Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
New investors, the statement “39% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 1597 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 6/15/2018
Current Short: 1,137,361
Previous Short: 1,059,202
Change: 78,159 (7.38%)
Average Daily Volume: 249,341
Days to Cover: 4.56
http://otce.finra.org/ESI
0.73% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 1597 shares (0.001% of the OS) were failure-to-deliver as of June 11.
20180611|45782D100|IPIX|1597|INNOVATION PHARMACEU|0.47
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
Article - Antibody-Directed Therapeutic Research Ongoing in Ovarian Cancer
Antibody-directed therapies have shown potential with investigational targets such as mesothelin and folate receptor alpha, but researchers are actively exploring approaches that target the prevalent p53, explained Sanaz Memarzadeh, MD, PhD.
“If you look at the top 5 cancers that affect men and women, a large proportion of these patients carry mutations in p53,” said Memarzadeh. “p53 is the most commonly mutated gene in cancer. It’s seen in upward of 40% of tumors.”
In an interview at the 2018 OncLive® State of the Science Summit™ on Ovarian Cancer, Memarzadeh, gynecologic oncologist and professor of obstetrics and gynecology, University of California, Los Angeles, detailed the early developments of antibody-directed therapies and restoring p53 function in the treatment of patients with ovarian cancer.
OncLive: What are some novel approaches that are being explored in ovarian cancer?
Memarzadeh: Some of the research we are doing in our laboratory, in collaboration with investigators at UCLA, tie into one of these novel treatment options. The first approach is an immune-biologic approach with antibody-directed treatments. It utilizes the ability of an antibody to recognize an antigen expressed on a cancer cell. The antibody also carries a cytotoxic drug or a targeted drug that can be taken up by the cell. When the antibody binds to the antigen on the cell, it can go inside of the cell and directly target the cancer cell. What’s unique about this is the idea that it targets the cancer cell without targeting the normal cell. That is a major advantage of this approach.
The key to this approach is that you have to pick antigens that are highly expressed in the cancer cell but have low expression in normal cells. Antibody-directed targeted therapies against mesothelin is one approach. Mesothelin is a cell-surface protein that is expressed in ovarian and other cancers, including mesotheliomas.
The general concept is that the antibody binds to mesothelin, carrying an agent that targets tubulin. That’s how it can specifically target the cancer cells. This approach has not been successful as a single therapeutic agent in mesotheliomas. That is the rationale as to why this approach is being combined with doxorubicin in a clinical trial for patients with platinum-resistant ovarian cancer.
The second approach takes a very similar idea: using antibody-directed therapies to target the folate receptor alpha. The folate receptor alpha can be overexpressed in ovarian cancer, but it’s not expressed at high levels in normal tissue. Therefore, it makes it a potential target. Similarly, an antibody that targets the folate receptor alpha can carry a drug conjugate that is taken up by the cancer cells. This is being explored in a series of ongoing clinical trials.
The FORWARD I trial looked at utilizing this approach with mirvetuximab soravtansine compared with clinician’s choice of chemotherapy for patients with platinum-resistant ovarian cancer. It is now being looked at in phase Ib/II trials where it’s being tested in combination with bevacizumab (Avastin), carboplatin, or doxorubicin. One arm looked at patients with platinum-resistant ovarian cancer and another arm looked at patients with platinum-sensitive disease.
The challenge with a biologic approach is that it relies on the fact that all of the tumor cells express this antigen or target to some extent. This may not be the case. If a small subset of the tumor cells do not express this antigen, they may evade this therapy. If those cells can regenerate the cancer, the cancer can relapse.
While these approaches are exciting, I’m excited about approaches that target ubiquitous pathways in the tumor. One such pathway is targeting p53, which is coined “the guardian of the genome.” It plays a very central role in making sure that the cell does not undergo chaos. It does this by sending a stress. If the stress is small, it tells the cell to repair DNA damage and the cell goes on to live. If the stress is severe, the cell then undergoes death. When it is mutated, it cannot do those normal functions.
It is a very exciting target to try to restore the p53 function, but to this day we don’t have an FDA-approved drug that can target p53.
There are 3 approaches that are being taken to restore p53 function. Many of these approaches are focused on restoring the folding of p53. In order to function properly, it has to be a tetramer and properly folded, so it can bind to DNA.
APR-246 is one of the first drugs being tested. It was found in a drug screen, speci cally targeting cells that carry a kind of p53 mutation. It is an interesting compound, but it has to be converted to methylene quinuclidone. The compound then binds to cystines in either p53 or any molecule that carries a cystine. By doing so, it’s thought that it can restore the folding and function of p53.
This is being tested in clinical trials in combination with other agents. One of the challenges is that the exact mechanism of action is not known. Therefore, biomarkers of response need to be discovered.
A second compound that is in clinical trials is a zinc-specific chelator. For p53 to be properly folded, it requires the right amount of zinc around it. Zinc chelators are thought to modulate p53 function folding by binding to zinc. This compound is also being tested in phase I clinical trials in ovarian cancer and heavily pretreated head and neck cancers.
One of the approaches that we are very excited about is using peptides to restore p53 function. It’s known that a subset of p53 mutations cause amyloid sheets to form. p53 mutations can cause this aggregation. They can stick to each other, aggregating amyloid sheets, so it cannot function normally.
We work with a brilliant group of collaborators at UCLA led by the lab of Drs David Eisenberg and Alice Soragni. They discovered a peptide that specifically targets a portion of p53 that is highly prone to this aggregation process. The peptide can sit at the groves of mutated p53 so newly synthesized [p53] can potentially be free to do its function. It also has a polyarginine tag that lets it get into the cell.
In collaboration with this group of scientists at UCLA, my lab has been doing a lot of preclinical testing. We are looking at the e cacy of what we call the “reactivator of p53” using a combination of in vitro and in vivo models. The ndings are promising; it certainly has efficacy as a single agent.
Now we are looking at combining it with carboplatin, which is a standard therapy for ovarian cancer. We want to see if these 2 drugs together can be synergistic. Since we know what the peptide’s mechanism of action is, we’re looking at biomarkers of response. We hope to be able to transition this to clinic soon.
Does doxorubicin hold the most promise in combination with antibody-drug conjugates?
Doxorubicin is used in many clinical trials because the patients who are enrolled in these trials are patients with platinum-resistant disease. It makes sense to use one of the most commonly used second-line agents.
Do you see the preclinical work transitioning to the clinic soon?
That’s our hope. We are actively working on pushing this project forward. It’s certainly a collaboration between different groups at UCLA. That is what is so exciting about science. We hope to be able to transition this to clinic within the next year or two. Sometimes translational science takes longer than you anticipate, but we clearly see efficacy. Most importantly, it seems to be well tolerated in the in vivo models.
https://www.onclive.com/web-exclusives/antibodydirected-therapeutic-research-ongoing-in-ovarian-cancer
New investors, the statement “13% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 1597 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 6/15/2018
Current Short: 1,137,361
Previous Short: 1,059,202
Change: 78,159 (7.38%)
Average Daily Volume: 249,341
Days to Cover: 4.56
http://otce.finra.org/ESI
0.73% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 1597 shares (0.001% of the OS) were failure-to-deliver as of June 11.
20180611|45782D100|IPIX|1597|INNOVATION PHARMACEU|0.47
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “49% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 1597 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 6/15/2018
Current Short: 1,137,361
Previous Short: 1,059,202
Change: 78,159 (7.38%)
Average Daily Volume: 249,341
Days to Cover: 4.56
http://otce.finra.org/ESI
0.73% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 1597 shares (0.001% of the OS) were failure-to-deliver as of June 11.
20180611|45782D100|IPIX|1597|INNOVATION PHARMACEU|0.47
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “33% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 1597 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 6/15/2018
Current Short: 1,137,361
Previous Short: 1,059,202
Change: 78,159 (7.38%)
Average Daily Volume: 249,341
Days to Cover: 4.56
http://otce.finra.org/ESI
0.73% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 1597 shares (0.001% of the OS) were failure-to-deliver as of June 11.
20180611|45782D100|IPIX|1597|INNOVATION PHARMACEU|0.47
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
BP's interests in the Big Three
“The Company is engaged in discussions and negotiations regarding the out-licensing of its mid-stage, first-in-class clinical assets to global and/or specialty pharmaceutical companies who have expressed an interest in one or more assets in our pipeline.”
https://www.sec.gov/Archives/edgar/data/1355250/000147793218002302/ipix_10q.htm
“The Confidential Disclosure Agreement (CDA) count toward partnering with global and specialty pharmaceutical companies interested in the Company’s first-in-class drug candidates is nearing 20, with additional Agreements in review."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
“These recent data, taken in aggregate, are what various actively engaged pharmaceutical companies have desired from us for some time—and they are what triggered a newfound flurry of inbound partnership discussions at the San Francisco conferences,”
http://www.ipharminc.com/press-release/2018/1/16/innovation-pharmaceuticals-presents-at-2018-biotech-conference-oral-mucositis-drug-candidate-garners-exceptional-interest-after-successful-phase-2-trial
"To advance our pipeline in an expeditious manner, this year we placed an emphasis on partnering—actively engaging global and regional pharmaceutical companies interested in licensing our novel drug candidates. These efforts have led to highly productive, mature (late-phase, ongoing) discussions, with multiple pharmaceutical companies, which we hope will bear fruit in the coming year." - CEO Letter to Shareholders
"We had said that confidential disclosure agreements have been signed with several pharmas interested in our drug candidates; to some, they're interested in one compound. Others have requested information on a combination of Brilacidin and Kevetrin and Prurisol."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
B-Dermatitis
"To further these efforts, the Company is in negotiations with a leading drug formulator to develop topical formulation(s) of Brilacidin for these three dermatology indications, starting in 1H2018. The goal of the negotiations is to reach terms on a strategic partnership for addressing these markets."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
B-OM
“Given the notable interest from global and specialty pharmaceutical companies that we have received following recent Brilacidin-OM data releases, we continue to carefully assess all of our potential alliance opportunities—toward cementing the best pathway forward,” said Leo Ehrlich, Chief Executive Officer of Innovation Pharmaceuticals.”
http://www.ipharminc.com/press-release/2018/5/9/innovation-pharmaceuticals-concludes-data-analysis-of-its-phase-2-clinical-trial-for-severe-oral-mucositis-in-head-and-neck-cancer-positioning-to-fill-a-substantial-void-in-supportive-cancer-care
“Formal collaboration with pharmaceutical companies that have expressed an interest in partnering Brilacidin-OM may well assist further with expediting the drug candidate’s development timetable. Some of these partnering conversations have matured to the point of potentially structuring mutually beneficial licensing agreements, pending the final Phase 2 study results.”
http://www.ipharminc.com/press-release/2017/11/16/innovation-pharmaceuticals-offers-perspectives-on-brilacidin-as-a-potential-preventative-treatment-for-oral-mucositis-in-head-and-neck-cancer-patients
B-UP
"We've actively engaged with multiple large pharmas, who have expressed an interest in Brilacidin inflammatory bowel disease. The pharma industry is quite interested in pursuing novel IBD therapies one of the more active therapeutic areas when it comes to dealmaking."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
Prurisol
"We have entered into multiple non-disclosure agreements with large pharmaceutical companies that enable us to continue ongoing discussions regarding potential partnering should the trial results support such a relationship."
https://www.sec.gov/Archives/edgar/data/1355250/000147793217004400/ipix_10k.htm
Kevetrin
"The Company is adhering to a value-building strategy born from discussions with larger pharmaceutical companies interested in Kevetrin, one of the world’s most advanced p53 drug candidates. Securing the right development partner for Kevetrin remains an important objective."
http://www.ipharminc.com/press-release/2018/2/8/innovation-pharmaceuticals-concludes-successful-phase-2a-trial-of-kevetrin-for-ovarian-cancer-intra-tumor-modulation-of-p53-observed
"What we and potential partners are extremely interested in is learning via tumor biopsies if Kevetrin is reaching its target and modulating pathways within the tumor that can deliver a clinically meaningful benefit; this would be a significant development in the p53/oncology dynamic,"
http://www.ipharminc.com/press-release/2017/9/14/innovation-pharmaceuticals-opens-new-clinical-site-for-novel-p53-drug-candidate-in-phase-2-ovarian-cancer-trial-1
Document - Getting Ready for a BioPharma Partnering Deal
A typical large pharmaceutical might see 500 to 1,000 opportunities in a given therapeutic area. Only 100 or so of those will go on to non-confidential meetings and only 50 or so of those will end up going to Confidential Disclosure Agreements (CDAs) and confidential meetings.
Unfortunately, getting a deal done can take a long time. After initial talks, getting a CDA can take a month or more. It might take another month for the other side to assemble appropriate team members and schedule team meetings.
It can then easily take another month for them to evaluate all the information and to decide that they will indeed go forward with the formal due diligence process. From the point when the other party has decided to go forward with term sheet negotiations, it can take another six to twelve months to get to a signature on a full agreement.
Diligence and a Good Virtual Data Room
When a potential partner begins the due diligence process, they’re trying to get to know you and your asset in greater detail, and they’re attempting to verify what they’ve been told.
Give them access to your experts, provide overview presentations, and answer questions. In short, spend the time it takes to make sure they really understand it.
IPIX Clinical Trial Summaries
B-OM (Phase 2)
Kaplan-Meier Curves

"Onset of Severe Oral Mucositis: Based on Kaplan-Meier curves, Brilacidin-OM oral rinse showed a clear separation from placebo in delaying the onset of SOM—particularly the period from approximately 28-42 days, after the initiation of treatment, during which the incidence of SOM rose strikingly in the placebo group while not in the group being treated with Brilacidin."
http://www.ipharminc.com/press-release/2018/1/3/innovation-pharmaceuticals-brilacidin-meets-key-secondary-endpoint-in-phase-2-trial-delays-onset-of-severe-oral-mucositis-som
Topline Results
· Brilacidin met primary endpoint of reduced incidence of severe OM experienced by patients during radiation therapy.
