Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$HEXO HEXO Corp., through its subsidiaries, produces, markets, and sells cannabis in Canada. The company offers its adult-use and medical products under the HEXO brand name. It provides cannabis beverages under the Little Victory, House of Terpenes, Mollo, Veryvell, and XMG brands; and cannabis products under UP Cannabis, Original Stash, and Up brand names. The company was formerly known as The Hydropothecary Corporation and changed its name to HEXO Corp. in August 2018. HEXO Corp. was founded in 2013 and is headquartered in Gatineau, Canada.
$TLRY Tilray, Inc. engages in the research, cultivation, production, marketing, and distribution of medical cannabis products. The company operates through five segments: Cannabis Business, Distribution Business, Beverage Alcohol Business, Wellness Business, and Business Under Development. It provides medical and adult-use cannabis products; pharmaceutical and wellness products; beverage alcohol products; and hemp-based food and other wellness products. The company offers its products to retailers, wholesalers, patients, physicians, pharmacies, clinics, hospitals, governments, and researchers. It has operations in Canada, the United States, Europe, Australia, New Zealand, Latin America, and internationally. The company was formerly known as Aphria Inc. Tilray, Inc. was incorporated in 2018 and is based in New York, New York.
CRON Cronos Group Inc. operates as a cannabinoid company. It manufactures, markets, and distributes hemp-derived supplements and cosmetic products through e-commerce, retail, and hospitality partner channels under the Lord Jones and Happy Dance brands in the United States. The company is also involved in the cultivation, manufacture, and marketing of cannabis and cannabis-derived products for the medical and adult-use markets. It sells cannabis and cannabis products, including dried cannabis, pre-rolls, and cannabis extracts through wholesale and direct-to-client channels under its wellness platform, PEACE NATURALS; and operates under two adult-use brands, COVE and Spinach. The company also exports dried cannabis and cannabis oils to Germany, Israel, and Australia. Cronos Group Inc. was founded in 2012 and is based in Toronto, Canada.
SNDL Sundial Growers has $985.89M in cash
SNDL Sundial Growers Inc. engages in production and marketing of cannabis products for the adult-use market in Canada. It produces and distributes inhalable products, such as flower, pre-rolls, and vapes. The company offers its products under the Top Leaf, Sundial Cannabis, Palmetto, and Grasslands brands. Sundial Growers Inc. was incorporated in 2006 and is headquartered in Calgary, Canada.
SNDL Next week will be epic imo
SUN.ca Wildflower Update
CTO Update
We wanted to provide a brief update on Wildflower's 2020 audit. The audit has been completed.
We advised the British Columbia Securities Commission and they requested the statements and the audit opinion letters be submitted to them for review prior to their filing.
We believe the rationale is to ensure they meet the criteria they established to ensure the exemptive relief application can move forward. The statements were submitted about 2 weeks ago. We have had no response thus far and have requested a time frame for their review but have not heard back.
We will continue to update you as we learn more.
CONTACT INFORMATION:
Krystian Wetulani, Director & CDO
ir@wildflowerbrands.co
1-604-559-0420
HempFusion CBD acquired Sagely Enterprises Inc, trading as Sagely Naturals, a provider of premium CBD products for women.
(TSE: CBD.U) (OTCQX: CBDHF) (FRA:8OO)
CEO Dr. Mitchell says Sagely Naturals generated net revenue of over US$4.19 million in 2020 and will boost HempFusion’s retail distribution by an additional 14,000 plus stores.
https://www.dropbox.com/s/bgd6ix3rj79aa02/Hem...l.pdf?dl=0
HempFusion CBD Quarter 1 Report, Q 2 Coming Up Soon.
HempFusion CBD Reports Q1, 2021 Financial Results
T.CBD.U | May 17, 2021
HempFusion Wellness Inc. (TSX: CBD.U) (OTCQX: CBDHF) (FWB:8OO) (“HempFusion” or the “Company”), an industry leader in full-spectrum cannabidiol (CBD) hemp extract wellness products and probiotics, today reported financial results for the first quarter ended March 31, 2021. All dollars are stated in US Dollars.
Q1-2021 Financial Highlights
Consolidated revenue increased 7.6% to $983,496 from $913,646 in Q4-2020
DTC sales increased 59.3% to $244,255 from $153,305 in Q4-2020
International sales increased to $75,994 from nil in Q4-2020
Gross profit of $280,205 or 28.5% of net revenue
Operating expenses were $6,306,703, an increase of 42.9% from $4,414,792 in Q1-2020, primarily attributed to IPO related expenses as well as marketing and digital related expenses
Strong cash position ending the quarter at $17,074,030
Q1-2021 Business Highlights
Completed a $17 million initial public offering (IPO) and commenced trading on the Toronto Stock Exchange (the “TSX”), becoming the first U.S.-based CBD and wellness products company to list directly on the TSX (senior board).
Became the No. 2 most recognized CBD brand in America according to Brightfield Group
As of the end of January 2021, NielsenIQ data rank HempFusion as the No. 1 in product sales and units sold in one of the largest food/drug/mass (“FDM”) retailers
E-Commerce success has shown a 62% increase in online traffic with a significant increase in conversion including a shift from 16.8% online sales in Q4-2020 as a percent of net revenue to 24.8% in Q1-2021
The Company launched the Probulin store on Amazon and early 2021 traction is encouraging. Consolidated net revenue for the three months ended March 31, 2021 increased 7.6% quarter-over-quarter to $983,496 versus $913,646 in Q4-2020. Direct to consumer (“DTC”) e-commerce net revenue increased 59.3% reflecting increased marketing efforts, a targeted DTC team and targeted digital promotions.
Not only did sales increase for both Probulin and HempFusion but the traffic has increased by 62% in Q1 2021 versus Q4 2020 with a conversion rate increase for Probulin of 2.1% to 3.2% and a current conversion rate for HempFusion of 3.4%. E-commerce accounted for 24.8% of net revenue in the first quarter of 2021 versus 16.8% for Q4, 2020. HempFusion's Probulin has seen 10% or greater month-over-month growth since launching on Amazon .
Officially launched Probulin on the world's largest e-commerce platform, Alibaba's Tmall Global, with HempFusion CBD following in a subsequent Q2 event!
Submitted dossier to the United Kingdom's Regulatory Food Safety Agency
"With nearly all international business paused due to COVID throughout 2020 and Q1 2021, and with slower than expected reopenings, the Company still achieved an incremental QoQ growth of 7.6% . Now, with vaccine rollouts taking place across the country, we are encouraged with reopening rates and excited that our sales, which currently take place in approximately 75% brick-and-mortar businesses, are beginning to reopen," commented Jason Mitchell, N.D., HempFusion's Co-Founder and Chief Executive Officer.
"We remain committed to developing new revenue channels and have entered additional countries including China, Ireland, and the United Kingdom, and we are excited with subsequent quarterly events in which new purchase orders were received, showing early signs of the return of international business. These international POs have the potential to drive millions in revenue. We are working feverishly to accelerate sales through organic channels and the continued pursuit of M&A opportunities; early indications have Q2 shaping up to be our best quarter yet," continued Dr. Mitchell.
Statements of Loss and Comprehensive Loss
The Below is from the Unaudited Condensed Interim Financial Statements for the Three Months Ended March 31, 2021 and 2020; Please Refer to the Complete Report, Including the Notes, Other Statements and Other Contents Therein.
Consolidated net revenue for the three months ended March 31, 2021 increased 7.6% quarter-over-quarter to $983,496 versus $913,646 in Q4-2020, and DTC e-commerce net sales increased 59.3% reflecting increased marketing efforts, a targeted DTC team, and targeted digital promotions. Site traffic increased by 62%, and conversion rates increased to 1.9%. DTC accounted for 24.8% of total revenue in the first quarter of 2021 versus 16.8% for Q4-2020.
