| Followers | 251 |
| Posts | 50151 |
| Boards Moderated | 47 |
| Alias Born | 08/29/2007 |
Thursday, July 27, 2023 5:41:09 AM
GoSymbol lookup
*Nasdaq FSI: *Deficient: Issuer Failed to Meet NASDAQ Continued Listing Requirements
Health Care : Biotechnology | Company profile
Carmell Therapeutics Corporation is a regenerative medicine platform company. The Company is engaged in the development of allogeneic plasma-based bioactive material (PBM) that is designed to boost innate regenerative pathways across a variety of bone and soft tissue indications. The PBM technology is licensed from Carnegie Mellon University and claims the ability to plasticize whole plasma and cross link with genipin, a derivative of the gardenia plant, to provide a controlled degradation profile in vivo. The Company’s lead product, CT-101 Bone Healing Accelerant (BHA), a biologic, is designated by the United States Food and Drug Administration (FDA) as a combination product, containing the Company’s core technology of PBM plus B Tri-Calcium Phosphate (B-TCP) an already approved medical device. The Company has FDA clearance for a Phase II-stage clinical trial designed to study accelerated healing and reduced infections in open tibia (shinbone) fractures with intramedullary rodding.
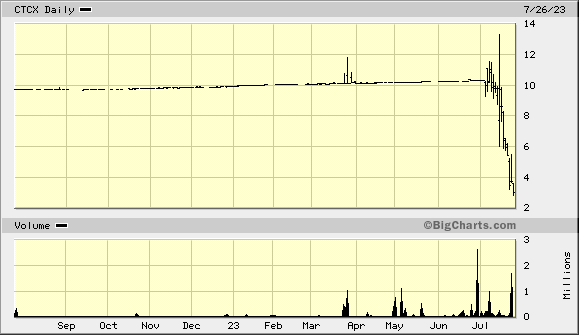
Carmell Therapeutics Corp NASDAQ: CTCX*
GoSymbol lookup
*Nasdaq FSI: *Deficient: Issuer Failed to Meet NASDAQ Continued Listing Requirements
Health Care : Biotechnology | Company profile
Carmell Therapeutics Corporation is a regenerative medicine platform company. The Company is engaged in the development of allogeneic plasma-based bioactive material (PBM) that is designed to boost innate regenerative pathways across a variety of bone and soft tissue indications. The PBM technology is licensed from Carnegie Mellon University and claims the ability to plasticize whole plasma and cross link with genipin, a derivative of the gardenia plant, to provide a controlled degradation profile in vivo. The Company’s lead product, CT-101 Bone Healing Accelerant (BHA), a biologic, is designated by the United States Food and Drug Administration (FDA) as a combination product, containing the Company’s core technology of PBM plus B Tri-Calcium Phosphate (B-TCP) an already approved medical device. The Company has FDA clearance for a Phase II-stage clinical trial designed to study accelerated healing and reduced infections in open tibia (shinbone) fractures with intramedullary rodding.
I am writing a book, American Cars of 1958. Check often for the latest addition. https://investorshub.advfn.com/American-Cars-of-1958-37252/
Recent CTCX News
- Carmell Announces Successful Closing of $3.0 Million Private Placement • GlobeNewswire Inc. • 04/11/2024 08:30:00 PM
- Carmell Announces $3.0 Million Private Placement • GlobeNewswire Inc. • 04/04/2024 12:30:00 PM
- Carmell Announces Successful Closing of Axolotl Biologix Sale Transaction • GlobeNewswire Inc. • 03/27/2024 01:15:00 PM
- Carmell Sharpens Aesthetics Focus with Execution of Definitive Agreement to Sell Axolotl Biologix Subsidiary • GlobeNewswire Inc. • 03/20/2024 08:39:36 PM
- Carmell to Host Investor Webinar, “Unveiling the Secretome: On the Eve of Product Launch” Featuring Key Opinion Leaders in the Medical Aesthetics Field • GlobeNewswire Inc. • 03/18/2024 07:16:22 PM
- Carmell Announces Product Development Completed for G.L.E.E. Launch in March 2024 • GlobeNewswire Inc. • 02/26/2024 01:30:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/05/2024 09:39:59 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/19/2023 10:00:05 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/18/2023 10:13:12 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/18/2023 10:12:55 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/14/2023 02:38:22 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/14/2023 02:37:00 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/09/2023 02:12:12 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/09/2023 02:08:51 AM
- Form 3 - Initial statement of beneficial ownership of securities • Edgar (US Regulatory) • 12/08/2023 09:46:47 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/01/2023 01:32:02 AM
- Carmell Corp Announces Addition of Scott Frisch and Gilles Spenlehauer to Board of Directors • GlobeNewswire Inc. • 11/16/2023 01:30:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/16/2023 02:54:32 AM
- Form NT 10-Q - Notification of inability to timely file Form 10-Q or 10-QSB • Edgar (US Regulatory) • 11/15/2023 09:56:30 PM
Greenlite Ventures Inks Deal to Acquire No Limit Technology • GRNL • May 17, 2024 3:00 PM
Music Licensing, Inc. (OTC: SONG) Subsidiary Pro Music Rights Secures Final Judgment of $114,081.30 USD, Demonstrating Strength of Licensing Agreements • SONGD • May 17, 2024 11:00 AM
VPR Brands (VPRB) Reports First Quarter 2024 Financial Results • VPRB • May 17, 2024 8:04 AM
ILUS Provides a First Quarter Filing Update • ILUS • May 16, 2024 11:26 AM
Cannabix Technologies and Omega Laboratories Inc. enter Strategic Partnership to Commercialize Marijuana Breathalyzer Technology • BLO • May 16, 2024 8:13 AM
Avant Technologies to Revolutionize Data Center Management with Proprietary AI Software Platform • AVAI • May 16, 2024 8:00 AM










