Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Most patents violate multiple other patents, and so must license the other patents or risk getting sued. For example say I patent a gadget for cars, and so I decide to start making cars with my gadget on them. I still need licenses, or implied licenses that come with other car parts for all the other patented goodies in the car.
Whose patents are good and/or violated is one thing. Another is which version is better and will dominate the market. I have no idea, myself.
There is no way that patent will ever be issued as written (based on the claims I see there). Sounds like they threw everything but the kitchen sink in there hoping someday, some piece of it might survive as written. I suspect it would violate AXPW's patents(s) before it ever reached the point of having something novel. But I need to read the entire mess before I can be sure.
The story behind them, their BK, and recent resurrection and new source of funding was very interesting to read.
WO 2010008966
ORGANIC ADDITIVES FOR IMPROVING PERFORMANCE OF LEAD-ACID BATTERIES
Abstract of WO 2010008966
The present invention includes small molecule organic additives for lead acid batteries, a lead acid battery and components thereof containing the small molecule organic additives of the invention, and methods for the use of such compounds. The batteries of the invention may optionally further contain carbon foam. The presence of carbon in the battery may generate some of the organic agents of the invention.
SUMMARY OF THE INVENTION
[0007] The present invention includes small molecule organic additives for lead acid batteries, a lead acid battery and components thereof containing the small molecule organic additives of the invention, and methods for the use of such compounds. The batteries of the invention may optionally further contain carbon in the form of foam.
[0008] The additives of the invention reduce some inefficiencies and effects noted above, and thereby improve battery capacity and utilization efficiency.
[0009] The organic additives may be added directly to battery paste or electrolyte as a production step, or may be added to preexisting batteries. Carbon precursors may be constructed and arranged in a battery to advantageously promote generation of organic molecules.
http://worldwide.espacenet.com/publicationDetails/description?CC=WO&NR=2010008966A1&KC=A1&FT=D&date=20100121&DB=EPODOC&locale=en_EP
Firefly’s carbon-graphite foam-based battery technology:
CLAIMS
What is claimed is:
1. A lead acid battery containing organic additives comprising: a lead acid battery said battery containing organic additives having a molecular weight smaller than about 10Ou.
2. The battery of claim 1 wherein said organic additives have a molecular weight smaller than about 500u.
3. A lead acid battery containing organic additives comprising carbonil compounds.
4. The battery of claim 3 wherein said carbonil compounds are selected from the group consisting of: aldehydes, R3C-CHO; ethers, R3C-O-CR3; ketones, R3C-CO-CR3; carboxylic acids, R-CO-OH; esters, R3C-CO-O-CR3; acid anhydride, R-CO-O-CO-Rl; enones, RC(O)C(R1)CR2'R3; hydroperoxides, R3C-O-O-CR3; carbonate esters, R3C-O-CO-O-CR3; metal carbonyls, Me-(CO)n.
5. A lead acid battery containing organic additives comprising liquids and solids including: formic acid, HCO2H, formate ions HCO2- or metal formates M-HCO2; carbonate or bicarbonate ions (CO32- and HCO3-), metal carbonates; formaldehyde; oxalic acid H2C2O4, oxalate and acid oxalate ions (C2O42- and HC2O4-), metal oxalates; acetic acid, CH3COOH; acetates, alcohols, such as R3C-OH; carboxyl compounds such as R3C-CO-OH.
6. The battery of claim 1 wherein said organic additives are selected from the group consisting of solutions of CH4, CO2, HC and CO in water or sulfuric acid.
7. The battery of claim 1 wherein organic additives are further comprised of lead and/or sulfur and/or oxygen and/or hydrogen as well as other organic groups and radicals.
8. The battery of claim 1 wherein said organic additives are selected from the group consisting of carbon containing gases including methane, carbon monoxide and carbon dioxide that can be dissolved into the electrolyte.
9. The battery of claim 1 wherein the battery is a carbon foam battery.
10. The battery of claim 1 wherein said organic additives are added to one or more of the following components: electrode material, lead paste, current collectors, separators, electrolyte, box, carbon foam, positive active material and negative active material.
11. The battery of claim 1 wherein the additives are one or more of the following: formic acid, formaldehyde, oxalic acid, acetic acid, compounds of these organics which may optionally contain lead and/or sulfur and/or oxygen and/or hydrogen, as well as other organic groups and radicals; carbon containing gases including methane, carbon monoxide and carbon dioxide that can be dissolved into the electrolyte.
12. The battery of claim 1 wherein the additives are resins.
13. The battery of claim 1 wherein the additives are Furfuryl.
14. A method of enhancing performance of a lead acid battery by adding to the battery during or after construction by adding organic additives, said addition of organic additives comprising: providing a lead acid battery having an electrode material, a lead paste, a current collector, a separator, an electrolyte and a box, and forming an organic additive comprised of material having a molecular weight of less than about 50Ou.
http://worldwide.espacenet.com/publicationDetails/claims?CC=WO&NR=2010008966A1&KC=A1&FT=D&date=20100121&DB=EPODOC&locale=en_EP
http://worldwide.espacenet.com/publicationDetails/biblio?CC=WO&NR=2010008966A1&KC=A1&FT=D&date=20100121&DB=EPODOC&locale=en_EP
No carbon in the form of foam in Axion's version.
I wonder what real difference their is from this outfits lead carbon electrode design and AXPWs?
http://www.fireflyenergy.com/index.php?option=com_content&task=view&id=205&Itemid=89
http://www.fireflyenergy.com/index.php?option=com_content&task=view&id=201&Itemid=96
http://www.fireflyenergy.com/index.php?option=com_content&task=view&id=204&Itemid=89
http://jcwinnie.biz/wordpress/?p=2347
Interesting info on bicycle regenerative breaking electric motors for hybrid E-bikes
In re FastCAP Systems
FastCAP’s ultracapacitor is built upon patent-pending electrodes made with vertically aligned carbon nanotubes, each one thirty-thousandth the width of a human hair. The regular shape and alignment of the nanotubes makes a very efficient and high surface area electrode. The process for making the nanotube electrodes also yields an increased capability to withstand voltage. FastCAP's ultracapacitors retain and even improve upon the benefits of today's ultracapacitors while adding the high energy density benefit that batteries historically monopolized.
Ultracapacitors to Boost the Range of Electric Cars
New energy-storage technology could surpass today's batteries in capacity and durability.
WEDNESDAY, MAY 4, 2011BY KEVIN BULLIS
A startup called Nanotune says its ultracapacitor technology could make electric cars cheaper and extend their range. The company, based in Mountain View, California, has developed a way to make electrodes that results in ultracapacitors with five to seven times as much storage capacity as conventional ones.
Conventional ultracapacitors, which have the advantage of delivering fast bursts of power and can be recharged hundreds of thousands of times without losing much capacity, are too expensive and store too little energy to replace batteries.
Nanotune, however, which has raised $3 million from the venture capital firm Draper Fisher Jurvetson, says its ultracapacitors are close to competing with batteries in terms of energy storage, and could soon surpass them. Using a conventional electrolyte, the company has demonstrated energy storage of 20 watt-hours per kilogram, as opposed to roughly five watt-hours for a conventional ultracapacitor. Using a more expensive ionic-liquid electrolyte, it has made ultracapacitors that store 35 watt-hours per kilogram. By the end of the year, the company hopes to approximately double this storage capacity, says Nanotune CEO Kuan-Tsae Huang. At 40 watt-hours per kilogram, the ultracapacitors would be an improvement over the batteries used in some hybrid vehicles.
In recent months, several startups have announced that they're using nanotechnology to make better ultracapacitors. Each hopes to help solve one of the biggest problems with electric cars today: their batteries' high cost and limited storage capacity. Nissan, for example, to make its electric Leaf affordable, had to limit the size of the battery pack, resulting in a range of just 73 miles.
Part of the reason battery systems are so expensive and bulky is that the batteries degrade as they're used, especially when exposed to extreme temperatures—so automakers often augment them with cooling and heating systems, and add extra battery cells to offset losses in performance over time. Ultracapacitors could sidestep this problem, because they can be recharged without degrading and can work well in a wide range of temperatures.
Eventually, Huang says, it may be possible to make ultracapacitors that store 500 kilowatt-hours per kilogram—about three to four times more than the lithium-ion batteries used in cars today. The practical benefit could be even greater. Cars are often engineered to use only half the storage capacity of their batteries, to keep them from degrading. But almost all of an ultracapacitor's storage capacity can be used.
Nanotune's technology is very expensive now—between $2,400 and $6,000 per kilowatt-hour. (The Department of Energy has proposed a goal of $250 per kilowatt-hour to make electric vehicles competitive with conventional ones.) Nanotune says, however, that its costs could come down to less than $150 per kilowatt-hour if the prices of some key materials, such as electrolytes, continue to fall, and as manufacturing is scaled up.
The company's energy-storage projections are based on several advances it is working on. Nanotune is currently making electrodes with pores that are about 4 to 5 nanometers across, but it says it can make them smaller (high porosity leads to high surface area, which makes it possible to store a large amount of charge) and tune them to match the needs of different electrolytes—the ion-conducting materials the electrodes are immersed in.
The company is also looking into using ionic liquids rather than conventional organic electrolytes. These increase the voltage of the system, greatly increasing energy storage, but typically they aren't compatible with conventional ultracapacitor electrodes. Finally, the company hopes to make use of recent academic findings that suggests that adding small amounts of ruthenium to the ultracapacitors can increase energy storage.