· Incidence of severe OM in Modified Intent to Treat (mITT) Population: Brilacidin 42.9%, Placebo 60.0%.
· Incidence of severe OM in Per Protocol (PP) Population: Brilacidin 36.8%, Placebo 60.0%.
· Trial results support continued and expedited development of Brilacidin-OM.
http://www.ipharminc.com/press-release/2017/12/11/innovation-pharmaceuticals-reports-positive-topline-results-from-phase-2-placebo-controlled-trial-of-brilacidin-for-the-prevention-of-oral-mucositis-in-head-and-neck-cancer-patients
Subgroup Analysis
“For the Modified Intent-to-Treat (mITT) population, Brilacidin-OM in the aggressive chemotherapy regimen reduced the incidence of SOM by 65.0% ([incidence control- incidence active]/incidence control) as compared with placebo (Brilacidin: 25.0%; placebo: 71.4%; p=0.0480). For the Per Protocol (PP) population, Brilacidin-OM in the aggressive chemotherapy regimen similarly reduced the incidence of SOM by 80.3% as compared with placebo (Brilacidin: 14.3%; placebo: 72.7%; p=0.0249).”
http://www.ipharminc.com/press-release/2018/4/9/innovation-pharmaceuticals-phase-2-oral-mucositis-trial-additional-data-show-brilacidin-om-demonstrated-a-significant-reduction-in-the-incidence-of-severe-oral-mucositis
B-UP (Phase 2 POC)
Before and after endoscopic images (Cohort B, 100 mg)

Topline Results
Rate of Clinical Remission (% of patients achieving)
- 60% Cohort A (3 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(i) #Endoscopy subscore ≤ 1 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(ii) Rectal Bleeding subscore of 0 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
(iii) Stool Frequency subscore, improvement or no change from baseline (% of patients achieving)
- 100% Cohort A (5 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
B-ABSSSI (Phase 2b)
Topline Results

"In treated patients assessed at 48-72 hours, 47/51 (92.2%), 46/48 (95.8%), 51/52 (98.1%), and 45/48 (93.8%) achieved clinical success in the Brilacidin 0.6 mg/kg single-dose group, Brilacidin 0.8 mg/kg single-dose group, Brilacidin 1.2 mg/kg 3-day group, and daptomycin 7-day group, respectively."
"On December 22nd 2014, Cellceutix also reported positive results in the Microbiological Intent-to-Treat (MITT) population. This is an important population that includes patients with baseline cultures positive for common ABSSSI pathogens, such as Staphylococcus aureus, including Methicillin-Resistant Staphylococcus aureus (MRSA). In this population, Clinical Success rates at 48-72 hours were again very high (above 90% across all treatment groups) and again very similar (with overlapping 95% confidence intervals)."
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-releases-confidence-interval-statistics-showing-clinical-success-rates-for-brilacidin-in-treatment-of-absssi
Prurisol (Phase 2a)
Topline Results (200 mg)

"Sub-population analyses further showed greater efficacy demonstrated in patients who had a baseline IGA score of 3 (“moderate”) as compared to those with a baseline score of 2 (“mild”). Some of these patients even experienced a 3-point reduction in their IGA score, going from “moderate” to “clear.” This suggests Prurisol may be more effective in treating moderate to severe psoriasis patients to a greater degree than those patients who exhibit less severe symptoms. In moderate to severe psoriasis studies, the placebo response also tends to be lower."
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
"Among patients participating in the study with the severest form of psoriasis, those having a baseline IGA score of 3 (“moderate”), the primary endpoint was met in 46.2% of patients who received Prurisol 200mg. This data was derived from analyses of all patients randomized across all 9 participating study sites."
"Additional preliminary data analyses of secondary endpoints show patients who received any dose of Prurisol, regardless of the treatment arm, had a 1-point improvement (using the IGA scoring system) at a higher rate than that of patients in the placebo arm. This is another clear indication of the drug’s efficacy."
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-provides-additional-insight-into-successful-phase-2-trial-for-treating-psoriasis
K-OC (Phase 2a)
P53 Modulation

"Modulation of the p53 protein was observed in response to administration of Kevetrin. Pathways analyses also point to concomitant cell cycle modulation at the level of gene expression. Importantly, these data are the first to directly support, in ovarian cancer patient tumors, Kevetrin’s ability to affect p53 and associated molecular pathways—a central gene signaling network involved in regulating cell growth and the cell cycle, helping to prevent cancer."
"In more detail, preliminary analyses used Western Blots to assess relative levels of key proteins extracted from tumor biopsies before and after a series of nine Kevetrin infusions administered over three weeks. The level of phospho-p53, the activated form of the protein, in addition to the noted p53 modulation, was also seen to change in response to Kevetrin administration. These findings confirm in patient tumors Kevetrin-induced anti-cancer effects similar to those demonstrated (pdf) preclinically in ovarian cancer cell-lines. These new data reinforce prior clinical data, from the earlier concluded Phase 1 study of Kevetrin in advanced solid tumors (see NCT01664000), in which observations of p21 expression in peripheral blood monocytes supported p53 involvement in Kevetrin’s mechanism of action."
http://www.ipharminc.com/press-release/2017/12/27/innovation-pharmaceuticals-obtains-direct-evidence-of-molecular-pathways-modulation-in-tumors-from-first-patients-in-kevetrin-ph2a-ovarian-cancer-trial
IPIX Milestones, Clinical Trials, Recent Articles, Corporate Overview, Patents
Q3 Potential Milestones
1. B-OM - Breakthrough Therapy Designation
2. Prurisol - Top-line Data (Phase 2b)
3. Licensing Agreement (double-digits millions upfront payment)
http://www.ipharminc.com/press-release/2018/6/28/innovation-pharmaceuticals-announces-7-million-milestone-funding-agreement-with-aspire-capital
Past Milestones

Kevetrin
- Positive primary endpoint (Phase 2a)
http://www.ipharminc.com/press-release/2018/2/8/innovation-pharmaceuticals-concludes-successful-phase-2a-trial-of-kevetrin-for-ovarian-cancer-intra-tumor-modulation-of-p53-observed
- Highly encouraging preliminary data (Phase 2a)
http://www.ipharminc.com/press-release/2017/12/27/innovation-pharmaceuticals-obtains-direct-evidence-of-molecular-pathways-modulation-in-tumors-from-first-patients-in-kevetrin-ph2a-ovarian-cancer-trial
- Positive primary endpoint and p21 data (Phase 1)
http://www.ipharminc.com/press-release/2016/11/12/assay-results-from-cellceutix-phase-1-clinical-trial-of-kevetrin-for-cancer-show-increased-p21-expression-in-675-of-evaluable-patients
- Orphan drug designation for ovarian cancer
http://www.ipharminc.com/press-release/2016/11/16/cellceutixs-kevetrin-receives-fda-orphan-drug-designation-for-the-treatment-of-ovarian-cancer
- Orphan drug designation for pancreatic cancer
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-receives-fda-orphan-drug-designation-for-kevetrin-for-the-treatment-of-pancreatic-cancer
- Orphan drug designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-fda-orphan-drug-designmation-for-kevetrin-for-the-treatment-of-retinoblastoma
- Rare pediatric disease designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-rare-pediatric-disease-designation-from-fda-for-kevetrin-for-the-treatment-of-retinoblastoma
Brilacidin
- B-OM positive secondary endpoint (Phase 2)
http://www.ipharminc.com/press-release/2018/1/3/innovation-pharmaceuticals-brilacidin-meets-key-secondary-endpoint-in-phase-2-trial-delays-onset-of-severe-oral-mucositis-som
- B-OM positive top-line data (Phase 2)
http://www.ipharminc.com/press-release/2017/12/11/innovation-pharmaceuticals-reports-positive-topline-results-from-phase-2-placebo-controlled-trial-of-brilacidin-for-the-prevention-of-oral-mucositis-in-head-and-neck-cancer-patients
- B-UP positive top-line data (Phase 2 POC)
http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
- B-ABSSSI positive top-line data (Phase 2b)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-positive-top-line-data-from-phase-2b-absssi-trial-single-dose-brilacidin-comparable-to-7-days-of-daptomycin
- QIDP designation for B-ABSSSI
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-antibiotic-brilacidin-receives-qidp-designation-from-fda
- Fast track designation for B-OM
http://www.ipharminc.com/press-release/2016/11/16/fda-grants-fast-track-designation-to-cellceutixs-brilacidin-om-for-oral-mucositis
- MTA extension for prophylactic testing in implanted devices
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-completes-lab-testing-of-brilacidin-for-planned-phase-3-trial-for-acute-bacterial-skin-and-skin-structure-infections
- Formulation stable at room temperature
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-breakthrough-in-the-formulation-of-novel-antibiotic-brlaicidin-plans-studies-to-treat-diabetic-foot-ulcers
- American Society for Microbiology journal publication
http://aac.asm.org/content/early/2014/06/11/AAC.02955-14#corresp-1
Prurisol
- Positive top-line data (Phase 2a)
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
- Positive primary endpoint (Phase 1)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-anti-psoriasis-drug-prurisol-meets-primary-endpoint-of-clinical-trial
- 505(b)(2) pathway
http://www.ipharminc.com/press-release/2016/11/17/cellceutix-informed-by-fda-that-505b2-approval-would-be-an-acceptable-approach-for-its-psoriasis-drug
Clinical Trials
1. K-OC (Phase 2a, Completed)
https://clinicaltrials.gov/ct2/show/NCT03042702?term=cellceutix&rank=1
2. P (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT02101216?term=cellceutix&rank=2
3. B-OM (Phase 2, Completed)
https://clinicaltrials.gov/ct2/show/NCT02324335?term=cellceutix&rank=3
4. P (Phase 2a, Completed)
https://clinicaltrials.gov/ct2/show/NCT02494479?term=cellceutix&rank=4
5. P (Phase 2b, Completed)
https://clinicaltrials.gov/ct2/show/NCT02949388?term=cellceutix&rank=5
6. B-ABSSSI (Phase 2b, Completed)
https://clinicaltrials.gov/ct2/show/NCT02052388?term=cellceutix&rank=6
7. K (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT01664000?term=cellceutix&rank=7
8. B-UP (Phase 2 POC, Completed)
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-receives-update-on-first-patient-enrollment-in-phase-2-proof-of-concept-study-of-brilacidin-for-ulcerative-proctitis
Pipeline
Recent Articles
- Taking a Page from Merck’s Gardasil and Glaxo’s Requip, These Small Companies Look to Cement Riches for Shareholders in New Category of Oral Mucositis
http://www.baystreet.ca/articles/stockstowatch/38594/Taking-a-Page-from-Mercks-Gardasil-and-Glaxos-Requip-These-Small-Companies-Look-to-Cement-Riches-for-Shareholders-in-New-Category-of-Oral-Mucositis
- A Billion Dollar Market Just Waiting for a New Oral Mucositis Drug: Five Companies That Want It
http://www.baystreet.ca/stockstowatch/2658/A-Billion-Dollar-Market-Just-Waiting-for-a-New-Oral-Mucositis-Drug-Five-Companies-That-Want-It
- Innovation Pharmaceuticals Stock May Be A Triple-Crown Winner With Brilacidin By Year End
https://seekingalpha.com/article/4088788-innovation-pharmaceuticals-stock-may-triple-crown-winner-brilacidin-year-end
- 2017 Q3 Conference Call Transcript
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
- 2017 Q1 Conference Call Transcript
http://seekingalpha.com/article/4023682-cellceutix-corporation-ctix-ceo-leo-ehrlich-q1-2017-results-earnings-call-transcript
- Interview with New Cellceutix President Dr A Bertolino
http://seekingalpha.com/article/3988240-interview-new-cellceutix-president-dr-bertolino
- Cellceutix Pipeline Continues to Hit Milestone After Milestone
https://www.streetwisereports.com/pub/na/cellceutix-pipeline-continues-to-hit-milestone-after-milestone
- A Small-Cap Biotech with Big Ideas for Acute Infections and Cancers: Cellceutix's Leo Ehrlich and Dr. Daniel Jorgensen
http://www.thelifesciencesreport.com/pub/na/a-small-cap-biotech-with-big-ideas-for-acute-infections-and-cancers-cellceutixs-leo-ehrlich-and-dr-daniel-jorgensen
Corporate Overview
- April 2018
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5acbbec2562fa79982ad5e22/1523302089564/2018+Apr+IPIX+Corporate+Overview.pdf
- 2018 Biotech Showcase
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5a4e50e39140b721186d1a6f/1515081962344/2018Jan+IPIX+Corporate+Overview.pdf
- BioCentury NewsMakers Conference
https://static1.squarespace.com/static/5715352e20c647639137f992/t/59b2f34946c3c498ebfe112e/1504899917915/IPIX+Company+Presentation+BioCentury+NewsMakers+Conference++8Sep2017.pdf
- Drug Discovery and Therapy World Congress
https://static1.squarespace.com/static/5715352e20c647639137f992/t/596cac7df14aa1a6e7118ed8/1500294274057/IPI+DDTWC+Brilacidin+presentation+%28final-u%29+13Jul2017.pdf
Patents
- Kevetrin
https://www.google.com/patents/US8338454
- Brilacidin
https://www.sec.gov/Archives/edgar/data/1355250/000147793214002418/ctix_ex1037.htm
https://www.google.com/patents/US20160243117
https://www.google.com/patents/EP2709619A2
- Prurisol
https://www.google.com/patents/WO2013103601A1
MP Advisors Report
https://drive.google.com/file/d/0Bz15O4eaX-asWVpoclB2WUhENFk/view
Abbreviations
K – Kevetrin
B – Brilacidin
P – Prurisol
OC – ovarian cancer
ABSSSI – acute bacterial skin and skin structure infections
OM – oral mucositis
UP – ulcerative proctitis
QIDP – qualified infectious disease product
Great catch, Daubers! I think Leo has two offers on the table for B-OM, one with BTD and one without. That’s probably why the BTD milestone was listed before a licensing agreement.
