Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
This may be possible, they do stuff with another large company. I hadn't actually considered it, but I am in the industry myself and so I get a lot of info from reliable sources. That's mainly why I went with them pretty early, I did some DD and liked what I saw.
Shares in MGC Pharmaceuticals, an Australian medical cannabis company, rose 60% on its first day trading on the LSE last week – and are now 2.5 times higher than their 1.75p debut.
https://www.theguardian.com/business/2021/feb/16/medicinal-cannabis-firm-buoyed-by-london-stock-market-debut
London Stock Exchange
Medicinal cannabis firm buoyed by London stock market debut
You may have something here. Great call so far!
GL
GW
$1 a shares ... that would be nice... and might even be higher...
Gw got picked up for billions ... market cap here is less then 100 m ...lol could get bought out at $1 a shares that would be crazy ..
ASX-listed MGC Pharma is the first medical cannabis producer to float on the main board of the London Stock Exchange, as Australian players rush into the rapidly opening European market.
https://www.afr.com/companies/healthcare-and-fitness/aussie-cannabis-players-blaze-a-trail-in-europe-20210210-p5712u
https://stockhead.com.au/health/mgc-pharma-makes-strong-debut-as-london-stock-exchanges-first-medicinal-cannabis-company/
I’m assuming this company will be bought ...
- MGC Pharmaceuticals (MXC) has appointed Nicole Godresse as Global Chief Sales Officer, effective immediately
- Nicole has more than 20 years experience in the pharmaceutical and healthcare industries, having worked with the likes of Johnson & Johnson and Eli Lilly
- Her most recent role at Tilray saw her play a part in launching the first medical cannabis brand legally in Australia and New Zealand
- She also helped launch one of the first medical cannabis clinics in the country and deliver some of Australia's first government-funded medical cannabis trials
https://themarketherald.com.au/mgc-pharmaceuticals-asxmxc-appoints-nicole-godresse-as-global-chief-sales-officer-2021-01-13/
Nicole's target (KPI) is for MGC Pharmaceuticals to reach $1million in sales per month.
https://www.proactiveinvestors.com.au/companies/news/939936/mgc-pharma-s-new-head-of-global-sales-has-big-ambitions-and-says-they-re--ready-to-go--939936.html
- MGC Pharmaceuticals (MXC) is set to become the first medicinal cannabis company to be admitted to the main market of the London Stock Exchange.
- The company will join the LSE on February 9 after a strongly supported placement was completed and £6.5 million (around A$12 million) was raised.
https://themarketherald.com.au/mgc-pharmaceuticals-asxmxc-to-list-on-lse-next-week-2021-02-04/
https://prohibitionpartners.com/2021/02/05/mgc-pharma-first-cannabis-company-london-stock-exchange/
nice move last couple of days congrats, hope it keeps up
Australian daily volume can be seen on Yahoo Finance, under MXC.AX
https://finance.yahoo.com/quote/MXC.AX/history?p=MXC.AX
Looks like profit taking from day before. Welcome to
Pinky land it appears
Here’s hoping. Thanks so much
I will watch Hope you make lots
During the quarter, MGC Pharma completed the 100% acquisition of the operating telehealth clinic-based assets, data and intellectual property of Medicinal Cannabis Clinics (MCC), a wholly owned subsidiary of Cannvalate Pty Ltd, with over 300 patient consults conducted since completion on the 23rd of November. This following the signing of a binding term sheet in July 2020. Consideration of $1m MGC Pharma Ordinary Shares (2/3 subject to trading restrictions) and $400,000 in cash has been paid for the acquisition.
Alongside revenue generated from consults, this acquisition provides MGC Pharma with an operating platform with both import and export capacity that will significantly expand market access and provide control of the supply chain from manufacturing through to patients.
The acquisition also allows the Company to continue providing its high-quality GMP certified medications to patients in Australia and further improves profit margins while keeping product at the current competitive prices.
The acquisition of the MCC Assets is the next step in building on-the-ground distribution assets allowing the Company to wholesale and distribute directly to other clinics and pharmacies to reduce storage and distribution costs.
Launch of CannEpil® App
As part of its ongoing work with the Royal Melbourne Institute of Technology (RMIT), MGC Pharma launched the CannEpil® App, and is providing medical access to the International Library of Cannabinoids (ILC).
The App is a cross platform application available to download from both the Apple App Store and Google Play Store and is designed to be used by patients (or the patient’s guardian) taking CannEpil® as a prescription treatment. The App will record patient responses to medical questionnaires as part of their treatment plan and the treating practitioner will be able to view the responses in real-time.
The ILC is a world first centralised platform compiling the diverse range of existing data on the therapeutic benefits of cannabinoids. The ILC database has been designed to collect comprehensive information about clinical trials, including details of diseases and follow up treatments, as well as product identifiers, including genetics, grow conditions and chemical profile to provide doctors with an encyclopaedia of exhaustive information on the best treatment for patients using cannabinoids.
First shipment of MP Line products directly to patients in Brazil
MGC Pharma’s first batch of MP Line products were shipped directly to patients in Brazil in October 2020, through its binding supply and distribution agreement with Brazil-based ONIX Empreendimentos e Participações (‘ONIX’). MGC Pharma is the first company globally to ship high THC formulations directly to a patient’s door in Brazil, without the need to visit a pharmacy.
The shipment was completed under Brazil’s Compassionate Use Program following the receipt of patients’ prescriptions provided by an ONIX referring doctor. ONIX currently has more than 100 referring doctors in Brazil able to prescribe cannabinoid products under the Compassionate Use Program and is targeting to have over 1,000 referring doctors by mid-2021.
Research and Development
Completion of Phase II clinical trial on COVID-19 patients
MGC Pharma’s Phase II double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of anti-inflammatory treatment, ArtemiCTM, on 50 patients diagnosed with COVID-19 has completed.
The trial included 50 patients of which 33 were in the treatment group and 17 in the placebo group and took place across three independent hospital sites across Israel and India.
