Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
RLBD , CBDY looking good both low float
DXY Breaks support around 92 and major support around $80. Currently DXY is at $94.12. This could allow overall markets to rise to many surprise.
USO break of around $12.50 could spell the beginning of a solid upward trend.
My big plays X AKS WYNN.
Always holding FNMA FMCC for big green.
I run a corporation...Nationally....
What National Corporation?
Slowing down on my posts as I am running my National Corporation...
I do see continued restoration of X and AKS and MT over the long term.
Also like WYNN and GRPN and LOCO.
FNMA FMCC could pop anytime...
Great day for my stocks though the overall market dipped and could not hold the gains. X FNMA FMCC AKS WYNN GRPN RUTH...BUT BIG GREEN FOR ME...LOVE IT.
Look into AVXL as well today another positive result
12 WEEK NEWS UPDATE!!!
Anavex Presents Preclinical Results of ANAVEX 2-73 in Rett Syndrome
- Data presentation at 2016 Epilepsy Pipeline Conference -
- Company exploring potential to advance ANAVEX 2-73 into human clinical studies
in rare disease indication -
New York, NY – February 25, 2016 – Anavex Life Sciences Corp. (“Anavex” or the “Company”) (Nasdaq: AVXL), a clinical-stage biopharmaceutical company developing differentiated therapeutics for the treatment of neurodegenerative diseases including Alzheimer’s Disease, other central nervous system (CNS) diseases, pain and various types of cancer, today announced positive data of ANAVEX 2-73 in an exploratory study in a Rett syndrome model at the 2016 Epilepsy Pipeline Conference being held February 25-26, 2016 in San Francisco, CA.
Chronic oral daily dosing of ANAVEX 2-73 starting at ~5.5 weeks of age was conducted in the MECP2 Rett syndrome disease mouse model and continued through a 12-week behavioral testing time point 60 minutes pre-treatment during the behavioral testing. Behavioral paradigms measure different aspects of muscular coordination, balance, motor learning and muscular strengths, some of the core deficits observed in Rett syndrome. The experiment was sponsored by the Rettsyndrome.org Scout Program and performed by PsychoGenics, Inc., NY.
“The data demonstrates dose related and significant improvements in an array of behavioral and gait paradigms in a mouse model with a MECP2-null mutation that causes neurological symptoms that mimic Rett syndrome. There is a tremendous need for therapeutic solutions for the individuals living with Rett syndrome and their families, and we are very encouraged by the data we have seen with ANAVEX 2-73,” said Steven Kaminsky, PhD, Chief Science Officer of Rettsyndrome.org.
Rett syndrome is rare non-inherited genetic postnatal progressive neurodevelopmental disorder that occurs almost exclusively in girls and leads to severe impairments, affecting nearly every aspect of the child’s life: their ability to speak, walk, eat, and even breathe easily. The hallmark of Rett syndrome is near constant repetitive hand movements while awake. It is characterized by normal early growth and development (6 to 18 months) followed by a slowing of development, loss of purposeful use of the hands, distinctive hand movements, slowed brain and head growth, problems with walking, seizures, and intellectual disability. There is currently no cure for Rett syndrome and treatment of the disorder is symptomatic. Management of the symptoms is done through a multidisciplinary approach utilizing medication for motor difficulties, breathing irregularities, and control of seizures through anticonvulsant drugs. Rett syndrome is caused by mutations in the MECP2 gene and strikes all racial and ethnic groups and occurs worldwide in approximately 1 in every 10,000-15,000 live female births.
Leader of the Rett syndrome Natural History Study, Alan Percy, MD, stated: “Given the strong clinical safety data profile of ANAVEX 2-73, it would be encouraging to explore the compound in patients with Rett syndrome, which is a very vulnerable patient population that in addition to its neurodevelopmental symptoms also experience a significant number of seizures.”
The presentation titled “Assessment of ANAVEX 2-73 in a MECP2 Rett Syndrome Mouse Model” will be presented by Christopher U. Missling, PhD at the 2016 Epilepsy Pipeline Conference in San Francisco, CA at 4:25 p.m. PST February 26, 2016 in the oral session and will be available on the publications page of the Anavex website.
“The positive preclinical signal in Rett syndrome seems to support the upstream mechanism of action of ANAVEX 2-73 with the potential to target not only neurodegenerative diseases such as Alzheimer’s, but also neurodevelopmental rare diseases like Rett syndrome,” said Christopher U. Missling, PhD, President and Chief Executive Officer of Anavex.
About Rettsyndrome.org
Rettsyndrome.org is accelerating research for treatments and a cure for Rett syndrome. As the world’s leading private funder of Rett research, they have funded more than $35 million in peer-reviewed research grants and programs to date. They are a 501(c) 3 organization, earning Charity Navigator’s most prestigious 4-star rating. Rettsyndorme.org empowers families to make a difference. Visit www.rettsyndrome.org to learn more, or call (800) 818-7388 (RETT).
About Anavex Life Sciences Corp.
