Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
>>> Coya Therapeutics, Inc. (COYA), a clinical-stage biotechnology company, engages in the development of proprietary medicinal products to modulate the function of regulatory T cells (Tregs). The company's product candidate pipeline is based on therapeutic modalities, such as Treg-enhancing biologics, Treg-derived exosomes, and autologous Treg cell therapy.
It is developing COYA 101, an autologous regulatory T-cell product candidate that has completed Phase 2a clinical trial for use in the treatment of Amyotrophic Lateral Sclerosis. The company's product candidates in IND-enabling studies include COYA 301, a low-dose interleukin 2 Treg-enhancing biologic for use in the treatment of Frontotemporal Dementia; and COYA 302, a biologic combination for subcutaneous and/or intravenous administration intended to enhance Treg function while depleting T effector function and activated macrophages for use in the treatment of neurodegenerative and autoimmune diseases. It is also developing COYA 201, an antigen directed Treg-derived exosome product candidate that is in preclinical stage for use in the treatment of neurodegenerative, autoimmune, and metabolic diseases; and COYA 206, an antigen directed Treg-derived exosome product candidate, which is in discovery stage. The company has a collaboration with Dr. Reddy's Laboratories SA for the development and commercialization of COYA 302, an investigational combination therapy for treatment of amyotrophic lateral sclerosis. The company was incorporated in 2020 and is headquartered in Houston, Texas.
<<<
---
AbbVie, Axsome - >>> 2 Biotech Stocks You Can Buy and Hold for the Next Decade
Motley Fool
January 2, 2024
https://finance.yahoo.com/news/2-biotech-stocks-buy-hold-134500692.html
There are many ways to grow one's wealth over a decade, but few are as accessible as investing in stocks. Even with a relatively modest sum, picking the right investments and letting time and compounding work their magic will generate the kinds of returns most people would be proud to achieve. However, with scores of options to choose from on equity markets, it can sometimes be challenging to know which stocks are worth investing in.
Let's consider two biotech stocks that could generate steady returns in the next 10 years: AbbVie (NYSE: ABBV) and Axsome Therapeutics (NASDAQ: AXSM).
1. AbbVie
AbbVie has been a terrific investment over the past decade. Revenue and earnings have generally grown at a good clip, and it has raised its dividend by 288%. Now that its blockbuster medicine Humira has seen its patent expire in the U.S., things are more complicated -- but that's no reason to give up on the company.
As Humira's sales drop, those of Skyrizi and Rinvoq, a pair of immunology medicines whose indications substantially overlap with those of Humira, are rising. Management expects this pair of drugs to exceed Humira's annual sales by 2027.
AbbVie is looking for growth elsewhere, too. It recently announced the acquisition of Cerevel Therapeutics, a neuroscience-focused biotech, for $8.7 billion in cash. The transaction will improve AbbVie's pipeline in that particular therapeutic area.
Back in 2020, AbbVie closed its acquisition of Allergan, which gave it access to key products, including its Botox franchise. There is nothing wrong with drugmakers relying on acquiring smaller companies to improve their lineups or pipelines, and that's what AbbVie is doing again.
The company does have an extensive pipeline of its own, with several dozen ongoing programs, many of which should result in brand-new approvals or label expansions. For instance, it launched a brand-new cancer medicine this year called Epkinly. So, despite its poor performance in the market this year, AbbVie isn't dead in the water -- far from it.
Patient investors can still expect the company to deliver solid returns and consistent dividend increases. AbbVie is part of the elite group of Dividend Kings: It has raised its payouts for 52 consecutive years, an impressive feat. The company's current yield is 4% -- much higher than the S&P 500's average of 1.62%.
With an underlying business that can still support dividend growth and a reasonable cash payout ratio of 42%, AbbVie can afford many more dividend hikes. That's another great reason to stick with this stock in the next decade.
2. Axsome Therapeutics
Axsome Therapeutics is a mid-cap biotech with just two products on the market. The company's recent financial results aren't great. In the third quarter, it posted revenue of just about $58 million, which soared by 243% year over year. Its net loss per share of $1.32 was worse than the net loss per share of $1.07 recorded in the prior-year period.
However, things could soon change for Axsome Therapeutics given its pipeline. The company has a string of potential approvals, regulatory submissions, and late-stage clinical trials due to start or release top-line data in the next 12 months. For instance, one of its products, depression treatment Auvelity, is seeking a label expansion in treating Alzheimer's disease agitation (aggressive and restless symptoms). The biotech is running a phase 3 clinical trial whose data should be revealed next year.
Auvelity is also being developed to help with smoking cessation, with a phase 2/3 study planned for next year. Axsome Therapeutics' other approved product, narcolepsy treatment Sunosi, is undergoing a phase 3 study targeting ADHD, with top-line results due in the second half of 2024. There is much more to the biotech's pipeline.
Axsome's lineup should look very different in the next three years. Considering that even its oldest approved product, Sunosi, has been on the market only since 2019, the company should be able to deliver solid top-line growth through the next decade.
That could translate into superior stock market returns, too -- especially since, with the company's market capitalization of just $3.5 billion, investors don't appear to fully appreciate the potential of Axsome's late-stage pipeline. That makes it an attractive biotech stock to buy now and hold.
<<<
---
>>> 11 Best Psychedelic Stocks to Buy in 2024
Insider Monkey
Sheryar Siddiq
December 29, 2023
https://finance.yahoo.com/news/11-best-psychedelic-stocks-buy-134746256.html
In this article, we discuss 11 of the best psychedelic stocks to buy in 2024. If you want to see more stocks in this selection, check out 5 Best Psychedelic Stocks to Buy.
Unlike substances like marijuana or cocaine, psychedelics are potent and serve as a form of gateway to an 'alternate reality' for those who use them. Psychedelics represent a category of hallucinogenic drugs capable of inducing non-ordinary states of consciousness. This class encompasses various chemical substances, including LSD and plant-derived compounds. Psychedelics have the potential to alter or intensify sensory perceptions, thought processes, and energy levels, often facilitating spiritual experiences. They are classified into two main groups: empathogens and dissociative drugs (e.g., PCP) and serotonergic substances (e.g., LSD). Beyond recreational use, these drugs are being explored for therapeutic purposes, showing promise in treating conditions such as major depressive disorders, treatment-resistant depression, panic disorder, post-traumatic stress disorder, and opiate addiction, among others.
Venture-capital investors have played a pivotal role in the resurgence of interest in psychedelics. In early 2020, startups in this field noticed a surge in investor appetite, marking the onset of a "psychedelic renaissance." Clara Burtenshaw, a partner at Neo Kuma Ventures, the largest venture capital fund in Europe focused on psychedelic investments, expressed her enthusiasm for psychedelic healthcare on February 16, 2023, stating:
“Psychedelic healthcare is a very exciting area because it’s really this apex of drugs, clinics, and experimental treatments.”
Numerous pharmaceutical companies specializing in psychedelics are pursuing regulatory pathways for the legal approval of their drugs. Hallucinogens, notably psilocybin, are demonstrating significant potential in addressing conditions like depression and schizophrenia. Some of these pharmaceutical firms are publicly listed, including the likes of Johnson & Johnson (NYSE:JNJ), Merck & Co., Inc. (NYSE:MRK), and AbbVie Inc. (NYSE:ABBV), which have garnered heightened interest from investors. This trend mirrors the trajectory observed with marijuana stocks in recent years, where cannabis, once on the periphery, has evolved into a substantial and expanding industry.
Our Methodology
We selected the following psychedelic stocks based on overall hedge fund sentiment toward each stock. We have assessed the hedge fund sentiment from Insider Monkey’s database of 910 elite hedge funds tracked as of the end of the third quarter of 2023. The list is arranged in ascending order of the number of hedge fund holders in each firm.
11. Cybin Inc. (NYSE:CYBN)
Number of Hedge Fund Holders: 1
Based in Toronto, Canada, Cybin Inc. (NYSE:CYBN) is a biopharmaceutical company with a primary focus on the development of therapeutic solutions using psychedelics. The company is engaged in the creation of medications for major depressive disorders, anxiety, and alcohol use disorders, along with addressing neuroinflammation. Additionally, Cybin Inc. (NYSE:CYBN) has introduced EMBARK, a program centered around psychedelic-assisted psychotherapy.
In late November, Cybin Inc. (NYSE:CYBN) disclosed encouraging results from the Phase 2 trial of CYB003, its exclusive deuterated psilocybin analog designed for treating major depressive disorder ("MDD"). The company reported that CYB003 met the primary efficacy endpoint, demonstrating swift and statistically significant enhancements in depression symptoms following a single dose. Additionally, a second dose conferred clear incremental benefits, leading to remission in 4 out of 5 patients from their depression within 6 weeks.
As of the end of Q3, Steve Cohen's Point72 Asset Management emerged as the primary and sole stakeholder, based on Insider Monkey's third-quarter database.
In addition to Johnson & Johnson (NYSE:JNJ), Merck & Co., Inc. (NYSE:MRK), and AbbVie Inc. (NYSE:ABBV), Cybin Inc. (NYSE:CYBN) ranks as one of the best psychedelic stocks to invest in.
10. Mind Medicine (MindMed) Inc. (NASDAQ:MNMD)
Number of Hedge Fund Holders: 3
Mind Medicine (MindMed) Inc. (NASDAQ:MNMD) is a biotechnology company based in New York that specializes in psychedelic medicine. The company is dedicated to the development of psychoplastogens and therapeutic solutions inspired by psychedelics to tackle mental illnesses and addiction.
Mind Medicine (MindMed) Inc. (NASDAQ:MNMD) recently reported that it is nearing the launch of its LSD-based treatment for anxiety patients. The company anticipates more phase two trial data by the fourth quarter of this year and proof-of-concept data by the end of Q1 next year. In its Q3 financial report released on Thursday, the psychedelic firm disclosed a net loss of $17.9 million for the period ending Sept. 30, up from the $16.5 million loss in the same quarter last year. However, MindMed also stated having $117.7 million in funds, expected to cover ongoing trials and research activities "into 2026, if certain milestones are achieved that unlock additional capital."
In 2023, Mind Medicine (MindMed) Inc. (NASDAQ:MNMD) also expended $43.8 million in operating expenses, including $6.4 million for MM-120 studies, their LSD version for treating generalized anxiety disorder. Results from a 198-patient study of MM-120 are anticipated before year-end, with "safety and efficacy results" expected in Q1 next year. The proof-of-concept study, involving 53 patients, is also projected to reveal results in Q1 2024.
Among the hedge funds being tracked by Insider Monkey, Israel Englander’s Citadel Investment Group is a leading shareholder in Mind Medicine (MindMed) Inc. (NASDAQ:MNMD) with 237,475 shares worth more than $743,297. Overall, 3 hedge funds reported holding stakes in Mind Medicine (MindMed) Inc. (NASDAQ:MNMD) as of the third quarter of 2023.
9. Atai Life Sciences N.V. (NASDAQ:ATAI)
Number of Hedge Fund Holders: 5
Founded in 2018 and headquartered in Berlin, Germany, Atai Life Sciences N.V. (NASDAQ:ATAI) is a clinical-stage biopharmaceutical company specializing in the development of treatments for mental health disorders. These include conditions such as treatment-resistant depression, schizophrenia, opioid use disorder, anxiety disorder, and mild traumatic brain injuries.
In its third quarter 2023 financial results, Atai Life Sciences (NASDAQ:ATAI) reported limited revenue as the company continues to develop its drug portfolio. The net income of $44.2 million was primarily attributed to a $69 million non-cash change in the fair value of other investments related to an accounting method change for its CCOMPASS Pathways plc (NASDAQ:CMPS) investment. Additionally, Atai Life Sciences (NASDAQ:ATAI) recorded an $8.3 million non-cash share-based compensation expense. Despite this, the company maintains a robust cash position of $209 million, anticipating that it, along with committed term loan funding, will be sufficient to sustain operations into the first half of 2026.
According to Insider Monkey’s third quarter database, 5 hedge funds were bullish on Atai Life Sciences N.V. (NASDAQ:ATAI), compared to 7 funds in the earlier quarter. John Overdeck And David Siegel’s Two Sigma Advisors is the largest stakeholder of the company, with 107,500 million shares worth $138,675.
8. Seelos Therapeutics, Inc. (NASDAQ:SEEL)
Number of Hedge Fund Holders: 5
Seelos Therapeutics, Inc. (NASDAQ:SEEL) is a clinical-stage biopharmaceutical company headquartered in New York. The company is dedicated to advancing the development and commercialization of therapeutics targeting central nervous system, respiratory, and other disorders.
Seelos Therapeutics, Inc. (NASDAQ:SEEL) has two programs currently undergoing clinical testing. In September 2023, the company disclosed outcomes from a phase 2 study evaluating the intranasal ketamine drug SLS-002 for the treatment of acute suicidal ideation and behavior in patients with major depressive disorder. Additionally, SLS-005 (intravenous trehalose, a sugar containing two glucose molecules) is in phase 2b/3 trials for addressing the neurological disease amyotrophic lateral sclerosis (ALS) and is in phase 2 testing for managing the neurodegenerative disorder spinocerebellar ataxia type 3.
According to Insider Monkey’s third quarter database, 5 hedge funds were bullish on Seelos Therapeutics, Inc. (NASDAQ:SEEL), compared to 3 funds in the earlier quarter.
7. GH Research PLC (NASDAQ:GHRS)
Number of Hedge Fund Holders: 10
GH Research PLC (NASDAQ:GHRS) is a clinical-stage biopharmaceutical company that was established in 2018 and is headquartered in Dublin, Ireland. The company is focused on the development of therapies for the treatment of psychiatric and neurological disorders.
According to Insider Monkey’s third quarter database, 10 hedge funds were long GH Research PLC (NASDAQ:GHRS), compared to 13 funds in the earlier quarter. Mark Lampert’s Biotechnology Value Fund / BVF Inc is the biggest position holder in the company, with 9.27 million shares worth approximately $93.2 million.
Much like Johnson & Johnson (NYSE:JNJ), Merck & Co., Inc. (NYSE:MRK), and AbbVie Inc. (NYSE:ABBV), GH Research PLC (NASDAQ:GHRS) is one of the best psychedelic stocks that investors should pay attention to.
6. Relmada Therapeutics, Inc. (NASDAQ:RLMD)
Number of Hedge Fund Holders: 13
Relmada Therapeutics, Inc. (NASDAQ:RLMD) is a biotechnology company based in Florida, specializing in the clinical-stage development of diverse products for the treatment of central nervous system diseases and other disorders. Analysts consider it one of the promising psychedelic stocks in the market.
In September of this year, Relmada Therapeutics, Inc. (NASDAQ:RLMD) announced encouraging safety and efficacy outcomes from the extended Phase III REL-1017-310 trial, which assessed REL-1017 in individuals diagnosed with major depressive disorder (MDD). Over the course of up to one year, patients receiving daily doses of REL-1017 demonstrated enduring enhancements in depressive symptoms and related functional challenges. The extended administration of REL-1017 exhibited favorable tolerability in patients, featuring low rates of adverse events and the absence of any newly identified safety concerns.
According to Insider Monkey’s third quarter database, 13 hedge funds were long Relmada Therapeutics, Inc. (NASDAQ:RLMD), compared to 11 funds in the prior quarter. David Kroin’s Deep Track Capital held the biggest position in the company.
5. COMPASS Pathways plc (NASDAQ:CMPS)
Number of Hedge Fund Holders: 21
COMPASS Pathways plc (NASDAQ:CMPS) is a mental healthcare company based in the United Kingdom. The company is actively engaged in the development of psilocybin therapy, progressing through late-stage clinical trials in both Europe and North America. The focus is on patients experiencing treatment-resistant depression (TRD). COMPASS Pathways plc (NASDAQ:CMPS) has formulated COMP360, a psilocybin formulation incorporating pharmaceutical-grade polymorphic crystalline psilocybin. A Phase IIb clinical trial for psilocybin therapy in TRD has been completed across 22 sites in Europe and North America, evaluating the safety and efficacy of COMP360 in three doses: 1mg, 10mg, and 25mg.
During 2023’s September quarter, 21 out of the 910 hedge funds profiled by Insider Monkey had held a stake in the company. COMPASS Pathways plc (NASDAQ:CMPS)’s biggest hedge fund investor is Catherine D. Wood’s ARK Investment Management due to its $18 million investment.
4. Intra-Cellular Therapies, Inc. (NASDAQ:ITCI)
Number of Hedge Fund Holders: 37
Intra-Cellular Therapies, Inc. (NASDAQ:ITCI) is a biopharmaceutical company specializing in the discovery, development, and commercialization of small molecule drugs targeting unmet medical needs, particularly in neuropsychiatric and neurological disorders. Caplyta, the sole approved drug in Intra-Cellular Therapies, Inc. (NASDAQ:ITCI)’s portfolio, received FDA approval in December 2019 for the treatment of schizophrenia in adults. Subsequently, in December 2021, it gained FDA approval for treating bipolar depression. Following these approvals, Caplyta’s sales have experienced substantial growth, and the company anticipates maintaining this positive momentum.
In the third quarter of 2023, Intra-Cellular Therapies, Inc. (NASDAQ:ITCI) reported a loss of $0.25 per share. The incurred loss was narrower than the $0.57 per share loss in the same quarter of the previous year, attributed to increased product sales. The total revenues, which include product sales and grant revenues, reached $126.2 million, marking a significant increase from $71.9 million in the corresponding period of the prior year.
By the end of this year’s third quarter, 37 out of the 910 hedge funds part of Insider Monkey’s research had invested in the company. Intra-Cellular Therapies, Inc. (NASDAQ:ITCI)’s biggest hedge fund investor is David Kroin’s Deep Track Capital through its $135 million stake.
3. AbbVie Inc. (NYSE:ABBV)
Number of Hedge Fund Holders: 73
AbbVie Inc. (NYSE:ABBV) operates as a specialized biopharmaceutical company dedicated to researching, developing, manufacturing, and distributing medications tailored for chronic and intricate illnesses. The company is renowned for its flagship drug, Humira, a crucial treatment for conditions like moderate-to-severe rheumatoid arthritis and Crohn’s disease. In May 2020, AbbVie Inc. (NYSE:ABBV) entered the psychedelic drugs market by acquiring a stake in Allergan PLC.
With an impressive 50-year streak of continuous dividend growth, the American pharmaceutical giant currently boasts a dividend yield of 4.48% as of December 13.
As of the close of the third quarter in 2023, Insider Monkey’s database, monitoring 910 hedge funds, indicated 73 holdings in AbbVie Inc. (NYSE:ABBV), marking a slight decrease from the 74 hedge funds in the previous quarter. The collective value of these holdings surpasses $3.27 billion.
2. Merck & Co., Inc. (NYSE:MRK)
Number of Hedge Fund Holders: 85
Merck & Co., Inc. (NYSE:MRK) is a distinguished American multinational pharmaceutical company with its headquarters in Rahway, New Jersey. Originally part of the Merck Group established in Germany in 1668, it retains the name of its former parent company. Operating as Merck Sharp & Dohme or MSD outside the United States and Canada, the company holds a prominent position in the pharmaceutical industry, specializing in the development of medicines, vaccines, biologic therapies, and animal health products.
Showcasing a consistent pattern of dividend growth for 11 consecutive years, the company currently provides a quarterly dividend of $0.77 per share, resulting in a dividend yield of 2.91% as of December 13.
Insider Monkey delved into the investment activities of 910 hedge funds in the third quarter of 2023, revealing that 85 had invested in Merck & Co., Inc. (NYSE:MRK), marking an increase from 78 in the previous quarter.
1. Johnson & Johnson (NYSE:JNJ)
Number of Hedge Fund Holders: 88
Established in 1886, Johnson & Johnson (NYSE:JNJ) is a prominent American multinational corporation known for its groundbreaking contributions to medical devices, pharmaceuticals, and consumer packaged goods. The company’s pharmaceutical division has consistently demonstrated impressive dividend growth for over 62 years. As of November 19, Johnson & Johnson (NYSE:JNJ) offers a quarterly dividend of $1.19 per share, reflecting a dividend yield of 3.18%.
On October 17, Johnson & Johnson (NYSE:JNJ) reported adjusted earnings and revenue that exceeded Wall Street expectations, prompting an upward revision of its full-year guidance. The robust sales performance in both pharmaceuticals and medical devices contributed to the positive results. The company posted a net income of $4.31 billion, equivalent to $1.69 per share, maintaining consistency with the previous year’s net income of $4.31 billion, or $1.62 per share, for the same period.
In the third quarter of 2023, the number of hedge funds tracked by Insider Monkey with holdings in Johnson & Johnson (NYSE:JNJ) decreased to 84, down from 88 in the previous quarter. The collective investments by these hedge funds surpass a total value of $4.15 billion. Bridgewater Associates, led by Ray Dalio, stands out as a prominent hedge fund investor in Johnson & Johnson (NYSE:JNJ) with a substantial stake valued at approximately $424.3 million.
>>>
---
>>> A potent antibiotic has emerged in the battle against deadly, drug-resistant superbugs
LA Times
by Corinne Purtill
January 3, 2024
https://www.yahoo.com/news/potent-antibiotic-emerged-battle-against-160058569.html
Under a microscope, this drug-resistant superbug looks as benign as a handful of pebbles. Yet carbapenem-resistant Acinetobacter baumannii, or CRAB, is a nightmare for hospitals worldwide, as it kills roughly half of all patients who acquire it.
Identified as a top-priority pathogen by both the World Health Organization and the U.S. Centers for Disease Control and Prevention, CRAB is the most common form of a group of bacteria that are resistant to nearly all available antibiotics. Victims are typically hospitalized patients who are already sick with blood infections or pneumonia. In the U.S. alone, the bug sickens thousands and kills hundreds every year.
But 2024 is starting with some encouraging news on the global health front: For the first time in half a century, researchers have identified a new antibiotic that appears to effectively kill A. baumannii.
The compound, zosurabalpin, attacks bacteria from a novel angle, disrupting the route that a key toxin takes on its journey from inside the bacterial cell to the outer membrane that shields the bug from the immune system’s defensive onslaughts.
No other antibiotic approved by the U.S. Food and Drug Administration takes this approach, and the element of surprise is an important advantage against even microscopic foes. A. baumannii has had no opportunity to develop resistance against the drug, which means that, for at least a little while, zosurabalpin could ward off severe illness and death.
“As far as I can tell, the scientific approach is brilliant,” said Dr. Oladele A. Ogunseitan, a professor of population health and disease prevention at UC Irvine who was not involved with the study.
The drug was developed jointly by scientists at the Swiss pharmaceutical company Roche and at Harvard University. Their findings were published Wednesday in the journal Nature.
Carbapenem-resistant A. baumannii is a type of gram-negative bacteria, a vexing category of superbugs. Encased in both an inner and outer membrane that antibiotics struggle to cross, gram-negative bacteria are resistant to most currently available therapies. They are also astonishingly canny for unicellular organisms, with the ability to rapidly develop new defenses against antibiotics and then pass them along to other bacteria through genetic material.
Antibiotic-resistant superbugs claim the lives of more than 1 million people globally each year. The rise of drug resistance is due in part to human folly — we have long over-prescribed and misused antibiotics — but it is also because bacteria are continually finding ways to evade threats. Over the last 50 years, these pathogens have evolved defenses faster than we can produce new drugs.
In their search for a new weapon, the Roche and Harvard scientists turned their attention to a group of compounds called tethered macrocyclic peptides. After testing a library of 45,000 MCPs, the researchers came across one that seemed especially lethal against A. baumannii. After some chemical tinkering, that compound became zosurabalpin.
“This is a very promising advance,” said Paul J. Hergenrother, a chemistry professor at the University of Illinois who was not involved in the research but wrote of the findings for Nature. “Zosurabalpin kills bacteria in a way that is different from all other approved antibiotics.”
The drug kicks into gear only in the presence of lipopolysaccharide, a bacterial toxin. LPS is made inside the bacterial cell and is carried by a dedicated transport system to the bug’s outer defenses.
“The bacterial outer membrane is important for bacteria because it helps them to live in harsh conditions and to survive attacks by our immune system,” said Kenneth Bradley, Roche’s global chief of discovery for infectious diseases.
Zosurabalpin essentially cuts off the LPS transport route. Without a way to get to the outer membrane of the cell, where it can get to work fighting off drugs and immune attacks, the toxin builds up inside the bacteria and eventually kills the cell.
In mice studies, the drug effectively killed off CRAB infections in the blood, lungs and thighs, a selection that mirrors the ways the bug infects humans.
It’s currently in Phase I trials in humans, where researchers are looking at the drug’s safety, tolerability and the amount of the chemical that remains in patients’ bodies over time, said Michael Lobritz, Roche’s infectious diseases chief.
“It has been more than 50 years since the last distinct class of antibiotic was launched that is capable of treating infections by Gram-negative bacteria,” Lobritz said in an email. “Any new antibiotic class that has the ability to treat infections caused by multidrug resistant (MDR) bacteria such as carbapenem-resistant Acinetobacter baumannii (CRAB) would be a significant breakthrough.”
