Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Damn...
Yahoo needs to find a solution to these bots....
Oh yeah...
I forgot...
Allowing people to have multiple IDs on a stock forum isn't all that respectable when you come to think of it...
Who am I?
What ID will I use now to post this message?
Lets use the one that best describes Yahoo....
Happy Trading,
Danny
Nanotechnology Roadmap
http://www.nanonextnl.nl/images/stories/downloads/Nanotechnology%20in%20the%20topsectors.pdf
This roadmap covers the whole of planned activities in the field of nanotechnology in relation to activities within the HTSM roadmaps and other top sectors for the period of 2012-2020 and is part of the Innovationcontract of the top sector HTSM. The proposed innovations items have been determined in close consultation between industry concerned, knowledge institutes, government and social institutions.
I'm not really maintaining this board anymore, so if anyone feels a calling to moderate this board, feel free to do so...
You can find me on the QTMM board.
Thanks for posting, company is working in solar technology which have lot of possibilities.
Top Ten reasons to buy QTMM
Author: ih8aloss
QTMM is the Quantum Materials Corp. and its subsidiary Solterra Renewable Technolgies, Inc. They are the GAME CHANGERS in the technology for Quantum Dot manufacturing and Solar Cells. This is a ground breaking developer and market leader for the production of Quantum Dots in the next generation of printed electronics technologies, Medical diagnostic applications and the Flexible Solar Power Industy for starters.
1. Quantity, Quality, Uniformity and Price of the Tetrapod Quantum Dots is unmatchable by any existing or foreseeable company. Not to mention they can make Quantum Dots (QD) heavy metal free (Cadmium Free) and of other biologically inert materials making them the #1 market leader in QD production.
2. The stock is SIGNIFICANTLY UNDERVALUED and affordable to the small investor looking for a chance at that life changing dream.
3. The single most important secret to building a stock market fortune is identifying a unique growth business poised to dominate a mass market. Parallels can be drawn between the silicon semiconductor revolution in the 70's by Intel and Quantum Materials Corp's new Tetrapod Quantum Dot semiconductors of the future.
4. QTMM - Quantum Materials Corp is still too new and small for institutional investors to be interested.
5. Quantum Dots are the future for nano technology advances projected to generate trillions (not billions per Lux research) in many industry discoveries that have literally been held back by the difficulty in manufacturing quantum dots. Quantum Materials Corp can now produce quantities (100KGrams per day) to support product commercialization for the various industries mentioned in reason #9
6. Quantum Materials Corp appears to be positioning to spin off Solterra Renewable Technologies, Inc. (http://www.solterrasolarcells.com). With the establishment of the 2nd web site (http://www.qdotss.com) this looks to be on track. Should that happen for every share of QTMM you own you will receive a % of the current subsidiary Solterra Renewable Technologies, Inc.
7. QTMM/Solterra's innovative Quantum Dot based solar cell technology for the Flexible Solar Power Industry is replacing the silicon wafer - based solar cell industry.
8. University of ARIZONA and RICE University have both issued EXCLUSIVE WORLDWIDE LICENSING agreements to the Quantum Materials Corp. and Solterra Renewable Technologie, Inc
9. This GAME CHANGER has a nice risk to rewards ratio, supported by additions like involvement in the LED/OLED technology, Optoelectronics, Biomed, Computer Memory, Lighting and Security industries, not to mention plans to develop a 1GW solar farm in the Middle East by 2015.
10. If you read the PRESS RELEASES and Solterra1 blog http://solterra1.wordpress.com/ you'll "See The Light" and why this company is so exciting.
Hi PK. If you like Solar, check out QTMM!
Quantum Materials Corporation (QTMM)
I just saw you're new to iHub... So Welcome!
You will love this board!
Solar energy is todays need, solar energy is renewable energy, petrol, diesel and gas are non renewable energy source one day these are finish so we should focus on renewable energy source and solar energy is the good way to get energy.
Hi Solar, welcome to my little board.
Doesn't Rice have to defend their patent? I don't think it's up to QMC to do this, right? So we shouldn't have the problems Evident had...
Free Patent Infringment in Texas??
This May be the Key Reason for Stealth Re: Dr. Wong's Patent..
I Referenced Evident Technologies Once Before from a DDHawk Post..
Thought I Would Bring Discussion Over Here vs QTMM Bd..
Researchers develop process to make cotton both water repellent and UV resistant
April 11, 2011 by Bob Yirka
Researchers at Northeast Normal University in China, have come up with a three-step process that when applied to cotton material results in a fabric that is both waterproof and very highly UV resistant.
In a recent paper published in ACS Applied Materials & Interfaces, Lingling Wang and his associates describe a method of using zinc oxide nanorods and zinc oxide crystallites to create a coating that bonds to cotton fiber resulting in a material that its creators hope will help meet the needs of a world that wants multi-functional clothes; in this case, clothes that are "green" (don't need cleaning) and help protect the wearer’s skin from harmful ultraviolet radiation.
