Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Update: TG Therapeutics 4.1M share Spot Secondary priced at $6.15 The deal size was increased to $25.2M in common stock from $15M in common stock. Cantor Fitzgerald acted as sole book running manager for the offering.
Nice price if this is what $TGTX gets and the size is reasonable. This is not confirmed news until management releases the news in a PR.
TG Therapeutics 4.1M share Spot Secondary priced at $6.15 »
Read more at:
https://thefly.com/landingPageNews.php?id=2873170
TG Therapeutics (TGTX)
In a report released today, Edward White from H.C. Wainwright reiterated a Buy rating on TG Therapeutics, with a price target of $20. The company’s shares closed yesterday at $4.80, close to its 52-week low of $3.32.
White said:
“Our $20 price target is based on the net present value of our revenue forecast through 2027, applying a 40% probability of success (POS) for ublituximab in CLL, a 40% POS for umbralisib in CLL, a 25% POS for both ublituximab and umbralisib in 40% POS for ublituximab in MS. We use a 4x price/sales multiple for these products and an early stage pipeline value of $0.23/share, which includes an anti-PD-L1 drug, and oral BTK inhibitor, and a CD47/CD19 bi-specific antibody, to arrive at our price target. Our P/S multiple of 4x is in-line with TG’s peers that range between 2-5x.”
https://www.markets.co/analysts-offer-insights-on-healthcare-companies-ocular-therapeutix-nasdaq-ocul-agile-therapeutics-nasdaq-agrx-and-tg-therapeutics-nasdaq-tgtx/199327/
Oral Duvelisib Potential New Option for Indolent Non-Hodgkin Lymphoma
Leah Lawrence Feb 26, 2019
The oral PI3K gamma inhibitor duvelisib produced an overall response rate of close to 50% in patients with heavily pretreated, double-refractory, indolent non-Hodgkin lymphoma (NHL) in the phase II DYNAMO study.
Results of the global trial showed that duvelisib may “provide a new oral treatment option for this patient population of which many are elderly and in need of additional therapies,” wrote Ian W. Flinn, MD, PhD, of Sarah Cannon Research Institute, and colleagues, in a study published in the Journal of Clinical Oncology.
Duvelisib is currently approved by the US Food and Drug Administration for relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (SLL), as well as follicular lymphoma after two or more prior systemic therapies.
This study in indolent NHL included 129 patients with either follicular lymphoma (42.2%), SLL (67.9%), or marginal zone B-cell lymphoma (38.9%). All patients were refractory to rituximab and chemotherapy or radioimmunotherapy. The treatment in the study consisted of twice-daily 25-mg duvelisib in 28-day cycles until progression, unacceptable toxicity, or death.
With a median follow-up of 32.1 months, the overall response rate was 47%. The majority of responses were partial responses (n = 59); only two patients had a complete response. Lymph node disease was reduced in 83% of patients.
Looking at each disease subtype, the overall response rates were 42%, 68%, and 39% for patients with follicular lymphoma, SLL, and marginal zone lymphoma, respectively.
“The efficacy demonstrated by duvelisib monotherapy is clinically meaningful, considering that nearly all patients had disease refractory to prior rituximab and chemotherapy, including the most recent prior therapy,” the researchers wrote.
According to the researchers, responses to duvelisib were both rapid and durable. The median time to response was 1.87 months, with the majority (84%) of patients responding by 4 months. Median duration of response was 10 months; about one-third of patients were estimated to remain in response by 1 year.
The most commonly occurring any-grade treatment-emergent adverse events were diarrhea (48.8%), nausea (29.5%), neutropenia (28.7%), fatigue (27.9%), and cough (27.1%). Among patients with at least one grade 3 or greater treatment-emergent adverse event (88.4%), the most common were neutropenia, diarrhea, anemia, and thrombocytopenia.
Commenting on the study, Ajay K. Gopal, MD, professor of medicine in the division of medical oncology at the University of Washington, told Cancer Network, “The data from the DYNAMO trial define another useful option with duvelisib for patients with follicular lymphoma who have received 2 prior therapies. In the long run the PI3K gamma inhibition of duvelisib may turn out to provide an important immunomodulatory role in combination with other agents compared with others in this class.
https://www.cancernetwork.com/news/oral-duvelisib-potential-new-option-indolent-non-hodgkin-lymphoma
Immutep to Present at Upcoming Industry Conferences
Tue February 26, 2019 8:00 AM|GlobeNewswire|About: IMMP
SYDNEY, Australia, Feb. 27, 2019 (GLOBE NEWSWIRE) -- Immutep Limited (IMMP) (Immutep (PRRUF) or the Company), a biotechnology company developing novel immunotherapy treatments for cancer and autoimmune diseases, today announced its participation in various upcoming industry conferences.
The Company will be presenting updated clinical data from the Phase I TACTI-mel trial in metastatic melanoma for its lead product candidate eftilagimod alpha (“efti” or “IMP321”) and new data relating to its pre-clinical product candidate, IMP761, a novel anti-LAG-3 agonist antibody for the treatment of auto-immune diseases, at industry conferences in March 2019.
Conference:
World Immunotherapy Congress USA 2019
Dates: 3-5 March 2019
Venue: Grand Hyatt, San Diego, USA
Presentation Title:
Two ACTive Immunotherapies (TACTI): Results of a Phase I trial with metastatic melanoma patients
Presenter:
Dr. Frédéric Triebel, CSO and CMO of Immutep
Conference:
14th Congress (CACOX) of ECCO (European Crohn’s and Colitis Organisation)
Dates: 6-9 March 2019
Venue: Bella Center, Copenhagen, Denmark
Presentation Title:
IMP761, a novel anti-LAG-3 agonist antibody for the treatment of auto-immune diseases
Presenter:
Mathieu Angin, Phd, Immutep Research Scientist
All presentations will be made available on the Company’s website.
About Immutep
Immutep is listed on the Australian Stock Exchange (IMM), and on the NASDAQ (IMMP) in the US.