Encouraging as the early results are, scientists stressed that it would be foolish to get cocky in the fight against a bug that, time and time again, has found ways to evade our most advanced pharmaceutical weaponry.
“Resistance has emerged to every antibiotic ever created, and it is likely that resistance will emerge to zosurabalpin in the future too, if it successfully becomes a clinical antibiotic,” Bradley said.
In their findings, the authors noted a few gene mutations in the lab that significantly decreased the drug’s success against A. baumannii. These were rare but worrying; one freak mutation reduced the drug’s effectiveness 256-fold.
“Although the rates of appearance of these resistant organisms is low, and comparable to standard-of-care antibiotics, the observation affirms the principle that we can never rest on our laurels with the chemical and biochemical warfare that we are waging on bacterial pathogens,” Ogunseitan said.
Zosurabalpin is essentially unknown to bacteria. If it proves safe and effective in humans, there’s probably a limited window in which it could effectively spare lives and suffering. But no matter how sophisticated our tools, scientists said, these potentially deadly cells will always have a major advantage against us.
“Bacteria have a big numbers advantage — billions can be in a flask,” said Hergenrother. “Bacteria will eventually evolve resistance to virtually every antibiotic, which is why we need a steady supply of new antibiotic candidates.”
<<<
---
>>> Acurx to present at the 42nd Annual J.P. Morgan Healthcare Conference
PR Newswire
January 3, 2024
https://finance.yahoo.com/news/acurx-present-42nd-annual-j-130000189.html
STATEN ISLAND, N.Y., Jan. 3, 2024 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company") today announced that President and Chief Executive Officer David P. Luci will present at the 42nd Annual J.P. Morgan Healthcare Conference on Thursday, January 11, 2024 at 12:00 pm PST. An audio webcast of the presentation will be available live at https://jpmorgan.metameetings.net/events/healthcare24/sessions/50099-acurx-pharmaceuticals-inc/webcast?gpu_only=true&kiosk=true and will be available upon completion of the conference on the Company's website at www.acurxpharma.com under the Investors section.
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates with a Gram-positive selective spectrum (GPSS®) that blocks the active site of the Gram+ specific bacterial enzyme DNA polymerase IIIC (pol IIIC), inhibiting DNA replication and leading to Gram-positive bacterial cell death. Its R&D pipeline includes antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
>>> Anixa Biosciences, Inc. (ANIX) is developing therapies and vaccines that are focused on critical unmet needs in oncology. Anixa’s programs are using the body’s immune system to take multiple approaches in fighting cancer—CAR-T cell therapy to treat cancer and vaccines to prevent cancer.
https://www.anixa.com/
Therapeutics
CAR-T
We are developing chimeric antigen receptor T-cell (CAR-T) based immuno-therapy drugs which genetically engineer a patient’s own immune cells to fight cancer.
Vaccines
Breast Cancer Vaccine
We are developing a revolutionary vaccine technology focused on preventing breast cancer, specifically triple negative breast cancer, the most lethal form of the disease.
Ovarian Cancer Vaccine
Using a similar mechanism of action as our breast cancer vaccine technology, we are developing a vaccine to prevent the occurrence of ovarian cancer.
<<<
---
>>> Acurx Announces Positive Phase 2b Results Showing 100% of Patients Who Had Clinical Cure with Ibezapolstat Also Had Sustained Clinical Cure
PR Newswire
December 11, 2023
https://finance.yahoo.com/news/acurx-announces-positive-phase-2b-120100375.html
All 15 ibezapolstat-treated patients in Phase 2b who achieved Clinical Cure (CC) at end of treatment (EOT) remained free of C. difficile Infection (CDI) recurrence through one month after EOT, for a Sustained Clinical Cure (SCC) rate of 100%
2 of 14 patients treated with standard of care, oral vancomycin, experienced recurrent infection within one month after EOT for a SCC of 86%
100% of the 25 ibezapolstat-treated patients in Phase 2 (Phase 2a and 2b) who had CC at EOT remained cured through one month after EOT
Further analyses will be forthcoming Q1 2024, as data become available, regarding other endpoints, from the Phase 2b trial, including Extended Clinical Cure (ECC) data up to 94 days and comparative effects vs vancomycin on the gut microbiome
Preparation underway for meetings with FDA, European Medicines Agency and other global regulatory agencies and advancement to international Phase 3 clinical trials
STATEN ISLAND, N.Y., Dec. 11, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a late-stage biopharmaceutical company developing a new class of small molecule antibiotics for difficult-to-treat bacterial infections, today announced positive Phase 2b results showing 100% of CDI patients who had CC with ibezapolstat, the company's late-stage antibiotic candidate, also had SCC.
The efficacy results from the Phase 2 trial (Phase 2a and Phase 2b) are summarized in the table below:
Clinical Cure
(CC)
at EOT
Sustained Clinical Cure
(SCC)
One Month After EOT for
all evaluable patients
Sustained
Clinical Cure* (SCC) One
Month After EOT
ibezapolstat Phase 2a
10/10 (100%)
10/10 (100%)
10/10 (100%)
ibezapolstat Phase 2b
15/16 (94%)
15/16 (94%)
15/15 (100%)
ibezapolstat Phase 2a +
Phase 2b Combined
25/26 (96%)
25/26 (96%)
25/25 (100%)
vancomycin
14/14 (100%)
12/14 (86%)
12/14 (86%)
*Sustained Clinical Cure was evaluated only for patients who were CC at EOT.
Kevin Garey, PharmD, MS, Professor and Chair, University of Houston College of Pharmacy, the Principal Investigator for microbiome aspects of the ibezapolstat clinical trial program and Acurx Scientific Advisory Board member stated: "These results help validate our ongoing scientific investigations into the anti-CDI recurrence properties of ibezapolstat including maintenance and regrowth of healthy gut microbes and bile acid homeostasis. I'm excited about our ongoing investigations into a new scientific paradigm optimizing C. difficile antibiotic development to effectively cure CDI and prevent recurrence."
According to Stuart Johnson, MD, Professor of Medicine, Loyola University (Infectious Disease) and Acurx Scientific Advisory Board member: "Treatment of CDI remains an important unmet medical need, for 2 reasons. First, the potential for development of resistance in C. difficile to currently available drugs like vancomycin threatens our standard therapeutic approach. Second, recurrent disease is a very serious problem with limited available treatment options. Although vancomycin is still an effective treatment, CDI patients treated with oral vancomycin experience a recurrence rate of 18-23%. Ibezapolstat, by virtue of its novel mechanism of action, lack of cross-resistance with any marketed antibiotics, narrow antibacterial spectrum, and selective effects on the gut microbiome, appears to be a promising potential new addition to our therapeutic armamentarium. I continue to be encouraged by the accumulating data showing that ibezapolstat is clinically comparable to vancomycin in treating CDI and preventing recurrence."
Robert J. DeLuccia, Executive Chairman of Acurx, stated: "The overall Phase 2 data demonstrate a high clinical cure rate of 96% together with this 100% recurrence-free rate is a promising one-two punch to C. difficile infection for a potential front-line treatment option for patients with CDI." He further stated: "These two clinical trial endpoints, together with the Phase 1 and Phase 2a clinical trial data and with additional data analyses to come, will form the basis for a comprehensive, solid data package to present to global regulatory authorities to support advancement to Phase 3 clinical trials during the second half next year and move one step closer on its pathway to commercialization."
David P. Luci, President & CEO of Acurx, stated: "Ibezapolstat continues to demonstrate success compared to a standard of care, oral vancomycin, to treat patients with CDI. We anticipate favorable separation between the two therapeutic options will continue in Q1 2024 with extended clinical cure and microbiome comparison data. We expect to leverage this success in a $1 billion plus US CDI market internationally as we move forward with an international Phase 3 clinical trial mandate." He added: "The Company also announced its "Made in America" policy initiative for manufacture of ibezapolstat capsules for Phase 3 clinical trials and commercial supply to ensure patients have uninterrupted access to this potentially life-saving antibiotic mitigating potential supply chain disruptions."
About the Ibezapolstat Phase 2 Clinical Trial
The completed multicenter, open-label single-arm segment (Phase 2a) study was followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542). This Phase 2 clinical trial was designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI
including pharmacokinetics and microbiome changes from baseline and continue to test for anti- recurrence microbiome properties seen in the Phase 2a trial, including the treatment- related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment. The Company's Scientific Advisory Board concurred with this recommendation.
In the now completed Phase 2b trial segment, 32 patients with CDI were enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments were identical in appearance, dosing times, and number of capsules administered to maintain the blind. The overall observed Clinical Cure rate in the combined Phase 2 trials in patients with CDI was 96% (25 out of 26 patients), based on 10 out of 10 patients (100%) in Phase 2a in the Modified Intent to Treat Population, plus 15 out of 16 (94%) patients in Phase 2b in the Per Protocol Population, who experienced Clinical Cure during treatment with ibezapolstat. Ibezapolstat was well-tolerated, with three patients each experiencing one mild adverse event assessed by the blinded investigator to be drug-related. All three events were gastrointestinal in nature and resolved without treatment. There were no drug-related treatment withdrawals or no drug-related serious adverse events, or other safety findings of concern. In the Phase 2b vancomycin control arm, 14 out of 14 patients experienced Clinical Cure. The Company is confident that based on the pooled Phase 2 ibezapolstat Clinical Cure rate of 96% and the historical vancomycin cure rate of approximately 81% (Vancocin® Prescribing Information, January 2021), we will demonstrate non-inferiority of ibezapolstat to vancomycin in Phase 3 trials in accordance with the applicable FDA Guidance for Industry (October, 2022).
The Phase 2b clinical trial segment was discontinued due to success. The Company made this decision in consultation with its medical and scientific advisors and statisticians based on observed aggregate blinded data and other factors, including the cost to maintain clinical trial sites and slow enrollment due to COVID-19 and its aftermath. The Company had determined that the trial performed as anticipated for both treatments, ibezapolstat and the control antibiotic vancomycin (a standard of care to treat patients with CDI), with high rates of clinical cure observed across the trial without any emerging safety concerns. Accordingly, an Independent Data Monitoring Committee was not required to perform an interim analysis of this Phase 2b trial data as originally planned. The Company anticipated that this decision would allow the Company to advance this first-in-class, FDA QIDP/Fast Track-designated antibiotic product candidate to Phase 3 clinical trials more expeditiously.
The Phase 2b trial was originally designed to be a non-inferiority (NI) trial and later amended to include an interim efficacy analysis with review by an Independent Data Monitoring Committee (IDMC). The decision to end the trial early based on blinded clinical observations obviated the need for an interim analysis, IDMC review, and NI assessment. The Company determined, in consultation with its clinical and statistical experts, that presenting clinical cure rates for the primary efficacy endpoint is the most appropriate representation for the clinical activity of ibezapolstat in treating CDI.
In the Phase 2 clinical trial, the Company will also evaluate pharmacokinetics (PK) and microbiome changes and test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy. Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin.
About Ibezapolstat
Ibezapolstat is a novel, orally administered antibiotic being developed as a Gram-Positive Selective Spectrum (GPSS™) antibacterial. It is the first of a new class of DNA polymerase IIIC inhibitors under development by Acurx to treat bacterial infections. Ibezapolstat's unique spectrum of activity, which includes C. difficile but spares other Firmicutes and the important Actinobacteria phyla, appears to contribute to the maintenance of a healthy gut microbiome.
In June 2018, ibezapolstat was designated by the U.S. Food and Drug Administration (FDA) as a Qualified Infectious Disease Product (QIDP) for the treatment of patients with CDI and will be eligible to benefit from the incentives for the development of new antibiotics established under the Generating New Antibiotic Incentives Now (GAIN) Act. In January 2019, FDA granted "Fast Track" designation to ibezapolstat for the treatment of patients with CDI. The CDC has designated C. difficile as an urgent threat highlighting the need for new antibiotics to treat CDI.
About Clostridioides difficile Infection (CDI). According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long-term care facilities and in the community. C. difficile is one of the most common causes of health care- associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate for the antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About the Microbiome in C. difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect. Beneficial effects of bile acids include a decrease in primary bile acids and an increase in secondary bile acids in patients with CDI, which was observed in the Company's Ph2a trial results and previously reported (CID, 2022).
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates with a Gram-positive selective spectrum (GPSS®) that blocks the active site of the Gram+ specific bacterial enzyme DNA polymerase IIIC (pol IIIC), inhibiting DNA replication and leading to Gram-positive bacterial cell death. Its R&D pipeline includes antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
>>> Anixa Biosciences and Cleveland Clinic Present Positive New Data from Phase 1 Study of Breast Cancer Vaccine
DECEMBER 06, 2023
https://ir.anixa.com/press-releases/detail/1030/anixa-biosciences-and-cleveland-clinic-present-positive-new?ftag=YHF4eb9d17
– Antigen-specific T cell responses were observed at all dose levels –
– IFN? and IL-17, immune-mediated biomarkers of T cell activation, increased over time from baseline –
– Vaccine was safe and well tolerated –
– Conference call to commence today at 6:30 p.m. ET –
SAN JOSE, Calif., Dec. 6, 2023 /PRNewswire/ -- Anixa Biosciences, Inc. ("Anixa" or the "Company") (NASDAQ: ANIX), a biotechnology company focused on the treatment and prevention of cancer, today announced new and updated positive results from the Phase 1 clinical trial of its breast cancer vaccine. The trial is being conducted in collaboration with Cleveland Clinic with funding by a grant from the U.S. Department of Defense.
The data were presented at the 2023 San Antonio Breast Cancer Symposium by G. Thomas Budd, M.D., staff physician at Cleveland Clinic Cancer Institute and principal investigator of the study, in a poster entitled "Phase I Trial of alpha-lactalbumin vaccine in high-risk operable triple negative breast cancer (TNBC) and patients at high genetic risk for TNBC."
Patients who had been curatively treated for TNBC received three vaccinations given once every two weeks. IFN? and IL-17, which are T cell immune response indicators (cellular immunity), and antibody production (B cell humoral immunity) were measured to evaluate the vaccination effect. Data from the 16 patients treated to date showed that:
The majority of patients developed ELISpot (T-cell) responses that met the rigorous protocol-specified definition of an immune response, with a measurable but lesser magnitude of response noted in the remaining patients.
12 (75%) of the women had antigen-specific IFN? and/or IL-17 ELISpot responses that were observed at all dose levels, while ELISA antibody responses were observed at Dose Level 2 and higher.
A statistically significant (P = 0.03) increase in IFN? over baseline (Day 0) was observed by Day 56; while a significant (P = 0.0001) increase in IL-17 over baseline was observed by Day 14.
Among the doses studied, Dose Level 1 (10 mcg a-lactalbumin/10 mcg zymosan) was determined to be a usable immunologic dose as well as the maximum tolerated dose (MTD).
No significant side effects were observed, at the MTD, besides irritation at the sites of injection. No myalgias, flu-like symptoms, or aberrant laboratory values were noted.
Anixa and Cleveland Clinic plan to investigate additional intermediate dose levels and continue studying the vaccine's safety and immunologic effects in two additional patient cohorts.
The first cohort, which opened for enrollment in August 2023, is evaluating the combination of the Company's breast cancer vaccine with Keytruda® (pembrolizumab) in post-operative patients found to have residual disease following neoadjuvant chemo-immunotherapy.
The second cohort will investigate the safety and immunologic effects of the vaccine in patients who are BRCA1, BRCA2, or PALB2 mutation positive and are planning prophylactic risk-reducing mastectomies.
"The data from our Phase 1 trial to date has exceeded our expectations, and we are pleased with our progress. This vaccine is designed to direct the immune system to destroy TNBC cancer cells through a mechanism that has never previously been utilized for cancer vaccine development," stated Dr. Amit Kumar, Chairman and CEO of Anixa Biosciences. "We look forward to reviewing additional data as the trial continues to completion, and we are in the planning stages of the Phase 2/3 studies of this vaccine. Our goal is to initially evaluate the vaccine's ability to prevent recurrence of cancer in survivors, and continue with extension studies to eventually determine its effectiveness in preventing the initial onset of TNBC."
"There is a large unmet need for preventing TNBC, an aggressive form of breast cancer with few targeted treatment options available," said Dr. Budd, Cleveland Clinic. "We are encouraged by the data gathered to date and look forward to determining the optimal vaccine dose in additional patient cohorts. Our hope is that future studies will demonstrate that the antigen-specific T cell responses we observed translate to the prevention of breast cancer recurrence."
Anixa is the exclusive worldwide licensee to the novel breast cancer vaccine technology invented at Cleveland Clinic, the site of the Phase 1 trial. The grant from the U.S. Department of Defense was made directly to Cleveland Clinic.
Conference Call Information
Anixa is pleased to invite all interested parties to participate in a conference call, during which this new data will be discussed.
Conference Call Details:
Presentation host:
Anixa management, with special guest speakers
Date and time:
Today, December 6, 2023, at 6:30 p.m. ET
Phone access:
Registration Link to receive your dial-in number and unique PIN
Webcast:
Available at www.anixa.com under "Events & Presentations"
About Triple-Negative Breast Cancer
One in eight women in the U.S. will be diagnosed with an invasive breast cancer at some point in their lives. Approximately 10-15% of those diagnoses are TNBC, however TNBC accounts for a disproportionately higher percentage of breast cancer deaths and has a higher rate of recurrence. This form of breast cancer is twice as likely to occur in African-American women, and approximately 70% to 80% of the breast tumors that occur in women with mutations in the BRCA1 genes are triple-negative breast cancer.
About Anixa Bioscience's Breast Cancer Vaccine
Anixa's breast cancer vaccine takes advantage of endogenously produced proteins that have a function at certain times in life, but then become "retired" and disappear from the body. One such protein is a breast-specific lactation protein, a-lactalbumin, which is no longer found post-lactation in normal, aging tissues, but is present in the majority of triple-negative breast cancers. Activating the immune system against this "retired" protein provides preemptive immune protection against emerging breast tumors that express a-lactalbumin. The vaccine also contains an adjuvant that activates an innate immune response, which allows the immune system to mount a response against emerging tumors to prevent them from growing. This vaccine technology was invented by the late Dr. Vincent Tuohy, who was the Mort and Iris November Distinguished Chair in Innovative Breast Cancer Research in the Department of Inflammation and Immunity at Cleveland Clinic's Lerner Research Institute. Dr. Tuohy was inventor of the technology, which Cleveland Clinic exclusively licensed to Anixa Biosciences. He was entitled to a portion of the commercialization revenues received by Cleveland Clinic and also held equity in Anixa.
About Anixa Biosciences, Inc.
Anixa is a clinical-stage biotechnology company focused on the treatment and prevention of cancer. Anixa's therapeutic portfolio consists of an ovarian cancer immunotherapy program being developed in collaboration with Moffitt Cancer Center, which uses a novel type of CAR- T, known as chimeric endocrine receptor T-cell (CER-T) technology. The Company's vaccine portfolio includes a novel vaccine being developed in collaboration with Cleveland Clinic to prevent breast cancer – specifically triple negative breast cancer (TNBC), the most lethal form of the disease – as well as a vaccine to prevent ovarian cancer. These vaccine technologies focus on immunizing against "retired" proteins that have been found to be expressed in certain forms of cancer. Anixa's unique business model of partnering with world-renowned research institutions on clinical development allows the Company to continually examine emerging technologies in complementary fields for further development and commercialization. To learn more, visit www.anixa.com or follow Anixa on Twitter, LinkedIn, Facebook and YouTube.
<<<
---
Anixa Biosciences - >>> An end to breast cancer? California company develops groundbreaking vaccine with promising future
CBS News
by Ashley Sharp
December 8, 2023
SACRAMENTO -- An estimated one in eight women will develop breast cancer in their lifetime, killing on average 42,000 women a year in the United States.
What if there was a vaccine that would significantly lower each woman's chance of ever getting it in the first place? In that question lies what could be the answer to one day eliminating the deadly disease.
A groundbreaking vaccine created through decades of research at the Cleveland Clinic and developed by Anixa Biosciences in San Jose, California is driving innovation by targeting triple-negative breast cancer, the disease's deadliest and most aggressive form.
"This vaccine could potentially eliminate breast cancer," said Dr. Amit Kumar, Anixa CEO.
The vaccine's findings from its first trial with 16 women were published Wednesday, with each participant reporting no bad side effects and no resurgence of their cancer so far.
Jennifer Davis, a brave woman from small-town Ohio, was the first woman in the world to get the vaccine in October 2021.
"This is how we advance medicine. It's important to be a part of those things," Davis said. "I am just beyond grateful."
When Davis heard the dreaded words "you have cancer" in September 2018, it came six months after her first alert to an abnormality on a routine mammogram and ultrasound.
At the time, her biopsy turned up negative for cancer.
"I really wanted to believe everything was OK, but I knew something wasn't right," Davis said.
At 41 years old, she had no history of cancer. Still, she could feel a lump growing and decided to go for a second opinion and a second biopsy.
She was diagnosed with triple-negative breast cancer. Her mind instantly went to her family and three children.
"It was very hard to tell them and try and be strong for them," Davis said. "With triple negative, there is nothing for us to take — no pill or anything to prevent recurrence. The rate is high and outcomes are poor if it does come back."
After chemotherapy, a double mastectomy and radiation, Davis was finally free of cancer, but she was not free of the fear that lingers.
"I was always nervous and afraid of it coming back," she said.
So when she learned of an experimental vaccine trial while receiving her cancer care at the Cleveland Clinic, she thought, "What do I have to lose?"
"It was something that was going to give me peace of mind," Davis said. "If this could work for me, then I wouldn't have to worry about a recurrence."
She became the first woman in the world to take the breast cancer vaccine.
A registered nurse herself, what eased her mind was the fact that in years of trials in animals, there had been no cancer recurrence and no anaphylactic reaction.
"That was all I needed to hear," said Davis, who reports that in the two years since taking the vaccine, she has never felt better.
The vaccine has been studied for more than two decades at the Cleveland Clinic, pioneered by pre-clinical research led by the late Dr. Vincent Tuohy.
Inspired by this and what it could mean for the future of cancer diagnosis, Dr. Kumar approached the clinic about developing the vaccine.
"I looked at it and I saw the vision," Kumar said.
So how does it work?
"Is it, in essence, teaching your body not to grow a tumor?" CBS13 reporter Ashley Sharp asked.
"That's exactly right. It's teaching your body to destroy the cells that can grow a tumor," Kumar said.
If a virus shows up in the body, the immune system teaches itself how to destroy it, knowing, easily, which cells are bad.
In cancer, it is more difficult, Dr. Kumar explained.
"All of the cells that become cancerous in your body came from normal, healthy cells," Kumar said. "The difference is not big, so the immune system has a harder time recognizing a cancer cell and distinguishing it from a healthy cell."
According to the Cleveland Clinic, the vaccine works by targeting a lactation protein called a-lactalbumin, which is no longer found after lactation in normal, aging tissues. It is, however, present in most triple-negative breast cancer patients. If breast cancer develops, the vaccine is designed to instruct the immune system to attack the tumor and keep it from growing entirely.
"The results are incredibly promising," Kumar said. "The vision is one day to be able to give this to any woman who wants to prevent cancer from ever occurring in her body. It's a small step and we have many more steps to go, but it's incredible if we can make this happen."
It's a promising find for the future of fighting cancer that started with one woman but hopefully ends with every woman.
"The bigger picture of this is overwhelming for me," Davis said.
The second vaccine trial is set to start in 2024, this time with 600 women instead of 16. This study will be on a much larger scale, where half the women will get the vaccine and the other half will get a placebo.
The hope is that within five years, they can get FDA approval to distribute the vaccine to the public.
<<<
---
>>> Anixa Biosciences, Inc. (ANIX), a biotechnology company, develops therapies and vaccines focusing on critical unmet needs in oncology and infectious diseases. The company's therapeutics programs include the development of a chimeric endocrine receptor T-cell therapy, a novel form of chimeric antigen receptor T-cell (CAR-T) technology focusing on the treatment of ovarian cancer; and the development of anti-viral drug candidates for the treatment of COVID-19 focused on inhibiting certain protein functions of the virus. Its vaccine programs comprise the development of a vaccine against triple negative breast cancer; and a preventative vaccine against ovarian cancer. The company is also developing immuno-therapy drugs against cancer. The company was formerly known as ITUS Corporation and changed its name to Anixa Biosciences, Inc. in October 2018. Anixa Biosciences, Inc. was incorporated in 1982 and is based in San Jose, California.
<<<
---
An Acurx interview tonight with Bloomberg TV (see below), so this could be part of the catalyst for today's jump in the stock.
As we know, the first batch of durability data should be released in Dec, per CEO Luci's previous guidance. Then the next batch (day 94 data) should be ~ Jan, and then the microbiome data. So a 3 stage release of data over the next few months to make things interesting :o)
I figure the 94 day durability data in Jan is the most important, with the Dec data being the lead up. If the Dec data is clearly positive the stock should zoom. If it's ambiguous, then we wait for the 94 day data in Jan, and the microbiome data. As a bio 'binary event' this is unusual, since a disappointment in Dec can be made moot by good news in Jan. So this de-risks the stock considerably. We already know that Ibex leaves the microbiome relatively intact, compared to Vancomycin, so Ibez's 'recurrence advantage' should increase over time, which makes for excellent odds with the 94 day data.
