Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Prior to September 15, 2023, the Series A Preferred shares of Enzolytics (that is, the Enzolytics shares with voting control) were owned as follows:
Zhabilov Trust - 18,900,000 shares - 31.50% of shares
Charles Cotropia - 14,917,500 shares - 24.86% of shares
Joseph Cotropia - 14,917,500 shares - 24.86% of shares
Gaurav Chandra - 5,265,000 shares - 8.78% of shares
Phil Phelizian - 6,000,000 shares - 10.00% of shares
source:
https://www.otcmarkets.com/otcapi/company/financial-report/387028/content (pages 28 - 29)
On September 15, 2023, Enzolytics closed the Business Combination Agreement with SAGA for the sale of VIRO and BGEN to SAGA. To facilitate the SAGA transaction, Charles Cotropia, Joseph Cotropia, and Guarav Chandra surrendered all of their Series A Preferred shares, and the Zhabilov Trust surrendered 11,700,000 of its Series A Preferred shares. As a result, the Series A Preferred shares of Enzolytics (that is, the Enzolytics shares with voting control) were owned as
follows:
Zhabilov Trust - 7,200,000 shares - 54.55% of shares
Phil Phelizian - 6,000,000 shares - 45.45% of shares
source:
https://www.otcmarkets.com/otcapi/company/financial-report/387028/content (page 42)
According to my Enzolytics SAFE (Simple Agreement for Future Equity), I am supposed to receive Enzolytics Common Stock if a "change of control" occurs. The SAFE defines "change of control" as follows:
“change of control” means (i) a transaction or series of related transactions in which any “person” or “group” (within the meaning of Section 13(d) and 14(d) of the Securities Exchange Act of 1934, as amended), becomes the “beneficial owner” (as defined in Rule 13d-3 under the Securities Exchange Act of 1934, as amended), directly or indirectly, of more than 50% of the outstanding voting securities of the Company having the right to vote for the election of members of the Company’s board of directors, (ii) any reorganization, merger or consolidation of the Company, other than a transaction or series of related transactions in which the holders of the voting securities of the Company outstanding immediately prior to such transaction or series of related transactions retain, immediately after such transaction or series of related transactions, at least a majority of the total voting power represented by the outstanding voting securities of the Company or such other surviving or resulting entity or (iii) a sale, lease or other disposition of all or substantially all of the assets of the Company.
Given the above information about who owns how many shares, it seems that, on September 15, 2023, the "group" known as the Zhabilov Trust became the "beneficial owner" of more than 50% of the outstanding voting securities. Therefore, on September 15, 2023, condition (i) of the definition of "change of control" was satisfied. Therefore, I am supposed to get Enzolytics Common Stock. In fact, everyone who owns an Enzolytics SAFE is supposed to get Enzolytics Common Stock.
So, am I correct in thinking that everyone who owns an Enzolytics SAFE is supposed to get Enzolytics Common Stock?
Also, why did the Cotropias, Chandra, and the Zhabilov Trust have to give up all of those Series A Preferred shares? How did their giving up the shares facilitate the SAGA transaction?
ENZC's securities lawyer is Morgan E. Petitti.
Her contact information is as follows:
118 W. Streetsboro Road
Suite #317
Hudson, OH 44236
330 - 697 - 8548
pettitilaw@gmail.com
sources:
https://www.otcmarkets.com/stock/ENZC/profile
https://www.otcmarkets.com/financialReportViewer?symbol=OSCI&id=376096 (page 15)
ENZC's accounting firm is Fruci & Associates.
This firm's contact information is as follows:
802 N. Washington St.
Spokane, WA 99201
509 - 624 - 9223
877 - 264 - 0486
mail@fruci.com
sources:
https://www.otcmarkets.com/stock/ENZC/profile
https://www.fruci.com/contact
I strongly suggest that we flood the lawyer's phone and email inbox and the accounting firm's phones and email inbox with our complaints about ENZC's behavior. Hopefully, pressure from a LOT of irate and angry investors will cause the lawyer and the accounting firm to pressure their clients (the people behind ENZC) to give the investors some honest answers about what is going on with ENZC. We should make sure that the lawyer and the accounting firm know that, if the people behind ENZC are doing something illegal, then the people behind ENZC, and possibly the lawyer and the accounting firm as well, can be brought up on criminal charges and/or sued in class-action lawsuits.
I believe that is what happened. Good luck to all here
This all points (as of now) to Harry being the problem, not the ones who left.
Chandra is a crooked POS. I have been calling him a crook for months. Nobody wanted to believe it.
Well, there you all have it. That’s the proof from Chandra on what I’ve suspected for weeks now, that the real valuable assets are gone from BGEN and ENZC and left when they did. And I’m 99.999999% sure Clone 3 left with Charles, just like I suspected as well. Again, zero chance the Cotropias life work didn’t leave with them.
I'm pretty sure my Top Secret clearance expired a LONG time ago, but thanks for the intel. Wish I didn't jump and sell all, but it is what it is. I will be watching.
ENZC$0.0035 WHERE ARE MY COMPADRES from the Onward and Forward fan club??
I told you all to be careful, this was Wolves territory and bags were the sheep. Bubble burst and all gone now. Everyone making a banging now in IGPK. Only dust in here
When Doc Chand said not to CEO position, that was the last red flag I got that it was over. Too many red flags anyways since a while but Chand saying not to his most loved HIV cure company? 😂 bad, very bad
Love love love seeing this!! Time for this to be back to trips right where it belongs!!
another great day for share holders at ENZC thanks to our world class management .