Anavex Life Sciences Corp. (Nasdaq: AVXL) is a publicly traded biopharmaceutical company dedicated to the development of differentiated therapeutics for the treatment of neurodegenerative diseases including Alzheimer’s disease, other central nervous system (CNS) diseases, pain and various types of cancer. Anavex’s lead drug candidates, ANAVEX 2-73 and ANAVEX PLUS, the combination of ANAVEX 2-73 and donepezil (Aricept®), are currently in a Phase 2a clinical trial for Alzheimer’s disease. The drug combination ANAVEX PLUS produced up to 80% greater reversal of memory loss in Alzheimer’s disease models versus when the drugs were used individually. ANAVEX 2-73 is an orally available drug candidate that targets sigma-1 and muscarinic receptors and successfully completed Phase 1 with a clean safety profile. Preclinical studies demonstrated its potential to halt and/or reverse the course of Alzheimer’s disease. It has also exhibited anticonvulsant, anti-amnesic, neuroprotective and anti-depressant properties in convulsive epileptic animal models, indicating its potential to treat additional CNS disorders, including epilepsy and others. The Michael J. Fox Foundation (MJFF) for Parkinson’s Research has awarded Anavex a research grant to develop ANAVEX 2-73 for the treatment of Parkinson’s disease to fully fund a preclinical study, which could justify moving ANAVEX 2-73 into a Parkinson’s disease clinical trial. ANAVEX 3-71, also targeting sigma-1 and M1 muscarinic receptors, is a promising preclinical drug candidate demonstrating disease modifications against the major Alzheimer’s hallmarks in transgenic (3xTg-AD) mice, including cognitive deficits, amyloid and tau pathologies, and also with beneficial effects on neuroinflammation and mitochondrial dysfunctions. Further information is available at www.anavex.com.
Forward-Looking Statements
Statements in this press release that are not strictly historical in nature are forward-looking statements. These statements are only predictions based on current information and expectations and involve a number of risks and uncertainties. Actual events or results may differ materially from those projected in any of such statements due to various factors, including the risks set forth in the Company’s most recent Annual Report on Form 10-K filed with the SEC. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement and Anavex Life Sciences Corp. undertakes no obligation to revise or update this press release to reflect events or circumstances after the date hereof.
For Further Information:
Anavex Life Sciences Corp.
Research & Business Development
Toll-free: 1-844-689-3939
Email: info@anavex.com
Investors:
Jenene Thomas
Jenene Thomas Communications, LLC
908- 938-1475
Email: JTC@jenenethomascommunications.com
Media:
Jules Abraham
JQA Partners, Inc.
917-885-7378
Email: jabraham@jqapartners.com
©2016 Anavex Life Sciences Corp. All Rights Reserved.
Anavex Life Sciences Corp. | ir@anavex.com | Anavex Life Sciences Corp. - Corporate Offices | 51 West 52nd Street, 7th floor | New York, NY 10019
Email Marketing by iContact - Try It Free!
Manage Your Subscription
Looks like $SCIE is finally ready to go from development into sales finally!
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=120533201
Great day today...big green. Or as Trump would say HUGE!
Monday China begins to trade after Chinese New Year.
Shall be interesting, indeed.
WYNN X AKS GRPN WYNMF LL
I will look into it.
It's a oil stock and Carl Ichan still holds shares They have Concerns over their cash which is overdone.
They also have a credit facility which they can draw on for about $4B.
Carl Icahn didn't aggressively buy into CHK for nothing. I feel Near term it'll be choppy but this stock could soar if they meet their debt obligations next month which I think they will.
If you hold it long enough without trading it, it can more then double. $6-$8 year end is not out of question. Oil is bottoming. If oil goes to $40-$50 then you have a $8-$10 stock
I did sell GoPro today at $11.50's to buy into CHK today in $1.93's today as it has a much higher upside...
I am not familiar with CHK...why do you like it?
GPRO certainly one of my few green stocks today. Coming back a little in power hour.
Uncertainty with presidential elections. But, I see president as not making that many changes to impact Wall Street.
The world is doing find with all the people we have in it...not that many wars and disputes...really. As compared to history.
Uncertainty remains in China and their stock market is closed all week and Hong Kong until Wednesday.
I believe we have an oversold condition today...a lot of fear in selling stocks as they are high risk investments.
I remain in my holdings and did not sell any today.
Will look to reposition if market turmoil continues tomorrow.
Europe will be key watch tonight...along with DXY and oil.
Feds could make a comment to the benefit to ease investor concerns.
CHK is one to buy too
Short squeeze on GPRO!!!
Up 13.66%
http://www.bidnessetc.com/62958-gopro-microsoft-to-collaborate-on-share-file-storage-patents/
Microsoft has explicitly listed that the exFAT file system is one of the technologies that can be licensed; meaning that this file system is most likely covered under the patent deal. The exFAT file system allows saving large files on storage media between 32GB and 256TB.
The latest models of GoPro Hero camera line support SD cards of up to 128GB of storage space. For larger storage media capacity, GoPro’s hardware uses exFAT while for smaller files and storage capacities of under 64GB, it uses the almost obsolete FAT format, which breaks down files into 4GB pieces to fit into the maximum file size available on FAT32 drives.
Glad I am in...with a grin.
Awesome GPRO up up
Got into gpro.
Yes FNMA dos awesome today and I am in AVXL as well and see GPRO is at the bottom too
Wonderful day in stocks today, must admit. Mr. PopnLock surprise visit to many retail investors. Missing out on a run is no fun. But taking some pain for a little gain is sure fun.
