Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
You forgot Quantum Material Corp (QTMM) in your list. Gonna be the world leader in Quantum dots...
Say Goodbye to Landfills, and Hello to Plasma Arc Gasification
VIDEO
With millions of tons of waste being generated every year and nowhere to put it, scientists have developed a method of converting any waste, no matter how toxic or dangerous, safely and efficiently into a clean fuel gas. Plasma Arc Gasification produces no pollution, and we will use it to make landfills obsolete, and restore planet earth’s environment.
Dirty Diapers, concrete, sludge, even hazardous biomedical waste can be transformed into energy and other useful elements using Plasmagasification technology.
According to a study by the Worldwatch Institute, growing prosperity and urbanization could double worldwide municipal solid waste production by 2025 – from 1.3 billion tons to 2.6 billion tons. Without Plasma Arc Gasification technology, this would be a worrisome statistic, but with it there is nothing to worry about, since there is no longer such a thing as garbage. What used to be considered garbage, is now a commercially valuable, renewable, and sustainable resource.
How does it work?
Plasma, also called the 4th state of matter, can exceed temperatures of 7,000 degrees centigrade – that’s three times hotter than petrochemical fuels burn, and hotter than the surface of the sun.
Inside the chamber, two electrodes create an electric arc so hot, that it breaks down materials into their atomic elements. Even though the materials are being exposed to intense heat, they are not being burnt or incinerated. According to Plasco Energy scientist Andreas Tsangaris, “Plasmagasification works in the absence, or near absence of oxygen. Therefore it’s a process of decomposition, and not combustion.” The result is energy produced from emission-free molecular deconstruction.
Useful Byproducts of Plasma Arc Gasification
The fuel gas produced by Plasma Arc Gasification can be used to generate electricity, produce fresh water, heat and cool buildings, power vehicles, and virtually anything else we need energy for.
A small amount of rock-like glassy by-product, called “vitrified slag”, can be safely used by the metals industry, the construction industry, or as road bed material for building highways.
How Much Energy Can We Produce Using This Technology?
The world produces approximately 1.3 billion tons of municipal solid waste annually, and plasma gasification of 1 ton of average municipal solid wastes would send about 815 Kilowatt-hours of electricity to the grid.
Let’s do a little math…
1,300,000,000 (tons of municipal solid waste per year) x 815 kWh (electricity)= 1,059,500,000,000 kWh
At the mid-peak time of day, hydro in the U.S. costs about 10.9 cents per kWh.
1,059,500,000,000 kWh x 10.9 cents/kWh
= $115,485,500,000
Therefore, if the world used all of their “garbage” to produce energy using Plasma Arc Gasification Technology, we would generate about $115.5 billion in clean energy every year, for free. Plasmagasification of municipal solid waste has the potential to create more energy than the projected energy from solar, wind, landfill gas and geothermal energies combined.
When Will it be Implemented?
Although we need many more Plasma Arc Gasification plants around the world, it is already in used in some places, and in places where it’s not, progress is quickly being made.
Developing rapidly across Europe, companies such as UK-headquartered Advanced Plasma Power (APP) has already formed a joint venture to gasify thousands of tonnes of waste unearthed from a landfill mining project in Belgium.
Since 2002, two commercial waste-to-energy plasma gasification plants have been operating successfully in Japan.
In December 2012, the city of Ottawa, Ontario signed a deal with Plasco Energy to build a 150,000 tonnes per year Plasma Arc Gasification waste-to-energy facility. According to the Ottawa Citizen, “Plasco has two major things it needs to do: sell its technology to other markets, China is very interested, and build a full scale working processing plant in Ottawa.” Newly appointed CEO Raymond Floyd has just what it takes to make that happen.
Plasco has already signed a letter of intent with one of China’s largest state owned enterprises called Shougang. When it goes through, the agreement will bring Plasco Energy technology on line by 2017, a plan that will convert about 3,000 tons a day of Beijing’s garbage into clean fuel.
http://www.endalldisease.com/say-goodbye-landfills-hello-plasma-arc-gasification/
Nanomaterial inspired by nature paves way for greener energy (w/video)
Posted: Dec 21st, 2012

(Nanowerk News) A new nanomaterial, which finds its inspiration in nature, will provide the potential for more efficient and greener vehicles, rechargeable batteries and solar cells.
Researchers at the University of Reading have patented a new method of making electrode coatings with a thousand-fold increase in surface area compared with a flat electrode. This larger surface means that conversion of fuel or sunlight into electricity can take place in a smaller, more compact cell, making the cells cheaper to produce. The chemical reaction to create the energy also takes place at room temperature allowing the cells to be fitted to cheap materials such as plastic for the first time.
Based on structures found in the natural world which occur within mitochondria and chloroplasts, nature's own "fuel cells" and "solar cells", the new nanostructure is formed of a network of tiny wires, millionths of a millimetre in size, and is created by growing the metal in a template made from a plant molecule. This creates a structure known as a "bicontinuous cubic phase".
Dr Adam Squires, of the Department of Chemistry at the University of Reading, said: "Making electrodes more efficient lies at the heart of making our energy production more sustainable. This novel electrode coating technique has applications for fuel cells¹ in the newest generation of hybrid cars, photovoltaic cells, rechargeable batteries or battery production for a wide range of green technologies."
The process works in water using a technique known as electrochemical deposition, similar to silver plating a coin, and can be applied to any conducting electrode, creating a low cost, mass manufacture component. The unique 3D nanostructure enables much better conductivity and is the ideal shape for a high area electrode to create a more effective energy supply. Potentially, the technique could lead to energy storage devices with much greater capacity than traditional cells.
Dr Squires continued: "The method of production is chemically mild, environmentally friendly and crucially takes place at room temperature, which means that electrodes could be fitted to a range of other materials. It will allow, for example, photovoltaic cells to be located on a plastic base rather than the currently used metal or glass, which will make renewable energy technologies more flexible, lightweight and cost effective."
The work was carried out by Dr Samina Akbar when she was a PhD student at the University of Reading, in a joint project between the research groups of Dr Adam Squires and Dr Joanne Elliott. The findings are published in the journal Advanced Materials ("Facile Production of Ordered 3D Platinum Nanowire Networks with “Single Diamond” Bicontinuous Cubic Morphology") this week.
Source: University of Reading
http://www.nanowerk.com/news2/newsid=28207.php
QTMM Patented TQD's, 3rd Party Endorsement.
Possibly up your alley..
FastCap Systems
Fueled by a multi-million dollar grant by the Department of Energy ( ARPA-E), and additional funding through the Chesonis Family Foundation, angel investment and the Massachusetts Clean Energy Center, FastCAP Systems seeks to transform the automotive and grid storage markets by commercializing its novel and enabling high-power, high-energy and low-cost energy storage device. FastCAP's proprietary carbon nanotube electrode technology has enabled breakthrough performance improvement in its cells when compared with commercially available ultracapacitors, a leap forward that could lead to dramatic reductions in the cost of hybrid electric vehicles. And because FastCAP's ultracapacitors are produced with abundant, eco-friendly raw materials and have extremely long life cycles, the environmental benefits associated with widespread deployment of the technology are unlimited.
http://www.fastcapsystems.com/
(WO2012170749) ENERGY STORAGE MEDIA FOR ULTRACAPACITORS
Applicants:
FASTCAP SYSTEMS CORPORATION [US/US]; 21 Drydock Avenue, 8th Floor Boston, MA 02210
Abstract:
An ultracapacitor includes at least one electrode that includes carbon nanotubes. The carbon nanotubes may be applied in a variety of ways, and a plurality of layers may be included. Methods of fabrication of carbon nanotubes and ultracapacitors are provided.
http://patentscope.wipo.int/search/en/detail.jsf?docId=WO2012170749&recNum=1&docAn=US2012041438&queryString=FP:%28WO2012170749%29&maxRec=1
Sponge-like graphene makes promising supercapacitor electrodes
October 12, 2012 by Lisa Zyga

SEM micrographs of the 3D sponge-like graphene, which can be used as electrodes in supercapacitors with ultrahigh power density and relatively good energy density. Image credit: Zhanwei Xu, et al. ©2012 American Chemical Society (Phys.org)
—While most of today's electric vehicles rely on batteries to store energy, supercapacitors have enjoyed significant improvements that have made them serious competitors to batteries. Batteries traditionally have the upper hand in terms of capacity, since supercapacitors' low capacities mean very short driving ranges for electric vehicles. Supercapacitors' biggest advantage lies in their much higher power density compared to batteries, enabling a quicker charge time and the ability to quickly discharge for fast acceleration.
A new study by a group of researchers at the University of Alberta and the National Research Council of Canada, both in Alberta, Canada, has shown that supercapacitors have great potential for continuing improvements.
The researchers have synthesized a new material that they call sponge-like graphene due to its 3D macroporous structure and demonstrated that it can be used to make supercapacitor electrodes. Supercapacitors with these new electrodes have fair energy density when operating at low power densities, but their biggest attraction is when operating at ultrahigh power densities of around 48,000 W/kg, where they're able to deliver an attractive energy density of 7.1 Wh/kg.
At first, an energy density of 7.1 Wh/kg may not sound remarkable compared to the energy density of the best Li-ion batteries, such as Envia System's record-breaking 400 Wh/kg announced earlier this year. However, in order to reduce the time it takes to charge Li-ion batteries for electric vehicles from hours to minutes, batteries need to have a higher power density than their current best values of around 10,000 W/kg. So the 48,000 W/kg power density of the supercapacitors reported here, coupled with a 7.1 Wh/kg energy density, shows that supercapacitors may offer batteries some competition.
"Supercapacitors and batteries are quite different electrochemical energy storage devices," coauthor Zhi Li, of both the University of Alberta and the National Research Council of Canada, told Phys.org. "Here is an example used quite often to demonstrate their differences. If you are driving an electric vehicle, you would like a high-energy-density battery to keep the vehicle running for many miles and you would probably also prefer a high-power-density supercapacitor to make the car start/accelerate faster. Supercapacitors are designed to work at much higher power density (fast charging/discharging). 7.1 Wh/kg is far from attractive for a battery. However, this energy is delivered in less than 2 seconds. I believe none of the existing batteries is ready to do that."

The synthesis process in which graphene is grown between CNTs, resulting in 3D sponge-like graphene. Image credit: Zhanwei Xu, et al. ©2012 American Chemical Society
http://phys.org/news/2012-10-sponge-like-graphene-supercapacitor-electrodes.html
http://dx.doi.org/10.1021/jz301207g
Batteries made from graphene could power tomorrow's electric cars
Posted: Aug 20th, 2012
(Nanowerk News) Engineering researchers at Rensselaer Polytechnic Institute made a sheet of paper from the world’s thinnest material, graphene, and then zapped the paper with a laser or camera flash to blemish it with countless cracks, pores, and other imperfections. The result is a graphene anode material that can be charged or discharged 10 times faster than conventional graphite anodes used in today’s lithium (Li)-ion batteries.
Rechargeable Li-ion batteries are the industry standard for mobile phones, laptop and tablet computers, electric cars, and a range of other devices. While Li-ion batteries have a high energy density and can store large amounts of energy, they suffer from a low power density and are unable to quickly accept or discharge energy. This low power density is why it takes about an hour to charge your mobile phone or laptop battery, and why electric automobile engines cannot rely on batteries alone and require a supercapacitor for high-power functions such as acceleration and braking.
The Rensselaer research team, led by nanomaterials expert Nikhil Koratkar, sought to solve this problem and create a new battery that could hold large amounts of energy but also quickly accept and release this energy. Such an innovation could alleviate the need for the complex pairing of Li-ion batteries and supercapacitors in electric cars, and lead to simpler, better-performing automotive engines based solely on high-energy, high-power Li-ion batteries. Koratkar and his team are confident their new battery, created by intentionally engineering defects in graphene, is a critical stepping stone on the path to realizing this grand goal. Such batteries could also significantly shorten the time it takes to charge portable electronic devices from phones and laptops to medical devices used by paramedics and first responders.

SEM image of the cross section of photo-thermally reduced graphene shows an expanded structure. The graphene sheets are spaced apart with an inter-connected network allowing for greater electrolyte wetting and lithium ion access for efficient high rate performance in lithium ions batteries.

http://pubs.acs.org/doi/abs/10.1021/nn303145j
“Li-ion battery technology is magnificent, but truly hampered by its limited power density and its inability to quickly accept or discharge large amounts of energy. By using our defect-engineered graphene paper in the battery architecture, I think we can help overcome this limitation,” said Koratkar, the John A. Clark and Edward T. Crossan Professor of Engineering at Rensselaer. “We believe this discovery is ripe for commercialization, and can make a significant impact on the development of new batteries and electrical systems for electric automobiles and portable electronics applications.”
Results of the study were published this week by the journal ACS Nano ("Photothermally Reduced Graphene as High-Power Anodes for Lithium-Ion Batteries").
http://dx.doi.org/doi:10.1021/nn303145j
Koratkar and his team started investigating graphene as a possible replacement for the graphite used as the anode material in today’s Li-ion batteries. Essentially a single layer of the graphite found commonly in our pencils or the charcoal we burn on our barbeques, graphene is an atom-thick sheet of carbon atoms arranged like a nanoscale chicken-wire fence. In previous studies, Li-ion batteries with graphite anodes exhibited good energy density but low power density, meaning they could not charge or discharge quickly. This slow charging and discharging was because lithium ions could only physically enter or exit the battery’s graphite anode from the edges, and slowly work their way across the length of the individual layers of graphene.
