Making $$ in the Market
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Immune Pharmaceuticals, Inc. (NASDAQ:IMNP) current consensus investment analysts is to Buy
http://theindependentrepublic.com/2017/01/12/immune-pharmaceuticals-inc-nasdaqimnp-current-consensus-investment-analysts-is-to-buy-3/
Investor 100
Immune Pharmaceuticals Inc (NASDAQ:IMNP): Here’s Our Take
https://www.insiderfinancial.com/immune-pharmaceuticals-inc-nasdaqimnp-heres-our-take/118988/
Investor 100
RECAP IMNP @ YouTube
December 8th, 2016
Compelling conversation update with CEO Tepper & Dr. Monica Luchi, Chielf Medical Officer.
Comments on financing starts at 10:57 on the tape.
Boston Biotech Conference-January 8th,2017.
East /West CEO
R&D Trends: What's Changed in the Pharma Pipeline?
Tom Heyman, President, Johnson & Johnson Innovation, JJDC; Vivek Ramaswamy, CEO, Roivant Sciences and Axovant Sciences; Daniel Teper, CEO, Immune Pharmaceuticals
Moderator: Joel Marcus, CEO, Alexandria Ventures
https://bbbiotechconference.com/conference-agenda.php?id=54
Investor 100
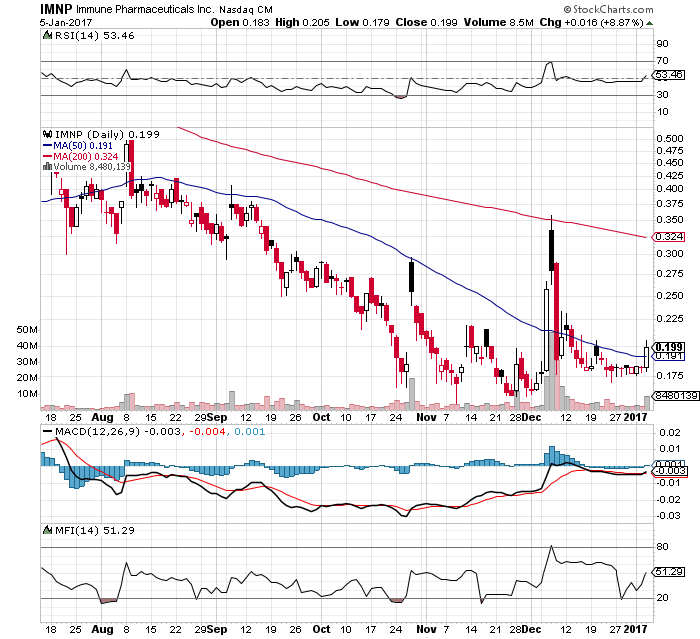
IMNP @ Sto0ckcharts
Happy New Year to IMNP traders / investors!
IMNP off to a good start with an inside purchase of 76k shares and todays activity looking good with RSI and MFI moving upward...follow the money...closing at .199 with over 8M shares traded or up 8.87%.
D. Tepper also posted this news earlier this week as well providing us some guidance on IMNP....good stuff.
http://www.immunepharma.com/strategic-focus-and-partnerships/
Looking forward to some positive developments at IMNP this year!
Investor 100
Happy Holidays @ IPCI
Pleased to be a shareholder having added shares throughout the year which from all accounts looks like a great 2017 /2018 coming up with major announcements. Holding shares well below $2.
Cheers,
Investor 100
Insider Trade Purchase @ IMNP
A vote of confidence from a director!
2016-12-20 20:44:44
2016-12-16
IMNP Kazado Daniel Dir P - Purchase $0.19 +161,377 1,068,667 +18% +$30,000
Investor 100
Pleased to be onboard FDA approval ship!
Investor 100
Depends on the company..reduces share count and creates opportunities for institutional buyers to come into play.
Investor 100
Management Invest's @ IMNP
As the saying goes "put your money where your mouth is" was proven back in August with a substancial personal investment into IMNP stock.
August 5th, 2016
http://finance.yahoo.com/news/immune-pharmaceuticals-inc-imnp-ceo-061503523.html;_ylt=AwrBT8FWV0tYDXwA2E1XNyoA;_ylu=X3oDMTEyaGJwcmJrBGNvbG8DYmYxBHBvcwM0BHZ0aWQDQjI5MTNfMQRzZWMDc3I-
Immune Pharmaceuticals Inc (IMNP) CEO Daniel Gedeon Teper Bought $356,250 of Stocks
CEO of Immune Pharmaceuticals Inc (IMNP) Daniel Gedeon Teper bought 937,500 shares of IMNP on 08/04/2016 at an average price of $0.38 a share. The total cost of this purchase was $356,250.