The full results have demonstrated to improve the health status of COVID-19 patients delivering a NEWS score of less than or equal to 2. None of the patients in the treatment group required additional oxygen, mechanical ventilation or admission to intensive care where all of these events were reported in the placebo group. The average NEWS score of patients in the placebo group was 2.25 statistically significantly higher (p<0.04) than in the treatment group – 0/5.
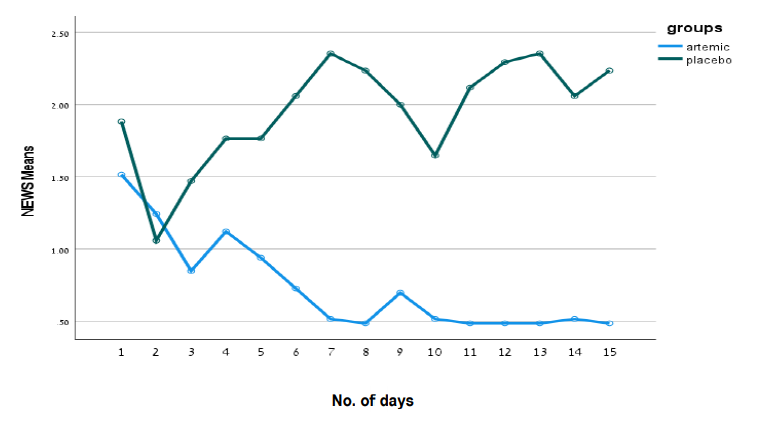
The Trial met all the FDA requirements for a COVID-19 study including population diversity (age, medical history, and genetic diversity) and demonstrated a full safety profile with no drug related adverse events. This resulted due to ArtemiCTM and the trial being focused on the immunomodulation specific for the prevention of cytokines storm, as opposed to other immunomodulators.
These results also follow safety and toxicity testing completed on mice and in line with FDA requirements for product registration requiring two types of rodents in pre-clinical trials. MGC Pharma completed an in vivo safety and toxicity pre-clinical study, including histology testing, on 24 rats. This included four groups with three study drug dosages being 48ug, 96ug and 196ug per kg rat and a control group.
The rats were observed and tested for clinical changes over seven days. This study included pathological examination of the organs: liver, heart, brain, spleen, spinal cord, sciatic nerve, kidney (L+R), lungs and tongue.
Further successful results from pre-clinical glioblastoma research
Results from the ongoing pre-clinical research program focused on evaluating cannabinoid formulations in the development of a treatment of the most aggressive and therapeutically resistant brain tumour, glioblastoma, have shown further successful results.
The pre-clinical in-vitro research program is being conducted in collaboration with the National Institute of Biology (‘NIB’) and the Neurosurgery Department at the University Medical Centre in Ljubljana, Slovenia.
The results from 18 patient tumour samples show for the first time that the Company’s proprietary formulation, CBG, exerts a superior effect in impairing the major hallmarks of glioblastoma progression, i.e. fast proliferation and invasion, and particularly enhancing glioblastoma cell death. Moreover, CBG can destroy therapy-resistant glioblastoma stem cells, which are the root of cancer development and extremely resistant to various treatments of this lethal cancer. CBG should present a new yet unexplored modality of glioblastoma therapy that could replace Tetrahydrocannabinol (THC) as a more acceptable add-on or adjuvant treatment strategy.
Financial and Corporate
Completion of unmarketable share parcel
The Company completed the sale of 50,696,634 fully paid ordinary shares pursuant to the Company’s Unmarketable Parcel Sale Facility (UMP Facility). MGC Pharma confirms its shareholder base has now been reduced by 5,067 shareholders. This will significantly reduce the Company’s administrative and corporate costs moving forward.
Progress towards completion of MGC Nutraceuticals sale to Onassis
As announced on 2nd December 2020, under the terms of the sale and purchase agreement for the 100% sale of the Company’s subsidiary MGC Nutraceuticals, MGC Pharma will receive shares equating to a value of US$6 million in Onassis Holdings Corp and the Company has secured an exclusive supply agreement for the provision of its CBD, raw materials and proprietary production intellectual property (IP). This follows the signing of a binding acquisition and exclusive CBD supply agreement as announced on 18 June 2020.
During the quarter MGC Pharma provided the 30 June 2020 audited financial statements for MGC Nutraceuticals to Onassis and full settlement of the acquisition is expected to complete over the next quarter. During the December quarter Onassis has commenced the process of finalising the offering submissions for the capital raising with the US SEC. The process from submission to completion is currently expected to complete in H1 2021. Once the offering submission has been approved, Onassis will complete the capital raising which will enable the full and complete settlement of the MGC Nutraceuticals acquisition with MGC Pharma.
Long terms benefits from UN vote to reschedule cannabis
In early December 2020, the United Nations (UN) voted in favour of the removal of cannabis and its derivatives from schedule IV in recommendations from the World Health Organisation (WHO). Cannabis and its derivativities are now contained under Schedule I of the 1961 UN Single Convention on Narcotic Drugs.
This creates a significant opportunity for MGC Pharma by removing red-tape that creates logistical limitations of the movement of products and creates an open pathway for easier and cheaper global distribution. This will also enable significantly more commercial opportunities for MGC Pharma by allowing it to deliver its Mercury Pharma product line to new markets going forward.
Appendix 4C
The Company had $1.57m cash at the end of the December 2020 quarter, with access to $9.25m undrawn from its $15m financing facility with Mercer Street Opportunity Fund LLC (as announced to the ASX on 10 September 2020). In accordance with Section 6 of the attached Appendix 4C, the Company confirms the total $480k was for executive director fees, non-executive director fees and corporate costs during the quarter.
As detailed in the Appendix 4C, expenditure for the quarter has been spent on $1.484m for research and development, $1.363m for manufacturing and operating costs, $152k for advertising and marketing, $258k staffing costs and $852k for administration and corporate costs.
https://stocknessmonster.com/announcements/mxc.asx-6A1016539/
Gives a link to a December quarterly report.