All IMHO :o)
>>> NEW YORK, Dec. 07, 2023 (GLOBE NEWSWIRE) -- FMW Media's New to The Street announces its corporate interview lineup for its business show airing as a sponsored program on Bloomberg TV tonight, Thursday, December 7, 2023, at 9:30 PM PT.
... 3) Biopharmaceutical - Acurx Pharmaceuticals, Inc.'s (NASDAQ: ACXP) ($ACXP) interview with David Luci, President/CEO.
https://finance.yahoo.com/news/street-tv-announces-episode-536-142000722.html
<<<
---
Repost from Dew's Revance board -- >>> RVNC 3Q23 financial summary:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173182593
https://www.businesswire.com/news/home/20231108710804/en
• 3Q23 Daxxify sales were $22.0M, -3% QoQ. (There is no meaningful YoY growth figure because Daxxify was not yet on the market during 3Q22.) Note: The slight QoQ decline during the seasonally slow third calendar quarter is effectively an increase in the projected annualized rate.
• 3Q23 dermal-filler sales were $32.1M, +1% QoQ and +23% YoY.
• 3Q23 non-GAAP operating expenses (excluding revenue and COGS) were $69.0. (RVNC’s guidance for full-year 2023 non-GAAP operating expenses is $315-335M.)
• Cash at 9/30/23 was $300.2M, a $19.5M decrease relative to 6/30/23. (RVNC raised $50M in Aug 2023 from the third tranche of the Athyrium Capital loan.)
<<<
---
Repost from Dew's ENTA board -- >>> ENTA reports FY4Q23* results and pipeline update:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173266700
https://ir.enanta.com/news-releases/news-release-details/enanta-pharmaceuticals-reports-financial-results-its-fiscal-37
FY4Q23 royalty revenue was $18.9M, unchanged from the prior quarter. 54.5% of ENTA’s Mavyret royalties are payable to OMERS†, and this amount is treated for accounting purposes as an amortization of debt (see discussion in #msg-172603887).
9/30/23 cash = $370M, a decrease of $23M relative to 6/30/23.
Today, ENTA issued FY2024* guidance as follows:
• R&D expense of $100-120M, down sharply from $163.5M in FY2023.
• SG&A expense of $45-50M, down modestly from $52M in FY2023.
Based on the reduced operating expenses described above, ENTA says its cash and expected royalty revenue are sufficient to fund operations for the next four years—i.e. through the end of FY2027.
Pipeline
• EDP-938 (RSV N-protein inhibitor): ENTA expects to report data from the phase-2b RSVPEDs (pediatric) and the phase-2b RSVHR (high-risk adults) trials in calendar 3Q23, assuming there is a normal RSV season in the Northern Hemisphere.
• EDP-323 (RSV L-protein inhibitor): ENTA just started a phase-2a “challenge” study; data expected in calendar 3Q23.
• The preclinical program for single-agent dual inhibition of RSV and hMPV has been terminated.
• EDP-235 (COVID protease inhibitor): If a partnership is secured for a phase-3 trial, the population tested will be standard-risk patients with a primary endpoint based on symptom relief.
• Non-virology: “The company will announce new therapeutic programs beginning in early 2024.” An analyst on today’s CC asked if one of these programs was GLP-1, but ENTA declined to answer.
*ENTA’s fiscal years end on September 30
†Ontario Municipal Employees Retirements System (Canada’s largest pension fund)
<<<
---
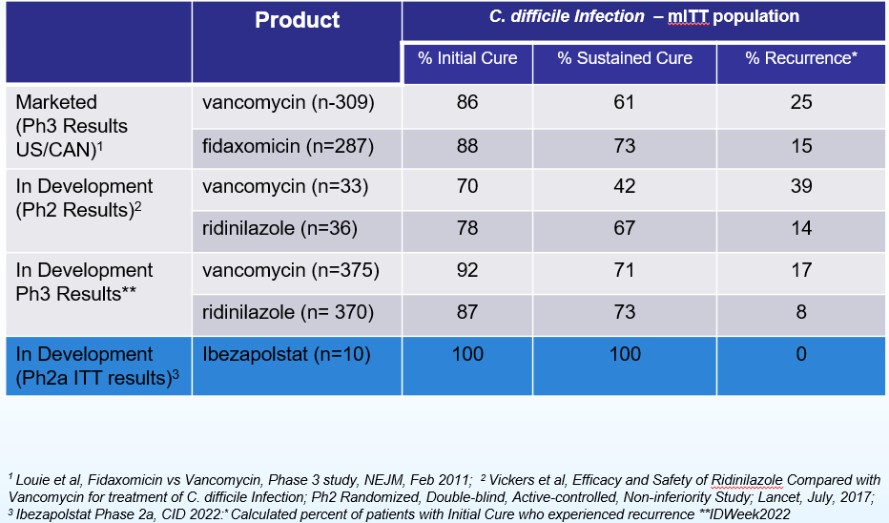
Acurx -- a summary slide of the various CDI trial results of the various antibiotics (below). Two are Phase 3 results, and one is from a Phase 2. In the Phase 2 (33 patients), the Vancomycin initial cure rate was 70%. Compare that to Acurx's Phase 2, where Vancomycin had a 100% initial cure rate. So it demonstrates just how limited the validity of all data is from a small Phase 2 -
---
>> MEIP <<
Looks like they decided to not pursue that Phase 3 program, after further talks with the FDA, though their partner will continue it in Japan. And the $41 mil in 'net income' turns out to be from "recognition of deferred revenue associated primarily with the termination of the Kyowa Kirin Commercialization Agreement in July 2023" (link below), so not real net income derived from actual product sales. The cash level is $81 mil, but looks like the burn rate is just under $20 mil per quarter, so they project having sufficient cash for approx 1 year. Clinical programs look like Phase 1 and 1b, so early stage. So I guess MEIP's low $39 mil market cap is appropriate, all things considered -
https://finance.yahoo.com/news/mei-pharma-reports-first-quarter-210500033.html
---
Blade, Just curious if you are still following MEIP? I noticed that MEIP's chart setup looks interesting for a possible upside breakout move, but I haven't kept up with the company's fundamentals. I remember last year the FDA wouldn't allow accelerated approval for MEIP's cancer drug based on the Phase 2, and would require the full Phase 3 to be completed for approval. But just curious if you might still be following the company? Yahoo Finance has their cash level at $82 mil, and revenues of $105 mil (!) and net income of $41 mil (!), which if accurate could make MEIP the bargain of the century, considering the current market cap is only $37 mil.
Thanks for any insights, and hope you are doing well :o)
---
>>> MEI Pharma, Inc. (MEIP), a clinical-stage pharmaceutical company, focuses on the development and commercialization of various therapies for the treatment of cancer. The company develops Zandelisib, an oral phosphatidylinositol 3-kinase delta inhibitor for the treatment of patients with relapsed/refractory follicular lymphoma; and Voruciclib, an oral cyclin-dependent kinase 9 inhibitor, which is in Phase I clinical trial for acute myeloid leukemia and B-cell malignancies. It also develops ME-344, a mitochondrial inhibitor targeting the oxidative phosphorylation complex which has completed Phase I clinical trial for the treatment of human epidermal growth factor receptor 2 negative breast cancer. MEI Pharma, Inc. has a license agreement with Presage Biosciences, Inc. The company was formerly known as Marshall Edwards, Inc. and changed its name to MEI Pharma, Inc. in July 2012. MEI Pharma, Inc. was incorporated in 2000 and is headquartered in San Diego, California.
<<<
---
>>> Halozyme Therapeutics, Inc. (HALO), a biopharma technology platform company, researches, develops, and commercializes proprietary enzymes and devices in the United States, Switzerland, Ireland, Belgium, Japan, and internationally. The company's products are based on the patented recombinant human hyaluronidase enzyme (rHuPH20) that enables delivery of injectable biologics, such as monoclonal antibodies and other therapeutic molecules, as well as small molecules and fluids. It offers Hylenex recombinant, a formulation of rHuPH20 to facilitate subcutaneous (SC) fluid administration for achieving hydration to enhance the dispersion and absorption of other injected drugs in SC urography and to enhance resorption of radiopaque agents; XYOSTED, an injection for SC administration of testosterone replacement therapy; NOCDURNA, a sublingual tablet to treat nocturia due to nocturnal polyuria; TLANDO, an oral formulation for testosterone replacement therapy; and ATRS-1902, a proprietary drug device combination product. The company also provides Herceptin (trastuzumab), Herceptin Hylecta, and Phesgo to treat breast cancer; Mabthera SC for the treatment of multiple blood cancer; HYQVIA to treat primary immunodeficiency disorders; and DARZALEX for patients with amyloidosis, smoldering myeloma, and multiple myeloma. In addition, it offers Epinephrine Injection to treat allergy and immunology; Sumatriptan injection for migraines; exenatide and teriparatide injections; Makena, a progestin drug to reduce the risk of preterm birth; and OTREXUP, a SC methotrexate injection for adults with severe active rheumatoid arthritis and severe recalcitrant psoriasis, as well as children with active polyarticular juvenile idiopathic arthritis. Further, the company provides ATRS-1902 for adrenal crisis rescue; ARGX-113; and ARGX-117 to treat severe autoimmune diseases in multifocal motor neuropathy. Halozyme Therapeutics, Inc. was founded in 1998 and is headquartered in San Diego, California.
<<<
---
>>> Corcept Therapeutics Incorporated (CORT) engages in discovery and development of drugs for the treatment of severe metabolic, oncologic, endocrine, and neurological disorders in the United States. It offers Korlym (mifepristone) tablets as a once-daily oral medication for the treatment of hyperglycemia secondary to hypercortisolism in adult patients with endogenous Cushing's syndrome, who have type 2 diabetes mellitus or glucose intolerance, and have failed surgery or are not candidates for surgery. The company is developing relacorilant to treat patients with Cushing's syndrome; and nab-paclitaxel in combination with relacorilant, which has completed Phase II clinical trial to treat patients with advanced ovarian tumors, as well as for the treatment of cortisol excess. It is also developing selective cortisol modulator to treat patients with metastatic castration-resistant prostate cancer; selective cortisol modulator for the treatment of antipsychotic-induced weight gain and other disorders; and FKBP5 gene expression assays. The company was incorporated in 1998 and is headquartered in Menlo Park, California.
<<<
---
>>> Ambrx Biopharma Inc. (AMAM), a clinical stage biopharmaceutical company, focuses on discovering and developing antibody drug conjugates (ADCs) and other engineered therapies to modulate the immune system. It focuses on portfolio of clinical and preclinical programs designed to optimize efficacy and safety in multiple cancer indications, including ARX517, its proprietary antibody-drug conjugate (ADC) targeting the prostate-specific membrane antigen (PSMA) and ARX788, its proprietary ADC targeting HER2. The company also has preclinical and clinical collaborations with multiple partners on drug candidates using Ambrx technology. The company was incorporated in 2003 and is headquartered in La Jolla, California.
<<<
---
>>> Iovance Biotherapeutics, Inc. (IOVA), a clinical-stage biotechnology company, focuses on developing and commercializing cancer immunotherapy products to harness the power of a patient's immune system to eradicate cancer cells. The company's lead product candidate is lifileucel that is in Phase II clinical trial for the treatment of metastatic melanoma and cervical cancer. It also develops LN-145 for the treatment of metastatic non-small cell lung cancer; IOV-4001 for the treatment of melanoma non-small cell lung cancer; IOV-2001; and IOV-3001. Iovance Biotherapeutics, Inc. has collaborations and licensing agreements with H. Lee Moffitt Cancer Center; M.D. Anderson Cancer Center; Ohio State University; Centre hospitalier de l'Université de Montreal; Cellectis S.A.; Novartis Pharma AG; Melanoma Institute Australia; and Beth-Israel Deaconess Medical Center. The company was formerly known as Lion Biotechnologies, Inc. and changed its name to Iovance Biotherapeutics, Inc. in June 2017. Iovance Biotherapeutics, Inc. was incorporated in 2007 and is headquartered in San Carlos, California.
<<<
---
>>> Sutro Biopharma, Inc. (STRO) operates as a clinical-stage oncology company. It develops site-specific and novel-format antibody drug conjugates (ADC) that enables its proprietary integrated cell-free protein synthesis platform, XpressCF and XpressCF+. The company's product candidates include STRO-001, an ADC directed against the cancer target CD74 for patients with multiple myeloma and non-Hodgkin lymphoma that is in Phase 1 clinical trials; and STRO-002, an ADC directed against folate receptor-alpha for patients with ovarian and endometrial cancers, which is in Phase 1 clinical trials. Its pre-clinical product candidate STRO-003, a ADC directed against an anti-receptor tyrosine kinase-like orphan receptor 1 (ROR1) for the treatment of solid tumors. It has collaboration and license agreements with Merck Collaboration to develop research programs focusing on cytokine derivatives for cancer and autoimmune disorders; Celgene Corporation to discover and develop bispecific antibodies and/or ADCs focused on the field of immuno-oncology; EMD Serono to develop ADCs for multiple cancer targets; and Astellas Pharma Inc. to develop ADC. The company was formerly known as Fundamental Applied Biology, Inc. Sutro Biopharma, Inc. was incorporated in 2003 and is headquartered in South San Francisco, California.
<<<
---
>>> Acurx Pharmaceuticals, Inc. (NASDAQ:ACXP) Q3 2023 Earnings Call Transcript November 14, 2023
Insider Monkey
https://www.insidermonkey.com/blog/acurx-pharmaceuticals-inc-nasdaqacxp-q3-2023-earnings-call-transcript-1223844/#q-and-a-session
Acurx Pharmaceuticals, Inc. beats earnings expectations. Reported EPS is $-0.23952, expectations were $-0.28.
Operator: Ladies and gentlemen, good morning, and welcome to the Acurx Pharmaceuticals Third Quarter 2023 Earnings Conference Call. At this time, all participants are in a listen-only mode. A brief question-and-answer session will follow the formal presentation. [Operator Instructions] As a reminder, this conference is being recorded. It is now my pleasure to introduce your host, Rob Shawah, Chief Financial Officer. Please go ahead.
Robert Shawah: Thank you, Ryan. Good morning, and welcome to our call. This morning, we issued a press release providing financial results and company highlights for the third quarter of 2023, which is available on our website at acurxpharma.com. Joining me today is Dave Luci, President and CEO of Acurx, who will give a corporate update and outlook; and Bob DeLuccia, our Executive Chairman, who will provide his perspective as the manager of our development programs, including the Phase II clinical trial. After Dave’s comments, I’ll provide some highlights of the financials for the quarter ended September 30, 2023, and then turn the call back over to Dave for his closing remarks. As a reminder, during today’s call, we’ll be making certain forward-looking statements.
A research laboratory arranged with a variety of test tubes filled with liquids for biopharmaceutical research.
These forward-looking statements are based on current information assumptions, estimates and projections about future events that are subject to change and involve a number of risks and uncertainties that may cause actual results to differ materially from those contained in forward-looking statements. Investors should consider these risks and other information described in our filings made with the Securities and Exchange Commission, including our quarterly report on Form 10-Q, which we filed yesterday, Monday, November 13, 2023. You are cautioned not to place undue reliance on these forward-looking statements and Acurx disclaims any obligation to update such statements at any time in the future. This conference call contains time-sensitive information that’s accurate only as of the date of this live broadcast today, November 14, 2023.
Acurx undertakes no obligation to revise or update any forward-looking statements to reflect events or circumstances after the date and time of this conference call. I’ll now turn the call over to Dave Luci. Dave?
David Luci: Thanks, Rob. Good morning, everyone and thanks for joining us to review our financial results for the third quarter and also to cover some exciting recent updates. Then we’d be pleased to take any questions. On October 2, 2023, we ended enrollment in our Phase IIb clinical trial of ibezapolstat, our lead antibiotic candidate for the treatment of patients with C. difficile infection or CDI. On November 2, 2023, we reported top line data from the Phase II clinical trial, including the ibezapolstat clinical cure rate at end of treatment or EOT of 96%, 25 out of 26 patients, including 100% in Phase IIa 10 for 10 and 94% in Phase IIb 15 for 16, as well as the cure rate for oral vancomycin at EOT of a 100% 14 of 14. No safety concerns were reported in either arm of the Phase IIb clinical trial or in the Phase IIb open-label trial.
Based on the Phase II data, in consultation with our scientific advisors, we determined that clear evidence of clinical cure has been established with ibezapolstat, and it is clinically comparable to vancomycin. Ibezapolstat will now move forward to Phase III clinical trials. Further data will be provided when available on all of the secondary and exploratory endpoints in the Phase IIb trial, including sustained clinical cure, extended clinical cure up to 94 days and the comparative impact on the microbiome. We anticipate that these secondary and exploratory endpoints will provide clear separation between these two therapeutic options and all of these endpoints will be disclosed when available over the next 90 days. The Phase IIb trial was originally designed to be a non-inferiority trial and later amended to include an interim efficacy analysis with review by an independent data monitoring committee or IDMC.
The decision to end the trial early based on the blinded clinical observations obviated the need for an interim analysis, the need for the IDMC review and the non-inferiority assessment. The company determined in consultation with its clinical and statistical experts that presenting clinical cure rates for the primary efficacy endpoint is the most appropriate representation for the clinical activity of ibezapolstat in treating C. diff infection. We remain particularly excited about the dual impact of ibezapolstat to treat the acute C. diff infection while appropriately managing the long-term care of each patient’s microbiome, which we believe is exceptional for antibiotic therapy. Other key highlights from the third quarter or in some cases shortly thereafter, include the following.
The World Anti-Microbial Resistance Congress convened its annual meeting in Philadelphia in September 2023 where experts in the field from both the public and private sectors weighed in on the latest innovations to address antimicrobial resistance. Our Executive Chairman with us today, Bob DeLuccia, presented an update entitled, Novel DNA Pol IIIC Inhibitors for Gram-Positive Bacterial Infections: Preparing for the Next Pandemic. This presentation as well as the others that I’ll describe is available on our website acurxpharmaceuticals.com. At ID Week which convened in Boston, October 11 to 15, Acurx was featured at two scheduled events. First, an oral presentation was provided by Dr. Kevin Garey, Professor and Chair, University of Houston College of Pharmacy, and the Principal Investigator for microbiome aspects of our ibezapolstat trial program entitled: Elucidating the Gram-Positive Selective Spectrum Activity of Ibezapolstat; Secondary Analysis from the Phase IIa trial.
Secondly, at ID Week Acurx presented at the symposium entitled New Antimicrobials in the Pipeline. At the symposium, Acurx presentation was entitled Novel DNA Pol IIIC inhibitors for Gram-Positive Bacterial Infections. Next up was the ClostPath meeting, the International Conference on Molecular Biology and Pathogenesis of Clostridia, at which there were three scientific posters presented during the conference in Banff, Canada from September 19 to 23. We provided new information supporting ibezapolstat’s unique pharmacologic profile. The first of the three was entitled ibezapolstat modulates Clostridioides difficile virulence factors in vitro showed ibezapolstat reduces toxin production by C. diff bacteria. The second entitled C. difficile In Vitro Biofilm Studies of Ibezapolstat and Comparator Antibiotics showed ibezapolstat was as effective as the currently used anti C.
diff antibiotics, fidaxomicin, vancomycin and metronidazole, reducing biofilm embedded C. difficile. The third entitled Metagenomic Evaluation of Ibezapolstat Compared to Other Anti-C. diff Agents showed ibezapolstat and fidaxomicin both caused favorable proportional increases in bacteroidetes, but distinct from vancomycin and metronidazole, which caused unfavorable proportional increases in proteobacteria. All the presentations again are available on our website. And now back to our CFO, Rob Shawah to guide you through the highlights of our financial results for the third quarter of 2023. Rob?
Robert Shawah: Thanks, Dave. Our financial results for the third quarter ended September 30, 2023 were included in our press release issued earlier this morning. The company ended the third quarter with cash totaling $7.1 million compared to $9.1 million, as of December 31, 2022. I’ll also note that subsequent to the quarter to the September 30, we did receive $2.2 million in cash from warrant conversions in October of 2023. Research and development expenses for the three months ended September 30, 2023 were $1.3 million compared to $1.6 million for the three months ended September 30, 2022. The decrease was due to the timing of Phase IIb trial related costs. For the nine months ended September 30, 2023, research and development expenses were $4.1 million versus $3.3 million for the nine months ended September 30, 2022.
The increase is due primarily to Phase IIb trial related costs and an increase in consulting costs. General and administrative expenses for the three months ended September 30, 2023 were $1.8 million compared to $2 million for the three months ended September 30, 2022. The decrease was due primarily to a $0.2 million decrease in professional fees. For the nine months ended September 30, 2023, general and administrative expenses were $5.4 million versus $5.5 million for the nine months ended September 30, 2022. The amounts reflect a decrease in professional fees of $0.3 million, offset by an increase of $0.2 million in share-based compensation. The company reported a net loss of $3.1 million or $0.24 per diluted share for the three months ended September 30, 2023, compared to a net loss of $3.5 million or $0.32 per diluted share for the three months ended September 30, 2022, and a net loss of $9.5 million or $0.77 per share for the nine months ended September 30, 2023, compared to a net loss of $8.8 million or $0.84 per diluted share for the nine months ended September 30, 2022, all for the reasons previously mentioned.
The company had 13,005,128 shares outstanding as of September 30, 2023. With that, I’ll turn the call back over to Dave. Dave?
David Luci: Thanks, Rob and to all of you joining us today. We outlined advances in several areas that we believe will spur continued momentum and growth to build on our strong fundamentals. We look forward to sustaining this momentum even during these challenging times and sharing future updates in the coming months. Now in advance of our customary Q&A, I’ll ask my Co-Founder and Executive Chairman, Bob DeLuccia to provide his perspective given Bob manages our research and development programs, including the recently completed Phase II clinical trial. Bob?
Robert DeLuccia: Thanks, Dave and thanks for updating our stakeholders on our recent progress and thanks to all for your continuing support to reach this important clinical development milestone, which takes ibezapolstat one step closer to commercialization for CDI patients in need of a promising new antibiotic with a novel bactericidal mechanism of action. And this is especially important in this age of emerging antimicrobial resistance to the currently used antibiotics. So from my perspective, we now have robust scientific evidence to present a strong data package to FDA for an end of Phase II meeting. The outcome of this meeting will confirm our readiness to advance the Phase III clinical trials with specifics on trial design and patient enrollment targets.
At the same time, we’ll submit our plans to the European Medicines Agency for conducting Phase III clinical trials outside the United States and we expect to have their guidance around midyear next year. Bottom line is, I think we have a new antibiotic, which is first in a new class and fast tracked by the FDA. It’s fully patented. It has regulatory exclusivity 10 years post market introduction in the U.S. as well. It works extremely well. It’s clinically comparable to the standard of care after 10 days old treatment in a serious and potentially life-threatening infection that demands antibiotic treatment. From what we’ve seen so far, we expect to further demonstrate favorable effects on the microbiome and less recurrence of infection. It’s also very well-tolerated and efficient to manufacture, so we can be cost-competitive in the marketplace.
Now since we’ll be the only C. diff antibiotic beginning Phase III next year, assuming success, we’ll be next up at that for approval and market introduction in the U.S. and countries outside the United States. In my over 50 years’ experience in antibiotic development and marketing, I think I’ve got a good rearview mirror that gives me a clear vision and a pathway forward to deliver a winner here, not only for patients with C. diff infection, but in general, for better public health and of course, for our shareholders. In my opinion, simply put, ibezapolstat kills the bug and preserves the microbiome. And back to you, Dave.
David Luci: Thank you, Bob. I’ll now open the call for questions. Operator?
Operator: Thank you. Ladies and gentlemen, we will now be conducting a question-and-answer session. [Operator Instructions] Our first question is from Jason McCarthy with Maxim Group. Please go ahead.
Michael Okunewitch: Hey, guys. How is it going? This is Michael Okunewitch on the line for Jason. And first off, I’d like to congratulate you on the progress.
David Luci: Thank you, Michael.
Michael Okunewitch: So I guess to start off, I’d like to for you to give us a bit more of an idea of what you’re thinking ahead of the end of Phase II meeting in terms of what a Phase III program could look like in terms of size and scope, potential costs? And then also if you would still be targeting non-inferiority as a primary endpoint. Can you just give me your thoughts on that?
David Luci: Sure. And yes, we would still be targeting non-inferiority to the control arm, which would be oral vancomycin, much like Summit did most recently in its effort to get an antibiotic approved to treat C. diff. What we’re looking at is we’re looking at two clinical trials in Phase III, two registration trials. And we’re considering, and all of this is preliminary, as you know, but we’re considering an imbalanced approach pursuant to which we would have fewer patients in the first trial and more patients in the second trial, so that we could potentially raise money to fund the smaller first trial. And with continued good data in hand, non-inferiority in this case for a Phase III registration trial, we would hope to see an uptick in our share price and use that uptick to raise money for the second trial.