Koratkar’s solution was to use a known technique to create a large sheet of graphene oxide paper. This paper is about the thickness of a piece of everyday printer paper, and can be made nearly any size or shape. The research team then exposed some of the graphene oxide paper to a laser, and other samples of the paper were exposed to a simple flash from a digital camera. In both instances, the heat from the laser or photoflash literally caused mini-explosions throughout the paper, as the oxygen atoms in graphene oxide were violently expelled from the structure. The aftermath of this oxygen exodus was sheets of graphene pockmarked with countless cracks, pores, voids, and other blemishes. The pressure created by the escaping oxygen also prompted the graphene paper to expand five-fold in thickness, creating large voids between the individual graphene sheets.
The researchers quickly learned this damaged graphene paper performed remarkably well as an anode for a Li-ion battery.
Whereas before the lithium ions slowly traversed the full length of graphene sheets to charge or discharge, the ions now used the cracks and pores as shortcuts to move quickly into or out of the graphene—greatly increasing the battery’s overall power density.
Koratkar’s team demonstrated how their experimental anode material could charge or discharge 10 times faster than conventional anodes in Li-ion batteries without incurring a significant loss in its energy density. Despite the countless microscale pores, cracks, and voids that are ubiquitous throughout the structure, the graphene paper anode is remarkably robust, and continued to perform successfully even after more than 1,000 charge/discharge cycles. The high electrical conductivity of the graphene sheets also enabled efficient electron transport in the anode, which is another necessary property for high-power applications.
Koratkar said the process of making these new graphene paper anodes for Li-ion batteries can easily be scaled up to suit the needs of industry. The graphene paper can be made in essentially any size and shape, and the photo-thermal exposure by laser or camera flashes is an easy and inexpensive process to replicate.
The researchers have filed for patent protection for their discovery. The next step for this research project is to pair the graphene anode material with a high-power cathode material to construct a full battery.
Source: Case Western Reserve University
http://www.nanowerk.com/news2/newsid=26421.php
(WO2012087497) POROUS CARBON FOR ELECTROCHEMICAL DOUBLE LAYER CAPACITORS
Applicants:
CORNING INCORPORATED
An electrochemical double layer capacitor electrode comprising microporous carbon, wherein the microporous carbon comprises a median pore width of 1.2 nanometers or less, and a ratio of BET surface area to total pore volume greater than 2200 m2/cm3.
What is claimed is:
1. An electrochemical double layer capacitor electrode comprising microporous carbon, wherein the microporous carbon comprises:
a median pore width of 1.2 nanometers or less; and
a ratio of BET surface area to total pore volume greater than 2200 m2/cm3.
SUMMARY
[0006] The performance of EDLCs comprising carbon based electrodes can be intimately related to the properties of the carbon. Disclosed herein is an improved level of understanding regarding activated carbon materials and their use in EDLCs. The carbon materials disclosed herein have a structure defined by the ratio of total BET surface area to total pore volume, independent of the type of activated carbon. In this disclosure, it is demonstrated that a ratio of total BET surface area to total pore volume of microporous carbon correlates to EDLC performance. Disclosed is a microporous carbon comprising median pore width of 1.2 nanometer or less and a ratio of total BET surface area to total pore volume from 2200 m2/cm3 to 20,000 m2/cm3.
http://patentscope.wipo.int/search/en/detail.jsf?docId=WO2012087497&recNum=1&docAn=US2011062287&queryString=FP:(WO2012087497)&maxRec=1
I wonder how their tech compares to ZBB.
I tried to convince my brother back in 1981 that flow batteries were the way to go. They seem to finally be gaining traction. At some point they will likely run into fuel cells, and at some point it will be hard to tell the difference between a flow battery and a fuel cell.
MVTG has a working electrochemical reactor that converts water and CO2 into formic acid, which can be used to run fuel cells among many other uses. Iceland has a million liter per year electrochemical reactor plant running that is making methanol out of CO2 water and geothermal steam power converted electricity. Methanol can also be used in fuel cells.
US energy storage company EnerVault raises $15.5m
23 Feb 2012
US energy storage company EnerVault has secured investment worth $15.5m through Series B fundraising.
Investors participating in the round include Mitsui Global Investment (MGI), Total Energy Ventures, 3M, TEL Venture Capital and Commercial Energy of California.
The company has also received a grant worth $5.5m from the US Department of Energy, California Energy Commission and the New York State Energy Research and Development Authority.
EnerVault is developing a storage solution based on redox flow battery technology. The company has now raised funding worth $24.5m.
Richard See, investment partner at MGI, said, ‘Redox flow batteries are creating a new paradigm for grid-scale energy storage. Because you scale the power independently of energy storage capacity, you can tailor the system to the needs of the application.
‘EnerVault’s approach expands upon the fundamental configuring and sizing benefits of flow batteries to yield the best approach for utility-scale electricity. The more energy needed, the lower the costs, so it’s ideally suited for a number of situations where more than an hour or two of storage is needed.’
Craig Horne, founder and CEO of EnerVault, added, ‘There is a high level of interest in our systems for addressing the stationary energy storage market. Utilising our patented engineered cascade technology, we are able to develop large-scale energy storage systems needed in a variety of applications with an unmatched combination of cost-effectiveness, safety and reliability.’
Copyright © 2012 NewNet
http://www.newenergyworldnetwork.com/investor-news/renewable-energy-news/by-technology/energy-storage/us-energy-storage-company-enervault-raises-15-5m.html
Take a look at the new hydrogen generator that converts methanol to Hydrogen, with what they claim is 70% reduced cost for hydrogen, using a catalyst....TTEG has the marketing rights, and a Tiawan company has the patents. Details in the TTEG chat room.
Ecomike
Nanowerk: Nanotechnology Nanomaterial Suppliers..
Quantum Materials Corporation (QMC) (Ticker QTMM) is now commercializing a low cost quantum dot technology of a superior quality and characteristics. This revolutionary new quantum dot technique, developed by Dr. Michael S. Wong and colleagues of William Marsh Rice University, Houston TX., has been acquired under an exclusive, world-wide license. Our new synthesis is mass productable using continous flow technology processes developed in conjunction with Access2Flow microreactor technology. QMC's research and development group was instrumental in developing this new scale-up process.
http://www.nanowerk.com/nanotechnology/nanomaterial/suppliers_cplist.php?country=USA&subcat1=qd
Towards artificial photosynthesis for solar hydrogen generation
Posted: Dec 19th, 2011
(Nanowerk News) Photosynthesis is considered the "Holy Grail" in the field of sustainable energy generation because it directly converts solar energy into storable fuel using nothing but water and carbon dioxide (CO2). Scientists have long tried to mimic the underlying natural processes and to optimize them for energy device applications such as photo-electrochemical cells (PEC), which use sunlight to electrochemically split water – and thus directly generate hydrogen, cutting short the more conventional approach using photovoltaic cells for the electrolysis of water.
Traditionally, PEC electrodes are made of semiconducting materials such as metal oxides, some of which are also known for their photocatalytic properties. For quite some time, researchers at Empa's Laboratory for High Performance Ceramics (LHPC) have been investigating nanoparticles of these materials, for instance titanium dioxide (TiO2), for the neutralization of organic pollutants in air and water. Collaborating with colleagues at the University of Basel and at Argonne National Laboratory in the US, they now succeeded in making a nano-bio PEC electrode, consisting of iron oxide conjugated with a protein from blue-green algae (also known as cyanobacteria), which is twice as efficient in water splitting as iron oxide alone (see paper in Advanced Functional Materials: ("Functionalization of Nanostructured Hematite Thin-Film Electrodes with the Light-Harvesting Membrane Protein C-Phycocyanin Yields an Enhanced Photocurrent").
http://dx.doi.org/doi:10.1002/adfm.201101830
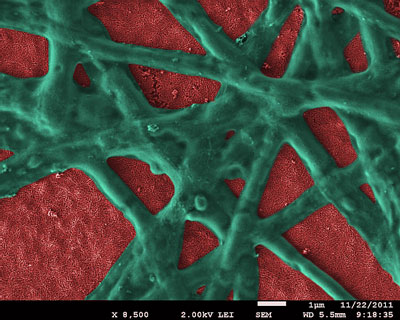
Hematite nanoparticle film (red) with functional phycocyanin network (green) attached. (Image by Dr. E. Vitol, Argonne National Laboratory)
Inspired by photosynthesis
Iron oxide, in particular hematite (a-Fe2O3), is a promising electrode material for PEC because it is susceptible to visible wavelengths and thus uses sunlight more efficiently than photocatalysts like TiO2, which can only use the UV part of solar radiation. What's more, hematite is a low-cost and abundant material.
The second ingredient in the novel electrode "recipe" is phycocyanin, a protein from blue-green algae. "I was inspired by the natural photosynthetic machinery of cyanobacteria where phycocyanin acts as a major light-harvesting component. I wanted to make artificial photosynthesis using ceramics and proteins", recalls Debajeet K. Bora who designed the new electrode during his PhD thesis at Empa. "The concept of hematite surface functionalization with proteins was completely novel in PEC research."
After Bora covalently cross-coupled phycocyanin to hematite nanoparticles that had been immobilized as a thin film, the conjugated hematite absorbed many more photons than without the algal protein. In fact, the induced photocurrent of the hybrid electrode was doubled compared to a "normal" iron oxide electrode.
One tough cookie
Somewhat surprisingly, the light harvesting protein complex does not get destroyed while in contact with a photocatalyst in an alkaline environment under strong illumination. Chemists would have predicted the complete denaturation of biomolecules under such corrosive and aggressive conditions. "Photocatalysts are designed to destroy organic pollutants, which are a burden to the environment. But here we have a different situation", says Artur Braun, group leader at Empa's LHPC and principal investigator of the study. "There seems to be a delicate balance where organic molecules not only survive harsh photocatalytic conditions, but even convey an additional benefit to ceramic photocatalysts: They double the photocurrent. This is a big step forward".
The project was fully funded by the Swiss Federal Office of Energy (SFOE). Bora who will soon have completed his PhD thesis says he will continue what he started at Empa during a postdoc at the University of California, Berkeley, which he will assume early next year.
Source: Empa
http://www.nanowerk.com/news/newsid=23783.php
Haven't nailed it down yet!
But the things I would expect from the market leader to be are 'nano' something and 'MIT' involvement.
In your opinion, what are the plays in this area?
Carbon nanotube-coated sponge makes an excellent supercapacitor
Posted: December 6, 2011
(Nanowerk Spotlight) Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy (for instance as power grid buffer). These devices combine the advantages of conventional capacitors – they can rapidly deliver high current densities on demand – and batteries – they can store a large amount of electrical energy.
Supercapacitors offer a low-cost alternative source of energy to replace rechargeable batteries for various applications, such as power tools, mobile electronics, and electric vehicles. A number of auto makers are exploring the concept of combining supercapacitors with Li-ion batteries as a next generation energy storage system for their electric hybrid vehicles. Although the energy density of capacitors is quite low compared to batteries, their power density is much higher, allowing them to provide bursts of electric energy that can help the new generation of cars to accelerate at comparable or better rates than traditional petrol-only engine vehicles, while achieving a significantly reduced fuel consumption (read more about supercapacitors and other nanotechnologies to mitigate global warming).
Researchers have now fabricated novel high-performance sponge supercapacitors using a simple and scalable method. Reporting their work in a recent edition of Nano Letters ("High-Performance Nanostructured Supercapacitors on a Sponge"), a team at King Abdullah University of Science and Technology (KAUST) in Saudi Arabia, led by Husam N. Alshareef, shows that three-dimensional electrodes potentially have a huge advantage over conventional mixed electrode materials.
http://dx.doi.org/doi:10.1021/nl2023433
"We showed supercapacitor electrodes fabricated on a macroporous structure give a boost to the speed and specific capacitance of the supercapacitors," Alshareef tells Nanowerk. "We worked with sponges because they offer novel exciting characteristics different from paper and textile: first, sponge has much more uniform size of macropores. Second, the cellulose or polyester fibers are interconnected virtually free of junctions. Therefore, continuous coating of conducting nanomaterials is much easier since there are no junctions to cross."

Characterizations of MnO2-CNT-sponge electrodes: (a) an overall view of 3D macroporous hierarchical MnO2-CNT-sponge electrode; (b) MnO2 uniformly deposited on the skeleton of CNT-sponge; (c) high magnification of porous MnO2 nanoparticles on CNT-sponge, inset shows morphology of an individual MnO2 flower-like particle. (Reprinted with permission from American Chemical Society)
The sponge-based supercapacitors were designed and fabricated by the KAUST team using a simple and scalable method. Their fabrication process consisted of four simple steps: First, a piece of commercially available sponge was cleaned by water and acetone; then, after drying, it was cut into small ribbons; these ribbons were coated with a carbon nanotube (CNT) ink; and finally, the researchers electrodeposited manganese dioxide (MnO2) nanoparticles onto the CNT-coated sponge.
"Due to the mechanical flexibility of carbon nanotubes and strong van der Waals interactions between the macroporous sponge cellulose and carbon nanotubes, the nanotubes can be easily coated onto the skeleton of a sponge, rendering the insulating sponge highly conductive by a simple dipping and drying process" explains Alshareef.