Seems like they are doing a Lazarus comeback.
thanks for the info , will see what happens?
With Shares Outstanding of 1,788,130,339 then 10MM could be a forgotten account. If the volume picks up over the next 2 weeks, we'll know for sure - something's up. Even today is about 1/2 MM over average. They do seem opportune for a 1 :100 Reverse split.
been tracking this , any idea why the drop today? thanks
MGC Pharma attracts $5m grant for its Malta-based production facility.
https://stockhead.com.au/health/mgc-pharma-attracts-5m-grant-for-its-malta-based-production-facility/
MGC Pharmaceuticals confirms Phase II trial success for anti-inflammatory COVID treatment
https://stockhead.com.au/health/mgc-pharmaceuticals-confirms-phase-ii-trial-success-for-anti-inflammatory-covid-treatment/
https://clinicaltrials.gov/ct2/show/NCT04382040?term=ArtemiC&draw=2&rank=1
Wow, Frankincense.
ArtemiC is a medical spray comprised of Artemisinin (6 mg/ml), Curcumin (20 mg/ml), Frankincense (=Boswellia) (15 mg/ml) and vitamin C (60 mg/ml) in micellar formulation for spray administration.
$MGCLF Global Arena Holdings Subsidiary Global Election Services Engages DigitalAMN for Strategic Advisory
Press Release | 10/09/2020
NEW YORK, NY, Oct. 09, 2020 (GLOBE NEWSWIRE) -- via NewMediaWire – Global Arena Holding, Inc. (the “Company”) (OTC PINK: GAHC) a growth oriented, acquisition minded Company, focused on specific niche markets offering exponential growth — is pleased to announce that its subsidiary, Global Election Services, Inc. (“GES”) has entered into a strategic advisory agreement with Digital Asset Monetary Network, Inc. (DigitalAMN), to help further the deployment of the GES’ business model.
Currently, GES is internally preparing its application to the Election Assistance Commission. If approved, GES would then have the ability to provide election services to municipal jurisdictions in states using absentee and mail-in ballots for their government elections. GES’ proprietary registration and tabulation system helps to mitigate fraud and outside interference, while allowing Americans voters to continue practicing social distancing during times of pandemic, confidently knowing that their votes ballots are being counted.
DigitalAMN (OTCMKTS: DATI) will look to help GES leverage the success that GES they had in providing election services to many clients including the Firehouse Caucus in March 2020, for the North Dakota Democratic-NPL Presidential Primary election – highlighting GES’ proprietary voter registration and voter tabulation software – whereby the state recognized its largest voter turnout for a primary in over a decade. Furthermore, given GES’ efficient and effective election process for ballots sent through the United States Postal Service and in-person ballots, GES was successful at ensuring a fair and safe election, void of any identifiable voter fraud, hacking, or outside interference that could have impeded the election process.
“We have a good and longstanding history with the CEO of DigitalAMN, who in the past, has been effective in assisting us with both GAHC and GES,” stated John Matthews, Chairman and CEO of Global Arena Holding, Inc. “We believe GES will greatly benefit from the strategic business development, capital sourcing, and market development management services that DigitalAMN will provide.”
GES intends to leverage the extensive expertise of its senior management and its proprietary software to administer election services at state and local levels. There are approximately 3,080 municipal jurisdictions located throughout the United States that are responsible for managing the election process at the state and local levels for an average of 8 to 12 elections that are held each year. These annual elections may include competitive races for School Board Elections, local Mayor, Village Officers, Supervisors, County Executives, County legislators, Governor, State Assembly, State Senate, and US House of Representatives and US Senate, to name a few. As more states increase the adoption of absentee and/or mail-in ballots as an effective option, the opportunity that GES may be retained by municipal jurisdictions grows, affording the Company the ability to increase its absentee, mail-in ballot and in-person business for government elections.
Ajene Watson, CEO of DigitalAMN stated, “I believe that globally, there is roughly $10B in virtually untapped market potential for Universal Absentee, Mail-In Ballots and in-person voting for governmental elections. I also know that GES’ senior staff has an expertise in this area of election services, has been supervising elections since 1981, and has had over 8,500 labor elections certified by the US Dept. of Labor with, impressively, not one labor election ever being overturned due to actions by GES’ senior management. Equally as exciting, GES is capable of providing election services on an International scale as well.
“This all encourages me to leverage the full force of our growing ecosystem to assist GES with its continued business development, visibility, capital formation requirements, and their next stage of growth into governmental elections.”
Management has indicated that the compensation to DigitalAMN for its strategic advisory services can be found in the Company’s most recent filings and includes a retainer, warrants, monthly compensation, and revenue share from new governmental election services.
For more information on these and other developments, please visit: http://GlobalArenaHolding.com, where you can also access all of the Company’s SEC filings, as management continues to pursue a more effective investor relations initiative.
ABOUT GAHC
Global Arena Holding, Inc. trades on the OTC Pink Sheets under the ticker symbol GAHC. The Company has been publicly traded since 2011 and holds a number of interests, including Global Elections Services, Inc., Tidewater Energy Group Inc., and GAHI Acquisition Corp. Please find the Company’s filings at SEC.gov
ABOUT Digital Asset Monetary Network, Inc.
Digital Asset Monetary Network, Inc. (OTCMKTS: DATI) is the first company to utilize the Public Accelerator-Incubator (PAI) model, with the intent to follow the global success of accelerators and incubators around the world, adding niche opportunities to both the microcap and startup communities. As a PAI, Digital Asset Monetary Network will develop and acquire innovations that solve problems through digital platforms and other electronic applications.