Further information can be found on the Company’s website www.immutep.com or by contacting:
U.S. Investors:
Jay Campbell, Chief Business Officer, Immutep Limited
+1 (917) 860-9404; jay.campbell@immutep.com
Australian Investors/Media:
Matthew Gregorowski, Citadel-MAGNUS
+61 2 8234 0105; mgregorowski@citadelmagnus.com
U.S. Media:
Garth Russell, LifeSci Advisors
+1 (646) 876-3613; garth@lifesciadvisors.com
Source: Immutep Limited 2019 GlobeNewswire, Inc.
https://seekingalpha.com/pr/17424272-immutep-present-upcoming-industry-conferences
Final Results of a Phase 2 Multicenter Study of Ublituximab, a Novel Glycoengineered Anti-CD20 Monoclonal Antibody, in Patients with RMS
Day: Thursday, February 28, 2019
https://actrims.confex.com/actrims/2019/meetingapp.cgi/Paper/3892
An excellent way to play double catch up in the top 300 China stocks. They have been out of favor since 2015, so a lot of growth here.
$CHAU Direxion Daily CSI 300 China A Share Bull 2X Shares
Nice to watch the accumulation going on and larger blocks of 5000 plus purchases. The ETF has a lot of room to move back up and even more so with the talks ongoing this week in Washington which seem to be going well with the current press conference highlights just disclosed today.
China, Emerging Markets Lead 2019 Rebound For Global Equities
Across-the-board gains continue to mark this year's results for global equity markets, based on a set of exchange-traded funds that track the world's major regions and countries. At the top of the performance list so far in 2019: stocks in China, Latin America and Africa.
The strongest increase year to date is currently held by iShares MSCI China (NYSEARCA:MCHI), which is up 13.6% through yesterday's close (Feb. 21). After suffering a sharp loss in 2018, the ETF has rallied this year and on Thursday closed above its 200-day moving average for a second straight day.
One factor that appears to be supporting Chinese stocks lately: upbeat reports in recent days that Sino-US trade talks may soon lead to a solution for resolving the trade war between the world's largest economies. Bloomberg reports that President Trump is scheduled to meet with China's top trade negotiator today (Feb. 22), an encounter that may produce a preliminary deal on a new round of tariffs that the US is planning to impose next month.
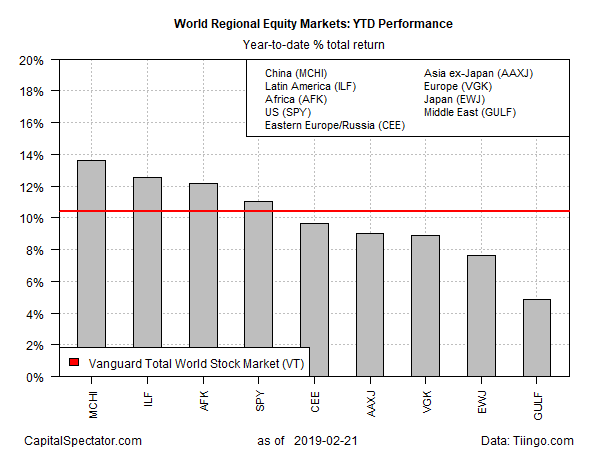
The second-strongest gain year to date is currently held by an ETF focused on Latin America. After a rough 2018 that took a hefty bite out of iShares Latin America 40 (NYSEARCA:ILF), the ETF is up 12.6% this year. In third place: VanEck Vectors Africa (NYSEARCA:AFK), which has also rebounded in 2019 and is currently posting a 12.1% return.
The US equity market is the fourth-strongest performer in this ranking. SPDR S&P 500 (NYSEARCA:SPY) is ahead by a solid 11.0% for 2019 - just slightly ahead of global equity market overall, based on the 10.4% year-to-date gain for Vanguard Total World Stock (NYSEARCA:VT).
The weakest year-to-date performer in the major region/country lineup: equity markets in the Middle East. After a strong run in 2018, WisdomTree Middle East Dividend (NASDAQ:GULF) has become a relative laggard this year. At Thursday's close, GULF was up 4.9% for 2019.
Foreign markets edged higher in Friday's trading, buoyed by expectations that the US-China trade war may be set to ease.
"Given that enough headway seems to have been made to warrant a meeting between Trump and the Chinese negotiator today, it appears more likely that the US will not raise the levies, which should help high-beta currencies and equities push higher," predicts Konstantinos Anthis, head of research at ADSS, a forex broker in Abu Dhabi.
https://seekingalpha.com/article/4243412-china-emerging-markets-lead-2019-rebound-global-equities
Seems Management has more faith in the company than does the market. The years i've owned/traded $VSTM, Management has not disappointed yet.
Verastem Oncology Announces Key Management Appointments
“Verastem Oncology continues to grow our operations across the U.S. in crucial functional areas, such as commercial management, clinical development, medical affairs, regulatory, quality assurance and investor relations and communications,” said Robert Forrester, President and Chief Executive Officer of Verastem Oncology. “We are delighted to welcome Amy, Bob and Erin to our team to support us in our mission of bringing new cancer therapies to patients and families battling these devastating diseases. We will greatly benefit from Amy’s successful product launch expertise and deep knowledge of the b-cell malignancies space. Bob is also an important addition that will help guide the organization and advancement of our pipeline of therapies to late-stage development. Erin’s extensive IR and communications experience in the biotechnology industry will be critical as we continue to increase our presence within the financial community.”
https://seekingalpha.com/pr/17417540-correcting-replacing-verastem-oncology-announces-key-management-appointments
IBB: This Is Only The Beginning
https://seekingalpha.com/article/4242004-ibb-beginning
Two good PR`s and the street does not care. It will be interesting when they are able to actually release some numbers going forward. On sidelines watching for now. GLTY
Trade optimism lifts stocks in Asia
Feb. 18, 2019 3:57 AM ET
Asian markets firmed overnight, after the Dow and Nasdaq boasted their eighth consecutive weekly gains, on hopes the U.S. and China would hammer out an agreement resolving their protracted trade war.
The two sides resume negotiations this week with President Trump saying he may extend a March 1 deadline for a deal.
China's credit growth also exceeded expectations in January, helping ease worries of a slowdown in the world's second largest economy.
Nikkei +1.8%; Shanghai +2.7%; Hang Seng +1.8%.
https://seekingalpha.com/news/3434176-trade-optimism-lifts-stocks-asia
Wonder if this had anything to do with the stocks momentum this week?
Resignation of CFO
On February 13, 2019, Mr. Joseph L. Damasio Jr., Vice President of Finance and Chief Financial Officer (“CFO”) of Pressure BioSciences, Inc. (the “Company”) informed the Company that he was resigning as CFO effective February 22, 2019.
