Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
leifsmith, there are government funded vaccines in clinical trials! Wishful thinking?
Thomas Duncan, the first person in the United States who was diagnosed with Ebola, was reported to have died yesterday after succumbing to the virus.
Thomas A. Duncan, who became ill with Ebola after arriving from West Africa in Dallas two weeks ago, succumbed to the virus today (Sunday), reports Reuters. Duncan was fighting for his life at a Dallas hospital on today after his condition worsened to critical, according to the director of the US Centers for Disease Control.
This is according to an Israeli news report. The information has not yet been released or confirmed in the United States at the time of publication.
This comes after a veritable comedy of errors. First, Duncan came into the country with the illness. Then, he was turned away from the hospital, even though he told them he had come from Liberia. After that, he went home and exposed numerous people, including children who subsequently attended school. When he returned to the hospital, he vomited on the sidewalk. The vomit was power washed away by workers using no protective gear. He was put in an ambulance and the paramedics were not told he’d been diagnosed with Ebola, as they continued to drive around in the contaminated vehicle for the next two days, hauling patients. The CDC keeps surreptitiously changing information on their website about transmission of the disease, and they refuse to close the border to travelers who’ve been in the area where Ebola has reached epidemic levels.
Given all of these mistakes, it’s no surprise that they may be reluctant to announce the patient has died.
Scientists have claimed the Ebola virus could reach the UK and France by the end of the month.
Following an analysis of disease spread patterns and airline traffic data, experts have predicted there is a 75 per cent chance the virus could be imported to France by October 24, while there is a 50 per cent chance it could have also hit Britain.
The deadly epidemic has killed more than 3,400 people since it began in West Africa in March and has now started to spread faster, infecting almost 7,200 people so far.
The estimates have been based on air traffic remaining at full capacity. An 80 per cent reduction in travel however would see France's risk remain at 25 per cent, while Britain's risk would still be at 15 per cent.
...
But Alex Vespignani, a professor at the Laboratory for the Modeling of Biological and Socio-Technical Systems at Northeastern University in Boston who led the research, said the risks change every day the epidemic continues.
He told Reuters: "This is not a deterministic list, it's about probabilities - but those probabilities are growing for everyone.
"It's just a matter of who gets lucky and who gets unlucky."
The latest calculations used data from October 1.
"Air traffic is the driver," Mr Vespignani said. "But there are also differences in connections with the affected countries (Guinea, Liberia and Sierra Leone), as well as different numbers of cases in these three countries - so depending on that, the probability numbers change."
It is common knowledge...
PEGylation, by increasing the molecular weight of a molecule, can impart several significant pharmacological advantages over the unmodified form, such as:
-Improved drug solubility
-Reduced dosage frequency, without diminished efficacy with potentially reduced toxicity
-Extended circulating life
-Increased drug stability
-Enhanced protection from proteolytic degradation
PEGylated drugs also have the following commercial advantages:
-Opportunities for new delivery formats and dosing regimens
-Extended patent life of previously approved drugs
PEG is a particularly attractive polymer for conjugation. The specific characteristics of PEG moieties relevant to pharmaceutical applications are:
-Water solubility
-High mobility in solution
-Lack of toxicity and low immunogenicity
-Ready clearance from the body
-Altered distribution in the body
Dr. Diwan also discussed the extremely high safety of NV-INF-1 observed in preliminary safety/toxicology studies. He noted that no significant changes in all observed parameters were found even at the maximum feasible dose of approximately 2,700 mg/kg/d repeatedly given for five consecutive days.
Ebola virus (EBOV) remains one of the most lethal transmissible infections and is responsible for high fatality rates and substantial morbidity during sporadic outbreaks. With increasing human incursions into endemic regions and the reported possibility of airborne transmission, EBOV is a high-priority public health threat for which no preventive or therapeutic options are currently available. Recent studies have demonstrated that cocktails of monoclonal antibodies are effective at preventing morbidity and mortality in nonhuman primates (NHPs) when administered as a post-exposure prophylactic within 1 or 2 days of challenge. To test whether one of these cocktails (MB-003) demonstrates efficacy as a therapeutic (after the onset of symptoms), we challenged NHPs with EBOV and initiated treatment upon confirmation of infection according to a diagnostic protocol for U.S. Food and Drug Administration Emergency Use Authorization and observation of a documented fever. Of the treated animals, 43% survived challenge, whereas both the controls and all historical controls with the same challenge stock succumbed to infection. These results represent successful therapy of EBOV infection in NHPs.
..."It's the single greatest concern I've ever had in my 40-year public health career," said Dr. Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota. "I can't imagine anything in my career - and this includes HIV - that would be more devastating to the world than a respiratory transmissible Ebola virus."
As the virus spreads and mutates, Osterhold and other experts warn, the likelihood increases.
Osterholm calls it "genetic roulette."
One thing is for certain: We have to be prepared if the problem escalates. And if it does, there's a very high chance U.S. technology will ultimately save the day.
...
The U.S. government is so motivated to prevent a widespread crisis that they've imposed what's called the "Animal Rule."
"The Animal Rule," says Paul Mampilly, editor of FDA Trader, "allows the FDA to give conditional approval to drugs if they work in animal studies."
Why, Paul explains to us, is "because we're going to need more than one drug to make sure, because the virus is adapting to us as we're adapting to it. It mutates into different forms."
Besides, we all have different DNA, and therefore different ways of reacting to certain drugs. Some drugs that work on one person may not work on another, and vice versa.
".we're going to need every single Ebola drug that we have to try to help these patients."
"That means we're going to need every single Ebola drug that we have to try to help these patients," Paul goes on. "And then pray that they work."
...The antiviral effect of the anti-HIV nanoviricide ("HIVCideT") continued throughout the 48 days of study even though HIVCide dosing was discontinued after only 20 days.
The clinical benefit of HIVCide was found to be sustained for at least four weeks after the last drug dose. Treatment with the lead anti-HIV nanoviricide both (1) reduced the HIV viral load and (2) also protected the human T cells (CD4+,CD8+), equally well as compared to treatment with the three-drug HAART cocktail, at 24-days as well as at 48-days, even though the HIVCide treatment was stopped at 20 days.
The Company believes that achieving this sustained clinical benefit after stopping nanoviricide treatment is highly significant. A similar sustained reduction in viral load even after discontinuation of the drug was also observed for the Company's previously reported anti-influenza FluCideT lead drug candidate. Such sustained, prolonged clinical effects can be expected on the basis of the design of nanoviricides. A nanoviricide is made from a polymeric micelle structure that is designed to provide long circulating half-life in the body that should enable sustained therapeutic effectiveness.
As part of the advanced IND–enabling development of our Injectable FluCide drug candidate, we have continued to scale up our production processes for both the backbone polymer and the ligands. We have been able to make up to 200g batches in our existing facility. We believe that we will be able to make as much as a few kilograms in a single batch in the new cGMP-capable facility. If the course of treatment of a successful Ebola drug candidate is assumed to be a few grams, we would be able to make as many as a thousand courses of treatment per batch. Our production capacity would thus be responsive to the current requirements for the containment of the Ebola epidemic in West Africa.
NanoViricides, Inc. therapeutic drugs have the science and bio-nanotechnology to boot. It is not so much about scientific theory anymore as we've been down this track before, in 2010.
It was not long ago we read the following in relation to the restart of the Ebola program,...
"...If Dr. Anil Diwan and his team are confident that the latest developed [second generation]EbolaCide candidate will yield a > 90% rescue of subject animals (in-vivo)....~ BigKahuna
Feds Provide Funding,...to Advance ZMapp Drug for Ebola
In a statement today[9/2/14], the federal health agency said it would provide $24.9 million to Mapp Bio through an initial, 18-month contract. The company will make a small amount of ZMapp for early stage clinical safety studies and for non-clinical animal studies needed to demonstrate its safety and effectiveness.
NanoViricides, Inc. now has its own drug manufacturing facility that is capable of producing sufficient quantities of an anti-Ebola drug after it is developed, for combating Ebola epidemics.
NanoViricides, Inc. (OTC BB: NNVC.OB) (the "Company") reports that the results of the evaluation of several of its nanoviricidesR anti-Ebola agents were presented July 17th at the Annual Meeting of the American Society for Virology, July 17-21, at Montana State University, Bozeman, MT. Dr. Corinne Scully delivered the presentation, which was entitled "Polymeric Micelle Nanomaterials as Antiviral Compounds For Ebola Virus Infection."
The studies were performed in the laboratory of Dr. Gene Olinger at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID), Frederick, MD.
In cell culture screening assays, the nanoviricidesR were evaluated for cytotoxicity and for inhibition of Ebola virus infection. Several of the nanoviricides demonstrated a dose-dependent inhibition of Ebola virus infectivity with no toxicity of uninfected cells at concentrations that were effective against the virus.
A subset of the nanoviricides that were effective in cell culture was selected for testing in vivo in a mouse model of Ebola infection. The compounds were well tolerated by the animals and showed some efficacy against the mouse-adapted Ebola virus. Efficacy of nanoviricides treatment against Ebola virus was demonstrated by an increase in lifespan in this uniformly 100% lethal animal model.
The Company developed a library of chemical ligands that were expected to bind to the Ebola virus envelope proteins. The results reported at this meeting will help guide the synthesis of next generation anti-Ebola nanoviricides in a lead optimization synthetic program. Optimized nanoviricides will also be evaluated against other similar Hemorrhagic Fever Viruses, e.g. Marburg virus.
Based on the current results and previous success with another hemorrhagic fever virus (viz. Dengue), the Company believes that it can develop a single nanoviricide drug that is highly effective against both Ebola and Marburg filoviruses. Developing a single drug against the various Ebola and Marburg viruses has been a major challenge for researchers in the field. Currently there are no approved vaccines or therapeutics for the prevention or treatment of Ebola and Marburg viruses.
Ebola/Marburg viruses are a major concern both as potential global health threats and as potential threat agents for Biosecurity and Biodefense, and are identified as Category A agents. Ebola outbreaks have resulted in fatality rates as high as 80% in humans ~ BusinessWire
[Marburg and Ebola virus] Entry
Niemann-Pick C1 (NPC1) appears to be essential for Ebola and Marburg virus infection. Two independent studies reported in the same issue of Nature (journal) showed that Ebola virus cell entry and replication requires the cholesterol transporter protein NPC1.[43][44] When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the virus, further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Ebola's cousin in the filovirus group, Marburg virus, showing that it too needs NPC1 to enter cells.[43][44] Furthermore, NPC1 was shown to be critical to filovirus entry because it mediates infection by binding directly to the viral envelope glycoprotein.[44] A later study confirmed the findings that NPC1 is a critical filovirus receptor that mediates infection by binding directly to the viral envelope glycoprotein and that the second lysosomal domain of NPC1 mediates this binding.[45]
In one of the original studies, a small molecule was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.[44][46] In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse adapted Ebola virus.[43] Together, these studies suggest NPC1 may be potential therapeutic target for an Ebola anti-viral drug.
In other news, NanoViricides reported that the synthesis of its anti-Ebola second generation drug candidates has started. We anticipate being able to evaluate these against Ebola virus with certain of our previous collaborators. The contracts to enable such evaluation are currently in progress. The Company's nanomedicine technology enables development of drugs that directly attack the virus, in a manner that a virus may not be able to overcome despite mutational changes. This is very important for the current epidemic-causing Ebola virus strain, which has been shown to be mutating rapidly.
NanoViricides has developed additional novel drug candidates against Ebola using its nanoviricide drug process. A nanoviricide drug consists of the combination of a virus-binding ligand that mimics the native receptor on the host cell and a backbone polymer that makes the drug look like the host cell surface to the virus.
"We believe the new anti-Ebola ligands should make the new drug candidates substantially superior to our older ones, based on the molecular modeling studies we have conducted using the structural information of interaction of Ebola virus glycoprotein with its cellular receptor Niemann-Pick C1 (NPC) protein," Anil Diwan, the president of NanoViricides, said. "We believe that Ebola virus will not be able to avoid our drug candidates in spite of mutations, because we are mimicking NPC1, the receptor to which the virus must bind in order to infect the host cell. Of course, we must await results from actual cell culture and animal testing to further develop these candidates."
As part of the advanced IND–enabling development of our Injectable FluCide drug candidate, we have continued to scale up our production processes for both the backbone polymer and the ligands. We have been able to make up to 200g batches in our existing facility. We believe that we will be able to make as much as a few kilograms in a single batch in the new cGMP-capable facility. If the course of treatment of a successful Ebola drug candidate is assumed to be a few grams, we would be able to make as many as a thousand courses of treatment per batch. Our production capacity would thus be responsive to the current requirements for the containment of the Ebola epidemic in West Africa.
(NaturalNews) Today Kurt Nimmo from Infowars.com is incorrectly reporting that "aerosol transmission is not possible" with Ebola. (2) That statement is part of an article entitled, "Don't Fear Ebola, Fear the State" which is, overall, a very compelling article.
Nimmo is a fantastic writer and a great researcher, but in this case his statement is factually incorrect and probably needs to be addressed. As clearly explained by the Public Health Agency of Canada: (3)
"INFECTIOUS DOSE: 1 - 10 aerosolized organisms are sufficient to cause infection in humans."
Ebola, you see, can "ride" on aerosolized particles of blood, mucous and other body fluids. Someone sneezing, for example, can cause Ebola viruses to be aerosolized where they land on other people's hands or faces. It only takes one virus entering the corner of your eye (or the corner of your mouth) to set off a full-blown infection.
In fact, a 2012 BBC article entitled "Growing concerns over 'in the air' transmission of Ebola" states: (3)
Canadian scientists have shown that the deadliest form of the ebola virus could be transmitted by air between species. In experiments, they demonstrated that the virus was transmitted from pigs to monkeys without any direct contact between them. In their experiments, the pigs carrying the virus were housed in pens with the monkeys in close proximity but separated by a wire barrier. After eight days, some of the macaques were showing clinical signs typical of ebola and were euthanised.
The problem now is that we have burned-out health care workers, 550 from the largest contingent from Medecins Sans Frontieres, or Doctors Without Borders, which has issued plea after plea in recent days to the international community saying, "We are exhausted. We are terrified. We want to leave. Can somebody else please come in and take over?"
You've had more than 60 health care workers succumb to the disease, including the most famous physicians in the battle against Ebola from Uganda, from Liberia, and from Sierra Leone. And we have seen resistance from the populations all over these three countries, but especially in Sierra Leone and Liberia, against all sorts of quarantine measures, efforts to remove the ailing from their households so that they don't infect household members and place them in quarantine, efforts to enforce quarantine, and efforts to deal with safe burials or cremations, rather than the traditional approaches of families preparing the bodies for burial and thereby getting exposed to the contaminating fluids.
I read and used the following post/information from 4theduration/connecticutmag.com and wrote to the U.S. Senators from my state exhorting them to join Connecticut/U.S. Senator Richard Blumenthal in moving the testing process along for a potentially safe, fast-acting, effective anti-Ebola drug.
Update from connecticutmag.com: Testing for a potentially life saving Ebola drug that would be developed and manufactured in Connecticut could begin as soon as a month from now, says Dr. Anil Diwan, chairman and president of NanoViricides, Inc., a biotech company based in Shelton . . .
As we initially reported in August, NanoViricides has plans for a drug that scientists at the company believe can be used to safely treat Ebola, and which could be manufactured in great enough quantity at the company’s Connecticut lab. At that time it appeared the company would not be able to test the concept until after the current Ebola outbreak was brought under control because there are only a handful of labs in the world with live Ebola strains available for testing—all of which are overwhelmed by the outbreak. However, Connecticut U.S. Senator Richard Blumenthal recently wrote a letter on the company’s behalf to help move the testing process along. Now Diwan says the company has a contract ready to be signed with a lab with Ebola-testing capabilities and that testing could begin soon (he says he can’t release the name of this lab until after the contract is signed).
In a statement today, the federal health agency said it would provide $24.9 million to Mapp Bio through an initial, 18-month contract. The company will make a small amount of ZMapp for early stage clinical safety studies and for non-clinical animal studies needed to demonstrate its safety and effectiveness.
The political becomes personal and deeply moving in 'The Good Lie,' a cross-cultural story of Sudanese refugees told in an engaging way~ USA Today
"Who wants to be locked up?" she said Thursday. Private security guards and sheriff's deputies blocked the entrance to the 300-unit apartment complex.
Dallas officials were waiting for a permit to allow hazardous material taken from the home to be transported along state highways for disposal, city spokeswoman Sana Syed said Friday.
A cleanup crew hired by the county and state arrived Thursday evening to assess the job but did not complete it. They expected to return Friday.
The first Ebola diagnosis in the nation has raised concerns about whether the disease that has killed 3,300 people in West Africa could spread in the U.S. Federal health officials say they are confident they can keep it in check.
...
The at-risk group includes 12 to 18 people who came in direct contact with the infected man, including an ambulance crew and a handful of schoolchildren, she said.
...
"This is a big spider web" of people involved, Neroes said.
The virus that causes Ebola is not airborne and can only be spread through close contact with someone who has symptoms. People must come into direct contact with the patient's bodily fluids — blood, sweat, vomit, feces, urine, saliva or semen — and those fluids must have an entry point.
Ebola dried on surfaces can survive for several hours, according to the CDC.
But I heard it could spread through the air.
A renowned infectious disease expert named Michael Osterholm wrote an opinion piece in the New York Times that said airborne Ebola is possible if the virus mutates enough. As TIME reported in the past, anything is possible with viruses, but there are many other mutations that are more likely than a change in the mode of transmission—meaning how you catch it. For instance, a virus could become more virulent—more contagious—or could develop incubation periods that are longer than the current estimate of 21 days. But in general, scientists are not very concerned about that.
The Economic Implications Of A Potential Ebola Pandemic In The United States
Predictably, many parents are already pulling their kids out of school in the Dallas area.
It shall be very interesting to see how many kids actually show up for school tomorrow morning.
But this is what happens to a society when the fear of Ebola takes hold. People almost immediately start shutting down their activities and staying home.
Over in West Africa, months of Ebola fear is starting to take a major toll on the economy. For example, the president of Guinea says that his economy is on the verge of complete collapse...
...
If thousands of people start getting Ebola in major cities all over America, the same thing will happen here too.
A major Ebola pandemic in America would mean an almost total economic shutdown and basic essentials would start disappearing from the marketplace almost immediately. Just check out what is happening in Liberia even as you read this...
...
Even though economic demand would drop through the floor for most things, prices for food and other essential supplies tend to skyrocket during a major emergency. The IMF says that the inflation rate will hit approximately 13 percent in Liberia by the end of the year even though economic activity has declined dramatically. It is going to become extremely challenging for most families over there to feed themselves.
And as economic activity withers, tax revenues also dry up. Liberia, Guinea and Sierra Leone are all facing massive revenue shortfalls, and they are asking for international assistance.
But if the same thing happened in the United States, do you think the rest of the world would send us lots of money to help us pay our bills?
Lvus, good to read your reaction. Yes, we are going to kick some serious Ebola butt soon. It was not long ago I read the following, "...If Dr. Anil Diwan and his team are confident that the latest developed [second generation]EbolaCide candidate will yield a > 90% rescue of subject animals (in-vivo)....
We should soon be on a fast war path with the killer virus! Nanopatent said on reply to your post, "...I'd say days, weeks tops.! Ditto on that!, or as Dr. Seymour once put it, "...I suspect that game-over for that virus, is in the cards...".
And how soon should we be reaching people afficted with the killer virus? With the speed afforded to ZMapp!
Feds Provide Funding,...to Advance ZMapp Drug for Ebola
In a statement today[9/2/14], the federal health agency said it would provide $24.9 million to Mapp Bio through an initial, 18-month contract. The company will make a small amount of ZMapp for early stage clinical safety studies and for non-clinical animal studies needed to demonstrate its safety and effectiveness.
NanoViricides, Inc. now has its own drug manufacturing facility that is capable of producing sufficient quantities of an anti-Ebola drug after it is developed, for combating Ebola epidemics.
NanoViricides, Inc. (OTC BB: NNVC.OB) (the "Company") reports that the results of the evaluation of several of its nanoviricidesR anti-Ebola agents were presented July 17th at the Annual Meeting of the American Society for Virology, July 17-21, at Montana State University, Bozeman, MT. Dr. Corinne Scully delivered the presentation, which was entitled "Polymeric Micelle Nanomaterials as Antiviral Compounds For Ebola Virus Infection."
The studies were performed in the laboratory of Dr. Gene Olinger at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID), Frederick, MD.
In cell culture screening assays, the nanoviricidesR were evaluated for cytotoxicity and for inhibition of Ebola virus infection. Several of the nanoviricides demonstrated a dose-dependent inhibition of Ebola virus infectivity with no toxicity of uninfected cells at concentrations that were effective against the virus.
A subset of the nanoviricides that were effective in cell culture was selected for testing in vivo in a mouse model of Ebola infection. The compounds were well tolerated by the animals and showed some efficacy against the mouse-adapted Ebola virus. Efficacy of nanoviricides treatment against Ebola virus was demonstrated by an increase in lifespan in this uniformly 100% lethal animal model.
The Company developed a library of chemical ligands that were expected to bind to the Ebola virus envelope proteins. The results reported at this meeting will help guide the synthesis of next generation anti-Ebola nanoviricides in a lead optimization synthetic program. Optimized nanoviricides will also be evaluated against other similar Hemorrhagic Fever Viruses, e.g. Marburg virus.
Based on the current results and previous success with another hemorrhagic fever virus (viz. Dengue), the Company believes that it can develop a single nanoviricide drug that is highly effective against both Ebola and Marburg filoviruses. Developing a single drug against the various Ebola and Marburg viruses has been a major challenge for researchers in the field. Currently there are no approved vaccines or therapeutics for the prevention or treatment of Ebola and Marburg viruses.
Ebola/Marburg viruses are a major concern both as potential global health threats and as potential threat agents for Biosecurity and Biodefense, and are identified as Category A agents. Ebola outbreaks have resulted in fatality rates as high as 80% in humans ~ BusinessWire
Marburg haemorrhagic fever
Key facts:
-The Marburg virus causes severe viral haemorrhagic fever in humans.
-Case fatality rates in Marburg haemorrhagic fever outbreaks have ranged from 24% to 88%.
-Rousettus aegypti, fruit bats of the Pteropodidae family, are considered to be natural hosts of Marburg virus. The Marburg virus is transmitted to people from fruit bats and spreads among humans through human-to-human transmission.
-No specific antiviral treatment or vaccine is available.
[Marburg and Ebola virus] Entry
Niemann-Pick C1 (NPC1) appears to be essential for Ebola and Marburg virus infection. Two independent studies reported in the same issue of Nature (journal) showed that Ebola virus cell entry and replication requires the cholesterol transporter protein NPC1.[43][44] When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the virus, further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Ebola's cousin in the filovirus group, Marburg virus, showing that it too needs NPC1 to enter cells.[43][44] Furthermore, NPC1 was shown to be critical to filovirus entry because it mediates infection by binding directly to the viral envelope glycoprotein.[44] A later study confirmed the findings that NPC1 is a critical filovirus receptor that mediates infection by binding directly to the viral envelope glycoprotein and that the second lysosomal domain of NPC1 mediates this binding.[45]
In one of the original studies, a small molecule was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.[44][46] In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse adapted Ebola virus.[43] Together, these studies suggest NPC1 may be potential therapeutic target for an Ebola anti-viral drug.
In other news, NanoViricides reported that the synthesis of its anti-Ebola second generation drug candidates has started. We anticipate being able to evaluate these against Ebola virus with certain of our previous collaborators. The contracts to enable such evaluation are currently in progress. The Company's nanomedicine technology enables development of drugs that directly attack the virus, in a manner that a virus may not be able to overcome despite mutational changes. This is very important for the current epidemic-causing Ebola virus strain, which has been shown to be mutating rapidly.
NanoViricides has developed additional novel drug candidates against Ebola using its nanoviricide drug process. A nanoviricide drug consists of the combination of a virus-binding ligand that mimics the native receptor on the host cell and a backbone polymer that makes the drug look like the host cell surface to the virus.
"We believe the new anti-Ebola ligands should make the new drug candidates substantially superior to our older ones, based on the molecular modeling studies we have conducted using the structural information of interaction of Ebola virus glycoprotein with its cellular receptor Niemann-Pick C1 (NPC) protein," Anil Diwan, the president of NanoViricides, said. "We believe that Ebola virus will not be able to avoid our drug candidates in spite of mutations, because we are mimicking NPC1, the receptor to which the virus must bind in order to infect the host cell. Of course, we must await results from actual cell culture and animal testing to further develop these candidates."
We've travelled down this road before, 2010...
NanoViricides, Inc. (OTC BB: NNVC.OB) (the "Company") reports that the results of the evaluation of several of its nanoviricidesR anti-Ebola agents were presented July 17th at the Annual Meeting of the American Society for Virology, July 17-21, at Montana State University, Bozeman, MT. Dr. Corinne Scully delivered the presentation, which was entitled "Polymeric Micelle Nanomaterials as Antiviral Compounds For Ebola Virus Infection."
The studies were performed in the laboratory of Dr. Gene Olinger at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID), Frederick, MD.
In cell culture screening assays, the nanoviricidesR were evaluated for cytotoxicity and for inhibition of Ebola virus infection. Several of the nanoviricides demonstrated a dose-dependent inhibition of Ebola virus infectivity with no toxicity of uninfected cells at concentrations that were effective against the virus.
A subset of the nanoviricides that were effective in cell culture was selected for testing in vivo in a mouse model of Ebola infection. The compounds were well tolerated by the animals and showed some efficacy against the mouse-adapted Ebola virus. Efficacy of nanoviricides treatment against Ebola virus was demonstrated by an increase in lifespan in this uniformly 100% lethal animal model.
The Company developed a library of chemical ligands that were expected to bind to the Ebola virus envelope proteins. The results reported at this meeting will help guide the synthesis of next generation anti-Ebola nanoviricides in a lead optimization synthetic program. Optimized nanoviricides will also be evaluated against other similar Hemorrhagic Fever Viruses, e.g. Marburg virus.
Based on the current results and previous success with another hemorrhagic fever virus (viz. Dengue), the Company believes that it can develop a single nanoviricide drug that is highly effective against both Ebola and Marburg filoviruses. Developing a single drug against the various Ebola and Marburg viruses has been a major challenge for researchers in the field. Currently there are no approved vaccines or therapeutics for the prevention or treatment of Ebola and Marburg viruses.
Ebola/Marburg viruses are a major concern both as potential global health threats and as potential threat agents for Biosecurity and Biodefense, and are identified as Category A agents. Ebola outbreaks have resulted in fatality rates as high as 80% in humans ~ BusinessWire
Marburg haemorrhagic fever
Key facts:
-The Marburg virus causes severe viral haemorrhagic fever in humans.
-Case fatality rates in Marburg haemorrhagic fever outbreaks have ranged from 24% to 88%.
-Rousettus aegypti, fruit bats of the Pteropodidae family, are considered to be natural hosts of Marburg virus. The Marburg virus is transmitted to people from fruit bats and spreads among humans through human-to-human transmission.
-No specific antiviral treatment or vaccine is available.
[Marburg and Ebola virus] Entry
Niemann-Pick C1 (NPC1) appears to be essential for Ebola and Marburg virus infection. Two independent studies reported in the same issue of Nature (journal) showed that Ebola virus cell entry and replication requires the cholesterol transporter protein NPC1.[43][44] When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the virus, further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Ebola's cousin in the filovirus group, Marburg virus, showing that it too needs NPC1 to enter cells.[43][44] Furthermore, NPC1 was shown to be critical to filovirus entry because it mediates infection by binding directly to the viral envelope glycoprotein.[44] A later study confirmed the findings that NPC1 is a critical filovirus receptor that mediates infection by binding directly to the viral envelope glycoprotein and that the second lysosomal domain of NPC1 mediates this binding.[45]
In one of the original studies, a small molecule was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.[44][46] In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse adapted Ebola virus.[43] Together, these studies suggest NPC1 may be potential therapeutic target for an Ebola anti-viral drug.
In other news, NanoViricides reported that the synthesis of its anti-Ebola second generation drug candidates has started. We anticipate being able to evaluate these against Ebola virus with certain of our previous collaborators. The contracts to enable such evaluation are currently in progress. The Company's nanomedicine technology enables development of drugs that directly attack the virus, in a manner that a virus may not be able to overcome despite mutational changes. This is very important for the current epidemic-causing Ebola virus strain, which has been shown to be mutating rapidly.
NanoViricides has developed additional novel drug candidates against Ebola using its nanoviricide drug process. A nanoviricide drug consists of the combination of a virus-binding ligand that mimics the native receptor on the host cell and a backbone polymer that makes the drug look like the host cell surface to the virus.
"We believe the new anti-Ebola ligands should make the new drug candidates substantially superior to our older ones, based on the molecular modeling studies we have conducted using the structural information of interaction of Ebola virus glycoprotein with its cellular receptor Niemann-Pick C1 (NPC) protein," Anil Diwan, the president of NanoViricides, said. "We believe that Ebola virus will not be able to avoid our drug candidates in spite of mutations, because we are mimicking NPC1, the receptor to which the virus must bind in order to infect the host cell. Of course, we must await results from actual cell culture and animal testing to further develop these candidates."
The opportunity to develop a offers the possibility of getting fast tracked to market with far fewer obstacles than otherwise. Bringing attention to EbolaCide II also means bringing attention to broad-spectrum FluCide which is currently advancing with their GLP full tox study
IF the new Ebolacide2 formulation(s) do work I think it will be a lot better for NNVC than most here imagine. A demonstrated effectiveness in this formulation in the hands of our BSL4 equipped partners' labs against Ebola model animals could (indeed) put a fast-track on Ebolacide that would not be possible with Flucide still needing to get TOX testing done, the IND together & approved, and the human testing done.
The Ebolacide fast-track (if that happens) could get NNVC on the map and generating revenue a year or more earlier that would the long slog to get Flucide through it's hoops.~ drkazmd65
So, yes, as some argue, there might not be much economic value and money in Ebola and other disease predominant in developing countries. But you don't want a few billion desperate and angry people. if we don't take care of the least fortunate, our nice and comfy society as we know it may not be around for our kids or grand kids.~ Kulnor
...NanoViricides reported that the synthesis of its anti-Ebola second generation drug candidates has started. We anticipate being able to evaluate these against Ebola virus with certain of our previous collaborators. The contracts to enable such evaluation are currently in progress. The Company’s nanomedicine technology enables development of drugs that directly attack the virus, in a manner that a virus may not be able to overcome despite mutational changes. This is very important for the current epidemic-causing Ebola virus strain, which has been shown to be mutating rapidly.~ 10-K
But I heard it could spread through the air.
A renowned infectious disease expert named Michael Osterholm wrote an opinion piece in the New York Times that said airborne Ebola is possible if the virus mutates enough. As TIME reported in the past, anything is possible with viruses, but there are many other mutations that are more likely than a change in the mode of transmission—meaning how you catch it. For instance, a virus could become more virulent—more contagious—or could develop incubation periods that are longer than the current estimate of 21 days. But in general, scientists are not very concerned about that.~ TIME
“Tekmira’s [drug] is one drug that has shown some promise, but like all of them, it still has a long way to go,” said Daniel Bausch, a professor at the Tulane University School of Public Health and Tropical Medicine, who is advising the WHO on experimental approaches in this outbreak. “None of them have what we really want, which is full safety testing and full evidence of efficacy.”
...
Teasing out what role TKM-Ebola played in Dr. Sacra’s recovery is virtually impossible. Along with his seven days of intravenous TKM-Ebola treatment, Dr. Sacra received plasma transfusions on days two and three of his treatment from Kent Brantly, an Ebola survivor whose type A blood matched Dr. Sacra’s.
"His whole family was screaming. He got outside and he was throwing up all over the place," resident Mesud Osmanovic, 21, said on Wednesday, describing the chaotic scene before the man was admitted to Texas Health Presbyterian Hospital on Sunday where he is in serious condition.
...
Texas health officials said that up to 18 people, including five children, had contact with the Ebola patient since he returned to the United States in late September. The children had gone to school early this week but have since been sent home and are being monitored for symptoms.
Ebola spreads through contact with bodily fluids such as blood or saliva, which health experts say limits its potential to infect others, unlike airborne diseases.~ Yahoo News
NewMoney, since you brought it up this morning, here is more information on TKM-Ebola so that you and NNVC board can better compare the two treatments.
The Company’s nanomedicine technology enables development of drugs that directly attack the virus, in a manner that a virus may not be able to overcome despite mutational changes. This is very important for the current epidemic-causing Ebola virus strain, which has been shown to be mutating rapidly.
Injectable FluCide was found to be extremely safe in mice in a preliminary safety study. This study showed no evidence of any adverse events even at the maximum tolerable dose level. No significant changes in all observed parameters were found even at the maximum feasible dose of approximately 2,700 mg/kg/d repeatedly given for five consecutive days.
TKM-Ebola is an intravenously infused RNAi Therapeutic that has been demonstrated to save the lives of monkeys infected with an otherwise fatal dose of Ebola. It is being developed under the ‘Animal Rule’ in efforts funded by the US government which is afraid that this virus could be weaponized and used as a bioterror agent. The ‘Animal Rule’ is a development pathway instituted by the US FDA for diseases such as Ebola for which it would either be impractical or unethical to conduct efficacy studies in humans.
Unfortunately, in the midst of the outbreak, the FDA instituted a Clinical Hold on the TKM-Ebola safety study because a case of dangerously high cytokine elevations was observed at the highest dose planned in this dose-escalating/dose-finding study (0.5mg/kg). I agree that this is to be considered a serious adverse event in a volunteer that is not infected with the virus.
The reason for the cytokine stimulation is likely due to TLR-mediated, lipid-amplified innate immune stimulation, a known risk of liposomal RNAi delivery, especially at doses of 0.5mg/kg and higher. It is also the reason why all other active development candidates by Tekmira and their licensee Alnylam are conducted in the presence of transient immune suppression with steroids and the like which in many cases is acceptable given the severe diseases these treatments go after.
“Tekmira’s [drug] is one drug that has shown some promise, but like all of them, it still has a long way to go,” said Daniel Bausch, a professor at the Tulane University School of Public Health and Tropical Medicine, who is advising the WHO on experimental approaches in this outbreak. “None of them have what we really want, which is full safety testing and full evidence of efficacy.”
“Tekmira’s [drug] is one drug that has shown some promise, but like all of them, it still has a long way to go,” said Daniel Bausch, a professor at the Tulane University School of Public Health and Tropical Medicine, who is advising the WHO on experimental approaches in this outbreak. “None of them have what we really want, which is full safety testing and full evidence of efficacy.”
...
Teasing out what role TKM-Ebola played in Dr. Sacra’s recovery is virtually impossible. Along with his seven days of intravenous TKM-Ebola treatment, Dr. Sacra received plasma transfusions on days two and three of his treatment from Kent Brantly, an Ebola survivor whose type A blood matched Dr. Sacra’s.
Before, now/2014 and coming soon is tomorrow, the conquest of Ebola and Marburg virus!
From Page 47 of 10-K:
In July 2010, our collaborators at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) presented the data on evaluation of anti-Ebola/Marburg nanoviricides. Significant efficacy was reported to have been achieved in cell culture studies. Animal studies indicated improvement in lifetime in the uniformly lethal mouse model. Further improvement in chemistry and dosage levels may be expected to lead to significant survival.
From Page 5 of 10-K:
In September 2014, we announced that we have already completed design of novel anti-Ebola drug candidates, based on “in silico” or molecular modeling approach, and synthesis has also begun. We believe that we will be able to work with our previous collaborators at the USAMRIID, and also at the NIH, to begin testing the potential utility of these drug candidates in cell cultures and animal models soon.
From Page 51 of 10-K:
To date, we have entered into the following collaborations.
Cooperative Research and Development Agreement for Material Transfer, dated October 15, 2007, between NanoViricides, Inc. and United States Army Medical Research Institute of Infectious Disease (“Laboratory”).
The term of the agreement was for one year initially and extended for an additional year. It has been extended again, based on positive results. The Company shall invent, develop, and provide to the laboratory, Nanoviricides® that are expected to be capable of attacking a multiplicity of different Ebola and Marburg viruses. The Laboratory shall assess in vitro and in vivo activity of the anti-Ebola Nanoviricides® provided against the virus.
There is no payment by the Company to the Laboratory, nor from the Laboratory to the Company. USAMRIID has federal funding to support their part of the work.
Before, now and coming soon is tomorrow, the conquest of Ebola and Marburg virus!
From Page 47 of 10K:
In July 2010, our collaborators at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) presented the data on evaluation of anti-Ebola/Marburg nanoviricides. Significant efficacy was reported to have been achieved in cell culture studies. Animal studies indicated improvement in lifetime in the uniformly lethal mouse model. Further improvement in chemistry and dosage levels may be expected to lead to significant survival.
In September 2014, we announced that we have already completed design of novel anti-Ebola drug candidates, based on “in silico” or molecular modeling approach, and synthesis has also begun. We believe that we will be able to work with our previous collaborators at the USAMRIID, and also at the NIH, to begin testing the potential utility of these drug candidates in cell cultures and animal models soon.
To date, we have entered into the following collaborations.
Cooperative Research and Development Agreement for Material Transfer, dated October 15, 2007, between NanoViricides, Inc. and United States Army Medical Research Institute of Infectious Disease (“Laboratory”).
The term of the agreement was for one year initially and extended for an additional year. It has been extended again, based on positive results. The Company shall invent, develop, and provide to the laboratory, Nanoviricides® that are expected to be capable of attacking a multiplicity of different Ebola and Marburg viruses. The Laboratory shall assess in vitro and in vivo activity of the anti-Ebola Nanoviricides® provided against the virus.
There is no payment by the Company to the Laboratory, nor from the Laboratory to the Company. USAMRIID has federal funding to support their part of the work.
The Ebola virus may be getting closer to Americans than ever, but don't expect big American drug companies to come to the rescue.
Instead, small biotech firms, academics and government agencies are leading the search for an Ebola cure.
...
Smaller biotech firms are more likely to be drawn to the project of finding a cure or vaccine thanks to the lure of government funding, good PR and the chance to make an impact, Geisbert said.
It appears that if EbolaCide works very well, then this could become a higher priority (and sooner into humans?). But it's not going to slow down the FluCide toxicity testing. It may delay overall FluCide development, however. ~ KMBJN