DXY is dropping for second day significantly. I like metal sector stocks X AKS MT AA. I like CAT MNTX MTW FNMA FMCC
$RBNWE
AMAZING Share Structure
A/S 200m
O/S 80m
*****Float 30m*****
50m Restricted
COP is a good one
.dxy is down 1.6% to 97.30, which is a good sign for some of my stocks... AKS X MT CAT...
Yep, I was right the V-shaped charts signaled bottom, IMO. FNMA up 12%...looking good.
Quite the battle going on at Power Hour...If we hold, I believe we saw the low at yesterday's V-shaped charts...simply wonderful is this is the bottom. Up from here, OK with me.
I believe the negative propaganda found on such web sites as MarketWatch were paid for by the billionaires of the world to scare retail out of their shares...scare tactics with bears always works...Dr. Doom and Gloom all over the headlines. Many assumptions were fabricated, yet today with the V-recovery could signal the bottom...I saw this same V-recovery at the bottom of AAMRQ and I doubled down, the best move of my career.
I could see we close green if the big guns pop and lock out the scared retail...which could happen. 293 points to go.
Watching CAR HTZ MTG FNMA X AKS MT SUNE LVS BYD HOG...may begin or add...
GSI was quite a surprise loaded around .9 and out just over $4. Loved it, but not the news as they selling the steel portion.
I do see hope for X, AKS, MT...so, I am in with a grin, someday drink some gin at the WYNN.
WYNN solid and news of large buyer of stock rumored.
FNMA FMCC still a long hold for me.
Happy New Years to you also- thanks for your posts
I am liking the steel sector...X AKS MT...
WYNN
FNMA FMCC
Take a look at AVXL great fundamental
http://www.anavex.com/about-us/scientific_advisors/
Abraham Fisher, PhD
A member of the Scientific Advisory Board, Dr. Fisher has nearly 40 years of experience in drug design and discovery. He has taken lead compounds including AF102B (EVOXAC®, cevimeline HCl) from concept to approval. AF102B is the first muscarinic agonist approved for sale in the U.S. (2000) and in Japan (2001) to treat dry mouth in patients with Sjoergen’s Syndrome. Dr. Fisher is also the inventor of AF710B (renamed ANAVEX 3-71). Dr. Fisher is co-founding Chairman and President of the Alzheimer’s and Parkinson’s Diseases Conference,is on the scientific review board of the Alzheimer’s Drug Discovery Foundation and is a reviewer of the Alzheimer’s Association. He is named on 21 patents worldwide, is the author of numerous peer-reviewed papers and academic book chapters and is associate editor of Current Alzheimer Research. He also serves on the editorial board of Neurodegenerative Diseases and was on the editorial boards of CNS Drug Reviews, Japanese Journal of Pharmacology and Drug Development Research. Currently Dr. Fisher is a visiting professor at the Department of Neurobiology at the Weizmann Institute in Israel. Previously he was senior scientist at the Israel Institute for Biological Research (IIBR) and held faculty appointments as adjunct professor in the Departments of Pharmacology and Psychiatry, Southern Illinois University and in the School of Medicine, Department of Molecular and Cellular Pharmacology, University of Miami.
Norman Relkin, MD, PhD
A member of the Scientific Advisory Board, Dr. Relkin is an internationally recognized expert on Alzheimer’s disease and related disorders. An American Board of Psychiatry and Neurology-certified neurologist, he graduated from Yale and earned MD and PhD degrees from New York’s Albert Einstein College of Medicine. Dr. Relkin is an Associate Professor of Neurology at the Weill Cornell Medical College and Founding Director of the Weill Cornell Memory Disorders Program. He has 20+ years of clinical trials experience, serving as principal investigator in over 20 therapeutic studies, including multi-center trials he designed. Over the past decade, Dr. Relkin pioneered the study of naturally occurring human antibodies for the treatment of Alzheimer’s. He led all three clinical trial phases of Intravenous Immunoglobulin as a potential Alzheimer’s treatment, including the pivotal National Institutes of Health (NIH) and Baxter co-sponsored study carried out at 45 sites (US and Canada). The author of numerous publications on neurodegenerative and traumatic disorders of the brain during the past 25 years, Dr. Relkin sits on editorial boards for three scientific journals. Additionally, he has been a reviewer for NIH and funding agencies in Europe and Australia, and has been a long-standing member of the Weill Cornell Institutional Review Board. In 2012, he was elected to the Board of Directors of The American Federation for Aging Research.
Michael Gold, MD
A member of the Scientific Advisory Board, Dr. Gold has over 20 years of experience in the clinical development of Alzheimer’s and other central nervous system (CNS) drugs, and currently serves as Vice President of the CNS practice at UCB, Inc., a global biopharmaceutical company. His background also includes leadership roles with GlaxoSmithKline (GSK), Johnson & Johnson (J&J) and Bristol-Myers Squibb (BMS). At GSK, Dr. Gold was responsible for the late-stage CNS and pain portfolio. He also led several clinical Phase II and Phase III Alzheimer’s disease-related project teams. At J&J, Dr. Gold served as the Compound Development Team Leader for Galantamine, an Alzheimer’s drug, culminating in FDA approval of Galantamine CR (Razadyne® ER). Prior to joining UCB, Dr. Gold served as the Chief Medical Officer of Allon Therapeutics, where he led a large pan-US and European clinical study for a neurodegenerative disease. Before joining the pharmaceutical industry, Dr. Gold was an Assistant Professor at the University of South Florida (USF) College of Medicine Department of Neurology and Director of the USF Memory Disorders Clinic. During this time, he was the Principal Investigator for several clinical trials for Alzheimer’s disease. He has also authored multiple publications related to Alzheimer’s and dementia. Dr. Gold also holds an academic appointment as an adjunct assistant professor in the Department of Medicine at the University of North Carolina at Chapel Hill.