The process, using off the shelf materials, including the cotton material, was begun by soaking a piece of cotton cloth in ZnO nano-crystals for 12 hours, then putting it in a Teflon autoclave (high pressure steamer) that was saturated in a zinc acetate hydrate solution for 48 hours at 95° C. This first step resulted in the dissolution and then re-crystallization of the ZnO nano-crystals which gave it a ZnO seed layer. Afterwards, the material was allowed to dry for ten minutes.
The next step was to place the seeded cotton back into the autoclave, this time in an aqueous solution of zinc nitrate hydrate and hexamethylenediamine (HMT) for 5 hours at 95° C. This step gives the material its water proofing abilities. Afterwards it’s rinsed and allowed to dry again.
The final step was to dip the material alternately in polyethylenimine (PEI) and silicate solutions creating a PEI/silica layer (shell).
The result is a material that is not only waterproof but is able to block out UV radiation from the sun to such an extent that it would rate a Ultraviolet Protection Factor (UPF) of 101.15 if the chart went that high. The rating is in effect, double the highest possible rating on the normal chart, and considerably higher than consumers are accustomed to seeing on suntan lotion bottles.
Though testing has not been done to see how the new process will hold up under real world conditions, the knowledge gained from the success of this procedure will no doubt advance the science of developing new clothes that will provide far more benefits to us all than simply covering our bodies.
Thin Sheet of Diamond Has Worlds of Uses
April 5 2011
A new technique from Argonne National Laboratory creates thin diamond films that are helping industry save energy and could even be used in heart and eye implants.
Even as royalty set diamonds into crowns and rings, engineers lusted after the gems for different reasons: diamonds are stronger than any other natural material and are excellent electrical insulators and heat conductors. Today they are widely used in industry and factories. But the diamond supply is limited, and while artificial diamonds can be made in gem form, they have been hard to synthesize in thin films.
A new technique invented at Argonne National Laboratory creates thin films of diamond with grains so small they're called ultrananocrystalline diamond films. The films can be applied to an astounding array of surfaces and uses, ranging from better seals on pumps to heart pump walls so smooth that dangerous blood clots don't form. The grains of diamond in the film are just five nanometers across—about a billion of them would fit inside one red blood cell.
The innovation sprang from DOE Office of Science-funded research, when materials scientists Dieter Gruen, Alan Krauss, Orlando Auciello, and John Carlisle developed a gas-based method to form the diamond films to precise standards.
Gruen filed the original patent on the concept in 1991 and the second patent two years later, after he demonstrated the concept worked. He got the idea in 1990 when a paper by Wolfgang Kratschmer and Donald Huffman appeared in the journal Nature describing how to make large amounts of Buckminsterfullerene, a carbon-60 molecule shaped like a soccer ball and named after Buckminster Fuller, an architect who popularized the geodesic dome. The discovery of Buckminsterfullerene, also known as "buckyballs," earned Robert Curl, Harold Kroto, and Richard Smalley the 1996 Nobel Prize in chemistry. Initially, buckyballs could be produced only in small amounts in the gas phase, but Kratschmer and Huffman's method showed how to make them in quantity.
"When I read that paper," Gruen said, "I immediately went into my lab and modified a used titanium sublimation pump by installing two graphite electrodes, one at either end. I filled it with an argon atmosphere and struck an arc between the carbon electrodes. The result was a sublimed carbon deposit that contained copious amounts of buckyballs."
Gruen quickly became a major supplier of buckyballs to his Argonne colleagues while continuing his own research, which focused on buckyball fragmentation—how they break up when they collide with other molecules and with surfaces. He found they break up by spitting out a series of carbon dimers—two carbon atoms linked together. For example, in an excited mixture of carbon-60 and argon gas, collisions cause carbon-60 to emit a dimer and become carbon-58. In subsequent collisions, carbon-58 becomes carbon-56, which becomes carbon-54, etc.—each time emitting a new carbon dimer.
Gruen linked this fragmentation method with recent results from Japanese studies of diamond structure and realized he could use buckyballs as a source of carbon dimers to grow a diamond surface using chemical vapor deposition.
His next step was to introduce buckyballs into a microwave plasma machine with an argon atmosphere. When he examined the deposit, he found a thin diamond film composed of nanocrystals and dubbed it "ultrananocrystalline diamond."
Gruen's discovery and implementation of this new growth mechanism for CVD diamond is responsible for the existence of ultrananocrystalline diamond (UNCD). The worldwide diamond community recognizes his newfound method for synthesizing UNCD films, as distinct from conventional microcrystalline diamond films, as having had a major impact on the field of diamond CVD. In 2000, the Materials Research Society presented him with the MRS Medal for the synthesis and characterization of UNCD films.
The UNCD coating technology captures most of the natural properties of diamond in thin-film form: the highest hardness of any diamond film demonstrated today, an extremely low friction coefficient and surface adhesion, very high electron emission, chemical inertness, biocompatibility, and extremely high conductivity when doped with nitrogen or boron. Each film's properties can be precisely tailored.