The June 2014 report from the Department of Justice on damages paid by the U.S. Government to vaccine victims was recently published on the U.S. Department of Health and Human Resources website. There were 120 cases of vaccine injuries decided. 78 cases received compensation, while 42 cases were denied.
Most of the U.S. public is unaware that a U.S. citizen, by law, cannot sue a pharmaceutical company for damages resulting from vaccines. Congress gave them total legal immunity in 1986, and that law was upheld by the U.S. Supreme Court in 2011. There is a special "vaccine court" called the National Vaccine Injury Compensation Program that is funded through a tax on vaccines. If you are injured or killed by a vaccine, you must hire an attorney and fight tax-funded government attorneys to seek damages, as you cannot sue the drug manufacturers. As you can see from the report below, it takes years to reach a settlement, with the longest case below being settled after 11 years. Therefore, this report probably only represents a tiny fraction of the actual number of people harmed or killed by vaccines, since it is so difficult to fight the government in court to win a settlement.
As part of the advanced IND-enabling development of our Injectable FluCide drug candidate, we have continued to scale up our production processes for both the backbone polymer and the ligands. We have been able to make up to 200g batches in our existing facility. We believe that we will be able to make as much as a few kilograms in a single batch in the new cGMP-capable facility. If the course of treatment of a successful Ebola drug candidate is assumed to be a few grams, we would be able to make as many as a thousand courses of treatment per batch. Our production capacity would thus be responsive to the current requirements for the containment of the Ebola epidemic in West Africa.
As I see it, the only way the FluCide program would be delayed would be if the following two conditions were present:
#1) EbolaCide2 works incredibly well
AND
#2) We receive a phenomenal amount of money from all of the agencies now committed to eradicating the disease in order to ramp up the production of the Ebola drug. ~ Dr. Eugene Seymour, CEO of NanoViricides, Inc. to drkazmd65
investorshub.advfn.com/boards/read_msg.aspx?message_id=106739780
Professor Boniuk joined the Company’s Board as an independent director in May, 2013, at the request of the Company’s executives. He says that his confidence in the Company has only continued to grow as he sees the Company’s management and execution now from a closer perspective.
When asked why he has made these investments, he explained that “As I became familiar with the technology and the various on-going programs that the Company has, it became apparent that the potential was nothing short of amazing. Dr. Diwan explained to me how it would be possible to create a novel drug against a previously unknown virus in a matter of weeks.
FORZANANO/nanopatent and other proactive NanoViricides, Inc. investors...
We are just a few weeks away from starting and completion of EbolaCide efficacy tests. First, we should soon find out who is going to be our BSL-4 partner to test EbolaCide for efficacy. Soon.
Since I do believe that NanoViricides, Inc. will soon have dramatically improved EbolaCide candidates I have, in anticipation of great efficacy results, drafted a petition to be posted in different places, government webpage. I want your opinion to improve on this petition that ultimately I hope "longs" will make it theirs.
Honorable _______,
The recent Ebola outbreak in West Africa has been the deadliest so far, per the World Health Organization (WHO). Ebola virus causes a deadly disease, with 60% to 90% of infected people dying, depending upon the strain of the virus. Fortunately, it is transmitted only through close contact with an infected person’s body fluids, and not through aerosol or the water route, thus limiting its transmission.
According to the WSJ:
Ebola’s economic impact has become so severe that the IMF is now warning that stricken countries could need emergency assistance. Guinea, Liberia and Sierra Leone have all been burning holes in their finances trying to curb the outbreak, and a dramatic downturn in trade—specifically timber and rubber—will compound those troubles.
And the NYT is along much the same lines:
Ebola — the reality and the hysteria over it — is having a serious economic impact on Guinea, Liberia and Sierra Leone, three nations already at the bottom of global economic and social indicators. Aggravating both the financial and social consequences, these countries and their frightened African neighbours are enacting concentric circles of quarantines, cutting off neighbourhoods, regions and even whole nations.
Currently, there are no licensed drugs or vaccines for Ebola, although some vaccines as well as some drug candidates have entered clinical trials.
Approximately 729 people have died and 1300 infected from Ebola virus in the current epidemic. It takes anywhere from 2 to 20 days for Ebola virus illness signs to appear after infection. In addition, the virus may persist up to 7 weeks in bodily fluids of recovered patients. Therefore, it is possible that transmission could sometimes occur far away with air travel, unknowingly.
(Sources: www.cdc.gov/vhf/ebola/ , “Why Ebola is So Dangerous” - http://www.bbc.com/news/world-africa-26835233 , “Ebola Virus Disease”- http://www.who.int/mediacentre/factsheets/fs103/en/).
Vaccines are 30%-50% effective on virus infected subjects and the Ebola virus is mutating rapidly, over 400 genetic modifications were found during the course of a recent study, and that is detrimental to current vaccines or future vaccines. Mutations of Ebola virus renders
current vaccines ineffective. A treatment is therefore, essential.
Ebola virus infects a wide variety of human cell types, by using various attachment receptors to enter endosome structures inside cells. Upon entry, the virus binds to its cognate receptor, the Niemann-Pick C1 cholesterol transporter protein inside the late endosomes, fuses with the endosomal membrane, and thus enters the cytoplasm. It replicates, buds out of the cell, and the cycle repeats itself. The virus shuts down several of the host’s immune system defenses, and thus gains an upper hand.
“We believe that with our ‘intelligent nanomachines’ approach we have the potential to develop superior therapeutics as compared to other approaches,” said Anil R. Diwan, PhD, President of the Company.
[[[“We are happy to restart the Ebola program, considering the public health impact of the Ebola virus infection,” said Dr. Eugene Seymour, MD, MPH, CEO of the Company, adding, “We are in a strong financial position now, enabling us to work on this project while we continue to advance our FluCide™ and DengueCide™ therapies further towards clinical trials. We hope to create highly effective drugs against Ebola, similar to what we have achieved with our FluCide™ Influenza drug candidate.”]]]
FDA recently fast tracked clinical trials of Zmapp developed by Mapp Biopharmaceuticals. This experimental drug was employed under the FDA "Animal Rule " on the American/Samaritans Purse health workers infected with Ebola and will undergo clinical trials. "In a statement...the federal health agency said it would provide $24.9 million to Mapp Bio through an initial, 18-month contract. The company will make a small amount of ZMapp for early stage clinical safety studies and for non-clinical animal studies needed to demonstrate its safety and effectiveness."
http://www.xconomy.com/san-diego/2014/09/02/feds-provide-funding-expertise-to-advance-zmapp-drug-for-ebola/
NanoViricides, Inc. now has its own drug manufacturing facility that is capable of producing sufficient quantities of an anti-Ebola drug now that it is developed, for combating Ebola epidemics.
From Business Week:
Ebola Spread Poses a Security Threat, President Barack Obama told a U.N. President Barack Obama urged world leaders to step up their nations’ efforts in helping to contain the Ebola virus outbreak, warning that the spread of the disease could pose a global security threat.
Please help us urge the Food and Drug Administration (FDA) to use the Accelerated Approval pathway for approval and access to safe, effective EbolaCide treatment for Ebola virus - the leading killer of 3,000 people in West Africa, and thousands more infected according to latest estimates from the World Health Organization.
Echo20...has the FluCide full tox study started?
"We are doing all of the preparatory pre-tox work now and the formal tox studies by BASI will start as soon as the pre-tox work is done and an [sic] sufficient amount of material is available from the existing lab. We're estimating a June start though that could go either way by 2-3 months depending on both BASI's schedule and ours." ~ Dr. E. Seymour, CEO NanoViricides, Inc. --- Apr 2014
....
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
....
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. One year to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is an absurd claim. Even highly toxic oncological drugs take 9 months on average. ~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
Under 505b, tox for FluCide becomes tox for everything that follows. For the second tox following FluCide, they will only need to show bioequivalence, that is, a bridge study would be required to demonstrate that a system processes EbolaCide, DengueCide, X-Cide, in the same way as FluCide. Since FluCide will most probably already be in clinical trials, the second and maybe third X-Cide for clinical trials will need only a bridge study in Phase 1 to advance to Phase 2/3, again that would be an abbreviated PK study showing bioequivalence in processing versions of nanomicelles. ~ BigKahuna
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange

...perhaps US $17.9 million with an additional US $24.9 million would be nicer...
In a statement today, the federal health agency said it would provide $24.9 million to Mapp Bio through an initial, 18-month contract. The company will make a small amount of ZMapp for early stage clinical safety studies and for non-clinical animal studies needed to demonstrate its safety and effectiveness.
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the [FluCide] tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
Excerpt from NanoBusiness Alliance Interview with Anil R. Diwan - 2011
SW: We've read stories in the past about the Ebola virus and how vicious and deadly it is. Do you think you’ll be able to come up with a treatment for Ebola in the future using your nanotechnology?
AD: Ebola virus is quite deadly. A single infectious particle can cause severe hemorrhagic disease in humans and about 50-80% of patients die. A related virus, called Marburg, is less infectious. These viruses are so deadly that they require BSL4+ facilities. Ebola is also a Category A pathogen on the CDC biodefense/terrorism category list.
We have been working with the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) to test nanoviricides against Ebola. In the first cell culture testing experiments we have obtained very good success. In the follow-on animal study, we obtained an excellent indication of safety. In the animal efficacy study, we have obtained indications that the test materials look promising but need improvements in efficacy. USAMRIID scientists have published these studies in international conferences.
Given the improvements we have been able to achieve in other projects, we believe that it is quite feasible to develop a nanoviricide drug against Ebola. A grant application to the US Department of Defense that we had sent in collaboration with USAMRIID passed the scientific review and went into "reserve” status". However, it did not receive funding. So at present, we are working on this project as a back burner project.
Ebola virus infects a wide variety of human cell types, by using various attachment receptors to enter endosome structures inside cells. Upon entry, the virus binds to its cognate receptor, the Niemann-Pick C1 cholesterol transporter protein inside the late endosomes, fuses with the endosomal membrane, and thus enters the cytoplasm. It replicates, buds out of the cell, and the cycle repeats itself. The virus shuts down several of the host’s immune system defenses, and thus gains an upper hand.
...
NanoViricides, Inc. now has its own drug manufacturing facility that is capable of producing sufficient quantities of an anti-Ebola drug after it is developed, for combating Ebola epidemics.
NanoViricides has developed additional novel drug candidates against Ebola using its nanoviricide drug process. A nanoviricide drug consists of the combination of a virus-binding ligand that mimics the native receptor on the host cell and a backbone polymer that makes the drug look like the host cell surface to the virus.
“We believe the new anti-Ebola ligands should make the new drug candidates substantially superior to our older ones, based on the molecular modeling studies we have conducted using the structural information of interaction of Ebola virus glycoprotein with its cellular receptor Niemann-Pick C1 (NPC) protein,” Anil Diwan, the president of NanoViricides, said. “We believe that Ebola virus will not be able to avoid our drug candidates in spite of mutations, because we are mimicking NPC1, the receptor to which the virus must bind in order to infect the host cell. Of course, we must await results from actual cell culture and animal testing to further develop these candidates.” ~ Dr. Anil Diwan
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange

When Thomas Jefferson became president in 1801, he decided that it was time to take military action to end the two-decades-old unprovoked Muslim terrorist attacks against Americans. [14] Using the brand new American Navy to transport the U. S. Marines overseas (President George Washington had called for the construction of a navy in 1795, and President John Adams had overseen its construction [15]), General William Eaton took the American military and proceeded to the same region of the world where Americans are still being attacked today. He then led a successful five-year campaign to free captured Americans and crush Muslim terrorist forces. [16] Tripoli (now called Libya) finally capitulated and signed a treaty on America’s terms in 1805, thus ending their aggressions – at least for a while. [17]~ WallBuilders
It's all scripted! Ebola outbreak and impossibly rapid vaccine response clearly scripted; U.S. govt. patented Ebola in 2010 and now owns all victims' blood
(NaturalNews) On the very same day that vaccine maker GlaxoSmithKline is being fined $490 million by Chinese authorities for running an illegal bribery scheme across China [3], the media is announcing the "astonishing" launch of human trials for an Ebola vaccine.
Care to guess who will be manufacturing this vaccine once it is whitewashed and rubber-stamped as "approved?" GlaxoSmithKline, of course. The same company that also admitted to a massive criminal bribery network in the United States, where felony crimes were routinely committed to funnel money to over 40,000 physicians who pushed dangerous prescription drugs onto patients.
This is the company that is now -- today! -- injecting 60 "volunteers" with an experimental Ebola vaccine.
What’s particularly disturbing about the present crisis, however, is that some infections could have been prevented. For years, the United States has been developing preventatives and treatments for Ebola, which would both provide defense if Ebola were used in warfare and reduce the spread of an outbreak of the disease. But, despite lobbying from scientists amid this latest outbreak, the drugs have not been put to the test...
“The whole thing has been very incompetently handled,” said Lansana Gberie, a historian from Sierra Leone. “If the government had quarantined this area” where the outbreak started, in the remote northeast, “they could have contained it. Instead they opened a treatment center in Kenema, a major population center.”
On PoliticoSL, a widely followed website in Sierra Leone, a journalist, Umaru Fofana, has written recently that “the country seems leaderless in the fight.” Now that cases have spread so widely, controlling the outbreak will be more costly and difficult.
Ebola virus infects a wide variety of human cell types, by using various attachment receptors to enter endosome structures inside cells. Upon entry, the virus binds to its cognate receptor, the Niemann-Pick C1 cholesterol transporter protein inside the late endosomes, fuses with the endosomal membrane, and thus enters the cytoplasm. It replicates, buds out of the cell, and the cycle repeats itself. The virus shuts down several of the host’s immune system defenses, and thus gains an upper hand.
...
NanoViricides, Inc. now has its own drug manufacturing facility that is capable of producing sufficient quantities of an anti-Ebola drug after it is developed, for combating Ebola epidemics.
NanoViricides has developed additional novel drug candidates against Ebola using its nanoviricide drug process. A nanoviricide drug consists of the combination of a virus-binding ligand that mimics the native receptor on the host cell and a backbone polymer that makes the drug look like the host cell surface to the virus.
“We believe the new anti-Ebola ligands should make the new drug candidates substantially superior to our older ones, based on the molecular modeling studies we have conducted using the structural information of interaction of Ebola virus glycoprotein with its cellular receptor Niemann-Pick C1 (NPC) protein,” Anil Diwan, the president of NanoViricides, said. “We believe that Ebola virus will not be able to avoid our drug candidates in spite of mutations, because we are mimicking NPC1, the receptor to which the virus must bind in order to infect the host cell. Of course, we must await results from actual cell culture and animal testing to further develop these candidates.”
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
There will always be a need and demand for vaccines, but many people won't use them. We know this from experience. However, the existence of a nanoviricide treatment for a virus-borne illness means that treatment can begin after symptoms for influenza or some other virus-borne disease appear.
Based on animal studies, we believe that virus populations will be so reduced that symptoms would disappear in only a few hours. Immunity, however, would develop in the normal 21-day period so that the patient could not get the same infection again.
Some viruses-notably hepatitis, HIV, and herpes-might not be cleared entirely from the system because they hide inside various tissues. I believe, however, that symptoms will disappear, and the patient would not be contagious. When the virus emerges, however, it will encounter nanoviricides if they are in the patient's system. Over time, there is a strong possibility that each reemergence will be smaller than the last, until the disease is gone. ~Mauldin Economics - Build Transformational Wealth from Three Tiny Companies