Nanotune isn't the first company to claim it can make ultracapacitors with very high energy storage. Others have found this promise hard to deliver. Increasing surface area can improve storage capacity only so much, since at some point the storage is limited by the ions in the electrolyte. Ionic liquids help with this, but they have significant shortcomings, says Joel Schindall, a professor of electrical engineering and computer science at MIT. (A company called FastCap Systems, which is developing ultracapacitors using carbon nanotubes, was spun out of his lab.) They're very expensive, for one thing, and some operate well only in a limited temperature range, making them impractical for cars.
Schindall says, however, that Nanotune can fall short of its very high energy goals and still improve the competitiveness of electric vehicles and hybrids. Given the durability of ultracapacitors, even achieving energy storage of 100 watt-hours per kilogram—close to that of lithium-ion batteries—"would be fantastic."
http://www.technologyreview.com/energy/37519/?nlid=4430
http://www.edn.com/article/517959-MIT_uses_virus_to_increase_solar_cell_efficiency.php
Gives a whole new meaning to GOING VIRAL!!!!
"MIT uses virus to increase solar-cell efficiency
The MIT research is based on the finding that carbon nanotubes can enhance the efficiency of electron collection from a solar cell's surface.
By Suzanne Deffree, Managing editor, news -- EDN, April 26, 2011
Researchers at MIT claim to have increased the power-conversion efficiency of solar cells by nearly one-third through the use of tiny viruses to perform detailed assembly work at the microscopic level.
The MIT research is based on the finding that carbon nanotubes can enhance the efficiency of electron collection from a solar cell's surface. The researchers used genetically engineered version of a virus called M13, which normally infects bacteria, to control the arrangement of the nanotubes on a surface. Doing so keeps the tubes separate so they can't short out the circuits and keeps the tubes apart so they don't clump -- two problems that have plagued previous attempts to use carbon nanotubes in solar cells.
The system the researchers tested used a type of solar cell known as dye-sensitized solar cells, a lightweight and inexpensive type where the active layer is composed of titanium dioxide (TiO2), rather than the silicon used in conventional solar cells. MIT pointed out that the same techniques could be applied to other types of solar cells, including quantum-dot and organic solar cells. In tests, adding the virus-built structures enhanced the power-conversion efficiency to 10.6% from 8%, almost a one-third improvement.
This efficiency improvement is achieved even though the viruses and the nanotubes make up only 0.1% by weight of the finished cell. "A little biology goes a long way," Angela Belcher, a WM Keck Professor of Energy who worked on the project, said in a statement. The researchers believe they can ramp up the efficiency even further with additional work."
See the link for images and more details.
Interesting. I looked into geothermal heating last year. It's already far more efficient than anything on the market. A solar-thermal method of collecting even more heat prior to entering the heat pump would make geothermal heating a home run. The efficiency would just be astounding. But what about the cost of the solar-thermal part of the sytem? Geothermal systems are already quite expensive.
First polymer solar-thermal device heats home, saves money
Posted: Apr 22nd, 2011
(Nanowerk News) A new polymer-based solar-thermal device is the first to generate power from both heat and visible sunlight – an advance that could shave the cost of heating a home by as much as 40 percent.
Geothermal add-ons for heat pumps on the market today collect heat from the air or the ground. This new device uses a fluid that flows through a roof-mounted module to collect heat from the sun while an integrated solar cell generates electricity from the sun's visible light.
"It's a systems approach to making your home ultra-efficient because the device collects both solar energy and heat," said David Carroll, Ph.D., director of the Center for Nanotechnology and Molecular Materials at Wake Forest University. "Our solar-thermal device takes better advantage of the broad range of power delivered from the sun each day."
Research showing the effectiveness of the device appears in the March issue of the peer-reviewed journal Solar Energy Materials and Solar Cells.
A standard, rooftop solar cell will miss about 75 percent of the energy provided by the sun at any given time because it can't collect the longest wavelengths of light – infrared heat. Such cells miss an even greater amount of the available daily solar power because they collect sunlight most efficiently between 10 a.m. and 2 p.m.
"On a rooftop, you have a lot of visible sunlight and heat from the infrared radiation," Carroll said. "The solar-cell industry has for the most part ignored the heat."
The design of the new solar-thermal device takes advantage of this heat through an integrated array of clear tubes, five millimeters in diameter. They lie flat, and an oil blended with a proprietary dye flows through them. The visible sunlight shines into the clear tube and the oil inside, and is converted to electricity by a spray-on polymer photovoltaic on the back of the tubes. This process superheats the oil, which would then flow into the heat pump, for example, to transfer the heat inside a home.
Unlike the flat solar cells used today, the curve of the tubes inside the new device allows for the collection of both visible light and infrared heat from nearly sunrise to sunset. This means it provides power for a much greater part of the day than does a normal solar cell.
Because of the general structure and the ability to capture light at oblique angles, this is also the first solar-thermal device that can be truly building-integrated – it can be made to look nearly identical to roofing tiles used today.
Tests of the solar-thermal device have shown 30 percent efficiency in converting solar energy to power. By comparison, a standard solar cell with a polymer absorber has shown no greater than 8 percent conversion efficiency.
The research team will build the first square-meter-size solar-thermal cell this summer, a key step in getting the technology ready for market.
Source: Wake Forest University
http://www.nanowerk.com/news/newsid=21128.php
I'll have to take your word on that!
I am not impressed with this one at all. Nothing really new here except an attempt to deceive with a fancy dance around the wording that was poorly done at best, nothing new except maybe a new separator material.
Scientists demonstrate novel ionic liquid batteries
April 15, 2011
Scientists demonstrate novel ionic liquid batteries
(PhysOrg.com) -- Scientists at the NRL Materials Science and Technology Division are providing solid evidence that there is a new route towards developing novel, lightweight energy storage devices. By moving away from centuries of caustic, hazardous aqueous-based battery cells and instead using non-volatile, thermally-stable ionic liquids, scientists predict multiple new types of batteries. Rather than depend on highly acidic electrolytes, ionic liquids are used to create a solid polymer electrolyte composed of an ionic liquid and polyvinyl alcohol, developing novel types of solid state batteries with discharge voltages ranging up to 1.8 volts.
The unique properties of ionic liquids have fostered this explosive interest in battery applications. Ionic liquids are room temperature molten salts that possess many important characteristics, such as nearly no vapor pressure, non- flammability and lack of reactivity in various electrochemical or industrial applications. "It is the high thermal and electrochemical stability of the ionic liquids which has fostered the growing interest in ionic liquids for use in various electrochemical processes," said Dr. Thomas Sutto. "These new types of solid-state cells mimic standard alkaline cells, but without the need for caustic electrolytes."
Limits imposed by using corrosive electrolytes often result in severe restrictions to standard battery geometry and the need for special corrosive-resistant battery containers. The use of reactive ionic liquids in non-aqueous cells replaces the more hazardous highly alkaline electrolytes such as manganese oxide (MgO) and zinc (Zn) found in traditional batteries.
The root of this work began during standard corrosion studies of different metals in ionic liquids. While working with ionic liquids based on mineral acids, such as hydrogen sulphates, it was observed that Zn metal would react to form zinc sulphate. Since this is similar to that observed for the zinc anode in a standard alkaline cell, a series of experiments were then performed to determine how different metal oxides reacted in these types of ionic liquids.
Electrochemical experiments demonstrate that not only can these reactive ionic liquids act as the electrolyte/separator in both solid state and liquid batteries, but they can also act as a reactive species in the cell's electrochemical makeup. Using a non-aqueous approach to primary and secondary power sources, batteries are designed using standard cathode and anode materials such as magnesium dioxide (MgO2), lead dioxide (PbO2) and silver oxide (AgO). The ionic liquid that is the main focus of this work is 1-ethyl-3-methylimidazolium hydrogen sulphate (EMIHSO4), however, other ionic liquids such as those based on the nitrate and dihydrogen phosphate anions (negatively charged ions) have also been found to work well in this type of a battery design.
The use of these electrolytes suggests the potential for new types of rechargeable systems, such as replacement electrolytes in nickel-metal hydride (NiMH) batteries, or even the standard lead-acid battery. Experimental work is currently underway to develop such a rechargeable ionic liquid power source. The ability to create solid separators also allows for the formation of many new types of batteries via a number of fabrication techniques.
Provided by Naval Research Laboratory
http://www.physorg.com/news/2011-04-scientists-ionic-liquid-batteries.html
Solar power without solar cells: A hidden magnetic effect of light could make it possible
April 14, 2011
(PhysOrg.com) -- A dramatic and surprising magnetic effect of light discovered by University of Michigan researchers could lead to solar power without traditional semiconductor-based solar cells.
The researchers found a way to make an “optical battery,” said Stephen Rand, a professor in the departments of Electrical Engineering and Computer Science, Physics and Applied Physics.
In the process, they overturned a century-old tenet of physics.
“You could stare at the equations of motion all day and you will not see this possibility. We’ve all been taught that this doesn’t happen,” said Rand, an author of a paper on the work published in the Journal of Applied Physics. “It’s a very odd interaction. That’s why it’s been overlooked for more than 100 years.”
Light has electric and magnetic components. Until now, scientists thought the effects of the magnetic field were so weak that they could be ignored. What Rand and his colleagues found is that at the right intensity, when light is traveling through a material that does not conduct electricity, the light field can generate magnetic effects that are 100 million times stronger than previously expected. Under these circumstances, the magnetic effects develop strength equivalent to a strong electric effect.