Immune Pharmaceuticals Inc is a clinical stage biopharmaceutical company specializing in the development and commercialization of targeted therapeutics for the treatment of inflammatory diseases and cancer. Immune Pharmaceuticals Inc has a market cap of $13.513 million; its shares were traded at around $0.33 .
CEO Recent Trades:
CEO, 10% Owner Daniel Gedeon Teper bought 937,500 shares of IMNP stock on 08/04/2016 at the average price of $0.38.
Directors and Officers Recent Trades:
EVP & CMO Monica E. Luchi bought 777,778 shares of IMNP stock on 08/04/2016 at the average price of $0.45.
Director Daniel Kazado bought 666,667 shares of IMNP stock on 08/04/2016 at the average price of $0.45.
For a complete lists of insider holdings:
http://openinsider.com/search?q=imnp
Management Team @ IMNP
Curious as to who the management team running this company!
http://www.immunepharma.com/who-we-are/management/
DANIEL TEPER, CHIEF EXECUTIVE OFFICER
Dr. DANIEL TEPER, CHIEF EXECUTIVE OFFICER
Dr. Daniel Teper is the founder and CEO of IMMUNE Pharmaceuticals. He was the Managing Director for North America at Bionest Partners, a global Strategy Consulting firm advising pharmaceutical and public biotechnology companies and previously a New York based Partner at ISO Healthcare Consulting (now part of Deloitte Monitor). Daniel started his career at Novartis Global Headquarters in Basel and then in the United States where he had growing responsibilities in sales, marketing and new product development. He held senior executive positions in Europe, first at GSK as Head of Commercial Operations for Glaxo France and then as President and President of Laboratoires Delagrange through the acquisition by Synthelabo (now part of Sanofi). He then became Global President HAVAS Health, a leading marketing and communication group, where he helped launch multiple industry blockbusters. Daniel was a cofounder of Novagali (NYSE Euronext: NOVA), which was acquired by Japan’s Santen. Daniel holds a Doctor of Pharmacy degree (PharmD) from Paris XI University and an MBA from INSEAD.
DR. MONICA E. LUCHI, EXECUTIVE VICE PRESIDENT GLOBAL DRUG DEVELOPMENT, CHIEF MEDICAL OFFICER
Dr. Monica E. Luchi is currently the Chief Medical officer for New York based biotech company Immune Pharmaceuticals, and Executive Vice President of Global Drug Development. She is a Board certified rheumatologist with 15 years industry experience at a mix of both large pharmaceutical and small biotech companies. Prior to joining Immune, Dr. Luchi held senior immunology and inflammation positions at Novartis, Incyte and most recently, Mesoblast, where she has led cross-functional teams and drove strategy across a variety of therapeutic areas and phases of development. In addition, Dr. Luchi has helped to drive the business interests of the biotech companies where she worked in order to engage development partners and to strengthen financial assets to support further progress of the companies’ development activities. She is adjunct Clinical Assistant Professor on faculty at the University of Pennsylvania where she did her fellowship, and continues to see patients there one half day per week. She has recently served on a local chapter board for the Healthcare Businesswoman’s Association. Dr. Luchi obtained a BA in Biology and in Health Science Policy at the University of Maryland, Baltimore Co. Campus, received her MD from Northeastern Ohio Universities, College of Medicine, and an MBA with a concentration in healthcare from George Washington University.
DR. MIRI BEN-AMI, PRESIDENT, IMMUNE ONCOLOGY PHARMACEUTICALS INC AND IMMUNE PHARMACEUTICALS LTD.