Keeping in mind that this sequential approach is very possible for us because we have 10 years of market exclusivity with our new molecular entity status and QIDP. So I think that’s what we would do. I think the first trial I think we would ballpark figures, we would try to have a 2:1 randomization potentially and preliminarily. If that gets through our science team and the FDA, it might look something like 133 patients on ibezapolstat and 66 on oral vancomycin. So it would be a much more discrete trial that I think a lot of people are thinking. So to pay for it won’t, for a small company won’t be that challenging.
Michael Okunewitch: All right. Thank you for that. And then one more for me and I’ll hop in the queue. Just as we’re getting up to those secondary analysis, could you talk a little bit about what you’re looking for specifically in terms of clinically relevant separation from Vanco? What kind of threshold do we need to reach in those secondary end points?
David Luci: So we think that the microbiome advantage is the key advantage because that’s the thing that most antibiotics don’t do. So if we can address the acute infection, while at the same time, fully restoring a healthy microbiome to baseline, that’s something which I don’t know of any other antibiotic that’s able to do that. We’re still studying the mechanism of action to see how that’s done. But I think that provides clear separation by itself. We’re also the only folks that have gone out formally 94 days for antibiotics and C. diff at least, to see that there are no reinfections that far out. So I’m particularly excited about that 94-day out data to see in a subset of patients how many patients of ours are reinfections 94 days out compared to vancomycin.
So those are two real exciting pieces. And what we’re going to do since all of the secondary and exploratory endpoints are so material from a corporate level perspective, and we don’t want to be holding material non-public information for any sort of period of time for the SEC purpose. So we’ll be getting the data out as it comes in, the sustained clinical cure, the extended clinical cure and of course the microbiome comparison. We won’t wait for it to be all together, and we’ll get it out as we receive it.
Michael Okunewitch: All right. Thank you very much.
David Luci: Thank you, Michael.
Operator: Thank you. Our next question is from Ed Arce with H.C. Wainwright. Please go ahead.
Thomas Yip: Hi. Good morning, everyone. This is Thomas Yip asking the [indiscernible] questions for Ed. Thank you for taking the questions. So perhaps first question, I believe you touched on it a little bit. When can we expect to see more Phase II data? Will this be in a purification — or will this be around major medical companies?
David Luci: I’m sorry, when can you expect to see the secondary endpoint information?
Thomas Yip: Yes. That’s right. This additional Phase II data additional analysis.
David Luci: I see. Okay. So we’ll have a first press — in terms — I’ll start out with the press release disclosure and then I’ll turn it over to Bob DeLuccia for the scientific conference disclosure. I think some of that is still a bit up in the air. But on the press releases, we’ll very likely come out with the first press release on sustained clinical cure in December. In either December or January, we’ll have the 94-day out data, the extended clinical cure data. And in January or February, we’ll have the data on the microbiome. Then, of course, in March, we’ll have the meeting with the FDA, and we’ll have a press release around that too after it’s completed. But Bob, did you want to mention the scientific presentations with the Phase IIb data?
Robert DeLuccia: Yeah. I think with some of the data we targeted in early next year as it becomes available, as Dave said. But concurrently, as we get the final study report, we’ll be preparing the data for publication as well.
Thomas Yip: Great. Thank you. We definitely look forward to that. Perhaps just one more question from us. This one is financial. You mentioned a little earlier Phase III is expected to be conducted in a sequential manner. Can you provide some preliminary thoughts, estimates on estimated costs for this first Phase III study? And what are some options to move forward this initial Phase III study?
David Luci: Yeah. I mean we have — we’re going to unveil our detailed plan in coming weeks. We think the first of the two trials, and again, this depends on the data and as you know, it is preliminary. But the first of the two trials will probably range between $20 million and $25 million. So right now, as we sit here with between $9 million and $9.5 million and we have about another $15 million in warrant exercises, which after more successful data is announced, we expect to see some of that coming in, in terms of cash for the warrant shares. But beyond that, we don’t have a very heavy lift and we have a detailed plan. And what I can tell you is it’s going to be as non-dilutive as humanly possible.
Thomas Yip: Understood. Thank you again for taking our questions. Looking forward to your updates in the next coming months.
David Luci: Very good. Well, thank you, Tom. Assume that and thanks to Ed as well.
Operator: Thank you. Our next question is from the line of James Molloy with Alliance Global Partners. Please go ahead.
James Molloy: Hi. Good morning. Thank you very much taking my questions. Could you walk us through a little bit, maybe a little competitive analysis where Summit their failure in ’21, but Dave said back in 11 (ph) obviously with the approval. Can you walk through a little bit to sort of the dosing, the reinfection rate what Summit did wrong with what Merck and Cubist did right on deficit and how that ties into ibezapolstat and what you guys are hoping to do here in your Phase III?
David Luci: Sure. The Merck example is the most clear example because they — Merck’s predecessor Optimer went to Phase III with 15 out of 16 cures in an open-label trial. We have 15 of 16 in IIb and another 10 of 10 in IIa. So we’re going at 25 of 26. So as Bob mentioned, it’s a robust package and is supplemented by our manufacturing, preclinical and other data. So we’re delighted with that and we’re following a successful pathway with the fidaxomicin pathway. What others have done wrong, so Summit Therapeutics, you mentioned, they conducted a superiority trial, as I understand it, and it wasn’t involved. But from what I gather from the public disclosure, they enrolled quite well through COVID at around 169 trial sites internationally.
And we like that model for our Phase III, which we expect to be international as well. And you can see their sites on clinicaltrial.gov. But they had a superiority trial. And while they are conducting that trial, Jim, they actually try — they changed their primary endpoint and unblinded the data and took a look. And only after that did they go to the FDA to try to get the FDA to kind of ratify what they did, and the FDA wasn’t comfortable with that. So I don’t know exactly how that series of decisions kind of happened. We didn’t do that. We contacted the FDA prior to ending the Phase IIb trial to make sure that everything was copacetic and they were very good at getting back to us quickly. And we also reached out and contacted our independent data monitoring committee and our Scientific Advisory Board to make sure that we are in unanimous agreement that this was the right decision.
And certainly, we think it was. So that’s kind of like an alternative to how Summit kind of ran their Phase III. So the thing about Summit’s Phase III that we like is the pace of their enrollment. I think they got about 750 patients internationally in about two years’ time. Our first study will be, I think, somewhere in the neighborhood of 200-ish.
James Molloy: Understood. And also, one of the — you talked about the healthy microbiome with the ibezapolstat, I know — again, not to pile on some of what, they are most recent relevant company to look at. They were talking about the microbiome and their data as well and obviously didn’t help them very much. How do you quantify sort of a healthy microbiome? And how much do you think that plays into the FDA’s decision vis-a-vis just really being non-inferior to Vanco?
David Luci: I think it’s a burgeoning fast-growing kind of business sector, the microbiome. And the reason why it’s so important is because when you have an imbalanced microbiome, just generally outside of C. diff, it leads to disease, whether it’s cancer or C. diff or diabetes, all kinds of things are triggered as we find more and more by an imbalanced microbiome. Now in terms of C. diff, the primary cause of reinfections is an imbalanced microbiome, right? And the C. diff reinfection market is best estimate $4.7 billion a year in the U.S. So if you can restore a healthy microbiome, you’re basically able to make a very nice dent in the public health cost in the recurrent C. Diff market. So we think that’s going to play an important role.
And it’s going to distinguish us from a broad spectrum antibiotic like oral vanc, which has — it just decimates the microbiome because it’s a broad spectrum. I think oral vanc was approved in 1986 to treat C. diff because there was so little out there that was useful to treat C. diff. It wasn’t that it was the best tool because it’s broad spectrum, not narrow spectrum, but there was just such a need that it got the approval and its first approval was in 1958. I hope that answers your question.
James Molloy: It does indeed. And just a couple of questions, if I could, please. Any updates on the PASTEUR Act?
David Luci: We haven’t had any updates on the PASTEUR Act specifically. We understand there’s a number of different legislative options out there that are being considered. But the more I watch Washington, the more I realize that I have no idea what’s going on. I mean, it looks like we’re coming up to another government shutdown. And I’m sure nobody is thinking ahead of the holidays. They’re trying to keep the government open right now.
James Molloy: Okay. Maybe just a last question for Robert. Keeping a close eye on the accrued expenses here in the third quarter. Can you walk us through what the 82% of the [indiscernible] vendor are on that, please?
David Luci: Rob, do you want to?
Robert Shawah: Yes, that’s our clinical research organization. Yes.
Robert DeLuccia: Our CRO, yes.
James Molloy: Great. Thanks for taking the questions.
David Luci: Thank you, Jim.
Operator: Thank you. Our next question is from the line of John Stinton (ph) an Investor. Please go ahead.
Unidentified Participant: Thank you for taking my questions.
David Luci: Good morning, John.
Unidentified Participant: Can you hear me now?
David Luci: Yes. I can hear you.
Unidentified Participant: Right. So with regard to the 94-day study, is it possible that, that study when fully digested will prove clinical superiority rather than non-inferiority?
David Luci: There will be numbers that people can interpret, but it won’t be statistically driven.
Robert DeLuccia: Correct. You’re right, Dave.
Unidentified Participant: Okay. Then follow-up is the original Phase IIa and b studies, you said were to prove non-inferiority, was it your expectation at the time that it would indeed prove clinically superior? And was that a disappointment that it did not or was that just not something you were measuring at all?
David Luci: Well, originally, that was the plan, to measure for statistical noninferiority, and if proven, to test for superiority. But just like with the independent data monitoring committee mechanism, those mechanisms kind of got put to bed when we decided to end the Phase IIb trial early because there are so few patients evaluable in the IIb. There was no mathematical mechanism to measure for non-inferiority or superiority. So in Phase II, unlike registration in Phase III trials, you need to establish clinical comparability to move on to Phase III. So we decided that since we are looking at the blinded data and it looks so positive, you could see how many failures there were, or in our case, there were not, that it was certain to us that we would be able to establish clinical comparability and move on to Phase III.
So we didn’t want to — for a number of reasons, we didn’t want to waste the time, the money and not have our drug at market as soon as possible. We figured we have a win on the table here, let’s take it off the table and move on.
Unidentified Participant: Okay. Thank you. One last question is since October 2, we’ve had some very wild swings in the price of this Acurx with the company with some days as many as and exceeding 8 million — almost 10 million shares being traded in a single day, which is pretty unusual. Do you have any comments on that?
David Luci: No. I mean, we came out with data, right? So we’re going to be coming out with even more data. Three to five solid press releases in the next kind of period of time ending at the end of the first quarter. So we expect to have a lot more high-volume days between now and the end of the first quarter. I will note that as part of the corporate maturation process, this is what happens. At the end of 2022, we were trading about 21,000 shares a day, if you recall, average daily trading volume. So now as you look at it in the rearview mirror, as Bob says, we’re now entering kind of the mid-life of microcap pharmaceutical company. And as we become Phase III ready in every sense, it’s – we expect the trend to continue.
What I like about it and what I will say as well, I like the notion that it seems to me, and this will be coming out more and more through public filings of our ownership. It seems to me that more and more of our shares are entering institutional hands, which is another thing that’s very healthy for the company.
Unidentified Participant: All right. Thank you.
David Luci: Thank you, John.
Operator: Thank you. Our next question is from the line of Ryan Mulholland (ph) with 50-50 LLC (ph). Please go ahead.
Unidentified Participant: Hi, David. Thank you very much for taking the time. Just a couple of questions. One regarding the Phase IIb trial and the randomization. Was that a block randomization that was used? And are we to assume that the two incomplete participants were then from the vancomycin arm? And then second question — you can go for it.
David Luci: Yeah. I was just going to say, I don’t know what a block randomization is, and I wouldn’t assume — I don’t know the two protocol violators, I don’t know which arm they were in. What I can tell you is that the randomization in the IIb was done at the local level as opposed to a centralized randomization. So I think that means it’s not a block randomization. But I’m not entirely sure where the two protocol vials have come out.
Unidentified Participant: Okay. Thank you. Do you know if that information will be forthcoming?
David Luci: I mean, I don’t expect to know. I haven’t asked because they’re protocol violators. So there’s nothing that I can assume if I had that information. So I don’t even think I asked, so the numbers are not evaluated.
Unidentified Participant: And then lastly, over the past year you’ve had several discussions, several interviews where you have discussed potential M&A participation and your kind of interest in not taking maybe ibezapolstat over to Phase III yourself, but finding a partner. Is that still something that’s on your radar? And are there interesting parties who have signed NDAs to investigate that interest on their own?
David Luci: Yes. So there’s a lot to bite off there. But yes, we do have NDAs signed in some cases, with interested parties. They being confidential, I can’t tell you the names. And yes, M&A is definitely on our calendar for 2024. We think sharing risk with a big pharma partner for Phase III in the commercial period is a prudent idea. Now it takes two parties to create a deal. I’m not certain whether or not a deal will come to fruition. And we won’t know what our value is until we unveil and find out what the secondary and exploratory endpoints are from the recently completed trial. So we kind of have to have that information in order to formally launch that process. But in the first quarter of the year, I’m sure we’ll formally start the process with an asterisk that if we were to get a term sheet in the meantime, then we would be forced — if they were within the ballpark that the Board of Directors find generally interesting, then they may form a special committee and have us move forward earlier than we expected.
But that’s about it. So for now, I would just refer you to the most recent deals in the C. diff space that have been consummated. And you can kind of get an idea of what evaluations are like. So one deal from November 2020 was when Astellas sold European rights to fidaxomicin, to Tillotts Pharma AG in Europe. And another deal was the Destiny Pharma deal with Sebela Pharmaceuticals, which look big, $540 million, but that was only $1 million upfront and the $540 million of all of that money isn’t payable until the very end of the marketing period, which, I don’t know, might be 15 or 20 years out. So those are the comparables that we see in the space. And we’ll look at our data. And hopefully our data shows a clear separation and we’re able to get something done in terms of M&A in 2024.
And we’ll be working on a parallel track with Phase III preparation, and we’ll see how far we get with each.
Unidentified Participant: Thank you and your team. I appreciate all your efforts and it’s a pretty great product you put out there.
David Luci: Thank you, Ryan.
Operator: Thank you. As there are no further questions, I would now hand the conference over to Dave Luci for his closing comments.
David Luci: Thank you very much, Ryan. We’re pleased for all of you coming to the conference today and expressing your thoughts and questions and we look forward to updating you soon. Let’s sit tight and buckle up and 2024 is going to be a great year, we expect. Thank you.
Operator: Thank you. The conference of Acurx Pharmaceuticals has now concluded. Thank you for your participation. You may now disconnect your lines.
<<<
---
Acurx conference call (Nov 14, 2023) --->
Some additional info from the Acurx conference call. Luci said that the durability and microbiome data from the Phase 2b will be released as the company gets it in the weeks and months ahead (see below), rather than all at once at the end. Here is his approx timeline for the news flow -
Dec ------------ Sustained Clinical Cure data
Dec or Jan --- Extended Clinical Cure data up to 94 days
Jan or Feb --- Impact on Microbiome data
March --------- End of Phase 2 meeting with FDA
On the partnership / M+A front, Luci said they have some NDAs (plural). As I understand it, an NDA is a Non-Disclosure Agreement, which (I think) allows sharing of non-public info like clinical data between Acurx and interested pharmas.
Luci has clearly said (in July) that he favors an M+A or partnering deal prior to the Phase 3s, and from the conf call it sounds like this process will advance in 2024 as the additional data becomes available. So a lot will probably depend upon the strength of the upcoming durability and microbiome data, the number of interested pharmas, etc.
With the Phase 3s, they will need two, and Luci said they are favoring (if possible) a smaller first Phase 3 trial, and then a larger second Phase 3. The first Phase 3 would be approx 200 patients (133 getting Ibez and 66 getting Vanc), and would likely cost in the $20-25 mil range, and will include some international sites, and will be a non-inferiority trial. He said the advantage of a smaller first Phase 3 is that it would be cheaper and faster, and should provide more than enough data to get an M+A or pharma deal (assuming good data of course). Ideally though, Luci said (in July) that he prefers doing a deal prior to Phase 3.
On the funding / cash side, he said they now have $9 mil range, and up to $15 mil potentially coming from the exercising of warrants. Luci says he wants to proceed - 'as non-dilutively as is humanly possible'.
Concerning the Pasteur Act, he said that he isn't expecting anything happening soon (is awaiting Congressional approval) since the government is currently in such disarray.
Anyway, this info is from my notes, so you may want to verify by listening to the conference call -
877-660-6853, and conf call code is - 13742354
---
>>> Acurx Pharmaceuticals, Inc. Reports Third Quarter 2023 Results and Provides Business Update
November 14, 2023
https://finance.yahoo.com/news/acurx-pharmaceuticals-inc-reports-third-120100928.html
STATEN ISLAND, N.Y., Nov. 14, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of antibiotics for difficult-to-treat bacterial infections, announced today certain financial and operational results for the third quarter ended September 30, 2023.
Highlights of the third quarter ended September 30, 2023, and in some cases shortly thereafter, include:
On October 2, 2023, Acurx ended enrollment in its Phase 2b clinical trial of ibezapolstat, its lead antibiotic candidate, for the treatment of patients with C. difficile infection, or CDI;
On November 2, 2023, Acurx reported top-line data from the Phase 2 clinical trial including the ibezapolstat clinical cure rate at end of treatment, or EOT, of 96% (25/26) including 100% in Phase 2a (10/10) and 94% in Phase 2b (15/16) as well as the cure rate for oral vancomycin at EOT of 100% (14/14);
Ibezapolstat will now move forward to Phase 3 clinical trials. Preparation underway for End-of-Phase 2 FDA Meeting and advancement to Phase 3
No safety concerns were reported in either arm of the Phase 2b clinical trial or in the Phase 2a open label trial;
In consultation with its scientific advisors, the Company determined that clear evidence of clinical cure was established with ibezapolstat and ibezapolstat is clinically comparable to vancomycin, the standard of care to treat CDI;
Further data will be provided when available on all of the secondary and exploratory endpoints in the Phase 2b trial, including sustained clinical cure data, extended clinical cure data up to 94 days and impact on the microbiome when compared to vancomycin.
The Company anticipates that these secondary and exploratory endpoints will provide clear separation between these two therapeutic options and provide validation for front-line use of ibezapolstat to treat patients with CDI;.
In September 2023, the World Antimicrobial Resistance (AMR) Congress convened its annual meeting in Philadelphia where experts in the field from both the public and private sectors weighed in on the latest innovations to address AMR. Our Executive Chairman, Bob DeLuccia, presented an update entitled: "Novel DNA pol IIIC Inhibitors for Gram-positive Bacterial Infections: Preparing for the Next Pandemic".
The IDSA (Infectious Diseases Society of America) convened its annual meeting, called ID Week, in Boston from October 11-15, 2023. Acurx was featured at two scheduled events:
First, an oral presentation by Dr. Kevin Garey, Professor and Chair, University of Houston College of Pharmacy, and the Principal Investigator for microbiome aspects of our ibezapolstat clinical trial program, was given on October 14 entitled: Elucidating the Gram-Positive Selective Spectrum Activity of Ibezapolstat; Secondary Analysis from the phase 2a trial.
Second, Acurx presented at the symposium entitled, "New Antimicrobials in the Pipeline" on October 12. At the symposium, Acurx presentation was entitled: "Novel DNA pol IIIC Inhibitors for Gram-positive Bacterial Infections."
Three scientific posters were presented during the CLOSTPATH conference held in Banff, Canada from September 19 to 23, 2023 and provided new information further supporting ibezapolstat's unique pharmacologic profile:
The first entitled: "Ibezapolstat modulates Clostridioides difficile virulence factors in vitro" showed Ibezapolstat reduces toxin production by the C. difficile bacteria…
The second entitled: "C. difficile In Vitro Biofilm Studies of Ibezapolstat And Comparator Antibiotics" showed ibezapolstat was as effective as the currently-used anti-C. difficile antibiotics fidaxomicin, vancomycin and metronidazole in reducing reduce biofilm-embedded C. difficile…
The third entitled: "Metagenomic Evaluation of Ibezapolstat Compared to Other Anti-C. difficile Agents" showed ibezapolstat and fidaxomicin both caused favorable proportional increases in Bacteroidetes but distinct from vancomycin and metronidazole, which caused unfavorable proportional increases in Proteobacteria.
All the presentations described above are available on our website.
Third Quarter 2023 Financial Results
Cash Position:
The Company ended the third quarter with cash totaling $7.1 million, compared to $9.1 million as of December 31, 2022. After the quarter end, the Company received an additional $2.2 million in cash associated with the conversion of approximately 680,000 warrants, which resulted in the issuance of approximately 680,000 shares.
R&D Expenses:
Research and development expenses for the three months ended September 30, 2023, were $1.3 million compared to $1.6 million for the three months ended September 30, 2022. The decrease was due to the timing of Phase 2b trial related costs. For the nine months ended September 30, 2023, research and development expenses were $4.1 million versus $3.3 million for the nine months ended September 30, 2022. The increase is due primarily to Phase 2b trial related costs and an increase in consulting costs.
G&A Expenses:
General and administrative expenses for the three months ended September 30, 2023, were $1.8 million compared to $2.0 million for the three months ended September 30, 2022. The decrease was due primarily to a $0.2 million decrease in professional fees. For the nine months ended September 30, 2023, general and administrative expenses were $5.4 million versus $5.5 million for the nine months ended September 30, 2022. The amounts reflect a decrease in professional fees of $0.3 million, offset by an increase of $0.2 million in share-based compensation.
Net Loss:
The Company reported a net loss of $3.1 million or $0.24 per diluted share for the three months ended September 30, 2023 compared to a net loss of $3.5 million or $0.32 per diluted share for the three months ended September 30, 2022, and a net loss of $9.5 million or $0.77 per share for the nine months ended September 30, 2023, compared to a net loss of $8.8 million or $0.84 per diluted share for the nine months ended September 30, 2022 for the reasons previously mentioned.
The Company had 13,005,128 shares outstanding as of September 30, 2023.
Conference Call
As previously announced, David P. Luci, President and Chief Executive Officer, and Robert G. Shawah, Chief Financial Officer, will host a conference call to discuss the results and provide a business update as follows:
Date:
Tuesday, November 14, 2023
Time:
8:00 a.m. ET
Toll free (U.S. and International):
877-790-1503
Conference ID:
13742354
About the Ibezapolstat Phase 2 Clinical Trial
On November 2, 2023, we reported top-line data from the Phase 2 clinical trial including the ibezapolstat clinical cure rate at end of treatment, or EOT, of 96% (25/26) including 100% in Phase 2a (10/10) and 94% in Phase 2b (15/16) as well as the cure rate for oral vancomycin at EOT of 100% (14/14). No safety concerns were reported in either arm of the Phase 2b clinical trial or in the Phase 2a open label trial. In consultation with its scientific advisors, the Company has determined that clear evidence of clinical cure has been established with ibezapolstat and is clinically comparable to vancomycin. Ibezapolstat will now move forward to Phase 3 clinical trials. Further data will be provided when available on all of the secondary and exploratory endpoints in the Phase 2b trial, including sustained clinical cure data, extended clinical cure data up to 94 days and impact on the microbiome compared to vancomycin.We anticipate that these secondary and exploratory endpoints will provide clear separation between these two therapeutic options.
The completed multicenter, open-label single-arm segment (Phase 2a) study was followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542).
This Phase 2 clinical trial was designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI including pharmacokinetics and microbiome changes from baseline and continue to test for anti-recurrence microbiome properties seen in the Phase 2a trial, including the treatment-related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment. In the now completed Phase 2b trial segment, 32 patients with CDI were enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments were identical in appearance, dosing times, and number of capsules administered to maintain the blind. This Phase 2 clinical trial will also evaluate pharmacokinetics (PK) and microbiome changes and test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy.
Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin.
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200;
doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect.