UNCD films can be used in a broad range of applications from macro to nanodevices, such as energy-saving ultra-low friction and wear coatings for mechanical pump seals and tools; high-performance microelectromechanical and nanoelectromechanical system-based telecommunication devices; the next generation of high-definition flat panel displays; in vivo biomedical implants; and biosensors.
The technology was licensed to Advanced Diamond Technologies Inc., based in Romeoville, Ill., which is providing UNCD film and materials integration solutions to a variety of industries developing a broad range of applications.
When used in mechanical pump seals, the diamond coating improves the pumps' reliability, durability, and ability to prevent fluids from escaping into the environment. The surface of UNCD coatings is so smooth that it reduces the friction present when the seals run one upon each other, and this saves up to 20 percent of the energy used running the pump. The UNCD-coated seals are now commercialized worldwide through several manufacturers of seals (see photo). The coating has also been successfully used to develop atomic force microscope tips, to coat wafers that can be used to develop a new generation of microelectromechanical and nanoelectromechanical systems and as coatings on bearings of mixers used in the manufacturing of pharmaceutical and food-grade chemicals.
A U.S. Department of Defense agency is working to use the films to create sensors that would take quick readings to detect chemical and biological threats in water. Diamond, unlike most other materials, can bond with biomolecules like E. coli, Salmonella, Listeria, and other pathogens in water to detect their presence. The detectors could even be miniaturized so that anyone, such as soldiers, police, or public health officers, could carry them.
Finally, because diamond is chemically inert, it doesn't react with biological human tissue or body fluids—and the body doesn't reject the diamond as a foreign material. For this reason, UNCD films can be used in implants, including heart pumps to treat heart failure.
By Jared Sagoff
Major advance in understanding how nanowires form
Public release date: 28-Mar-2011
New insights into why and how nanowires take the form they do will have profound implications for the development of future electronic components. PhD student Peter Krogstrup from the Nano-Science Center at the University of Copenhagen is behind the sensational new theoretical model, which is developed in collaboration with researchers from CINAM-CNRS in Marseille.
One of the most important components in future electronic devices will likely be based on nanocrystals, which are smaller than the wavelength of the light our eyes can detect. Nanowires, which are extremely thin nanocrystal wires, are predicted to have a predominant role in these technologies because of their unique electrical and optical properties. Researchers around the world have been working for years to improve the properties of these nanowires. With his research, PhD student Peter Krogstrup at the Niels Bohr Institute, University of Copenhagen has laid the foundations for a greater understanding of nanowires. With that comes the potential for improving their performance, which will bring the research closer to being applied in the development of solar cells and computers. In the latest edition of Physical Review Letters he describes how, under certain conditions, nanowires form a crystal structure that really should not be possible, seen from an energy perspective.
"Crystals will always try to take the form in which their internal energy is as little as possible. It is a basic law of physics and according to it these nanowires should have a cubic crystal structure, but we almost always see that a large part of the structure is hexagonal," explains Peter Krogstrup, who has been working with the theory in recent years.
Catalyst particle shape is the key
In order to explain why and when these crystals become hexagonal, Peter Krogstrup has, as part of his doctoral dissertation, examined the shape of the catalyst particle (a little nano-droplet), which controls the growth of the nanowires. It appears that the shape of the droplet depends on the amount of atoms from group 3 in the periodic system, which make up half of the atoms in the nanowire crystal. The other half, atoms from group 5 in the periodic system, are absorbed by the drop and hence the atoms organize themselves into a lattice, and the nanowire crystal will grow.
"We have shown that it is the shape of the droplet, which determines what kind of crystal structure the nanowires obtain and with this knowledge it will be easier to improve the properties of the nanowires," explains Peter Krogstrup and continues: "The crystal structure has an enormous influence on the electrical and optical properties of the nanowires and you would typically want them to have a certain structure, either cubic or hexagonal. The better nanowires we can make the better electronic components we can make to the benefit of us all," says Peter Krogstrup, whose research is conducted in collaboration with the firm SunFlake A/S, which is located at the Nano-Science Center at the University of Copenhagen. The company is working to develop solar cells of the future based on nanowires.
TU Delft identifies huge potential of nanocrystals in fuel cells
Published on 28 March 2011
The addition of extremely small crystals to solid electrolyte material has the potential to considerably raise the efficiency of fuel cells. Researchers at TU Delft were the first to document this accurately. Their second article on the subject in a very short time was published in the scientific journal, Advanced Functional Materials.
Electrolyte
The researchers at the Faculty of Applied Sciences at TU Delft were concentrating their efforts on improving electrolyte materials. This is the material between two electrodes, for example in a fuel cell or a battery. The better the characteristics of the electrolyte, the better, more compactly or more efficiently the fuel cell or battery works.
Solid matter
The electrolyte is usually a liquid, but this has a number of drawbacks. The liquid has to be very well enclosed, for example, and it takes up a relatively large amount of space. "It would therefore be preferable to have an electrolyte made of solid matter," says PhD student Lucas Haverkate. "Unfortunately though, that has disadvantages as well. The conductivity in solid matter is not as good as it is in a liquid."