Miri Ben-Ami’s background includes more than 15 years in executing broad management and drug development responsibilities for the healthcare industry. Most recently, she served as the CEO for Aposense Ltd., a clinical stage public biotech company developing therapeutics and molecular imaging diagnostics in the field of oncology. Under her leadership, the company refocused its drug pipeline, entered into partnerships with big pharma, and received approval from the FDA for accelerated development of its lead compound. Before that, Miri held senior leadership roles during her 9-year tenure at Teva Pharmaceuticals. As Global Head of Neurology, Miri spearheaded development strategies for Copaxone, a lead immunomodulator in multiple sclerosis, underlying the successful expansion of the product’s indications and market. Currently, Miri serves on the governing board of the Israeli Public Companies Association, as Chairperson of the organization’s life-sciences division. Miri obtained her B.Sc.(Hon.) degree in Medical Biochemistry and her M.D. degree from the University of the Witwatersrand, Johannesburg, and gained clinical experience during her residency in Obstetrics and Gynecology in the UK and Israel.
Screen Shot 2016-02-08 at 3.08.12 PM
JOHN MILITELLO, VP FINANCE, CONTROLLER, CHIEF ACCOUNTING OFFICER AND PRINCIPAL FINANCIAL OFFICER
John joined the Company’s in April 2015 as our Executive Director of Finance and Corporate Controller. Prior to that, Mr. Militello was an Assistant Controller with Retrophin, Inc., a San Diego based biotech company, and the Manager, External Reporting & Compliance at Volt Information Sciences, Inc., a publicly traded staffing company. Prior to Volt Information Sciences, Inc., Mr. Militello was a Senior Manager with BDO USA, LLP serving multi-national SEC registrants. Mr. Militello is a Certified Public Accountant and earned his Bachelor of Science degree in Accounting from St. Joseph’s College.
DR. JEAN KADOUCHE, SVP, SCIENTIFIC DEVELOPMENT
Co-founder of Immune Pharmaceuticals and a scientific advisor focusing on the development and application of novel antibody technologies, including fully human antibody generation, bi-specific antibodies and antibody drug conjugates. With 25 years of experience in the development of mAbs in both academia and industry, Dr. Kadouche was the developer of the bispecific antibody platform and co-developer (with Prof. Simon Benita) of the NanoMAbs technology – both licensed by Immune Pharmaceuticals from Atlante Biotech and the Hebrew University of Jerusalem respectively. Dr. Kadouche is the former CEO of MAT BioPharma, an antibody company where Kadouche developed and partnered several clinical and pre-clinical antibodies. He is also the founder of Clonatec and a co-founder of Novagali (NYSE Euronext: NOVA) and has been an advisor to SangStat, Roche, Merck AG and J&J. Dr. Kadouche holds a PhD in immunology from the Pasteur Institute and was an Assistant Professor at St Louis Hospital. He has about 100 publications and registered over 15 patents which have led to company creations.
DR. MARK LEVITT, SVP, CLINICAL AFFAIRS, ONCOLOGY
Mark L. Levitt, MD, PhD is a Board Certified Medical Oncologist and has a strong academic background in both basic and clinical oncology research as well as extensive experience in small and big pharma. His area of focus in the pharmaceutical industry has been in clinical development, business development and clinical and scientific evaluation and strategy.
Following his residency, he did his fellowship in Medical Oncology at the National Cancer Institute (NIH) and subsequently became Director of the University of Pittsburgh Cancer Institute Pulmonary Oncology Program. During this time he was co-PI on one of the first SPORE grants in lung cancer and a leader in ECOG, serving on several steering committees as well as the Laboratory Science Advisory Committee. In 2006, Mark joined Teva Pharmaceutical Industries Ltd. as the Global Clinical Oncology Platform Leader in Innovative R&D following which he became the Global Head of the Oncology Therapeutic Area and helped lead Teva to the establishment of an Oncology Franchise with the acquisition in 2011 of Cephalon.
CEO Tepper @ IMNP
One particular area of interest I have with IMNP is this notion of "partnership" and returning to this weeks interview he speaks at length about this as well in a previous interviews thus I find his message consistent with the belief we will hear more about those partnership opporunities throughout 2017 as well as trial updates!
December 8th, 2016
http://www.immunepharma.com/investor-information/investor-events/immuno-oncology-rd-update-december-2016/
CEO Speaks on behalf of Commercial Sales/ Partnerships Opportunities/( minute 10:50-13:34 )
December 4, 2015
Strong Rebound @ IMNP
PPS at .22 / 14M shares traded.
Yesterday's knee jerk reaction created a buy opportunity that I took advantage of given this time next year this company will have progressed. High Risk with High Reward.