John Harrison, PhD
A member of the Scientific Advisory Board, Dr. Harrison is an internationally acknowledged specialist for design of human clinical outcome measurement in Alzheimer’s disease and other cognitive impairments. Dr. Harrison has successfully integrated cognitive testing into drug development programs for many pharmaceutical and biotechnology companies including eight of the current ‘Fortune’ top 10 pharmaceutical companies. He is Honorary Senior Lecturer in the Department of Medicine at Imperial College in London, focusing on investigating cognitive change that may show disease progression in Alzheimer’s and other related indications. Dr. Harrison is a member of the American Psychological Association, holds Chartered Psychologist status with the British Psychological Society and Chartered Scientist status with the UK Science Council. He has authored/co-authored more than 60 books and scientific articles and has been invited as a specialist for cognitive tests at many international meetings, including the European Task Force for Alzheimer’s disease and American Alzheimer’s Association roundtable events. Dr. Harrison’s background also includes prior positions as Head of Neuropsychology at CeNeS Pharmaceuticals, Principal Consultant at CPC Pharma Services, and Principal Scientist at CogState Ltd. In addition to his current academic appointment, he is principal consultant at Metis Cognition Ltd and holds a PhD in neuroscience from the University of London.
Ottavio Arancio, MD, PhD
A member of the Scientific Advisory Board, Dr. Arancio is a cellular neurobiologist who has pioneered the field of mechanisms of synaptic dysfunction in Alzheimer’s disease. He is Associate Professor of Pathology and Cell Biology at the Columbia University Medical Center and The Taub Institute for Research on Alzheimer’s Disease and the Aging Brain, Columbia University. Over the last 10 years, Dr. Arancio has raised more than $25 million in grant funding and published more than 100 peer-reviewed scientific papers. Dr Arancio’s honors include the “G. Moruzzi Fellowship” (Georgetown University), the “Anna Villa Rusconi Foundation Prize” (Italy), the “INSERM Poste vert Fellowship” (France), the Edward N. and Della L. Thome Memorial Foundation Award (2010), the Margaret Cahn Research Award (2008), the American Health Assistance Foundation Centennial Award (2007) and the Zenith Award (2007). He also founded Citta Pharmaceuticals, a biotech company for development of small molecules to treat Alzheimer’s disease.
Tangui Nicolas Maurice, Ph.D.
A member of the Scientific Advisory Board, Dr. Maurice has spent 15 years in the field of neurosciences, including behavioral and molecular neuropharmacology, sigma receptors, neuropeptides, neurosteroids, neurotrophic factors, normal/pathological aging models for Alzheimer’s and related disorders, and behavioral phenotyping of rodent models. Dr. Maurice is a researcher at the Institut national de la sante et de la recherche medicale (INSERM) U710 at Montpellier. He has also held research positions at the Centre National de la Recherche Scientifique (CNRS), INSERM U336, the department of neuropsychopharmacology and hospital pharmacy at Meijo University (Nagoya, Japan), and Jouveinal Research Institute (Fresnes, France).
Jeffrey Cummings, MD
A clinical expert on the Anavex Scientific Advisory Board, Dr. Jeffrey Cummings is Professor of Neurotherapeutics and Drug Development in the Neurological Institute, Cleveland Clinic. He is Director of the Cleveland Clinic Lou Ruvo Center for Brain Health, Las Vegas, Nevada and Cleveland, Ohio. Dr. Jeffrey Cummings graduated magna cum laude from the University of Wyoming, Laramie and obtained his MD degree (with thesis) from the University of Washington, Seattle. He completed internship at Hartford Hospital in Hartford, Connecticut and did his Neurology residency at Boston University, Boston, Massachusetts. Dr. Cummings was formerly the Augustus S. Rose Professor of Neurology and Professor of Psychiatry and Biobehavioral Sciences at the David Geffen School of Medicine at UCLA, Los Angeles, California, USA. He was the Director of the Mary S. Easton Center for Alzheimer’s Disease Research at UCLA and Director of the Deane F. Johnson Center for Neurotherapeutics at UCLA. Dr. Cummings has expertise in neuropsychiatric assessment, outcomes in clinical trials, clinical trial design and analysis, and global clinical trials. He is a frequent consultant to industry. Dr. Cummings has authored more than 500 peer-reviewed papers and 30 books on Alzheimer’s disease, neuropsychiatry, and clinical trials. Dr. Cummings’ interests embrace the neuropsychiatry of neurologic disorders, biomarkers for neurodegenerative diseases, and clinical trials and drug development for neurologic diseases.