Examining the CNT-coated sponge, the researchers found that it still maintained a hierarchical macroporous nature where its intricate assembly of pores remained open to allow the flow of electrolyte. The carbon nanotubes formed a thin layer wrapped around the skeleton of the sponge.
"We also tested the mechanical resilience of the CNT-sponge skeleton by folding, twisting, and stretching it repeatedly," says Alshareef. "After all the mechanical tests, the sponge always reverted to its original shape without any permanent deformation."
The team also tested the electrochemical performance of their sponge supercapacitors. "The macroporous nature of the sponge along with the porous nature of the electrodeposited MnO2 nanoparticles provided a double porous electrode structure giving good conductivity and full accessibility of electrolyte to MnO2, improving the performances of MnO2-CNT-sponge supercapacitors dramatically," says Alshareef. "We found that our sponge supercapacitor exhibits high specific capacitance, ultrafast charge-discharge rate, excellent cycling stability, and good energy and power density, making it a promising electrode for future energy storage systems."
In what could be an interesting aspect for small mobile electronic appliances, the finished supercapacitor sponge turns out to be much lighter than a rigid metal or other flexible substrates with the same area: a sponge with an area of 2 square centimeters and a thickness of 1 millimeter weighs only about 10 mg.
By Michael Berger. Copyright © Nanowerk
http://www.nanowerk.com/spotlight/spotid=23649.php
Batteries get a quick charge with new anode technology
November 3, 2011 By Jared Sagoff
(PhysOrg.com) -- A breakthrough in components for next-generation batteries could come from special materials that transform their structure to perform better over time.
A team of researchers at the U.S. Department of Energy's Argonne National Laboratory, led by Argonne nanoscientist Tijana Rajh and battery expert Christopher Johnson, discovered that nanotubes composed of titanium dioxide can switch their phase as a battery is cycled, gradually boosting their operational capacity. Laboratory tests showed that new batteries produced with this material could be recharged up to half of their original capacity in less than 30 seconds.
By switching out conventional graphite anodes for ones composed of the titanium nanotubes, Rajh and her colleagues witnessed a surprising phenomenon. As the battery cycled through several charges and discharges, its internal structure began to orient itself in a way that dramatically improved the battery's performance.
"We did not expect this to happen when we first started working with the material, but the anode spontaneously adopted the best structure," Rajh said. "There's an internal kind of plasticity to the system that allows it to change as the battery gets cycled."
According to Argonne nanoscientist Hui Xiong, who worked with Rajh to develop the new anode material, titanium dioxide seemed like it would be unlikely to adequately substitute for graphite. "We started with a material that we never thought would have provided a functional use, and it turned into something that gave us the best result possible," she said.
One of the other researchers in Rajh's group, Sanja Tepavcevic, has adopted a similar approach to make a self-improving structure for a sodium-ion nanobattery.
"This is highly unusual material behavior," added Jeff Chamberlain, an Argonne chemist who leads the laboratory's energy storage major initiative. "We're seeing some nanoscale phase transitions that are very interesting from a scientific standpoint, and it is the deeper understanding of these materials' behaviors that will unlock mysteries of materials that are used in electrical energy storage systems."
The reason that titanium dioxide seemed like an implausible solution for battery development lies in the amorphous nature of the material. Because amorphous materials have no internal order, they lack the special electronic properties of highly ordered crystalline materials. However, amorphous materials have not been known to undergo such profound structural transformations during cycling, according to Rajh. Most of the known battery materials undergo the opposite transition: they start out as highly crystalline and pulverize to an amorphous state upon cycling.
Having anodes composed of titanium dioxide instead of graphite also improves the reliability and safety of lithium-ion batteries. In certain cases, lithium can work its way out of solution and deposit on the graphite anodes, causing a dangerous chain reaction known as thermal runaway. "Every type of test we've conducted on titanium anodes has shown them to be exceptionally safe," Chamberlain said.
The Argonne discovery came from collaboration between two of the laboratory's flagship user facilities: the Center for Nanoscale Materials and the Advanced Photon Source. By combining state-of-the-art nanofabrication techniques with high-intensity X-rays to characterize the nanotubes, the Argonne researchers were able to quickly observe this unusual behavior.
Provided by Argonne National Laboratory
http://www.physorg.com/news/2011-11-batteries-quick-anode-technology.html
Shuttle fluoride increases storage capacity
Press Release 151/2011
[GOOGLE TRANSLATION]
KIT scientists are developing a new concept for rechargeable batteries
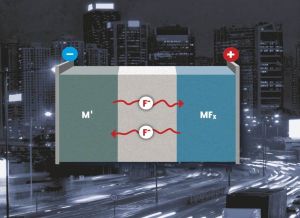
Structure of the fluoride-ion battery: a fluoride electrolyte separates the anode from the cathode metal and metal fluoride. (Photo: KIT)
A new concept for rechargeable batteries, researchers have developed the kit. Based on a fluoride-shuttle - the transfer of fluoride anions between the electrodes - it promises to be a multiple of the memory as it is possible for lithium-ion batteries. Even the reliability can be increased by reducing the need for lithium. In the "Journal of Materials Chemistry" are Dr. Maximilian Fichtner and Dr. Anji Reddy Munnangi the fluoride-ion battery before the first time.[ http://pubs.rsc.org/en/Content/ArticleLanding/2011/JM/c1jm13535j ]
Lithium-ion batteries are widely available - but their capacity is limited. In the future, be in demand with higher energy density, especially for mobile applications, battery systems, which can save more energy with less weight. KIT scientists conduct research, therefore, to alternative systems. A totally new concept for secondary batteries based on metal fluorides have developed Dr. Maximilian Fichtner, head of energy storage systems, and Dr. Anji Reddy Munnangi at the Institute of Nanotechnology (INT) of the KIT.
Metal fluorides can be used both as a conversion materials in lithium-ion batteries. They also allow lithium batteries with fluoride-free electrolyte, anode and cathode made of metal, metal fluoride, which have significantly higher storage capacity and enhanced security features. It takes the fluoride anion in place of lithium-cation the charge transfer. At anode and cathode, it is each to form a metal fluoride, or to its reduction. "There can be several electrons transferred per metal atom, this concept allows extremely high energy densities - up to ten times as high as with current lithium-ion batteries," explained Dr. Maximilian Fichtner.
The KIT researchers are now working to develop battery materials design and architecture to the initial capacity and to improve the cycle stability of the fluoride-ion battery. Another challenge is the development of electrolyte: The solid electrolyte used until now is only suitable for applications at elevated temperatures. The aim is therefore to find a suitable electrolyte for application liquid at room temperature.
M. Anji Reddy and M. Fichtner: Batteries based on fluoride shuttle. Journal of Materials Chemistry. 2011, Advance Article. DOI: 10.1039/C1JM13535J.
The Karlsruhe Institute of Technology (KIT) is a public corporation under the laws of the State of Baden-Wuerttemberg. It fulfills the mission of a university and the mission of a national research center of the Helmholtz Association. The KIT focuses on the knowledge triangle of research - teaching - innovation.
or, 19.10.2011
http://www.kit.edu/besuchen/pi_2011_8281.php
http://www.nanowerk.com/news/newsid=23143.php
Researchers dramatically increase the energy density of graphene-based supercapacitors

Posted: Sep 21st, 2011
(Nanowerk News) Professor Jie Tang, Group Leader of the 1D Nanomaterials Research Group of the Materials Processing Unit, National Institute for Materials Science, and Mr. Qian Cheng, a doctoral student and NIMS Junior Researcher in the same Group, have succeeded in dramatically increasing the energy density of supercapacitors, which are used to store electrical energy. This was realized by developing a new electrode in which graphene nanosheets are stacked in a layered structure with carbon nanotubes sandwiched between the graphene layers. This research achievement is published in the latest issue of Physical Chemistry Chemical Physics.
( http://pubs.rsc.org/en/content/articlelanding/2011/cp/c1cp21910c )
Various new batteries, such as nickel metal hydride batteries, are currently being developed with the aims of achieving higher efficiency and higher energy storage for electric power supplies. In comparison with batteries, capacitors have a larger output power density to enable rapid charging, excellent durability to allow operations in both higher and lower extreme temperatures, better cyclicity for recharging repeatedly over a long period, and are also safer. However, it has been as a great technical challenge to realize high energy density due to the relatively low specific capacity of the conventional capacitor devices.
In order to achieve a revolutionary increase in density of energy storage, Professor Tang and her team, in collaboration with Professor Lu-Chang Qin of the University of North Carolina at Chapel Hill in the United States, have designed and developed a graphene-based composite structure, in which graphene is used as the base material of the capacitor electrodes and carbon nanotubes (CNT) are inserted between the graphene sheets. In this structure graphene offers a far larger specific surface area (2630 m2/g) than the conventional materials and the CNTs function as spacers as well as conducting paths to enable adsorption of a larger quantity of electrolyte ions on the graphene surface. With this graphene-CNT composite as the capacitor electrodes, Professor Tang has obtained a high energy density of 62.8 Wh/kg and output power density of 58.5 kW/kg using organic electrolyte. By using an ionic liquid as the electrolyte, they have achieved an energy density of 155.6 Wh/kg, which is comparable to that of nickel metal hydride batteries.
Among the many industrial applications of capacitors, the new capacitors developed in this research offer promises as power sources for electric and hybrid vehicles, which require high energy density. As the current production processes are also inexpensive and can be scaled up, large expectations are placed on practical applications.
Source: NIMS
http://www.nanowerk.com/news/newsid=22807.php
Solar rays could replace petroleum fuels, research shows
September 13, 2011 By Ken Klabunde
(PhysOrg.com) -- Alternative fuel sources for cars may have a glowing future as a Kansas State University graduate student is working to replace petroleum fuels with ones made from sunlight.
Yen-Ting Kuo, a doctoral candidate in chemistry, Taiwan, has spent several years in K-State's chemistry program working to create new materials that better use sunlight in chemical reaction processes to generate energy.
"People tend to think of chemistry as test-tube experiments and not really creating practical things. That's just not true," Kuo said. "A big focus now is on 'green chemistry.' This means wanting to have the same quality of life that we have right now, but using chemistry to replace some things with materials that are more eco-friendly, such as biodegradable products or clean fuel."
As a way to advance the clean fuel research, Kuo is making and studying metal-oxide catalysts that react with light. These catalysts, called photocatalysts, cause a chemical reaction when triggered by sunlight, but are not destroyed during the reaction. Photocatalysts are crucial to producing new fuels, like solar gasoline, which use hydrogen.
To make solar gasoline, sunlight is channeled into a tank of water that contains photocatalysts. The sunlight triggers the photocatalysts to react with the water. This reaction causes the water to split into hydrogen and oxygen. When the hydrogen is combined with carbon monoxide it forms a synthetic gas -- called syngas -- that is the basic building block in fossil fuel and can be used to power cars.
In recent years solar gasoline has been getting more mileage as more international laboratories attempt to improve and perfect the process. But developing a photocatalyst that efficiently uses sunlight to create a chemical reaction and produce hydrogen is proving difficult for researchers. It also is needed for production to reach commercial levels. Kuo is working to solve that problem by creating and analyzing new photocatalysts in the lab.
To make a photocatalyst, Kuo mixes various elements in powdered form, and then cooks them at temperatures between 700 degrees Celsius and 850 degrees Celsius.
Once the material is made, its structure is studied with a transmission electron microscope and ultraviolet spectrums. Doing this allows Kuo to look at ways to structurally improve the photocatalyst and its performance.
In addition to improving the material's photocatalytic properties -- which will intensify reaction with the sunlight -- Kuo focuses on increasing the material's surface area. An increased surface area means bigger and better reactions, and a material with a high surface area and with high photocatalytic properties could mean a bright future for solar gasoline and other alternative fuels.
Engineering a photocatalyst that efficiently splits water into hydrogen and oxygen could also be a boon to fuel cell technology, Kuo said. Fuel cells operate by essentially reversing the chemical reaction that's used to split water. Hydrogen is converted into electrical power, and water is given off as a byproduct.
"Even though the mature technology of fuel cells is in the near future, the source of hydrogen is still a question since most of the hydrogen sources now are from petroleum," Kuo said. "Therefore, water splitting using photocatalysts is one of the solutions providing a new pathway to obtain hydrogen."
Kuo came to K-State after reading work published from his adviser, Ken Klabunde. Klabunde, a university distinguished professor of chemistry, is an expert in turning chemistry into new environmentally-friendly materials. He's created inorganic materials and nanotechnology that filter water and air; control odor, bacteria and viruses; and detoxify hazardous chemical spills.
Kuo will defend his dissertation, "Novel photocatalytic water splitting with the N-doped In2O3/TiO2 D10-D0 configuration composite oxide semiconductors," in mid-September. He then will begin a postdoctoral position at the University of Michigan, one of the few universities in the U.S. to study solar gasoline.
Provided by Kansas State University
http://www.physorg.com/news/2011-09-solar-rays-petroleum-fuels.html
Breakthrough in hydrogen fuel cells: Chemists develop way to safely store, extract hydrogen
August 30, 2011

A team of USC scientists has developed a robust, efficient method of using hydrogen as a fuel source.
Hydrogen makes a great fuel because of it can easily be converted to electricity in a fuel cell and because it is carbon free. The downside of hydrogen is that, because it is a gas, it can only be stored in high pressure or cryogenic tanks.
