Thursday, September 14, 2017 6:23:42 PM
Upcoming Milestones
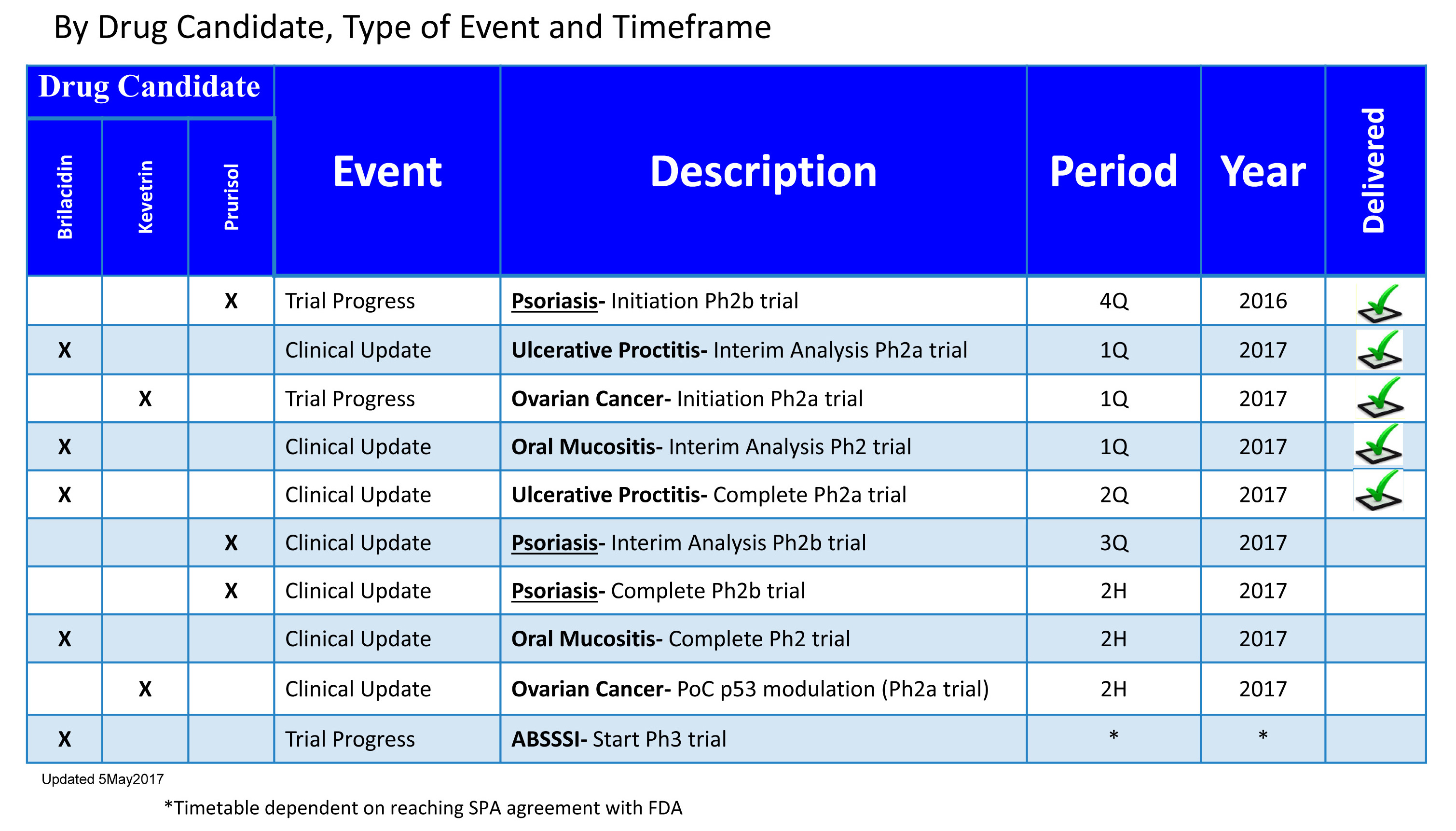
Past Milestones
Kevetrin
- Positive primary endpoint and p21 data (Phase 1)
http://www.ipharminc.com/press-release/2016/11/12/assay-results-from-cellceutix-phase-1-clinical-trial-of-kevetrin-for-cancer-show-increased-p21-expression-in-675-of-evaluable-patients
- Orphan drug designation for ovarian cancer
http://www.ipharminc.com/press-release/2016/11/16/cellceutixs-kevetrin-receives-fda-orphan-drug-designation-for-the-treatment-of-ovarian-cancer
- Orphan drug designation for pancreatic cancer
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-receives-fda-orphan-drug-designation-for-kevetrin-for-the-treatment-of-pancreatic-cancer
- Orphan drug designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-fda-orphan-drug-designmation-for-kevetrin-for-the-treatment-of-retinoblastoma
- Rare pediatric disease designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-rare-pediatric-disease-designation-from-fda-for-kevetrin-for-the-treatment-of-retinoblastoma
Brilacidin
- B-UP positive top-line data (Phase 2 POC)
http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
- B-OM encouraging interim data (Phase 2)
http://www.ipharminc.com/press-release/2017/3/27/cellceutix-reports-very-encouraging-interim-analysis-of-phase-2-drug-candidate-brilacidin-for-severe-oral-mucositis-om-in-head-and-neck-cancer-patients-high-potential-for-preventative-treatment
- B-ABSSSI positive top-line data (Phase 2b)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-positive-top-line-data-from-phase-2b-absssi-trial-single-dose-brilacidin-comparable-to-7-days-of-daptomycin
- QIDP designation for B-ABSSSI
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-antibiotic-brilacidin-receives-qidp-designation-from-fda
- Fast track designation for B-OM
http://www.ipharminc.com/press-release/2016/11/16/fda-grants-fast-track-designation-to-cellceutixs-brilacidin-om-for-oral-mucositis
- MTA extension for prophylactic testing in implanted devices
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-completes-lab-testing-of-brilacidin-for-planned-phase-3-trial-for-acute-bacterial-skin-and-skin-structure-infections
- Formulation stable at room temperature
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-breakthrough-in-the-formulation-of-novel-antibiotic-brlaicidin-plans-studies-to-treat-diabetic-foot-ulcers
- American Society for Microbiology journal publication