“This could lead to a new kind of solar cell without semiconductors and without absorption to produce charge separation,” Rand said. “In solar cells, the light goes into a material, gets absorbed and creates heat. Here, we expect to have a very low heat load. Instead of the light being absorbed, energy is stored in the magnetic moment. Intense magnetization can be induced by intense light and then it is ultimately capable of providing a capacitive power source.”
What makes this possible is a previously undetected brand of “optical rectification,” says William Fisher, a doctoral student in applied physics. In traditional optical rectification, light’s electric field causes a charge separation, or a pulling apart of the positive and negative charges in a material. This sets up a voltage, similar to that in a battery. This electric effect had previously been detected only in crystalline materials that possessed a certain symmetry.
Rand and Fisher found that under the right circumstances and in other types of materials, the light’s magnetic field can also create optical rectification.
“It turns out that the magnetic field starts curving the electrons into a C-shape and they move forward a little each time,” Fisher said. “That C-shape of charge motion generates both an electric dipole and a magnetic dipole. If we can set up many of these in a row in a long fiber, we can make a huge voltage and by extracting that voltage, we can use it as a power source.”
The light must be shone through a material that does not conduct electricity, such as glass. And it must be focused to an intensity of 10 million watts per square centimeter. Sunlight isn’t this intense on its own, but new materials are being sought that would work at lower intensities, Fisher said.
“In our most recent paper, we show that incoherent light like sunlight is theoretically almost as effective in producing charge separation as laser light is,” Fisher said.
This new technique could make solar power cheaper, the researchers say. They predict that with improved materials they could achieve 10 percent efficiency in converting solar power to useable energy. That’s equivalent to today’s commercial-grade solar cells.
“To manufacture modern solar cells, you have to do extensive semiconductor processing,” Fisher said. “All we would need are lenses to focus the light and a fiber to guide it. Glass works for both. It’s already made in bulk, and it doesn’t require as much processing. Transparent ceramics might be even better.”
In experiments this summer, the researchers will work on harnessing this power with laser light, and then with sunlight.
The paper is titled “Optically-induced charge separation and terahertz emission in unbiased dielectrics.” The university is pursuing patent protection for the intellectual property.
Provided by University of Michigan
http://www.physorg.com/news/2011-04-solar-power-cells-hidden-magnetic.html
Scientists aim to improve photosynthesis to increase food and fuel production
April 12, 2011
(PhysOrg.com) -- Two new initiatives at the University of Cambridge aim to address the growing demand on the Earth’s resources for food and fuel by improving the process of photosynthesis.
As part of a new collaboration, the scientists have been awarded the major component of a $4M initiative to improve the process of photosynthesis, which allows biological systems to convert sunlight into food and is also the source of fossil fuels.
Four transatlantic research teams – two of which include academics from Cambridge’s Department of Plant Sciences – will explore ways to overcome limitations in photosynthesis which could then lead to ways of significantly increasing the yield of important crops for food production or sustainable bioenergy.
Professor Howard Griffiths from the Department of Plant Sciences said: “Plants really matter, and for the next generation, plant and microbial productivity will become the focus of key global issues: the basis for feeding an additional 2-3 billion mouths, to drive forward an economy currently trading on past sunlight, and maintain biodiversity in the face of climate change.”
The funding has been awarded by the UK Biotechnology and Biological Sciences Research Council (BBSRC) and the US National Science Foundation (NSF) in a pioneering undertaking for the best minds from the USA and UK to join forces to explore this important research.
Despite the fact that photosynthesis is the basis of energy capture from the sun in plants, algae and other organisms, it has some fundamental limitations. There are trade-offs in nature which mean that photosynthesis is not as efficient as it could be – for many important crops such as wheat, barley, potatoes and sugar beet, the theoretical maximum is only 5%, depending on how it is measured. There is scope to improve it for processes useful to us, for example increasing the amount of food crop or energy biomass a plant can produce from the same amount of sunlight.
Some of the research will focus on improving a reaction driven by an enzyme called Rubisco, which is a widely recognised bottleneck in the photosynthesis pathway. By attempting to transfer parts from algae and bacteria into plants, the researchers hope to make the environment in the plants’ cells around Rubisco richer in carbon dioxide which will allow photosynthesis to produce sugars more efficiently.
Professor Griffiths added: “The enzymatic powerhouse Rubisco takes carbon dioxide from the atmosphere and uses light energy to produce sugars and other building blocks of life. However, the enzyme is rather flawed and somewhat promiscuous: it engages with oxygen as well as carbon dioxide, to the detriment of potential plant productivity.
“Some plants have evolved mechanisms, which act like biological turbochargers, to concentrate CO2 around Rubisco and improve the enzyme’s operating efficiency. These carbon concentrating mechanisms have evolved in certain key crops, such as sugar cane and maize. Other plants, such as aquatic algae, have developed mechanism in parallel which actively concentrate bicarbonate as a source of CO2 for Rubisco.”
The research projects have been funded by BBSRC and NSF following a multidisciplinary workshop held by the funders in California in September 2010. The workshop, called the Ideas Lab, enabled scientists from different disciplines and institutions in the UK and USA to explore ideas and potential projects before submitting them to BBSRC and NSF.
The Ideas Lab experience was likened by Professor Griffiths to be a combination of Big Brother, The Weakest Link and The Apprentice. Professor Griffiths is the consortium leader for one of the joint proposals funded, which will be exploring the operation of an algal carbon concentrating mechanism, and the possibility for introducing components into higher plant cells.
Dr. Julian Hibberd from the Department of Plant Sciences is part of one of the other initiatives which is seeking to increase the efficiency of light harvesting by broadening the wavelengths of light, as used by bacteria, to power biophysical transport processes in higher plants.
This research will consolidate a major Plant Sciences initiative at Cambridge, which is exploring the means to improve photosynthesis from the perspective of sustainable plant productivity and crop yields for the future. Additional work is also being undertaken by Dr. Hibberd to investigate the potential introduction of C4 photosynthetic traits into crops such as rice. This programme is part of a broader sweep of strategic research relevant to sustainable crop development, involving RNAi, pathogen suppression and epidemiological controls to maintain yields in a changing climate.
Provided by University of Cambridge
http://www.physorg.com/news/2011-04-scientists-aim-photosynthesis-food-fuel.html
Research into batteries will give electric cars the same range as petrol cars
April 6, 2011
Li-air batteries are a promising opportunity for electric cars. "If we succeed in developing this technology, we are facing the ultimate breakthrough for electric cars, because in practice, the energy density of Li-air batteries will be comparable to that of petrol and diesel, if you take into account that a combustion engine only has an efficiency of around 30 percent," says Tejs Vegge, senior scientist in the Materials Research Division at Risø DTU, Denmark. If batteries with an energy density this great become a reality, one could easily imagine electrically powered trucks.
The electric car was introduced by Edison as early as 1900. But, as we all know, Henry Ford's vehicle concept with a noisy, smelly combustion engine won the race to become people's most treasured individual means of transport, despite the fact that in principle, the combustion engine is hopeless.
Then, as now, the Achilles' heel of the electric car was the limited energy density of the batteries, which will only sustain short drives. Now – 110 years later – the battery technology, combined with the effect electronics and the electric engine, have come so far in performance, size and price that the electric car is again becoming interesting. The electric car does not pollute locally and it can, if used cleverly, be utilised to introduce more renewable energy into the electricity supply.
Electric cars are the perfect match for a society that has abandoned the use of fossil fuels.
This is why electric cars have been reborn as an important factor in the vision of a society without fossil fuels, and the first electric cars have already hit the roads, albeit in very limited numbers and with very short ranges between recharges.
The advantages of the electric car are first and foremost that it can be integrated into the electricity system and potentially serve as a buffer in the electricity system of tomorrow, where most of our electricity originates from fluctuating renewable energy. Where there is excess electricity from e.g. wind turbines, the electric cars can be charged. When there is a shortage of electricity, some of the power can be returned to the electricity grid. The other major advantage is that, if mass-produced, the electric car could be cheaper to produce than the current cars.
2 tonnes of batteries or 50 litres of petrol
Today, battery packs are expensive and are only able to store a relatively low amount of energy. Researchers all over the world are working to change that. In the current setting, an electric car is no good if you are taking the family on holiday to Lake Garda in Italy. For electric cars to become the consumers' preferred mode of transport, the battery capacity must be significantly increased. In Risø Energy Report 9, page 58, you can read that the energy density in today's batteries is almost two orders lower than that of fossil fuels. This means that a battery pack containing energy corresponding to 50 litres of petrol, would weigh between 1.5 and 2 tonnes.
The most promising electric car batteries are based on the metal lithium (Li). Lithium is a soft, silver-white metal – the lightest of all metals. Lithium is extremely reactive and corrodes quickly in a humid atmosphere. There, lithium is typically stored under kerosene to avoid contact with oxygen and water. The lightness is one of the strengths of lithium. Traditional car batteries are based on lead (Pb), which is one of the heaviest metals in existence. To reduce the weight of batteries, lithium is the way to go, which is also substantiated by the prominence of rechargeable Li-ion batteries in e.g. mobile phones, cameras and MP3 and MP4 players. These batteries have the highest energy density among rechargeable batteries.
The lithium battery market is going to grow exponentially, and a discussion has already emerged whether there is going to be enough lithium to electrify the entire world's car park. Lithium is naturally occurring with approx. 65 g per tonne in top soil and approx. 0.1 g per tonne of water and can be extracted from soil as well as water, but if the lithium content is small, the extraction is costly.