Traffic jam on the motorway
"In a solid matter you have a network of ions, in which virtually every position in the network is taken. This makes it difficult for the charged particles (protons) to move from one electrode to another. It’s a bit like a traffic jam on a motorway. What you need to do is to create free spaces in the network."
Nanocrystals
One of the ways of achieving this, and therefore of increasing conductivity in solid electrolytes, is to add nanocrystals (of seven nanometres to around fifty nanometres), of Titanium Dioxide. "A characteristic of these TiO2 crystals is that they attract protons, and this creates more space in the network." The nanocrystals are mixed in the electrolyte with a solid acid (CsHSO4). This latter material 'delivers' the protons to the crystals. "The addition of the crystals appears to cause an enormous leap in the conductive capacity, up to a factor of 100," concludes Haverkate.
Similarity
This remarkable achievement by TU Delft has already led to two publications in the scientific journal Advanced Functional Materials. Last December, Haverkate published an article on the theory behind the results. His fellow PhD student, Wing Kee Chan, is the main author of a second item that appeared in the same publication this week. Chan focused on the experimental side of the research. "The nice thing about these two publications is that the experimental results and the theoretical underpinning strongly complement each other," says Haverkate.
Neutrons
Chan carried out measurements on the electrolyte material using the neutron diffraction method. This involves sending neutrons through the material. The way in which the neutrons are dispersed makes it possible to deduce certain characteristics of the material, such as the density of protons in the crystals. Haverkate: "It is the first time that measurements have been taken of solid-material electrolytes in this way, and on such a small scale. The fact that we had nuclear research technologies at the Reactor Institute Delft at our disposal was tremendously valuable."
Temperature
However, the combination of TiO2 and CsHSO4 does not mark the end of the search for a suitable solid-material electrolyte. Other material combinations will be tested that may achieve better scores in the area of stability, for example. Professor Fokko Mulder, who is Haverkate’s and Chan’s PhD supervisor, says. "At this stage, we are more concerned about acquiring a fundamental understanding and a useful model, than the concrete issue of finding out what the most suitable material is. It is important that we identify the effect of nanocrystals, and give it a theoretical basis. I think there is great potential for these electrolytes. They also have the extra benefit of continuing to function well over a wide range of temperatures, which is of particular relevance for applying them in fuel cells."
Cellulosic nanocrystals from biomass waste for thermoplastics for automotive applications
By Green Car Congress on 03/28/2011
Scientists in Brazil are studying the production and incorporation of cellulosic nanocrystals (also known as cellulose whiskers) derived from several sources such as pineapple, curaua, banana and coir into thermoplastics matrix composites for automotive applications. They described the work, which could lead to stronger, lighter, and more sustainable materials for cars and other products, at the 241st National Meeting & Exposition of the American Chemical Society (ACS) in Anaheim.
Cellulose nanocrystals are the crystalline domains of cellulosic sources. They have the potential to improve, among others, the mechanical, optical, and dielectric properties of these polymeric matrices, and have been of significant research interest for a number of years.
Some of these nano-cellulose fibers are almost as stiff as Kevlar, but unlike Kevlar and other traditional plastics, which are made from petroleum or natural gas, nano-cellulose fibers are completely renewable.
The properties of these plastics are incredible. They are light, but very strong—30% lighter and 3-to-4 times stronger. We believe that a lot of car parts, including dashboards, bumpers, side panels, will be made of nano-sized fruit fibers in the future. For one thing, they will help reduce the weight of cars and that will improve fuel economy.
So far, we’re focusing on replacing automotive plastics. But in the future, we may be able to replace steel and aluminum automotive parts using these plant-based nanocellulose materials.
—Alcides Leão, Ph.D., study leader, São Paulo State University
Welcome to the board Gnike!
The natural world is a gift to science, to explore and imitate.
The nanoworld is just opening up to us, and amazing things are about to happen!
Nanotechnology is the next revolution, after the digital revolution!
I created this board to discus all nanotechnology, but my focus is mainly on QTMM, Solterra.
Check it out, you won't regret it!
Make sure you look at our management team, which is world class!
Solterra Renewable Technologies Inc. is singularly positioned to lead the development of truly sustainable and cost-effective solar technology as the first company to introduce a new dimension of cost reduction by replacing silicon wafer based solar cells with low cost highly efficient Quantum Dot based solar cells.
I just saw that this was your first message on iHub. You will love this board! And don't worry, after a few days you'll be able to post 15 free messages a day. Just post a messages ones in a while.
Welcome and enjoy!
Here are a few video's about Nanotech to start with.
Nanomaterials for next-generation ultrafast lithium storage
Nanowerk Spotlight, January 21, 2011
Nanomaterials are key in developing high-performance energy storage technologies for mobile phone batteries and notebooks providing long battery times. Lithium-ion batteries are not only used in mobile gadgets but are currently being intensively developed worldwide for use in electric vehicles. As lithium-ion batteries are charged, large amounts of lithium ions are held in the anode. When the battery is used, these ions migrate to the cathode, sending electrons through the circuit. Traditional anode materials like graphite have a fairly low storage capacity and release rate, so finding alternatives is key to making batteries that last longer and produce more power.