Appointment of Acting CFO
On February 13, 2019, the Company appointed Mr. Richard P. Thomley as the Company’s Acting Chief Financial Officer.
Richard Thomley, age 67, was the Company’s Acting CFO from September 2013 through November 6, 2015 and was an independent financial consultant to the Company from March 2013 through September 2013. Prior to March 2013, Mr. Thomley was the Director of Finance of Kiva Systems, Inc. from June 2007 until January 2012. Mr. Thomley was the Chief Accounting Officer at Spire Corp. from January to June 2007. Mr. Thomley served as the Corporate Controller for Implant Sciences, Inc. from November 2004 until January 2007. Mr. Thomley was Chief Financial Officer at SynQor, Inc. from July 2000 to November 2004. Mr. Thomley has also held senior management positions in finance at Genesis Technical and Financial, Catamount Manufacturing, Inc., ChemDesign Corp., and Ansul Fire Protection Co. He has over 30 years of experience in U.S. GAAP accounting, internal management, financial reporting and analysis, and SEC reporting. Mr. Thomley is a Certified Public Accountant.
Family Relationships
There are no family relationships between Mr. Thomley and any other executive officer or member of the board of directors of the Company.
Related Party Transactions
There are no related party transactions with regard to Mr. Thomley reportable under Item 404(a) of Regulation S-K.
Material Plans, Contracts, or Arrangements
The Company does not have a written employment agreement with Mr. Thomley, however, the Company has orally agreed to pay Mr. Thomley $100 per hour for his services. Mr. Thomley is expected to devote approximately fifty (50) hours per month to oversee the Company’s financial and administrative operations.
https://ih.advfn.com/stock-market/USOTC/pressure-biosciences-inc-PBIO/stock-news/79284416/current-report-filing-8-k
Biotech Bonanza: The Mergers And Acquisitions Theme
https://seekingalpha.com/article/4240702-biotech-bonanza-mergers-acquisitions-theme
A recent analysis by EvaluatePharma revealed that based on the historical data over the past 5 years, a majority of the buyouts have occurred for products in the Pre-clinical and Phase II stages.
The lower number of Phase III buyouts is understandable as such assets are also more scarce, besides being at a higher valuation, compared to the volume of assets available in Pre-clinical and Phase II.
The analysis also indicated that the leading therapy area for buyouts remains Oncology, as it has been for many years.
BBC Daily Chart
insert-text-here
If earnings release is on time tomorrow I think they easily beat which will push up the stock.
Their Ultra Shear technology the company uses for separating CBD oil for consumables could be a market standard and used industry wide. For this small company that would be a huge stock mover.
$PBIO Pressure Biosciences is scheduled to release their next quarterly earnings announcement on Wednesday, February 13th 2019.
Pressure BioSciences, Inc. develops pressure cycling technology (PCT) solutions. Its PCT technology uses alternating cycles of hydrostatic pressure between ambient and ultra-high levels to control the actions of molecules in biological samples, such as cells and tissues from human, animal, plant, and microbial sources. The company offers Barocycler instrumentations, including Barocycler 2320EXT, Barozyme-HT48, Barocycler NEP3229, HUB440, HUB880, and The Shredder SG3. It also distributes cell disruption equipment, parts, and consumables. In addition, the company offers Barocycler consumable products, such as PCT MicroTubes, PCT-MicroCaps, PCT-Micro-Pestle, PULSE Tubes, and BaroFlex 8-well Processing Strips, as well as application specific kits. Pressure BioSciences, Inc. serves researchers at academic laboratories; government agencies; and biotechnology, pharmaceutical, and other life sciences institutions in the United States, Europe, and Asia. The company was formerly known as Boston Biomedica, Inc. Pressure BioSciences, Inc. was founded in 1978 and is based in South Easton, Massachusetts.
$PBIO CEO Ric Schumacher of Pressure BioSciences, Inc. (OTCQB: PBIO), returns to Stock Day to discuss holding over 20 patents that utilize high pressure, the many applications in life-sciences, and the multiple instruments that they create for various industries including nutraceuticals.
The CEOs interviewed on the Stock Day Podcast
https://upticknewswire.com/featured-interview-ceo-ric-schumacher-of-pressure-biosciences-inc-otcqb-pbio-11/
Hemp's FDA Declassification Opens Door to Massive CBD Market Growth
Pressure BioSciences, Inc. (OTCQB: PBIO) just announced breaking cannabis news earlier this morning that, "a collaboration to advance the development of a new generation of health and wellness nutraceutical products based on processing by PBI's proprietary Ultra Shear Technology (UST™) platform. The Companies believe that nanoemulsions prepared by the UST Platform will have improved quality and effectiveness compared to current emulsions, which will help to facilitate the development of a new generation of improved nutraceutical and other emulsion-based products, such as cosmetics and pharmaceuticals.
https://www.prnewswire.com/news-releases/hemp-s-fda-declassification-opens-door-to-massive-cbd-market-growth-897133713.html
From a post over at Yahoo message board by William:
Consumers Turn to CBD Products Due to their Non-Psychoactive Nature NEW YORK, Feb. 8, 2019 /PRNewswire
Cannabidiol, or CBD-based, products are appearing everywhere throughout the U.S., whether it's for healthcare and wellness, beauty, pets, medicinal, or even just purely recreational use. CBD is known to offer therapeutic effects just like tetrahydrocannabinol, or THC, however, CBD is the non-psychoactive derivative of cannabis. CBD is also legally available within the U.S. as long as it contains 0.3% or less THC, making it very accessible for consumers. Due to its current status under federal law, CBD is expected to be the more explosive market compared to its counterparts. According to data compiled by Brightfield Group, the U.S. hemp CBD market is expected to reach USD 22 Billion by 2022, outpacing the rest of the cannabis market combined. Recently, the U.S. Food and Drug Administration decided to delist CBD as a Schedule 1 drug. Shortly after, the U.S. House passed the Farm Bill, allowing for the cultivation of hemp, which CBD is primarily derived from. The passing of these two legislations positions the industry to be overly attractive for investors and businesses.