In Italy, the famous Fashion designers Domenico Dolce and Stefano Gabbana were sentenced to 18 months in prison this week for keeping hundreds of millions of euros from Italian tax authorities offshore. When I say there is a worldwide hunt for capital that is destroying the world economy - this is NO JOKE! Politicians have spent whatever they like and then imprison citizens for not handing over whatever they demand. This is not democracy - it is totalitarianism. They have NO right to take money from people and criminalize refusing to pay unreasonable sums. People come together to form societies because a synergy emerges that creates an economy from the Invisible Hand that is larger than the sum of the parts. It has historically be VOLUNTARY. Government has abused its power and looks upon the people as a herd of unwashed wild animals for them to drive in whatever direction they desire for their own self-interest. They retain that power by preaching to the ignorant that they are NEVER the problem, it is always the "rich" who refuse to turnover everything they own so politicians can live high and mighty. ~ Martin Armstrong
How is it possible you forgot that the accusers of ENRON mega house of cards showed up in the U.S. Court of Law to face the accused? Where is NanoViricides, Inc. dirty, low down, "perp gang", pseudo named Pump Terminator? Hit and run accident?
============================================
In this lecture, tech investor Juan Enriquez explains how the FDA's extreme risk aversion hurts us, and why that behaviour is our fault.
Another_voice_2, keep winning tennis matches for now. It will be just weeks and EbolaCide II will demonstrate what it can do, fast acting, destroy Ebola virus machinery, in the trillions, in a matter of hours. I do have confidence in the genius of Dr. Anil Diwan.
My post this morning began with Leifsmith but I'm sure he knows it was directed to the "big government" mouthpiece that wrote the article, claiming it is not "big government's" fault. But if our 'big government" is betting on Big Pharma vaccine mules you bet I am going to fault the bunch of tin horn dictators.
If the "wild bunch" is hoofs and all into our economy, with all sorts of laws and regulations, you bet I am going to fault the control freaks that came up with the ill conceived laws.
Technology companies were hit hardest. Apple dropped nearly 4 percent following its announcement late Wednesday that it had pulled a software update which prevented users from making phone calls. Others complained that they bent their new iPhones by sitting on them. Apple lost $3.88 to $97.87 in heavy trading.
"Two economic reports out Thursday were little help. Claims for unemployment benefits crept up last week. But the less volatile four-week average fell. A separate report said businesses orders for equipment plunged last month, mainly a result of falling orders for commercial aircraft.
Why has the WHO been unable to assume an assertive lead role? Budget cuts?
The WHO's response has been abysmal. It's just shameful. Yes, WHO is a financial shadow of its former self, operating on an inflation-adjusted budget that is less than it had in 1990. The WHO's legislative body, the World Health Assembly, has consistently voted to downgrade the institution's capacity to deal with outbreaks and infectious disease in favor of increasing commitment to noncommunicable disease programs such as cancer and heart disease.
When asked "Where is the WHO?" Margaret Chan, the director-general, said, "We are just a normative agency." In other words, the WHO's job was to set standards for health, but not to actually intervene. That represented a very significant change over prior WHO policies, and in this case, since [no one intervened] except an NGO, MSF, it put the world in a very awkward situation.
Now the WHO is racing to catch up, to have some leadership role, and there are many institutions that want the WHO in the driver's seat. But institutionally, and in terms of personnel, skills, and budget, it's not the kind of WHO most people imagine. The moment for the WHO to claim a [credible] leadership role has already passed. I'm afraid [it] looks incompetent.

Is there a threat, as some experts have said, of the virus mutating into an airborne contagion?
It would be a pretty profound leap in mutation. To put this in perspective, we know the virus has already made more than 300 mutations in the last five months. And that's significant. However, most of the changes have made no difference whatsoever in the ability of the virus to perform its biological function [in regard to its transmissibility].
I'm less worried about the virus becoming airborne than I am about it being able to outmaneuver any vaccine we throw at it. My concern is that Ebola is mutating and that, even if we find a vaccine, it may have very limited efficacy because of the virus's capacity to constantly change its code.
"I do have reason to distrust the government."
"Why did our government imposed finding the toxic dose on our "agnostic to the host", low toxicity, nanoviricides/FluCide?"
One does not have to be a staunch supporter of government regulation to see the justification for safety standards.
The traditional comparison is unavoidable.
Health Effects
Marijuana:
Heightened sensory perception; euphoria, followed by drowsiness/relaxation; impaired short-term memory, attention, judgment, coordination and balance; increased heart rate; increased appetite
Alcohol:
Alcohol affects every organ in the drinker's body and can damage a developing fetus. Intoxication can impair brain function and motor skills; heavy use can increase risk of certain cancers, stroke, and liver disease. Alcoholism or alcohol dependence is a diagnosable disease characterized by a strong craving for alcohol, and/or continued use despite harm or personal injury. Alcohol abuse, which can lead to alcoholism, is a pattern of drinking that results in harm to one's health, interpersonal relationships, or ability to work.
www.drugabuse.gov/drugs-abuse
"When our government had the chance to call on Pump Terminator and ask them to come out of the shadows, face their accused, they failed to uphold the law. "
Surely the company knows who Pump Terminator is or they shouldn't have said this in a court filing:
"...he is a devious market manipulator who made upward of $1,000,000 by selling NNVC stock short when the share price plummeted following publication of his derogatory comments."
If they know who he is they can carry out this threat, which appears to have been an idle one:
"NanoViricides, Inc. is fully prepared to seek appropriate legal action against parties involved in this libelous attack."
Leifsmith, I do have reason to distrust the government.
1- Pump Terminator attacked our small biotech company and caused the pps to tumble. Damage was done to our small company, our property. When our government had the chance to call on Pump Terminator and ask them to come out of the shadows, face their accused, they failed to uphold the law. If you damage private property you should have your days in court and face the music.
2- If the law/regulations must apply equally to all, and they should, why is toxic Cannabis given a pass and is not required to have clinical trials? Everyone else has to spend time and money on clinical trials but not a drug pushing and lawless drug cartel? Trust our government and unelected FDA officials?
Equality before the law, also known as equality under the law, equality in the eyes of the law, or legal equality, is the principle under which all people are subject to the same laws of justice (due process). ~ Wikipedia
“ When you look at 19th century America or 18th and 16th century Europe, all of a sudden it’ll become clearer that … the thing that broke the back of poverty and privilege in developed countries in the past was when property rights came around and destroyed feudal title.” ~ Hernando de Soto
Dr. Rick Sacra started receiving TKM-Ebola, manufactured by Canadian drug firm Tekmira, on Sept. 5, the day he arrived at Nebraska Medical Center in Omaha, hospital officials said in a statement. The drug, which is designed to stop the Ebola virus from replicating, was administered to Sacra over seven days.
Sacra had also received blood plasma transfusions from Dr. Kent Brantly, a fellow missionary who has recovered from the disease and was discharged from Emory University Hospital last month. The World Health Organization earlier this month recommended blood transfusions from survivors as a way of boosting an Ebola patient's immune system by pumping in antibodies that successfully fought the virus.
Doctors at the hospital, who last week said they expect Sacra to make a full recovery, warned Tuesday against assuming the drug is a “magic bullet.”
“We don’t know if it was Dr. Sacra’s own immune system, the supportive therapy we provided, the blood transfusion from Dr. Brantly, TKM-Ebola or a combination of all these factors that helped Dr. Sacra recover,” said Dr. Angela Hewett, associate medical director of the hospital’s biocontainment unit.
"In frenzied excitement, he eats up the ground. He paws fiercely, rejoicing in his strength and charges into the fray, afraid of nothing, when the trumpet sounds." (Excerpt from Job 39: 21-25)
NanoViricides, Inc. is not just a biotech healthcare stock. It is a small biotech company with great science. I call it Bones "23rd century science" today! Science that could bring Ebola virus to a screaching halt! Why? Because when nanoviricides enters the host it will encounter virus structures in the billions, perhaps trillions, destroy them and stop the menacing virus advance in a matter hours. This quickly translates to alleviation from pain and suffering to the host of Ebola virus, or any other virus. Whatever is left of the virus will be addressed by the host's immune system. Soon to be confirmed, once again, we are makers of low toxity antiviral therapeutic drugs. Unlike other antiviral drug makers, we will soon have a new validated 5kg CGMP Pilot Plant, capable of producing 1000 doses of EbolaCide II to address the killing Ebola virus. We are agnostic to the host, we only care to find and destroy the life threatening virus.
The whole world will soon be in frenzied excitement because the people of NanoViricides, Inc. was there (the 10,000+ and Institutional Investors), when most needed, to prevent a catastrophy of epic proportions. We are not a horse in a horse race but there are some similarities with our small biotech company, in a race: We charge into the fray, afraid of nothing because we know our science and how effective it is. NanoViricides, Inc. paws fiercely, rejoicing in its strength with approximately $41 million in cash and other instruments, enough to move forward with EbolaCide II, FluCide and DengueCide, our triple crown! When the trumpet sounds is because we smell the war from afar, and we will soon be ready to destroy viruses and their machinery, in the trillions, in a matter of hours.
Enough said, fired up, "...lets roll"!!!
==============================================
"In frenzied excitement, he eats up the ground. He paws fiercely, rejoicing in his strength and charges into the fray, afraid of nothing, when the trumpet sounds." (Excerpt from Job 39: 21-25)
Fresh Graves Point to Undercount of Ebola Toll
By ADAM NOSSITERSEPT. 22, 2014
“We will need much more space,” said James C. O. Hamilton, the chief gravedigger, as a colleague cleared the bush with his machete.
The Ebola epidemic is spreading rapidly in Sierra Leone’s densely packed capital — and it may already be far worse than the authorities acknowledge.

“I’m working with the burial team, and the first question I ask them is, ‘Are they Ebola-positive?’ ” said Mr. Parker, adding that the figures were based on medical certificates that he had seen himself. The deaths are carefully recorded by name and date in a notebook headed “Ebola Burials.”
The majority of the recent deaths recorded at the cemetery were young people — young adults, people in early middle age, or children — with very few elderly people on the list. Several of the deaths also occurred in a concentrated area, sometimes in the same house, suggesting that a virulent infection had struck.
“The way my Mummy died was pathetic,” said the son, Michael Foday, clearly frustrated by the quarantine. “How do you expect us to get food?”