In addition to the use in batteries, lithium is used in anti-depressants, ceramics, glass, aluminium production, lubricants and synthetic rubber. In the future (after 2050), lithium will probably also be used in fusions reactors for electricity production. The world's lithium reserves are found in countries such as Chile, China, Australia, Russia, Argentina, the USA, Zimbabwe and Bolivia. Lately, large deposits have been found in Afghanistan – so large that the USA has dubbed the country 'the Saudi Arabia of lithium'. In Bolivia, lithium is found in large quantities under Salar de Uyuni – the world's largest salt lake. Last year, Bolivia's president Morales announced that the country is going to invest DKK 5 billion in extracting lithium from the dried-out salt lake that covers more than 10,000 square kilometres and contains more than a quarter of the world's total lithium deposits.
The fight over the world's lithium resources will intensify in the future, but the upside is that the lithium part of batteries can be recycled, so when the batteries are worn out, the lithium can be extracted and form part of a new battery.
Batteries, a research theme at Risø DTU
At Risø DTU, two divisions possess great expertise which can be used to develop better electric car batteries. One is the Materials Research Division and the other is the Fuel Cells and Solid State Chemistry Division.
Together, these two divisions have excellent competencies within modelling and characterisation of synthesis as well as electrochemistry, all of which are vital aspects in the development of new batteries and other forms of chemical energy storage.
Research is being conducted at Risø DTU into the development and manufacture of new battery materials, e.g. improved electrode and electrolyte materials for the next generation of Li-ion and Li-air batteries. This requires insight and 'nano-scale engineering' as well as detailed understanding of the underlying processes.
Risø DTU thus has the best possible prerequisites for delivering exactly the package required to boost battery research considerably. It's all about skills within durability and product life, energy density as well as stability and safety.
Li-air batteries could have the same efficient energy density as petrol
Li-air batteries are a promising opportunity in the long term. "If we succeed in developing this technology, we are facing the ultimate break-through for electric cars, because in practice, the energy density of Li-air batteries will be comparable to that of petrol and diesel, if you take into account that a combustion engine only has an efficiency of around 30 per cent," says Tejs Vegge, senior scientist in the Materials Research Division. If batteries with an energy density this great become a reality, one could easily imagine electrically powered trucks.
Li-air batteries are thus a promising research area, but there are many research challenges to overcome before the batteries find their way to the electric cars.
The development of rechargeable batteries has moved slowly since the invention of the traditional lead-acid batteries, which are still used in the majority of e.g. starter batteries for conventional cars. The development of the Li-ion batteries marked a significant leap in the energy density of the rechargeable batteries. The final break-through may belong to the Li-air batteries which, in practice, could have the same efficient energy density as petrol. Source: Lithium – Air Battery: Promise and Challenges, G. Girishkumar, B. McCloskey, A.C. Luntz, S. Swanson and W. Wilcke, IBM Research, published in J.Phys.Chem.Lett. 2010, 1, 2193-2203.
The Li-air battery is designed with a lithium electrode (the anode), and electrolyte and a porous carbon electrode (the cathode), which attracts the oxygen from the air when the battery is in operation. The battery is therefore, so to speak, open at one end, or it has an oxygen supply of its own. During discharge, oxygen reacts with lithium to form lithium peroxide (Li2O2), and during charging, this process is reversed to release oxygen. Both reactions take place on the surface of the porous carbon electrode.
Battery resembles humans: Gains weight and becomes short of breath
The interaction with air requires the electrode to have a very large surface area. The prototypes being worked on now have a current density of approx. 1 milliamp per square centimetre surface area, and this has to be increased by at least one order before the batteries are ready to be used in real life.
The fact that the battery absorbs oxygen atoms from the air means that the battery gains weight as it being discharged. Theoretically, the battery can more than double its weight.
At the same time, the electrode could become short of breath, so to speak. The oxygen absorbed by the battery reacts with lithium to form lithium peroxide, which may cause clogging of aggregates in the battery's channels, causing them to become blocked and preventing the supply of further oxygen. "In our trials, we use pure oxygen, so we are okay, but the problems accumulate when the oxygen has to be extracted from ordinary air," says Søren Højgaard Jensen from the Fuel Cells and Solid State Chemistry Division. Ordinary air also contains moisture, and it must be taken into consideration that, as mentioned above, lithium and humidity do not make an attractive combination.
Difficult to charge
En extremely high overvoltage is required to recharge the battery again after a discharge. The so-called equilibrium voltage for the Li-air battery is 3 volts. When the battery is discharged, the voltage drops to 2.6-2.7 volts. But when you want to recharge the battery, the voltage must be increased to 4.5 volts. In comparison, a Li-ion battery can be recharged at an overvoltage of only 10 per cent.
"The discharge process is proceeding really well. Our problem is that the reverse process has a very high energy loss," says senior scientist Poul Norby, Materials Research Division. "The high overvoltage for recharging is hard going for the current battery components, which limits the number of times the battery can be recharged," says Poul Norby. The cyclic energy loss in charging/recharging is about 40 per cent in Li-air batteries. The challenge is to reduce this number to 10 per cent, corresponding to Li-ion batteries.
In order to solve this issue, Tejs Vegge performs extensive computer calculations, so-called DFT calculations (Density Functional Theory), on the Li-air batteries. Using this method, it is possible – at atom level applying an approximation to the famous Schrödinger equation, to calculate how the lithium and oxygen atoms interact. "In this way, we hope to find an explanation of the high overvoltage and a solution to what we can do to reduce it, e.g. by adding an appropriate catalyst," says Tejs Vegge.
In addition to the computer calculations, the batteries are examined using X-ray and neutron rays. These techniques allow the scientists to study how ions and electrons move in the electrode-electrolyte interfaces when the battery is charged and discharged. "We focus particularly on solid-state electrolytes because they offer safety and transport advantages. Large lithium batteries with liquid electrolytes could pose a safety risk in the event of accidents," says Tejs Vegge.
Finally, the battery properties are tested in practice. Testing of large lithium batteries takes place in a converted chest freezer in the laboratories of the Fuel Cells and Solid State Chemistry Division. "The batteries have to be able to withstand heavy frost and extreme heat, and we can subject them to that in our converted chest freezer, which is able to cool objects down to -60°C and heat them to around 50°C," says Søren Højgaard Jensen.
Must recharge quickly – and at least 300 times
Today, metal-air batteries are only used as disposable batteries for special purposes with high energy density requirements, e.g. for military equipment, and zinc-air batteries are used as disposable batteries in e.g. hearing aids.
If the battery is to withstand a car running e.g. 250,000 kilometres during its lifetime, and the battery is able to deliver approx. 800 kilometres from one charge, it must be able to handle full charging and discharging at least 300 times. Li-air battery prototypes can currently handle 50 charges, so the researchers are faced with other scientific challenges.
In addition to the number of charges the battery must be able to withstand, it must also be possible to charge it quickly. "Think about the volume of energy transferred when you put petrol into your car. It takes a couple of minutes, and then you can go another 800-1000 kilometres. This is a true challenge for the Li-air batteries, because they may potentially be able to contain the same amount of energy as petrol, but it takes considerably longer to refuel," says Tejs Vegge.
Provided by Technical University of Denmark
http://www.physorg.com/news/2011-04-batteries-electric-cars-range-petrol.html
IBM To Offer Nanotechnology Assuring Long Battery Life
Filed in archive Computing & Infotech on March 21, 2011
IBM is soon going to offer a nanotechnology ensuring longer battery life. Designed by Illinois researchers the nanotechnology, comprising a 3-D nanostructure for battery cathodes allowing for speedy charge and discharge without sacrificing capacity, will considerably extend the battery life of portable devices.
People often forgetting to recharge their batteries will benefit the most through IBM's nanotechnology, requiring them to recharge their batteries just once a month! Power consumption in the flash memory of portable devices is reduced significantly, so it is power saving. And since it makes use of carbon nanotubes that do not get spoilt like metal wires, it assures longevity.
Illinois' Professor, Paul Braun, is also optimistic for the potential of the nanotechnology's use in batteries of electric vehicles, medical devices, lasers and military applications.
http://www.nanotechbuzz.com/41765700/ibm_to_offer_nanotechnology_assuring_long_battery_life.php
3D Nanostructure for Cathodes in Batteries Could Mean Cell Phones that Charge in Seconds
POSTED BY: Dexter Johnson / Tue, March 22, 2011
No sooner do I discuss University of Illinois researchers who have created 3D antennas for mobile phones using nanotechnology than another group of researchers at the University of Illinois (this time at Urbana-Champaign) have developed 3D material for batteries that combines the qualities of supercapacitors with those of batteries and could change the entire battery paradigm.
Professor Paul Braun and his colleagues just published in the March 20th edition of the journal Nature Nanotechnology their results that showed ultra fast charge and discharge rates by “using cathodes made from a self-assembled three-dimensional bicontinuous nanoarchitecture consisting of an electrolytically active material sandwiched between rapid ion and electron transport pathways.”
What this could mean, according to the excited science and technology press, are electric cars that could be charged in five minutes, a laptop in just a couple of minutes and a cell phone in seconds.
While thin film technology has allowed faster charging capabilities than seen in your typical li-ion batteries but it can’t store the energy well, meaning that a mobile device would run out of power in mere seconds.
What Braun and his team have done essentially is to take the thin film technology but built it up through self-assembly into a three-dimensional structure thereby increasing its surface area and its ability to store energy.
The actual structure apparently resembles a lattice of tightly packed spheres. Metal is used to fill in the spaces around the spheres and then it is all melted leaving a 3D scaffold that appears like a sponge. Then the structure is electropolished that increases the size of the pores.
