Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Many years, HL45, and I ain't stoppin' till the final chapter is written and I get to finally buy those 1st 2 rounds in Tustin at an ASM!
CP, that's one excellent post on PPHM+MSKCC/Wolchok. The whole thing is blowing my mind. I will followup with some addl. thoughts that have been rattling around my head about the new AACR'17/Biomarker#3(IFN-Y), the upcoming NCCN trials(esp. the 2 Merck/Keytruda's), the AZN Mult-Solid-Tumors Trial ("Design Under Eval." per pipeline), and how I think Dr. Rachael Sanborn (Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland OR; Ph3 Sunrise Site P.I. too) is right at the center of it all with Dr. Wolchok. Just look at who Dr. Sanborn is working with… More later when I have some time to try to assemble my thoughts better.

This is where it all started IMO… PPHM, Mem.Sloan/Wolchok & Providence/Page/Sanborn working together on a new Bavi trial in Jan’16 before Sunrise stopped for futility...
1-11-16 PR, “Peregrine Provides Update on Planned Expansion of Bavi Clinical Pgm in Lung, Breast and Other Cancers”… http://tinyurl.com/zhdy37a
PLANNED TRIALS...
#4. Phase II Trial in Early Stage TNBC in Combination with Chemotherapy
Peregrine is planning to initiate a Phase II trial of bavituximab in combination with neoadjuvant chemotherapy in early stage TNBC. The primary endpoint of this study is to determine the pathologic complete response rate (pCR), an accepted surrogate endpoint in early stage TNBC. The concept for this neoadjuvant setting trial, which will be conducted at a few select U.S. sites, originated from Peregrine's ongoing collaboration with Memorial Sloan Kettering Cancer Center (MSKCC). The company has filed a study protocol to its existing bavituximab IND application in the U.S. and is currently working to open clinical trial sites, including one that will be led by David B. Page, MD, at the Providence Cancer Center in Oregon [ http://oregon.providence.org/our-services/p/providence-cancer-center => http://oregon.providence.org/clinical-trials ].
...Note: ProvidenceCC is a Ph.3 SUNRISE site, PI=Rachel Sanborn MD, Dir./Thoracic-ONCO, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md
Dr. R.Sanborn's Conflicts of interest: DNA, AZN
- - - - - - - -
Note DR. DAVID PAGE's prior work with Dr. Jedd Wolchok, chief of Mem.Sloan's Melanoma & Immunotherapeutics Service who “investigates novel approaches for cancer immunotherapy and mechanisms of tumor cell–immune cell interactions”… http://www.bcrfcure.org/researchers/david-page
SENIOR AUTHOR of PPHM’s new Biomarker Abstract coming 4-3-17 at AACR17: Yep, Dr. Rachel E. Sanborn;

Known Upcoming Events (incl. AACR’17 w/New MSK/Wolchok, NEW-BIOMARKER)
Mar13(Monday): FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Mar14/Avid: Repligen Corp's "Driving Bioprocessing Efficiency Seminar", Carlsbad http://www.repligen.com/sandiego2017 PR: http://tinyurl.com/zbflrcf
...2:45pm: Dr. David Briggs (Avid Scientist), "Case study: Positive Impact of Pre-packed Columns in a Multi-product Mfg. Facility"
Apr1-5: AACR 2017, WashDC http://tinyurl.com/zdsbds8 [See DETAILS below]
SUMMARY of PPHM’s 5 AACR’17 ABSTRACTS:
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study, see: http://tinyurl.com/h3ylrku
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com
5. PPHM+VANDERBILT+PRECISIONMEDICINE+PROVIDENCECC: 4-3-17/1pm #CT159/25 (Session: Ph2/3 Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (abstract embargoed, Sunrise Biomarker #3?)
??Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/MidAPR, Full=May17)
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
.
.
= = = = = = = = = = = = = = = = = =AACR’17(Apr2-4) DETAILS...
Apr1-5 2017: “AACR 2017”, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Abstracts: http://www.abstractsonline.com/pp8/#!/4292
5 PPHM ABSTRACTS: 2/MemSloan, 1/Immunovaccine, 1/PPHM-Only. There’s also a 5th one (#CT159/25: PPHM, Vanderbilt, Precision-for-Medicine, Providence CC) in the “Ph2/3 Clinical Trials” session, whose Abstract is still embargoed: “CT159/25: IFN-y Analysis in Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” – is this a new (3rd) Sunrise Biomaker analysis?? - see below.
...Add ImmunoVaccine Inc. (Halifax https://www.imvaccine.com ) to the list of collaborators.
...#1651(Apr3) is the newly revealed 2nd joint PPHM+Mem.Sloan/Wolchok preclin. study: “PS Targeting + Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Model” - see more below.
5 AACR’17 PPHM ABSTRACTS (DETAIL):
= = = = = = = = = = = = = = = = = = =
1. 4-2-17/1pm #574 - Session: CHECKPOINTS 1
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
NOTE: SAME TITLE AS WAS PRESENTED BY Mem.Sloan 11-14-16 SITC’16: see http://tinyurl.com/js3fca4
ABSTRACT:
Phosphatidylserine (PS) is a phospholipid that is exposed on surface of apoptotic cells, tumor cells and tumor endothelium. PS has been shown to promote immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by polarizing tumor associated macrophages into a pro-inflammatory M1 phenotype, reducing the number of MDSCs in tumors and promoting the maturation of dendritic cells into functional APCs. In a mouse B16 melanoma model, targeting PS in combination with immune checkpoint blockade promoted greater anti-tumor activity than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy (RT) is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that RT induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in treated tumors. The abscopal effect, a phenomenon in which tumor regression occurs outside the site of RT, has been observed in both preclinical and clinical trials when RT is combined with immunotherapy. In this study, we show that irradiation treatment of B16 melanoma causes an increase in PS expression on the surface of viable tumor and immune infiltrates. We subsequently examined the effects of combining RT with an antibody that targets PS (mch1N11) [“Mouse version of Bavituximab”] and immune checkpoint blockade (anti-PD-1) in B16 melanoma. We found that treatment with mch1N11 synergizes with RT to improve anti-tumor activity and overall survival in tumor bearing mice. In addition, the triple combination of mch1N11, RT and anti-PD-1 treatment displayed even greater anti-tumor and survival benefit. Analysis of local immune responses in the tumors of treated animals revealed an increase in tumor-associated macrophages with a shift towards a pro-inflammatory M1 phenotype after treatment with RT and mch1N11. In addition, analysis of the systemic immune responses in the spleen and tumor draining lymph nodes revealed an increase in CD8 T cell activation, effector cytokine production and differentiation into effector memory cells in the triple combination. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and other cancers and may inform the design of clinical studies combining PS-targeting antibodies with RT and/or checkpoint blockade.
= = = = = = = = = = = = = = = =
2. 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY$$$
…...$$$See: More on the Significance of the New MSK/Wolchok+PPHM “ACT” study: http://tinyurl.com/h3ylrku
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
ABSTRACT:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors or chimeric antigen receptors. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockadedid not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
----------
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
= = = = = = = = = = = = = = = =
3. 4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
ABSTRACT:
Previous studies utilizing NanoString immune profile analysis demonstrated that intratumoral levels of LAG3 (lymphocyte activation gene 3) mRNA increased in response to phosphatidylserine (PS) and PD-1 targeting antibodies in murine triple negative breast cancers (TNBC). This suggests LAG3 acts to attenuate immune system activation during I/O therapies - and that PD-1 and LAG3 function cooperatively in suppressing immune system activation. Here we show that adding PS targeting antibodies can further enhance the effectiveness of antibodies targeting LAG3 and/or LAG3+PD-1. We first examined expression of LAG3 and PD-1 in the murine TNBC model E0771 and found that tumor associated T-cells (CD4+ and CD8+) have expression of both markers. Mice implanted with TNBC tumors were next treated with antibodies targeting PS, PD-1, and LAG3 alone and in combination with each other. Interestingly, the addition of PS targeting antibodies not only increased the effectiveness anti-PD-1 effectiveness as previously observed, but also enhanced anti-LAG3 treatment, showing that PS targeting antibodies are capable of augmenting additional I/O therapeutic regimens. Comparison of anti-PD-1+LAG3 combination vs. single anti-PD-1 or anti-LAG3 treatments showed moderately more anti-tumor activity than single treatments, however the addition of PS targeting antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the anti-LAG3+PD-1 treatment. Further comparison of antibody treatments targeting PD-1+LAG3 vs. PS+PD-1+LAG3 demonstrated that the addition of PS targeting antibodies resulted in a significant decrease in tumor growth with complete tumor regression in 80% of the animals (along with the ability to completely reject secondary TNBC challenge) compared to 0% in the anti-PD-1+LAG3 treatment group. Immunoprofiling showed that the addition of PS targeting antibodies to these checkpoint therapies, including the combination of anti-PD-1+LAG3, resulted in a phenotype associated with enhanced immune system activation and immune-surveillance including increased tumor infiltrating lymphocytes (TILs) with upregulation of T-cell associated activation pathways, increased Th1 to Th2 profile, and enhanced antigen presentation processing /presentation mechanisms along with cytokines associated with immune system activation. Overall our data demonstrate that adding PS targeting antibodies to clinically relevant therapies, including PD-1 and LAG3, may significantly enhance their ability to activate and redirect the host immune system into recognition and elimination of tumor cells compared to single and combinational treatments that lack PS targeting antibodies.
= = = = = = = = = = = = = = = =
4. 4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
ABSTRACT:
Antibodies targeting phosphatidylserine (PS) have been shown to induce anti-tumor responses by induction of tumor-specific T cells. Based on this observation, we evaluated the responses of PS and PD-1 targeting antibody therapy to enhance anti-tumor responses of a HPV16 peptide vaccine formulated in DepoVax (DPX) in mice bearing HPV-transformed C3 mouse tumors. The addition of PS-targeting antibody (mch1N11) [“Mouse version of Bavituximab”] to DPX/metronomic cyclophosphamide (mCPA) immunotherapy prolonged survival in comparison to mice receiving an isotype control in combination with DPX/mCPA. When anti-PD-1 was added to mch1N11 + mCPA, there was no increase in survival. The addition of mch1N11 to DPX/mCPA immunotherapy had no effect on tumor growth or survival in the aggressive B16-F10 model. TIL analysis revealed an increase in CD8+ T cells, antigen specific CD8+ T cells and PD-1+ T cells in the tumor with mch1N11 treatment. The expression of surface markers for macrophages (CD68high, F4/80) and dendritic cells (CD11c) were also increased in the tumors of mice treated with mch1N11. RT-qPCR analysis of the tumor confirmed higher mRNA expression of T cells markers (CD8, Granzyme B, PD-1) and antigen presenting cell markers (F4/80, CD74). In the spleen, expression of cell surface markers for monocytes (CD11b) and PD-1+ T cells (CD8) were elevated in groups treated with mch1N11 in combination with anti-PD-1. Combined, these findings indicate that in this model, PS-targeting antibodies can enhance the activity of phagocytic cells involved in antigen presentation. We have found that PD-1 expression increases as anti-tumor activity increases, therefore these results also provide an indication that antibodies targeting PS enhance the anti-tumor immune response induced by DPX/mCPA therapy. The observations suggest that PS-targeting antibodies may enhance therapeutic vaccines for the treatment of cancer.
= = = = = = = = = =IS THIS A NEW (3RD) SUNRISE BIOMAKER ANALYSIS??
#5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn (Providence CC)*** 4
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
***Rachel Sanborn MD, Dir./Thoracic-Oncology, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN
ABSTRACT: Embargoed – until 4/3/17??
-------
Is this AACR’17 #5 perhaps the 3rd Sunrise Biomarker??? It does not look like #2 (12-7-16 WCLC’16(IASLC)/Vienna) that was canceled due to “work not complete”...
Known Ph.3 Sunrise Biomarkers/UTSW’s Dr. David Gerber et al:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=???
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5


- - - - - - - - - - - - - - - - - - -
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(P1+3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”
= = = = = = = = = = = = =
2-28-17: Collabs with Mem.Sloan(Wolchok), Duke, MDA, Rutgers, ImmunoVaccine, UTSW… http://tinyurl.com/heg9t3v
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
“PS Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (same as AACR’17 4-2-17 #574)
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchock states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub states, "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current & next gen. of immune modulators is likely to increase the extent & amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next gen. of clinical studies with bavituximab.”
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
SITC’16 PPHM/Mem.Sloan Poster – same as AACR’17 4-2-17 #574…

I checked AACR'17 Abstracts for Dr.Wolchok, and 4 more Abstracts he is co-author on popped up (All CTxxx Clin.Trials Abstracts - I guess recently added). I have updated my Mem.Sloan/Wolchok AACR'17 summary:
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(P1+3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”
PPHM’s Sunrise Biomarkers (#1/Oct’16, #2/Dec’16, #3?/AACR17)
I’m not sure if the 3-1-17 revealed AACR’17 Biomarker Abstract is the same as #2 or a brand new #3. One diff. between #2 and AACR’17 is BIODESIX dropped and PRECISION-FOR-MEDICINE added.
Summary of the 3 known Ph3/Sunrise Biomarker Data Presentations:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.”
David R. Spigel: LEAD AUTHOR: CSO/Dir. Lung Cancer Pgm/Sarah Cannon Res.
David E. Gerber: SENIOR AUTHOR: UTSW/Dallas (Sunrise PI)
CO-AUTHORS: R.Natale/CEDARS-SINAI, R.Sanborn/PROVCC, PPHM’s N.Kallinteris, J.Lai, M.Tang, J.Shan, and 7 Intl. Sunrise PI’s: Ger/2, Ukraine, Greece, Spain, Korea, Hungry.
RESULTS:
~200 of 600 pts (~30%) tested positive for B2GPI(200-240). Those 200 received Stat.Sig. Improvement in O/S 7.7=>13.2mos; ie, lived +70% longer.
MORE DETAILS: http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce”
PRESENTATION CANCELLED – IR said,”Data analysis not completed in time.”
David E. Gerber: LEAD AUTHOR: UTSW/Dallas (Sunrise PI)
Rachael Sanborn: SENIOR AUTHOR: Dir./Thoracic-Oncology, Providence CC/Portland
CO-AUTHORS: L.Horn/VANDY, G.Losonczy/BUDAPEST, R.Natale/CEDARS-SINAI, H.Roder, J.Roder/BIODESIX, PPHM’s N.Kallinteris, M.Tang, J.Shan.
RESULTS:
104 of 193 pts tested high for complement activation, and from that, a 2nd subgrp isolated that tested low IL-10: O/S 5.9=>12.5mos. Remaining 397pts will be presented at WCLC’16.
MORE DETAILS: http://tinyurl.com/z8cq8vx
#3??? - REVEALED BY AACR’17 ABSTRACT REL. 3-1-17:
4-3-17/1pm #CT159/25 (Session: Phase II/III Clinical Trials in Progress)
“IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
Abstract still embargoed a/o 3-2-17 – will we have to wait until 4-3-17?
Nikoletta Kallinteris (PPHM): LEAD AUTHOR: Peregrine
Rachael Sanborn: SENIOR AUTHOR: Dir./Thoracic-Oncology, Providence
CO-AUTHORS: L.Horn/VANDY, T.Guennel/PRECISION-FOR-MEDICINE, PPHM’s N.Kallinteris, M.Tang, S.Yin, J.Lai, J.Shan.
“IFNy, or type II interferon gamma, is a cytokine that is critical for innate & adaptive immunity against viral, some bacterial & protozoal infections.”
MORE DETAILS: http://tinyurl.com/zdsbds8
QUESTION: Is this Sunrise Biomarker #3, or a followup to BioMarker #2 from 12-7-16 IASLC/WCLC16 (Complement & IL-10 Pathways) that was cancelled because “data analysis not completed in time” per IR?


