Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
7-14-15 Qtly CC-Transcript, PR(Fins/Devs Q4FY15/fye4-30-15), updated Avid Revenues History Table By Quarter…
=> Total Revs May06-Apr15: $129.2mm/Avid + $24.1mm/Govt + $2.1mm/Lic. = $155.5mm
This large post has 3 sections:
I. 7-14-15 Q4/FY15 Qtly. Earnings Conf. Call TRANSCRIPT (q/e 4-30-15)
II. 7-14-15 PPHM Press Release: Q4/FY15 Earnings & Developments
III. Updated Table of Avid Revenues By Quarter (May’06-Current)
…Recall: Peregrine’s FY runs May-Apr, so FY’15 = May’14-Apr’15.
((( Orig. transcript from SeekingAlpha.com [ http://tinyurl.com/q5sj9zo ], with numerous corrections made. )))
Link to webcast replay:
http://ir.peregrineinc.com/events.cfm => http://edge.media-server.com/m/p/jzohnozt
FULL TRANSCRIPT…
7-14-2015 Q4 FY’15 Earnings Conf. Call (q/e 4-30-15)
WELCOME & FWD-LOOKING STATEMENTS: Jay Carlson (IR) http://www.peregrineinc.com
Speakers: Steve King, Joe Shan, Jeff Hutchins, Paul Lytle; Q&A session.
CEO STEVE KING – OPENING COMMENTS:
Thanks to all of you who have dialed in or participating via webcast in this afternoon’s Peregrine update. This past year was marked by clinical progress, scientific validation and business growth for Peregrine. Clinically, our SUNRISE trial in NSCLC remains on target to complete patient enrollment by calendar year-end. While we continue to advance the SUNRISE trial toward completion, we are also taking important steps to expand the commercial potential of bavituximab in the NSCLC and Breast Cancer indications by initiating new later stage clinical trials in both areas by calendar year-end. These new studies are supported by compelling data from several prior clinical trials showing the potential of bavituximab in combination with chemotherapy in HER2-negative breast cancer, and by exciting preclinical and translational data supporting the potential of bavituximab with anti-PD-1 targeting compounds like Opdivo. We believe supportive data from these new trials, combined with the SUNRISE trial data, will set the stage for commercial success for bavituximab into very important cancer indications. In addition to the flurry of activities on the later stage clinical development front, exciting data from the bavituximab clinical and preclinical programs was consistent and building throughout this past year.
Within oncology, there is a need almost across the board for new drugs that will improve existing therapies. Whether as chemotherapy, radiation or immune-oncology agents, most patients are still not getting the level of anti-tumor activity that is needed to have a significant impact. We believe bavituximab has the potential to make a difference in patient outcome when used in combination with all of these therapeutic approaches to treating patients. We have already seen across a number of clinical trials the classical immunotherapy survival tail when bavi is given in combination with chemotherapy. Recent preclinical data firmly supports the concept that targeting and blocking the PS-signaling pathway with antibodies, like bavituximab, allows more subjects to initiate a T-cell immune response, resulting in longer treatment duration in combination with PD-1 targeting antibodies that resulted in more subjects responding to therapy and as a result, statistically significant improvement in anti-tumor activity. In addition, translational clinical data supports the potential of bavituximab to have an immune stimulating impact on PD-L1 negative tumors. Data presented by number of investigators at both ASCO & AACR showed convincingly that patients with PD-L1 negative tumors did not fare well when treated with either PD-1 or PD-L1 targeting antibodies. So, bavituximab has shown the potential to help stimulate a T-cell anti-tumor immune response in the tumors that absolutely need a better immune response in order to respond to PD-1 therapy.
We recently entered into collaboration with investigators at Memorial Sloan Kettering Cancer Center [5-29-15: http://tinyurl.com/qxu4w2x ] to continue expanding on this important work, as well as to explore other potential applications of bavituximab and other agents that target PS-signaling pathway.
On the manufacturing front, I am very pleased to acknowledge that our wholly-owned subsidiary, Avid Bioservices, achieved record revenue this year, growing as a successful business while providing equally important infrastructure that can support commercial launch of bavituximab. I will continue my comments later, but first, the other members of our team will give a detailed overview of our clinical, preclinical and operational achievements.
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
I’d like to first provide an update on the company’s ongoing Phase3 SUNRISE trial, which is evaluating the use of bavituximab in the treatment of non-squamous NSCLC. SUNRISE continues to progress according to plan with more than 150 active clinical centers spanning 14 countries. We remain on track to complete enrollment by the end of this calendar year. Meanwhile, we continue to receive positive feedback from investigators who are excited by both the safety profile and immunomodulating properties of bavituximab. As a reminder, SUNRISE is designed as a Phase 3 Registration Trial [ http://www.clinicaltrials.gov/ct2/show/NCT01999673 ] and has 2 planned interim analyses. The 1st interim analysis, which will be conducted when 33% of the targeted overall survival events are reached, is for futility, and the 2nd interim analysis for futility or predictive success, will be conducted at 50% of events. To protect the integrity of this double-blind trial, an independent data monitoring committee has been established to evaluate safety data on an ongoing basis and make recommendations to Peregrine as to when the trial should be unblinded. As the interim analyses are triggered after pre-specified numbers of trial events are reached, in this case, deaths, we cannot, at this point, guide to when these might be reached, but we will be sure to provide updates as soon as we can.
Let me now discuss the expansion of our clinical programs in NSCLC and Breast Cancer. As we announced last month, while we wrap up enrollment in SUNRISE, we are planning a few new clinical trials, which we expect to initiate before year end. Peregrine’s decision to launch these new trials is driven by the considerable and growing body of data supporting bavituximab’s therapeutic potential when combined with chemotherapeutic drugs or immunotherapeutic agents such as checkpoint inhibitors.
One of these trials will build on the foundation we are laying in the NSCLC setting with the SUNRISE trial. The decision to initiate another lung cancer trial is supported by recent robust preclinical data supporting the combination of bavituximab with immune checkpoint inhibitors, which is very timely as the anti-PD-1 agent, nivolumab, has recently been approved in previously-treated squamous NSCLC. To that end, we are planning to initiate an open label multi-center randomized Phase2 trial of nivolumab (which is marketed as Opdivo) vs. nivolumab+bavituximab in patients with previously treated locally advanced or metastatic NSCLC who have not received a prior PD-1 or PD-L1 inhibitor. The primary endpoint of this trial is expected to be ORR, with secondary endpoints including duration of response, PFS, OS, and safety. Importantly, as translational studies have demonstrated that bavituximab enhances multiple markers of immune activation, even in tumors that express low levels of PD-L1, we plan to retrospectively examine patient outcomes based on pre-treatment PD-L1 expression level to better understand bavituximab’s effects on this potential biomarker.
With regards to our overall lung cancer & clinical development strategy, our goal is to have the opportunity for preliminary readout from this new trial prior to the un-blinding of the SUNRISE trial.
In addition, based on the totality of our clinical experience in advanced Breast cancer to date, we are also compelled to advance the program by initiating a late stage trial in BREAST CANCER. Promising data from an investigator-sponsored Phase 1 trial of bavituximab+paclitaxel to patients with metastatic HER2-negative Breast cancer were published during the quarter in the peer reviewed journal, Cancer Medicine [3-31-15: http://tinyurl.com/nm5oog4 ].
Results demonstrated that 85% of evaluable patients achieved an objective tumor response, including a complete response rate of 15% by RECIST. Data from this IST, combined with 2 prior Peregrine-sponsored trials (bavituximab+taxane-based combination treatments that yielded 61-74% overall response rates, a median overall survivals of over 20 months in advanced or metastatic Breast cancer patients) provided strong rationale to advance this indication. Thus, we are planning a seamless Phase 2/3 trial in patients with metastatic HER2-negative Breast cancer, with all patients receiving physician’s choice of paclitaxel or docetaxel alone or in combination with bavituximab. If the primary endpoint of ORR in the Phase 2 is reached, the Phase 3 part of the trial will be activated, which has a primary endpoint of PFS. As with our planned lung cancer trial, we expect to initiate this Breast cancer trial before the end of the calendar year, and the open-label nature of the trial may provide us the opportunity for data updates prior to SUNRISE unblinding.
Additionally, a randomized trial that of neoadjuvant paclitaxel with or without bavituximab is also planned to further elucidate bavituximab’s immune modulating mechanism in early stage HER2-negative Breast cancer.
Now, beyond Lung & Breast cancers, bavituximab has shown promise in several addl. oncology indications. Data from an IST evaluating bavituximab+sorafenib in patients with advanced Hepatocellular [Liver] carcinoma has demonstrated promising signs of activity, an acceptable safety profile, and multiple signs of immune activation as measured by pre- and post-treatment tumor samples.
Finally, 2 IST continue to enroll patients, evaluating bavituximab combination treatment regimens in patients with advanced Melanoma or Rectal adenocarcinoma. While it’s important to note that these studies are being conducted independently under investigator-held INDs and thus we do not have control over the timing of the data, with that said data from the Rectal adenocarcinoma trial will be presented at the American Society for Radiation Oncology’s (ASTRO) Annual Meeting in October [Oct18-21 2015, San Antonio http://www.astro.org/Meetings-and-Events/2015-Annual-Meeting/Index.aspx ]. I look forward to providing you an update on our future clinical advancements. And I will now turn the call over to Jeff Hutchins to discuss our preclinical programs.
JEFF HUTCHINS (VP/Preclinical Research):
Our group is working to further delineate bavituximab’s unique immune-stimulating mechanism of action and find the most promising immune activating treatment combinations for bavituximab by a series of preclinical studies that have also recently led into translational studies utilizing human lung tumor samples. I am pleased to say that the results of these studies have been impressive. Data from these studies presented at the ASCO Annual Meeting in June demonstrated the ability of the company’s PS-targeting antibodies to significantly increase the prevalence of tumor infiltrating CDA+ T cells and immune activating cytokines, while decreasing tumor promoting macrophages and myeloid cells, thereby enhancing the antitumor effects of chemotherapy as well as immune checkpoint inhibitors. Measurements of cellular immune activation markers and cytokine profiles in multiple tumor models consistently support the potential of our PS-targeting antibodies’ ability to work synergistically with approved & investigational immunotherapies. Our preclinical studies show that the combination treatment with an anti-PD-1 antibody yields superior growth inhibition in a large percentage of subjects, while also exhibiting multiple immuno-stimulatory changes associated with an anti-immune response as compared to anti-PD-1 alone. Taken together, these results support the potential of bavituximab to increase the number of subjects susceptible to respond to immune checkpoint blockade treatments and provide a rationale for the clinical evaluation of bavituximab with PD-1 or PD-L1 targeting drugs in Lung cancer and other indications.
Data from addl. studies, presented at the ASCO Annual Meeting [6-1-15: http://tinyurl.com/qxu4w2x ], measured changes in immune parameters in human lung tumor samples treated with the combination of bavituximab and the chemotherapeutic drug, docetaxel. These clinical translational studies demonstrated that bavituximab alone and in combination with docetaxel activates tumor-infiltrating CD8+ T cells, as demonstrated by increases in immune stimulatory cytokines and cellular activation markers. Interestingly, these positive results were correlated with low PD-L1 expression in the tumor lung tissue, a measurement common to the majority of NSCLC patients, and which is typically associated with poor response to checkpoint therapies. This further increases our enthusiasm regarding the Phase 3 SUNRISE trial, which is evaluating the same treatment combination used in these promising translational studies. We are excited to see these data and the consistency in which we see superior tumor growth inhibition with bavituximab alone and in combination with other therapies.
We are not alone, as Peregrine has generated and presented more data in recent months. There has been growing interest in the PS-signaling pathway in bavituximab. This interest is evidenced by the research agreement that Peregrine recently signed with Memorial Sloan Kettering Cancer Center [5-29-15: http://tinyurl.com/qxu4w2x ]. This research, which will be conducted in the lab of Dr. Jed Wolchok, a leader in the field of cancer immunotherapy, will examine the combination of bavituximab alongside models of checkpoint blockades that are unresponsive to inhibition or co-stimulation. This is not only a collaboration that is validating, but it will also create a better understanding of how PS-targeting agents may enhance immune activation & antitumor responses combined with other immunotherapies. We look forward to providing an update on this collaboration as well as the translational work we continued to conduct at Peregrine.
CFO Paul Lytle:
Turning to our financials, it’s important to note that we continue to closely manage our operations in line with our cash position, while balancing our various sources of capital. And one important source of capital is derived from our contract mfg business, Avid Bioservices, which generated $9.3mm in revenue this qtr and $26.7mm for the full FY2015. This represents a 44% increase in qtr-over-qtr revenue and a 20% increase in yr-over-yr revenue. As we look to the future, I would also like to emphasize that we currently have a strong backlog for future services. Over the recent periods, our backlog has ranged from $20mm to $30mm and today this backlog has grown to approx. $40mm. As a result of this growing backlog, we expect contract mfg revenue for FY2016 to increase to $30-35mm. This projected revenue growth is primarily due to 2 reasons: 1st, we have seen an increase in demand from existing & new customers; and 2nd, we now have new mfg capacity to offer our customers that is already being booked.
This is an exciting time for our contract mfg business, so let me take a few moments to share with you our plans and progress in building our new facility. Last December, we laid out strategic plans to expand our mfg capacity to help support the revenue growth of Avid as well as creating sufficient mfg capacity for the potential commercial launch of bavituximab. We also mentioned that growing this revenue-generating business is very important to us, as it reduces the amount of capital and funding we would need to raise by other means. This strategy is coming to fruition very quickly. I am excited to say that the new facility is close to being ready for production. The core mfg suite has been built, the equipment has been installed, and it is now undergoing some final testing to ensure all systems are operating effectively. We remain on track to commence production in this new facility in the very near future.
Now, turning to expenses, we saw an expected increase in R&D spending this qtr and FY as we continue to invest in the Phase 3 SUNRISE trial. This resulted in an increase in our reported cash burn rate representing our reported net loss minus non-cash expenses to approximately $10.5mm for Q4. This cash burn rate has been consistent over the past 4 qtrs of FY2016, leading to a burn rate of $42.6mm for the full FY. A more detailed analysis of our statement of operations is included in our Form 10-K that will be filed shortly [ http://tinyurl.com/ocrtkuj ]. In conclusion, let me say that our financial goals are centered on maintaining a solid cash position and investing these proceeds into our novel immuno-oncology program led by bavituximab and our revenue-generating mfg business. We will continue to closely manage our operations in line with our cash position, while balancing our various sources of capital.
CEO STEVE KING – MILESTONES:
I started today’s call by stating that we have continued to make progress across our entire business. As Joe, Jeff, and Paul have detailed, these achievements are significant. We are more confident than ever in the potential of bavituximab to help other cancer treatments work better and we are expanding our clinical programs to capture the value that we believe exists in new therapeutic combinations & indications. Specifically, as Joe detailed, we will be initiating a Phase 2 study in NSCLC, combining bavituximab with Opdivo, an FDA-approved PD-1 inhibitor, while also initiating a Phase 2/3 preclinical trial in Breast cancer, combining bavituximab with chemotherapy. The intent of these trials and all future trials of bavituximab is to expand the utility to ultimately address multiple cancers and to improve the lives of cancer patients worldwide by improving upon current and evolving standard of care.
As discussed by Jeff, the compelling preclinical data we have been presenting, as well as a growing body of nonaffiliated research into the PS signaling pathway and how it is exploited by tumors for survival and growth, has greatly heightened interest in bavituximab from the scientific community. As a result, we recently entered into collaboration with established immune-oncology experts at Memorial Sloan Kettering Cancer Center to continue expanding the potential of the PS-targeting platform.
Beyond our therapeutic platforms, the Avid Bioservices business has been growing steadily, achieving record revenue of over $26mm this year. And, with the $40mm backlog, we anticipate our revenue next year to grow to between $30-35 mm. This business is strong and gaining momentum and the revenue generated by Avid provides a firm foundation for its continued success.
With that, I will outline a number of upcoming milestones. Regarding our SUNRISE Ph3 2nd-Line NSCLC study, on track - we are on track for completing patient enrollment by yr-end 2015, with planned IDMC interim analyses at 33% & 50% of targeted overall survival events as those are reached, with estimated unblinding of the trial to occur approx. near calendar yr-end 2016.
Regarding our planned Ph2 2nd-Line NSCLC, bavituximab+nivo, and our planned Phase 2/3 HER2-neg. Breast cancer trial evaluating docetaxel or paclitaxel with bavituximab, we estimate trial initiation from both of those studies during the 2nd-half of 2015. And in these open label studies, we will have the potential for interim data from both studies as early as 2016.
For the ongoing Phase 1 Rectal adenocarcinoma IST evaluating bavituximab in combination with capecitabine & radiation, the investigator is continuing to enroll and treat patients and we do expect interim data to be presented at the ASTRO Meeting coming up in October.
And at Avid, we expect launch of production in our new facility to happen in the immediate future, and this launch will be a pivotal event as we continue to grow the Avid business and to prepare for bavituximab commercialization. This concludes our prepared remarks and we would now like to open the line for questions.
Q&A: [26:25 mark]
1. Roy Buchanan for Charles Duncan – Piper Jaffray [http://www.piperjaffray.com – 3-5-13 Initiates PPHM: http://tinyurl.com/bxhntk3 ]
RB: ” Nice top line results… On this Ph2 with nivolumab, do you guys plan to screen for PD-L1 expression on enrollment and intend to balance between the arms for that expression?”
Joe Shan: At this point, we are not planning to select patients based on that status, but we will be collecting that information for retrospective.
RB: ”Have you guys applied for breakthrough and do you intend to for SUNRISE?”
Joe Shan: For SUNRISE, as you know, we have a fast track designation, and so that affords us the regulatory benefits of a priority review.
Steve King: To expand on that, I think as we learn more about bavituximab, particularly through these upcoming studies, that’s certainly a possibility for the future. I think it will depend really on the particular targeted indication or set of patients. And that’s going to be very interesting, as you mentioned earlier, to take a look at the PD-L1 neg. patients or low patients and see how the outcome of those patients is as compared to the PD-L1 higher positive patients. There appears to be a developing need for better treatments, or better combinations, in those PD-L1 neg. patients, and so that’s an example of a potential indication that could trigger some add. regulatory strategies beyond the Fast Track.
2. Thomas Yip (MLV & Co.): http://www.mlvco.com
TY: ”Congratulations on the very nice qtr for Avid. My 1st question pertains to your preclinical collab. with Memorial Sloan Kettering regarding the development of checkpoint inhibitor combinations. It maybe a little bit early, but just wondering whether you guys have explored any specific oncology indications that you are going after while you’re screening, perhaps indications such as Melanoma?
Jeff Hutchins: Melanoma is their lab specialty and they are heavily invested in that, so that certainly will be our first look, and then expanding that based on the results. What we are really looking for is what are the contributions of each of the combination partners and that sort of thing, and the MSK Wolchok Lab has become just world-renown in understanding those dynamics, and so that’s what we are really anticipating to benefit from.
Steve King: In addition to that, obviously there is lot of interest now in a number of indications outside of just simply NSCLC as well as Melanoma. The collaboration at Memorial Sloan Kettering is definitely broader than just Melanoma, but really we want to look at other potential indications. A lot of interest coming to the top - Ovarian cancer, Bladder cancer are other prime targets. So, we will be looking at a number of different indications, and some of those may end up being things we do on the preclinical front to set stage for clinical studies and some maybe more even potentially moving forward with collaborators right into clinical trials. All that thinking is currently being discussed with the people from Memorial Sloan Kettering, and I do think we’ll have collaborations at different levels which will include preclinical as well eventually, clinical studies.
TY: ”You mentioned that the new Avid facility is very close to being completed. What does the addl. capacity mean to the backlog? It’s very nice that you guys have grown the backlog from $29mm just a couple of months ago to $40mm, and you guys have the new revenue target, everything sounds very good. But, will the extra capacity bite into the backlog or do you expect more new customers and more orders from existing customers as well?”
Steve King: Almost all the backlog is really related to the current facility, because as the new facility comes online, obviously we have a lot of addl. activities for bavituximab planned for there in the early parts of the opening of the facility. But we do have a lot of expressed interest from clients in moving into the space and actually being able to utilize that and that’s particularly for later-stage & commercial products. So right now it’s a nice mix - a lot of backlog is just related to the existing facility, but some of that backlog is now starting to be associated with the new facility that’s coming online, and we do expect that to grow as time goes by and as we really reach full capacity over in that facility.
Paul Lytle: The new facility more than doubles what our current capacity is in terms of space and bioreactor space that we have over there. So we do have an opportunity here to really to grow the business, even much larger than what our current backlog is. So we are excited about the new facility.
3. George Zavoico – Jones Trading
GZ: ”Congrats on a good quarter. I guess if you build this, they will come. The number of antibodies and other biologics that are being developed now by multiple potential customers is growing quite rapidly – you are there. It seems like you are there to capitalize on it, that’s great, so we expect to see that backlog increase but also the revenue increase as you fill the orders. Joe, you mentioned in your prepared remarks that some of the physicians that you are talking to that were involved in SUNRISE or participating in SUNRISE have been impressed by the immuno-modulatory properties that they have seen. Could you expand on that a little bit? What exactly are they measuring, what are they seeing that you might be able to talk about that they’re relating to or you can’t disclose that?”
Joe Shan: It’s more of a general impression. I think they’ve been briefed on the data that was presented at ASCO and the consistency across the systems, the species that we looked as all pointing to the same immune-stimulating properties. I think that’s what I was referring to in terms of them being impressed with the potential.
GZ: ”So you weren’t speaking specifically to the patients?”
Joe Shan: Not SUNRISE, it’s still blinded.
GZ: That’s why I thought it was a little confusing when you mentioned that. But I am sorry about that, I misunderstood.”
Steve King: If I can expand on that – I’ve had a chance to meet with a number of the physicians, and in general, a lot of the enthusiasm is, obviously it’s a blinded study, so they can’t relate to their direct experience in the SUNRISE trial other than how the trial is going overall, but I do think there is a lot of enthusiasm because of bavituximab’s mechanism of action & safety profile and the potential for combinability even outside of the docetaxel combination. That’s also driving a lot of interest because it’s had a lot of great data over the last 6mos, including the translational data, which is really important because it ties together what we’ve seen in the preclinical models with what we expect to see in the clinic, and it’s really matching up very nicely and that consistency I think is very exciting to them. But also, as physicians want to treat their patients and want to see their patients treated better, a lot of the enthusiasm is fueled by the fact that bavituximab really has potential in chemotherapy, which is going to continue to be an important part of how patients are treated in Lung cancer, in Breast cancer, but also that as new & emerging treatments, such as Opdivo & Keytruda, coming to the marketplace that bavituximab has a place there as well. I think they like the fact that they are working on a drug that really does have a potential to be used throughout the continuum when they think about how their patients might get treated in the future.
GZ: ”Thanks, that’s helpful. With regard to Breast cancer, you are combining chemotherapy now with an immune-checkpoint. Breast cancer has been noticeably absent for most immune-checkpoint studies, can you comment on that? Are there emerging immunotherapies in breast cancer?”
Steve King: The focus from an immunotherapy standpoint has been on Melanoma, more recently Lung, Bladder. There are a number of indications which have historically been kind of hot spots for immunotherapies. Breast cancer is relatively unexplored. It’s one of those tumor types that tends to have less of mutations that would generate some robust immune responses. What’s needed is changing that tumor microenvironment and getting those immune responses started, and that’s where we see a role for bavituximab. As we continue to move forward, while our initial focus, because of our great clinical data to date, was in chemotherapy combinations, we are evaluating immunotherapy combinations in Breast cancer, and you will just have to stay tuned to see how that evolves.
Jeff Hutchins: What we saw at ASCO was that the T-cell CD8+ immune cell infiltrate is becoming a key marker in breast cancer for a positive prognosis. So, the underlying value clinically for bavituximab for immune stimulation in breast cancer is certainly there.
GZ: ”And in that regard are you reconsidering Pancreatic cancer? You had good results in that before.”
Steve King: That’s interesting. We’ve had a lot of interest from collaborators in Pancreatic cancer because when you go back and look at our data, we had nice tumor responses, but we did see the survival tail in the overall survival curve, and that is something that drives interest, because if we can combine that with downstream checkpoint inhibitor like PD-1, PD-L1 inhibitor, we think that, that’s absolutely an indication, where we potentially could have some impact. So it’s on the list along with Breast and a few others. There is definitely a lot of interest in Pancreatic cancer, not just from Peregrine but from others in the immuno-oncology space.
GZ: ”On the costs in the MSKCC collaboration, any terms that you can disclose or how that might increase your R&D spending?”
Paul Lytle: Good question. We haven’t disclosed the details of all of our collaborations. But it’s a typical sponsored research agreement, where we provide drug and funding and they perform research for us. So that’s about all I can say about that because we don’t detail it out specifically within our filings.
Steve King: I think in the scheme of things with all the clinical trials going on or what have you, it’s not...
Paul Lytle: An insignificant portion of our R&D spending.
GZ: ”Final question on the significant spending on your upcoming Ph2 and Ph2/3 and your continuing SUNRISE trial. Part of it seems to be offset by the Avid revenue. Paul, you mentioned how you are managing the various sources of capital – can you talk about the expected burn rate? Might you consider partnering any of these programs in the near future to help offset some of those costs?”
Paul Lytle: We’re very common with other biotech companies in that we operate in a fairly capital-intensive industry. That being said, we are actually a very unique company in that we actually generate revenues, and as I mentioned earlier, we are increasing our revenue guidance this year to $30-35mm, with a $40mm backlog. We have our bavituximab program, which is a I/O compound in Ph3 development that we own 100% of the rights to. That brings tremendous partnering opportunities to the table, and one of our goals here internally is to partner ex-U.S. and to drive the value here in the U.S. for ourselves. So, I think we are going to be looking at lot of those things to kind of balance our financial resources and these are just a couple of sources that we have available to us that not all companies have.
Steve King: In addition to that, if you look at it from a timing standpoint, as we’re completing enrollment in the SUNRISE trial which moves us more & more towards completion of that study, we’ll actually be ramping up these other studies. That’s important for a couple of reasons, but also from an expenditure standpoint, that as we expect to see the cost associated with SUNRISE go down, we’ll see the costs of these other trials go up to sort of replace it, but to add to that potential value as we go forward.
Paul Lytle: We actually strategically aligned the completion of enrollment with the current SUNRISE trial with the initiation of these other Phase 2 trials.
GZ: ”I suppose the addl. trials also act as leverage in any partnering discussions you might be engaged in right now?”
Paul Lytle: Absolutely.
4. Rahul Jasuja - Noble Life Science Partners http://noblelsp.com/research
RJ: ”Looking at PS blockade as the PS’s immune checkpoint and comparing that to the other immune checkpoints, PD-1 & CTLA-4 and so on, could you comment on the fact that you are getting such a broad repertoire of an immune response vs. blocking just PD-1 or CTLA-4? So, you are probably connecting innate adaptive immunity. Can you comment on that sort of differential? And, how does that affect your opportunity for clinical development?”
Steve King: I will start off and turn over to Jeff. I really view the immune system as, it’s a balance, right? What’s happening is that patients whose balance has tipped toward immune-activation, particularly T-cell activation, are the patients who are now responding well to PD-1, PD-L1 therapy, and it’s because those agents now have an immune active component to work with, namely the T-cells. What bavituximab does is it helps create that same balance in patients that otherwise might not have it by changing the tumor microenvironment, activating T-cells, and now you’ve got a perfect environment for these downstream checkpoint inhibitors to work. So, we view it as a nice synergistic relationship with these downstream checkpoint inhibitors, because they’re helping keep going what bavituximab can get started. So, it can really get that immune system kicked off, and now we can keep that going with PD-1, PD-L1 therapy. So, effectively, anywhere PD-1, PD-L1 are, there’s still going to be a need for converting more patients over to responders, and we see that as very broadly in the I/O combinations where the potential is from a development standpoint in the clinic. And the same can be said for chemotherapy. Chemotherapy, by killing cells and causing more PS exposure, inhibits the immune system from being able to respond to those tumors adequately. Anywhere where chemotherapy is effective, we think we have a great potential to combine with that and drive the immune component to go along with the direct tumor killing of the chemotherapy. And particularly like the Taxanes, where we’ve seen great clinical data and so we’re following that lead, a that’s what we’ve learned from our own clinical experience. Joe or Jeff, care to expand?
Jeff Hutchins: I like to think of PS targeting kind of as a catalyst or an enzyme catalyst. So, it’s not necessarily the enzyme itself, but it lowers the barrier to activation. And you can kind of think of that in the tumor microenvironment terms is that there is such a high barrier to activation in this local microenvironment, and by coming in and blocking the PS signaling, sending activation signals to FcR gamma, now you are turning the tables and, as Steve said, reestablishing that balance to an immune-activation balance and now you have an environment that’s capable of recognizing tumor cells and killing it.
Joe Shan: I will just add from the clinic, this phenomenon really seems to be localized to tumor microenvironment. So, we see great safety profiles and the combinability of bavituximab with virtually any of the antineoplastic treatments, including immunotherapies, is certainly an advantage and that’s what we hope to be able to capitalize on.
RJ: ”It’s tempting to say that maybe comparing to PD-1, CTLA-4, where it seems like you only need to have tumors that have TILs [tumor infiltrating lymphocytes] out there, you probably are getting [?????] presentation and really sort of getting more [????] and so on because of the way your antibody works. Is that OK to say?”
Joe Shan: Yes.
RJ: ”We talked about combining with checkpoint inhibitors. If you’ve got MedImmune’s OX40 to the co-stimulator in the pipeline; 4-1BB is getting a lot of traction. What about in combination with co-simulators? Is that on the cards with the Wolchok Lab or is that not on the cards?”
Jeff Hutchins: Absolutely. That’s some of our main intent. We have looked at the checkpoint inhibitors quite extensively internally. That’s the kind of data that attracted this kind of collaboration and the logical follow-on now is to look at the agonist through the immune stimulator, co-stimulators in that realm.
RJ: ”A question on kinase inhibitors. There are tons of tumors and cancer subtypes, and many of them are genetically defined that are based on kinase inhibitors. You’ve talked about sorafenib, and I know that sorafenib treatment also flips PS. So, is that another subset of tumors that you could look at combination approaches with kinase inhibitors?”
Joe Shan: Absolutely. Anything that stresses the tumor microenvironment out I think will work nicely with PS blockade. We’re not excluding patients who have known EGFR mutations or ALK gene rearrangements in the SUNRISE trial, for example. We will have an opportunity at the end of the study to look at patients who have those genetic mutations, [???] mutations and see if they fare better on the bavi containing arm.