Pipeline @ IMNP
http://www.immunepharma.com/what-we-do/products-3/
Investor 100
News Coverage @ IMNP
News article coming in from Stockholm ( Direkt):
https://www.avanza.se/placera/telegram/2016/12/08/immune-pharma-knoppar-av-ceplene-nytt-forsok-i-usa-vd.html
Translation into English
STOCKHOLM (AFX) Immune Pharmaceuticals buds of cancer drug Ceplene and form a new company that will make a new attempt to get the drug on the US market. It says immunes CEO Daniel Teper to the news agency Direkt before the company's webcast R & D update at 17:00 CET on Thursday, Swedish time.
The company's shares rose yesterday by more than 70 percent without any news had been presented. Rate increase is according to CEO on selling pressure in immunes shares declined and investors look to the dormant project Ceplene alive again.
"We have been going around and met with investors in recent months, and people are now looking more to the fundamentals of the company than before. Research and development projects are developing rapidly and now bertilimumab our only focus," says Daniel Teper.
Ceplene is an immune stimulant drugs developed against acute myeloid leukemia and is approved in Europe. 2012 out-licensed the drug to the Swedish Meda, which this year was bought by Mylan. Attempts to launch the drug in the US market were up after the FDA demanded additional Phase 3 study, but now the tests will be taken up again.
During the evening will update Daniel Teper talk more about the spin-off of Ceplene in the new company which Immune Pharmaceutical become the largest shareholder. New money should be brought in for the continued development, how much is yet to be decided.
What makes that Ceplene will have better chances of reaching the United States now?
"We published a new study with Ceplene early in 2016 that showed positive results in progression-free survival. The development includes testing of biomarker and the effect can be seen after six weeks, which is advantageous in a health economic perspective," said Daniel Teper and continues:
"In addition, we will in 2017 launch a phase 3 clinical study in which we have collaborated with the FDA on the study design," he says.
Ceplene reaches the US market, sales may exceed $ 200 million, according to Daniel Teper.
Regarding bertilimumab, which is the only substance that remains in immunes portfolio in the future, two ongoing studies in various indications in which results are expected in 2017. The company took in November, up $ 11 million from existing investors and will in 2017 examine more indications of bertilimumab.
Parallel to the development of the company, talks with "several potential partners," according to Daniel Teper. Probably covering a partnership all the indications bertilimumab developed within instead of signing contracts for the various disease area, he says.
Speculation in the market raises the question whether Pfizer is getting ready to devour the research company, but Daniel Teper would not comment on market rumors. However, he certifies that the company has been in dialogue with several major pharmaceutical companies, including Pfizer, but it will not affect the information in tonight's R & D update.
Anna Sundström +46 8 5191 7911
News Agency Direkt
Analysts expectation about IMMUNE PHARMACEUTICALS INC. (IMNP) to Strike High potential price target at $3.00
Looking forward to 2017!
http://www.hotstockspoint.com/analysts-expectation-immune-pharmaceuticals-inc-imnp-strike-high-potential-price-target-3-00/
MNP Stock Price Comparison to Moving Averages:
USA based company, Immune Pharmaceuticals, Inc.’s (IMNP)’s latest closing price distance was at -20.29% from the average-price of 200 days while it maintained a distance from the 50 Day Moving Average at 30.58% and 52.18% compared with the 20 Day Moving Average.
Analysts Mean Rating:
Analysts’ mean recommendation for Immune Pharmaceuticals, Inc.’s (IMNP) stands at 1.50. Rating Scale; where 1.0 rating means Strong Buy, 2.0 rating signify Buy, 3.0 recommendation reveals Hold, 4.0 rating score shows Sell and 5.0 displays Strong Sell signal.
Performance Review:
To review the IMNP previous performance, look at its past history, which highlighted below:
During last 5 trades the stock sticks almost 67.87%.
During last one month it showed the change of 55.30%.
During last 3 month it remained at -22.22%.
Along with these its year to date performance is standing at -61.64%.
Important Technical Indicators:
ATR value of company was 0.03. Relative Strength Index (RSI) was 69.69. Developed J. Welles Wilder, the Relative Strength Index (RSI) is a momentum oscillator that measures the speed and change of price movements. RSI oscillates between zero and 100. Traditionally, and according to Wilder, RSI is considered overbought when above 70 and oversold when below 30. Signals can also be generated by looking for divergences, failure swings and centerline crossovers. RSI can also be used to identify the general trend.