International sales increased to $75,994 from nil showing a return to normal from Q4-2020, and early indication of future international orders accelerating and producing incremental quarterly gains. H empFusion maintains a leadership position in retailers across the United States, including the #1 in sales by dollar volume and SKUs in one of the top five largest FDM channel retailers.
Gross profit of $280,205 or 28.5% of net revenue is primarily attributed to improvement in inventory and supply chain management.
Operating expenses were $6,306,703, an increase of 42.9% from $4,414,792 in Q4-2020, primarily attributed to IPO related expenses, including D&O insurance and listing fees, as well as marketing and digital expenses.
Balance Sheet | Cash Position
The Below is from the Unaudited Condensed Interim Financial Statements for the Three Months Ended March 31, 2021 and 2020; Please Refer to the Complete Report, Including the Notes, Other Statements and Other Contents Therein.
The Company ended Q1-2021 with a strong cash position of $17,074,030 ensuring the capital requirements of the Company are met, with ample room for strategic M&A. The Company incurred $2,655,134 in general and administrative expenses and $3,651,569 in sales and marketing expenses.
“In 2019, HempFusion (combined with Probulin on a pro forma basis with respect to 2019) achieved net revenue of $4,627,544, followed by $3,823,638 in 2020. The impact COVID has had on our business is clear. We have adapted. It’s important to note we have strategically bolstered the Company’s online presence with the help of new channels such as Amazon for Probulin and Alibaba’s Tmall for both brands. Our efforts have resulted in early traction and success, as we continue to strengthen our foothold in the direct-to-consumer CBD market. This quarter showed a strong DTC sales increase of 59.3%. We expect this trend to continue as DTC now accounts for 24.8% of total revenue in the first quarter of 2021 versus 16.8% for Q4-2020,” commented Dr. Mitchell.
Conference Call
Management of the Company will host a conference call to discuss the Company's first quarter 2021 results at Monday, May 17, 2021 at 4:15 p.m., Eastern Time. To participate in the call, please dial 888-506-0062. For international callers, dial 973-528-0011. Use Participant Code: 795055. To join the webcast: https://www.webcaster4.com/Webcast/Page/2710/41342 .
A replay will be available from the Investor Relations section of HempFusion’s website at HempFusion.com/corporate-information .
ABOUT HEMPFUSION
HempFusion is a leading health and wellness CBD Company utilizing the power of whole-food hemp nutrition. HempFusion distributes its family of brands, including HempFusion, Probulin Probiotics, Biome Research, and HF Labs, to approximately 4,000 retail locations across all 50 states of the United States and select international locations. Built on a foundation of regulatory compliance and human safety, HempFusion’s diverse product portfolio comprises 48 SKUs including tinctures, proprietary FDA Drug Listed Over-The-Counter (OTC) Topicals, Doctor/Practitioner Lines and more. With a strong focus on research and development, HempFusion has an additional 30 products under development. HempFusion is a board member of the US Hemp Roundtable, and HempFusion’s wholly-owned subsidiary, Probulin Probiotics, is one of the fastest-growing probiotics companies in the United States, according to SPINs reported data. HempFusion’s CBD products are based on a proprietary Whole Food Hemp Complex™ and are available in-store or by visiting HempFusion online at www.hempfusion.com or www.probulin.com .
OXIHF- can't give it away because their filings are behind (but submitted according to management)
CEO owns 80 million shares so I think this is one to hold on to
$CVSI CV Sciences, Inc. is a life science company. The Company operates in two segments: specialty pharmaceuticals and consumer products. The Company's specialty pharma business segment is focused on developing and commercializing therapeutics utilizing synthetic Cannabidiol (CBD) across several therapeutic areas. The consumer product business segment is focused on manufacturing, marketing and selling plant-based CBD products to a range of market sectors.
Company Contact
Headquarters
10070 Barnes Canyon Rd
San Diego, CA 92121-2722
https://cvsciences.com/
agree. look at 6 month chart.
Nice- MJNA looks ready for a substantial run...
$MJNA Medical Marijuana, Inc. became the first company to have its CBD products listed among the healthcare resources provided in the U.S. Physicians’ Desk Reference. Two products from our portfolio – Real Scientific Hemp Oil™ (RSHO™) Gold Label and Kannaway Pure Gold CBD – were added to the 2019 edition, providing healthcare professionals guidelines when discussing or recommending CBD products to their patients.
Not traded in the U.S.? Interesting ...
Tough day today. Was buying $0.50 TLLTF.
Medical Marijuana, Inc. $MJNA subsidiary Kannalife™ Sciences holds the first two licenses awarded to develop and commercialize drug treatments under the U.S. government’s only cannabinoid patent. Kannalife™ Sciences is a phyto-medical company specializing in the research and development of cannabinoid based pharmaceuticals
https://www.medicalmarijuanainc.com/company-of-firsts/the-first-to-license-to-commercialize-the-us-national-institues-of-health-patent/
$MJNA First with a License to Commercialize the U.S. National Institutes of Health Patent “Cannabinoids as Antioxidants and Neuroprotectants.”
https://www.medicalmarijuanainc.com/company-of-firsts/the-first-to-license-to-commercialize-the-us-national-institues-of-health-patent/
$MJNA First to Deliver Prescription CBD Products to Epilepsy, Alzheimer’s Disease, Parkinson’s Disease, Chronic Pain, and Migraine Sufferers
HempMeds® Brasil, a subsidiary of Medical Marijuana, Inc., became the first company to have a cannabis product, Real Scientific Hemp Oil™(RSHO™), prescribed for a number of indications when it was exported to Brazil for all forms of epilepsy, Alzheimer’s disease, Parkinson’s disease, chronic pain and migraines. https://www.medicalmarijuanainc.com/company-of-firsts/the-first-to-deliver-prescription-cbd-products-to-epilepsy-alzheimers-parkinsons-chronic-pain-migraines/
$MJNA The global nutraceutical market size is projected to reach USD $722.49 billion by 2027, expanding at a CAGR of 8.3% over the forecast period. "Medical Marijuana, Inc., an investment holding company, operates in the cannabinoid-based nutraceutical industry."
$MJNA Medical Marijuana, Inc. (MJNA) is a development-stage company, which operates in the cannabis and industrial hemp space in America. Through its subsidiaries and investment holdings, the Company focuses on the development, sale and distribution of hemp oil that contains naturally occurring cannabinoids, including cannabidiol (CBD) and other products containing CBD-rich hemp oil (Legal Hemp). The Company's products are formulated for the pharmaceutical, nutraceutical and cosmeceutical industries, including dietary supplements, prescription-based hemp oil for sale for the treatment of illnesses and conditions, and skin care products. The Company's services include development of cannabinoid based health and wellness products, and the development of medical grade compounds. It operates through subsidiaries, including HempMedsPX, LLC, Wellness Managed Services, LLC, Red Dice Holdings, LLC, CanChew Biotechnologies, LLC, Hempwire, LLC, Kannaway, LLC, HempVap, LLC and HempMeds Brasil.
$MJNA First to Address the United Nations About CBD
In November of 2017, Medical Marijuana, Inc. subsidiary HempMeds® Mexico became the first company to address the United Nations and the World Health Organization (W.H.O.) about cannabidiol (CBD). HempMeds® Mexico President Raul Elizalde was invited to Geneva to speak at the W.H.O Meeting of the Expert Committee on Drug Scheduling, where he urged the committee to recognize CBD as non-psychoactive and to classify it as a nutritional supplement.
https://www.medicalmarijuanainc.com/company-of-firsts/first-address-united-nations-cbd/
$MJNA First with a License to Commercialize the U.S. National Institutes of Health Patent “Cannabinoids as Antioxidants and Neuroprotectants.”