Various types of businesses are seen entering into the CBD industry as well. Products are most commonly found in forms of oils, tinctures, and concentrates, but the recent shift in the marketplace has caused new sectors to dip their toes into the market. Products such as beverages, creams and flowers are becoming widely popular whether they are being used for medicinal or recreational purposes. "CBD is the next healthcare phenomenon," said Bethany Gomez, Managing Director at Brightfield Group. "Over the past decade I've covered a wide range of consumer markets, from foods and drinks to telecom, gaming and even cannabis. I have never seen anything like the explosion that we're seeing right now in CBD, even with the bizarre legal gray area where CBD now operates."
If you listen to their investor podcast management buys stock on the open market when they can afford too. Not a lot, but they do buy.
https://www.marketwatch.com/investing/stock/pbio/insideractions
From your chart looks like possible double top short term and overbought but that did not come until the last 1/2 hr of trading.
Strange push EOD move not sure what to make of it so took a little off the table. Maybe this is becoming more of a momentum play after today. Will be interesting to see what trading brings tomorrow.
Are the short given it a rest? Time will tell a visual aid.
The short sale volume percent (not short interest) for stock ticker TGTX is 35% on Feb 06, 2019. The short sale volume is 284,980. The total volume is 808,398. The short sale volume percent is down -31% compare to Feb 05, 2019.
http://shortvolumes.com/?t=tgtx
The stock is consolidating just as it should. If your interested, this is when you should add shares to a position or even start one. GLTY
Combo emerges as bridge to transplant in rel/ref PTCL
Publish date: February 5, 2019
Audio link:
https://www.mdedge.com/hematology-oncology/article/193912/lymphoma-plasma-cell-disorders/combo-emerges-bridge-transplant
Combo emerges as bridge to transplant in rel/ref PTCL
Publish date: February 5, 2019
REPORTING FROM TCLF 2019
LA JOLLA, CALIF. – The combination of duvelisib and romidepsin is active and can provide a bridge to transplant in relapsed or refractory peripheral T-cell lymphoma (PTCL), according to researchers.
In a phase 1 trial, duvelisib plus romidepsin produced an overall response rate (ORR) of 59% in patients with PTCL. Sixteen patients achieved a response, nine had a complete response (CR), and six complete responders went on to transplant.
“So we think that you can achieve remission deep enough to then move on to a potentially curative approach,” said study investigator Neha Mehta-Shah, MD, of Washington University in St. Louis.
She and her colleagues evaluated romidepsin plus duvelisib, as well as bortezomib plus duvelisib, in a phase 1 trial (NCT02783625) of patients with relapsed or refractory PTCL or cutaneous T-cell lymphoma (CTCL).
Dr. Mehta-Shah presented the results at the annual T-cell Lymphoma Forum.
She reported results in 80 patients – 51 with PTCL and 29 with CTCL. The patients’ median age was 64 years (range, 28-83), and 57% of the study population were men. Patients had received a median of 3 (range, 1-16) prior therapies, and 16% had received a prior transplant.
Treatment
Dr. Mehta-Shah noted that patients and providers could choose whether patients would receive romidepsin or bortezomib.
Patients in the romidepsin arm received romidepsin at 10 mg/m2 on days 1, 8, and 15 of each 28-day cycle. Patients in the bortezomib arm received bortezomib at 1 mg/m2 on days 1, 4, 8, and 11 of each cycle.
Duvelisib dosing was escalated, so patients received duvelisib at 25 mg, 50 mg, or 75 mg twice daily.
In the bortezomib arm, there was one dose-limiting toxicity – grade 3 neutropenia – in a patient who received duvelisib at the 25-mg dose. There were no dose-limiting toxicities in the romidepsin arm.
The researchers determined that the maximum tolerated dose (MTD) of duvelisib was 75 mg twice daily in the romidepsin arm and 25 mg twice daily in the bortezomib arm.
Lead-in phase
The study also had a lead-in phase during which patients could receive single-agent duvelisib.
“Because the original phase 1 study of duvelisib did not collect as many prospective tumor biopsies or on-treatment biopsies, we built into this study a lead-in phase so that we could characterize on-treatment biopsies to better understand mechanisms of response or resistance,” Dr. Mehta-Shah said.
Patients and providers could choose to be part of the lead-in phase, she noted. Patients who did not achieve a CR during this phase went on to receive either combination therapy, which was predetermined before the monotherapy began.
There were 14 patients who received duvelisib monotherapy at 75 mg twice daily. Four of them achieved a CR, and three had a partial response (PR). Ten patients went on to receive romidepsin as well. One of them achieved a CR, and three had a PR.
There were 12 patients who received duvelisib monotherapy at 25 mg twice daily. Three of them achieved a CR, and two had a PR. Nine patients went on to receive bortezomib as well. This combination produced one CR and two PRs.
Efficacy with romidepsin
Among all evaluable PTCL patients in the romidepsin arm, the ORR was 59% (16/27), and the CR rate was 33% (9/27).
Responses occurred in seven patients with PTCL not otherwise specified (NOS), six with angioimmunoblastic T-cell lymphoma (AITL), one with hepatosplenic T-cell lymphoma, one with aggressive epidermotropic CD8+ T-cell lymphoma, and one with primary cutaneous PTCL.
RELATED
Duvelisib combos show promise for PTCL, CTCL
CRs occurred in five patients with AITL and four with PTCL-NOS. Six patients who achieved a CR went on to transplant.
Among evaluable CTCL patients in the romidepsin arm, the ORR was 45% (5/11), and there were no CRs. Responses occurred in three patients with mycosis fungoides and two with Sézary syndrome.
The median progression-free survival was 5.41 months in CTCL patients and 6.72 months in PTCL patients.
Efficacy with bortezomib
Among evaluable PTCL patients in the bortezomib arm, the ORR was 44% (7/16), and the CR rate was 25% (4/16).
Responses occurred in three patients with AITL and four with PTCL-NOS. CRs occurred in two patients with each subtype.
Among evaluable CTCL patients in the bortezomib arm, the ORR was 27% (4/15), and there were no CRs. Responses occurred in one patient with mycosis fungoides and three with Sézary syndrome. One CTCL patient went on to transplant.
The median progression-free survival was 4.56 months among CTCL patients and 4.39 months in PTCL patients.
Safety
Dr. Mehta-Shah said both combinations were considered safe and well tolerated. However, there was a grade 5 adverse event (AE) – Stevens-Johnson syndrome – that occurred in the bortezomib arm and was considered possibly related to treatment.