The stock volatility for week was 21.95% while for month was 13.85%.The stock, as of last close, traded 80.53% to its 52 week low and was changed -67.82% from its 52 week high. Beta factor, which measures the riskiness of the security, was observed as 1.57. IMNP’s value Change from Open was at -16.17% with a Gap of 29.61%.
Earnings per share Details about IMNP:
EPS growth in past 5 year was 41.30% .
EPS growth in next year is estimated to reach 198.10% while EPS growth estimate for this year is set at 38.20%.
The forward P/E ratio stands at 0.55. The price to sales ratio is 17.
Immune Pharmaceuticals, Inc.’s (IMNP)’s Stock Price Trading Update:
Immune Pharmaceuticals, Inc.’s (IMNP), a part of Healthcare sector and belongs to Drug Manufacturers – Major industry; ended its day with a gain of 8.65% and finalized at the price of $0.28. During its last trading session, Stock traded with the total exchanged volume of 48.6 million shares. The average volume stands around 4.01 million shares. The average numbers of shares are traded in a security per day, during the recent 3-month period. The stock has relative volume of 12.13. Relative volume is ratio between current volume and 3-month average value, intraday adjusted.
Analysts are expecting that Immune Pharmaceuticals Inc. (IMNP ) to achieve $3.00 Price Target in next 52-weeks, average price is come up through the consensus of analysts. High potential price target is set at $3.00 however minimum price target advised by analysts is $3.00. The Median price target for the stock is measured at $3.00.
Immuno Oncology R&D Update, December 2016
For those that missed the interview today.
http://www.immunepharma.com/investor-information/investor-events/immuno-oncology-rd-update-december-2016/
http://www.immunepharma.com/focus-on-acute-myeloid-leukemia-at-the-american-society-of-hematology/
Focus on Acute Myeloid Leukemia at the American Society of Hematology
The Immune Pharma team traveled this weekend to the annual meeting of the American Society of Hematology (ASH) in San Diego. This was an opportunity to learn about the latest developments in Acute Myeloid Leukemia(AML) and to hold a Scientific Advisory Board for our AML drug, Ceplene®. Acute Myeloid Leukemia (AML) is the most frequent form of leukemia in adults. An estimated 19,950 patients were diagnosed in 2016 with AML in the US, with a 26.6% 5-year survival rate (2006-2012) and an estimated 10,430 deaths in 2016. This is a stern reminder of the urgent need for new therapies.
The bi-partisan Congress 21st Century Cures, provides additional funding to the FDA to accelerate approval of needed therapies, to help fund the Cancer Moonshot initiatives, and to enable acceleration of cancer research programs. New data is promising and comes from a mix of novel breakthrough approaches, such as targeting specific cytogenetic characteristics. Molecular genetic analysis of the Flt3 gene, CEBPA, and NPM1 among others is becoming the standard of care for the diagnosis, prognostic understanding, and treatment guidance in AML. Targeted therapies, such as Agios IDH1 and IDH2 inhibitors also reported favorable early clinical data.
Vyxeos (CPX-351) is a new 5+1 liposomal formulation of the 7+3 cytarabine-daunorubicin standard of care in induction therapy. After a disappointing phase 2 study, Celator (now part of Jazz Pharmaceuticals) conducted a phase 3 study that has demonstrated impressive overall survival benefit and should lead to approval in 2017. Mylotarg, which targets CD33, was the first Antibody Drug Conjugate and was developed for AML. However, toxicity led to its withdrawal from the market. Seattle Genetics is now targeting CD33 but with a novel toxin and linker system, vadastuximab talirine, where phase 1b data appears to support further development.
While new induction or consolidation approaches show promise, a gap remains in remission maintenance of AML patients, which is also being explored with vadastuximab and with Celegene’s oral azacitidine. Ceplene in combination with low dose Interleukin 2 (IL2) is approved in Europe to maintain remission in AML patients. Ceplene enables IL2 growth factor-like activity on T-Cells and NK cells, helping prevent relapse of leukemia blasts post first remission (CR1). The over 1500 patient Ceplene clinical trial data includes a positive pivotal phase 3 Leukemia Free Survival study published in Blood, ASH’s weekly medical journal, and a phase 4 study presented at the American Academy of Cancer Research earlier this year. Now Immune Pharmaceuticals is planning a global pivotal Overall Survival (OS) study which will compare Ceplene+ IL2 to IL2 in AML patients post CR1. The primary end point will be OS at 2 years while intermediary end points will be assessed at 1 year. The Ceplene SAB included pioneers of Ceplene+IL2 therapy like Kristoffer Hellstrand, MD (Gotenborg, Sweden) and Jacob Rowe (Jerusalem, Israel) as well as several of the leading US key opinion leaders, Martin Tallman ( MSK, New York), Elihu Estey ( Fred Hutchinson, Seattle), Gail Roboz ( Weill Cornell, New York), Janice Dutcher (Cancer Research Foundation, New York), Selina Luger ( University of Pennsylvania, Philadelphia) and Jessica Altman ( Northwestern University, Chicago). We are grateful for their guidance and their passion to bring new therapies to AML patients.