P result could also be a factor for a B-OM deal. If the trial is positive, Leo might ask for less unfront payment and more royalty. If the result is negative, Leo might ask for more upfront payment and less royalty.
New investors, the statement “49% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
Repost - Doctor-Founder-Billionaire Talks About Biotech Investment, Stocks
Not a lot of biotechnology entrepreneurs go on to count their wealth in billions, build supercomputers or own a significant piece of the Los Angeles Lakers basketball team.
But that’s been the trajectory of Dr. Patrick Soon-Shiong, who launched and sold two biotech startups–together worth nearly $8 billion. He now is heading up a new endeavor featuring artificial intelligence, semiconductors, cloud databases, a supercomputer, nano-optics and fiber-optic cable, all in the name of improving the country’s health-care system.
Dr. Soon-Shiong is also a heavy investor in publicly traded biotech stocks, he said.
The public markets last year welcomed dozens of new entrants from the world of biotech, but many have not been able to sustain the enthusiasm from investors. Many today are trading below their offering price, prompting some to speculate that there could be a biotech bubble.
Dr. Soon-Shiong says that is not the case, and that pressing medical needs will buoy many stocks beyond their initial ups and downs.
And while he stopped short of saying that his new company–Los Angeles-based Nantworks LLC–will definitely file to go public, he said he expects the company to become a powerful standalone player in the high-tech health system that the country is moving toward. He talked to Venture Capital Dispatch about the complex picture for new biotech issues on the public markets.
Q: What is your view of a situation where a lot of companies go public over a short time, and then have a very mixed performance? Is it something people should worry about?
A: No, absolutely not. The evolving tools of science, and their promise, have never been as exciting as right now. There are some real breakthrough drugs out now, particularly in cancer. It’s not a bubble, but people have followed each other [onto the public markets]. There’s been a pent-up demand for growth companies, and there’s capital out there. You will definitely see some of these companies stand the test of time.
As far as the ones that aren’t performing as well, sometimes it’s not the companies, it’s the market. The market is not scientifically driven. People should analyze the fundamentals, not the stock price.
Q: So how does one do that? How can people spot a healthy biotech company, putting aside its stock price?
A: There’s no way to generalize. But you ask: Is the science sound? Is there an unmet need? Has the company built a translational application that is demonstrable in trials? Can you do this at scale? Does the technology improve or impact outcomes for the patient?
If it brings value to the patient, it’s a company of value. We spend $4.3 trillion a year on health-care in the U.S. If the focus of your company is to increase value to the patient, then you will organically grow. A lot of these companies have zero revenue, so the only way to measure them, really, is to ask: Is there forward movement of this technology platform?
Q: What other advice would you give to biotech investors?
A: It’s science. It’s a 10-year horizon. Not everyone wants to invest with a 10-year horizon. There are long lead times, there are trials. If you’re impatient, as an investor, it can be bumpy. Choose wisely, because it takes a decade. It’s never a short-term thing.
When I took American Pharmaceutical Partners public in 1998, it opened at $14 a share. It dropped to $8 a share. Later on, we were so confident of the technology and the business model that we bought $30 million of the stock back. This was eight months after our IPO.
Our drug [a formulation of cancer drug Heparin] came out in 2001, and our stock hit $45 per share. It later split into two companies, and they were both acquired. In 2008, APP was the only safe supplier of Heparin in the country. That’s not a business model. That’s a company focused on benefitting the patient.
Q: So what does the future hold for these biotech stocks?
A: The strong will survive. Not strong as in financial strength, but sustainable value–value to the patient.
http://blogs.wsj.com/venturecapital/2014/06/26/doctor-founder-billionaire-talks-about-biotech-investment-stocks/?KEYWORDS=Dr+Patrick+Soon-Shiong
New investors, the statement “59% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “48% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
Document - Getting Ready for a BioPharma Partnering Deal
A typical large pharmaceutical might see 500 to 1,000 opportunities in a given therapeutic area. Only 100 or so of those will go on to non-confidential meetings and only 50 or so of those will end up going to Confidential Disclosure Agreements (CDAs) and confidential meetings.
Unfortunately, getting a deal done can take a long time. After initial talks, getting a CDA can take a month or more. It might take another month for the other side to assemble appropriate team members and schedule team meetings.
It can then easily take another month for them to evaluate all the information and to decide that they will indeed go forward with the formal due diligence process. From the point when the other party has decided to go forward with term sheet negotiations, it can take another six to twelve months to get to a signature on a full agreement.
Diligence and a Good Virtual Data Room
When a potential partner begins the due diligence process, they’re trying to get to know you and your asset in greater detail, and they’re attempting to verify what they’ve been told.
Give them access to your experts, provide overview presentations, and answer questions. In short, spend the time it takes to make sure they really understand it.
New investors, the statement “38% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “49% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
B-UP Due Diligence
IBD Development Update (10/24/2017)
"Oral formulation plans are underway to deliver Brilacidin more widely throughout the GI tract so as to enable treatment of more extensive forms of IBD—Crohn’s Disease and Ulcerative Colitis. A meeting with the Food and Drug Administration (FDA) is being requested to help inform appropriate next steps for our Brilacidin-IBD program, such as the size and scoape of subsequent clinical trials. Finally, the Company plans to continue exploration of Brilacidin’s various mechanisms of action, including its anti-inflammatory and anti-bacterial properties, as well as its immunomodulatory and wound healing capabilities—all of which likely have a role in Brilacidin’s ability to treat IBD."
http://www.ipharminc.com/press-release/2017/10/24/innovation-pharmaceuticals-provides-development-update-on-brilacidin-for-inflammatory-bowel-disease
Positive Trial Result (7/13/2017)
Rate of Clinical Remission (% of patients achieving)
- 60% Cohort A (3 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(i) #Endoscopy subscore ≤ 1 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(ii) Rectal Bleeding subscore of 0 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
(iii) Stool Frequency subscore, improvement or no change from baseline (% of patients achieving)
- 100% Cohort A (5 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
Endoscopic Image (Cohort B, 100 mg)

http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
Patient Treatment Update (6/26/2017)
"With the study treatments now completed, Brilacidin has consistently shown strong signs of efficacy while exhibiting a good safety profile even as dosing increased, supporting its potential as a novel, non-corticosteroid, non-biologic anti-inflammatory drug candidate in treating IBD. Innovation Pharmaceuticals’ formulation development plans for Brilacidin include foam and/or gel for the treatment of UP/UPS, and pills for oral dosing for the treatment of Ulcerative Colitis and Crohn’s Disease."
http://www.ipharminc.com/press-release/2017/6/24/innovation-pharmaceuticals-completes-treatments-in-phase-2-poc-trial-for-induction-of-remission-in-inflammatory-bowel-disease-topline-results-with-endoscopic-response-to-be-presented-july-13-2017
Third Cohort Update (5/18/2017)
“What is perhaps most remarkable about this trial is that Brilacidin was administered to patients in water as a retention enema,” said Leo Ehrlich, Chief Executive Officer at Cellceutix. “The logical next step is transitioning to a foam and gel/lotion formulation, with planning already underway, which would greatly facilitate Brilacidin’s adhesion to the mucosal lining of the gastrointestinal tract and may lead to even greater efficacy. Brilacidin for UP/UPS is well-positioned to become first-in-class.”
http://www.ipharminc.com/press-release/2017/5/15/cellceutix-completes-patient-enrollment-of-final-cohort-in-phase-2-trial-of-brilacidin-for-inflammatory-bowel-disease-topline-results-anticipated-in-july
Investor & Shareholder Update (4/20/2017)
https://www.youtube.com/watch?v=kzFu-jItfMw&t=474s
First & Second Cohort Top-line Data (3/21/2017)
Primary Efficacy Endpoint of Clinical Remission (accounting for Stool Frequency, Rectal Bleeding and Endoscopy Findings subscores)—50 percent of patients in Cohort A (3 of 6) and 50 percent of patients in Cohort B (3 of 6) met this endpoint; all 6 of the remaining patients in Cohorts A and B met 2 of the 3 criteria (Partial Response).
Full MMDAI (accounting for Stool Frequency, Rectal Bleeding, Physician’s Global Assessment and Endoscopy Findings subscores)—notable improvements observed in 10 of 11 patients (one patient declined endoscopy at end of treatment):
- 100 percent reduction in 2 patients in Cohort A and 2 patients in Cohort B
- 50-75 percent reduction in 2 patients in Cohort A and 4 patients in Cohort B
- 20 percent reduction in 1 patient in Cohort A
Partial MMDAI (accounting for Stool Frequency, Rectal Bleeding and Physician’s Global Assessment subscores)—notable improvements observed in 11 of 12 patients:
- 100 percent reduction in 3 patients in Cohort A and 3 patients in Cohort B
- 50-83 percent reduction in 2 patients in Cohort A and 3 patients in Cohort B
- 0 percent reduction in 1 patient in Cohort A
Patient Quality of Life (as assessed by the Short Inflammatory Bowel Disease Questionnaire, SIBDQ)—notable improvements in all 12 patients; 50 percent of patients in Cohort A (3 of 6) and 80 percent of patients in Cohort B (5 of 6) reported significant improvements of 15 points to more than 50 points higher on the 70-point SIBDQ scale.
Other Clinically Meaningful Observations
- Endoscopy subscore of ≤ 1 met in 7 of 11 patients (1 patient declined final endoscopy).
- PGA subscore improved for 10 of 12 patients.
- Rectal Bleeding subscore improved for all 12 patients.
- Stool Frequency subscore demonstrated improvement by study end for all patients who were abnormal at study start.
http://www.ipharminc.com/press-release/2017/3/19/cellceutix-releases-favorable-topline-findings-as-part-of-interim-analysis-of-phase-2-drug-candidate-brilacidin-for-the-treatment-of-inflammatory-bowel-disease
Second Cohort Update (3/8/2017)
All 12 patients experienced a beneficial response, as measured by the Modified Mayo Disease Activity Index (MMDAI).
- At Day 42, the Primary Efficacy Endpoint of Clinical Remission (accounting for Stool Frequency, Rectal Bleeding, and Endoscopy sub-scores) was met in half (n=6) of all patients (3 of 6 in Cohort A; 3 of 6 in Cohort B).
- Among the remaining patients (n=6) in Cohorts A and B not meeting all three criteria for Clinical Remission, two of the three criteria were achieved by all of these patients (defined as a Partial Response).
Patient Quality-of-Life, as assessed by the Short Inflammatory Bowel Disease Questionnaire (SIBDQ), was improved in all 12 patients after six weeks of daily Brilacidin treatment. Specifically, over 40 percent of patients reported significant improvements, ranging approximately from 20 points to more than 50 points higher on the 70-point SIBDQ scale.
http://www.ipharminc.com/press-release/2017/3/8/cellceutix-releases-preliminary-efficacy-and-safety-data-in-interim-analysis-of-first-two-cohorts-in-phase-2-trial-of-brilacidin-for-the-induction-of-remission-of-mild-to-moderate-ulcerative-colitis
Second Cohort Update (1/23/2017)
"Site investigators report enthusiastic feedback from patients currently receiving treatment in the second cohort with Brilacidin at 100 mg daily (by retention enema) for six weeks —comments included how the treatment already has greatly improved or eliminated their symptomatic complaints, daily functioning and overall quality of life. There is significant interest by new patients wishing to participate in the third (final) cohort once enrollment opens."
http://www.ipharminc.com/press-release/2017/1/22/cellceutixs-brilacidin-demonstrates-promise-in-treating-ulcerative-colitis-supported-by-endoscopic-assessment-patient-reported-outcomes
First & Second Cohort Update (12/7/2016)
"Review of safety data from the first cohort revealed that Brilacidin, administered for 6 weeks as a retention enema, at 50 mg once daily, appeared well-tolerated, with no measurable systemic absorption detected. This drug profile, at the lowest dose, allowed the approval of dose escalation to the second cohort (100 mg, once daily). Clinically meaningful improvements in symptoms of UP/UPS were also demonstrated in the first cohort, as measured by physician assessments and patient reported outcomes, further supported with endoscopic evaluation of disease activity."
http://www.ipharminc.com/press-release/2016/12/6/cellceutix-phase-2-trial-dose-escalates-in-2nd-cohort-for-brilacidin-as-a-novel-anti-inflammatory-drug-candidate-for-ulcerative-colitis
First Cohort Update (10/10/2016)
At Day 42, on the Partial MMDAI (accounting for Stool Frequency, Rectal Bleeding, and Physician’s Global Assessment scores), 2 of 4 patients achieved full response (100% reduction) and the other 2 patients had notable improvement (50% reduction).
At Day 42, on the MMDAI (equivalent to Partial MMDAI + Endoscopy score; completed by 3 of 4 patients), 1 of 3 patients achieved full response (100% reduction) and 2 of 3 patients had notable improvement (50% reduction). One patient who had demonstrated a full response on the Partial MMDAI (as defined in first bullet point) did not consent to the final endoscopy, so their data cannot be included in these initial results.
Patient quality of life, as assessed by the Short Inflammatory Bowel Disease Questionnaire (SIBDQ), was improved after 6-weeks of treatment with Brilacidin.
Brilacidin was generally well-tolerated and patients maintained stable normal vital signs during treatment.
http://www.ipharminc.com/press-release/2016/10/27/cellceutix-phase-2-trial-initial-data-shows-potential-of-brilacidin-as-a-novel-anti-inflammatory-drug-candidate-for-induction-of-remission-of-mild-to-moderate-ulcerative-colitis
First Patient Update (07/18/2016)
"Cellceutix has been advised that the first patient in Cohort A remains on study. The clinical site administering Brilacidin to this patient has informed the Company, “Both the patient and the site staff have been amazed how the study drug works as all the symptoms decreased significantly within the 1st week of treatment.” "
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-receives-update-on-first-patient-enrollment-in-phase-2-proof-of-concept-study-of-brilacidin-for-ulcerative-proctitis
About Ulcerative Proctitis
Ulcerative proctitis (UP), a limited type of ulcerative colitis (UC), is a mucosal inflammatory disease of unknown cause involving only the rectum. When it involves both the rectum and the distal colon, it is called Ulcerative Proctosigmoiditis (UPS). It is characterized by inflammation, redness, and ulcerations of the mucosa. The course of the disease is variable and ranges from complete resolution to easily maintained remission to chronic relapses or refractory disease. Diagnosis can occur at any point in life, with approximately 30-50 percent of patients developing more extensive UC. There is currently no cure. According to estimates provided by GlobalData, the worldwide UC market, which includes products for UP/UPS, is expected to increase at a compound annual growth rate of 4.7 percent, from $4.2 billion in 2012 to approximately $6.6 billion by 2022.