Paul Aisen, MD
A clinical expert on the Anavex Scientific Advisory Board, Dr. Aisen is a leading clinician and researcher in Alzheimer’s disease clinical trials and is founding director of the University of Southern California (USC) Alzheimer’s Therapeutic Research Institute in San Diego. Previously, he was on the faculty of the University of California, San Diego (UCSD) School of Medicine’s Department of Neurosciences. His primary research interests focus on the development of new strategies for the treatment of Alzheimer’s disease. Since 2007, Dr. Aisen has been Director of the Alzheimer’s Disease Cooperative Study, a consortium funded by the National Institute on Aging (NIA) to develop assessment instruments and conduct clinical trials. Dr. Aisen is Associate Editor of Alzheimer’s Research and Therapy, a major international peer-reviewed journal, and sits on the editorial board of BMC Medicine. He has published more than 180 peer-reviewed papers.
Corinne Lasmézas, DVM, PhD
A member of the Scientific Advisory Board, Professor at The Scripps Research Institute for the past 10 years and frequent TED Speaker, Dr. Lasmézas is an internationally recognized expert in the field of neurodegenerative diseases with a focus now on studying the mechanisms of neurodegeneration. Since her appointment at Scripps in 2005, Dr. Lasmézas has focused on how misfolded proteins lead to neuronal dysfunction and loss in diseases including Alzheimer’s, Parkinson’s and prion diseases. Additionally, Dr. Lasmézas is a reviewer for national and private funding agencies worldwide, including the US National Institutes of Health (NIH) and the UK Medical Research Council, and an advisor for the US Food and Drug Administration (FDA), the US Environmental Protection Agency (EPA) and the US Department of Agriculture (USDA). She has published more than 60 original scientific papers. Earlier in her career, Dr. Lasmézas’ research provided the first experimental evidence that the prion disease “mad cow disease” had been transmitted to humans, causing variant Creutzfeldt-Jakob disease. This fatal disease belongs to the same group of age-related neurodegenerative diseases as Alzheimer and Parkinson’s diseases, caused by aggregates of misfolded proteins. At the peak of the mad cow crisis, Dr. Lasmézas became an advisor to the World Health Organization (WHO) as well as several governmental and public health committees. Dr. Lasmézas holds a PhD in Neurosciences from the University Pierre & Marie Curie in Paris and obtained her Doctorate of Veterinary Medicine and Diploma of Aeronautic and Space Medicine from the University of Toulouse, France.
Jacqueline French, MD, FAAN
A member of the Scientific Advisory Board, professor in the Department of Neurology at New York University (NYU), Co-Director of Epilepsy Research and Clinical Trials at NYU’s Comprehensive Epilepsy Center and Director of the Epilepsy Study Consortium, Dr. French is an award-winning, internationally recognized expert on epilepsy, new therapeutic interventions and clinical trial methodology. She plays an ongoing leadership role in the area of development of new therapeutics for epilepsy, including co-directing a bi-annual symposium on trial design and its implications and holds positions on committees of the American Academy of Neurology (AAN), where she has also co-authored several AAN clinical practice guidelines. Broadly published, including numerous research articles, editorials and chapters, her writings have been featured in respected publications including The New England Journal of Medicine, Neuron, Neuro Image, Epilepsy Currents and Lancet Neurology. She has also edited two books on epilepsy and is an in-demand global speaker on antiepileptic drug therapeutics and related topics. Previously, Dr. French was Assistant Dean for Clinical Trials at the University of Pennsylvania, a recent President of the American Epilepsy Society, Secretary of the American Society of Experimental Neurotherapeutics, and worked with the US Food and Drug Administration (FDA) developing new trial designs for the approval of antiepileptic drugs. Dr. French trained in Neurology at Mount Sinai Hospital (New York), and did her fellowship training in EEG and epilepsy at Mount Sinai Hospital and Yale University. She was the recipient of the Epilepsy Foundation’s 2013 Hero of Epilepsy Award, honoring her longtime contributions to epilepsy research and clinical trials, alongside her significant impact on the epilepsy community.
Harald Hampel, MD, MA, PhD, MSc
A member of the Scientific Advisory Board and an internationally recognized expert on Alzheimer’s and related neurodegenerative diseases, Dr. Hampel is Professor and AXA Research Fund Chair at Sorbonne Universities’ Pierre and Marie Curie University (UPMC) in Paris, the leading university in science, technology and medicine in France. The AXA-UPMC Chair is hosted within the highly specialized Institute for Memory and Alzheimer’s Disease (IM2A), a reference center for Alzheimer’s and other neurodegenerative diseases, and affiliated with a leading neuroscience institution, the Brain and Spine Institute (ICM), as well as the Department of Neurology at the Pitié-Salpétrière University Hospital. He has conducted more than 50 clinical trials over the past 20 years and has developed international programs focused on Alzheimer’s therapy, diagnosis and the development of biological and imaging biomarkers, including his current work as AXA-UPMC Chair, which aims to improve early detection at the preclinical ‘silent stage’ before the onset of any clinical signs and symptoms. After a post-doctoral fellowship focused on structural and functional neuroimaging of the healthy aging and Alzheimer’s disease brain at the NIH/NIA Laboratory of Neurosciences in Bethesda, MD, Dr. Hampel became founding director of the Alzheimer Memorial Center and Professor of Psychiatry at the University of Munich. In 2006, he was appointed as a Professor and Chair of Psychiatry at Trinity College, University of Dublin, Ireland. During this time, he was a leading Principal Investigator at the Trinity College Institute of Neuroscience (TCIN). In 2010, Dr. Hampel was appointed as Professor, Chair and Head of Department of Psychiatry and Co-Director of the Brain Imaging Center (BIC) at the University of Frankfurt. Dr. Hampel has published more than 500 peer-reviewed research papers, 80 book chapters and eight books, many in world-leading scientific journals, such as the New England Journal of Medicine, JAMA, The Lancet, The Lancet Neurology, Nature Genetics, Nature Reviews Drug Discovery and Nature Reviews Neurology. He has won numerous awards for his scientific work on healthy and diseased brains as well as biomarker and therapy discovery in Alzheimer’s. Additionally, Dr. Hampel has been a reviewer for leading international scientific journals and funding agencies in the US, Canada and Europe and is Senior Associate Editor of the leading international Alzheimer’s journal Alzheimer’s & Dementia, the journal of the Alzheimer’s Association.
