Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Updated PPHM REVS-BY-QTR TABLE, now thru FY16'Q3(qe1-31-16). Cust.Deposits=$22.4mm & Deferred.Revs=$15.4mm bode well for FY16/Q4 (q/e 4-30-16), with FY'16 Guidance raised to “over $40mm” and 1st 3 qtrs now at Revs=$25.6mm.
• Total Revs since May’06: ($154.8mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $181.4mm
• 3-9-16: FY'16 (May'15-Apr'16) Avid revs guidance raised from $35-40mm to “over $40mm”.
• Deferred-Revs at 1-31-16 total $15.4mm, UP from $9.7mm at 10-31-15.
• Cust.Deposits at 1-31-16 total $22.4mm, UP from $14.9mm at 10-31-15.
• Inventories at 1-31-16 total $15.2mm, UP from $12.6mm at 10-31-15.
• Avid’s Gross-Profit over last 3 qtrs: $12.3mm on revs of $25.6mm (GP%=48%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
FY16Q2 10-31-15 9523 0 0 9523 9688 0 12554
FY16Q3 1-31-16 6672 0 37 6709 15418 0 15189
Totals: 154786 24149 2453 181388 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
FY16Q2 10-31-15 13,198,000
FY16Q3 1-31-16 16,847,000
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
2-26-16/Dr.Raymond.Birge article: “Phosphatidylserine is a Global Immunosuppressive Signal...” - Cell Death & Differentiation, online pub. 2-26-16.
2-26-16: “Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer”
'Cell Death & Differentiation Jrnl' online pub. 2-26-16 (rec.10-31-15; acc.1-7-16)
Raymond Birge 1; S Boeltz 2; S Kumar 1; J Carlson 3; J Wanderley 4; D Calianese 1; M Barcinski5; Rolf Brekken 6,7; Xianming Huang 6,7; Jeff Hutchins 3; Bruce Freimark 3; C Empig 3; J Mercer 8; A J Schroit 9; G Schett 2; Martin Herrmann 2
1. Dept of Microbiology, Biochemistry & Molecular Genetics, CC, Rutgers NJ Medical School, Newark, NJ
2. Dept of Internal Medicine, Rheumatology & Immunology, Friedrich-Alexander-Univ., Erlangen, Germany
3. Peregrine Pharma., Tustin, CA
4. Universidade Federal do Rio de Janeiro, Brazil
5. Lab de Biologia Celular, Instituto Oswaldo Cruz, Rio de Janeiro, Brazil
6. Div. of Surgical Oncology, Hamon Ctr for Therapeutic Oncology Res., UTSW-MC/Dallas
7. Dept of Pharmacology, UTSW-MC/Dallas
8. Medical Research Council Lab for Molecular Cell Biology, Univ. College London
9. Simmons Cancer Center & Dept of Immunology, UTSW-MC/Dallas
ABSTRACT
Apoptosis is an evolutionarily conserved and tightly regulated cell death modality. It serves important roles in physiology by sculpting complex tissues during embryogenesis and by removing effete cells that have reached advanced age or whose genomes have been irreparably damaged. Apoptosis culminates in the rapid and decisive removal of cell corpses by efferocytosis, a term used to distinguish the engulfment of apoptotic cells from other phagocytic processes. Over the past decades, the molecular and cell biological events associated with efferocytosis have been rigorously studied, and many eat-me signals and receptors have been identified. The externalization of phosphatidylserine (PS) is arguably the most emblematic eat-me signal that is in turn bound by a large number of serum proteins and opsonins that facilitate efferocytosis. Under physiological conditions, externalized PS functions as a dominant and evolutionarily conserved immunosuppressive signal that promotes tolerance and prevents local and systemic immune activation. Pathologically, the innate immunosuppressive effect of externalized PS has been hijacked by numerous viruses, microorganisms, and parasites to facilitate infection, and in many cases, establish infection latency. PS is also profoundly dysregulated in the tumor microenvironment and antagonizes the development of tumor immunity. In this review, we discuss the biology of PS with respect to its role as a global immunosuppressive signal and how PS is exploited to drive diverse pathological processes such as infection and cancer. Finally, we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.
FULL ARTICLE: http://www.nature.com/cdd/journal/vaop/ncurrent/full/cdd201611a.html

= = = = = = = = = =UPCOMING:
June2-4: ICDS 2016: Translational Implications in Cell Death", Cork Ireland http://tinyurl.com/h2gvyld
...Dr. Rolf Brekken (USA, UTSE, PPHM SAB): ”Blockade of PS & Immune Activation in Cancer” (Chair: Dr. Raymond Birge/Rutgers – see http://tinyurl.com/j3whbx3 )
Jun19-22: “ICDS'16 - From Molecules To Diseases”, Istanbul Turkey http://tinyurl.com/h4zjvo4
….Rutgers' Dr. Raymond Birge ( http://birgelab.org ) ”Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer” - see http://tinyurl.com/j3whbx3
= = = = = = = = = = = = =PREV:
June2015: DR RAYMOND BIRGE & Dr. Xianming Huang (Dr.Rolf.Brekken-Lab) at Gordon Res. Conference in Maine on 6-15-15 – speaking next to each other in “Consequences of Lipid Alterations” Session. Peregrine was the only corp. contributor to this conference, specified as, “In Memory of Dr. Philip E. Thorpe.”...
June 14-19, 2015: “Apoptotic Cell Recognition & Clearance”, Gordon Research Conference, Biddeford, ME
Chair: David S. Ucker, Vice Chairs: Peter Henson & Kirsten Lauber
http://www.grc.org/programs.aspx?id=13127
”Moreover, apoptotic cells are potently immunosuppressive, and their clearance occurs in the absence of inflammation. Recognition & inflammatory modulation represent key elements of an innate immunity that discriminates live from effete cells.”
**The only Corp. Sponsor: PEREGRINE PHARM, “Generous financial support from Peregrine Pharmaceuticals is in memory of Dr. Philip E. Thorpe.”
- - - - - - - -
SESSION: “Consequences of Lipid Alterations for Apoptotic Cell Clearance
Discussion Leader: Christopher Gregory (Univ. of Edinburgh, UK)
7:30pm: Ian Dransfield (Univ. of Edinburgh, UK), “Protein S Binding to Apoptotic Cells...”
8:10pm: Dr. Raymond Birge (Rutgers NJMS), “Apoptotic Cell Recognition Receptors, Tyro3/Axl/Mer...”
8:50pm: Dr. Xianming Huang** (UTSW-MC/Dallas, Brekken Lab), “PS-Targeting Antibodies Overcome Tumor Immunosuppression and Synergize with Immune Checkpoint Blockade"
**Dr. Xianming Huang: formerly in Dr. Philip Thorpe's Lab (RIP, Dr. Thorpe, 1951-2013 http://tinyurl.com/l9gqmt5 ), now in Dr. Rolf Brekken's (PPHM SAB) Lab.
http://www.utsouthwestern.edu/labs/brekken/
= = = = = = = = = = = = = =DR. BIRGE:
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html
====================DR. RAY BIRGE (RUTGERS) & PEREGRINE:
June19-22 2016: “15th Intl. Congress of Histochemistry & Cytochemistry - from Molecules to Diseases”, Istanbul Turkey
“The main goal of ICHC 2016 is to bring the worldwide histochemists together and provide an environment for close cooperation, exchange of information, and collaborations.”
http://www.ichc2016.com
INVITED SPEAKER – Session: “Cellular Aging & Cell Death”
Dr. Raymond Birge (Rutgers Univ. http://birgelab.org ), “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”
Nov4-8 2015: “(SITC) Society for Immunotherapy of Cancer 30th Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2015 Meeting: http://www.sitcancer.org/2015
EXHIBITOR: Peregrine Pharm. - booth #121 (directly across from #120/ASCO & BMY/124; AstraZeneca/108 & DNA/113 are close by)
...Floorplan: http://www.eventscribe.com/2015/sitc/exhibitors/index.asp
...Join Peregrine for a Scientific Session - FRI Nov6 2015 7:30-9:00pm
“The PS Signaling Pathway: A Promising
Therapeutic Target Exploited by
Tumors for Immune Evasion”
PGM:
* Raymond Birge (PhD, Rutgers) http://birgelab.org
* Douglas Graham (MD, PhD, Emory) http://choa.org/Childrens-Hospital-Services/Cancer-and-Blood-Disorders/Meet-the-Team/Physicians-and-Researchers/Douglas-Graham
* Dmitry Gabrilovich (MD, PhD, Wistar Inst.) http://www.wistar.org/our-science/scientists/dmitry-gabrilovich-md-phd
* Rolf Brekken (PhD, UTSW/Dallas) http://www.utsouthwestern.edu/labs/brekken
* Maria Karasarides (PhD, AstraZeneca - Sr. Director, ImmunoOncology, Global Medicines Dev.) http://www.linkedin.com/pub/maria-karasarides-ph-d/6/769/136
...Brochure handout (not sure if exactly it): http://www.personalizedmedonc.com/article/phosphatidylserine-an-immune-modulating-checkpoint-ushers-in-the-next-wave-of-immuno-oncology-targets/
- - - - - - - -
SITC'15 Track: “Optimizing Combination Immunotherapy”
11-7-15/Sat./12:45-2:00pm
PPHM#1: Poster P357, “Targeting of Phosphatidylserine by Monoclonal Antibodies Augments the Activity of Paclitaxel & anti-pd1/pd-L1 Therapy in the Murine Breast Model E0771”
Presenting Author: Michael Gray, PhD (Sr.Scientist, Peregrine Pharmaceuticals)
Michael Gray 1, Jian Gong 1, Van Nguyen 1, Takuya Osada 2, Zachary Hartman 2, Jeff Hutchins 1, Bruce Freimark 1, Kim (Herbert K.) Lyerly, Duke Univ.** 2
...1=Peregrine Pharmaceuticals, Tustin CA
...2=Duke University, Durham, NC
**Dr. Herbert Kim Lyerly: http://surgery.duke.edu/faculty/details/0117267 (Professor of Surgery, Assistant Professor in Immunology, Associate Professor of Pathology, Duke Univ. MC)
The other 2 Duke co-author scientists:
* Dr. Takuya Osada (MD/PhD – Duke Cancer Inst. - Hematology, Oncology, Clinical Immunology http://www.researchgate.net/profile/Takuya_Osada2 )
* Dr. Zachary Hartman (PhD) – Duke MC, Section of Applied Therapeutics http://surgery.duke.edu/faculty/details/0281172 ”My group is also involved in strategies to modulate the immune response to tumors, which involves the use of novel immunotherapeutic strategies & dev. of vaccines to specific oncogenic targets.”
Oct29 2015: “John-Hopkins Molecular Microbiology & Immunology Seminar”, Baltimore http://tinyurl.com/oks5uo6
...12-1:00pm: "Phosphatidylserine is a Global Immune Checkpoint In Cancer"
Raymond Birge, PhD, Vice Chair, Professor, Dept of Molecular, Biochemistry & Molecular Genetics, Rutgers Univ., Newark, NJ