Well, it's not ideal but at least we know where Chandra believes the IP is currently (not within BGEN or ENZC). Which would actually make sense for the Novation agreement that removed BGEN from the business combination. It would be hard to spin this as a positive on any aspect from a common shareholders perspective.
I expect we will see legal papers revealed to the public relatively soon attempting to stop Harry and his used medical supply business ideas.

I really doubt that docsetc is attempting to pump with his recent posts. If he was attempting to pump the stock, why wouldn't he have given us a positive report about his visit to the lab? I don't know what he found that caused him change his mind about the stock from earlier this week.
Correction: Exhibit 9, not 8.
Exhibit 9
TO THE BUSINESS COrvIBINATION AGREEl'vIENT AMONG SAGALIAM ACQUISTION CORP.
BIOGENYSIS, INC., VIROGENTICS, INC., AND THE SUBSIDIARY SHAREHOLERS
PA TENT LICENSE AGREEMENTS
Log In
Skip to content
?
The Company protects its technology through an aggressive strategy to cover its intellectual property. The Company’s intellectual property includes:
Issued Patents
Enzolytics is the owner of the following issued U.S. Patents covering its technology relating to a peptide that has been demonstrated in clinical trials to provide anti-HIV-1 retroviral benefit in vivo.
U.S. Patent No. 7,479,538, issued January 20, 2009, entitled Irreversibly-Inactivated Pepsinogen Fragment and Pharmaceutical Compositions Comprising the Same for Detecting, Preventing and Treating HIV
U.S. Patent No. 8,066,982, issued November 29, 2011, entitled Irreversibly-Inactivated Pepsinogen Fragment and Pharmaceutical Compositions Comprising the Same for Detecting, Preventing, and Treating HIV.
These patents cover an Inactivated Pepsin Fragment (IPF) identified and characterized by the amino acid sequence GDEPLENYLDTEYF. This peptide has demonstrated a significant in vitro binding affinity for HIV-1 gp 120 and gp 41 and human CD4 cells. The peptide exhibits antiretroviral activity in vivo, particularly anti-HIV-1 activity. IPF appears to modulate helper T1 cells’ expression of elaborate cytokines INFy, IL-2, which selectively promote cell-mediated immune response and subsequently stimulate cytotoxic lymphocytes. These lymphocytes have a prominent role in the host’s immunologic response to HIV infection. Proteins encoded by these pathogens enter the endogenous pathway for antigen presentation and are expressed on the surface of the infected cell as a complex with class I MHC – proteins. IPF appears to present a novel mechanism to reduce the viral burden and stimulate innate immune responses to the virus for patients with significant antiretroviral resistance.
Proprietary Cell Line Producing Clone 3 anti-HIV monoclonal antibody
The Company’s Clone 3 cell line, which produces fully human monoclonal antibodies (mAbs) that specifically target and neutralize the HIV-1 virus, is proprietary to the Company.
Pending Patent Applications
The Company has the following pending patents covering its anti-HIV monoclonal antibody technology:
Pending Patent covering the recombinant of the Clone 3 antibody. This form of the Clone 3 antibody is prepared using the known amino acid sequence of the antibody in conjunction with a high producing CHO cell line for generating recombinant form of the monoclonal antibodies that will ultimately be used in patient application.
Pending Patent coverage of small molecules (mini-peptides) for commercial use derived from the structure of the Clone 3 antibody for interrupting and preventing binding between the HIV virus and the human CD4+ cell.
Coverage is directed to blocking peptides that bind to and neutralize the HIV virus, and
Competitive peptides that bind to the target CD4+ cells at the point of virus access into the human cell to prevent infection.
Pending Patent covering the proprietary methodology for producing fully human neutralizing monoclonal antibodies against infectious diseases, including Rabies, influenza A, influenza B, Tetanus, Diphtheria, HIV-2, Anthrax, Smallpox, H1N1 influenza, Herpes Zoster, Varicella Zoster and Ebola.
Pending Patent covering the proprietary methodology for produced anti-antigen monoclonal antibodies to produce vaccines to achieve a broad and durable humoral protective antibody response against the corresponding infectious agent.
Proprietary Methodology for Producing Fully Human Monoclonal Antibodies
The Company proprietary mAb methodologies & immunotherapeutic technologies platform will be used to create therapeutics for treatments of viral infectious diseases against:
HIV-2
Anthrax
Smallpox
H1N1 Influenza
Herpes Zoster
Varicella Zoster
Ebola
The Company antibody-based immunotherapeutic platform can be utilized for both human and all animal infectious-disease applications.
In addition to the anti-HIV monoclonal antibodies produced by the Company, the Company currently has also produced fully human monoclonal antibodies against:
Rabies
Influenza A
Influenza B
Tetanus
Diphtheria
The Significance of the Methodology for Producing Fully Human Therapeutic Monoclonal Antibodies
Biologics, specifically fully human neutralizing monoclonal antibodies directed against infectious disease, is considered the new frontier in biotechnology. In the past, the initial starting products were “humanized” rat and mouse mAbs being created for therapeutic use. “Humanized” immunoglobulins are the major immunotherapeutic that is prevalent today. What sets the Company’s antibody technology apart from that employed by other pharma? The Company’s technology permits the cloning of human or animal immune system cells. With the Company’s proprietary methodologies, stable parent hybridomas cell lines can be created that produce fully human antibodies.