Grade 3/4 AEs observed in the 31 patients treated at the MTD in the romidepsin arm were transaminase increase (n = 7), diarrhea (n = 6), hyponatremia (n = 4), neutrophil count decrease (n = 10), and platelet count decrease (n = 3).
Grade 3/4 AEs observed in the 23 patients treated at the MTD in the bortezomib arm were transaminase increase (n = 2) and neutrophil count decrease (n = 5).
Grade 3/4 transaminitis seemed to be more common among patients who received duvelisib alone during the lead-in phase, Dr. Mehta-Shah said.
Among patients treated at the MTD in the romidepsin arm, grade 3/4 transaminitis occurred in four patients treated during the lead-in phase and three who began receiving romidepsin and duvelisib together. In the bortezomib arm, grade 3/4 transaminitis occurred in two patients treated at the MTD, both of whom received duvelisib alone during the lead-in phase.
Based on these results, Dr. Mehta-Shah and her colleagues are planning to expand the romidepsin arm to an additional 25 patients. By testing the combination in more patients, the researchers hope to better understand the occurrence of transaminitis and assess the durability of response.
This study is supported by Verastem. Dr. Shah reported relationships with Celgene, Kyowa Kirin, Bristol-Myers Squibb, Verastem, and Genentech.
The T-cell Lymphoma Forum is held by Jonathan Wood & Associates, which is owned by the same company as this news organization.
jensmith@mdedge.com
Pressure Biosciences (PBIO) Upgraded by ValuEngine to “Buy”
Posted by Lisa Matthews on Feb 5th, 2019
Pressure Biosciences (OTCMKTS:PBIO) was upgraded by research analysts at ValuEngine from a “hold” rating to a “buy” rating in a note issued to investors on Monday.
Shares of OTCMKTS PBIO traded up $0.37 during trading on Monday, hitting $2.67. The company had a trading volume of 56,278 shares, compared to its average volume of 11,622. Pressure Biosciences has a 12 month low of $1.52 and a 12 month high of $5.00.
Pressure Biosciences (OTCMKTS:PBIO) last posted its quarterly earnings data on Wednesday, November 14th. The company reported ($1.92) EPS for the quarter, missing the consensus estimate of ($0.62) by ($1.30). The firm had revenue of $0.52 million during the quarter, compared to analysts’ expectations of $1.04 million. On average, equities analysts predict that Pressure Biosciences will post -3.29 EPS for the current fiscal year.
Pressure Biosciences Company Profile
Pressure BioSciences, Inc develops pressure cycling technology (PCT) solutions. Its PCT technology uses alternating cycles of hydrostatic pressure between ambient and ultra-high levels to control the actions of molecules in biological samples, such as cells and tissues from human, animal, plant, and microbial sources.
https://www.fairfieldcurrent.com/news/2019/02/05/pressure-biosciences-pbio-upgraded-by-valuengine-to-buy.html
I must of missed this one:
HC Wainwright restated their buy rating on shares of TG Therapeutics (NASDAQ:TGTX) in a research note released on Tuesday, January 22nd.
“Our $20 price target is based on the net present value of our revenue forecast through 2026, applying a 45% probability of success (POS) for ublituximab in CLL, a 45% POS for umbralisib in 25% POS for both ublituximab and umbralisib in NHL. We use a 4x price/sales multiple for these products, an early stage pipeline value of $2.84/share, and fully diluted net cash of $0.75/share to arrive at our price target. Our P/S multiple of 4x is in-line with TG’s peers that range between 2-5x.”,” the firm’s analyst wrote.
In related news, CFO Sean A. Power sold 37,275 shares of the company’s stock in a transaction that occurred on Wednesday, January 2nd. The stock was sold at an average price of $4.13, for a total value of $153,945.75. The transaction was disclosed in a filing with the SEC, which is available at this hyperlink. 15.10% of the stock is currently owned by corporate insiders.
Hedge funds have recently modified their holdings of the stock. Forbes J M & Co. LLP bought a new position in shares of TG Therapeutics during the fourth quarter valued at about $44,000. Nisa Investment Advisors LLC grew its stake in shares of TG Therapeutics by 1,891.9% during the fourth quarter. Nisa Investment Advisors LLC now owns 18,425 shares of the biopharmaceutical company’s stock valued at $76,000 after buying an additional 17,500 shares during the last quarter. Claraphi Advisory Network LLC purchased a new stake in shares of TG Therapeutics during the third quarter worth about $123,000. MetLife Investment Advisors LLC boosted its holdings in shares of TG Therapeutics by 35.2% during the second quarter. MetLife Investment Advisors LLC now owns 28,284 shares of the biopharmaceutical company’s stock worth $372,000 after purchasing an additional 7,358 shares during the period. Finally, Belpointe Asset Management LLC purchased a new stake in shares of TG Therapeutics during the third quarter worth about $243,000. Institutional investors own 57.38% of the company’s stock.
But look who's in.
Largest Quarterly Institutional Transactions
https://money.cnn.com/quote/shareholders/shareholders.html?symb=FNJN&subView=institutional
Lil BD
From what I remember management said all that was under control so i`m going to say zero. The selling likely is long term holders getting out on this price spike when they should be doing the opposite.
Now with the NutraFuels collaboration, $PBIO can prove out its Ultra Shear Technology (UST) platform with CBD oils, which could be hugh. Look to the whole consumables industry, but CBD oils alone, that will put the company on the MJ map first. GLYU
Duvelisib Effective After Progression on Ofatumumab in CLL
https://www.targetedonc.com/news/duvelisib-effective-after-progression-on-ofatumumab-in-cll

Featured Interview: CEO Ric Schumacher of Pressure BioSciences, Inc. 02/04/2019
The CEOs interviewed on the Stock Day Podcast
https://upticknewswire.com/featured-interview-ceo-ric-schumacher-of-pressure-biosciences-inc-otcqb-pbio-11/
Verastem: A Compelling Risk/Reward Profile Feb. 1, 2019 4:50 PM ET
Summary
Small oncology concern Verastem has seen its stock drop by half since its first product was approved by the FDA in September.
However, the company seems significantly undervalued here as the compound Duvelisib gains traction.
We take an in-depth look at Verastem's prospects in the quarters ahead in the paragraphs below.