Safe Harbor Statement
The Private Securities Litigation Reform Act of 1995 provides a "safe harbor" for forward-looking statements. Certain information included in this press release contains statements that are forward-looking, such as statements related to the future anticipated direction of the industry, plans for future expansion, various business development activities, planned or required capital expenditures, future funding sources, anticipated sales growth, and potential contracts. Such forward-looking information involves important risks and uncertainties that could significantly affect anticipated results in the future and, accordingly, such results may differ from those expressed in any forward-looking statements made by, or on behalf of, the company. These risks and uncertainties include, but are not limited to, those relating to development and expansion activities, dependence on existing management, financing activities, domestic and global economic conditions, and other risks and uncertainties described in the Company's periodic filings with the Securities and Exchange Commission.
CONTACT
Global Arena Holding, Inc.
208 East 51st St.
Suite 112
New York, NY 10022
kathryn@globalarenaholding.com
Tel: 646-801-5524
www.globalarenaholding.com
$MGCLF
Trading Halt
Home Country News Release | 09/07/2020
September 7, 2020
OTC Disclosure & News Service
West Perth, WA, Australia —
This release includes additional documents. Select the link(s) below to view.
Trading Halt.pdf
Wondering what the benefits and effect of this news will be on the share price.
$MGCLF-prosped has terms of a long squeeze propabally percents.
https://www.otcmarkets.com/stock/MGCLF/news/MGC-Pharma-to-acquire-100-of-Medicinal-Cannabis-Clinic?id=268023
What will be the chances of getting off the grey market in the near future.
MGC Pharmaceuticals Ltd (ASX: MXC, ‘MGC’ or ‘the Company’), a European based ‘Seed to Medicine’ bio-pharma company specialising in the production and development of phytocannabinoid-derived medicines, is pleased to announce successful research results from the ongoing pre-clinical research program that supports and directs novel cannabinoid formulations in the development of treatment for glioblastoma multiforme (‘GBM’), the most aggressive, and so far therapeutically resistant, primary
brain tumour.
The cannabinoid formulations used in this collaborative research program are proprietary and are core to the intellectual property of MGC Pharmaceuticals.
Key Highlights
• This pre-clinical research focuses on the development of cannabinoid formulations specifically tailored for the effective treatment of high-grade brain tumours
• Recent data from NIB, conducted on 14 additional GBM tumour tissue samples, support and direct novel cannabinoid formulations in the treatment of glioblastoma
• Results to date on brain tissue samples from a total of 24 patients (including the 10 patients reported on in July 2019) confirm that the cannabinoid preparations can successfully inhibit tumour cell viability and cause a significant percentage of glioblastoma cells to undergo
“programmed cell death” i.e. apoptosis, after a short time following application
Research also shows CBG termination effects specifically on glioblastoma stem cells, which tend to be extremely resistant to irradiation and chemotherapy treatments
https://themarketherald.com.au/mgc-pharmaceuticals-asxmxc-successful-results-from-ongoing-brain-cancer-research-2020-04-22/
Problem is the stock is trading in the Grey market.
excellent news, thanks Cash4.
I have shares in this company in Australia for the past few years, maybe coming is a pay off
MGC Pharmaceuticals (ASX:MXC) receives Phase II trial approval for COVID-19 patients
https://themarketherald.com.au/mgc-pharmaceuticals-asxmxc-receives-phase-ii-trial-approval-for-covid-19-patients-2020-04-17/?fbclid=IwAR3SJ9Okpd9XHjtBCOEJv8CCQ9uaTMxT5WrLtosJNOmak7tFss0SPMfh-uI
Ashleigh Melanko
Markets Reporter
ashleigh.melanko@themarketherald.com.au
17 April 2020 13:09
MGC Pharma (ASX:MXC) - Co Founder and Managing Director, Roby Zomer - The Market Herald
Co Founder and Managing Director, Roby Zomer
Sourced: FinFeed
MGC Pharmaceuticals (MXC) will be conducting a Phase II placebo-controlled clinical trial to evaluate the safety and efficacy of ArtemiC in COVID-19 patients
This trial follows the company's recent approval from an Israel-based hospital
ArtemiC, which is a natural anti-infective formulation, is used to target viral infections
The trial is expected to begin this month and finish in September
Results will then be available the following month
Despite there being a pause in trade, MGC Pharma shares have soared 36 per cent and are currently trading for 3.4 cents each
MGC Pharmaceuticals (MXC) will be conducting a Phase II placebo-controlled clinical trial to evaluate the safety and efficacy of ArtemiC in COVID-19 patients.
This trial comes after the company was granted Human Research Ethics Committee approval from Nazareth Hospital EMMS in Israel.
Due to the trial being classified as a "Special Clinical Trial", no more approvals need to be granted before it can begin.
On April 4 2020, MGC Pharma entered a binding agreement with Micelle Technology to provide research support, and commercial manufacturing and distribution of the product.
ArtemiC is a natural, anti-infective formulation that is made up of Artemisinin (a drug derived from a fern-like plant and is used against malaria) and Curcumin (bright yellow chemical which provides anti-inflammatory and antioxidant effects).
It is then mixed with Vitamin C and Boswellia serrata (herbal extract used to treat inflammatory illnesses) as taking Artemisinin by itself may do more harm than good.
Both MGC Pharma and Micelle have begun planning for testing programs to evaluate ArtemiC's anti-infective, anti-viral and anti-inflammatory properties, including on patients with COVID-19.
The Trial
The Phase II trial will evaluate the safety and efficacy of ArtemiC in patients with COVID-19.
The trial is expected to begin this month, following the recruitment of 50 patients with COVID-19.
The trial will be conducted over a period of two weeks per patient and it is expected to finish in September 2020 with results available in October 2020.
If the trial is successful then both MGC Pharma and Micelle will make a decision, based off the results, about future trials.
The end goal for both companies is to achieve full marketing authorisation, however, it was announced that successful outcomes do not necessarily guarantee regulatory approval.
If the trial is unsuccessful then MGC Pharma will examine the results and decide whether to proceed.