There are those who complain that the tox studies are delayed. I don't believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014
Nanoviricides says early results indicate that FluCide® was extremely safe in a non-GLP small animal tox study
WEST HAVEN, CONNECTICUT -- Monday, October 21st, 2013 -- NanoViricides, Inc. (NYSE MKT: NNVC) (the "Company"), reports that its optimized injectable FluCide® drug candidate was found to be well tolerated in a non-GLP small animal safety/toxicology study. This study is an important step in the drug development pathway for FluCide. This study will provide guidance for the IND-enabling GLP safety and toxicology study. The Company previously had a pre-IND meeting with the U.S. FDA to discuss and receive guidance on the FluCide drug development pathway. This non-GLP safety and toxicology study was begun in late September at KARD Scientific in Massachusetts.
Systemic toxicity and toxicokinetics of a high dose of polyethylene glycol 400 in dogs following intravenous injection.
Abstract
Polyethylene glycol 400 (PEG-400) has been used in injections. However, limited data are available concerning the toxicity of a high dose of PEG-400 following intravenous (i.v.) injection. The aim of the present study was to estimate the systemic toxicity and toxicokinetics of a high dose of PEG-400 in dogs following i.v. injection. Twenty-four dogs were divided into four groups: a control group receiving normal saline and three test groups receiving 4.23, 6.34, and 8.45 g/kg of PEG-400, respectively, by i.v. injection once a day for 30 days. The repeated-dose toxicity of PEG-400 was assessed. Toxicokinetic parameters of PEG-400 in dogs were estimated on days 1 and 30. Dry mouth and dry nasal mucus membrane were observed in dogs treated with 6.34 and 8.45 g/kg of PEG-400. Cloudy swelling of kidney cell and increased glomerular volume were observed in dogs treated with 8.45 g/kg of PEG-400 when the animals were sacrificed 24 hours after the last injection. No significant histological changes were found 21 days later. Repeated dosing did not affect the toxicokinetic profile of PEG-400 in dogs. This study has shown that the toxicity of a high dose of PEG-400 following repeated intravenous injections is low, and alterations produced are reversible.
The non-GLP toleration/dose range finding study: design and methodology used in an early toxicology screening program
A major directive of Pharmaceutical Research and Development (R&D) is to efficiently advance potential new chemical entities (NCEs) from the Discovery therapeutic area into Global Preclinical Development (GPCD), where a safety profile can be established. To facilitate the transition a comprehensive toxicity evaluation is required. In order to support both the R&D Discovery teams and GPCD, investigative (non-GLP) tolerance/dose range finding studies are conducted. These studies are designed to provide a quality toxicological and toxicokinetic assessment of potential NCEs early in the drug development process. During tolerance evaluations, compounds are first assessed in a single dose escalation (SDE) phase where rodents (or canines) receive a single dose anticipated to achieve relevant multiples of the efficacious dose. Data from this phase evaluates NCE absorption, and assists in estimating the maximum tolerated dose for a single administration and establish doses for a repeat dose (RD) phase. Data from the RD phase are used to determine potential target tissues of toxicity and also select doses for future GLP Toxicology studies. Thus, a rapid assessment of the toxicological profile of the NCE can be made to establish initial safety facilitating conduct of subsequent regulatory Toxicological studies and potentially earlier entry into clinical trials.
NanoViricides, Inc. Reports Excellent Safety Profile of Its Broad-Spectrum anti-Influenza Drug Candidate, FluCide™ , in a Non-GLP Study
WEST HAVEN, CONNECTICUT -- Monday, December 2, 2013 -- NanoViricides, Inc. (NYSE MKT: NNVC) reports that it has received results of detailed lab analysis studies from the initial non-GLP toxicology studies of intravenously administered FluCide™. No overt adverse safety and toxicology effects were observed in this study of the Company's optimized FluCide broad-spectrum anti-influenza drug candidate, even at the maximum feasible dose level. These results are consistent with the preliminary findings of this study that the Company has previously reported, and provide greater details of the safety of FluCide.
The study was an important step in the progress of the FluCide drug gaining approval from the U.S. Food and Drug Administration. The study was conducted at KARD Scientific in Massachusetts.
During the course of the study, laboratory mice were treated with the maximum dose of FluCide within a one hour timeframe. The mice remained healthy throughout the course of the study. Even at the highest possible dose, no visible signs of toxicity were observed.
Nano Viricides reported no safety or toxicology concerns in the mice. The research team found the injectable FluCide drug candidate to be highly effective in non-GLP small animal models against an array of influenza A viral strains. Nano Viricides will soon receive the complete report of all clinical and laboratory data, including blood chemistry and histopathology, to confirm its findings.
Nano Viricides is currently reconfiguring its studies to reproduce the study with larger quantities of the FluCide drug candidate. It is also continuing research toward two injectable FluCide administration options to create doses appropriate for both hospitalized patients with severe influenza and outpatients with influenza infections...
Dr. Diwan further discussed logistical issues – that influenza vaccines in particular rely heavily on highly specialized antibodies and tight quality control standards for manufacturing. As these antibodies are highly specific to a particular strain, tailoring an effective vaccine to a specific strain is a very expensive – and some would argue, quixotic challenge – that brings minimal benefit and renders many of the vaccines useless.
For example, a vaccine being developed must demonstrate efficacy against four “type zero” strains and four “type one” strains. According to Dr. Diwan, this is easier said than done: each type zero and type one is different, and thus the likelihood to demonstrate success among all eight is rare.
Dr. Diwan estimated the cost of influenza vaccine R&D and production to be between $1.7-2 BN per candidate over 10 years.
“If you are lucky, one passes”, Dr. Diwan said, referring to one of only two commercially available influenza treatments, but more importantly, to the countless failed treatments over the years.
Failed, or ‘useless’ does not simply mean therapies that do not reach market. Many vaccines produced are purchased for government stockpile, and go unused – only 2-3% of vaccines purchased are actually used, according to Dr. Diwan.
Clearly, there exists a need for a new approach that can be more easily adapted to new strains.
"We are doing all of the preparatory pre-tox work now and the formal tox studies by BASI will start as soon as the pre-tox work is done and an [sic] sufficient amount of material is available from the existing lab ~ Dr. Eugene Seymour, CEO of NanoViricides, Inc.
AZ ABST Case Study
'ABST' stands for Automated Blood Sampler and Telemetry, the combined solution for integrative pharmacology. Through a collaborative effort between BASi, the maker of the Culex® Automated In Vivo Sampling System, and Data Sciences International (DSI), the gold standard in physiologic monitoring, it is now possible to collect more data from individual animals than ever before. ABST can be used to measure multiple variables in a single animal including heart rate, blood pressure, temperature, blood, urine and feces, dialysates, locomotion, activity and various biopotentials e.g. ECG, EEG, EMG, or ERG. Protocols requiring automated infusion can be easily incorporated and controlled with the BASi system. Contact BASi for further details.
Save Money
This combined system makes it possible to collect multiple streams of data simultaneously from individual animals. The ability to collect blood at the same time as other safety data eliminates the use of satellite groups. Subjects can be re-used after a washout period, leading to further reduction in the use of animals. Since the system is fully automated, it is possible to reduce staff hours compared to traditional methods. Additional money savings arise thanks to better data; the ABST makes it possible to make earlier go/no-go decisions so that money isn’t wasted on compounds that will not make it to market.
We're estimating a June start though that could go either way by 2-3 months depending on both BASI's schedule and ours." ~ Dr. E. Seymour, CEO NanoViricides, Inc. --- Apr 2014
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014
"...by the way, I'm sure that when you think human trials for drugs you think of hundreds of millions of dollars and years of time, well in this case because the disease only lasts a week, two weeks,...that it is possible to complete human trials in the space of a few short months...four parts to the human trials" ~ Dr. Eugene Seymour, CEO Nanoviricides, Inc.
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. Thirteen weeks or less to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is about right. Moreover, highly toxic oncological drugs take 9 months on average.~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
Definition of Integrative and Organ Systems Pharmacology: "Pharmacological research using in vivo animal models or substantially intact organ systems that are able to display the integrated responses characteristic of the living organism that result from complex interactions between molecules, cells, and tissues."
AZ ABST Case Study
'ABST' stands for Automated Blood Sampler and Telemetry, the combined solution for integrative pharmacology. Through a collaborative effort between BASi, the maker of the Culex® Automated In Vivo Sampling System, and Data Sciences International (DSI), the gold standard in physiologic monitoring, it is now possible to collect more data from individual animals than ever before. ABST can be used to measure multiple variables in a single animal including heart rate, blood pressure, temperature, blood, urine and feces, dialysates, locomotion, activity and various biopotentials e.g. ECG, EEG, EMG, or ERG. Protocols requiring automated infusion can be easily incorporated and controlled with the BASi system. Contact BASi for further details.
Save Money
This combined system makes it possible to collect multiple streams of data simultaneously from individual animals. The ability to collect blood at the same time as other safety data eliminates the use of satellite groups. Subjects can be re-used after a washout period, leading to further reduction in the use of animals. Since the system is fully automated, it is possible to reduce staff hours compared to traditional methods. Additional money savings arise thanks to better data; the ABST makes it possible to make earlier go/no-go decisions so that money isn’t wasted on compounds that will not make it to market.
Save Time
The ABST system offers full automation for dosing, blood sampling, and the collection of safety data. This allows technicians to use time effectively, for example, an ABST experiment can commence in the morning, followed by a more time consuming manual PK study. Taking this approach can make your staff twice as effective as before. Elimination of satellite groups further reduces the amount of time that is spent on each compound. Finally, early decisions can be made with high quality data, reducing the overall time spent on non-viable compounds.
Gather Better Data
With satellite groups it is difficult to ensure that all study parameters are the same for each group. Indeed, manual pharmacokinetic sampling is often very stressful for research animals, while great care is taken to minimize human intervention during the collection of safety data. The ability to eliminate satellite groups with ABST allows for better control over study variables. Data collection is further refined by achieving a within subject experimental design and capturing inter-animal variability. Additionally, the ABST makes it possible to compare information across organ systems to determine time and exposure-dependent effects. All of these features make it possible to form early and informed decisions during drug development.
With the ABST, it is possible to simultaneously collect:
Urine/Feces for:
DMPK
Metabonomics
Clinical Pathology
Terminal tissue collections for:
Clinical Pathology
Molecular Toxicology
Other biofluids:
Bile
Dialysates
Telemetry Data including:
Heart Rate
Blood Pressure
Temperature
Locomotion
EMG, ERG, ECG or EEG
Serum/Plasma for:
DMPK
Metabonomics
Biomarkers
Clinical Pathology