Titanium dioxide is regarded as one of the ideal candidates for high-rate anode materials, owing not only to its structural characteristics and special surface activity, but also to its low cost, safety, and relatively low environmental impact.
Researchers in Singapore have developed a facile system to fabricate sandwich-like carbon-supported stacked titanium dioxide nanosheets, in which carbon pillars create open channels for fast lithium ion diffusion and the ultrathin framework renders the storage of lithium almost exclusively on the surface. This work provides a new route to design the electrode materials for quick-charging lithium ion batteries.
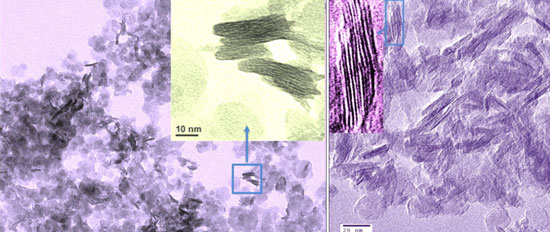
TEM images of the as-synthesized layered titanate nanosheets (left) and the carbon-supported stacked titanium dioxide nanosheets obtained after annealing at 350 °C for 2 hours (right). (Source: Xuewei Liu, Nanyang Technological University)
"The lack of open channels in bulk titanium dioxide is the main drawback that restricts its capacity and rate capability for reversible lithium insertion and extraction," Xuewei Liu, an assistant professor at Nanyang Technological University (NTU), explains to Nanowerk. "A reduction in the effective size and construction of open channels in the material are the main strategies currently employed to increase the rate performance. The capacity of ultrafine TiO2 nanocrystals and nanotubes, for example, is significantly enhanced at lower rates. However, their capacity and cycle life deteriorate dramatically at higher rates."
Liu notes that, in this respect, significant efforts have recently been made on the fabrication of anatase TiO2 nanosheets with exposed highly reactive (001) facets. These TiO2 nanosheets are shown to be an excellent host structure for lithium insertion and extraction due to the presence of exposed (001) facets and short path along the [001] direction for lithium ion diffusion.
"However, high-pressure systems including flammable solvent and corrosive additives (bases or acids) were often employed in solvothermal methods" says Liu. "There is still potential danger in the high-temperature and high-pressure systems when low boiling-point inflammable solvents are used. In addition, the nanosheets with exposed smooth facets easily overlap with each other because of the Ostwald ripening process, which leads to a decrease of surface area. To conquer these obstacles, we designed our simple ionic-liquid system."
Reporting their findings in a recent issue of Advanced Materials ("Sandwich-Like, Stacked Ultrathin Titanate Nanosheets for Ultrafast Lithium Storage"), Liu's group, working with Lou Xiong Wen's team at NTU's College of Engineering, synthesized sandwich-like carbon-supported stacked TiO2 nanosheets (CTNSs) on a large scale by a facile and clean route.
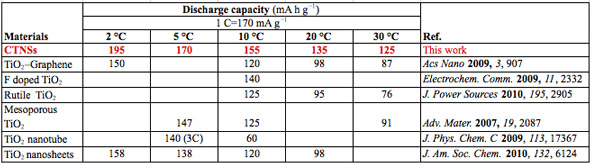
Comparison with existing methods and materials (1 C = 170 mA g-1) (Source: Xuewei Liu, Nanyang Technological University)
Owing to the unique textural features, i.e., stacked nanosheets with open channels, the storage of lithium in these CTNSs mainly takes place on surface, which is completely different from the insertion of lithium into bulk anatase TiO2 materials.
"This pseudocapacitive insertion and extraction renders remarkable improvement in high-rate charging and discharging" says Liu. "Moreover, the in situ derived carbon can significantly stabilize these stacked TiO2 nanosheets, leading to an excellent cycle life. We thus anticipate the promising use of this material in high-power lithium-ion batteries."
Liu summarizes the advantages of the new anode material: 1) Clean synthetic systems: Low pressure and safe; 2) The ionic liquid system is low-cost and facile to be synthesized; 3) High yield: the product could be synthesized in large scale and easy to mass-production; 4) Unique structure with superior performance: The sandwich structure provides open channels that are very suitable for fast lithium storage. The table above shows that the material has excellent capacities at low rates and high rates. The full charging time at 30 °C is only ~1.3 minutes.
Since the development of ultrafast-charging and high-rate electrode materials with low-cost is still a big challenge for next-generation lithium ion batteries, the NTU team will continue to focus their research on the development of novel and low-cost electrode materials for next-generation lithium ion batteries.
Nanocrystals of rock salt into lead telluride create breakthrough thermoelectric
January 18, 2011
Iridium is attractive for improving flash memory chips
One of the rarest metals on Earth may be an excellent option for enabling future flash memory chips to continue to increase in speed and density, according to a group of researchers in Taiwan.