About Clostridioides difficile Infection (CDI)
According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long term care facilities and in the community. C. difficile is one of the most common causes of health care- associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate of two of the three antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates that target the DNA polymerase IIIC enzyme and its R&D pipeline includes early-stage antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
With Acurx, here's what Dew had to say about the company when I asked him several weeks ago (link below). He makes some excellent points about the dwindling cash level, as well as the potential for warrants being exercised soon, which could bring in some cash -
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173027440
The CEO Luci is a finance guy, and they did a $4 mil money raise in May, and a similar one in 2022. Acurx was also in the running for approx $11 mil in funding for a pre-clinical stage program, but that fell through in August -
>>> The Company was notified by CARB-X that its application for a non-dilutive grant to fund its pre-clinical antibiotic candidate, ACX375C, was not approved. CARB-X noted that the 2023 round of funding was very competitive and that their Scientific Advisory Board was enthusiastic about pol IIIC as the bacterial target of our molecules and that the sufficiently good PK and safety properties of the compounds justified the proposed lead optimization plan. CARB-X encouraged us to re-apply for potential future requests for proposals or RFPs that CARB-X will continue to promulgate from time to time for CARB-X funding consideration. <<<
https://www.acurxpharma.com/news-media/press-releases/detail/60/acurx-pharmaceuticals-inc-reports-second-quarter-2023
---
>>> Acurx Announces Positive Top-Line Ibezapolstat Phase 2 Efficacy Results with 96% Clinical Cure Rate in Patients with C. difficile Infection
PR Newswire
November 2, 2023
https://finance.yahoo.com/news/acurx-announces-positive-top-line-110100342.html
In a total of 26 ibezapolstat-treated patients in Phases 2a and 2b, the Clinical Cure rate is 96%
Meets protocol primary objective of assessing the primary efficacy endpoint of the Clinical Cure rate after 10 days of oral treatment
Ibezapolstat was well tolerated; no drug-related Serious Adverse Events
Further analyses forthcoming regarding secondary and exploratory endpoints, including Sustained Clinical Cure data, Extended Clinical Cure data up to 94 days and comparative effects on gut microbiome
Preparation underway for End-of-Phase 2 FDA Meeting and advancement to Phase 3
Ibezapolstat has previously received FDA Qualified Infectious Disease Product and Fast-Track Designation
Management will be available for Q&A on November 14, 2023 earnings call https://www.acurxpharma.com/news-media/press-releases/detail/65/acurx-pharmaceuticals-to-discuss-third-quarter-2023
STATEN ISLAND, N.Y., Nov. 2, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of small molecule antibiotics for difficult-to-treat bacterial infections, today announced efficacy results from the Phase 2 clinical trial (Phase 2a segment and Phase 2b segment) of ibezapolstat for the treatment of C. difficile Infection (CDI).
The Company previously announced that it decided to terminate the Phase 2b vancomycin-controlled trial segment early based on aggregate blinded data showing a high observed clinical cure rate with no emerging safety concerns. The Company made this decision in consultation with its medical and scientific advisors based on the compelling clinical observation that the clinical cure rate for ibezapolstat was projected to be at least 90% pooled across the open-label Phase 2a and the blinded Phase 2b segments with no safety concerns noted. Other factors also influenced the decision, including the cost of maintaining clinical trial sites and the challenging enrollment environment in the U.S. due to the COVID-19 pandemic and its aftermath.
The Phase 2b trial was originally designed to be a non-inferiority (NI) trial and later amended to include an interim efficacy analysis with review by an Independent Data Monitoring Committee (IDMC). The decision to end the trial early based on blinded clinical observations obviated the need for an interim analysis, IDMC review, and NI assessment. The Company determined, in consultation with its clinical and statistical experts, that presenting clinical cure rates for the primary efficacy endpoint is the most appropriate representation for the clinical activity of ibezapolstat in treating CDI.
The overall observed Clinical Cure rate in the combined Phase 2 trials in patients with CDI was 96% (25 out of 26 patients), based on 10 out of 10 patients (100%) in Phase 2a in the Modified Intent to Treat Population, plus 15 out of 16 (94%) patients in Phase 2b in the Per Protocol Population, who experienced Clinical Cure during treatment with ibezapolstat. Ibezapolstat was well-tolerated, with three patients each experiencing one mild adverse event assessed by the blinded investigator to be drug-related. All three events were gastrointestinal in nature and resolved without treatment. There were no drug-related treatment withdrawals or no drug-related serious adverse events, or other safety findings of concern. In the Phase 2b vancomycin control arm, 14 out of 14 patients experienced clinical cure. The Company is confident that based on the pooled Phase 2 ibezapolstat clinical cure rate of 96% and the historical vancomycin cure rate of approximately 81% (Vancocin® Prescribing Information, January 2021), we will demonstrate non-inferiority of ibezapolstat to vancomycin in Phase 3 trials in accordance with the applicable FDA Guidance for Industry (October, 2022).
According to Stuart Johnson, MD, Professor of Medicine, Loyola University (Infectious Disease) and Acurx Scientific Advisory Board member: "I am very encouraged by the accumulating data showing that ibezapolstat is clinically comparable to vancomycin in treating CDI. Since there is only one other antibiotic besides vancomycin approved for treatment of this serious disease, there is a clear need for more first-line therapeutic agents in our armamentarium. Moreover, the advancement of a small synthetic molecule with a novel bactericidal mechanism to treat CDI is especially important in this era of emerging antimicrobial resistance. I am very supportive of the decision by the Company to end the Phase 2 trial and plan for Phase 3."
Kevin Garey, PharmD, MS, Professor and Chair, University of Houston College of Pharmacy, the Principal Investigator for microbiome aspects of the ibezapolstat clinical trial program and Acurx Scientific Advisory Board member stated: "These results confirm and extend our data from the open-label component of the Phase 2 study and demonstrate the potent activity of ibezapolstat against C. difficile. I anticipate our ongoing microbiome studies will confirm the microbiologic eradication of C. difficile and also compare microbiome changes to advance knowledge of anti-CDI recurrence properties of ibezapolstat. Our previous data showed that ibezapolstat unexpectedly spares other Firmicutes along with the important Actinobacteria phylum necessary for maintaining a healthy microbiome. These characteristics, in conjunction with ibezapolstat's ability to favorably increase the ratio of secondary-to-primary bile acids in the colon, suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to standard of care vancomycin."
Robert J. DeLuccia, Executive Chairman of Acurx, stated: "These ibezapolstat observed clinical cure rate results are impressive for an investigational antibiotic in a Phase 2 trial for CDI. They greatly enhance our scientific evidence base and, in our view, provide robust support for an anticipated End-of-Phase 2 FDA Meeting to occur towards end of first quarter next year with advancement into Phase 3 clinical trials to follow. Additional analyses of secondary and exploratory endpoints of the Phase 2b clinical trial will be forthcoming as soon as available and will include: Sustained and Extended Clinical Cure rates up to 94 days, systemic exposure to ibezapolstat, comparative effects on gut microbiome and, patient reported outcomes."
Mr. DeLuccia added: "I thank all of our clinical trial investigators and their staffs and patients who participated in the trial, as well as all our stakeholders whose support contributed to reaching this important clinical development milestone which is one step closer to commercialization for patients-in-need of a promising new class of antibiotic for treatment of CDI."
David P. Luci, the Company's President and Chief Executive Officer, stated: "Ultimately the marketplace will determine the antibiotic of choice for front-line treatment of CDI. But, in our view, as we plan to enter Phase 3 pivotal clinical trials, ibezapolstat appears to have the properties for ultimate competitive advantage including high clinical cure rates, low recurrent infection, minimal microbiome disruption and manufacturing efficiencies to allow competitive pricing. We believe the market will recognize and appreciate these advantages."
The Company recognizes the month of November as C. difficile Awareness Month as designated by the US Centers for Disease Control and Prevention (CDC) and supports the work of the Peggy Lillis Foundation and the C Diff Foundation in raising awareness, educating and advocating for the Prevention, Treatments, Clinical Trials, and Environmental Safety of Clostridioides difficile (C. difficile) Infections worldwide. Please visit their websites:
Peggy Lillis Foundation: https://cdiff.org/
Cdiff Foundation: https://cdifffoundation.org/.
About the Ibezapolstat Phase 2 Clinical Trial
The completed multicenter, open-label single-arm segment (Phase 2a) study was followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542). This Phase 2 clinical trial was designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI including pharmacokinetics and microbiome changes from baseline and continue to test for anti- recurrence microbiome properties seen in the Phase 2a trial, including the treatment-related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment. The Company's Scientific Advisory Board concurred with this recommendation.
The Phase 2b clinical trial segment was discontinued due to success. The Company made this decision in consultation with its medical and scientific advisors and statisticians based on observed aggregate blinded data and other factors, including the cost to maintain clinical trial sites and slow enrollment due to COVID-19. The Company has determined that the trial performed as anticipated for both treatments, ibezapolstat and the control antibiotic vancomycin (a standard of care to treat patients with CDI), with high rates of clinical cure observed across the trial without any emerging safety concerns. Accordingly, an Independent Data Monitoring Committee will not be required to perform an interim analysis of this Phase 2b trial data as originally planned. The Company anticipates that this decision will allow the Company to advance this first-in-class, FDA QIDP/Fast Track-designated antibiotic product candidate to Phase 3 clinical trials more expeditiously.
In the now completed Phase 2b trial segment, 32 patients with CDI were enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments were identical in appearance, dosing times, and number of capsules administered to maintain the blind.
In the Phase 2 clinical trial, the Company will also evaluate pharmacokinetics (PK) and microbiome changes and test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy. Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin.
About Ibezapolstat
Ibezapolstat is a novel, orally administered antibiotic being developed as a Gram-Positive Selective Spectrum (GPSS™) antibacterial. It is the first of a new class of DNA polymerase IIIC inhibitors under development by Acurx to treat bacterial infections. Ibezapolstat's unique spectrum of activity, which includes C. difficile but spares other Firmicutes and the important Actinobacteria phyla, appears to contribute to the maintenance of a healthy gut microbiome.
In June 2018, ibezapolstat was designated by the U.S. Food and Drug Administration (FDA) as a Qualified Infectious Disease Product (QIDP) for the treatment of patients with CDI and will be eligible to benefit from the incentives for the development of new antibiotics established under the Generating New Antibiotic Incentives Now (GAIN) Act. In January 2019, FDA granted "Fast Track" designation to ibezapolstat for the treatment of patients with CDI. The CDC has designated C. difficile as an urgent threat highlighting the need for new antibiotics to treat CDI.
About Clostridioides difficile Infection (CDI). According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long-term care facilities and in the community. C. difficile is one of the most common causes of health care-associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate for the antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect. Beneficial effects of bile acids include a decrease in primary bile acids and an increase in secondary bile acids in patients with CDI, which was observed in the Company's Ph2a trial results and previously reported (CID, 2022).
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates with a Gram-positive selective spectrum (GPSS®) that blocks the active site of the Gram+ specific bacterial enzyme DNA polymerase IIIC (pol IIIC), inhibiting DNA replication and leading to Gram-positive bacterial cell death. Its R&D pipeline includes antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
>>> Acurx Announces Ibezapolstat Scientific Posters and Presentations at ClostPath 2023 and IDWeek 2023 Scientific Conferences
PR Newswire
October 19, 2023
https://finance.yahoo.com/news/acurx-announces-ibezapolstat-scientific-posters-110100090.html
Three scientific posters highlighting novel anti-virulence pharmacologic properties of oral ibezapolstat for C. difficile Infection; effects on toxin production, biofilm and the gut microbiome
A podium presentation entitled First of a New Class of Antibiotics (pol IIIC Inhibitors) Targeting CDC/FDA/WHO Priority Pathogens; Preparing for the Next Pandemic: Antimicrobial Resistance in Gram-positive Bacterial Infections
Ibezapolstat has previously received FDA QIDP and Fast-Track Designation
STATEN ISLAND, N.Y., Oct. 19, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of antibiotics for difficult-to-treat bacterial infections, today announced three scientific posters were presented during the 13th International Conference on Molecular Biology and Pathogenesis of Clostridia (ClostPath) held in Banff, Canada from September 19 to 23, 2023. Additionally, two podium presentations were made at the Infectious Disease Society of America (IDSA) IDWeek™ 2023 Conference held October 11-15, 2023 in Boston, MA. Highlights of each are shown below.
Robert J. DeLuccia, Executive Chairman of Acurx, stated: "In light of our recent decision to discontinue the Phase 2b ibezapolstat clinical trial earlier than planned and prepare for Phase 3 clinical trials, the new information contained in these scientific posters and presentations at these conferences will add to our evidence-based briefing package for an End of Phase 2 FDA meeting planned for in the first half of next year." He also added: "We are currently compiling and verifying all data from the Phase 2b trial and we will report topline clinical efficacy for the primary clinical endpoint and safety data in the coming weeks, with other outcome data available later this year".
ClostPath:
Ibezapolstat modulates Clostridioides difficile virulence factors in vitro
Presented by Eugenie Basseres, et al; University of Houston College of Pharmacy
Ibezapolstat reduces toxin production by C. difficile
C. difficile In Vitro Biofilm Studies of Ibezapolstat And Comparator Antibiotics
Presented by M. Jahangir Alam et al; University of Houston College of Pharmacy
Ibezapolstat was as effective as the currently-used anti-C. difficile agents fidaxomicin, vancomycin and metronidazole to reduce biofilm-embedded C. difficile quantity and biofilm biomass
Metagenomic Evaluation of Ibezapolstat Compared to Other Anti-Clostridioides difficile Agents
Presented by Jinhee Jo, University of Houston College of Pharmacy
Ibezapolstat and fidaxomicin caused proportional increases in Bacteroidetes distinct from vancomycin and metronidazole, which caused proportional increases in Proteobacteria
IDWeek:
First of a New Class of Antibiotics (pol IIIC Inhibitors) Targeting CDC/FDA/WHO Priority Pathogens
Presented by Michael Silverman, MD, FACP, Acurx's Medical Director; at the New Antimicrobials in the Pipeline session
Among the promising data for ibezapolstat in the treatment of C. difficile are in vitro potency, anti-virulence activities, high human fecal concentrations, 100% Clinical Cure rate in a 10-patient open-label trial, favorable safety profile to date, and potentially beneficial effects on the gut microbiome
Elucidating the Gram-Positive Selective Spectrum Activity of Ibezapolstat; Secondary Analysis from the Phase 2a trial; Presented by Kevin Garey, PharmD, MS, Professor& Chair, University of Houston, School of Pharmacy
Ibezapolstat showed variable selectivity against Firmicutes helping to elucidate its narrow spectrum of activity against certain pathogenic Firmicutes including C. difficile
The posters and presentations are available on the Company's website www.acurxpharma.com.
About Ibezapolstat
Ibezapolstat is a novel, orally administered antibiotic being developed as a Gram-Positive Selective Spectrum (GPSS™) antibacterial. It is the first of a new class of DNA polymerase IIIC inhibitors under development by Acurx to treat bacterial infections. Ibezapolstat's unique spectrum of activity, which includes C. difficile but spares other Firmicutes and the important Actinobacteria phyla, appears to contribute to the maintenance of a healthy gut microbiome.
In June 2018, ibezapolstat was designated by the U.S. Food and Drug Administration (FDA) as a Qualified Infectious Disease Product (QIDP) for the treatment of patients with CDI and will be eligible to benefit from the incentives for the development of new antibiotics established under the Generating New Antibiotic Incentives Now (GAIN) Act. In January 2019, FDA granted "Fast Track" designation to ibezapolstat for the treatment of patients with CDI. The CDC has designated C. difficile as an urgent threat highlighting the need for new antibiotics to treat CDI.
About the Ibezapolstat Phase 2 Clinical Trial
The completed multicenter, open-label single-arm segment (Phase 2a) study was followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542). This Phase 2 clinical trial was designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI including pharmacokinetics and microbiome changes from baseline and continue to test for anti-recurrence microbiome properties seen in the Phase 2a trial, including the treatment-related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment.
The Phase 2b clinical trial segment has been discontinued due to success. The Company made this decision in consultation with its medical and scientific advisors and statisticians based on observed aggregate blinded data and other factors, including the cost to maintain clinical trial sites and slow enrollment due to COVID-19. The Company has determined that the trial performed as anticipated for both treatments, ibezapolstat and the control antibiotic vancomycin (a standard of care to treat patients with CDI), with high rates of clinical cure observed across the trial without any emerging safety concerns. Accordingly, an Independent Data Monitoring Committee will not be required to perform an interim analysis of this Phase 2b trial data as originally planned. Acurx will analyze the data and report topline efficacy results promptly. The Company anticipates that this decision will allow the Company to advance this first-in-class, FDA QIDP/Fast Track-designated antibiotic product candidate to Phase 3 clinical trials more expeditiously.
In the now completed Phase 2b trial segment, 32 patients with CDI were enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments were identical in appearance, dosing times, and number of capsules administered to maintain the blind.
This Phase 2 clinical trial will also evaluate pharmacokinetics (PK) and microbiome changes and test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy. In the event noninferiority of ibezapolstat to vancomycin is demonstrated, further analysis will be conducted to test for superiority. Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin.
About ClostPath
The ClostPath conferences, which began in 1995, have been a leading venue to bring together top scientists and clinicians studying the molecular biology of clostridia and their role in health and disease. The scientific program of ClostPath 13 included lectures by internationally recognized leaders in clostridial research and clinical practice. In addition to state-of-the-art invited talks on the most recent and exciting discoveries in the field, short oral contributions were selected from submitted abstracts. Poster presentations gave attendees the opportunity to discuss their ongoing work with a broad audience in line with the goal to bring together basic science with clinical and translational research issues.
About the IDSA and IDWeek
The Infectious Diseases Society of America (IDSA) is a community of over 12,000 physicians, scientists and public health experts who specialize in infectious diseases. Our mission is to improve the health of individuals, communities, and society by promoting excellence in patient care, education, research, public health, and prevention relating to infectious diseases. IDWeek is the joint annual meeting of the Infectious Diseases Society of America (IDSA), Society for Healthcare Epidemiology of America (SHEA), the HIV Medicine Association (HIVMA), the Pediatric Infectious Diseases Society (PIDS), and the Society of Infectious Diseases Pharmacists (SIDP). Over 9,500 participants attended this conference in October 2022.
About Clostridioides difficile Infection (CDI). According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long-term care facilities and in the community. C. difficile is one of the most common causes of health care- associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate of two of the three antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect. Beneficial effects of bile acids include a decrease in primary bile acids and an increase in secondary bile acids in patients with CDI, which was observed in the Company's Ph2a trial results and previously reported. (CID, 2022)
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates that target the DNA polymerase IIIC enzyme and its R&D pipeline includes antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
Acurx Pharma -- Pasteur Act -- >>> Bennet, Young, Bipartisan House Colleagues Reintroduce Bipartisan PASTEUR Act to Fight Antimicrobial Resistance
April 27, 2023
https://www.bennet.senate.gov/public/index.cfm/2023/4/bennet-young-bipartisan-house-colleagues-reintroduce-bipartisan-pasteur-act-to-fight-antimicrobial-resistance
Bipartisan, Bicameral Legislation Would Support Development of Innovative Antibiotics to Treat Resistant Infections and Improve Appropriate Antibiotic Use
Washington, D.C. — Today, Colorado U.S. Senator Michael Bennet and U.S. Senator Todd Young (R-Ind.), alongside U.S. Representatives Scott Peters (D-Calif.), Drew Ferguson (R-Ga.), Mike Levin (D-Calif.), and Jake LaTurner (R-Kan.) reintroduced the Pioneering Antimicrobial Subscriptions to End Upsurging Resistance (PASTEUR) Act to encourage innovative drug development targeting the most threatening infections, improve the appropriate use of antibiotics, and ensure domestic availability of antibiotics when needed.
“Right now, we don’t have the tools to address the threat posed by antimicrobial resistance – and infectious disease experts are warning us that it will only get worse,” said Bennet. “The bipartisan PASTEUR Act is the strongest bill ever written to strengthen antibiotic development and use. It will fix our market failures, expand the pipeline for next generation antibiotics, and save lives. We can’t sit on our hands as this public health crisis arrives – we have to act now.”
“Americans understand that we must take every reasonable and responsible measure to prevent future public health crises. Antimicrobial resistance has become a growing crisis in recent years. Market failures have resulted in a lack of needed research and development in this field which is a threat to public health. Our bill would incentivize the development of new innovative antibiotics and focus on educating health care providers on how to avoid overuse or misuse of these life-saving medications in order to slow the emergence of antibiotic-resistant pathogens,” said Young.
“Antimicrobial resistance poses a growing and significant threat to Americans’ health,” said Peters. “The PASTEUR Act will help us develop better antibiotics to counter resistant infections and help doctors ensure these drugs are used responsibly to stop the emergence of new superbugs. In the wake of the COVID-19 pandemic, we must do everything in our power to prevent the next public health crisis.”
“Antibiotics make modern medicine possible and the U.S. is at risk of losing these critical drugs. Antibiotic resistant infections are becoming more commonplace, and Congress must take action so that the foundation of modern medicine doesn’t crumble,” said Ferguson. “The PASTEUR Act brings together the public and private sectors to address these drug development market failures, increase public health preparedness, and help usher in a new era of antibiotic development. This essential legislation will also improve appropriate antibiotic use across the healthcare system while enhancing and safeguarding new antibiotic development. Simply put, we must act now to keep research and development from falling behind.”
“Each year in the United States, at least 2.8 million people become infected with pathogens that are resistant to treatment and for which advanced antimicrobials are needed. Unfortunately, as the COVID-19 pandemic made clear, our country needs stronger resources to develop those antimicrobials and prevent another global pandemic,” said Levin. “Our PASTEUR ACT empowers the Department of Health and Human Services to seek expertise on the development of antimicrobials and devise a plan to make them widely available. I thank Sen. Bennet and Rep. Ferguson for leading this bicameral, bipartisan legislation and look forward to it moving through the legislative process.”
“The COVID-19 pandemic reminded us how crucial it is for our nation to continue investing in healthcare research to prevent future public health emergencies,” said LaTurner. “America can't afford to be asleep at the wheel when it comes to the threat of antimicrobial resistance. That's why I'm proud to join my colleagues in introducing the bipartisan PASTEUR Act to bolster new antibiotic development and help medical professionals prevent the overuse of lifesaving drugs.”
According to the Centers for Disease Control and Prevention’s (CDC) Antibiotic Resistance Threats in the United States report, more than 2.8 million antibiotic-resistant infections occur in the United States each year, and at least 35,000 people die as a result. In March 2015, the U.S. National Action Plan for Combating Antibiotic-Resistant Bacteria directed federal agencies to accelerate a coordinated, full government response to antibiotic resistance and take action to expand the ability of our health care system to prevent, identify, and respond to the infection pandemic threat posed by antimicrobial resistance. Part of this plan was to increase and incent development of innovative antimicrobial drugs to treat resistant infections. Because of severe market failures in the health care system, many of the innovative antibiotic companies doing this work have filed for bankruptcy and stopped producing their critical drugs completely.
The PASTEUR Act would address this market failure and increase public health preparedness by keeping novel antibiotics on the market and improving appropriate use across the health care system. While current contracts between the government and drug makers base payment on volume, the PASTEUR Act would establish a subscription-style model which would offer antibiotic developers an upfront payment in exchange for access to their antibiotics, encouraging innovation and ensuring our health care system is prepared to treat resistant infections.
Statements of Support
“Millions will continue to die from resistant bacteria because we are out of treatment options. Antibiotics aren't working any more for most people who contract a superbug. The science is extraordinary, it's the business model that's broken. We desperately need a new way to pay for these drugs - antibiotics, antifungals, and phage therapy. The Pasteur Act is that rare, bipartisan idea that solves an incredible problem for an affordable price,” said Professor Kevin Outterson, Boston University.
“Antibiotics play a vital role in modern medicine, and we know that preserving access to these drugs is essential to any pandemic or public health emergency response. Yet the medicines that the U.S. relies on to treat serious infections have remained largely the same for nearly 40 years and are increasingly ineffective against quickly evolving bacteria. In 2023, the U.S. has already experienced several alarming antibiotic-resistant threats—and the emergence of new superbugs will continue and will only get worse. The bipartisan PASTEUR Act has the support of a diverse group of more than 230 public health and health care organizations, because it will help us fix the broken antibiotic drug pipeline and deliver important new therapies to physicians and the patients who need them. Reintroduction of the bill is an encouraging sign that policymakers remain committed to ensuring that lifesaving antibiotics are available when Americans need them most,” said David Hyun, director of The Pew Charitable Trusts’ Antibiotic Resistance Project.
"The need for legislative solutions to address the public health challenges posed by antimicrobial resistance (AMR) has been mounting for quite some time now, and we applaud the sponsors of the PASTEUR Act for their leadership. This bill will make new novel antibiotics a reality for patients and providers and fortify our healthcare system for future generations. AMR impacts us all and protection against the increasing threats of infection is not a partisan issue. We encourage broad support and quick passage of the PASTEUR Act,” said Candace DeMatteis, Vice President of Policy, Partnership to Fight Infectious Disease.
"For decades, we have seen antimicrobial resistance (AMR) soar around the world, while the pipeline for new treatments slows to a trickle due to the broken ecosystem for antimicrobial innovation. The PASTEUR Act is an integral solution to addressing the global public health crisis of AMR. The bipartisan bill will help repair the foundational challenges of the antimicrobial marketplace and drive the development of new, innovative treatments for patients,” said Rachel King, Interim President and CEO of the Biotechnology Innovation Organization.
“Infectious diseases physicians see firsthand the devastating impact of antimicrobial-resistant infections on our patients. We urgently need novel antimicrobials and investments in antimicrobial stewardship to preserve the efficacy of these precious drugs and optimize patient outcomes. The PASTEUR Act will deliver the tools we need to protect modern medicine and strengthen our preparedness for future emergencies,” said Carlos del Rio, MD, FIDSA, President, Infectious Diseases Society of America; and Interim Dean, Emory University School of Medicine.