http://www.datasci.com/solutions/integrated-pharmacology/automated-blood-sampler-and-telemetry
Other related source:
A modern in vivo pharmacokinetic paradigm: combining snapshot, rapid and full PK approaches to optimize and expedite early drug discovery.
http://www.ncbi.nlm.nih.gov/pubmed/22982770
========================================
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date.
There will always be a need and demand for vaccines, but many people won’t use them. We know this from experience. However, the existence of a nanoviricide treatment for a virus-borne illness means that treatment can begin after symptoms for influenza or some other virus-borne disease appear.
Based on animal studies, we believe that virus populations will be so reduced that symptoms would disappear in only a few hours. Immunity, however, would develop in the normal 21-day period so that the patient could not get the same infection again.
Some viruses—notably hepatitis, HIV, and herpes—might not be cleared entirely from the system because they hide inside various tissues. I believe, however, that symptoms will disappear, and the patient would not be contagious. When the virus emerges, however, it will encounter nanoviricides if they are in the patient’s system. Over time, there is a strong possibility that each reemergence will be smaller than the last, until the disease is gone. ~Mauldin Economics
Our small biotech company/technology is one that will be stepping on big toes of some big players in Big Pharma. We really don't know who is who posting on this board however, I do know who Scott Gotliebb, MD is. He was once the no. 2 at the FDA and he recently wrote the following when it became evident that Ebola was spinning out of control in West Africa:
The World Health Organization said last Thursday that the scope of the Ebola outbreak may be vastly underestimated, and will grow worse before they are able to get the epidemic under control.
Amid these dire warnings, and facing a virus with no proven remedies, treatments, or vaccines, a panel of WHO ethicists approved the use of experimental therapies in stricken patients.
It was the right decision for a host of reasons. In fact, it shouldn't have been ethicists who reached such a conclusion. Drug regulators should have taken this decision.
Patients facing a deadly virus with no proven treatments deserve access to experimental drugs, so long as they provide informed consent to receive them. Such forward deployed drugs should already have established basic proof of safety, and there should be a plausible reason to believe that these agents could also prove effective.
“We definitely would like to ramp up to have an impact on the Ebola epidemic,” Dr. Whaley said. But he added, “We’re not decision makers on many of these issues. There are regulatory and legal issues that have to be addressed.”
The drug had never before been tried in people, though it and some predecessor drugs had been tested in monkeys, showing some effectiveness. Mapp was only now gearing up to start the larger animal toxicity studies typically needed before testing it in humans, with an eye to doing the first human safety studies in healthy volunteers next year. For that reason there were very few doses available when the Ebola outbreak started.
Now the company is trying to move faster and it is likely the drug will be tested in patients rather than healthy volunteers.
The only way we're going to find out for sure if these experimental Ebola drugs are effective is to use them in sick patients. Given the state of science, this initial use is going to based on little more than information about their effectiveness in animals.
FDA issued a so-called "animal rule" almost a decade ago (amid fears of bioterrorism after the deadly anthrax attacks) outlining how a drug could be approved on the basis of only efficacy studies in animals. It was rightly acknowledged that animal testing alone would have to form the basis of approval when we were dealing with therapeutics that targeted rare and deadly diseases that could be used as weapons.
You are citing the FDA rule/regulations. Fine. Should FDA allow toxic Cannabis go to market without clinical trials? The law/regulations should apply equally to all companies. Why the exception, the double-standard, with toxic Cannabis? We are a small biotech company with low toxicity, fast acting, effective nanoviricides. Six thousand animal subjects survived lethal doses viruses like Influenza, Dengue, etc., thanks to nanoviricides.
Why does the FDA put hurdles on our small biotech company??? Why find the toxic dose? FDA could have requested the toxic dose for later and that would have been ok. All of this while you see vaccine makers getting all kinds of taxpayers money and I am referring to big cap companies in comparison to our small biotech. And how generous of some companies/governments to donate vaccine doses that were made possible/manufactured with taxpayers money. It is as if they were saying, "...use those vaccine doses quickly because if the virus mutates they will become worthless...". And whatever happened with Tamiflu data? Billions were spent by governments just to find out later that the vaccine was marginally effective.
The PEG molecule is common to all nanoviricides, yes? The difference among nanoviricides What products are out there in the market with PEG that have been, by FDA regulations, required to undergo clinical studies. What were their results and are they in the market today? Perhaps it may be easier to cite the failures, if any.
If EbolaCide tests in BSL-4 facility and if found to be highly efficient, why not employ it right away to fight Ebola? How far did Zmapp go, in meeting goals according to FDA regulations, before it was employed in the US and in Liberia, Africa?
If we are not delayed for FluCide, we are not delayed for 2015. Why would any one say "not delayed" and skip the entire 2015? You said it in one of your posts, time will wait for no one, not even NNVC.
Thirteen weeks (13 weeks)? We are advancing with FluCide in paralell to EbolaCide. Are FluCide GLP full tox studies worth anything in relation to EbolaCide and FDA regulations/requirements?
I don't have the time to post any other questions this morning. Perhaps in the evening.
Here it is nanopatent, niemann-pick c1
Search with the NPC1 mimic (ligand), encapsulate the filovirus Glycoproteins(GP) and destroy most, if not all, in the trillions, in a matter of hours.
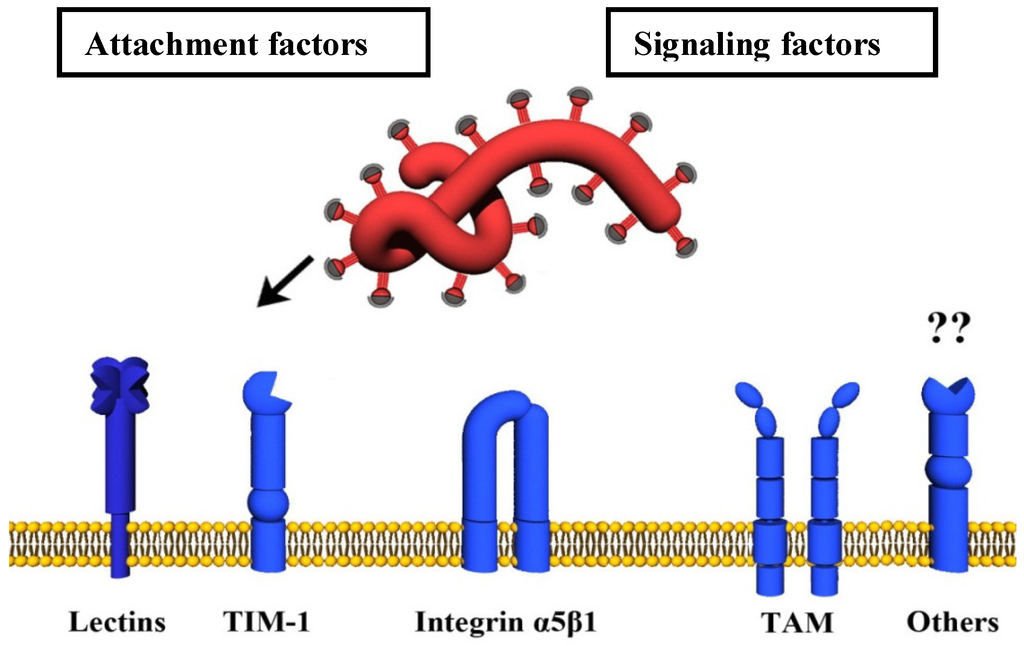
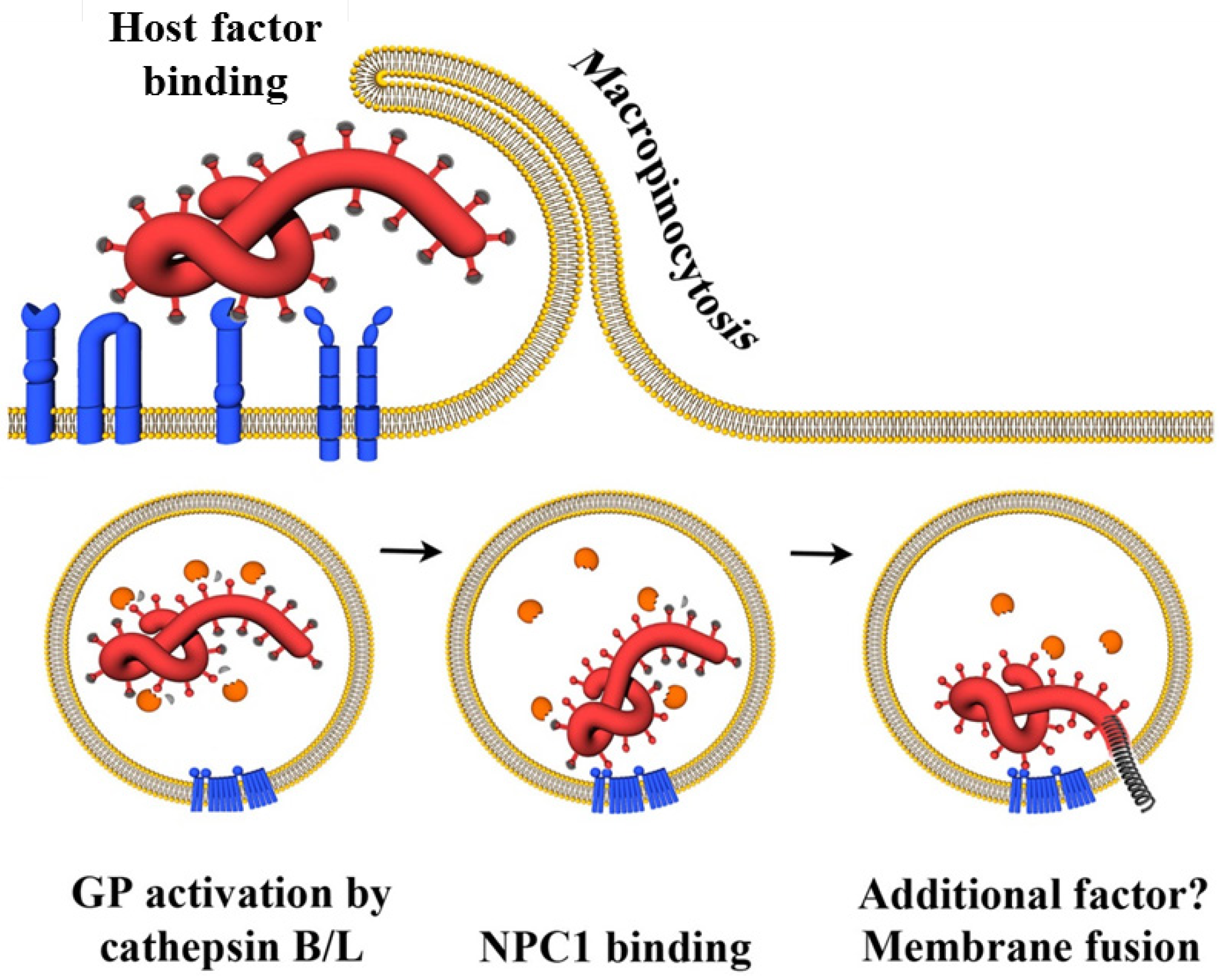
http://www.mdpi.com/1999-4915/4/12/3336/htm
So, we do have news coming Monday? Perhaps on low toxicity, fast acting and effective FluCide or more on the low toxicity EbolaCide (that can be dramatically improved) that could soon be fast tracked to Clinical Trials. Of course, synthesis completion, in about 3 weeks (from date started), comes first.
...we feel that we can dramatically improve the efficacy with our new improved techniques.~ Dr. E. Seymour, CEO NanoViricides, Inc.

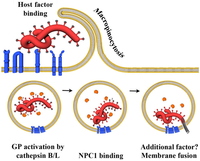
...treated with class 2 amphiphiles to mimic NPC cells

Echo20, if Gilead Sofosbuvir is worth $84,000 for a 12 month treatment period, once we get HCVCide to market it could be licenced to Gilead or other company, maybe. What if the 12 month treatment period cost were to be slashed down to 1/2 or a 1/3 of what it is today?
NanoViricides, Inc. has transformational technology that could reduce the cost of healthcare, the cost of health insurance, etc., slashing them dramatically.
============================================
Judge orders Internet commenter's name revealed
By Joseph A. Slobodzian, Inquirer Staff Writer
Posted: March 09, 2014
A warning to people who post comments online: Anonymous is not forever.
A Philadelphia judge has ordered the owners of Philly.com - who also own The Inquirer and the Philadelphia Daily News - to disclose the identity of a person who posted a comment online.
The ruling came in a defamation suit filed by John J. Dougherty, the powerful head of Local 98 of the International Brotherhood of Electrical Workers.
FluCide GLP full tox study and BASi
BASi has the Culex-L (large animals) and Culex ABC (for small animals).
From the International Society of Study of Xenobiotics
The aim of the current research was to demonstrate that pharmacokinetic data in plasma and Dried Blood Spots (DBS) can yield equivalent results. Four male beagle dogs were implanted with a subcutaneous access port (VAP) with a catheter inserted into the femoral vein. After surgery the dogs were allowed to recover 7 days. During the recovery period the dogs were acclimatized to metabolism cages and dog jackets for 2 hours a day. On the study start day the dogs were placed in jackets and individual metabolism cages. The dogs were connected to the Culex-L automated blood collector (Culex-L ABC), a computer-controlled robotic blood collection system, and dosed once orally with 50 mg/kg of acetaminophen. Samples of 600 ?L whole blood were collected into chilled vials prior to dosing and at 0.5, 1, 2, 4, 6 and 8 hours post dosing. Within two hours after collection, four 20 ?L spots of whole blood from each time point were pipetted onto Culex DBS collection cards. The DBS cards were allowed to air dry overnight at room temperature and stored at room temperature in a sealed bag containing a desiccant. The remaining blood was processed to plasma and frozen. Twenty eight whole blood samples were collected, seven from each dog. Twelve samples were several minutes later than scheduled (from 5 to 14min) due to either a software problem, or dislodgement of the needle accessing the VAP. Samples were analyzed by liquid-liquid extraction with separation and detection by LC-MS/MS. The pharmacokinetic profiles from each sampling method indicate that there is no difference between the sampling techniques. The mean concentrations from both sampling methods across the time points were indistinguishable. DBS methods offer many advantages for the collection, storage and shipping of blood samples for drug analysis. Smaller volume of sample is required for analysis allowing more frequent and multiple samples compared to a traditional PK method. Processing, handling, and shipping of DBS samples is less time consuming, less laborious, and incurs lower cost than frozen plasma. Automation of the blood sampling and delivery process ensures consistent volume, quality and location of DBS samples on the collection cards. It can also reduce labeling and handling of the cards.

Dr. Eugene Seymour, 9/27/11
...This is wonderful news for us, particularly with regard to the flu since I feel we can cut 5-6 days from the average length of hospitalization with FluCide!!! So whatever we charge, it's irrelevant because of the hospital days saved. For a sick elderly patient with the flu, I would imagine that the daily hospital costs in the ICU will exceed $5000 per day. Saving $25K while spending $2K for our drug is a no brainer (drug charges for the intravenous preparation for extremely ill patients and daily hospital costs are just estimates for the purpose of comparisons. Outpatient drug cost would be 10% of that figure {all estimates})
Hope that I have answered everyone's concerns
Sincerely,
EUGENE SEYMOUR, MD, MPH
Chief Executive Officer
NANOVIRICIDES, INC
Nanotechnology-based targeted anti-viral therapeutics
www.nanoviricides.com
eugene@nanoviricides.com
Important flu statistics, based on the best available data.
...
Percentage of the U.S. population that will get the flu, on average, each year: between 5% and 20%.
Number of Americans hospitalized each year because of flu complications: 200,000, on average.
The number of people who die each year from flu-related causes in the U.S.: ranges from 3,000 to 49,000.
In the U.S., influenza and pneumonia were the eighth leading cause of death in males in 2009.
Number of flu vaccine doses available in the U.S. for the 2013-2014 flu season: Between 135 and 139 million.
The CDC recommends that everyone over six months of age get a flu vaccine as soon as it's available.
Flu activity usually peaks in January and February.
...
It takes about two weeks after vaccination for an adult to develop antibodies against the flu.
The typical incubation period for the flu is one to four days. Adults can be contagious from the day before symptoms begin through five to 10 days after the illness starts.
A regular case of the flu typically resolves after three to seven days for the majority of people, although cough and fatigue can persist for more than two weeks.

thefamilyman, from an article 2008...
In 2007, the U.S. National Academy of Sciences released a report, Toxicity Testing in the 21st Century: A Vision and a Strategy, that envisions a not-so-distant future in which virtually all routine toxicity testing would be conducted in human cells or cell lines in vitro by evaluating cellular responses in a suite of toxicity pathway assays using high-throughput tests, that could be implemented with robotic assistance. Risk assessment based on results of these types of tests would shift towards the avoidance of significant perturbations of these pathways in exposed human populations. Dose-response modeling of perturbations of pathway function would be organized around computational systems biology models of the circuitry underlying each toxicity pathway. In vitro to in vivo extrapolations would rely on pharmacokinetic models to predict human blood and tissue concentrations under specific exposure conditions. All of the scientific tools needed to affect these changes in toxicity testing practices are either currently available or in an advanced state of development. A broad scientific discussion of this new vision for the future of toxicity testing is needed to motivate a departure from the traditional high dose animal-based toxicological tests, with its attendant challenges for dose and species extrapolation, towards a new approach more firmly grounded in human biology. The present paper, and invited commentaries on the report that will appear in Toxicological Sciences over the next year, are intended to initiate a dialog to identify challenges in implementing the vision and address obstacles to change.
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. One year to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is an absurd claim. Even highly toxic oncological drugs take 9 months on average. ~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
FDA issued a so-called "animal rule" almost a decade ago (amid fears of bioterrorism after the deadly anthrax attacks) outlining how a drug could be approved on the basis of only efficacy studies in animals. It was rightly acknowledged that animal testing alone would have to form the basis of approval when we were dealing with therapeutics that targeted rare and deadly diseases that could be used as weapons.
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
...vaccines usually fail with the emergence of mutated viruses. And when a person develops a disease, we still need to have a treatment ~ Dr. Anil Diwan, President of NanoViricides, Inc.