"Following our recently announced agreement with Micelle, this approval to proceed immediately with a Phase II clinical trial of ArtemiC is a major milestone," Co-Founder and Managing Director Roby Zomer commented.
"This trial will evaluate the safety and efficacy of ArtemiC on patients diagnosed with COVID-19 and we look forward to updating the market with developments," he added.
Despite there being a pause in trade, MGC Pharma shares have soared 36 per cent and are trading for 3.4 cents each at 12:45 pm AEST.
Tweet received. "Wait until you see the announcement from @MGC_Pharma tomorrow."
Cannepil now available in Ireland. Pretty big step as it's doorway in having the whole EU on board. Steps are continuing in a forward fashion. This ticker is almost completely unknown in the US, or anywhere outside Australia for that matter!
MGCLF
Company is moving along and checking off boxes. Need to keep the dilution in check and quit paying their officers top dollar when the pps is at 0.03. But, lots of potential with this ticker.
MGCLF
"The Company has now passed the 1,000 prescribed products milestone ahead of schedule in Australia and the United Kingdom, through prescriptions issued to more than 500 unique patients."
http://www.aspecthuntley.com.au/docserver/02167030.pdf?fileid=02167030&datedir=20191031&edt=MjAxOS0xMS0wNCsxNDowNTo0Mys0ODArNzUyOTAxK2FuZHJld3dlc3QrcmVkaXJlY3QraHR0cDovL3d3dy5hc3BlY3RodW50bGV5LmNvbS5hdS9pbWFnZXNpZ25hbC9lcnJvcnBhZ2VzL3BkZnRpbWVvdXQuaHRtbCtodHRwOi8vd3d3LmFzcGVjdGh1bnRsZXkuY29tLmF1L2ltYWdlc2lnbmFsL2Vycm9ycGFnZXMvcGRmZGVsYXllZC5qc3A=
'MGC Lands Key Australian Cannabis Licence'
https://thewest.com.au/business/public-companies/mgc-lands-key-australian-cannabis-cultivation-licence-c-138467
Official ASX announcement:
http://www.aspecthuntley.com.au/docserver/02109587.pdf?fileid=02109587&datedir=20190529&edt=MjAxOS0wNS0yOSsxOToyNzozOCs0ODArNzUyOTAxK2FuZHJld3dlc3QrcmVkaXJlY3QraHR0cDovL3d3dy5hc3BlY3RodW50bGV5LmNvbS5hdS9pbWFnZXNpZ25hbC9lcnJvcnBhZ2VzL3BkZnRpbWVvdXQuaHRtbCtodHRwOi8vd3d3LmFzcGVjdGh1bnRsZXkuY29tLmF1L2ltYWdlc2lnbmFsL2Vycm9ycGFnZXMvcGRmZGVsYXllZC5qc3A=
An EU-based BioPharma company, MGC Pharmaceuticals Ltd (ASX: MXC) is an expert in the medical cannabis industry. The company’s strategy is to deliver high-quality cannabinoids-based pharmaceuticals products in the European, North American and Australasian medical markets.
MGC Pharmaceuticals announced on 6 May 2019 that it has entered into an exclusive distribution agreement with Grow Biotech and IPS (‘the Parties’) – leaders in UK medical cannabis market. The two parties have an extensive distribution network of above 5,500 pharmacies. As per the agreement, MXC can now have direct access to their distribution channels in the growing UK medical cannabis market.
As per the agreement, Grow Biotech and IPS would render medical import and distribution services to MXC. This would allow the patients residing in the UK to have access to MGC’s propriety products like CannEpilTM for people suffering from epilepsy and CogniCannTM for people suffering from Dementia and Alzheimer.
On 12th April 2019, the MGC Pharmaceuticals obtained a permit to import epilepsy drug into the UK. The company also got its first formal purchase orders from Australia and the UK for CannEpilTM.
Post-UK regulation changes on 1st November 2018, Grow Biotech and IPS provided medicines to the patients directly on 15th February 2019, due to which the specialist doctors got permission to prescribe medical cannabis to their patients. Grow Biotech and IPS were the first companies that imported a bulk shipment of medical cannabis to the UK market. They have an on-site pharmacy to deliver prescriptions directly to a patient’s door.
As indicated in the agreement, Grow Biotech and IPS would ensure that MGC Pharma’s proprietary products would be registered, imported and delivered directly to the end patient. In an effort to support the products’ reception in the UK market, Grow Biotech would offer educational support and would provide product information to UK prescribing doctors.
The initial prescriptions for CannEpilTM have already been received by the Parties. MGC Pharma would help the board Centre for Medical Cannabis, a UK industry membership body in the formation of a supportive regulatory framework. This would enable patients to have smooth access to cannabis-based medications.
According to Roby Zomer, Co-founder and Managing Director of MGC Pharma, this partnership has placed the company as a proven expert in importing medical cannabis products into the UK market.
The terms of the agreement are highlighted below:
Grow Biotech and IPS – exclusive importer and distributor of MGC Pharma’s proprietary Investigational Medicinal Products (‘IMPs’) in the UK.
Grow Biotech and IPS would offer scientific education services and clinic support during the duration of the agreement.
The term of the agreement spans across 5 years of timeframe.
Existence of termination rights in certain cases.
The company is entitled to pay success fees upon specified sales-based milestones.
No specified minimum contract amounts or volumes in the agreement.
The UK medical cannabis market is anticipated to reach €8.8B by 2028, which provides a significant opportunity for MGC Pharma in the UK market. Also, over 500,000 people in the UK are currently diagnosed with epilepsy that can benefit from the CannEpilTM product.
Recently, MGC Pharma had also signed two strategic supply agreements with two medicinal cannabis distribution and logistics companies – Cannvalate and Health House International. Following the contract, MXC’s phytocannabinoid-based products would be accessible under the Special Access Scheme.
A few days ago, the company also partnered with Chinese e-commerce import platform, YuShop Global. The partnership enabled MXC to market and distribute its nutraceuticals CBD based products in the Chinese health products market.