http://aac.asm.org/content/early/2014/06/11/AAC.02955-14#corresp-1
Prurisol
- Positive top-line data (Phase 2a)
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
- Positive primary endpoint (Phase 1)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-anti-psoriasis-drug-prurisol-meets-primary-endpoint-of-clinical-trial
- 505(b)(2) pathway
http://www.ipharminc.com/press-release/2016/11/17/cellceutix-informed-by-fda-that-505b2-approval-would-be-an-acceptable-approach-for-its-psoriasis-drug
Clinical Trials
1. K-OC (Phase 2a, Recruiting)
https://clinicaltrials.gov/ct2/show/NCT03042702?term=cellceutix&rank=1
2. P (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT02101216?term=cellceutix&rank=2
3. B-OM (Phase 2, Active, not recruiting)
https://clinicaltrials.gov/ct2/show/NCT02324335?term=cellceutix&rank=3
4. P (Phase 2a, Completed)
https://clinicaltrials.gov/ct2/show/NCT02494479?term=cellceutix&rank=4
5. P (Phase 2b, Active, not recruiting)
https://clinicaltrials.gov/ct2/show/NCT02949388?term=cellceutix&rank=5
6. B-ABSSSI (Phase 2b, Completed)
https://clinicaltrials.gov/ct2/show/NCT02052388?term=cellceutix&rank=6
7. K (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT01664000?term=cellceutix&rank=7
8. B-UP (Phase 2 POC, Completed)
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-receives-update-on-first-patient-enrollment-in-phase-2-proof-of-concept-study-of-brilacidin-for-ulcerative-proctitis
Pipeline
Recent Interviews
- 2017 Q3 Conference Call Transcript
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
- 2017 Q1 Conference Call Transcript
http://seekingalpha.com/article/4023682-cellceutix-corporation-ctix-ceo-leo-ehrlich-q1-2017-results-earnings-call-transcript
- Interview with New Cellceutix President Dr A Bertolino
http://seekingalpha.com/article/3988240-interview-new-cellceutix-president-dr-bertolino
- Cellceutix Pipeline Continues to Hit Milestone After Milestone
https://www.streetwisereports.com/pub/na/cellceutix-pipeline-continues-to-hit-milestone-after-milestone
- A Small-Cap Biotech with Big Ideas for Acute Infections and Cancers: Cellceutix's Leo Ehrlich and Dr. Daniel Jorgensen
http://www.thelifesciencesreport.com/pub/na/a-small-cap-biotech-with-big-ideas-for-acute-infections-and-cancers-cellceutixs-leo-ehrlich-and-dr-daniel-jorgensen
Corporate Overview
- BioCentury NewsMakers Conference https://static1.squarespace.com/static/5715352e20c647639137f992/t/59b2f34946c3c498ebfe112e/1504899917915/IPIX+Company+Presentation+BioCentury+NewsMakers+Conference++8Sep2017.pdf
