex, we are committed to developing life-changing gene therapies for patients afflicted with cancer and diabetes. We are dedicated in our efforts to helping patients with limited or no treatment options to have access to new and advanced therapies to more effectively treat their disease.
Ryan Confer Appointed Genprex President and CEO and to its Board of Directors
AUSTIN, Texas — (May. 1, 2024) — Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a clinical-stage gene therapy company focused on developing life-changing therapies for patients with cancer and diabetes, today announced the Company’s participation in the following upcoming investor and industry conferences to be held in May 2024.
Compelling Data Validates the Potential of Reqorsa® Immunogene Therapy and the Oncoprex® Delivery System as Innovative Cancer Treatments
Multiple clinical trial sites to be opened under collaboration with large network of community-based oncology practices
Genprex, Inc. is a clinical-stage gene therapy company focused on developing life-changing therapies for patients with cancer and diabetes. Genprex’s technologies are designed to administer disease-fighting genes to provide new therapies for large patient populations with cancer and diabetes who currently have limited treatment options. Genprex works with world-class institutions and collaborators to develop drug candidates to further its pipeline of gene therapies in order to provide novel treatment approaches. Genprex’s oncology program utilizes its proprietary, non-viral ONCOPREX® Nanoparticle Delivery System, which the Company believes is the first systemic gene therapy delivery platform used for cancer in humans. ONCOPREX encapsulates the gene-expressing plasmids using lipid nanoparticles. The resultant product is administered intravenously, where it is then taken up by tumor cells that express tumor suppressor proteins that are deficient in the body. The Company’s lead product candidate, REQORSA™ (quaratusugene ozeplasmid), is being evaluated as a treatment for non-small cell lung cancer (NSCLC) (with each of these clinical programs receiving a Fast Track Designation from the Food and Drug Administration) and for small cell lung cancer. Genprex’s diabetes gene therapy approach is comprised of a novel infusion process that uses an endoscope and an adeno-associated virus (AAV) vector to deliver Pdx1 and MafA genes to the pancreas. In models of Type 1 diabetes, the genes express proteins that transform alpha cells in the pancreas into functional beta-like cells, which can produce insulin but are distinct enough from beta cells to evade the body’s immune system. In Type 2 diabetes, where autoimmunity is not at play, it is believed that exhausted beta cells are also rejuvenated and replenished.
We are committed to fighting cancer and diabetes by continuing to develop unique and innovative gene therapies.
Our pioneering technologies are at the forefront of gene therapy and we stand behind our high-quality technology platform.
We have integrity and high ethical standards, and we believe in responsibly transforming the lives of those affected with cancer and diabetes.
Our commitment, excellence in technology, and integrity behind what we do enables us to be trailblazers in the effort to change the lives of patients afflicted by cancer and diabetes.
We have assembled a multidisciplinary team of executives and advisors with broad business experience in the biotech and pharmaceutical industries, and research and clinical experience at preeminent medical and academic institutions around the world. Our management team pairs an unmatched expertise with a knack for innovation in research.
Our lead drug candidate, REQORSA® Immunogene Therapy (quaratusugene ozeplasmid) for non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), is designed to interrupt cell signaling pathways that cause replication and proliferation of cancer cells, target and kill cancer cells, and stimulate the natural immune responses against cancer. REQORSA is an immunogene therapy in that it combines features of gene therapy and immunotherapy. It up-regulates TUSC2 expression in the cell, and also increases the anti-tumor immune cell population and down-regulates PD-L1, thereby potentially boosting the immune response to cancer.
In 2020, the FDA granted Fast Track Designation for REQORSA in combination with AstraZeneca’s Tagrisso® (osimertinib) in late-stage NSCLC patients with EFGR mutations whose tumors progressed after treatment with Tagrisso. In 2021, the FDA granted Fast Track Designation for REQORSA in combination with Merck & Co’s Keytruda® (pembrolizumab) in late-stage NSCLC patients whose disease progressed after treatment with Keytruda. In 2023, the FDA granted Fast Track Designation for REQORSA in combination with Genentech, Inc.’s Tecentriq® in patients with extensive-stage small cell lung cancer (ES-SCLC) who did not develop tumor progression after receiving Tecentriq and chemotherapy as initial standard treatment. The FDA also granted Orphan Drug Designation for REQORSA for the treatment of SCLC.