December 14, 2010
"Incorporating nanocrystals of iridium into the critical floating gate portion of flash memory designs shows both excellent memory properties as well as stability in the high temperatures used in processing such semiconductor devices," says the research team leader, Wen-Shou Tseng of Taiwan's Center for Measurement Standards, Industrial Technology Research Institute. The research results appears in the journal Applied Physics Letters, which is published by the American Institute of Physics. His colleagues included students and professor at the nearby National Chiao Tung University and Chung Hua University.
This team chose iridium -- a hard, dense and corrosion-resistant metal in the platinum family that is one of the rarest metals found in the earth's crust -- because unlike most alternatives, it has two desired properties: Iridium holds its electrons strongly (it has a high "work function", which is well-known to correlate with excellent memory properties), and its melting point of nearly 2,500 degrees Celcius is well beyond the 900 C annealing temperature that many chips must survive during manufacturing. Fortunately only a billionth of a billionth of a gram of iridium would be needed for each gate.
Researchers worldwide are investigating new ways to improve the popular flash memory, which is the nonvolatile memory chip design used in virtually all digital cameras and mobile electronics and, increasingly, in solid-state drives for laptop computers. The easiest way for future flash memories to hold more data and read/write faster, is to shrink the dimensions of the existing chip design, including the floating gate. But today's gate design has already progressed to the point where it cannot get much smaller before it can no longer retain the electrical charges that actually store the data. Nanocrystals have been proposed as a rather simple change that can improve memory chip performance without changing the tried-and-true floating-gate design.
In recent years, many different metals have been investigated for their nanocrystal potential. Nickel and tungsten, for example, are attractive for, respectively, a high work function and thermal stability. But they and other elements lack both needed properties. It is rare, indeed, that iridium has both needed qualities, Tseng says.
Sigma-Aldrich and Nanosys Enter Into Distribution Agreement to Supply Materials to the Research Community.
Global Availability of Silicon Nanowires via Aldrich(R) Materials Science Paves Way for Innovation and Application Development Across Areas Such as Bio-Informatics, Electronics and Clean Energy
Nov. 29, 2010
Sigma-Aldrich today announced a distribution agreement with Nanosys, Inc., an advanced materials architect. Under the agreement, Sigma-Aldrich, through its Aldrich Materials Science business, is now distributing Nanosys-created silicon nanowires to the global scientific research community for the development of new applications in technologies such as solid-state lighting, electronics and energy storage.
"As Aldrich Materials Science continues to expand its nanomaterials offer, we are pleased to introduce an innovative line of silicon nanowires that includes undoped silicon nanowires as well as the higher conductivity, boron-doped nanowires," said Dr. Kaushik Patel, Product Manager, Aldrich Materials Science. "The applications of these nanowires are, we believe, truly innovative and revolutionary. The fabrication of nanowires is challenging; therefore, a consistent and high-quality supply of these materials from Aldrich Materials Science is expected to enable scientists to accelerate their research to further develop a variety of new high-technology applications that can continue to open up exciting end product markets. In all aspects of the Aldrich Materials Science business, from manufacturing and custom R&D to distribution of materials from leading-edge technology companies such as Nanosys, we strive to fulfill our mission of focusing on materials so that our research customers can focus on results."
"Architected and synthesized at the atomic level, our materials deliver exponential improvements in efficiency and performance," added Jason Hartlove, CEO of Nanosys. "This new distribution agreement with Sigma-Aldrich puts our technology directly into the hands of researchers who are doing exciting work in labs around the world, driving the use of architected material solutions in a new generation of markets and applications."
Nanosys silicon nanowires, which demonstrate semiconductor properties, are comprised of single crystal silicon grown using the vapor liquid solid (VLS) process to a diameter of 150 nm and a length of 20 micrometer. The materials are available undoped or as activated boron-doped p-type silicon nanowires with 1x10(19) /cm(3) doping at ends and a 3 micrometer intrinsic region in the center of the wire. Polydispersed silicon nanowires of various lengths that are designed for use in energy research are also expected to be available beginning January 2011.
Across an increasingly diverse range of applications, silicon nanowires can deliver tangible benefits and significant advances. Examples include:
Electronics applications - sensors can be constructed from nanowire arrays that act as a highly sensitive 'e-skin' which could be used to restore the sense of touch to patients with prosthetic limbs or to enable robotic systems to 'sense' pressure.
Solar cells - Research conducted earlier in 2010 found that fabricating thin films from ordered arrays of vertical silicon nanowires significantly increased the light-trapping properties in solar cells, compared to conventional silicon thin film photovoltaics.
Batteries - Nanowire technology is enabling the production of batteries that are more economical, longer lasting, smaller and more environmentally friendly. Nanowires are used to increase the surface area for electrolyte reaction, thereby enabling greater power in a smaller overall footprint.
To learn more about Aldrich Materials Science, silicon nanowires and other new nanomaterials, please visit http://www.sigma-aldrich.com/newnano.