Looking for more? I update all of my investing ideas and strategies to members of The Biotech Forum. Start your free trial today »
A writer who is afraid to overreach himself is as useless as a general who is afraid to be wrong." - Raymond Chandler, Pearls are a Nuisance
Today, we look at a recently minted 'Tier 3' oncology concern whose stock has cratered over the past six months after the approval of its first compound (where have we heard this story before?). However, the shares now appear beyond cheap, the stock seems to maybe found a bottom and the shares has seen some 'shout outs' from analyst firms recently. Today, we revisit our investment case on this promising small-cap concern.
Company Overview:
Verastem (VSTM) is a small, Boston-based biotech company focused on developing and commercializing medicines to improve the survival of life of cancer patients. On September 24th, the company's primary drug candidate Duvelisib, also known as COPIKTRA, was approved for use in third line chronic lymphocytic leukemia (CLL) and follicular lymphoma (FL) by the FDA. The stock currently trades at just over $3.25 a share and has a market cap of approximately $240 million.
Duvelisib also showed encouraging Phase 1 results in combination with a drug from Celgene (CELG) to treat relapsed/refractory T-cell lymphomas in early December. In early November, Verastem announced a deal with the Leukemia & Lymphoma Society (LLS) aimed at accelerating the development of Duvelisib for peripheral T-cell lymphoma, an aggressive type of non-Hodgkin lymphoma.
Analyst Commentary & Balance Sheet:
Since Duvelisib was approved, a half dozen analyst firms have reissued Buy ratings on the stock. Price targets proffered have ranged from the low to high teens. Two weeks ago, H.C. Wainwright was the latest analyst firm to maintain VSTM as a Buy with a $13 price target. Here is the commentary from that call.
We maintain our Buy rating of VSTM and adjust our 12-month price target to $13.00 per share, down from $15.00. We derive our price target based on the average of two valuation methods: 1) price-sales multiple analysis applying an 8x multiple to our 2025 sales estimate discounted at 12%; and 2) price-earnings multiple analysis applying an 18x multiple to our 2025 estimated earnings discounted at 12%.
The most interesting analyst comments came from BTIG, however, when they reissued their Buy rating and $17 price target the day after Duvelisib was approved.
Verastem's Copiktra (duvelisib), its novel PI3K ?/? inhibitor, received FDA approval for relapsed/refractory CLL/SLL after at least 2 prior therapies, and accelerated approval for relapsed/ refractory follicular lymphoma (FL) after at least 2 prior therapy (a confirmatory trial in FL is required). We continue to estimate US peak revenue >$500mm, and $>350mm internationally in these initial indications."
In the approved 3rd line CLL/SLL population, mPFS for Copiktra was 16.4 mo. vs 9.1 mo. for control, compared to 13.3 and 9.9 mo., respectively, for the overall study. Verastem set a list price of $11,800/mo., generally in-line with our estimate. We continue to model peak US penetration of Copiktra in CLL/SLL of ~1,900 patients, or ~42% of the ~4,500 patients treated in 3rd line (in 2016), which we feel adequately reflects the potential of its label."
While that analysis is somewhat technical, it means BTIG thinks eventually 1,900 patients will be treated annually at $11,800 per month, each patient projected to be on treatment for approximately 16 months. If one models 1,000 patients for 12 months as a more conservative estimate, that gets you to just under $150 million in annual revenue.
Verastem ended the third quarter with approximately $145 million in cash and marketable securities on hand. It then raised another $145 via a direct offering of convertible notes in October, so its funding needs for the foreseeable have been completely addressed.
Verdict:
Duvelisib got off to a nice start with just over $500,000 in revenues during the last week of September. We should get a better picture of the compound's ramp-up when Verastem reports Q4 results in a week or so as well. In September, the company signed a deal with CSPC Pharmaceutical Group Limited for the development and commercialization of COPIKTRA for all oncology indications in China. Verastem received $15 million in an upfront payment and is eligible for tiered royalties and milestone payouts from this deal.
If Duvelisib just does a fraction of BTIG's peak sales estimates, the stock seems woefully undervalued, given that cash on hand and strong analyst support. Several insiders also made small purchases in the shares in late September when the stock traded just over $7.00 a share.
The stock looks like it might have formed a bottom over the past five to six weeks as well. Given all of this, the risk/reward profile of Verastem looks very favorable at current trading levels.
I satirize at all times, and my hyperboles are as nothing compared to the events to which they refer." ? Marshall McLuhan
Bret Jensen is the Founder and author of articles on The Biotech Forum, The Busted IPO Forum, and The Insiders Forum. To receive these articles as published on Seeking Alpha, just click the appropriate link and hit the orange follow button.
The biotech sector has gotten off to a solid start in 2019 after deep losses in the fourth quarter. M&A has picked up as two major acquisitions have happened early this year. We just provided our 2019 Outlook to Biotech Forum members. It contains what we expect in this high beta sector for the rest of the year as well as a couple of possible logical takeout candidates. To get it, access to our model portfolio and our investment analysis archives, sign up for your free 14 day trial into The Biotech Forum by clicking here. Hope to see you on Live Chat soon!
Disclosure: I am/we are long VSTM. I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.
Editor's Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.
https://seekingalpha.com/article/4237589-verastem-compelling-risk-reward-profile
As of November 2nd 2018 CSPC Pharmaceutical Group
Investment thesis
NBP plant visit: The Shijiazhuang NBP plant is at full capacity now for ~HK$5bn in NBP sales (vs.our projected NBP 2018E total revenue of HK$5.1bn). This implies NBP needs new capacity to support future growth. According to the plant manager, the new NBP plant will commence production in 1H2019 and will double NBP capacity to ~HK$10bn in value, i.e. fulfilling next three years’ projected production requirements. Furthermore, NBP shipments and channel inventory remain
healthy, so we expect NBP to continue its solid growth momentum in Q3 2018. Overall, we expect NBP to deliver ~43% YoY top-line growth in 2018E (+42.6% YoY in 1H2018).
Visit to CSPC’s Central Institute of Pharmaceutical Research: We visited CSPC’s Central Institute of Pharmaceutical Research in Shijiazhuang. Following are the key takeaways:
? Innovative - COPIKTRA (licensed from Verastem for leukemia) is under final human race trial.