Immune Pharmaceuticals is committed to fully funding the further development of Ceplene through a recently announced new immuno-oncology company, Cytovia Oncology.
Investor 100
Short and Sweet @ IMNP
The news was a recapp on their meeting earlier this month at ASH and from all indications studies continue despite positive results as well as NO DILUTION for shareholders in order for additional financing. For those looking for FDA approval or a buy-out that was not on the table this morning. Stock traded down at .18 from .28 thus a loss for short term traders and an opportunity to buy the dip for those willing to buy and hold for 2017!
Investor 100
Dr. Daniel Teper and Dr. Monica Luchi Interviewed by John McCamant, December 2016
Ceplene Scientific Presentation with Mark Levitt
Someone’s Loading Up On Immune Pharmaceuticals Inc (NASDAQ:IMNP) Ahead Of A Major Update
https://www.insiderfinancial.com/someones-loading-up-on-immune-pharmaceuticals-inc-nasdaqimnp-ahead-of-a-major-update/118575/
mmune Pharmaceuticals Inc (NASDAQ:IMNP) is a big runner this week, up more than 55% on its Monday open. The company announced back on November 22 that it will host an R&D update event on December 8, and it looks as though someone is loading up big ahead of the event in anticipation of wider markets taking note, and the buy volume flooding in.
So, ahead of management taking the stage, what is on the table for discussion, and what might drive speculative volume to boost the company’s market capitalization near term?
Let’s take a look.
The focus of the event is a drug called Ceplene, which is one of a couple of leads assets in the company’s pipeline, with the other being bertilimumab. The latter is an ulcerative colitis indication, and investors will be hoping that management serves up some degree of insight into the progress that that drug is making through its so-far promising program. However, if the led in info is to be relied upon, chances are we won’t see any major bertilimumab news. That doesn’t mean it’s not going to be interesting, however. Ceplene in itself is a promising asset, and could be a big revenue generator for Immune if it can carry it through to commercialization.
What’s it all about?
The Ceplene moniker is a brand name for the chemical histamine dihydrochloride, and it’s designed to improve the efficacy of IL-2 in patients with Acute Myeloid Leukemia (AML). IL-2 is a cytokine that derives from T cells (which are basically the attack cells in the human immune system). It initiates the T cell response against cancerous cells, which would otherwise be hidden from view of the immune system as a result of the cloaking mechanism these cancerous cells employ to avoid detection and – by proxy – removal. In this specific indication, IL-2 isn’t overly effective (at least, not as effective as it is in some other cancers) because it gets inhibited by what are called myeloid-cell derived reactive oxygen species (ROS). Ceplene essentially blocks these ROSs from inhibiting the IL-2, and in turn, enhances the latter’s impact on T cell activation towards the cancer cells in question.
It’s a pretty neat mechanism of action (MOA) in that the rationale behind including Ceplene as part of a combo therapy with IL-2 in AML patients is simple, and clearly beneficial.
The EMA in Europe has already cleared the drug, and its at-market right now. The US market is bigger, however, and an FDA nod is what the company is seeking with its Ceplene development program. And that’s what this update is going to address – the next stage of the program.
Here’s what we think is going to happen.
The Ceplene Scientific Advisory Board (SAB) met on Sunday to discuss the next steps ahead of the update on the 8th. At this meeting, we think the board will have put together a protocol for a pivotal phase III trial of Ceplene in combination with IL-2 in AML, and that company management are going to both confirm the agreement of, and relay the details of, this program at the update. The SAB meeting comes following FDA guidance, and with the clear loading up that’s taking place ahead of management reporting on the guidance and the subsequent protocol decision, we’re guessing it’s pretty favorable. We’re almost definitely going to see an overall survival endpoint (it’s a remission maintenance study, so no other endpoint would make that much sense) and we’re looking to a relatively condensed patient population as being key to getting this drug in front of the FDA by way of an NDA as soon as possible. We’ll be watching the event, so stick with us and we’ll bring our interpretation to the table as soon as possible.