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-starts-phase-2-trial-of-brilacidin-as-a-novel-therapy-for-ulcerative-proctitis
Trial Design
The primary objective of the P-o-C trial is to assess the frequency of clinical and endoscopic remission with Brilacidin administered per rectum in subjects with active UP or UPS after 6 weeks of treatment. Secondary objectives include evaluation of safety and tolerability of Brilacidin when administered per rectum, evaluation of clinical remission at Week 2 and Week 4, assessment of systemic exposure and/or pharmacokinetics of Brilacidin when administered per rectum, assessment of the efficiency of Brilacidin by biomarker evaluation of biopsy samples for interleukin (IL)-6 and IL-1beta, and estimation of statistical power for subsequent trial(s) in UP and UPS.
The P-o-C trial will include 18 patients divided evenly into three cohorts. Cohort A is receiving 50 milligrams (mg) of Brilacidin once daily for 42 days. Dosing will be increased to 100mg and 200mg once daily for 42 days for Cohort B and Cohort C, respectively. Endoscopic evaluation of the rectum and mucosa up to 40 cm from the anal verge will be performed at screening and at the end of treatment/Day 42 (± 3 days). Per protocol, a safety committee will review safety and retention data (clinical laboratory findings, vital signs and adverse events) after 21 days of therapy for all six patients in each cohort before proceeding with initiating enrollment in the subsequent cohort.
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-receives-update-on-first-patient-enrollment-in-phase-2-proof-of-concept-study-of-brilacidin-for-ulcerative-proctitis
Q1 2017 Conference Call
During September we received initial comments on the first patient enrolled in our Phase 2 trial of Brilacidin for the inflammatory bowel disease, IBD, ulcerative proctitis or ulcerative proctosigmoiditis which detailed a significant decrease in symptoms within one week of Brilacidin activity. Early in the current quarter we followed that up with even better news of clinically meaningful improvements in the first four patients of the first cohort who completed the trial at the lowest dosing level. Yes, the lowest grossing level. Dr. Bertolino will provide some more color on the results. But the key is the fact that Brilacidin concentrations in the patient's plasma at all timeframes were basically undetectable. This is incredibly important in the safety profile that is being built for the FDA. We believe that a highly effective new drug with minimal systemic exposure will have competitive advantages to capture market share in a market for IBD medicine, the vision gain forecast to client to $9.3 billion in 2019.
Unidentified Analyst
Brilacidin seems to have very broad potential across a number of indications. Can you comment on why you chose to start with inflammatory bowel disease and ulcerative proctitis? Can you expand on the critical path for these indications and what other indications would you consider pursuing?
Leo Ehrlich
There is currently a significant medical need for effective treatment of inflammatory bowel disease. We look for massive markets with unmet needs, so IBD certainly qualified. The simplest way for us to do a proof-of-concept evaluation was to first explore ulcerative proctitis, ulcerative proctosigmoiditis. We have certainly been encouraged by preliminary results if it holds true that we can achieve significant clinical benefits with localized treatment and no significant systemic drug levels. We also anticipate potential for use in expanding IBD indications and in a number of dermatological diseases.
Unidentified Analyst
Dr. Bertolino, can you explain more about what is known of the science underlying the anti-inflammatory actions of Brilacidin?
Arthur P. Bertolino
Brilacidin is an immunomodulatory and anti-inflammatory agent. It inhibits the production of a number of key molecular targets, TNF-alpha, IL- 1 beta, MCP-1, MMP-9 and IL-6. Recent encouraging results in ulcerative proctitis, ulcerative proctosigmoiditis reassure us that the science is meaningful.
http://seekingalpha.com/article/4023682-cellceutix-corporation-ctix-ceo-leo-ehrlich-q1-2017-results-earnings-call-transcript
Interim Data
"A: Let me first touch on the ulcerative proctitis (UP) and oral mucositis (OM) trials. The UP trial is progressing smoothly and barring any unforeseen complications, we think that we will have some interim results sometime next quarter. This is a proof-of-concept study, which I believe can have a significant impact on the value of the Brilacidin franchise by serving as a gateway to other gastrointestinal diseases and conditions.”
http://seekingalpha.com/article/3988240-interview-new-cellceutix-president-dr-bertolino#alt2
Modified Mayo Disease Activity Index (MMDAI)
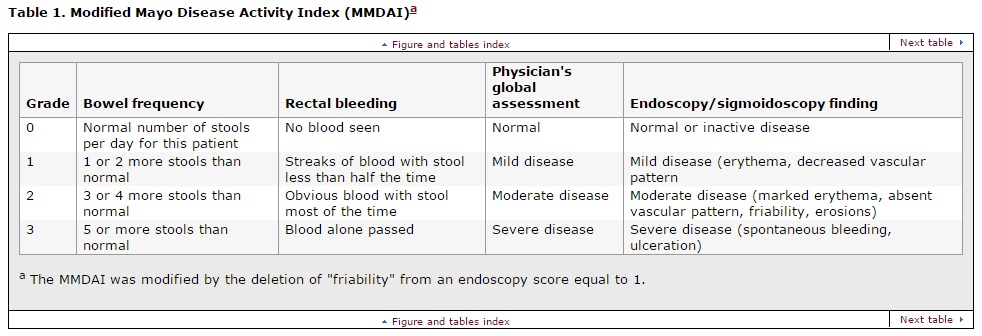
Clinical improvement was defined as a greater than or equal to 3 point improvement from baseline in the total MMDAI score and a greater than or equal to 1 point improvement from baseline in the rectal bleeding subscale of the MMDAI. Secondary efficacy end points included the proportion of patients in clinical remission, defined as a score of 0 for rectal bleeding and a combined score of less than or equal to 2 for bowel frequency and physician's assessment using the MMDAI subscales.
https://www.ncbi.nlm.nih.gov/pubmed/19491859
Ulcerative Proctitis
Abstract
Ulcerative proctitis is an idiopathic mucosal inflammatory disease involving only the rectum and is therefore an anatomically limited form of ulcerative colitis. Diagnosis is made based on clinical presentation, endoscopic appearance, and histopathology. Additionally, other etiologies of proctitis are excluded. The course of the disease is variable ranging from complete resolution to easily maintained remission to frequent relapses or refractory disease. Extension of inflammatory changes involving the proximal colon occurs in some cases. Rectal 5-aminosalicylic acid (5-ASA) or steroids are the initial treatments of choice with oral 5-ASA, sulfasalazine, or steroids used for treatment failures or patients unable to tolerate rectally administered drugs. Immunomodulators like azathioprine and 6-mercaptopurine have been used successfully in small groups of patients who have not responded to 5-ASA or steroids. Oral or rectal 5-ASA products maintain remission but long-term steroid use should be avoided. Rare cases may require surgical therapy.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2780078/
Foam Preparations
Abstract
Patients with ulcerative colitis uniformly have disease involving the distal colon. When patients have disease limited to the left colon or symptoms suggestive of active rectal inflammation, guidelines recommend topical rectal therapies as first-line agents either as monotherapy or in conjunction with oral products. Rectal delivery modalities offer the advantage of delivering high local concentrations of active medication to the site of maximal inflammation with minimization of systemic side effects. Methods of rectal administration include suppositories, liquid enemas and foams. Suppositories are limited to the treatment of rectal disease, and patients often have difficulty retaining the liquid enema secondary to its high volume and consistency. Rectal foams reliability extend to the descending and sigmoid colon with application. Foams are further characterized by increased viscosity, lower volumes, finer dispersion on the colonic mucosa, and increased adhesiveness to the colonic mucosa compared with liquid enemas. Additionally, rectal foam agents demonstrate equal efficacy to their liquid enema counterparts yet consistently yield better patient tolerance, lower incidence of side effects, and increased patient acceptability. Currently available agents include 5-aminosalicylic acid and corticosteroids, both first and newer generation. This review focuses on clinical trials assessing efficacy, tolerability, and patient preferences for these agents as well as describing the currently available rectal foam products.
http://www.ncbi.nlm.nih.gov/pubmed/21235478
Placebo Response Rate
Experience with foam preparations has demonstrated better retention and adherence, as well as more uniform distribution of medication in the distal colon and rectum, as compared with enemas and suppositories.
Analysis of the individual trials showed that 38.3% and 44% of patients in the budesonide arm achieved remission at 6 weeks as compared with 25.8% and 22.4% of the placebo groups (P=0.0324 and P<0.0001, respectively). Combined results showed a remission rate of 41.2% with budesonide foam and 24% with placebo (P<0.0001).
http://www.medpagetoday.com/meetingcoverage/acg/42311
Current and Future Therapies
Ulcerative Colitis and Crohn’s Disease
UC is a gastrointestinal (GI) disease that is localized to the large intestine, or colon, where inflammation can affect either the entire organ or a portion of it. Approximately 1.86 billion patients have been diagnosed with UC globally, with 1.54 billion patients currently receiving treatment. Traditional therapies have yielded $4.18 billion in annual sales around the world, a figure expected to increase to $6.85 billion by 2022 with the approval of various pipeline drugs.
CD can affect any part of the GI tract, but most commonly involves both the large and small intestines. Although CD is more severe than UC, the global prevalence is much lower, with only 1.3 million patients diagnosed and 0.8 million who currently receive treatment. Still, traditional therapies have resulted in an impressive $3.17 billion in global market sales, which is predicted to increase to $4.20 billion by 2022.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4123809/
Mesalamine in the Treatment and Maintenance of Remission of Ulcerative Colitis
Abstract
Ulcerative colitis (UC) is a chronic disease of the GI tract that is characterized by mucosal inflammation in the colon. Mesalamine (mesalazine) is a 5-aminosalicylic acid compound that is the first-line treatment for patients with mild-to-moderate UC. There are multiple formulations of mesalamine available, primarily differentiated by their means of delivering active mesalamine to the colon. Mesalamine has been demonstrated in randomized controlled trials to induce both clinical response and remission, and maintain clinical remission, in these patients. It has few serious adverse effects and is generally well tolerated by patients. The main areas of uncertainty with use of mesalamine in patients with UC center on the optimal dose for induction of response, how to maintain patient adherence and the role of mesalamine in cancer chemoprophylaxis. Generic forms of mesalamine have yet to be approved by regulatory bodies in the USA.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3314328/
Potential Market
The global ulcerative colitis (UC) market value will increase steadily over the coming years, growing from almost $4.2 billion in 2012 to approximately $6.6 billion by 2022, at a Compound Annual Growth Rate (CAGR) of 4.7%, according to a new report from research and consulting firm GlobalData.
Johnson & Johnson (J&J) has been continually dominating the UC market due to the success of Remicade. However, Remicade’s patent is set to expire between 2015 and 2018, which will knock J&J’s sales down from approximately $2 billion in 2012 to $1.5 billion by 2022, according to GlobalData.
Another key player in the UC industry, AbbVie’s Humira, will also lose its patent during the forecast period, with sales slipping from $1.2 billion in 2012 to $569 million by 2022.
https://healthcare.globaldata.com/media-center/press-releases/pharmaceuticals/global-ulcerative-colitis-market-simponi-and-entyvio-launches-to-boost-growth-by-2022-says-globaldata
http://www.prnewswire.com/news-releases/anti-inflammatory-therapeutics-market-is-expected-to-reach-1061-billion-globally-by-2020---allied-market-research-528652061.html
Recent IBD Deals
1. Janssen, Theravance ink GI pact worth up to $1B
“Janssen and Theravance Biopharma are teaming up to develop a JAK inhibitor for inflammatory bowel disease. Theravance will pick up $100 million up front, with the potential to earn another $900 million in milestone payments and royalties.”
“Under the agreement, Theravance will conduct a phase 2 study of the candidate, TD-1473, in Crohn’s disease, as well as a phase 2b/3 induction and maintenance study in ulcerative colitis, both slated to start this year. If all goes well, Janssen may pull the trigger on an exclusive license agreement for the program and take the lead on developing the drug in Crohn’s, according to a statement.”
“If the drug is approved, Janssen will be responsible for ex-U.S. marketing, with Theravance receiving tiered royalties, while the pair has the option of co-commercializing within the U.S. They will also split profits in the U.S., as well as development costs, with Janssen picking up 67% of the tab.”
https://www.fiercebiotech.com/biotech/janssen-theravance-ink-gi-pact-worth-up-to-1b
2. Johnson & Johnson commits $990M to land Protagonist’s oral Crohn’s drug (preclinical)
"Johnson & Johnson has landed rights to Protagonist Therapeutics’ preclinical Crohn’s disease asset PTG-200. The deal gives J&J a stake in an oral interleukin-23 (IL-23) receptor antagonist in return for $50 million upfront and up to $940 million in milestones."