= = = = = = = = = = = = = = = = = = = = = = = = = =
DR. BIRGE, CLEARLY IN STEP WITH DR. THORPE, DR. BREKKEN, AND PEREGRINE PHARMACEUTICALS...
5-1-12/NYAS: Dr. Philip Thorpe (UTSW, RIP/1951-2013) on Bavi MOA: http://tinyurl.com/9792gl5
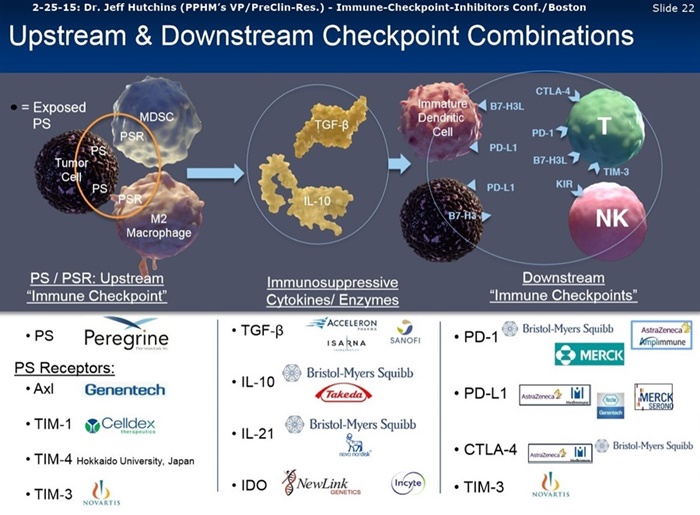
“As a 2nd mechanism, bavituximab can overcome the immunosuppressive processes observed with cells that externalize PS. In tumors the expression of PS on cell surfaces keeps macrophages in their M2 state, an activation state that suppresses inflammation and supports angiogenesis. It also prevents myeloid-derived suppressor cells (MDSCs) from differentiating into macrophages and dendritic cells (DCs). In addition, PS inhibits the maturation of DCs, which prevents the presentation of antigens to T cells. Bavituximab repolarizes macrophages to their M1 state, which allows them to kill cells that express PS, including the tumor vascular endothelium. It also leads to differentiation of the MDSCs and prompts DCs to mature and to present tumor antigens to T cells.”
5-28-14/NYAS: Dr. Rolf Brekken (UTSW) http://tinyurl.com/lq9stnk
“Phosphatidylserine (PS) is a potent immunosuppressive lipid typically segregated to the inner leaflet of the plasma membrane. PS is externalized on tumor vasculature, tumor-derived exosomes, and tumor cells in the tumor microenvironment and externalization is enhanced by therapy. Externalized PS interacts with immune cells where it actively promotes expansion of myeloid derived suppressor cells (MDSCs) and M2-like tumor associated macrophages (TAMs), which drive immunosuppression and tumor progression. In preclinical studies, treatment of tumor-bearing mice with murine-versions of bavituximab significantly depleted M2-likeTAMs & MDSCs and increased the presence of M1-like TAMs and mature dendritic cells. In addition, PS blockade shifted the cytokine balance in the tumor microenvironment from immunosuppressive to immunostimulatory. Furthermore, in the immune-competent tumor models combination of standard of care therapy with PS blockade induced potent durable tumor-specific T-cell immunity and significantly improved tumor free long-term survival. These data suggest that externalized PS defines a global immune checkpoint in tumors and support that antibody-mediated PS blockade can reverse PS-mediated immune checkpoint suppression, revitalize innate and the adaptive immunity, and promote therapeutically effective anti-tumor immunity.”
1-16-16/PR: About Bavituximab: A Targeted Investigational Immunotherapy”
“Bavituximab is an investigational immunotherapy designed to assist the body's immune system by targeting and modulating the activity of phosphatidylserine (PS), a highly immune-suppressive signaling molecule expressed broadly on the surface of cells in the tumor microenvironment. Peregrine's PS targeted inhibitor, bavituximab, is thought to reverse the immunosuppressive environment that many tumors establish in order to proliferate and spread, while also fighting cancer by activating immune cells that target and fight cancer.”
“Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab blocks PS and, in turn, is believed to remove this immunosuppressive signal and send an alternate immune activating signal. PS targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in robust anti-tumor immune responses.”
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=949704
3-1-2014/Youtube: “Cancer Research” - Dr. Raymond Birge Laboratory, Rutgers http://birgelab.org
http://www.youtube.com/watch?v=HdacTc_I7Js
Dr. Ray Birge/2:54: "The immune system is very complex and if it can recognize tumors as being foreign to the body, then it can reactivate & eliminate these tumors to treat as a chronic disease, or in some cases eliminate the tumors. One of the projects that our lab is interested in is how to reactivate the immune system toward particular cancers, and we believe that the type of cell death that cancer cells undergo will determine whether or not they'll be an immune reaction against the cancer. If cells die, through a process called apoptosis, in a certain way, they can reactivate / activate the immune system and give a durable, anti-tumor response. The problem that we're trying to understand is how the tumor cells evade the immune system and therefore block these immune-suppressing signals to reactivate the immune system against cancer..."
S.King/2-25-16/CC, “Yes, we've never seen these #'s reported...”
2-25-16: IDMC Halts SUNRISE at 1st Look-in. Bavi+Doce arm “OS performing as expected”; Doce arm “dramatically outperforming OS expectations” http://tinyurl.com/jbg48vs
http://tinyurl.com/jbg48vs
[2-25-16 CC Replay(16mins): http://edge.media-server.com/m/p/xfa9fy5v ]
EXCERPTS:
11:10/Q&A: RahulJasuja/Noble, “You said that the performance of the Doce/Ctl arm was higher than ever seen in such a trial?” - SK “Yes, we've never seen these #'s reported. It's quite perplexing… at this point, the performance of the Doce arm was just completely out in left field.”
2-26-16/Dr.Ray.Birge article: “Phosphatidylserine is a Global Immunosuppressive Signal...” - Cell Death & Differentiation, online pub. 2-26-16.
2-26-16: “Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer”
'Cell Death & Differentiation Jrnl' online pub. 2-26-16 (rec.10-31-15; acc.1-7-16)
Raymond Birge 1; S Boeltz 2; S Kumar 1; J Carlson 3; J Wanderley 4; D Calianese 1; M Barcinski5; Rolf Brekken 6,7; Xianming Huang 6,7; Jeff Hutchins 3; Bruce Freimark 3; C Empig 3; J Mercer 8; A J Schroit 9; G Schett 2; Martin Herrmann 2
1. Dept of Microbiology, Biochemistry & Molecular Genetics, CC, Rutgers NJ Medical School, Newark, NJ
2. Dept of Internal Medicine, Rheumatology & Immunology, Friedrich-Alexander-Univ., Erlangen, Germany
3. Peregrine Pharma., Tustin, CA
4. Universidade Federal do Rio de Janeiro, Brazil
5. Lab de Biologia Celular, Instituto Oswaldo Cruz, Rio de Janeiro, Brazil
6. Div. of Surgical Oncology, Hamon Ctr for Therapeutic Oncology Res., UTSW-MC/Dallas
7. Dept of Pharmacology, UTSW-MC/Dallas
8. Medical Research Council Lab for Molecular Cell Biology, Univ. College London
9. Simmons Cancer Center & Dept of Immunology, UTSW-MC/Dallas
ABSTRACT
Apoptosis is an evolutionarily conserved and tightly regulated cell death modality. It serves important roles in physiology by sculpting complex tissues during embryogenesis and by removing effete cells that have reached advanced age or whose genomes have been irreparably damaged. Apoptosis culminates in the rapid and decisive removal of cell corpses by efferocytosis, a term used to distinguish the engulfment of apoptotic cells from other phagocytic processes. Over the past decades, the molecular and cell biological events associated with efferocytosis have been rigorously studied, and many eat-me signals and receptors have been identified. The externalization of phosphatidylserine (PS) is arguably the most emblematic eat-me signal that is in turn bound by a large number of serum proteins and opsonins that facilitate efferocytosis. Under physiological conditions, externalized PS functions as a dominant and evolutionarily conserved immunosuppressive signal that promotes tolerance and prevents local and systemic immune activation. Pathologically, the innate immunosuppressive effect of externalized PS has been hijacked by numerous viruses, microorganisms, and parasites to facilitate infection, and in many cases, establish infection latency. PS is also profoundly dysregulated in the tumor microenvironment and antagonizes the development of tumor immunity. In this review, we discuss the biology of PS with respect to its role as a global immunosuppressive signal and how PS is exploited to drive diverse pathological processes such as infection and cancer. Finally, we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.
FULL ARTICLE: http://www.nature.com/cdd/journal/vaop/ncurrent/full/cdd201611a.html
= = = = = = = = = =UPCOMING:
June2-4: ICDS 2016: Translational Implications in Cell Death", Cork Ireland http://tinyurl.com/h2gvyld
...Dr. Rolf Brekken (USA, UTSE, PPHM SAB): ”Blockade of PS & Immune Activation in Cancer” (Chair: Dr. Raymond Birge/Rutgers – see http://tinyurl.com/j3whbx3 )
Jun19-22: “ICDS'16 - From Molecules To Diseases”, Istanbul Turkey http://tinyurl.com/h4zjvo4
….Rutgers' Dr. Raymond Birge ( http://birgelab.org ) ”Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer” - see http://tinyurl.com/j3whbx3
= = = = = = = = = = = = =PREV:
June2015: DR RAYMOND BIRGE & Dr. Xianming Huang (Dr.Rolf.Brekken-Lab) at Gordon Res. Conference in Maine on 6-15-15 – speaking next to each other in “Consequences of Lipid Alterations” Session. Peregrine was the only corp. contributor to this conference, specified as, “In Memory of Dr. Philip E. Thorpe.”...
June 14-19, 2015: “Apoptotic Cell Recognition & Clearance”, Gordon Research Conference, Biddeford, ME
Chair: David S. Ucker, Vice Chairs: Peter Henson & Kirsten Lauber
http://www.grc.org/programs.aspx?id=13127
”Moreover, apoptotic cells are potently immunosuppressive, and their clearance occurs in the absence of inflammation. Recognition & inflammatory modulation represent key elements of an innate immunity that discriminates live from effete cells.”
**The only Corp. Sponsor: PEREGRINE PHARM, “Generous financial support from Peregrine Pharmaceuticals is in memory of Dr. Philip E. Thorpe.”
- - - - - - - -
SESSION: “Consequences of Lipid Alterations for Apoptotic Cell Clearance
Discussion Leader: Christopher Gregory (Univ. of Edinburgh, UK)
7:30pm: Ian Dransfield (Univ. of Edinburgh, UK), “Protein S Binding to Apoptotic Cells...”
8:10pm: Dr. Raymond Birge (Rutgers NJMS), “Apoptotic Cell Recognition Receptors, Tyro3/Axl/Mer...”
8:50pm: Dr. Xianming Huang** (UTSW-MC/Dallas, Brekken Lab), “PS-Targeting Antibodies Overcome Tumor Immunosuppression and Synergize with Immune Checkpoint Blockade"
**Dr. Xianming Huang: formerly in Dr. Philip Thorpe's Lab (RIP, Dr. Thorpe, 1951-2013 http://tinyurl.com/l9gqmt5 ), now in Dr. Rolf Brekken's (PPHM SAB) Lab.
http://www.utsouthwestern.edu/labs/brekken/
= = = = = = = = = = = = = =DR. BIRGE:
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html
Dr. Raymond Birge spoke at Peregrine's 11-2015 “Sci. Session” at SITC'15/Maryland ( http://tinyurl.com/pbof95w ); will be the chair in June 2016 of the ICDS'16/Ireland session where Dr. Rolf Brekken (PPHM SAB) is speaking on “Blockade of PS & Immune Activation in Cancer”; and now also speaking in late June'16 at ICHC'16/Istanbul on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”.
3-1-14/Youtube: “Cancer Research” - Dr. Raymond Birge Laboratory, Rutgers http://birgelab.org
2-25-16/PR: IDMC Halts SUNRISE at 1st Look-in. Bavi+Doce arm “OS performing as expected”; Doce arm “dramatically outperforming OS expectations”…
2-25-16: Peregrine Pharmaceuticals Provides Update on Phase III SUNRISE Trial of Bavituximab
- Conference Call With Management Scheduled for 4:30pm ET
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=957281
TUSTIN, Feb. 25, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM, PPHMP), a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer, today announced that it is discontinuing the company's Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC). The decision to stop the trial was based on the recommendation of the study's Independent Data Monitoring Committee (IDMC) following a pre-specified interim analysis performed after 33% of targeted overall events (patient deaths) in the study were reached. Results of the analysis demonstrated that the bavituximab plus docetaxel group did not show a sufficient improvement in overall survival as compared to the docetaxel group to warrant continuation of the study. The interim analysis showed that the bavituximab combination group is performing as expected according to the original trial assumptions in terms of overall survival, while the docetaxel group is dramatically outperforming overall survival expectations based on the original trial assumptions and as compared to recently published studies.
"Let me start by taking this opportunity to thank all of the patients, their families, and the physicians who participated in the SUNRISE trial. While we are deeply disappointed by this early outcome from the SUNRISE trial, we plan to take a deliberate and detailed approach in reviewing and verifying all available data from the trial in order to understand what subgroups or other patient characteristics may have impacted the performance of the study. While we perform this analysis, we plan to put our other chemotherapy combination studies on hold until we have a clear understanding of the SUNRISE study results," said Steven W. King, President & CEO of Peregrine. "While this is an unexpected and disappointing setback for the bavituximab chemotherapy combination clinical program, we have not seen anything in this trial result that diminishes our enthusiasm for advancing our immuno-oncology (I-O) combination trials. The I-O combination studies are based on different mechanistic synergies that are clearly separate from the chemotherapy combination being evaluated in the SUNRISE study. In addition, it is important to note that in no way do these results have any impact on our contract manufacturing business conducted through our wholly owned subsidiary, Avid Bioservices. This business has shown consistent revenue growth and has been instrumental in maintaining a strong cash position and our plan is to continue growing this business."
As of February 1, 2016, Avid Bioservices had a revenue backlog in excess of $58 million under committed contracts from existing clients. In addition, Peregrine had $67.5 million in cash and equivalents as of January 31, 2016.
Conference Call Today
Peregrine will host a conference call today beginning at 4:30PM ET (1:30PM PT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the archived webcast please visit: http://ir.peregrineinc.com/events.cfm .
About Bavituximab: A Targeted Investigational Immunotherapy
Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and robust anti-tumor immune responses.
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer. Bavituximab is the company's lead immunotherapy candidate. In addition to its drug development programs, Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http:/www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Safe Harbor*snip*
Contacts: Jay Carlson, Peregrine Pharmaceuticals, Inc., (800) 987-8256 info@peregrineinc.com
Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401 sdiaz@vidasp.com
Tim Brons (Media), Vida Strategic Partners, 415-675-7402 tbrons@vidasp.com
= = = = = = = = = = = = = = =
BAVITUXIMAB "SUNRISE" PHASE III TRIAL: http://www.SunriseTrial.com (Start=Dec'13 EstPrimComp=Dec’16)
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673 <=chgd. to "Active, Not Recruiting." 2-1-16
...2 ARMS: A=BAVI/3mg+DOCE(Weekly), B=Doce+Placebo(Weekly)
...ECOG's: Ph3SUNRISE/0-1, Ph2Bavi+Doce(n=120)/0-1-2, Ph2Bavi+Durva(AZN)/0-1, Ph3Herbst2010/0-1 (2L/NSCLC)
...161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
...EU/EEA Registries: (pts world=582, pts/EU=345, planned EU sites=100) http://www.clinicaltrialsregister.eu/ctr-search/search?query=2013-003953-13
2-25-16: IDMC Halts SUNRISE at 1st Look-in. Bavi+Doce arm “OS performing as expected”; Doce arm “dramatically outperforming OS expectations” http://tinyurl.com/jbg48vs
2-1-16: SUNRISE Trial status updated: “Study ongoing, but not recruiting.” http://www.clinicaltrials.gov/ct2/show/NCT01999673
12-10-15: “Sunrise >90% enrolled; sufficient to allow the 2 planned interim looks (33%/50%) & final readout based on PrimEndPt=OS” http://tinyurl.com/jkp885g
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
3-7-14: PR & Conf-Call: "first patients enrolled and dosed." http://tinyurl.com/kh9cnrg
1-6-14: FDA grants FAST TRACK status to Bavi in 2ndLine NSCLC http://tinyurl.com/l799ukk
12-30-13: Pivotal Ph.3 ‘SUNRISE’ NSCLC Trial Initiated (n=~600, sites=100+) http://tinyurl.com/kdjb9qz
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
Yes, don't rule out Bavi + IDO pathway inhibitors...
7-14-14/CC: Steve Worsley (VP/BusDev) said that Peregrine is ”seeking collaborators that build on other checkpoints of pathways such as PD-1, IDO, immune-stimulatory drugs, and even vaccines…” http://tinyurl.com/o2e4a4g
Interesting: PPHM KOL/SRB’r Dr. Scott Antonia’s (Moffitt-CC/Tampa) role in IDO dev: http://tinyurl.com/mol86st <=“1st to report in 2002 that IDO inhibitors delay growth of human lung cancer cells”
• Incyte’s immune checkpoint inhibitor INCB24360 is an oral inhibitor of IDO1 (indoleamine dioxygenase-1) – http://www.incyte.com
• NewLink Genetics’ immune checkpoint inhibitor indoximod is an oral inhibitor of IDO (indoleamine 2,3-dioxygenase) – http://newlinkgenetics.com
...“IDO pathway inhibitors are another class of immune check point inhibitors akin to the recently developed antibodies targeting CTLA-4 & PD-1 that represent potential breakthrough approaches to cancer therapy.”
= = = = = = = =
2-23-15: “Bristol-Myers bags IDO immunotherapy in $1.25B buyout of fledgling Flexus”
http://www.fiercebiotech.com/story/bristol-myers-bags-ido-immunotherapy-125b-buyout-fledgling-flexus/2015-02-23
12-2009/SITC: “Small-Molecule Inhibitors of the IDO Pathway as Immune Modulators”
...Slide18 ref's Ph1 Trial of IDO Inhibitor 1-D-MT (NewLink Genetics) with PI=Dr. Scott Antonia/Moffitt (PPHM SAB)
PDF: http://www.sitcancer.org/meetings/am09/presentations/fri/Munn.pdf ...See ASCO'12: http://meetinglibrary.asco.org/content/96504-114
= = = = = = = = = = = = = = = = = = = = = = =
Sep6-9 2015: IASLC’s 16th World Conf. on Lung Cancer, Denver http://tinyurl.com/p9eduac (PPHM KOL/SAB Dr. David Carbone becomes President of IASLC eff. 9-9-2015)
…Peregrine Exhibiting Booth #1724 and 3 Bavi presentations (1 Oral, 1 Mini-Oral, 1 poster) by UTSW (David Gerber, Rolf Brekken, Xianming Huang) and Moffitt CC (Scott Antonia), all with Peregrine co-authors.
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
...**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
I love Peregrine's new Logo unveiled for Wistar's June'16 Conference in Philly. ( http://myeloidsuppressors.com/sponsors & http://tinyurl.com/hwrogdk )

= = = = = = = = = = = = =
4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
"Phil will be sorely missed as a colleague and friend. At Peregrine, each of us is pursuing Phil's dream of bringing important drugs to patients who need them based on his basic research. We are more motivated than ever to see this dream through."
…Dr. Thorpe’s successor at UTSW: long-time colleague, co-inventor of 2C3, Dr. Rolf A. Brekken, PhD - http://www.utsouthwestern.edu/labs/brekken