= = = = = = = = = = = = =
Biomarker #2: 12-7-16 IASLS/WCLC’16 ABSTRACT pic:

Biomarker #1 (ESMO’16 10-10-16):
--------------
** Data presented at ESMO’16 demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approx. 30% of randomized patients) achieved a statistically significant, 5.5-mo. improvement (13.2 mos. vs. 7.7 mos.) in MOS as compared to patients in the ctl. group with the same range of B2GP1 levels [p = 0.049; HR=.67]. http://tinyurl.com/hp73njt
JakeD/BioBS/etal, I still can't figure out if the AACR'17 Biomarker Abstract revealed at AACR.com yesterday is the same as 12-7-16/IASLC/Vienna or a brand new #3!! Study my next post please and give me the definitive answer please! ![]()
PurpleD, Thx for the find of AACR #5. Was so busy, no time to thank you till now. No doubt it's a Sunrise Biomarker Analysis. It could be the same as the 2nd "Cancelled, work not finished" 12-7-16/Vienna/WCLC'16 one, but I think it's a new 3rd one - just too many differences in the authors and the title is so different. We'll see though!
Apr1-5: AACR 2017, WashDC http://tinyurl.com/zdsbds8 <=See #5.
= = = = = = = = = =IS THIS A NEW (3RD) SUNRISE BIOMAKER ANALYSIS??
#5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
"IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn (Providence CC)*** 4
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
***Rachel Sanborn MD, Dir./Thoracic-Oncology, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN
ABSTRACT: Still Embargoed a/o 3-1-17 5:45pmET.
------
Is this AACR’17 #5 perhaps the 3rd Sunrise Biomarker??? It does not look like #2 (12-7-16 WCLC’16(IASLC)/Vienna) that was canceled due to “work not complete”, but certainly I could be wrong...
Sunrise Biomarkers/UTSW’s Dr. David Gerber:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=???
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5
...Recall: #2: 12/7/16/Vienna/WCLC’16/Canceled:
”A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder[CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
AACR’17(Apr2-4), 5 PPHM ABSTRACTS: 2/MemSloan, 1/Immunovaccine, 1/PPHM-Only. There’s also a 5th one (PPHM, Vanderbilt, Precision-for-Medicine, Providence CC) in the “Ph2/3 Clinical Trials” session, whose Abstract is still embargoed: “CT159/25: IFN-y Analysis in Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” – is this a new (3rd) Sunrise Biomaker analysis?? - see below.
...Add ImmunoVaccine Inc. (Halifax https://www.imvaccine.com ) to the list of collaborators.
...#1651(Apr3) is the newly revealed 2nd joint PPHM+Mem.Sloan/Wolchok preclin. study: “PS Targeting + Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Model” - see more below.
...Memorial Sloan’s Dr. Jedd. Wolchok is co-author of 5 AACR’17 Posters: 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA.
= = = = = = = = = = = = = = = = = = = = = = = = = =
Apr1-5 2017: “AACR 2017”, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Abstracts: http://www.abstractsonline.com/pp8/#!/4292
SUMMARY of PPHM’s 5 AACR’17 ABSTRACTS:
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study, see: http://tinyurl.com/h3ylrku
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com
5. PPHM+VANDERBILT+PRECISIONMEDICINE+PROVIDENCECC: 4-3-17/1pm #CT159/25 (Session: Phase II/III Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (Abstracts still embargoed a/o 3-1-17 5:45pmET)
AACR’17 DETAILS (5 PPHM ABSTRACTS)...
= = = = = = = = = = = = = = = = = = =
1. 4-2-17/1pm #574 - Session: CHECKPOINTS 1
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
NOTE: SAME TITLE AS WAS PRESENTED BY Mem.Sloan 11-14-16 SITC’16: see http://tinyurl.com/js3fca4
ABSTRACT:
Phosphatidylserine (PS) is a phospholipid that is exposed on surface of apoptotic cells, tumor cells and tumor endothelium. PS has been shown to promote immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by polarizing tumor associated macrophages into a pro-inflammatory M1 phenotype, reducing the number of MDSCs in tumors and promoting the maturation of dendritic cells into functional APCs. In a mouse B16 melanoma model, targeting PS in combination with immune checkpoint blockade promoted greater anti-tumor activity than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy (RT) is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that RT induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in treated tumors. The abscopal effect, a phenomenon in which tumor regression occurs outside the site of RT, has been observed in both preclinical and clinical trials when RT is combined with immunotherapy. In this study, we show that irradiation treatment of B16 melanoma causes an increase in PS expression on the surface of viable tumor and immune infiltrates. We subsequently examined the effects of combining RT with an antibody that targets PS (mch1N11) [“Mouse version of Bavituximab”] and immune checkpoint blockade (anti-PD-1) in B16 melanoma. We found that treatment with mch1N11 synergizes with RT to improve anti-tumor activity and overall survival in tumor bearing mice. In addition, the triple combination of mch1N11, RT and anti-PD-1 treatment displayed even greater anti-tumor and survival benefit. Analysis of local immune responses in the tumors of treated animals revealed an increase in tumor-associated macrophages with a shift towards a pro-inflammatory M1 phenotype after treatment with RT and mch1N11. In addition, analysis of the systemic immune responses in the spleen and tumor draining lymph nodes revealed an increase in CD8 T cell activation, effector cytokine production and differentiation into effector memory cells in the triple combination. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and other cancers and may inform the design of clinical studies combining PS-targeting antibodies with RT and/or checkpoint blockade.
= = = = = = = = = = = = = = = =
2. 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY$$$
…...$$$See: More on the Significance of the New MSK/Wolchok+PPHM “ACT” study: http://tinyurl.com/h3ylrku
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
ABSTRACT:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors or chimeric antigen receptors. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockadedid not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
----------
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
= = = = = = = = = = = = = = = =
3. 4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
ABSTRACT:
Previous studies utilizing NanoString immune profile analysis demonstrated that intratumoral levels of LAG3 (lymphocyte activation gene 3) mRNA increased in response to phosphatidylserine (PS) and PD-1 targeting antibodies in murine triple negative breast cancers (TNBC). This suggests LAG3 acts to attenuate immune system activation during I/O therapies - and that PD-1 and LAG3 function cooperatively in suppressing immune system activation. Here we show that adding PS targeting antibodies can further enhance the effectiveness of antibodies targeting LAG3 and/or LAG3+PD-1. We first examined expression of LAG3 and PD-1 in the murine TNBC model E0771 and found that tumor associated T-cells (CD4+ and CD8+) have expression of both markers. Mice implanted with TNBC tumors were next treated with antibodies targeting PS, PD-1, and LAG3 alone and in combination with each other. Interestingly, the addition of PS targeting antibodies not only increased the effectiveness anti-PD-1 effectiveness as previously observed, but also enhanced anti-LAG3 treatment, showing that PS targeting antibodies are capable of augmenting additional I/O therapeutic regimens. Comparison of anti-PD-1+LAG3 combination vs. single anti-PD-1 or anti-LAG3 treatments showed moderately more anti-tumor activity than single treatments, however the addition of PS targeting antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the anti-LAG3+PD-1 treatment. Further comparison of antibody treatments targeting PD-1+LAG3 vs. PS+PD-1+LAG3 demonstrated that the addition of PS targeting antibodies resulted in a significant decrease in tumor growth with complete tumor regression in 80% of the animals (along with the ability to completely reject secondary TNBC challenge) compared to 0% in the anti-PD-1+LAG3 treatment group. Immunoprofiling showed that the addition of PS targeting antibodies to these checkpoint therapies, including the combination of anti-PD-1+LAG3, resulted in a phenotype associated with enhanced immune system activation and immune-surveillance including increased tumor infiltrating lymphocytes (TILs) with upregulation of T-cell associated activation pathways, increased Th1 to Th2 profile, and enhanced antigen presentation processing /presentation mechanisms along with cytokines associated with immune system activation. Overall our data demonstrate that adding PS targeting antibodies to clinically relevant therapies, including PD-1 and LAG3, may significantly enhance their ability to activate and redirect the host immune system into recognition and elimination of tumor cells compared to single and combinational treatments that lack PS targeting antibodies.
= = = = = = = = = = = = = = = =
4. 4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
ABSTRACT:
Antibodies targeting phosphatidylserine (PS) have been shown to induce anti-tumor responses by induction of tumor-specific T cells. Based on this observation, we evaluated the responses of PS and PD-1 targeting antibody therapy to enhance anti-tumor responses of a HPV16 peptide vaccine formulated in DepoVax (DPX) in mice bearing HPV-transformed C3 mouse tumors. The addition of PS-targeting antibody (mch1N11) [“Mouse version of Bavituximab”] to DPX/metronomic cyclophosphamide (mCPA) immunotherapy prolonged survival in comparison to mice receiving an isotype control in combination with DPX/mCPA. When anti-PD-1 was added to mch1N11 + mCPA, there was no increase in survival. The addition of mch1N11 to DPX/mCPA immunotherapy had no effect on tumor growth or survival in the aggressive B16-F10 model. TIL analysis revealed an increase in CD8+ T cells, antigen specific CD8+ T cells and PD-1+ T cells in the tumor with mch1N11 treatment. The expression of surface markers for macrophages (CD68high, F4/80) and dendritic cells (CD11c) were also increased in the tumors of mice treated with mch1N11. RT-qPCR analysis of the tumor confirmed higher mRNA expression of T cells markers (CD8, Granzyme B, PD-1) and antigen presenting cell markers (F4/80, CD74). In the spleen, expression of cell surface markers for monocytes (CD11b) and PD-1+ T cells (CD8) were elevated in groups treated with mch1N11 in combination with anti-PD-1. Combined, these findings indicate that in this model, PS-targeting antibodies can enhance the activity of phagocytic cells involved in antigen presentation. We have found that PD-1 expression increases as anti-tumor activity increases, therefore these results also provide an indication that antibodies targeting PS enhance the anti-tumor immune response induced by DPX/mCPA therapy. The observations suggest that PS-targeting antibodies may enhance therapeutic vaccines for the treatment of cancer.
= = = = = = = = = =IS THIS A NEW (3RD) SUNRISE BIOMAKER ANALYSIS??
5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”CT159 / 25 - IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn (Providence CC)*** 4
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
***Rachel Sanborn MD, Dir./Thoracic-Oncology, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN
ABSTRACT: Still Embargoed a/o 3-1-17 5:45pmET.
------
Is this AACR’17 #5 perhaps the 3rd Sunrise Biomarker??? It does not look like #2 (12-7-16 WCLC’16(IASLC)/Vienna) that was canceled due to “work not complete”...
Sunrise Biomarkers/UTSW’s Dr. David Gerber:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=???
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5
...Recall: #2: 12/7/16/Vienna/WCLC’16/Canceled:
”A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder[CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
- - - - - - - - - - - - - - - - - - -
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 34-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
= = = = = = = = = = = = =
2-12-17: Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, ImmunoVaccine Inc… http://tinyurl.com/gof4kua
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
“PS Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (same as AACR’17 4-2-17 #574)
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchock states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub states, "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current & next gen. of immune modulators is likely to increase the extent & amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next gen. of clinical studies with bavituximab.”
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
SITC’16 PPHM/Mem.Sloan Poster – same as AACR’17 4-2-17 #574…