Hemp's FDA Declassification Opens Door to Massive CBD Market Growth
9:00 am ET January 31, 2019 (PR Newswire) Print
FinancialBuzz.com News Commentary
Cannabis is a family of plants that splits into two categories, marijuana and hemp. However, marijuana contains the psychoactive components of the plant while hemp has more practical uses such as clothing. Due to the effects of marijuana, it is currently more regulated as opposed to hemp. Hemp, being the non-psychoactive derivative of cannabis currently enjoys significantly less Federal regulations around the country, and, due to the current status of hemp, the industry is positioned to see significant growth. According to data compiled by Hemp Business Journal, a division of New Frontier Data, the U.S. hemp industry registered sales of USD 820 Million in 2017 and is expected to reach USD 1.9 Billion by 2022. Additionally, the market is projected to grow at a CAGR of 14.4% during the forecast period. Pressure BioSciences, Inc. (OTC: PBIO), GW Pharmaceuticals plc (NASDAQ: GWPH), Cronos Group Inc. (NASDAQ: CRON), Organigram Holdings Inc. (OTC: OGRMF), Emerald Health Therapeutics, Inc. (OTC: EMHTF)
Within the hemp industry, CBD products held the largest market value in 2017, delivering USD 190 Million in sales. CBD was also recently delisted from its status of a Schedule 1 drug by the U.S. Food and Drug Administration, prompting a robust growth. By 2022, the segment is expected to drive in sales of USD 646 Million and continue to dominate the overall U.S. hemp industry. Hemp-derived products include tinctures, topicals and capsules, which are all being used for both medical and recreational applications due to their relaxing and therapeutic effects. "I have never seen anything like the explosion that we're seeing right now in CBD, even with the bizarre legal gray area where CBD now operates. A few years ago almost no one knew what hemp-derived CBD was," said Bethany Gomez, Managing Director at Brightfield Group. "All of a sudden, CBD is everywhere - it is both a trendy new ingredient in drinks, face creams and pet treats and an answer to the prayers of so many people suffering from medical conditions ranging from epilepsy to anxiety and chronic pain."
Pressure BioSciences, Inc. (OTCQB: PBIO) just announced breaking cannabis news earlier this morning that, "a collaboration to advance the development of a new generation of health and wellness nutraceutical products based on processing by PBI's proprietary Ultra Shear Technology (UST(TM)) platform. The Companies believe that nanoemulsions prepared by the UST Platform will have improved quality and effectiveness compared to current emulsions, which will help to facilitate the development of a new generation of improved nutraceutical and other emulsion-based products, such as cosmetics and pharmaceuticals.
PBI is a leader in the development and sale of enabling high pressure-based instruments, consumables, and related services for the life sciences industry. PBI has more than 300 high-pressure instruments installed in over 200 life sciences laboratories worldwide, including in some of the world's leading academic, government, and biopharmaceutical laboratories.
NTFU manufactures and distributes nutritional and dietary supplements focusing on in-house product development and the highest manufacturing standards. All quality control testing and manufacturing processes are in compliance with FDA and cGMP standards. NTFU has developed multiple products for the wellness and nutraceutical markets, including formulas to support energy and focus, sleep, stress, joints, and weight loss.
Edgar J. Ward, President and CEO of NTFU, said, 'We pride ourselves in ensuring that we incorporate the highest level of quality possible in our manufactured products. When we heard that PBI was developing their new, proprietary UST processing platform, and learned of its potential to significantly increase the quality and effectiveness of nutraceutical products, we spoke with PBI and offered to help accelerate its commercial introduction. We are thrilled to be working with such experienced scientific leaders and innovators, in a program that we believe can change lives worldwide for the better.'
Mr. Ward continued, 'We believe PBI's UST platform has the potential to create long-term room temperature stable, water-soluble nanoemulsions of oil-based solutions. Nanoemulsions are known to offer greater stability and bioavailability than the standard macroemulsions used today in nutraceuticals, cosmetics, and other industries. We are excited to have the opportunity to work with a life science industry leader in the optimization of a process that has the potential to bring higher quality not just to our products, but to nutraceutical products worldwide.'
Dr. Bradford A. Young, Chief Commercial Officer of PBI, commented, 'We are pleased to have the opportunity to work with NTFU's scientists and manufacturing personnel in the development of new and improved nutraceutical products utilizing our UST platform. This proprietary technology employs ultra-high pressure and extreme shearing forces to create nano-scale emulsions of oil and water with long-term stability. For many oil-based products, the ability to create very small, nanometer-sized oil droplets that can effectively dissolve in water (nanoemulsions) can improve a product's appearance, sensory and medicinal benefits. There is a large and growing market opportunity for nutraceutical products with proven health and wellness benefits. We believe PBI's UST platform can help manufacturers accelerate growth and success in this market with higher quality, water-soluble, oil-based products with superior dietary absorption and shelf-life.'
Mr. Richard T. Schumacher, President and CEO of PBI, added, 'We are excited to work with Edgar and his NTFU team in the optimization of our UST platform, which we believe will result in the development of new and beneficial health and wellness products. The staff at NTFU has years of experience in manufacturing nutraceutical products in a quality environment. They also have access to both raw materials and finished goods, both of which are needed for optimization. Finally, they have an existing analytical laboratory with state-of-the-art equipment and well-trained chemists who can perform testing on both pre and post-processed materials, which will be invaluable to the optimization process. This collaboration clearly supports both company's strategic objectives: we look forward to an exciting and mutually beneficial relationship with our colleagues at NutraFuels.'
About Pressure BioSciences, Inc. - Pressure BioSciences, Inc. (PBIO) is a leader in the development and sale of innovative, broadly enabling, pressure-based solutions for the worldwide life sciences industry. Our products are based on the unique properties of both constant (i.e., static) and alternating (i.e., pressure cycling technology, or PCT) hydrostatic pressure. PCT is a patented enabling technology platform that uses alternating cycles of hydrostatic pressure between ambient and ultra-high levels to safely and reproducibly control bio-molecular interactions (e.g., cell lysis, biomolecule extraction). Our primary focus is in the development of high pressure-based products for biomarker and target discovery, drug design and development, biotherapeutics characterization and quality control, food science, soil & plant biology, forensics, and counter-bioterror applications. Additionally, we are actively expanding the use of our pressure-based technologies in the following areas: (1) the use of our recently acquired protein disaggregation and refolding technology from BaroFold, Inc. to allow entry into the biologics manufacturing and contract research services sector, and (2) the use of our recently-patented, scalable, high-efficiency, pressure-based Ultra Shear Technology (UST) platform to (i) create stable nanoemulsions of otherwise immiscible fluids (e.g., oils and water) and to (ii) prepare higher quality, homogenized, extended shelf-life or room temperature stable low-acid liquid foods that cannot be effectively preserved using existing non-thermal technologies.