8-2014: UK colleagues from Chester Beatty Labs(ICR/Univ-London), ICRF(IsraelCancerRes), & UTSW create memorial for Dr.Thorpe in London’s Chelsea Physic Garden: http://tinyurl.com/lon73l7
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
PPHM SAB'ers Drs. Brekken+Gabrilovich June16-19 WISTAR “Myeloid Suppressors” Conf. In Philadelphia. General Sponsors are: Peregrine & Genentech.
Jun16-19 2016: “WISTAR's Regulatory Myeloid Suppressor Cells Conf.”, Philadelphia
“From Basic Discovery To Therapeutic Application”
http://myeloidsuppressors.com
“This conference will focus on the pathological functions of myeloid-derived suppressor cells, dendritic cells, macrophages and neutrophils, and provide a forum for in-depth discussion of the most pressing issues associated with the biology & clinical application of these cells. The conference will bring together scientists from academia & industry interested in the basic and translational aspects of these cells in cancer and other pathological conditions.”
OVERVIEW: Myeloid cells are comprised of populations of mature terminally differentiated macrophages, dendritic cells and neutrophils, as well as immature myeloid cells including granulocytes, monocytes, and myeloid progenitors. These cells are evolutionarily designed to protect the host from bacteria and viruses by utilizing mechanisms of innate & adaptive immunity. They are also major contributors to tissue remodeling after injuries or resolved inflammation. In cancer, chronic infections, and inflammation, these cells are undergoing extensive changes, which make them immunosuppressive, able to actively promote angiogenesis, tumor cell invasion, and formation of pre-metastatic niches. Recent data demonstrates the association of accumulation of these cells in cancer patients and clinical outcomes of the diseases. With the development of novel immunotherapeutics, it became apparent that regulatory myeloid cells play a major role in limiting therapeutic efficacy of treatment. This conference is focused on pathological functions of myeloid-derived suppressor cells, dendritic cells, macrophages and neutrophils. The specific goals of this meeting are to develop a better understanding of the mechanisms regulating the accumulation of these cells, markers that allow for detection of these cells in cancer patients and patients with chronic infections & inflammation, and approaches to therapeutic targeting of these cells.
ABSTRACTS: titles & authors will be posted on the conference website before the conference begins. No data will be posted online before the conference.
- - - - - - -
Conf. Chair: Dmitry Gabrilovich, MD, PhD [PPHM SAB]
Keynote: Robert A. Weinberg, Whitehead Inst. for Biomedical Research, MIT
40 Speakers, including:
Rolf A. Brekken [PPHM SAB], UTSW-MC/Dallas
Dmitry I. Gabrilovich [PPHM SAB], The Wistar Institute, Philadelphia
TWO GENERAL SPONSORS: PEREGRINE & GENENTECH (Session: Juno, Galera; Day: Syndax)

= = = = = = = = = = = = = = = = = = = = =
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: ”Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
RRdog passed away over the weekend. I have no details. Fitting we made his last post a stickie. A man who was so dedicated to seeing Peregrine's ultimate Success come true. So many contributions here. Sincerest condolences to his Family. R.I.P., you good & outstanding man.
Stoney: Pg.17(2-19-16/Dr.Gerber): "The ORR of 17% (Bavi/3mg arm) - although falling short*** of the 26% required to achieve the primary endpoint (ORR) of the trial - represents an approx. doubling of rates typically seen with docetaxel. The clinical efficacy of docetaxel + bavituximab 3 mg/kg also appears comparable to that of other docetaxel combinations, including the vascular endothelial growth factor receptor antibody ramucirumab and the multitargeted angiokinase inhibitor nintedanib."
***Pg.2: "Due to a labeling discrepancy, the placebo & bavituximab 1 mg/kg arms were combined and compared to the bavituximab 3 mg/kg arm. This exploratory analysis showed similar toxicity between arms, and albeit without meeting the pre-set statistical endpoints due to the pooled control arm data, had trends favoring bavituximab 3 mg/kg."
= = = = = = = = = = =
2-19-16: "(Ph2) Docetaxel + Bavituximab in Previously Treated, Advanced Nonsquamous NSCLC"
Clinical Lung Cancer Jrnl (Elsevier)
Rec. 11-4-15; Rev. 2-5-16; Acc. 2-9-16; Online 2-19-16
http://www.sciencedirect.com/science/article/pii/S1525730416300079
FULL article: http://www.sciencedirect.com.sci-hub.io/science/article/pii/S1525730416300079
David E. Gerber 1, David R. Spigel 2, David Giorgadze 3, Mikhail Shtivelband 4, Olga V. Ponomarova 5, Joseph S. Shan 6, Kerstin B. Menander 7, Chandra P. Belani 8
1 Harold C. Simmons Comprehensive Cancer Center, UTSW-MC/Dallas
2 Tennessee Oncology, PLLC, 250 25th Ave North, Nashville, TN
3 Medulla Chemotherapy and Immunotherapy Clinic, Tbilisi, Rep. of Georgia
4 Ironwood Cancer and Research Center, Chandler, AZ
5 Kyiv City Oncology Hospital, Kyiv, Ukraine
6/7 Peregrine Pharmaceuticals, Inc., Tustin, CA
8 Penn. State Hershey Cancer Institute, Hershey, PA
ABSTRACT:
BACKGROUND: Bavituximab is a phosphatidylserine-targeting antibody with a selective tumor, vascular-directed immune response. This phase 2 trial evaluated efficacy & safety of bavituximab combined with docetaxel for previously treated, advanced nonsquamous NSCLC.
METHODS: Patients were randomized 1:1:1 to receive docetaxel 75 mg/m2 every 21 days for up to 6 cycles combined with weekly, blinded infusions of placebo, bavituximab 1 mg/kg, or bavituximab 3 mg/kg until progression or unacceptable toxicity. The primary endpoint was overall response rate (ORR), with a predefined endpoint of 26% in the bavituximab arms. After study unblinding, vial-coding discrepancies were discovered in the placebo and bavituximab 1 mg/kg groups. In exploratory analyses, data from these groups were pooled to form the control group and compared with the 3 mg/kg group.
RESULTS: Efficacy endpoints in the bavituximab 3 mg/kg group (n=41) and in the placebo/bavituximab 1 mg/kg group (n=80), respectively, were as follows: ORR 17.1% (95% CI 5.6%, 28.6%) and 11.3% (95% CI 4.3%, 18.2%); median progression-free survival 4.5 and 3.3 months (HR 0.74 [95% CI 0.45, 1.21], P=.24); median overall survival 11.7 and 7.3 months (HR 0.66 [95% CI 0.40, 1.10], P=.11). Toxicities were manageable and similar between arms.
CONCLUSIONS: The combination of bavituximab and docetaxel is well tolerated. Although no firm efficacy conclusions can be drawn and the trial did not meet the predefined primary endpoint, exploratory analyses suggest trends favoring the addition of bavituximab 3 mg/kg to docetaxel. This regimen is being evaluated in the ongoing, global, Ph.3 SUNRISE trial [ http://www.clinicaltrials.gov/ct2/show/NCT01999673 ].
= = = = = = = = = = = = = = = = = = = = =
F. LEAD IND: Phase IIb Bavi+Doce vs. 2nd-Line NSCLC (randomized, double-blinded, placebo-ctl'd, n=120, 'registrational')
Protocol: http://clinicaltrials.gov/ct2/show/NCT01138163 (24 U.S. + 15 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 53 as of 8-12-11)
Enrolled Oct2010 - Oct2011 at 40 global sites (per J.Shan 9-7-12 webcast ( http://tinyurl.com/8cn87la ) ;
8-2012: Compare Bavi+Doce's MOS=11.7mos (Bavi/3mg) to the 4 Curr-Approved 2Line/NSCLC Drugs http://tinyurl.com/cgnkvpa
• Taxotere/docetaxel => MOS=6.3mos (meta-analysis of 5 trials, 865 pts)
• Altima/pemetrexed => No diff. vs. Docetaxel (Ph.3 non-inferiority vs. Doce, 571 pts)
• Tarceva/erlotinib => MOS=5.3mos (TITAN Ph.III n=424 trial - see http://tinyurl.com/8w8lo93 )
• Iressa/gefitinib => "Iressa does not improve OS"
9-8-15: CMS SETTLEMENT EXECUTED (CSM pays PPHM only $600k for their “breach of contract, negligence & constructive fraud”, due to “limitation of liability contract clauses”) http://tinyurl.com/pemub47 (10Q/pg.17)
6-23-14: PPHM files Opposition to CSM’s Motion For Partial Summary Judgment - excerpts: http://tinyurl.com/q8xwd4v ;
. . .Declaration of Joseph Shan (VP/Clin+RegAffairs): http://tinyurl.com/kdgllxn
. . .Declaration of Jeffery Masten (VP/Quality): http://tinyurl.com/oru9p5q #6: “up to 25%" of CTL had 1mg, and “up to 25%" of 1mg had CTL(DoceOnly).
3-28-14: Peregrine files 1st Amended Complaint vs. CSM (13pgs) http://tinyurl.com/lsgf5lz
...Pg6: ”[as of 4-15-10], CSM had already secretly & unilaterally swapped the A & B arms so that those patients that were randomized in the A arm (CTL) and supposed to receive placebo treatments, were actually receiving 1MG Bavi treatments, and vice-versa. Peregrine’s Fall’12 investigation revealed that CSM committed other labeling & distribution errors affecting the A & B arms above & beyond the swap of the A& B arms noted above.”
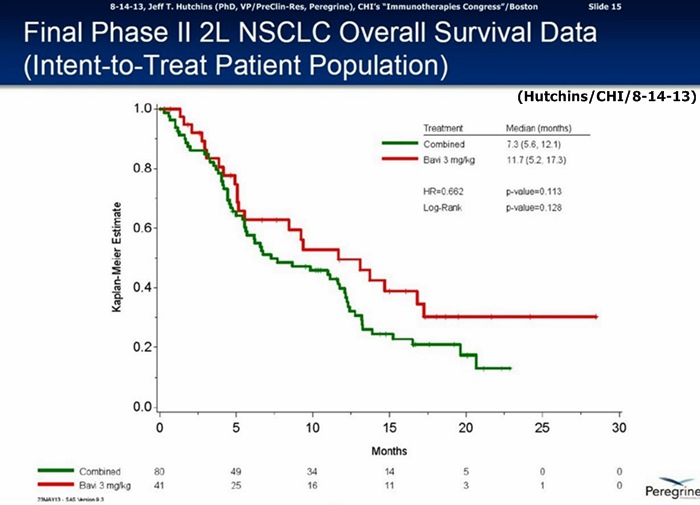
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
2-19-13: Topline Data Update from 2nd-Line NSCLC Trial after data discrepancies review http://tinyurl.com/ansqcea
…60% improvement in MOS: 3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined 1mg & Doxy+placebo arms), HR=.73, p=.217
6-5-13: FTM's table of MOS data in 15 prior Doxy 2nd-Line NSCLC trials (Bavi's 60% MOS Improvement is Tops) http://tinyurl.com/m886ctb

1-25-13: MLV's George Zavoico recaps 2ndLine/NSCLC data errors & current status of PPHM's review http://tinyurl.com/b9u4pk8
...GZ: "This means that patients randomized into the high dose arm were administered Bavi correctly, whereas some of the patients in the placebo arm were administered low dose Bavi and some in the low dose Bavi arm were administered placebo. More importantly, the findings suggested that the MOS of 13.1 mos. in the high dose arm was likely to be valid. Even by historical measures, this is a remarkable result, since docetaxel's product insert lists the MOS of NSCLC patients receiving this widely used drug as 2nd-Line therapy in 2 trials as 5.7 & 7.5 mos. In effect, adding Bavi doubled the MOS. In our view, this was an extraordinary stroke of luck. If the high dose arm had been affected by the coding discrepancy, Peregrine would have been in a completely different & unfortunate position… Moreover, Peregrine must determine how best to present its case to the FDA. Will the historical controls be sufficient to justify moving Bavi into a Phase III pivotal trial, or will Peregrine have to pool the results of the placebo & low-dose arms and use that as a comparator to argue for moving ahead? A simple average of the placebo & low-dose arms results in a new control MOS of about 8.4 mos., still several months less than that of the high dose arm. This quick analysis results in about a 5-mo. survival advantage, a substantial prolongation for patients with second-line NSCLC and likely to justify moving Bavi into a pivotal Phase III trial in 2013, in our view."
1-7-13 PPHM PR - Review Update: "discrepancies are isolated to the placebo and 1 mg/kg arms; no evidence of discrepancies in the 3 mg/kg arm… Peregrine is taking a very conservative approach toward analyzing the results by combining the placebo & 1mg/kg arms into one treatment arm (control arm), and comparing to the 3mg/kg arm. This analysis indicates that the 3 mg/kg arm continues to show favorable TRR's, PFS, and OS over the new combined control arm. Peregrine expects to announce more detailed results from the analysis in the near term when it is completed." http://tinyurl.com/asup54d
9-24-12: Major Discrepancies found in 2nd-Line NSCLC Ph.2B Treatment Group Coding by Indep. 3rd-Party Vendor CMS/FargoND http://tinyurl.com/8r9zcqy
…"Investors should not rely on clinical data that the company disclosed on or before Sept. 7, 2012 from its Ph.2 Bavi trial in patients with 2nd-Line NSCLC or any presentations or other documents related to this Ph.2 trial."
9-24-12: Peregrine sues CSM Over Bavi Ph.2B 2nd-Line NSCLC Clinical Trial Mix-Up http://tinyurl.com/8fpgngu
…CSM = Clinical Supplies Management Inc., Fargo ND http://www.csmondemand.com
...1-17-13: Peregrine's lawsuit against CSM for "breach of contract & negligence" SERVED http://tinyurl.com/a7zrgys
…9-10-12 CEO Steve King, QtlyCC ( http://tinyurl.com/8nkwrml )
……"These are truly remarkable results (statistically doubling MOS) that are not only great for the pgm… but also great news for the NSCLC patients in the trial…"
…9-10-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/8nkwrml )
……"The NSCLC data we announced 9-7-12 has far exceeded our expectations, and I hope that you're as excited as I am with bavituximab's potential. I feel strongly that Peregrine should be recognized for having the corporate courage to conduct the rigorous, randomized placebo-controlled Phase II trial that provided these robust data and that provide the basis for us to plan for a pivotal Phase III program."
...9-7-12: PPHM Press Release about Dr. Gerber's plenary at ASTRO/Thoracic/Chicago: http://tinyurl.com/96wrrso
…"The interim data showed a statistically significant improvement in OS (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS (11.1/13.1mos. vs. 5.6mos.) in the Bavituximab-containing arms compared to the [Docetaxel] ctl-arm."
......VP Joe Shan's 15min. Webcast & Slideshow recapping Dr. David Gerber's 9-7-12 ASTRO/Chicago Plenary: http://tinyurl.com/96wrrso
…8-15-12 CEO Steve King, Wedbush/NYC ( http://tinyurl.com/8mhrtld )
......"As we're sitting here today, we have still not reached the # of events for MOS in either of the Bavituximab arms - and, in fact, we still have patients that are on treatments." Q&A: "it's going to be a very positive MOS result, it's just a matter now of magnitude."
…7-16-12 CEO Steve King, QtlyCC ( http://tinyurl.com/cs7spbz )
......"The strength of this 2nd-Line NSCLC data (esp. MOS trends) in this large area of high unmet medical need has also sparked a surge in partnering discussions that has included over 15 in-person partnering meetings since that time with major players in oncology, with all discussions ongoing and addl. parties showing interest. Our goal for the program is to position ourselves, along with a potential partner, to initiate Ph3 by mid-2013, which means an EOP2 meeting by yr-end'12. It would be ideal to have a partner on board to participate in the EOP2 meeting, and we have communicated this to interested parties and they agree."
…7-16-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/cs7spbz )
……"We've been working very hard and very actively on the next steps in our Bavi 2nd-Line NSCLC pgm, given the favorable data that we've seen. As you can imagine, with data like this, there are many things that we need to consider. One consideration is that, should the data continue to trend the way it is, particularly in survival, this opens a door for potential discussions around a pathway for Accelerated Approval. At this point, all options are being considered, with Peregrine working towards the most efficient path forward from a regulatory standpoint." Q&A: "…all in all, I think the data is extremely compelling and I think it makes a really good case. Certainly, I think, I've seen a lot of Ph2 & Ph3 data, and this is as compelling Ph2 data as I've ever seen. So, I'm very comfortable proposing a meeting with the FDA for Q4'12."
…7-12-12 CEO Steve King, JMP-Conf/NYC ( http://tinyurl.com/csdclwb )
……"Re: 2nd-Line/NSCLC trial, the most thrilling thing is the fact that, even though we'd reached MOS for the ctl-arm(Doce) at end of Apr'12 of LESS THAN 6MOS, the majority of patients are still alive (today) in both Bavi arms, and we expect that to continue for some period of time still. Ph3 planning is underway already; our goal is to start this Ph3 by mid'13, meaning an EOP2 meeting with the FDA in Q4'12; our goal is to bring a partner on board, ideally in time for that EOP2 meeting, certainly before the beg. of the Ph3 trial."
…5-21-12: TopLine data n=117 for Bavi/3mg+Doce arm: ORR=17.9%/PFS=4.5mos (vs. CTL 7.9%/3mos) http://tinyurl.com/73aeyxj
......Importantly, MOS for CTL-arm "< 6 mos", but not yet reached in both Bavi arms.
...10-6-11: Enrollment complete. http://tinyurl.com/3m9re39
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to ~45 sites, expect enroll. comp. "early in Q4/2011", data unblinding 1H'12. http://tinyurl.com/6k6y2as
…3-17-10/Roth, CEO S.King: "We refer to this trial as a Registrational Phase II Study, because we believe that if we have results anywhere near approaching what we saw in the earlier [India] study, it could be a conduit for Accelerated Approval."
...6-4-10: Ph.2b randomized reg. trial Open for enrollment: http://tinyurl.com/25v22qk
……"up to 120 refractory patients at ~30 clinical sites; goal: fully-enroll by mid'11, topline data by y/e'11."
= = = = = = = = = = = = = = = = = = = = = = = = = =
Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673 <=chgd. to "Active, Not Recruiting." 2-1-16
...2 ARMS: A=BAVI/3mg+DOCE(Weekly), B=Doce+Placebo(Weekly)
...ECOG's: Ph3SUNRISE/0-1, Ph2Bavi+Doce(n=120)/0-1-2, Ph2Bavi+Durva(AZN)/0-1, Ph3Herbst2010/0-1 (2L/NSCLC)
...161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
...EU/EEA Registries: (pts world=582, pts/EU=345, planned EU sites=100) http://www.clinicaltrialsregister.eu/ctr-search/search?query=2013-003953-13
2-1-16: SUNRISE Trial status updated: “Study ongoing, but not recruiting.” http://www.clinicaltrials.gov/ct2/show/NCT01999673
12-10-15: “Sunrise >90% enrolled; sufficient to allow the 2 planned interim looks (33%/50%) & final readout (80%) based on PrimEndPt=OS” http://tinyurl.com/jkp885g
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
3-7-14: PR & Conf-Call: "first patients enrolled and dosed." http://tinyurl.com/kh9cnrg
1-6-14: FDA grants FAST TRACK status to Bavi in 2ndLine NSCLC http://tinyurl.com/l799ukk
12-30-13: Pivotal Ph.3 ‘SUNRISE’ NSCLC Trial Initiated (n=~600, sites=100+) http://tinyurl.com/kdjb9qz
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
Clock hasn't “reset” - it's TURNED OFF (as of yesterday, BTW). The only thing that could turn it back on is if we CLOSE below $1.00 in the future.
Bavi+Taxol MSKCC/Collab. Triple- Breast Cancer Ph2 Trial hits ClinicalTrials.gov: n=42, Start=Mar.2016
“A Study of Neoadjuvant Paclitaxel in Combination With Bavituximab in Early-Stage Triple- Negative Breast Cancer – Ph.2”
http://clinicaltrials.gov/ct2/show/NCT02685306 (rec. 2-2-16, upd. 2-12-16)
PURPOSE: The primary purpose of this research study is to see whether adding bavituximab (an investigational drug) to the standard chemotherapy drug taxane, will improve the results of the treatment for early-stage Triple Negative Breast Cancer followed by Standard-of-Care surgery
Est. Enrollment: 42
Start Date: March 2016
Est. Study Completion Date: Sept 2017
Est. Prim. Comp. Date: April 2017 (Final data collection date for primary outcome measure)
PI: David Page, MD - Providence Portland / Robert W. Franz Cancer Res. Center
Est.Init Q1/2016: Ph2, Bavi+Paclitaxel(Taxol), Early Stage Triple- Breast Cancer (TNBC), (randomized, open-label, ~27pts), "originated from Peregrine's ongoing collab. with MSKCC" http://tinyurl.com/zhdy37a
= = = = = = = = = = = = = = = = = = = = = = = = = =
1-11-16 PR, “Peregrine Provides Update on Planned Expansion of Bavi Clinical Pgm in Lung, Breast and Other Cancers”… http://tinyurl.com/zhdy37a
PLANNED TRIALS...
#4. Phase II Trial in Early Stage TNBC in Combination with Chemotherapy
Peregrine is planning to initiate a Phase II trial of bavituximab in combination with neoadjuvant chemotherapy in early stage TNBC. The primary endpoint of this study is to determine the pathologic complete response rate (pCR), an accepted surrogate endpoint in early stage TNBC. The concept for this neoadjuvant setting trial, which will be conducted at a few select U.S. sites, originated from Peregrine's ongoing collaboration with Memorial Sloan Kettering Cancer Center (MSKCC). The company has filed a study protocol to its existing bavituximab IND application in the U.S. and is currently working to open clinical trial sites, including one that will be led by David B. Page, M.D., at the Providence Cancer Center in Oregon. http://tinyurl.com/zhdy37a
- - - - - - - -DR. DAVID PAGE: note his prior work with Dr. Jedd Wolchok, chief of Mem.Sloan's Melanoma & Immunotherapeutics Service who “investigates novel approaches for cancer immunotherapy and mechanisms of tumor cell–immune cell interactions”… http://www.bcrfcure.org/researchers/david-page
5-29-15: Peregrine/MSKCC Collab. Announced: http://tinyurl.com/zkvebh6
“The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.”
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok (MSKCC).
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub (MSKCC).