RJ: ”On using PD-L1 as a biomarker - as I understand it, you are going to look at PD-L1 prior to therapy - I think you talked about that in the Breast cancer trial, or is it in NSCLC? You are going to look at PD-L1 and then post-treatment, you are going to look for increasing the PD-L1, which would indicate positive anti-PS therapy, is that right?”
Joe Shan: Yes, we’re going to retrospectively look at this to see if we see CD8 activation, and therefore PD-1 increase, along with the PD-L1 increase that we’ve seen in our Liver cancer translational data as well as in the ex-vivo microsphere project, and of course, in preclinical studies.
Steve King: That’s actually a component we have been building into a lot of these studies, so it’s also built into the Melanoma IST as well as a Rectal adenocarcinoma IST, and it’s going to be built into all these studies going forward, because we recognize that’s an important validation point for each of these studies. It’s one of the data points that can come from these studies, which is nice because they are open-label studies – we’ll have surrogate endpoints and regular endpoints, but also we will have these translational data points coming from those trials as well. We think each of these studies we start can be rich with data points that are all basically validating for this mechanism of action and for these particular combinations.
Jeff Hutchins: And it’s not just PD-L1 that we are looking at – we’re really just building an immune profile in this pre & post-tissues in these patients to look at the condition of the lymphocytes, the myeloid cells, and the dendritic cells.
RJ: ”For the 2nd-Line NSCLC SUNRISE trial, obviously these metrics prospectively were not studied on medically for each patient or is there any way you can look at retrospectively these metrics?”
Joe Shan: Yes, we are collecting, as available, archival issues of pretreatment. So at least we should have some kind of hint of prognostic, if you will. These are terms that we value.
Steve King: While we may get some data from the SUNRISE trial, we also recognized early on that that’s Phase 3, and the goal there is to get us through and execute, and hopefully get to an approval endpoint at the end of the study. The more complicated you make it, it’s more difficult it is to enroll a study like that, so what’s nice about these upcoming studies is they are a little more amenable to the sort of analysis and planned data analysis right up front.
RJ: ”You talked about not having any autoimmunity with PS blockade vs. the fact that there is the risk of autoimmunity with PD-1 & PD-L1 and probably more with CTLA-4. Trying to understand the mechanism - is it because maybe the bigger effect of blocking PS really comes from not just preventing or worsen the immunosuppression, but actually because of the FcR gamma-mediated effect and so on, that instead of revitalizing the immune systems and not what only affects immunosuppressive pathway, that is the case in PD-1 & CTLA-4?”
Jeff Hutchins: That’s certainly our view, and we are engaging in preclinical & ex-vivo experiments to look at that closer. Certainly, the dogma around FcR gamma activation is that it’s a very cross-linked dependent, concentration dependent event. And so if there are enough cells there, apoptotic cells that are expressed in PS, then that activation won’t take place, and you will also need a lot of myeloid cells there at the same time. So, we believe that it is very cell & receptor concentration dependent, and that’s why we are not seeing the autoimmunity in a systemic type way in our animal models, and we haven’t seen it, as Joe mentioned, clinically manifest itself on the safety profiles.
Steve King: Another point there is that with blocking PS, we’re simply restoring what should be the normal immune response to the tumor. So we’re actually pushing the immune system beyond what it’s normally supposed to do - we’re actually just recreating that balance that allows the immune system to activate. If you look at other checkpoints, you are basically pushing the immune system beyond what it’s actually supposed to be doing, you supposed to have those checks in place to protect the body, and when you take them all off, then of course, you are going to have these complications. One of the very promising things about bavituximab combination therapy is we are simply restoring the right balance of immune response to the tumor that allows that immune response to take place – we’re not pushing it beyond its limits. If we can then take these downstream checkpoints and push that immune response as far as their safety will allow, then we shouldn’t be adding to the toxicity because it’s already there in those patients that are probably responding the best. That’s one of the things when you talk to physicians, they really like the concept, ‘hey, we’ve basically got a pretty safe drug, we can start monitoring these other things’, vs. just adding two toxic things together and what happens, why we’ve seen some of the clinical data that you just get double the toxicity, and that’s getting to the edge of I think what patients want to see at the end of the day.
RJ: ”Sure, that makes sense. Well, thank you guys and congratulations on the Avid revenue, a pleasant surprise.”
MR. KING’S CLOSING COMMENTS:
I’d like to thank all of you for participating in today’s phone call. In closing, I’d like to say that Peregrine’s preclinical, translational, and clinical programs are contributing significantly to the promise of immune-oncology and the impact it will have on millions battling cancer. More than ever, we believe bavituximab can help standard and emerging cancer treatments to overcome the immune suppression so commonly found in the tumor environment. In closing, as always I want to thank our stockholders for their continued support, and I’d like to especially thank the patients and their families that are participating in our bavituximab clinical trials. With that, we’ll conclude the call.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = == = = =
7-14-15 PR: “Peregrine Pharmaceuticals Reports Fourth Quarter and Year-End FY 2015 Financial Results and Recent Developments”
• Phase III SUNRISE Clinical Trial on Track to Complete Patient Enrollment by Calendar Year-End 2015
• Initiation of Later-Stage Bavituximab Trials to Expand Commercial Potential in NSCLC and Breast Cancer Planned for Second Half of Calendar Year 2015
• Avid's Contract Manufacturing Revenue Exceeds $26 Million for FY 2015
TUSTIN, July 14, 2015: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM) (NASDAQ:PPHMP), a biopharmaceutical company focused on developing novel investigational products that help harness the body's own immune system to fight cancer, today announced financial results for the fourth quarter and the fiscal year (FY) 2015 ended April 30, 2015 and provided an update on its advancing clinical pipeline and other corporate developments.
HIGHLIGHTS SINCE JANUARY 31, 2015:
"During the fourth quarter, Peregrine achieved multiple milestones spanning all areas of the business. Most importantly, we remain on schedule to complete patient enrollment in the SUNRISE Phase III trial in NSCLC by the end of calendar year 2015, while also planning for the initiation of two new trials designed to further expand our breast and non-small cell lung cancer clinical programs," said Steven W. King, President and CEO of Peregrine. "Our promising new collaboration with Memorial Sloan Kettering Cancer Center [5-29-15: http://tinyurl.com/qxu4w2x ], together with the considerable amount of pre-clinical and clinical data that has been generated recently, serves to further validate bavituximab and its potential to enhance the effects of chemotherapy, as well as immune checkpoint targeting treatments. Today, we are more confident than ever in bavituximab and we are strategically expanding our clinical programs to capture the value that we believe exists in new therapeutic combinations and indications. Specifically, we are expanding our NSCLC clinical program to include a planned Phase II study combining bavituximab with Opdivo®, an FDA-approved PD-1 inhibitor, while also initiating a planned Phase II/III clinical trial in breast cancer combining bavituximab with chemotherapy. In addition to our drug development efforts, Avid achieved record revenue during this FY 2015 and is on track to grow its revenue in FY 2016 based on the growing backlog of services and the near-term launch of the new manufacturing facility. We look forward to providing updates on our bavituximab clinical program, data from currently enrolling clinical trials and collaborative development efforts in the coming months."
Clinical Highlights:
• Continued progress enrolling ongoing SUNRISE clinical trial in non-small cell lung cancer (NSCLC); study remains on schedule to complete enrollment by end of calendar 2015.
• Peregrine announced plans to expand the bavituximab clinical development program to include a Phase II trial to evaluate the combination of bavituximab and Opdivo® (nivolumab), an anti-PD-1 antibody, in NSCLC, and a Phase II/III trial to evaluate bavituximab with chemotherapy combinations in HER2-negative metastatic breast cancer. These trials are expected to be initiated during the second half of 2015.
• Phase I study results from an investigator-sponsored trial evaluating bavituximab plus paclitaxel in patients with HER2-negative metastatic breast cancer were published in the peer-reviewed journal, Cancer Medicine. Findings showed that the combination produced an objective tumor response in 85% of evaluable patients, with 15% of patients achieving a complete response, measured in accordance with published Response Evaluation Criteria In Solid Tumors (RECIST).
• Data presented at the 2015 ASCO annual meeting from a Phase I/II study of bavituximab and sorafenib in advanced hepatocellular carcinoma (HCC) demonstrated that the treatment combination induced multiple signs of immune activation with a corresponding reduction of T-regulatory cells in the tumor environment. The treatment was well-tolerated with no indications of autoimmune adverse events that have been seen with other checkpoint immunotherapies.
Preclinical Highlights:
• Peregrine and Memorial Sloan Kettering Cancer Center entered into a research agreement to explore the potential of Peregrine's proprietary PS-targeting antibody platform. The goal of the research is to identify effective treatments combining bavituximab with other checkpoint inhibitors or immune stimulating agents. [5-29-15: http://tinyurl.com/qxu4w2x ]
• Data from preclinical studies presented at the 2015 ASCO annual meeting [6-1-15/ASCO: http://tinyurl.com/qxu4w2x ] demonstrated the ability of the company's PS-targeting antibodies to significantly increase the prevalence of tumor infiltrating CD8+ T-cells and immune-activating cytokines, while decreasing tumor-promoting macrophages and myeloid cells. These findings highlight the ability of the antibodies to enhance the anti-tumor effects of both chemotherapy and immune checkpoint inhibitors.
• Two preclinical abstracts and one clinical translation abstract were presented at the 106th Annual Meeting of the American Association for Cancer Research (AACR). Most notably, initial data from a pilot study of clinical translational ex vivo cultures show that bavituximab, both alone and with docetaxel, elicits evidence of a tumor-specific immune response in patients with human adenocarcinoma of the lung including tumors with low PD-L1 expression.
• Preclinical data presented at the Keystone Tumor Immunology Symposium [2-9-15: http://tinyurl.com/q6cx4w6 ] showed that a phosphatidylserine (PS)-targeting antibody equivalent to bavituximab combined with an anti-PD-1 antibody displayed statistically significant increases in tumor fighting immune cells, activation signals and inflammatory cytokines in a model of melanoma compared to anti-PD-1 alone. Moreover, cells that suppress the immune system from recognizing tumors, such as myeloid-derived suppressor cells (MDSCs), were reduced by more than 40% in the combination with the PS-targeting antibody versus anti-PD-1 alone.
Commenting on data presented at the 2015 ASCO meeting, Jeff T. Hutchins, Ph.D., VP of Preclinical Research stated, "Measurements of cellular immune activation markers and cytokine profiles in multiple tumor models consistently support the potential of our PS-targeting antibodies to work synergistically with approved and investigational immunotherapies. Our preclinical studies show that combination treatment with an anti-PD-1 antibody yields superior tumor growth inhibition in a larger percentage of subjects while also exhibiting multiple immunostimulatory changes generally associated with anti-tumor immune responses as compared to anti-PD-1 alone. Taken together, these results support the potential of bavituximab to increase the number of subjects whose tumors express increased levels of PD-1 positive T-cells and provide rationale for the clinical evaluation of bavituximab with PD-1 or PD-L1 targeting drugs in lung cancer and other indications."
Avid Bioservices Highlights:
• Avid Bioservices reports revenue growth of 20% for FY 2015 with revenues of more than $26 million from contract manufacturing business.
• Contract manufacturing committed backlog hits $40 million from existing customers.
• Avid makes significant progress toward launching its new state-of-the-art contract manufacturing facility.
"Avid Bioservices had a strong fourth quarter and record FY generating $9.3 million in contract manufacturing revenue in the fourth quarter of FY 2015 and $26.7 million in contract manufacturing revenue for the full FY 2015," said Paul Lytle, CFO of Peregrine. "We have also continued to see a strong demand for contract manufacturing services that has grown our committed backlog to approximately $40 million. With the new manufacturing facility coming online in the near future, Avid is positioned to meet the growing demand of existing and potential future clients while also preparing for our potential commercial launch of bavituximab."
Corporate - Intellectual Property
The European Patent Office (EPO) granted Patent Number 2,269,656, licensed to Peregrine titled "Selected Antibodies Binding to Aminophospholipids and their Use in Treatment, Such as Cancer." The patent covers bavituximab as a composition of matter and for use in therapy, such as for treating cancer including in combination with radiotherapy or chemotherapy, e.g., with docetaxel. This important patent expands upon the company's intellectual property portfolio, which now numbers more than 140 worldwide issued patents and pending applications for the bavituximab oncology program.
FINANCIAL RESULTS
Total revenues for the fourth quarter FY 2015 were $9,308,000, compared to $6,474,000 for the same quarter of the prior fiscal year. For FY 2015, total revenues were $26,781,000, compared to $22,401,000 for the prior fiscal year. The fourth quarter FY 2015 and FY 2015 increases were attributed to an increase in contract manufacturing revenue.
Contract manufacturing revenue from Avid's clinical and commercial biomanufacturing services provided to its third-party clients increased 44% to $9,308,000 for the fourth quarter FY 2015 compared to $6,474,000 for the fourth quarter FY 2015 and increased 20% to $26,744,000 for FY 2015 compared to $22,294,000 for FY 2014. The fourth quarter FY 2015 and FY 2015 increases were primarily attributed to an increase in demand for contract manufacturing services. Current contract manufacturing commitments from Avid's third-party customers are approximately $40 million, covering services to be provided during FY 2016 and into FY 2017. Based on this current backlog, Peregrine expects contract manufacturing revenue for FY 2016 to be between $30 and $35 million. In addition to providing biomanufacturing services to its third-party customers, Avid will continue to prepare for the potential commercialization of bavituximab.
Total costs and expenses for the fourth quarter FY 2015 were $21,477,000, compared to $17,003,000 for the fourth quarter FY 2014. For FY 2015, total costs and expenses were $77,280,000 compared to $58,107,000 for FY 2014. These increases for both fourth quarter FY 2015 and FY 2015 were primarily attributable to an increase in research and development expenses associated with the Phase III SUNRISE trial. For the fourth quarter FY 2015, research and development expenses were $11,531,000, compared to $8,813,000 for the fourth quarter FY 2014, and for FY 2015 were $42,996,000 compared to $27,723,000 for FY 2014. In addition, cost of contract manufacturing increased 24% to $4,758,000 and 19% to $15,593,000 for the fourth quarter FY 2015 and FY 2015, respectively, primarily due to higher reported revenue compared to the same prior year periods. For the fourth quarter FY 2015, selling, general and administrative expenses were $5,188,000, compared to $4,361,000 for the fourth quarter FY 2014 and for FY 2015 were $18,691,000 compared to $17,274,000 for FY 2014.
Peregrine's consolidated net loss attributable to common stockholders was $13,513,000 or $0.07 per share, for the fourth quarter of FY 2015, compared to a net loss attributable to common stockholders of $10,649,000, or $0.06 per share, for the same prior year quarter. For FY 2015, net loss attributable to common stockholders was $54,054,000, or $0.30 per share, compared to $35,763,000, or $0.22 per share, for FY 2014.
Peregrine reported $68,001,000 in cash and cash equivalents as of April 30, 2015, compared to $77,490,000 at fiscal year ended April 30, 2014.
More detailed financial information and analysis may be found in Peregrine's Annual Report on Form 10-K, which will be filed with the Securities and Exchange Commission today.
CONFERENCE CALL
Peregrine will host a conference call and webcast this afternoon, July 14, 2015, at 4:30 PM EDT (1:30 PM PDT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.peregrineinc.com/events.cfm .
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of previously-treated non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Opdivo® is a registered trademark of Bristol-Myers Squibb Company
Safe Harbor *snip*
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
Three Months Ended
April 30,
Twelve Months Ended
April 30,
2015 2014 2015 2014
Unaudited Unaudited
REVENUES:
Contract manufacturing revenue $ 9,308,000 $ 6,474,000 $ 26,744,000 $ 22,294,000
License revenue - - 37,000 107,000
Total revenues 9,308,000 6,474,000 26,781,000 22,401,000
COSTS AND EXPENSES:
Cost of contract manufacturing 4,758,000 3,829,000 15,593,000 13,110,000
Research and development 11,531,000 8,813,000 42,996,000 27,723,000
Selling, general and administrative 5,188,000 4,361,000 18,691,000 17,274,000
Total costs and expenses 21,477,000 17,003,000 77,280,000 58,107,000
LOSS FROM OPERATIONS (12,169,000 ) (10,529,000 ) (50,499,000 ) (35,706,000 )
OTHER INCOME (EXPENSE):
Interest and other income 34,000 281,000 142,000 349,000
Interest and other expense - - (1,000 ) (5,000 )
NET LOSS $ (12,135,000 ) $ (10,248,000 ) $ (50,358,000 ) $ (35,362,000 )
COMPREHENSIVE LOSS $ (12,135,000 ) $ (10,248,000 ) $ (50,358,000 ) $ (35,362,000 )
Series E preferred stock accumulated dividends (1,378,000 ) (401,000 ) (3,696,000 ) (401,000 )
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS
$ (13,513,000 ) $ (10,649,000 ) $ (54,054,000 ) $ (35,763,000 )
WEIGHTED AVERAGE COMMON SHARES OUTSTANDING:
Basic and Diluted 188,747,579 177,264,434 182,558,332 161,579,649
BASIC AND DILUTED LOSS PER COMMON SHARE $ (0.07 ) $ (0.06 ) $ (0.30 ) $ (0. 22 )
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED BALANCE SHEETS
AS OF APRIL 30, 2015 AND 2014
2015 2014
ASSETS
CURRENT ASSETS:
Cash and cash equivalents $ 68,001,000 $ 77,490,000
Trade and other receivables, net 3,813,000 1,332,000
Inventories 7,354,000 5,530,000
Prepaid expenses and other current assets, net 1,355,000 1,419,000
Total current assets 80,523,000 85,771,000
PROPERTY AND EQUIPMENT:
Leasehold improvements 1,538,000 1,538,000
Laboratory equipment 5,965,000 5,646,000
Furniture, fixtures, office equipment and software 3,991,000 2,679,000
Construction-in-progress 11,819,000 -
23,313,000 9,863,000
Less accumulated depreciation and amortization (8,189,000 ) (7,416,000 )
Property and equipment, net 15,124,000 2,447,000
Other assets 1,817,000 2,327,000
TOTAL ASSETS $ 97,464,000 $ 90,545,000
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED BALANCE SHEETS
AS OF APRIL 30, 2015 AND 2014 (continued)
2015 2014
LIABILITIES AND STOCKHOLDERS' EQUITY
CURRENT LIABILITIES:
Accounts payable $ 10,385,000 $ 2,434,000
Accrued clinical trial and related fees 3,910,000 4,433,000
Accrued payroll and related costs 4,606,000 3,837,000
Deferred revenue, current portion 6,630,000 5,241,000
Customer deposits 11,363,000 5,760,000
Other current liabilities 437,000 502,000
Total current liabilities 37,331,000 22,207,000
Deferred revenue, less current portion - 292,000
Deferred rent, less current portion 1,098,000 347,000
Commitments and contingencies
STOCKHOLDERS' EQUITY:
Preferred stock - $.001 par value; authorized 5,000,000 shares; issued and outstanding - 1,574,764 and 775,000, respectively 2,000 1,000
Common stock - $.001 par value; authorized 325,000,000 shares; issued and outstanding - 193,346,627 and 178,871,164, respectively 193,000 179,000
Additional paid-in-capital 512,464,000 470,785,000
Accumulated deficit (453,624,000 ) (403,266,000 )
Total stockholders' equity 59,035,000 67,699,000
TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY $ 97,464,000 $ 90,545,000
Contact: Jay Carlson, Peregrine Pharmaceuticals, 800-987-8256, info@peregrineinc.com
- - - - - - - - -
[ From 10-Q header: “As of Jul. 10, 2015, there were 199,934,918 shares of issuer’s common stock.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-15 iss. 7-14-15 http://tinyurl.com/ocrtkuj PR: http://tinyurl.com/nw2v5u6 (Cash 4-30-15=$68.0mm)
Latest 10Q 1-31-15 iss. 3-12-15 http://tinyurl.com/mwedt8w PR: http://tinyurl.com/q78oxvm (Cash 1-31-15=$55.2mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY15/Q4 (fye 4-30-15), per the 4-30-15 10-K (http://tinyurl.com/ocrtkuj ) issued 7-14-15. Deferred-Revs at 4-30-15, going fwd into FY’16/Q1 (q/e 7-31-15), total $6.6mm, up from the $5.8mm of Deferred-Revs at 1-31-15 that drove into FY’15/Q4.
Total Revs since May’06: ($129.2mm/Avid + $24.1mm/Govt + $2.1mm/Lic.) = $155.5mm
Avid’s Gross-Profit over last 3 qtrs: $9.4mm on revs of $21.2mm (GM% = 44%)
=> Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GM%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 5752 0 6148
Totals: 129212 24149 2124 155485 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY14 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
AVID “Total Services”:
AVID OUTPUT$ 3rd-PARTY + PEREGRINE = TOTAL-OUTPUT$
FY09 4-30-09 13mm 10mm $23mm #
FY10 4-30-10 13mm 17mm $30mm #
FY11 4-30-11 9mm 11mm $20mm @
FY12 4-30-12 15mm 11mm $26mm @
FY13 4-30-13 21mm ~10mm ~$31mm ^
LTM ended 1/2010 3rd/$15.3mm + Govt/$8.3mm + PPHM/$8.8mm = $32.4mm *
@SKing 3-18-2013 RothOC/DanaPT (Slide21) http://tinyurl.com/cebtwen
#SKing 7-12-2012 JMP/NYC Conf. (Slide27) http://tinyurl.com/csdclwb
*SKing 3-17-2010 RothOC/DanaPT Conf. (Slide18) http://tinyurl.com/ye9v7jq
^PLytle 7-11-2013 Qtly-CC “Avid did ~$10mm in equivalent services for Peregrine in FY13, which doesn’t get reflected into the fin. statements, it's eliminated in consolidation.”
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’14: 42,613,000 10K pg.54)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
7-30-15: HeartWare Reports $73.6mm in Q2’15 Revs, Driven By Strong Global Unit Growth…
• Record 773 HeartWare HVAD Systems sold worldwide in Q2’15, with record unit sales in both U.S. & Intl. markets
• U.S. revenue grew 16% to $42.9mm, compared to Q2’14
• Intl. rev. of $30.7mm, an increase of 10% from Q2’14 on a constant-currency basis
• Conference call today at 8:00amET
http://ir.heartware.com/phoenix.zhtml?c=187755&p=irol-newsArticle&ID=2072761
Did the thought occur to you that the 79% covers May’14-Apr15, whereas Revs for the new customer may not have begun (or been up to full speed) until as much as ½-way thru FY’15 – maybe even later??
= = = = = = =
9-9-14 PR: "Avid Bioservices started FY15 on a strong note, generating $5.5mm in contract mfg. revenue for Q1," said Paul Lytle, CFO of Peregrine. "In addition, Avid has been successful in expanding its client roster, while also continuing to evaluate mfg. options that would create new mfg. capacity for the potential commercial launch of bavituximab in addition to providing Avid with increased capacity for its clients."
http://tinyurl.com/ktrfswj
CEO Steve King, 9-9-14 C.Call:
“In addition of these development efforts, we also continue to see a solid performance from our wholly-owned manufacturing subsidiary Avid Bioservices that, coming off a record year, has already begun 2015 strongly with $5.5mm in 3rd-party contract revenue for the quarter and the expansion of our client base…”
http://tinyurl.com/ktrfswj
HTWR's Q2'15 Financials 7-30-15, CC @8amET
FRAMINGHAM, July 21, 2015: HeartWare Intl. (HTWR) has scheduled a conf. call to discuss its financial results for the three months ended June 30, 2015, at 8:00amET on Thursday, July 30, 2015. The Company plans to release the financial results prior to the conference call.
3's, this might help with understanding the "censoring" of patients... The only thing that actually makes up the shape of the K-M OS curve is confirmed deaths. The censored (lost to followup for whatever reason but confirmed death) patients are overlaid on the graph with 'x' tick marks, but don't affect the shape of the curve or the latest MOS estimate in a ongoing trial.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=110018191
JGMS, SK said “as those ARE reached”, not WERE… I’ll be posting the corrected SA transcript soon…
SK 7-14-15/CC (24:40):
“…With that, I will outline a number of upcoming milestones. Regarding our SUNRISE Ph3 2nd-Line NSCLC study, on track - we are on track for completing patient enrollment by yr-end 2015, with planned IDMC interim analyses at 33% & 50% of targeted overall survival events as those are reached, with estimated unblinding of the trial to occur approx. near calendar yr-end 2016…”
http://edge.media-server.com/m/p/jzohnozt
Going thru fixing the transcript errors, Joe Shan’s SUNRISE “positive feedback from investigators” comment (his 3rd prepared sentence) really hit me – the “and immunomodulating properties”. Am I inferring more than he is implying??
7-14-15/CC:
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
I’d like to first provide an update on the company’s ongoing Phase3 SUNRISE trial, which is evaluating the use of bavituximab in the treatment of non-squamous NSCLC. SUNRISE continues to progress according to plan with more than 150 active clinical centers spanning 14 countries. We remain on track to complete enrollment by the end of this calendar year. Meanwhile, we continue to receive positive feedback from investigators who are excited by both the safety profile and immunomodulating properties of bavituximab. As a reminder, SUNRISE is designed as a Phase 3 Registration Trial and has 2 planned interim analyses…
Replay: http://edge.media-server.com/m/p/jzohnozt
REVS since May’06: Tot=$155.5mm, Avid=$129.2mm(Q4 GP%=51%), #FT-Emps now=211, Cash(Apr30)=$68mm, OS-Shares(Jul10)=199.9mm
Updated PPHM REVS-BY-QTR TABLE, now thru FY15/Q4 (fye 4-30-15), per the 4-30-15 10-K (http://tinyurl.com/ocrtkuj ) issued 3-12-15. Deferred-Revs at 4-30-15, going fwd into FY’16/Q1 (q/e 7-31-15), total $6.6mm, up from the $5.8mm of Deferred-Revs at 1-31-15 that drove into FY’15/Q4.
Total Revs since May’06: ($129.2mm/Avid + $24.1mm/Govt + $2.1mm/Lic.) = $155.5mm
Avid’s Gross-Profit over last 3 qtrs: $9.4mm on revs of $21.2mm (GM% = 44%)
=> Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GM%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 51%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 5752 0 6148
Totals: 129212 24149 2124 155485 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY14 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’14: 42,613,000 10K pg.54)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
Cures4, the Duke/PPHM HIV Collab. (Haynes/Moody/Thorpe/PPHM/Affitech, CHAVI-NIH/CAVD-Gates) original 23-pg. article was published 4-2010 in the “Journal of Experimental Medicine” (JEM), as announced by the 4-5-10 Duke Medicine News article titled, “Scientists Identify How a Novel Class of Antibodies Inhibits HIV Infection”. Subsequent to that, in 12-2011 Duke patent app#20110318360 was published titled "Anti-Lipid Antibodies", describing using Peregrine’s PGN632 mab (fully-human, B2GPI-indep.) to inhibit HIV infection. In 3-2014 at the Keystone Conf. in Alberta, a PS-Targeting/HIV Poster by PPHM’s Cyril Empig & Duke/Peregrine Co-Authors B.Haynes/A.Moody/etc stated, ”We expanded the characterization of PGN632 in the current study; PGN631 & PGN633, ‘sister clones’ of PGN632, are also able to inhibit HIV-1 in the PBMC assay.”.
II: THE DUKE/HAYNES/CAVD/GATES/CHAVI/NIAID HIV COLLABORATION:
Duke's B.Haynes CAVD-Reports outline Thorpe's Role in the CAVD-Gates HIV-Vaccine Initiative: http://tinyurl.com/5xwjk4
…"Philip Thorpe is determining the role of lipid binding of anti-HIV antibodies and anti-phosphatidylserine (PS) autoantibodies to protection from HIV infection."
3-2014: Keystone Conf./Alberta PS-Targeting/HIV Poster by PPHM’s Cyril Empig & Duke/Peregrine Co-Authors Haynes/Moody/etc http://tinyurl.com/m6kuqpr
…”We expanded the characterization of PGN632 in the current study; PGN631 & PGN633, “sister clones” of PGN632, are also able to inhibit HIV-1 in the PBMC assay.”
12-29-11: Duke patent app#20110318360 "Anti-Lipid Antibodies" describes using PGN632 to inhibit HIV infection http://tinyurl.com/84vluj7
Sept'10: LWW article by Barton Haynes ref's 4-2010 Duke/PPHM JEM antiPS article: http://tinyurl.com/2bsgv8m
......"HIV vaccines after STEP: Strategies for Eliciting HIV-1 Inhibitory Antibodies"
4-11-10: CHAVI Newsletter highlights PPHM-Duke JEM article http://tinyurl.com/35eelvw
..."The study is significant because it shows that components of the adaptive immune system can activate antiviral innate immunity. Investigators believe that a vaccine that elicits polyreactive anti-PS antibodies could potentially be used to engage both the innate and adaptive immune response against HIV."
4-5-10: DukeHealth announces the 1st Duke/PPHM HIV Collab. article in JEM: http://tinyurl.com/yzr2tax
...Duke's Bart Haynes: "While the findings still have to be tested clinically, they do suggest a new way the immune system might be manipulated to thwart HIV."
...The full 4-5-10 JEM Duke-PPHM HIV article (23pgs): http://jem.rupress.org/content/207/4/763.full
Ph.3/NSCLC SUNRISE #Sites=161 a/o 7-9-15 (39US+122Intl)
• 5-2-14: 25 (All USA)
• 5-29-14: 62 (USA/29 Aus/3 Fr/1 Ger/6 Hungary/4 Korea/4 Spain/13 Taiwan/2)
• 6-9-14: 73 (USA/31 Aus/5 Fr/1 Ger/6 Hungary/4 Korea/4 Rus/7 Spain/13 Taiwan/2)
• 6-19-14: 88 (USA/34 Aus/5 Bel/6 Fr/1 Ger/6 Hungary/6 Italy/1 Korea/5 Rom/1 Rus/6 Spain/14 Taiwan/3)
• 6-25-14: 91 (USA/34 Aus/5 Bel/7 Fr/1 Ger/6 Hungary/6 Italy/0 Korea/7 Rom/1 Rus/6 Spain/14 Taiwan/4)
• 7-2-14: 100 (USA/32 Aus/6 Bel/7 Fr/4 Ger/8 Hungary/6 Korea/7 Rom/4 Rus/6 Spain/15 Taiwan/5)
• 7-9-14: 108 (USA/33 Aus/6 Bel/7 Fr/7 Ger/9 Hungary/7 Korea/7 Rom/6 Rus/6 Spain/15 Taiwan/5)
• 7-23-14: 115 (USA/32 Aus/6 Bel/7 Fr/9 Ger/9 Greece/5 Hungary/7 Korea/7 Rom/6 Rus/6 Spain/15 Taiwan/6)
• 7-28-14: 118 (USA/34 Aus/6 Bel/7 Fr/9 Ger/9 Greece/5 Hungary/7 Korea/7 Rom/6 Rus/6 Spain/15 Taiwan/7)
• 7-31-14: 121 (USA/34 Aus/6 Bel/7 Fr/9 Ger/10 Greece/6 Hungary/7 Korea/7 Rom/6 Rus/6 Spain/15 Taiwan/8)
• 8-7-14: 124 (USA/34 Aus/7 Bel/7 Fr/9 Ger/11 Greece/6 Hungary/7 Korea/7 Rom/6 Rus/6 Spain/16 Taiwan/8)
• 8-13-14: 128 (USA/34 Aus/7 Bel/7 Fr/10 Ger/12 Greece/6 Hungary/7 Italy/2 Korea/7 Rom/6 Rus/6 Spain/16 Taiwan/8)
• 8-20-14: 131 (USA/35 Aus/7 Bel/7 Fr/9 Ger/12 Greece/7 Hungary/7 Italy/2 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/9)
• 8-26-14: 133 (USA/36 Aus/7 Bel/7 Fr/9 Ger/12 Greece/7 Hungary/7 Italy/2 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10)
• 9-4-14: 135 (USA/37 Aus/7 Bel/7 Fr/9 Ger/12 Greece/7 Hungary/7 Italy/3 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10)
• 9-17-14: 138 (USA/37 Aus/8 Bel/7 Fr/9 Ger/12 Greece/7 Hungary/7 Italy/3 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/2)
• 9-23-14: 141 (USA/37 Aus/8 Bel/7 Fr/9 Ger/12 Greece/8 Hungary/7 Italy/3 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/4)
• 10-1-14: 143 (USA/37 Aus/8 Bel/7 Fr/9 Ger/12 Greece/8 Hungary/7 Italy/4 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/5)
• 10-17-14: 145 (USA/37 Aus/8 Bel/7 Fr/9 Ger/12 Greece/8 Hungary/7 Italy/5 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 11-7-14: 148 (USA/37 Aus/8 Bel/7 Fr/9 Ger/13 Greece/8 Hungary/7 Italy/7 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 12-1-14: 150 (USA/37 Aus/8 Bel/7 Fr/9 Ger/14 Greece/8 Hungary/7 Italy/8 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 12-19-14: 152 (USA/38 Aus/8 Bel/7 Fr/9 Ger/14 Greece/9 Hungary/7 Italy/8 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 2-17-15: 154 (USA/38 Aus/8 Bel/7 Fr/9 Ger/14 Greece/9 Hungary/7 Italy/10 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 3-23-15: 156 (USA/38 Aus/8 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/7 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 3-31-15: 158 (USA/38 Aus/8 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6)
• 7-9-15: 161 (USA/39* Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) – *NOTE: BaltMD was added 5-6-15.
BAVITUXIMAB PHASE III TRIAL: (see http://PeregrineTrials.com => http://www.SunriseTrial.com )
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582) (Start=Dec’13 Est PrimaryComp=Dec’16)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673 - 161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
…EU/EEA Registries: (pts world=582, pts/EU=345, planned EU sites=100) http://www.clinicaltrialsregister.eu/ctr-search/search?query=2013-003953-13
• GER: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/DE (planned sites=14, pts=43)
• SPAIN: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/ES (planned sites=16, pts=100)
• HUNGARY: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/HU (planned sites=9, pts=28)
• GREECE: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/GR (planned sites=9, pts=120)
• ITALY: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/IT (planned pts=55)
• BELGIUM: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/BE (planned sites=7 pts=31)
...EU= European Union (29 countries – see http://www.gov.uk/eu-eea ), EEA=European Economic Area (all the EU, plus Iceland, Liechtenstein, Norway)
…Regions where sites are planned: Australia, Belgium, France, Germany, Greece, Hungary, Italy, Korea, Romania, Russia., Spain, Taiwan, Ukraine, USA
- - - - - -
6-1-15: “Completion of enrollment is anticipated by calendar yr-end.” http://tinyurl.com/qxu4w2x
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
3-7-14: PR & Conf-Call: "first patients enrolled and dosed." http://tinyurl.com/kh9cnrg
1-6-14: FDA grants FAST TRACK status to Bavi in 2ndLine NSCLC http://tinyurl.com/l799ukk
12-30-13: Pivotal Ph.3 ‘SUNRISE’ NSCLC Trial Initiated (n=~600, sites>100) http://tinyurl.com/kdjb9qz
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
Contact: Jennifer Lai, MBA 1-855-291-SUNR (7867) lungcancertrial@peregrineinc.com
Peregrine Pharmaceuticals’ Clinical Development Group
• A.J. Leyco, RN - Associate Director, Clinical Trials
• Jennifer Lai, MBA, CCRA - Manager, Clinical Research
N40K, the latest Kenneth-Dart/13G I see shows 9,921,760sh a/o 12-13-14 (up from the ~8mm you stated, which was a/o 12-31-12)…
2-13-15: Kenneth-Dart’s (Eastern-Capital) PPHM stake now 9,921,760sh=5.4% a/o 12-31-14
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=110845168
11-8-10: Kenneth Dart (Eastern Capital) acquires 7.1% stake (3,961,568) in PPHM http://tinyurl.com/24qctos
...2-14-12: Kenneth Dart adds 3,460,192, now 7,421,760 (8.55% a/o 12-9-11) http://tinyurl.com/7bb26j4 (13G)
...2-13-13: Kenneth Dart adds 500,000, now 7,921,760 (5.9% a/o 12-31-12) http://tinyurl.com/b25a93j (13G)
...2-13-15: Kenneth Dart PPHM stake now 9,921,760 (5.4% a/o 12-31-14) http://tinyurl.com/k4nsfuu (13G)
- - - - - - -
All Eastern Capital 13G’s:
http://www.sec.gov/cgi-bin/browse-edgar?company=EASTERN+CAPITAL&match=&CIK=&filenum=&State=&Country=&SIC=&owner=exclude&Find=Find+Companies&action=getcompany
DD, is this how you define Credibility re:Peregrine?
By DEWDILIGENCE 12-11-12 7:23am BVals #104208
No trial with scrambled data has ever been salvaged to the satisfaction of the FDA, as far as I know; hence, what PPHM purports to be attempting is either delusion or misdirection, probably the latter.
PS: What PPHM purports to be attempting is completely different from what VRTX did with its CF data, which was never scrambled.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=82330647
= = = = = = = = = =
1-6-14: FDA grants FAST TRACK status to Bavi in 2ndLine NSCLC http://tinyurl.com/l799ukk
12-30-13: Pivotal Ph.3 ‘SUNRISE’ NSCLC Trial Initiated (n=~600, sites=100+) http://tinyurl.com/kdjb9qz
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...Steve King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…Rob Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
Avid’s John Haney – pretty solid guy (understatement) giving the talk today at the BIO-INTL. Conf. in Philly on the Design Strategy of the New Avid Expansion…