Adrian Carsini: This is dreadful. Don't you realize that a great wine is like a great work of art? It has to be nurtured, it has to be taken care of. You have subjected this port to a temperature in excess of 150 degrees. Such disdain can not and must not be tolerated!
FNMA FMCC have been mistreated by the government, instead of nurturing them, they have tried to destroy them, though they have been the golden goose for Treasury.
Golden Bolden Holden...
WYNN X ICA SUNE LOCO HOV LL USO COP AKS GRPN
FNMA FMCC Totally manipulated OTC. Restoration will bring tremendous upside...Ackman probably shorting the box and will add where he feels is bottom. Bottom will come, in the mean time we chew gum.
Distressed Stock to watch=RCAP
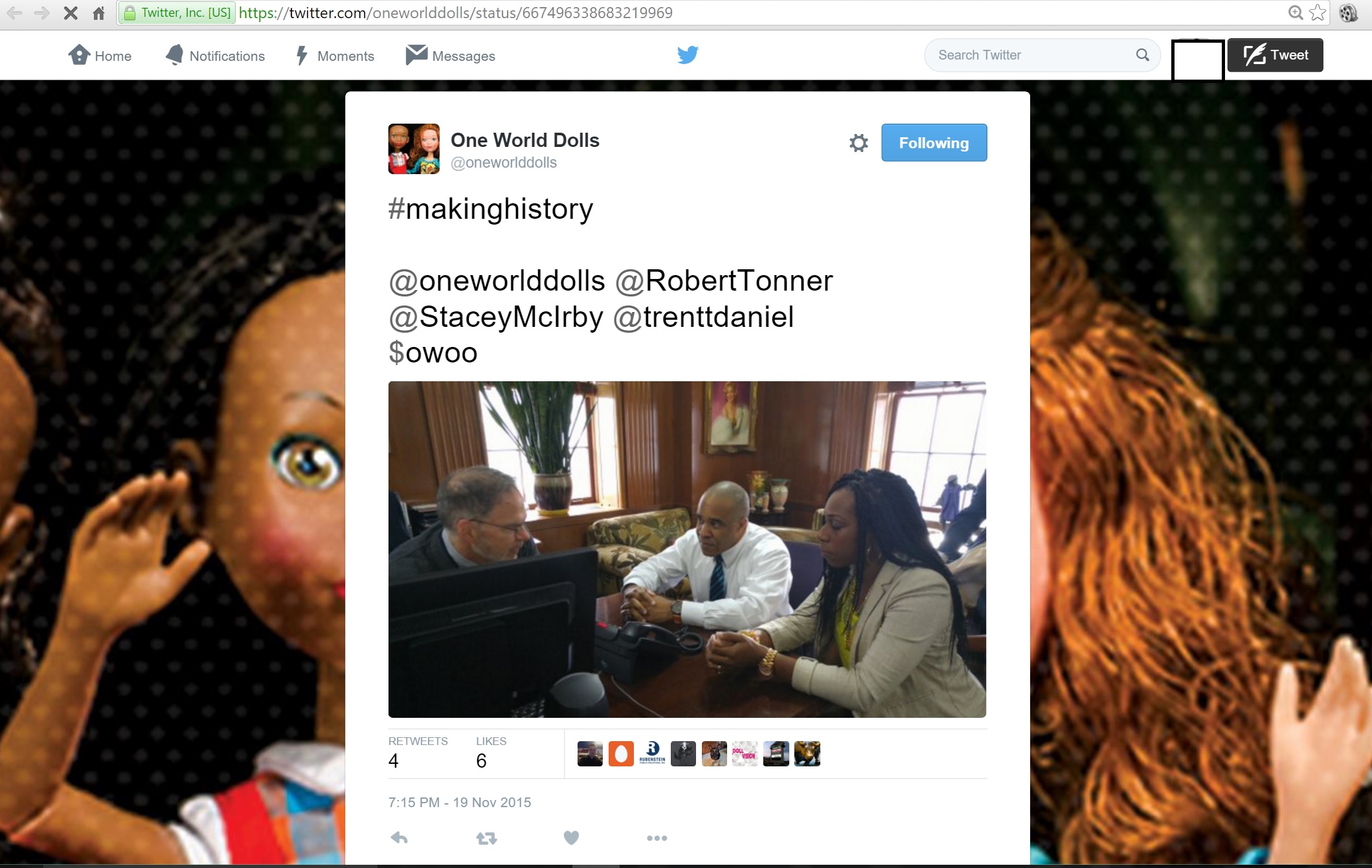
OWOO First this Tweet!!!