12-10-15/CC: “Sunrise >90% enrolled; sufficient to allow the 2 planned interim looks (33%/50%) & final readout based on PrimEndPt=OS” http://tinyurl.com/jkp885g
12-10-15/CC: CEO STEVE KING – OPENING COMMENTS: http://tinyurl.com/jkp885g
“...today’s development discussions will focus on these efforts, including upcoming completion enrollment in the SUNRISE trial which is evaluating a chemotherapy combination, and, as SUNRISE wraps up, our plans for a smooth transition of the key SUNRISE clinical sites directly to the next Phase II lung cancer trial evaluating an IO combination [init Q1/2016: Phase2, Bavi+durvalumab(AZN's anti-PD-L1 MEDI4736), 2ndLine NSCLC, squam+nonsquam (randomized, open-label, ~200pts)]. In addition, we are expanding our potential cancer indications through initiation of a Phase II/III metastatic breast cancer study. All of this while continuing to work through several other clinical trial concepts actively under development or initiation in the new year. On the development front, I’m pleased to report today that we are nearing completion of enrollment in the cornerstone of our bavituximab development strategy, our Phase III SUNRISE trial. In fact, with over 90% of the expected enrollment complete, we currently have sufficient patient enrollment based on the assumptions of the study to allow the trial’s planned interim evaluations and final readout based on the primary endpoint of overall survival. Having said that, we do expect to complete enrollment of at least the pre-specified 582 patients over the coming weeks. At this point, the next big milestones really are the interim data analysis from the study, expected to take place during early & mid 2016, with trial unblinding expected toward the end of 2016.”
12-10-15.CC: JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS: http://tinyurl.com/jkp885g
“I’d like to start by quickly addressing our Phase III SUNRISE trial, which is evaluating the use of bavituximab & docetaxel in patients with previously treated locally advanced or metastatic non-squamous NSCLC. As Steve stated, we have already enrolled the number of patients required to achieve the trial’s main objectives, and expect to complete enrollment of the target sample size of 582 in the coming weeks. The next milestones are the interim analyses that will be conducted when 33% & 50% of the targeted number of deaths are reached. While these are event driven, it is our expectation that the 1st interim analysis will read out in early 2016 and the 2nd interim analysis around mid-2016. The final analysis [1-18-16/SK/Noble: “Unblinding at 80% of Events” http://tinyurl.com/j9dkekm ], which will trigger study unblinding, is currently projected to occur at the end of 2016. With the SUNRISE enrollment nearing completion, the Peregrine clinical team is shifting focus to a number of new clinical projects, including those just referenced by Steve. In each case, our goal is to generate clinical evidence of bavituximab’s ability to improve patient outcomes when combined with chemotherapy and immuno-oncology agents. With this goal in mind, we are very pleased to be collaborating with AstraZeneca. Through this partnership, we will be conducting 2 clinical trials, both of which will be initiated in 2016. One trial, which we expect to initiate early 2016, is a global randomized Phase II study in approx. 200 patients with previously treated NSCLC. This trial will evaluate the combination of bavituximab and AZ’s anti-PD-L1 immune checkpoint inhibitor durvalumab, or MEDI4736. As part of this combination trial, patients will also be evaluated retrospectively for the correlation between their PD-L1 levels & clinical outcomes. As the remaining patients are enrolled into SUNRISE, we have already begin laying the groundwork to quickly initiate this new Phase II combination study at a number of our most active sites participating in SUNRISE. These investigators are very familiar with bavituximab and have access to the appropriate patient populations, and we believe this experience will greatly benefit our new study.”
= = = = = = = = = = = = =
BAVITUXIMAB "SUNRISE" PHASE III TRIAL: see http://PeregrineTrials.com => http://www.SunriseTrial.com (Start=Dec'13 EstPrimComp=Dec’16)
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
2 ARMS: A=BAVI3mg+DOCE(Weekly), B=Doce+Placebo(Weekly)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673 <=chgd. to "Active, Not Recruiting." 2-1-16
Geo/Map view of Worldwide Ph3 SUNRISE Sites: http://www.medbravo.org/study?study=NCT01999673
...161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
Q. Phase II Bavi+durvalumab(anti-PD-L1 MEDI4736), 2ndLine NSCLC(squa+nonsq.), randomized, open-lab, n=198, AZN Collab.)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT02673814 <=added 2-2-16
3 ARMS: A./DurvaOnlyBiWK, B./BaviWEEKLY+DurvaBiWK, C./BaviBiWK+DurvaBiWK
...Est. Enrollment: 198, Study Start: Feb. 2016, Est.Prim.Comp: Apr.2017 (Final data coll. for prim.outcome), Est.StudyComp: Apr.2018
.
.
SKing/8:50 Slide16 (1-18-16): “Unblinding at 80% of Events” http://tinyurl.com/j9dkekm

JohnR: “NASDAQ Last Sale Price: The price at which a stock last traded during regular market hours."
http://www.nasdaq.com/quotes/real-time.aspx
Right, def. 2 Arms in SUNRISE (50% get Bavi+Doce, 50% get DoceOnly – All Weekly). Maybe he got confused with the upcoming AZN-Collab. Bavi+Durva/NSCLC N=198 Ph2 trial, which will have 3 arms (A./DurvaOnlyBiWK, B./BaviWEEKLY+DurvaBiWK, C.BaviBiWK+DurvaBiWK)...
PS: I'm personally very happy that in the new AZN/Bavi+Druva trial, 67% will get the benefit of Bavi, rather than 50% in Sunrise.
= = = = = = = = = = = =
Q. Phase II Bavi+durvalumab(anti-PD-L1 MEDI4736), 2ndLine NSCLC(squa+nonsq.), randomized, open-lab, n=198, AZN Collab.)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT02673814 <=added 2-2-16
...3 Arms: A./DurvaOnlyBiWK, B./BaviWEEKLY+DurvaBiWK, C.BaviBiWK+DurvaBiWK
...Est. Enrollment: 198, Study Start: Feb. 2016, Est.Prim.Comp: Apr.2017 (Final data coll. for prim.outcome), Est.StudyComp: Apr.2018
= = = = = = = = = = =
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673 <=chgd. to "Active, Not Recruiting." 2-2-16
Geo/Map view of Worldwide Ph3 SUNRISE Sites: http://www.medbravo.org/study?study=NCT01999673
...161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
2-17-16/BiotechStocks: 4 Stocks to Consider for Biotech Turnaround. Don't know anything about BiotechStocks, but a straightforward overview, 'tho no mention of the 3 new 2015-16 collabs, MSKCC, AstraZeneca, and NCCN...
2-17-16/BiotechStocks: 4 Stocks to Consider for Biotech Turnaround
http://finance.yahoo.com/news/4-stocks-consider-biotech-turnaround-165500882.html
...Curis, Inc. (CRIS $1.50), Del Mar Pharm. (DMPI $.78), Immune Design (IMDZ $10.73)...
Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM)
Peregrine's lead candidate is bavituximab, which is a chimeric monoclonal antibody. Essentially, the drug binds to receptors on the surface of cancerous cells and signals the immune system to come and attack endothelial cells. Endothelial cells are cells that line blood vessels, and so through inducing this attack, bavituximab aims to cut off the blood supply to a tumor and halt its growth. Data from a Ph. II trial in lung cancer [6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7 “…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113” ] demonstrated superiority to docetaxel, a multi billion dollar blockbuster currently marketed by Sanofi (NYSE: SNY) - and Peregrine is currently running a large Ph. III of 600 enrolled patients in the same indication. The trial is again pitching bavituximab against Docetaxel, with a 3rd placebo arm thrown in. If the drug can replicate the Ph. II data in this trial (called SUNRISE - all details here: https://www.clinicaltrials.gov/ct2/show/NCT01999673 ), Peregrine could have a real blockbuster on its hands. The trial is expected to complete in December this year, so again look for any interim updates to provide upside catalysts, and end of the year top-line efficacy to form the basis of an early 2017 NDA submission.
Source: Biotech Stocks ( http://www.BiotechStocks.com )
Direct link: http://biotechstocks.com/4-stocks-to-consider-for-a-biotech-turnaround
The MSK collab's already resulted in upcoming Triple-/Breast Cancer Trial…
1-11-16 PR, “Peregrine Provides Update on Planned Expansion of Bavi Clinical Pgm in Lung, Breast and Other Cancers”… http://tinyurl.com/zhdy37a
PLANNED TRIALS...
#4. Phase II Trial in Early Stage TNBC in Combination with Chemotherapy
Peregrine is planning to initiate a Phase II trial of bavituximab in combination with neoadjuvant chemotherapy in early stage TNBC. The primary endpoint of this study is to determine the pathologic complete response rate (pCR), an accepted surrogate endpoint in early stage TNBC. The concept for this neoadjuvant setting trial, which will be conducted at a few select U.S. sites, originated from Peregrine's ongoing collaboration with Memorial Sloan Kettering Cancer Center (MSKCC). The company has filed a study protocol to its existing bavituximab IND application in the U.S. and is currently working to open clinical trial sites, including one that will be led by David B. Page, M.D., at the Providence Cancer Center in Oregon. http://tinyurl.com/zhdy37a
- - - - - - - -DR. DAVID PAGE: note his prior work with Dr. Jedd Wolchok, chief of Mem.Sloan's Melanoma & Immunotherapeutics Service who “investigates novel approaches for cancer immunotherapy and mechanisms of tumor cell–immune cell interactions”… http://www.bcrfcure.org/researchers/david-page
5-29-15: Peregrine/MSKCC Collab. Announced: http://tinyurl.com/zkvebh6
“The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.”
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok (MSKCC).
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub (MSKCC).
= = = = = = = = = = = = = = = = = = =
http://tinyurl.com/zkvebh6 1-16-16: Summary of Peregrine's New Collab's: Mem.Sloan-Kettering, AstraZeneca, NCCN
Post in 3 sections:
I. Memorial Sloan Kettering (MSKCC) – 5-2015/Investigate Novel PS-Targeting Immunotherapy Combos
II. AstraZeneca (Bavi+Durvalumab) - 8-2015/multiple solid tumors, 10-2015/expanded to NSCLC
III. Natl-Comprehensive-Cancer-Network (NCCN) – 1-2016/$2mm grant to NCCN's Oncology Res. Pgm (ORP), “26 of the world's leading cancer centers”
The Ph2/n=120 2Line-NSCLC Trial basically doubled MOS ~6=>~12mos prior to the CMS screwup found in the CTL & 1Mg arms forced Peregrine to combine the 1Mg & CTL arms for comparison with the CMS-untouched 3Mg arm for “Final Data” and FDA approval to proceed with Ph3 SUNRISE. Note that the 9-7-12 interim data (showing stat-sig. doubling of MOS) reported just before CSM Ctl/1mg screwup was found/reported would have been EVEN BETTER had the CTL (DoceOnly) arm not received ~25% of BAVI and the 1Mg arm hadn't received ~25% of DoceOnly...
I. Just Before the CSM CTL<=>1Mg Screwup was found/announced (3mg OK)...
...9-7-12: PPHM PR (& Slides) about Dr. Gerber's plenary at ASTRO/Thoracic/Chicago (INTERIM Ph2 NSCLC Data): http://tinyurl.com/96wrrso
…”The interim data showed a statistically significant improvement in OS (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS (12.1/13.1mos. vs. 5.6mos.) in the Bavituximab-containing arms compared to the [Docetaxel] ctl-arm."
......VP Joe Shan's 15min. Webcast & Slideshow recapping Dr. David Gerber's 9-7-12 ASTRO/Chicago Plenary: http://tinyurl.com/96wrrso
…9-10-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/8nkwrml )
……"The Ph2 NSCLC data we announced 9-7-12 has far exceeded our expectations, and I hope that you're as excited as I am with bavituximab's potential. I feel strongly that Peregrine should be recognized for having the corporate courage to conduct the rigorous, randomized placebo-controlled Ph2 trial that provided these robust data and that provide the basis for us to plan for a pivotal Ph3 pgm."
- - - - - ->Ph2 Interim B4 CMS CTL/1mg Screwup found:
DoceONLY n=38: ECOG0-1-2=25%-62%-13% MOS=5.6mos.
Bavi/1mg: n=40 ECOG0-1-2=45%-43%-12% MOS=11.1mos.
Bavi/3mg n=39: ECOG0-1-2=20%-56%-24% MOS=13.1mos.
***FOUND OUT LATER: ~25% of CTL had 1mg, and 25% of 1mg had CTL(DoceOnly). Ie, CTL arm was HELPED and 1mg arm was HURT. Ie, Results would have been even better w/o CMS screwup. Also, note 3MG Arm had a hurdle of more ECOG=2 pts (24%).
II. FDA APPROVED SUNRISE AFTER PEREGRINE COMBINED DATA IN CTL & 1MG ARMS DUE TO CSM SCREWUP:
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
=>ECOG=2: Combined-Placebo+1mgBavi: 12.5%, 3mgBavi: 24.4%
= = = = = = = = = = = = = = =
ECOG: Ph3SUNRISE/0-1, Ph2Bavi+Doce(n=120)/0-1-2, Ph2Bavi+Durva(AZN)/0-1, Ph3Herbst2010/0-1 (2L/NSCLC)
0=FullyActive, 1=CompletelyAmbulatory/NoStrenuous, 2=Symptomatic/<50%inBed@Day/NoWork, 3=>50%inBed@Day/LimitedSelfCare, 4=CompletelyDisabled
http://radiopaedia.org/articles/ecog-performance-status
6-2012: FTM's charts of MOS Data from CtlArms(chemo) of Comp.Trials for 1NSCLC 2NSCLC PANCRE trials http://tinyurl.com/757plm7
Sunstar, look @Ph2/Bavi+Doce: 24.4% ECOG=2's in 3mgBavi Arm vs. 12.5% in combined placebo+1mgBavi Arm - and yet 3mg arm bested MOS 7.3 => 11.7mos...
I added this to my ECOG post a few mins ago - will Delete this post in a few mins.
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
=>ECOG=2: Combined-Placebo+1MgBavi: 12.5%, 3mgBavi: 24.4%
ECOG: Ph3SUNRISE/0-1, Ph2Bavi+Doce(n=120)/0-1-2, Ph2Bavi+Durva(AZN)/0-1, Ph3Herbst2010/0-1 (2L/NSCLC)
0=FullyActive, 1=CompletelyAmbulatory/NoStrenuous, 2=Symptomatic/<50%inBed@Day/NoWork, 3=>50%inBed@Day/LimitedSelfCare, 4=CompletelyDisabled
http://radiopaedia.org/articles/ecog-performance-status
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
=>ECOG=2: Combined-Placebo+1MgBavi: 12.5%, 3mgBavi: 24.4%
6-2012: FTM's charts of MOS Data from CtlArms(chemo) of Comp.Trials for 1NSCLC 2NSCLC PANCRE trials http://tinyurl.com/757plm7