Details on Avid's presentation at the BioProcess Theater within the BioProcess Zone (BIO-INTL’15):
Title: "Designing & Implementing a New State-of-the-Art Single-Use Facility for Late Phase III & Commercial Production"
Speaker: John Haney, Senior Project Manager, Avid Bioservices, Inc.
Date: Wednesday, June 17, 2015
Time: 3:30PM - 3:55PM Eastern Time
[**John Haney: ex-Genentech(3yrs), ex-Pfizer(8yrs) https://www.linkedin.com/pub/john-haney/2a/b9a/188 ]
In addition, as part of the biopartnering forum, Avid Bioservices (Booth #533) will be conducting one-on-one partnering meetings with industry colleagues. If you are a company wishing to meet with Avid during BIO, please submit a partnering request through the BIO partnering system.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114684127
= = = = = = = = = = = =
AVID BIOSERVICES, Inc. (Peregrine's Mfg. Subsidiary): http://www.avidbio.com (Aug'13 ad: http://tinyurl.com/mhhs4e2 )
12-10-14: PPHM's Revs & Burns By Qtr Table, FY'07/Q1 thru FY'15/Q3 (q/e 1-31-15): http://tinyurl.com/q78oxvm ($146.2mm since 5-2006, incl.Govt)
12-10-14: Avid to Double Mfg. Capacity (“expanding client roster; potential commercial launch of bavituximab”) http://tinyurl.com/mmc3qgy & http://tinyurl.com/kmdgq8t
3-24-15: Avid Receives CMO Leadership Awards for Its Commitment to Innovation & Reliability http://tinyurl.com/psep47f
6-17/15/Avid PR: Launch of New State-of-the-Art-Facility highlighted at the June15-18 BIO Intl. Conf., incl. presentation by Avid Senior Project Manager JOHN HANEY at 3:30pmET 6-17-15…
6-17/15: Avid Bioservices to Highlight the Upcoming Launch of Its New State-of-the-Art-Facility at the BIO International
Convention
• Presentation in BioProcess Theater Program Will Highlight the Strategy for Designing and Building a Late Phase III and Commercial Production Facility
• Meet Our Biomanufacturing Experts at Booth #533
http://news.avidbio.com/press-releases/avid-bioservices-to-highlight-the-upcoming-launch-of-its-new-state-of-the-art-fa-1201872
TUSTIN, Jun 17, 2015: Avid Bioservices, Inc., a wholly owned subsidiary of Peregrine Pharmaceuticals, Inc., today announced its participation at the Biotechnology Industry Organization's (BIO) International Convention being held at the Philadelphia Convention Center, in Philadelphia, Pennsylvania, June 15-18, 2015. At this global biotechnology event, Avid will present in the BioProcess theater forum its strategy in designing and building a state-of-the-art commercial manufacturing facility that was designed to utilize 100% single-use, disposable technologies.
Avid's new facility is designed for late phase III and commercial production for biological products expressed from mammalian cell lines. The facility incorporates monolithic modular clean room walls, dedicated support utilities for each key processing step, and the latest in single-use technology, which can accommodate multiple single-use bioreactors, up to 2,000 liter scale. The facility is located adjacent to the company's current campus in Orange County, California and is expected to begin production by mid-2015.
"The annual gathering at BIO allows us to share the newest additions to Avid's services portfolio and highlight the upcoming launch of our new state of the art single-use facility. The new production facility was designed to be one of the first US late phase III clinical and commercial production facilities for the exclusive operation of single-use equipment to produce biologics," said Steve King, President and CEO at Avid Bioservices. "The new facility uses the most advanced modular cleanroom and single-use production systems and was designed to meet both US and international regulatory requirements. We have already seen tremendous interest for production in the new facility, both from new and existing clients and we look forward to launching production in the near future."
The presentation in the BioProcess Theater program will enable attendees to hear about the strategy and considerations that went into designing this new facility for late phase III and commercial production and the flexibility offered by the modular cleanroom bioproduction suites that allowed the company to construct this new facility in a shorter timeframe than the typical facility build-out and commissioning.
Details on Avid's presentation at the BioProcess Theater within the BioProcess Zone:
Title: "Designing & Implementing a New State-of-the-Art Single-Use Facility for Late Phase III & Commercial Production"
Speaker: John Haney**, Senior Project Manager, Avid Bioservices, Inc.
Date: Wednesday, June 17, 2015
Time: 3:30PM - 3:55PM Eastern Time
[**John Haney: ex-Genentech(3yrs), ex-Pfizer(8yrs) https://www.linkedin.com/pub/john-haney/2a/b9a/188 ]
In addition, as part of the biopartnering forum, Avid Bioservices (Booth #533) will be conducting one-on-one partnering meetings with industry colleagues. If you are a company wishing to meet with Avid during BIO, please submit a partnering request through the BIO partnering system.
For additional information on the BIO International Convention, please visit: http://convention.bio.org/
ABOUT AVID BIOSERVICES
Avid Bioservices provides a comprehensive range of high quality cGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With over 15 years of experience producing monoclonal antibodies and recombinant proteins in batch, fed-batch and perfusion modes, Avid's services include cGMP clinical and commercial product manufacturing, purification, bulk packaging, final product filling, stability testing and regulatory strategy, submission and support. The company also provides a variety of process development activities, including cell line development and optimization, cell culture and feed optimization, analytical methods development and product characterization. For more information about Avid, please visit http://www.avidbio.com .
ABOUT PEREGRINE PHARMACEUTICALS
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Contact: Kelly Pisarev Lord (800) 987-8256 http://www.avidbio.com
= = = = = = = = = = = =
AVID BIOSERVICES, Inc. (Peregrine's Mfg. Subsidiary): http://www.avidbio.com (Aug'13 ad: http://tinyurl.com/mhhs4e2 )
12-10-14: PPHM's Revs & Burns By Qtr Table, FY'07/Q1 thru FY'15/Q3 (q/e 1-31-15): http://tinyurl.com/q78oxvm ($146.2mm since 5-2006, incl.Govt)
12-10-14: Avid to Double Mfg. Capacity (“expanding client roster; potential commercial launch of bavituximab”) http://tinyurl.com/mmc3qgy & http://tinyurl.com/kmdgq8t
3-24-15: Avid Receives CMO Leadership Awards for Its Commitment to Innovation & Reliability http://tinyurl.com/psep47f
PPHM SAB’r Dr.Xianming.Huang(Brekken Lab) speaks tonight at Gordon-Research-Conf., Biddeford ME
Jun14–19 2015: “GRC: Apoptotic Cell Recognition & Clearance: Physiological Significance & Pathological Consequences”, Biddeford ME
GRC = Gordon Research Conferences (Univ. of New England) http://www.grc.org
”This 7th meeting of the Apoptotic Cell Recognition & Clearance Gordon Research Conf. will explore molecular details of apoptotic cell recognition and clearance mechanisms throughout evolution, including apoptotic immune regulation. We also will focus on the pathogenic subversion of mechanisms of apoptotic cell clearance. This meeting will bring together investigators who are at the forefront of these fields to present and discuss new findings & important issues…”
• Peregrine is the only corporate contributor to this conference: ”Generous financial support from Peregrine Pharmaceuticals is in memory of Dr. Philip E. Thorpe.” [Dr. Thorpe spoke & chaired at prior GRC’s many times before his passing on 3-30-13.]
Conf: http://www.grc.org/programs.aspx?id=13127
JUNE15 SESSION 7:30-9:30pm “Consequences of Lipid Alterations for Apoptotic Cell Clearance”
…Discussion Leader: Christopher Gregory (Univ. of Edinburgh, UK)
• 7:30-7:55pm Ian Dransfield (Univ/Edinburgh UK), "Protein S Binding to Apoptotic Cells & Subsequent Phagocytosis by Macrophages"
• 8:10-8:35pm Raymond Birge (Rutgers NJ Med-School), "Apoptotic Cell Recognition Receptors, Tyro3/Axl/Mer, Demonstrate Distinct Patterns of Ligand Recognition & Biological Function"
• June15 8:50-9:15pm Xianming Huang (UTSW-MC/Dallas – PPHM SAB/Brekken-Lab), "Phosphatidylserine-Targeting Antibodies Overcome Tumor Immunosuppression & Synergize with Immune Checkpoint Blockade"
= = = = = = = = = = = =KNOWN UPCOMING:
Jun15: GRC’s “7th Apoptotic Cell Recognition & Clearance Conf.”, Biddeford ME http://www.grc.org/programs.aspx?id=13127
…8:50-9:15pm: Xianming Huang (UTSW), “PS-Targeting Antibodies Overcome Tumor Immunosuppression & Synergize with Immune Checkpoint Blockade"
~Jul13: FY'15/Q4 (fye 4-30-15) Financials & Conf. Call http://ir.peregrineinc.com/events.cfm
Jun15-18/Avid/Booth533: BIO Intl. Convention, Philadelphia http://convention.bio.org/2015
Aug26: CHI’s 10th Annual Immunotherapy & Vaccine Summit (IMVACS), Boston http://tinyurl.com/lfdoksl
… 9-9:30 Jeff T. Hutchins (VP/Preclin-Res.): “Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
Sep6-9: IASLC’s 16th World Conf. on Lung Cancer”, Denver http://tinyurl.com/qdteoc8
…Peregrine Exhibiting, Booth #1724 (KOL/SAB Dr. David Carbone will be President of IASLC eff. 9-2015)
Oct26-29/Avid/Booth909: IBC's BioProcess Intl. Conf. & Exhib, Boston http://www.ibclifesciences.com/BPI
Dec7-10/Avid/Booth307: IBC's Antibody Eng. & Therapeutics, SanDiego http://www.ibclifesciences.com/AntibodyEng
Mar15-17 2016: “Immune Checkpoint Inhibitors Conf.”, Boston http://immune-checkpoint.com (Peregrine is 1 of 4 Corp. Sponsors)
Opdivo works in only ~20-35% of NSCLC Patients, those that express PD-L1 > 10%. In BMY’s Checkmate-057 NSCLC trial, Opdivo DID NOT increase survival vs. DOCE in the 64% of All Pts that didn’t express PD-L1 > 10% (see BMY’s K-M Charts below). Thus, the window of opportunity for Bavi is essentially 2/3+ of the NSCLC market. Of course, the other 1/3 of the patients where Opdivo works well might live even longer if Bavi were added to the mix, or possibly Bavi will allow Opdivo at reduced dosages to cut back on its considerable side-effects…
- - - - - - -
PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit 20-25% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Bavi’s goal is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = = = = = =
BMY’s Ph3 Opdivo NSCLC trial – look at the KM-Curve and OS #’s for those pts that express < 10% PD-L1. The OS separation in the tail just collapses and Opdivo (nivolumab) dead standstill with Doce in MOS => That’s ~64% of ALL the pts in the n=582 trial!
http://investor.bms.com/files/doc_presentations/2015/ASCO-Investor-Presentation-June-1-FINAL_v001_p9iq7v.pdf (pg.25)


= = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
Follow-up on BMY’s Ph3 Opdivo NSCLC trial – look at the KM-Curve and OS #’s for those pts that express < 10% PD-L1. Tthe OS separation in the tail just collapses and Opdivo (nivolumab) dead standstill with Doce in MOS => That’s ~64% of ALL the pts in the n=582 trial! …Thus the window of opportunity for Bavi is essentially 2/3 of the NSCLC market. Of course, the other 1/3 of the patients where Opdivo works well might live even longer if Bavi were added to the mix….
http://investor.bms.com/files/doc_presentations/2015/ASCO-Investor-Presentation-June-1-FINAL_v001_p9iq7v.pdf (pg.25)


= = = = = = = = = = = = = = = = = = = =
5-30-15: PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit 20-25% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Enter BAVI, stage left, whose objective is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
Peregrine Exhibiting Sept6-9 at IASLC World Conf. in Denver – at that point PPHM KOL/SAB’r Dr. David Carbone will have become President of The IASLC…
Sept6-9 2015: “IASLC’s 16th World Conf. on Lung Cancer”, Denver
http://wclc2015.iaslc.org/
“The World Conference on Lung Cancer (WCLC) is the world’s largest meeting dedicated to lung cancer and other thoracic malignancies. More than 7,000 delegates come from more than 100 countries to discuss the latest developments in thoracic malignancy research. Attendees include surgeons, medical oncologists, radiation oncologists, pulmonologists, radiologists, pathologists, epidemiologists, basic research scientists, nurses, allied health professionals, advocates, and patients.”
http://www.iaslc.org/events/16th-world-conference-lung-cancer
2015 Exhibitor: Peregrine Pharm. (1 of 39) - booth #1724 (adjacent to Abbvie)
• RECALL: PPHM KOL/SAB’r Dr. David Carbone becomes President of IASLC 9/2015 ( https://www.iaslc.org/about-us/board ). “…I also personally find it rewarding to participate in the global fight against cancer, and am honored to have been elected President of the IASLC, eff. Sept. 2015." http://www.supportohiostate.org/site/TR?px=1009121&fr_id=1151&pg=personal
• IASLC Faculty & Staff Disclosures: Heather A. Wakelee, MD - Grant/Research Support: Genentech/Roche, Lilly, Pfizer, Novartis, Exelixis, Xcovery, AstraZeneca, Clovis, Regeneron; Consultant: Peregrine. http://www.gotoper.com/conferences/ilc/meetings/15th-international-lung-cancer-congress “Associate Professor of Medicine, Div. of Oncology; Faculty Director, Cancer Clinical Trials Office; Co-leader, Lung Cancer Disease Mgt Group, Stanford Univ. School of Medicine, Stanford CA”
= = = = = = = =NEW ALSO:
Mar15-17 2016: “Immune Checkpoint Inhibitors Conf.”, Boston
http://immune-checkpoint.com
Organized by: Hanson Wade http://hansonwade.com “Among our many events, we bring together world leading experts who are delivering brand new medical treatments…”
Peregrine Pharm. is 1 of 4 Corp. Sponsors.
- - - - - - - - - - - -
KNOWN UPCOMING AT THIS POINT:
Jun15: GRC’s “7th Apoptotic Cell Recognition & Clearance Conf.”, Biddeford ME http://www.grc.org/programs.aspx?id=13127
…8:50-9:15pm: Xianming Huang (UTSW), “PS-Targeting Antibodies Overcome Tumor Immunosuppression & Synergize with Immune Checkpoint Blockade"
~Jul13: FY'15/Q4 (fye 4-30-15) Financials & Conf. Call http://ir.peregrineinc.com/events.cfm
Jun15-18/Avid/Booth533: BIO Intl. Convention, Philadelphia http://convention.bio.org/2015
Aug26: CHI’s 10th Annual Immunotherapy & Vaccine Summit (IMVACS), Boston http://tinyurl.com/lfdoksl
… 9-9:30 Jeff T. Hutchins (VP/Preclin-Res.): “Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
Sep6-9: IASLC’s 16th World Conf. on Lung Cancer”, Denver http://wclc2015.iaslc.org
…Peregrine Exhibiting, Booth #1724 (KOL/SAB Dr. David Carbone will be Pres. of IASLC eff. 9-2015)
Oct26-29/Avid/Booth909: IBC's BioProcess Intl. Conf. & Exhib, Boston http://www.ibclifesciences.com/BPI
Dec7-10/Avid/Booth307: IBC's Antibody Eng. & Therapeutics, SanDiego http://www.ibclifesciences.com/AntibodyEng
Mar15-17 2016: “Immune Checkpoint Inhibitors Conf.”, Boston http://immune-checkpoint.com
…Peregrine Pharm. is 1 of 4 Corp. Sponsors.
.
.

6-8-15, NATURE-REVIEWS Article – Dr. Thorpe’s PS-Viral-Research recognized again by Paris/UK authors who conclude, “these results suggest that future efforts should be directed at exploiting viral apoptotic mimicry, PS exposure and PS receptors for the development of antiviral therapies."…
6-8-15: “Viral Apoptotic Mimicry” - Nature Reviews Microbiology (Advance Online Pub.)
Ali Amara, PhD (Saint-Louis Hosp., Paris); Jason Mercer, PhD (Univ. College London, UK)
http://www.nature.com/reviews/micro
http://www.nature.com/nrmicro/journal/vaop/ncurrent/abs/nrmicro3469.html
INTRO: “Viral apoptotic mimicry, defined by the exposure of phosphatidylserine on the pathogen surface, is emerging as a common theme used by enveloped viruses to promote infection. In this Progress article, Amara & Mercer discuss how viruses acquire phosphatidylserine and how this mimicry might facilitate cell entry and evasion of the immune response.”
LAST SENTENCE: “Collectively, these results suggest that future efforts should be directed at exploiting viral apoptotic mimicry, phosphatidylserine exposure and phosphatidylserine receptors for the development of antiviral therapies."
- - - -
REF#31: MM_Soares, SW_King, PE_Thorpe, “Targeting Inside-Out Phosphatidylserine as a Therapeutic Strategy for Viral Diseases” Nat. Med. 14, 1357–1362 (2008)**.
**[11-24-08: Dr.Thorpe publishes in Dec.2008 NATURE Medicine on BaviAV vs. Lassa/CMV: http://tinyurl.com/9ktvsb
...Dr. Thorpe, "This is a whole new strategy for making antiviral drugs."… CEO S.King, "We think it has tremendous potential."
...Dr. B.Haynes(Duke), "Targeting a host cell lipid such as PS as an anti-viral strategy is an intriguing concept that may have relevance for new therapeutic and possibly prophylactic innovations in a number of virus infections."]

- - - - -AUTHORS:
• Ali Amara obtained his PhD from the Univ. of Bordeaux, France, and conducted his postdoctoral research at the Pasteur Institute, Paris, France. He is now the Institut National de la Sante et de la Recherche Medicale (INSERM) Research Director of the Biology of Emerging Viruses group in the Saint-Louis Hospital, Paris. His research aims to decipher how viruses enter target cells and how they exploit the host cell machinery to accomplish their infectious life cycle. He currently uses mosquito-borne viruses (dengue virus, yellow fever virus and West Nile virus) as models. Ali Amara's homepage: http://www.jeanbernard.univ-paris-diderot.fr/en/research-teams/biology-of-emerging-viruses-ali-amara
• Jason Mercer obtained his PhD in the laboratory of Paula Traktman at the Medical College of Wisconsin, Milwaukee, and conducted his postdoctoral research in the laboratory of Ari Helenius at ETH Zurich, Switzerland. He is now Associate Professor of Virus Cell Biology in the Medical Research Council Laboratory for Molecular Cell Biology at Univ. College London, UK. His research group focuses on deciphering the complex interactions between poxviruses and their host cells during infection. Jason Mercer's homepage: http://www.ucl.ac.uk/lmcb/research-group/jason-mercer-research-group
= = = = = = = = = = =PPHM/THORPE VIRAL NEWS:
10-15-14: Public-Health-England/PPHM article on Bavi/Ebola in Hindawi’s Jrnl-of-Immunology-Res http://tinyurl.com/mg3va35 (also see: http://tinyurl.com/ofa5zlt )
3-2014: Keystone Conf./Alberta PS-Targeting/HIV Poster by PPHM’s Cyril Empig & DUKE/Peregrine Co-Authors Haynes/Moody/etc http://tinyurl.com/m6kuqpr
…”We expanded the characterization of PGN632 in the current study; PGN631 & PGN633, “sister clones” of PGN632, are also able to inhibit HIV-1 in the PBMC assay.”
6-2013: NIAID is “particularly interested how PS & PE expression… which is key to regulation of the immune response.” http://tinyurl.com/mvzqn26
…the above from John Coligan’s (PhD, Chief, Receptor Cell Biology Section) NIAID lab webpage.
11-16-12: 'Antiviral-Research' article by Bavi-AV collaborator HPA/UK & PPHM researchers on Bavi vs. EBOLA http://tinyurl.com/aa98xl3
…"Virus induced externalization of PS raises the possibility that PGN401 (Bavi) has broad spectrum antiviral activity."
11-16-12: 'Parasite Immunology' article by Dr. Phillip Thorpe & Brazilian Bavi-AV collaborators on AntiPS vs. Leishmania http://tinyurl.com/a8xtmny
..."This study provides new information regarding the mechanism of immune suppression in Leishmania infection."
12-29-11: Duke patent app#20110318360 "Anti-Lipid Antibodies" describes using PGN632 to inhibit HIV infection http://tinyurl.com/84vluj7......"HIV vaccines after STEP: Strategies for Eliciting HIV-1 Inhibitory Antibodies"
4-11-10: CHAVI Newsletter highlights PPHM-Duke JEM article http://tinyurl.com/35eelvw
..."The study is significant because it shows that components of the adaptive immune system can activate antiviral innate immunity. Investigators believe that a vaccine that elicits polyreactive anti-PS antibodies could potentially be used to engage both the innate & adaptive immune response against HIV."
4-5-10: DukeHealth announces the 1st Duke/PPHM HIV Collab. article in JEM: http://tinyurl.com/yzr2tax
...Duke's Bart Haynes: "While the findings still have to be tested clinically, they do suggest a new way the immune system might be manipulated to thwart HIV."
...The full 4-5-10 JEM Duke-PPHM HIV article (23pgs) posted to PPHM's homepage: http://tinyurl.com/ydqdk59
HTWR Webcast 6-11-15 8amPT at Goldman-Sachs Conf.
HeartWare Presentation At The Goldman Sachs 36th Annual Global Healthcare Conference To Be Webcast
FRAMINGHAM, June 4 2015: HeartWare International (NASDAQ: HTWR), a leading innovator of less invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure, today announced that its presentation at the Goldman Sachs 36th Annual Global Healthcare Conference will be webcast live on Thur. June 11 2015 at 8amPT (11amET). The conference is being held June 9-11 at Terranea in Rancho Palos Verdes, CA...
PD-1/PD-L1 blockade therapies like Opdivo/Keytruda benefit ~20% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Enter BAVI, stage left, whose objective is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan Kettering’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
- - - - - - - - - - - - -
5-29-15/PR: Peregrine & Sloan Kettering Enter Into Collaboration
=> http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
ASCO’15(May30-Jun2): PPHM’s 3 Abstracts & May31/6pmCT I-O Roundtable Webcast (7 on I-O panel, incl. 3 Sloan-Kettering Researchers)
=> http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114181197
6-1-15/PR’s: ASCO’15 and Planned Expansion of Bavi Clinical-Pgm => Ph2 Bavi+Opdivo/2L-NSCLC(Squamous+NonSquamous), Ph2/3 Bavi+Doce/PAC/Her2-Breast), Sunrise Enrollment anticipated Y/E’15, Avid Exp. s/b done 7-2015, Cash 4-30-15=$68mm…
6-1-15: Peregrine Pharmaceuticals Provides Update on Planned Expansion of Bavituximab Clinical Program
• Phase II Trial to Evaluate Combination of Bavituximab and Opdivo® (Nivolumab) in Non-Small Cell Lung Cancer
• Phase II/III Combination Trial to Advance Bavituximab with Chemotherapy Combinations in HER2 Negative Breast Cancer
• New Studies Expected to be Underway as Phase III SUNRISE Trial in Lung Cancer Completes Enrollment by Calendar Year-End
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915699
TUSTIN, June 1, 2015: Peregrine Pharmaceuticals, Inc. (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on oncology and the treatment of lung and breast cancers through the development of bavituximab, a novel immunotherapy currently in Phase III, today provided a corporate update on its immuno-oncology pipeline, its wholly-owned contract manufacturing business, Avid Bioservices, as well as anticipated upcoming milestones. This update outlines the expansion of the bavituximab clinical pipeline with a focus on exploring expanded indications and combinations in lung and breast cancers. These new trials build on recently published clinical data of bavituximab in combination with paclitaxel in HER2 negative breast cancer and clinical translational and preclinical data presented at the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting that support the potential combination of bavituximab with the anti-PD-1 checkpoint inhibitor, nivolumab (marketed as Opdivo®). These clinical trials are expected to begin enrollment later this year.
"With many exciting near-term opportunities for Peregrine and Avid, we are pleased to share several strategic decisions aimed at expanding the potential clinical indications for bavituximab. These new trials represent large market opportunities for the bavituximab clinical program that can add significant value," said Steven King, President and CEO of Peregrine. "As we near completion of enrollment in the SUNRISE pivotal Phase III trial by year-end, we are looking to expand bavituximab's market potential and market position in key indications that are supported by strong clinical, translational, and preclinical data. Taken together with the research collaboration we announced last week, we believe we are setting the stage for many significant value driving events throughout 2015 and into 2016."
SUNRISE Phase III Pivotal Trial
The company's Phase III SUNRISE (Stimulating ImmUne RespoNse thRough BavItuximab in a PhaSE III Lung Cancer Study) is an approximately 600 patient trial that continues to enroll at over 150 sites worldwide. Completion of enrollment is anticipated by calendar year-end. This Phase III, randomized, double-blind, placebo-controlled clinical trial is designed to evaluate the safety, tolerability and efficacy of bavituximab as a second-line treatment in patients with non-squamous, non-small cell lung cancer (NSCLC). Enrollment is proceeding according to plan with two planned interim efficacy analyses which will be reviewed by the trial's Independent Data Monitoring Committee (IDMC). The first interim analysis, which will be conducted when 33% of the targeted overall survival events are reached, is for futility and the second interim analysis, for futility or superiority, will be conducted at 50% of events. As these analyses are event driven, the exact timing of each is unknown, however the company plans to provide updates as these events are reached. For additional information about the SUNRISE trial, please visit http://www.sunrisetrial.com or ClinicalTrials.gov using the Identifier NCT01999673 [ http://www.clinicaltrials.gov/ct2/show/NCT01999673 ].
Beyond SUNRISE: Exploring Checkpoint Inhibitor Combinations in Lung Cancer
Joe Shan said: "Following recent encouraging preclinical data supporting the combination of bavituximab with immune checkpoint inhibitors, today we are supplementing our strategy in lung cancer with the second clinical trial to emerge from our Immuno-Oncology Development Program, a combination trial of bavituximab with nivolumab, an immune checkpoint inhibitor of PD-1. Specifically, recent preclinical data show that the addition of bavituximab could result in turning patients that do not respond to monotherapy into patient responders. This supports our long held knowledge of the synergistic properties of the combination of bavituximab and chemotherapies, thus making this new trial the next logical step."
The company plans to initiate an open-label multi-center, randomized Phase II trial of the anti-PD-1 monoclonal antibody nivolumab (marketed as Opdivo®) versus nivolumab plus bavituximab in patients with previously treated locally advanced or metastatic NSCLC. Enrollment will include patients with squamous and non-squamous NSCLC who have not received a prior PD-L1 or PD-1 inhibitor. The primary endpoint of this trial will be overall response rate (ORR) with secondary endpoints including tumor response and duration, progression free survival, overall survival (OS) and safety. The trial is in the final stages of design and the details of which will be made available once details are completed. Trial initiation is anticipated in the second half of 2015.
Leveraging Positive Data in Breast Cancer
"Today we are pleased to be detailing our strategy in breast cancer which is built upon very promising data which were recently published in the peer-reviewed journal Cancer Medicine," said Joseph Shan, VP of Clinical & Regulatory Affairs at Peregrine Pharmaceuticals. "The clinical development pathway that we are outlining today is based upon the strong clinical experience that supports synergies between bavituximab and docetaxel and paclitaxel, our deep and consistently positive clinical experience in breast cancer to date, as well as the support of leading breast cancer physicians."
The company plans to initiate a Phase II/III open-label trial of physician's choice of either docetaxel or paclitaxel with or without bavituximab in patients with locally advanced or metastatic HER2 negative breast cancer. The primary endpoint for the Phase II trial will be ORR. Results from this trial are expected to inform decisions on future studies in breast cancer and add value to the overall program. The initiation of this trial is anticipated in the second half of 2015.
Fiscal Year End Cash Position
Peregrine today reported $68 million in cash and cash equivalents, as of fiscal year ended April 30, 2015. The company will review full financial results for the fourth quarter and fiscal year 2015 during its quarterly call on Tuesday, July 14, 2015 after the close of markets, details of which will be included in a forthcoming press release.
Avid Bioservices' Expansion on Track and Strongly Positioned to Meet Increased Client Demand
In December 2014, the company announced the expansion of Avid's Current Good Manufacturing Practice (cGMP) current manufacturing capacity with a build out that is utilizing an existing 40,000 square foot facility located adjacent to the company's current campus. Designed to more than double Avid's current manufacturing capacity, this state-of-the-art biotechnology facility will employ an innovative and flexible modular clean room design and the latest in single-use technologies to meet the growing needs of Avid's existing and future clients as well as the planned commercialization of bavituximab. Avid's new facility is anticipated to be ready for cGMP production of biotechnology products in July 2015.
Added Steve King: "We are very pleased with the rapid progress by which this expansion is advancing as this is a key component to further demonstrating Avid as a leading provider of high quality biotechnology contract manufacturing services. The design and the features that we have employed in this expansion will truly be an integral part to the future growth of this unique part of our business. We look forward to sharing specific details on the expansion upon its formal unveiling later this year."
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Safe Harbor <snip>
Opdivo® is a registered trademark of Bristol-Myers Squibb Company
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = = = =
6-1-15/PR: Peregrine’s 3 ASCO’15 Presentations (NSCLC, Melanoma, Breast, Liver)…
6-1-15: Data Presentations at ASCO Demonstrate the Ability of Peregrine Pharmaceuticals' Bavituximab to Activate Tumor Targeting Immune Cells in PD-L1 Negative NSCLC Tumors
• New Data Show that Bavituximab Alone and in Combination with Docetaxel Elicits a Tumor-Specific Immune Response in PD-L1 Negative Tumors Extracted from NSCLC Patients
• PS-Targeting Antibodies Combined with Anti-PD-1 Reverses Tumor Immunosuppression and Activates Tumor Fighting CD8+ Immune Cells in Models of Melanoma and Breast Cancer
• Encouraging Data in Patients with Advanced Liver Cancer Warrants Further Clinical Study
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915719
TUSTIN, June 1, 2015: Peregrine Pharmaceuticals, Inc. (Nasdaq:PPHM) (Nasdaq:PPHMP), a biopharmaceutical company focused on oncology and the treatment of lung and breast cancers through the development of bavituximab, a novel immunotherapy currently in Phase III, today announced the presentation of preclinical and clinical translation data highlighting its phosphatidylserine (PS) targeting platform and bavituximab at the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting, being held May 29-June 2, 2015 in Chicago, Illinois.
"These presentations bridge the preclinical and clinical portions of our Immuno-Oncology Development Program and support our recently announced strategic decision to expand our clinical program aimed at the development of the most promising potential immunotherapy combinations with bavituximab," said Jeff Hutchins, VP of Preclinical Development of Peregrine. "The data generated to date reinforces the potential of bavituximab to enhance current immuno-oncology treatment modalities aimed at creating a greater group of patients responding to currently available therapies."
In poster #385 titled, "Activation of CD8+ Tumor Infiltrating Lymphocytes by Bavituximab in a 3D Ex Vivo System of Lung Cancer Patients", researchers from Nilogen Oncosystems [=Moffit Startup: http://otmc.moffitt.org/startups.aspx ] and H. Lee Moffitt Cancer Center presented initial data from a pilot translation study analyzing tumor tissue from six lung cancer patients to evaluate the immunomodulatory effects of bavituximab in a human ex vivo model of non-small cell lung cancer (NSCLC). New data generated from additional assays further validates previous data showing that ex vivo drug treatment with bavituximab, alone and in combination with docetaxel, elicits an immune response in tumors from NSCLC patients with negative PD-L1 and low PD-1 expression. Specifically, data showed activation of tumor infiltrating lymphocytes (TILs) by polarization of the tumor microenvironment from immunosuppressive to immuno-stimulatory. Results in this pilot translational study also identified PD-L1 and PD-1 expression as a potential biomarker of response to bavituximab treatment, suggesting that the interruption of the PD-1/PD-L1 axis may enhance the bavituximab effect in lung cancer.
[POSTER: http://www.peregrineinc.com/images/stories/pdfs/asco_2015_lung.pdf ]
"These preliminary gene profiling data are consistent with previously published data on bavituximab's immunotherapeutic mechanism of action," said Dr. Soner Altiok, MD, PhD, Chief Scientific Officer at Nilogen Oncosystems and Senior Member at the H. Lee Moffitt Cancer Center. "Building upon encouraging translational findings of a cytokine profile previously reported, these new data support the finding that M1 polarization of tumor associated macrophages is involved in bavituximab-mediated activation of tumor infiltrating lymphocytes in the ex vivo model of lung cancer."
In poster #386 titled, "Phosphatidylserine Targeting Antibody in Combination with anti-PD-1 Antibody Treatment Activates Infiltrating T Lymphocytes of the Spleen and Tumor Microenvironment in Pre-Clinical Models of Melanoma and Breast Cancer", researchers from The University of Texas Southwestern Medical Center in Dallas, Texas led by Xianming Huang, PhD, found that blocking PS with PS-Targeting antibody enhances the anti-tumor activity of combination therapies including anti-PD-1 and anti-CTLA-4 antibodies in an immune competent model of breast cancer and in preclinical models of melanoma. In the breast cancer model, researchers found that the combination of PS blockade and an anti-PD-1 antibody promoted strong and localized anti-tumor responses without the side-effects of systemic immune activation. In models of melanoma, the combination of PS blockade with either an anti-PD-1 or anti-CTLA-4 antibody showed significantly superior tumor growth inhibition over single treatment, with many subjects achieving complete tumor regressions. No toxicity was observed in any of the treatment groups following multiple treatment doses.
[POSTER: http://www.peregrineinc.com/images/stories/pdfs/asco_2015_melanoma_breast.pdf ]
Added Dr. Hutchins: "These data support our view of bavituximab as an immunomodulatory treatment in PD-1 sensitive and anti-PD-1 resistant/unresponsive tumors. We look forward to the execution of additional clinical trials aimed at further exploring these potential combinations."
In poster #220 titled: "A phase I/II study of Bavituximab and Sorafenib in Advanced Hepatocellular Carcinoma (HCC)", researchers led by Adam Yopp, M.D., Assistant Professor of Surgery at The University of Texas Southwestern Medical Center in Dallas, Texas found that the combination of bavituximab and sorafenib is associated with an improved time to progression (TTP) of 6.7 months, a disease specific survival (DSS) of 8.7 months, a disease control rate (DCR) of 58% (22 out of 58 patients) and a 4-month progression-free survival (PFS) of 62%. Two patients (5%) achieved a partial response according to Response Evaluation Criteria In Solid Tumors (RECIST). The secondary endpoint of median overall survival (OS) was 6.2 months. The trial's patient population had unfavorable disease biology as demonstrated by a high rate of previous treatment and macrovascular invasion. The combination of bavituximab and sorafenib was well-tolerated in patients with advanced HCC with no indications of autoimmune adverse events that have been seen with other checkpoint immunotherapies.
[POSTER: http://www.peregrineinc.com/images/stories/pdfs/asco_2015_hcc_utsw_y_.pdf ]
A link to copies of these presentations can be found on the home page of Peregrine's website at http://www.peregrineinc.com .
Webcast Replay: Roundtable Discussion "Raising the Immuno-Oncology Bar"
Last night, Peregrine hosted a roundtable discussion of immunotherapy thought leaders in conjunction with the 2015 Annual Meeting of the American Society of Clinical Oncology (ASCO) in Chicago, Illinois. A link to the replay of this event titled: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors" can be found on the home page of Peregrine's website at http://www.peregrineinc.com . [ http://edge.media-server.com/m/p/h2toyjv4 ]
About Bavituximab: A Targeted Investigational Immunotherapy
Scientific research has shown that tumors evade immune detection due partly to the expression of phosphatidylserine, or PS, a highly immunosuppressive molecule. Peregrine's immuno-oncology development program has developed bavituximab, an investigational monoclonal antibody that targets and binds to PS, blocking its immunosuppressive effects while activating tumor fighting immune cells, thus enabling the immune system with the ability to better recognize and fight cancer. Bavituximab's immune-stimulatory mechanism-of-action data is the subject of a manuscript published in the October 2013 issue of the American Association for Cancer Research's (AACR) peer-reviewed journal, Cancer Immunology Research. Bavituximab is currently being evaluated in several solid tumor indications, including non-small cell lung cancer (the SUNRISE Phase III trial), breast cancer, liver cancer, rectal cancer and advanced melanoma. In January 2014, bavituximab received Fast Track designation by the U.S. Food and Drug Administration (FDA) for the potential second-line treatment of patients with non-small cell lung cancer.
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., 800-987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = = = = = = = =
Event Brochure for Peregrine’s 5-31-15 ASCO’15 I-O Roundtable – sent to me my CK@IR…
Sunday 5-31-15 6pmCT: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
…7 panel members, incl. 3 Mem. Sloan Kettering researchers – recall the 5-29-15 announced collab. with MSK “to Investigate Novel PS-Targeting Immunotherapy Combinations”…
WEBCAST Replay: http://edge.media-server.com/m/p/h2toyjv4