REQORSA consists of the TUSC2 gene expressing plasmid encapsulated in non-viral nanoparticles made from lipid molecules (our ONCOPREX® Nanoparticle Delivery System) with a positive electrical charge. REQORSA is injected intravenously and specifically targets cancer cells, which generally have a negative electrical charge. REQORSA is designed to deliver the functioning TUSC2 gene to cancer cells while minimizing their uptake by normal tissue.
Tumor biopsy studies conducted at MD Anderson show that, in three patients, the uptake of TUSC2 in tumor cells after REQORSA treatment was 10 to 33 times the uptake in normal cells. We believe that REQORSA is the first systemic gene therapy to be used for cancer in humans.
Unlike many other gene therapies, REQORSA is administered intravenously and it does not need to integrate into the patient’s DNA. Many other gene therapies require complex procedures that result in permanent changes in a patient’s DNA, including the removal of cells from a patient and the modification of those cells which are then reinfused into the patient.
Multimodal Mechanism of Action
Many approved cancer therapeutics target only single molecules or a single specific genetic abnormality related to driving the proliferation and survival of cancer cells. In contrast, REQORSA has been shown to have a multimodal mechanism of action whereby it interrupts cell signaling pathways that cause replication and proliferation of cancer cells, re-establishes pathways for programmed cell death (apoptosis) in cancer cells, and modulates the immune response against cancer cells. REQORSA also has been shown to be complementary with targeted drugs and immunotherapies.

Overcoming Drug Resistance
Resistance to targeted drugs and checkpoint inhibitors develop through activation of alternate bypass pathways. For example, when PD-1 is blocked, the TIM-3 checkpoint is up-regulated. We believe that REQORSA’s multimodal activity will block emerging bypass pathways, thereby potentially reducing the probability that drug resistance develops.
Combination Therapies
Our preclinical and clinical data indicate that REQORSA is well tolerated and may be effective alone or in combination with targeted small molecule therapies. Preclinical data indicate that REQORSA may also be effective with immunotherapies, and in a three-drug combination with immunotherapy and chemotherapy. These data suggest that REQORSA, when combined with other therapies, may be effective in a large population of lung cancer patients.
To learn more about scientific evidence and studies supporting REQORSA and the TUSC2 gene, please refer to our Clinical Trials and TUSC2 Bibliography pages.
In diabetes, we have exclusively licensed from the University of Pittsburgh multiple technologies relating to the development of a gene therapy product for each of Type 1 and Type 2 diabetes.
The same general novel approach is used in each of Type 1 and Type 2 whereby an adeno-associated virus (AAV) vector containing the Pdx1 and MafA genes is administered directly into the pancreatic duct. In humans, this can be done with a routine endoscopy procedure. Our diabetes product candidates are currently being evaluated and optimized in preclinical studies at the University of Pittsburgh.
GPX-002 is designed to work in Type 1 diabetes by transforming alpha cells in the pancreas into functional beta-like cells, which can produce insulin but may be distinct enough from beta cells to evade the body’s immune system.

GPX-002 has been tested in vivo in mice and nonhuman primates. Earlier studies in diabetic mouse models showed that an earlier version of GPX-002 restored normal blood glucose levels for an extended period of time, which lasted approximately four months, and markedly increased the mass of insulin producing beta cells. According to the researchers, the duration of restored blood glucose levels in mice could potentially translate to decades in humans.
In Type 2 diabetes, GPX-003 is believed to work by replenishing and rejuvenating the beta cells that make insulin.
In August 2022, we entered into a one-year sponsored research agreement with the University of Pittsburgh for the use of GPX-003 in a non-human primate (NHP) model in Type 2 diabetes.
In February 2023, the Company’s research collaborators at the University of Pittsburgh presented preclinical data in a NHP model of Type 1 diabetes highlighting the therapeutic potential of GPX-002 at the 16th International Conference on Advanced Technologies & Treatments for Diabetes (ATTD 2023) in Berlin, Germany. The statistically significant study results showed that after infusion of the AAV engineered construct all eight of the NHPs had:
We believe the data in NHPs demonstrate the potential for this gene therapy treatment to eliminate the need for insulin replacement therapy for Type 1 and Type 2 diabetic patients.
To learn more about scientific evidence and studies supporting GPX-002, GPX-003 and the Pdx1/MafA genes, please refer to our Clinical Trials and Pdx1/MafA Bibliography pages.