Then USA Today Coverage on OWOO ???
Worldwide DOLL MAKER LEGEND ROBERT Tonner as a couple of days ago Tweeted "making history"
#makinghistory@oneworlddolls @RobertTonner @StaceyMcIrby @trenttdaniel$owoo pic.twitter.com/tTrJrRS7rt
— Tonner-One World (@oneworlddolls) November 20, 2015
Hi detearing
wondering if you ares till focusing on distressed stocks or enjoying your AMR winnings?
Have you looked at Magnum Hunter MHR, now MHRC after de-listing, missed initial bounce play but not sure if to expect more or falling back until concrete resolution to debt is announced.
Global Digital Solutions Inc. ( GDSI ) Acquires Privately Held Rontan Group of Brazil
One of the Largest Specialty Vehicles OEM in the World with Leading Market Positions in a Variety of Segments Including Police Cars, Ambulances, Fire Trucks, Heavy Duty Vehicles, Acoustic and Visual Signaling Equipment, Motorcycle Helmets and Telecommunication Systems (Refer to GDSI's October 19, 2015 8-K Filing for Details)
At an upcoming special board meeting, GDSI's board of directors is expected to elect Rontan's CEO, Edigimar A. Maximiliano Jr., Chief Executive Officer of GDSI and Richard J. Sullivan Non-Executive Chairman