Neato: H.McArthur/CedSinai, D.Page/ProvCC => MBC Advisors for PPHM
Nov2016: “Immunotherapy for the Treatment of Breast Cancer: Checkpoint Blockade, Cancer Vaccines, and Future Directions in Combination Immunotherapy”
Clinical Advances in Hematology & Oncology
Authors(equal contributors):
* Heather L. McArthur MD/MPH: Medical Director of Breast Oncology at Cedars Sinai Medical Ctr, Los Angeles
* David B. Page MD: Oncologist at the Providence Cancer Ctr and a researcher at the Earle A. Chiles Res. Inst., Portland
DISCLOSURES:
* Dr H.McArthur has participated in advisory boards for Celgene, Merck, Spectrum Pharm., OBI Pharma, Peregrine Pharmaceuticals, Syndax Pharm, and has received research support from Bristol-Myers Squibb, MedImmune/AstraZeneca, Eli Lilly, Ziopharm Oncology, Merck.
* Dr D.Page has participated in advisory boards for Celgene and Peregrine Pharmaceuticals, and has received research support from MedImmune and Merck.
http://www.hematologyandoncology.net/index.php/archives/november-2016/immunotherapy-for-the-treatment-of-breast-cancer-checkpoint-blockade-cancer-vaccines-and-future-directions-in-combination-immunotherapy
= = = = = = = = = = = = = = = = = = = = = = = = = =
1-11-16 PR, “Peregrine Provides Update on Planned Expansion of Bavi Clinical Pgm in Lung, Breast and Other Cancers”… http://tinyurl.com/zhdy37a
PLANNED TRIALS...
#4. Phase II Trial in Early Stage TNBC in Combination with Chemotherapy
Peregrine is planning to initiate a Phase II trial of bavituximab in combination with neoadjuvant chemotherapy in early stage TNBC. The primary endpoint of this study is to determine the pathologic complete response rate (pCR), an accepted surrogate endpoint in early stage TNBC. The concept for this neoadjuvant setting trial, which will be conducted at a few select U.S. sites, originated from Peregrine's ongoing collaboration with Memorial Sloan Kettering Cancer Center (MSKCC). The company has filed a study protocol to its existing bavituximab IND application in the U.S. and is currently working to open clinical trial sites, including one that will be led by David B. Page, M.D., at the Providence Cancer Center in Oregon. http://tinyurl.com/zhdy37a
- - - - - - - -DR. DAVID PAGE: note his prior work with Dr. Jedd Wolchok, chief of Mem.Sloan's Melanoma & Immunotherapeutics Service who “investigates novel approaches for cancer immunotherapy and mechanisms of tumor cell–immune cell interactions”… http://www.bcrfcure.org/researchers/david-page
= = = = = = = = = = = = = =
Cedars-Sinai Researcher Dr. Heather McArthur (Medical Director, Breast Oncology) lists PPHM in her Disclosures in this Joint Mem.Sloan/Cedars-Sinai AACR’17 Abstract, where she’s the Lead Author, along with co-authors Jedd Wolchok, Taha Merghoub, and several other Mem. Sloan researchers. So, 2 of Dr. Wolchok’s 5 AACR’17 abstracts are Joint PPHM+MemSloan, and a 3rd is Joint CedarsSinai+MemSloan whereby the Lead Author lists Peregrine in her disclosures. Maybe we need to add Cedars-Sinai to our collabs list along with Memorial Sloan Kettering, Duke, MDA, Rutgers, ImmunoVaccine, and UTSW.
SEE: 3-1-17: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=129115855
= = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly & PPHM poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Title: ‘Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”’… Data showed that a combination of anti-PS & anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations w/paclitaxel, in the E0771 murine model of TNBC.”
Kensuke Kaneko 1, Takuya Osada 1, Bruce D. Freimark 2, Herbert Kim Lyerly ** (Duke Univ.)
1=Duke University, Durham, NC
2=Peregrine Pharmaceuticals, Inc.
**Dr. Herbert Kim Lyerly: https://immunology.duke.edu/people/herbert-kim-lyerly-md (George Barth Geller Professor, Duke Univ. MC)
PPHM’s Dr. Jeff Hutchins 10-24-16: “We plan to continue to work with our collaborators at Duke Univ. Medical Center to further study the therapeutic potential of PS-targeting agents in combination with checkpoint inhibitors like anti-PD-L1 and conventional therapies that augment immunotherapy mechanisms."
Cedars-Sinai Researcher lists PPHM in her Disclosures in this Joint Mem.Sloan/Cedars-Sinai AACR’17 Abstract, where she's the Lead Author, along with co-authors Jedd Wolchok, Taha Merghoub, and several other Mem. Sloan researchers. So, 2 of Dr. Wolchok’s 5 AACR’17 abstracts are Joint PPHM+MemSloan, and a 3rd is Joint CedarsSinai+MemSloan whereby the Lead Author lists Peregrine in her disclosures. Maybe we need to add Cedars-Sinai to our collabs list along with Memorial Sloan Kettering, Duke, MDA, Rutgers, ImmunoVaccine, and UTSW.
AACR’17 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing Rad therapy (RT) for HER2+ breast cancer brain metastases”
Session: Immunomodulatory Agents & Therapeutics
Presenter/Authors:
Heather L. McArthur 1[Lead author, MD/MPH], Kathryn Beal 2, Darragh Halpenny 2, Micaela Henrich 2, Shanu Modi 2, Sujata Patil 2, Robert Young 2, Thomas Kaley 2, Taha Merghoub 2, Christopher Barker 2, Phillip Wong 2, Nicola Hamilton 2, Clifford Hudis 2, Jedd Wolchok 2, Larry Norton 2
1=Cedars-Sinai Medical Center, Los Angeles
2=Memorial Sloan Kettering Cancer Ctr., NYC
Disclosures:
Heather McArthur: Merck; OBI Pharma; Bristol Myers Squibb; Eli Lilly; MedImmune, AstraZeneca; Roche; Celgene; Calithera; Peregrine; Spectrum Pharm.
http://www.abstractsonline.com/pp8/#!/4292/presentation/6249
------
Breast Cancer Res. Foundation (BCRF) Bio: Heather L. McArthur, MD, MPH
Medical Director, Breast Oncology, Cedars-Sinai Medical Center, Los Angeles
https://www.bcrfcure.org/researchers/heather-l-mcarthur
.
.
NOTE: Mem. Sloan’s JEDD WOLCHOK is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=same as SICT’16/11-14-16
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=New(2nd) PPHM+MSK/Wolchok
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
=>5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing Rad therapy for HER2+ breast cancer brain metastases” <=Note: Lead Author Heather L. McArthur, Medical Dir./Breast Oncology at Cedars-Sinai MC, lists PPHM in her disclosures.
The 4 PPHM/MemSloan/Immunovaccine Abstracts - Pub. Today/4:30pmET at http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105&DetailItemID=539
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/Nov'16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study, see: http://tinyurl.com/h3ylrku
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com
------------------
Mem. Sloan’s DR. JEDD. WOLCHOK is co-author of 5 AACR’17 Posters.
...2 of the 5 are w/PPHM re: PS-Targeting.
...Only one other is with a biotech: Genocea Biosciences.
...The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model”
NON-PEREGRINE:
3.(MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4.(MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5.(MSK & Cedars-Sinai/LA) 34-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
Collabs with Mem.Sloan(Wolchok), Duke, MDA, Rutgers, ImmunoVaccine, UTSW…
...(updated with further info. on the new(2nd) joint PPHM+MemSloan/Wolchok study, “PS-Targeting + Adoptive T Cell Transfer (ACT) Eliminates Advanced Melanoma Tumors”, revealed by the AACR’17 abstracts. Also added to Summary at the top).
QUICK SUMMARY:
* The PS+ Exosomes Program: esp. given UTSW’s Dr. Alan Schroit’s reputation; initial POC done/advancing via partnership. See: http://tinyurl.com/zbwr7cc
* Sunrise Biomarkers/UTSW’s Dr. David Gerber: #1=B2GPI, #2=Complement & IL-10 Pathways(12-7-26/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”, #3=???. The results of this may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5 & http://www.peregrineinc.com/pipeline/overview.html
* The MSK/Wolchok collab: incl. revelation by the new(2nd) AACR’17 MSK abstract stating, “PPHM’s PS-Targeting + Adoptive Tcell Transfer(ACT) Eliminates Advanced Tumors without Off-Target Toxicities”. Plus, Dr. Wolchok co-author of 5 AACR’17 Posters: 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. See: http://tinyurl.com/z47hb2s
* The Rutgers/R.Birge collab: ex: “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk) Regulates AKT-dep. Chemoresistance & PD-L1 Expression”) - 2-9-17/AACR. See: http://tinyurl.com/h4gdke3
* The Duke/H.Lyerly collab: ex: “Anti-PS + anti-PD-L1 leads to greater anti-tumor responses in TNBC” - latest is 10-22-16/AACR I-O Conf. – see: http://tinyurl.com/zzryfok
* The MDA/JB.Fleming collab: ex: “PreClin. Eval of DNAtrix’s DNX2401+FhuBAVI(1N11) for Pancreatic Cancer - significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”, 1-30-17 AACR(Mol.Cancer). See: http://tinyurl.com/hov4hfb
* The ImmunoVaccine Inc. collab: “PS Targeting Enhances Anti-Tumor Activity of DepoVax Tumor Vaccine” - per AACR’17 4-4-17 poster. See: http://tinyurl.com/jxfm3hb
------------------------------------------------------------------
MORE DETAIL (CHRONOLOGICAL):
AACR’17: 4-3-17 #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(2nd known study - revealed by AACR’17 Abstract):
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer (ACT) Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Lead author: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK), Co-author Jedd Wolchok, PPHM & MSKCC scientists. ( http://www.mskcc.org/research-areas/labs/jedd-wolchok )
NOTES:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
-----
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
-----
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
-----
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow ["Immunologic Checkpoint Blockade"]. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-2-17 #574 - Session: CHECKPOINTS 1 http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(1st known study – revealed at Nov’16/SITC16):
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
...OBVIOUSLY THIS IS A FOLLOWUP TO THE 11-14-16 SITC’16 PRESENTATION:
SITC’16: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (Joint Mem. Sloan Kettering/Wolchok Lab & PPHM)
...DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
-------
EYE OPENER: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. ).http://www.abstractsonline.com/pp8/#!/4292
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model”
NON-PEREGRINE:
3.(MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4.(MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5.(MSK & Cedars-Sinai/LA) 34-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-4-17 #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model” (PPHM & Immunovaccine Inc.)
=> Genevieve Weir 1, Tara Quinton 1, Jeff Hutchins 2, Bruce Freimark 2, Marianne Stanford (VP/Res., Immunovaccine Inc.)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]; 2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
[Note: POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB?? 11-9-15 SITC'15: ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.” http://tinyurl.com/pbof95w ]
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 2-9-17/AACR article (Rutgers’ Dr. Raymond Birge, PPHM, Advanced Proteome Therapeutics, etal): “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk) Regulates AKT-dependent Chemoresistance & PD-L1 Expression” http://tinyurl.com/h4gdke3
…”These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors. Implications: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.”

= = = = = = = = = = = = = = = = = = = = = = = = = =
PPHM's EXOSOME-BASED (PS Positive) CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") – Led by UTSW’s Dr. Alan Schroit (PPHM SAB)
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17/PR: PS+ Exosomes Proof-of-Concept Data (N=44, Ovarian, Dr. Alan Schroit/UTSW etal) Published 1-22-17 in OncoTarget http://tinyurl.com/jhv57ua
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies.
...Dr. Stephen Worsley (VP/BusDev): “...we believe this has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
...PPHM’s PS Exosomes platform advantages outlined: UTSW/website & World Patent #WO/2016/201064: http://tinyurl.com/zbwr7cc
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
...Dr. Jeff Hutchins (VP/PreclinRES): "Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing pgm. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this pgm in 2017."
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 1-30-17 AACR(Mol.Cancer) article: MDA/UTSW/Brekken/etal, “PreClin. Eval of DNX2401+FhuBAVI(1N11) for Pancreatic Cancer” http://tinyurl.com/hov4hfb
...Combo Delta-24-RGD + FhuBAVI “significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”
http://mct.aacrjournals.org/content/early/2017/01/28/1535-7163.MCT-16-0526
**DNX-2401: DNAtrix Corp., Houston TX http://www.dnatrix.com (private company)
“DNAtrix’s lead product, DNX-2401, has demonstrated that DNX viruses exhibit a potent anti-tumor mechanism of action by (1) replicating in human tumors (2) eliciting tumor necrosis and (3) triggering an immune response.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
PPHM’s Sunrise Biomarker Data (#1/Oct’16, #2/Dec’16/Delayed) – UTSW’s Dr. David Gerber
See: 2-14-17: http://tinyurl.com/jbv3ms5
...Summary of the 2 known Ph3/Sunrise Biomarker Data Presentations:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce” <=PRESENTATION CANCELLED/”Anal.Not.Finished(IR)” http://tinyurl.com/z8cq8vx
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly & PPHM poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Title: ‘Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”’… Data showed that a combination of anti-PS & anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations w/paclitaxel, in the E0771 murine model of TNBC.”
Kensuke Kaneko 1, Takuya Osada 1, Bruce D. Freimark 2, Herbert Kim Lyerly ** (Duke Univ.)
1=Duke University, Durham, NC
2=Peregrine Pharmaceuticals, Inc.
**Dr. Herbert Kim Lyerly: https://immunology.duke.edu/people/herbert-kim-lyerly-md (George Barth Geller Professor, Duke Univ. MC)
PPHM’s Dr. Jeff Hutchins 10-24-16: “We plan to continue to work with our collaborators at Duke Univ. Medical Center to further study the therapeutic potential of PS-targeting agents in combination with checkpoint inhibitors like anti-PD-L1 and conventional therapies that augment immunotherapy mechanisms."
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray (PPHM): Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 7-27-16 Shaul/Brekken/Thorpe/etal PLOS ONE article: Fhu/Bavi vs. APS-related Pregnancy Complications & Thrombosis http://tinyurl.com/jlhrdg2
...”The potential clinical impact of 1N11 (Fhu Bavi) in patients with APS is substantial. Spring-boarding from the present discovery of 1N11 as a highly-effective, mechanism-based treatment for APS in a comprehensive series of mouse models of APS-related disorders, clinical studies of 1N11 now warrant consideration.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes(UCal-Irvine)-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article (PPHM/Friemark, U.Cal-Irvine/CW-Hughes - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
= = = = = = = = = = = = = = = = = = = = = = = = = =
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
...Authors: Rutgers(Birge/Kumar/Calianese), Peregrine(4), UTSW(Brekken/Schroit/Huang), Friedrich-Alexander-Univ./Germany(Martin Herrmann/Boeltz/Schett), Rio de Janeiro, Univ. College London
...”we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
= = = = = = = = = = = = = = = = = = = = = = = = = =
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Jedd Wolchok states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab & Swim Across America Lab at Memorial Sloan Kettering) states, ”A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab.”
.
.

.
.