In a vehicle with a tank full of hydrogen, "if you got into a wreck, you'd have a problem," said Travis Williams, assistant professor of chemistry at the USC Dornsife College.
A possible solution is to store hydrogen in a safe chemical form. Earlier this year, Williams and his team figured out a way to release hydrogen from an innocuous chemical material — a nitrogen-boron complex, ammonia borane — that can be stored as a stable solid.
Now the team has developed a catalyst system that releases enough hydrogen from its storage in ammonia borane to make it usable as a fuel source. Moreover, the system is air-stable and re-usable, unlike other systems for hydrogen storage on boron and metal hydrides.
The research was published this month in the Journal of the American Chemical Society.
"Ours is the first game in town for reusable, air stabile ammonia borane dehydrogenation," Williams said, adding that the USC Stevens Institute is in the process of patenting the system.
The system is sufficiently lightweight and efficient to have potential fuel applications ranging from motor-driven cycles to small aircraft, he said.
More information: A Robust, Air-Stable, Reusable Ruthenium Catalyst for Dehydrogenation of Ammonia Borane, J. Am. Chem. Soc., Article ASAP.
DOI: 10.1021/ja2058154
http://dx.doi.org/10.1021/ja2058154
Abstract
We describe an efficient homogeneous ruthenium catalyst for the dehydrogenation of ammonia borane (AB). This catalyst liberates more than 2 equiv of H2 and up to 4.6 system wt % H2 from concentrated AB suspensions under air. Importantly, this catalyst is robust, delivering several cycles of dehydrogenation at high [AB] without loss of catalytic activity, even with exposure to air and water.
Provided by University of Southern California
http://www.physorg.com/news/2011-08-breakthrough-hydrogen-fuel-cells-chemists.html
Novel alloy could produce hydrogen fuel from sunlight
Posted: Aug 30th, 2011
(Nanowerk News) Scientists from the University of Kentucky and the University of Louisville have determined that an inexpensive semiconductor material can be "tweaked" to generate hydrogen from water using sunlight.
The research, funded by the U.S. Department of Energy, was led by Professors Madhu Menon and R. Michael Sheetz at the UK Center for Computational Sciences, and Professor Mahendra Sunkara and graduate student Chandrashekhar Pendyala at the UofL Conn Center for Renewable Energy Research. Their findings were published Aug. 1 in the Physical Review Journal ("Visible-light absorption and large band-gap bowing of GaN1-xSbx from first principles") http://dx.doi.org/doi:10.1103/PhysRevB.84.075304 .
The researchers say their findings are a triumph for computational sciences, one that could potentially have profound implications for the future of solar energy.
Using state-of-the-art theoretical computations, the UK-UofL team demonstrated that an alloy formed by a 2 percent substitution of antimony (Sb) in gallium nitride (GaN) has the right electrical properties to enable solar light energy to split water molecules into hydrogen and oxygen, a process known as photoelectrochemical (PEC) water splitting. When the alloy is immersed in water and exposed to sunlight, the chemical bond between the hydrogen and oxygen molecules in water is broken. The hydrogen can then be collected.
"Previous research on PEC has focused on complex materials," Menon said. "We decided to go against the conventional wisdom and start with some easy-to-produce materials, even if they lacked the right arrangement of electrons to meet PEC criteria. Our goal was to see if a minimal 'tweaking' of the electronic arrangement in these materials would accomplish the desired results."
Gallium nitride is a semiconductor that has been in widespread use to make bright-light LEDs since the 1990s. Antimony is a metalloid element that has been in increased demand in recent years for applications in microelectronics. The GaN-Sb alloy is the first simple, easy-to-produce material to be considered a candidate for PEC water splitting. The alloy functions as a catalyst in the PEC reaction, meaning that it is not consumed and may be reused indefinitely. UofL and UK researchers are currently working toward producing the alloy and testing its ability to convert solar energy to hydrogen.
Hydrogen has long been touted as a likely key component in the transition to cleaner energy sources. It can be used in fuel cells to generate electricity, burned to produce heat, and utilized in internal-combustion engines to power vehicles. When combusted, hydrogen combines with oxygen to form water vapor as its only waste product. Hydrogen also has wide-ranging applications in science and industry.
Because pure hydrogen gas is not found in free abundance on Earth, it must be manufactured by unlocking it from other compounds. Thus, hydrogen is not considered an energy source, but rather an "energy carrier." Currently, it takes a large amount of electricity to generate hydrogen by water splitting. As a consequence, most of the hydrogen manufactured today is derived from non-renewable sources such as coal and natural gas.
Sunkara says the GaN-Sb alloy has the potential to convert solar energy into an economical, carbon-free source for hydrogen.
"Hydrogen production now involves a large amount of CO2 emissions," Sunkara said. "Once this alloy material is widely available, it could conceivably be used to make zero-emissions fuel for powering homes and cars and to heat homes."
Menon says the research should attract the interest of other scientists across a variety of disciplines.
"Photocatalysis is currently one of the hottest topics in science," Menon said. "We expect the present work to have a wide appeal in the community spanning chemistry, physics and engineering."
Source: University of Kentucky
http://www.nanowerk.com/news/newsid=22579.php
A microporous polymer is an unusually powerful supercapacitor
Posted: Aug 22nd, 2011
(Nanowerk News) For future electric vehicles, powerful notebook computers, and other portable devices, we need a new generation of energy storage materials that are better suited to modern needs than current rechargeable batteries. The best materials for this are known as supercapacitors. A team led by Dinglin Jiang at the National Institutes of Natural Sciences in Okazaki (Japan) has now introduced a new material with outstanding supercapacitor properties in the journal Angewandte Chemie ("Supercapacitive Energy Storage and Electric Power Supply Using an Aza-Fused p-Conjugated Microporous Framework") http://onlinelibrary.wiley.com/doi/10.1002/anie.201103493/abstract;jsessionid=7C17789C27B6042C836FEE7B56F8A365.d03t03 .
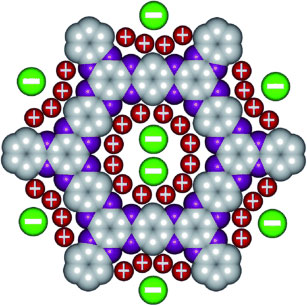
A p-conjugated microporous polymer with aza-fused framework is reported. The porous framework is conductive and allows electrolyte ions to move into the pores because of structural features.
Emission-free electric cars are well suited for drives around the city; for long stretches, however, this has not been the case. The problem stems from the small amount of energy that can be stored, which covers only short distances before requiring a charge, and the amount of current that can be delivered, which limits the speed and acceleration of the vehicles. Supercapacitors could overcome these challenges because they combine the advantages of earlier capacitors and batteries: like a capacitor, they can deliver high current densities on demand while storing a large amount of electric energy like a battery.
Supercapacitors work on a different charge-storage principle than rechargeable batteries, and consist of electrochemical double layers on electrodes, which are wetted by an electrolyte. When a voltage is applied, ions of opposite charge collect on both electrodes to form wafer-thin zones of immobilized charge carriers. In contrast to a battery, there is only a shift of charge; no chemical transformation occurs. Various materials are suitable for supercapacitors, but the truly perfect material has yet to be found. The researchers in Japan have now reached an important milestone along the way.
There is one class of materials with interesting properties: special microporous, framework-like, organic polymers. Their double bonds are arranged in such a way that some of their electrons can move freely over extended regions of the framework as an "electron cloud". Such materials are thus conducting. A large inner surface area is important for the formation of electrostatic charge-separation layers in the pores. Jiang and his team have now synthesized a nitrogen-containing framework with a pore size optimal for allowing ions to flow in and out rapidly – a requirement for rapid charging and discharging. The nitrogen centers interact with the electrolyte ions, thus favoring the accumulation of charge and the movement of ions.
The interplay between these different advantageous properties provides the new material with an unusually high charge storage capacity and high energy density. Jiang and his co-workers were able to demonstrate that their microporous frameworks can withstand many charge/discharge cycles.
Source: Wiley-VCH Verlag
http://www.nanowerk.com/news/newsid=22487.php
New energy storage device could recharge electric vehicles in minutes
August 19, 2011 by Lisa Zyga

Compared with supercapacitors and batteries, SMCs (with three different electrode thicknesses shown) offer both a high power density and high energy density. Image copyright: Jang, et al. ©2011 American Chemical Society
(PhysOrg.com) -- It has all the appearances of a breakthrough in battery technology, except that it’s not a battery. Researchers at Nanotek Instruments, Inc., and its subsidiary Angstron Materials, Inc., in Dayton, Ohio, have developed a new paradigm for designing energy storage devices that is based on rapidly shuttling large numbers of lithium ions between electrodes with massive graphene surfaces. The energy storage device could prove extremely useful for electric vehicles, where it could reduce the recharge time from hours to less than a minute. Other applications could include renewable energy storage (for example, storing solar and wind energy) and smart grids.
The researchers call the new devices "graphene surface-enabled lithium ion-exchanging cells," or more simply, "surface-mediated cells" (SMCs). Although the devices currently use unoptimized materials and configurations, they can already outperform Li-ion batteries and supercapacitors. The new devices can deliver a power density of 100 kW/kgcell, which is 100 times higher than that of commercial Li-ion batteries and 10 times higher than that of supercapacitors. The higher the power density, the faster the rate of energy transfer (resulting in a faster recharge time). In addition, the new cells can store an energy density of 160 Wh/kgcell, which is comparable to commercial Li-ion batteries and 30 times higher than that of conventional supercapacitors. The greater the energy density, the more energy the device can store for the same volume (resulting in a longer driving range for electric vehicles).
“Given the same device weight, the current SMC and Li-ion battery can provide an electric vehicle (EV) with a comparable driving range,” Bor Z. Jang, co-founder of Nanotek Instruments and Angstron Materials, told PhysOrg.com. “Our SMCs, just like the current Li-ion batteries, can be further improved in terms of energy density [and therefore range]. However, in principle, the SMC can be recharged in minutes (possibly less than one minute), as opposed to hours for Li-ion batteries used in current EVs.”
Jang and his coauthors at Nanotek Instruments and Angstron Materials have published the study on the next-generation energy storage devices in a recent issue of Nano Letters. Both companies specialize in nanomaterial commercialization, with Angstron being the world’s largest producer of nano graphene platelets (NGPs).
As the researchers explain in their study, batteries and supercapacitors each have their respective strengths and weaknesses when it comes to energy storage. While Li-ion batteries provide a much higher energy density (120-150 Wh/kgcell) than supercapacitors (5 Wh/kgcell), the batteries deliver a much lower power density (1 kW/kgcell compared to 10 kW/kgcell). Many research groups have made efforts to increase the power density of Li-ion batteries and increase the energy density of supercapacitors, but both areas still have significant challenges. By providing a fundamentally new framework for energy storage devices, the SMCs could enable researchers to bypass these challenges.
“The development of this new class of energy storage devices bridges the performance gap between a Li-ion battery and a supercapacitor,” Jang said. “More significantly, this fundamentally new framework for constructing energy storage devices could enable researchers to achieve both the high energy density and high power density without having to sacrifice one to achieve the other.”

The large surface areas of the SMCs’ electrodes enable rapid shuttling of large numbers of ions between electrodes, resulting in a fast recharge time. Image copyright: Jang, et al. ©2011 American Chemical Society
The key to the SMCs’ performance is a cathode and anode that contain very large graphene surfaces. When fabricating the cell, the researchers put lithium metal (in the form of particles or foil) at the anode. During the first discharge cycle, the lithium is ionized, resulting in a much larger number of lithium ions than in Li-ion batteries. As the battery is used, the ions migrate through a liquid electrolyte to the cathode, where the ions enter the pores and reach the large graphene surface inside the cathode. During recharging, a massive flux of lithium ions quickly migrates from the cathode to the anode. The electrodes’ large surface areas enable the rapid shuttling of large numbers of ions between electrodes, resulting in their high power and energy densities.
As the researchers explain, the exchange of lithium ions between the porous electrodes’ surfaces (and not in the bulk of the electrode, as in batteries) completely removes the need for the time-consuming process of intercalation. In this process, the lithium ions must be inserted inside the electrodes, which dominates the charging time of batteries.
Although in this study the researchers prepared different types of graphene (oxidized, and reduced single-layer and multilayer) from a variety of different types of graphite, further analysis of the materials and configuration is needed for optimizing the device. For one thing, the researchers plan to further investigate the cells’ cycling lifetime. So far, they found that the devices could retain 95% capacity after 1,000 cycles, and even after 2,000 cycles showed no evidence of dendrite formation.
The researchers also plan to investigate the relative roles of different lithium storage mechanisms on the device’s performance.
“We do not anticipate any major hurdle to commercialization of the SMC technology,” Jang said. “Although graphene is currently sold at a premium price, Angstron Materials, Inc., is actively engaged in scaling up the production capacity of graphene. The production costs of graphene are expected to be dramatically reduced within the next 1-3 years.”
More information: Bor Z. Jang, et al. “Graphene Surface-Enabled Lithium-Ion Exchanging Cells: Next-Generation High-Power Energy Storage Devices.” Nano Letters. DOI:10.1021/nl2018492
http://dx.doi.org/10.1021/nl2018492
http://www.physorg.com/news/2011-08-energy-storage-device-recharge-electric.html
when is Covanta going to make something out of that piece of garbage Alphakat system GEYI is trying to get working?