Enzolytics specializes in the creation of human neutralizing monoclonal antibodies, not “humanized” mouse or rat counterparts, as are many mAb therapeutics in pharmaceutical use today. The Company’s technological methodologies have developed an effective, strong, and robust portfolio of biologics that have a pharmaceutical application with significant benefits to patients or animals in the global marketplace. From an identified and created parent hybridoma cell line, four distinct and effective products can be produced: (1) fully human neutralizing monoclonal antibody—directed against any pathogen based disease entity—through use in passive immunotherapy; (2) an effective humoral active vaccine that is safe and effective; (3) an oral mini-antibody peptide-based medication with an efficacy that is equivalent to the immunologic capacity of the monoclonal antibody produced by parent hybridoma cell; and (4) an entry-fusion inhibitor that is immunologic in character and scope. The applications are broad, effective, and beneficial for immunotherapeutic use.
The methodologies and processes for creating immortalized cells that stably produce human monoclonal antibodies are Company proprietary trade secrets.
Anti-HIV Technology and Products
Using its proprietary technology, The Company has created a proprietary cell line that produces a fully human monoclonal antibody (known as Clone 3) that specifically targets and neutralizes the HIV virus (i.e., renders the virus incapable of reproduction). This capability to neutralize the virus means that Clone 3 may be used successfully to treat those infected with HIV and provide an ideal immunogen in the development of an active anti-HIV/AIDS vaccine that is both prophylactic and therapeutic.
Additional supplemental products may be produced as well, for example, vaginal creams for prevention of transmission of the virus. Additionally, Clone 3 has the potential to enormously reduce the incidence of mother-to-child transmission (MTCT) of the virus. Not only might it prevent in-utero viral transmission but, postnatally, effective treatment can be administered without fear of antiretroviral (ARV) toxicity or resistance.
Each of these distinct products flows from the Company’s technology. The first is the use of the antibody itself or a recombinant of the antibody. The second is the Clone 3 vaccine, the third is a mini-antibody or paratope of the Clone 3 antibody, and the fourth is a competitive binding entry-fusion inhibitor that prevents infectivity.
In addition to the Clone 3 monoclonal antibody against HIV, the Company has also created human monoclonal antibodies against other infectious diseases, including rabies, influenza A, influenza B, tetanus, and diphtheria. This immunotherapeutic technology platform can be readily applied to creating monoclonal antibodies against other pathogenic diseases, including the SARAS-CoV-2 (CoronaVirus), HIV-2, anthrax, smallpox, H1N1 influenza, herpes zoster, varicella zoster, Rh (+) auto=immune disease, and the Ebolavirus. The Company’s antibody-based Immunotherapeutics platform can be utilized across both human and animal infectious-disease applications. For example, the technology can be used to produce treatment for elephant and equine diseases that threaten these species.
?
Home
Our Mission
Our Technology
Our Science
Who We Are
Intellectual Property
Company News
Contact Us
Sitemap
© 2024 Enzolytics.
All rights reserved.
Website by web.com.
I suspect you and I-glow know that what matters is the ownership of the rights to make, produce, use and sell, etc. under protection of a patent, not the actual ownership of a patent itself. JMHO. In fact, once an exclusive right is granted (assigned), the owner typically has no rights other than those reserved in the terms of the assignment. JMHO. So, to keep saying that ENZC doesn't own the patents may be true, but it is misleading because what they do own are the rights to those patents according to the licensing documents (assignments), assuming the documentation matches ENZC's representations. JMHO.
In this case, Harry Z's trust has assigned those rights to VIRO, which was and still may be a subsidiary of ENZC. Those assignments and an application for a multiple sclerosis patent assigned by Harry individually are attached to the BCA as exhibit 8. Note: the multiple sclerosis application has been reported in the ENZC reports as a provisional patent application, but the assignment is for "U.S. Application No. 63/123,341" filed 12/9/2020 and is a different number than the provisional patent application. Presumably, the new application was filed after the provisional expired. JMHO.
If you can find that these assignments are not legitimate, then you may have something, and I might agree with you, otherwise IMO you're talking BS about the patents and what ENZC or VIRO own.
(1) U.S. Patent No. 7,479538, Irreversibly - Inactivated pepsinogen fragment and
Pharmaceutical composition the same for detecting preventing and treating IIlV; U.S. Patent
No. 8,066982, Irreversibly - Inactivated pepsinogen :fragment and Pharmaceutical composition
compressing the same for detecting preventing and treating HIV (herein the "Licensed
Patent"), including all patents issuing therefrom and any foreign counterparts thereof;
WHEREAS Licensee wishes to license the Licensed Patent and Licensor is willing to grant to
Licensee a license to the Licensed Patent on the terms and conditions set forth in this Agreement.
n December 9, 2020 the company filed a provisional patent with the U.S. Patent Office for an Immunotherapy
treatment of Multiple Sclerosis developed by Harry Zhabilov, titled NUCLEAR PROTEINS ISOLATED FROM
MAMALIAN SPINAL CORD (SCNP) IMMUNE FACTOR, Ser. No. 62/123341. The Company received confirmation of
filing from the U.S. Patent Office on December 10, 2020. On January 19, 2021 the Company announced the receipt
for the Multiple Sclerosis Patent Application.
Harry the Bulgarian bandit probably pumped it so he can dump it for big returns.
Crazy huge dump. Almost 50M shares in a half hour.