"The asset is due to enter the clinic this year. Protagonist will run and fund the trial with J&J taking over—and covering 80% of the costs—once it advances to phase 2. Beyond this, a series of major paydays await Protagonist. J&J will pay Protagonist $125 million if it moves the drug into phase 2b and a further $200 million if it sticks with the asset after getting a look at data from the trial. The rest of the cash is tied to regulatory and sales milestones."
http://www.fiercebiotech.com/biotech/j-j-commits-990m-to-land-protagonist-s-oral-crohn-s-drug
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=138464059
UP Presentation
2017 Drug Discovery and Therapy World Congress
https://static1.squarespace.com/static/5715352e20c647639137f992/t/596cac7df14aa1a6e7118ed8/1500294274057/IPI+DDTWC+Brilacidin+presentation+%28final-u%29+13Jul2017.pdf
UP Blog
10/24/2017 - Deficient Defensins and Missing Mucins in the Pathogenesis of Inflammatory Bowel Disease
http://www.ipharminc.com/new-blog/2017/10/24/deficient-defensins-and-missing-mucins-in-the-pathogenesis-of-inflammatory-bowel-disease
7/13/2017 - Ulcerative Proctitis / Ulcerative Proctosigmoiditis — Opportunity for Newer Therapies Like Brilacidin to Emerge
http://www.ipharminc.com/new-blog/2017/7/13/ulcerative-proctitisulcerative-proctosigmoiditissubstantial-opportunity-exists-for-newer-therapies-like-brilacidin-to-emerge
7/6/2017 - Growing Interest in Novel Treatments for Inflammatory Bowel Disease
http://www.ipharminc.com/new-blog/2017/7/3/growing-interest-in-novel-non-biologic-treatments-for-inflammatory-bowel-disease
6/2/2017 - Dr. Francis A. Farraye on the Use of Endoscopy in Inflammatory Bowel Disease (IBD)
http://www.ipharminc.com/new-blog/2017/6/2/dr-francis-farraye-on-the-use-of-endoscopy-in-ibd
4/6/2017 - IBD Inpatient Costs Skyrocketing
http://www.ipharminc.com/new-blog/2017/2/2/ibd-inpatient-costs-skyrocketing-1
2/17/2017 - Endoscopic Remission in the Treatment of Ulcerative Colitis
http://www.ipharminc.com/new-blog/2017/2/2/endoscopic-remission-in-the-treatment-of-ulcerative-colitis
Dr. Francis A. Farraye – Scientific Advisor, Gastroenterology
Dr. Francis A. Farraye serves as Professor of Medicine, Clinical Director, Section of Gastroenterology and Co-Director, Center for Digestive Disorders, at Boston University School of Medicine. Dr. Farraye is a Fellow of the American College of Physicians, American Society of Gastrointestinal Endoscopy, American Gastroenterological Association and the American College of Gastroenterology. Across his professional career, he has published over 350 original manuscripts, abstracts and book chapters, with particular clinical interest in inflammatory bowel disease and colorectal cancer. Dr. Farraye has been named Humanitarian of the Year by the New England Chapter of the Chrohn’s & Colitis Foundation of America. Dr. Farraye received a MD from Albert Einstein College of Medicine in New York and a MSc (Epidemiology) from Harvard University.
http://www.cellceutix.com/senior-management-1/
Books
Questions & Answers About Ulcerative Colitis
https://books.google.com/books?id=45iIz7k14pwC&printsec=frontcover&dq=ulcerative+colitis+farraye&hl=en&sa=X&ved=0ahUKEwi5lJHKndHPAhVHQSYKHTgBCCwQ6AEINDAA#v=onepage&q=ulcerative%20colitis%20farraye&f=false
Curbside Consultations in IBD: 49 Clinical Questions
https://books.google.com/books?id=JQaR2Sk2Ek4C&printsec=frontcover&dq=curbside+consultation&hl=en&sa=X&ved=0ahUKEwiruMDTnNHPAhXIVyYKHd68A3gQ6AEIHjAA#v=onepage&q=curbside%20consultation&f=false
Presentations
Health Maintenance for your IBD Patient (Slide 104)
http://www.galamericas.org/dallas/library/PPTS/June2014/Inflammatory_Bowel_Disease.pdf
The role of chromoendoscopy in ulcerative colitis patients: Should we incorporate the SCENIC recommendations? (Slide 10)
http://advancesinibd.com/archive/2015/presentations/Friday/Clinical/Session-IVA/1030_Challenges%20in%20IBD_Med.pdf
Publications
https://www.researchgate.net/profile/Francis_Farraye/publications
YouTube
How can we best determine endoscopic severity in ulcerative colitis and Crohn's disease
https://www.youtube.com/watch?v=mdR7UCm33xg&t=23s
Diagnosis and Management of Colorectal Neoplasia in Inflammatory Bowel Disease
https://www.youtube.com/watch?v=kV5-9qwetmc
B-OM Due Diligence
Lastest Update
5/9/2018 - Innovation Pharmaceuticals Concludes Data Analysis of its Phase 2 Clinical Trial for Severe Oral Mucositis in Head and Neck Cancer; Positioning to Fill a Substantial Void in Supportive Cancer Care
“Given the notable interest from global and specialty pharmaceutical companies that we have received following recent Brilacidin-OM data releases, we continue to carefully assess all of our potential alliance opportunities—toward cementing the best pathway forward,” said Leo Ehrlich, Chief Executive Officer of Innovation Pharmaceuticals. “The expedient advancement of Brilacidin-OM into later clinical development is a priority, as is the broader expansion of the complete Brilacidin Franchise.”
http://www.ipharminc.com/press-release/2018/5/9/innovation-pharmaceuticals-concludes-data-analysis-of-its-phase-2-clinical-trial-for-severe-oral-mucositis-in-head-and-neck-cancer-positioning-to-fill-a-substantial-void-in-supportive-cancer-care
4/23/2018 - Innovation Pharmaceuticals Signs Drug Supply Contract with Evonik to Bulk Produce Commercial-Grade Brilacidin
"Securing a commercial-grade drug supply of Brilacidin with a leading Contract Development and Manufacturing Organization (CDMO), such as Evonik, is an important and necessary step that will enable Innovation Pharmaceuticals to more rapidly advance the Brilacidin Franchise, toward getting much-needed treatments to patients."
http://www.ipharminc.com/press-release/2018/4/23/innovation-pharmaceuticals-signs-drug-supply-contract-with-evonik-to-bulk-produce-commercial-grade-brilacidin
4/16/2018 - Innovation Pharmaceuticals Data from Phase 2 Brilacidin Oral Mucositis (OM) Trial in Head and Neck Cancer Show Notable Reductions in Median Duration of Severe OM and in Number of Unplanned Visits/Hospital Admissions Due to OM
· Median Duration of Severe Oral Mucositis (SOM) Reduced to Less Than One Day
· No Unplanned Office Visits, Emergency Department Visits, and/or Hospital Admissions Due to OM in Brilacidin-OM Arm vs. Four in Placebo Arm
· Data Align with Previously Reported 80.3% Risk Reduction in Incidence of Severe OM in Per Protocol Population Receiving Aggressive Chemotherapy Regimen Compared to Placebo
http://www.ipharminc.com/press-release/2018/4/16/innovation-pharmaceuticals-data-from-phase-2-brilacidin-oral-mucositis-om-trial-in-head-and-neck-cancer-show-notable-reductions-in-median-duration-of-severe-om-and-in-number-of-unplanned-visit
4/9/2018 - Innovation Pharmaceuticals Phase 2 Oral Mucositis Trial Additional Data Show Brilacidin-OM Demonstrated A Significant Reduction in the Incidence of Severe Oral Mucositis in Patients with Head and Neck Cancer (HNC) Receiving Aggressive Chemotherapy Regimen
“For the Modified Intent-to-Treat (mITT) population, Brilacidin-OM in the aggressive chemotherapy regimen reduced the incidence of SOM by 65.0% ([incidence control- incidence active]/incidence control) as compared with placebo (Brilacidin: 25.0%; placebo: 71.4%; p=0.0480). For the Per Protocol (PP) population, Brilacidin-OM in the aggressive chemotherapy regimen similarly reduced the incidence of SOM by 80.3% as compared with placebo (Brilacidin: 14.3%; placebo: 72.7%; p=0.0249).”
http://www.ipharminc.com/press-release/2018/4/9/innovation-pharmaceuticals-phase-2-oral-mucositis-trial-additional-data-show-brilacidin-om-demonstrated-a-significant-reduction-in-the-incidence-of-severe-oral-mucositis
3/15/2018 - Innovation Pharmaceuticals Views Brilacidin-OM as Clearly Differentiated Compared to Limited Competition for Preventing Severe Oral Mucositis
“The Company is currently focused on completing the Clinical Study Report for its Phase 2 clinical trial (see NCT02324335) evaluating Brilacidin’s ability to safely prevent and attenuate SOM in HNC patients receiving chemoradiation and compiling a Briefing Package for discussion with the FDA.”
http://www.ipharminc.com/press-release/2018/3/15/innovation-pharmaceuticals-views-brilacidin-om-as-clearly-differentiated-compared-to-limited-competition-for-preventing-severe-oral-mucositis
1/29/2018 - Innovation Pharmaceuticals Brilacidin Franchise Anchored in Three Clinical Indications — Oral Mucositis, Inflammatory Bowel Disease and Serious Skin Infections; Expands into Dermatologic Diseases
"A leading international drug manufacturer has been engaged with to bulk produce commercial-quality Brilacidin, aimed at lowering patient drug cost and anticipating future drug needs in preparation for expedient market introduction. This critical step also proactively facilitates future patient and insurance reimbursement adoption through favorable cost savings;"
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
1/3/2018 - Innovation Pharmaceuticals Brilacidin Meets Key Secondary Endpoint in Phase 2 Trial, Delays Onset of Severe Oral Mucositis (SOM)
"Onset of Severe Oral Mucositis: Based on Kaplan-Meier curves, Brilacidin-OM oral rinse showed a clear separation from placebo in delaying the onset of SOM—particularly the period from approximately 28-42 days, after the initiation of treatment, during which the incidence of SOM rose strikingly in the placebo group while not in the group being treated with Brilacidin."
http://www.ipharminc.com/press-release/2018/1/3/innovation-pharmaceuticals-brilacidin-meets-key-secondary-endpoint-in-phase-2-trial-delays-onset-of-severe-oral-mucositis-som
Kaplan-Meier Curves

12/18/2017 - Innovation Pharmaceuticals Brilacidin Oral Mucositis Program Moving Forward Based Upon Positive Anchoring Phase 2 Results and Increased Brilacidin Franchise Value
"Towards commercial planning, the Company has begun exploring potential unit dose drug product packaging in the form of a sachet. Sachets, which people are very familiar with and use on an almost daily basis (e.g., sugar packets, artificial sweeteners) increasingly are being developed as a novel means of patient-friendly, drug delivery. Manufacturing plans for drug substance appropriate for late phase testing and eventual market introduction are also underway."
http://www.ipharminc.com/press-release/2017/12/18/innovation-pharmaceuticals-brilacidin-oral-mucositis-program-moving-forward-based-upon-positive-anchoring-phase-2-results-and-increased-brilacidin-franchise-value
12/12/2017 - Innovation Pharmaceuticals Granted European Patent for Brilacidin in the Prevention of Oral Mucositis
"The European patent supplements other Brilacidin-OM patents that have been granted in the United States, Asia (Japan, Taiwan, China), Oceania (Australia) and South Africa. All currently issued patents have an expiration date of 2032. Additional Brilacidin-OM patent applications are pending in other key markets including Russia and South Korea."
http://www.ipharminc.com/press-release/2017/12/12/innovation-pharmaceuticals-granted-european-patent-for-brilacidin-in-the-prevention-of-oral-mucositis
12/11/2017 - Innovation Pharmaceuticals Reports Positive Topline Results from Phase 2 Placebo-Controlled Trial of Brilacidin for the Prevention of Oral Mucositis in Head and Neck Cancer Patients
Summary of Topline Results from the Placebo-Controlled Phase 2 Trial
· Brilacidin met primary endpoint of reduced incidence of severe OM experienced by patients during radiation therapy.
· Incidence of severe OM in Modified Intent to Treat (mITT) Population: Brilacidin 42.9%, Placebo 60.0%.
· Incidence of severe OM in Per Protocol (PP) Population: Brilacidin 36.8%, Placebo 60.0%.
· Trial results support continued and expedited development of Brilacidin-OM.
http://www.ipharminc.com/press-release/2017/12/11/innovation-pharmaceuticals-reports-positive-topline-results-from-phase-2-placebo-controlled-trial-of-brilacidin-for-the-prevention-of-oral-mucositis-in-head-and-neck-cancer-patients
11/16/2017 - Innovation Pharmaceuticals Offers Perspectives on Brilacidin as a Potential Preventative Treatment for Oral Mucositis in Head and Neck Cancer Patients
"Formal collaboration with pharmaceutical companies that have expressed an interest in partnering Brilacidin-OM may well assist further with expediting the drug candidate’s development timetable. Some of these partnering conversations have matured to the point of potentially structuring mutually beneficial licensing agreements, pending the final Phase 2 study results."
http://www.ipharminc.com/press-release/2017/11/16/innovation-pharmaceuticals-offers-perspectives-on-brilacidin-as-a-potential-preventative-treatment-for-oral-mucositis-in-head-and-neck-cancer-patients
10/26/2017 - Innovation Pharmaceuticals Aims to Develop First Drug for Approval in Prevention of Oral Mucositis in Head and Neck Cancer Patients as Phase 2 Clinical Trial of Brilacidin Completes
"The Company has begun the process of closing trial study sites and is now aggregating patient data for top-line analysis, to be reported this quarter."
http://www.ipharminc.com/press-release/2017/10/25/jk5qd8ltvc4ed00ekrbqwbej1vz0x1
10/18/2017 - Innovation Pharmaceuticals to Complete Phase 2 Trial of Brilacidin for Oral Mucositis in Cancer Patients
"After the final patient post-treatment follow-up is conducted next week, the Company will focus on the process of concluding the study, including closing clinical sites and aggregating and analyzing data."
http://www.ipharminc.com/press-release/2017/10/18/innovation-pharmaceuticals-to-complete-phase-2-trial-of-brilacidin-for-oral-mucositis-in-cancer-patients
10/2/2017 - Final Patient Completes Treatment in Innovation Pharmaceuticals Phase 2 Trial of Brilacidin for Preventing Oral Mucositis in Cancer Patients
"... today announces that the last patient has completed study treatment in the Company’s Phase 2 clinical trial of Brilacidin for the prevention and treatment of severe Oral Mucositis (OM) in patients undergoing chemoradiation for Head and Neck Cancer."
