Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and we are not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
PRELIMINARY PROSPECTUS SUBJECT TO COMPLETION, DATED AUGUST 21, 2018
Shares of Common Stock
Pre-Funded Warrants to Purchase Shares of Common Stock
We are offering ???? shares of our common stock. We are also offering to certain purchasers whose purchase of shares of common stock in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if any such purchaser so chooses, pre-funded warrants, in lieu of shares of common stock that would otherwise result in such purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. The purchase price of each pre-funded warrant will be equal to the price per share at which shares of common stock are sold to the public in this offering, minus $0.01, and the exercise price of each pre-funded warrant will be $0.01 per share. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants sold in this offering. The pre-funded warrants will be exercisable immediately and may be exercised at any time until all of the pre-funded warrants are exercised in full. For each pre-funded warrant we sell, the number of shares of common stock we are offering will be decreased on a one-for-one basis. Our common stock is listed on the NYSE American under the symbol “APHB.” On August 20, 2018, the last reported sale price of our common stock on the NYSE American was $1.16 per share. The public offering price per share and any pre-funded warrant will be determined between us and the underwriter at the time of pricing, and may be at a discount to the current market price. There is no established public trading market for the pre-funded warrants, and we do not expect a market to develop. In addition, we do not intend to apply for a listing of the pre-funded warrants on any national securities exchange or other nationally recognized trading system.
We are an “emerging growth company” as that term is used in the Jumpstart Our Business Startups Act of 2012 and, as such, we have elected to comply with certain reduced public company reporting requirements for this prospectus and future filings.
Need to see what they will be setting the share price at, could weigh on stock. I still have large holdings, but no real sense of current trials until further progression. I`m in it for a trade that is taking longer to prove out than expected....Thin float makes this attractive, but little daily volume.......
Thinly traded nano cap AmpliPhi Biosciences (NYSEMKT:APHB) has filed a preliminary prospectus for a $12M public offering of stock and warrants. Prices, volumes and terms have yet to be determined.
https://www.sec.gov/Archives/edgar/data/921114/000157104918000447/tv501435_s1.htm
There are 30 states that have given the green light to medical marijuana, with the heavily “red” state of Oklahoma becoming the latest to send the smoke signals for sales. The industry is also witnessing nine of these 30 states give the go ahead for adult use of cannabis, including Vermont, which became the first state to approve recreational marijuana entirely through the legislative process in January.
The passage of Canada’s Parliament bill C-45 will allow Canada to become the first industrialized country in the world to legalize recreational marijuana and in doing so, add roughly $5 billion in annual sales.
If you’re an investor watching the marijuana industry grow, make sure to be paying close attention to this market especially during the next few months leading up to Canada’s time when the “switch is flipped” on for legal recreational marijuana retail sales at a national (and international) level.
https://smallcapreporter.wordpress.com/2018/08/14/canadas-big-boom-marijuana-stocks-to-watch/
Way overdone triple bottom shoot through. Will it go lower? Adding again down here to lower my cost basis......
Highlights from quarter going forward:
Key Remaining 2018 Milestones
Present top-line overall response rate results from our UNITY-CLL Phase 3 trial in front line and relapsed or refractory Chronic Lymphocytic Leukemia (CLL).
Prepare and potentially file the Company’s first BLA and/or NDA.
Complete enrollment in the current arms of the UNITY-NHL trial, including the Follicular Lymphoma, Marginal Zone Lymphoma, and Diffuse Large B-Cell Lymphoma cohorts.
Present updated clinical data from ongoing oncology trials and final results from the Phase 2 trial of ublituximab in Multiple Sclerosis (MS) at major medical meetings during 2018.
Financial Results for the Second Quarter 2018
Cash Position: Cash, cash equivalents, investment securities, and interest receivable were $126.3 million as of June 30, 2018.
Thanks, they all have skin in the game and Michael S. Weiss has at this point run the company up to SEC standards. He may not of run a very forthright investment firm but has done well with $TGTX and in the article link Weiss served as chairman of the National Holdings board from shortly after Fortress acquired a controlling stake in the company until he resigned in June. I have no problem with any conflicts of interest otherwise.
Can you point to where this is the case in any respects? TY
Reads like a hit piece!!!!! Nothing inditing Mike and his management of TGTX......
No opinion. I still believe in this market that most all the MJ plays will in time be strong winners going forward. It's early on still for the whole sector so there will be a lot of gains going forward after this consolidation.
This sell off/consolidation has really been going on since late February and being the newest disruptive play people still need reasons to buy. Problem is this will take time now to grow into the fundamentals so not a lot of catalyst for traders and time now is on the side of buy the dips and hold people IMO........
Added more today !!!!!!!!!!!
Added a few at $26.50 to bring down my current cost basis. It's all about time and disruption. Now it's sit and wait out all the future money makers in the fund.....Looked to be shorts covering down here finally, sure will see if correct !!!!!
No I got stopped out. Not buying back so moved on. GL
Very likely, this rolling bear has been a bit long in the tooth. Been accumulating throughout so it’s eased my cost average. This is one of my few long term plays so not looking to trade.
No opinion as I have trouble understanding this bio any more. GL
MakinBank
I posted this on the thread last night after market close:
Anyone that read it was able to get out or in using after hours trading......
$AVEO Oncology Shifts TIVO-3 Topline Data for Renal Cancer to Q4 2018 -
Stock Slumps 32% After-Hours
04:39 PM EDT, 07/16/2018 (MT Newswires) -- AVEO Oncology (AVEO) said on Monday it now expects to report TIVO-3 topline data in Q4 of 2018 due to the progression-free survival events (PFS) events occurring slower than forecasted, combined with some patients being removed from the PFS event count, which often occurs once the data cleaning and review process is initiated.
Prior to reporting data, Aveo said it plans to announce when 255 PFS events have occurred and topline data analysis for the trial has been initiated.
The company had expected to report topline data from the TIVO-3 trial, its phase 3 trial of tivozanib as a third-line treatment for advanced renal cell carcinoma, 6-8 weeks after the trial records 255 PFS events.
Price: 1.93, Change: -0.92, Percent Change: -32.28
Too much risk for this play, out.......
$AVEO Oncology Shifts TIVO-3 Topline Data for Renal Cancer to Q4 2018 - Stock Slumps 32% After-Hours
04:39 PM EDT, 07/16/2018 (MT Newswires) -- AVEO Oncology (AVEO) said on Monday it now expects to report TIVO-3 topline data in Q4 of 2018 due to the progression-free survival events (PFS) events occurring slower than forecasted, combined with some patients being removed from the PFS event count, which often occurs once the data cleaning and review process is initiated.
Prior to reporting data, Aveo said it plans to announce when 255 PFS events have occurred and topline data analysis for the trial has been initiated.
The company had expected to report topline data from the TIVO-3 trial, its phase 3 trial of tivozanib as a third-line treatment for advanced renal cell carcinoma, 6-8 weeks after the trial records 255 PFS events.