Our oncology platform utilizes our non-viral Oncoprex® Nanoparticle Delivery System. Using this system, anti-cancer genes expressing DNA plasmids contained in lipid nanoparticles are delivered intravenously to the patient. This platform, originally developed through collaborative research between the University of Texas MD Anderson Cancer Center and the National Institutes of Health, has been optimized to work with our initial drug candidate, Reqorsa® Immunogene Therapy (quaratusugene ozeplasmid).
REQORSA® utilizes the ONCOPREX® Nanoparticle Delivery System to encapsulate the TUSC2 gene in positively charged nanoparticles that bind to negatively charged cancer cells, and then enter the cancer cell through selective endocytosis, a process by which cells take in substances from outside the cell by engulfing them in a vesicle. The nanoparticles in our system differ significantly from liposomes historically used for drug delivery in that they are true particles encapsulating the therapeutic payload within a bilamellar lipid coat.
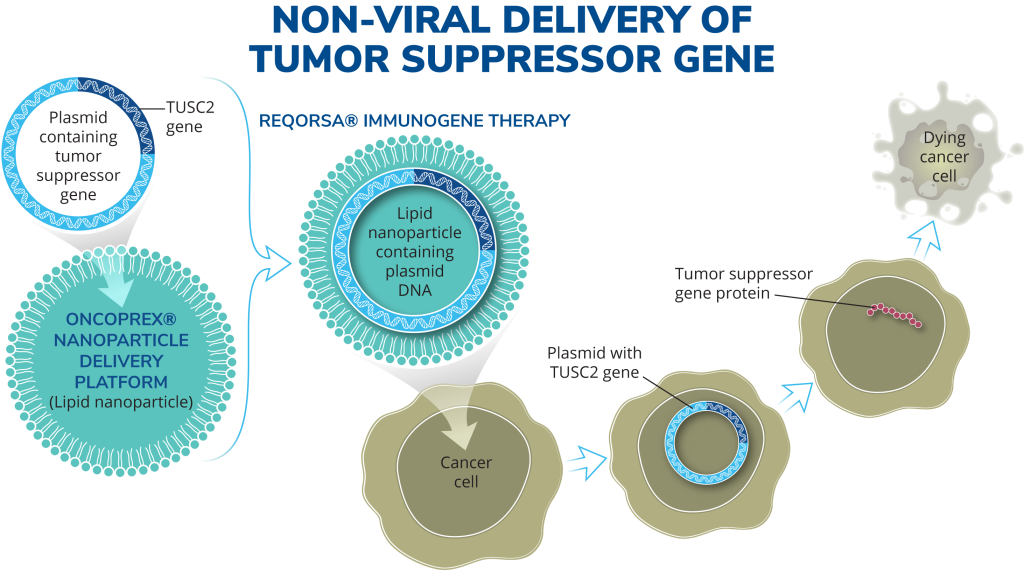
Our systemic, nanoparticle, non-viral delivery system, which is being used in our clinical trials for the treatment of non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), is designed to be small enough to cross tight barriers in the lung, but large enough to avoid accumulation in the liver, spleen and kidney. The cationic (positive) charge of the nanoparticles target cancer cells. A Phase 1 clinical trial showed that intravenous REQORSA therapy selectively and preferentially targeted tumor cells, resulting in anticancer activity. The nanoparticles are non-immunogenic, allowing repetitive therapeutic dosing and providing extended half-life in the circulation.
In mice studies, the nanoparticles have been shown to be taken up by tumor cells after REQORSA administration at 10 to 33 times the rate they are taken up by normal cells.
We have administered REQORSA to more than 50 patients in Phase 1 and 2 clinical trials using our systemic, proprietary, ONCOPREX non-viral delivery system.
A Phase 1 clinical trial showed that systemic, intravenous REQORSA therapy using the ONCOPREX Nanoparticle Delivery System selectively and preferentially targeted tumor cells, resulting in clinically significant anticancer activity. The nanoparticles are non-immunogenic, allowing repetitive therapeutic dosing and providing extended half-life in the circulation.
Our earlier clinical trials have also shown that REQORSA therapy using the ONCOPREX Nanoparticle Delivery System is well tolerated in humans and can be delivered at high therapeutic doses. We believe the ONC-001 clinical trial was the first systemic gene therapy clinical trial using a nanoparticle delivery system to deliver a tumor suppressor gene.