That craps been spewed since it's pump 3 or 4 years ago. They milking this for all it's worth lol. "not selling-i know what i own!!" At least there hasn't been any champagne bottle poppin talk lately
On this board, you'll be surprised.
But don't take my word for it, rail on. Other investor boards, I'm just not going to worry about.
BigTalon will be just fine, and so will patient ENZC shareholders.
Criticism is warranted, but as you said, if the stock is useless move on, leave it for the interested parties.
The tough times ENZC shareholders have suffered through does stoke justified frustration, major frustration, but good news will be upcoming.
I personally, would pay no heed to ENZC PR's, but wait until those you are concerned about, post their own announcements. And they will.
GLTA.
Sorry, but looks like investors are rolling with my sentiment.
"Take the complaints elsewhere."
"GLTY, elsewhere"
These are the tactics they pull when the end is near
Yes, and the best part being that its already expected that no reply will be given.
Priceless!
Timing, considering that the shares are basically worthless, and if you tried to sell off a bunch it would crater the stock even more, the real question is how much $$ the gang milked from investors on the salary side of things over al this time.
Agreed..Also I have not seen any proof that the IP and assets of BGEN have been removed by Charles and Joseph. I hear a lot of discussion about this on the other board also..But I'm not convinced it has been separated. As you and I agree..These guys own a LOT of Shares of ENZC.
Doggone, Mutat and all
--
Do NOT be surprised if those A Shares for CC, GC and JC are still in their possession now, why the SAGA deal did not go through. Plus the commons they had before . All One Knows is "What Buttons" Harry Z is pushing at this point. In any case the 3 C's still have a large , LARGE Vested interest here and for sure Charles has protected their interests (TBD) and will play those cards when the time is right .
--
I along with Many have worked at company's in an Executive Position ,moved on , and still hold shares.
Lets See
Mutat is correct. Not a single share to Chandra, Catropias or Fellegy have been released or sold off in any way.
There's a really good reason for that.
Stay tuned and stay patient. I'd also advise not putting too much into any PR coming out prior to BIG NEWS coming out later.
GLTA
All IP was and still is under the trust that I'm aware of. Shareholders never have, and never will own squat.
Was their ever a filing where the owner was listed as the company (ie shareholders) themselves?
If so, there's your breaking news.
The last I looked, the expiration on those two wasn't far out.
Do you recall the M/Y that they expire?
It certainly is, but not how you describe it.
Take the complaints elsewhere. There's stuff going on, but here's two facts I can give you.
Charles and Chandra are gone from ENZC yes, so are their BGEN IP's, patent apps, and research assets. Harry, ENZC and SAGA can no longer touch them or ever file a claim to them.
Charles and Chandra have not sold or released even 1 of their stock shares in ENZC since they got them. Put that back into your calculus.
Brighter futures for ENZC shareholders ahead. Might not be with ENZC, but for the shareholders of ENZC.
Based upon that, I'm going to be patient for a while longer and hold.
GLTY, elsewhere.
I wasn't challenging your integrity, as I was suggesting to everyone in the room that you have none.
That won't change as long as you pump this pos turd and claim it to be a real thing.
Chandra = Gone
Charles = Gone
Started a new company Adnexus completely unrelated to Enzolytics
ENZC is now a marketing company selling medical devices explained in recent PR
Enzolytics twitter is a joke.. Pushing a delisted SAGA = criminal
The rug has been pulled
Just assumed Charles would never surrender his life’s work.
I must have missed something. Where do you see that the IP left BGEN?
April 26th PR...
Sagaliam Acquisition Corp. Announces Novation Agreement with Enzolytics, Inc.
Sagaliam Acquisition Corp.
Fri, Apr 26, 20246 min read
NEW YORK, NY / ACCESSWIRE / April 26, 2024 / Sagaliam Acquisition Corp. ("SAGAU", "SAGA", SAGAR") (the "Company") announced that Sagaliam entered into a Novation Agreement with Enzolytics, Inc. (OTC PINK:ENZC), Biogenysis, Inc. (BGEN), and Virogentics, Inc. (VIRO) (together the "Parties"). ?Under this restructured agreement SAGA retains 100% ownership of VIRO, ITV-1, the nutraceutical products and license along with VIRO's other assets. This agreement transferred ownership of Biogenysis, Inc. ("BGEN") and it's IP, patents and associated asset being developed back to ENZC in exchange for a reduction of the price from $450,000,000 to $290,000,000 with the dividend of the purchase shares to come directly to the ENZC shareholders from SAGA. The Parties believe this will significantly shorten the time needed to obtain regulatory approval to initiate the dividend of the 29 million SAGA shares issued for the purchase of VIRO. All other terms and conditions of the purchase agreement, including the make-whole provision relating to the 29 million SAGA shares, remain in place.
Barry Kostiner, the CEO of Sagaliam, commented, "After the Sagaliam - Enzolytics transaction was closed in September, we have worked together with the Enzolytics management team to tweak the details for mutual benefit. We are excited about moving forward with the Virogentics team. The ITV-1 therapy has potential to significantly improve effectiveness and reduce costs of the standard of care for HIV. Additionally, ITV-1 is believed to have therapeutic application to other chronic conditions including diabetes, cancer and other conditions known to be impacted by peptide therapies targeting inflammation. Our IPF nutraceutical has received strong consumer approval, and we will be ramping up our marketing efforts, in parallel with our other nutraceutical products. Through the Novation Agreement, return of Biogenysis to Enzolytics, and reduction of shares issued, we believe that we have a strong foundation for building a valuable pharmaceutical company with global impact, in partnership with our colleagues at Enzolytics. We are also working with Enzolytics on building the sales, marketing and distribution infrastructure for our nutraceutical products."