- Drug Discovery and Therapy World Congress
https://static1.squarespace.com/static/5715352e20c647639137f992/t/596cac7df14aa1a6e7118ed8/1500294274057/IPI+DDTWC+Brilacidin+presentation+%28final-u%29+13Jul2017.pdf
- July 2017
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5967d1c92994ca6bed362ade/1499976142099/2017+July+IPIX+Corporate+Overview.pdf
Patents
- Kevetrin
https://www.google.com/patents/US8338454
- Brilacidin
https://www.sec.gov/Archives/edgar/data/1355250/000147793214002418/ctix_ex1037.htm
https://www.google.com/patents/US20160243117
https://www.google.com/patents/EP2709619A2
- Prurisol
https://www.google.com/patents/WO2013103601A1
MP Advisors Report
https://drive.google.com/file/d/0Bz15O4eaX-asWVpoclB2WUhENFk/view
Abbreviations
K – Kevetrin
B – Brilacidin
P – Prurisol
OC – ovarian cancer
ABSSSI – acute bacterial skin and skin structure infections
OM – oral mucositis
UP – ulcerative proctitis
QIDP – qualified infectious disease product
Recent IPIX News
- Form 8-K - Current report • Edgar (US Regulatory) • 02/01/2024 01:30:25 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 12/05/2023 09:25:58 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/20/2023 09:05:44 PM
- Form NT 10-Q - Notification of inability to timely file Form 10-Q or 10-QSB • Edgar (US Regulatory) • 11/15/2023 01:00:19 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2023 08:15:25 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 09/28/2023 01:00:08 PM
Lingerie Fighting Championships Signs Broadcast Deal With Maybacks Global Entertainment • BOTY • Sep 26, 2024 9:00 AM
Maybacks Global Entertainment and Lingerie Fighting Championships Enter Into Broadcast And Revenue Sharing Agreement • AHRO • Sep 26, 2024 8:30 AM
North Bay Resources Commences Operations at Bishop Gold Mill, Inyo County, California; Engages Sabean Group Management Consulting • NBRI • Sep 25, 2024 9:15 AM
CEO David B. Dorwart Anticipates a Bright Future at Good Gaming Inc. Through His Most Recent Shareholder Update • GMER • Sep 25, 2024 8:30 AM
Cannabix Technologies and Omega Laboratories Inc. Advance Marijuana Breathalyzer Technology - Dr. Bruce Goldberger to Present at Society of Forensic Toxicologists Conference • BLOZF • Sep 24, 2024 8:50 AM
Integrated Ventures, Inc Announces Strategic Partnership For GLP-1 (Semaglutide) Procurement Through MedWell USA, LLC. • INTV • Sep 24, 2024 8:45 AM