Cautionary statement: The foregoing release contains forward-looking statements that can be identified by terminology such as "is expected to enable," "can continue to open," "are also expected to be available," "can deliver" or similar expressions, or by expressed or implied discussions regarding potential future revenues from products derived therefrom. You should not place undue reliance on these statements. Such forward-looking statements reflect the current views of management regarding future events, and involve known and unknown risks, uncertainties and other factors that may cause actual results to be materially different from any future results, performance or achievements expressed or implied by such statements. There can be no guarantee that silicon nanowires will achieve any particular levels of revenue in the future. In particular, management's expectations regarding silicon nanowires could be affected by, among other things, unexpected regulatory actions or delays or government regulation generally; the Company's ability to obtain or maintain patent or other proprietary intellectual property protection; competition in general; government, industry and general public pricing pressures; the impact that the foregoing factors could have on the values attributed to the Company's assets and liabilities as recorded in its consolidated balance sheet, and other risks and factors referred to in Sigma-Aldrich's current Form 10-K on file with the US Securities and Exchange Commission. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those anticipated, believed, estimated or expected. Sigma-Aldrich is providing the information in this press release as of this date and does not undertake any obligation to update any forward-looking statements contained in this press release as a result of new information, future events or otherwise.
About Sigma-Aldrich: Sigma-Aldrich is a leading Life Science and High Technology company committed to Enabling Science to Improve the Quality of Life. Its chemical and biochemical products and kits are used in scientific research, including genomic and proteomic research, biotechnology, pharmaceutical development and as key components in pharmaceutical, diagnostic and other high technology manufacturing. The Company has customers in life science companies, university and government institutions, hospitals, and in industry. Over one million scientists and technologists use its products. Sigma-Aldrich operates in 40 countries and has 7,700 employees providing excellent service worldwide. For more information about Sigma-Aldrich, please visit its award-winning Web site at http://www.sigma-aldrich.com.
About Nanosys, Inc.: Nanosys, Inc. is an advanced material architect, harnessing the fundamental properties of inorganic materials into process ready systems that can integrate into existing manufacturing to produce vastly superior products in lighting, electronic displays and energy storage. In 2010, Nanosys commercialized its quantum dot technology with the QuantumRail(TM), a process-ready component that improves LED backlit display color gamut and efficiency, signing partnerships with LG Innotek and Samsung. Nanosys is currently working with major battery manufacturers to improve lithium-ion battery capacity using its SiNANOde(TM) silicon composite additive that, when added to the anode side, improves capacity by 40 percent. SiNANOde(TM) -enhanced batteries are expected to be available in 2011.
Ever-Sharp Urchin Teeth May Yield Tools That Never Need Honing
ScienceDaily (Dec. 26, 2010)
To survive in a tumultuous environment, sea urchins literally eat through stone, using their teeth to carve out nooks where the spiny creatures hide from predators and protect themselves from the crashing surf on the rocky shores and tide pools where they live.

Sea urchin teeth are pictured in situ. New research by Pupa Gilbert, a physics professor at the University of Wisconsin-Madison, and her colleagues reveals how the sea urchin's teeth are always sharp, despite constant grinding and scraping to create the nooks that protect the marine animal from predators and crashing waves. (Credit: Photo courtesy of Pupa Gilbert)
The rock-boring behavior is astonishing, scientists agree, but what is truly remarkable is that, despite constant grinding and scraping on stone, urchin teeth never, ever get dull. The secret of their ever-sharp qualities has puzzled scientists for decades, but now a new report by scientists from the University of Wisconsin-Madison and their colleagues has peeled back the toothy mystery.
Writing in the journal Advanced Functional Materials, a team led by UW-Madison professor of physics Pupa Gilbert describes the self-sharpening mechanism used by the California purple sea urchin to keep a razor-sharp edge on its choppers.
The urchin's self-sharpening trick, notes Gilbert, is something that could be mimicked by humans to make tools that never need honing.
"The sea urchin tooth is complicated in its design. It is one of the very few structures in nature that self-sharpen," says Gilbert, explaining that the sea urchin tooth, which is always growing, is a biomineral mosaic composed of calcite crystals with two forms -- plates and fibers -- arranged crosswise and cemented together with super-hard calcite nanocement. Between the crystals are layers of organic materials that are not as sturdy as the calcite crystals.
"The organic layers are the weak links in the chain," Gilbert explains. "There are breaking points at predetermined locations built into the teeth. It is a concept similar to perforated paper in the sense that the material breaks at these predetermined weak spots."
The crystalline nature of sea urchin dentition is, on the surface, different from other crystals found in nature. It lacks the obvious facets characteristic of familiar crystals, but at the very deepest levels the properties of crystals are evident in the orderly arrangement of the atoms that make up the biomineral mosaic teeth of the sea urchin.
To delve into the fundamental nature of the crystals that form sea urchin teeth, Gilbert and her colleagues used a variety of techniques from the materials scientist's toolbox. These include microscopy methods that depend on X-rays to illuminate how nanocrystals are arranged in teeth to make the sea urchins capable of grinding rock. Gilbert and her colleagues used these techniques to deduce how the crystals are organized and melded into a tough and durable biomineral.