Price: 1.93, Change: -0.92, Percent Change: -32.28
http://www.mtnewswires.com
July 16, 2018
4:38 pm ET
*Aveo Pharma 8-K Shows Warrant Deal With Computershare, Computershare Trust For Purchase Of 2M Shares Of Common Stock Related To Terms Of Stipulation Of Settlement OF Securities Class Action Suit
Benzinga
July 03, 2018
7:45 am ET
*AVEO Pharmaceuticals Received Notice From Novartis On Jun. 29th Of A Termination Of The AV-380 License Agreement Dated August 13, 2015; Termination Will Be Effective Aug. 28 And Will Not Impact Co.'s Cash Guidance
Newton's Law Of Inertia, that states "an object in motion tends to remain in motion unless acted upon by an unbalanced force."
In other words, when markets begin strongly trending in one direction, that direction will continue until an "unbalanced" force stops it. Momentum strategies, which are trend following strategies by nature, have been proven to work well across extreme market environments, multiple asset classes and over historical time frames.
$TNA Direxion Small Cap Bull 3X to bust out to new highs this coming week: The $IWMs have fallen back after attempting to hit a new high in the upper 160s, looks to be an inverted head and shoulders setup om $TNA & $IWM charts.
Overall, the technical side of things is positive. EMAs are advancing and momentum is rising. Combine that with the potential support of a strong earnings season, we could see this rally continue into the summer.

Melinta Therapeutics: Could The H.R. 6294 Bill Improve The Prospects Of Antibiotic Innovators?
Jul. 5, 2018 11:45 AM ET|5 comments | About: Melinta Therapeutics (MLNT), Includes: CDTX, MDCO, PRTK, TTOO
BioSci Capital Partners
BioSci Capital Partners
Integrated biosci research, consultant
MARKETPLACEIntegrated BioSci Investing
Summary
Despite that most companies in our various Specialty Reports have outperformed, the performance of the infectious disease innovators like Melinta are subpar to date.
There are hurdles that deter newly approved antibiotics to generate blockbuster sales.
Novel antibiotics are maintained as the last-line reserves to prevent antibiotic resistance.
Stellar new policies pioneered by the FDA Commissioner (Dr. Scott Gottlieb) can significantly improve the fundamentals for these companies.
We present a fundamental analysis and pertinent updates on Melinta. And we wish to share this article with all readers in celebration of July 4th.
This idea was discussed in more depth with members of my private investing community, Integrated BioSci Investing.
Life will have terrible blows in it, horrible blows, unfair blows. It doesn't matter. And some people recover and others don't. And there I think the attitude of Epictetus is the best. He thought that every missed chance in life was an opportunity to behave well. Every missed chance in life was an opportunity to learn something and that your duty was not to be submerged in self-pity. But instead to utilize the terrible blow in constructive fashion. That is a very good idea. - Charlie Munger
We recently increased our focus on the Specialty Reports, as part of our increasing emphasis to cover various niches in bioscience for subscribers of Integrated BioSci Investing. Most equities in our five Specialty Reports have outperformed the market. Nevertheless, the infectious disease innovators - Melinta Therapeutics (MLNT), Paratek Pharmaceuticals (PRTK), Cidara Therapeutics (CDTX), Achaogen (AKAO), and T2 Biosystems (TTOO) - are lagging behind. Regardless of a heighten demand for novel anti-infectious agents, there is a key reimbursement issue that makes investing in this niche unprofitable. Interestingly, highly promising policy changes - initiated by the stellar FDA Commissioner (Dr. Scott Gottlieb) - can substantially improve the investing prospects of these firms while delivering hopes to patients. In this research, we'll feature an update of our investing thesis on Melinta.
Figure 1: Melinta stock chart. (Source: StockCharts).
About The Company
First thing first, we'd like to briefly go over the background on Melinta for new investors. If you are already familiar with the firm, we recommend that you skip to the next section on the H.R. 6294 Bill. Melinta Therapeutics operates out of Chapel Hill, NC. The company is the product of the merger with Cempra that completed on Nov. 06, 2017. The reorganization gave Melinta and Cempra stockholders 51.6% and 48.4% ownership of the new company, respectively. We explicated in the prior research:
With the FDA approved delafloxacin (Baxdela) from Cempra, Melinta now has more option to execute the mission, in developing and commercializing antibiotics for the treatment of serious infectious diseases. Moreover, the mutually beneficial union enabled Cempra with Melinta's abundant cash and resources. Accordingly, the firm now has a highly enriched pipeline of antibiotics to service the infectious diseases market (one that is strongly in demand of better molecules to treat bugs increasingly-resistant to available medicines).
Figure 2: Therapeutic Pipeline. (Source: Melinta Therapeutics). Note: ABSSSI: Acute bacterial skin and skin structure infections || CABP: Community-acquired bacterial pneumonia || cUTI: Complicated urinary tract infections || ESKAPE: The ESKAPE pathogen program includes a focus on multidrug- and extremely-drug-resistant versions of the following pathogens: Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter species and Escherichia coli || QIDP: Qualified Infectious Disease Product.
Increasing Antibiotic Resistance
With rising elderly population, more people are becoming sick and thereby increases antibiotic use. The more prescription, the higher the chances that bacteria will develop resistance. The statistics from the Center for Disease Control and Prevention ("CDC") showed that annually, there are at least 2M people who get serious bacterial infection requiring one or more antibiotics. Due to their increasing utility, the "work-horse" antibiotics that we currently have will become obsolete in the future.
From the economic view, antibiotic-resistant raises the financial burdensome on the U.S.'s healthcare spending. Accordingly, to the CDC's Antibiotic Resistance Threat Report, the total annual cost of antibiotic resistance amounts to the staggering $20B. Moreover, the additional society costs from productivity loss can be as high as $35B.
Reimbursement Keeping The Lid On Melinta Shares
Notably, the reimbursement for a novel antibiotic is strikingly different from that of other therapeutics. Physicians are strongly encouraged to prescribe a newly approved drug. Therefore, this translates into increased earnings for the company to offset (the approximate $1B used to innovate a molecule from bench research to commercialization).
In contrast, a newly approved antibiotic is being kept as the last-line reserve in the effort to deter resistance. Hence, this lowers the sales volume. Without much sales, the companies cannot get back their capital invested. Ultimately, the current situation will deter further innovation that, in and of itself, is much needed in this space. And, the health consequences will boil down to an increased mortality and morbidity for us all.
H.R. 6294 To Potentially Ameliorate The Situation
In the prior Rounds Report, we note that the FDA is working with Congress in the development and launch of various campaigns to help the antibiotic innovators. One policy is the "qualified infectious disease product ("QIDP") designation. Interestingly, new applications considered as QIDP can receive the fast track designation, priority review, and possibly a 5-year extension of any exclusivity. Second is the new ("LPAD") pathway that streamlines the approval process. LPAD requires a much smaller and shorter clinical trial yet still ensuring the adequate safety for approval.
That aside, the most notable development is the new incentive program being pushed by the FDA Commissioner (Dr. Scott Gottlieb): hospitals can buy the newly-approved antibiotics for a flat licensing fee rather than conventional sales based reimbursement. Under the new structure, a company can procure significant revenues on a novel antibiotic (regardless of how much of the drug is being prescribed). Just weeks into Dr. Gottlieb's announcement, notable politicians drafted a new bill that pushes for greater exclusivity extension for new antibiotics while favoring the flat licensing fee structure. According to a research by BioCentury:
Two members of the House Energy and Commerce Committee introduced legislation that would create a new "pull" incentive pathway for priority antibiotics, providing manufacturers a transferable award that could be worth $1B or more. The bill, the Re-Valuing Anti-Microbial Products (REVAMP) Act of 2018, aims to de-link the returns an antibiotics company receives from the volume of drugs sold by providing a high-value award upon approval of antibiotics to treat multidrug-resistant pathogens. The bill, H.R. 6294, is sponsored by Rep. John Shimkus (R-Ill.) and co-sponsored by Rep. Tony Cárdenas (D-Calif.).