The result is that lithium ions can move rapidly through the material with a high electrical conductivity.
According to Braun this could revolutionize the battery. "We like that it's very universal,” Braun is quoted as saying in a number of articles covering the report. “This is not linked to one very specific kind of battery, but rather it's a new paradigm in thinking about a battery in three dimensions for enhancing properties."
TAGS: battery technology // li-ion batteries // nanostructure // self-assembly // supercapacitors
http://spectrum.ieee.org/nanoclast/semiconductors/nanotechnology/3d-nanostructure-for-cathodes-in-batteries-could-mean-cell-phones-that-charge-in-seconds
My brother was a PhD Nuclear Physics guru (Rice Univ) (sadly he passed away last year), but I was talking recently with a close friend and associate of his (also a top of the field physicist) who claims the Brussard drive (Polywell) is the real deal:
http://en.wikipedia.org/wiki/Polywell
The Google founders nearly funded it with a VC deal. It may be what you are talking about?
Nano!
Have you ever looked at ERC? CO2 ElectroReduction of CO2 to formic acid, which can be used as a fuel in fuel cells?
MVTG owns the tech, and is a penny stock.
Or CABN which is trying to commercialize CO2 & CH4 conversion to syngas for conversion to gasoline? Both use what I consider to be novel nanotech catalysts, but they do not promote the nanotech angle. I own some of both.
One of my companies - Applied Nanotech (formerly - Nanoproprietary and SI Diamond) - has/had an involvement in that very thing some years back - let me see if I can find it:
R.L. Fink is the guy who now works at Applied Nanotech:
http://aerospace.engin.umich.edu/people/faculty/gallimore/AGallimoreJanuary2011.pdf
http://en.wikipedia.org/wiki/Electrostatic_ion_thruster
Rather than a jet of hot gas as the thrust force they use a jet of high velocity ions - not much force but long lasting!
Have you ever heard of ion drive? A ship travels through space and uses ion fuel from the stars.The speed of the ship approaches light speed. Supposedly there are companies working on these engines and they should be ready to test before the next decade. If you're interested I will try and research but you probably have info already, Mr Info man!
As long as the sun keeps shining! The sun is the source of energy. So no perpetual motion is involved.
Magnetism? We use that to generate electricity but one needs to input energy from falling water, wind or steam to move a wire through a magnetic field to get electricity generated.
What I figure would be great would be to have a black hole energy source which though tiny would be a great energy source - trouble is how to contain it in a box or whatever - being a black hole it just sucks everything in!
This is close to perpetual motion, don't you think? I have to hand it to you this is incredible. I have always invisioned a small black box with a receptical and from that box you get unlimited electricity. Now it's about to become reality. The ocean plus sewage and nano catalyst all inside the box. What's next harnessing magnetism?
Hydrogen and oxygen are generated so they could have a hydrogen range to cook with.
When?
By next year the team expects to be able to power a small home with only roughly a bottle and a half of water.
How soon before Sun Catalytix has a product to market? This could be enormous. Supplied power as it now exists is about to be obsolete. I never thought electric cars were overall very practical, but with this new cheap source of power all electric will be our future, except for cooking. We have to have gas ranges.
Nanotechnology battery uses salinity difference between fresh- and seawater to create electricity
Posted: Mar 28th, 2011
(Nanowerk News) Stanford researchers have developed a battery that takes advantage of the difference in salinity between freshwater and seawater to produce electricity.
Anywhere freshwater enters the sea, such as river mouths or estuaries, could be potential sites for a power plant using such a battery, said Yi Cui, associate professor of materials science and engineering, who led the research team.
The theoretical limiting factor, he said, is the amount of freshwater available. "We actually have an infinite amount of ocean water; unfortunately we don't have an infinite amount of freshwater," he said.
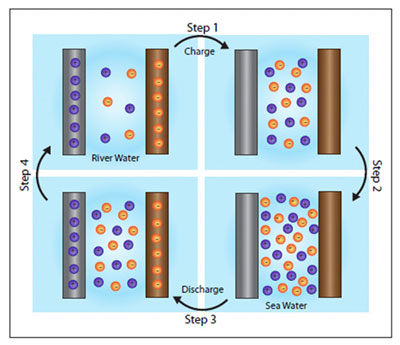
The cycle that generates electricity in Cui's new battery.
As an indicator of the battery's potential for producing power, Cui's team calculated that if all the world's rivers were put to use, their batteries could supply about 2 terawatts of electricity annually – that's roughly 13 percent of the world's current energy consumption.
The battery itself is simple, consisting of two electrodes – one positive, one negative – immersed in a liquid containing electrically charged particles, or ions. In water, the ions are sodium and chlorine, the components of ordinary table salt.
Initially, the battery is filled with freshwater and a small electric current is applied to charge it up. The freshwater is then drained and replaced with seawater. Because seawater is salty, containing 60 to 100 times more ions than freshwater, it increases the electrical potential, or voltage, between the two electrodes. That makes it possible to reap far more electricity than the amount used to charge the battery.
"The voltage really depends on the concentration of the sodium and chlorine ions you have," Cui said. "If you charge at low voltage in freshwater, then discharge at high voltage in sea water, that means you gain energy. You get more energy than you put in."
Once the discharge is complete, the seawater is drained and replaced with freshwater and the cycle can begin again. "The key thing here is that you need to exchange the electrolyte, the liquid in the battery," Cui said. He is lead author of a study published in the journal Nano Letters earlier this month ("Batteries for Efficient Energy Extraction from a Water Salinity Difference") [ http://pubs.acs.org/doi/abs/10.1021/nl200500s?journalCode=nalefd ].
In their lab experiments, Cui's team used seawater they collected from the Pacific Ocean off the California coast and freshwater from Donner Lake, high in the Sierra Nevada. They achieved 74 percent efficiency in converting the potential energy in the battery to electrical current, but Cui thinks with simple modifications, the battery could be 85 percent efficient.
To enhance efficiency, the positive electrode of the battery is made from nanorods of manganese dioxide. That increases the surface area available for interaction with the sodium ions by roughly 100 times compared with other materials. The nanorods make it possible for the sodium ions to move in and out of the electrode with ease, speeding up the process.
Other researchers have used the salinity contrast between freshwater and seawater to produce electricity, but those processes typically require ions to move through a membrane to generate current. Cui said those membranes tend to be fragile, which is a drawback. Those methods also typically make use of only one type of ion, while his battery uses both the sodium and chlorine ions to generate power.
Cui's team had the potential environmental impact of their battery in mind when they designed it. They chose manganese dioxide for the positive electrode in part because it is environmentally benign.
The group knows that river mouths and estuaries, while logical sites for their power plants, are environmentally sensitive areas.
"You would want to pick a site some distance away, miles away, from any critical habitat," Cui said. "We don't need to disturb the whole system, we just need to route some of the river water through our system before it reaches the ocean. We are just borrowing and returning it," he said.
The process itself should have little environmental impact. The discharge water would be a mixture of fresh and seawater, released into an area where the two waters are already mixing, at the natural temperature.
One of Cui's concerns is finding a good material for the negative electrode. He used silver for the experiments, but silver is too expensive to be practical.
His group did an estimate for various regions and countries and determined that South America, with the Amazon River draining a large part of the continent, has the most potential. Africa also has an abundance of rivers, as do Canada, the United States and India.
But river water doesn't necessarily have to be the source of the freshwater, Cui said.
"The water for this method does not have to be extremely clean," he said. Storm runoff and gray water could potentially be useable.
A power plant operating with 50 cubic meters of freshwater per second could produce up to 100 megawatts of power, according to the team's calculations. That would be enough to provide electricity for about 100,000 households.
Cui said it is possible that even treated sewage water might work.
"I think we need to study using sewage water," he said. "If we can use sewage water, this will sell really well."
Source: Stanford University (Louis Bergeron)
http://www.nanowerk.com/news/newsid=20754.php
Huge Potential of Nanocrystals to Raise Efficiency in Fuel Cells
ScienceDaily (Mar. 28, 2011) — The addition of extremely small crystals to solid electrolyte material has the potential to considerably raise the efficiency of fuel cells. Researchers at TU Delft were the first to document this accurately, and this week their second article on the subject in a very short time was published in the scientific journal, Advanced Functional Materials.
Electrolyte
The researchers at the Faculty of Applied Sciences at TU Delft were concentrating their efforts on improving electrolyte materials. This is the material between two electrodes, for example in a fuel cell or a battery. The better the characteristics of the electrolyte, the better, more compactly or more efficiently the fuel cell or battery works.
Solid matter
The electrolyte is usually a liquid, but this has a number of drawbacks. The liquid has to be very well enclosed, for example, and it takes up a relatively large amount of space. "It would therefore be preferable to have an electrolyte made of solid matter," says PhD student drs. Lucas Haverkate. "Unfortunately though, that has disadvantages as well. The conductivity in solid matter is not as good as it is in a liquid."
Traffic jam on the motorway
"In a solid matter you have a network of ions, in which virtually every position in the network is taken. This makes it difficult for the charged particles (protons) to move from one electrode to another. It's a bit like a traffic jam on a motorway. What you need to do is to create free spaces in the network."
Nanocrystals
One of the ways of achieving this, and therefore of increasing conductivity in solid electrolytes, is to add nanocrystals (of seven nanometres to around fifty nanometres), of Titanium Dioxide in this case. "A characteristic of these TiO2 crystals is that they attract protons, and this creates more space in the network." The nanocrystals are mixed in the electrolyte with a solid acid (CsHSO4). This latter material 'delivers' the protons to the crystals. "The addition of the crystals appears to cause an enormous leap in the conductive capacity, up to a factor of 100," concludes Haverkate.