Medical Marijuana, Inc. subsidiary Kannalife™ Sciences holds the first two licenses awarded to develop and commercialize drug treatments under the U.S. government’s only cannabinoid patent. Kannalife™ Sciences is a phyto-medical company specializing in the research and development of cannabinoid based pharmaceuticals
https://www.medicalmarijuanainc.com/company-of-firsts/the-first-to-license-to-commercialize-the-us-national-institues-of-health-patent/
RYAH Group,Medical Cannabis Report On Use for Pain
RYAH Group, Inc Publishes Report on Medical Cannabis Use for Pain
TORONTO, June 10, 2021 (GLOBE NEWSWIRE) -- via InvestorWire -- RYAH Group, Inc. (CSE:RYAH) (“RYAH” or the “Company”) is pleased to announce that its wholly owned subsidiary, RYAH Medtech Inc. ("RYAH Medtech", has published a data report on use of medical cannabis among patients with pain conditions.
The data report features insights into the use of cannabis to treat pain conditions by patients within the RYAH Data ecosystem and whether cannabis may be one of the alternative treatment solutions to reduce opioid use-related problems associated with existing treatment options. The publication is based on user feedback provided by more than 40,000 patients that have reported to the RYAH Data ecosystem.
The report provides a breakdown based on patient gender, main conditions related to pain management and corresponding strain types and reveals that pain is one of the top three conditions for medical cannabis treatment within the RYAH Data ecosystem and also seems to be one of the most interesting symptoms for researchers. As of the date hereof, one of the world’s largest clinical trials in cannabis is focused specifically on pain management, and further to RYAH's recent press release, the RYAH Smart Dry-Herb Inhaler has been selected as the device for such study (with RYAH Medtech recently having completed its initial shipment of RYAH Smart Dry Herb Inhalers and RYAH Cartridges to the U.K. clinic conducting the study).
“The RYAH data report on cannabis use for pain management comes at a turning point as studies on alternatives to opioids in treatment and potential efficacy of chronic pain start to accelerate. The opioid crisis, which began with initially good intentions to more effectively treat pain, created a flood of opioids being prescribed, up from an estimated 70 million prescriptions in the 1990s to more than 255 million by 2012. RYAH is committed to providing the tools needed to effectively monitor and measure dosing in plant-based medicine treatments and to providing data analytics in order for the industry to make more informed decisions around alternative treatments to opioids."
Gregory Wagner, CEO of RYAH
View the Report
Learn more
About RYAH Group, Inc.
RYAH Group, Inc. (“RYAH”) is a connected device and big data and technology company focused on valuable predictive analysis in the global medical plant and nutraceutical intake industry. Its robust artificial intelligence platform aggregates and correlates HIPAA-compliant patient data, which is intended to help doctors and patients personalize plant-based treatments to better predict treatment outcomes. The data collection is relevant for clinics, doctors, dispensaries and pharmaceutical companies and licensed processors (LPs) to monitor and manage formulation effects on patients and demographics. With a strong IP portfolio, RYAH gathers deep and insightful data on the complete patient session and formulation life cycle.
$HEMP Hemp, Inc. focuses on the provision of industrial hemp. The company is involved in processing and farming industrial hemp; extracting hemp CBD oil; and educating and empowering hemp farmers and entrepreneurs with knowledge, processing, infrastructure, and support. It also engages in the sale of hemp accessories, such as extractors, harvesters, storage bags, containers, fertilizer, soil amendments, humidifiers, dehumidifiers, balers, greenhouses, and greenhouse equipment; and drying, trimming, curing, storing, and brokering for other farmers harvesting hemp, as well as provision of research and development, hemp consulting, and educational entertainment services. The company was formerly known as Marijuana, Inc. and changed its name to Hemp, Inc. in June 2012. Hemp, Inc. was founded in 2008 and is based in Las Vegas, Nevada. https://finance.yahoo.com/quote/HEMP?p=HEMP&.tsrc=fin-srch
TILT last quarterly. Solid.
I do not like the chart. TILT.
Lexaria Biotech BREAKTHROUGH NEWS Will Pay Of?
Lexaria’s Newest DehydraTECHTM 2.0 Formulation Tested in Study HYPER-A21-2 Demonstrates Its Strongest CBD Absorption Results Ever
New formulation delivers up to 2,708% more CBD into bloodstream*
Human clinical hypertension study HYPER-H21-1 also progressing
Kelowna, British Columbia, May 20, 2021 – Lexaria Bioscience Corp. (Nasdaq: LEXX) (Nasdaq: LEXXW) (CSE: LXX) (the “Company” or “Lexaria”), a global innovator in drug delivery platforms, is pleased to announce successful test results of two new “DehydraTECH™ 2.0” cannabidiol (“CBD”) formulations in its second 2021 applied research and development study program, HYPER-A21-2.
“One of our latest DehydraTECH 2.0 formulations gave us the strongest absorption enhancement results we’ve ever recorded, at 2,708% more CBD into the bloodstream during the study period than the representative industry standard MCT control formulation. It was also 174% more effective than the original DehydraTECH 2.0 formulation from 2019,” said Chris Bunka, CEO of Lexaria. “This is a 27-fold improvement in CBD delivery compared to the control formulation.”
Study HYPER-A21-2 included three additional new DehydraTECH 2.0 formulation variations designed to enhance CBD delivery performance and pharmacokinetic optimization. Two of the three new DehydraTECH 2.0 formulations delivered improved performance when compared to both Lexaria’s original DehydraTECH 1.0 and 2019 DehydraTECH 2.0 concentration-matched formulations, as well as to a medium chain triglyceride (“MCT”) oil based control formulation representative of standard industry practices. The final formulation provided useful data but did not deliver enhanced performance compared to the original DehydraTECH 2.0. Summary data is shown below:
Lexaria continues to build a robust body of evidence demonstrating that its patented DehydraTECH technology can significantly enhance the delivery of lipophilic active ingredients such as CBD across a range of uptake levels, with ultimate applications to wide ranging areas including consumer packaged goods as well as drugs with potential for disease treatment applications.
In each arm of the study, ten male Sprague-Dawley rats were dosed orally at a level of 25 mg/Kg CBD, and over the next 120 minutes multiple measurements were taken to assess delivery into the bloodstream and tissues comparing the DehydraTECH formulations to certain controls. There were a total of 120 animals for the new DehydraTECH 2.0 formulations evaluated in studies HYPER-A21-1 and HYPER-A21-2. Both studies HYPER-A21-1 and HYPER-A21-2 were conducted by an independent, premier animal testing laboratory located in the United States.
Lexaria also confirms that its HYPER-H21-1 human clinical hypertension study is currently underway and dosing is nearly complete, utilizing a formulation most closely resembling the original 2019 DehydraTECH 2.0. Along with a more than 3-fold increase in dose quantity, these formulation improvements are expected to be more effective than the original DehydraTECH 1.0 formulation used in Lexaria’s foundational 2018 human clinical study that nonetheless evidenced significant blood pressure reduction, as published and available at PubMed. Lexaria thus expects improved pharmacokinetic performance in its current human clinical study to translate into further improved pharmacodynamic performance. Lexaria will provide details on the outcomes of study HYPER-H21-1 when they become available.
* Compared with control formula utilizing medium chain triglycerides (coconut oil) representative of standard industry practices
About Lexaria Bioscience Corp.