About NutraFuels, Inc. - NutraFuels, Inc was founded in 2010 and has evolved into a branded and private label developer, distributor and Food and Drug Administration registered (FDA) manufacturer. NTFU's products include a range of nutraceutical, wellness, and CBD products as well as a cosmetics line. NutraFuels' manufacturing process received the Good Manufacturing Processes Standard (GMP) certification. Its products adhere to high manufacturing standards throughout every step of the manufacturing and extraction process. NTFU's product testing and research and development is conducted by four chemists under the direction of NTFU's founder and Chief Executive Officer, Edgar Ward."
GW Pharmaceuticals plc (NASDAQ: GWPH), founded in 1998, is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. GW Pharmaceuticals plc recently announced positive top-line results of the second randomized, double-blind, placebo-controlled Phase 3 clinical trial of EPIDIOLEX(R) (cannabidiol or CBD) CV in the treatment of seizures associated with Dravet syndrome, a rare and severe form of childhood-onset epilepsy. In this trial, EPIDIOLEX, when added to the patient's current treatment, achieved the primary endpoint of reduction in convulsive seizures for both dose levels (10 mg/kg per day and 20 mg/kg per day) with high statistical significance compared to placebo. Both EPIDIOLEX doses also demonstrated statistically significant improvements on all key secondary endpoints. "The positive results from this trial follow the recent FDA approval, DEA rescheduling and U.S. launch of EPIDIOLEX for the treatment of seizures associated with Dravet syndrome and Lennox-Gastaut Syndrome in patients two years and older. These data show an effective dose range in Dravet syndrome that is consistent with our FDA approved label, and which allows for dosing flexibility to address individual patient needs," stated Justin Gover, GW's Chief Executive Officer. "We are proud to have recently launched EPIDIOLEX, the first FDA-approved plant-derived cannabinoid medicine and are excited about its potential to help the lives of patients and their families."
Cronos Group Inc. (NASDAQ: CRON) is a globally diversified and vertically integrated cannabis company with a presence across five continents. Cronos Group Inc. recently announced that it has entered into a sponsored research agreement with the Technion Research and Development Foundation of the Technion - Israel Institute of Technology ("Technion") to explore the use of cannabinoids and their role in regulating skin health and skin disorders. The preclinical studies will be conducted by Technion over a three-year period and will focus on three skin conditions: acne, psoriasis and skin repair. "We believe that the potential applications of cannabinoids to regulate skin health and treat skin disorders are vast, and we are excited to begin exploring these applications through our partnership with Technion," said Mike Gorenstein, Chief Executive Officer of Cronos Group. "Using rigorous data to develop efficacious topical and transdermal formulations will be key to creating differentiated products that provide quality treatments to our consumers and strengthen our brand portfolio."
Organigram Holdings Inc. (OTCQX: OGRMF) is a TSX Venture Exchange listed company whose wholly owned subsidiary, Organigram Inc., is a licensed producer of cannabis and cannabis-derived products in Canada. Organigram Holdings Inc. announced the Company has entered into an agreement with 1812 Hemp, a New Brunswick based industrial hemp research company to secure supply and support research and development on the genetic improvement of hemp through traditional plant breeding methods. 1812 Hemp is focused on further developing a line of Canadian cultivars (specific varieties of plants cultivated to enhance desirable qualities) of high cannabidiol (CBD) yielding hemp for the Canadian climate. "Maximizing the CBD yield of hemp and cannabis means being able to meet increasing consumer demand for CBD while reducing overall production costs," explains Greg Engel, Chief Executive Officer Organigram. "We are proud to work with other industry-leading companies and researchers to continue to explore the potential of hemp and cannabis plants and the efficiency with which we can deliver CBD to our customers."
Emerald Health Therapeutics, Inc. (OTCQX: EMHTF) is a Licensed Producer under Canada's Access to Cannabis for Medical Purposes Regulations and produces and sells dried cannabis and cannabis oil for medical purposes. Recently, Emerald Health Therapeutics, Inc. agreed to purchase from Emerald Health Hemp Inc. (EHH) CBD-containing hemp biomass for extraction into CBD oil. EHH has established agreements to purchase approximately 500 acres of harvested hemp chaff, plant material consisting mainly of flower and leaf, in Manitoba and Prince Edward Island in 2018, with a plan to expand field operations in 2019. Emerald has signed an agreement with EHH to purchase approximately 500 acres of chaff in 2018 and approximately 1,000 acres in 2019 to 2022, with an option to renew for an additional two-year period. The hemp chaff will be processed via Emerald's exclusive alliance with Factors R&D Technologies Ltd., a division of Factors Group of Nutritional Companies Inc., which has extensive extraction experience and large capacity. "We see non-smoked, non-psychoactive CBD consumption as a major growth opportunity in both the medical and adult-use market in Canada, and our Factors Group alliance uniquely positions us with the largest biomass extraction and softgel encapsulation facility in Canada to produce value-added products to serve this market," said Chris Wagner, Chief Executive Officer of Emerald. "Our intent is to secure significant low-cost cannabinoid supply to serve all our manufacturing and marketing goals and hemp is expected to be our lowest cost source of CBD. We are very pleased with the rapid advancement of our Pure Sunfarms production joint venture with Village Farms and our St. Eustache facility in Quebec. Our collaboration with Emerald Health Hemp now expands our ability to produce CBD products."
https://www.prnewswire.com/news-releases/hemp-s-fda-declassification-opens-door-to-massive-cbd-market-growth-897133713.html
Lets see how fast this can mature into the CBD oils which I believe is their main goal or should be top priority near term.
This event is intended for genuine institutional and retail investors.
Wed, January 30, 2019
https://www.eventbrite.co.uk/e/invest-in-cannabis-tickets-50979096851
Tonight Jim Cramers pot prediction.
https://www.cnbc.com/video/2019/01/28/cramers-pot-predictions-for-2019-legalization-deals-brands.html