Of note, it is not far from the truth that the whole process of drafting a bill to its passage into its approval can take 2-3 weeks. And yet, only 10% to 12% of the bills are eventually voted into laws. Therefore, the statistics are seemingly not favorable. Despite their small chances of success, Dr. Gottlieb is known for getting things done. His skills and determination to approve more drugs, ensuring public safety and access to medicine are stellar. If H.R. 6294 eventually becomes law, this can be a major catalyst for the infectious disease innovators like Melinta.
Financial Assessment
On May 08, 2018, Melinta reported the Q1 2018 earnings (that ended on March 31). As follows, the company posted $11.8M in product sales. This included the addition of Vabomere, Minocin, and Orbactiv as of Jan. 5, 2018, from The Medicines Company (MDCO) acquisition. Per figure 3, the total revenues came in at $14.8M compared to $22.5M for the same period a year prior. The higher figure for last fiscal was due to the upfront payment from Menarini (re the ex-US Baxdela licensing agreement).
Figure 3: Key financial metrics for Q1 2018. (Source: Melinta)
The research and development (R&D) logged in at $16.1M compared to $12.9M for the similar comparison. The higher spending was due to the more employee count (as a result of the Cempra merger and the new business from The Medicine Company). Moreover, the ongoing community-acquired bacterial pneumonia ("CABP") registration trial for Baxdela added more cost. For the respective periods, there were $29.4M ($0.95 per share) and $5.8M in net losses.
Pertaining to the balance sheet, there were $91.5M in cash and equivalent for Q1. The firm also raised $123M in the offering of 22.0M common shares on May 29. Therefore, the cash position is substantially improved to $214.5M. Based on the $34.6M quarterly burn rate, there should be adequate funding for operations into early 2020 (prior to any additional offering).
Potential Risks
As with any investment, there are pertinent risks. At this point in its growth cycle, the main concern for Melinta is whether various approved molecules will generate increasingly significant sales. As elucidated, the hurdles to blockbuster sales for newly approved antibiotics are nearly insurmountable. The chances of H.R. 6294 to pass is quite low. Nevertheless, if it can be passed, the fundamentals of the company will increase by leaps and bounds. The other risk is the increasing cash burn that will subject the stock to further dilution.
Conclusions
In all, we maintain our buy recommendation on Melinta but lowered its stars rating to a three out of five stars rating. And, we decreased the price target to $10 (to be reached within two to three years). As a merger with Cempra, Melinta has an ample cash to fund further innovation. The cash position was strengthened with the public offering that raised $123M. The three molecules acquired from The Medicine Company (Orbactiv, Minocin, and Vabomere) enabled Melinta to leverage on The Medicine Company's sales team to launch its newly approved molecule (Baxdela) for ABSSSI. The various programs being pushed by the FDA serve as the catalysts to improve the company's fundamentals. The most important is H.R. 6294 that was recently introduced. Despite its small chances of being passed, if it gets voted into law the investing story on Melinta can change drastically. Of note, our reservation on raising the star rating is due to the small chances of passage for the aforesaid bill (and the reimbursement issue centering the infectious disease innovators).
Last but not least, we wish to send our July 4th greeting to our subscribers and all readers. May we remember those who sacrificed their lives for our freedom. God bless America and those who sacrificed for us.
Author's Notes: We're honored that you took the time to read our market intelligence. Founded by Dr. Hung Tran, MD, MS, CNPR, (in collaborations with Analyst Vu, and other PhDs), Integrated BioSci Investing ("IBI") is delivering stellar returns. To name a few, Nektar, Spectrum, Atara, Madrigal, and Kite procured over 155%, 177%, 279%, 258%, and 83% profits, respectively. Our secret sauce is extreme due diligence with expert data analysis. The service features a once-weekly exclusive Alpha-Intelligence article, daily analysis/consulting, and model portfolios. And, we invite you to subscribe to our marketplace now to lock in the current price and save money for the future.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.
Additional disclosure: Our research articles are best used as starting points in your own due diligence. We are not registered investment advisors and our articles are not construed as professional investment advice.
Editor's Note: This article covers one or more stocks trading at less than $1 per share and/or with less than a $100 million market cap. Please be aware of the risks associated with these stocks.
https://seekingalpha.com/article/4185550-melinta-therapeutics-h-r-6294-bill-improve-prospects-antibiotic-innovators
Melinta Therapeutics: Undervalued With Upcoming Milestones
Jul. 5, 2018 12:15 PM ET|4 comments | About: Melinta Therapeutics (MLNT), Includes: MDCO Bret Jensen
Specializing in biotech stocks, Small Caps, managing optimized portfolios
MARKETPLACEThe Biotech Forum
Summary
Today we look at Melinta Therapeutics, a small biopharma concern that made a huge acquisition late last year.
The company is picking up positive analyst support and had myriad potential milestones in the second half of 2018.
We take a deeper look at the company's prospects and outlook in the paragraphs below.
This idea was discussed in more depth with members of my private investing community, The Biotech Forum.
A concept is a brick. It can be used to build a courthouse of reason. Or it can be thrown through the window.” ? Gilles Deleuze
Today we look at a 'Tier 3' biopharma concern that is undergoing a transformation due to a deal late in 2017. The shares look undervalued and ideal for accumulation or as a Buy-Write option candidate
Company Overview:
Melinta Therapeutics (MLNT) is a New Haven based commercial-stage pharmaceutical company. It is focused on discovering and commercializing various anti-infectives for the treatment of bacterial infectious diseases. The company was transformed late last year when it purchased the infectious disease business of The Medicines Company (MDCO). This transaction closed early this year. This business included approved products Vabomere, Orbactiv® (oritavancin) and Minocin® for Injection. This deal called for Melinta to pay $165 million in cash upfront. It also provided The Medicines Company with $50 million in common stock and is on the hook for $25 million in cash 12 months early in 2019 and $25 million more early next summer as well as tiered royalties on net sales. The stock currently has a market capitalization just less than $400 million and sells right at $6.50 a share.
Product Portfolio & Pipeline:
These recently acquired products delivered almost all of the just under ~$15 million in revenue the company reported in the first quarter. Melinta has an evolving pipeline as well which can be seen here.
The company should have a busy second half of 2018 and it listed numerous milestones that should take place before the year closes out on its last conference call including:
Pivotal Phase 3 data for Baxdela in CABP
Vabomere EMA regulatory approval decision
TANGO-2 additional data and potential publication
Additional ex-U.S. submissions for Baxdela in Central and South America
Ex-U.S. partnership opportunities for Vabomere, Orbactiv and Minocin for Injection
IND-enabling studies for the lead ESKAPE compound
Analyst Commentary & Balance Sheet:
Cantor Fitzgerald came out Monday with a new Outperform rating and $15 price target on Melinta Therapeutics. Cantor's analyst provide the following commentary to support his positive view on the company and stock
"We are initiating coverage of Melinta’s stock with an Overweight rating and $15 PT. Upward earnings revisions and multiple expansion will drive the shares higher, in our view. We think a greater appreciation for the company’s four commercial, antibiotic drugs (Baxdela, and its growing pipeline (new drugs, international expansion, additional indications) will increase earnings estimates to levels not yet reflected in consensus expectations. Also, MLNT is considering adjacent opportunities, which means it could become a diversified hospital company. This should improve its trading multiple, in our view.”