Which will break 1st? What order the rest?
* The PS+ Exosomes Program: esp. given UTSW’s Dr. Alan Schroit’s reputation; initial POC done/advancing via partnership. See: http://tinyurl.com/zbwr7cc
* Sunrise Biomarkers: #1=B2GPI, #2=Complement & IL-10 Pathways(12-7-26/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos.”, #3=???. See http://tinyurl.com/jbv3ms5
* The MSK/Wolchok collab: incl. revelation by the new(2nd) AACR’17 MSK abstract stating, “PPHM’s PS-Targeting + Adoptive Tcell Transfer(ACT) Eliminates Advanced Tumors without Off-Target Toxicities”. Plus, Dr. Wolchok co-author of 5 AACR’17 Posters: 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. See: http://tinyurl.com/z47hb2s
* The Rutgers/R.Birge collab: ex: “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk) Regulates AKT-dep. Chemoresistance & PD-L1 Expression”) - 2/9/17/AACR. See: http://tinyurl.com/h4gdke3
* The Duke/H.Lyerly collab: ex: “AntiPS + antiPD-L1 leads to greater anti-tumor responses in TNBC” - latest is 10-22-16/AACR I-O Conf. – see: http://tinyurl.com/zzryfok
* The MDA/JB.Fleming collab: ex: “PreClin. Eval of DNAtrix’s DNX2401+FhuBAVI(1N11) for Pancreatic Cancer - significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”, 1-30-17 AACR(Mol.Cancer). See: http://tinyurl.com/hov4hfb
* The ImmunoVaccine Inc. collab: “PS-Targeting Enhances Anti-Tumor Activity of DepoVax Tumor Vaccine” - per AACR’17 4-4-17 poster. See: http://tinyurl.com/jxfm3hb
* The AZN & NCCN clinical collabs: Sunrise Biomarkers adjusted?
* Continuing Avid expansion/revs-growth.
MORE ON PRE-CLIN. COLLABS HERE: http://tinyurl.com/gwa2gkn
.
.
.
= = = = = = = = = = = KNOWN UPCOMING EVENTS:
Feb27-Mar2/Avid Booth #816: BioProcess Intl. West, SanFran https://lifesciences.knect365.com/bpi-west PR: http://tinyurl.com/zbflrcf
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Mar14/Avid: Repligen Corp's "Driving Bioprocessing Efficiency Seminar", Carlsbad http://www.repligen.com/sandiego2017 PR: http://tinyurl.com/zbflrcf
...2:45pm: Dr. David Briggs (Avid Scientist), "Case study: Positive Impact of Pre-packed Columns in a Multi-product Mfg. Facility"
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=2nd Joint MemSloan/PPHM study, see: http://tinyurl.com/h3ylrku
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
INTERESTING:
Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. This is presuming that I searched properly (I think I did): http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model”
NON-PEREGRINE:
3.(MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4.(MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5.(MSK & Cedars-Sinai/LA) 34-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins***(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
***Note: 1-4-17: Dr. Hutchins left Peregrine to become CSO of Heat-Biologics.
??Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/MidAPR, Full=May17)
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
SunS, I doubt Drs. Wolchok & Merghoub and the rest of the Mem. Sloan Wolchok Lab throw around the phrase “Eliminates Advanced Tumors” just any ‘ol place, for any ‘ol reason, in their presentations. We’ll know a lot more on March 1st at ~4:30pmET.
#2: AACR’17(4-3-17) http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17.
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
"...Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
Reminder/Mar1: On/Feb12, AACR’17 revealed a 2nd(new) joint PPHM+MemSloan poster (study) that the Wolchok Lab will present on 4-3-17, “Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”. We only know the TITLE at this point. On Wed. 3-1-17 4:30pmET, AACR’17 will publish the Abstract Summaries at http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105&DetailItemID=539 . This has obviously be known for some time by Mem.Sloan & Peregrine – the deadline for “regular abstracts” was Nov. 17th, and for “late breakers” was Jan. 12th.
= = = = = = = = = = = = =WHAT WE DO KNOW:
The New PPHM+Mem.Sloan/Wolchok PS-Targeting Study was revealed via AACR’17 Abstracts pub. 2-12-17 at http://www.abstractsonline.com/pp8/#!/4292 . I’m learning more about this cancer immunotherapy approach called “Adoptive Cell Transfer (ACT)” that The Wolchok Lab & Peregrine are working on. It’s a bigger deal than I thought...
THIS IS THE NEW MSK/PPHM STUDY REVEALED BY THE AACR’17 ABSTRACTS: (see http://tinyurl.com/z47hb2s ):
#2: AACR’17(4-3-17) http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17.
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”...
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow ["Immunologic Checkpoint Blockade"]. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = =
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW MSK STUDY
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
MOST INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine targeting antibody in combination with tumor radiation and immune checkpoint blockade promotes anti-tumor activity in mouse B16 melanoma”
2. 4-3-17 #1651 “Targeting phosphatidylserine in combination with adoptive T cell transfer eliminates advanced tumors without off-target toxicities in a melanoma preclinical model”
NON-PEREGRINE:
3.(MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4.(MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5.(MSK & Cedars-Sinai/LA) 34-4-17 #4705 “CTLA4 blockade with HER2-directed therapy (H) yields clinical benefit in women undergoing radiation therapy (RT) for HER2-positive (HER2+) breast cancer brain metastases (BCBM)”
.
.
SITC’16 PPHM/Mem.Sloan Poster – same as AACR’17 4-2-17 #574…

Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, ImmunoVaccine…
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=128740654
Hutschi, the MDA/UTSW Bavi+DNX2401/Pancreatic article pub. 1-30-17 in AACR’s Mol-Cancer-Ther. was reported here on 2-1-17… This is MDA collaborating w/UTSW(R.Brekken) on Bavi preclin. studies, combining DNAtrix Corp's drug DNX-2401 w/Bavi.
DNAtrix: Houston TX ( http://www.dnatrix.com , Private Co.).
MORE here: http://tinyurl.com/hov4hfb
1-30-17/AACR(MolCancer): “Preclinical Evaluation of Sequential Combination of Oncolytic Adenovirus Delta-24-RGD and Phosphatidylserine-targeting Antibody [1N11=FhuBAVI] in Pancreatic Ductal Adenocarcinoma”
http://mct.aacrjournals.org/content/early/2017/01/28/1535-7163.MCT-16-0526
Rec/8-5-16, Rev/12-1-16, Acc/12-15-16, PUB/1-30-17
**DNX-2401: DNAtrix Corp., Houston TX
http://www.dnatrix.com (private company)
“DNAtrix’s lead product, DNX-2401, has demonstrated that DNX viruses exhibit a potent anti-tumor mechanism of action by (1) replicating in human tumors (2) eliciting tumor necrosis and (3) triggering an immune response.”
INTERESTING: Dr.Jedd.Wolchok is co-author of 5 AACR’17 Posters. 2 of the 5 are w/PPHM re: PS-Targeting. Only one other is with a biotech: Genocea Biosciences. The 4th is MSKCC ONLY, and the 5th is jointly w/Cedars-Sinai/LA. This is presuming that I searched properly (I think I did): http://www.abstractsonline.com/pp8/#!/4292 . If anybody can find any I missed, let me know and I’ll fix.
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine targeting antibody in combination with tumor radiation and immune checkpoint blockade promotes anti-tumor activity in mouse B16 melanoma”
2. 4-3-17 #1651 “Targeting phosphatidylserine in combination with adoptive T cell transfer eliminates advanced tumors without off-target toxicities in a melanoma preclinical model”
NON-PEREGRINE:
3.(MSK ONLY): 4-2-17 #874 Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy
4.(MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 Genome-scale neoantigen screening using ATLAS™ prioritizes candidate antigens for immunotherapy in a NSCLC patient
5.(MSK & Cedars-Sinai/LA) 34-4-17 #4705 CTLA4 blockade with HER2-directed therapy (H) yields clinical benefit in women undergoing radiation therapy (RT) for HER2-positive (HER2+) breast cancer brain metastases (BCBM)
= = = = = = = = = = = = = = = = = = = = = = = = = =
Apr1-5 2017: “AACR 2017”, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Full Abstracts to be posted to the AACR Online Itinerary Planner 3-1-17 4:30pm.
...TITLES Only ( http://www.abstractsonline.com/pp8/#!/4292 ):
4-2-17/1pm #574 - Session: CHECKPOINTS 1
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW MSK STUDY$$$
…...$$$See: More on the Significance of the New MSK/Wolchok+PPHM “ACT” study: http://tinyurl.com/h3ylrku
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
= = = = = = = = = = = = =
2-12-17: Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, ImmunoVaccine Inc… http://tinyurl.com/gof4kua
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
...MSK’s Dr. Jedd Wolchok states, ”Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchock states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
Re: The 2-23-17 StockNewsUnion “What’s Next” snippet… Most of the “articles” that pop up when Googling “Peregrine” are just robot things, and I ignore 98% of them. But this one has some real human input concerning potential partners for PS+ Exosomes Cancer Detection: AstraZeneca, Tesaro, Clovis...
2-23-17/StockNewsUnion.com: “What Next For Peregrine Pharmaceuticals After Exosomes Proof of Concept?”
…What comes next? With the positive proof of concept data [ 2-9-17: http://tinyurl.com/jhv57ua ], Peregrine Pharmaceuticals (NASDAQ:PPHM) has said that its sight is on developing the technology into an optimized diagnostic platform for clinical testing of cancer. To reach that goal, Peregrine said it is seeking strategic partners to help it develop & commercialize the platform. Those partners would be expected to inject money into the project for an equity stake in Peregrine Pharmaceuticals (NASDAQ:PPHM) or a cut of revenue from the sale of the platform. There are several large pharmaceutical companies developing their own ovarian cancer diagnostic systems are in need of such technology, so PPHM could pitch to them to join its exosomes-based diagnostic efforts.
POTENTIAL PARTNERS:
* AstraZeneca plc (NYSE:AZN https://www.astrazeneca.com - http://finance.yahoo.com/quote/AZN MktCap=$72B)
* Tesaro Inc. (NASDAQ:TSRO http://www.tesarobio.com - http://finance.yahoo.com/quote/TSRO MktCap=$9.5B)
* Clovis Oncology Inc. (NASDAQ:CLVS http://clovisoncology.com - http://finance.yahoo.com/quote/CLVS MktCap=$2.6B)
...are some of the major pharma brands that could be a fit for PPHM. The pharma giants are at various stages in the development of their ovarian cancer assets.”
http://stocknewsunion.com/what-next-for-peregrine-pharmaceuticals-nasdaqpphm-after-proof-of-concept/2043/
= = = = = = == = = = = = = = = = = =
PPHM's EXOSOME-BASED (PS Positive) CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17/PR: PS+ Exosomes Proof-of-Concept Data (N=44, Ovarian, Dr. Alan Schroit/UTSW etal) Published 1-22-17 in OncoTarget http://tinyurl.com/jhv57ua
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies.
...Dr. Stephen Worsley (VP/BusDev): “...we believe this has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
...PPHM’s PS Exosomes platform advantages outlined: UTSW/website & World Patent #WO/2016/201064: http://tinyurl.com/zbwr7cc
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
...Dr. Jeff Hutchins (VP/PreclinRES): "Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing pgm. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this pgm in 2017."
Exosomes-RNA.com posted the Exosomes PPHM/Schroit World-Patent App today. I sense that http://www.exosome-rna.com has a substantial readership...
http://www.exosome-rna.com/new-patent-application-published-diagnostic-test-for-early-stage-cancer/

Recall, on 2-10-17, they posted the news of the 2-9-17 PPHM Exosomes POC PR...
http://www.exosome-rna.com/proof-of-concept-data-for-a-novel-exosome-based-cancer-detection-platform-published/
...2-9-17: Press Release on 1-22-17 OncoTarget Article: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223

= = = = = = = = = = = = = = = = =
PPHM's EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17: Press Release on 1-22-17 OncoTarget Article: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/jg2k8pt
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
.
.
= = = = = = = = = = = = = = = = = = =
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, Filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“Detecting may comprise contacting the sample of step (b) with an exosome binding agent that may be the same or other phosphatidylserine binding agent than the one used in step (b), or a non-phosphatidylserine binding agent. Given that only PS-positive tumor exosomes are captured and available at this stage (after step b), the detecting agent may be an agent that binds to all exosomes regardless of their source… In general, normal tumor-free individuals have undetectable levels of PS-exosomes in an unconcentrated sample. In contrast, patients with tumors will typically exhibit values above 100 pg/50 \xh of sample in an unconcentrated sample. Thus, values greater than 50 pg/50 ui, plasma (or serum) are indicative of a malignancy. The method may further comprise obtaining said sample from the subject prior to step (a). The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy… In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

6-30-16: UTSW WEBSITE - “AVAILABLE TECHNOLOGIES” (#2932):

“A Blood-based Diagnostic Test for the Early Detection of Cancer” (Alan Schroit, S.Ward, A.Gazdar)
ALL: http://www.utsouthwestern.edu/about-us/administrative-offices/technology-development/industry/available-technologies.html
SCHROIT/EXOSOMES #2932: http://www.utsouthwestern.edu/edumedia/edufiles/about_us/admin_offices/technology_development/available_technologies/utsd-2932.pdf
This invention is a highly sensitive, blood-based assay system for the diagnosis of cancer. Tumors secret large quantities of cell membrane-derived exosomes into the blood stream of cancer patients. While all exosomes, whether from tumor cells or normal cells, share membrane epitopes similar to those found on the parent cell membrane, only tumor cell-derived exosomes express the membrane phospholipid, phosphatidylserine (PS), in the outer leaflet of the particle membrane. Based on data clearly showing that PS exposure is a unique signature of tumor-derived exosomes, the investigators developed a discovery platform for the screening & detection of tumor-derived exosomal PS signatures in patient blood. This novel tool provides:
1) A diagnostic marker that could dramatically improve the early detection of cancer.
2) Predicts whether a tumor is benign or malignant.
3) Could provide a predictive marker for response to therapy. Importantly, early detection of indolent neoplastic disease could significantly improve patient survival.
Currently, there are very few blood tests for cancer. These include alpha-fetoprotein for hepatocellular cancer, CA125 for ovarian cancer, and prostate specific antigen for prostate cancer. Although, high levels of these proteins might indicate the presence of neoplastic disease, none are diagnostic. Because our system detects a shared universal marker of neoplastic disease, it provides a viable method for detecting cancer of any type, in any organ.
PPHM Scientist Dr.Michael.Gray speaking today: CHI I-O Conference/SanFran
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & Immuno-Oncology”, SanFran
“Models & Approaches to Bring Combination Therapies to the Clinic”
Part of the 24th Intl. Molecular Medicine Tri-Conference
http://www.triconference.com/Pre-Clinical-Oncology-Models
CHI = Cambridge Healthtech Institute http://www.healthtech.com
Track: TUMOR MODELS FOR CANCER IMMUNOTHERAPY
2-21-17 11:15-11:45am: Michael Gray (Sr.Res.Sci.), Peregrine Pharmaceuticals - “Case Study: Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
SUMMARY: Phosphatidylserine (PS) exposure in tumors induces non-inflammatory signals which contribute to an immunosuppressive environment. Antibody blockade of PS activates immune responses by promoting M1 macrophages, maturation of dendritic cells and inducing adaptive T-cell responses. PS targeting antibodies enhance the anti-tumor activity of checkpoint antibodies in preclinical tumor models.
- - - - -
DR. MICHAEL GRAY (Principal Scientist, Peregrine 12-2014) - Prev: Quanticel Pharm, OSI; PhD/U-Pitt, Post-Doc/MDA https://www.linkedin.com/in/michael-gray-18447328
- - - - - -
BAVI MOA 5-11-16 Breast Cancer Res. article, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
Lead author: Michael Gray; Senior author: Bruce Freimark; Co-author: Christopher C. W. Hughes(U-Cal/Irvine)
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
PS+ Exosomes World-Patent: “Exosome-specific biomarkers can help distinguish the nature of the type of cancer… Some exemplary exosome biomarkers can be found above in Table 1”. I would think Dr. Schroit & Team are working on the “can help distinguish” part as we speak.
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“. . .The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy. . .
In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