http://www.ipharminc.com/press-release/2017/10/2/final-patient-completes-treatment-in-innovation-pharmaceuticals-phase-2-trial-of-brilacidin-for-oral-mucositis
8/7/2017 - Innovation Pharmaceuticals Completes Patient Enrollment in Phase 2 Study of Brilacidin for the Prevention of Severe Oral Mucositis
"... today announces that it has completed patient enrollment for its ongoing Phase 2 clinical study of Brilacidin for the prevention of Severe Oral Mucositis (OM). A total of 61 patients have been enrolled, with topline results anticipated in 4Q2017."
http://www.ipharminc.com/press-release/2017/8/6/innovation-pharmaceuticals-completes-patient-enrollment-in-phase-2-study-of-brilacidin-for-the-prevention-of-severe-oral-mucositis
3/27/2017 - Cellceutix Reports Very Encouraging Interim Analysis of Phase 2 Drug Candidate Brilacidin for Severe Oral Mucositis (OM) in Head and Neck Cancer Patients; High Potential for Preventative Treatment
"Study showed a markedly reduced rate of Severe OM (WHO Grade ≥ 3): Active Arm (Brilacidin): 2 of 9 patients (22.2 percent); Control Arm (Placebo): 7 of 10 patients (70 percent)"
Primary Efficacy Results: Incidence of Severe OM (WHO Grade ≥ 3)
• Active Arm (Brilacidin): 2 of 9 patients (22.2 percent)
• Control Arm (Placebo): 7 of 10 patients (70 percent)
Secondary Efficacy Results: Duration of Severe OM (WHO Grade ≥ 3)
• Active Arm (Brilacidin): Mean 10.5 days (Range 3 to 18 days; 2 patients)
• Control Arm (Placebo): Mean 14 days (Range 3 to 39 days; 7 patients)
http://www.ipharminc.com/press-release/2017/3/27/cellceutix-reports-very-encouraging-interim-analysis-of-phase-2-drug-candidate-brilacidin-for-severe-oral-mucositis-om-in-head-and-neck-cancer-patients-high-potential-for-preventative-treatment
Fast Track
11/25/2015 - FDA Grants Fast Track Designation to Cellceutix’s Brilacidin-OM for Oral Mucositis
"The FDA established the Fast Track Designation process to facilitate the development, and expedite the review of, drugs that have the potential to treat serious and life threatening conditions and fill an unmet medical need. Drugs developed under the Fast Track program are afforded increased access to the FDA and could qualify for other programs to expedite development, including priority review and accelerated approval."
http://www.ipharminc.com/press-release/2016/11/16/fda-grants-fast-track-designation-to-cellceutixs-brilacidin-om-for-oral-mucositis
Study Design
CTIX-BRI-205 is a Phase 2 randomized, double-blind, placebo-controlled study evaluating the safety and efficacy of Brilacidin as an oral rinse in preventing and controlling OM in patients receiving chemoradiation therapy for Head and Neck Cancer. The study is anticipated to enroll approximately 60 patients in the United States, 30 each to Brilacidin treatment or to placebo (water). Brilacidin (45 mg/15 ml oral rinse—“swish and spit”) is administered 3 times daily across 7 weeks (49 days). Pharmacokinetics of Brilacidin are to be evaluated if there is measurable systemic exposure (from drug concentrations in plasma).
The Brilacidin OM trial uses a World Health Organization (WHO) OM Grading Scale, a common measurement tool in assessing the presence and severity of OM, as defined below.
WHO Scale for OM
• Grade 0 = None
• Grade 1 = Erythema and Mouth Pain Soreness; no Ulceration/Pseudomembrane formation
• Grade 2 = Ulceration/Pseudomembrane formation; solid diet
• Grade 3 = Ulceration/Pseudomembrane formation; liquid diet
• Grade 4 = Ulceration/Pseudomembrane formation; not able to tolerate a solid or liquid diet (except enough liquid for medication)
About Oral Mucositis
Oral Mucositis (OM) is a frequent, painful and debilitating complication of chemoradiation commonly manifesting in the treatment of Head and Neck Cancer. Characterized by inflammation and ulceration, patients suffering from OM are often unable to speak or eat (requiring the insertion of a feeding tube) and are more susceptible to bacterial infections, with severe cases leading to hospitalization and increased treatment costs of up to $25,000. Affecting over 500,000 people in the United States, there currently are no approved medications for the prevention of OM in this population, with only limited palliative care options available. Worldwide, the potential market for OM is expected to exceed $1 billion in the next few years.
For more information on the CTIX-BRI-205 Phase 2 study, please visit:
https://clinicaltrials.gov/ct2/show/NCT02324335
OM Market
"Head and Neck Cancer (HNC) patients -- comprising an estimated 65,000 newly diagnosed cases in the U.S. alone in 2017, and an estimated 700,000 worldwide (source: GLOBOCAN) -- are at greatest risk of developing OM (a 90 to 100 percent rate of occurrence). By 2030, the global incidence of HNC cases is expected to exceed 1 million per year. Moreover, between 25 and 60 percent of cancer patients, regardless of cancer type, also will experience OM during the course of their chemo/radiotherapy.
Estimates vary as to the market size (in dollars) of an effective OM treatment, for HNC-only patients, across major markets (U.S., Europe and Japan), ranging between $500 million and $1.5 billion on an annual basis (sources: GlobalData; Redington Inc., pdf). One company in the OM space projects the worldwide OM market opportunity to be as high as $2.6 billion annually."
http://www.ipharminc.com/new-blog/2017/10/17/the-market-opportunity-in-oral-mucositis
Potential Competitors
Taking a Page from Merck’s Gardasil and Glaxo’s Requip, These Small Companies Look to Cement Riches for Shareholders in New Category of Oral Mucositis
http://www.baystreet.ca/articles/stockstowatch/38594/Taking-a-Page-from-Mercks-Gardasil-and-Glaxos-Requip-These-Small-Companies-Look-to-Cement-Riches-for-Shareholders-in-New-Category-of-Oral-Mucositis
A Billion Dollar Market Just Waiting for a New Oral Mucositis Drug: Five Companies That Want It
http://www.baystreet.ca/stockstowatch/2658/A-Billion-Dollar-Market-Just-Waiting-for-a-New-Oral-Mucositis-Drug-Five-Companies-That-Want-It
Some of the players operating for the oral mucositis are
- Izun Pharmaceutical Ltd (US)
- Shoreline Pharmaceuticals Inc. (USA)
- Himalaya (India)
- AMAG Pharmaceuticals Inc. (USA)
- Kinnear Pharmaceuticals (US)
- Celleutix Corporation(USA)
- Soligenix Inc. (US)
- Oragenics (US)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135072984
Amgen Kepivance (IV)
"In addition to a lower incidence of severe oral mucositis, patients receiving palifermin had almost one week less severe mucositis compared to those receiving placebo (10.4 days vs 3.7 days). Furthermore, palifermin-treated patients reported 60 percent less soreness of the mouth and throat, required lower doses of painkillers and less total parenteral nutrition use (11 percent versus 40 percent with placebo)."
http://www.amgen.com/media/news-releases/2003/06/phase-3-data-suggest-palifermin-significantly-reduces-the-duration-and-incidence-of-oral-mucositis-in-cancer-patients/
"The cost for Kepivance intravenous powder for injection 6.25 mg is around $14,401 for a supply of 6 powder for injection, depending on the pharmacy you visit."
https://www.drugs.com/price-guide/kepivance
Soligenix SGX942 (IV)
"n the 1.5 mg/kg treatment group, the median duration of severe oral mucositis was decreased by 50%, from 18 days to 9 days (p=0.099), meeting the prospectively defined statistical threshold of p<0.1 in the study protocol. Further, patients receiving the most aggressive CRT in this dose group had even more striking reductions in their median duration of severe oral mucositis of 67%, from 30 days to 10 days (p=0.040)."
http://www.soligenix.com/news/soligenix-announces-positive-preliminary-results-from-its-phase-2-clinical-trial-of-sgx942-for-the-treatment-of-oral-mucositis-in-head-and-neck-cancer-patients/
Validive (applies to gum)
"The incidence of SOM (primary endpoint) was reduced by 26.3% (40% relative to placebo) in OPC patients treated with Validive (100 µg) (p=0.09)"
http://monopartherapeutics.com/phase_II
"Onxeo Grants Exclusive Worldwide License of Validive® developed for the treatment of oral severe mucositis to Monopar Therapeutics"
https://www.onxeo.com/onxeo-grants-exclusive-worldwide-license-validive-developed-treatment-oral-severe-mucositis-monopar-therapeutics/
Izun IZN-6N4 (oral rinse)
"Not only did patients treated with IZN-6N4 have less mouth and throat pain and soreness than controls, but importantly, they were also more able to maintain their weights throughout the course of radiotherapy. The data supports that the best efficacy of IZN-6N4 was related to its initiation of use at the start of chemoradiation."
http://www.businesswire.com/news/home/20171011005127/en/Izun-Pharmaceuticals-Announces-Positive-Results-Phase-2
Oragenics AG013 (oral rinse)
"Data from the Phase 1B trial published in the journal Cancer showed AG013 was safe, well tolerated, and demonstrated preliminary efficacy with a 35% reduction in the duration of ulcerative OM compared to placebo."
https://www.oragenics.com/news-media/press-releases/detail/37/oragenics-announces-positive-results-from-confirmatory
OM Presentation
January 2018 Corporate Overview
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5a4e50e39140b721186d1a6f/1515081962344/2018Jan+IPIX+Corporate+Overview.pdf
2017 Drug Discovery and Therapy World Congress
https://static1.squarespace.com/static/5715352e20c647639137f992/t/596cac7df14aa1a6e7118ed8/1500294274057/IPI+DDTWC+Brilacidin+presentation+%28final-u%29+13Jul2017.pdf
25th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID 2015)
https://static1.squarespace.com/static/5715352e20c647639137f992/t/583f83821b631be3d85bc071/1480557442978/ECCMID-2015-OM-poster.pdf
OM Blog
5/4/2018 - Brilacidin-OM in the News: Oral Mucositis Market Untapped—Framed as “Blue Sky” Opportunity
http://www.ipharminc.com/new-blog/2018/5/4/brilacidin-om-in-the-news-oral-mucositis-market-untappedframed-as-blue-sky-opportunity
12/11/2017 - Brilacidin-OM Called a “Top Contender” in Oral Mucositis Market
http://www.ipharminc.com/new-blog/2017/12/11/brilacidin-om-called-a-top-contender-in-oral-mucositis-market
10/17/2017 - The Market Opportunity in Oral Mucositis
http://www.ipharminc.com/new-blog/2017/10/17/the-market-opportunity-in-oral-mucositis
8/7/2017 - Brilacidin for the Prevention of Severe Oral Mucositis
http://www.ipharminc.com/new-blog/2017/8/7/brilacidin-for-oral-mucositis
5/19/2017 - On the Unmet Medical Need in Oral Mucositis: Brilacidin as a Potential First-in-Class Preventative Treatment
http://www.ipharminc.com/new-blog/2017/5/17/on-the-unmet-need-in-oral-mucositis-and-brilacidin-as-a-promising-one-of-a-kind-treatment
Stephen Sonis, MD
Scientific Advisor, Oral Mucositis
Dr. Sonis is one of the world’s foremost experts in the research and clinical treatment of cancer-related oral mucosal toxicities. He currently holds appointments at the Harvard School of Dental Medicine as Professor of Oral Medicine (part-time), and is a Senior Surgeon at the Dana-Farber Cancer Institute and Brigham and Women’s Hospital. Dr. Sonis also is a Founder and Chief Scientific Officer of Biomodels, LLC, a preclinical contract research organization. Widely respected by his professional peers, he is the author of over 250 original publications, reviews and chapters, 11 books, and 5 patents, serves on a number of editorial boards, and is a founding member of the International Society of Oral Oncology and the International Academy of Oral Oncology. Dr. Sonis is a graduate of Tufts University and Harvard University and completed his post-doctoral education at Oxford University.
http://www.ipharminc.com/senior-management-1/
Cellceutix Corporation Welcomes Dr. Stephen T. Sonis to its Scientific Advisory Board
http://www.ipharminc.com/press-release-2/2016/9/19/cellceutix-corporation-welcomes-dr-stephan-t-sonis-to-its-sceintific-advisory-board
Stephen T. Sonis, DMD, DMSc, on Radiotherapy-Induced Oral Complications
https://vimeo.com/182309692
Q3 Potential Milestones
1. B-OM - Breakthrough Therapy Designation
2. Prurisol - Top-line Data (Phase 2b)
3. Licensing Agreement (double-digits millions upfront payment)
http://www.ipharminc.com/press-release/2018/6/28/innovation-pharmaceuticals-announces-7-million-milestone-funding-agreement-with-aspire-capital
Your ability to link Regulation Fair Disclosure ‘impresses’ me.
The REAL way to impress me would have been to accommodate my simple request instead of telling me "Sorry pal but you're gonna have to find another dance partner".
Good point. I was being conservative because Phase 2b has almost twice as many patients as Phase 2a. July is here already so we don’t need to wait long.
I expect to see the Phase 2b top-line result in August. Phase 2a was locked on 4/19/2016 and top-line result was released on 5/24/2016, exactly five weeks later.