NBOB, thank you for your thoughts this morning. Peregrine's Mission: “Delivering Innovative and Exceptional Biopharmaceutical Products to Improve Patient Lives” (PeregrineInc.com).

4-2-13: Peregrine Mourns Loss of Dr. Philip E. Thorpe (1951-2013) http://tinyurl.com/l9gqmt5
Phil will be sorely missed as a colleague and friend. At Peregrine, each of us is pursuing Phil's dream of bringing important drugs to patients who need them based on his basic research. We are more motivated than ever to see this dream through."
…Dr. Thorpe’s successor at UTSW: long-time colleague, co-inventor of 2C3, Dr. Rolf A. Brekken, PhD - http://www.utsouthwestern.edu/labs/brekken

PS: Hope to see KBob soon – his surgery is set for March9, a week after his annual Nip Open – are you competing this year? (just kidding, that'd give no one else a chance!)
The AstraZeneca deal IS a Cash Deal as far as I'M concerned. AZN is providing durvalumab in the trials “free of charge”. To me, that's the same as CASH to Peregrine.
12-15-15/BioWorld: “If You Can't Beat 'em, Join 'em; Looking for Immuno-onc Boost”
By Brian Orelli, BioWorld Staff Writer
...Earlier this year, Peregrine partnered up with Astrazeneca to test the pharma's anti-PD-L1 immune checkpoint inhibitor, durvalumab, with bavituximab, after preclinical data suggested that the combination might help even if tumors don't initially express PD-L1. By removing the phosphatidylserine checkpoint, the immune system is activated, but the tumor can then use PD-L1 to inhibit the immune system. Durvalumab should help perpetuate the bavituximab-induced immune response, while boosting the opportunity for durvalumab in tumors that are not expressing PD-L1 until the bavituximab treatment. "It's sort of a win-win situation," King said. ...
King said he thinks that bavituximab will likely work with any of the drugs targeting the PD-1 pathway, but decided to work with London-based Astrazeneca rather than one of the FDA-approved drugs, in part, because Astrazeneca was willing to provide the drug free of charge. ”We save probably as much as we're spending because of the cost of these drugs," King said.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=119200003
= = = = = = = = = = = = =
10-15-15: Peregrine & AstraZeneca Expand Collab. w/Ph2/2ndLine-NSCLC Trial, Bavi+durvalumab(MEDI4736), squamous or non-squamous. http://tinyurl.com/q79bkam
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
...Durvalumab=MEDI4736(anti-PD-L1 immune checkpoint inhibitor). AZN’s Head/I-O(Robert Iannone): “Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
Q. Phase II Bavi+durvalumab(anti-PD-L1 MEDI4736), 2ndLine NSCLC(squa+nonsq.), randomized, open-lab, n=198, AZN Collab.)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT02673814 <=added 2-2-16
Est. Enrollment: 198, Study Start: Feb. 2016
Est.Prim.Completion: Apr.2017 (Final data coll. for prim.outcome), Est.StudyCompletion: Apr.2018
2-4-16 Annals of Surgical Oncology: Bavi+Sorafenib vs. Liver Cancer/HCC - murine xenograft tests and Ph.1 human data from Dr. Yopp's UTSW IST trial...
2-4-16: “Antibody-Mediated Blockade of Phosphatidylserine Enhances the Antitumor Effect of Sorafenib in Hepatocellular Carcinomas Xenografts”
Annals of Surgical Oncology - Translational Research & Biomarkers (pp 1-9)
Xiaoyun Cheng, Li Li, Philip E. Thorpe, Adam C. Yopp, Rolf A. Brekken, Xianming Huang
http://link.springer.com/article/10.1245%2Fs10434-016-5107-5
ABSTRACT - Background
Currently, the only FDA-approved systemic therapy for hepatocellular carcinoma (HCC) is the multi-receptor tyrosine kinase inhibitor, sorafenib, which provides only modest clinical benefit. We recently showed that treatment with a phosphatidylserine (PS) targeting agent suppresses tumor growth by targeting tumor vasculature & reactivating antitumor immunity.
METHODS
We tested the hypothesis that sorafenib increases PS exposure on tumor vasculature, thereby enhancing the antitumor efficacy of PS targeting. We evaluated the efficacy of combining a PS targeting agent (2aG4) with sorafenib in murine xenograft models of human HCC.
RESULTS
Our results demonstrate that combination of 2aG4 & sorafenib had a superior therapeutic effect over single agent therapy. Mechanistic studies showed that sorafenib significantly increased PS exposure on tumor vasculature; the percentage of PS-positive vessels increased from 19 to 52, 23 to 68, and 30 to 55 % in PLC/PRF/5, C3A, and Huh7 tumors, respectively. Combination therapy significantly decreased tumor microvessel density and the level of M2 macrophages, while increasing the apoptotic index of tumor endothelial cells and the frequency of M1 macrophages. Furthermore, we report the findings of a Phase I clinical study of bavituximab, a chimeric version of 2aG4, combined with sorafenib in HCC patients. The Phase I results demonstrate the appropriate dose of bavituximab to be given with sorafenib in future clinical trials.
CONCLUSIONS
Overall, these results strongly support the combination of bavituximab with sorafenib as a promising systemic therapeutic strategy for the treatment for advanced HCC patients.
--------
Xiaoyun Cheng & Li Li have contributed equally to this work.
Rolf Brekken & Xianming Huang are consultants to, have equity interest in, and have a sponsored research agreement with Peregrine Pharmaceuticals Inc.
= = = = = = = = = = = = = = = = = = = = = = = =
H. 1st Investigator-Sponsored (IST) Ph.I/II Trial (Bavi+Sorafenib vs. Liver Cancer/HCC, open-label, n=9+38=47)
...Note: Sorafenib = Onyx/Bayer's Nexavar - see http://www.nexavar.com
Protocol: http://clinicaltrials.gov/ct2/show/NCT01264705 UTSW: http://tinyurl.com/mwdc2ql ; (5 sites: 3/UTSW, Parkland-Hosp, Dallas/VA, PI=Dr. Adam Yopp)
2-4-16/Annals.Surg.Onco.(Brekken/Thorpe/Yopp/Xianming) article: Murine xenograft & Ph.1/human data http://tinyurl.com/gtk2qg8
...”Bavi+sorafenib combo had a superior therapeutic effect over single agent therapy.”
3-25-15: Dr. Adam Yopp (UTSW) Oral-pres./SSO (Liver-IST/Ph2-data) http://tinyurl.com/opkh5qy N=38(79%HepC, ECOG/0=34%): MTTP=6.7mos, MOS=6.1, MDSS=8.7, DCR=58%
=>Dr.Yopp's PDF: http://www.peregrineinc.com/images/stories/pdfs/yopp_sso_03-28-2015.pdf
1-16-15 ASCO Gastro-Symposium: Ph.2 data/n=38 (Adam Yopp), ”These clinical outcomes of TTP=6.7/DCR=58%/PFS=4mo are quite encouraging…” http://tinyurl.com/m9uz9mo
11-8-14 SITC'14: Ph.2 Correlative Studies data (biopsies B4/After) on 6pts, incl. KOL Dimitry Gabrilovich’s comments: http://tinyurl.com/pchzr6h
9-9-14 Enrollment complete. http://tinyurl.com/ktrfswj ; (ph2=38 Ph1=9)
4-4-12 AACR'12: Dr. Adam Yopp, ”promising safety profile to-date" http://tinyurl.com/7yrwqm7 (see #5591)
Feb'12-Sep'14 10+ times: CEO Steve King hints of future ex-US partner-driven Bavi+Sorafenib/LIVER trial in Asia-Pacific: http://tinyurl.com/nkaxtcc
......Articles & Data describe Liver Cancer challenges in Asian populations: http://tinyurl.com/7z7o8j9 & http://tinyurl.com/7z99cy4
12-1-10: PPHM's 1st IST (Liver Cancer) initiated at UTSW, ~56 patients - http://tinyurl.com/3xd3e6c
…Per S.King, 5-18-10/R&R, "We've had a lot of interest in running clinical trials with the compound from investigators who have either had prior experience with the drug or would like to study the drug in various settings. Potential IST indications include all the major solid tumor types. Of particular interest is Liver Cancer, in which we have a natural tie-in with our HCV program, Ovarian Cancer and Pancreatic Cancer, also very nicely supported by the prior data.”
SUNRISE at >90%: they have Both Lookins & Topline Data/Unblinding covered. Since Unblinding & Topline-Data at 80% of Events(Deaths, 582x.80=466), I take it that further/new Patients enrolled at 91%+ won't contribute to those since most will fortunately survive beyond when those 3 points are tallied. And, there's the transition of SUNRISE sites to the New AZN Collab. Ph2 Bavi+durvalumab NSCLC trial… These 12-10-15/CC comments by SK & JS explain much...
12-10-15/CC: “Sunrise >90% enrolled; sufficient to allow the 2 planned interim looks (33%/50%) & final readout based on PrimEndPt=OS” http://tinyurl.com/jkp885g
12-10-15/CC: CEO STEVE KING – OPENING COMMENTS: http://tinyurl.com/jkp885g
“...today’s development discussions will focus on these efforts, including upcoming completion enrollment in the SUNRISE trial which is evaluating a chemotherapy combination, and, as SUNRISE wraps up, our plans for a smooth transition of the key SUNRISE clinical sites directly to the next Phase II lung cancer trial evaluating an IO combination [init Q1/2016: Phase2, Bavi+durvalumab(AZN's anti-PD-L1 MEDI4736), 2ndLine NSCLC, squam+nonsquam (randomized, open-label, ~200pts)]. In addition, we are expanding our potential cancer indications through initiation of a Phase II/III metastatic breast cancer study. All of this while continuing to work through several other clinical trial concepts actively under development or initiation in the new year. On the development front, I’m pleased to report today that we are nearing completion of enrollment in the cornerstone of our bavituximab development strategy, our Phase III SUNRISE trial. In fact, with over 90% of the expected enrollment complete, we currently have sufficient patient enrollment based on the assumptions of the study to allow the trial’s planned interim evaluations and final readout based on the primary endpoint of overall survival. Having said that, we do expect to complete enrollment of at least the pre-specified 582 patients over the coming weeks. At this point, the next big milestones really are the interim data analysis from the study, expected to take place during early & mid 2016, with trial unblinding expected toward the end of 2016.”
12-10-15.CC: JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS: http://tinyurl.com/jkp885g
“I’d like to start by quickly addressing our Phase III SUNRISE trial, which is evaluating the use of bavituximab & docetaxel in patients with previously treated locally advanced or metastatic non-squamous NSCLC. As Steve stated, we have already enrolled the number of patients required to achieve the trial’s main objectives, and expect to complete enrollment of the target sample size of 582 in the coming weeks. The next milestones are the interim analyses that will be conducted when 33% & 50% of the targeted number of deaths are reached. While these are event driven, it is our expectation that the 1st interim analysis will read out in early 2016 and the 2nd interim analysis around mid-2016. The final analysis [1-18-16/SK/Noble: “Unblinding at 80% of Events” http://tinyurl.com/j9dkekm ], which will trigger study unblinding, is currently projected to occur at the end of 2016. With the SUNRISE enrollment nearing completion, the Peregrine clinical team is shifting focus to a number of new clinical projects, including those just referenced by Steve. In each case, our goal is to generate clinical evidence of bavituximab’s ability to improve patient outcomes when combined with chemotherapy and immuno-oncology agents. With this goal in mind, we are very pleased to be collaborating with AstraZeneca. Through this partnership, we will be conducting 2 clinical trials, both of which will be initiated in 2016. One trial, which we expect to initiate early 2016, is a global randomized Phase II study in approx. 200 patients with previously treated NSCLC. This trial will evaluate the combination of bavituximab and AZ’s anti-PD-L1 immune checkpoint inhibitor durvalumab, or MEDI4736. As part of this combination trial, patients will also be evaluated retrospectively for the correlation between their PD-L1 levels & clinical outcomes. As the remaining patients are enrolled into SUNRISE, we have already begin laying the groundwork to quickly initiate this new Phase II combination study at a number of our most active sites participating in SUNRISE. These investigators are very familiar with bavituximab and have access to the appropriate patient populations, and we believe this experience will greatly benefit our new study.”
.
.
SKing/8:50 Slide16 (1-18-16): “Unblinding at 80% of Events” http://tinyurl.com/j9dkekm