Steve Sharabura, the CEO of Enzolytics, commented, "The Novation Agreement is an exciting development for Enzolytics. It serves as a solid foundation for building both Enzolytics and Sagaliam as significant biotech companies that are independent entities, but that work closely together. Enzolytics is proud to be a major shareholder in Sagaliam. The two companies have several initiatives in the works, specifically in the areas of medical testing, medical devices and an expanded nutraceutical product line. Negotiations are ongoing between Sagaliam and Enzolytics on these matters."
Harry Zhabilov, the Chief Science Officer of Virogentics, commented, "With the completion of the Novation Agreement, we have visibility to funding and the success of both companies. We now have a platform that allows us to implement our vision of bringing our technology to commercial implementation. We are also in advanced discussions with multiple synergistic biotech companies that are interested in joining our platform. After much anticipation, we are excited about moving forward on bringing our therapies to improve the lives of many patients with multiple chronic conditions, as well as increasing the near-term revenue of our nutraceutical products."
About Sagaliam Acquisition Corp.
We are a blank check company incorporated under the laws of the State of Delaware on March 31, 2021 for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses or entities. Sagaliam intends to continue to pursue the consummation of a business combination with an appropriate target.
Virogentics, a subsidiary of Sagaliam, is advancing multiple therapeutics targeting numerous infectious diseases. One patented and clinically tested compound, ITV-1 (Immune Therapeutic Vaccine-1), is a suspension of Inactivated Pepsin Fraction (IPF), covered by U.S. Patent Nos. 8,066,982 and 7,479,538. Studies have shown it to be effective in treating HIV/AIDS. ITV-1 has also been shown to modulate the immune system.
DOGONE all tha IP left with Charles and Chandra and unless we have a miracle happen we may not have a ticket to ride. I didn’t invest in HARRY or anything he has. Clone 3 is what I put my money on and the AI was to be the bonus.
You're a class act docsetc.
Thanks for all of your efforts.
Well, since I'm the one that found the lab-lights had been turned off myself and posted the pictures here, and now tell you that its best to sit and wait on ENZC, I must have reasons for it, right basher?
Look, we all had good discussions about what was going wrong at ENZC, and I can tell you on good authority, we, for some good conjecture and fact-sharing, did have a lot of it right, as to what was happening, but it was a flipped-situation on how it was happening.
So now, on good information that became available, I will wait and see about ENZC. I was ready to sell off on a spike, but now I will wait until I see the right PR or public announcement that rings true before I act. I have good reason to hold and believe that the work on Clone3 and Clone7 will see the light of day sometime in the not-so-forboding future.
I will tell you, like I posted right after the last PR, I have no faith in the new 'Medical Device Sales and Distribution' angle, but I have faith in some other factors I believe will arise.
GLTY And don't ever challenge my integrity again.
continued.....
Enzolytics, Inc. Overview
Enzolytics, Inc. is a drug development company committed to commercializing its proprietary monoclonal antibodies and proteins to treat debilitating infectious diseases.
Biogenysis, a subsidiary of Enzolytics, uses proprietary techniques to produce fully human monoclonal antibodies (mAbs) against infectious diseases. The technology is currently being employed to produce monoclonal antibody therapeutics for treating the CoronaVirus (SARS-CoV-2), HIV-1 and the Feline Leukemia Virus. The Company has also identified conserved epitopes on and has plans to produce mAbs targeting many other viruses, including H1N1 Influenza, HTLV1 & HTLV2, Leukemia Virus, Herpes Simplex I Virus, Respiratory Syncytial Virus (RSV), Smallpox Virus, Rabies, Influenza A, Influenza B, Ebola Virus, Epstein Barr Virus, and Monkey Pox Virus. The Company has also analyzed epitopes of animal viruses and plans to produce mAbs for treating these animal viruses.
Virogentics, a subsidiary of Enzolytics, is advancing multiple therapeutics targeting numerous infectious diseases. One patented and clinically tested compound, ITV-1 (Immune Therapeutic Vaccine-1), is a suspension of Inactivated Pepsin Fraction (IPF), covered by U.S. Patent Nos. 8,066,982 and 7,479,538. Studies have shown it to be effective in treating HIV/AIDS. ITV-1 has also been shown to modulate the immune system.
This Aug 24th 2023 PR gives us a breakdown of VIRO and BEGN and what they contain.
ACCESSWIRE
Biogenysis’ International Patent Application, Covering Its Anti-SARS-CoV-2 Monoclonal Antibodies, Advances to Prosecution in Europe and as a U.S. National Stage Application.
ACCESSWIRE· Enzolytics, Inc.
Enzolytics, Inc.
Thu, Aug 24, 202310 min read
COLLEGE STATION, TX / ACCESSWIRE / August 24, 2023 / Biogenysis, Inc. (OTC PINK:ENZC) (or the "Company"). Biogenysis, a subsidiary of Enzolytics, Inc., announces the progress of its Patent Cooperation Treaty (PCT) application covering its inventions relating to Anti-SARS-CoV-2 Monoclonal Antibodies. The PCT application has now advanced to filing as a U.S. National Stage Application in the U.S. Patent Office and as a formal European Patent Organization (EPO) application. The EPO application makes possible coverage in the 39 contracting countries within the EPO. These countries include all of the countries in the European Union, the United Kingdom and the extensive list totally 39 member countries throughout Europe and adjacent regions. https://www.epo.org/about-us/foundation/member-states.html.