The company also announced its 2019 commercialisation strategy on 2nd April 2019. The strategy was centred around the company’s two core divisions, Seed-to-Pharmacy Manufacturing and Research & Development.
The company’s stock is currently trading at AUD 0.051 (as on 6th May 2019, 3:15 PM AEST), up by 2 percent.
Cannepil making it's debut in Aussieland and the UK!! Great development going on here!!
MGCLF
The world-leading clinical trial, aimed at improving the quality of life for thousands of people with dementia, will be undertaken by the university’s Institute for Health Research in partnership with @MGC_Pharma .
2 April 2019 ASX Code: MXC
MXC’s 2019 Pharma Product Commercialisation Strategy
Delivering Strong Pipeline of GMP Medicinal Cannabis Products
MGC Pharmaceuticals Ltd (ASX: MXC) (OTC: MGCLF) (“MGC Pharma” or “the Company”), a pure bio-pharma company focused on developing and commercialising a portfolio of cost-effective phytocannabinoid based medicines, is pleased to provide a positive update on its pharmaceutical operations and seed-to-pharmacy business plan.
• Established commercialisation strategy of focusing exclusively on two core divisions, Seed- to-Pharmacy Manufacturing and Research & Development, with the objective of becoming a leading supplier of cannabinoid based pharmaceutical products for medical markets in Europe, UK, and Australasia
• R&D division to deliver the production of cost-effective and affordable medicines in collaboration with leading internationally renowned research institutions and ensuring that MGC Pharma remains at the forefront of the sector
• Seed-to-pharmacy manufacturing focused on bringing affordable phytocannabinoid-based medicines to the global markets via cannabis cultivation, extraction, isolation and compounding through to a finished medicinal product
• Strong existing pipeline of phytocannabinoid based medicines targeting conditions such as epilepsy, Alzheimer’s, cancer and irritable bowel disease that all at present have no effective treatment. Products are poised for release into the European and Australian markets upon completion of testing and clinical trials, currently underway
• 2019 priority already commenced with patient recruitment into the CogniCannTM clinical trial and the progression and expansion of all RMIT projects following recent approval from the Office of Drug Control in Australia
Roby Zomer, Co-founder and Managing Director, MGC Pharmaceuticals commented
“MGC Pharma has delivered a very productive first quarter with key operational milestones being achieved for the commercialisation of its bio-pharma business. The recent sale of MGC Derma to CannaGlobal has enabled us to position ourselves as a pure pharmaceutical company as we direct our research and focus into our two core divisions.
“Receiving the necessary approvals from the Office of Drug Control in Australia to possess and handle phytocannabinoids for research purposes is integral to the work being undertaken by our R&D division as they continue the rapid development of treatments focusing on the neurological, oncological, dermatological and gastroenterological sectors.
1 - 5
“Having secured an EU licence to produce non-sterile schedule two medicines, which includes all cannabinoids and the consolidation of our European activities in Malta, we are in a strong position to grow and develop our Seed-to-Pharmacy manufacturing division and make excellent progress in 2019.”
Research and Development (R&D) Division
The R&D division’s primary function is to ensure MGC Pharma remains at the forefront of pharmaceutical innovation within the cannabis for medicinal use sector. The R&D division is spearheaded by a team of highly experienced medical practitioners working alongside scientific departments and genetics teams from world leading universities. Importantly, a number of ground-breaking achievements have already been made including the establishment of several pioneering research programs and the development of new proprietary genetic strain MXC-10, containing industry high levels of THC.
Current research and development activities underway to create a pipeline of pharmaceutical and nutraceutical products include; the development of CannaHub, in collaboration with the Royal Melbourne Institute of Technology (RMIT) and the Hebrew University of Jerusalem (HUJ); the CogniCannTM clinical trial taking place with the University of Notre Dame in Western Australia; the CannEpilTM clinical trial taking place with the University of Ljubljana, Slovenia; the C4E education platform in collaboration with Epilepsy Action Australia and; MGC Pharma’s genetics development and registration activities with the University of Ljubljana’s biotechnical faculty in Slovenia.
The R&D division is currently focused on three sectors:
1. Neurological disorders
Based on the experience of three of our leading doctors Prf. Uri Kramer, Prof David Neubauer and Dr. Jonathan Grunfeld, MGC Pharma established its first clinical arm with Investigational Medicinal Product (IMP) CannEpilTM to treat Drug Resident Epilepsy (refectory epilepsy). It has subsequently developed a second product CogniCannTM, to improve the quality of life in mild cases of dementia and Alzheimer’s. Further products will be produced as part of a continuous development to target additional neurological indications.
2. Oncology and treatment side effects
Under the guidance of the Group’s CSO Dr. Jonathan Grunfeld, and utilising his vast experience in treatment of cancer patients in Israel, and the support equipment and sciences of RMIT and HUJ, MGC Pharma’s second clinical arm has been established to focus on oncological treatments. TetrinolTM, is the first drug in development for use in the treatment of cancer side effects such as cachexia and acute nausea. Additionally, MGC Pharma is developing medicine for the treatment of Melanoma and Prostate cancer which are part of a pre-clinical research with CannaHub (RMIT and HUJ) and a brain cancer research project with the National Institute of Biology (NIB) Slovenia.
3. Inflammatory and Autoimmune
Utilising data collected over time with CannaHub, MGC Pharma’s third clinical arm aims to treat one of the most rapidly emerging problems in the western but also in the eastern side of the world, which is the chronic Inflammation and Inflammation of the immune system. The Company’s first Anti-Inflammatory products are InCannTM, a BiActive microspheres capsule used to treat Inflammatory bowel disease (IBD) Crohn’s disease and colitis, and TopiCannTM, an Anti-inflammatory topical cream to treat Eczema and inflamed skin.