Hybrid Solar System Makes Rooftop Hydrogen

This is the hybrid system schematic. (Credit: Nico Hotz)
ScienceDaily (Aug. 9, 2011) — While roofs across the world sport photovoltaic solar panels to convert sunlight into electricity, a Duke University engineer believes a novel hybrid system can wring even more useful energy out of the sun's rays.
Instead of systems based on standard solar panels, Duke engineer Nico Hotz proposes a hybrid option in which sunlight heats a combination of water and methanol in a maze of glass tubes on a rooftop. After two catalytic reactions, the system produces hydrogen much more efficiently than current technology without significant impurities. The resulting hydrogen can be stored and used on demand in fuel cells.
For his analysis, Hotz compared the hybrid system to three different technologies in terms of their exergetic performance. Exergy is a way of describing how much of a given quantity of energy can theoretically be converted to useful work.
"The hybrid system achieved exergetic efficiencies of 28.5 percent in the summer and 18.5 percent in the winter, compared to 5 to 15 percent for the conventional systems in the summer, and 2.5 to 5 percent in the winter," said Hotz, assistant professor of mechanical engineering and materials science at Duke's Pratt School of Engineering.
The paper describing the results of Hotz's analysis was named the top paper during the ASME Energy Sustainability Fuel Cell 2011 conference in Washington, D.C. Hotz recently joined the Duke faculty after completing post-graduate work at the University of California-Berkeley, where he analyzed a model of the new system. He is currently constructing one of the systems at Duke to test whether or not the theoretical efficiencies are born out experimentally.
Hotz's comparisons took place during the months of July and February in order to measure each system's performance during summer and winter months.
Like other solar-based systems, the hybrid system begins with the collection of sunlight. Then things get different. While the hybrid device might look like a traditional solar collector from the distance, it is actually a series of copper tubes coated with a thin layer of aluminum and aluminum oxide and partly filled with catalytic nanoparticles. A combination of water and methanol flows through the tubes, which are sealed in a vacuum.
"This set-up allows up to 95 percent of the sunlight to be absorbed with very little being lost as heat to the surroundings," Hotz said. "This is crucial because it permits us to achieve temperatures of well over 200 degrees Celsius within the tubes. By comparison, a standard solar collector can only heat water between 60 and 70 degrees Celsius."
Once the evaporated liquid achieves these higher temperatures, tiny amounts of a catalyst are added, which produces hydrogen. This combination of high temperature and added catalysts produces hydrogen very efficiently, Hotz said. The resulting hydrogen can then be immediately directed to a fuel cell to provide electricity to a building during the day, or compressed and stored in a tank to provide power later.
The three systems examined in the analysis were the standard photovoltaic cell which converts sunlight directly into electricity to then split water electrolytically into hydrogen and oxygen; a photocatalytic system producing hydrogen similar to Hotz's system, but simpler and not mature yet; and a system in which photovoltaic cells turn sunlight into electricity which is then stored in different types of batteries (with lithium ion being the most efficient).
"We performed a cost analysis and found that the hybrid solar-methanol is the least expensive solution, considering the total installation costs of $7,900 if designed to fulfill the requirements in summer, although this is still much more expensive than a conventional fossil fuel-fed generator," Hotz said.
Costs and efficiencies of systems can vary widely depending on location -- since the roof-mounted collectors that could provide all the building's needs in summer might not be enough for winter. A rooftop system large enough to supply all of a winter's electrical needs would produce more energy than needed in summer, so the owner could decide to shut down portions of the rooftop structure or, if possible, sell excess energy back to the grid.
"The installation costs per year including the fuel costs, and the price per amount of electricity produced, however showed that the (hybrid) solar scenarios can compete with the fossil fuel-based system to some degree," Hotz said. 'In summer, the first and third scenarios, as well as the hybrid system, are cheaper than a propane- or diesel-combusting generator."
This could be an important consideration, especially if a structure is to be located in a remote area where traditional forms of energy would be too difficult or expensive to obtain.
Hotz's research was supported by the Swiss National Science Fund. Joining him in the study were UC-Berkeley's Heng Pan and Costas Grigoropoulos, as well as Seung H. Ko of the Korea Advanced Institute of Science and Technology, Daejon.
http://www.sciencedaily.com/releases/2011/08/110809132232.htm
See also EP 2143490:
http://worldwide.espacenet.com/publicationDetails/biblio?FT=D&date=20100113&DB=EPODOC&locale=en_EP&CC=EP&NR=2143490A1&KC=A1

Power pillars: This battery electrode, shown in cross section under an electron microscope, consists of nanoscale tin pillars sandwiched between sheets of graphene.
Credit: Lawrence Berkeley
Nanoscale Pillars Could Have a Big Role in Future Batteries
A new fabrication technique lets batteries use tin electrodes, and store more energy.
TUESDAY, AUGUST 2, 2011
BY KATHERINE BOURZAC
Tin, silicon, and a few other elements have long been languishing on chemists' list of electrode materials that could, in theory, help lithium-ion batteries hold more energy. A new way of structuring these materials could at last allow them to be used in this way.
Researchers at the Lawrence Berkeley National Laboratory made tin electrodes by using layers of graphene to protect the normally fragile tin. These first tin electrodes are a sign that materials scientists have made a great deal of progress in using nanoscale structures to improve batteries.
Making battery electrodes from tin or silicon can boost the battery's overall energy storage. That's because such materials can take in more lithium during charging and recharging than carbon, which is normally used. But silicon and tin tend to be unstable as electrodes. Tin takes up so much lithium that it expands in volume by a factor of two to three during charging. "This forms cracks, and the tin leaks into the electrolyte and is lost," says Yuegang Zhang, a scientist at Lawrence Berkeley.
Zhang's clever solution is to layer the tin between sheets of graphene, single-atom-thick sheets of carbon mesh. Graphene is highly conductive, and while it's flexible, it's also the strongest material ever tested.
The tin-graphene electrode consists of two layers of tin nanopillars sandwiched between three sheets of graphene. The pillars help the electrode remain stable: instead of fracturing, the tin expands and contracts during charging without breaking. The space between the pillars means there's plenty of room for the battery's electrolyte to move around, which ensures fast charging speeds.
Zhang's group has made prototype batteries featuring these electrodes. The prototype tin-graphene batteries can charge up in about 10 minutes and store about 700 milliamp-hours per gram of charge. This storage capacity is maintained over 30 charge cycles. The batteries will ultimately need to hold their performance for hundreds of charge cycles. "The performance they have is quite reasonable, and this has a pretty clear application in existing batteries," says Yi Cui, associate professor of materials science at Stanford University. Cui was not involved with the work.
Several other research groups are working on promising battery materials that include nanoscale structures. Cui has founded a company called Amprius to commercialize another kind of battery anode that features silicon nanowires. The nanostructure of these wires also helps the fragile material remain stable as it takes up and releases lithium. Another group, led by Pulickel Ajayan at Rice, recently built a nanostructured battery that incorporates a tin electrode, in this case integrating the electrodes and the electrolyte on individual nanowires. Arrayed together, these nanowires could make long-lasting microbatteries for small devices such as sensors.
Zhang is working to demonstrate the use of the nanopillar structure with other fragile electrode materials, including silicon. The process may add to the cost of battery production, but the performance gains could offset the potential additional cost. "People typically assume that a fancy nanoscale structure will cost more, but it may not," says Zhang.
http://www.technologyreview.com/energy/38205/?nlid=nldly&nld=2011-08-03
Graphite + water = the future of energy storage
Posted: Jul 15th, 2011
(Nanowerk News) A combination of two ordinary materials – graphite and water – could produce energy storage systems that perform on par with lithium ion batteries, but recharge in a matter of seconds and have an almost indefinite lifespan.
Dr Dan Li [ http://www.monash.edu.au/news/search?q=%22Dan%20Li%22 ], of the Monash University Department of Materials Engineering, and his research team have been working with a material called graphene, which could form the basis of the next generation of ultrafast energy storage systems.
"Once we can properly manipulate this material, your iPhone, for example, could charge in a few seconds, or possibly faster." said Dr Li.
Graphene is the result of breaking down graphite, a cheap, readily available material commonly used in pencils, into layers one atom thick. In this form, it has remarkable properties.
Graphene sheets.
Graphene is strong, chemically stable, an excellent conductor of electricity and, importantly, has an extremely high surface area.
Dr Li said these qualities make graphene highly suitable for energy storage applications.
"The reason graphene isn't being used everywhere is that these very thin sheets, when stacked into a usable macrostructure, immediately bond together, reforming graphite. When graphene restacks, most of the surface area is lost and it doesn't behave like graphene anymore."
Now, Dr Li and his team have discovered the key to maintaining the remarkable properties of separate graphene sheets: water. Keeping graphene moist – in gel form – provides repulsive forces between the sheets and prevents re-stacking, making it ready for real-world application.
"The technique is very simple and can easily be scaled up. When we discovered it, we thought it was unbelievable. We're taking two basic, inexpensive materials – water and graphite – and making this new nanomaterial with amazing properties," said Dr Li.
When used in energy devices, graphene gel significantly outperforms current carbon-based technology, both in terms of the amount of charge stored and how fast the charges can be delivered.
Dr Li said the benefits of developing this new nanotechnology extend beyond consumer electronics.
"High-speed, reliable and cost-effective energy storage systems are critical for the future viability of electricity from renewable resources. These systems are also the key to large-scale adoption of electrical vehicles.
"Graphene gel is also showing promise for use in water purification membranes, biomedical devices and sensors."
Dr Li has been working with graphene since 2006 and his team's research findings have recently been published in a number of prestigious journals including Advanced Materials, Angewandte Chemie and Chemical Communications.
Source: Monash University
http://www.nanowerk.com/news/newsid=22128.php
US Patent 7,974,075 Peng, et al., July 5, 2011
Electrode of supercapacitor and method for manufacturing the same
Abstract
A method for manufacturing an electrode of a supercapacitor is provided. First, a poly(acrylonitrile) (PAN) fabric is provided. The PAN fabric includes a plurality of PAN fibers each having a diameter of about 50-500 nm. Then, the PAN fabric undergoes a heat treatment so that the PAN fibers are carbonized to form a carbon fiber textile. The carbon fiber fabric includes a plurality of carbon fibers each having a diameter of about 50-500 nm. The surface of each carbon fiber is nano-porous having a plurality of nano pores of about 1-50 nm in diameter. The total surface area of the nano pores account for about 85-95% of the total surface area of the carbon fibers. The carbon fiber fabric is then cut to acquire the electrode of the supercapacitor.
Inventors:
Peng; Chao-Chun (Hsinchu, TW), Chang; Haw-Jer (Hsinchuang, TW), Lin; Wen-Ting (Caotun Township, Nantou County, TW)
Assignee:
Taiwan Textile Research Institute (Tu-Chen, Taipei Hsien, TW)
Appl. No.:
12/334,737
Filed:
December 15, 2008
Foreign Application Priority Data
Dec 31, 2007 [TW]
96151466 A
What is claimed is:
1. A method for manufacturing an electrode of a supercapacitor, comprising: providing a poly(acrylonitrile) fabric, the poly(acrylonitrile) fabric comprising a plurality of poly(acrylonitrile) fibers each having a diameter of about 50-500 nm; heat treating the poly(acrylonitrile) fabric to form a carbon fiber fabric comprising a plurality of carbon fibers each having a diameter of about 50-500 nm, wherein a surface of each of the carbon fibers has a plurality of nano pores of about 1-50 nm in diameter, and a total surface area of the nano pores accounts for about 85-95% of a total surface area of the carbon fibers; and cutting the carbon fiber fabric to acquire the electrode.
http://is.gd/gAgvzR
Nanostructures Could Result In Cheaper Electric-Car Batteries
Chinese startup develops new, low-cost ways to improve the properties of lithium-iron phosphate, a leading electrode material.
TUESDAY, JULY 12, 2011
BY KEVIN BULLIS
A Beijing startup called Wuhe is making electrode materials and batteries that could lower the cost of electric vehicles. The company uses nanostructures for battery materials that, like other recent nanostructures, let the materials deliver the large bursts of power needed for acceleration while maintaining energy storage capacity. But the Wuhe advance also makes the materials easier to work with than similar electrode materials, and as a result, it could cut battery-cell manufacturing costs by 10 percent.
Battery packs are the most expensive item on electric cars such as the Tesla Roadster and the Nissan Leaf. The cost either makes electric cars too expensive for most people, or it prompts automakers to use small battery packs, which limits the range of the cars.
To reduce battery costs and improve their performance, Wuhe founder Yu-Guo Guo, a professor of chemistry at the Chinese Academy of Sciences in Beijing, has developed new, low-cost ways to improve the properties of lithium-iron phosphate, one of the leading lithium-ion battery electrode materials, and other promising electrode materials.
Ordinarily, the conductivity of lithium-iron phosphate is too low to be useful. The conductivity can be increased by milling it into extremely fine nanoscale powders—as companies such as A123 Systems do. Because the particles are small, electrons or lithium ions—both of which are necessary to create current—can move in and out of them quickly. But this powder is difficult to work with, which raises manufacturing costs.