PGG76109, RRDog reposted his post after realizing he had a blank line at the top, ie no subject. It's out there as #251028:
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=120195918
(Delete)
Ao q/e10-31-15, HALO at 56%, other U.S. custs at 41%. (the q/e 7-31-15 split was 84%-15%).
Avid's New Customer – hints from 9/2014 on – definitely hit the revenue stream in FY16Q2(qe 10-31-15). We do not know who this new customer is at this point. ...And on 12-10-15, Rob Garnick disclosed, “We are already contemplating our options to increase further our mfg. capacity [beyond Myford=Avid II]”, due to ”what appears to be a growing opportunity in this important area of our business.”
Q/E10-31-15 Q/E7-31-15 FY4-30-15 FY4-30-14
Halozyme 56% 84% 79% 91%
Cust-A U.S. 41% 15% 12% 1%
Other Custs 3% 1% 9% 8%
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*

(DELETE) Cp, my #250776 gave me the biggest kick out of any I've ever made I think, because, thanks to SK's adding Slide11 to 1-16-18/Nobel, for THE FIRST TIME I've been able to understand exactly WHY our Direct PS-Targeting method is so superior to the others that have to go after all the little indiv. receptors on PS. ...All those Axl MerTIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. receptor-targeting substances I've read about over the years. It was just a very uplifting thing for me to FINALLY UNDERSTAND!!
SKing/1-18-16 explains PPHM's PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others, like: Axl MerTIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc etc. (maybe why Dr. Raymond Birge is spending lots of time with Dr.Brekken & Peregrine these days?? => http://tinyurl.com/j3whbx3 )
CEO S.King/1-18-16/NobleConf Slide11 7:54: “It turns out that the PS receptors are very well studied, and a number of companies are going after the individual receptors [ex: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8...], depicted on Slide11 as the little pacman symbols. But, the bottom-line is that other companies are going after the individual receptors – our approach is to go after the ligand itself, PS (phosphatidylserine). So, effectively by blocking PS with a good safety profile, we're now able to affect the entire PS-signaling pathway, and we think that's a real advantage over other approaches, since basically we can simultaneously impact the ability of PS to shutdown the immune system.”

1-18-16: CEO Steve King's 1-18-16 presentation at Noble-Financial's Investor Conf. (21min replay, 31 slides) http://tinyurl.com/j9dkekm
- - - - - - - - - - - - - - - - - - - -
I ASKED A SCIENTIST FRIEND TO EXPLAIN THIS FURTHER – HIS 1-28-16 REPLY:
“Look at SK's Slide11 at Noble/1-18-16. The apoptotic or tumor cells, also the blood vessel cells of tumors, have PS expressed on their surface, this is the ligand. All those other cells, macrophages, dentritic cells etc, have receptors that bind the ligand (PS) on their surface. So if you have an antibody (bavi) which binds to PS it blocks the binding of PS with ANY of those receptors on the cells on which those receptors are expressed. The alternative is to develop antibodies which bind to each of those receptors, so you would need to develop a lot of different antibodies to accomplish the same result, i.e. blocking the ligation (binding) of PS to all the receptors. This is also true for receptors that don't bind to PS directly. For example, the receptor MerTK binds to the protein Gas6 (its ligand), not to PS, but Gas6 in turn binds with PS on apoptotic cells. So you can block the activation of MerTK by blocking PS with bavi so that Gas6 can't bind to PS and complete the chain with MerTK.”
- - - - - - - - - - - - - - - - - - - -
BY MASSHYSTERIA 1-29-16 IHUB #250754
“...evidence is strongly mounting support your comments from complimentary sources. In a type of concurrence with importance of PS blockade - except across its receptors on the T-Cell side of things. Many are aware that TIM-3 & LAG-3 are 2 of several checkpoints that bind to PS. Not sure how many are aware that the rate of clinical trials evaluating these two checkpoint inhibitors is rising, and that multiple smaller scale trials are showing great results using that combo. That is driving the greater investment and expansion of trials. Why is that a good thing? All these combo trials are conducted by large pharma companies who are seeing supporting signals to increase investment. Peregrine holds the molecule that achieves the complimentary results (blocking at PS site) holistically and without having to chase a multitude of checkpoints that bind to PS to stop/reduce immune response. BP's excitement about TIM-3 and LAG-3 should be a very good cause for upbeat expectations from bavituximab, and a likely reason that world-class institutions and AZN are in the game. The beauty is having a single solution that blocks PS, vs. possibly dozens that block the several other T-Cell checkpoints with which it binds. Commercially, clinically and logistically, there is no better winner in this competition if one can do the work of many. Provided this mechanism works and is shown to do what so many diverse professionals seem to be suggesting - its becoming a game of timing and finesse. The more good news I hear from Industry on TIM-3 & LAG-3, the happier I get. Definitely happier now than 12mos ago.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=120163782
= = = = = = = = = = = = = = =ARTICLES:
11-2014: “Phosphatidylserine receptors: Enhancers of Enveloped Virus Entry & Infection”
http://www.sciencedirect.com/science/article/pii/S0042682214004206
“Six PVEERs have been identified to date: TIM-1, TIM-4, Gas6 or Protein S/Axl, Mer, and Tyro3, and MFG-E8/integrin avB3 & avB5. “
9-2015/Mucosal Immunology: “The Axl Receptor Tyrosine Kinase is a Discriminator of Macrophage Function in the Inflamed Lung”
http://www.nature.com/mi/journal/v8/n5/full/mi2014129a.html
“The TAM receptor family of receptor tyrosine kinases (Tyro3, Axl and MerTK) mediates the non-inflammatory removal of apoptotic cells by phagocytes through the bridging phosphatidylserine-binding molecules growth arrest-specific 6 (Gas6) or Protein S.”
.
.
.


11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
BAVI MOA 8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow http://tinyurl.com/qz64pzg
…“Expansion & Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
Funny Youtube by Immunotherapy-World-Conf./WashDC re: The Storm.
We were Ready for a Nice Sunny Meeting in Washington.
...AND THEN…
We're right in the middle of The Blizzard of 2016…
BUT WHY LET A LITTLE SNOW SPOIL THE MEETING?
- - - - - -
Thank You to Everyone Who Made It.
- - - - - -
And, See You Next Year in... MIAMI !!
SEE YOU NEXT YEAR!!!
.
.
= = = = = = = = = = = = =
Jan25-27 2016: “Phacilitate's Immunotherapy World Conf.”, WashDC
http://www.immunotherapyforum.com
“After years of posting impressive clinical results and pushing analyst projections to greater and greater heights, immuno-oncology has solidified its status as the most exciting development in cancer treatment for a long time. Now that checkpoint inhibitors have uncloaked cancer cells, drug developers are investigating dozens of immune drug cocktails (everything from CAR-T to antibodies, vaccines and other cellular therapies) to find well-tolerated treatments which could become the first-in-line to fight cancer in patients. We have all heard of the stunning breakthroughs at ASCO'15. BMS’ Opdivo + Yervoy study which lead to a 58% shrinkage of tumors and a PFS superiority of 8.6mos. against monotherapies in patients with melanoma. AstraZeneca’s own PD-L1 (MEDI4736/durvalumab) [see 10-15-15 AZN/PPHM Collab. Expansion: http://tinyurl.com/q79bkam ] and CTLA-4 (Tremelimumab) inhibitor combination, which demonstrated an ORR of 27% in the treatment of NSCLC… On Jan25-27 2016, 750 deal-makers will gather in the Grand Hyatt Hotel to discuss the direction, future and value of cell, gene and immunotherapy portfolios.”
1-24-16 Agenda Update due to storm – everything moved back ~30mins:
http://www.immunotherapyforum.com/files/new_immunotherapy_agenda.pdf
---------
Jan25(Day1): Focus Session1, “Cellular Cancer Immunotherapy: Optimizing Combination Therapy Development Strategy”
3:15pm: Chair's intro, David Lebwohl, NOVARTIS
3:20: Michael Hanna, VACCINOGEN, “The Provocative Issues of Tumor Heterogeneity on Active Specific Immunotherapy...”
3:35: Laura Benjamin, ELI LILLY, “Challenges/Opportunities for Translational Science to Guide Angiogenesis/Combos w/Immunotherapy”
3:50: Taylor Schreiber, HEAT BIOLOGICS, “Dual-Acting Immunotherapy with ComPACT - Vaccination & Co-Stimulation...”
-------
1-25-16 4:05-4:20pm: Jeff T. Hutchins (PEREGRINE'S VP/PreClinRes), “Combination Immunotherapies - Opening the Gate: Increasing Tumor Infiltrating Activated T-cells to Optimize and Expand the Benefits of Immune Checkpoint Therapies”
* Analyzing complimentary mechanisms in therapeutics and building solid partnerships
* Utilizing scientifically-driven data to build a product value story
* Demonstrating how a product fits within a treatment in combination(s) and against competitive therapies
-------
4:20pm: Robert Preti, PCT(CALADRIUS), “What Does Commercialization Look Like?”

- - - - - - - - - - - - - - - -From PPHM's 1-20-16 PR: http://tinyurl.com/z65avfw
“In his talk, Dr. Hutchins will discuss strategies for expanding the therapeutic benefit seen with immuno-oncology monotherapies to a broader range of patients using combination treatment approaches. Specifically, he will highlight the strategy of leveraging treatments capable of increasing the number & activity of T-cells in the tumor microenvironment to optimize the therapeutic benefit of immune checkpoint inhibitors such as anti-PD-1/anti-PDL-1 agents. Dr. Hutchins will draw on the company's experience in working with preclinical equivalents of bavituximab, Peregrine's lead investigational phosphatidylserine (PS)-targeting immunotherapy candidate. PS-targeting antibodies have been shown to shift the immunosuppressive functions of immune cells in tumors, resulting in anti-tumor immune responses. Peregrine has generated results from multiple preclinical & clinical-translational studies demonstrating enhanced anti-tumor activity and immune activation when combining equivalent PS-targeting antibodies with conventional chemotherapy or checkpoint inhibitors such as anti-PD-1 agents.”
CFH, I def. think MSKCC's working with Betabodies too - otherwise they would have just said "Bavi" instead of "our PS-Targeting Platform, incl Bavi". Again, Betabodies are "2nd Gen. Bavi". At some point down the line, I trust & hope they'll be a 3rd gen. too - all part of moving the science forward!
= = = = = = = = =
5-29-15: Sloan-Kettering's Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Wolchok.
http://tinyurl.com/qxu4w2x
MSKCC Collab: “PS-Targeting agents (incl. Bavi)” mentioned over & over in the 5-29-15 PPHM/MSKCC Collab. PR, by Mem.Sloan's Drs. Jedd Wolchok & Taha Merghoub, and PPHM's Dr. Hutchins. CEO S.King refers to “our PS-targeting Platform”...
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/qxu4w2x
No doubt BETABODIES are part of the expanson of the PS-Targeting Platform beyond Bavi:
BETABODIES (ex: KL15), “potentially the next generation of PS-targeting cancer therapeutics” - generated by fusing domains of the PS-binding the plasma protein, B2-glycoprotein I (B2GPI), to the Fc region of mouse IgG2a. Such ‘betabodies’ potentially have the following advantages: they bind directly to PS and do not require a cofactor protein (B2GPI) for binding; they can be made fully human; they are smaller in size (100 vs. 250 KDa); and they have slower blood clearance rates (half-life of ~5days vs. Bavi’s ~1day).
…See 7-6-13, “Entdoc, here’s a bunch of stuff on Betabodies” http://investorshub.advfn.com/boards/read_msg.aspx?message_id=89680206http://tinyurl.com/khopa3d
BETABODIES PATENT, USPTO Patent #8,956,616: http://tinyurl.com/nj4jpry (U.S. Granted 2-17-15)
Maybe even PGN632, the Mab that 1st showed up in the Duke HIV/Collab:
• PGN632 (B2GPI-indep.) is the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p
1-18-16: Steve King, Noble-Financial's Investor Conf. (21min replay, 31 slides) http://tinyurl.com/j9dkekm

Awesome presentation by SK imo – extremely confident!
1-18-16: CEO Steve King's 1-18-16 presentation at Noble-Financial's Investor Conf. (21min replay, 31 slides) http://tinyurl.com/j9dkekm

.
.

.
.

CEO Steve King's 1-18-16 presentation @Noble-Financial's Investor Conf. - 21min. video replay and 31 slides captured...
Jan17-20 2016: NobleCon12 - Noble Financial's 12th Annual Emerging Growth Investor Conf., Port St. Lucie, FL
1-18-16/SK: “Delivering Innovative & Exceptional Biopharmaceutical Products to Improve Patient Lives”
REPLAY/21min. Video & 31 Slides (CEO Steve King): http://tinyurl.com/jr4vr36














SKing/8:50 Slide16 (1-18-16): “Unblinding at 80% of Events”