Lexaria Bioscience Corp.’s proprietary drug delivery technology, DehydraTECH™, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier oral ingestion methods and increasing the effectiveness of fat-soluble active molecules, thereby lowering overall dosing. The Company’s technology can be applied to many different ingestible product formats, including foods, beverages, oral suspensions, tablets, and capsules. DehydraTECH has repeatedly demonstrated since 2016 with cannabinoids and nicotine the ability to increase bio-absorption by up to 5-10x, reduce time of onset from 1 - 2 hours to minutes, and mask unwanted tastes; and is planned to be further evaluated for orally administered bioactive molecules, including anti-virals, cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs), and nicotine. Lexaria has licensed DehydraTECH to multiple companies including a world-leading tobacco producer for the development of smokeless, oral-based nicotine products and for use in industries that produce cannabinoid beverages, edibles, and oral products. Lexaria operates a licensed in-house research laboratory and holds a robust intellectual property portfolio with 19 patents granted and approximately 60 patents pending worldwide. For more information, please visit www.lexariabioscience.com
Lexarias CBD Trial On Hypertension VERY Promising Tests News.
https://www.barchart.com/story/news/1656715/lexaria-bioscience-corp-discusses-potential-of-dehydratechtm-cbd-for-revolutionary-hypertension-solution-with-the-stock-day-podcast
$SNDL From candy and chocolate to infused beverages, the cannabis edibles market will hit $4.1 billion by 2022 according to a research report from ArcView.
CANN.ca $HERTF Heritage Cannabis Announces the Upcoming Launch of CBD Product Applications Utilizing the Patented VESIsorb® Delivery System Technology in the United States, Including ArthroCBD and Topical CBD Products
by @businesswire on 25 Mar 2021, 04:00
Heritage Cannabis Holdings Corp. (CSE: CANN) (OTCQX: HERTF) (“Heritage” or the “Company”), is pleased to announce that, through a phased approach in 2021, the Company will be launching several new medical products formulated with the patented VESIsorb® delivery system technology in the U.S. through its subsidiary Opticann, including ArthroCBD softgel capsules as well as topical cannabinoid applications that have been formulated to address some of the most pressing consumer health concerns, such as eczema, psoriasis, and local pain.
The first product, launching by April 19, 2021 on eCommerce channels is ArthroCBD, a 25 mg CBD and 6 mg beta-caryophyllene (BCP) softgel capsule formulated with VESIsorb® for optimized absorption, bioavailability, and rapid onset of action. In addition, CBD topical applications formulated with VESIsorb® for optimized dermal penetration and permeation are in the final stages of product development with a planned launch in the summer on opticann.com as well as at leading retailers in the food, drug, and mass channel of distribution. Opticann’s CBD/CBDA sublingual filmstrip which is already planned for launch in Canada and Australia, will also be launched through the eCommerce effort later this year.
To support this effort, Opticann will present its products and brands at the National Association of Chain Drug Stores (“NACDS”) annual meeting in late April, with the expectation to be listed for launch at major U.S. retailers.
“We continue to innovate and expand our clinically proven cannabinoid product offerings as we launch our initial suite of products formulated with the VESIsorb® drug delivery system technology,” commented Umar Syed, Heritage’s President, Medical Division. “With our products, we aim to reach the broader healthcare market that includes sophisticated consumers and healthcare professionals who are seeking highly effective products that have substantial safety and efficacy data to support their utility for medical applications.”
VESIsorb® is a leading, innovative delivery system technology for dramatically improving the stability, absorption, and bioavailability of natural ingredients like cannabinoids, and is backed by a robust body of supporting scientific evidence spanning several decades. Most recently, the peer reviewed journal, Molecules, published the results of a double-blind, cross-over study (Molecules 2019,24(16), 2967) comparing the pharmacokinetic performance of a CBD extract formulated with VESIsorb® to that of the same CBD extract formulated with industry standard (MCT) oil. Overall, the VESIsorb® formulation showed statistically significant improvements for all measured parameters, including much higher total absorption (a 440% increase in maximum plasma CBD concentration (Cmax), and a 285% increase in total CBD exposure over 8 hours [area under the curve (AUC)]) and the time to reach the peak concentration 300% faster than the standard formulation (1 hour for VESIsorb® versus 3 hours for the standard to reach Tmax).
About Heritage Cannabis Holdings Corp.
Heritage is a cannabinoid company focused on the production and sale of medical and recreational hemp-based and cannabis-based products and services. In Canada, Heritage operates through its subsidiaries Voyage Cannabis Corp. and CannaCure Corp., both regulated under the Cannabis Act Regulations. Working under these subsidiaries and the Purefarma, Pura Vida, Premium 5, RAD and feelgood. brands, Heritage focuses on extraction and the creation of derivative products for recreational consumers, as well as the formulation of cannabis based medical solutions. In the U.S., Heritage operates under Opticann Inc., a Colorado based oral and topical cannabinoid company with the rights to exclusively sell CBD and CBG products made with the patented VESIsorb® drug delivery system for optimized absorption and stability. As the parent company, Heritage is focused on providing the resources for its subsidiaries to advance their products or services to compete both domestically and internationally.
ON BEHALF OF THE BOARD OF DIRECTORS OF HERITAGE CANNABIS HOLDINGS CORP.
“Clint Sharples”
Clint Sharples
CEO
The Canadian Securities Exchange does not accept responsibility for the adequacy or accuracy of this release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210325005271/en/
Did it!
Fidelity trading platform
HMJI:CA BETAPRO MARIJUANA COS INVERSE NEW ETF SHS
$10.77 CA
$8.55 US
$19.00 commission Canadian = $15.08 US
accesswire.com/637358/World-Class-Extractions-Reports-Operating-and-Financial-Results-for-the-Nine-Month-Period-Ended-January-31-2021
$PUMP.CN $PUMP $WCEXF
World Class Extractions Reports Operating and Financial Results for the Nine-Month Period Ended January 31, 2021
Thursday, March 25, 2021 3:30 AM
Share this article now
Topic:
Company Update
World-Class continues to show substantial revenue growth quarter-over-quarter with revenue of $2,649,140 for the quarter ended January 31, 2021 compared to revenue of $75,562 for the quarter ended January 31, 2020
VANCOUVER, BC / ACCESSWIRE / March 25, 2021 / World-Class Extractions Inc. (CSE:PUMP)(FRA:WCF)(OTCQB:WCEXF) (the "Company" or "World-Class"), is pleased to report the operating and financial results for its third quarter ended January 31, 2021.
Financial (unaudited)
Revenue of $2,649,140 for the quarter ended January 31, 2021 compared to $2,289,799 for the quarter ended October 31, 2020 and $75,562 revenue for the quarter ended January 31, 2020 - a 2,653% revenue growth from January 2020 to January 2021. The increase in revenue is primarily attributable to the operations of PED.
Gross margin for the quarter ended January 31, 2021 was 27%, consistent with the gross margin figure for the month of January 2021 reported in the Company's news release dated March 2, 2021, but down slightly from the gross margin figures reported for the individual months of November and December 2020.
The Company had cash at January 31, 2021 of $3,017,799 (April 30, 2020 - $5,632,160).
Working capital as at January 31, 2021 is $911,231.
The Company has long-term debt consisting of $819,216 in non-current portion of lease obligations and $92,061 in non-current loans payable.