Prior to that, WBB Securities upgraded MLNT from Hold to Speculative Buy with a $8 price target on May 25th. Two weeks before WBB's call, Robert W. Baird upgraded this name to Outperform from Neutral with a $15 price target. This is the only analyst commentary I can find on this small cap concern so far in 2018.
The company ended the first quarter with cash and cash equivalents of $91.5 million. It then raised just under $125 million via a secondary offering in late May, bringing its cash balance just past the $200 million level.
Verdict:
The company is probably a few years from profitability. However, a transformation seems to be underway at Melinta and the stock is significantly under the median analyst price target. The company is also receiving its fair share of positive analyst commentary of late as well. With its funding needs addressed and numerous potential catalysts schedule for the rest of the year, a small stake within a well-diversified biotech portfolio seems warranted at current trading levels.
Option Strategy:Image result for Stock Purchase
I personally would accumulate an initial stake or to increase exposure to MLNT via a Buy-Write order. Using the January 2019 $7.50 call strikes, fashion a Buy-Write order with a net debit of between $5.30 to $5.40 a share range (net stock price - option premium). This mitigates some downside risk and sets up a more than solid potential return for its six and half month 'hold' period.
Those who deny freedom to others, deserve it not for themselves” ? Abraham Lincoln
Author's Note: To get these types of articles and Instablogs on attractive biotech and pharma stocks as soon as they are published, just click here for my profile. Hit the big orange "Follow" button and choose the real-time alerts option.
Disclosure: I am/we are long mdco,mlnt.
I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.
https://seekingalpha.com/article/4185554-melinta-therapeutics-undervalued-upcoming-milestones
Wish I understood more about the science behind it all. What I do understand it will likely get to market as the results will be good.
Analyst Wangzhi highlights that TOCA is a Phase 3 clinical-stage biotech company developing novel viral gene therapy for cancer. It has developed a proprietary Retroviral Replicating Vector (RRV) platform to selectively deliver therapeutic genes in tumor cells. Lead program Toca 511 & Toca FC delivers cytosine deaminase to convert pro-drug 5-FC (5-fluorocytosine) to active 5-FU (5-Fluorouracil) selectively in tumor cells, which kills the tumor cells and also induces immune response.
Li views TOCA as an attractive investment opportunity with multiple upcoming catalysts and significant upside potential:
I. We see a good probability of success (POS) for Toca 511 & Toca FC pivotal trial with potential market launch in 2020 to address the high unmet need of rHGG ($1.2B+ market), given:
1. Toca 511 & Toca FC Phase 1 trials showed good safety and strong efficacy signal indicated by: 1) 5/23 (21.7%) pivotal trial eligible rHGG pts achieved rare durable CR (38.0+ to 57.5+ m) in rHGG with impressive mOS of 14.4m vs 6-10m of current TRx; 2) Late response onset and gradual improvement from PR to CR in 2 pts is consistent with the immune mechanism of action of Toca 511 & Toca FC; 3) Trend of dose response in OS; 4) Both FDA and EMA have granted Toca 511 & Toca FC BTD and PRIME designations after their diligent review of the Phase 1 results.
2. We see a good POS for the Toca5 pivotal trial to recapitulate the promising Phase 1 results in rHGG, based on: 1) Design of Toca5 pivotal trial recapitulates key Phase 1 signal with reasonable assumptions to provide decent buffer room for success; 2) Potential variables (injection procedures, resection extent and distribution of rGBM vs rAA pts, etc.) for Toca5 outcome appear to be in good control; 3) Strong regulatory support from FDA and EMA with BTD and PRIME designations; 4) Enrollment on track to complete by YE2018 with two interim analysis to provide earlier readouts in 2H18 and 1H19.
3. Toca 511 & Toca FC results appear highly competitive among products currently in active clinical development for rGBM/rHGG (page 15).
II. We see significant upside potential of expanding Toca 511 & Toca FC into other solid tumors.
III. We also see broad upside potential of expanding RRV platform to deliver a variety of anti-cancer viral gene therapy (e.g., local aPD-L1 by Toca 521).
Interesting and should raise some plays in the ETF`s portfolio...
The Biggest Irony Behind Recreational Marijuana Legalization in Canada
Motley Fool•July 1, 2018
In 108 more days, Canada will make history. According to Prime Minister Justin Trudeau, and following the passage of the Cannabis Act (officially Bill C-45) less than two weeks ago, adult-use marijuana will go on sale in approved dispensaries throughout Canada on Oct. 17. This will make Canada only the second country in the world, and the first industrialized nation, to have legalized recreational weed.
What does legal marijuana mean from a financial perspective, you wonder? How about the addition of somewhere in the neighborhood of $5 billion in annual sales. This comes atop the revenue marijuana businesses are already generating from the sale of cannabis to medical patients in Canada and via exports to countries where medical pot has been legalized. This expected meteoric rise in sales over the short and intermediate term, along with rapidly expanding capacity, is what's lured investors in droves to Canada's pot stocks.
The irony behind Canada's marijuana legalization
Yet there's an interesting irony that lies behind the legalization of marijuana in Canada, and the expected green rush that's less than four months away from hitting the market. Namely, that dried cannabis isn't the product that growers are necessarily going to be focus on.
Don't get me wrong, in the first couple of months to perhaps a year following legalization, more traditional cannabis products are liable to be strong sellers. With the proverbial green flag waving on Oct. 17, it appears very likely that there will not be enough marijuana available to meet demand. The reason for this being that growers had to wait until it appeared certain that the Cannabis Act would pass before they spent exorbitant amounts of money on capacity expansion. This has left most pot stocks with staggered completion dates for their major capacity-expansion projects that range between the summer of 2018 and the end of 2020.
For example, Aurora Cannabis (NASDAQOTH: ACBFF) has suggested that it could lead the pack by producing 570,000 kilograms of cannabis-equivalent production yearly, when at full capacity. But when Oct. 17 rolls around, it's not going to be anywhere near this level. Its 800,000-square-foot Aurora Sky facility will be done by then, and it's capable of just over 100,000 kilograms of cannabis production annually, but its recently announced Aurora Sun facility in Alberta, and its partnered Aurora Nordic project, won't be completely online until a later date.
This is the case with pretty much every grower in Canada, which should lead to a domestic cannabis shortage through perhaps 2019. Assuming this thesis is correct, a shortage of cannabis with strong demand should buoy or lift the per-gram price for dried cannabis.
https://finance.yahoo.com/news/biggest-irony-behind-recreational-marijuana-112100826.html
Buying the dips is the only rational for me as the sector is even more established than 6 months ago, let alone 24 months back.
The whole sector is being sold as profits are locked in due to the big gains as opposed to the overall markets.