Yes, EbolaCide II is bound to join the group of new drugs and vaccines to treat Ebola and possibly take the lead from Flucide in the race to FDA review/market (WHO review by November 2014). Thank you MDPH1.
Experts will be meeting this week at the World Health Organization (WHO) to discuss the role of new drugs and vaccines to help control the Ebola epidemic in West Africa. Last month, the WHO said that it is ethical to provide experimental drugs and vaccines for Ebola, but that there’s also a “moral duty” to conduct clinical trials of these experimental drugs and vaccines to determine whether they’re safe and effective. At the same time, a new study released last week shows that the Ebola virus is mutating rapidly, which could make it more transmissible or reduce the effectiveness of drugs and vaccines in the pipeline. ~ Reuters
...vaccines usually fail with the emergence of mutated viruses. And when a person develops a disease, we still need to have a treatment ~ Dr. Anil Diwan, President of NanoViricides, Inc.
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
There will always be a need and demand for vaccines, but many people won’t use them. We know this from experience. However, the existence of a nanoviricide treatment for a virus-borne illness means that treatment can begin after symptoms for influenza or some other virus-borne disease appear.
Based on animal studies, we believe that virus populations will be so reduced that symptoms would disappear in only a few hours. Immunity, however, would develop in the normal 21-day period so that the patient could not get the same infection again.
Some viruses—notably hepatitis, HIV, and herpes—might not be cleared entirely from the system because they hide inside various tissues. I believe, however, that symptoms will disappear, and the patient would not be contagious. When the virus emerges, however, it will encounter nanoviricides if they are in the patient’s system. Over time, there is a strong possibility that each reemergence will be smaller than the last, until the disease is gone.

cGMP Pilot Plant has been realized and scientists are racing to get it validated. The plant is critical for Clinical Trials in Australia (2015) and possibly EbolaCide/WHO fast tracked clinical trials this year. That cGMP plant is real and critical requirements to be considered as one of the treatments for deadly Ebola, provided we prove dramatic efficacy over other drugs/vaccines. We are a small biotech company now, not a pharmaceutical giant like for example, Glaxo Smith Kline or Merck, but I believe we will be. Save for yourself the shorts narrative. I am long on this small biotech company.
============================================
Stepping on some big toes...
There will always be a need and demand for vaccines, but many people won’t use them. We know this from experience. However, the existence of a nanoviricide treatment for a virus-borne illness means that treatment can begin after symptoms for influenza or some other virus-borne disease appear.
Based on animal studies, we believe that virus populations will be so reduced that symptoms would disappear in only a few hours. Immunity, however, would develop in the normal 21-day period so that the patient could not get the same infection again.
Some viruses—notably hepatitis, HIV, and herpes—might not be cleared entirely from the system because they hide inside various tissues. I believe, however, that symptoms will disappear, and the patient would not be contagious. When the virus emerges, however, it will encounter nanoviricides if they are in the patient’s system. Over time, there is a strong possibility that each reemergence will be smaller than the last, until the disease is gone.
Do you want the CEO to spell plan B because plan A (6 to 9 months) spells trouble for longs? Patience, I honestly believe that the "good news" are coming soon. The new cGMP Pilot Plant is a reality and will soon enough be validated. The new cGMP Pilot Plant is critical for Clinical Trials (2015) and possibly Ebolacide if efficacy is dramatically increased over that of vaccines.
On FluCide full tox study...start of plan B?...
"We are doing all of the preparatory pre-tox work now and the formal tox studies by BASI will start as soon as the pre-tox work is done and an [sic] sufficient amount of material is available from the existing lab
We're estimating a June start though that could go either way by 2-3 months depending on both BASI's schedule and ours." ~ Dr. E. Seymour, CEO NanoViricides, Inc. --- Apr 2014
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. One year to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is an absurd claim. Even highly toxic oncological drugs take 9 months on average. ~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
§ The new technology enables rapid drug development against an emerging virus, which would be important for global biosecurity against natural as well as man-made (bioterrorism) situations. It is possible to develop a research drug against a novel life-threatening viral disease within 3-6 weeks after the infection is found, i.e. as soon as an antibody from any animal source is available.~ Jain Report
A couple of months ago I spoke with a surgeon, briefly described the NanoViricides, Inc. technology and that they were going to Clinical Trials (2015). That was all, I didn't say buy some shares. Immediately he said, do they have a drug for HIV?
Impact of HIV and AIDS on Surgical Practice
...the largest study to audit the impact of HIV/AIDS in general surgical practice in the UK retrospectively. Surgery for HIV patients can be safely conducted with a low complication rate for the diagnostic and anorectal procedures that comprise the vast majority of surgery in HIV/AIDS patients. Medical treatment for patients with HIV/AIDS has developed dramatically over the last two decades. In parallel, this has resulted in a heavy, new and varied workload for general surgeons, who have also had to adapt in order to deal with the challenging spectrum of this disease.
...the lead anti-HIV candidate achieved anti-HIV efficacy equivalent to a HAART (highly active anti-retroviral therapy) triple drug cocktail in its recently completed animal study. Treatment with the lead anti-HIV nanoviricide reduced HIV levels and protected the human T cells (CD4+/CD8+) to the same extent as treatment with the HAART cocktail. The three drug HAART cocktail used for comparison in this study is one of the combination therapies recommended for initial therapy in humans. No evidence of drug toxicity was observed. The lead candidate will now undergo further optimization.
"...I was also impressed by their receiving orphan dPushing the rule of law asiderug designation for the dengue/dengue hemorrhagic fever drug candidate, the interest shown by the British Government Agency in a collaboration on MERS and H7N9, as well as the progress they’ve made in the construction of the new R&D and cGMP pilot production facility. Although I have known Dr. Seymour professionally for many years, I was introduced to the Company by my sister, Vivien Boniuk, MD, also an ophthalmologist, who had performed the animal studies on the Company’s drug candidates for adenovirus and herpes virus infections of the eye.~ Dr. Milton Boniuk, MD”
Rawnoc, are you implying projections (forecasts) cannot move and must happen within a year? Oh, I see. If projections were not exact in 2013 and moved, they wont be this time arround. Patience, it is 2014 an the state-of-the-art cGMP Pilot Plant is a reality, critical and operational. Scientists are inside racing to get it validated. Once validated it will be ready for Clinical Trials in Australia (2015) and possibly for fast tracked EbolaCide clinical trials. You do expect the cGMP Pilot Plant to be validated or do you have information to the contrary? Don't forget to reference/quote your sources. By the way, PT is not a reference/source if he/she/them are not willing to come out of the shadows.
Regarding FluCide GLP full tox study, perhaps you could persuade someone with credentials to refute in detail the following. Why not the full tox study in 13 weeks or less?
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. One year to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is an absurd claim. Even highly toxic oncological drugs take 9 months on average. ~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange

Thank you for the opportunity to re-post reasons why am I optimistic.
On FluCide full tox study...
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
The longest study in the nonclinical safety assessment is that for genotoxicity, the duration ranging from 13 to 26 weeks. Most studies take 13 weeks or less, including dose ranging studies. One year to complete tox studies for a drug that has shown no toxicity in the histology of 6,000 animals is an absurd claim. Even highly toxic oncological drugs take 9 months on average. ~ BigKahuna
Source: Nonclinical Safety Assessment: A Guide to International Pharmaceutical Regulations.
edited by William J. Brock, Kenneth L. Hastings, Kathy M. McGown
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
"We are doing all of the preparatory pre-tox work now and the formal tox studies by BASI will start as soon as the pre-tox work is done and an [sic] sufficient amount of material is available from the existing lab
We're estimating a June start though that could go either way by 2-3 months depending on both BASI's schedule and ours." ~ Dr. E. Seymour, CEO NanoViricides, Inc. --- Apr 2014
There are those who complain that the tox studies are delayed. I don’t believe that it is the case at all. Had our drug system not produced such amazing initial tox results, we would be well into the BASi studies at this time. The FDA mandates that we find the toxic dose. To do that requires an inordinate amount of material. When the amount of material needed is produced, the studies will start. I feel that it will be quite soon but I cannot, in good conscience, give a hard date. ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Jul 20, 2014
To nanopatent on BASi - FluCide full GLP tox study, "...ready to go as soon as we ship..." ~ Dr. E. Seymour, CEO of NanoViricides, Inc. --- Aug 26, 2014

"You can fool all of the people some of the time, and some of the people all of the time, but you can not fool all of the people all of the time." ~ Abraham Lincoln
Fast tracked clinical trials, efficacy and safety, possible and soon!
Experts will be meeting this week at the World Health Organization (WHO) to discuss the role of new drugs and vaccines to help control the Ebola epidemic in West Africa. Last month, the WHO said that it is ethical to provide experimental drugs and vaccines for Ebola, but that there’s also a “moral duty” to conduct clinical trials of these experimental drugs and vaccines to determine whether they’re safe and effective. At the same time, a new study released last week shows that the Ebola virus is mutating rapidly, which could make it more transmissible or reduce the effectiveness of drugs and vaccines in the pipeline.
A vaccine failure is when an organism develops a disease in spite of being vaccinated against it. Primary vaccine failure occurs when an organism's immune system does not produce enough antibodies when first vaccinated. Secondary vaccine failure occurs when enough antibodies are produced immediately after the vaccination, but the levels fall over time. While antibody levels always fall over time, this would be a more rapid loss of immunity than expected for that vaccine.
1. The vaccine virus may be a different serotype from the challenged virus.
2. Maternal antibody which protects neonates may interfere with vaccine presentation.
3. Some viruses notably Herpes Viruses are poorly neutralized and once established can spread between cells by fusion.
4. The vaccine virus may become denatured or inactivated during storage or administration.
5. The vaccine virus may be ineffective if it is manufactured incorrectly, for example containing insufficient antigen or live virus.
~ Wikipedia
Get the measles vaccine, and you won’t get the measles—or give it to anyone else. Right? Well, not always. A person fully vaccinated against measles has contracted the disease and passed it on to others. The startling case study contradicts received wisdom about the vaccine and suggests that a recent swell of measles outbreaks in developed nations could mean more illnesses even among the vaccinated.
When it comes to the measles vaccine, two shots are better than one. Most people in the United States are initially vaccinated against the virus shortly after their first birthday and return for a booster shot as a toddler. Less than 1% of people who get both shots will contract the potentially lethal skin and respiratory infection. And even if a fully vaccinated person does become infected—a rare situation known as “vaccine failure”—they weren’t thought to be contagious.
Whooping cough outbreaks have occurred with increasing frequency in the last year and are beginning to become major news items, as highlighted by major outbreaks on-going in California. In virtually every article, the disease is described as “vaccine preventable” and blame is often cast upon Jenny McCarthy and autism-crazed parents that frequently refuse vaccination. But the facts of the matter are not that simple.
Despite the recent press on the vaccine-autism debate, infant vaccination rates remain at or near all-time highs with 96.2% of infants getting at least three doses of DTaP in 2008 versus just 61% getting at least three doses of DTP in 1991 [1]. Furthermore, in recent outbreaks, a majority of affected have in fact been completely vaccinated (see NJ outbreak in which ALL affected children had been completely vaccinated). So given that the most at-risk population (children under 3 months) is not eligible for vaccination and their protection depends on the protection of older children, the real question to emerge from the recent pertussis outbreaks should be “Why is the pertussis vaccine no longer as effective or as long lasting as it once was?” People and public health agencies should be DEMANDING the answer to this question. They instead seem content with the possible addition of another booster of the DTaP to be given at seven years of age, thus increasing the number of doses of DTaP now given to American children to 6 and unnecessarily boosting tetanus and diphtheria in the bargain.
...vaccines usually fail with the emergence of mutated viruses. And when a person develops a disease, we still need to have a treatment
All we do is make the drug. The partners do the testing.
We will announce a partnership agreement when everything is signed.
There are very few BSL4 facilities in the world with whom we can work
I think that we explained in the PR why we feel that we can dramatically improve the efficacy with our new improved techniques.
We need a partner to try to get a piece of the available $100M EHO (sic WHO) grant.
This WILL NOT delay the start of the tox studies.
Sent from my iPhone
Eugene Seymour MD MPH
Chief Executive Officer
NanoViricides, Inc
eugene@nanoviricides.com
www.nanoviricides.com
310-486-5677
"NNVC" on the New York Stock Exchange
There will always be a need and demand for vaccines, but many people won’t use them. We know this from experience. However, the existence of a nanoviricide treatment for a virus-borne illness means that treatment can begin after symptoms for influenza or some other virus-borne disease appear.
Based on animal studies, we believe that virus populations will be so reduced that symptoms would disappear in only a few hours. Immunity, however, would develop in the normal 21-day period so that the patient could not get the same infection again.
Some viruses—notably hepatitis, HIV, and herpes—might not be cleared entirely from the system because they hide inside various tissues. I believe, however, that symptoms will disappear, and the patient would not be contagious. When the virus emerges, however, it will encounter nanoviricides if they are in the patient’s system. Over time, there is a strong possibility that each reemergence will be smaller than the last, until the disease is gone.

"You can fool all of the people some of the time, and some of the people all of the time, but you can not fool all of the people all of the time." ~ Abraham Lincoln