Cellceutix Initiates "Database Soft Lock" On Its Phase 2 Psoriasis Clinical Trial, Top Line Results Expected in May; Additional Company Updates
http://www.ipharminc.com/press-release/2016/11/15/cellceutix-initiates-database-soft-lock-on-its-phase-2-psoriasis-clinical-trial-top-line-results-expected-in-may-additional-company-updates
Cellceutix Phase 2 Trial of Prurisol for Mild to Moderate Psoriasis Meets Primary Endpoint
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
New investors, the statement “16% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “37% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “20% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “29% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
BP's interests in the Big Three
“The Company is engaged in discussions and negotiations regarding the out-licensing of its mid-stage, first-in-class clinical assets to global and/or specialty pharmaceutical companies who have expressed an interest in one or more assets in our pipeline.”
https://www.sec.gov/Archives/edgar/data/1355250/000147793218002302/ipix_10q.htm
“The Confidential Disclosure Agreement (CDA) count toward partnering with global and specialty pharmaceutical companies interested in the Company’s first-in-class drug candidates is nearing 20, with additional Agreements in review."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
“These recent data, taken in aggregate, are what various actively engaged pharmaceutical companies have desired from us for some time—and they are what triggered a newfound flurry of inbound partnership discussions at the San Francisco conferences,”
http://www.ipharminc.com/press-release/2018/1/16/innovation-pharmaceuticals-presents-at-2018-biotech-conference-oral-mucositis-drug-candidate-garners-exceptional-interest-after-successful-phase-2-trial
"To advance our pipeline in an expeditious manner, this year we placed an emphasis on partnering—actively engaging global and regional pharmaceutical companies interested in licensing our novel drug candidates. These efforts have led to highly productive, mature (late-phase, ongoing) discussions, with multiple pharmaceutical companies, which we hope will bear fruit in the coming year." - CEO Letter to Shareholders
"We had said that confidential disclosure agreements have been signed with several pharmas interested in our drug candidates; to some, they're interested in one compound. Others have requested information on a combination of Brilacidin and Kevetrin and Prurisol."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
B-Dermatitis
"To further these efforts, the Company is in negotiations with a leading drug formulator to develop topical formulation(s) of Brilacidin for these three dermatology indications, starting in 1H2018. The goal of the negotiations is to reach terms on a strategic partnership for addressing these markets."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
B-OM
“Given the notable interest from global and specialty pharmaceutical companies that we have received following recent Brilacidin-OM data releases, we continue to carefully assess all of our potential alliance opportunities—toward cementing the best pathway forward,” said Leo Ehrlich, Chief Executive Officer of Innovation Pharmaceuticals.”
http://www.ipharminc.com/press-release/2018/5/9/innovation-pharmaceuticals-concludes-data-analysis-of-its-phase-2-clinical-trial-for-severe-oral-mucositis-in-head-and-neck-cancer-positioning-to-fill-a-substantial-void-in-supportive-cancer-care
“Formal collaboration with pharmaceutical companies that have expressed an interest in partnering Brilacidin-OM may well assist further with expediting the drug candidate’s development timetable. Some of these partnering conversations have matured to the point of potentially structuring mutually beneficial licensing agreements, pending the final Phase 2 study results.”
http://www.ipharminc.com/press-release/2017/11/16/innovation-pharmaceuticals-offers-perspectives-on-brilacidin-as-a-potential-preventative-treatment-for-oral-mucositis-in-head-and-neck-cancer-patients
B-UP
"We've actively engaged with multiple large pharmas, who have expressed an interest in Brilacidin inflammatory bowel disease. The pharma industry is quite interested in pursuing novel IBD therapies one of the more active therapeutic areas when it comes to dealmaking."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
Prurisol
"We have entered into multiple non-disclosure agreements with large pharmaceutical companies that enable us to continue ongoing discussions regarding potential partnering should the trial results support such a relationship."
https://www.sec.gov/Archives/edgar/data/1355250/000147793217004400/ipix_10k.htm
Kevetrin
"The Company is adhering to a value-building strategy born from discussions with larger pharmaceutical companies interested in Kevetrin, one of the world’s most advanced p53 drug candidates. Securing the right development partner for Kevetrin remains an important objective."
http://www.ipharminc.com/press-release/2018/2/8/innovation-pharmaceuticals-concludes-successful-phase-2a-trial-of-kevetrin-for-ovarian-cancer-intra-tumor-modulation-of-p53-observed
"What we and potential partners are extremely interested in is learning via tumor biopsies if Kevetrin is reaching its target and modulating pathways within the tumor that can deliver a clinically meaningful benefit; this would be a significant development in the p53/oncology dynamic,"
http://www.ipharminc.com/press-release/2017/9/14/innovation-pharmaceuticals-opens-new-clinical-site-for-novel-p53-drug-candidate-in-phase-2-ovarian-cancer-trial-1
Document - Getting Ready for a BioPharma Partnering Deal
A typical large pharmaceutical might see 500 to 1,000 opportunities in a given therapeutic area. Only 100 or so of those will go on to non-confidential meetings and only 50 or so of those will end up going to Confidential Disclosure Agreements (CDAs) and confidential meetings.
Unfortunately, getting a deal done can take a long time. After initial talks, getting a CDA can take a month or more. It might take another month for the other side to assemble appropriate team members and schedule team meetings.
It can then easily take another month for them to evaluate all the information and to decide that they will indeed go forward with the formal due diligence process. From the point when the other party has decided to go forward with term sheet negotiations, it can take another six to twelve months to get to a signature on a full agreement.
Diligence and a Good Virtual Data Room
When a potential partner begins the due diligence process, they’re trying to get to know you and your asset in greater detail, and they’re attempting to verify what they’ve been told.
Give them access to your experts, provide overview presentations, and answer questions. In short, spend the time it takes to make sure they really understand it.
IPIX Clinical Trial Summaries
B-OM (Phase 2)
Kaplan-Meier Curves

"Onset of Severe Oral Mucositis: Based on Kaplan-Meier curves, Brilacidin-OM oral rinse showed a clear separation from placebo in delaying the onset of SOM—particularly the period from approximately 28-42 days, after the initiation of treatment, during which the incidence of SOM rose strikingly in the placebo group while not in the group being treated with Brilacidin."
http://www.ipharminc.com/press-release/2018/1/3/innovation-pharmaceuticals-brilacidin-meets-key-secondary-endpoint-in-phase-2-trial-delays-onset-of-severe-oral-mucositis-som
Topline Results
· Brilacidin met primary endpoint of reduced incidence of severe OM experienced by patients during radiation therapy.
· Incidence of severe OM in Modified Intent to Treat (mITT) Population: Brilacidin 42.9%, Placebo 60.0%.
· Incidence of severe OM in Per Protocol (PP) Population: Brilacidin 36.8%, Placebo 60.0%.
· Trial results support continued and expedited development of Brilacidin-OM.
http://www.ipharminc.com/press-release/2017/12/11/innovation-pharmaceuticals-reports-positive-topline-results-from-phase-2-placebo-controlled-trial-of-brilacidin-for-the-prevention-of-oral-mucositis-in-head-and-neck-cancer-patients
Subgroup Analysis
“For the Modified Intent-to-Treat (mITT) population, Brilacidin-OM in the aggressive chemotherapy regimen reduced the incidence of SOM by 65.0% ([incidence control- incidence active]/incidence control) as compared with placebo (Brilacidin: 25.0%; placebo: 71.4%; p=0.0480). For the Per Protocol (PP) population, Brilacidin-OM in the aggressive chemotherapy regimen similarly reduced the incidence of SOM by 80.3% as compared with placebo (Brilacidin: 14.3%; placebo: 72.7%; p=0.0249).”
http://www.ipharminc.com/press-release/2018/4/9/innovation-pharmaceuticals-phase-2-oral-mucositis-trial-additional-data-show-brilacidin-om-demonstrated-a-significant-reduction-in-the-incidence-of-severe-oral-mucositis
B-UP (Phase 2 POC)
Before and after endoscopic images (Cohort B, 100 mg)

Topline Results
Rate of Clinical Remission (% of patients achieving)
- 60% Cohort A (3 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(i) #Endoscopy subscore ≤ 1 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 67% Cohort B (4 of 6 patients)
- 75% Cohort C (3 of 4 patients)
(ii) Rectal Bleeding subscore of 0 (% of patients achieving)
- 80% Cohort A (4 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
(iii) Stool Frequency subscore, improvement or no change from baseline (% of patients achieving)
- 100% Cohort A (5 of 5 patients)
- 100% Cohort B (6 of 6 patients)
- 100% Cohort C (4 of 4 patients)
http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
B-ABSSSI (Phase 2b)
Topline Results

"In treated patients assessed at 48-72 hours, 47/51 (92.2%), 46/48 (95.8%), 51/52 (98.1%), and 45/48 (93.8%) achieved clinical success in the Brilacidin 0.6 mg/kg single-dose group, Brilacidin 0.8 mg/kg single-dose group, Brilacidin 1.2 mg/kg 3-day group, and daptomycin 7-day group, respectively."
"On December 22nd 2014, Cellceutix also reported positive results in the Microbiological Intent-to-Treat (MITT) population. This is an important population that includes patients with baseline cultures positive for common ABSSSI pathogens, such as Staphylococcus aureus, including Methicillin-Resistant Staphylococcus aureus (MRSA). In this population, Clinical Success rates at 48-72 hours were again very high (above 90% across all treatment groups) and again very similar (with overlapping 95% confidence intervals)."
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-releases-confidence-interval-statistics-showing-clinical-success-rates-for-brilacidin-in-treatment-of-absssi
Prurisol (Phase 2a)
Topline Results (200 mg)

"Sub-population analyses further showed greater efficacy demonstrated in patients who had a baseline IGA score of 3 (“moderate”) as compared to those with a baseline score of 2 (“mild”). Some of these patients even experienced a 3-point reduction in their IGA score, going from “moderate” to “clear.” This suggests Prurisol may be more effective in treating moderate to severe psoriasis patients to a greater degree than those patients who exhibit less severe symptoms. In moderate to severe psoriasis studies, the placebo response also tends to be lower."
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
"Among patients participating in the study with the severest form of psoriasis, those having a baseline IGA score of 3 (“moderate”), the primary endpoint was met in 46.2% of patients who received Prurisol 200mg. This data was derived from analyses of all patients randomized across all 9 participating study sites."
"Additional preliminary data analyses of secondary endpoints show patients who received any dose of Prurisol, regardless of the treatment arm, had a 1-point improvement (using the IGA scoring system) at a higher rate than that of patients in the placebo arm. This is another clear indication of the drug’s efficacy."
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-provides-additional-insight-into-successful-phase-2-trial-for-treating-psoriasis
K-OC (Phase 2a)
P53 Modulation

"Modulation of the p53 protein was observed in response to administration of Kevetrin. Pathways analyses also point to concomitant cell cycle modulation at the level of gene expression. Importantly, these data are the first to directly support, in ovarian cancer patient tumors, Kevetrin’s ability to affect p53 and associated molecular pathways—a central gene signaling network involved in regulating cell growth and the cell cycle, helping to prevent cancer."
"In more detail, preliminary analyses used Western Blots to assess relative levels of key proteins extracted from tumor biopsies before and after a series of nine Kevetrin infusions administered over three weeks. The level of phospho-p53, the activated form of the protein, in addition to the noted p53 modulation, was also seen to change in response to Kevetrin administration. These findings confirm in patient tumors Kevetrin-induced anti-cancer effects similar to those demonstrated (pdf) preclinically in ovarian cancer cell-lines. These new data reinforce prior clinical data, from the earlier concluded Phase 1 study of Kevetrin in advanced solid tumors (see NCT01664000), in which observations of p21 expression in peripheral blood monocytes supported p53 involvement in Kevetrin’s mechanism of action."