= = = = = = = = = = = = = = = = =
PPHM's EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17: Press Release on 1-22-17 OncoTarget Article: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/jg2k8pt
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
I don’t think they’ll be news next week either. I think that the big buying is from people looking 6+MOS AHEAD (as the general stock market tends to do), to things like…
* The Exosomes Program(esp. given Dr. Alan Schroit’s reputation; POC done/advancing via partnership)
* Sunrise Biomarkers(#1=B2GPI, #2=Complement & IL-10 Pathways, #3=????)
* The MSK/Wolchok collab(incl. revelation by the new AACR’17 MSK abstract stating, “PPHM’s PStargeting + Adoptive Tcell Transfer(ACT) Eliminates Advanced Tumors without Off-Target Toxicities”)
* The Rutgers/Birge collab(ex: “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk) Regulates AKT-dep. Chemoresistance & PD-L1 Expression”)
* The Duke/Lyerly collab(ex: “Anti-PS + anti-PD-L1 leads to greater anti-tumor responses in TNBC")
* The ImmunoVaccine Inc. collab(“PStargeting Enhances Anti-Tumor Activity of DepoVax Tumor Vaccine”)
* The AZN & NCCN clinical collabs, and continuing Avid expansion/growth.
= = = = = = = = = = = = =Updated:
PPHM's EXOSOME-BASED (PS Positive) CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17/PR: PS+ Exosomes Proof-of-Concept Data (N=44, Ovarian, Dr. Alan Schroit/UTSW etal) Published 1-22-17 in OncoTarget http://tinyurl.com/jhv57ua
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies.
...Dr. Stephen Worsley (VP/BusDev): ”...we believe this has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
...PPHM’s PS Exosomes platform advantages outlined: UTSW/website and World Patent #WO/2016/201064: http://tinyurl.com/zbwr7cc
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
...Dr. Jeff Hutchins (VP/PreclinRES): "Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing pgm. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this pgm in 2017.”
NCCN Bavituximab Trials Announced 9-6-16 - To Begin "Early 2017" http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular (Bavi+RAD+Sorafenib), MOFFITT CANCER CENTER - PI: Jessica Frakes, MD - https://clinicaltrials.gov/ct2/show/NCT02989870 (start=4/30/17)
...#2: P1-2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. CANCER CENTER - PI: Elizabeth Gerstner, MD
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHN-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD
.
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-3-17 #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(2nd known study - revealed by AACR’17 Abstract):
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer (ACT) Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Lead author: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK), Co-author Jedd Wolchok, PPHM & MSKCC scientists. ( http://www.mskcc.org/research-areas/labs/jedd-wolchok )
NOTES:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
-----
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
-----
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
-----
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow ["Immunologic Checkpoint Blockade"]. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-2-17 #574 - Session: CHECKPOINTS 1 http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(1st known study – revealed at Nov’16/SITC16):
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
...OBVIOUSLY THIS IS A FOLLOWUP TO THE 11-14-16 SITC’16 PRESENTATION:
SITC’16: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (Joint Mem. Sloan Kettering/Wolchok Lab & PPHM)
...DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-4-17 #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model” (PPHM & Immunovaccine Inc.)
=> Genevieve Weir 1, Tara Quinton 1, Jeff Hutchins 2, Bruce Freimark 2, Marianne Stanford (VP/Res., Immunovaccine Inc.)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]; 2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
[Note: POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB?? 11-9-15 SITC'15: ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.” http://tinyurl.com/pbof95w ]
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 2-9-17/AACR article (Rutgers’ Dr. Raymond Birge, PPHM, Advanced Proteome Therapeutics, etal): “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk) Regulates AKT-dependent Chemoresistance & PD-L1 Expression” http://tinyurl.com/h4gdke3
…”These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors. Implications: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.”

= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 1-30-17 Mol.Cancer article: MDA/UTSW/Brekken/etal, “PreClin. Eval of DNX2401+FhuBAVI(1N11) for Pancreatic Cancer” http://tinyurl.com/hov4hfb
...Combo Delta-24-RGD + FhuBAVI “significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly & PPHM poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Title: ‘Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”’… Data showed that a combination of anti-PS & anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations w/paclitaxel, in the E0771 murine model of TNBC.”
Kensuke Kaneko 1, Takuya Osada 1, Bruce D. Freimark 2, Herbert Kim Lyerly ** (Duke Univ.)
1=Duke University, Durham, NC
2=Peregrine Pharmaceuticals, Inc.
**Dr. Herbert Kim Lyerly: https://immunology.duke.edu/people/herbert-kim-lyerly-md (George Barth Geller Professor, Duke Univ. MC)
PPHM’s Dr. Jeff Hutchins 10-24-16: “We plan to continue to work with our collaborators at Duke Univ. Medical Center to further study the therapeutic potential of PS-targeting agents in combination with checkpoint inhibitors like anti-PD-L1 and conventional therapies that augment immunotherapy mechanisms."
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray (PPHM): Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 7-27-16 Shaul/Brekken/Thorpe/etal PLOS ONE article: Fhu/Bavi vs. APS-related Pregnancy Complications & Thrombosis http://tinyurl.com/jlhrdg2
...”The potential clinical impact of 1N11 (Fhu Bavi) in patients with APS is substantial. Spring-boarding from the present discovery of 1N11 as a highly-effective, mechanism-based treatment for APS in a comprehensive series of mouse models of APS-related disorders, clinical studies of 1N11 now warrant consideration.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes(UCal-Irvine)-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article (PPHM/Friemark, U.Cal-Irvine/CW-Hughes - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
= = = = = = = = = = = = = = = = = = = = = = = = = =
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
...Authors: Rutgers(Birge/Kumar/Calianese), Peregrine(4), UTSW(Brekken/Schroit/Huang), Friedrich-Alexander-Univ./Germany(Martin Herrmann/Boeltz/Schett), Rio de Janeiro, Univ. College London
...”we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
= = = = = = = = = = = = = = = = = = = = = = = = = =
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Jedd Wolchok states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab & Swim Across America Lab at Memorial Sloan Kettering) states, ”A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab.”
.
.

.
.

Says Wolchok’s Lab: we’ve got something that “Eliminates-Advanced-Tumors Without Off-Target Off-Target Toxicities in a Melanoma Preclinical Model”… Quite an AACR’17 attention grabber!
All we know so far is that the MSKCC/Wolchok.Lab will be presenting on 4-3-17 at AACR’17 an abstract titled, “Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”.
We only know the TITLE at this point. On 3-1-17, AACR’17 will publish the Abstract Summaries at http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105&DetailItemID=539 . This has obviously be known for some time by Mem.Sloan & Peregrine – the deadline for “regular abstracts” was Nov. 17th, and for “late breakers” was Jan. 12th.
.
.
= = = = = = = = = = = = =WHAT WE DO KNOW:
The New PPHM+Mem.Sloan/Wolchok PS-Targeting Study revealed-via AACR’17 Abstracts – I’m learning more about this cancer immunotherapy approach called “Adoptive Cell Transfer (ACT)” that The Wolchok Lab & Peregrine are working on. It’s a bigger deal than I thought...
THIS IS THE NEW MSK/PPHM STUDY REVEALED BY THE AACR’17 ABSTRACTS: (see http://tinyurl.com/z47hb2s ):
#2: AACR’17(4-3-17) http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17. http://www.abstractsonline.com/pp8/#!/4292
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow ["Immunologic Checkpoint Blockade"]. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = =
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW MSK STUDY
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
...MORE on AACR'17: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=128740654
Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, ImmunoVaccine…
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=128740654
Known Upcoming Events (incl. AACR’17 New PPHM+MSK/Wolchok Study) => 4-3-17 #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities in Melanoma” (J.Wolchok, T.Merghoub, PPHM). Also added 2 new Avid events.
= = = = = = = = = = = = = = = =
Feb20-22: CHI’s 5th Translational Models in Oncology & I-O, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Feb21 11:15-11:45am: Michael Gray(Sr.Res.Sci.), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
Feb27-Mar2/Avid Booth #816: BioProcess Intl. West, SanFran https://lifesciences.knect365.com/bpi-west
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW MSK STUDY$$$
…...$$$SEE: More on the Significance of the New MSK/Wolchok+PPHM “ACT” study: http://tinyurl.com/h3ylrku
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins***(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
***Note: 1-4-17: Dr. Hutchins left Peregrine to become CSO of Heat-Biologics.
??Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/MidAPR, Full=May17)
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
.
.
= = = = = = = = = = = = = = =
MORE AACR’17 DETAILS (4 Abstracts):
AACR’17(4-3-17) reveals a 2nd Mem.Sloan Wolchok Lab/PPHM study.
Both PPHM+MemSloan abstracts are for Melanoma, and the lead author for both is Dr. Taha Merghoub, Co-Dir., Ludwig Collaborative Lab at MSK...
This new one, #1651, sounds exciting: “Targeting PS in Combination with Adoptive T Cell Transfer (ACT) Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”. Awaiting 3-1-17 for Full AACR abstract.
-----
AACR’17: 4-3-17 #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(2nd known study - revealed by AACR’17 Abstract):
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Lead author: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK), Co-author Jedd Wolchok, PPHM & MSKCC scientists. ( http://www.mskcc.org/research-areas/labs/jedd-wolchok )
NOTES:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
-----
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
-----
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
-----
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = =THE 1st KNOWN PPHM+MSK STUDY:
AACR’17(4-2-17 http://tinyurl.com/jxfm3hb ), same as SITC’16(Nov2016)
SITC’16: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AACR’17: “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
AACR’17 AUTHORS: Sadna Budhu, Olivier De Henau, Roberta Zappasodi, Rachel Giese, Luis F. Campesato, Christopher Barker, Bruce Freimark, Jeff Hutchins, Jedd D. Wolchok, Taha Merghoub (same list as SITC’16, except K.Schlunegger dropped/now at Pfenex, and MSKCC’s L.Campesato & R.Geise added)
...From the SITC’16 abstract: http://tinyurl.com/js3fca4
“We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer & survival benefit. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers.”
...From PPHM’s 11-14-16 SITC’16 PR: http://tinyurl.com/js3fca4
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
= = = = =THE OTHER 2 (of 4 total) AACR’17 PPHM ABSTRACTS:
4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
.
.

Preclin. Anti-PS work w/collab’s MemSloan,Duke,MDA,Rutgers,Wistar,ImmunoVaccine…
(updated with further info. on the new PPHM+MemSloan/Wolchok “PS-Targeting + Adoptive T Cell Transfer (ACT)” study revealed by the AACR’17 abstracts).
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-3-17 #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(2nd known study - revealed by AACR’17 Abstract):
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer (ACT) Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Lead author: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK), Co-author Jedd Wolchok, PPHM & MSKCC scientists. ( http://www.mskcc.org/research-areas/labs/jedd-wolchok )
NOTES:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
-----
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
-----
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
-----
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-2-17 #574 - Session: CHECKPOINTS 1 http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(1st known study – revealed at Nov’16/SITC16):
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
...From the SITC’16 abstract: http://tinyurl.com/js3fca4 **SEE 11-14-16 BELOW**
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: 4-4-17 #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff Hutchins 2, Bruce Freimark 2, Marianne Stanford (VP/Res., Immunovaccine Inc.)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]; 2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
[Note: POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB??? 11-9-15 SITC'15: ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.” http://tinyurl.com/pbof95w ]
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 2-9-17/AACR article (Rutgers’ Dr. Raymond Birge, PPHM, Advanced Proteome Therapeutics, etal): “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk)...” http://tinyurl.com/h4gdke3
…”These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors. Implications: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.”

= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA: 1-30-17 Mol.Cancer article: MDA/UTSW/Brekken/etal, “PreClin. Eval of DNX2401+FhuBAVI(1N11) for Pancreatic Cancer” http://tinyurl.com/hov4hfb
...Combo Delta-24-RGD + FhuBAVI “significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
...DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Title: ‘Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”’… Data showed that a combination of anti-PS & anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations w/paclitaxel, in the E0771 murine model of TNBC.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray (PPHM): Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 7-27-16 Shaul/Brekken/Thorpe/etal PLOS ONE article: Fhu/Bavi vs. APS-related Pregnancy Complications & Thrombosis http://tinyurl.com/jlhrdg2
...”The potential clinical impact of 1N11 (Fhu Bavi) in patients with APS is substantial. Spring-boarding from the present discovery of 1N11 as a highly-effective, mechanism-based treatment for APS in a comprehensive series of mouse models of APS-related disorders, clinical studies of 1N11 now warrant consideration.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes(UCal-Irvine)-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article (PPHM/Friemark, U.Cal-Irvine/CW-Hughes - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
= = = = = = = = = = = = = = = = = = = = = = = = = =
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
...Authors: Rutgers(Birge/Kumar/Calianese), Peregrine(4), UTSW(Brekken/Schroit/Huang), Friedrich-Alexander-Univ./Germany(Martin Herrmann/Boeltz/Schett), Rio de Janeiro, Univ. College London
...”we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
= = = = = = = = = = = = = = = = = = = = = = = = = =
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
= = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Jedd Wolchok states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab & Swim Across America Lab at Memorial Sloan Kettering) states, ”A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab.”
.
.

.
.