= = = = = = = = = = = =
5-27-15/PR: Peregrine Pharmaceuticals Announces Webcast of Immunotherapy Roundtable at ASCO Annual Meeting
• Immunotherapy Thought Leaders to Discuss Next Wave of Therapies Aimed at Increasing the Number of Patients That Respond to Immuno-Oncology Combination Therapies
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915098
TUSTIN, May 27, 2015: Peregrine Pharmaceuticals (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology (I-O) agent in Phase III development for the treatment of lung cancer, invites investors and the general public to listen to a webcast of its roundtable discussion in conjunction with the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting [ http://am.asco.org ]. This roundtable is comprised of key opinion leaders in the field of Immunotherapy who will discuss novel I-O combination therapies that may improve patient outcomes.
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6:00-7:00pm CT
The event is being hosted by Peregrine Pharmaceuticals and will be webcast live and archived for 60 days. The event may be accessed from the home page of Peregrine Pharmaceuticals' website at http://www.peregrineinc.com . For the live event, listeners are encouraged to login 5-10 minutes ahead of the scheduled start in order to verify system requirements and complete registration.
Lori Kunkel, MD will moderate this event. Dr. Kunkel serves on the Board of Directors of Loxo Oncology [ http://www.loxooncology.com ], where she held the position of Acting Chief Medical Officer. Previously, Dr. Kunkel was Chief Medical Officer at Pharmacyclics and Proteolix and has also held senior roles at ACT Therapeutics, Syndax, Xencor, and Genitope. Dr. Kunkel received her MD from the Univ. of Southern California and her BA degree from the Univ. of California San Diego.
The panel includes:
Scott J. Antonia[PPHM SAB], MD, PhD, Department Chair, Program Leader, Thoracic Oncology Dept Associate Professor Dept of Interdisciplinary Oncology and Program Leader of the Immunology Program, H. Lee Moffitt Cancer Center and Research Institute, Tampa, Florida
[ http://moffitt.org/research--clinical-trials/individual-researchers/scott-j--antonia-md-phd ]
Dmitry I. Gabrilovich, MD, PhD, Christopher M. Davis Professor in Cancer Research and Program Leader, Translational Tumor Immunology, The Wistar Institute, Philadelphia, PA
[ http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd ]
Kathy D. Miller, MD, Ballve Lantero Scholar in Oncology, Professor, Dept of Medicine, Co-Director, Simon Cancer Center Breast Cancer Program, Indiana University School of Medicine, Indianapolis, IN
[ http://www.bcrfcure.org/researchers/kathy-d-miller ]
Dmitriy Zamarin, MD, PhD, Assistant Attending Physician, Gynecologic Medical Oncology & Immunotherapeutics, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/dmitriy-zamarin http://www.youtube.com/watch?v=hGL__YKuOLY ]
Matthew D. Hellman, MD, Assistant Attending Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/cancer-care/doctors/matthew-hellmann “History of Immunotherapy”: http://www.youtube.com/watch?v=2KWsYmXhclw ]
Alexander M. Lesokhin, MD, Assistant Member, Assistant Attending Physician, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/alexander-lesokhin-01 ]
Rolf A. Brekken[PPHM SAB], PhD, Effie Marie Cain Research Scholar in Angiogenesis Research, Professor, Depts of Surgery & Pharmacology, Univ. of Texas Southwestern Medical Center, Dallas, TX
[ http://profiles.utsouthwestern.edu/profile/10808/rolf-brekken.html ]
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. Additional information about Peregrine can be found at http://www.peregrineinc.com .
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., 800-987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
= = = = = =
5-30-15: PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit ~20% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Enter BAVI, stage left, whose objective is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = =
BAVITUXIMAB PHASE III TRIAL: see http://PeregrineTrials.com => http://www.SunriseTrial.com (Start=Dec'13 EstPrimComp=Dec’16)
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673
Geo/Map view of Worldwide Ph3 SUNRISE Sites: http://www.medbravo.org/study?study=NCT01999673
...159 sites a/o 5-6-15 (USA/39 Aus/8 Bel/7 Fr/9 Ger/15 Greece/9 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/7 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/nrpbqhl
...EU/EEA Registries: (pts world=582, pts/EU=345, planned EU sites=100) http://www.clinicaltrialsregister.eu/ctr-search/search?query=2013-003953-13
• GER: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/DE (planned sites=14, pts=43)
• SPAIN: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/ES (planned sites=16, pts=100)
• HUNGARY: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/HU (planned sites=9, pts=28)
• GREECE: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/GR (planned sites=9, pts=120)
• ITALY: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/IT (planned pts=55)
• BELGIUM: http://www.clinicaltrialsregister.eu/ctr-search/trial/2013-003953-13/BE (planned sites=7 pts=31)
...EU= European Union (29 countries – see http://www.gov.uk/eu-eea ), EEA=European Economic Area (all the EU, plus Iceland, Liechtenstein, Norway)
…Regions where sites are planned: Australia, Belgium, France, Germany, Greece, Hungary, Italy, Korea, Romania, Russia., Spain, Taiwan, Ukraine, USA
...3-2014: Eur.Med.Assoc.(EMA) appears to approve Bavi/2L-NSCLC for possible future trial in Children: http://tinyurl.com/lqsjfj2
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
3-7-14: PR & Conf-Call: "first patients enrolled and dosed." http://tinyurl.com/kh9cnrg
1-6-14: FDA grants FAST TRACK status to Bavi in 2ndLine NSCLC http://tinyurl.com/l799ukk
12-30-13: Pivotal Ph.3 ‘SUNRISE’ NSCLC Trial Initiated (n=~600, sites=100+) http://tinyurl.com/kdjb9qz
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
= = = = = = = = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s

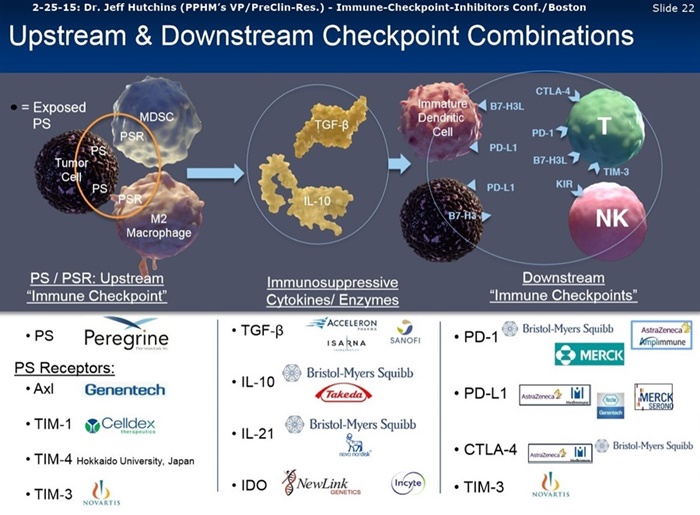
BAVI MOA 5-21-15: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
. . .MORE/iBox: http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
Event Brochure for Peregrine’s 5-31-15 ASCO’15 I-O Roundtable – sent to me my CK@IR…
Sunday 5-31-15 6pmCT: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
…7 panel members, incl. 3 Mem. Sloan Kettering researchers – recall the 5-29-15 announced collab. with MSK “to Investigate Novel PS-Targeting Immunotherapy Combinations”…
WEBCAST Replay link: http://edge.media-server.com/m/p/h2toyjv4

= = = = = = = = = = = =
5-27-15/PR: Peregrine Pharmaceuticals Announces Webcast of Immunotherapy Roundtable at ASCO Annual Meeting
• Immunotherapy Thought Leaders to Discuss Next Wave of Therapies Aimed at Increasing the Number of Patients That Respond to Immuno-Oncology Combination Therapies
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915098
TUSTIN, May 27, 2015: Peregrine Pharmaceuticals (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology (I-O) agent in Phase III development for the treatment of lung cancer, invites investors and the general public to listen to a webcast of its roundtable discussion in conjunction with the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting [ http://am.asco.org ]. This roundtable is comprised of key opinion leaders in the field of Immunotherapy who will discuss novel I-O combination therapies that may improve patient outcomes.
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6:00-7:00pm CT
The event is being hosted by Peregrine Pharmaceuticals and will be webcast live and archived for 60 days. The event may be accessed from the home page of Peregrine Pharmaceuticals' website at http://www.peregrineinc.com . For the live event, listeners are encouraged to login 5-10 minutes ahead of the scheduled start in order to verify system requirements and complete registration.
Lori Kunkel, MD will moderate this event. Dr. Kunkel serves on the Board of Directors of Loxo Oncology [ http://www.loxooncology.com ], where she held the position of Acting Chief Medical Officer. Previously, Dr. Kunkel was Chief Medical Officer at Pharmacyclics and Proteolix and has also held senior roles at ACT Therapeutics, Syndax, Xencor, and Genitope. Dr. Kunkel received her MD from the Univ. of Southern California and her BA degree from the Univ. of California San Diego.
The panel includes:
Scott J. Antonia[PPHM SAB], MD, PhD, Department Chair, Program Leader, Thoracic Oncology Dept Associate Professor Dept of Interdisciplinary Oncology and Program Leader of the Immunology Program, H. Lee Moffitt Cancer Center and Research Institute, Tampa, Florida
[ http://moffitt.org/research--clinical-trials/individual-researchers/scott-j--antonia-md-phd ]
Dmitry I. Gabrilovich, MD, PhD, Christopher M. Davis Professor in Cancer Research and Program Leader, Translational Tumor Immunology, The Wistar Institute, Philadelphia, PA
[ http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd ]
Kathy D. Miller, MD, Ballve Lantero Scholar in Oncology, Professor, Dept of Medicine, Co-Director, Simon Cancer Center Breast Cancer Program, Indiana University School of Medicine, Indianapolis, IN
[ http://www.bcrfcure.org/researchers/kathy-d-miller ]
Dmitriy Zamarin, MD, PhD, Assistant Attending Physician, Gynecologic Medical Oncology & Immunotherapeutics, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/dmitriy-zamarin http://www.youtube.com/watch?v=hGL__YKuOLY ]
Matthew D. Hellman, MD, Assistant Attending Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/cancer-care/doctors/matthew-hellmann “History of Immunotherapy”: http://www.youtube.com/watch?v=2KWsYmXhclw ]
Alexander M. Lesokhin, MD, Assistant Member, Assistant Attending Physician, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/alexander-lesokhin-01 ]
Rolf A. Brekken[PPHM SAB], PhD, Effie Marie Cain Research Scholar in Angiogenesis Research, Professor, Depts of Surgery & Pharmacology, Univ. of Texas Southwestern Medical Center, Dallas, TX
[ http://profiles.utsouthwestern.edu/profile/10808/rolf-brekken.html ]
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. Additional information about Peregrine can be found at http://www.peregrineinc.com .
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., 800-987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
= = = = = =
5-30-15: PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit ~20% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Enter BAVI, stage left, whose objective is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s


BAVI MOA 5-21-15: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
The Inventor of the PS-Targeting Mabs, like 3G4/bavituximab, is Dr. Philip E. Thorpe of UTSW-MC/Dallas.
4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
Dr. Thorpe's LAB TEAM (UTSW Hamon Center): http://tinyurl.com/yuxemu
…2013: Dr. Thorpe’s lab taken over by UTSW’s Dr. Rolf A. Brekken: http://www.utsouthwestern.edu/labs/brekken
Dr. Thorpe's Patents: GRANTED: http://tinyurl.com/m232k PENDING: http://tinyurl.com/845p2
…Dr. Thorpe's Articles: http://tinyurl.com/csjsl Also see: http://www.researchgate.net/profile/Philip_Thorpe
Dr. Brekken’s Patents: GRANTED: http://tinyurl.com/q2joy3v PENDING: http://tinyurl.com/l5jngmw
…Dr. Brekken’s Articles: http://tinyurl.com/oyf5r9p Also see: http://www.researchgate.net/profile/Rolf_Brekken
Dr. Alan Schroit's Patents (MDA, PPHM SRB, “Clipped B2GP1”): GRANTED: http://tinyurl.com/n4pnk8p PENDING: http://tinyurl.com/mrf2nsb
…Dr. Schroit’s Articles: http://tinyurl.com/lqbd8kt Also see: http://tinyurl.com/lx2p32b (MDA profile)
Incorrect statement, ExWanna => “This board (the long side) was near universal in predictions that failed to come about. SUNRISE enrollment complete by early 2015.”
Completely untrue. Please explain why you said this.
Link to Today’s ASCO I-O Webcast @6pmCT/7pmET – 7 panel members, incl. 3 Mem. Sloan Kettering researchers – recall the 5-29-15 announced collab. with MSK “to Investigate Novel PS-Targeting Immunotherapy Combinations”…
Sunday May31 6pmCT (7pmET, 4pmPT): “Raising the Immuno-Oncology Bar”
WEBCAST link: http://edge.media-server.com/m/p/h2toyjv4
= = = = = = = = = = = = = =
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6pmCT (7pmET, 4pmPT)
MORE about this as posted earlier today: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114163787
Link to Today’s ASCO I-O Webcast @6pmCT/7pmET – 7 panel members, incl. 3 Mem. Sloan Kettering researchers – recall the 5-29-15 announced collab. with MSK “to Investigate Novel PS-Targeting Immunotherapy Combinations”…
WEBCAST link: http://edge.media-server.com/m/p/h2toyjv4
…Sunday May31 6pmCT,7pmET, 4pmPT: “Raising the Immuno-Oncology Bar”
5-27-15/PR: Peregrine Pharmaceuticals Announces Webcast of Immunotherapy Roundtable at ASCO Annual Meeting
• Immunotherapy Thought Leaders to Discuss Next Wave of Therapies Aimed at Increasing the Number of Patients That Respond to Immuno-Oncology Combination Therapies
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915098
TUSTIN, May 27, 2015: Peregrine Pharmaceuticals (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology (I-O) agent in Phase III development for the treatment of lung cancer, invites investors and the general public to listen to a webcast of its roundtable discussion in conjunction with the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting [ http://am.asco.org ]. This roundtable is comprised of key opinion leaders in the field of Immunotherapy who will discuss novel I-O combination therapies that may improve patient outcomes.
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6:00-7:00pm CT
The event is being hosted by Peregrine Pharmaceuticals and will be webcast live and archived for 60 days. The event may be accessed from the home page of Peregrine Pharmaceuticals' website at http://www.peregrineinc.com . For the live event, listeners are encouraged to login 5-10 minutes ahead of the scheduled start in order to verify system requirements and complete registration.
Lori Kunkel, MD will moderate this event. Dr. Kunkel serves on the Board of Directors of Loxo Oncology [ http://www.loxooncology.com ], where she held the position of Acting Chief Medical Officer. Previously, Dr. Kunkel was Chief Medical Officer at Pharmacyclics and Proteolix and has also held senior roles at ACT Therapeutics, Syndax, Xencor, and Genitope. Dr. Kunkel received her MD from the Univ. of Southern California and her BA degree from the Univ. of California San Diego.
The panel includes:
Scott J. Antonia[PPHM SAB], MD, PhD, Department Chair, Program Leader, Thoracic Oncology Dept Associate Professor Dept of Interdisciplinary Oncology and Program Leader of the Immunology Program, H. Lee Moffitt Cancer Center and Research Institute, Tampa, Florida
[ http://moffitt.org/research--clinical-trials/individual-researchers/scott-j--antonia-md-phd ]
Dmitry I. Gabrilovich, MD, PhD, Christopher M. Davis Professor in Cancer Research and Program Leader, Translational Tumor Immunology, The Wistar Institute, Philadelphia, PA
[ http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd ]
Kathy D. Miller, MD, Ballve Lantero Scholar in Oncology, Professor, Dept of Medicine, Co-Director, Simon Cancer Center Breast Cancer Program, Indiana University School of Medicine, Indianapolis, IN
[ http://www.bcrfcure.org/researchers/kathy-d-miller ]
Dmitriy Zamarin, MD, PhD, Assistant Attending Physician, Gynecologic Medical Oncology & Immunotherapeutics, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/dmitriy-zamarin http://www.youtube.com/watch?v=hGL__YKuOLY ]
Matthew D. Hellman, MD, Assistant Attending Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/cancer-care/doctors/matthew-hellmann “History of Immunotherapy”: http://www.youtube.com/watch?v=2KWsYmXhclw ]
Alexander M. Lesokhin, MD, Assistant Member, Assistant Attending Physician, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/alexander-lesokhin-01 ]
Rolf A. Brekken[PPHM SAB], PhD, Effie Marie Cain Research Scholar in Angiogenesis Research, Professor, Depts of Surgery & Pharmacology, Univ. of Texas Southwestern Medical Center, Dallas, TX
[ http://profiles.utsouthwestern.edu/profile/10808/rolf-brekken.html ]
<snip>
= = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
<snip>
- - - - - - - - - - - - -
ASCO’15(May30-Jun2): PPHM’s 3 Abstracts & May31/6pmCT I-O Roundtable Webcast (7 on I-O panel, incl. 3 Sloan-Kettering Researchers)
=> http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114081170
= = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s