2- 5
ODC Approval Received
Royal Melbourne Institute of Technology (RMIT) has received approval from the Office of Drug Control (“ODC”) to possess and handle phytocannabinoids for research purposes. This is a significant milestone achieved for the advancement of all projects currently underway.
Following receipt of this approval, the core research activities at RMIT are:
1. Cancer
The use of nanotechnology in the development of drug delivery systems for the treatment of cancer using existing knowledge from HUJ. Current projects are focussed on researching treatments to inhibit the development of angiogenesis and inflammation in the progression of cancer, antiangiogenic and anti-cancer efficacy of CB2 agonists and Cannabinoid-induced targeted facilitated entry of cytotoxic compounds selectively into tumour cells.
2. Nanotechnology based drug delivery
Both RMIT and HUJ have divisions focussed on nanotechnology-based drug delivery for treatments of various disorders, such as Controlled Release Gastro-Retentive Swelling Dosage Forms for Optimising Therapy of Cannabinoids, targeting of cannabinoids to inflamed tissue: Elucidating the mechanism of immunomodulation and Targeting of nanoparticles containing cannabinoids to peripheral neuronal cells or the extracellular matrix.
3. Combined treatment: traditional medicine and cannabinoid therapy
A current project is underway at RMIT, focussed on researching the benefits and possibility of developing a new treatment that combines medicinal cannabis using traditional medical principles. The objective is to look at multiple compounds and assess how each can be enhanced and developed to create new innovative medicines.
3- 5
Clinical Trials Progress on Track
Recruitment for the Phase IIB CogniCannTM clinical trial has commenced. The trial remains on track and is scheduled to last 16 weeks focussing on the effects of CogniCannTM on 50 patients aged 65 and over with mild dementia and Alzheimer’s.
Seed-to-Pharmacy Manufacturing Division
The Seed-to-Pharmacy division capitalises on the intellectual property generated by the R&D division. Its operations encompass supply chain management and end-to-end cannabis production including cultivation, extraction, isolation and compounding the finished medicinal product. Furthermore, it is focussed on bringing a series of phytocannabinoid based medicines to market that are specifically designed to treat a range of neurological, inflammatory and physiological disorders.
Strategic overview
In order to commercialise its research findings, MGC Pharma has centralised its European operations and retained several operational branches in other strategic territories. Furthermore, the Company has been awarded an EU licence to produce non-sterile schedule two medicines, which includes all cannabinoids and noting this includes THC in particular. MGC Pharma is one of only a few companies to be awarded this licence and it enables the production of compound phytocannabinoid-based medicines.
This licence was issued to the Company’s Slovenian facility that contains a CO2 super-critical extraction and separation unit that isolates cannabinoids into purified compounds. These purified compounds can then be used as an Active Pharmaceutical Ingredient (API) and be compounded into end product medicines such as Investigational Medicinal Products (IMP).
Additionally, MGC Pharma’s operational strategy is to implement a fully integrated supply chain in order to provide cost effective medicines.
2019 to deliver strong progress in Malta – upgraded commercial facility approval
In support of its seed-to-pharmacy strategy, the Company will construct a facility in Malta on the land it was awarded by Malta Enterprise, which is expected to commence very shortly. The original approved 4,000m2 site footprint has been increased to 6,000m2 as a result of strong relationships established with the government agencies, and will be used for 5,000m2 EU GMP certified production and a 5,000m2 fully equipped cultivation site, all on the same plot in a multi-story facility. The land will be leased on a long-term basis at preferable cost to the Company with an option to acquire; this is testament to the support for the Company from Malta Enterprise and Malta Industrial Parks.
Aside from an ideal climate of extended sunlight hours and optimal temperatures, Malta also provides the most effective business “climate” due to its low corporate tax, which makes it suitable to be the “market-release” site for MGC Pharma’s products to all Europe and MENA countries, as it is part of the European Union.
4- 5
Malta is also home to one of the largest sea-ports in the Mediterranean and has direct access to Italy, providing the ideal central location for operations and logistics within Europe. As a result, MGC Pharma’s Maltese facility site will become its primary operation site, fully integrating its supply chain and providing easy access to European the global markets.
In preparation for the commencement of construction, the Company has successfully finished its geological survey on the land and has submitted the architectural plans to the relevant authorities for approval. MGC Pharma’s is now waiting for the Acquisition of Immovable Property, by Non-Residents (AIP) in order to sign the final agreement with Maltese Industrial Parks. On execution of these final agreements, the first stage construction activities will then commence.
The Company will update the market on developments in due course.
Strong commercial relationships for growth
As a bio-pharma company, MGC Pharma’s core focus is on the development and manufacturing of cutting edge phytocannabinoid based medicines and to support this, MGC Pharma has partnered with global pharmaceutical companies to generate global exposure within the sector.
MGC’s distribution network covers Europe, the United Kingdom, MENA
countries, Australia and New Zealand, providing it access to patients for its
medicinal cannabis products. Maintaining strong relationships with leading
pharmaceutical distributors globally also provides MGC Pharma; with the
ability to leverage these relationships and expand its operations, distribution and footprint globally.
Due to changes in TGA regulation pertaining to the promotion of IMPs and other non-registered medicinal products, the Company is no longer permitted to provide an update of the current status of any of its IMPs in market. All commercial successes will be reflected as cash inflows from ordinary operations in future Appendix 4C, Half Year and Full Year reporting submissions.
MXC’s Distribution footprint
For further information, please contact:
Media Enquiries
Justin Kelly
Media and Capital Partners
+61 408 215 858 Justin.kelly@mcpartners.com.au
About MGC Pharma
--End-- UK IR/Media Advisors
Gaby Jenner/ Catherine Leftley
St Brides Partners Ltd
+44 (0) 207 236 1177 gaby@stbridespartners.co.uk catherine@stbridespartners.co.uk
MGC Pharmaceuticals Ltd
Brett Mitchell
Executive Chairman
+61 8 6382 3390 info@mgcpharma.com.au
MGC Pharmaceuticals Ltd (ASX: MXC) is an EU based BioPharma company with many years of technical clinical and commercial experience in the medical cannabis industry. The Company’s founders were key figures in the global medical cannabis industry and the core business strategy is to develop and supply high quality Cannabinoids based pharmaceuticals products for the growing demand in the medical markets in Europe, North America and Australasia.