The net loss and comprehensive loss for the three months ended January 31, 2021 was $626,878 (as compared to January 31, 2020 of $33,144,218). The loss for the three-month quarter ended January 31, 2021 (with comparisons for the quarter ended January 31, 2020) is mainly attributed to the following:
depreciation and amortization expense of $157,600 (2020 - $775,844), consulting fees of $8,500 (2020 - $132,102),; filing fees of $25,592 (2020 - $26,075); development and research expenses of $97,600 (2020 - $830,180); lease interest of $66,151 (2020 - $42,348); office expense of $140,684 (2020 - $156,933); professional fees of $166,328 (2020 - $195,315); management fees of $55,500 (2020 - $57,675), remuneration and benefits of $544,140 (2020 - $640,550); share-based payments of $11,865 (2020 - $241,131) and impairment loss of $Nil (2020 - 29,364,656)
Company Summary and Highlights
Pineapple Express Delivery Inc.
World-Class holds a controlling interest in Pineapple Express Delivery Inc. ("PED"), a company which offers compliant and secure delivery of government regulated products, including medical and recreational cannabis in Ontario, Manitoba and Saskatchewan, and liquor delivery in certain jurisdictions in Saskatchewan.
Since the Company's acquisition of a controlling interest in PED, PED has achieved the following milestones:
A total of 580,732 cannabis and alcohol deliveries for the nine months ended January 31, 2021 were made. The fluctuations in deliveries quarter over quarter are due to the COVID-19 lockdown measures put in place in the Greater Toronto Area with volumes increasing during these lockdowns. Total deliveries per quarters: 209,676 - three months ended January 31, 2021, 181,569 - three months ended October 31, 2020 and 189,487 - three months ended July 31, 2020.
The number of drivers changed from 55 at the beginning of 2020, to approximately 190 by the end of July 31, 2020, 180 by October 31, 2020 and 200 by January 31, 2021. Efficiencies have also been created by routing deliveries through hub expansions and concentrating drivers to tighter geographical areas, leading to less hours used by drivers for the timely delivery of packages.
Offering same day cannabis delivery service in 37 cities (183 postal code regions) and 3-day service in 65 cities (288 postal code regions) in Ontario, PED expanded cannabis delivery services to Ottawa, Barrie, Kingston, London, and the Windsor/Chatham region.
Expanded liquor delivery services to provide same-day and next-day delivery of wine, spirits and beer products from Saskatoon Co-op's Wine-Spirits-Beer retail stores in Saskatoon, Warman and Martensville.
Partnered with Medical Cannabis by Shoppers™ to offer same-day delivery to their medical cannabis patients in cities in and around the Greater Toronto Area (GTA).
Partnered with Spectrum Therapeutics, the medical division of Canopy Growth Corporation to bring same-day delivery to medical cannabis patients in the GTA.
Partnered with CannTrust Inc. to provide deliveries for estoraTM medical cannabis products to CannTrust patients in Ontario, under the name "Trust Delivery - Powered by Pineapple Express".
Several contracts in place with Health Canada license holders for delivery within select provinces in Canada (B2B cannabis delivery services).
Late-stage applicant to Health Canada to obtain a ‘Sale for Medical Purposes' license for its facility located in the GTA. With a population of 6-million people, this sales license gives PED the ability to store inventory for other license holders, allowing PED to leverage its facility in the GTA and offer same day delivery services to the patients of those LPs.
The Company cautions that figures for revenue, gross margin, and number of deliveries generated from the services offered by Pineapple Express Delivery have not been audited beyond April 30, 2020 and any monthly updates are based on calculations prepared by the Company and management of Pineapple Express Delivery. Actual results may differ from those reported in this release once figures have been audited.
Soma Labs
The Company's subsidiary, Soma Labs Scientific Inc. ("Soma"), has focused on production, quality and compliance of systems, technologies, processes and procedures for cannabis and hemp extraction and processing, including the BOSS CO2 Extraction System and the BEAST Ethanol Extraction System. World-Class continues to focus on monetizing its Soma Labs' assets, including the extraction and processing systems, technology and processes to produce cannabis and hemp concentrates and end-products.
Patent Applications and Information
The Company has previously filed, and will continue to file, patent applications directed to its proprietary systems and methods for producing cannabis and hemp extracts. Such systems and methods are believed to be novel and non-obvious, based on the ability of the World-Class Technologies to produce extracts that have substantially greater concentrations of target cannabinoids and possess minimal contaminants. Additional advantages of the World-Class Technologies include the ability to process very large volumes of cannabis and hemp plant material.
The United States Patent and Trademark Office ("USPTO") has awarded the Company the following U.S. Patents:
U.S. Patent No. 10,851,077, issued on December 1, 2020 in relation to the Company's methods for extracting and concentrating cannabinoids and other target compounds from cannabis using ultrasound-enhanced solvent extraction. WCE's patented plant processing technology allows for prompt cannabinoid extraction following harvest, including the root and all other parts of the hemp plant and further utilizes ethanol for extraction, as opposed to the use of toxic hydrocarbon solvents. WCE's patented process additionally uses precisely focused ultrasound to release cannabinoids from the hemp plant that produces CBD yields that are believed to be among the highest in the industry while also minimizing biomass waste and solvent usage.
U.S. Patent No. 10,946,306, issued on March 16, 2021 in relation to the Company's methods for the systematic extraction and concentration of cannabinoids and other target compounds via solvent extraction from large scale harvests of cannabis and hemp crops. World-Class' patent process minimizes energy consumption, conserves the volume of solvent utilized in the extraction process, and maximizes the quantity of cannabinoids sought to be extracted on a large-scale, commercial basis, than other known cannabinoid extraction techniques.
These patents continue to supplement World-Class' goal to achieve the most sustainable, efficient, and economical method of large-scale commercial production of cannabis plant material.
Update regarding Cobra Ventures & HydRx Farms Debenture Investment
During the period ended January 31, 2021, World-Class provided a $2.5M loan to Cobra Ventures Inc. ("Cobra") with respect to a debenture purchase of HydRx Farms Ltd. operating as Scientus Pharma("HydRx"). World-Class subsequently sold its 50% interest in Cobra to the remaining shareholder of Cobra, the sale of which included the receipt of all funds ($2.5 million) previously advanced by World-Class to Cobra. The World-Class loan was paid out in full, and World-Class has no further interest in Cobra and no ongoing role or involvement with HydRx or the HydRx debenture.
About World-Class Extractions Inc.
World-Class is an innovation-driven company with a principle focus on the rapidly evolving cannabis and hemp industries. World-Class offers compliant and secure delivery of government regulated products through its subsidiary Pineapple Express Delivery Inc., including medical and recreational cannabis in Ontario, Manitoba and Saskatchewan, and liquor delivery in certain jurisdictions in Saskatchewan. World-Class continues to investigate opportunities to deploy and manage custom-built extraction centres utilizing its custom systems, technology, and processes to efficiently produce high-quality cannabis and hemp concentrates and end-products.
Contact
World-Class Investor Relations
1-437-266-1968
ir@worldclassextractions.com
https://worldclassextractions.com
Neither the Canadian Securities Exchange nor its Regulation Service Provider (as that term is defined in the policies of the Canadian Securities Exchange) accepts responsibility for the adequacy or accuracy of this press release, which has been prepared by management of the Company.
Cautionary Note Regarding Forward-Looking Statements
All statements in this press release, other than statements of historical fact, are "forward-looking information" with respect to the Company within the meaning of applicable securities laws, including with respect to the deployment of extraction and processing systems for cannabis and hemp and the ability of Pineapple Express Delivery to expand its delivery services and to provide discrete shipment of products. The Company provides forward-looking statements for the purpose of conveying information about current expectations and plans relating to the future and readers are cautioned that such statements may not be appropriate for other purposes. By its nature, this information is subject to inherent risks and uncertainties that may be general or specific and which give rise to the possibility that expectations, forecasts, predictions, projections or conclusions will not prove to be accurate, that assumptions may not be correct and that objectives, strategic goals and priorities will not be achieved. These risks and uncertainties include but are not limited those identified and reported in the Company's public filings under the Company's SEDAR profile at www.sedar.com. Although the Company has attempted to identify important factors that could cause actual actions, events or results to differ materially from those described in forward-looking information, there may be other factors that cause actions, events or results not to be as anticipated, estimated or intended. There can be no assurance that such information will prove to be accurate as actual results and future events could differ materially from those anticipated in such statements. The Company disclaims any intention or obligation to update or revise any forward-looking information, whether as a result of new information, future events or otherwise unless required by law.
