| Followers | 246 |
| Posts | 49771 |
| Boards Moderated | 69 |
| Alias Born | 08/29/2007 |
Sunday, August 06, 2023 12:29:54 PM
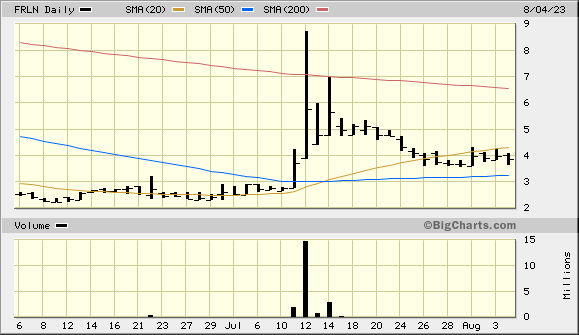
GoSymbol lookup
Health Care : Biotechnology | | Based in United KingdomCompany profile
Freeline Therapeutics Holdings plc is a United Kingdom-based clinical-stage biotechnology company. The Company is developing transformative adeno-associated virus (AAV) vector-mediated gene therapies. It is focused on improving patient lives through one-time treatments for chronic debilitating diseases. It uses its rationally designed AAV vector and capsid (AAVS3), along with promoters and transgenes, to deliver a functional copy of a therapeutic gene into human liver cells, thereby expressing a persistent functional level of the missing or dysfunctional protein into the patient's bloodstream. Therapeutic AAV vectors consist of two main components, the capsid and the expression cassette (that contains the gene and the promoter). The Company is advancing clinical programs in Fabry disease and Gaucher disease Type 1. The Company's pipeline includes Fabry Disease FLT190 for the treatment of Fabry disease and Gaucher Disease FLT201 for the treatment of type 1 Gaucher disease.
I am writing a book, American Cars of 1958. Check often for the latest addition. https://investorshub.advfn.com/American-Cars-of-1958-37252/
Recent FRLN News
- Freeline to Present New Data on its Gaucher and Parkinson’s Disease Programs at the American Society of Gene and Cell Therapy (ASGCT) 27th Annual Meeting • GlobeNewswire Inc. • 04/24/2024 08:00:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 02/26/2024 05:15:23 AM
- Form S-8 POS - Securities to be offered to employees in employee benefit plans, post-effective amendments • Edgar (US Regulatory) • 02/22/2024 09:38:00 PM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 02/22/2024 09:28:12 PM
- Form SC 13E3/A - Going private transaction by certain issuers: [Amend] • Edgar (US Regulatory) • 02/20/2024 08:22:44 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 02/20/2024 12:13:56 PM
- Acquisition of Freeline by Syncona Becomes Effective • GlobeNewswire Inc. • 02/20/2024 12:00:00 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 02/12/2024 09:01:50 PM
- Freeline Shareholders Approve Acquisition by Syncona • GlobeNewswire Inc. • 02/12/2024 05:00:00 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 01/18/2024 09:15:56 PM
- Form SC 13E3/A - Going private transaction by certain issuers: [Amend] • Edgar (US Regulatory) • 01/17/2024 09:17:39 PM
- Form SC 13E3 - Going private transaction by certain issuers • Edgar (US Regulatory) • 12/08/2023 10:21:15 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 12/01/2023 01:56:00 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 11/22/2023 02:20:57 PM
- Syncona to Acquire Freeline Therapeutics • GlobeNewswire Inc. • 11/22/2023 12:30:00 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 10/25/2023 10:01:03 AM
- Freeline Presents Positive New Data from Phase 1/2 GALILEO-1 Trial of FLT201, Its Novel Gene Therapy Candidate, in Gaucher Disease at ESGCT 30th Annual Congress • GlobeNewswire Inc. • 10/25/2023 06:01:00 AM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 10/18/2023 11:00:05 AM
- Freeline to Present New Clinical Data for FLT201 in Gaucher Disease in Oral Presentation at Upcoming ESGCT 30th Annual Congress • GlobeNewswire Inc. • 10/16/2023 11:00:00 AM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 10/06/2023 08:52:15 PM
- Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16] • Edgar (US Regulatory) • 10/04/2023 11:00:02 AM
- Freeline Reports Positive Initial Clinical Data from First Cohort of Phase 1/2 GALILEO-1 Trial of FLT201, Its Novel Gene Therapy Candidate, in Gaucher Disease • GlobeNewswire Inc. • 10/04/2023 11:00:00 AM
NanoViricides Reports that the Phase I NV-387 Clinical Trial is Completed Successfully and Data Lock is Expected Soon • NNVC • May 2, 2024 10:07 AM
ILUS Files Form 10-K and Provides Shareholder Update • ILUS • May 2, 2024 8:52 AM
Avant Technologies Names New CEO Following Acquisition of Healthcare Technology and Data Integration Firm • AVAI • May 2, 2024 8:00 AM
Bantec Engaged in a Letter of Intent to Acquire a Small New Jersey Based Manufacturing Company • BANT • May 1, 2024 10:00 AM
Cannabix Technologies to Deliver Breath Logix Alcohol Screening Device to Australia • BLO • Apr 30, 2024 8:53 AM
Hydromer, Inc. Reports Preliminary Unaudited Financial Results for First Quarter 2024 • HYDI • Apr 29, 2024 9:10 AM