Shorts have been building positions, end of month selling and stop losses being triggered.
The future of MJ companies is still extremely bright and this short term downtrend and selling is hard to watch but if you believe, adding in here should be more rewarding long term still given no recession or Bear market happening.
Tocagen Initiated at Buy by Ladenburg Thalmann
7:48 am ET June 27, 2018 (Dow Jones)
Ratings actions from Benzinga:
Molson Coors in talks with marijuana companies - Bloomberg
Jun. 22, 2018 2:32 PM ET|By: Stephen Alpher, SA News Editor
With pot legalization looming in Canada, and aiming for growth as beer sales decline, Molson Coors (TAP +1.9%) has been in talks with up to four Canadian cannabis companies, according to Bloomberg. The discussions, says the report, are "serious," with a deal maybe announced by year-end.
Among the companies with which TAP has talked with are Aphria (OTCQB:APHQF -4.4%) and Aurora Cannabis (OTCQX:ACBFF -3.4%).
Molson Coors wouldn't be the first alcohol-related company to enter the marijuana space, but - with a market cap near $15B - it would be the largest. Constellation Brands (STZ +0.2%) last year, purchased a 9.9% stake in Canopy Growth (CGC -6.7%) for $245M.
Inpatient investors that will get out on any run up since the extended sell off. Hoping it finds its bottom and moves back up after today. Stupidally oversold again after positive news.
The breakdown and $MJ closes above its 200 MA.
June 2018 – On June 1, while debating issues related to the sale and distribution of marijuana and packaging, senators passed an amendment from Conservative Sen. Judith Seidman.
Her amendment restricts the sale of marijuana merchandise and promotional products that could appeal to youth. The amendment passed by a vote of 34 to 28.
Debate and possible changes to the bill will continue following this thematic schedule up until the scheduled vote:
Monday, June 4 senators are set to tackle international and border issues related to legalization;
Tuesday, June 5 senators will debate and bring any amendments related to criminal penalties related to the new regime; and
Wednesday June 6 senators will discuss public and mental health matters, consumption, as well as impacts from an Indigenous perspective.
Pending any unforeseen circumstances, Bill C-45, the Cannabis Act is set to be voted on, on Thursday June 7. Woo, citing the over 200 hours of testimony heard on Bill C-45, said he hopes most senators show restraint day-of and offer their contribution to the debate during one of the allotted days spelled out above. He said it would be "highly unexpected," procedurally if anything prevented senators from voting.
Even if it's not until late Thursday, senators may have some parliamentary company, as due to an unrelated government decision, the House of Commons is sitting until midnight that night.
While the Conservative contingent in the Senate has already stated it plans to oppose the passage of the bill, it is yet to be seen what the breakdown of support will be among Independent senators, many of whom have expressed concern with the bill, but have given their approval to see it pass through all stages with the intent of improving the legislation. Woo said he is planning to vote in favour of Bill C-45, because the amendments that senators have passed have improved the legislation, but insisted there will be no whipping of other Independents’ votes.
If Bill C-45 passes, given it has been amended, it will go back to the House of Commons, where the aforementioned legislative ping pong could ensue. It is yet to be seen what if any changes the Senate will insist on, but it’s expected that dealing with the bill if it is sent back will be the Senate's top priority.
Late August or early September 2018 – This is the window of time the federal government is anticipating that marijuana will be fully legalized across the country.
Immunotherapy Clinical Trial at Overlook Medical Center Reveals Brain Tumor Shrinkage, Continues with Phase 3 Study
By TAPINTO SUMMIT STAFF June 4, 2018
SUMMIT, NJ — Following successful Phase 1 clinical trials, the Toca regimen – a new immunotherapy for patients with recurrent brain cancer – has advanced to a Phase 3 study, which is currently underway. The trial is being conducted at 68 sites across the United States, Canada, Israel and South Korea, including at Overlook Medical Center’s Gerald J. Glasser Brain Tumor Center, one of three clinical trial sites in New Jersey.
In this surgical study, patients are randomized to either standard of care treatment or the Toca regimen. Enrollment is expected to complete in this trial by the end of 2018.
“Toca 5 uses a virus to stimulate a patient’s own immune system and attack recurring high-grade gliomas – glioblastoma and anaplastic astrocytoma,” explains Yaron Moshel, MD, PhD, a neurosurgeon with Atlantic Neurosurgical Specialists and co-director of the Gerald J. Glasser Brain Tumor Center who is Principal Investigator (PI) for the local arm of the study at Overlook Medical Center.
Patients randomized to the Toca regimen receive an investigational treatment that involves two discrete steps. During the first step, patients receive Toca 511 (vocimagene amiretrorepvec), a replicating virus that selectively infects cancer cells, at the time of surgery. After a few weeks, they then take cycles of Toca FC (extended-release 5-fluorocytosine), a potent anti-cancer pill that kills cancerous cells and activates immune cells selectively against cancerous ones, leaving healthy cells unharmed.
“Data from Phase 1 testing of the Toca regimen showed a favorable safety profile, complete tumor shrinkage, and extended patient longevity compared to other therapies,” Dr. Moshel continues.
With the current standard of care, newly diagnosed patients with glioblastomas have a median survival of approximately 14 to 16 months. After recurrence, median survival is typically seven to nine months.
“In contrast, Phase 1 results of the immunotherapy trial showed median longevity of 14.4 months for patients with recurrent disease and patients with complete tumor shrinkage are still alive almost three years after starting the Toca regimen,” adds Dr. Moshel. “These results are encouraging – for patients, their loved ones and the medical community – and we look forward to sharing further findings from Phase 3 within the next 18 months.
Specialists at the Gerald J. Glasser Brain Tumor Center treat a variety of brain tumors and related conditions, including acoustic neuromas and schwannomas; anaplastic astrocytomas, oligodendrogliomas and low-grade gliomas; brain, epidural and leptomeningeal metastases; ependymomas; glioblastomas; low-grade astrocytomas; lymphomas; meningiomas; medulloblastomas; peripheral nerve, pineal and pituitary tumors; rare glial and neuronal tumors; sarcomas; and spinal cord tumors.
The center features a multidisciplinary panel of experts who specialize in neurosurgery, skull base surgery, neuroradiology, radiation oncology, neuro-oncology, medical oncology, neurology, neuropathology and social work. Overlook Medical Center also has the largest CyberKnife program in the tri-state area for the treatment of brain tumors. The brain tumor center recently moved into its own space on the main level of Overlook Medical Center, in order to foster an optimal, convenient patient-centered experience.
For more information, visit research.atlantichealth.org/Toca511, or contact Patrice Light, research nurse coordinator, Gerald J. Glasser Brain Tumor Center, at 908-522-5768.
Editor's Note: This advertorial content is being published by TAPinto.net as a service for its marketing partners. For more information about how to market your business on TAPinto, please email contact@tapinto.net
https://www.tapinto.net/towns/westfield/articles/immunotherapy-clinical-trial-at-overlook-medical-22
Look out dot.com era! Here comes the pot.com era!

With a medical and recreational marijuana industry about to fly, there’s a whole lot of buzz going on. Globally-competitive, lucrative sectors and commercial opportunities are predicted to grow like a weed!