Guo's solution has been to incorporate iron-phosphate nanoparticles, which are easier to pack closely, and are less likely become airborne, but retain high conductivity. He isn't giving precise details, but he says the technology is based on some of his earlier published work. In one example of that work, he embedded the nanoparticles in larger particles made of porous carbon. The carbon conducts electricity well, and the pores host electrolyte materials that conduct lithium ions well.
Guo says the materials are only 10 to 20 percent more expensive to make than bulk lithium-iron phosphate. But they can deliver about twice as much power as the bulk material, and make twice as much of the energy in lithium-iron phosphate available, roughly doubling the energy storage capacity. Per watt-hour, the materials cost the same as other lithium-iron-phosphate electrode materials, he says. But since the material is easier to work with, it will cut the cost of incorporating the materials into battery cells.
Wuhe, which Guo founded at the end of last year, already has the capacity to produce 300 metric tons of electrode material a year, enough for about 30 million standard lithium-ion battery cells. It also makes battery cells, with the first application being electric bicycles. It can currently make enough cells for roughly 500 electric cars a year.
Jeff Dahn, professor of physics and chemistry at Dalhousie University, says that, based on the company's performance figures, the iron-phosphate batteries will be "very useful" and could perform better than the batteries used now in the electric Chevrolet Volt. And he predicts Wuhe will find a market.
http://www.technologyreview.com/energy/38005/?nlid=nldly&nld=2011-07-12
Carbon nanofibers on graphene make promising high-performance electrode materials
Posted: Jul 11th, 2011
(Nanowerk News) Lithium-ion batteries are an important energy storage technology for portable electronics and electric vehicles. The performance of these batteries depends on the density at which they can store energy and how fast they can be charged and discharged. One way to improve these properties is through the use of nanostructured electrodes. Zhuang-Jun Fan and colleagues from Harbin Engineering University and other institutions in China have now developed three-dimensional, carbon-based nanostructures with enhanced capacity for storing lithium ions and high-rate charge/discharge cycling characteristics ("Nanographene-Constructed Carbon Nanofibers Grown on Graphene Sheets by Chemical Vapor Deposition: High-Performance Anode Materials for Lithium Ion Batteries"insert-text-here).
Graphene is a promising energy-storage material because of its high specific surface area and chemical stability. However, as Zhuang-Jun explains, graphene has not yet been used to its full potential in battery technologies because of the loss of surface area available for energy storage due to graphene aggregation. To address this issue, the researchers used a chemical vapor deposition approach in a fluidized bed reactor to grow one-dimensional carbon nanofibers on graphene nanosheets.
"The carbon nanofibers improve not only graphene aggregation," says Zhuang-Jun, "but also the overall conductivity of the graphene networks." The resulting graphene–carbon nanofiber hybrid structure contains many cavities, open tips and graphene platelets with exposed edges (see image), providing high capacity for lithium ion storage.
Zhuang-Jun and his co-workers grew the carbon nanofibers from nanoparticles of cobalt dispersed on sheets of graphene oxide. The cobalt nanoparticles act as catalysts for the growth of the nanofibers as the graphene oxide breaks up in the high-temperature growth reactor chamber. The outer shell of the resultant fibers consists of graphene platelets with a spacing of 0.342 nm. According to Zhuang-Jun, the graphene platelets arrange themselves roughly perpendicular to the fiber axis, which is favorable for the efficient diffusion of lithium ions. When used as an electrode in lithium-ion batteries, this structure allows for efficient charging and discharging processes, which occur through the insertion and extraction of lithium ions.
"The as-prepared hybrid material shows high reversible capacity, high-rate performance and cycling stability superior to pure graphene, natural graphite and carbon nanotubes," Zhuang-Jun remarks. These properties make the hybrid material a promising candidate as an electrode material for high-performance lithium-ion batteries, full cell and supercapacitors.
Source: Tokyo Institute of Technology
http://www.nanowerk.com/news/newsid=22057.php
Rectenna and CNTs for solar cells
I have high and fond hopes for these!
United States Patent Application 20110163920
Kind Code A1
CUTLER; PAUL A. July 7, 2011
METHOD AND APPARATUS FOR AN OPTICAL FREQUENCY RECTIFIER
Abstract
The present invention is a method and apparatus for receiving and converting incident radiation into DC current. The method begins with selection of an antenna having a terminal tip with a sharp edge. The antenna is layered with a substrate and a first coating. A first electrode and a metallic/mCNT antenna are layered on the first coating, and a plasmonic layer is then added. A gap is formed which is bounded on one side by the terminal ends of the plasmonic layer and the first coating, and a second electrode and a second coating on the other. The second electrode is layered upon the second coating which is layered upon the substrate. A set of AC currents is induced along the length of the antenna. The method then calculates whether or not the induced AC currents are large enough to create voltages for field emission. If the voltages are large enough, then a forward bias and a reverse bias are initiated. Due to the asymmetry in these tunneling barriers, a positive net DC current is delivered to an external circuit.
Inventors: CUTLER; PAUL A.; (State College, PA)
Serial No.: 973262
Series Code: 12
Filed: December 20, 2010
BACKGROUND OF THE INVENTION
[0004] 1. Field of the Invention
[0005] The present invention relates to a technique for performing optical frequency rectification so as to more efficiently harvest radiation from the infrared to the visible. More specifically, the present invention relates to a technique of optical frequency rectification based on a geometric asymmetry of the antenna tip, or other shaped edges such as on patch antennas.
USPTO
http://is.gd/OuErlt
Metal nanoparticle generates new hope for hydrogen energy
Posted: Jun 29th, 2011
(Nanowerk News) Tiny metallic particles produced by University of Adelaide chemistry researchers are bringing new hope for the production of cheap, efficient and clean hydrogen energy. Led by Associate Professor Greg Metha, Head of Chemistry, the researchers are exploring how the metal nanoparticles act as highly efficient catalysts in using solar radiation to split water into hydrogen and oxygen.
"Efficient and direct production of hydrogen from solar radiation provides a renewable energy source that is the pinnacle of clean energy," said Associate Professor Greg Metha. "We believe this work will contribute significantly to the global effort to convert solar energy into portable chemical energy."
The latest research is the outcome of 14 years of fundamental research by Associate Professor Metha's research group investigating the synthesis and properties of metal nanoparticles and how they work as catalysts at the molecular level.
The group works with metal "clusters" of about one-quarter of a nanometre in size – less than 10 atoms. Associate Professor Metha said these tiny "magic clusters" act as super-efficient catalysts. Catalysts drive chemical reactions, reducing the amount of energy required.
"We've discovered ways of producing these tiny metallic clusters, we've explored their fundamental chemical activity, and now we are applying their catalytic properties to reactions which have great potential benefit for industrial use and the environment," said Associate Professor Metha.
PhD student Jason Alvino is exploring splitting water to make hydrogen (and oxygen) using solar energy – a process that is not viable for industry development at the moment.
"We know this catalysis works very efficiently at the molecular level and now need to demonstrate it works on the macroscopic scale," said Associate Professor Metha.
"Splitting water to make hydrogen and oxygen requires a lot of energy and is an expensive process. We will be using solar radiation as the energy source, so there will be no carbon emissions and because the clusters work so efficiently as a catalyst, it will be a much better process.
"The ultimate aim is to produce hydrogen from water as a cheap portable energy source."
Associate Professor Metha said there were also other industrial chemical reactions that could be made feasible by these catalysts, using solar radiation as the energy source - with potentially significant environmental benefits. One example was converting carbon dioxide into methane or methanol with water.
Source: University of Adelaide
http://www.nanowerk.com/news/newsid=21908.php
Scientists Generates Hydrogen as an Energy Source from Ethanol and Sunlight
ScienceDaily (May 26, 2011) — A team of researchers from the Universitat Politècnica de Catalunya, the University of Aberdeen (Scotland) and the University of Auckland (New Zealand) uses ethanol and sunlight to generate hydrogen as an energy source.
The results of the study have been published in Nature Chemistry.
Jordi Llorca, director of the Institute of Energy Technology and researcher at the Universitat Politècnica de Catalunya's Nanoengineering Research Centre, is one of the authors of the study, which represents a major step towards using hydrogen as an alternative to fossil fuels. In the framework of the research, a fully scalable powder photocatalyst was created that makes the hydrogen production process simpler and cheaper as it takes place at ambient temperature and pressure.
A solid photocatalyst is placed in a container with ethanol and exposed to ultraviolet light by agitation, simulating the most energetic part of the solar spectrum. The device contains a titanium dioxide semiconductor that in contact with sunlight generates electrons captured by metallic gold nanoparticles, which react with the alcohol molecules to produce hydrogen.
According to Llorca, the semiconductor's structure and the contact with the nanoparticles are crucial features in the design of the photocatalyst.
The amount of hydrogen and energy generated depends on the amount of catalyst used and the area exposed to solar radiation. Researchers have generated up to 5 litres of hydrogen per kilogram of catalyst in one minute. If 9 kg of catalyst were put in an ethanol tank and exposed to sunlight and the hydrogen generated were used to power a fuel cell, 3 kW of electricity would be obtained, an amount similar to that which is used in a home.
Llorca plans to design reactors with real-life applications such as providing electricity to the home, which he sees as an important step towards introducing hydrogen as an energy vector and gradually gaining independence from fossil fuels. One of the advantages of hydrogen compared with electricity is that it can be stored.
An economical process based on renewable resources Until now, solar-generated hydrogen techniques have largely relied on water. However, despite water being cheap and abundant, these techniques have garnered poor results and the materials they require are expensive. As an alternative, the researchers suggest using ethanol, a renewable and economical resource that is easily obtained from agricultural and forest waste (100 grams of glucose generate approximately 50 grams of ethanol).
The photocatalyst is also much cheaper and simpler to use than the materials employed in techniques with water as it uses very small gold particles, ranging in size from 2 to 12 nanometres (1 metre = 1 million nanometres). These nanoparticles capture the free electrons generated when titanium oxide -- used as a support base -- comes into contact with sunlight.
During the process, which is based on solar energy, the team also discovered that the size of the gold nanoparticles has no influence on the production of hydrogen, unlike what occurs during the more widespread processes in which the catalyst powder must be heated to reaction temperature (usually over 500ºC) and therefore incurs an energy cost. In addition, the catalyst is more durable because it works at ambient temperature and pressure.
http://www.sciencedaily.com/releases/2011/05/110526102350.htm

At the heart of the Cougar™ Platform is the Innovalight™ Silicon Ink. The proprietary material is comprised of silicon nano-particles dispersed in an environmentally friendly blend of chemicals. The result of over 100 engineering-years of ink development, Innovalight has optimized silicon particle size and dopant concentration to maximize the conversion efficiency of the Cougar Platform. Additionally, the material was formulated to be compatible with low-cost industry standard screen printers – the same tool cell manufacturers use for printing metal contacts.
To meet the demand of the Cougar Platform, Innovalight is ramping Silicon Ink production to support over 2,000 MW of Cougar Cells by June 2011.
http://innovalight.com/technology_solarink.htm
This is very interesting:
http://www.japanfs.org/en/pages/030912.html
Japan Advanced Institute of Science and Technology (JAIST) announced on February 7, 2011, that the Institute successfully developed thin film amorphous silicon with good semiconducting properties using liquid silicon for the first time in the world. JAIST also noted that it made a prototype of an amorphous silicon solar cell and confirmed its high performance.
As part of its problem-solving type basic studies of the Japan Science and Technology Agency (JST), JAIST studied basic properties and the control technology of liquid silicon materials and successfully developed a liquid ink that has a stable coating ability. Then it developed a high efficiency thin film solar cell by establishing coating and annealing technologies to uniformly fabricate thin film amorphous silicon on a substrate.
Manufacturing amorphous solar cells using liquid silicon was considered a revolutionary low-cost manufacturing method, but it had been impossible due to substantial technical difficulties. JAIST thoroughly studied the properties of liquid silicon and exhibited this new possibility.
In the future, JAIST will organize collaborative research on higher quality silicon thin film and lower cost liquid silicon with companies, aiming for the commercialization of silicon solar cells with an equivalent cost to current commercial electricity.
Ultra-Thin Solar Panel Attachable to Wavy Slate Roofing Launched (Related JFS article)
http://www.japanfs.org/en/pages/030104.html
Mitsubishi Chemical Testing Solar-Powered Air-Conditioning System for Truck Cabs (Related JFS article)
http://www.japanfs.org/en/pages/029627.html
Sanyo to Increase HIT Solar Cell Production(Related JFS article)
http://www.japanfs.org/en/pages/029190.html
Teaching algae to make fuel
May 24, 2011 by David L. Chandler
Postdoctoral researcher Iftach Yacoby holds vials containing two of the materials used in the research: On the right, green photosynthetic membranes derived from plants, and on the left, brown ferredoxin protein, one of two enzymes the team combined to increase hydrogen production. Photo: Patrick Gillooly
Many kinds of algae and cyanobacteria, common water-dwelling microorganisms, are capable of using energy from sunlight to split water molecules and release hydrogen, which holds promise as a clean and carbon-free fuel for the future. One reason this approach hasn’t yet been harnessed for fuel production is that under ordinary circumstances, hydrogen production takes a back seat to the production of compounds that the organisms use to support their own growth.
But Shuguang Zhang, associate director of MIT’s Center for Biomedical Engineering, and postdocs Iftach Yacoby and Sergii Pochekailov, together with colleagues at Tel Aviv University in Israel and the National Renewable Energy Laboratory in Colorado, have found a way to use bioengineered proteins to flip this preference, allowing more hydrogen to be produced.