http://www.ipharminc.com/press-release/2017/12/27/innovation-pharmaceuticals-obtains-direct-evidence-of-molecular-pathways-modulation-in-tumors-from-first-patients-in-kevetrin-ph2a-ovarian-cancer-trial
IPIX Milestones, Clinical Trials, Recent Articles, Corporate Overview, Patents
Potential Milestones
1. Prurisol - Topline Data (Phase 2b)
2. B-OM - Breakthrough Therapy Designation
Past Milestones

Kevetrin
- Positive primary endpoint (Phase 2a)
http://www.ipharminc.com/press-release/2018/2/8/innovation-pharmaceuticals-concludes-successful-phase-2a-trial-of-kevetrin-for-ovarian-cancer-intra-tumor-modulation-of-p53-observed
- Highly encouraging preliminary data (Phase 2a)
http://www.ipharminc.com/press-release/2017/12/27/innovation-pharmaceuticals-obtains-direct-evidence-of-molecular-pathways-modulation-in-tumors-from-first-patients-in-kevetrin-ph2a-ovarian-cancer-trial
- Positive primary endpoint and p21 data (Phase 1)
http://www.ipharminc.com/press-release/2016/11/12/assay-results-from-cellceutix-phase-1-clinical-trial-of-kevetrin-for-cancer-show-increased-p21-expression-in-675-of-evaluable-patients
- Orphan drug designation for ovarian cancer
http://www.ipharminc.com/press-release/2016/11/16/cellceutixs-kevetrin-receives-fda-orphan-drug-designation-for-the-treatment-of-ovarian-cancer
- Orphan drug designation for pancreatic cancer
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-receives-fda-orphan-drug-designation-for-kevetrin-for-the-treatment-of-pancreatic-cancer
- Orphan drug designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-fda-orphan-drug-designmation-for-kevetrin-for-the-treatment-of-retinoblastoma
- Rare pediatric disease designation for retinoblastoma
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-receives-rare-pediatric-disease-designation-from-fda-for-kevetrin-for-the-treatment-of-retinoblastoma
Brilacidin
- B-OM positive secondary endpoint (Phase 2)
http://www.ipharminc.com/press-release/2018/1/3/innovation-pharmaceuticals-brilacidin-meets-key-secondary-endpoint-in-phase-2-trial-delays-onset-of-severe-oral-mucositis-som
- B-OM positive top-line data (Phase 2)
http://www.ipharminc.com/press-release/2017/12/11/innovation-pharmaceuticals-reports-positive-topline-results-from-phase-2-placebo-controlled-trial-of-brilacidin-for-the-prevention-of-oral-mucositis-in-head-and-neck-cancer-patients
- B-UP positive top-line data (Phase 2 POC)
http://www.ipharminc.com/press-release/2017/7/13/innovation-pharmaceuticals-phase-2-poc-trial-for-inflammatory-bowel-disease-achieves-induction-of-remission-in-a-majority-of-patients-treated-with-brilacidin
- B-ABSSSI positive top-line data (Phase 2b)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-positive-top-line-data-from-phase-2b-absssi-trial-single-dose-brilacidin-comparable-to-7-days-of-daptomycin
- QIDP designation for B-ABSSSI
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-antibiotic-brilacidin-receives-qidp-designation-from-fda
- Fast track designation for B-OM
http://www.ipharminc.com/press-release/2016/11/16/fda-grants-fast-track-designation-to-cellceutixs-brilacidin-om-for-oral-mucositis
- MTA extension for prophylactic testing in implanted devices
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-completes-lab-testing-of-brilacidin-for-planned-phase-3-trial-for-acute-bacterial-skin-and-skin-structure-infections
- Formulation stable at room temperature
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-announces-breakthrough-in-the-formulation-of-novel-antibiotic-brlaicidin-plans-studies-to-treat-diabetic-foot-ulcers
- American Society for Microbiology journal publication
http://aac.asm.org/content/early/2014/06/11/AAC.02955-14#corresp-1
Prurisol
- Positive top-line data (Phase 2a)
http://www.ipharminc.com/press-release/2016/11/12/cellceutix-phase-2-trial-of-prurisol-for-mild-to-moderate-psoriasis-meets-primary-endpoint
- Positive primary endpoint (Phase 1)
http://www.ipharminc.com/press-release/2016/11/16/cellceutix-anti-psoriasis-drug-prurisol-meets-primary-endpoint-of-clinical-trial
- 505(b)(2) pathway
http://www.ipharminc.com/press-release/2016/11/17/cellceutix-informed-by-fda-that-505b2-approval-would-be-an-acceptable-approach-for-its-psoriasis-drug
Clinical Trials
1. K-OC (Phase 2a, Completed)
https://clinicaltrials.gov/ct2/show/NCT03042702?term=cellceutix&rank=1
2. P (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT02101216?term=cellceutix&rank=2
3. B-OM (Phase 2, Completed)
https://clinicaltrials.gov/ct2/show/NCT02324335?term=cellceutix&rank=3
4. P (Phase 2a, Completed)
https://clinicaltrials.gov/ct2/show/NCT02494479?term=cellceutix&rank=4
5. P (Phase 2b, Completed)
https://clinicaltrials.gov/ct2/show/NCT02949388?term=cellceutix&rank=5
6. B-ABSSSI (Phase 2b, Completed)
https://clinicaltrials.gov/ct2/show/NCT02052388?term=cellceutix&rank=6
7. K (Phase 1, Completed)
https://clinicaltrials.gov/ct2/show/NCT01664000?term=cellceutix&rank=7
8. B-UP (Phase 2 POC, Completed)
http://www.ipharminc.com/press-release/2016/11/10/cellceutix-receives-update-on-first-patient-enrollment-in-phase-2-proof-of-concept-study-of-brilacidin-for-ulcerative-proctitis
Pipeline
Recent Articles
- Taking a Page from Merck’s Gardasil and Glaxo’s Requip, These Small Companies Look to Cement Riches for Shareholders in New Category of Oral Mucositis
http://www.baystreet.ca/articles/stockstowatch/38594/Taking-a-Page-from-Mercks-Gardasil-and-Glaxos-Requip-These-Small-Companies-Look-to-Cement-Riches-for-Shareholders-in-New-Category-of-Oral-Mucositis
- A Billion Dollar Market Just Waiting for a New Oral Mucositis Drug: Five Companies That Want It
http://www.baystreet.ca/stockstowatch/2658/A-Billion-Dollar-Market-Just-Waiting-for-a-New-Oral-Mucositis-Drug-Five-Companies-That-Want-It
- Innovation Pharmaceuticals Stock May Be A Triple-Crown Winner With Brilacidin By Year End
https://seekingalpha.com/article/4088788-innovation-pharmaceuticals-stock-may-triple-crown-winner-brilacidin-year-end
- 2017 Q3 Conference Call Transcript
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
- 2017 Q1 Conference Call Transcript
http://seekingalpha.com/article/4023682-cellceutix-corporation-ctix-ceo-leo-ehrlich-q1-2017-results-earnings-call-transcript
- Interview with New Cellceutix President Dr A Bertolino
http://seekingalpha.com/article/3988240-interview-new-cellceutix-president-dr-bertolino
- Cellceutix Pipeline Continues to Hit Milestone After Milestone
https://www.streetwisereports.com/pub/na/cellceutix-pipeline-continues-to-hit-milestone-after-milestone
- A Small-Cap Biotech with Big Ideas for Acute Infections and Cancers: Cellceutix's Leo Ehrlich and Dr. Daniel Jorgensen
http://www.thelifesciencesreport.com/pub/na/a-small-cap-biotech-with-big-ideas-for-acute-infections-and-cancers-cellceutixs-leo-ehrlich-and-dr-daniel-jorgensen
Corporate Overview
- April 2018
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5acbbec2562fa79982ad5e22/1523302089564/2018+Apr+IPIX+Corporate+Overview.pdf
- 2018 Biotech Showcase
https://static1.squarespace.com/static/5715352e20c647639137f992/t/5a4e50e39140b721186d1a6f/1515081962344/2018Jan+IPIX+Corporate+Overview.pdf
- BioCentury NewsMakers Conference
https://static1.squarespace.com/static/5715352e20c647639137f992/t/59b2f34946c3c498ebfe112e/1504899917915/IPIX+Company+Presentation+BioCentury+NewsMakers+Conference++8Sep2017.pdf
- Drug Discovery and Therapy World Congress
https://static1.squarespace.com/static/5715352e20c647639137f992/t/596cac7df14aa1a6e7118ed8/1500294274057/IPI+DDTWC+Brilacidin+presentation+%28final-u%29+13Jul2017.pdf
Patents
- Kevetrin
https://www.google.com/patents/US8338454
- Brilacidin
https://www.sec.gov/Archives/edgar/data/1355250/000147793214002418/ctix_ex1037.htm
https://www.google.com/patents/US20160243117
https://www.google.com/patents/EP2709619A2
- Prurisol
https://www.google.com/patents/WO2013103601A1
MP Advisors Report
https://drive.google.com/file/d/0Bz15O4eaX-asWVpoclB2WUhENFk/view
Abbreviations
K – Kevetrin
B – Brilacidin
P – Prurisol
OC – ovarian cancer
ABSSSI – acute bacterial skin and skin structure infections
OM – oral mucositis
UP – ulcerative proctitis
QIDP – qualified infectious disease product
New investors, the statement “34% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “47% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “52% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “52% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “52% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “49% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “6% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “30% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “37% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
New investors, the statement “35% of today's volume was short sales!" wasn’t in the source file. The most recent short interest is < 1% of the OS and the most recent failure-to-deliver (naked short) # is a measly 64,587 shares. IMHO interpreting short volume as actual short sales is incorrect. This is what FINRA, the exact same source which you used to post the #, said.
"Thank you for contacting FINRA’s Office of the Ombudsman. Regarding your question, it would be incorrect to assume that short sales comprise 32% of the total volume on that trading day for IPIX. FINRA publishes the daily short sale volume pursuant to an SEC mandate; however, there are various trading anomalies that result in the reported short sale volume often being exaggerated. Thus, the true total short sales volume may be significantly lower than the volume that is reported. Third parties sometimes ignore this and try to claim the ratio is valid to further their own objectives."
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=135537542
MMs mark shares short temporarily to execute trades and they are almost always covered in the same day, therefore the actual short % is much lower than the # suggests. If the short volume indicates actual naked shorting, would the criminals be stupid enough to leave incriminating evidence in the FINRA file?
You can also check out these two posts for some detailed explanations.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=133263103
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=132584168
Here are the official short interest and failure-to-deliver data.
Settlement Date: 5/31/2018
Current Short: 1,059,202
Previous Short: 1,194,881
Change: -135,679 (-11.36%)
Average Daily Volume: 325,736
Days to Cover: 3.25
http://otce.finra.org/ESI
0.68% of the outstanding shares were short interest.
“In a “naked” short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard two-day settlement period. As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a ”failure to deliver” or “fail”).”
https://www.sec.gov/investor/pubs/regsho.htm
According to the SEC, only 64,587 shares (0.042% of the OS) were failure-to-deliver as of May 14.
20180514|45782D100|IPIX|64587|INNOVATION PHARMACEU|0.42
https://www.sec.gov/data/foiadocsfailsdatahtm
News - SEC makes rule on 'naked' short-selling permanent
https://abcnews.go.com/Business/story?id=8186474&page=1
BP's interests in the Big Three
“The Company is engaged in discussions and negotiations regarding the out-licensing of its mid-stage, first-in-class clinical assets to global and/or specialty pharmaceutical companies who have expressed an interest in one or more assets in our pipeline.”
https://www.sec.gov/Archives/edgar/data/1355250/000147793218002302/ipix_10q.htm
“The Confidential Disclosure Agreement (CDA) count toward partnering with global and specialty pharmaceutical companies interested in the Company’s first-in-class drug candidates is nearing 20, with additional Agreements in review."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
“These recent data, taken in aggregate, are what various actively engaged pharmaceutical companies have desired from us for some time—and they are what triggered a newfound flurry of inbound partnership discussions at the San Francisco conferences,”
http://www.ipharminc.com/press-release/2018/1/16/innovation-pharmaceuticals-presents-at-2018-biotech-conference-oral-mucositis-drug-candidate-garners-exceptional-interest-after-successful-phase-2-trial
"To advance our pipeline in an expeditious manner, this year we placed an emphasis on partnering—actively engaging global and regional pharmaceutical companies interested in licensing our novel drug candidates. These efforts have led to highly productive, mature (late-phase, ongoing) discussions, with multiple pharmaceutical companies, which we hope will bear fruit in the coming year." - CEO Letter to Shareholders
"We had said that confidential disclosure agreements have been signed with several pharmas interested in our drug candidates; to some, they're interested in one compound. Others have requested information on a combination of Brilacidin and Kevetrin and Prurisol."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
B-Dermatitis
"To further these efforts, the Company is in negotiations with a leading drug formulator to develop topical formulation(s) of Brilacidin for these three dermatology indications, starting in 1H2018. The goal of the negotiations is to reach terms on a strategic partnership for addressing these markets."
http://www.ipharminc.com/press-release/2018/1/29/innovation-pharmaceuticals-brilacidin-franchise-anchored-in-three-clinical-indications-oral-mucositis-inflammatory-bowel-disease-and-serious-skin-infections-expands-into-dermatologic-diseases
B-OM
“Given the notable interest from global and specialty pharmaceutical companies that we have received following recent Brilacidin-OM data releases, we continue to carefully assess all of our potential alliance opportunities—toward cementing the best pathway forward,” said Leo Ehrlich, Chief Executive Officer of Innovation Pharmaceuticals.”
http://www.ipharminc.com/press-release/2018/5/9/innovation-pharmaceuticals-concludes-data-analysis-of-its-phase-2-clinical-trial-for-severe-oral-mucositis-in-head-and-neck-cancer-positioning-to-fill-a-substantial-void-in-supportive-cancer-care
“Formal collaboration with pharmaceutical companies that have expressed an interest in partnering Brilacidin-OM may well assist further with expediting the drug candidate’s development timetable. Some of these partnering conversations have matured to the point of potentially structuring mutually beneficial licensing agreements, pending the final Phase 2 study results.”
http://www.ipharminc.com/press-release/2017/11/16/innovation-pharmaceuticals-offers-perspectives-on-brilacidin-as-a-potential-preventative-treatment-for-oral-mucositis-in-head-and-neck-cancer-patients
B-UP
"We've actively engaged with multiple large pharmas, who have expressed an interest in Brilacidin inflammatory bowel disease. The pharma industry is quite interested in pursuing novel IBD therapies one of the more active therapeutic areas when it comes to dealmaking."
https://seekingalpha.com/article/4080068-innovation-pharmaceuticals-ipix-ceo-leo-ehrlich-q3-2017-results-earnings-call-transcript
Prurisol
"We have entered into multiple non-disclosure agreements with large pharmaceutical companies that enable us to continue ongoing discussions regarding potential partnering should the trial results support such a relationship."
https://www.sec.gov/Archives/edgar/data/1355250/000147793217004400/ipix_10k.htm
Kevetrin
"The Company is adhering to a value-building strategy born from discussions with larger pharmaceutical companies interested in Kevetrin, one of the world’s most advanced p53 drug candidates. Securing the right development partner for Kevetrin remains an important objective."
http://www.ipharminc.com/press-release/2018/2/8/innovation-pharmaceuticals-concludes-successful-phase-2a-trial-of-kevetrin-for-ovarian-cancer-intra-tumor-modulation-of-p53-observed
"What we and potential partners are extremely interested in is learning via tumor biopsies if Kevetrin is reaching its target and modulating pathways within the tumor that can deliver a clinically meaningful benefit; this would be a significant development in the p53/oncology dynamic,"
http://www.ipharminc.com/press-release/2017/9/14/innovation-pharmaceuticals-opens-new-clinical-site-for-novel-p53-drug-candidate-in-phase-2-ovarian-cancer-trial-1
Document - Getting Ready for a BioPharma Partnering Deal
A typical large pharmaceutical might see 500 to 1,000 opportunities in a given therapeutic area. Only 100 or so of those will go on to non-confidential meetings and only 50 or so of those will end up going to Confidential Disclosure Agreements (CDAs) and confidential meetings.
Unfortunately, getting a deal done can take a long time. After initial talks, getting a CDA can take a month or more. It might take another month for the other side to assemble appropriate team members and schedule team meetings.
It can then easily take another month for them to evaluate all the information and to decide that they will indeed go forward with the formal due diligence process. From the point when the other party has decided to go forward with term sheet negotiations, it can take another six to twelve months to get to a signature on a full agreement.
Diligence and a Good Virtual Data Room
When a potential partner begins the due diligence process, they’re trying to get to know you and your asset in greater detail, and they’re attempting to verify what they’ve been told.
Give them access to your experts, provide overview presentations, and answer questions. In short, spend the time it takes to make sure they really understand it.