CANN.ca $HERTF Heritage Cannabis Announces the Expansion of its US Supply and Distribution Agreement with Geocann for Products Utilizing VESIsorb® Technology for the Canadian Medical and Retail Markets
by @businesswire on 23 Mar 2021, 04:00
Heritage Cannabis Holdings Corp. (CSE: CANN) (OTCQX: HERTF) (“Heritage” or the “Company”), is pleased to announce it has signed an expansion of its US supply and distribution agreement (the “Agreement”) with Geocann LLC (“Geocann”) for a select portfolio of oral and topical cannabinoid products utilizing the patented VESIsorb® delivery system technology for optimized absorption, bioavailability, and therapeutic benefits. The expansion of the Agreement now includes strategic channels in the Canadian marketplace that are currently accessible via the Canadian Cannabis Act as well as a platform for growth as regulatory conditions continue to develop.
Heritage, through its U.S. subsidiary Opticann, signed a supply and distribution agreement in 2020 to exclusively brand and market a select portfolio of VESIsorb® formulated oral and topical CBD and CBG products to major U.S. retailers, including CVS, Walgreens, Rite-Aid, Kroger, Costco and Walmart.
“Obtaining the distribution rights for select VESIsorb® products in Canada adds to our fast-growing suite of retail and medical product offerings across the country,” commented Umar Syed, President of Heritage’s Medical and International business. “With this agreement, we will leverage the significant scientific research, customer targeting, market planning and branding activity underway for large U.S. retailers to market these medical CBD products in Canada. These are highly differentiated products with published data to support their utility for medical applications. Our portfolio of VESIsorb® formulated products is designed to fulfill the unmet needs of sophisticated consumers and healthcare professionals who are demanding science-backed and clinically proven cannabinoid product offerings.”
VESIsorb® is a leading, innovative delivery system technology for dramatically improving the stability absorption, and bioavailability of natural ingredients like cannabinoids, and is backed by a robust body of supporting scientific evidence spanning several decades. Most recently, the peer reviewed journal, Molecules, published the results of a double-blind, cross-over study (Molecules 2019,24(16), 2967; https://doi.org/10.3390/molecules24162967) comparing the pharmacokinetic performance of a CBD extract formulated with VESIsorb® to that of the same CBD extract formulated with industry standard (MCT) oil. Overall, the VESIsorb® formulation showed statistically significant improvements for all measured parameters, including much higher total absorption (a 440% increase in maximum plasma CBD concentration (Cmax), and a 285% increase in total CBD exposure over 8 hours [area under the curve (AUC)]) and the time to reach the peak concentration 300% faster than the standard formulation (1 hour for VESIsorb® versus 3 hours for the standard to reach Tmax).
“Geocann is pleased to expand our agreement with Heritage as their leadership team has a proven track record of pioneering innovative cannabis products with strong brands and a strategic distribution infrastructure,” said Jesse Lopez, CEO and founder of Geocann. “We look forward to supporting their efforts in the U.S. and Canada by building on the supporting scientific evidence of the superiority of VESIsorb® formulated cannabis products with new PK studies for cannabinoids and terpenes, topical penetration and permeation studies, and clinical end-points studies – all of which are currently underway.”
About Heritage Cannabis Holdings Corp.
Heritage is a cannabinoid company focused on the production and sale of medical and recreational hemp-based and cannabis-based products and services. In Canada, Heritage operates through its subsidiaries Voyage Cannabis Corp. and CannaCure Corp., both regulated under the Cannabis Act Regulations. Working under these subsidiaries and the Purefarma, Pura Vida, Premium 5, RAD and feelgood. brands, Heritage focuses on extraction and the creation of derivative products for recreational consumers, as well as the formulation of cannabis based medical solutions. In the U.S., Heritage operates under Opticann Inc., a Colorado based oral and topical cannabinoid company with the rights to exclusively sell CBD and CBG products made with the patented VESIsorb® drug delivery system for optimized absorption and stability. As the parent company, Heritage is focused on providing the resources for its subsidiaries to advance their products or services to compete both domestically and internationally.
About Geocann
Geocann is a global cannabis organization led by experienced leadership with scientific and technical expertise for pioneering new innovations in natural health products and solutions. Its focus is combining clinically-researched cannabinoids with technologically-advanced and patented innovations that provide product differentiation with superior therapeutic performance and unmatched supporting evidence. Geocann has the exclusive global rights to the patented VESIsorb® drug delivery technology for cannabinoids, terpenes, and flavonoid formulations. For more information, please visit www.geocann.com.
ON BEHALF OF THE BOARD OF DIRECTORS OF HERITAGE CANNABIS HOLDINGS CORP.
“Clint Sharples”
Clint Sharples
CEO
The Canadian Securities Exchange does not accept responsibility for the adequacy or accuracy of this release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210323005385/en/
High Tide: Strong Growth Suggests Undervaluation
Mar. 17, 2021
Seeking Alpha
By Main Street Investor
While the Canadian cannabis producer jumped 300% from its initial IPO price in November, I believe the company continues to trade at a discount.
The legal cannabis market is gigantic and growing quickly; High tide is simultaneously seeing strong revenue growth.
High Tide has a chance to multiply many times from current levels, considering valuations of peers and levels reached in 2018.
Various risks remain, including high competition, low liquidity, and slower than expected adaptation of cannabis.
Overview
*All financial numbers in this article are denominated in Canadian Dollars, unless otherwise specified. USD prefixes to any US$ amounts in the article.
High Tide (OTCQB:HITIF) is a relatively new cannabis producer and distributor that held its IPO on the Canadian stock exchange in November 2020. After Canada announced the legalization of Cannabis in 2018, it became the second large nation that completely allows the possession of Marijuana following Uruguay in 2013. As a result, marijuana producers saw shares surge by hundreds and even thousands of percent in a short period of time.
The legalization served as a benchmark for further legalization in other countries, putting pressure on other democracies around the world. Here, various states in the United States began to legalize pot, including California, New York, New Jersey, and Vermont. The wave of cannabis legalization is swapping across other continents too, as Europe increasingly adopts marijuana for medical use.
The penny stock quickly jumped, now trading 300% higher than when it first hit public markets. While the company is currently under the radar of many cannabis investors due to its low share price and only accessible through the Canadian stock exchange or over-the-counter markets, the company has submitted an application to list on Nasdaq, which would benefit the company offering wider exposure to more investors. The application could be successful, considering that High Tide fulfills most of NASDAQ's listing requirements, yet it might need to do a reverse stock split in order to reach a minimum of $3 per share.
Strong Growth
High Tide reported blockbuster Q4 earnings, as revenue jumped 118% to $24.9 million in the quarter and a staggering 166% to $83.3 million for the entire fiscal year 2020. The company also announced gross margins of 35% for the quarter and 37% for the fiscal year. Geographically, $68.4 million was generated in Canada, while $14.3 million (17%) came from the United States. Furthermore, the company's cash balance increased to $38 million.