The New PPHM+Mem.Sloan/Wolchok PS-Targeting Study revealed-via AACR’17 Abstracts – I’m learning more about this cancer immunotherapy approach called “Adoptive Cell Transfer (ACT)” that The Wolchok Lab & Peregrine are working on. It’s a bigger deal than I thought...
THIS IS THE NEW MSK/PPHM STUDY REVEALED BY THE AACR’17 ABSTRACTS: (see http://tinyurl.com/z47hb2s ):
#2: AACR’17(4-3-17) http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17. http://www.abstractsonline.com/pp8/#!/4292
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
**From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
**Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
**12-12-2012: MSK Researchers Jedd Wolchok & Michel Sadelain Appointed to “Stand Up To Cancer” Immunology “Dream Team”
TWO APPROACHES TO ATTACKING CANCER:
The Immunology Dream Team will pursue 2 research techniques.
The 1st, being led by Dr. Wolchok, involves studying how a type of white blood cell called a T lymphocyte, or T cell, can kill cancer cells. Sometimes, the natural function of the T lymphocyte is blocked or not activated enough to attack cancer cells, allowing the cancer to grow. Part of the Dream Team’s focus will be to investigate ways to ensure that the T lymphocytes work properly in recognizing and killing cancer cells.
The 2nd immunotherapy approach, known as Adoptive Cell Transfer (ACT), involves removing some of a patient’s T cells, enhancing their cancer-fighting abilities and growing them in the laboratory, and then infusing the enhanced cells back into the patient. This can provide a patient with an army of immune cells specifically programmed to fight against cancer. This part of the Dream Team’s research, led by Dr. Sadelain, will investigate several ways to use ACT as a cancer therapy.
https://www.mskcc.org/blog/msk-researchers-appointed-stand-immunology-dream-team
= = = = = = = = = = = = =
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW MSK STUDY
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, ImmunoVaccine…
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=128637091
PD, Sunrise wasn't cancelled. The scheduled 12-7-16 Presentation on Biomarker #2 (“Complement & IL-10 Pathways) at WCLC’16(IASLC) by Drs. Gerber & Sanborn was Cancelled.
12-20-16/S.Diaz(per Cheynew post #282068): “There was no poster in Vienna. The team was working on a very tight timeframe and, despite their best efforts, couldn’t complete the data analysis in time. Given the crunch, we knew there was a possibility that we wouldn’t meet the timing, so we never issued a PR announcing that we would present. Hence no PR announcing that we did not present.” S.DIAZ/FOLLOWUP: ”Unfortunately, I don’t have a timeline for completion of the analysis or where/when it might be presented. We’ll certainly announce this as soon as we know for sure. Only Joe Shan attended the conference for Peregrine.”
PPHM’s Sunrise Biomarker Data (#1/Oct’16, #2/Dec’16/Delayed)
Summary of the 2 known Ph3/Sunrise Biomarker Data Presentations:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce” <=CANCELLED/”Anal.Not.Done(IR)” http://tinyurl.com/z8cq8vx
= = = = = = = = = = = = = = =DETAILS:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
10-10-16/PR:
“Peregrine Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at Eur. Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Addl. Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Addl. patient sample testing & analysis is ongoing and may result in other biomarkers of importance.
--------------
** Data presented at ESMO’16 demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approx. 30% of randomized patients) achieved a statistically significant, 5.5-mo. improvement (13.2 mos. vs. 7.7 mos.) in MOS as compared to patients in the ctl. group with the same range of B2GP1 levels [p = 0.049; HR=.67].
-------------
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP/Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific & medical conferences as the more results become available.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
= = = = = = = = = = = = = = = =
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefitting from Bavi+Doce” <=CANCELLED/”Anal.Not.Done(IR)”
...Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
ABSTRACT CONCLUSION:
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...” [Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9mos => 12.5mos.]
- - - - - - -
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
http://wclc2016.iaslc.org Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
ABSTRACT Book PDF: http://wclc2016.iaslc.org/wp-content/uploads/2016/12/WCLC2016-Abstract-Book_vF-WEB_revDec12.pdf
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with high complement activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [MOS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded indep. validation.
= = = = = = = = = =NOTE:
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ] “Founded in 2005, Biodesix discovers & commercializes cancer tests (diagnostics) that help patients & their doctors make more informed decisions about treatment based on a patient’s unique molecular profile.”
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
- - - - - - - - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
Summary of PPHM+RUTGERS(Dr. Raymond Birge) Collaborative Work
2-9-17/AACR/MolCancerRes, Birge+PPHM: “Phosphatidylserine Sensing by TAM Receptors Regulates AKT-dependent Chemoresistance & PD-L1 Expression”
Rec. 10-27-16, rev. 12-30-16, Acc 1-13-17, PUB. 2-9-17
*RUTGERS: Canan Kasikara, Sushil Kumar, Stanley Kimani, Wen-I Tsou, Ke Geng, Viralkumar Davra, Connor Devoe, Khanh-Quynh Nguyen, Anita Antes, Sergei V Kotenko, Raymond B. Birge [Lead Author http://birgelab.org ]
*MIT: Ganapathy Sriram [prev: Rutgers]
*ADVANCED PROTEOME THERAPEUTICS: Allen Krantz, Andrzej Wilczynski http://advancedproteome.com
*BOSTON UNIV: Grzegorz Rymarczyk(Consultant to Adv. Proteome Ther.)
*PEREGRINE: Cyril Empig, Bruce D Freimark, Michael Gray, Jeff Hutchins
*PFENEX INC: Kyle Schlunegger [prev: Peregrine]
http://mcr.aacrjournals.org/content/early/2017/02/09/1541-7786.MCR-16-0350
https://www.ncbi.nlm.nih.gov/pubmed/28184013
ABSTRACT:
Tyro3, Axl, and Mertk (collectively TAM receptors) are 3 homologous receptor tyrosine kinases (RTKs) that bind vitamin K-dependent endogenous ligands, Protein S (ProS) and Growth arrest specific factor 6 (Gas6), and act as bridging molecules to promote phosphatidylserine (PS)-mediated clearance of apoptotic cells (efferocytosis). TAM receptors are overexpressed in a vast array of tumor types, whereby the level of expression correlates with the tumor grade and the emergence of chemo- and radio-resistance to targeted therapeutics, but also have been implicated as inhibitory receptors on infiltrating myeloid-derived cells in the tumor microenvironment (TME) that can suppress host anti-tumor immunity. In the present study, we utilized TAM-IFNgammaR1 reporter lines and expressed TAM receptors in a variety of epithelial cell model systems to show that each TAM receptor has a unique pattern of activation by Gas6 or ProS, as well as unique dependency for PS on apoptotic cells and PS liposomes for activity. In addition, we leveraged this system to engineer epithelial cells that express WT TAM receptors, and show that while each receptor can promote PS-mediated efferocytosis, AKT-mediated chemo-resistance, as well as up-regulate the immune checkpoint molecule PD-L1 on tumor cells, Mertk is most dominant in the aforementioned pathways. Functionally, TAM receptor-mediated efferocytosis could be partially blocked by PS-targeting antibody 11.31 [***] and Annexin V, demonstrating the existing of a PS/PS-Receptor (i.e. TAM-receptor) /PD-L1 axis that operates in epithelial cells to foster immune escape. These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors.
IMPLICATIONS: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.
[***NOTE: Mab 11.31 is PGN632 (B2GPI-indep.), the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p ]

= = = = = = = = = = = = = =DR. BIRGE: “Cancer Metastasis and Immune Evasion”
Rutgers’ Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html

Interesting that one Birge/Rutgers Lab Member’s (Ke Geng) Theme/Mission is, “Regulation of Phosphatidylserine (PS) Externalization in the Tumor Micro-Environment; Role of PS as an Immunosuppressive Signal”.
http://birgelab.org/lab.html
Ke Geng - BS: Zhengzhou Univ. Medical School

= = = = = = = = = = = = =PRIOR PPHM+RUTGERS/R.BIRGE WORK:
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article('Cell Death & Differentiation Jrnl'), ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
Authors: Rutgers(Birge/Kumar/Calianese), Peregrine(4), UTSW(Brekken/Schroit/Huang), Friedrich-Alexander-Univ./Germany(Martin Herrmann/Boeltz/Schett), Rio de Janeiro, Univ. College London
ABSTRACT:
Apoptosis is an evolutionarily conserved and tightly regulated cell death modality. It serves important roles in physiology by sculpting complex tissues during embryogenesis and by removing effete cells that have reached advanced age or whose genomes have been irreparably damaged. Apoptosis culminates in the rapid and decisive removal of cell corpses by efferocytosis, a term used to distinguish the engulfment of apoptotic cells from other phagocytic processes. Over the past decades, the molecular and cell biological events associated with efferocytosis have been rigorously studied, and many eat-me signals and receptors have been identified. The externalization of phosphatidylserine (PS) is arguably the most emblematic eat-me signal that is in turn bound by a large number of serum proteins and opsonins that facilitate efferocytosis. Under physiological conditions, externalized PS functions as a dominant and evolutionarily conserved immunosuppressive signal that promotes tolerance and prevents local and systemic immune activation. Pathologically, the innate immunosuppressive effect of externalized PS has been hijacked by numerous viruses, microorganisms, and parasites to facilitate infection, and in many cases, establish infection latency. PS is also profoundly dysregulated in the tumor microenvironment and antagonizes the development of tumor immunity. In this review, we discuss the biology of PS with respect to its role as a global immunosuppressive signal and how PS is exploited to drive diverse pathological processes such as infection and cancer. Finally, we outline the rationale that agents targeting PS could have significant value in cancer & infectious disease therapeutics.
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w ...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
Known Upcoming Events (incl. AACR’17 New PPHM+MSK/Wolchok Study) - see details below.
Feb20-22: CHI’s 5th Translational Models in Oncology & I-O, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Feb21 11:15-11:45am: Michael Gray(Sr.Res.Sci.), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Apr1-5: AACR 2017, WashDC http://tinyurl.com/jxfm3hb (Abstract Summaries pub. 3-1-17)
...MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma”
...MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer Elims. Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW MSK STUDY(see below)
...PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
...IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor”
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins***(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
***Note: 1-4-17: Dr. Hutchins left Peregrine to become CSO of Heat-Biologics.
??Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/MidAPR, Full=May17)
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
= = = = = = = = = = = = = = = = = = = = = = = =
AACR’17(4-3-17) reveals a 2nd Mem.Sloan Wolchok Lab/PPHM study.
Both PPHM+MemSloan abstracts are for Melanoma, and the lead author for both is Dr. Taha Merghoub, Co-Dir., Ludwig Collaborative Lab at MSK...
This new one sounds exciting: “Targeting PS in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”. Awaiting 3-1-17 for Full AACR abstract.
NOTE:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
#2: AACR’17(4-3-17) **NEW PPHM/MSKCC STUDY** http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17. http://www.abstractsonline.com/pp8/#!/4292
Summary of the 2 known MSKCC/PPHM Anti-PS studies to date:
#1: SITC’16(Nov2016) => AACR’17(4-2-17 http://tinyurl.com/jxfm3hb )
SITC’16: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AACR’17: “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
AACR’17 AUTHORS: Sadna Budhu, Olivier De Henau, Roberta Zappasodi, Rachel Giese, Luis F. Campesato, Christopher Barker, Bruce Freimark, Jeff Hutchins, Jedd D. Wolchok, Taha Merghoub (same list as SITC’16, except K.Schlunegger dropped/now at Pfenex, and MSKCC’s L.Campesato & R.Geise added)
...From the SITC’16 abstract: http://tinyurl.com/js3fca4
“We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer & survival benefit. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers.”
...From PPHM’s 11-14-16 SITC’16 PR: http://tinyurl.com/js3fca4
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
= = = = =THE OTHER 2 (of 4 total) AACR’17 PPHM ABSTRACTS:
4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
.
.

Preclin. Anti-PS work w/collab’s MemSloan,Duke,MDA,Rutgers,Wistar,ImmunoVaccine…
AACR’17: 4-3-17 #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS http://tinyurl.com/z47hb2s
MemSloan Jedd Wolchok Lab+PPHM(2nd known study): “Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
Lead author: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK), Co-author Jedd Wolchok, PPHM & MSKCC scientists. ( http://www.mskcc.org/research-areas/labs/jedd-wolchok )
NOTE:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient. AACR’17: http://tinyurl.com/jxfm3hb
AACR’17: 4-4-17 #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff Hutchins 2, Bruce Freimark 2, Marianne Stanford (VP/Res., Immunovaccine Inc.)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]; 2=Peregrine Pharmaceuticals
[Note: POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB??? 11-9-15 SITC'15: ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.” http://tinyurl.com/pbof95w ]
BAVI MOA: 2-9-17/AACR article (Rutgers’ Dr. Raymond Birge, PPHM, Advanced Proteome Therapeutics, etal): “PS Sensing by TAM Receptors (Tyro3, Axl, Mertk)...” http://tinyurl.com/gna9q4v
…”These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors. Implications: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.”
BAVI MOA: 1-30-17 Mol.Cancer article: MDA/UTSW/Brekken/etal, “PreClin. Eval of DNX2401+FhuBAVI(1N11) for Pancreatic Cancer” http://tinyurl.com/hov4hfb
...Combo Delta-24-RGD + FhuBAVI “significantly inhibited tumor growth; further enhanced its anticancer activity; warrants further clinical evaluation...”
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
...MSK’s Dr. Jedd Wolchok states, ”Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Title: ‘Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”’… Data showed that a combination of anti-PS & anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations w/paclitaxel, in the E0771 murine model of TNBC.”
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray (PPHM): Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
BAVI MOA 7-27-16 Shaul/Brekken/Thorpe/etal PLOS ONE article: Fhu/Bavi vs. APS-related Pregnancy Complications & Thrombosis http://tinyurl.com/jlhrdg2
...”The potential clinical impact of 1N11 (Fhu Bavi) in patients with APS is substantial. Spring-boarding from the present discovery of 1N11 as a highly-effective, mechanism-based treatment for APS in a comprehensive series of mouse models of APS-related disorders, clinical studies of 1N11 now warrant consideration.”
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes(UCal-Irvine)-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article (PPHM/Friemark, U.Cal-Irvine/CW-Hughes - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchok states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
.
.

.
.

AACR’17(4-3-17) reveals a 2nd Mem.Sloan Wolchok Lab/PPHM study. Both are for Melanoma, and the lead author for both is Dr. Taha Merghoub, Co-Dir., Ludwig Collaborative Lab at MSK... This new one sounds exciting: “Targeting PS in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”. Awaiting 3-1-17 for Full AACR abstract.
NOTE:
“Adoptive Cell Transfer (ACT)” is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system, with the goal of improving immune functionality and characteristics. In cancer immunotherapy, T cells are extracted from the patient, genetically modified and cultured in vitro and returned to the same patient.
Summary of the 2 known MSKCC/PPHM Anti-PS studies to date:
#1: SITC’16(Nov2016) => AACR’17(4-2-17 http://tinyurl.com/jxfm3hb )
SITC’16: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AACR’17: “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
AACR’17 AUTHORS: Sadna Budhu, Olivier De Henau, Roberta Zappasodi, Rachel Giese, Luis F. Campesato, Christopher Barker, Bruce Freimark, Jeff Hutchins, Jedd D. Wolchok, Taha Merghoub (same list as SITC’16, except K.Schlunegger dropped/now at Pfenex, and MSKCC’s L.Campesato & R.Geise added)
...From the SITC’16 abstract: http://tinyurl.com/js3fca4
“We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer & survival benefit. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers.”
...From PPHM’s 11-14-16 SITC’16 PR: http://tinyurl.com/js3fca4
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
#2: AACR’17(4-3-17) http://tinyurl.com/jxfm3hb
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Jeff Hutchins 2, Bruce Freimark 2, Michael Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
***FULL ABSTRACTS TO PUB. AT AACR.COM 3-1-17. http://www.abstractsonline.com/pp8/#!/4292
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
.
.