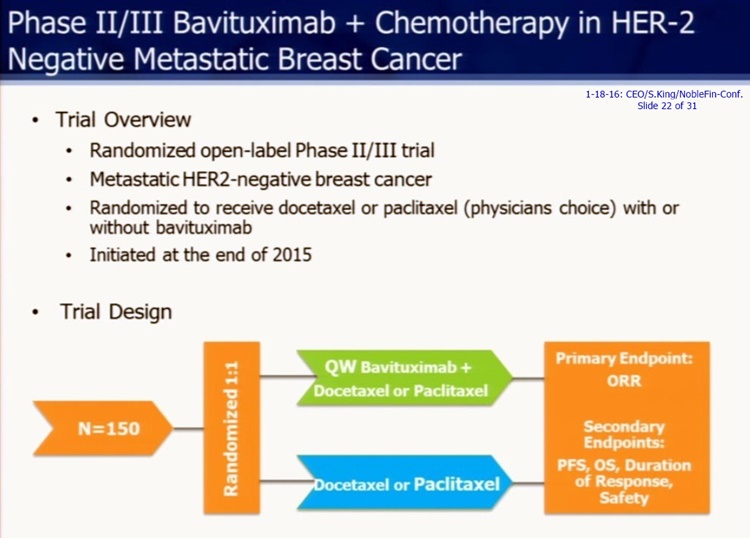









= = = = = = = = = = = = = = = = = = = = = = = = = = =
1-11-16/PR: Update on 4 New Bavi Clinical Trials (Lung/AZN, Breast/1Co./1MSKCC, Other Cancers/AZN). SUNRISE estimates: Interim1=Early'16, Interim2=Mid'16, FinalUnblinding=End'16… http://tinyurl.com/zhdy37a
1-6-16: Peregrine enters into Research Collab. with Natl-Comprehensive-Cancer-Network (NCCN) http://tinyurl.com/zmxtpsb
...$2mm res. grant to NCCN's Oncology Res. Pgm (ORP), will “significantly expand our clinical evaluation of Bavi and augment Peregrine's IST pgm at 26 of the world's leading cancer centers”.
12-10-15 Qtly. Conf. Call (King/Shan/Worsley/Garnick/Lytle) Transcript http://tinyurl.com/jkp885g
...CEO SK: “Although our SUNRISE enrollment milestone has been reached, we have no intention of slowing down, quite the opposite. We are aggressively moving to initiate new clinical trials [Lung, Breast, Mult-Types] that will allow us to build the most robust oncology business possible… With each of these studies our goal is the same - we are committed to identifying key indications, patient populations, and therapeutics that can benefit from combination treatment with bavituximab. From what we have seen to-date, the opportunity appears vast and we are hard at work converting the most promising prospects into true value.”
10-15-15 Peregrine's ASM: ATTENDEE Reports & Link to SK's 18min/16slide webcast: http://tinyurl.com/o6z4bm4
10-15-15: Peregrine & AstraZeneca Expand Collab. w/Ph2/2ndLine-NSCLC Trial, Bavi+durvalumab(MEDI4736), squamous or non-squamous. http://tinyurl.com/q79bkam
9-9-15 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/ph22vdn
...CEO S.King: “The Memorial Sloan Kettering & AstraZeneca collaborations are an important part of our announced plans to expand our bavituximab clinical pgm.”
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
...Durvalumab=MEDI4736(anti-PD-L1 immune checkpoint inhibitor). AZN’s Head/I-O(Robert Iannone): “Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
7-14-15 Qtly. Conf. Call (King/Shan/Hutchins/Lytle) Transcript http://tinyurl.com/nw2v5u6
...CEO S.King: “We recently entered into collaboration with investigators at Memorial Sloan Kettering Cancer Ctr to continue expanding on this important work, as well as to explore other potential applications of bavituximab and other agents that target PS-signaling pathway.”
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/qxu4w2x
= = = = = = = = = = = = = = = = =RECALL:
12-10-15 Qtly. Conf. Call (King/Shan/Worsley/Garnick/Lytle) Transcript http://tinyurl.com/jkp885g
Q&A:
3. Rahul Jasuja - Noble Life Science Partners http://noblelsp.com/research
RJ: ”In planning your combination studies going forward, we are looking at PD-L1 low tumors because they are likely to be non-responsive to anti-PD-1's, like in Keytruda & Opdivo. Is that the only the rational, or is it also likely that PD-L1 low tumors also more responsive to bavituximab?”
Steve King: That’s what we want to show out in some of these studies we’re starting. We’re not planning on selecting for PD-L1 neg. patients in the Phase II study or the initial combination of bavi with chemo & durvalumab, but rather taking all-comers and then doing subset analysis and determining which patient populations we’re having the biggest impact in. Because, based on our translational data that’s been presented this year at ESMO & SITC'15 [11-9-15/SITC: http://tinyurl.com/pbof95w ], we’ve shown that we can take PD-L1 neg. tumors and actually elicit an immune response in those tumors. That’s the reason that we think that we have the potential to turn those into better responders on a PD-1 or PD-L1 therapy. As I mentioned during the prepared remarks it’s basically, “use bavi to get the immune response going and then use durvalumab to keep it going.” That's the reason there is a great scientific rationale right now for why we may have the biggest impact in those patients who don’t do well, because the delta between how they would normally do and how they might do with bavituximab may be the largest. We’re going to have some great insight into that from the studies we’re planning on running and the ability to go in and do subset analysis. Joe, do you want to add any more to that?
Joe Shan: I was going to use the word delta, but you beat me to it. I think in the PD-L1 neg. patient you have more opportunities to demonstrate the delta.
RJ: ”One of the concepts that’s evolving pretty rapidly is that you’ve got the TILs- & TILs+, and TILs+ are responding to PD-1 checkpoint immunotherapy. So is it fair to say, or is it an extrapolation, that PD-L1 positive tumors are more likely the ones that are TILs+? And in your case are you also seeing that TILs- tumors are probably the ones that are going to respond to chemo combination therapy better than the other ones that are the non-responders in combination with PS you can make them responders.”
Steve King: That’s certainly what our evidence has shown so far. The general assumption is that TILs- or TILs/Low tumors the ones that have low levels of the need for PD-L1, right, that’s really meant to stop an ongoing immune response. I think that’s generally true. Now it gets a little bit more complex because you’ve got Tregs and all kinds of T-cells present inside the tumor, so it also depends on the particular makeup. What we know is that when you get bavituximab, we seem to see a nice change in the levels of MDSCs (Myeloid-Derived Suppressor Cells) who are really the cell type that’s responsible for controlling the immune response in the tumor. It's been shown that patients with high levels of MDSCs have very poor prognosis. So as much as probably getting new TILs into the tumors is important, it’s probably more important to change the makeup of those cells into a more productive immune response positive phenotype. That’s exactly what we see with when you get bavituximab treatment; when we take a look a look at our translational data it shows an increase in CD4+ T-Cells and an increase in CD8+ T-Cells along with the changes in the suppressive cells types and the expression of immuno-suppressor cytokines, in which both decrease after treatment. So, it’s a matter of getting everything moving in the right direction. Again this is the role we think PS plays, by blocking it and activating an immune response, we're able to turn that around.
RJ: ”That does make sense. I think you’re looking at patient selection as being helpful in defining the population as well as the data you showedat SITC'15 [11-9-15/Duke's Herbert K. Lyerly http://tinyurl.com/pbof95w ], where you talked about immuno-profiling and looking at response to bavituximab, those are very interesting datasets. The other question I have is, there are a couple of trials running that are ISTs [at UTSW] - one of them is bavi in combination with Yervoy(ipilimumab) in melanoma, and the other one is a rectal cancer IST. Any updates on those?”
Steve King: The Rectal adenocarcinoma, we expect to have some data coming up this year from that study. As in any IST, it’s always a bit difficult, we do whatever we can to encourage them. But we do expect data to be becoming out of that study in 2016, which I think will be a real positive because in that study we did have the opportunity to collect pre- and post-treatment biopsies - #1, so we can see what happens following bavituximab treatment, but also that was a combination with radiation which we expect to be a very strong inducer of tumor antigens, which can then be taking advantage of by bavituximab treatment to make CD8+ T-Cells or killer T-Cells. For the Melanoma study, obviously the since that investigator (UTSW) started this study the SOC has changed pretty substantially. So right now, we're trying to work with that investigator, as well as some others, to probably change the profile of that study or start a new multi-center study in which we can then look at little bit more closely at what is the current SOC and make sure it’s an attractive trial for patients. As the treatment options for patient’s change, you need to be able to change with it and luckily it's a small IST trial and we may have an opportunity here to run a trial that I think would be very attractive for patients and will allow us to answer some key questions, because that was really the point of that study was to answer some of the key questions of combining with Yervoy. In addition, we’re starting the combination of durvalumab [AZN collab.] in multiple solid tumor types after the beginning of the year, so one way or another we’ll have lots of information coming from those studies.
RJ: ”Any updates any more color or comments on the collaboration with Sloan Kettering and Jed Wolchok on combination approaches - any data coming that way in the next few months?”
Steve King: That's an ongoing process. We’re very actively working with them to study a new combinations, looking for potential and different indications as well as those different combinations. We fully expect that it will be a fruitful collaboration; there will be a lots of data coming from that that we’ll see a very scientific meetings coming up. We already see a lot of data coming out of all for other collaborators nad we’ve had examples of presentations in multiple conferences this year. It always takes a while for them to get started, but then once they are going you tend to have a lot of data that continuously comes through them because the all the systems are up and running.
RJ:”Great. Thank you and congratulations on the healthy revenues at Avid.”
WOW/BioBS/Missed it: CEO S.King's 1-18-16 presentation at Noble Financial's Emerging Growth Investor Conf.
Jan17-20 2016: NobleCon12 - Noble Financial's 12th Annual Emerging Growth Investor Conf., Port St. Lucie, FL
1-18-16/SK: “Delivering Innovative & Exceptional Biopharmaceutical Products to Improve Patient Lives”
REPLAY/21min. Video & 31 Slides (CEO Steve King) http://tinyurl.com/jr4vr36
= = = = = = =RECALL:
12-10-15 Qtly. Conf. Call (King/Shan/Worsley/Garnick/Lytle) Transcript http://tinyurl.com/jkp885g
Q&A:
3. Rahul Jasuja - Noble Life Science Partners http://noblelsp.com/research
RJ: ”In planning your combination studies going forward, we are looking at PD-L1 low tumors because they are likely to be non-responsive to anti-PD-1's, like in Keytruda & Opdivo. Is that the only the rational, or is it also likely that PD-L1 low tumors also more responsive to bavituximab?”
Steve King: That’s what we want to show out in some of these studies we’re starting. We’re not planning on selecting for PD-L1 neg. patients in the Phase II study or the initial combination of bavi with chemo & durvalumab, but rather taking all-comers and then doing subset analysis and determining which patient populations we’re having the biggest impact in. Because, based on our translational data that’s been presented this year at ESMO & SITC'15 [11-9-15/SITC: http://tinyurl.com/pbof95w ], we’ve shown that we can take PD-L1 neg. tumors and actually elicit an immune response in those tumors. That’s the reason that we think that we have the potential to turn those into better responders on a PD-1 or PD-L1 therapy. As I mentioned during the prepared remarks it’s basically, “use bavi to get the immune response going and then use durvalumab to keep it going.” That's the reason there is a great scientific rationale right now for why we may have the biggest impact in those patients who don’t do well, because the delta between how they would normally do and how they might do with bavituximab may be the largest. We’re going to have some great insight into that from the studies we’re planning on running and the ability to go in and do subset analysis. Joe, do you want to add any more to that?
Joe Shan: I was going to use the word delta, but you beat me to it. I think in the PD-L1 neg. patient you have more opportunities to demonstrate the delta.
RJ: ”One of the concepts that’s evolving pretty rapidly is that you’ve got the TILs- & TILs+, and TILs+ are responding to PD-1 checkpoint immunotherapy. So is it fair to say, or is it an extrapolation, that PD-L1 positive tumors are more likely the ones that are TILs+? And in your case are you also seeing that TILs- tumors are probably the ones that are going to respond to chemo combination therapy better than the other ones that are the non-responders in combination with PS you can make them responders.”
Steve King: That’s certainly what our evidence has shown so far. The general assumption is that TILs- or TILs/Low tumors the ones that have low levels of the need for PD-L1, right, that’s really meant to stop an ongoing immune response. I think that’s generally true. Now it gets a little bit more complex because you’ve got Tregs and all kinds of T-cells present inside the tumor, so it also depends on the particular makeup. What we know is that when you get bavituximab, we seem to see a nice change in the levels of MDSCs (Myeloid-Derived Suppressor Cells) who are really the cell type that’s responsible for controlling the immune response in the tumor. It's been shown that patients with high levels of MDSCs have very poor prognosis. So as much as probably getting new TILs into the tumors is important, it’s probably more important to change the makeup of those cells into a more productive immune response positive phenotype. That’s exactly what we see with when you get bavituximab treatment; when we take a look a look at our translational data it shows an increase in CD4+ T-Cells and an increase in CD8+ T-Cells along with the changes in the suppressive cells types and the expression of immuno-suppressor cytokines, in which both decrease after treatment. So, it’s a matter of getting everything moving in the right direction. Again this is the role we think PS plays, by blocking it and activating an immune response, we're able to turn that around.
RJ: ”That does make sense. I think you’re looking at patient selection as being helpful in defining the population as well as the data you showedat SITC'15 [11-9-15/Duke's Herbert K. Lyerly http://tinyurl.com/pbof95w ], where you talked about immuno-profiling and looking at response to bavituximab, those are very interesting datasets. The other question I have is, there are a couple of trials running that are ISTs [at UTSW] - one of them is bavi in combination with Yervoy(ipilimumab) in melanoma, and the other one is a rectal cancer IST. Any updates on those?”
Steve King: The Rectal adenocarcinoma, we expect to have some data coming up this year from that study. As in any IST, it’s always a bit difficult, we do whatever we can to encourage them. But we do expect data to be becoming out of that study in 2016, which I think will be a real positive because in that study we did have the opportunity to collect pre- and post-treatment biopsies - #1, so we can see what happens following bavituximab treatment, but also that was a combination with radiation which we expect to be a very strong inducer of tumor antigens, which can then be taking advantage of by bavituximab treatment to make CD8+ T-Cells or killer T-Cells. For the Melanoma study, obviously the since that investigator (UTSW) started this study the SOC has changed pretty substantially. So right now, we're trying to work with that investigator, as well as some others, to probably change the profile of that study or start a new multi-center study in which we can then look at little bit more closely at what is the current SOC and make sure it’s an attractive trial for patients. As the treatment options for patient’s change, you need to be able to change with it and luckily it's a small IST trial and we may have an opportunity here to run a trial that I think would be very attractive for patients and will allow us to answer some key questions, because that was really the point of that study was to answer some of the key questions of combining with Yervoy. In addition, we’re starting the combination of durvalumab [AZN collab.] in multiple solid tumor types after the beginning of the year, so one way or another we’ll have lots of information coming from those studies.
RJ: ”Any updates any more color or comments on the collaboration with Sloan Kettering and Jed Wolchok on combination approaches - any data coming that way in the next few months?”
Steve King: That's an ongoing process. We’re very actively working with them to study a new combinations, looking for potential and different indications as well as those different combinations. We fully expect that it will be a fruitful collaboration; there will be a lots of data coming from that that we’ll see a very scientific meetings coming up. We already see a lot of data coming out of all for other collaborators nad we’ve had examples of presentations in multiple conferences this year. It always takes a while for them to get started, but then once they are going you tend to have a lot of data that continuously comes through them because the all the systems are up and running.
RJ:”Great. Thank you and congratulations on the healthy revenues at Avid.”
I still find this Steve King 1-13-16 Cancer-Moonshot-2020 quote in EP-Vantage interesting… Just has a positive tone to it to me, and funny the EP-Vantage writer chose only to write abut Peregrine/SK. I can't tell if “this Cancer MoonShot 2020 coalition might serve to stimulate an expansion of (Peregrine's) clinical work” is just a thought of writer Jonathan Gardner's, or if SK contributed in any way to that positive twist.
1-13-16: “JP Morgan - Jumping In Front Of The Cancer Immunotherapy Parade”
EP Vantage Newsletter Provider
. . .
Steven King, CEO of Peregrine Pharmaceuticals, sees the potential for this (Cancer MoonShot 2020) initiative to offer an opportunity for smaller companies to run more combination trials. While Peregrine is planning a trial in combination of its immuno-oncology agent bavituximab with AstraZeneca’s durvalumab and is collaborating with the National Comprehensive Cancer Network (NCCN) for other combination trials, this coalition might serve to stimulate an expansion of clinical work. “It’s often access to drugs. If you want to run a combination with Keytruda or Opdivo, you’re going to have to buy the drug,” he told EP Vantage in an interview on the sidelines of the JP Morgan meeting. “It becomes a cost issue. You’re not going to be able to run as many studies.”
MORE: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=119836127
June2015: Dr.Raymond.Birge & Dr.Xianming.Huang (Dr.Rolf.Brekken-Lab) at Gordon Res. Conference in Maine – speaking next to each other in “Consequences of Lipid Alterations” Session on 6-15-15. Peregrine was the only corp. contributor to this conference, specified as, “In Memory of Dr. Philip E. Thorpe.”...
June 14-19, 2015: “Apoptotic Cell Recognition & Clearance”, Gordon Research Conference, Biddeford, ME
Chair: David S. Ucker, Vice Chairs: Peter Henson & Kirsten Lauber
http://www.grc.org/programs.aspx?id=13127
”Moreover, apoptotic cells are potently immunosuppressive, and their clearance occurs in the absence of inflammation. Recognition & inflammatory modulation represent key elements of an innate immunity that discriminates live from effete cells.”
**The only Corp. Sponsor: PEREGRINE PHARM, “Generous financial support from Peregrine Pharmaceuticals is in memory of Dr. Philip E. Thorpe.”
- - - - - - - -
SESSION: “Consequences of Lipid Alterations for Apoptotic Cell Clearance
Discussion Leader: Christopher Gregory (Univ. of Edinburgh, UK)
7:30pm: Ian Dransfield (Univ. of Edinburgh, UK), “Protein S Binding to Apoptotic Cells...”
8:10pm: Dr. Raymond Birge (Rutgers NJMS), “Apoptotic Cell Recognition Receptors, Tyro3/Axl/Mer...”
8:50pm: Dr. Xianming Huang** (UTSW-MC/Dallas, Brekken Lab), “PS-Targeting Antibodies Overcome Tumor Immunosuppression and Synergize with Immune Checkpoint Blockade"
**Dr. Xianming Huang: formerly in Dr. Philip Thorpe's Lab (RIP, Dr. Thorpe, 1951-2013 http://tinyurl.com/l9gqmt5 ), now in Dr. Rolf Brekken's (PPHM SAB) Lab.
http://www.utsouthwestern.edu/labs/brekken/
= = = = = = = = = = = = = =
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html
Dr. Raymond Birge spoke at Peregrine's 11-2015 “Sci. Session” at SITC'15/Maryland ( http://tinyurl.com/pbof95w ); will be the chair in June 2016 of the ICDS'16/Ireland session where Dr. Rolf Brekken (PPHM SAB) is speaking on “Blockade of PS & Immune Activation in Cancer”; and now also speaking in late June'16 at ICHC'16/Istanbul on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”.
3-1-14/Youtube: “Cancer Research” - Dr. Raymond Birge Laboratory, Rutgers http://birgelab.org
Futility=Inferiority, Efficacy=Superiority – these are official FDA clinical trial words. For SUNRISE, the 1st-Look-in (33%/Events) will test ONLY for “Futility”(inferiority), meaning the trial will be Stopped IF very bad (statistical) results to date vs. Control. The 2nd-Look-in (50%/Events) will again test for Futility, and also for “Efficacy”(superiority), meaning the trial will be Stopped IF very excellent (statistical) results to date vs. Control. Of course, SAFETY is always looked at by the DMC. These Look-in rules happen to the ones Dr. Garnick & Mr. Shan & Peregrine Team negotiated with the FDA.
= = = = = = = = = = = = = = = = =
3 Types of Interim Analyses: SAFETY, FUTILITY, EFFICACY. (SAFETY being a continuous DMC concern).
During a clinical trial, we can perform interim analysis (or DMC, DSMB review) for 3 different reasons:
Interim analysis for SAFETY:
1) with pre-specified stopping rule (for example stop the trial if we see # of cases of Serious Adverse Events)
2) without pre-specified stopping rule (rely on DMC members to review the overall safety)
Interim analysis for FUTILITY ["INFERIORITY"]: To see if the new treatment is unlikely to beat the control – then stop the trial for futility - this is called ‘futility analysis’.
Interim analysis for EFFICACY ["SUPERIORITY"]: To see if the new treatment is overwhelmingly better than control - then stop the trial for efficacy.
In situations 2 & 3, the criteria for stopping rule for efficacy could be different from the stopping rule for futility, but need to be pre-specified.
More: http://onbiostatistics.blogspot.com/2012/03/futility-analysis-in-clinical-trials.html
= = = = = = =
6-1-15 PR: BAVI CLINICAL TRIALS EXPANSION http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=915699
SUNRISE Phase III Pivotal Trial
The company's Phase III SUNRISE (Stimulating ImmUne RespoNse thRough BavItuximab in a PhaSE III Lung Cancer Study) is an approximately 600 patient trial that continues to enroll at over 150 sites worldwide. Completion of enrollment is anticipated by calendar year-end. This Phase III, randomized, double-blind, placebo-controlled clinical trial is designed to evaluate the safety, tolerability and efficacy of bavituximab as a second-line treatment in patients with non-squamous NSCLC. Enrollment is proceeding according to plan with 2 planned interim efficacy analyses which will be reviewed by the trial's Independent Data Monitoring Committee (IDMC). The 1st interim analysis, which will be conducted when 33% of the targeted overall survival events are reached, is for futility and the 2nd interim analysis, for futility or superiority [Superiority=EFFICACY], will be conducted at 50% of events. As these analyses are event driven, the exact timing of each is unknown, however the company plans to provide updates as these events are reached. For additional information about the SUNRISE trial, please visit http://www.sunrisetrial.com or ClinicalTrials.gov using the Identifier NCT01999673. => http://www.clinicaltrials.gov/ct2/show/NCT01999673
12-10-15: “Sunrise >90% enrolled; sufficient to allow the 2 planned interim looks (33%/50%) and final readout based on PrimEndPt=OS”
“The next milestones are the interim analyses that will be conducted when 33% & 50% of the targeted number of deaths are reached. While these are event driven, it is our expectation that the 1st interim analysis will read out in early 2016 and the 2nd interim analysis around mid-2016. The final analysis, which will trigger study unblinding, is currently projected to occur at the end of 2016.
http://tinyurl.com/jkp885g
Immunotherapy World/WashDC(Dr.Hutchins) delayed by 1day =>TUE.