Despite the global slump in retail sales associated with the pandemic, and thanks to the tireless efforts of our team, we closed the year with approximately $8 million in Adjusted EBITDA making 2020 the best year in High Tide's history. We continued to run our operations tightly, ending the year off with the record levels of revenue and Adjusted EBITDA. Our integrated value chain which includes Cannabis Bricks & Mortar stores, e-commerce platforms for consumption accessories and hemp-derived CBD products, along with manufacturing and distribution of licensed and proprietary consumption accessories, experienced sizeable growth on all fronts. - CEO Raj Grover
High Tide Revenue
The strong growth is expected to continue well into 2021, as the company expects Q1 revenue of $38 million, representing top-line growth of 355% YOY. For FY 2021, consensus estimates are calling for roughly $186 million in revenue, further growing to $268 million by 2022.
The Cannabis Market is Huge
The legal cannabis market for medical and recreation use is gigantic and growing quickly. In the North American market alone, which is High Tide's main operational market, the cannabis market should reach a staggering $38 billion by 2024 if current trends continue. As of 2019, the continent held the largest global share with 88%, which will drop as other continents are increasingly legalizing recreational cannabis use.
The global legal marijuana market size is expected to reach USD 73.6 billion by 2027, according to a new report published by Grand View Research, Inc. It is anticipated to expand at a CAGR of 18.1% during the forecast period. The increasing legalization of cannabis for medical as well as adult-use is expected to promote growth. - Grand View Research
On a basis type, the medical segment accounts for the largest share of 71% with wide applications to treat severe medical conditions such as Alzheimer's, cancer, arthritis, and Parkinson's disease. Moreover, it can reduce chronic pain, mental disorders and anxieties, a gigantic market opportunity when fully adapted as a medicine.
However, what gets Investors most excited is the opportunity of Cannabis as a recreational drug, similar to Alcohol. This would outstrip the medical segment by a wide margin if legalized. Recent statistics reveal that at least 94.5 million Americans admitted to trying cannabis at least once in their lifetimes, representing 30% of the entire population. If Cannabis was legal that figure would likely be even greater.
Cheap Valuation
High Tide is among the cheapest cannabis stocks on the market, despite rallying over 300% from its lows of just $0.17. Here, High Tide trades at 2.4 times forward EV to Sales, significantly lower than major peers including Aphria (NASDAQ:APHA), Cronos (NASDAQ:CRON), Canopy (NYSE:CGC), and Tilray (NASDAQ:TLRY). Moreover, High Tide is growing the quickest from the list and has comparable gross margins of around 40%. What's striking is that High Tide is trading at just 1.3x projected sales in 2022, whereas peers are trading at around 10 times that figure.
I also want to point out that the Cannabis market is nowhere near valuations reached in 2018. Stocks such as Tilray and Aurora (NYSE:ACB) were trading at above 100 times revenue, though valuations dropped significantly in the following years. Still, there is nothing holding back the market to spike again, as seen in early February when Cannabis stocks jumped by a few hundred percent. Thus, a valuation of at least 20x sales does not seem absurd for High Tide to reach at least for a short period of time. If that happens, High Tide could triple or more from current levels. I thus believe, there is significantly more upside to High Tide than downside. As pointed out before, if High Tide lists on the Nasdaq and the stock become visible to more investors, shares could skyrocket.
Risks
Although I believe High Tide is an attractive investment, various risks remain. The cannabis market is exciting to investors as it offers high growth, with the hope of being widely adopted as a medicine to treat health conditions and disrupting the recreational drug market in North America. While the current situation is developing positively with more states legalizing cannabis, as a result of the presidential election, it is unclear how fast cannabis will be fully adopted across society. For instance, it is argued that when cannabis will be legalized overall consumption will drop significantly as the 'thrill' disappears. Prices of cannabis will also drop significantly, as the value chain is practically eliminated. As a result, overall margins should decrease and competition will increase drastically.
High Tide could drop back to below $0.10 if the company sees slower than expected growth and if its Nasdaq listing application is rejected. Moreover, if retail interest for the stock decreases, shares could plunge additionally. Also, the stock benefitted from an overall bull market in Cannabis stocks, yet in a bear market shares will likely underperform the broader market due to high volatility.
Takeaways
High Tide is an underrated penny stock that benefits from the enormous growth in the North American cannabis industry, reflected in its quarterly earnings. The stock may not stay a penny stock for long: Share surged from just $0.17 to $1 in a matter of months, before dropping back to $0.68. What matters most, however, are extremely attractive fundamentals. The company's sales and are surging, a trend that is unlikely to reverse anytime soon. At the same time, High Tide is trading well below its peers, and another spike in the overall cannabis market similar to 2018 could cause High Tide to multiply many times from current levels. Despite the rosy fundamentals, small risks remain as competition will increase and High Tide's liquidity is still somewhat low, standing at $38 million.
CANN.ca $HERTF Heritage Cannabis Announces Receipt of the First Health Canada Cannabis Export License Granted for Brazil
by @businesswire on 18 Mar 2021, 04:04
Heritage Cannabis Holdings Corp. (CSE: CANN) (OTCQX: HERTF) (“Heritage” or the “Company”), today announced that Health Canada has granted Heritage with the first ever license to export cannabis products from Canada to Brazil.
In December 2020, Heritage entered into an arrangement (the “Arrangement”) with CannaBR Exports Corp. (“CannaBR”), a Brazilian focused pharmaceutical CBD product importation and distribution company, for the distribution and bulk sale of formulated CBD products to the Brazilian cannabis market. The Arrangement calls for minimum order quantities agreed to by both parties, to be delivered to CannaBR’s large distribution network in Brazil with the option to establish long-term distribution contracts.
Heritage, in conjunction with CannaBR, has focused on filing and achieving approvals from the Brazil Health Authority (ANVISA) to distribute and sell various formulated CBD products in Brazil and is currently in advanced stages of product testing, enabling it to meet ANVISA’s requirements.
Through its partnership in Brazil, CannaBR already holds an import license, and with Heritage receiving its export license from Health Canada, shipments of CBD products are set to begin within a week with the intention to satisfy the ANVISA requirements. Upon achieving all necessary testing requirements, the Company will commence final negotiations to complete a long-term sales agreement with minimum order quantities.
“We are very pleased to be the first cannabis company in Canada to be granted an export license for Brazil by Health Canada,” stated, Clint Sharples, CEO of Heritage. “Our work with CannaBR is the second extension of the Company beyond the Canadian and U.S. markets, and with the Brazilian market projected to grow dynamically over the next five to seven years, we are poised to take advantage of the many opportunities in the premium CBD product space.”
About Heritage Cannabis Holdings Corp.
Heritage is a cannabinoid company focused on the production and sale of medical and recreational hemp-based and cannabis-based products and services. In Canada, Heritage operates through its subsidiaries Voyage Cannabis Corp. and CannaCure Corp., both regulated under the Cannabis Act Regulations. Working under these subsidiaries and the Purefarma, Pura Vida, Premium 5, RAD and feelgood. brands, Heritage focuses on extraction and the creation of derivative products for recreational consumers, as well as the formulation of cannabis based medical solutions. In the U.S., Heritage operates under Opticann Inc., a Colorado based oral and topical cannabinoid company with the rights to exclusively sell CBD and CBG products made with the patented VESIsorb® drug delivery system for optimized absorption and stability. As the parent company, Heritage is focused on providing the resources for its subsidiaries to advance their products or services to compete both domestically and internationally.
ON BEHALF OF THE BOARD OF DIRECTORS OF HERITAGE CANNABIS HOLDINGS CORP.