The new PPHM/ImmunoVaccine collab via AACR’17: looking at IV’s website https://www.imvaccine.com , it’s clear preclin. work has been done combining their “DepoVax” drug with PPHM’s Anti-PS, either Bavi or Fully-Human Bavi (PGN635=1N11), or the “Duke” B2GPI-indep. mab PGN632=11.31, or maybe even BetaBodies? We’ll probably find out 3-1-17 when the AACR’17 full abstracts are released.
ImmunoVaccine’s DepoVax Platform https://www.imvaccine.com/depovax.php
“The cornerstone of all vaccine dev. efforts at Immunovaccine is the company’s DepoVax vaccine adjuvanting platform. DepoVax is a patented vaccine delivery formulation that provides controlled & prolonged exposure of antigens + adjuvant to the immune system. The result is a strong, specific and sustained immune response with the capability for single-dose effectiveness.
DEPOVAX IN CANCER:
Maintains remission and combats micro-metastases
*Provides strong & sustained polyfunctional T cell responses
*Specifically targets tumor cells”
= = = = = = = = = = = = = =
Apr1-5 2017: AACR 2017, WashDC http://tinyurl.com/jxfm3hb
PPHM+IMMUNOVACCINE:
AACR’17 4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
Thanks for finding the AACR’17 Abstract Titles, Cheynew. Didn’t realize the Titles already out. The Full Abstracts will be released March 1st. DELETE
AACR’17(April2-4): 4 PPHM Abstracts (2 w/MemSloan, 1 w/Immunovaccine). Add Immunovaccine Inc. (Halifax https://www.imvaccine.com ) to the list of collaborators.
Apr1-5 2017: “AACR 2017”, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Full Abstracts to be posted to the AACR Online Itinerary Planner 3-1-17 4:30pm.
...TITLES Only ( http://www.abstractsonline.com/pp8/#!/4292 ):
4-2-17/1pm #574 - Session: CHECKPOINTS 1
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
= = = = = = = = = = = = =
2-10-17: Preclin. Anti-PS work w/collab’s MemSloan, Duke, MDA, Rutgers, Wistar, UTSW, U.Cal/Irvine, Immunovaccine Inc. http://tinyurl.com/h5avlw3
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
2-9-17/AACR Article: R.Birge+PPHM, “PS Sensing by TAM Receptors Regulates AKT-dependent Chemoresistance & PD-L1 Expression”. Lead author, Rutgers' Dr. Raymond Birge. Also, 2 co-authors from Advanced Proteome Therapeutics, in addition to PPHM scientists.
2-9-17/AACR: “Phosphatidylserine Sensing by TAM Receptors Regulates AKT-dependent Chemoresistance and PD-L1 Expression”
Rec. 10-27-16, rev. 12-30-16, Acc 1-13-17, PUB. 2-9-17
*RUTGERS: Canan Kasikara, Sushil Kumar, Stanley Kimani, Wen-I Tsou, Ke Geng, Viralkumar Davra, Ganapathy Sriram, Connor Devoe, Khanh-Quynh Nguyen, Anita Antes, Sergei V Kotenko, Raymond B. Birge [Lead Author http://birgelab.org ]
*ADVANCED PROTEOME THERAPEUTICS: Allen Krantz, Andrzej Wilczynski http://advancedproteome.com
*BOSTON UNIV: Grzegorz Rymarczyk
*PEREGRINE: Cyril Empig, Bruce D Freimark, Michael Gray, Kyle Schlunegger, Jeff Hutchins
http://mcr.aacrjournals.org/content/early/2017/02/09/1541-7786.MCR-16-0350
https://www.ncbi.nlm.nih.gov/pubmed/28184013
ABSTRACT:
Tyro3, Axl, and Mertk (collectively TAM receptors) are three homologous receptor tyrosine kinases (RTKs) that bind vitamin K-dependent endogenous ligands, Protein S (ProS) and Growth arrest specific factor 6 (Gas6), and act as bridging molecules to promote phosphatidylserine (PS)-mediated clearance of apoptotic cells (efferocytosis). TAM receptors are overexpressed in a vast array of tumor types, whereby the level of expression correlates with the tumor grade and the emergence of chemo- and radio-resistance to targeted therapeutics, but also have been implicated as inhibitory receptors on infiltrating myeloid-derived cells in the tumor microenvironment (TME) that can suppress host anti-tumor immunity. In the present study, we utilized TAM-IFNgammaR1 reporter lines and expressed TAM receptors in a variety of epithelial cell model systems to show that each TAM receptor has a unique pattern of activation by Gas6 or ProS, as well as unique dependency for PS on apoptotic cells and PS liposomes for activity. In addition, we leveraged this system to engineer epithelial cells that express WT TAM receptors, and show that while each receptor can promote PS-mediated efferocytosis, AKT-mediated chemo-resistance, as well as up-regulate the immune checkpoint molecule PD-L1 on tumor cells, Mertk is most dominant in the aforementioned pathways. Functionally, TAM receptor-mediated efferocytosis could be partially blocked by PS-targeting antibody 11.31 [***] and Annexin V, demonstrating the existing of a PS/PS-Receptor (i.e. TAM-receptor) /PD-L1 axis that operates in epithelial cells to foster immune escape. These data provide a rationale that PS-targeting, anti-TAM receptor, and anti-PD-L1 based therapeutics will have merit as combinatorial checkpoint inhibitors. Implications: Many tumor cells are known to up-regulate the immune checkpoint inhibitor PD-L1. This study demonstrates a role for PS and TAM receptors in the regulation of PD-L1 on breast cancers cells.
---------
[***NOTE: Mab 11.31 is PGN632 (B2GPI-indep.), the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p ]
= = = = = = = = = = = = = = = = = = = =
Interesting that one Birge/Rutgers Lab Member’s Theme/Mission is, “Regulation of Phosphatidylserine (PS) Externalization in the Tumor Micro-Environment; Role of PS as an Immunosuppressive Signal”. http://birgelab.org/lab.html
Raymond Birge, PhD - Univ. of Connecticut, Post-doc: Rockefeller Univ.
“Cancer Metastasis and Immune Evasion”
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
http://birgelab.org => http://birgelab.org/biography.html
“The Birge laboratory conducts basic science focused on the eradication of cancer.”

Ke Geng - BS: Zhengzhou Univ. Medical School
“Regulation of Phosphatidylserine (PS) Externalization in the Tumor Micro-Environment; Role of PS as an Immunosuppressive Signal”

= = = = = = = = = = = = = = = = = = ==
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
CP, interesting that one Birge/Rutgers Lab Member’s Theme/Mission is, “Regulation of Phosphatidylserine (PS) Externalization in the Tumor Micro-Environment; Role of PS as an Immunosuppressive Signal”. http://birgelab.org/lab.html
Raymond Birge, PhD - Univ. of Connecticut, Post-doc: Rockefeller Univ.
“Cancer Metastasis and Immune Evasion”
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
http://birgelab.org => http://birgelab.org/biography.html
“The Birge laboratory conducts basic science focused on the eradication of cancer.”

Ke Geng - BS: Zhengzhou Univ. Medical School
“Regulation of Phosphatidylserine (PS) Externalization in the Tumor Micro-Environment; Role of PS as an Immunosuppressive Signal”

= = = = = = = = = = = = = = = = = = ==
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
2-9-17/PR: PS+ Exosomes Proof-of-Concept Data Published
...See below for known history of PPHM’s Exosome’s pgm – chronological order.
2-9-17: ”Recent Publication Highlights Proof-of-Concept Data Supporting the Diagnostic Potential of Phosphatidylserine-Positive Exosomes in Ovarian Cancer”
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
TUSTIN, Feb. 9, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the publication of positive proof-of-concept data for a novel exosome-based cancer detection platform. Results of the study, conducted at University of Texas (UT) Southwestern Medical Center, showed researchers were able to distinguish between healthy subjects and patients with ovarian tumors based on the levels of exosomes containing phosphatidylserine (PS) found in their plasma. Furthermore, analysis of the PS-positive exosome levels allowed researchers to distinguish between malignant and benign tumors. These data were recently published online by the peer-reviewed journal, Oncotarget, in a paper titled, "Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof-Of-Concept Study."
Peregrine is currently advancing the proprietary exosome-based cancer diagnostic technology, licensed from UT Southwestern Medical Center in July 2016 [7-14-16: http://tinyurl.com/zszd4fj ], with the goal of developing an optimized test for further clinical testing. As part of these efforts, the company is in the process of seeking a strategic partner for collaboration on developing and commercializing the technology. The platform is based on the diagnostic potential of tumor exosomes, which are small vesicles from tumor cells that are released into the blood as tumors grow. Tumor derived exosomes have PS on their surface as a detectable marker. It is believed that even small tumors begin to release PS-positive exosomes and thus the ability to detect these exosomes in the blood may be an indicator of the presence of a tumor.
In the study published by Oncotarget [1-22-17 FULL article: http://tinyurl.com/hlc68tq ], plasma samples from 34 patients with ovarian tumors and 10 healthy subjects were analyzed for the presence of PS-expressing exosomes in a blinded test. Results demonstrated that those patients with malignant ovarian cancer displayed significantly higher blood PS exosome levels than those with benign tumors (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups displayed significantly higher blood PS exosome levels than the healthy subjects (median 0.237 vs -0.158, p < 0.0001 and -0.027 vs -0.158, p=0.0002, respectively).
"These initial proof-of-concept results are encouraging as they appear to support the underlying concept that the measurement of PS-positive exosome levels in blood could be a simple way to detect and monitor cancer. While the work is still early, we think these data serve as an important first step in highlighting the diagnostic potential of this platform," said Steven W. King, President and CEO of Peregrine. "This type of diagnostic technology is particularly important in an area such as ovarian cancer, in which screening options are limited and the ability to detect the disease at an early stage is inadequate. We look forward to continuing to explore the potential of the technology platform in ovarian as well as other types of cancer."
"There is a significant and growing interest in the healthcare industry around the ability to detect cancer and monitor its progression with more readily accessible blood tests. With this area being one of the fastest growing segments of the oncology diagnostics market, we believe that our exosome-based technology represents a significant product development and licensing opportunity," stated Stephen Worsley, VP of Business Development at Peregrine. "Based on the fact that PS is a marker associated with a broad range of cancer types, we believe our platform has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = = = =
PPHM’s PS+ EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") – Chron. History
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
May2014/Article: J Immunol Methods, Thorpe/Schroit/etal, “A Novel ‘Salting-Out’ Procedure for the Isolation of Tumor-Derived Exosomes”
https://www.ncbi.nlm.nih.gov/pubmed/24735771
ABSTRACT:
“The last decade has seen an exponential growth in the number of exosome-related publications. Although many of these studies have used exosomes from biological fluids (blood, and ascites or pleural effusions) the vast majority employed vesicles isolated from large volumes of tissue culture supernatants. While several techniques are available for their isolation, all require a significant reduction in volume to obtain sufficient concentrations for study. One approach is to concentrate the medium before proceeding with their isolation, however, these procedures are very time consuming and require specialized laboratory equipment. Here we provide a new & effective method for the isolation of tumor-derived exosomes based on "charge neutralization" with acetate. We show that titration of tissue culture supernatants with 0.1M acetate to pH4.75 results in immediate precipitation of virtually all the exosomes. The precipitated exosomes can be washed to remove residual media and are readily "resolubilized" upon resuspension in acetate-free buffer at neutral pH. This simple cost effective method significantly increases the yield of exosomes from an unlimited quantity of culture supernatants. Exosomes isolated by this technique are indistinguishable from exosomes recovered by direct ultracentrifugation.”
2-27-15/U.S.PATENT: App#20150241431, filed 2-27-15 (Schroit/Thorpe)
Title: “Methods & Compositions for Isolating Exosomes”
http://tinyurl.com/Schroit-Thorpe20150241431
ABSTRACT:
“Disclosed are surprising new methods & compositions for isolating extracellular microvesicles such as exosomes, particularly disease-related and phosphatidylserine (PS)-positive extracellular microvesicles as exemplified by tumor- and viral-derived exosomes. The methods of the invention are rapid, efficient, cost-effective and, importantly, are suitable for use with large volumes of biological fluids and produce antigenically intact extracellular microvesicles & exosomes. The methods & compositions are based on the surprising use of acetate buffers to isolate large quantities of extracellular microvesicles, particularly tumor-derived exosomes, from solution, without damaging their morphological or functional properties or antigenicity.”
Inventors: Alan J. Schroit; Philip E. Thorpe; Shelley Fussey
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, Filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“Detecting may comprise contacting the sample of step (b) with an exosome binding agent that may be the same or other phosphatidylserine binding agent than the one used in step (b), or a non-phosphatidylserine binding agent. Given that only PS-positive tumor exosomes are captured and available at this stage (after step b), the detecting agent may be an agent that binds to all exosomes regardless of their source… In general, normal tumor-free individuals have undetectable levels of PS-exosomes in an unconcentrated sample. In contrast, patients with tumors will typically exhibit values above 100 pg/50 \xh of sample in an unconcentrated sample. Thus, values greater than 50 pg/50 ui, plasma (or serum) are indicative of a malignancy. The method may further comprise obtaining said sample from the subject prior to step (a). The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy… In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

6-30-16: UTSW WEBSITE - “AVAILABLE TECHNOLOGIES” (#2932):