The pending applications contain 23 separate patent claims covering (1) the 19 discovered highly conserved antigens and epitopes (sites) on the SARS CoV-2 virus, (2) antibodies that bind to the disclosed antigens and epitopes, (3) vaccines based on the antigens, (4) methods of treating, preventing, or reducing the risks of SARS CoV-2 infection with the antigens or binding proteins, and (5) methods and kits for detecting or diagnosing infection by SARS CoV-2 using the antigens or binding proteins.
Through computer analysis (Artificial Intelligence (AI)), the Company discovered 19 immutable conserved virus sites on the COVID-19 virus by analyzing 2.8 million SARS CoV-2 isolates. This analysis identified these critical, conserved, immutable epitopes of the virus. These sites are now claimed as patentable based on their novel specificity and the finding that they are conserved on SARS-CoV-2. This is significant because antibodies produced to bind to these conserved, immutable sites can neutralize the virus without the possibility of "virus escape" due to virus mutation. Specifically, virus mutants will still contain these target sites allowing these specialized monoclonal antibodies to neutralize the virus even as it mutates.
The Company's patent applications claim the use of any one identified epitope or any combination of any of the multiple identified epitopes in any of the following ways:
For producing a therapeutic monoclonal antibody to treat the CoronaVirus.
For making a vaccine against the CoronaVirus.
For creating related prophylactic/therapeutic methods relating to the epitopes/antigens.
For use in any diagnostic test to identify whether a person has the CoronaVirus.
The Company has produced and is producing fully human monoclonal antibodies against these claimed sites. The International Patent Office has confirmed these discoveries as being novel and inventive, capable of being patented and claimed exclusively for a 20-year term in each member country under the Patent Cooperation Treat in which the Company pursues these claims.
The Company also has similar PCT applications pending covering discovered immutable conserved epitopes (binding sites) on the HIV virus and the animal Feline (cat) Leukemia Virus (FeLV). The Company has also analyzed the amino acid sequences on numerous other human and animal viruses and plans to also produce anti-mAbs targeting these sites and will file for U.S. and international patents on these discoveries. These discoveries include the Company's findings from analyzing the human viruses H1N1 Influenza, HTLV1 & HTLV2, Leukemia Virus, Herpes Simplex I Virus, Respiratory Syncytial Virus (RSV), Smallpox Virus, Rabies, Influenza A, Influenza B, Ebola Virus, Epstein Barr Virus, and Monkey Pox Virus.
The Company has also analyzed and identified conserved and immutable epitopes in animal viruses including Equine Infectious Anemia Virus, Feline Immunodeficiency Virus, Koala Retrovirus Virus, and Elephant Endotheliotropic Herpes Virus. Using the Company's technology currently being used to produce monoclonal antibodies against human viruses, therapeutic mAbs may be produced to treat these animal viruses.
The Company fully expects the issuance of multiple international patents covering these discoveries accomplished through the Company's unique process using computer analysis (AI) to identify conserved, immutable epitopes on the target virus. Biogenysis' proprietary AI platform, invented by Dr. Gaurav Chandra, Biogenysis' CEO, has been used to analyze the sequences (structure) of 2.8 Million SARS-CoV-2 virus isolates to identify conserved sites. As a part of this process, 3-dimensional models of these conserved targets were generated, and the targets were analyzed for linearity, accessibility by antibodies, and neutralizability by antibodies. From this, Biogenysis scientists are producing multiple broadly neutralizing antibodies targeting these multiple conserved, immutable epitopes on the targeted virus. The monoclonal antibodies directed against these targets will expectedly be universal, durable, broadly neutralizing, and unaffected by virus mutation.
The Company considers the expected patent protection to be highly significant, recognizing that for a monoclonal antibody to be effective (that is, to be fully capable of neutralizing a virus), it must target an immutable site on the virus. Otherwise, virus mutation will render the therapeutic ineffective. Biogenysis' methodology for producing monoclonal antibodies is unique and innovative, unlike those other biotech companies employ. The Company's antibodies are made from human "immune-B cells" obtained from convalescent individuals (or animals in the case of veterinarian monoclonal antibodies) who have recovered from the target virus. The Company's monoclonal antibodies are not "humanized" but are fully human monoclonal antibodies where the original antibody affinity and specificity are maintained, and the chances of immunogenicity are minimized.
The Company believes its discoveries will be significant to the success of the Company. Although the COVID-19 pandemic was reported to be over, it is now recognized that COVID-19 will be with us from year to year. The global market size for effective treatments in 2022 was $16.43 billion. The global Monoclonal Antibodies market size was $185.50 Billion in 2021, with an expected compound annual growth rate (CAGR) of 11.30% from 2022 to 2030.
The Company's near-term objective is to collaborate with pharma entities and license its technology or partner with major pharma entities now producing therapies for COVID-19.