Cannabusiness is set to be smoking – forecasts of the medical marijuana market predict US$31 billion in annual sales by 2021. A Deloitte study suggests recreational marijuana sales alone in Canada could be as high as $8.7 billion per year.
Who consumes marijuana and the many ways it can be used is about to be reinvented. You only have to look to the U.S. where cannabis is legal to see the range of pot-abilities for cannabis use and consumption – edibles are a huge success with everything from brownies and beer to beef jerky and candy infused with pot. The pending Cannabis Act provides for edibles to be allowed recreationally in Canada by 2019.
Canadian companies are busy developing pills, inhalers and creams to compete with the pharmaceutical giants to treat medical conditions, including anxiety and chronic pain to childhood epilepsy and multiple sclerosis.
From pills to drinks to creams, cannabis is certainly looking to go mainstream, and even part of the luxe beauty experience. Ste. Anne’s Spa in Grafton, Ont., will be offering hemp-infused herbal remedies in a new skincare line and as part of their spa package. Massage products will be made with the company’s own hemp CBD products and “will include an aromatherapy element to induce and even deeper level of relaxation,” says spa director Natalie Koshowski.
Their weedy wonders include lotions, balms, body butters and oils that are infused with CBD extracted from hemp – there are no psychogenic effects.
The positive therapeutic powers of the cannabis plant extracts like CBD are renowned, yet sadly its use is highly stigmatized, says Ste. Anne’s Spa owner Jim Corcoran. “More people worldwide view it as socially acceptable and medically important.”
Corcoran’s company is also one of the first to cover medical marijuana for qualifying employees through their health benefits plans for issues including chronic pain, depression and anxiety. Loblaws was the first large Canadian employer to cover medicinal pot for employees, limited to treating the symptoms of multiple sclerosis, and the side-effects of chemotherapy for cancer patients. Some progressive insurers have added medical marijuana as a regular medicine to group benefits plans, including Sun Life Financial, a provider of health benefits coverage to one in six Canadians.
Corcoran says that covering medical marijuana for his staff gets them back to work sooner, staying at work, and contributes to their overall wellness and productivity.
But some HR officials worry about impairment at work and safety issues as well as a decrease in attendance and productivity. Even when accommodating medicinal use, there is no accommodation for impairment in the workplace, says Alison McMahon, owner of Cannabis at Work which educates employers and helps them comply with changing legislation.
“We are seeing more of a trend towards offering coverage for medical marijuana than we have ever before,” says the Edmonton-based cannabis expert at cannabisatwork.com. “Employers have a duty to accommodate medical cannabis to the point of undue hardship and they also have to restrict recreational cannabis in the workplace in the same way they would restrict alcohol.”
Employers who are worried represent more of a lack of knowledge about cannabis, and about the solutions they can put in place in the workplace, says McMahon, adding that cannabis use is in workplaces today and this is not brand new.
When it comes to smoking pot, it does matter when you start. New Canadian research shows that boys who start smoking pot before 15 are much more likely to have a drug abuse problem by the age of 28 than those who start at 15 or after – by an astounding 68%!
It’s mindboggling what a year or two can make. Université de Montréal researchers report that “the odds of developing any drug abuse symptoms by age 28 were reduced by 31% per cent for each year of delayed onset of cannabis use in adolescence.” Those who started before age 15 were at higher risk regardless of how often they consumed.
With the pending legalization of pot and pot’s ever-increasing potency, the researchers stress that prevention strategies need implementing in elementary school and kids need to be educated early about the risks of starting pot smoking.
Other research published in the journal Addictive Behaviors, 10% of Canadian adolescents consumed cannabis in Grade 8. By Grade 12, that percentage nearly tripled to 29%.
“The odds of developing any drug abuse symptoms by age 28 were non-significant if cannabis use had its onset at ages 15 to 17, but were significant and almost doubled each year if onset was before age 15,” the study says.
http://torontosun.com/health/diet-fitness/healing-with-hemp-cannabis-in-spas
Biotechs: 4 Reasons And A Catalyst To Move Stocks Higher
https://seekingalpha.com/article/4178638-biotechs-4-reasons-catalyst-move-stocks-higher
Still here and actually picked up more shares on the move below $0.30 even though i'm overweight in this issue.
Today looked more in favor of long term holders than usual as the share price was walked up for once.
Doubt it was the news today as it really told us very little unfortunately....
Attend this session at the 2018 ASCO Annual Meeting
Session: Hematologic Malignancies—Lymphoma and Chronic Lymphocytic Leukemia
Type: Poster Session
Time: Monday June 4, 8:00 AM to 11:30 AM
Location: Hall A
http://abstracts.asco.org/214/AbstView_214_223539.html
A phase 2 study to assess the safety and efficacy of umbralisib (TGR-1202) in pts with CLL who are intolerant to prior BTK or PI3Kd inhibitor therapy.
Sub-category: Chronic Lymphocytic Leukemia (CLL)
Category: Hematologic Malignancies—Lymphoma and Chronic Lymphocytic Leukemia
Meeting:2018 ASCO Annual Meeting
Abstract No: 7530
Poster Board Number: Poster Session (Board #167)
Citation: J Clin Oncol 36, 2018 (suppl; abstr 7530)
Author(s): Anthony R. Mato, Stephen J. Schuster, Nicole Lamanna, Ian Flinn, Jacqueline Claudia Barrientos, Suman Kambhampati, Bruce D. Cheson, Paul M. Barr, John M. Pagel, James Andrew Reeves, Frederick Lansigan, Jeffrey J. Pu, Alan P. Skarbnik, Gustavo A. Fonseca, Colleen Dorsey, Eline Luning Prak, Dana Paskalis, Peter Sportelli, Hari P. Miskin, Danielle M. Brander; Memorial Sloan Kettering Cancer Center, New York, NY; Abramson Cancer Center, Philadelphia, PA; New York-Presbyterian, Columbia University Medical Center, Manhasset, NY; Sarah Cannon Research Institute, Nashville, TN; Northwell Health/CLL Research and Treatment Program, New Hyde Park, NY; Sarah Cannon Research Institute at Research Medical Center, Kansas City, MO; Georgetown University Hospital, Lombardi Comprehensive Cancer Center, Washington, DC; University of Rochester Medical Center, Rochester, NY; Swedish Cancer Institute, Seattle, WA; Florida Cancer Specialists South / Sarah Cannon Research Institute, Ft Myers, FL; Dartmouth-Hitchcock Medical Center, Lebanon, NH; Penn State Hershey Cancer Institute, Hershey, PA; City of Hope, New York, NY; Florida Cancer Specialists/Sarah Cannon Research Institute, St. Petersburg, FL; Memorial Sloan Kettering Cancer Center / CLL Program, Leukemia Service, New York, NY; Clinical Immunology Laboratory at the Hospital of the University of Pennsylvania, Philadelphia, PA; TG Therapeutics, Inc., New York, NY; Duke University School of Medicine, Durham, NC
Abstract Disclosures
Abstract:
Background: Although KI therapies are generally well-tolerated, intolerance is the most common reason for discontinuation, thus representing an unmet medical need. Umbralisib (TGR-1202), a next generation PI3Kd inhibitor, has a discontinuation rate due to adverse events (AEs) of < 10%. Methods: We report results from a Ph 2 study assessing the safety/efficacy of umbralisib in CLL pts who were intolerant (defined per protocol) to a prior KI within 12 mos. AEs must have resolved to ≤ GR 1 prior to umbralisib therapy. Umbralisib (800mg QD) administered until progression or toxicity. Primary endpoint is progression-free survival (PFS). Results: 40 pts were treated as of 2/2018 (36 BTK & 4 PI3Kd intolerant). Baseline demographics: median (med) age 69 yrs (range 52-96), med prior therapies 2 (1-7), 55% male, ECOG 0-1 (92%), del17p (20%), del11q (23%), IGHV unmutated (60%). 80% required treatment within 6 mos of prior KI discontinuation. Most AE’s leading to KI discontinuation were: arthralgia, rash (9 events each), A-fib (6), diarrhea (4), bleeding, fatigue and weight loss (3 each). AEs on umbralisib are listed in the table: GR ≥3 PI3Kd-associated AE’s were limited: AST/ALT (3%); diarrhea (7.5%); rash (3%). 4 pts discontinued umbralisib due to intolerance (rash, pneumonia, pneumonitis, pancreatitis), and 1 pt due to study noncompliance. No pt discontinued umbralisib as a result of a prior KI intolerant AE. 3 pts (7.5%) had dose reductions (headache, hematologic, and colitis) and were successfully re-challenged (colitis pt on study in PR now 12 mos). Med PFS not reached with 90% of pts progression free at a med follow up 6.5 mos (range 1–15). Conclusions: Umbralisib appears to be safe and effective in KI intolerant CLL pts. These are the first data to confirm that switching from KI to an alternate PI3Kd (umbralisib) can result in disease control (PFS) without recurrence of KI intolerance toxicities. BTK mutations and CYP polymorphisms are being assessed. Clinical trial information: NCT02742090
Umbralisib AEs (all causality) in > 15% pts (n = 40).