“For people living with cystic fibrosis, difficult-to-treat infections are an unfortunate but common occurrence, and the fear of not having enough treatment options is an all too familiar concern. There is an urgent need to pass the PASTEUR Act to help ensure availability of novel antibiotics, not only for the CF community today, but for patients everywhere who could face a public health crisis tomorrow if Congress refuses to take action now,” said Mary Dwight, Senior Vice President and Chief Policy and Advocacy Officer, Cystic Fibrosis Foundation.
“Patients with drug resistant diseases are defenseless without new treatments, many of us are fighting rare diseases and we desperately need the treatments supported by the PASTEUR Act. PASTEUR is a bill for patients, and without it, too many of us will not survive our fight and those that do are facing a reduced quality of life. The new treatments created through the PASTEUR Act could one day cure me and others fighting disease with limited or no treatment options. Until then, I wake up every day hoping the medications available do not fail me again,” said Rob Purdie, Cofounder, MyCARE (MyCology Advocacy, Research & Education).
Specifically, the PASTEUR Act would:
Establish a subscription model to encourage innovative antimicrobial drug development aimed at treating drug-resistant infections. This model will be fully delinked, meaning that participating developers would not receive income, as a part of their subscription payments, based on volume or quantity of sales.
Subscription contracts would contain terms and conditions including product availability to individuals on a government health insurance plan, supporting appropriate use, and completion of postmarketing studies. These contracts could be valued between $750 million and $3 billion.
Build on existing frameworks to improve usage of the CDC National Healthcare Safety Network, the Emerging Infections Program, and other programs to collect and report on antibiotic use and resistance data.
Include transition measures such as smaller subscription contracts to support novel antimicrobial drug developers that need a financial lifeline.
Form a Committee on Critical Need Antimicrobials, consisting of representatives from federal agencies, doctors, patients, and outside experts, to develop and implement necessary guidance regarding infections of concern, and the favored characteristics of potential treatments.
Bennet and Young first introduced the PASTEUR Act in September 2020.
<<<
---
>>> Acurx Pharmaceuticals, Inc. Reports Second Quarter 2023 Results and Provides Business Update
August 14, 2023
https://finance.yahoo.com/news/acurx-pharmaceuticals-inc-reports-second-110100020.html
STATEN ISLAND, N.Y., Aug. 14, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of antibiotics for difficult-to-treat bacterial infections, announced today certain financial and operational results for the second quarter ended June 30, 2023.
Highlights of the second quarter ended June 30, 2023 include:
Acurx continues to enroll patients in its Phase 2b clinical trial, which includes 28 U.S. clinical trial sites, for patients with C. difficile infection (CDI);
The Phase 2b clinical trial will compare the efficacy of oral ibezapolstat, the Company's lead antibiotic candidate, to oral vancomycin, the current standard of care for patients with CDI;
Acurx anticipates completing enrollment of the 36 patients required for an interim review of the Phase 2b data by a newly appointed Independent Data Monitoring Committee (IDMC) in the coming months, with only 5 patients to enroll forward;
In April 2023 two presentations were made at the 33rd Annual European Congress of Clinical Microbiology and Infectious Disease (ECCMID) in Copenhagen. First, a scientific poster entitled "Novel Pharmacology and Susceptibility of Ibezapolstat Against C. difficile Isolates with Reduced Susceptibility to C. difficile-directed Antibiotics" was presented by Dr. Kevin Garey, Professor and Chair, University of Houston College of Pharmacy, and Principal Investigator for microbiome aspects of our ibezapolstat clinical trial program. Second, Acurx Executive Chairman, Bob DeLuccia, presented an update regarding the Company's preclinical, systemic oral and IV program for treatment of other gram-positive infections caused by MRSA, VRE and DRSP at the "Pipeline Corner" featured session at ECCMID, organized by Dr. Ursula Theuretzbacher, a world-renowned microbiology expert involved in antibacterial drug research, discovery and development strategies and policies for clinical and public health needs. These presentations are available on the Company's website at www.acurxpharma.com .
Acurx announced that it has been approved for presentations in 2H 2023 at two of the most prestigious scientific conferences in our sector; namely, the World Antimicrobial Resistance Conference (Philadelphia, PA) in September 2023 as well as at ID Week sponsored by the Infectious Disease Society of America (Boston, MA) in October 2023.
The Company is continuing its R&D collaboration with Leiden University Medical Center (Holland) under a previously awarded grant from the Dutch Government of approximately $500,000 USD to further evaluate the mechanism-of-action of Acurx's inhibitors against the DNA pol IIIC enzyme, which is the bacterial target of our antibiotic pipeline for the systemic treatment (IV and oral) of gram-positive bacterial infections;
The Company was notified by CARB-X that its application for a non-dilutive grant to fund its pre-clinical antibiotic candidate, ACX375C, was not approved. CARB-X noted that the 2023 round of funding was very competitive and that their Scientific Advisory Board was enthusiastic about pol IIIC as the bacterial target of our molecules and that the sufficiently good PK and safety properties of the compounds justified the proposed lead optimization plan. CARB-X encouraged us to re-apply for potential future requests for proposals or RFPs that CARB-X will continue to promulgate from time to time for CARB-X funding consideration.
Second Quarter 2023 Financial Results
Cash Position:
The Company ended the second quarter with cash totaling $9.1 million compared to $9.1 million as of December 31, 2022.
R&D Expenses:
Research and development expenses for the three months ended June 30, 2023 were $1.7 million compared to $0.9 million for the three months ended June 30, 2022. The increase was due to an increase in Phase 2b trial related costs. For the six months ended June 30, 2023 research and development expenses were $2.8 million compared to $1.7 million for the six months ended June 30, 2022. The increase is due primarily to an increase in Phase 2b trial related costs and an increase in consulting costs.
G&A Expenses:
General and administrative expenses for the three months ended June 30, 2023 were $1.7 million compared to $1.7 million for the three months ended June 30, 2022. Professional fees decreased by $0.1 million, offset by an increase of $0.1 million in employee related compensation costs. For the six months ended June 30, 2023, general and administrative expenses were $3.6 million compared to $3.6 million for the six months ended June 30, 2022. Professional fees decreased by $0.2 million, offset by an increase of $0.2 million in employee related compensation costs.
Net Income/Loss:
The Company reported a net loss of $3.4 million or $0.28 per diluted share for the three months ended June 30, 2023, compared to a net loss of $2.6 million or $0.26 per diluted share for the three months ended June 30, 2022, and a net loss of $6.3 million or $0.53 per diluted share for the six months ended June 30, 2023, compared to a net loss of $5.3 million or $0.52 per diluted share for the six months ended June 30, 2022 for the reasons previously mentioned.
Conference Call
As previously announced, David P. Luci, President and Chief Executive Officer, and Robert G. Shawah, Chief Financial Officer, will host a conference call to discuss the results and provide a business update as follows:
Date:
Monday, August 14, 2023
Time:
8:00 a.m. ET
Toll free (U.S. and International):
877-790-1503
Conference ID:
13740293
About the Ibezapolstat Phase 2 Clinical Trial
The completed multicenter, open-label single-arm segment (Phase 2a) study is now followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542). This Phase 2 clinical trial is designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI including pharmacokinetics and microbiome changes from baseline and continue to test for anti-recurrence microbiome properties seen in the Phase 2a trial, including the treatment-related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment. In the currently enrolling Phase 2b, trial segment, patients with CDI will be enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments will be identical in appearance, dosing times, and number of capsules administered to maintain the blind. This Phase 2 clinical trial will also evaluate pharmacokinetics (PK) and microbiome changes and continue to test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy. In the event non-inferiority of ibezapolstat to vancomycin is demonstrated, further analysis will be conducted to test for superiority.
Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect.
About Clostridioides difficile Infection (CDI)
According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long-term care facilities and in the community. C. difficile is one of the most common causes of health care- associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate of two of the three antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates that target the DNA polymerase IIIC enzyme and its R&D pipeline includes early-stage antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
Acurx CEO presentation from earlier in the year. This provides an excellent overview -
Acurx - >>> Discovery and development of DNA polymerase IIIC inhibitors to treat Gram-positive infections
Wei-Chu Xu 1, Michael H Silverman 2, Xiang Yang Yu 2, George Wright 2, Neal Brown 2
PMID: 31221610 DOI: 10.1016/j.bmc.2019.06.017
Abstract
Despite the growing global crisis caused by antimicrobial drug resistance among pathogenic bacteria, the number of new antibiotics, especially new chemical class of antibiotics under development is insufficient to tackle the problem. Our review focuses on an emerging class of antibacterial therapeutic agents that holds a completely novel mechanism of action, namely, inhibition of bacterial DNA polymerase IIIC. The recent entry of this new class into human trials may herald the introduction of novel drugs whose novel molecular target precludes cross-resistance with existing antibiotic classes. This review therefore examines the evolution of DNA pol IIIC inhibitors from the discovery of 6-(p-hydroxyphenylazo)uracil (HPUra) in the 1960s to the development of current first-in-class N7-substituted guanine drug candidate ACX-362E, now under clinical development for the treatment of Clostridioides difficile infection.
<<<
---
Acurx - >>> Here's Why We're Watching Acurx Pharmaceuticals' (NASDAQ:ACXP) Cash Burn Situation
Simply Wall St
October 8, 2023
https://finance.yahoo.com/news/heres-why-were-watching-acurx-140059801.html
There's no doubt that money can be made by owning shares of unprofitable businesses. For example, although software-as-a-service business Salesforce.com lost money for years while it grew recurring revenue, if you held shares since 2005, you'd have done very well indeed. But while history lauds those rare successes, those that fail are often forgotten; who remembers Pets.com?
So, the natural question for Acurx Pharmaceuticals (NASDAQ:ACXP) shareholders is whether they should be concerned by its rate of cash burn. For the purposes of this article, cash burn is the annual rate at which an unprofitable company spends cash to fund its growth; its negative free cash flow. Let's start with an examination of the business' cash, relative to its cash burn.
See our latest analysis for Acurx Pharmaceuticals
Does Acurx Pharmaceuticals Have A Long Cash Runway?
You can calculate a company's cash runway by dividing the amount of cash it has by the rate at which it is spending that cash. As at June 2023, Acurx Pharmaceuticals had cash of US$9.1m and no debt. Importantly, its cash burn was US$7.2m over the trailing twelve months. So it had a cash runway of approximately 15 months from June 2023. That's not too bad, but it's fair to say the end of the cash runway is in sight, unless cash burn reduces drastically. You can see how its cash balance has changed over time in the image below.
How Is Acurx Pharmaceuticals' Cash Burn Changing Over Time?
Acurx Pharmaceuticals didn't record any revenue over the last year, indicating that it's an early stage company still developing its business. Nonetheless, we can still examine its cash burn trajectory as part of our assessment of its cash burn situation. As it happens, the company's cash burn reduced by 10% over the last year, which suggests that management are maintaining a fairly steady rate of business development, albeit with a slight decrease in spending. Clearly, however, the crucial factor is whether the company will grow its business going forward. For that reason, it makes a lot of sense to take a look at our analyst forecasts for the company.
Can Acurx Pharmaceuticals Raise More Cash Easily?
Even though it has reduced its cash burn recently, shareholders should still consider how easy it would be for Acurx Pharmaceuticals to raise more cash in the future. Generally speaking, a listed business can raise new cash through issuing shares or taking on debt. Many companies end up issuing new shares to fund future growth. By looking at a company's cash burn relative to its market capitalisation, we gain insight on how much shareholders would be diluted if the company needed to raise enough cash to cover another year's cash burn.
Since it has a market capitalisation of US$25m, Acurx Pharmaceuticals' US$7.2m in cash burn equates to about 29% of its market value. That's fairly notable cash burn, so if the company had to sell shares to cover the cost of another year's operations, shareholders would suffer some costly dilution.
So, Should We Worry About Acurx Pharmaceuticals' Cash Burn?
On this analysis of Acurx Pharmaceuticals' cash burn, we think its cash runway was reassuring, while its cash burn relative to its market cap has us a bit worried. Even though we don't think it has a problem with its cash burn, the analysis we've done in this article does suggest that shareholders should give some careful thought to the potential cost of raising more money in the future. Separately, we looked at different risks affecting the company and spotted 6 warning signs for Acurx Pharmaceuticals (of which 2 can't be ignored!) you should know about.
<<<
---
>>> Acurx Announces Presentation and Update of Its pol IIIC R&D Pipeline at the World Antimicrobial Resistance Scientific Congress
PR Newswire
September 12, 2023
https://finance.yahoo.com/news/acurx-announces-presentation-pol-iiic-110100399.html
Ibezapolstat is currently enrolling in a Phase 2b trial for C. difficile infection in U.S. centers across the country and nearing its goal to reach a targeted 36 patients at which point an Interim Analysis of the unblinded primary clinical endpoint and safety data will be reviewed by an Independent Data Monitoring Committee
Ibezapolstat has received FDA QIDP and Fast-Track Designation
Also presented was an update on the Company's pre-clinical antibiotic program in Lead Optimization stage for systemic gram-positive bacterial infections, including Acute Bacterial Skin and Skin Structure Infections caused by MRSA
The company's preclinical pipeline also targets systemic infections caused by other gram-positive bacteria such as VRE and DRSP which are expected to be QIDP and Fast-Track eligible as product candidates advance in development
STATEN ISLAND, N.Y., Sept. 12, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of antibiotics for difficult-to-treat bacterial infections, today announced that a presentation was given by Acurx Executive Chairman, Robert J. DeLuccia, at the World Antimicrobial Resistance Scientific Congress on September 7, 2023. In his presentation at the Innovation Showcase session, he highlighted that the Company anticipates completing enrollment of the 36 patients required for an interim review of the Phase 2b data by the Independent Data Monitoring Committee (IDMC) in the coming months.
Mr. DeLuccia also presented an update on the Company's preclinical GPSS™ (Gram Positive Selective Spectrum) program for systemic oral and IV treatment of other gram-positive infections including MRSA, VRE and DRSP. Mr. DeLuccia summarized the progress stating that "Our potential lead compound meets Theurezbacher's criteria for antibiotic innovation in that it is a new chemical class, has novel mechanism and bacterial target, and has not shown cross-resistance in early in vitro microbiology studies." He further stated: "Having established clinical validation of the pol IIIC bacterial target in a Ph2a proof-of-principal trial showing 100% cure of C. difficile Infection, with no recurrence after 30 days' follow up, we have made substantial progress toward lead compound selection of our gram-positive IV and oral compounds. We've made significant improvements in invitro and invivo safety and have demonstrated oral and IV efficacy in a number of mouse infection models. Our current focus is to prioritize the oral form for acute bacterial skin and skin structure staph infections, including MRSA, to speed lead product selection and advancement to the clinic."
The presentation is available on the Company's website www.acurxpharma.com.
About Ibezapolstat
Ibezapolstat is a novel, orally administered antibiotic being developed as a Gram-Positive Selective Spectrum (GPSS™) antibacterial. It is the first of a new class of DNA polymerase IIIC inhibitors under development by Acurx to treat bacterial infections. Ibezapolstat's unique spectrum of activity, which includes C. difficile but spares other Firmicutes and the important Actinobacteria phyla, appears to contribute to the maintenance of a healthy gut microbiome.
The Company currently is enrolling patients in a Ph2b clinical trial of ibezapolstat to treat patients with C. difficile infection (CDI). The Company successfully completed Phase 1 and Phase 2a clinical trials of ibezapolstat. The Phase 2a trial demonstrated 100% clinical cure and 100% sustained clinical cure in patients with CDI, along with beneficial microbiome changes during treatment including overgrowth of Actinobacteria and Firmicutes phylum species while on therapy and new findings which demonstrate potentially beneficial effects on bile acid metabolism. The Ph2b clinical trial is designed to enroll 64 patients and is a randomized (1:1), non-inferiority, double-blind trial of oral ibezapolstat compared to oral vancomycin, a standard of care to treat CDI.
The FDA has accepted the Company's plan to have an Independent Data Monitoring Committee (IDMC) conduct an interim review of clinical outcome from the ongoing Ph2b clinical trial of patients with C. difficile Infection (CDI). The interim review will be conducted upon reaching enrollment of 36 patients in total. FDA's acceptance was based on the Company's filing of a protocol amendment to its Investigational New Drug Application (IND) with FDA in January 2023. The Company's filing and intention for the IDMC to conduct an interim review of data was based on the observed blinded data to date from the ongoing Ph2b clinical trial at that time. Upon conducting the interim review, the IDMC will determine and recommend to the Company whether the most appropriate course of action is to terminate the Ph2b clinical trial early due to success, as the Company had done with the Ph2a clinical trial, or to continue patient enrollment. The Company intends to report available data promptly after the IDMC conducts this interim review. The IDMC initial organizational meeting was conducted in March 2023 and it has completed all organizational matters required to ensure readiness for data review.
In June 2018, ibezapolstat was designated by the U.S. Food and Drug Administration (FDA) as a Qualified Infectious Disease Product (QIDP) for the treatment of patients with CDI and will be eligible to benefit from the incentives for the development of new antibiotics established under the Generating New Antibiotic Incentives Now (GAIN) Act. In January 2019, FDA granted "Fast Track" designation to ibezapolstat for the treatment of patients with CDI. The CDC has designated C. difficile as an urgent threat highlighting the need for new antibiotics to treat CDI.
About the World Antimicrobial Resistance Congress
Since 2015, the World Antimicrobial Resistance Congress has attracted top thought leaders, hospitals, companies, and policymakers as the annual, go-to event in the Antimicrobial Resistance (AMR) space. It has grown into the most impactful event in advancing solutions to combat current and future pressing global health crises. Diagnostic developers, antibiotic biotechs & pharmaceutical companies, stewardship technologies, access firms, and many more, rely on our event for business development opportunities, networking and showcasing of new products and solutions. Over 1,300 attendees were expected to attend with over 200 speakers presenting over the two-day conference held in Philadelphia, PA on September 7-8, 2023.
About Clostridioides difficile Infection (CDI). According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society or Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long-term care facilities and in the community. C. difficile is one of the most common causes of health care-associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate of two of the three antibiotics currently used to treat CDI is between 20% and 40% among approximately 150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect. Beneficial effects of bile acids include a decrease in primary bile acids and an increase in secondary bile acids in patients with CDI, which was observed in the Company's Ph2a trial results and previously reported. (CID, 2022)
About the Ibezapolstat Phase 2 Clinical Trial
The multicenter, open-label single-arm segment of this study (Phase 2a) is to be followed by a double- blind, randomized, active-controlled segment (Phase 2b) which, together, comprise the Phase 2 clinical trial. The Phase 2 clinical trial is designed to evaluate ibezapolstat in the treatment of CDI. Phase 2a of this trial is completed and was an open- label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment, the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study. Based on the recommendation of Acurx's Scientific Advisory Board (SAB) and Trial Oversight Committee, we terminated enrollment in Phase 2a early and are now advancing to Phase 2b. The SAB unanimously supported the early termination of the Phase 2a trial after 10 patients were enrolled in the trial instead of 20 patients as originally planned. The early termination was based on the evidence of meeting the primary and secondary endpoints of eliminating the infection (100%), with no recurrences of infection (100%), and with an acceptable adverse event profile. In the Phase 2b, approximately 64 additional patients with CDI will be enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments will be identical in appearance, dosing times, and number of capsules administered to maintain the blind. This Phase 2 clinical trial also will evaluate pharmacokinetics (PK) and microbiome changes and continue to test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy.
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates that target the DNA polymerase IIIC enzyme and its R&D pipeline includes antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
>>> Acurx Pharmaceuticals Announces Successful Completion and Early Discontinuation of the Ibezapolstat Phase 2b Trial for Treatment of C. difficile Infection
PR Newswire
October 2, 2023
https://finance.yahoo.com/news/acurx-pharmaceuticals-announces-successful-completion-110100751.html
Based on observed aggregate blinded data the Company has determined that both treatments, ibezapolstat and the control antibiotic vancomycin, have performed as expected
High rates of Clinical Cure were observed without any emerging safety concerns
Data will be analyzed and topline efficacy results will be reported as soon as possible
This successful milestone will allow advancement of this first-in-class, FDA QIDP/Fast Track-designated antibiotic candidate to Phase 3 clinical trials more expeditiously
STATEN ISLAND, N.Y., Oct. 2, 2023 /PRNewswire/ -- Acurx Pharmaceuticals, Inc. (NASDAQ: ACXP) ("Acurx" or the "Company"), a clinical stage biopharmaceutical company developing a new class of antibiotics for difficult-to-treat bacterial infections, announced today that the Company has discontinued the Phase 2b clinical trial of its lead antibiotic candidate, ibezapolstat, for the treatment of patients with Clostridioides difficile infection (CDI) due to success. The Company made this decision in consultation with its medical and scientific advisors and statisticians based on observed aggregate blinded data and other factors, including the cost to maintain clinical trial sites and slow enrollment due to COVID-19. The Company has determined that the trial performed as anticipated for both treatments, ibezapolstat and the control antibiotic vancomycin (a standard of care to treat patients with CDI), with high rates of clinical cure observed across the trial without any emerging safety concerns.
Accordingly, the Independent Data Monitoring Committee will not be required to perform an interim analysis of this Phase 2b trial data as originally planned and the Company has discontinued the trial. Acurx will analyze the data and report topline efficacy results promptly. The Company anticipates that this decision will allow the Company to advance this first-in-class, FDA QIDP/Fast Track-designated antibiotic product candidate to Phase 3 clinical trials more expeditiously.
Robert J. DeLuccia, Executive Chairman of Acurx, stated: "Considering the totality and weight of evidence of our preclinical, Phase 1 and Phase 2a clinical results and now with the observed aggregate blinded data, we determined it was in the best interests of the Company and its shareholders to discontinue the Phase 2b clinical trial early and prepare for Phase 3 clinical trials. Mr. DeLuccia stated further, "We look forward to compiling, analyzing the data and reporting topline results for the study's primary clinical endpoint and safety aspects as soon as possible". He further stated: "We thank the clinical trial investigators and patients across the country who participated in this study allowing advancement of this promising new antibiotic into late-stage clinical trials for this serious and life-threating infection which is classified by FDA and CDC as an urgent priority for which new classes of antibiotics are needed."
David P. Luci, the Company's President and Chief Executive Officer, stated: "We also look forward to reporting the full ibezapolstat data which will include the most extensive data for any antibiotic on sustained clinical cure to date in patients with CDI, as well as a comparison of the effect on the microbiome between oral ibezapolstat and oral vancomycin. We believe that, if approved by FDA for marketing, these attributes will support the use of ibezapolstat for front-line treatment of CDI."
About the Ibezapolstat Phase 2 Clinical Trial
The completed multicenter, open-label single-arm segment (Phase 2a) study was followed by a double-blind, randomized, active-controlled, non-inferiority, segment (Phase 2b) at 28 US clinical trial sites which together comprise the Phase 2 clinical trial (see https://clinicaltrials.gov/ct2/show/NCT04247542). This Phase 2 clinical trial was designed to evaluate the clinical efficacy of ibezapolstat in the treatment of CDI including pharmacokinetics and microbiome changes from baseline and continue to test for anti-recurrence microbiome properties seen in the Phase 2a trial, including the treatment-related changes in alpha diversity and bacterial abundance and effects on bile acid metabolism.
The completed Phase 2a segment of this trial was an open label cohort of up to 20 subjects from study centers in the United States. In this cohort, 10 patients with diarrhea caused by C. difficile were treated with ibezapolstat 450 mg orally, twice daily for 10 days. All patients were followed for recurrence for 28± 2 days. Per protocol, after 10 patients of the projected 20 Phase 2a patients completed treatment (100% cured infection at End of Treatment), the Trial Oversight Committee assessed the safety and tolerability and made its recommendation regarding early termination of the Phase 2a study and advancement to the Ph2b segment. In the now discontinued Phase 2b trial segment, 32 patients with CDI were enrolled and randomized in a 1:1 ratio to either ibezapolstat 450 mg every 12 hours or vancomycin 125 mg orally every 6 hours, in each case, for 10 days and followed for 28 ± 2 days following the end of treatment for recurrence of CDI. The two treatments were identical in appearance, dosing times, and number of capsules administered to maintain the blind. This Phase 2 clinical trial will also evaluate pharmacokinetics (PK) and microbiome changes and test for anti-recurrence microbiome properties, including the change from baseline in alpha diversity and bacterial abundance, especially overgrowth of healthy gut microbiota Actinobacteria and Firmicute phylum species during and after therapy. In the event non-inferiority of ibezapolstat to vancomycin is demonstrated, further analysis will be conducted to test for superiority.