Neutrons
Chan carried out measurements on the electrolyte material using the neutron diffraction method. This involves sending neutrons through the material. The way in which the neutrons are dispersed makes it possible to deduce certain characteristics of the material, such as the density of protons in the crystals. Haverkate: "It is the first time that measurements have been taken of solid-material electrolytes in this way, and on such a small scale. The fact that we had nuclear research technologies at the Reactor Institute Delft at our disposal was tremendously valuable."
Temperature
However, the combination of TiO2 and CsHSO4 does not mark the end of the search for a suitable solid-material electrolyte. Other material combinations will be tested that may achieve better scores in the area of stability, for example. Professor Fokko Mulder, who is Haverkate's and Chan's PhD supervisor, says. "At this stage, we are more concerned about acquiring a fundamental understanding and a useful model, than the concrete issue of finding out what the most suitable material is. It is important that we identify the effect of nanocrystals, and give it a theoretical basis. I think there is great potential for these electrolytes. They also have the extra benefit of continuing to function well over a wide range of temperatures, which is of particular relevance for applying them in fuel cells."
This achievement by TU Delft has already led to two publications in the scientific journal Advanced Functional Materials. Last December, Haverkate published an article on the theory behind the results. His fellow PhD student, Wing Kee Chan, is the main author of a second item that appeared in the same publication this week. Chan focused on the experimental side of the research. "The nice thing about these two publications is that the experimental results and the theoretical underpinning strongly complement each other," says Haverkate.
Story Source:
The above story is reprinted (with editorial adaptations by ScienceDaily staff) from materials provided by Delft University of Technology.
Journal References:
Wing K. Chan, Lucas A. Haverkate, Wouter J. H. Borghols, Marnix Wagemaker, Stephen J. Picken, Ernst R. H. van Eck, Arno P. M. Kentgens, Mark R. Johnson, Gordon J. Kearley, Fokko M. Mulder. Direct View on Nanoionic Proton Mobility. Advanced Functional Materials, 2011; DOI: 10.1002/adfm.201001933
[ http://dx.doi.org/10.1002/adfm.201001933 ]
Lucas A. Haverkate, Wing K. Chan, Fokko M. Mulder. Ionic Nanosystems: Large Space-Charge Effects in a Nanostructured Proton Conductor (Adv. Funct. Mater. 23/2010). Advanced Functional Materials, 2010; 20 (23): 4018 DOI: 10.1002/adfm.201090105
[ http://dx.doi.org/10.1002/adfm.201090105 ]
http://www.sciencedaily.com/releases/2011/03/110328093059.htm
In re heat resistance - like in automotive catalytic converters - I note this:
The nanocomposite can be catalytic. Catalytic functionality can be provided by either the inorganic phase or the nanoparticle. In certain embodiments, the nanocomposite can withstand high temperature applications (such as catalytic conversion) without sintering. The catalyst can be a photocatalyst. The catalyst can be an oxidation catalyst.
Vive Nano Inc. = Private company.
http://en.wikipedia.org/wiki/Vive_Nano
Not likely that PDDA could survive the temperature extremes in auto exhaust. But the carbon nano tubes might, so no doubt someone is testing a carbon nanotube modification for the auto and diesel exhaust application, possibly with nickel....etc. But the PDDA-CNT might do interesting things in a battery.
Conceptually, I like to look at fuel cells as flow through batteries, and visa versa!
This prior art is the closest I could find:
Preparation method of electrode modified by carbon nano-tube
Publication number:
CN101158663 (A)
Publication date:
2008-04-09
Inventor(s):
LIYI SHI [CN]; SHUPING ZHANG [CN]; LIANGANG SHAN [CN] +
Applicant(s):
UNIV SHANGHAI SCIENCE & TECH [CN] +
Application number:
CN20071170699 20071121
Priority number(s):
CN20071170699 20071121
Also published as:
CN101158663 (B)
Abstract of CN 101158663 (A)
The invention discloses a carbon nanometer tube modified electrode preparation method, which comprises raw materials solution preparation, carbon nanometer tube galvanic deposit at a glassy carbon electrode surface and layer upon layer static electricity self-assemble process. The invention deposits the carbon nanometer tube with negative charge to the glassy carbon electrode surface by using galvanic deposit technology, thus solving the problem that the belt negative charge quantity is insufficient after the glassy carbon electrode surface pretreatment, then layer upon layer static electricity self-assemble process is carried on in PDDA solution and carbon nanometer tube dispersing liquid, finally the high self-assembled efficiency and stabile modified electrode is obtained. The invention effectively modifies carbon nanometer tube in the glassy carbon electrode surface.; The formed modified material layer has the catalytic effect to the electrochemistry active material, enhances the examination sensitivity, reduces the oxidizing reaction electric potential, and enhances electrode selectivity and stability.
http://v3.espacenet.com/publicationDetails/biblio?DB=EPODOC&adjacent=true&locale=en_EP&FT=D&date=20080409&CC=CN&NR=101158663A&KC=A
And this:
(WO/2011/033377) MULTIFUNCTIONAL NANOCOMPOSITES
Pub. No.:
WO/2011/033377
International Application No.:
PCT/IB2010/002341
Publication Date:
24.03.2011
International Filing Date:
17.09.2010
Applicants:
VIVE NANO, INC. [CA/CA]; 700 Bay Street, Suite 1100 Toronto, Ontario M5G 1Z6 (CA) (All Except US).
LOUKINE, Nikolai [CA/CA]; (CA) (US Only).
DAS, Anjan [IN/CA]; (CA) (US Only).
NORTON, Danielle [CA/CA]; (CA) (US Only).
ANDERSON, Darren, J. [CA/CA]; (CA) (US Only).
Inventors: LOUKINE, Nikolai; (CA).
DAS, Anjan; (CA).
NORTON, Danielle; (CA).
ANDERSON, Darren, J.; (CA).
Priority Data:
61/243,349
17.09.2009 US
Title:
MULTIFUNCTIONAL NANOCOMPOSITES
Abstract:
The present invention provides a multifunctional nanocomposite with at least two components, at least one component of which is a nanoparticle that includes a polymer.
Claims
1. A multifunctional nanocomposite comprising at least two components, at least one component of which is a nanoparticle comprising a polymer and the other component comprises an inorganic phase.
11. The multifunctional nanocomposite of claim 1, wherein the secondary inorganic phase is selected from the group consisting of amorphous carbon, pyrolytic carbon, activated carbon, charcoal, ash, graphite, fullerenes, nanotubes and diamond.
19. The multifunctional nanocomposite of claim 1, wherein the nanocomposite is a catalyst.
http://is.gd/BhYZhH
VIVE NANO, INC:
http://www.nntech.com/index.html
Just thinking out loud - would this Pt replacement PDDA-CNT catalyst also replace the Pt catalyst in automotive catalytic mufflers? And anywhere else for that matter that Pt is used as a catalyst?
I found a ton of news hits googling the polymer compound. Looks like a full court news press this week on it!
http://www.google.com/search?q=polydiallyldimethylammoniumn+chloride&ie=utf-8&oe=utf-8&aq=t&rls=org.mozilla:en-US:official&client=firefox-a
Debut of the first practical 'artificial leaf'
Posted: Mar 27th, 2011
(Nanowerk News) Scientists today claimed one of the milestones in the drive for sustainable energy — development of the first practical artificial leaf. Speaking at the 241st National Meeting of the American Chemical Society, they described an advanced solar cell the size of a poker card that mimics the process, called photosynthesis, that green plants use to convert sunlight and water into energy.
"A practical artificial leaf has been one of the Holy Grails of science for decades," said Daniel Nocera, Ph.D., who led the research team. "We believe we have done it. The artificial leaf shows particular promise as an inexpensive source of electricity for homes of the poor in developing countries. Our goal is to make each home its own power station," he said. "One can envision villages in India and Africa not long from now purchasing an affordable basic power system based on this technology."
The device bears no resemblance to Mother Nature's counterparts on oaks, maples and other green plants, which scientists have used as the model for their efforts to develop this new genre of solar cells. About the shape of a poker card but thinner, the device is fashioned from silicon, electronics and catalysts, substances that accelerate chemical reactions that otherwise would not occur, or would run slowly. Placed in a single gallon of water in a bright sunlight, the device could produce enough electricity to supply a house in a developing country with electricity for a day, Nocera said. It does so by splitting water into its two components, hydrogen and oxygen.
The hydrogen and oxygen gases would be stored in a fuel cell, which uses those two materials to produce electricity, located either on top of the house or beside it.
Nocera, who is with the Massachusetts Institute of Technology, points out that the "artificial leaf" is not a new concept. The first artificial leaf was developed more than a decade ago by John Turner of the U.S. National Renewable Energy Laboratory in Boulder, Colorado. Although highly efficient at carrying out photosynthesis, Turner's device was impractical for wider use, as it was composed of rare, expensive metals and was highly unstable — with a lifespan of barely one day.
Nocera's new leaf overcomes these problems. It is made of inexpensive materials that are widely available, works under simple conditions and is highly stable. In laboratory studies, he showed that an artificial leaf prototype could operate continuously for at least 45 hours without a drop in activity.