Adverse Events All Grades % All Grades GR 3/4 % GR 3/4
Diarrhea 17 43% 3 8%
Nausea 17 43%
Thrombocytopenia 11 28% 4 10%
Insomnia 9 23%
Neutropenia 9 23% 7 18%
Dizziness 8 20%
Fatigue 8 20%
Rash 7 18% 1 3%
Am a little surprised at the stock selling off on small volume trades after a good move up, but happy to accumulate today at these lower prices. GL and thanks for the link.......
As a new company I am averaging in with their promising drugs..Double bottom in place.........
Aileron Therapeutics ($ALRN)
Therapy: ALRN-6924
Disease: T- and NK-cell lymphomas
News: ALRN has published preclinical findings for their stapled peptide ALRN-6924 in patient-derived xenograft models of T- and natural killer (NK)-cell lymphomas. The findings were published in Nature Communications. The activity of ALRN-6924 was shown to be favorable compared with standard romidepsin across a number of subforms of these tumors, and the paper also mentions that the researchers have observed a complete remission in a patient with angioimmunoblastic T-cell lymphoma, providing a rationale for ongoing development.
Looking forward: The preclinical work demonstrates that the interface between Mdm2 and p53 is an actionable target in preclinical models of lymphoma, which is a pretty marvelous feat in my book. We've known for a very long time that p53 is a promising target, since it is a potent tumor suppressor when active. Mdm2 inhibits p53 activity in vivo, which can help to drive tumor growth. The observation of a complete remission in a patient is definitely favorable, as well, and support the ongoing work in this space.
Cash Position and Guidance: Cash, cash equivalents and investments as of
March 31, 2018 were $43.3 million, compared to $50.8 million as of
December 31, 2017. The Company believes that its cash, cash equivalents
and investments as of March 31, 2018 will enable the Company to fund its
operating expenses and capital expenditure requirements into the second
half of 2019.
Here's Why The Marijuana ETF Could Be A Short-Covering Idea
Published: May 23, 2018 11:54 a.m. ET
The ETFMG Alternative Harvest ETF MJ, +0.09% the only U.S.-listed exchange-traded fund that's a pure play on marijuana-related equities, is enjoying a solid rebound effort in May with a gain of about 7 percent this month.
There are some catalysts, political and otherwise, that have the potential to generate more near-term upside for MJ and pot equities.
“A key development for the US Marijuana industry came on April 13th, when President Trump confirmed he had promised Colorado Senator Cory Gardner that US AG Jeff Sessions revocation of the Cole memo will not affect the state's legal Marijuana industry,” said IHS Markit in a recent research note. “That was a very timely update, with the combined market cap for Marijuana related stocks hitting a 2018 low on April 9th. Since then the market cap of those stocks has risen by $3.4 billion, a 13 percent increase.”
Home to over $380 million in assets under management, MJ tracks the Prime Alternative Harvest Index. The ETF holds 38 stocks.
Markit data indicate a slew of marijuana stocks are currently sold short in significant fashion.
“Short sellers have not backed down, increasing the combined short position in Marijuana related stocks to nearly $2bn, a level just below the all-time high on January 24th,” according to the research firm. “Since that last high point in short positions, the total market cap has fallen by $5.5 billion, or 15 percent. Short sellers have generally increased positions on a lag to advances in share prices, but have kept up the overall pace with the total market cap increasing by 92 percent since the start of Q4 2017, while short balances have increased by 102 percent.”
Markit points to 10 marijuana-related stocks with significant short interest, a group that includes several of MJ's top 10 holdings, indicating the ETF could be well-positioned for a possible short-covering rally.
“On June 7 Canada's Senate will vote on bill C-45, legislation which would make the recreational use of Marijuana legal throughout the country, in essence regulating recreational Marijuana use in a manner similar to tobacco,” said Markit. “The road to recreational legality in Canada received a boost on March 23rd when a group of Senators who opposed the bill were unable to muster the votes to prevent the bill from reaching this final stage of legislative approval.”
This is important because Canada is where many of the more credible marijuana stocks, including some MJ holdings, are listed. For its part, MJ allocates over 61 percent of its weight to Canadian stocks, by far the ETF's largest geographic exposure.
https://www.marketwatch.com/story/heres-why-the-marijuana-etf-could-be-a-short-covering-idea-2018-05-23-11465441
Yes the right to try should help the sector overall and we never should have needed Congress or the President to have to pass something that should be an individual's right already.
No more buying for me until the outright manipulations stops. I topped off my position the other day and hold 100,000 shares now. I have too many other stocks that are good swing trades and will hold off buying any more for now as we wait on Management to show their hand.
This could help the stocks in $MJ`s ETF:
Right to Try' bill heads to Trump's desk after House OK
https://www.npr.org/sections/health-shots/2018/05/22/613106777/house-passes-right-to-try-bill-for-experimental-drugs
Right-to-try' bill passes Congress
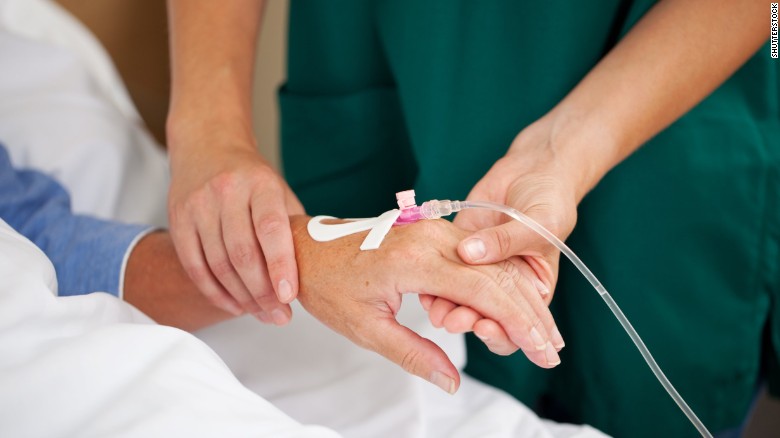
The bill would give terminally ill patients the right to seek drug treatments that remain in clinical trials.