Phase 2a data demonstrated complete eradication of colonic C. difficile by day three of treatment with ibezapolstat as well as the observed overgrowth of healthy gut microbiota, Actinobacteria and Firmicute phyla species, during and after therapy. Very importantly, emerging data show an increased concentration of secondary bile acids during and following ibezapolstat therapy which is known to correlate with colonization resistance against C. difficile. A decrease in primary bile acids and the favorable increase in the ratio of secondary-to-primary bile acids suggest that ibezapolstat may reduce the likelihood of CDI recurrence when compared to vancomycin
About the Microbiome in Clostridioides difficile Infection (CDI) and Bile Acid Metabolism
C. difficile can be a normal component of the healthy gut microbiome, but when the microbiome is thrown out of balance, the C. difficile can thrive and cause an infection. After colonization with C. difficile, the organism produces and releases the main virulence factors, the two large clostridial toxins A (TcdA) and B (TcdB). (Kachrimanidou, Microorganisms 2020, 8, 200; doi:10.3390/microorganisms8020200.) TcdA and TcdB are exotoxins that bind to human intestinal epithelial cells and are responsible for inflammation, fluid and mucous secretion, as well as damage to the intestinal mucosa.
Bile acids perform many functional roles in the GI tract, with one of the most important being maintenance of a healthy microbiome by inhibiting C. difficile growth. Primary bile acids, which are secreted by the liver into the intestines, promote germination of C. difficile spores and thereby increase the risk of recurrent CDI after successful treatment of an initial episode. On the other hand, secondary bile acids, which are produced by normal gut microbiota through metabolism of primary bile acids, do not induce C. difficile sporulation and therefore protect against recurrent disease. Since ibezapolstat treatment leads to minimal disruption of the gut microbiome, bacterial production of secondary bile acids continues which may contribute to an anti-recurrence effect.
About Clostridioides difficile Infection (CDI)
According to the 2017 Update (published February 2018) of the Clinical Practice Guidelines for C. difficile Infection by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA), CDI remains a significant medical problem in hospitals, in long- term care facilities and in the community. C. difficile is one of the most common causes of health care- associated infections in U.S. hospitals (Lessa, et al, 2015, New England Journal of Medicine). Recent estimates suggest C. difficile approaches 500,000 infections annually in the U.S. and is associated with approximately 20,000 deaths annually. (Guh, 2020, New England Journal of Medicine). Based on internal estimates, the recurrence rate of two of the three antibiotics currently used to treat CDI is between 20% and 40% among approximately150,000 patients treated. We believe the annual incidence of CDI in the U.S. approaches 600,000 infections and a mortality rate of approximately 9.3%.
About Acurx Pharmaceuticals, Inc.
Acurx Pharmaceuticals is a clinical stage biopharmaceutical company focused on developing new antibiotics for difficult to treat infections. The Company's approach is to develop antibiotic candidates that target the DNA polymerase IIIC enzyme, and its R&D pipeline includes early-stage antibiotic product candidates that target Gram-positive bacteria, including Clostridioides difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant Enterococcus (VRE) and drug-resistant Streptococcus pneumoniae (DRSP).
<<<
---
>>> Being vegetarian may be partly determined by your genes
New Scientist
by Clare Wilson
https://www.msn.com/en-us/health/other/being-vegetarian-may-be-partly-determined-by-your-genes/ar-AA1hHHA2?OCID=ansmsnnews11
People’s likelihood of being vegetarian appears to be influenced by several genetic variants, and two of the three most important genes found so far seem to be involved in fat metabolism.
This hints that some people find it easier to give up meat because they naturally produce certain fat molecules, says Nabeel Yaseen at Northwestern University in Chicago. “Maybe there’s some fat that’s essential for some people to have in their diet but not for others,” he says.
About 5 per cent of people in the UK and US avoid all meat and fish. But surveys indicate that some people who say they are vegetarian still eat meat sometimes. That suggests that some people would like to be vegetarian but find it too hard, says Yaseen.
Genetic factors are known to influence other aspects of diet, such as whether people like coffee or alcohol. To see if genes also affect vegetarianism, Yaseen and his team turned to the UK Biobank, a large study where people filled in lifestyle and medical surveys and had their DNA sequenced. They analysed about 5300 people who said they were strict vegetarians and another 330,000 people who were meat eaters.
Three gene variants were more common in vegetarians. Two, called NPC1 and RMC1, are involved in the transport and metabolism of cholesterol and other fatty molecules called glycolipids. The third gene, called RIOK3, has various functions, including affecting the immune system.
It isn't known exactly how these genes could relate to vegetarianism. But one of the chief differences between animal-based foods and plant-based ones is the chemical make-up of their fats or oils, collectively known as lipids. Yaseen and his colleagues speculate that some people may function better on a vegetarian diet because they are more able to synthesise certain lipid molecules that are present in meat.
People who try vegetarianism but give up may be doing so because the body becomes deficient in the postulated essential lipids, says Yaseen. “They decide that this diet is not for them or gradually creep back into an omnivore diet. Some people might think they just don't have the willpower.”
Yaseen says, however, that another possibility is that the apparently vegetarianism-promoting gene variants affect people’s taste. “A lot of information about genes is yet to be known,” he says.
Albert Koulman at the University of Cambridge says most research into how food nutrients influence satiety and food choices has focused on proteins rather than fats. “We don’t know enough about [this idea] to either accept or dismiss it,” he says.
Richard McIlwain at the UK Vegetarian Society says the number of vegetarians has been rising in recent years, almost doubling in the UK between 2012 and 2019. “That would seem to suggest something other than underlying genetic factors are at play,” he says.
“People go vegetarian because, more and more, they are concerned about climate, about animal welfare or about their health. Psychological factors, such as tradition, education and awareness of animal suffering in food production, and ‘taste preferences’ are far more important determinants of vegetarianism than any physiological factors,” says McIlwain.
<<<
---
CRDF reports 2nd quarter results. Much expanded relationship with PFE. Stock up 22% in AH.
- Advance to first-line RAS-mutated mCRC follows the strong signal from new clinical and preclinical data, and agreement with FDA -
- First-line mCRC represents substantial increase in patient impact and market opportunity over second-line -
- Pfizer Ignite will be responsible for the clinical execution of new first-line mCRC trial with interim topline data expected in mid-2024 -
- Cash position on June 30, 2023 was $89.4 million; sufficient to fund operations into 2025 and through interim topline results from mCRC trial -
- Company will hold a conference call today at 5:00 p.m. ET/2:00 p.m. PT -
SAN DIEGO, Aug. 7, 2023 /PRNewswire/ -- Cardiff Oncology, Inc. (Nasdaq: CRDF), a clinical-stage biotechnology company leveraging PLK1 inhibition, a well-validated oncology drug target, to develop novel therapies across a range of cancers, today announced plans to advance the company's lead program to the first-line setting of metastatic colorectal cancer (mCRC) and conduct its new CRDF-004 trial with study execution support from Pfizer Ignite, a new end-to-end service for biotech companies.
Cardiff Oncology is a clinical-stage biotechnology company leveraging PLK1 inhibition to develop novel therapies across a range of cancers. Our lead asset is onvansertib, a PLK1 inhibitor we are evaluating in combination with standard-of-care (SoC) therapeutics in clinical programs targeting indications such as KRAS/NRAS-mutated metastatic colorectal cancer (mCRC) and metastatic pancreatic ductal adenocarcinoma (mPDAC), as well as in investigator-initiated trials in triple negative breast cancer (PRNewsfoto/Cardiff Oncology, Inc.)
Cardiff Oncology is a clinical-stage biotechnology company leveraging PLK1 inhibition to develop novel therapies across a range of cancers. Our lead asset is onvansertib, a PLK1 inhibitor we are evaluating in combination with standard-of-care (SoC) therapeutics in clinical programs targeting indications such as KRAS/NRAS-mutated metastatic colorectal cancer (mCRC) and metastatic pancreatic ductal adenocarcinoma (mPDAC), as well as in investigator-initiated trials in triple negative breast cancer (PRNewsfoto/Cardiff Oncology, Inc.)More
"Our advance to the first-line mCRC setting is the result of a comprehensive data-driven review coupled with the agreement and support of the FDA. Ultimately, this decision moves Cardiff Oncology into a stronger position to realize the promise of onvansertib for the benefit of patients and all of our stakeholders," said Mark Erlander, Ph.D., Chief Executive Officer of Cardiff Oncology. "We are delighted to expand our relationship with Pfizer and conduct this new first-line trial beginning this fall through Pfizer Ignite, leveraging its clinical execution capabilities and expertise."
The company estimates that there are 48,000 new patients in the U.S. annually in the first-line RAS-mutated mCRC setting for whom there are no ongoing clinical trials and no new treatments approved in the past 20 years.
Dr. Erlander continued: "Key to today's decision has been our discovery of a novel mechanism of action by which onvansertib inhibits angiogenesis by turning off a 'survival switch' for tumorigenesis. This has helped us understand onvansertib's interaction with bevacizumab, and the compelling clinical results we observed in our Phase 1b/2 second-line KRAS-mutated mCRC trial."
The clinical activities of the company's new CRDF-004 trial in first-line RAS-mutated mCRC will be conducted with support from Pfizer Ignite. This expands the relationship established in November 2021 when Pfizer made an equity investment in Cardiff Oncology and nominated Adam Schayowitz, Ph.D., Vice President & Medicine Team Group Lead for Breast Cancer, Colorectal Cancer and Melanoma, Pfizer Global Product Development as a Scientific Advisory Board member.
Pfizer Ignite is a new end-to-end service for biotech companies with high potential science that leverages Pfizer Inc.'s significant R&D capabilities, scale and expertise to accelerate the development of breakthrough therapies.
Cardiff Oncology will maintain full economic ownership and control of onvansertib.
"We believe onvansertib, by inhibiting PLK1, has the potential to play a meaningful role in the treatment of several types of cancer, including the lead program in RAS-mutated mCRC," said Dr. Schayowitz. "We believe that by combining Pfizer's clinical development capabilities and expertise, with onvansertib's promising novel clinical findings, we have an opportunity to accelerate the advancement of this program for the benefit of the many patients in the RAS-mutated mCRC setting."
Cardiff Oncology's new lead program in first-line RAS-mutated mCRC will consist of two trials that will be conducted sequentially. The first trial will be CRDF-004, a Phase 2 randomized trial generating preliminary safety and efficacy data and evaluating two different doses of onvansertib to confirm an optimal dose. Onvansertib will be added to standard-of-care consisting of FOLFIRI plus bevacizumab, or FOLFOX plus bevacizumab. A total of 90 patients will be randomized in a 1:1:1 ratio to either 20mg of onvansertib plus standard-of-care, 30mg of onvansertib plus standard-of-care, or standard-of-care alone. Interim topline results from this trial are expected in mid-2024.
Contingent upon the results of CRDF-004, Cardiff Oncology will initiate a Phase 3, randomized trial with registrational intent. The FDA has agreed that a seamless trial with objective response rate (ORR) at an interim point is an acceptable endpoint to pursue accelerated approval, with progression-free survival (PFS) and trend in overall survival being the endpoints for full approval.
"The stand-out results from our Phase 1b/2 second-line mCRC trial of onvansertib were observed in a well-defined subset of patients, namely those who had not previously been treated with bevacizumab in the first-line setting," said Fairooz Kabbinavar, MD, Chief Medical Officer of Cardiff Oncology. "Bev naïve patients in our Phase 1b/2 trial who received FOLFIRI, bevacizumab and onvansertib had a remarkable 73% ORR and 15-month mPFS, comparing favorably against historical controls that report an ORR of approximately 25% with a 7 to 8-month mPFS. Such high levels of efficacy have not been previously observed in 2nd line mCRC. The clinical and preclinical data we are reporting today confirm our initial finding, and based on highly encouraging interactions with the FDA and Pfizer, we are moving into first-line RAS-mutated mCRC where we believe enrollment should occur more quickly given the significantly larger number of first-line patients versus second-line."
Consistent with the strategic decision to focus on first-line RAS-mutated mCRC, Cardiff Oncology will discontinue enrollment in its ONSEMBLE second-line trial to focus resources on its new lead first-line program. This decision is driven by the fact that both trials essentially test the same clinical hypothesis, the importance of deploying the Company's capital efficiently, and the FDA's suggestion that Cardiff Oncology consider focusing on the first-line RAS-mutated mCRC setting.
All other Cardiff Oncology programs remain unaffected by this decision.
Conference Call and Webcast
Cardiff Oncology will host a corresponding conference call and live webcast at 5:00 p.m. ET/2:00 p.m. PT on August 7, 2023. Individuals interested in listening to the live conference call may do so by using the webcast link in the "Investors" section of the company's website at www.cardiffoncology.com. A webcast replay will be available in the investor relations section on the company's website for 30 days following the completion of the call.
About Cardiff Oncology, Inc.
Cardiff Oncology is a clinical-stage biotechnology company leveraging PLK1 inhibition, a well-validated oncology drug target, to develop novel therapies across a range of cancers. The company's lead asset is onvansertib, a PLK1 inhibitor being evaluated in combination with standard-of-care (SoC) therapeutics in clinical programs targeting indications such as RAS-mutated metastatic colorectal cancer (mCRC) and metastatic pancreatic ductal adenocarcinoma (mPDAC), as well as in investigator-initiated trials in triple negative breast cancer (TNBC) and small cell lung cancer (SCLC). These programs and the company's broader development strategy are designed to target tumor vulnerabilities in order to overcome treatment resistance and deliver superior clinical benefit compared to the SoC alone. For more information, please visit https://www.cardiffoncology.com.
Bladerunner
>>> Vistagen Therapeutics, Inc. (VTGN), a late clinical-stage biopharmaceutical company, primarily focus to transform the treatment landscape for individuals living with anxiety, depression, and other central nervous system (CNS) disorders. The company's pipeline includes six clinical stage product candidates, including five investigational agents belonging to drugs known as pherines. Its product pipeline comprises PH94B, a fasedienol nasal spray, which is in Phase III development for the treatment of social anxiety disorder; and PH10, a Ituvone nasal spray which is in Phase II development for the treatment of major depressive disorder. In addition, the company is also developing PH15, an early-stage investigational synthetic neuroactive steroid for the treatment of cognition improvement; PH80, an odorless and tasteless synthetic investigational pherine for the treatment of menopausal hot flashes and migraine; PH284, an early-stage investigational synthetic neuroactive steroid for the treatment of wasting syndrome Cachexia; and AV-101, an oral nmdr glycine site antagonist for depression and neurological disorders. Further, it has a license and collaboration agreement with EverInsight Therapeutics Inc. to develop and commercialize to address ophthalmologic and CNS disorders. The company was founded in 1998 and is headquartered in South San Francisco, California. <<<
---
Blade, Instead of Ozempic for weight loss, you should check out cardiologist Dr. Steve Gundry (link below). Using his approach, I lost over 50 lbs (from 260 down to 205), and have easily kept it off, just by changing the types of foods consumed and by shortening the 'eating window' down to 8 hours/day (which replicates the natural eating cycle).
Gundry explains why most Americans are 'metabolically inflexible', and literally cannot burn fat at the mitochondrial level. He has numerous best selling books dealing with the various aspects, but following the info in his videos is sufficient to get the basic idea. It's a variation on the Mediterranean diet, but you need to avoid most grains (wheat, corn, soy, etc) since they contain 'lectins' (which cause leaky gut), and avoid processed foods, junk food, etc.
The ancient grains like sorghum and millet are fine, and beans are good but must be pressure cooked. Also lean meats and lots of organic vegetables. On the vegetable side, there are some to be avoided due to their lectin content, but most are fine. Peanuts and cashew are to be avoided (extremely high in lectins), but true nuts like pistachios and walnuts are great. Fruit intake is limited to in season (late summer, fall), although fruits like kiwis and avocados are good to eat all year.
Anyway, check out Gundry. Not only did I easily lose 50 lbs (less than 1 year), but my high blood pressure returned to normal, and autoimmune problems (primarily related to leaky gut) disappeared. Most of Gundry's diabetes and autoimmune patients (thousands) have been able to go off their meds, as the source of the immune system overactivity (leaky gut) is resolved.
A big key in Gundry's approach is reversing 'leaky gut' (intestinal hyperpermeability) and rebuilding the person's intestinal microbiome, which is extremely critical for good health.. Between over usage of antibiotics in medicine, and glyphosate herbicide residues on wheat, corn, the average American's gut microbiome has been absolutely devastated. Glyphosate / Roundup kills the intestinal flora by blocking the shikimate pathway, which is the same pathway associated with its weed killing ability. Glyphosate was originally patented by Monsanto as an antibiotic.
Gundry also has a lot to say about heart disease and cancer. Check him out -
>>> Moolec Science Presents 'Piggy Sooy', a Soybean Platform That Can Produce Significantly High Amounts of Pork Proteins
Accesswire
Moolec Science SA
June 26, 2023
https://finance.yahoo.com/news/moolec-science-presents-piggy-sooy-123000813.html
LUXEMBOURG / ACCESSWIRE / June 26, 2023 / Moolec Science SA ((NASDAQ:MLEC) "Company"; "Moolec"), a science-based food ingredient company focused on producing animal proteins in plants through Molecular Farming technology, announced today an outstanding achievement in its Meat Replacements Program for the Soybean platform, as its new "Piggy Sooy" produced a significantly high amount of pork protein.
Piggy Sooy

Piggy Sooy
The animal protein reached a high expression level up to 26.6% of total soluble protein in soy seeds, 4x higher than initially projected by the Company. The result can be directly observed due to the pink color of Moolec's soybeans, the same color as the pig (access the picture by clicking here). After this achievement, the Company's soybean platform was renamed "Piggy Sooy".
The breakthrough accomplishment has led Moolec to file a new patent utilizing a novel approach aiming to provide the Company with a frictionless regulatory pathway going forward.
Moolec's CEO & Co-Founder Gastón Paladini said: "Piggy Sooy represents tangible and visual proof that Moolec's technology has the capacity to achieve significant yields in plants to produce meat proteins. With this groundbreaking achievement, Moolec consolidates its position as a category creator and a pioneer in Molecular Farming for the food industry. Our plant biology team is writing the history of science in food, I couldn't be prouder of them."
This scientific milestone consolidates the Molecular Farming path as one of the most valuable alternative technologies to produce animal proteins, given that plants can function as animal protein factories in a more efficient manner than initially expected. This enhanced efficiency of plants has the potential to improve the economics of the Company's business model.
Moolec Science is producing several meat proteins in plants as functional ingredients to improve the taste, appearance, texture, and nutrition of meat alternatives. Due to its enhanced functionality and final application, the Company also highlighted that these food ingredients could also be potentially commercialized within the ~$600 billion traditional processing meat industry.
Amit Dhingra, Ph.D., Chief Science Officer of Moolec said: "This achievement opens up a precedent for the entire scientific community that is looking to achieve high levels of protein expression in seeds via Molecular Farming." He further emphasized: "Moolec has developed a unique, successful, and patentable platform for the expression of highly valuable proteins in the seeds of economically important crops such as soybeans. This platform has the potential to be used across a wide variety of proteins of interest for a broad range of industries, such as the pharma, cosmetic, diagnostic reagents, and other food industries."
About Moolec Science SA
Moolec is a science-based food ingredient company focused on producing animal proteins in plants through Molecular Farming, a disruptive technology in the alternative protein landscape. Its purpose is to upgrade the taste, nutrition, and affordability of alternative protein products while building a more sustainable and equitable food system. The Company's technological approach aims to have the cost structure of plant-based solutions with the organoleptic properties and functionality of animal-based ones. Moolec's technology has been under development for more than a decade and is known for pioneering the production of a bovine protein in a crop for the food industry. The Company's product portfolio and pipeline leverages the agronomic efficiency of broadly used target crops, like safflower, soybean, and pea. Moolec has a growing international patent portfolio (24, both granted and pending) for its Molecular Farming technology. The Company is run by a diverse team of Ph.Ds and Food Insiders, and operates in the United States, Europe, and South America. For more information, visit moolecscience.com.
<<<
---
OCGN getting a nudge. Corp deck https://ocugen.gcs-web.com/static-files/e4bb4434-d94d-4d09-b119-ccfcb30af927
Candida Auris - >>> A deadly fungal infection is spreading in hospitals. Here's what to know.
by Jennifer Hassan and Fenit Nirappil
The Washington Post
March 21, 2023
https://www.yahoo.com/news/deadly-fungal-infection-spreading-hospitals-164045552.html
A deadly fungal infection is spreading at "an alarming rate" inside health facilities and long-term-care hospitals across the United States, the Centers for Disease Control and Prevention said Monday.
Here's what to know about the highly drug-resistant fungus - a strain of a kind of yeast known as Candida auris (or C. auris for short) that the CDC says "presents a serious global health threat."
While healthy people are not likely to contract the infection, those with lower immunity and people living in nursing homes are more likely to fall sick and be unable to fight the infection, and the outcome can be fatal.
What is Candida auris?
Candida is a family of yeasts that can be found on the skin and inside the body. Usually, the fungus lives in areas such as the mouth, throat, gut and vagina, without causing health problems. Common types of Candida include "Candida albicans," which causes the yeast infection thrush.
Sometimes, however, certain types of Candida can cause infection in older people and those battling other health issues.
C. auris was first discovered in the ear canal of a patient in Tokyo in 2009. It can enter the body during medical treatment, including operations or when urinary catheters, tubes or drips are inserted. It can also infect surgical wounds.
While the number of cases recorded by the CDC is only in the thousands, it is considered a serious global public health threat because it is drug resistant and has the potential to spread among the most medically vulnerable. It is also difficult to identify using standard laboratory methods, and it has caused outbreaks in health-care environments across the country.
What are the symptoms?
Candida auris can enter the bloodstream and spread throughout the body, causing invasive infections. Fever and chills that don't improve after antibiotic treatment for suspected bacterial infections are the most common symptoms of invasive candidiasis, the CDC notes.
The infection can aggravate various parts of the body, including the ears, heart, kidneys, eyes and brain.
How is the fungus transmitted?
The infection spreads easily from person to person - especially within hospital environments and among the medically vulnerable. It can also be spread through contact with infected surfaces and lingers on objects including hospital equipment such as bed rails, chairs and windowsills.
The infection can also be resistant to certain cleaning products, making it harder for the fungus to be eradicated from hospital wards.
People can also carry the infection without experiencing symptoms, unknowingly spreading the infection to other people who may be more at risk, such as people in long-term health-care facilities, including nursing home patients on ventilators, those with diabetes and cancer patients.
Those who take lots of antibiotics or antifungal medications appear to be at highest risk of infection, according to the CDC.
Between 30 percent and 60 percent of hospitalized people who develop bloodstream infections are estimated to die, according to CDC data. However, the CDC notes that many of these patients had other serious illnesses that increased their risk of death.
The CDC says more work is needed to understand how the infection spreads.
Which U.S. states has the fungus been identified in so far?
C. auris was diagnosed in a handful of patients in the United States in 2016, where clinicians on American soil had been warned by health officials to be on the lookout for the infection.
It has since been detected in more than 20 states, with the most cases recorded in Nevada, California, Florida New York, Illinois and Texas in the past 12 months, according to case counts provided to the CDC by local and state health departments.
Fungal infections from Candida auris tripled nationally from 476 in 2019 to 1,471 in 2021, the CDC said Monday.
Where else in the world have outbreaks been reported?
Since it was first reported in Asia in 2009, the fungus has been reported in a slew of countries, including Colombia, India, Israel, Kenya, Kuwait, Pakistan, South Korea, Venezuela and Britain.
Transmission of the infection was facilitated by international travel, according to the CDC, though experts say that climate change may also be fueling the infection. The infection also worsened amid the coronavirus pandemic, which hospitalized more people around the world, overburdening health-care staff who were forced to reuse personal protective equipment.
Clinical cases of C. auris soared about 60 percent in 2020 compared to 2019, according to a 2022 report from the CDC that noted the global pandemic "likely intensified spread of C. auris and hindered detection of additional cases."
One leading theory suggests that Candida has evolved to survive in a warming world, while other theories suggest that widespread use of antifungal drugs along with heavy use of fungicide on crops may have sparked the emergence of the fungus.
How can the spread be prevented?
The CDC says that several infection control measures can help prevent C. auris, including adherence to hand hygiene and thoroughly cleaning hospital environments. Equipment that is shared among patients, such as blood pressure cuffs, temperature probes and ultrasound machines, should be thoroughly disinfected frequently.
Alcohol-based hand sanitizer, gowns and gloves should also be used to reduce the spread of infection inside health-care facilities.
According to the CDC, those who have the infection and their close contacts do not have to self-isolate and can participate in social activities as long as they maintain good hand hygiene.
How is the infection diagnosed and treated?
The infection is difficult to identify with standard laboratory methods because the yeast can be mistaken for other organisms, so specific technology is needed, the CDC says.
While the CDC says most C. auris infections are treatable with antifungal medications called echinocandins, some strains of the infection have developed a resistance to antifungal drugs, making it harder to treat patients, and cases of reinfection have also been recorded.
<<<
---
>>> Researchers discover way to combat superfungi
The News Glory
06/07/2023
https://thenewsglory.com/researchers-discover-way-to-combat-superfungi/
Researchers discover way to combat superfungi
A study conducted at USP reveals that brilacidin, a new drug tested for illnesses ranging from bacterial skin infections to Covid-19, can kill resistant strains of fungi when combined with two classes of antifungals available on the market.
The new potential application of the medicine, now patented and described in the journal Nature Communications, was discovered by researchers from the Faculty of Pharmaceutical Sciences of Ribeirão Preto (FCFRP-USP), in the interior of São Paulo, supported by FAPESP.