Disclosure: We have no position in IMNP and have not been compensated for this article.
PM @ IMNP
Pre-market price at .30 with over 200K shares traded...tic..tock til 11am on Facebook!
GLTA
Investor 100
Immune Pharmaceuticals Announces Immuno-Oncology R&D Update on December 8, 2016
Discussion on the topics listed below is what I anticipate on the Facebook platform discussion!
http://www.prnewswire.com/news-releases/immune-pharmaceuticals-announces-immuno-oncology-rd-update-on-december-8-2016-300367275.html
Confirms plans for pivotal phase 3 Overall Survival study with Ceplene in AML
NEW YORK, Nov. 22, 2016 /PRNewswire/ -- Immune Pharmaceuticals (NASDAQ: IMNP) (Immune) announced today that it will hold an Immuno-Oncology R&D Update for investors on December 8, 2016 in New York City. Immune management and several Oncology Key Opinion leaders will review development plans and milestones for Ceplene® and other pipeline assets to be funded separately in the recently announced Immune subsidiary, Cytovia Oncology.
Prior to the Immuno-Oncology R&D Update, the Ceplene Scientific Advisory Board (SAB) will meet on December 4, 2016 in San Diego where the American Society of Hematology is holding its annual meeting. Following the guidance from the Food and Drug Administration, the SAB will review the proposed clinical protocol for a pivotal phase 3 Overall Survival clinical study of Ceplene in combination with low dose IL2 in the remission maintenance of patients with Acute Myeloid Leukemia (AML).
Investor 100
Financing Immune Pharmaceuticals
http://www.immunepharma.com/financing-immune-pharmaceuticals/
Like many micro-cap companies, Immune Pharmaceuticals (NASDAQ:IMNP) has had to overcome the challenges of structured financings. Stock price and liquidity are key drivers for an early stage biotech company to raise sufficient capital to advance its pipeline. The good news is that IMNP finally has a clean cap table with no outstanding preferred shares, low percentage of warrants, and relatively low debt level. Additionally, IMNP has excellent daily liquidity. The reverse split is an option that IMNP will consider based on market conditions as well as company performance and prospects. IMNP will also consider the potential impact of any reverse split on the liquidity of the stock.
So how does IMNP finance its pipeline going forward? The recently announced At The Market financing from a single investor limits any short term selling pressure and minimizes dilution, while securing access to proper funding. There are no discounts, warrants, or resets.
Lastly, we have announced the formation of subsidiaries and potential spinoffs for each of our product lines which will unlock the value of IMNP’s assets by allowing for dedicated financing while focusing IMNP’s resources on our lead product candidate, bertilimumab. IMNP shareholders may benefit through the company’s direct shareholding and possibly through share dividends that will allow them to hold shares in both IMNP and the new companies focused respectively on Oncology, Pain & Neurology, and Dermatology. Due to their confidence in the company, insider buying from management and board has exceeded $1.5 Million in IMNP shares over the past 15 months. This includes significant investment from the Chief Executive Officer and Chief Medical Officer.
Investor 100
Last Hour @ IMNP
Less than an hour of trading with 46M+ shares trading hands, .2949 / +14.3% / high .3574...IMNP will publish an update tomorrow using its Facebook platform...I gather with yesterdays 20M shares traded and today's many shareholders/ traders expect some strong news..
GLTA
Investor 100
Facebook @ IMNP
Tomorrows news will be announced on their Facebook page!
Investor 100
This is a high risk /high reward with pending news so its anybodies guess but from all indications 40M+ shares has traded thus far...
Investor 100
MOMO @ IMNP
Out of the gates with 14M shares traded/ +14% / high .3571 without any news that is pending from IMNP!
GLTA
Investor 100
PM @ IMNP
Looking for a strong day with the stock trading at .32 this PM!
GLTA
Investor 100
MOMO AH @ IMNP
Closed at .31 / 1.7M shares traded / +18.35%...well over 20M shares traded today....I think Santa is going to deliver some great news!
GLTA
Investor 100