BAVI MOA 5-21-15: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
The Inventor of the PS-Targeting Mabs, like 3G4/bavituximab, is Dr. Philip E. Thorpe of UTSW-MC/Dallas.
4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
Dr. Thorpe's LAB TEAM (UTSW Hamon Center): http://tinyurl.com/yuxemu
…2013: Dr. Thorpe’s lab taken over by UTSW’s Dr. Rolf A. Brekken: http://www.utsouthwestern.edu/labs/brekken
Dr. Thorpe's Patents: GRANTED: http://tinyurl.com/m232k PENDING: http://tinyurl.com/845p2
…Dr. Thorpe's Articles: http://tinyurl.com/csjsl Also see: http://www.researchgate.net/profile/Philip_Thorpe
Dr. Brekken’s Patents: GRANTED: http://tinyurl.com/q2joy3v PENDING: http://tinyurl.com/l5jngmw
…Dr. Brekken’s Articles: http://tinyurl.com/oyf5r9p Also see: http://www.researchgate.net/profile/Rolf_Brekken
Dr. Alan Schroit's Patents (MDA, PPHM SRB, “Clipped B2GP1”): GRANTED: http://tinyurl.com/n4pnk8p PENDING: http://tinyurl.com/mrf2nsb
…Dr. Schroit’s Articles: http://tinyurl.com/lqbd8kt Also see: http://tinyurl.com/lx2p32b (MDA profile)
PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit ~20% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: BMY's Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Enter BAVI, stage left, whose objective is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
- - - - - - - - - - - - -
5-29-15/PR: Peregrine & Sloan Kettering Enter Into Collaboration
=> http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
ASCO’15(May30-Jun2): PPHM’s 3 Abstracts & May31/6pmCT I-O Roundtable Webcast (7 on I-O panel, incl. 3 Sloan-Kettering Researchers)
=> http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114081170
5-29-15/PR: Peregrine & Sloan Kettering Enter Into Collaboration to Investigate Novel PS-Targeting Immunotherapy Combinations. “Researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses.”…
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915472
TUSTIN, May 29, 2015: Peregrine Pharmaceuticals, Inc. (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology agent in Phase III development for the treatment of lung cancer, today announced that the company has entered into a sponsored research agreement with Memorial Sloan Kettering Cancer Center (MSK) to explore the potential of Peregrine's proprietary phosphatidylserine (PS)-targeting antibody platform. The goal of the research is to identify effective treatment combinations based on Peregrine's PS-targeting agents, including Peregrine's lead clinical agent bavituximab, with other checkpoint inhibitors or immune stimulating agents that will further guide the bavituximab clinical development program.
The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Wolchok.
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Merghoub.
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. "Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. "Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
Peregrine's antibodies target and bind to phosphatidylserine (PS), a highly immunosuppressive molecule normally located on the interior of cellular membranes, but, following stresses in the tumor environment, becomes exposed on tumor cells and cells that line tumor blood vessels, helping tumors to evade immune detection. PS-targeting antibodies block this immunosuppressive signal, thereby enabling the immune system to better recognize and fight the tumor. Preclinical data show that the combination of bavituximab and inhibitors of immune checkpoints reduce tumor-suppressive factors including myeloid-derived suppressor cells and confer increased tumor-specific immunity when compared to either treatment alone.
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
= = = = = = = =Note 3 Sloan Kettering Panelists on May31 Webcast:
ASCO’15(May30-Jun2): PPHM’s 3Abstracts & May31/6pmCT I-O Webcast, 7 I-O KOL’s on panel, “Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"…
= = = = = = = = = = = =
5-27-15/PR: Peregrine Pharmaceuticals Announces (May 31st 6pmCT) Webcast of Immunotherapy Roundtable at ASCO Annual Meeting
• Immunotherapy Thought Leaders to Discuss Next Wave of Therapies Aimed at Increasing the Number of Patients That Respond to Immuno-Oncology Combination Therapies
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915098
TUSTIN, May 27, 2015: Peregrine Pharmaceuticals (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology (I-O) agent in Phase III development for the treatment of lung cancer, invites investors and the general public to listen to a webcast of its roundtable discussion in conjunction with the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting [ http://am.asco.org ]. This roundtable is comprised of key opinion leaders in the field of Immunotherapy who will discuss novel I-O combination therapies that may improve patient outcomes.
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6:00-7:00pm CT
The event is being hosted by Peregrine Pharmaceuticals and will be webcast live and archived for 60 days. The event may be accessed from the home page of Peregrine Pharmaceuticals' website at http://www.peregrineinc.com . For the live event, listeners are encouraged to login 5-10 minutes ahead of the scheduled start in order to verify system requirements and complete registration.
Lori Kunkel, MD will moderate this event. Dr. Kunkel serves on the Board of Directors of Loxo Oncology [ http://www.loxooncology.com ], where she held the position of Acting Chief Medical Officer. Previously, Dr. Kunkel was Chief Medical Officer at Pharmacyclics and Proteolix and has also held senior roles at ACT Therapeutics, Syndax, Xencor, and Genitope. Dr. Kunkel received her MD from the Univ. of Southern California and her BA degree from the Univ. of California San Diego.
The panel includes:
Scott J. Antonia[PPHM SAB], MD, PhD, Department Chair, Program Leader, Thoracic Oncology Dept Associate Professor Dept of Interdisciplinary Oncology and Program Leader of the Immunology Program, H. Lee Moffitt Cancer Center and Research Institute, Tampa, Florida
[ http://moffitt.org/research--clinical-trials/individual-researchers/scott-j--antonia-md-phd ]
Dmitry I. Gabrilovich, MD, PhD, Christopher M. Davis Professor in Cancer Research and Program Leader, Translational Tumor Immunology, The Wistar Institute, Philadelphia, PA
[ http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd ]
Kathy D. Miller, MD, Ballve Lantero Scholar in Oncology, Professor, Dept of Medicine, Co-Director, Simon Cancer Center Breast Cancer Program, Indiana University School of Medicine, Indianapolis, IN
[ http://www.bcrfcure.org/researchers/kathy-d-miller ]
Dmitriy Zamarin, MD, PhD, Assistant Attending Physician, Gynecologic Medical Oncology & Immunotherapeutics, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/dmitriy-zamarin http://www.youtube.com/watch?v=hGL__YKuOLY ]
Matthew D. Hellman, MD, Assistant Attending Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/cancer-care/doctors/matthew-hellmann “History of Immunotherapy”: http://www.youtube.com/watch?v=2KWsYmXhclw ]
Alexander M. Lesokhin, MD, Assistant Member, Assistant Attending Physician, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/alexander-lesokhin-01 ]
Rolf A. Brekken[PPHM SAB], PhD, Effie Marie Cain Research Scholar in Angiogenesis Research, Professor, Depts of Surgery & Pharmacology, Univ. of Texas Southwestern Medical Center, Dallas, TX
[ http://profiles.utsouthwestern.edu/profile/10808/rolf-brekken.html ]
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. Additional information about Peregrine can be found at http://www.peregrineinc.com .
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., 800-987-8256, info@peregrineinc.com
May29-Jun2: ASCO Annual Meeting 2015, Chicago
http://am.asco.org
Abstracts: https://iplanner.asco.org/am2015/AM2015.aspx (pub. 5-13-15 5pmET)
• #3059/Sat-May30/8-11:30am, Track: Dev. Therapeutics & Translational Res. - “PS Targeting Antibody in Combination with Anti-PD-1 Antibody Treatment Activates Infiltrating T lymphocytes of the Spleen & Tumor Microenvironment in Pre-Clinical Models of Melanoma & Breast Cancer” - Xianming Huang, PhD (UTSW-MC/Dallas, Brekken Lab)
• #3060/Sat-May30/8-11:30am, Track: Dev. Therapeutics & Translational Res. - “Activation of CD8+ Tumor Infiltrating Lymphocytes by Bavituximab in a 3D ex vivo System of Lung Cancer Patients” - Soner Altiok, MD, PhD (H. Lee Moffitt CCC & Res. Institute, Tampa)
• #4109/Mon-June1/8:11:30am, Track: Gastrointestinal (NonColorectal) Cancer - “A Phase I/II Study of Bavituximab & Sorafenib in Advanced Hepatocellular Carcinoma (HCC)” - Adam Yopp, MD (UTSW-MC/Dallas)
“In addition, Peregrine will be hosting ASCO conf. attendees at Booth #22107” http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=913039
= = = = = = = = = = = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s

BAVI MOA 5-21-15: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
The Inventor of the PS-Targeting Mabs, like 3G4/bavituximab, is Dr. Philip E. Thorpe of UTSW-MC/Dallas.
4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
Dr. Thorpe's LAB TEAM (UTSW Hamon Center): http://tinyurl.com/yuxemu
…2013: Dr. Thorpe’s lab taken over by UTSW’s Dr. Rolf A. Brekken: http://www.utsouthwestern.edu/labs/brekken
Dr. Thorpe's Patents: GRANTED: http://tinyurl.com/m232k PENDING: http://tinyurl.com/845p2
…Dr. Thorpe's Articles: http://tinyurl.com/csjsl Also see: http://www.researchgate.net/profile/Philip_Thorpe
Dr. Brekken’s Patents: GRANTED: http://tinyurl.com/q2joy3v PENDING: http://tinyurl.com/l5jngmw
…Dr. Brekken’s Articles: http://tinyurl.com/oyf5r9p Also see: http://www.researchgate.net/profile/Rolf_Brekken
Dr. Alan Schroit's Patents (MDA, PPHM SRB, “Clipped B2GP1”): GRANTED: http://tinyurl.com/n4pnk8p PENDING: http://tinyurl.com/mrf2nsb
…Dr. Schroit’s Articles: http://tinyurl.com/lqbd8kt Also see: http://tinyurl.com/lx2p32b (MDA profile)
ASCO’15(May30-Jun2): PPHM’s 3Abstracts & May31/6pmCT I-O Roundtable Webcast - 7 I-O KOL’s on panel, “Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"…
May29-Jun2: ASCO Annual Meeting 2015, Chicago
http://am.asco.org
Abstracts: https://iplanner.asco.org/am2015/AM2015.aspx (pub. 5-13-15 5pmET)
• #3059/Sat-May30/8-11:30am, Track: Dev. Therapeutics & Translational Res. - “PS Targeting Antibody in Combination with Anti-PD-1 Antibody Treatment Activates Infiltrating T lymphocytes of the Spleen & Tumor Microenvironment in Pre-Clinical Models of Melanoma & Breast Cancer” - Xianming Huang, PhD (UTSW-MC/Dallas, Brekken Lab)
• #3060/Sat-May30/8-11:30am, Track: Dev. Therapeutics & Translational Res. - “Activation of CD8+ Tumor Infiltrating Lymphocytes by Bavituximab in a 3D ex vivo System of Lung Cancer Patients” - Soner Altiok, MD, PhD (H. Lee Moffitt CCC & Res. Institute, Tampa)
• #4109/Mon-June1/8:11:30am, Track: Gastrointestinal (NonColorectal) Cancer - “A Phase I/II Study of Bavituximab & Sorafenib in Advanced Hepatocellular Carcinoma (HCC)” - Adam Yopp, MD (UTSW-MC/Dallas)
“In addition, Peregrine will be hosting ASCO conf. attendees at Booth #22107” http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=913039
= = = = = = = = = = = =
5-27-15/PR: Peregrine Pharmaceuticals Announces Webcast of Immunotherapy Roundtable at ASCO Annual Meeting
• Immunotherapy Thought Leaders to Discuss Next Wave of Therapies Aimed at Increasing the Number of Patients That Respond to Immuno-Oncology Combination Therapies
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915098
TUSTIN, May 27, 2015: Peregrine Pharmaceuticals (Nasdaq:PPHM, PPHMP), a biopharmaceutical company focused on advancing bavituximab, a novel immuno-oncology (I-O) agent in Phase III development for the treatment of lung cancer, invites investors and the general public to listen to a webcast of its roundtable discussion in conjunction with the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting [ http://am.asco.org ]. This roundtable is comprised of key opinion leaders in the field of Immunotherapy who will discuss novel I-O combination therapies that may improve patient outcomes.
Title: "Raising the Immuno-Oncology Bar: The Next Wave of Immune Modulating Checkpoint Inhibitors"
Date/Time: Sunday, May 31, 2015 from 6:00-7:00pm CT
The event is being hosted by Peregrine Pharmaceuticals and will be webcast live and archived for 60 days. The event may be accessed from the home page of Peregrine Pharmaceuticals' website at http://www.peregrineinc.com . For the live event, listeners are encouraged to login 5-10 minutes ahead of the scheduled start in order to verify system requirements and complete registration.
Lori Kunkel, MD will moderate this event. Dr. Kunkel serves on the Board of Directors of Loxo Oncology [ http://www.loxooncology.com ], where she held the position of Acting Chief Medical Officer. Previously, Dr. Kunkel was Chief Medical Officer at Pharmacyclics and Proteolix and has also held senior roles at ACT Therapeutics, Syndax, Xencor, and Genitope. Dr. Kunkel received her MD from the Univ. of Southern California and her BA degree from the Univ. of California San Diego.
The panel includes:
Scott J. Antonia [PPHM SAB], MD, PhD, Dept Chair, Program Leader, Thoracic Oncology Department Associate Professor Dept of Interdisciplinary Oncology and Program Leader of the Immunology Program, H. Lee Moffitt Cancer Center and Research Institute, Tampa, Florida
[ http://moffitt.org/research--clinical-trials/individual-researchers/scott-j--antonia-md-phd ]
Dmitry I. Gabrilovich, MD, PhD, Christopher M. Davis Professor in Cancer Research and Program Leader, Translational Tumor Immunology, The Wistar Institute, Philadelphia, PA
[ http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd ]
Kathy D. Miller, MD, Ballve Lantero Scholar in Oncology, Professor, Dept of Medicine, Co-Director, Simon Cancer Center Breast Cancer Program, Indiana University School of Medicine, Indianapolis, IN
[ http://www.bcrfcure.org/researchers/kathy-d-miller ]
Dmitriy Zamarin, MD, PhD, Assistant Attending Physician, Gynecologic Medical Oncology & Immunotherapeutics, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/dmitriy-zamarin http://www.youtube.com/watch?v=hGL__YKuOLY ]
Matthew D. Hellman, MD, Assistant Attending Thoracic Oncology Service, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/cancer-care/doctors/matthew-hellmann “History of Immunotherapy”: http://www.youtube.com/watch?v=2KWsYmXhclw ]
Alexander M. Lesokhin, MD, Assistant Member, Assistant Attending Physician, Memorial Sloan Kettering Cancer Center, NYC
[ http://www.mskcc.org/research-areas/labs/members/alexander-lesokhin-01 ]
Rolf A. Brekken [PPHM SAB], PhD, Effie Marie Cain Research Scholar in Angiogenesis Research, Professor, Depts of Surgery & Pharmacology, Univ. of Texas Southwestern Medical Center, Dallas, TX
[ http://profiles.utsouthwestern.edu/profile/10808/rolf-brekken.html ]
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. Additional information about Peregrine can be found at http://www.peregrineinc.com .
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., 800-987-8256, info@peregrineinc.com
= = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s

BAVI MOA 5-21-15: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
The Inventor of the PS-Targeting Mabs, like 3G4/bavituximab, is Dr. Philip E. Thorpe of UTSW-MC/Dallas.
4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
Dr. Thorpe's LAB TEAM (UTSW Hamon Center): http://tinyurl.com/yuxemu
…2013: Dr. Thorpe’s lab taken over by UTSW’s Dr. Rolf A. Brekken: http://www.utsouthwestern.edu/labs/brekken
Dr. Thorpe's Patents: GRANTED: http://tinyurl.com/m232k PENDING: http://tinyurl.com/845p2
…Dr. Thorpe's Articles: http://tinyurl.com/csjsl Also see: http://www.researchgate.net/profile/Philip_Thorpe
Dr. Brekken’s Patents: GRANTED: http://tinyurl.com/q2joy3v PENDING: http://tinyurl.com/l5jngmw
…Dr. Brekken’s Articles: http://tinyurl.com/oyf5r9p Also see: http://www.researchgate.net/profile/Rolf_Brekken
Dr. Alan Schroit's Patents (MDA, PPHM SRB, “Clipped B2GP1”): GRANTED: http://tinyurl.com/n4pnk8p PENDING: http://tinyurl.com/mrf2nsb
…Dr. Schroit’s Articles: http://tinyurl.com/lqbd8kt Also see: http://tinyurl.com/lx2p32b (MDA profile)
N40K, that quote by Nilogen Oncosystems' (Moffitt startup) Dr. Sigrid M. Volko about clinical translational data from the UTSW/Yopp Liver IST as applied to Lung Cancer came from Peregrine's 4-20-15 AACR'15 (Abstract #274) press release:
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=112903784
"Our translational findings of a cytokine profile that reflects an immune response following either bavituximab single-agent or combination treatment are encouraging. Furthermore, these preliminary translational data show that tumors with negative PD-L1 expression and low levels of PD-1 expression on CD8+ tumor infiltrating T cells showed immune response to bavituximab treatment ex vivo," said Sigrid M. Volko, Ph.D., CLP the lead investigator on the study and President & CEO of Nilogen Oncosystems [=Moffit Startup: http://otmc.moffitt.org/startups.aspx ]. "It is an exciting time for the field of immunotherapy and the data we have been generating support that bavituximab has the potential to activate a tumor specific immune response in patients with PD-L1 negative tumors that generally do not respond as well to PD-1 or PD-L1 inhibitors."
2 Bavi MOA Videos added 3-2014 (from iBox)…
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14
Loofman: cjgaddy: 12/31/15=$2.55, 6/30/16=$3.80 (6-30-17=$45.00)
…Looking fwd to taking home the grand prize of a jug of Old Loofman's Pure Kentucky Korn Licker, and I trust you’ll throw in a large package of Loofman's Famous Fresh Kill Squirrel Jerky to boot!! Go, Peregrine, GO!
5-13-15/SeanWilliams/MotleyFool: “3 Reasons Peregrine's Stock Could Rise”
“Without further ado, 3 reasons Peregrine's stock could rise.
1. Positive SUNRISE data
2. The announcement of licensing partner or immunotherapy partner programs
3. Buyout interest from a growth-hungry Big Pharma or biotech blue-chip stock”
. . .
Conclusion: “Overall, I believe Peregrine has potential, but I don't think it belongs in anyone's portfolio unless you have an extremely high appetite for risk & reward. Potentially positive data from its IST-based studies could help stem the negative impact of a stock offering to raise cash, but all eyes should remain focused on the SUNRISE data release expected in December 2016.”
“Sean Williams has no material interest in any companies mentioned in this article.”
FULL ARTICLE: http://www.investopedia.com/stock-analysis/051315/3-reasons-peregrines-stock-could-rise-pphm.aspx
Sean Motley profile: http://my.fool.com/profile/TMFUltraLong/info.aspx
Known Upcoming events, incl. ASCO May30-June1(2 Abstracts)
May21: NYAS: Emerging Approaches to Cancer Immunotherapy, NYC http://tinyurl.com/lfdoksl
…3:10pm: Dr. Rolf Brekken, “Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
May29-Jun2: ASCO Annual Meeting 2015, Chicago http://am.asco.org - see: http://tinyurl.com/lfdoksl
...May30 #3060/S.Altiok(Moffitt): “Activation of CD8+ Tumor Infiltrating Lymphocytes by Bavituximab in a 3D ex vivo System of Lung Cancer Patients”
...Jun1 #4109/Adam Yopp(UTSW): “A Phase I/II Study of Bavituximab & Sorafenib in Advanced Hepatocellular (Liver) Carcinoma (HCC)”
Abstracts: https://iplanner.asco.org/am2015/AM2015.aspx (pub. 5-13-15 5pmET)
Jun15: GRC’s “7th Apoptotic Cell Recognition & Clearance Conf.”, Biddeford ME http://www.grc.org/programs.aspx?id=13127
…8:50-9:15pm: Xianming Huang (UTSW), “PS-Targeting Antibodies Overcome Tumor Immunosuppression & Synergize with Immune Checkpoint Blockade"
Jun15-18/Avid/Booth533: BIO Intl. Convention, Philadelphia http://convention.bio.org/2015
~Jul13: FY'15/Q4 (fye 4-30-15) Financials & Conf. Call http://ir.peregrineinc.com/events.cfm
Aug26: CHI’s 10th Annual Immunotherapy & Vaccine Summit (IMVACS), Boston http://tinyurl.com/lfdoksl
… 9-9:30 Jeff T. Hutchins (VP/Preclin-Res.): “Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
Oct26-29/Avid/Booth909: IBC's BioProcess Intl. Conf. & Exhib, Boston http://www.ibclifesciences.com/BPI
Dec7-10/Avid/Booth307: IBC's Antibody Eng. & Therapeutics, SanDiego http://www.ibclifesciences.com/AntibodyEng
Upcoming: NYAS/Brekken ASCO/Yopp,Moffitt GRC/Huang CHI/Hutchins
May21 2015: “NYAS’ Emerging Approaches to Cancer Immunotherapy Symposium”, NYC
NYAS=New York Academy of Sciences: http://www.nyas.org/AboutUs/aboutus.aspx
“Over the past several years, cancer immunotherapy has made extraordinary progress both conceptually and in clinical practice. Led by landmark studies in the setting of metastatic melanoma, many of the findings of this field are being generalized both to hematological malignancies and to other solid tumors. Areas of investigation such as checkpoint blockade inhibitors, advanced tumor vaccination studies, and adoptive T-cell therapies are changing the face of cancer treatment. This symposium will bring together leading experts in immunotherapy & immunology to discuss these emerging approaches, along with the challenges & opportunities faced in this evolving field.”
Symposium: http://www.nyas.org/Events/Detail.aspx?cid=8029254f-aa2f-4373-894c-8c8a335a2e82
Presented by: Cancer & Signaling Discussion Group
Academy Friend: Peregrine Phram. - Grant Support: AstraZeneca
5-21-15 3:10-3:40pm: Dr. Rolf Brekken, PhD (UTSW, PPHM SAB) - one of 9 speakers
”Lifting the Veil of Immune Suppression and Inducing Immune Activation through Antibody-mediated Blockade of Phosphatidylserine Signaling”
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
May29-Jun2: ASCO Annual Meeting 2015, Chicago
http://am.asco.org
Abstracts: https://iplanner.asco.org/am2015/AM2015.aspx
• #3060/Sat-May30/8-11:30am, Track: Dev. Therapeutics & Translational Res.
“Activation of CD8+ Tumor Infiltrating Lymphocytes by Bavituximab in a 3D ex vivo System of Lung Cancer Patients” - Soner Altiok, MD, PhD (H. Lee Moffitt CCC & Research Institute, Tampa)
• #4109/Mon-June1/8:11:30am, Track: Gastrointestinal (NonColorectal) Cancer
“A Phase I/II Study of Bavituximab & Sorafenib in Advanced Hepatocellular Carcinoma (HCC)” - Adam Yopp, MD (UTSW-MC/Dallas)
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
Jun14–19 2015: “GRC: Apoptotic Cell Recognition & Clearance: Physiological Significance & Pathological Consequences”, Biddeford ME
GRC = Gordon Research Conferences (Univ. of New England) http://www.grc.org
”This 7th meeting of the Apoptotic Cell Recognition & Clearance Gordon Research Conf. will explore molecular details of apoptotic cell recognition and clearance mechanisms throughout evolution, including apoptotic immune regulation. We also will focus on the pathogenic subversion of mechanisms of apoptotic cell clearance. This meeting will bring together investigators who are at the forefront of these fields to present and discuss new findings & important issues…”
*Peregrine is the only corporate contributor to this conference.
Conf: http://www.grc.org/programs.aspx?id=13127
SESSION 7:30-9:30pm “Consequences of Lipid Alterations for Apoptotic Cell Clearance”
…Discussion Leader: Christopher Gregory (Univ. of Edinburgh, UK)
• 7:30-7:55pm Ian Dransfield (Univ/Edinburgh UK), "Protein S Binding to Apoptotic Cells & Subsequent Phagocytosis by Macrophages"
• 8:10-8:35pm Raymond Birge (Rutgers NJ Med-School), "Apoptotic Cell Recognition Receptors, Tyro3/Axl/Mer, Demonstrate Distinct Patterns of Ligand Recognition & Biological Function"
• 8:50-9:15pm Xianming Huang (UTSW-MC/Dallas – PPHM SAB/Brekken-Lab), "Phosphatidylserine-Targeting Antibodies Overcome Tumor Immunosuppression & Synergize with Immune Checkpoint Blockade"
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
Aug 24-28 2015: “CHI’s 10th Annual Immunotherapy & Vaccine Summit”, Boston
CHI = Cambridge Healthtech Institute, http://www.healthtech.com
“…This year's event has been expanded to 5 days with coverage on adjuvants, vaccine & immunotherapy technologies, immunomodulatory therapeutic antibodies for cancer, combination cancer immunotherapy, and T cell target discovery. ImVacS 2015 promises to be a must-attend event for commercial and academic entities to continue advancing immunotherapies and vaccines through technology and innovation.”
Event: http://www.imvacs.com/
PGM: Rational Combination Cancer Immunotherapy (1 of 6)
SESSION: COMBINATION IMMUNOTHERAPY STRATEGIES
8-26-15 9-9:30 Jeff T. Hutchins, PhD, VP/Preclin-Res., Peregrine Pharmaceuticals
“Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand Phosphatidylserine (PS): Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
SUMMARY:
The underlying cause for the failure of current therapies is the persistent and multifocal immune suppression in the tumor microenvironment that drives the absence of pre-existing antitumor T cells. Bavituximab blocks PS-mediated immunosuppression (decreasing MDSCs) by reprograming immune cells in the tumor microenvironment to enhance anticancer activity. Pre-clinical, translational, and clinical results using bavituximab with conventional and immunotherapy combinations promotes a robust, anti-tumor T cell mediated response to enhance cancer therapy.
http://ir.peregrineinc.com/events.cfm
= = = = = = =AVID:
Jun15-18 2015: “BIO Intl. Convention”, Philadelphia
http://convention.bio.org/2015
Avid: booth #533
Oct26-29 2015: “IBC's BioProcess Intl. Conf. & Exhibition”, Boston
http://www.ibclifesciences.com/BPI
Avid: booth #909
Dec7-10 2015: ”IBC's 26th Annual Antibody Engineering & Therapeutics”, SanDiego
http://www.ibclifesciences.com/AntibodyEng
Avid: booth #307
HL45, yes, 9-7-12 Ph2/NSCLC “STATSIG extension(doubling) in OS” BEFORE the CSM switching was discovered – ie BEFORE it was realized that the CTL-ARM had some 1mg/ActiveDrug in it and the 1MG ARM had some Placebo In it – ie, without the Switching, the results clearly would have been even better!
9-7-12: “The interim data showed a statistically significant improvement in overall survival (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS in the bavituximab-containing arms compared to the control arm. The following interim data was presented as part of a late-breaking plenary presentation at the 2012 Chicago Multidisciplinary Symposium in Thoracic Oncology by David Gerber, M.D., Associate Prof. of Internal Medicine at UTSW-MC, a principal investigator in the trial.”
9-7-12: "This study was a rigorous trial designed to minimize bias and we are encouraged that this trial yielded such positive results in the most important endpoint, overall survival. The positive overall response rates and progression free survival in both bavituximab-containing arms seen earlier in the study has now translated into a statistically significant extension in overall survival for patients, a result rarely achieved in phase II clinical trials." said Joseph Shan, VP of clinical and regulatory affairs at Peregrine. "The quality of this data gives us a solid foundation for designing a phase III trial with an increased probability of success. We are planning for an end-of-phase II meeting with the FDA as we plan to initiate this trial by mid-2013."
9-7-12: "The median overall survival [MOS] results from the Proof-of Concept study are truly outstanding and great news for patients. Statistically significant overall survival results at this stage of development are rare and have put us in an excellent position for advancing the program. Our attention is now turned to an end of phase II meeting by year end which will help us define the most efficient path forward to potential regulatory approval," said Robert Garnick, PhD, Head of Regulatory Affairs at Peregrine. "A global Phase III trial designed very similarly to the robust design of this Phase II trial greatly increases bavituximab's likelihood of success."
= = = = = = = = = =
9-7-12/PR: Bavituximab Doubles MOS in 2nd-Line NSCLC, interim n=121 Ph.IIB data presented by Dr. David Gerber at the 2012 Chicago Multidisciplinary Symposium in Thoracic Oncology (ASCO/ASTRO) shows.
This post contains:
• 9-7-12 Press Release recapping Dr. David Gerber’s Plenary talk in Chicago
• 9-7-12 Slides from VP/Clin.Affairs Joe Shan’s 15 min. video recapping Dr. Gerber’s talk
• Comparison data from prev. trials of currently-approved 2nd-Line NSCLC drugs
= = = = = = = = = = = = = = = = = = = = = = =
9-7-12: ”Interim Data from Peregrine's Phase II Trial in Second-Line Non-Small Cell Lung Cancer Demonstrate Doubling of Median Overall Survival in Bavitiximab-Containing Arms”
• Interim Data from Double-Blind, Placebo-Controlled Trial Presented at Late-Breaking Plenary Session at Leading Oncology Symposium
• Data Show Statistically Significant Improvement in Overall Survival for Patients Receiving Bavituximab Plus Docetaxel Versus Docetaxel Alone
• Clinical Data Strongly Support Advancing Program into Phase III Clinical Development
• Company to Host Conference Call on Monday, Sept. 10, at 11:00AM EDT
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=705521
TUSTIN, 9/7/2012: Peregrine Pharmaceuticals, Inc. (NASDAQ: PPHM), today announced that interim results were presented from its 121 patient randomized, double-blind, placebo-controlled Phase IIb trial in patients with refractory non-small cell lung cancer (NSCLC). The blinded study evaluated two dose levels of bavituximab (bavituximab-containing arms) given with docetaxel versus docetaxel plus placebo (control arm). The interim data showed a statistically significant improvement in overall survival (Hazard Ratio 0.524, p-value .0154) and a doubling of median overall survival (OS) in the bavituximab-containing arms compared to the control arm. The following interim data was presented as part of a late-breaking plenary presentation at the 2012 Chicago Multidisciplinary Symposium in Thoracic Oncology by David Gerber, M.D., Associate Professor of Internal Medicine at the University of Texas Southwestern Medical Center, a principal investigator in the trial.