“The algae are really not interested in producing hydrogen, they want to produce sugar,” Yacoby says — the sugar is what they need for their own survival, and the hydrogen is just a byproduct. But a multitasking enzyme, introduced into the liquid where the algae are at work, both suppresses the sugar production and redirects the organisms’ energies into hydrogen production. The work is described in a paper being published online this week in the Proceedings of the National Academy of Sciences, and was supported in part by a European Molecular Biology Organization postdoctoral fellowship, the Yang Trust Fund and the U.S. Department of Energy’s National Renewable Energy Laboratory.
Adding the bioengineered enzyme increases the rate of algal hydrogen production by about 400 percent, Yacoby says. The sugar production is suppressed but not eliminated, he explains, because “if it went to zero, it would kill the organism.”
The research demonstrates for the first time how the two processes carried out by algae compete with each other; it also shows how that competition could be modified to favor hydrogen production in a laboratory environment. Zhang and Yacoby plan to continue developing the system to increase its efficiency of hydrogen production.
“It’s one step closer to an industrial process,” Zhang says. “First, you have to understand the science” — which has been achieved through this experimental work. Now, developing it further — through refinements to produce a viable commercial system for hydrogen-fuel manufacturing — is “a matter of time and money,” Zhang says.
Ultimately, such a system could be used to produce hydrogen on a large scale using water and sunlight. The hydrogen could be used directly to generate electricity in a fuel cell or to power a vehicle, or could be combined with carbon dioxide to make methane or other fuels in a renewable, carbon-neutral way, the researchers say.
Bengt Nordén, chair of the physical chemistry department at the Chalmers University of Technology in Gothenburg, Sweden, says, “The results are convincing, although it is difficult to judge, at this stage, how useful this system could be for practical energy purposes.”
Nordén adds, “Hydrogen will no doubt be the future’s fuel,” either in pure form or combined to make methane. This particular method of making hydrogen, he says, “is a both simple and environmentally friendly bio-inspired system.”
In the long run, “the only viable way to produce renewable energy is to use the sun, [either] to make electricity or in a biochemical reaction to produce hydrogen,” Yacoby says. “I believe there is no one solution,” he adds, but rather many different approaches depending on the location and the end uses.
This particular approach, he says, is simple enough that it has promise “not just in industrialized countries, but in developing countries as well” as a source of inexpensive fuel. The algae needed for the process exist everywhere on Earth, and there are no toxic materials involved in any part of the process, he says.
“The beauty is in its simplicity,” he says.
More information: Photosynthetic electron partitioning between [FeFe]-hydrogenase and ferredoxin:NADP+-oxidoreductase (FNR) enzymes in vitro, PNAS, Published online before print May 23, 2011, doi: 10.1073/pnas.1103659108
http://www.pnas.org/content/early/2011/05/19/1103659108.full.pdf
Abstract
Photosynthetic water splitting, coupled to hydrogenase-catalyzed hydrogen production, is considered a promising clean, renewable source of energy. It is widely accepted that the oxygen sensitivity of hydrogen production, combined with competition between hydrogenases and NADPH-dependent carbon dioxide fixation are the main limitations for its commercialization. Here we provide evidence that, under the anaerobic conditions that support hydrogen production, there is a significant loss of photosynthetic electrons toward NADPH production in vitro. To elucidate the basis for competition, we bioengineered a ferredoxin-hydrogenase fusion and characterized hydrogen production kinetics in the presence of Fd, ferredoxin:NADP+-oxidoreductase (FNR), and NADP+. Replacing the hydrogenase with a ferredoxin-hydrogenase fusion switched the bias of electron transfer from FNR to hydrogenase and resulted in an increased rate of hydrogen photoproduction. These results suggest a new direction for improvement of biohydrogen production and a means to further resolve the mechanisms that control partitioning of photosynthetic electron transport.
Provided by Massachusetts Institute of Technology
http://www.physorg.com/news/2011-05-algae-fuel.html
Stevens Students Develop Cheaper, Greener, Alternative Energy Storage
Every year, the world consumes 15 Terrawatts of power. Since the amount of annual harvestable solar energy has been estimated at 50 Terrawatts, students at Stevens Institute of Technology are working on a supercapacitor that will allow us to harness more of this renewable energy through biochar electrodes for supercapacitors, resulting in a cleaner, greener planet.
Supercapacitors are common today in solar panels and hydrogen fuel cell car batteries, but the material they use to store energy, activated carbon, is unsustainable and expensive. Biochar, on the other hand, represents a cheap, green alternative. The Chemical Engineering Senior Design team of Rachel Kenion, Liana Vaccari, and Katie Van Strander has designed biochar electrodes for supercapacitors, and is looking to eventually bring their solution to market. The group is advised by Dr. Woo Lee, the George Meade Bond Professor of Chemical Engineering and Materials Science.
For their project, the team designed, fabricated, and tested a prototype supercapacitor electrode. The group demonstrated biochar's feasibility as an alternative to activated carbon for electrodes, which can be used in hybrid electric automobile batteries or home energy storage in solar panels.
Follow up:
"While the team's findings are preliminary, the approach taken by us represents a small, but potentially very important step in realizing sustainable energy future over the next few decades," says Dr. Lee.
Biochar is viewed as a green solution to the activated carbon currently used in supercapacitor electrodes. Unlike activated carbon, biochar is the byproduct of the pyrolysis process used to produce biofuels. That is, biochar comes from the burning of organic matter. As the use of biofuels increases, biochar production increases as well. "With our process, we are able to take that biochar and put it to good use in supercapacitors. Our supply comes from goldenrod crop, and through an IP-protected process, most organics, metals, and other impurities are removed. It is a more sustainable method of production than activated carbon," Liana says. Another significant advantage: biochar is nontoxic and will not pollute the soil when it is tossed out. The team estimates that biochar costs almost half as much as activated carbon, and is more sustainable because it reuses the waste from biofuel production, a process with sustainable intentions to begin with.
One of the largest concerns for solar panel production today is the sheer cost of manufacturing supercapacitors. Current photovoltaic arrays rely on supercapacitors to store the energy that is harnessed from the sun. And while the growth rate of supercapacitors is advancing at 20 percent a year, their cost is still very high, in part because they require activated carbon. Biochar, on the other hand, is cheaper and readily available as a byproduct of a process already used in energy production.
"My favorite part of this project was seeing the creation of the prototype," Katie says. "It was cool to be able to hold it in my hand and test it and say that I made this."
"Using this technology, we can reduce the cost of manufacturing supercapacitors by lowering the cost of the electrodes," Katie says. "Our goal is eventually to manufacture these electrodes and sell them to a company that already makes supercapacitors. Once supercapacitors become cheaper, they will become more common and be integrated into more and more devices."
http://www.stevens.edu/news/content/stevens-students-develop-cheaper-greener-alternative-energy-storage
http://www.sciencedaily.com/releases/2011/05/110523101909.htm
BTW, Axion uses activated carbon:
Conventional lead-acid batteries use negative electrodes made of sponge lead pasted onto a lead grid current collector. In comparison, our technology uses negative electrodes made of microporous activated carbon with very high surface area. The result is a battery-supercapacitor hybrid that uses less lead.
After 34 years Voyager 1 and 2 are still working!!! Now leaving the solar system!
The spacecraft are powered by a radioisotope thermoelectric generator, in essence a nuclear battery. Pellets of plutonium release heat through natural decay and are converted into electricity using a series of thermocouples. It is a safe, reliable and long lasting power source expected to be finally depleted somewhere around 2025. Voyager's thrusters are fueled by hydrazine and there is enough on board to last another 60 years as fuel is only used to reorient the spacecraft. It uses about 1.6 grams per day.
Voyager carries various scientific instruments along with three computers that share just 68 KB of memory. Engineers developed a self repairing, programmable command processor with multiple modules that are able to determine errors by comparing past data.
The last time we checked on the Voyager 1 & 2 they were hurtling towards the edge of the solar system at over 37,000 mph (60,000 km/h). The car-sized spacecraft are now and incredible 11 billion miles (17 billion km) and 8 billion miles (14 billion km) from Earth respectively - they are the longest continuously operated spacecraft in deep space and, having traveled further than any man-made object, they will soon become the first to enter the realm of interstellar space.
Built by NASA's Jet Propulsion Laboratory in Pasadena, California, Voyager 2 was launched on Aug 20, 1977, closely followed by Voyager 1 on Sept 5. It was a special year because there was a rare planetary alignment of the outer planets, which meant the spacecraft would be able to visit all four giant gas planets; Jupiter, Saturn, Uranus and Neptune.
The Voyager twins' primary five year mission was to explore Jupiter (which was reached in 1979) and Saturn (1980). The spacecraft captured images of the planets, their larger moons and Saturn's rings, making some amazing discoveries. Jupiter's Red Spot was found to be a massive hurricane three times the diameter of Earth and just one of many huge storms on the planet. Saturn's rings were revealed to be made of a mish mash of icy particles - some as large as a house. Io, one of Jupiter's moons, was observed to have eight active volcanoes, where previously volcanic eruptions were only known on Earth.
Voyager 2, propelled further by Jupiter and Saturn's gravity, continued to Uranus in 1986 and Neptune in 1989. Breathtakingly close up pictures were taken of the planets, their moons and the system of rings and magnetic fields surrounding each. Notable discoveries included the fact that Uranus spins on a horizontal axis. Neptune the most remote giant planet has the fastest winds in the solar system that travel at over 1,200 mph (2,000 kph). Like Jupiter, it has a Great Dark Spot of its own, a giant storm about the size of Earth. Triton, a moon of Neptune is the coldest measured object in the Solar System. It has a surface temperature of -390°F (-235°C) with geysers spewing nitrogen gas into the atmosphere.
The Voyager spacecraft rewrote the textbooks on the four gas giants revealing the complexity and diversity of our solar system. On its way out of our solar system, Voyager 1 was asked to look back and take more photograph before the cameras were turned off. The "Pale Blue Dot" photograph was taken from a record distance of more than four billion miles from Earth and is part of the resulting is 60 frame mosaic of the solar system. In the photo, the Earth appears as a tiny dot 0.12 pixels in size, giving us a mind-boggling reminder of just how small we are in the vastness of space.
More on the history and story!!!!
http://www.gizmag.com/voyager-spacecraft-interstellar-space/18557/?utm_source=Gizmag+Subscribers&utm_campaign=7dae3cd16b-UA-2235360-4&utm_medium=email
The spacecraft are still functioning and thriving after more than 30 years. Robust, durable and built from 1970s components, the transistors are still functioning today. They have exceeded their design specifications and the loftiest dreams of their makers ... and despite having already made an amazing contribution to our civilization, many great discoveries are still to come.
Voyager 1 is on a trajectory to reach star AC+79 in about 40000 years. Voyager 2 is on its way to the vicinity of star Sirius, a mere 296,000 years away.
FYI - United States Patent Application 20110110019 Varade; Ashish; et al. May 12, 2011
Composition Of Electrode Material In The Form Of A Coating And A Process Theeof
Abstract
The present invention is in relation to a composition of electrode material in the form of a coating, said composition represented by formula Mn.sub.1-xO/C, wherein Mn.sub.1-xO is the monoxide of manganese with x is .gtoreq.0 and .ltoreq.0.1 and C is carbon. In addition, the invention also provides a process for deposition of aforementioned composition in the form of a nanocomposite coat on the electrode of an electrochemical capacitor in the fields of automobile, aerospace engineering and applications, very large scale integrated circuits (VLSI) technology, micro-electro-mechanical systems (MEMS) and combinations thereof.
Inventors:
Varade; Ashish; (Karnataka, IN) ; Shivashankar; Ajjampur Srinivasarao; (Karnataka, IN) ; Dhar; Sukanya; (Karnataka, IN) ; Sampath; Srinivasan; (Karnataka, IN)
Serial No.:
673726
Filed:
August 29, 2007
PCT Filed:
August 29, 2007
PCT NO:
PCT/IN07/00372
371 Date:
February 16, 2010
http://is.gd/R1cGMO
I can remember reading a good while back about a home unit that took in natural gas and converted it into a car fuel or electricity - I can't remember which now - damned memory - needs an overhaul! But this device clearly makes H2 from methane (aka natural gas) and does it at 99.5% efficiency.
I've always wanted to use MORE natural gas and figure THIS development could well power my home and car and everything else with H2 generating electricity in my home and car in fuel cells! And what with cell phones - no wires - pull down those ugly telephone/power poles everywhere. Underground will be reserved for GAS! And no more power outages from wonky electric company feed problems!! No more nuclear, no more coal, oil and OPEC, no more cancer from those and Bob's your uncle.
Where and when do I get to buy one?
And invest in the company making and selling them?
Re --Mohamed Halabi--
http://www.sciencedaily.com/releases/2011/05/110512103946.htm
Extract from patent:
The presently disclosed embodiments generally relate to the use of nanoscale coaxial transmission lines (NCTL) to fabricate a nanoscale optics apparatus. The nanoscale optics apparatus is a multifunctional nano-composite material made of a metallic film having a top surface and a bottom surface and a plurality of cylindrical channels filled with a photovoltaic material. An array of nanorods penetrate the metallic film through the plurality of cylindrical channels. The array of nanorods has a protruding portion that extends beyond a surface of the metallic film and an embedded portion that is within the metallic film. The protruding portion acts as a [b]nano-antenna and is capable of receiving and transmitting an electromagnetic radiation at a visible frequency. The embedded portion acts as a nanoscale coaxial transmission line (CTL) and allows for propagation of external radiation with a wavelength exceeding the perpendicular dimensions of the nanorod.