Regarding the Company's therapeutic against HIV, the Company has produced a fully human anti-HIV monoclonal Antibody, Clone 3. The Company is now producing additional anti-HIV mAbs to be used in combination. Experts agree that a cure for HIV may be found in the production and administration of multiple anti-HIV monoclonal antibodies. This is the Company's goal. The Company has successfully produced and tested, in vitro, one HIV monoclonal antibody (Clone 3) and has identified 7 additional immutable conserved sites on the HIV virus against which it is in the process of producing anti-monoclonal antibodies. These antibodies will be produced as fully human, with the starting point being human "immune-B cells" from long-term non-progressor HIV patients. This production is being conducted with pharmaceutical entities to accelerate the process of production and testing of the monoclonal antibodies.
In 2021, the global HIV drugs market was $30.46 billion, with this market projected to grow to $45.58 billion by 2028. The global Monoclonal Antibody market size was $185.50 Billion in 2021, with an expected compound annual growth rate (CAGR) of 11.30% from 2022 to 2030.
The global market for Human Monoclonal Antibodies in 2022 was $55 Billion.
The Company's near-term objective is collaboration with pharma companies producing treatments for HIV. The current HIV treatments available do not cure and must be taken for a lifetime. The Company is focused on licensing or partnering with major pharma entities now producing treatments for HIV. To accelerate and fully execute the successful production of the multiple monoclonal antibodies, the subject of the Company's intellectual property, the Company continuously engages with multiple pharma entities to accomplish the successful goal of production, testing, and delivery of successful therapeutics.
Gaurav Chandra, Biogenysis' CEO, stated, "Amid alarming concerns regarding COVID mutations, it is crucial to prioritize efforts towards achieving broader immunity against variants, longer-lasting protection, and preventing infection and transmission. Utilizing intranasal IgA monoclonal antibodies effectively attains high secretory IgA levels, reflecting mucosal immunity. The Biogenysis anti-COVID monoclonal antibodies, both IgG and IgA, being universal, broadly neutralizing, durable, and unaffected by mutations, are a critical solution to this pressing issue."
Charles Cotropia, Enzolytics' CEO, said, "We have a well-defined strategy for developing an Intellectual Property portfolio to secure significant patent protection covering our therapeutic discoveries in numerous countries around the world. With these rights secured, we will focus on potential partnering or licensing the technology to international pharmaceutical companies working in the areas in which we are focused. Obtaining international patent coverage will be significant in achieving this objective.
"The necessary technology is now in hand for producing these therapeutics for use in the treatment of many infectious diseases. The earlier announced plans for the sale of Biogenysis and Virogentics, subsidiaries of Enzolytics, to Sagaliam Acquisition Corp. (NASDAQ: SAGA), a special purpose acquisition company ("SPAC"), comes at an opportune time. Once completed, this transaction will provide Biogenysis and Virogentics with the capital that will make possible final development of the therapeutics now being produced."
lol, I think he's sleeping with BM, she has a hot passion for emailing hirer ups.
Bigtalan wants to send emails lol, imagine thinking you should have to send an email to get a remotely honest response, or any information at all, about the company you've invested in lol. I guess they're too stupid to communicate in any proper way, but an email should solve that!
No there aren't.
And please stop mentioning that you're from the great state of Texas.
Its an embarrassment.
I know we probably wont get a reply , but has anyone sent emails to Harry and Barry , asking simple questions that as a long time shareholders are due answers and not the same BS pr's we have been getting? Question on when the dividend date was or will be , will SAGA be tradeable and on what trading platform and when along with a few more. Still waiting on mine GLTA
So we're going from saving humanity, medical break-through antibodies to hawking vitamin C enhanced health food store supplements! C'mon! And if you haven't al ready, check out Chandra's latest happy tweets. What have they been doing for four years? For the love of God, When are we going to get some real information? There....I had to say it. LOL
Third Coast Fulfillment - http://www.thirdcoastfulfillment.com/
The address from the website is:
3810 Dacoma St
Houston, TX 77092
When I looked that up I found:
Wahlberg-McCreary Parts Plus
Auto parts & supply store
Permanently closed
3810 Dacoma St, Houston, TX 77092
Then I checked Third Coast Fulfillment with the Texas SOS
Mailing Address 3701 AVENUE F BAY CITY, TX 77414-7605
3701 Avenue F
Bay City, TX 77414
40,040 SF
Vacant Industrial Building
For Sale.
Before blindly believing a press release that Quotes Kostiner - verify everything then run.