Great day yesterday for many of my stocks!
X WYNN AKS MNTX RUTH
FNMA FMCC most upside potential.
Big Green: WYNN X CAT MNTX WTW RAD ICA
FNMA FMCC will within a year...imo.
ANY THOUGHTS TO AUDIT FINANCIAL for a Pink?
NEW IGEX DD what do you think?
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=117350818
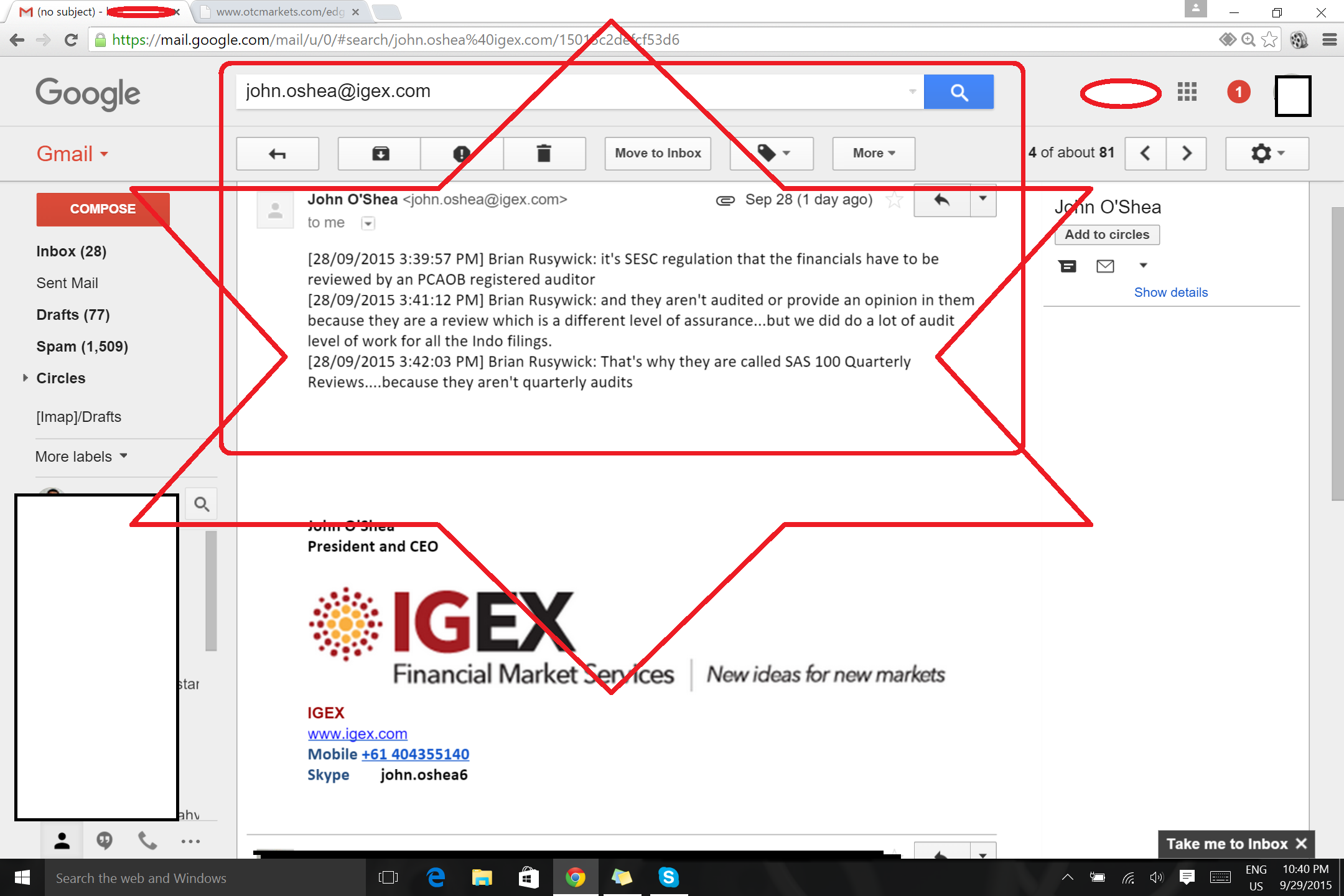
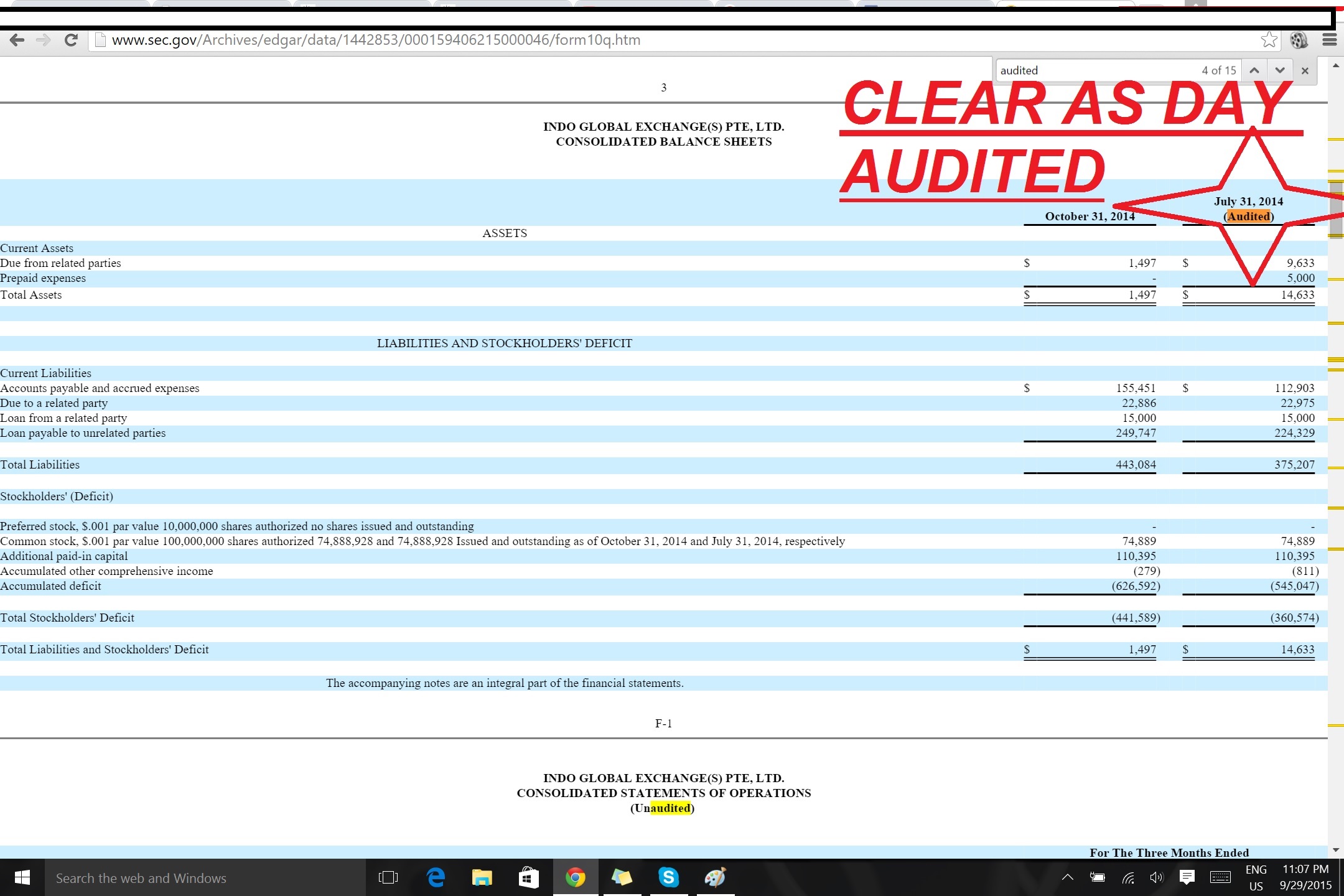
OMG CRAZY DD LOOK IGEX AUDIT FINANCIALS!!!
http://www.sec.gov/Archives/edgar/data/1442853/000159406215000046/form10q.htm
PAGE 3 OF THIS FILING LOOK!!!
X and WYNN falling hard. Added yesterday to both. Long term I see a turn.
HOV GRPN CAT as well. SHLD added near recent lows for swing trade play.
Dollar weakens and this would be fine, so watching that. Oil stocks looking promising...UWTI USO COP BP XOM; STEEL TOO: X AKS MT MTL SID GSI
FNMA FMCC LONG TERM MOST UPSIDE IMO.
MTW MNTX CAT IR DE
yes an agree/ 'Detearing Toastmaster of Winning Distressed Stocks'
Toastmaster
make mine medium /////\\\\\
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