The nanoscale optics apparatus can concentrate light, and therefore enhance a field up to about 10.sup.3 times. The array of optical nano-antennas, with nano-CTL embedded in a metallic film, effectively compresses light into nanoscopic dimensions. The nano-antennas are capable of receiving and transmitting electromagnetic radiation at the visible frequencies. The extreme compression of light in the nano-CTL leads to an asymmetric tunneling of electrons between the electrodes of the nano-CTL, and thus provides a rectifying action at the light frequencies, and thus conversion of the light into a direct current (DC) voltage. This property leads to a new class of efficient, and low cost rectenna solar cells. The extreme compression of light in the nano-CTL is quick, and is not limited by the usual parasitic capacitances that make the conventional diode rectification inefficient, if not impossible, at the light frequencies.
http://is.gd/SBD8EZ
United States Patent 7,943,847 Kempa, et al., May 17, 2011
Apparatus and methods for solar energy conversion using nanoscale cometal structures
Abstract
An apparatus and methods for solar conversion using nanoscale cometal structures are disclosed herein. The cometal structures may be coaxial and coplanar. A nanoscale optics apparatus for use as a solar cell comprises a plurality of nanoscale cometal structures each including a photovoltaic material located between a first electrical conductor and a second electrical conductor. A method of fabricating solar cells comprises preparing a plurality of nanoscale planar structures; coating a plurality of planar surfaces of the plurality of planar structures with a photovoltaic semiconductor while leaving space between the plurality of planar surfaces; and coating the photovoltaic semiconductor with an outer electrical conductor layer, wherein a portion of the outer electrical conductor layer is located between the planar structures to form coplanar structures.
Inventors:
Kempa; Krzysztof J. (Billerica, MA), Naughton; Michael J. (Norwood, MA), Ren; Zhifeng (Newton, MA), Rybczynski; Jakub A. (Cambridge, MA)
Assignee:
The Trustees of Boston College (Chestnut Hill, MA)
Appl. No.:
11/509,269
Filed:
August 24, 2006
http://is.gd/SBD8EZ
Will this be in man's future? Will it be in ANI's future?? Will it power our cars??? Provide our power needs for all things?
Perhaps.
Will China be involved?
New Solar Product Captures Up to 95 Percent of Light Energy
5/17/2011 9:34:08 AM
Efficiency is a problem with today’s solar panels; they only collect about 20 percent of available light. Now, a University of Missouri engineer has developed a flexible solar sheet that captures more than 90 percent of available light, and he plans to make prototypes available to consumers within the next five years.
Patrick Pinhero, an associate professor in the MU Chemical Engineering Department, says energy generated using traditional photovoltaic (PV) methods of solar collection is inefficient and neglects much of the available solar electromagnetic (sunlight) spectrum. The device his team has developed – essentially a thin, moldable sheet of small antennas called nantenna – can harvest the heat from industrial processes and convert it into usable electricity. Their ambition is to extend this concept to a direct solar facing nantenna device capable of collecting solar irradiation in the near infrared and optical regions of the solar spectrum.
Working with his former team at the Idaho National Laboratory and Garrett Moddel, an electrical engineering professor at the University of Colorado, Pinhero and his team have now developed a way to extract electricity from the collected heat and sunlight using special high-speed electrical circuitry. This team also partners with Dennis Slafer of MicroContinuum, Inc., of Cambridge, Mass., to immediately port laboratory bench-scale technologies into manufacturable devices that can be inexpensively mass-produced.
“Our overall goal is to collect and utilize as much solar energy as is theoretically possible and bring it to the commercial market in an inexpensive package that is accessible to everyone,” Pinhero said. “If successful, this product will put us orders of magnitudes ahead of the current solar energy technologies we have available to us today.”
As part of a rollout plan, the team is securing funding from the U.S. Department of Energy and private investors. The second phase features an energy-harvesting device for existing industrial infrastructure, including heat-process factories and solar farms.
Within five years, the research team believes they will have a product that complements conventional PV solar panels. Because it’s a flexible film, Pinhero believes it could be incorporated into roof shingle products, or be custom-made to power vehicles.
Once the funding is secure, Pinhero envisions several commercial product spin-offs, including infrared (IR) detection. These include improved contraband-identifying products for airports and the military, optical computing, and infrared line-of-sight telecommunications.
A study on the design and manufacturing process was published in the Journal of Solar Energy Engineering.
http://nanotechwire.com/news.asp?nid=12063
Carbon-Based Supercapacitors Produced by Activation of Graphene
Published Online 12 May 2011
Science DOI: 10.1126/science.1200770
REPORT
Carbon-Based Supercapacitors Produced by Activation of Graphene
Yanwu Zhu1, Shanthi Murali1, Meryl D. Stoller1, K. J. Ganesh1, Weiwei Cai1 , Paulo J. Ferreira1, Adam Pirkle2, Robert M. Wallace2 , Katie A. Cychosz3, Matthias Thommes3, Dong Su4, Eric A. Stach4, and Rodney S. Ruoff1,*
- Author Affiliations
1Department of Mechanical Engineering and Materials Science and Engineering Program, University of Texas at Austin, One University Station C2200, Austin, TX 78712, USA.
2Department of Materials Science and Engineering, University of Texas at Dallas, 800 W. Campbell Road, Richardson, TX 75080, USA.
3Quantachrome Instruments, 1900 Corporate Drive, Boynton Beach, FL 33426, USA.
4Center for Functional Nanomaterials, Brookhaven National Laboratory, Upton, NY 11973, USA.
*?To whom correspondence should be addressed. E-mail: r.ruoff@mail.utexas.edu
ABSTRACT
Supercapacitors, also called ultracapacitors or electrochemical capacitors, store electrical charge on high surface area conducting materials. Their widespread use is limited by their low energy storage density and relatively high effective series resistance. Using chemical activation of exfoliated graphite oxide, we synthesized a porous carbon with a BET surface area of up to 3100 square meters per gram, a high electrical conductivity, and a low O and H content. This sp2-bonded carbon has a continuous three-dimensional network of highly curved, atom thick walls that form primarily 0.6- to 5-nm-width pores. Two-electrode supercapacitor cells constructed with this carbon yielded high values of gravimetric capacitance and energy density with organic and ionic liquid electrolytes. The processes used to make this carbon are readily scalable to industrial levels.
Received for publication 22 November 2010.
Accepted for publication 2 May 2011.
http://www.sciencemag.org/content/early/2011/05/11/science.1200770.abstract
Activated Graphene Makes Superior Supercapacitors for Energy Storage
Abstract:
Scientists at the U.S. Department of Energy's Brookhaven National Laboratory have helped to uncover the nanoscale structure of a novel form of carbon, contributing to an explanation of why this new material acts like a super-absorbent sponge when it comes to soaking up electric charge. The material, which was recently created at The University of Texas - Austin, can be incorporated into "supercapacitor" energy-storage devices with remarkably high storage capacity while retaining other attractive attributes such as superfast energy release, quick recharge time, and a lifetime of at least 10,000 charge/discharge cycles.
Activated Graphene Makes Superior Supercapacitors for Energy Storage: New material combines high storage capacity with quick energy release and unlimited recharge
Upton, NY | Posted on May 13th, 2011
"Those properties make this new form of carbon particularly attractive for meeting electrical energy storage needs that also require a quick release of energy — for instance, in electric vehicles or to smooth out power availability from intermittent energy sources, such as wind and solar power," said Brookhaven materials scientist Eric Stach, a co-author on a paper describing the material published in Science on May 12, 2011.
Supercapacitors are similar to batteries in that both store electric charge. Batteries do so through chemical reactions between metallic electrodes and a liquid electrolyte. Because these chemicals take time to react, energy is stored and released relatively slowly. But batteries can store a lot of energy and release it over a fairly long time.
Supercapacitors, on the other hand, store charge in the form of ions on the surface of the electrodes, similar to static electricity, rather than relying on chemical reactions. Charging the electrodes causes ions in the electrolyte to separate, or polarize, as well — so charge gets stored at the interface between the electrodes and the electrolyte. Pores in the electrode increase the surface area over which the electrolyte can flow and interact — increasing the amount of energy that can be stored.
But because most supercapacitors can't hold nearly as much charge as batteries, their use has been limited to applications where smaller amounts of energy are needed quickly, or where long life cycle is essential, such as in mobile electronic devices.
The new material developed by the UT-Austin researchers may change that. Supercapacitors made from it have an energy-storage capacity, or energy density, that is approaching the energy density of lead-acid batteries, while retaining the high power density — that is, rapid energy release — that is characteristic of supercapacitors.
"This new material combines the attributes of both electrical storage systems," said University of Texas team leader Rodney Ruoff. "We were rather stunned by its exceptional performance."
The UT-Austin team had set out to create a more porous form of carbon by using potassium hydroxide to restructure chemically modified graphene platelets — a form of carbon where the atoms are arrayed in tile-like rings laying flat to form single-atom-thick sheets. Such "chemical activation" has been previously used to create various forms of "activated carbon," which have pores that increase surface area and are used in filters and other applications, including supercapacitors.
But because this new form of carbon was so superior to others used in supercapacitors, the UT-Austin researchers knew they'd need to characterize its structure at the nanoscale.
Ruoff had formed a hypothesis that the material consisted of a continuous three-dimensional porous network with single-atom-thick walls, with a significant fraction being "negative curvature carbon," similar to inside-out buckyballs. He turned to Stach at Brookhaven for help with further structural characterization to verify or refute this hypothesis.
Stach and Brookhaven colleague Dong Su conducted a wide range of studies at the Lab's Center for Functional Nanomaterials (CFN), the National Synchrotron Light Source (NSLS), and at the National Center for Electron Microscopy at Lawrence Berkeley National Laboratory, all three facilities supported by the DOE Office of Science. "At the DOE laboratories, we have the highest resolution microscopes in the world, so we really went full bore into characterizing the atomic structure," Stach said.
"Our studies revealed that Ruoff's hypothesis was in fact correct, and that the material's three-dimensional nanoscale structure consists of a network of highly curved, single-atom-thick walls forming tiny pores with widths ranging from 1 to 5 nanometers, or billionths of a meter."
The study includes detailed images of the fine pore structure and the carbon walls themselves, as well as images that show how these details fit into the big picture. "The data from NSLS were crucial to showing that our highly local characterization was representative of the overall material," Stach said.
"We're still working with Ruoff and his team to pull together a complete description of the material structure. We're also adding computational studies to help us understand how this three-dimensional network forms, so that we can potentially tailor the pore sizes to be optimal for specific applications, including capacitive storage, catalysis, and fuel cells," Stach said.
Meanwhile, the scientists say the processing techniques used to create the new form of carbon are readily scalable to industrial production. "This material — being so easily manufactured from one of the most abundant elements in the universe — will have a broad range impacts on research and technology in both energy storage and energy conversion," Ruoff said.
The work at Brookhaven was supported by DOE's Office of Science; the UT - Austin team's research was supported by the Office of Science, the National Science Foundation, and the Advanced Technology Institute.
####
About Brookhaven National Laboratory
The Center for Functional Nanomaterials at Brookhaven National Laboratory is one of the five DOE Nanoscale Science Research Centers (NSRCs), premier national user facilities for interdisciplinary research at the nanoscale. Together the NSRCs comprise a suite of complementary facilities that provide researchers with state-of-the-art capabilities to fabricate, process, characterize and model nanoscale materials, and constitute the largest infrastructure investment of the National Nanotechnology Initiative. The NSRCs are located at DOE’s Argonne, Brookhaven, Lawrence Berkeley, Oak Ridge and Sandia and Los Alamos national laboratories. For more information about the DOE NSRCs, please visit nano.energy.gov.
One of ten national laboratories overseen and primarily funded by the Office of Science of the U.S. Department of Energy (DOE), Brookhaven National Laboratory conducts research in the physical, biomedical, and environmental sciences, as well as in energy technologies and national security. Brookhaven Lab also builds and operates major scientific facilities available to university, industry and government researchers. Brookhaven is operated and managed for DOE's Office of Science by Brookhaven Science Associates, a limited-liability company founded by the Research Foundation of State University of New York on behalf of Stony Brook University, the largest academic user of Laboratory facilities, and Battelle, a nonprofit, applied science and technology organization.
For more information, please click here
Contacts:
Rodney Ruoff:
The University of Texas
Austin Cockrell School of Engineering
Communications Office
Maria Arrrellaga
512-232-8060
maria.arrellaga@austin.utexas.edu
Karen McNulty Walsh
(631) 344-8350
kmcnulty@bnl.gov
or
Peter Genzer
(631) 344-3174
genzer@bnl.gov
Copyright © Brookhaven National Laboratory
http://www.nanotech-now.com/news.cgi?story_id=42451
It all depends who has the dominating patent.
In this situation there likely is NO dominating patent - they are exclusive to each other - peculiar, if you like - no common ground. It then remains to be seen which will dominate the market on their business abilities and technical prowess.
Energy production and storage developments using nanotechnology.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