"This study was a rigorous trial designed to minimize bias and we are encouraged that this trial yielded such positive results in the most important endpoint, overall survival. The positive overall response rates and progression free survival in both bavituximab-containing arms seen earlier in the study has now translated into a statistically significant extension in overall survival for patients, a result rarely achieved in phase II clinical trials." said Joseph Shan, VP of clinical and regulatory affairs at Peregrine. "The quality of this data gives us a solid foundation for designing a phase III trial with an increased probability of success. We are planning for an end-of-phase II meeting with the FDA as we plan to initiate this trial by mid-2013."
The trial enrolled 121 patients (117 evaluable per the study protocol) with second-line non-squamous NSCLC following one prior chemotherapy regimen at over 40 clinical centers. Patients were equally randomized to 1 of the 3 treatment arms, docetaxel (75mg/m2) plus either placebo, 1 mg/kg bavituximab, or 3 mg/kg bavituximab until disease progression. Approximately 50% of the patients were enrolled in the U.S. and 50% were enrolled internationally with equal distribution between all treatment groups.
"Robust data from this Phase II trial clearly demonstrate a significant benefit in overall survival with a good safety profile in patients receiving bavituximab plus docetaxel compared to those receiving docetaxel plus placebo," said Steven W. King, president and CEO of Peregrine. "We are currently in discussions with several potential pharmaceutical partners who have expressed great interest in our bavituximab oncology program. It is our goal to identify the optimal partner to assist with the design and logistics of a multinational Phase III pivotal trial."
The interim results from the study showed no significant safety differences between the three treatment arms as determined by the trial's independent data monitoring committee. Baseline characteristics were well balanced across all three treatment arms of the study, including performance (ECOG) status, age, gender, and race. Tumor responses were determined in accordance with Response Evaluation Criteria In Solid Tumors (RECIST 1.1) based on blinded central radiology review.
"The median overall survival [MOS] results from the Proof-of Concept study are truly outstanding and great news for patients. Statistically significant overall survival results at this stage of development are rare and have put us in an excellent position for advancing the program. Our attention is now turned to an end of phase II meeting by year end which will help us define the most efficient path forward to potential regulatory approval," said Robert Garnick, PhD, Head of Regulatory Affairs at Peregrine. "A global Phase III trial designed very similarly to the robust design of this Phase II trial greatly increases bavituximab's likelihood of success."
AUDIO WEBCAST
In conjunction with Dr. Gerber's presentation in Chicago, Peregrine has posted an audio webcast and slide deck to Peregrine's website. The webcast will be hosted by Joseph Shan, VP of clinical and regulatory affairs. This event will be pre-recorded. Access to the audio and corresponding slides can be found on Peregrine's website [ http://www.peregrineinc.com ].
CONFERENCE CALL
Peregrine will host a conference call and webcast to discuss these data and financial results for the first quarter fiscal year 2013 on Monday, Sept. 10, 2012, at 11:00 AM ET (8:00 AM PT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals call. A replay of the call will be available starting approximately two hours after the conclusion of the call through September 24, 2012 by calling (855) 859-2056, or (404) 537-3406 and using passcode 27367274.
ABOUT LUNG CANCER
According to the American Cancer Society, lung cancer is the second most commonly diagnosed cancer in the U.S., with approximately 226,160 new cases and 160,340 deaths each year, representing approximately 28% of all cancer deaths. NSCLC is the most common type of lung cancer, accounting for approximately 85-90% of lung cancer cases. Unfortunately, the 5-year survival rate for NSCLC patients is only 1%.
ABOUT BAVITUXIMAB
Bavituximab is a first-in-class phosphatidylserine (PS)-targeting monoclonal antibody that represents a new approach to treating cancer. Bavituximab is the lead drug candidate from the company's PS technology platform and is currently being tested in eight clinical trials including three randomized Phase II trials in front-line and second-line non-small cell lung cancer, front-line pancreatic cancer and five investigator-sponsored trials (ISTs) in additional oncology indications.
PS is a highly immunosuppressive molecule usually located inside the membrane of healthy cells, but "flips" and becomes exposed on the outside of cells that line tumor blood vessels, creating a specific target for anti-cancer treatments. PS-targeting antibodies target and bind to PS and block this immunosuppressive signal, thereby enabling the immune system to recognize and fight the tumor.
ABOUT PEREGRINE PHARMACEUTICALS
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a portfolio of innovative monoclonal antibodies in clinical trials focused on the treatment and diagnosis of cancer. The company is pursuing multiple clinical programs in cancer with its lead product candidate bavituximab and novel brain cancer agent Cotara®. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and outside customers. Additional information about Peregrine can be found at http://www.peregrineinc.com .
Safe Harbor *snip*
Contact: Christopher Keenan or Jay Carlson - Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = =
VP Joe Shan’s 9-7-12 15min. Audio Webcast recapping Dr. David Gerber’s 2nd-Line NSCLC Interim Data Presentation on 9-7-12 at the ASTRO-ASCO Thoracic-Oncology Symposium in Chicago ( http://clinicaltrials.gov/ct2/show/NCT01138163 ):
“In conjunction with Dr. Gerber's presentation in Chicago, Peregrine has posted an audio webcast and slide deck to Peregrine's website. The webcast will be hosted by Joseph Shan, VP of Clinical & Regulatory Affairs. This event will be pre-recorded. Access to the audio & corresponding slides can be found on Peregrine's website.”
• Access replay here: http://www.peregrineinc.com
• Or directly: http://www.peregrineinc.com/nsclc-presentation-astro-9-7-12.html?tmpl=video

9-7-12 Joe Shan Slideshow (Dr. Gerber’s 9-7-12 ASTRO/Chicago Bavi 2nd-Line NSCLC Interim Data):










From the 9-7-12 Press Release:

= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
Per Cancer.gov (NCI) - last modified: 8/15/2012…
Std. treatment options for Recurrent NSCLC include the following:
Chemotherapy or kinase inhibitors alone, including the following for patients who have previously received platinum chemotherapy:
• Taxotere (docetaxel) => MOS=6.3mos (meta-analysis of 5 trials, 865 pts)
• Altima (pemetrexed) => No diff. vs. Docetaxel (Ph.3 non-inferiority vs. Doce, 571 pts)
• Tarceva (erlotinib) => MOS=5.3mos (TITAN** Ph.III n=424 trial – see http://tinyurl.com/8w8lo93 )
• Iressa (gefitinib) => “Iressa does not improve OS”
• Xalkori (crizotinib) => very limited, only for ALK-positive pts (2-7% of pop.)
http://www.cancer.gov/cancertopics/pdq/treatment/non-small-cell-lung/healthprofessional/page12
- - - - - - - -
NOTE 10-8-08: “2nd-Line NSCLC: Avastin/Tarceva Improves PFS but Not Overall Survival vs. Tarceva” ( http://cancergrace.org/lung/2008/10/08/beta-neg-for-os )
** "The TITAN (Tarceva) study (treatment 4/2006-2/2010) is the first prospective head-to-head study to specifically investigate Tarceva (erlotinib) vs. systemic chemotherapy in 2nd-Line NSCLC for patients who progressed during 1st-line platinum-doublet chemotherapy.”
http://www.egfr.roche.es/TITAN/TITAN_EMCTO_Poster.pdf
= = = = = = = = = = =
F. LEAD IND: Phase IIb Bavi+Doce vs. 2nd-Line NSCLC (randomized, double-blinded, placebo-ctl'd, n=120, 'registrational')
Protocol: http://clinicaltrials.gov/ct2/show/NCT01138163 (24 U.S. + 15 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 53 as of 8-12-11)
Enrolled Oct2010 - Oct2011 at 40 global sites (per J.Shan 9-7-12 webcast ( http://tinyurl.com/8cn87la )
8-2012: Compare Bavi+Doce's MOS=11.7mos (Bavi/3mg) to the 4 Curr-Approved 2Line/NSCLC Drugs http://tinyurl.com/cgnkvpa
• Taxotere/docetaxel => MOS=6.3mos (meta-analysis of 5 trials, 865 pts)
• Altima/pemetrexed => No diff. vs. Docetaxel (Ph.3 non-inferiority vs. Doce, 571 pts)
• Tarceva/erlotinib => MOS=5.3mos (TITAN Ph.III n=424 trial - see http://tinyurl.com/8w8lo93 )
• Iressa/gefitinib => "Iressa does not improve OS"
6-23-14: PPHM files Opposition to CSM’s Motion For Partial Summary Judgment - excerpts: http://tinyurl.com/q8xwd4v
. . .Declaration of Joseph Shan (VP/Clin+RegAffairs): http://tinyurl.com/kdgllxn
. . .Declaration of Jeffery Masten (VP/Quality): http://tinyurl.com/oru9p5q
3-28-14: Peregrine files 1st Amended Complaint vs. CSM (13pgs) http://tinyurl.com/lsgf5lz
...Pg6: ”[as of 4-15-10], CSM had already secretly & unilaterally swapped the A & B arms so that those patients that were randomized in the A arm (CTL) and supposed to receive placebo treatments, were actually receiving 1MG Bavi treatments, and vice-versa. Peregrine’s Fall’12 investigation revealed that CSM committed other labeling & distribution errors affecting the A & B arms above & beyond the swap of the A& B arms noted above.”
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
2-19-13: Topline Data Update from 2nd-Line NSCLC Trial after data discrepancies review http://tinyurl.com/ansqcea
…60% improvement in MOS: 3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined 1mg & Doxy+placebo arms), HR=.73, p=.217
6-5-13: FTM's table of MOS data in 15 prior Doxy 2nd-Line NSCLC trials (Bavi's 60% MOS Improvement is Tops) http://tinyurl.com/m886ctb
1-25-13: MLV's George Zavoico recaps 2ndLine/NSCLC data errors & current status of PPHM's review http://tinyurl.com/b9u4pk8
...GZ: "This means that patients randomized into the high dose arm were administered Bavi correctly, whereas some of the patients in the placebo arm were administered low dose Bavi and some in the low dose Bavi arm were administered placebo. More importantly, the findings suggested that the MOS of 13.1 mos. in the high dose arm was likely to be valid. Even by historical measures, this is a remarkable result, since docetaxel's product insert lists the MOS of NSCLC patients receiving this widely used drug as 2nd-Line therapy in 2 trials as 5.7 & 7.5 mos. In effect, adding Bavi doubled the MOS. In our view, this was an extraordinary stroke of luck. If the high dose arm had been affected by the coding discrepancy, Peregrine would have been in a completely different & unfortunate position… Moreover, Peregrine must determine how best to present its case to the FDA. Will the historical controls be sufficient to justify moving Bavi into a Phase III pivotal trial, or will Peregrine have to pool the results of the placebo & low-dose arms and use that as a comparator to argue for moving ahead? A simple average of the placebo & low-dose arms results in a new control MOS of about 8.4 mos., still several months less than that of the high dose arm. This quick analysis results in about a 5-mo. survival advantage, a substantial prolongation for patients with second-line NSCLC and likely to justify moving Bavi into a pivotal Phase III trial in 2013, in our view."
1-7-13 PPHM PR - Review Update: "discrepancies are isolated to the placebo and 1 mg/kg arms; no evidence of discrepancies in the 3 mg/kg arm… Peregrine is taking a very conservative approach toward analyzing the results by combining the placebo & 1mg/kg arms into one treatment arm (control arm), and comparing to the 3mg/kg arm. This analysis indicates that the 3 mg/kg arm continues to show favorable TRR's, PFS, and OS over the new combined control arm. Peregrine expects to announce more detailed results from the analysis in the near term when it is completed." http://tinyurl.com/asup54d
9-24-12: Major Discrepancies found in 2nd-Line NSCLC Ph.2B Treatment Group Coding by Indep. Third-Party Vendor http://tinyurl.com/8r9zcqy
…"Investors should not rely on clinical data that the company disclosed on or before Sept. 7, 2012 from its Ph.2 Bavi trial in patients with 2nd-Line NSCLC or any presentations or other documents related to this Ph.2 trial."
9-24-12: Peregrine sues CSM Over Bavi Ph.2B 2nd-Line NSCLC Clinical Trial Mix-Up http://tinyurl.com/8fpgngu
…CSM = Clinical Supplies Management Inc., Fargo ND http://www.csmondemand.com
...1-17-13: Peregrine's lawsuit against CSM for "breach of contract & negligence" SERVED http://tinyurl.com/a7zrgys
…9-10-12 CEO Steve King, QtlyCC ( http://tinyurl.com/8nkwrml )
……"These are truly remarkable results (statistically doubling MOS) that are not only great for the pgm… but also great news for the NSCLC patients in the trial…"
…9-10-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/8nkwrml )
……"The NSCLC data we announced 9-7-12 has far exceeded our expectations, and I hope that you're as excited as I am with bavituximab's potential. I feel strongly that Peregrine should be recognized for having the corporate courage to conduct the rigorous, randomized placebo-controlled Phase II trial that provided these robust data and that provide the basis for us to plan for a pivotal Phase III program."
...9-7-12: PPHM Press Release about Dr. Gerber's plenary at ASTRO/Thoracic/Chicago: http://tinyurl.com/96wrrso
…"The interim data showed a statistically significant improvement in OS (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS (12.1/13.1mos. vs. 5.6mos.) in the Bavituximab-containing arms compared to the [Docetaxel] ctl-arm."
......VP Joe Shan's 15min. Webcast & Slideshow recapping Dr. David Gerber's 9-7-12 ASTRO/Chicago Plenary: http://tinyurl.com/96wrrso
…8-15-12 CEO Steve King, Wedbush/NYC ( http://tinyurl.com/8mhrtld )
......"As we're sitting here today, we have still not reached the # of events for MOS in either of the Bavituximab arms - and, in fact, we still have patients that are on treatments." Q&A: "it's going to be a very positive MOS result, it's just a matter now of magnitude."
…7-16-12 CEO Steve King, QtlyCC ( http://tinyurl.com/cs7spbz )
......"The strength of this 2nd-Line NSCLC data (esp. MOS trends) in this large area of high unmet medical need has also sparked a surge in partnering discussions that has included over 15 in-person partnering meetings since that time with major players in oncology, with all discussions ongoing and addl. parties showing interest. Our goal for the program is to position ourselves, along with a potential partner, to initiate Ph3 by mid-2013, which means an EOP2 meeting by yr-end'12. It would be ideal to have a partner on board to participate in the EOP2 meeting, and we have communicated this to interested parties and they agree."
…7-16-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/cs7spbz )
……"We've been working very hard and very actively on the next steps in our Bavi 2nd-Line NSCLC pgm, given the favorable data that we've seen. As you can imagine, with data like this, there are many things that we need to consider. One consideration is that, should the data continue to trend the way it is, particularly in survival, this opens a door for potential discussions around a pathway for Accelerated Approval. At this point, all options are being considered, with Peregrine working towards the most efficient path forward from a regulatory standpoint." Q&A: "…all in all, I think the data is extremely compelling and I think it makes a really good case. Certainly, I think, I've seen a lot of Ph2 & Ph3 data, and this is as compelling Ph2 data as I've ever seen. So, I'm very comfortable proposing a meeting with the FDA for Q4'12."
…7-12-12 CEO Steve King, JMP-Conf/NYC ( http://tinyurl.com/csdclwb )
……"Re: 2nd-Line/NSCLC trial, the most thrilling thing is the fact that, even though we'd reached MOS for the ctl-arm(Doce) at end of Apr'12 of LESS THAN 6MOS, the majority of patients are still alive (today) in both Bavi arms, and we expect that to continue for some period of time still. Ph3 planning is underway already; our goal is to start this Ph3 by mid'13, meaning an EOP2 meeting with the FDA in Q4'12; our goal is to bring a partner on board, ideally in time for that EOP2 meeting, certainly before the beg. of the Ph3 trial."
…5-21-12: TopLine data n=117 for Bavi/3mg+Doce arm: ORR=17.9%/PFS=4.5mos (vs. CTL 7.9%/3mos) http://tinyurl.com/73aeyxj
......Importantly, MOS for CTL-arm "< 6 mos", but not yet reached in both Bavi arms.
...10-6-11: Enrollment complete. http://tinyurl.com/3m9re39
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to ~45 sites, expect enroll. comp. "early in Q4/2011", data unblinding 1H'12. http://tinyurl.com/6k6y2as
…3-17-10/Roth, CEO S.King: "We refer to this trial as a Registrational Phase II Study, because we believe that if we have results anywhere near approaching what we saw in the earlier [India] study, it could be a conduit for Accelerated Approval."
...6-4-10: Ph.2b randomized reg. trial Open for enrollment: http://tinyurl.com/25v22qk
……"up to 120 refractory patients at ~30 clinical sites; goal: fully-enroll by mid'11, topline data by y/e'11."
5-5-15 Webcast/workshop: “New Perspectives in Immuno-Oncology in Lung-Cancer” (pt1 of 2) – overview/general level, targeted to PATIENTS…
5-5-15/Webcast – CancerCare ( http://CancerCare.org )
“New Perspectives in Immuno-Oncology in the Treatment of Lung Cancer”
http://www.cancercare.org/connect_workshops/464-immuno_oncology_treatment_lung_cancer_2015-05-05
WEBCAST: http://engage.vevent.com/index.jsp?eid=2860&seid=630 (1hr.)
Supported by: BMS, Lilly, Genentech, AstellasUS (650+ participants)
• Matthew D. Hellmann, MD - Memorial Sloan Kettering CC
• Scott N. Gettinger, MD - Yale-New Haven (Smilow Cancer Hosp)
• Justin F. Gainor, MD - Harvard Medical School (Mass-Gen-Hosp)
Objective: Drugs that “Take away the Breaks that Cancer puts on the Immune System”
Dr.Gainor: PD-1 / PD-L1 Inhibitors – a sizable % of patients don’t respond – can we find ways to improve the # of people that respond? => Use trials to guide us to COMBINATIONS w/vaccines. or w/other I-O drugs, or w/Targeted-Therapies.
The immune system is so complex that there is actually a series of gas pedals and a series of brakes. Which brakes should we be applying, or should we combine one drug that releases one of the brakes or one drug that punches on the gas? So, Combinations may be a fruitful strategy – clinical trials will help answer this.
Whoever posted “focus on ASCO” last-yr admitted later it was posted in jest and not true – don’t remember who the poster was, but it’s that sort of thing that irritates me, because it results in the same misinformation being repeated over & over – perfect example, TODAY.
4-30-15: HTWR Q1/Fins q/e3-31-15 Revs=$70mm
HeartWare International Reports $70.0mm In Q1/2015 Revenue
• 713 HeartWare® Systems sold worldwide in Q1/2015
• U.S. revenue increased 25% over Q1/2014 to $42.2mm
• Intl. revenue of $27.8mm; reflects unfavorable currency trends
• Next-gen. MVAD® System targeted for 1st clinical use in Q2 2015
• Conference call today at 8:00amET
http://ir.heartware.com/phoenix.zhtml?c=187755&p=irol-newsArticle&ID=2042157
FRAMINGHAM, April 30, 2015: HeartWare Intl. (NASDAQ: HTWR), a leading innovator of less invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure, today announced revenue of $70.0mm for Q1 ended March 31, 2015, a 5% increase compared to $66.5mm in revenue for the same period of 2014. Currency fluctuations negatively impacted total revenue growth by approx. $5mm, or nearly 8%, in the Q1/15, as compared to the same period in 2014.
"Continued U.S. commercial expansion drove the second-largest unit sales quarter for HeartWare, although year-over-year U.S. growth was offset by a decrease in intl. unit sales when compared to the exceptionally strong intl. sales we achieved in Q1/14," said Doug Godshall, President and CEO. "We continue to see enthusiastic support of the HeartWare® System around the world, with the addition of 6 intl. and 5 U.S. customers during Q1, which increases our global customer base to more than 280 hospitals."
During Q1/15, 713 HeartWare® Ventricular Assist Systems were sold globally, which represents a 7% increase from 665 units sold in Q1/2014. During the quarter, U.S. revenue, generated through the sale of 381 units, was $42.2mm, a 25% increase from $33.8mm in Q1/2014. Revenue from international markets was $27.8mm, a decrease of 15% from $32.7mm in Q1/2014, while intl. unit sales decreased 6% to 332 units in Q1/15. Intl. unit sales decreased primarily in distributor territories, which have a greater tendency to fluctuate on a quarterly basis.
"During the quarter, we continued to advance our clinical trials for the HVAD System, with enrollment in the supplemental destination therapy study nearing completion; follow-up concluded for our Japan clinical trial; and commencement of HVAD LATERAL, a trial designed to evaluate a less-invasive thoracotomy implant technique," noted Mr. Godshall. "Regarding our pipeline, we recently completed training with investigators and are eager to commence first implants in the CE Mark study for our next-generation MVAD System later this quarter, and we plan to submit our Investigational Device Exemption protocol to FDA for review in the coming weeks."
. . .
GM% improved to 68.5% in Q1/2015, compared to 65.5% in Q1/2014, reflecting lower cost of goods and efficiencies associated with increased mfg.
. . .
Net loss for Q1/2015 was $14.5mm, or $.85 per basic & diluted share, compared to a net loss of $19.4mm, or $1.15 for Q1/2014.
. . .
At 3-31-15, HeartWare had ~ $174mm of cash, cash equivalents and investments, compared to $180mm at 12-31-14. . .
AACR’15 (3 Abstracts, 2 PR’s): Bavi Clinical TransData/Lung & PreClinData/Melanoma/Breast – Moffitt/S.Antonia, UTSW, Univ. of Calif/Irvine, Peregrine
AACR2015 PR #1 4-20-15:
Data Presented at AACR Demonstrate Peregrine Pharmaceuticals' Bavituximab Induces Immune Activation in PD-L1 Negative NSCLC Tumors
• Bavituximab Alone and in Combination with Docetaxel Elicit a Tumor-Specific Immune Response in PD-L1 Negative Tumors Extracted from NSCLC Patients
• Immune Modulating Results Consistent with Previously Conducted Translational Studies in Liver Cancer
• Presented Data Further Support Clinical Studies Evaluating the Effectiveness of Bavituximab Immunotherapy Combinations in Patients with PD-L1 Negative Tumors
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=907312
TUSTIN, April 20, 2015: Peregrine Pharmaceuticals, Inc. (Nasdaq:PPHM) (Nasdaq:PPHMP) today announced the presentation of data from clinical translational studies of the company's phosphatidylserine (PS)-targeting immunotherapy bavituximab. Initial data from a pilot study of clinical translational ex vivo cultures show that bavituximab, both alone and with docetaxel, elicits evidence of a tumor-specific immune response in patients with human adenocarcinoma of the lung and that bavituximab exhibits an impact on tumors with negative PD-L1 expression. These data were presented yesterday, Sunday, April 19th, in a poster presentation at the 106th Annual Meeting of the American Association for Cancer Research (AACR) being held in Philadelphia, Pennsylvania from April 18-22, 2015. Bavituximab is currently being evaluated in second-line non-small cell lung cancer (NSCLC) as part of the SUNRISE pivotal Phase III clinical trial.
"Our translational findings of a cytokine profile that reflects an immune response following either bavituximab single-agent or combination treatment are encouraging. Furthermore, these preliminary translational data show that tumors with negative PD-L1 expression and low levels of PD-1 expression on CD8+ tumor infiltrating T cells showed immune response to bavituximab treatment ex vivo," said Sigrid M. Volko, Ph.D., CLP the lead investigator on the study and President & CEO of Nilogen Oncosystems [=Moffit Startup: http://otmc.moffitt.org/startups.aspx ]. "It is an exciting time for the field of immunotherapy and the data we have been generating support that bavituximab has the potential to activate a tumor specific immune response in patients with PD-L1 negative tumors that generally do not respond as well to PD-1 or PD-L1 inhibitors."
In a poster [ AACR’15 Abstract #274] titled: "Bavituximab Modulates Tumor Microenvironment and Activates CD8+ Tumor Infiltrating Lymphocytes in a Patient-derived 3D ex vivo System of Lung Cancer," researchers from Moffitt Cancer Center [Dr. Scott Antonia et al], Nilogen Oncosystems, and Peregrine present initial data from a pilot translation study analyzing tumor tissue from 6 lung cancer patients to evaluate the immunomodulatory effects of bavituximab in a human ex vivo model of NSCLC. Researchers generated 3D tumor microspheres from tumor tissues produced at the time of surgical resection. These microspheres were treated ex vivo for 36 hours with bavituximab, bavituximab and docetaxel, or controls in the presence of Interleukin-2 (IL-2), a cytokine that regulates the activities of white blood cells that are responsible for immunity. Data show that bavituximab as a single agent or in combination with docetaxel induces lymphocyte activation in tumors as demonstrated by a significant increase in interferon-gamma (IFNy), TNF-alpha, and GM-CSF secretion when compared to tumors treated with IL-2 control. Researchers also concluded that the immune response to treatment with bavituximab correlates with negative PD-L1 expression in the resected tumor tissue and low PD-1 expression on CD8+ tumor infiltrates thus serving as a potential prognostic biomarker of positive response to bavituximab treatment.
"These data are exciting as the immune responses seen in this translational lung cancer study mirror what we saw in translational data from a liver cancer clinical trial and is perfectly aligned with what has been seen preclinically to date. It was also encouraging to see how well these data further support our ongoing Phase III SUNRISE trial in that the combination of bavituximab and docetaxel induces immune activity," said Joseph Shan, MPH, VP of Clinical and Regulatory Affairs at Peregrine. "These data are playing a key role in advancing the clinical portion of an Immuno-Oncology Development Program which has been built upon a growing body of favorable data supporting the potential combination of bavituximab and checkpoint inhibitors. Specifically, these data show that activating the immune system in this negative PD-L1 patient population provides a strong rational for combining bavituximab with inhibitors of the PD-1/PD-L1 pathway. We look forward to detailing our plans for additional immuno-oncology combination clinical trials in the near future."
Liver Cancer Translational Data
In November, clinical translational data from the company's Immuno-Oncology Development Program were presented assessing changes in immune response pre- and post-treatment in 6 patients participating in a Phase II IST evaluating bavituximab in combination with sorafenib in advanced hepatocellular carcinoma. Data from this translational sub-study of patients, show that half of the patients had an increase in tumor fighting immune cells following one cycle of treatment, similar to what has been shown for PS-targeting antibodies in multiple preclinical cancer models. In addition, the increase in immune response was associated with patients that remained on study treatment for longer time periods, consistent with an immunotherapeutic mechanism and suggest the possibility of a clinically meaningful anti-tumor immune response. Immune responding patients also had increased infiltration of CD8 T-cells in the tumor microenvironment which correlated with a prolonged time to disease progression. In addition, these immune responders expressed lower levels of PD-1, an established marker of T cell activation and disease outcome, prior to the initiation of therapy, followed by a measurable rise.
A link to this presentation is located on the front page of Peregrine's website at http://www.peregrineinc.com .
http://www.peregrineinc.com/publications/posters-and-presentations.html
POSTER #274 PDF: http://www.peregrineinc.com/images/stories/pdfs/aacr_2015_lung.pdf