The key to this breakthrough is Nocera's recent discovery of several powerful new, inexpensive catalysts, made of nickel and cobalt, that are capable of efficiently splitting water into its two components, hydrogen and oxygen, under simple conditions. [Would those polymer dipped CNTs work??] Right now, Nocera's leaf is about 10 times more efficient at carrying out photosynthesis than a natural leaf. However, he is optimistic that he can boost the efficiency of the artificial leaf much higher in the future.
"Nature is powered by photosynthesis, and I think that the future world will be powered by photosynthesis as well in the form of this artificial leaf," said Nocera, a chemist at Massachusetts Institute of Technology in Cambridge, Mass.
Source: American Chemical Society
http://www.nanowerk.com/news/newsid=20717.php
Researchers claim fuel cell breakthrough
[For the record!]
Published: March. 22, 2011 at 6:29 PM
CLEVELAND, March 22 (UPI) -- U.S. researchers say they've made a breakthrough in the development of low-cost hydrogen fuel cells that one day could power electric cars.
Researchers at Case Western Reserve University in Cleveland say catalysts made of carbon nanotubes dipped in a polymer solution can outperform traditional platinum catalysts in fuel cells at a fraction of the cost.
The scientists say the new technology can remove one of the biggest roadblocks to widespread cell use: the cost of the catalysts.
Platinum, which represents at least a quarter of the cost of fuel cells, currently sells for about $30,000 per pound, while the activated carbon nanotubes cost about $45 per pound, a Case release said Tuesday.
"This is a breakthrough," Liming Dai, a professor of chemical engineering and the research team leader, said.
Soaking carbon nanotubes in a water solution of the polymer for a couple of hours coats the nanotube surface and pulls an electron partially from the carbon, creating a net positive charge, researchers said.
When placed on the cathode of an alkaline fuel cell, the charged material acts as a catalyst for the oxygen-reduction reaction that produces electricity by electrochemically combining hydrogen and oxygen.
In testing, the researchers' carbon catalyst fuel cell produced as much power as an identical cell using a platinum catalyst.
Dai said he's confident his lab can increase the energy output of the new process.
"We have not optimized the system yet," he said.
One widely researched use for such cells would be to produce electricity to power an electric car, using hydrogen and oxygen from the air. The only emission from such a vehicle, researchers say, would be water.
http://www.upi.com/Science_News/2011/03/22/Researchers-claim-fuel-cell-breakthrough/UPI-73011300832974/
Cheap Catalyst Made Easy
ScienceDaily (Mar. 22, 2011) — Catalysts made of carbon nanotubes dipped in a polymer solution equal the energy output and otherwise outperform platinum catalysts in fuel cells, a team of Case Western Reserve University engineers has found.
The researchers are certain that they'll be able to boost the power output and maintain the other advantages by matching the best nanotube layout and type of polymer.
But already they've proved the simple technique can knock down one of the major roadblocks to fuel cell use: cost.
Platinum, which represents at least a quarter of the cost of fuel cells, currently sells for about $65,000 per kilogram. These researchers say their activated carbon nanotubes cost about $100 per kilogram.
Their work is published in the online edition of Journal of the American Chemical Society.
"This is a breakthrough," said Liming Dai, a professor of chemical engineering and the research team leader.
Dai and research associates Shuangyin Wang and Dingshan Yu found that by simply soaking carbon nanotubes in a water solution of the polymer polydiallyldimethylammoniumn chloride for a couple of hours, the polymer coats the nanotube surface and pulls an electron partially from the carbon, creating a net positive charge.
They placed the nanotubes on the cathode of an alkaline fuel cell. There, the charged material acts as a catalyst for the oxygen-reduction reaction that produces electricity while electrochemically combining hydrogen and oxygen.
In testing, the fuel cell produced as much power as an identical cell using a platinum catalyst.
But the activated nanotubes last longer and are more stable, the researchers said. Unlike platinum, the carbon-based catalyst: doesn't lose catalytic activity and, therefore, efficiency, over time; isn't fouled by carbon monooxide poising; and is free from the crossover effect with methanol. Methanol, a liquid fuel that's easier to store and transport than hydrogen, reduces activity of a platinum catalyst when the fuel crosses over from the anode to the cathode in a fuel cell.
The new process builds on the Dai lab's earlier work using nitrogen-doped carbon nanotubes as a catalyst. In that process, nitrogen, which was chemically bonded to the carbon, pulled electron partially from the carbon to create a charge. Testing showed the doped tubes tripled the energy output of platinum.
Dai said the new process is far simpler and cheaper than using nitrogen-doped carbon nanotubes and he's confident his lab will increase the energy output as well. "We have not optimized the system yet."
Story Source:
The above story is reprinted (with editorial adaptations by ScienceDaily staff) from materials provided by Case Western Reserve University, via EurekAlert!, a service of AAAS.
Journal Reference:
Shuangyin Wang, Dingshan Yu, Liming Dai. Polyelectrolyte Functionalized Carbon Nanotubes as Efficient Metal-free Electrocatalysts for Oxygen Reduction. Journal of the American Chemical Society, 2011; : 110317101957076 DOI: 10.1021/ja1112904 [ http://dx.doi.org/10.1021/ja1112904 ]
http://www.sciencedaily.com/releases/2011/03/110322151252.htm
http://www.upi.com/Science_News/2011/03/22/Researchers-claim-fuel-cell-breakthrough/UPI-73011300832974/
Replacing platinum catalyst in fuel cells with polymer dipped carbon nanotubes!
Sun Cataytix IP
5 results found in the Worldwide database for:
SUN CATALYTIX as the applicant
http://v3.espacenet.com/searchResults?PA=SUN+CATALYTIX&DB=EPODOC&submitted=true&locale=en_EP&ST=advanced&compact=false
I notice that the TATA people are involved with Nocera of MIT! TATA developed the $2500 TATA NANO car for the masses in India and appear to want to provide the masses in India and elsewhere with a fridge sized unit to power their home and auto using local sunlight - POWER TO THE PEOPLE gets a brand new meaning!! And what with the problems with nuclear power in Japan, none too soon.
Thank you for forming this Board. I am very interested in this subject and believe we will have cheap power soon.
Nanopatent - Thanks for alerting me about this board. This is the concept video from Dan Nocera FWIW...made a year or two back. http://vimeo.com/8194089
They do have some major money backers!!!!
http://www.suncatalytix.com/Sun_Catalytix_Series_B_Press_Release.pdf
WOW
Here's more on Nocera - I've followed him for years:
http://donpatent.blogspot.com/2008/08/solar-power-breakthrough.html
I love the way the press and scientists leave out critical details like the rate. It says it can power a house with 1.5 bottles of water. It does not say how big the bottles are (volume or mass), and does not say how long it powers the house, i.e. 1.5 bottles per minute, hour, day, week, month year or decade, LOL!
I also do no not buy the using grey water plan. They will have all kinds of serious problems with contamination, catalyst poising, scaling etc if they do not use DI water!!!! But that might not be a deal killer if they catch clean enough rain water?
I think I have seen a photo catalytic water splitting catalyst before (I assume that is what they are doing although it does not say that precisely).
I like that expanded title!!!! Sounds great!!!
I just posted a stab at my vision on the meaning of nano tech to me on Axion. Here it is for us. Might help explain why I do not see a clean clear cut, discrete "old is not Nano, new is nano" vision. I see it as old nano, and then new, perhaps better nano just for future reference.
I found a working definition that may helpful to this discussion:
http://www.epa.gov/ncer/nano/questions/index.html
But as with so many things the EPA does, they missed the boat here:
Quote:Nanotechnology is really something new:
Many things we are already familiar with are nanoscale and analogous to applications of nanotechnology. For instance, living organisms from bacteria to beetles rely on nanometer-sized protein machines that do everything from whipping flagella to flexing muscles. All biological cells are comprised of smart materials that self-assemble. Nanometer-sized carbon (carbon black) that improves the mechanical properties of tires, nanometer silver particles that initiate photographic film development, and nanometer particles that are the basis of catalysts critical to the petrochemical industry have contributed to commercial products for many years.
However, all of the above examples of technologies are not considered nanotechnology because they do not involve specific atomic manipulations to achieve desired properties and functions of materials or products. Nanotechnology does involve purposeful atomic manipulations and structural assembly to achieve predetermined properties and functions.
Being a part of the government they love to re write the english language and ignore the obvious for the purpose of legalize regulatory purposes. I propose that they got it right in the first half, and then should have distinguished between naturally occurring (evolution, natural selection....and so on), natural nanotechnology and man made, synthetic nanotechnology.
Secondly their last statement is simply silly (absurd even) to any real scientist. In my mind, the question should be whether or not the manipulation produces something not previously found in nature!
On that basis I propose that the nano scale molecular structures and conditions in a lead acid battery are (1) not found in nature (hydrated lead sulfate particles for instance), and indeed (2) are nano scale and there for are in fact early examples of nanotechnology. I could also argue the plating and re-dissolving of lead is a nano scale process at the surfaces in a lead acid battery.
Perhaps at some point, they will come up with a better definition and separation of what was (past), and what is being invented that is truly new.
I can offer hoards of examples of old tech that even fit the EPA definition above, that defeat the EPA's intended purpose of writing that definition. Simply adding or removing one atom from a naturally occurring protein or enzyme molecule would fit that definition they used above, which would mean that every synthetic bio-organic compound produced in the last 200 years was nano technology.
Who needs batteries!!
Creating power from water
[With a little help from the sun!]
March 25, 2011 by Katie Gatto

Image credit: J. Am. Chem. Soc., 2009, 131 (11), pp 3838-3839. DOI: 10.1021/ja900023k [ http://pubs.acs.org/doi/abs/10.1021/ja900023k ]
VIDEOS:
Sorry - that link requires a paying member.