(CNN)With a House of Representatives vote Tuesday, Congress passed legislation that could give terminally ill patients a way to independently seek drugs that are still experimental and not fully approved by the US Food and Drug Administration.
The House voted 250-169 in favor of the bill, which the Senate passed in August. The bill will now be sent to President Trump, who is expected to sign it.
"This is an extraordinarily great day," Democratic Sen. Joe Donnelly of Indiana, one of the original cosponsors of the bill, said in a press conference after the event. Donnelly said he met with Vice President Mike Pence a few weeks ago and urged him to push for a House vote on the bill.
After a failed attempt, the House passed its own version of the bill in March after making changes to the Senate bill, which would have required the Senate to vote anew. But House Republicans announced a change of course last week, saying they would vote on the Senate bill.
"It is time for the House to do what Senate Democrats won't and send a right-to-try bill to the President's desk, bringing hope to terminally-ill patients across the country," House Energy and Commerce Committee Chairman Greg Walden said in a statement Thursday, when the vote was announced.
What you need to know about right-to-try legislation
The bill gives terminally ill patients the right to seek drug treatments that remain in clinical trials and have passed phase one of the FDA's approval process, but have not been fully approved.
Advocates for the legislation say it opens a door for terminally ill people in states that haven't passed such a law. Critics argue that the legislation disempowers the FDA and won't make it easier for terminally ill people to access these drugs.
Trump has expressed his support for right-to-try legislation and is expected to sign the measure into law.
"We also believe that patients with terminal conditions should have access to experimental treatments that could potentially save their lives," he said in his 2018 State of the Union address. "It is time for the Congress to give these wonderful Americans the 'right to try.' "
Frank Mongiello, who was diagnosed with ALS and whose name is one of four on the bill, has been a lead advocate on the issue and was at the Capitol Tuesday for the vote.
"I think it's the first time I've had butterflies going to the Capitol," Mongiello, who is unable to use his voice, told CNN while using eye-tracking technology.
"It's very important to me to be able to look my wife and children in the eye and say we did everything possible and say we never gave up," he added. "Unfortunately with today's contentious Congress, I won't really celebrate until after President Trump signs the bill."
Where's the obstacle?
"This issue is about real people who are terminally ill, facing the end of the line, and want to have one more shot at life," said Starlee Coleman, senior policy adviser at the Goldwater Institute, a conservative public policy think tank based in Phoenix that supports right-to-try legislation.
Right-to-try laws exist in 40 states: Alabama, Arizona, Arkansas, California, Colorado, Connecticut, Florida, Georgia, Idaho, Illinois, Indiana, Iowa, Kentucky, Louisiana, Maine, Maryland, Michigan, Minnesota, Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, South Carolina, South Dakota, Tennessee, Texas, Utah, Virginia, Washington, West Virginia, Wisconsin and Wyoming.
Explaining "Right to Try" legislation
Explaining "Right to Try" legislation 02:56
With the new federal legislation, Coleman argues, patients in states without these laws could save time accessing experimental treatments by eliminating FDA application requirements."You don't have to file an application with the federal government. You just get to work directly with your doctor and a drug company if you meet the criteria," Coleman said.Opponents of the bill, including over 100 patient and provider advocacy groups, say it won't have a major impact on accessing treatments; on the contrary, it could have a detrimental effect on how the FDA safeguards people's health.
"By removing the FDA oversight, you're counting on physicians and manufacturers to serve as the gatekeeper and protector of our patients. I simply don't buy that that's going to work," New Jersey Rep. Frank Pallone Jr. said on the floor shortly before the vote.
"The law is ... targeting the wrong obstacle," said Art Caplan, director of the Division of Medical Ethics at the New York University School of Medicine. "It's basically saying the FDA is in the way, and that's why people can't try unapproved medicines."
"It's companies that control access to their drugs," he added.
For decades, there have been measures in place for terminally ill patients to access experimental medical products outside clinical trials. These measures are often referred to as expanded-access policies or compassionate use.
"I'm in favor of pathways that allow patients with terminal disease to get access to products that might be in development," FDA Commissioner Dr. Scott Gottlieb told CNN Chief Medical Correspondent Dr. Sanjay Gupta in April. "That's why we have an expanded-access pathway."
About 99% of submitted applications for expanded access to almost 9,000 investigational drugs were allowed to proceed over a 10-year period between January 2005 and December 2014, according to a study by FDA researchers, published in the journal Therapeutic Innovation & Regulatory Science in 2016.
Gottlieb previously said in a statement that "emergency requests for individual patients are usually granted immediately over the phone and non-emergency requests are generally processed within a few days."
Caplan said, "I think the FDA does fine with compassionate use requests. But let's be clear: FDA doesn't do anything until a doctor goes to a company -- say, a big pharmaceutical company -- and the company says, 'We're going to give you the drug.' If they don't say yes, there is no FDA. So let's be clear where the obstacles are."
'Flying blind'
It is unclear how many people have taken advantage of right-to-try laws because the vast majority of states do not have central reporting requirements, Coleman said.
She also said there's no guarantee that insurance companies would pay for these treatments, and she has seen both outcomes at the state level. The legislation, she said, "is really about giving patients control over the treatments and the options that they have at the end of their life" when they have "exhausted all available treatment options and ... cannot qualify for a clinical trial."
Some doctors worry that removing the FDA from the equation leaves too
much uncertainty surrounding drugs that haven't been tested -- especially when it comes to medically fragile patients taking drugs with unknown risks and benefits.
"The rest of us doctors are kind of flying blind with respect to things like how much of the drug to give, how to give it, what kind of side effects to look for," Dr. Steven Joffe, professor in medical ethics and health policy at the University of Pennsylvania's Perelman School of Medicine, previously told CNN. "So by cutting the FDA out of the loop, you are failing to take advantage of the knowledge that it has about how to use the drug.
"About 10% of the time, the FDA, when it gets requests for expanded access, will say, 'Yes, but here's the way you can do it more safely' or 'here's the side effects to look out for,' " he said, referencing an agency document about patient access to investigational drugs.
Even the bill's opponents, though, have found some positives about the right-to-try push -- including more data about experimental drugs and greater awareness about other options for the terminally ill.
"I think the publicity helps and lets more people know 'maybe there's a last ditch thing I ought to pursue,' " Caplan said.
"But at the end of the day, creating a world in which you say there's a right to try, if it's nothing more than a right to beg a company, that right already exists, and you're really not doing much to help anybody gain access to much of anything."
CNN's Ben Tinker and Ashley Killough contributed to this report.
https://www.cnn.com/2018/05/22/health/right-to-try-legislation-congress/index.html