…ABOUT BAVITUXIMAB & ABOUT PEREGRINE: see PR#2 below.
= = = = = = = = = = = = = = = = = = =
AACR2015 PR #2 4-21-15:
Preclinical Data Presented at AACR Demonstrate Synergistic Anti-Tumor Effects of Peregrine Pharmaceuticals' Phosphatidylserine (PS)-Targeting Antibodies With Immune Checkpoint Inhibitors in Models of Melanoma and Breast Cancer
• Combination Treatment Reduces Tumor Immune System Blockade and Enhances Tumor Specific Immune Responses
• Studies Reveal Significant Increases in Tumor-Infiltrating CD8+ T Cells and Immune-Activating Cytokines while Decreasing Tumor-Promoting Macrophages and Myeloid Cells
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=907609
TUSTIN, April 21, 2015: Peregrine Pharmaceuticals, Inc. (Nasdaq:PPHM) (Nasdaq:PPHMP) today announced the presentation of data from preclinical studies demonstrating the combination of phosphatidylserine (PS) blockade with anti-PD-1 or anti-CTLA-4 immune checkpoint inhibitors promoted strong, localized and enhanced efficacy in models of melanoma and breast cancer. These data were presented at the 106th Annual Meeting of the American Association for Cancer Research (AACR) being held in Philadelphia, Pennsylvania from April 18-22, 2015. Peregrine's lead PS-blocking antibody, bavituximab, is currently being evaluated in second-line non-small cell lung cancer (NSCLC) in a Phase III clinical trial named Sunrise.
"The data presented this week at AACR showed in much greater detail the collective ability of PS and PD-1 blockade to change the immune response in melanoma and breast cancer models," said Jeff T. Hutchins, Ph.D., VP of Preclinical Research at Peregrine. "These data showed that blocking PS resulted in a decrease in immune-blocking cell types such as myeloid-derived suppressor cells and M2 macrophages while increasing the number of activated T-cells that are able to specifically recognize and kill tumor cells which set the stage for anti-PD-1 therapy that keeps the immune response going. The result were synergistic anti-tumor effects in established melanoma and breast cancers. The consistency of the data generated from pre-clinical experiments, and, more recently, in clinical translational studies is impressive. When taken together with the additional lung cancer translational data presented Sunday, we are obtaining a clearer picture as to the potential of bavituximab in different immuno-oncology combinations. We look forward to presenting additional supporting data over the coming months."
In a poster [ AACR’15 Abstract #252] titled: "Antibody-mediated Phosphatidylserine Blockade Significantly Enhances the Efficacy of Immune Checkpoint Blockades in K1735 & B16 Mouse Melanoma Models," researchers from Peregrine, UT Southwestern Medical Center and the University of California at Irvine presented data assessing the antitumor effect of the combination of PS blockade and anti-CTLA-4 or anti-PD-1 antibodies in preclinical models of melanoma. Both combinations showed significantly superior tumor growth inhibition over single treatment, with many subjects achieving complete tumor regressions. The combination treatment showed significantly greater total and functional tumor-infiltrating CD8+ T, more IL-2- and interferon gamma (IFN?)-producing splenic T cells, and lower number of splenic myeloid derived suppressor cells myeloid-derived suppressor cells (MDSCs) than did single treatment. In addition, the ratio of M2 to M1 macrophages in the tumor was significantly lower in the combination treatment than that in single treatment. Finally, no toxicity was observed in any of the treatment groups following multiple treatment doses.
In a poster [ AACR’15 Abstract #4289] titled: "Targeting of Phosphatidylserine by Monoclonal Antibodies Enhances the Activity of Immune Checkpoint Inhibitors in Breast Tumors," Peregrine researchers presented data demonstrating that PS blockade enhances the anti-tumor activity of combination therapies including anti-PD-1 antibodies in an immune competent model of breast cancer. Tumor growth inhibition correlates with statistically significant increases in the infiltration of CD8+ T cells and a reduction of myeloid-derived suppressor cells (MDSCs). The combination of these mechanisms promotes strong and localized anti-tumor responses without the side-effects of systemic immune activation.
Copies of these presentations can be found on the front page of Peregrine's website at http://www.peregrineinc.com .
POSTER #252 PDF: http://www.peregrineinc.com/images/stories/pdfs/aacr_2015_melanoma.pdf

POSTER #4289 PDF:
http://www.peregrineinc.com/images/stories/pdfs/gong_2015_aacr_p.pdf

About Bavituximab: A Targeted Investigational Immunotherapy
Scientific research has shown that tumors evade immune detection due partly to the expression of phosphatidylserine, or PS, a highly immunosuppressive molecule. Peregrine's immuno-oncology development program has developed bavituximab, an investigational monoclonal antibody that targets and binds to PS, blocking its immunosuppressive effects while activating tumor fighting immune cells, thus enabling the immune system with the ability to better recognize and fight cancer. Bavituximab's immune-stimulatory mechanism-of-action data is the subject of a manuscript published in the October 2013 issue of the American Association for Cancer Research (AACR) peer-reviewed journal, Cancer Immunology Research [8-19-13: http://tinyurl.com/mhjftka PDF]. Bavituximab is currently being evaluated in several solid tumor indications, including non-small cell lung cancer (the SUNRISE Phase III trial), breast cancer, liver cancer, rectal cancer and advanced melanoma. In January 2014, bavituximab received Fast Track designation by the U.S. Food and Drug Administration (FDA) for the potential second-line treatment of patients with non-small cell lung cancer.
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials for the treatment and diagnosis of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. The company is also advancing a molecular imaging agent, 124I-PGN650, in an exploratory clinical trial for the imaging of multiple solid tumor types. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com.
Safe Harbor *snip*
CONTACT: Christopher Keenan, Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
= = = = = = = = = = = = = = = = = = =
Apr18-22 2015: AACR 106th Annual Meeting, Phily http://www.aacr.org/2015
Abstracts: http://www.abstractsonline.com/plan/start.aspx
PEREGRINE’s Posters:
• Abstract #274(Apr19): “Bavituximab Modulates Tumor Microenvironment and Activates CD8+ Tumor Infiltrating Lymphocytes in a Patient-Derived 3D Ex-Vivo System of Lung Cancer”
• Abstract #252(Apr19): “Antibody-Mediated Phosphatidylserine Blockade Significantly Enhances the Efficacy Of Immune Checkpoint Blockades in K1735 & B16 Mouse Melanoma Models” [Bavi+Ipilimumab(BMS’s Yervoy)]
• Abstract #4289(Apr21): “Targeting of Phosphatidylserine by Monoclonal Antibodies Enhances the Activity of Immune Checkpoint Inhibitors in Breast Tumors”
Sun. Apr 19, 2015, 1-5pm, Abstract#274, Session: TUMOR MICROENVIRONMENT/INNATE IMMUNE ACTIVATORS
”Bavituximab Modulates Tumor Microenvironment and Activates CD8+ tumor Infiltrating Lymphocytes in a Patient-derived 3D Ex Vivo System of Lung Cancer”
Soner Altiok 1, Melanie Mediavilla-Valera 2, Jenny Kreahling 2, David Noyes 2, Tiffany N. Razabdouski 2, Nikoletta L. Kallinteris 3, Joseph Shan 3, Scott Antonia 2
1=Nilogen Oncosystems, Tampa, FL; 2=Moffitt CC/Tampa; 3=Peregrine Pharm.
ABSTRACT: Bavituximab is a monoclonal antibody directed against the membrane phospholipid phosphatidylserine exposed on the outer leaflet of tumor and vascular endothelial cells of the tumor microenvironment. Bavituximab modulates the tumor microenvironment by blocking PS-mediated immune suppression and activating cytotoxic T lymphocyte anti-tumor responses.
In this study, we tested the immunomodulatory effect of bavituximab using a proprietary 3D ex vivo tumor microsphere technology. Upon obtaining informed consent, fresh tumor tissue from lung cancer patients were collected at the time of surgical resection. Tissue was processed for characterization of the tumor microenvironment and potential immunosuppressive mechanisms such as expression of PD-1, CTLA4, LAG3, TIM3, BTLA, and Adenosine A2AR. 3D tumor microspheres were prepared and cells were treated ex vivo with f(ab)’2 version of bavituximab, bavituximab, docetaxel, and a combination of bavituximab and docetaxel for 36 hours within the 3D tumor microsphere simulating an intact tumor microenvironment made up of tumor infiltrating lymphocytes (TIL) and myeloid cells. At the end of the treatment, TILs were analyzed by flow cytometry for cell activation and changes in CD4, CD8 and Treg (CD25+/CD127-) subpopulations. A multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFN-gamma in culture media as a surrogate of TIL activation.
Preliminary results indicate the combination of bavituximab & docetaxel can induce TIL activation as demonstrated by a significant increase in IFN-gamma secretion when compared to tumors treated with control or either agent alone. Flow cytometry analysis revealed that this effect was associated with low PD-1 expression on CD8 cells, but did not correlate with other known immune-modulating receptors.
This lung patient derived ex-vivo approach indicates that bavituximab in combination with docetaxel can elicit a tumor specific immune response in human adenocarcinoma of the lung. This effect involves, at least in part, activation of CD8+ TIL and increased inflammatory cytokine production by lymphoid and myeloid cells. In addition, we have observed low PD1 expression as a potential prognostic biomarker of positive response to bavituximab treatment.
POSTER #274 PDF: http://www.peregrineinc.com/images/stories/pdfs/aacr_2015_lung.pdf
Sun. Apr 19, 2015, 1-5pm, Abstract#252, Session: IMMUNE CHECKPOINTS
“Antibody-Mediated Phosphatidylserine Blockade Significantly Enhances the Efficacy of Immune Checkpoint Blockades in K1735 & B16 Mouse Melanoma Models”
Bruce Freimark 1, Jian Gong 1, Dan Ye 2, Rolf Brekken 2, Shen Yin 1, Jeff Hutchins 1, Van Nguyen 1, Chris Hughes 3, Xianming Huang 2
1=Peregrine Pharm.; 2=UTSW-MC/Dallas; 3=Univ. of Calif/Irvine
ABSTRACT: Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane and becomes exposed abundantly on tumor endothelial cells, tumor-secreted microvesicles, and tumor cells in response to chemo- and radiotherapy. Extensive studies have shown that PS is a global immune checkpoint and major contributor to tumor immunosuppression, which promotes the expansion of immunosuppressive cells, such MDSC & M2 macrophages, inhibits DCs maturation, while stimulates them to secrete immunosuppressive mediators. We have shown that PS targeting can override PS-mediated immunosuppression, reactivate innate tumor immunity, and evoke adaptive antitumor immunity. In the present study, we assessed the antitumor effect of the combination of PS blockade and anti-CTLA-4 or anti-PD-1 antibodies in B16 & K1735 melanoma models. Both combinations showed significantly superior tumor growth inhibition over single treatment, with many mice achieving complete tumor regression. Flow cytometry analysis showed that the combination treatment had significantly greater total and functional tumor-infiltrating CD8+ T, more IL-2- and IFNy-producing splenic T cells, and lower number of splenic MDSCs than did single treatment. In addition, the ratio of M2 to M1 in the tumor was significantly lower in the combination treatment than that in single treatment. Finally, no toxicity was observed in any of the treatment groups following multiple treatment doses. These data suggest that combination of PS blockade with immune checkpoint blockade promotes strong, localized, enhanced therapeutic efficacy without the side-effects of systemic immune activation and represents a promising combinatorial strategy for cancer immunotherapy. A chimeric PS-targeting antibody, bavituximab, is being used in combination with chemotherapy to treat patients with solid tumors in multiple late-stage clinical trials. These data support our Phase I IST evaluation of our PS-targeting antibody bavituximab in combination with ipilimumab in advanced melanoma patients.
POSTER #252 PDF: http://www.peregrineinc.com/images/stories/pdfs/aacr_2015_melanoma.pdf
NOTES:
Ipilimumab=BMS’s Yervoy (anti-CTLA-4) http://www.yervoy.com ]
O. PPHM’s 6th IST Trial: Bavi+Ipilimumab(Yervoy) vs. Adv.Melanoma (Ph1b, random, open-label, 2arms, n=24)
Protocol (UTSW): http://www.clinicaltrials.gov/ct2/show/NCT01984255 (PI: Dr. Arthur Frankel - see "Researching for Cures" http://youtu.be/0zLAxjFny5Q )
UTSW's listing: http://www.utsouthwestern.edu/research/fact/detail.html?studyid=STU%20102013-007
...4-23-14: Bavi+Yervoy IST trial initiated: http://tinyurl.com/km7krcm
Tue. Apr 21, 2015, 1-5pm, Abstract#4289, Session: NOVEL IMMUNOMODULATORS
”Targeting of Phosphatidylserine by Monoclonal Antibodies Enhances the Activity of Immune Checkpoint Inhibitors in Breast Tumors”
Jian Gong, Shen Yin, Van Nguyen, Jeff Hutchins, Bruce D. Freimark - Peregrine Pharm.
ABSTRACT: Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on tumor vascular endothelial cells (ECs) and tumor cells. PS exposure becomes enhanced in response to chemotherapy, irradiation, and oxidative stresses in the tumor microenvironment. PS exposure in tumors promotes an immunosuppressive microenvironment which includes the recruitment of myeloid derived suppressor cells (MDSCs), immature dendritic cells and M2-like macrophages, as well as, the production of anti-inflammatory cytokines. Binding of PS targeting antibodies on tumor endothelial cells, tumor cells and their secreted microparticles triggers an Fc-FcR mediated pro-inflammatory cellular and cytokine response that reverses the immunosuppressive PS meditated checkpoint, thereby enhancing anti-tumor immunity. A chimeric PS-targeting antibody, bavituximab, is being used in combination with chemotherapy to treat patients with solid tumors in multiple late-stage clinical trials. Using breast tumors in immune competent mice, we demonstrate PS targeting antibodies enhance the anti-tumor activity of combination therapies including anti-PD-1 antibodies. Tumor growth inhibition correlates with statistically significant increases in the infiltration of CD8+ T cells and a reduction of myeloid-derived suppressor cells (MDSCs). The combination of these mechanisms promotes strong, localized, anti-tumor responses without the side-effects of systemic immune activation.
POSTER #4289 PDF: http://www.peregrineinc.com/images/stories/pdfs/gong_2015_aacr_p.pdf
= = = = = = = = = =PIPER JAFFRAY 4-20-15 COMPANY NOTE:
”Bavi – Not Just Another IO Candidate”

= = = = = = = = = =ROTH CAPITAL 4-20-15 UPDATE:
”Positive on PPHM Following Bavituximab AACR’15 Presentation”
Roth Capital affirmed Peregrine Pharma (Nasdaq: PPHM) at Buy with a price target of $5 after the company announced the presentation of data from clinical translational studies of the company's phosphatidylserine (PS)-targeting immunotherapy bavituximab. Peregrine said that initial data from a pilot study of clinical translational ex vivo cultures show that bavituximab, both alone and with docetaxel, elicits evidence of a tumor-specific immune response in patients with human adenocarcinoma of the lung and that bavituximab exhibits an impact on tumors with negative PD-L1 expression.
Analyst Joseph Pantginis commented, ”We generally do not write on preclinical data, however we believe these data continue to represent important incremental steps in defining bavituximab's immunotherapy mechanism of action. In the lung model, immune response to treatment correlates with negative PD-L1 expression and low PD-1 expression on CD8+ tumor cells, which could represent a prognostic biomarker. We believe these data represent the closest ex-vivo model of the patient population being assessed in the ongoing Phase III SUNRISE study in 2nd-line NSCLC. Nilogen’s drug discovery platform is described in the figure below. Over the next 12-mos, we expect to see an increase in visibility on the shares based on 1) completion of enrollment into the Phase III SUNRISE study, 2) incremental data updates from earlier clinical studies, and 3) increasing visibility for bavituximab's mechanism of action within the immuno-oncology landscape. The ~600 patient SUNRISE study in 2nd-line NSCLC is expected to complete enrollment by the end of 2015…”
http://www.streetinsider.com/Analyst+Comments/Roth+Capital+Remains+Positive+on+Peregrine+Pharma+%28PPHM%29+Following+Bavituximab+Presentation/10471273.html
http://www.roth.com – Universe: http://roth.bluematrix.com/docs/pdf/BLUE.pdf

= = = = = = = = = = = = = = = = =
Bavituximab MOA & Clinical Data: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
BAVI MOA: 5-26-11 Dr.Thorpe's keynoter at Recombinant-Mabs/Barcelona http://tinyurl.com/3klpodc & http://tinyurl.com/3m33h33