Follow us through our social media channels
5- 5
Great news!
Go MGC!!!
Welcome to 2019 MGC'ers!! It should be a very good year for continued new developments and execution of projects already in the works. Today, the fins came across US newswires which is a first for this Australian company. New eyes will be exposed to MGC today and going forward which is huge as is being off the pinks and finally on the QB. With any luck, we can maybe move up to the QX by EOY/early 2020. Good luck to us all!!
MGC
As we progress further into 2019, we’d like to update you on the progress of our MGC Botanic division:
In 2018, our cannabis crop in the Czech Republic delivered 30% more flowers than the 2017 harvest and is being prepared for immediate transportation to its Slovenian extraction facility.
In addition, the Slovenian Ministry of Health has awarded us a permit for the extraction of phytocannabinoids for the creation of MXC’s own proprietary APIs (Active Pharmaceutical Ingredients).
We’re also pleased to report that we have successfully developed a new proprietary genetic strain, MXC-10, containing the highest level of THC (>35%) of any MXC strand launched to date.
The next step for our Botanic division is to begin the process of registering our four unique Cannabis Sativa strains (bred in the University of Slovenia Biotechnical faculty) in Europe for medical purposes — an enormous production and distribution milestone for MGC Pharma.
See the full update here: http://snip.ly/46iz47
"Today we’re pleased to report that three landmark milestones have now been met by MGC Pharma.
1. Our CTN (Clinical Trial Notification) for CogniCann™ into Alzheimer’s and dementia at the University of Notre Dame is now available in the Clinical Trials Repository.
2. We are one of the first EU facilities to be granted a full API extraction permit to develop our own THC, CBD and other Phytocannabinoids for use in CannEpil™, CogniCann™ and future phytomedicines.
3. We have received SME certification from the European Medicines Agency (EMA) for the scientific advice, drug evaluation and registration of CannEpil™, CogniCann™ and additional Phytomedicines that the Company is developing.
These are landmark milestones for the MGC Pharma company and are a testament to the continuous hard work we’re putting in to become a global market leader in phytocannabinoids."
Read the full announcement here: http://snip.ly/bx35d9
|
Followers
|
20
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1011
|
|
Created
|
11/02/17
|
Type
|
Free
|
| Moderators | |||
Our three pioneering divisions allow MGC to be a key player in all significant levels of production in various global locations. It also allows MGC to progress with simultaneous high level research and development, clinical trials around the world and to embrace new emerging verticals in CBD markets.
Founded in 2014 by experts in the Medical Cannabis field with the intention of capitalising on the emerging global Medical Cannabis industry, founders Nativ Segev and Roby Zomer bring a combination of over 10 years in the Medical Cannabis and the BioTech industry. MGC Pharma was created to become a leading BioPharma company based on Medical Cannabis with operations spanning all aspects of the value chain, from cultivation, manufacturing and distribution of products in a global market.
Today, MGC Pharma has operations spanning Europe and Australia. It has cultivated and harvested crops in its growing facilities in the Czech Republic and Slovenia and is manufacturing active pharmaceutical ingredients (APIs) at its European GMP extraction facility. The Company has also developed a CBD cosmetics range that is available for purchase throughout Europe and online via the Company’s eShop www.mgcderma.com
The Botanic Division engages with multiple cultivation sites to meet increasing global demand, alongside research into breeding and genetics with leading institutions and academia. We will continue to harvest the product in Slovenia and Czech Republic, and use the raw material at our production facility, as well as provide the product globally.
Our research & development:
The Innovation Division is focused on our medical product pipeline and is our largest division. This division focuses on products that emerge from our supply line. These include neutraceuticals, which will be able to provide the additional needs and requirements that our body demands on a daily basis to improve our immune system, detoxify and enhance wellness.
Additionally, this includes our extracts, at various levels of purity, and with the ability to extract various phytocannabinoids and terpenes to the level of Active Pharmaceutical Ingredient.
The Innovation division is focused on three specific areas of treatment:
MGC Derma produces CBD enhanced cosmetics, DermaPlus dermatological line and private label manufacturing.
Our research & development:

MGC Derma is the brand leading the way in utilising this year’s ‘it’ skin care ingredient: cannabis. Before you get the wrong end of the stick… we’re talking about cannabidiol (CBD) a totally non-psychoactive compound derived from the cannabis plant, which shares part of the ‘fingerprint’ of our genetics, allowing the body to instantly recognise it – and utilise it accordingly. It is MGC Derma’s unique, meticulous extraction and purification process that keeps this ‘fingerprint’ intact; the brand uses only the highest quality cannabidiol in this range of pure yet potent serums, masks, toners and cream. Renowned for its healing, sebum-regulating and moisturising capabilities, CBD is particularly adept at addressing eczema, acne and other problematic skin conditions, as well as minimising signs of ageing, hydrating and nourishing.

MGC Derma is the brand leading the way in utilising this year’s ‘it’ skin care ingredient: cannabis. Before you get the wrong end of the stick… we’re talking about cannabidiol (CBD) a totally non-psychoactive compound derived from the cannabis plant, which shares part of the ‘fingerprint’ of our genetics, allowing the body to instantly recognise it – and utilise it accordingly. It is MGC Derma’s unique, meticulous extraction and purification process that keeps this ‘fingerprint’ intact; the brand uses only the highest quality cannabidiol in this range of pure yet potent serums, masks, toners and cream. Renowned for its healing, sebum-regulating and moisturising capabilities, CBD is particularly adept at addressing eczema, acne and other problematic skin conditions, as well as minimising signs of ageing, hydrating and nourishing.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