The problem of drug resistance is a challenge recognized by the WHO (World Health Organization), but the process of developing a new drug is very expensive and time-consuming.
“For this reason, we sought to identify the antifungal activity of chemical molecules that were already known, but which until then had not been studied in terms of their effects on controlling fungal growth. In this case, we started by exploring 1,400 chemical compounds until we arrived at this one”, says Thaila Fernanda dos Reis, postdoctoral fellow at FCFRP-USP and first author of the article.
Thanks to the use of different methods, the researchers concluded that the combination of brilacidin with two different antifungal drugs (caspofungin or voriconazole) has the ability to kill resistant strains of several species of fungus that cause infections in humans, such as Aspergillus fumigatus, the causative agent of invasive pulmonary aspergillosis.
Aspergillosis is a common infection in patients admitted to intensive care units (ICUs), which can lead to death in between 60% and 90% of individuals. It also affects patients with a certain degree of immune impairment, such as those undergoing cancer treatments.
In addition to combinations with antifungals for lung infections, brilacidin alone blocked the growth of the A. fumigatus and disease development in an animal model of keratitis, an infection that affects the cornea.
The eye disease affects 1 to 2 million people a year worldwide, especially in tropical countries with great agricultural activity. In the United States and other developed countries, the use of contact lenses contaminated with mold is the main risk factor.
Mechanism of action
Drug resistance occurs when the microorganism (fungus, bacteria or virus) finds a way to survive and continue to multiply even in the presence of the drug that should have stopped its growth.
Therefore, it is important to have drug options that act in different ways on the pathogen, in order to eradicate the infection even when the strain is resistant to some drug. However, while there are nine classes of antibacterials, there are only four classes of antifungals commercially available.
Caspofungin, for example, is an antifungal that has been available on the market for a long time. Its mechanism of action consists of inhibiting the synthesis of the cell wall, a structure that surrounds the plasma membrane and maintains the integrity of the fungal cell.
When in contact with the drug, however, not infrequently, the fungus activates a repair system, which bypasses the action of the drug and allows it to survive in its presence. Hence the potential of the combination of caspofungin and brilacidin. In tests, the presence of the new molecule disabled the repair system triggered by caspofungin.
“Caspofungin does not kill the fungus A. fumigatus, but hinders its multiplication. This is often enough for the host’s immune system to control the infection, but not always. That is why it is important to identify drugs capable of acting in synergy with One of the options would be to create a single drug that combined caspofungin and brilacidin simultaneously, so that they could act together”, summarizes Gustavo Henrique Goldman, a professor at FCFRP-USP who coordinated the study.
Superfungi
Another advantage of brilacidin is that the combination with caspofungin or voriconazole had action against different species of fungus.
In tests with animal models, in addition to A. fumigatus, the combination of brilacidin with caspofungin was effective in inhibiting other fungal species such as Candida albicans, Candida auris and Cryptococcus neoformans.
Called “superfungi” because of their high drug resistance, some of these strains have been blamed for serious nosocomial infections. Recently, they have become more common due to the large number of hospitalizations in ICUs due to the COVID-19 pandemic.
The synergistic action of brilacidin with voriconazole, in turn, was effective both against A. fumigatus and against Mucorales, a fungus that occurs mainly in India and Pakistan and causes serious deformations of the face.
For the effects to be proven in humans, however, clinical trials are needed. Together with the company that owns the brilacidin patent, the North American Innovation Pharmaceuticals Incorporated, the researchers are now looking for a Brazilian company that can license the medicine in the country and carry out the clinical tests, necessary to prove the effects in humans and, in case of successfully make the drug available on the market.
<<<
---
>>> Axsome aims to give its balance sheet a boost
https://www.fool.com/investing/2023/06/28/as-the-bull-roars-shareholders-hate-how-these-2-co/?source=eptyholnk0000202&utm_source=yahoo-host&utm_medium=feed&utm_campaign=article
Shares of Axsome Therapeutics were down 9% early Wednesday morning. The biopharmaceutical company specializing in treatments for central nervous system disorders announced that it would sell shares of common stock.
Axsome's motivation is clear. As a biotech with a pipeline of candidate drugs, Axsome needs cash in order to run late-stage clinical trials on its most promising treatments, and the proceeds from the offering will go toward both existing and potential new clinical programs in the future. In addition, Axsome will need capital to expand its commercialization efforts for its already-approved Sunosi drug for sleep disorders and its antidepressant drug Auvelity.
The filed prospectus did not indicate a specific number of shares that Axsome intends to sell. However, the offering will fall under an existing shelf registration statement.
In the past couple of weeks, Axsome has told investors that it expects its top treatments to generate as much as $11.5 billion in revenue in the U.S. market at peak sales, which dramatically exceeds what most analysts following the stock had projected. Moreover, if Auvelity pans out as a possible treatment that could help Alzheimer's disease as well as those looking to quit smoking, then it could dramatically expand its addressable market and add to Axsome's overall success.
<<<
---
>>> Has the Serotonin Hypothesis Been Debunked?
Not really. It never meant anything.
Psychology Today
Nassir Ghaemi M.D., M.P.H.
October 1, 2022 |
https://www.psychologytoday.com/us/blog/mood-swings/202210/has-the-serotonin-hypothesis-been-debunked#:~:text=The%20serotonin%20hypothesis%20of%20depression%2C%20popular%20from%20the%201990s%20until,was%20proven%20to%20begin%20with.
KEY POINTS
The serotonin hypothesis of depression never was a legitimate scientific hypothesis that could be proven or disproven.
It was meaningless in the sense of being too broad, and clearly false when defined more narrowly.
A recent review paper simply points out the absence of evidence for it, which was well-known for decades.
The serotonin hypothesis of depression, popular from the 1990s until now, is false, and has been known to be false for a long time, and never was proven to begin with. The norepinephrine hypothesis of depression, which preceded the serotonin hypothesis in the 1960s to the 1980s, also was false, and has been known to be false for a long time, and never was proven to begin with. The same holds for the dopamine hypothesis of schizophrenia, which began in the 1960s and 1970s, and more generally for the “chemical imbalance” metaphor for all mental illness.
All of these are major oversimplifications, which most scientists realize are major oversimplifications, but which the general public and many clinicians have assumed to be true. A recent review paper merely documents the absence of much if any scientific evidence for these oversimplified false hypotheses. So it’s not new scientifically at all. The first author of the review has been a major critic of psychiatric medications in general, especially serotonin reuptake inhibitors (SRIs), and thus the main purpose of the paper may be to seek to undermine the use of SRIs. It may indeed do so for the general public and those clinicians who have believed the false concepts of chemical imbalance and/or the serotonin hypothesis of depression. But for scientists and researchers, the use of SRIs is completely unrelated to these false metaphors. The use of SRIs should be based solely on the efficacy data shown for those agents in randomized clinical trials. Those data are indeed weak, and thus, I hold the view that SRIs should be used much less than they are, and for shorter durations, but this view has nothing to do with the already known false concepts of a serotonin theory of depression.
Another feature of the paper, which usually isn’t acknowledged, is that the absence of a relationship between measures of serotonin and “depression” also is expected and routine because that is the case with any biological marker of any kind in psychiatry in the past 40 years and any DSM-based diagnosis. By “depression” these studies usually mean DSM-defined “major depressive disorder” (MDD), and almost all studies for the last 40 years find that no biological marker correlates with most DSM diagnoses (with important exceptions in schizophrenia and bipolar illness). The problem is that DSM diagnoses are not biologically valid because they are not scientifically based; they are not based solely on scientific evidence but rather are social constructions of the American psychiatric profession. Hence they are not useful for biological research and almost always produce negative results, as in this paper. The NIMH leadership acknowledged this major problem in 2013 when the DSM-5 came out and since that time the NIMH policy has been to not use DSM diagnoses for biological research. So again this review is only documenting what is already known in general: Most DSM diagnoses, like MDD, do not correlate with any biological measure, like serotonin.
More generally: It is obvious that these false views are based on backward logic. Since SRIs improved depressive symptoms somewhat in clinical trials (though much less than people believe), it was assumed that depression had a basis in “low” serotonin. This would be like saying that since aspirin is a prostaglandin inhibitor, and it reduces fever, then fever is a prostaglandin disorder. In fact, prostaglandin effects is just one way to reduce fever, and it is only a last step to reducing fever. The real way to reduce fever is to stop the cause of fever earlier in the process, as with antibiotics for the bacteria that cause infections that produce fever.
Similarly with SRIs and serotonin. SRIs are only symptomatic drugs; they reduce symptoms of depression somewhat, just as aspirin reduces symptoms of fever. Their mechanism, increasing serotonin, may have nothing at all to do with the psychiatric diseases that cause depression, such as manic-depressive illness, just as the mechanism of aspirin has nothing to do with the infectious diseases that cause fever.
Also, the brain just doesn’t work that way. It’s not about “high” this or “low” that. There are many chemicals in the brain interacting with each other in a very complex manner, with negative and positive feedback loops, so that there is no sense at all to say that anything in the brain relevant to any illness has to do with simply having too much or too little of any chemical. Further, besides the chemicals often discussed, like serotonin, norepinephrine, and dopamine, there are hundreds of other proteins, called second messengers, that relay information inside neurons related to these chemicals. And those second messengers interact with each other in a myriad of ways. If SRIs partially influence depression by their effects on serotonin – even if we accept this simple statement – those effects are then transmitted by hundreds of other proteins and second messengers in ways that are far too complex to describe.
So how should clinicians explain depression to their patients? This is what I do and recommend:
“Depression is not a disease; it is a set of symptoms, like fever, chills, and night sweats. I would be a bad doctor if I just gave you anti-fever pills and anti-chill pills and anti-night sweat pills, instead of treating the infection that caused all those symptoms. Similarly, antidepressants like SRIs improve the symptoms of depression somewhat, but don’t get at the cause. Just like aspirin and Tylenol can improve the symptoms of fever somewhat, but don’t get at the cause. We might use antidepressants short-term for symptom benefit, but we should also try to find the disease that is causing your depression, such as manic-depressive illness.”
THE BASICS
What Is Depression?
Further, I could say: “Depression is not a ‘chemical imbalance’ because there is no ‘chemical balance.’ These are false metaphors. The brain is complex and many chemicals are active in many different directions in the brain. Your depression may be biological disease, in which chemicals are functioning abnormally, but it’s not about just having too much or too little of anything. And it’s not about getting everything into some ‘balance.’ It’s about treating the disease itself.”
<<<
(also - the 'placebo effect' can be a very significant factor with antidepressant meds. The placebo effect is sometimes in the 50% range in clinical trials)
---
>>> Study drugs make people worse at problem solving, not better
The Economist
June 20, 2023
https://finance.yahoo.com/news/study-drugs-people-worse-problem-171714158.html
FOR MORE THAN six months Americans have been struggling to get their hands on medications like dextroamphetamine (better known as Adderall) and methylphenidate (Ritalin). Officially, these stimulant drugs—alongside another, Modafinil (Provigil)—are used to treat attention-deficit hyperactivity disorder (ADHD).
Unofficially, the drugs are also popular with devotees of “nootropics”—chemicals that supposedly boost brainpower. Students and workers in industries from tech to finance take the medications in the hope they will improve concentration and ability to get things done. But a new paper suggests that this may be ill-advised. The drugs seem to make people slightly worse at solving problems, not better.
In a paper published on June 14th in Science Advances, a group of researchers led by Peter Bossaerts, an economist at the University of Cambridge, tested how Adderall, Provigil and Ritalin affected 40 healthy people’s ability to perform optimisation problems. They used the “knapsack task”, in which participants had to work out which items to put into a bag. The idea was to maximise the value of the items without exceeding the carrying weight of the sack. The researchers used several trials of varying difficulty, each with different weight limits and lists of items.
The participants visited the lab on four separate days. On each day they were given either a placebo pill or one of the drugs under study. The study was double-blind, meaning neither the participants taking the pills nor the experimenters handing them out knew which had been administered on which day. They found that participants achieved slightly worse end results on the task after taking a drug. The drugs did not impair people’s ability to find an optimal solution. Participants managed this in around half of the trials, whether they took the drugs or the placebo pills. But they did cause a small drop in the value of participants’ knapsacks across all trials, by making the non-optimal solutions worse.
Perhaps more striking was how drugs changed the way people attacked the task. After taking Adderall or Ritalin (but not Provigil) the participants spent far longer working on their knapsacks than they did when they had taken the placebo pill. (Participants were given four minutes to complete each trial but could submit an answer earlier if they thought they had found a good solution). When given Ritalin in particular, subjects were around 50% slower at completing trials. That was roughly equivalent to the delay expected from going from the easiest to the most difficult trial in the placebo session.
This extra time was spent moving items in and out of the knapsack, somewhat erratically. The authors assessed the productivity of each move by measuring how much it increased the value of a sack, and found that participants were about 9% less productive when they had taken one of the study drugs compared with a placebo pill. “It was like they were trying to solve a jigsaw puzzle by randomly throwing pieces in the air,” says Dr Bossaerts.
The authors argue that although the drugs made people more motivated and helped them put more effort into the task, this was more than cancelled out by the fact that the drugs decreased the quality of all that effort. In other words, although people tried harder, they became far less competent. Just how much the drugs hindered performance seemed to depend on how good a participant was without them. Star performers during the placebo session fell to the bottom of the pack when they had taken the drugs.
Popping stimulants is commonplace in industries like software and finance. One survey of 6,500 American college students reported that 14% had used the drugs for non-medical reasons. This latest study adds to a growing pile of evidence suggesting that such drugs do little to improve cognitive performance in people who do not need them. For tech bosses looking for efficient employees, and workers hoping to clock off at a reasonable hour, the stimulant shortage may be a good thing.
<<<
---
>>> Aaron Rodgers talks about mental heath at a psychedelics conference
San Diego Union Tribune
by JESSE BEDAYN
6-21-23
https://www.msn.com/en-us/sports/nfl/aaron-rodgers-talks-about-mental-heath-at-a-psychedelics-conference/ar-AA1cPMMC
An eclectic crowd of thousands — podcasters, vendors, startups, seekers — swarmed a psychedelics conference in Denver this week to experience everything from a dimly lit hall packed with kaleidoscope art and a wide-ranging lineup of speakers from a former Republican governor to NFL star quarterback Aaron Rodgers.
The conference, put on by a psychedelic advocacy group, took place months after Colorado’s voters decided to join Oregon in decriminalizing psychedelic mushrooms. While it’s a sign of growing cultural acceptance for substances that proponents say may offer benefits for things like post-traumatic stress disorder and alcoholism, medical experts caution that more research is needed on the drugs’ efficacy and the extent of the risks of psychedelics, which can cause hallucinations.
Rodgers, who’ll soon debut with the New York Jets after years with the Green Bay Packers, spoke Wednesday night with podcaster Aubrey Marcus. Rodgers described taking ayahuasca with his teammates as “radically life-changing,” and claimed many other pro athletes have reached out to him.
“I found a deeper self love,” said Rodgers of his ayahuasca experience. “It unlocked that whole world of what I’m really here to do is to connect, to connect with those guys, and to make those bonds and to inspire people.”
The organization hosting the conference, the Multidisciplinary Association for Psychedelic Studies, is the largest U.S. advocacy group. It has strategized to reach the full political spectrum, said Nicolas Langlitz, a historian of science who’s researched the boom and bust of psychedelic movements.
“At the time when any topic gets politically polarized, ironically, these super-polarizing substances now get bipartisan support," Langlitz said. Still, he added, the conference is “purely designed to promote the hype."
“Any kind of overselling is not good for science because science should be accurate rather than pushing things," he said. “It’s a tradeoff. (The conference) generates interest, it generates ultimately more research, even though the research might be skewed toward positive results.”
Psychedelics are illegal at the federal level, though acceptance and interest in studying their potential benefits has grown. For example, some researchers believe psilocybin, the compound in psychedelic mushrooms, changes the way the brain organizes itself and can help users overcome things like depression and alcoholism.
The drugs themselves — and the interest in them — are not new. Mid-last century, Aldous Huxley, Timothy Leary and Ken Kesey helped spur the use of psychedelics during the counterculture movement, and optimism brimmed among some psychologists over the drugs’ potential.
But the Nixon administration criminalized psychedelics, pushing them underground.
“In both cases you have this upwelling of exuberance that may or may not be irrational,” said author Michael Pollan, who wrote a book on psychedelics and will be speaking at the conference. “But I think a big difference (now) is that the enthusiasm for the potential of psychedelics cuts across a much more representative slice of the population — it’s not about a counterculture."
Republican strongholds, including Utah and Missouri, have or are considering commissioning studies into the drugs, partly inspired by veterans’ stories. Former Texas Republican Gov. Rick Perry spoke Wednesday about helping get a bill passed in the Texas legislature in 2021 to fund a study of psilocybin for veterans, though he doesn't support recreational use. In Congress, similar veteran-focused proposals brought progressive Democratic Rep. Alexandria Ocasio-Cortez from New York and far-right Rep. Matt Gaetz from Florida into an unlikely alignment.
Public interest also appears to be growing. Just six years ago in Oakland, California, the Multidisciplinary Association for Psychedelic Studies held a conference with roughly 3,000 attendees and a smattering of lesser-known speakers and die-hard proponents.
This time, organizers estimate at least 10,000 attendees. Other famous speakers will include former NHL player Daniel Carcillo, who owns a company specializing in psychedelic therapies; Olympic silver-medal figure skater Sasha Cohen; rapper and actor Jaden Smith; comedians Reggie Watts and Eric Andre, top-10 podcaster Andrew Huberman; and Carl Hart, the chair of Columbia University's psychology department.
Recruiting that celebrity support for psychedelics is part of MAPS' public relations strategy, founder Rick Doblin said. When asked whether platforming a non-expert like Rodgers could mislead the public, Doblin demurred, adding it would be “dangerous” for anyone to claim that there are no risks to taking psychedelics.
Doblin said taking MDMA should happen “only under the direct supervision of a therapist, it's never a take-home medicine.” He also emphasized what many speakers echoed during the first day about psychedelics being paired with mental health professional: “The treatment is not the drug, it’s the therapy that the drug makes more effective."
That was a more tempered approach than his introductory speech, when, to an overflowing theater, Doblin espoused grandiose goals such as “net-zero” trauma by 2070 through the use of psychedelics.
The American Psychiatric Association has not endorsed the use of psychedelics in treatment, noting the Food and Drug Administration has yet to offer a final determination. The FDA did designate psilocybin as a “breakthrough therapy” in 2018, a label that’s designed to speed the development and review of drugs to treat a serious condition. MDMA, often called ecstasy, also has that designation for PTSD treatment.
Both Pollan and Langlitz believe further research is key — especially as the nation faces an unprecedented mental health crisis and people struggle to find adequate treatment. But, Langlitz said, it's important to let research shape the narrative.
“I would just try to keep my mind open to the possibility that in retrospect we will tell a very different story from the one that the protagonists of psychedelic therapies are currently predicting,” he said.
<<<
---
>>> Synthetic human embryos created in groundbreaking advance
Exclusive: Breakthrough could aid research into genetic disorders but raises serious ethical and legal issues
Analysis: advances leave legislators needing to catch up
The Guardian
Hannah Devlin
14 Jun 2023
https://www.theguardian.com/science/2023/jun/14/synthetic-human-embryos-created-in-groundbreaking-advance
Scientists have created synthetic human embryos using stem cells, in a groundbreaking advance that sidesteps the need for eggs or sperm.
Scientists say these model embryos, which resemble those in the earliest stages of human development, could provide a crucial window on the impact of genetic disorders and the biological causes of recurrent miscarriage.
However, the work also raises serious ethical and legal issues as the lab-grown entities fall outside current legislation in the UK and most other countries.
The structures do not have a beating heart or the beginnings of a brain, but include cells that would typically go on to form the placenta, yolk sac and the embryo itself.
Prof Magdalena Zernicka-Goetz, of the University of Cambridge and the California Institute of Technology, described the work in a plenary address on Wednesday at the International Society for Stem Cell Research’s annual meeting in Boston.
“We can create human embryo-like models by the reprogramming of [embryonic stem] cells,” she told the meeting.
There is no near-term prospect of the synthetic embryos being used clinically. It would be illegal to implant them into a patient’s womb, and it is not yet clear whether these structures have the potential to continue maturing beyond the earliest stages of development.
The motivation for the work is for scientists to understand the “black box” period of development that is so called because scientists are only allowed to cultivate embryos in the lab up to a legal limit of 14 days. They then pick up the course of development much further along by looking at pregnancy scans and embryos donated for research.
Robin Lovell-Badge, the head of stem cell biology and developmental genetics at the Francis Crick Institute in London, said: “The idea is that if you really model normal human embryonic development using stem cells, you can gain an awful lot of information about how we begin development, what can go wrong, without having to use early embryos for research.”
Previously, Zernicka-Goetz’s team and a rival group at the Weizmann Institute in Israel showed that stem cells from mice could be encouraged to self-assemble into early embryo-like structures with an intestinal tract, the beginnings of a brain and a beating heart. Since then, a race has been under way to translate this work into human models, and several teams have been able to replicate the very earliest stages of development.
The full details of the latest work, from the Cambridge-Caltech lab, are yet to be published in a journal paper. But, speaking at the conference, Zernicka-Goetz described cultivating the embryos to a stage just beyond the equivalent of 14 days of development for a natural embryo.
The model structures, each grown from a single embryonic stem cell, reached the beginning of a developmental milestone known as gastrulation, when the embryo transforms from being a continuous sheet of cells to forming distinct cell lines and setting up the basic axes of the body. At this stage, the embryo does not yet have a beating heart, gut or beginnings of a brain, but the model showed the presence of primordial cells that are the precursor cells of egg and sperm.
“Our human model is the first three-lineage human embryo model that specifies amnion and germ cells, precursor cells of egg and sperm,” Zernicka-Goetz told the Guardian before the talk. “It’s beautiful and created entirely from embryonic stem cells.”
The development highlights how rapidly the science in this field has outpaced the law, and scientists in the UK and elsewhere are already moving to draw up voluntary guidelines to govern work on synthetic embryos. “If the whole intention is that these models are very much like normal embryos, then in a way they should be treated the same,” Lovell-Badge said. “Currently in legislation they’re not. People are worried about this.”
There is also a significant unanswered question on whether these structures, in theory, have the potential to grow into a living creature. The synthetic embryos grown from mouse cells were reported to appear almost identical to natural embryos. But when they were implanted into the wombs of female mice, they did not develop into live animals. In April, researchers in China created synthetic embryos from monkey cells and implanted them into the wombs of adult monkeys, a few of which showed the initial signs of pregnancy but none of which continued to develop beyond a few days. Scientists say it is not clear whether the barrier to more advanced development is merely technical or has a more fundamental biological cause.
“That’s very difficult to answer. It’s going to be hard to tell whether there’s an intrinsic problem with them or whether it’s just technical,” Lovell-Badge said. This unknown potential made the need for stronger legislation pressing, he said.
<<<
---
OCGN 5 min volume. Holding cash shares.
| Name | Symbol | % Assets |
|---|---|---|
| Regeneron Pharmaceuticals Inc | REGN | 2.77% |
| Moderna Inc | MRNA | 2.65% |
| Gilead Sciences Inc | GILD | 2.43% |
| Vertex Pharmaceuticals Inc | VRTX | 2.32% |
| Biogen Inc | BIIB | 2.28% |
| United Therapeutics Corp | UTHR | 2.26% |
| Biomarin Pharmaceutical Inc | BMRN | 2.25% |
| Seattle Genetics Inc | SGEN | 2.16% |
| Exelixis Inc | EXEL | 2.06% |
| ACADIA Pharmaceuticals Inc | ACAD | 2.04% |
| Name | Symbol | % Assets |
|---|---|---|
| Regeneron Pharmaceuticals Inc | REGN | 4.88% |
| Gilead Sciences Inc | GILD | 4.58% |
| Qiagen NV | QGEN | 4.48% |
| Biogen Inc | BIIB | 4.28% |
| Seattle Genetics Inc | SGEN | 4.08% |
| United Therapeutics Corp | UTHR | 3.93% |
| Vertex Pharmaceuticals Inc | VRTX | 3.89% |
| ACADIA Pharmaceuticals Inc | ACAD | 3.75% |
| Biomarin Pharmaceutical Inc | BMRN | 3.69% |
| Alnylam Pharmaceuticals Inc | ALNY | 3.53% |
| Name | Symbol | % Assets |
|---|---|---|
| Pacific Biosciences of California Inc | PACB | 6.85% |
| Teladoc Health Inc | TDOC | 5.94% |
| CRISPR Therapeutics AG | CRSP | 5.77% |
| Twist Bioscience Corp | TWST | 5.72% |
| CareDx Inc | CDNA | 3.87% |
| Iovance Biotherapeutics Inc | IOVA | 3.59% |
| Exact Sciences Corp | EXAS | 3.58% |
| Fate Therapeutics Inc | FATE | 3.47% |
| Invitae Corp | NVTA | 3.42% |
| Personalis Inc | PSNL | 3.26% |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