Genprex’s Chief Medical Officer discusses the Company’s collaboration with a large network of community-based oncology practices to open additional sites for the Acclaim-3 clinical trial in SCLC
The KOL event, “Bringing Gene Therapy to the Fight Against Lung Cancers,” features discussions by Alexander I. Spira, MD, PhD, FACP; Daniel Morgensztern, MD; and Mark S. Berger, MD, Chief Medical Officer at Genprex, highlighting REQORSA® as a potential treatment for both NSCLC and SCLC.
Innovators with Jane King profiles tech companies and thought leaders in the space. Genprex (NASDAQ: GNPX): is focused on developing life-changing therapies for cancer & diabetes patients.
In this exclusive Global One Media interview, Genprex, Inc. (NASDAQ: GNPX) Chief Financial Officer, Ryan M. Confer, delves into Genprex’s groundbreaking work so far in clinical-stage gene therapy.
Video Highlights Validation of the ONCOPREX® Nanoparticle Delivery System with a Second Tumor Suppressor Gene
Watch Genprex’s KOL event, titled, “Groundbreaking Data From Preclinical Study Reported at ATTD 2023: Novel Gene Therapy To Treat Type 1 Diabetes.” This discussion is moderated by Genprex’s Chief Medical Officer, Mark Berger, M.D., and the data is presented by George Gittes, M.D., Professor of Surgery and Pediatrics and Chief of the Division of Pediatric Surgery at the University of Pittsburgh School of Medicine.
Genprex, Inc. CEO Rodney Varner tells Proactive the FDA has granted Fast Track Designation to the company’s lead drug candidate, REQORSA Immunogene Therapy, in combination with Merck & Co Inc (NYSE:MRK)’s Keytruda in patients with stage III or IV non-small cell lung cancer (NSCLC), where disease progressed after treatment with Keytruda.
Note: When you click on media articles below, a new page will open indicating that you are leaving the Genprex, Inc. website and entering a third-party website not affiliated with Genprex Inc. or any of its affiliates. No information contained in a linked site has been endorsed or approved by Genprex, Inc. and Genprex, Inc. is not responsible for the content of such third-party websites.
Genprex’s Chief Financial Officer, Ryan Confer, shares his insight on partnering with academic institutions, the benefits, and best practices
Diabetes is the most expensive chronic illness in the U.S., with $1 of every $4 in healthcare costs going to care for diabetics. The National Center for Chronic Disease Prevention and Health Promotion (NCCDPHP) estimates that $237 billion is spent directly on diabetes-related medical costs every year, while another $90 billion is lost each year due to reduced productivity associated with diabetes.
Bell2Bell’s latest podcast features Chairman, President and CEO Rodney Varner and CFO Ryan Confer of Genprex, Inc. (NASDAQ: GNPX), a clinical-stage gene therapy company focused on developing life-changing therapies for patients living with cancer and diabetes. In this interview, Varner and Confer discuss the journeys that led them to Genprex and the promise of the company’s drug development pipeline.
Bell2Bell’s latest podcast features Dr. Mark Berger, Chief Medical Officer of Genprex Inc. (NASDAQ: GNPX), a clinical-stage gene therapy company focused on developing life-changing therapies for patients living with cancer and diabetes.
On the heels of the release of recent positive safety and early efficacy data related to the phase 1 portion of its Acclaim-1 phase 1/2 clinical trial for non-small cell lung cancer evaluating REQORSA in combination with Tagrisso, Genprex has released a new patient video featuring an NSCLC patient from the phase 1 portion of its Acclaim-1.
Genprex Inc., a clinical-stage gene therapy company, is focused on developing for these patients with serious medical conditions and unmet need new and better treatment options that are life-changing.
Reprogramming the course of cancer and diabetes, Austin-based Genprex is on the cutting edge of technologies to address the biggest health concerns
Genprex, a clinical-stage gene therapy company, has recently released groundbreaking data from a non-human primate study evaluating a novel gene therapy to treat Type 1 diabetes.
With results of an animal study published in February 2023, Genprex aims to add credibility to another genome editing approach in the hunt for a functional cure to T1D.
How one company seeks to provide hope to cancer patients through more effective treatments.
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