PPHM's Jeff Hutchins(VP/PreClinRes) speaking 1-25-16 (<=DELAYED 1 DAY!) at Immunotherapy World Conf. (WashDC)
“750 deal-makers will gather to discuss the direction, future and value of cell, gene and immunotherapy portfolios.”
Jan25-27 2016: “Phacilitate's Immunotherapy World Conf.”, WashDC
http://www.immunotherapyforum.com
“After years of posting impressive clinical results and pushing analyst projections to greater and greater heights, immuno-oncology has solidified its status as the most exciting development in cancer treatment for a long time. Now that checkpoint inhibitors have uncloaked cancer cells, drug developers are investigating dozens of immune drug cocktails (everything from CAR-T to antibodies, vaccines and other cellular therapies) to find well-tolerated treatments which could become the first-in-line to fight cancer in patients. We have all heard of the stunning breakthroughs at ASCO'15. BMS’ Opdivo + Yervoy study which lead to a 58% shrinkage of tumors and a PFS superiority of 8.6mos. against monotherapies in patients with melanoma. AstraZeneca’s own PD-L1 (MEDI4736/durvalumab) [see 10-15-15 AZN/PPHM Collab. Expansion: http://tinyurl.com/q79bkam ] and CTLA-4 (Tremelimumab) inhibitor combination, which demonstrated an ORR of 27% in the treatment of NSCLC… On Jan25-27 2016, 750 deal-makers will gather in the Grand Hyatt Hotel to discuss the direction, future and value of cell, gene and immunotherapy portfolios.”
CONF. DELAYED by ONE DAY DUE TO STORM:
http://www.immunotherapyforum.com/files/new_immunotherapy_agenda.pdf
---------
Jan25(Day1): Focus Session1, “Cellular Cancer Immunotherapy: Optimizing Combination Therapy Development Strategy”
3:15pm: Chair's intro, David Lebwohl, NOVARTIS
3:20: Michael Hanna, VACCINOGEN, “The Provocative Issues of Tumor Heterogeneity on Active Specific Immunotherapy...”
3:35: Laura Benjamin, ELI LILLY, “Challenges/Opportunities for Translational Science to Guide Angiogenesis/Combos w/Immunotherapy”
3:50: Taylor Schreiber, HEAT BIOLOGICS, “Dual-Acting Immunotherapy with ComPACT - Vaccination & Co-Stimulation...”
-------
1-25-16 4:05-4:20pm: Jeff T. Hutchins (PEREGRINE'S VP/PreClinRes), “Combination Immunotherapies - Opening the Gate: Increasing Tumor Infiltrating Activated T-cells to Optimize and Expand the Benefits of Immune Checkpoint Therapies”
* Analyzing complimentary mechanisms in therapeutics and building solid partnerships
* Utilizing scientifically-driven data to build a product value story
* Demonstrating how a product fits within a treatment in combination(s) and against competitive therapies
-------
4:20pm: Robert Preti, PCT(CALADRIUS), “What Does Commercialization Look Like?”

- - - - - - - - - - - - - - - -From PPHM's 1-20-16 PR: http://tinyurl.com/z65avfw
“In his talk, Dr. Hutchins will discuss strategies for expanding the therapeutic benefit seen with immuno-oncology monotherapies to a broader range of patients using combination treatment approaches. Specifically, he will highlight the strategy of leveraging treatments capable of increasing the number & activity of T-cells in the tumor microenvironment to optimize the therapeutic benefit of immune checkpoint inhibitors such as anti-PD-1/anti-PDL-1 agents. Dr. Hutchins will draw on the company's experience in working with preclinical equivalents of bavituximab, Peregrine's lead investigational phosphatidylserine (PS)-targeting immunotherapy candidate. PS-targeting antibodies have been shown to shift the immunosuppressive functions of immune cells in tumors, resulting in anti-tumor immune responses. Peregrine has generated results from multiple preclinical & clinical-translational studies demonstrating enhanced anti-tumor activity and immune activation when combining equivalent PS-targeting antibodies with conventional chemotherapy or checkpoint inhibitors such as anti-PD-1 agents.”
= = = = = = = = = = = = = = = = = = = =
Bruce Freimark (Dir/PreClin-Oncology) speaking 1-26-16 at GTCbio's Novel-Immunotherapeutics-Summit, SanDiego
Jan25-26 2016: “GTCbio's Novel Immunotherapeutics Summit”, San Diego
https://www.gtcbio.com/conferences/immunotherapeutics-summit-overview
GTC = Global Technology Community https://www.gtcbio.com/about-us
4 Sub-Conferences, including: 8TH IMMUNOTHERAPEUTICS & IMMUNOMONITORING
1-26-15: SESSION: ”Combinatorial Immunotherapies” - Moderator: Bruce Freimark, Peregrine Pharm.
4 Speakers:
*9:35am “DNA Vaccines with T-Cell Checkpoint Blockade”, Douglas McNeel, UNIV./WISCONSIN
*10:00am “Blockade of Phosphatidylserine Enhances the Anti-Tumor Activity of Targeted Therapy & Immune Checkpoint Inhibitors by Reducing Immunosuppressive Cells in the Tumor Microenvironment”, Bruce Freimark (Res.Dir./Preclin.Oncology), PEREGRINE PHARMACEUTICALS
Abstract: The underlying cause for the failure of immune checkpoint blockade is persistent, multifocal immune suppression in the tumor microenvironment. This is due to the absence of pre-existing antitumor Teff because of the action of immune checkpoints that induce immunosuppressive cytokines and recruit tumor infiltrating myeloid-derived suppressor cells (MDSCs), regulatory T cells (Treg), and M2 macrophages. Phosphatidylserine (PS) is segregated to the inner leaflet of the plasma membrane but becomes externalized to the outer leaflet in tumor cells where it contributes as an upstream checkpoint inhibitor to an immunosuppressive environment. Antibody blockade of PS reprograms the immune cells in the tumor microenvironment to support immune activation. Antibody-mediated PS blockade reduces the levels of MDSCs, TGF-beta, and IL-10 and increases the levels of TNF-alpha and IL-12. PS blockade also re-polarizes tumor-associated M2 macrophages to a M1 phenotype which promotes the maturation of dendritic cells and induces of adaptive T-cell responses. PS targeting antibodies enhance the anti-tumor activity of anti-CTLA-4 and anti-PD-1 antibodies in models of melanoma and breast cancer and correlates with an increase in the infiltration of activated T cells and the induction of adaptive immunity. In summary, PS blockade in combination with targeted therapy and other immune checkpoint inhibitors promotes a localized, anti-tumor response and represents a promising strategy to enhance cancer immunotherapy.
*10:25am “Cancer Vaccines in the Era of Checkpoint Blockade”, Willem Overwijk, MD.ANDERSON
*11:20am “Myeloid-Derived IL-10 & PD-1 Create a Vicious Immune...”, Keith Knutson, MAYO CLINIC

- - - - - - - - - - - - - - - -From PPHM's 1-20-16 PR: http://tinyurl.com/z65avfw
“Dr. Freimark will highlight data showing that blocking PS signaling in combination with immune checkpoint inhibitors promotes a localized, anti-tumor response. He will share research findings demonstrating that PS-targeting antibodies enhance the anti-tumor activity of anti-CTLA-4 & anti-PD-1 antibodies in models of melanoma & breast cancer and correlate with an increase in the infiltration of activated T-cells and the induction of adaptive immunity. Both [Dr.Hutchins/1-25-16 & Dr.Freimank/1-26-16] presentations will also highlight key recent research findings showing that PS-signaling pathway inhibitors demonstrate multiple signs of immune activation in low or negative PD-L1 tumors. This suggests that PS-targeting antibodies have the potential to show a clinical benefit in patients with low PD-L1 levels and who do not generally benefit from checkpoint treatment alone. The potential for bavituximab to improve the clinical outcome of checkpoint inhibitors will be evaluated as part of Peregrine's ongoing clinical research collaboration with AstraZeneca. To this end, a global Phase II study of bavituximab in combination with AstraZeneca's durvalumab, an anti-PD-L1 immune checkpoint inhibitor, in patients with previously treated squamous or non-squamous NSCLC is expected to begin during Q1/2016.”
= = = = = = = = = =
Jan26-28 2016: “20th Symposium on the Interface of Regulatory & Analytical Sciences for Biotechnology Health Products”, WashDC
http://www.casss.org/?WCBP1600
CASSS = Calif. Separation Science Society (“the detailed study & controlled separation of mixtures - also referred to as “chromatography”. Major advances in separation science have enabled biologists, chemists, pharmacists and environmentalists to make breakthroughs of their own. Genomics, drug discovery, DNA fingerprinting, and ultra-trace residue analysis, for instance, would not be possible without recourse to the findings generated by separation science.
Visit Avid at table #20
BioPh, what gets me about Dr. Birge is that this man's cancer lab of 10 or so at Rutgers has been working on “the eradication of cancer” for ~15yrs, and all of a sudden he hooks up with Dr. Brekken & Peregrine, and now he's heading to Istalbul in June to give a talk ICHC 2016, and this is the subject he chooses: “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”.
Raymond Birge, PhD, is Professor of Biochemistry at Rutgers, The State Univ. of NJ, and member of the NJ Medical School Cancer Center. Dr. Birge received a BS from the Univ. of Connecticut (1984), and a PhD in Biochemical Toxicology (1989), also from the Univ. of Connecticut. He completed postdoctoral training at The Rockefeller Univ. in New York with Professor Hidesaburo Hanafusa 1989-94, was Assistant Professor in the Lab. of Molecular Oncology at Rockefeller Univ. 1994-98, and acting head of that same lab. 1998-00. Dr. Birge joined UMDNJ-NJ Medical in 2000 and currently holds a position of Professor in the Dept. of Biochemistry & Molecular Biology. Dr. Birge’s research focuses on apoptosis & cancer biology, emphasizing genes and signaling pathways that promote tumor progression, metastasis, and immune escape.
...Dr. Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html

Rutgers' Dr. Raymond Birge's 3-1-14 Youtube on Cell-Death/Apoptosis & “blocking these immune-suppressing signals to reactivate the immune system against cancer…".
Dr. Birge spoke at Peregrine's 11-2015 “Sci. Session” at SITC'15/Maryland; will be the chair in June'16 of the ICDS'16/Ireland session where Dr. Rolf Brekken (PPHM SAB) is speaking on “Blockade of PS & Immune Activation in Cancer”; and now also speaking in late June'16 at ICHC'16/Istanbul on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”.
Rutgers Univ: http://birgelab.org “The Birge laboratory conducts basic science focused on the eradication of cancer.”
3-1-14/Youtube: “Cancer Research” - Dr. Raymond Birge Laboratory, Rutgers http://birgelab.org