“A Blood-based Diagnostic Test for the Early Detection of Cancer” (Alan Schroit, S.Ward, A.Gazdar)
ALL: http://www.utsouthwestern.edu/about-us/administrative-offices/technology-development/industry/available-technologies.html
SCHROIT/EXOSOMES #2932: http://www.utsouthwestern.edu/edumedia/edufiles/about_us/admin_offices/technology_development/available_technologies/utsd-2932.pdf
This invention is a highly sensitive, blood-based assay system for the diagnosis of cancer. Tumors secret large quantities of cell membrane-derived exosomes into the blood stream of cancer patients. While all exosomes, whether from tumor cells or normal cells, share membrane epitopes similar to those found on the parent cell membrane, only tumor cell-derived exosomes express the membrane phospholipid, phosphatidylserine (PS), in the outer leaflet of the particle membrane. Based on data clearly showing that PS exposure is a unique signature of tumor-derived exosomes, the investigators developed a discovery platform for the screening & detection of tumor-derived exosomal PS signatures in patient blood. This novel tool provides:
1) A diagnostic marker that could dramatically improve the early detection of cancer.
2) Predicts whether a tumor is benign or malignant.
3) Could provide a predictive marker for response to therapy. Importantly, early detection of indolent neoplastic disease could significantly improve patient survival.
Currently, there are very few blood tests for cancer. These include alpha-fetoprotein for hepatocellular cancer, CA125 for ovarian cancer, and prostate specific antigen for prostate cancer. Although, high levels of these proteins might indicate the presence of neoplastic disease, none are diagnostic. Because our system detects a shared universal marker of neoplastic disease, it provides a viable method for detecting cancer of any type, in any organ.
7-14-16/PR: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
VP JEFF HUTCHINS: “We are excited to enter into this licensing agreement with our long-term collaborators at UTSW. This technology offers a promising product development opportunity and aligns directly with the company's expertise with our proprietary PS-targeting platform and our longstanding CDMO capabilities around the development, qualification, and validation of in vitro analytical assays. As such, there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts. It is important to note that this dev. program will require minimal capital investment and has the potential to create significant value over the next 18mos., including potential partnering opportunities. As a result, we feel that today's licensing deal provides yet another important driver in our ongoing efforts to achieve profitability."
7-14-16/CC S.King PREPARED: “...In addition to bavituximab program which continues to have tremendous potential value, the company announced earlier today that it has licensed-in a Novel PS Exosome Technology, with the potential to detect & monitor cancer at an early stage through a simple blood test [ 7-14-16: http://tinyurl.com/zszd4fj ]. I recognize that some of you might think this theme is contrary to controlling spending as we move toward profitability, but in fact we believe that given our already existing knowledge base in Targeting PS and our already available infrastructure for developing & validating tests, that so a very modest capital investment we can quickly reach proof-of-concept with a goal of partnering this technology, which could then bring in addl. revenue, with another potential upside of the technology being that it can possibly be useful in the continued dev. of bavituximab. Jeff will talk more about this during his prepared remarks. Given the strategy of R&D targeted toward early partnering, will allow the company to continue its R&D activities with significant upside coming from partnering as we move to our profitability.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Hutchins PREPARED: “I'm very happy to be able to discuss a number of exciting developments in our preclinical group. First, as Steve discussed, we have executed the licensing agreement with UT Southwestern Medical Center, for novel exosome technology [ 7-14-16: http://tinyurl.com/zszd4fj ]. While many of you are familiar with exosomes, I’ll provide a brief overview for those who are not. Exosomes are cell-secreted vesicles, or mini-cells if you will, that are present in nearly all bodily fluids, including blood. Likewise, tumor-derived exosomes represent small pieces of tumor cells that are released into the blood as tumors grow. As well, these tumor-derived exosomes have Phosphatidylserine or PS on their surface as a marker and can also contain DNA, RNA and proteins as markers of malignant disease. It is believed that even small tumors begin to release PS-positive exosomes, and thus the ability to detect these exosomes in the blood may be an indicator of presence or progression of a tumor. The licensed technology is designed to detect & monitor PS-positive exosomes in a patient's blood sample, providing clinicians with detection & monitoring information regarding the presence & prevalence of cancer. These exosomes have PS flipped to the outside of the surface and demonstrate immunosuppressive activity, just as we find with tumor cells. Preliminary studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS-positive exosomes that are detected in the blood of cancer patients and the severity or extent of their disease burden. Given our in-house expertise in PS-targeting, we believe that we are uniquely qualified to advance this technology. As Steve stated, there are significant opportunities to use this technology as both a complimentary tool in bavituximab's ongoing development, which Joe will address later, as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of our partnering efforts. It is our goal to develop, optimize, and validate a functional detection & monitoring assay capable of detecting PS-positive exosomes from a simple blood sample, and, given the company's extensive experience in developing assays of this type, we do not anticipate the need for added personnel or any specialized equipment for this project. Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.”
...SUMMARY: “And lastly, we've in-licensed a new exosome technology for a minimal cost that leverages our existing in-house expertise and provides us with another opportunity for us to create value to product development. Together, we believe the strategy will provide success, as it will allow us to focus the majority of our resources on achieving our primary corporate goal, future sustainable profitability within 24mos. At same time, we will focus our R&D efforts on small early stage trials and development of the exosome technology in an effort to attract partners. We believe this strategy will allow us to build near-term revenues through Avid, while maintaining the potential for significant addl. value creation associated with our R&D efforts.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Shan PREPARED: “I'd first like to comment on our new exosome program. One of the most exciting aspects of this technology is the potential synergy that it offers with our bavituximab clinical dev. program. Through our ongoing work with bavituximab, we have gained significant understanding of PS-mediated immuno-suppression in cancer. The availability of the PS specific biomarker, which can be implemented in our planned future bavituximab clinical trials, aligns nicely with our refocused bavituximab dev. strategy aimed at generating the most meaningful data possible from small, early stage clinical trials to support partnering efforts. We are very anxious to bring this new technology to Peregrine and we look forward to the value it brings to our bavituximab program.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/Pantginis – SK: “(On the Exosome pgm), …it fits right in; we’re not having to hire addl. people, we’re not having to bring in addl. equipment. This really fits in with everything we are doing on both studying PS, as well as on the assay development side of the business. We think it’s actually going to be complementary. There are other technologies out there looking at exosomes; they're are all taking a very different approach to what we’re doing and we actually think they could be very complementary to each other. We also see a need, even as interest in exosomes begins to pick up, to actually utilize this in conjunction with other things that are in development.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/T.Yip – SK: “(On the Exosome pgm), the next step will be to validate that through patient samples. The beauty of this is that while you do need IRB approval of course, you’re not running really clinical trials, so this can be done in conjunction with either our ongoing trials or partners' trials, or there are many other sources of just receiving these types of blood samples. This gives us the ability to very quickly go through and test hundreds or thousands of patient samples as part of the validation process. At that point, we can zero in on what are the potential applications of the technology, outside of what we might do with our own PS Targeting programs – what would be the potential utility of this for patients. Our goal is not to become a diagnostics company, but to put this in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself. Our benefit at that point would become, hopefully, some residual royalties, milestones and what have you, which feeds back into our revenue goals of becoming profitable. So, we thought that this is a very attractive technology that just fits right in with what we’re doing and requires almost no addl. resources whatsoever…
Q2:/TY: ”when should we expect to see more preclin. data on this front?”… SK: “Our goal is, probably towards for the end of this year, to be in a position to have data that we can present. That will come in a lot of different formats, so it will be in conjunction with other ongoing studies that maybe taking place already to be standalone just on the diagnostic itself. So, you'll be hearing a lot about this, and one of the reasons when to get this news out there is because what you think sooner than later we will build a talk about this technology.” http://tinyurl.com/h8eqtg5
9-8-16-PR/FY17Q1: “Peregrine in-licensed a novel exosome technology from UTSW that has potential for cancer detection and monitoring applications. This technology aligns directly with the company's expertise, its proprietary PS-targeting platform and the bavituximab dev. program. As such, there are opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts.” http://tinyurl.com/jydtkoy
9-8-16/CC J.Hutchins PREPARED: “1st, an an update on our PS Exosome Program. As we announced in July, we executed a licensing agreement with UT Southwestern Medical Center for a novel exosome technology designed to detect PS-positive exosomes in a patient blood sample. It is our belief that this technology can provide clinicians with detection & monitoring information regarding the presence and the prevalence of cancer. Preliminary indep. studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS positive exosomes detected in the blood of cancer patients and the severity and extent of their disease burden. Given our in-house expertise in PS targeting, we believe that we are uniquely qualified to advance this technology. We believe there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of partnering efforts. It is our goal to develop, optimize and validate a functional screening assay capable of detecting PS positive exosomes in a blood sample and to initiate partnering discussions for commercialization of the pgm in 2017. We are on track to achieve this goal and we look forward to providing further updates.” http://tinyurl.com/jydtkoy
10-13-16/ASM REPORT BY ATTENDEE COPPER888: “...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...” http://tinyurl.com/jx7ouay

12-12-16/CC: No mention of Exosomes! http://tinyurl.com/hhn4gga
1-22-17/OncoTarget Article: PS-Exosomes: PPHM SAB’r Dr. Alan Schroit: Proof-of-Concept Datasupports the “high diagnostic power” of PS-Positive Exosomes in Ovarian Malignancies. ...Basically, Dr. Schroit (cell membrane guru) & his UTSW team used a variant of fully-human Bavi (1N11) to detect the PS on the exosomes…
FROM PG.8: “The data summarized in Fig. 4 [N=44] show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
1-22-17 Oncotarget:
“Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study”
Lea J 1, Sharma R 2, Yang F 2, Zhu H 1, Ward ES 3,4, Alan J. Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/jlrkxma ) 1,3
1 Harold Simmons Comprehensive CC, UTSW-MC/Dallas
2 Hamon Center for Therapeutic Oncology Res., UTSW-MC/Dallas
3 Dept of Immunology, UTSW-MC/Dallas
4 Dept of Molecular & Cellular Medicine, Texas A&M Univ. Health Science Ctr
https://www.ncbi.nlm.nih.gov/pubmed/28122335
ABSTRACT:
There are no suitable screening modalities for ovarian carcinomas (OC) and repeated imaging and CA-125 levels are often needed to triage equivocal ovarian masses. Definitive diagnosis of malignancy, however, can only be established by histologic confirmation. Thus, the ability to detect OC at early stages is low, and most cases are diagnosed as advanced disease. Since tumor cells expose phosphatidylserine (PS) on their plasma membrane, we predicted that tumors might secrete PS-positive exosomes into the bloodstream that could be a surrogate biomarker for cancer. To address this, we developed a highly stringent ELISA that detects picogram quantities of PS in patient plasma. Blinded plasma from 34 suspect ovarian cancer patients and 10 healthy subjects were analyzed for the presence of PS-expressing vesicles. The nonparametric Wilcoxon rank sum test showed the malignant group had significantly higher PS values than the benign group (median 0.237 vs. -0.027, p=0.0001) and the malignant & benign groups had significantly higher PS values than the healthy group (median 0.237 vs -0.158, p<0.0001 and -0.027 vs -0.158, p=0.0002, respectively). ROC analysis of the predictive accuracy of PS-expressing exosomes/vesicles in predicting malignant against normal, benign against normal & malignant against benign revealed AUCs of 1.0, 0.95 and 0.911, respectively. This study provides proof-of-concept data that supports the high diagnostic power of PS detection in the blood of women with suspect ovarian malignancies.
FULL ARTICLE (13pgs):
HTML: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=14795&path%5B%5D=47251
PDF: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=download&path%5B%5D=14795&path%5B%5D=47248
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
.
PDF EXCERPTS 1-22-17/OncoTarget/A.Schroit/PPHM-Exosomes (5 pgs of the 13):

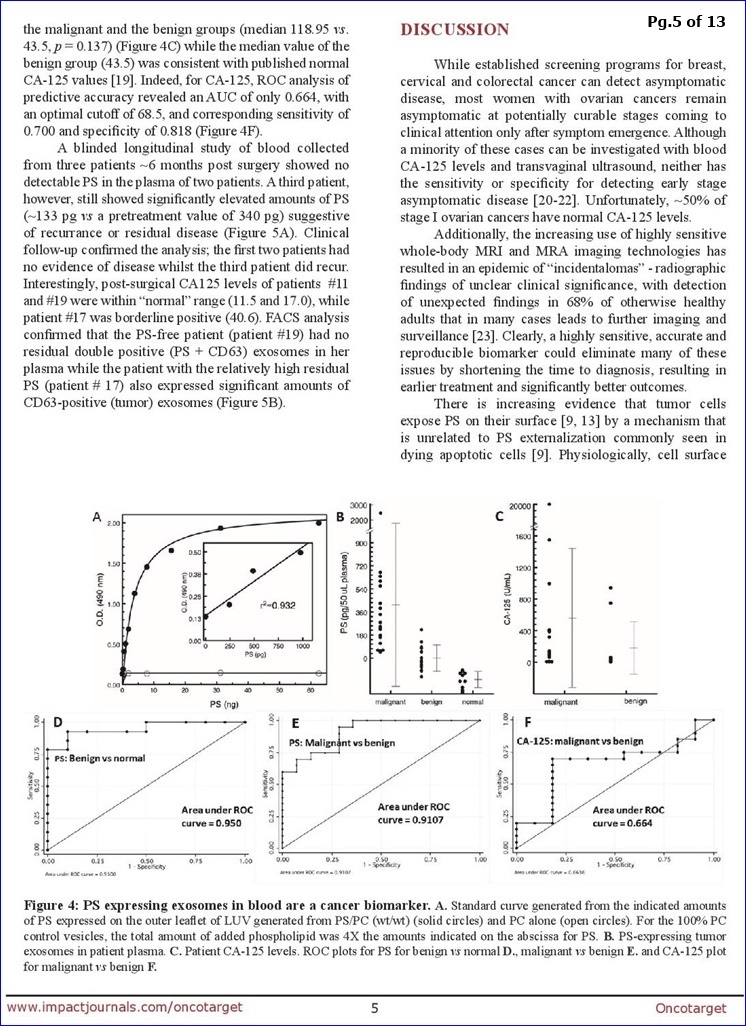



2-9-17: Press Release on 1-22-17 OncoTarget Article http://tinyurl.com/jhv57ua
...Stephen Worsley (VP/BusDev): “Based on the fact that PS is a marker associated with a broad range of cancer types, we believe our platform has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
PPHM’s Exosomes Technology: UTSW-DESC & World Patent #WO/2016/201064
6-30-16: UTSW WEBSITE - “AVAILABLE TECHNOLOGIES” (#2932):

“A Blood-based Diagnostic Test for the Early Detection of Cancer” (Alan Schroit, S.Ward, A.Gazdar)
ALL: http://www.utsouthwestern.edu/about-us/administrative-offices/technology-development/industry/available-technologies.html
SCHROIT/EXOSOMES #2932: http://www.utsouthwestern.edu/edumedia/edufiles/about_us/admin_offices/technology_development/available_technologies/utsd-2932.pdf
This invention is a highly sensitive, blood-based assay system for the diagnosis of cancer. Tumors secret large quantities of cell membrane-derived exosomes into the blood stream of cancer patients. While all exosomes, whether from tumor cells or normal cells, share membrane epitopes similar to those found on the parent cell membrane, only tumor cell-derived exosomes express the membrane phospholipid, phosphatidylserine (PS), in the outer leaflet of the particle membrane. Based on data clearly showing that PS exposure is a unique signature of tumor-derived exosomes, the investigators developed a discovery platform for the screening & detection of tumor-derived exosomal PS signatures in patient blood. This novel tool provides:
1) A diagnostic marker that could dramatically improve the early detection of cancer.
2) Predicts whether a tumor is benign or malignant.
3) Could provide a predictive marker for response to therapy. Importantly, early detection of indolent neoplastic disease could significantly improve patient survival.
Currently, there are very few blood tests for cancer. These include alpha-fetoprotein for hepatocellular cancer, CA125 for ovarian cancer, and prostate specific antigen for prostate cancer. Although, high levels of these proteins might indicate the presence of neoplastic disease, none are diagnostic. Because our system detects a shared universal marker of neoplastic disease, it provides a viable method for detecting cancer of any type, in any organ.
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, Filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“Detecting may comprise contacting the sample of step (b) with an exosome binding agent that may be the same or other phosphatidylserine binding agent than the one used in step (b), or a non-phosphatidylserine binding agent. Given that only PS-positive tumor exosomes are captured and available at this stage (after step b), the detecting agent may be an agent that binds to all exosomes regardless of their source… In general, normal tumor-free individuals have undetectable levels of PS-exosomes in an unconcentrated sample. In contrast, patients with tumors will typically exhibit values above 100 pg/50 \xh of sample in an unconcentrated sample. Thus, values greater than 50 pg/50 ui, plasma (or serum) are indicative of a malignancy. The method may further comprise obtaining said sample from the subject prior to step (a). The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy… In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

= = = = = = = = = = = = = = = = =
PPHM's EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
2-9-17: Press Release on 1-22-17 OncoTarget Article: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/jg2k8pt
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”