| Followers | 248 |
| Posts | 49913 |
| Boards Moderated | 63 |
| Alias Born | 08/29/2007 |
Sunday, August 06, 2023 11:57:59 AM
GoSymbol lookup
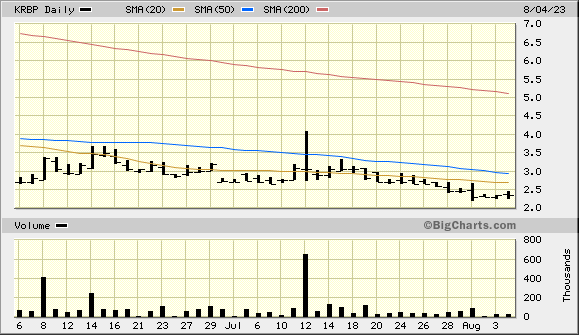
*Nasdaq FSI: *Deficient: Issuer Failed to Meet NASDAQ Continued Listing Requirements
Health Care : Biotechnology | Company profile
Kiromic BioPharma, Inc. is an artificial intelligence (AI) driven, end-to-end allogenic cell therapy company. The Company is engaged in developing the multi-indication allogeneic T cell therapy that exploits the natural potency of the Gamma Delta T cell (GDT) to target solid tumors. The Procel, Isocel, and Deltacel product platform candidates consist of allogeneic cell therapy candidates that are in the preclinical development stage. Its Procel product candidate consists of engineered GDTs targeting PD-L1. Its Isocel product candidate consists of engineered GDTs targeting Mesothelin Isoform 2 positive tumors (Iso-Meso). Its Deltacel product candidate consists of non-engineered GDTs used to treat solid tumors. Its Allogenic Lead Exogenous Isoforms (ALEXIS)-PRO-1 trial candidate is its allogeneic GDT therapy product targeting PD-L1. Its ALEXIS-ISO-1 trial candidate is its allogenic GDT therapy product targeting an isoform of Mesothelin that is present in tumor cells, namely Isomeso.
I am writing a book, American Cars of 1958. Check often for the latest addition. https://investorshub.advfn.com/American-Cars-of-1958-37252/
Recent KRBP News
- Kiromic BioPharma Reports Consistent Favorable Safety, Tolerability, and Efficacy in Deltacel-01 Clinical Trial First Three Patients • Business Wire • 04/24/2024 12:08:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/19/2024 08:30:26 PM
- Form RW - Registration Withdrawal Request • Edgar (US Regulatory) • 04/10/2024 08:30:09 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/05/2024 08:30:28 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/02/2024 08:30:20 PM
- Kiromic BioPharma Reports Favorable Safety, Tolerability, and Early Efficacy from Third Patient in Deltacel-01 Clinical Trial • Business Wire • 04/02/2024 12:18:00 PM
- Form 10-K/A - Annual report [Section 13 and 15(d), not S-K Item 405]: [Amend] • Edgar (US Regulatory) • 03/21/2024 09:21:31 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 03/20/2024 09:58:13 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 03/20/2024 09:57:02 PM
- Early Efficacy Results Show Primary Lung Tumor Stabilization and Complete Disappearance of Brain Metastases in Second Patient in Kiromic BioPharma’s Deltacel-01 Clinical Trial • Business Wire • 03/19/2024 12:08:00 PM
- Kiromic BioPharma Adds Virginia Oncology Associates, PC as the Second Clinical Trial Site in the Ongoing Phase 1 Deltacel-01 Trial • Business Wire • 03/01/2024 01:08:00 PM
- Kiromic BioPharma to Present at the OTC Markets’ Virtual Life Science Investor Forum on March 7th • Business Wire • 02/28/2024 11:30:00 PM
- Kiromic BioPharma to Present at the OTC Markets’ Virtual Life Science Investor Forum on March 7th • GlobeNewswire Inc. • 02/28/2024 09:09:06 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/14/2024 09:30:45 PM
- Results from First Patient in Kiromic BioPharma’s Deltacel-01 Clinical Trial Indicate Tumor Reduction at Two Months • Business Wire • 02/14/2024 01:08:00 PM
- Kiromic BioPharma Reports Favorable Early Efficacy Results from First Patient in Deltacel-01 Phase 1 Clinical Trial in Non-Small Cell Lung Cancer • Business Wire • 01/29/2024 12:47:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/11/2024 09:15:21 PM
- Kiromic BioPharma Reports Favorable Early Safety and Tolerability Data from First Patient Enrolled in the Phase 1 Deltacel-01 Clinical Trial • Business Wire • 01/05/2024 01:08:00 PM
- Kiromic BioPharma Announces First Patient Dosed in Phase 1 Study Evaluating Deltacel™ for the Treatment of Non-Small Cell Lung Cancer • Business Wire • 12/14/2023 01:17:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/09/2023 02:49:14 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 09/13/2023 08:45:22 PM
- Kiromic BioPharma Common Stock to Begin Trading on the OTC Pink Sheets on September 14 • Business Wire • 09/13/2023 08:35:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/29/2023 08:15:48 PM
- Form S-8 - Securities to be offered to employees in employee benefit plans • Edgar (US Regulatory) • 08/15/2023 08:16:11 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2023 08:52:28 PM
VAYK Discloses Strategic Conversation on Potential Acquisition of $4 Million Home Service Business • VAYK • May 9, 2024 9:00 AM
Bantec's Howco Awarded $4.19 Million Dollar U.S. Department of Defense Contract • BANT • May 8, 2024 10:00 AM
Element79 Gold Corp Successfully Closes Maverick Springs Option Agreement • ELEM • May 8, 2024 9:05 AM
Kona Gold Beverages, Inc. Achieves April Revenues Exceeding $586,000 • KGKG • May 8, 2024 8:30 AM
Epazz plans to spin off Galaxy Batteries Inc. • EPAZ • May 8, 2024 7:05 AM
Moon Equity Holdings, Corp. Announces Acquisition of Wikolo, Inc. • MONI • May 7, 2024 9:48 AM