IG
If this was what docset was having us wait for, it’s disappointing. Let’s hope something more is mentioned soooonn
|
Followers
|
1251
|
Posters
|
|
|
Posts (Today)
|
1
|
Posts (Total)
|
199574
|
|
Created
|
01/07/10
|
Type
|
Free
|
| Moderators Homebrew Timing101 archilles SeniorApollo | |||




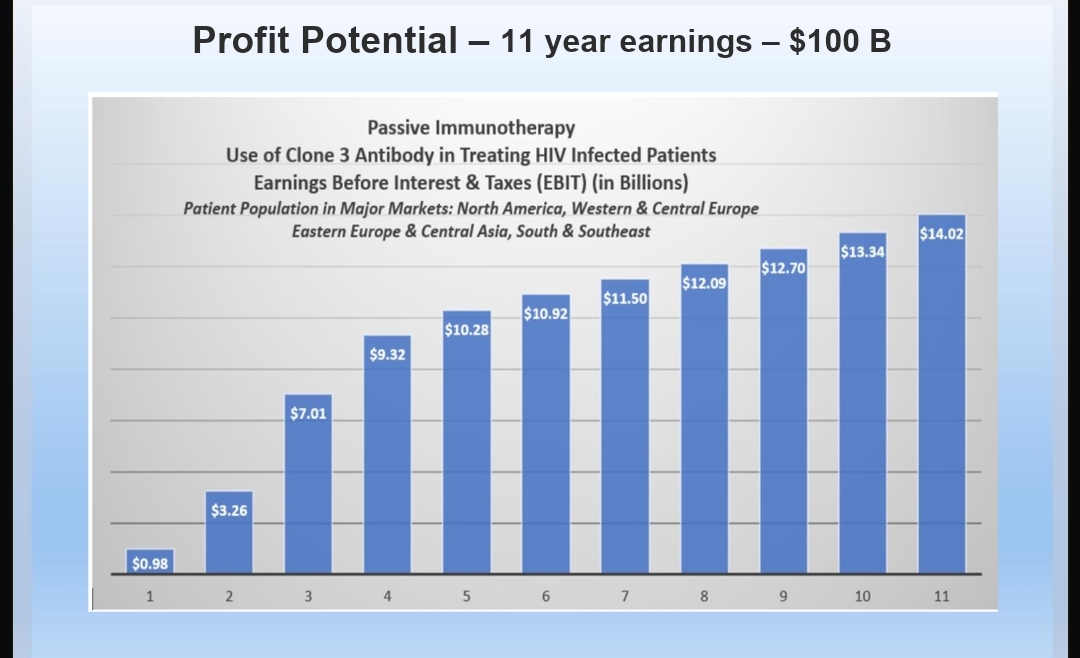
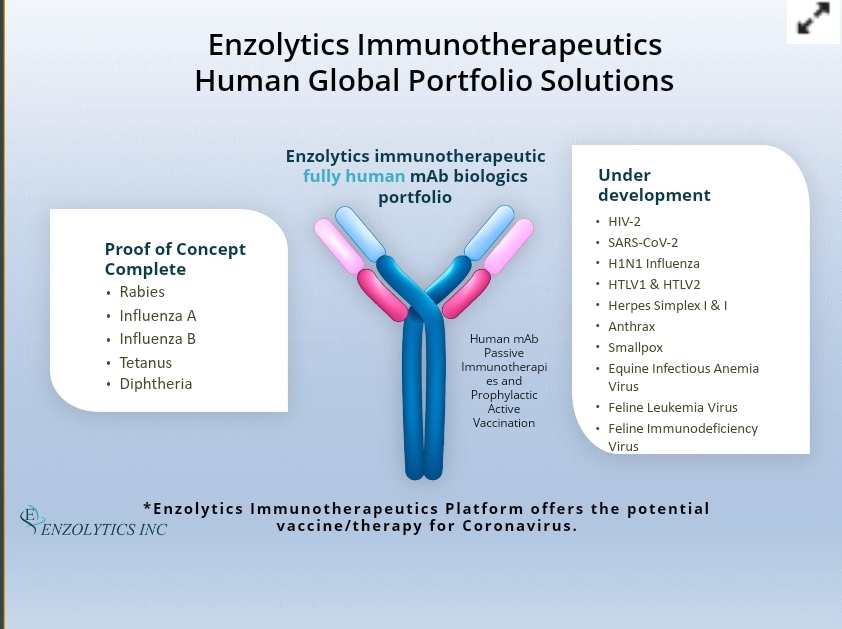






Independent Valuation Report Pre Clinical Studies
Investments
___________________________________________________________________________________
Press Release July 20-2021
Enzolytics Announces the Signing of a Letter of Intent with Creative Biolabs, Inc.
for Licensing and Commercialization of anti-HTLV-1 Monoclonal Antibodies
https://www.creative-biolabs.com/
https://finance.yahoo.com/news/enzolytics-announces-signing-letter-intent-113000339.htm

________________________________________
Press release July 21-2021
Enzolytics and Lonza Bioscience Combine Technologies to Fast-track Development and
Production of Anti-HIV and Anti-SARS-CoV-2 Monoclonal Antibodies produced by Enzolytics
https://www.lonza.com/
https://www.bloomberg.com/press-releases/2021-07-21/enzolytics-and-lonza-bioscience-combine-technologies-to-fast-track-development-and-production-of-anti-hiv-and-anti-sar
_________________________________________
July 22 INTERVIEW
with
James Hicks, Charles Cotropia and Gurav Chandra
http://www.youtube.com/watch?v=mdljO9VLgj0
______________________________________________________________
COLLEGE STATION, TX August 25, 2021
Important Excerpt from this news progress update.
>>>>>><<<<<<
"The monoclonal antibodies being produced by Enzolytics against the CoronaVirus target 19 conserved immutable sites on the virus, sites which the Company has now confirmed are conserved sites not only on the initial virus strains but also existing in the Delta and Lambda variants, as well as in the Alpha, Beta and Gamma variants."
Enzolytics Inc. and Samsung Biologics Announce
Development and Manufacturing Agreement for Anti-
HIV and Anti-SARS-CoV-2
Important Excerpt from this news progress update.
>>>>>><<<<<<
"Samsung Biologics (KRX: 207940. K.S.), a leading contract development and manufacturing organization and Enzolytics (ENZC), a drug development company committed to commercializing multiple proprietary therapeutics to treat debilitating infectious diseases, announced the signing of a strategic CDMO partnership agreement."___
 ____
____

2000 North Central Expressway
Plano, TX 75074
972-292-9414
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