Perhaps I could expand the realm of coverage with this heading - "Energy production and storage using nanotechnology".
I requested that change from iHub - request pending.
These are those messages involved:
Search for: nano* on AXION POWER INTERNATIONAL
Subject Author Date/Time
Batteries using nanotechnology nanopatent 03/25/2011 09:43:55 AM
nanopatent - jimbonano 03/25/2011 08:07:25 AM
Fair enough - jimbonano 03/25/2011 08:03:15 AM
Axion is not currently involved with nanotechnology - kirktierney 03/25/2011 04:48:09 AM
Axion is not currently involved with nanotechnology - Ecomike 03/24/2011 11:26:27 PM
Axion is not currently involved with nanotechnology - jimbonano 03/24/2011 05:29:29 PM
In case anyone mis-understands me -- I (for kirktierney 03/24/2011 04:59:54 PM
I agree - jimbonano 03/24/2011 09:53:30 AM
I don't see that the issues of the kirktierney 03/22/2011 10:33:33 AM
Batteries charge quickly and retain capacity ... kirktierney 03/20/2011 07:58:37 PM
Batteries charge quickly and retain capacity, thanks to nanopatent 03/20/2011 04:16:19 PM
Will the electrodes lose performance in mass production? kirktierney 03/08/2011 10:50:34 AM
One real good reason for Axion Power to Ecomike 02/28/2011 04:58:56 PM
Funny that you mention BCON, it is up Ecomike 02/28/2011 03:46:38 PM
The Solar Venture nanopatent 02/27/2011 06:07:13 PM
Hi, Ecomike, kirktierney 02/27/2011 01:58:49 PM
Speculation: CNT's are an ultimate additive candidate. But just kirktierney 02/27/2011 09:50:02 AM
Re--The simplest additive is high aspect ratio (read nanopatent 02/27/2011 09:20:57 AM
Catching up on posts re PbC technology. 2 kirktierney 02/26/2011 08:42:07 PM
One of these days I may need to Ecomike 02/26/2011 05:47:48 PM
Author Date/Time
John, Nice to see you posting here!!! Thanks Ecomike 02/26/2011 05:32:28 PM
I am glad to hear another the voice Ecomike 02/26/2011 05:25:26 PM
It's a tough balancing act in the battery John Petersen 02/26/2011 04:39:30 AM
You need to remember that Dr. Schmidt was John Petersen 02/25/2011 02:32:19 PM
JP - now this is what I've been nanopatent 02/24/2011 09:25:45 AM
(WO/2011/019431) POROUS CARBON OXIDE NANOCOMPOSITE ELECTRODES FOR HIGH nanopatent 02/17/2011 09:28:14 AM
Prieto Battery nanopatent 02/16/2011 07:50:56 PM
Revolutionary nano-sheets to boost battery power nanopatent 02/15/2011 06:14:08 PM
Search the USPTO files, perhaps - that's where nanopatent 02/10/2011 09:58:12 AM
Sorry for not be clearer in my request jimbonano 02/10/2011 09:18:15 AM
I did a "nano" search for you here nanopatent 02/08/2011 12:16:04 PM
I would like to read more on the jimbonano 02/08/2011 09:20:53 AM
That improvement is on lead acid not their nanopatent 02/07/2011 03:31:35 PM
Jimbo - as one 'nano' to another - nanopatent 02/07/2011 11:03:23 AM
New nanomaterials unlock new electronic and energy technologies nanopatent 02/03/2011 03:15:59 PM
How does Axion compare to this? Maybe an nanopatent 01/29/2011 02:57:25 PM
The problem with all this is scaling it up from the nano scale to the macro scale with out loosing the cool advantages in the scale up process. It is easy to things fast and efficiently in 2 dimensions (surface effects). When you add depth or volume, then it is another story.
Superb Post !!
The recharge time is very impressive !!
Can you imagine a 5-minute recharging station -
Nano,
That link only works for paid members.
I am on board!!!
Might consider adding fuel cells to the topic? I am finding that fuel cells, in many ways are like batteries (especially primary batteries).
I am studying the shared chemical engineering domains of organic electrochemistry, batteries (primary and secondary) and fuel cells (been buying and plowing through texts books on all three lately, getting real serious again about it), where they are similar and where they differ.
I am a Bio-chemical engineer, and I have been working in and around the plating electrochemistry industry (metal finishing) for 37 years, and relelated industrial water and waste water treatment chemistry.
I was asked last week to consider starting a board for technologies for the smart electric grid. If I did it would include batteries, smart devices, flywheels, solar, inverters, and what ever. If this one works out here for you, I might take the jump on it.
Batteries charge very quickly and retain capacity, thanks to new structure

Paul Braun, professor of materials science and engineering, center, led the research group of graduate student Xindi Yu, left, and postdoctoral researcher Huigang Zhang that developed a three-dimensional nanostructure for battery cathodes that allows for dramatically faster charging and discharging without sacrificing energy storage capacity.
Photo by
L. Brian Stauffer
Paul Braun, professor of materials science and engineering, center, led the research group of graduate student Xindi Yu, left, and postdoctoral researcher Huigang Zhang that developed a three-dimensional nanostructure for battery cathodes that allows for dramatically faster charging and discharging without sacrificing energy storage capacity.
3/21/11 | Liz Ahlberg, Physical Sciences Editor | 217-244-1073; eahlberg@illinois.edu
CHAMPAIGN, Ill. — The batteries in Illinois professor Paul Braun’s lab look like any others, but they pack a surprise inside.
Braun’s group developed a three-dimensional nanostructure for battery cathodes that allows for dramatically faster charging and discharging without sacrificing energy storage capacity. The researchers’ findings will be published in the March 20 advance online edition of the journal Nature Nanotechnology.
Aside from quick-charge consumer electronics, batteries that can store a lot of energy, release it fast and recharge quickly are desirable for electric vehicles, medical devices, lasers and military applications.
“This system that we have gives you capacitor-like power with battery-like energy,” said Braun, a professor of materials science and engineering. “Most capacitors store very little energy. They can release it very fast, but they can’t hold much. Most batteries store a reasonably large amount of energy, but they can’t provide or receive energy rapidly. This does both.”
The performance of typical lithium-ion (Li-ion) or nickel metal hydride (NiMH) rechargeable batteries degrades significantly when they are rapidly charged or discharged.
Making the active material in the battery a thin film allows for very fast charging and discharging, but reduces the capacity to nearly zero because the active material lacks volume to store energy.
Braun’s group wraps a thin film into three-dimensional structure, achieving both high active volume (high capacity) and large current. They have demonstrated battery electrodes that can charge or discharge in a few seconds, 10 to 100 times faster than equivalent bulk electrodes, yet can perform normally in existing devices.
This kind of performance could lead to phones that charge in seconds or laptops that charge in minutes, as well as high-power lasers and defibrillators that don’t need time to power up before or between pulses.
Braun is particularly optimistic for the batteries’ potential in electric vehicles. Battery life and recharging time are major limitations of electric vehicles. Long-distance road trips can be their own form of start-and-stop driving if the battery only lasts for 100 miles and then requires an hour to recharge.
“If you had the ability to charge rapidly, instead of taking hours to charge the vehicle you could potentially have vehicles that would charge in similar times as needed to refuel a car with gasoline,” Braun said. “If you had five-minute charge capability, you would think of this the same way you do an internal combustion engine. You would just pull up to a charging station and fill up.”
All of the processes the group used are also used at large scales in industry so the technique could be scaled up for manufacturing.
The key to the group’s novel 3-D structure is self-assembly. They begin by coating a surface with tiny spheres, packing them tightly together to form a lattice. Trying to create such a uniform lattice by other means is time-consuming and impractical, but the inexpensive spheres settle into place automatically.
Then the researchers fill the space between and around the spheres with metal. The spheres are melted or dissolved, leaving a porous 3-D metal scaffolding, like a sponge. Then a process called electropolishing uniformly etches away the surface of the scaffold to enlarge the pores and make an open framework. Finally, the researchers coat the frame with a thin film of the active material.
The result is a bicontinuous electrode structure with small interconnects, so the lithium ions can move rapidly; a thin-film active material, so the diffusion kinetics are rapid; and a metal framework with good electrical conductivity.
The group demonstrated both NiMH and Li-ion batteries, but the structure is general, so any battery material that can be deposited on the metal frame could be used.
“We like that it’s very universal, so if someone comes up with a better battery chemistry, this concept applies,” said Braun, who is also affiliated with the Materials Research Laboratory and the Beckman Institute for Advanced Science and Technology at Illinois. “This is not linked to one very specific kind of battery, but rather it’s a new paradigm in thinking about a battery in three dimensions for enhancing properties.”
The U.S. Army Research Laboratory and the Department of Energy supported this work. Visiting scholar Huigang Zhang and former graduate student Xindi Yu were co-authors of the paper.
Editor's note: To contact Paul V. Braun, call 217-244-7293; e-mail pbraun@illinois.edu.
The paper, "Three-Dimensional Bicontinuous Ultrafast-Charge and -Discharge Bulk Battery Electrodes." is available online.
[ http://www.nature.com/nnano/journal/vaop/ncurrent/full/nnano.2011.38.htm ]
http://news.illinois.edu/news/11/0321batteries_PaulBraun.html
See also:
http://www.engadget.com/2011/03/25/styrofoam-touches-electrodes-to-create-incredibly-fast-charging/
Energy production and storage developments using nanotechnology.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