“Clint Sharples”
Clint Sharples
CEO
The Canadian Securities Exchange does not accept responsibility for the adequacy or accuracy of this release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210318005236/en/
TILT HOLDINGS TLLTF
Yes OXIHF is still under the radar- at the moment- It will get noticed imo as Florida hemp industry gets rolling because they are first movers down there. Banking industry is starting to recognize hemp growers and so on-
OXIHF- trading around .004- A Canadian based company growing hemp in Florida- Pioneer Green Canada
CANN.ca $HERTF Heritage Cannabis Announces Manitoba to Now Carry RAD and Newly Introduced feelgood. Brands
by @businesswire on 10 Mar 2021, 04:01
Heritage Cannabis Holdings Corp. (CSE: CANN) (OTCQX: HERTF) (“Heritage” or the “Company”), is pleased to announce that Heritage’s RAD brand and its newest brand – feelgood., a high-quality, affordable health and wellness brand are now available for order in Manitoba. The Company confirms that the feelgood. Extra Strength CBD Oil has shipped and is already on the way to select provincial retailers.
Additional products available for order from feelgood. include CBD Facial Rejuvenation Cream, Extra Strength CBD Muscle Cream, and Extra Strength THC Muscle Cream. These products contain the highest potency of active ingredients available in the Canadian market today. As announced previously, the widely popular RAD brand offers affordable extract and extract derivative products including RAD vapes, RAD shatter, RAD crumble, and RAD isolate.
Heritage also continues to receive recurring orders for products from their Pura Vida, Purefarma and Premium 5 brands that have been available in the Manitoba market since the latter half of 2020, indicating strong support for these brands in the province.
Heritage’s Recreational Division President David Schwede said, “Manitobans will soon have access to two of our newer brands – RAD and feelgood., which both offer consumers high-quality products at affordable prices. With our growing availability of SKUs – now over 80, consumers in Manitoba will have improved choice in the products they purchase and access to a high-potency health and wellness brand that is new to the province. We couldn’t be happier with the early success of RAD and the positive feedback we have on the products. We believe consumers will be equally happy with feelgood. product quality and price.”
Follow Heritage’s Purefarma, Pura Vida, Premium 5, and RAD brands on Instagram and Facebook for more information.
About Heritage Cannabis Holdings Corp.
Heritage is a cannabinoid company focused on the production and sale of medical and recreational hemp-based and cannabis-based products and services. In Canada, Heritage operates through its subsidiaries Voyage Cannabis Corp. and CannaCure Corp., both regulated under the Cannabis Act Regulations. Working under these subsidiaries and the Purefarma, Pura Vida, Premium 5, RAD and feelgood. brands, Heritage focuses on extraction and the creation of derivative products for recreational consumers, as well as the formulation of cannabis based medical solutions. In the U.S., Heritage operates under Opticann Inc., a Colorado based oral and topical cannabinoid company with the rights to exclusively sell CBD and CBG products made with the patented VESIsorb® drug delivery system for optimized absorption and stability. As the parent company, Heritage is focused on providing the resources for its subsidiaries to advance their products or services to compete both domestically and internationally.
ON BEHALF OF THE BOARD OF DIRECTORS OF HERITAGE CANNABIS HOLDINGS CORP.
“Clint Sharples”
Clint Sharples
CEO
The Canadian Securities Exchange does not accept responsibility for the adequacy or accuracy of this release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210310005168/en/
High Tide Opens New Canna Cabana Store in Calgary's Beltline Neighbourhood
BY Canada NewsWire
03/08/2021
CALGARY, AB, March 8, 2021 /CNW/ - High Tide Inc. ("High Tide" or the "Company") (TSXV: HITI) (FRA: 2LY), a retail-focused cannabis corporation enhanced by the manufacturing and distribution of consumption accessories, announced today that the Canna Cabana retail store located at Unit 1245 - 1230 11 Ave. SW, in Calgary's Beltline neighbourhood, has begun selling recreational cannabis products for adult use. This opening represents High Tide's 74th branded retail location across Canada selling recreational cannabis products and consumption accessories.
High Tide Opens New Canna Cabana Store in Calgary's Beltline Neighbourhood (CNW Group/High Tide Inc.)
"While we aggressively pursue opportunities to expand the reach of our ancillary businesses in the United States and Europe, we also remain committed to growing our Canadian retail footprint," said Raj Grover, President and Chief Executive Officer of High Tide. "As we continue to grow our presence in emerging markets, we intend to keep building on our Canadian momentum which is powered by our differentiated one-stop shop model that allows consumers to purchase all of their cannabis and accessory needs under one roof," added Mr. Grover.
Not bad. They are in need of CASH. The O/S number may take a hit.
https://worldclassextractions.com/wp-content/uploads/2020/06/wce-presentation-june-2020.pdf
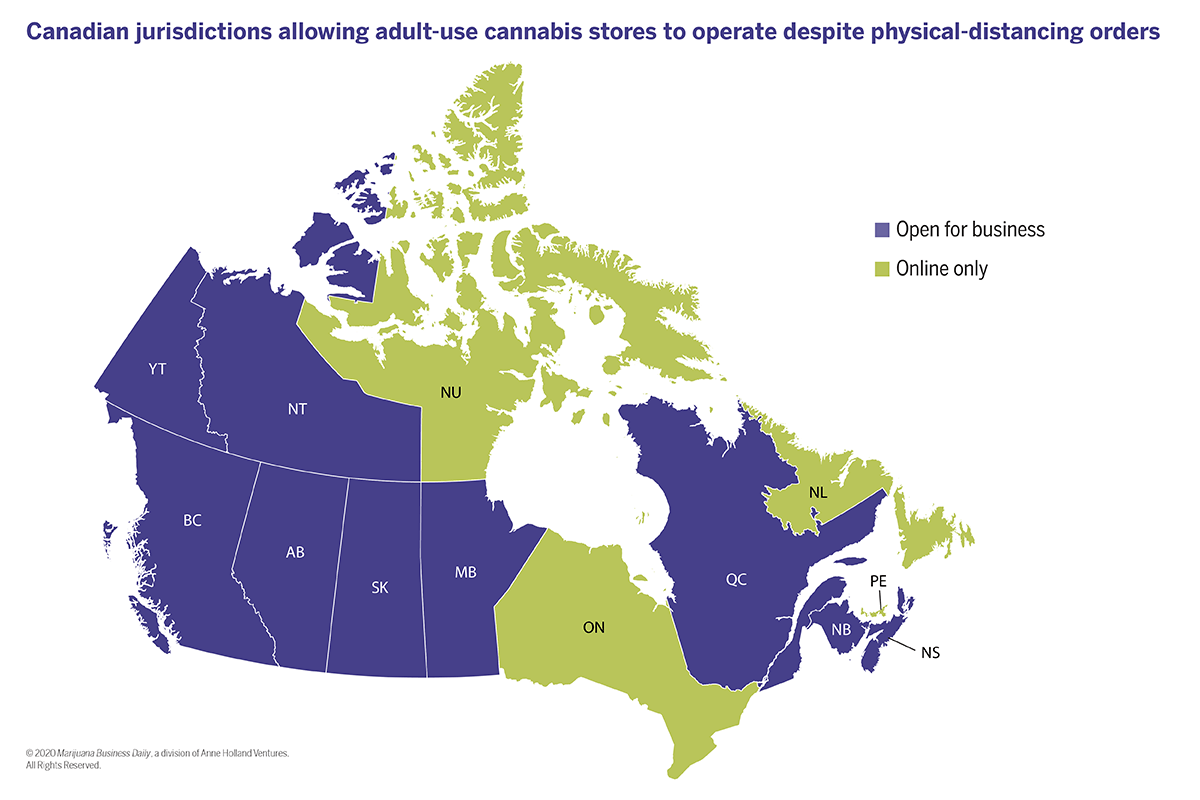


| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