Knowing the secret of the ever-sharp sea urchin tooth, says Gilbert, could one day have practical applications for human toolmakers. "Now that we know how it works, the knowledge could be used to develop methods to fabricate tools that could actually sharpen themselves with use," notes Gilbert. "The mechanism used by the urchin is the key. By shaping the object appropriately and using the same strategy the urchin employs, a tool with a self-sharpening edge could, in theory, be created."
The new research was supported by grants from the U.S. Department of Energy and the National Science Foundation. In addition to Gilbert, researchers from the University of California, Berkeley; Argonne National Laboratory; the Weizmann Institute of Science; and the Lawrence Berkeley National Laboratory contributed to the report.
First observation of new type of 'sea urchin' shaped nano-crystalline aggregate
Oct 4th, 2010
Dr. Ujjal Gautam, an ICYC-MANA Researcher at the International Center for Materials Nanoarchitectonics (MANA), National Institute for Materials Science, in joint research with the Jawaharlal Nehru Center for Advanced Scientific Research (JNCASR), which is located in Bangalore, India, discovered a "new type of crystalline aggregate," in which small rod-shaped crystals are mutually attached according to a unique relationship.
This crystalline aggregate consists of rod-shaped nanocrystals of zinc oxide (ZnO). Zinc oxide is an excellent semiconductor,absolutely non-toxic and compatible with living organisms and can be employed in numerous applications, such as transparent electronics, light condensers, piezoelectronics, cosmetics, and others. In the crystalline aggregates described here, a large number of ZnO nanorods bristle from the central core like pencil-leads, ultimately taking a shape that resembles a sea urchin. Crystalline aggregates exist everywhere and can be used in many devices. For example, in the field of thin film technology, research is being carried out with the aim of creating ordered stacked layers of crystalline substances. In the natural world, astonishing control can be seen in the biomineralization process which produces elaborate crystalline aggregates such as those in Coccolithophores. Research on crystalline aggregates does not aim simply at understanding and imitating the mechanism of growth of crystals in the natural world. It is also extremely important for organizing substances as required by many applications, particularly in small regions where manipulation of the individual constituents is difficult.
The newly-discovered law in connection with the formation of this crystalline aggregate is based on a property of zinc oxide called polarity. This means that the two opposite ends of a ZnO rod-shaped crystal are positively electrically charged and negatively electrically charged respectively. Until now, it had been unknown that when ZnO rod-shaped crystals form aggregates, whether alignment of this polarization occurs or not. It is now discovered that, in one aggregate, all of the ZnO rods grew with a polarization orientation, so that all the positively charged ends point outward (and in this case, the negatively charged tips are fixed at the center), or vice versa. As another important fact, the two types of aggregates are mutually related by a unique and simple law of symmetry. If the respective ZnO rods in one aggregate are turned upside down, their polarity reverses, and that aggregate changes to the other type of aggregate. This unique relationship between crystalline aggregates was not known previously.
This new law of aggregation is important in the development of new properties. Although all of the crystal rods are the same (i.e., have identical physical properties), because the charges of their respective tips are different, different properties exist in aggregates with positive tips and those with negative tips. It is not possible to obtain the same properties with a single piece substance or a random assembly of crystal rods without considering these polarities.
A large number of other important substances exist which also possess polarity and form aggregates with similar shapes, for example, gallium nitride, which is used in light emitting diodes. Accordingly, it is possible that the property of natural polarity control is applicable to all such crystalline aggregates. This is a completely new and unprecedented phenomenon. High expectations are placed on the future development of applications as substances with unique properties which had not existed in the past.
New Nanowires Act Like LEDs
September 30, 2010 in Energy Efficiency, General Technology
In one of those happy research accidents that lead to new breakthroughs, scientists at the National Institute of Standards and Technology (NIST) have developed a new kind of nanowire that produces light, much like the glow produced by LED technology. The team was actually fine tuning a new method for manufacturing ultra thin or nanoscale wires, hoping to create a process that yields a product with uniform traits, when their experiment began to light up.
The Trouble with Nanowires
Typically, nanowires are grown on a base material or substrate. The process involves depositing molecules such as zinc oxide in the form of a gas. The nanowires then “grow” vertically, like bristles on a brush. The problem is, they grow so densely that it is difficult to pick out the ones with better characteristics. Also, since the wires only touch the substrate at one end, their properties are not uniformly distributed.
A New Method of Growing Nanowires That Glow
The NIST team came up with a solution, which is to grow the nanowires horizontally. They converted gold into nanoparticles by superheating it, then manipulated zinc oxide nanocrystals into pushing the gold particles along the substrate, forming nanowires. Because the wire touches the substrate at all points, its characteristcs are more uniformly influenced than in the vertical growth method. When the researchers increased the size of the gold particle, the wires grew a fin-like nanowall which allowed electrons to flow, giving off a light similar to that of an LED.
Glowing Nanowires vs. LEDs
The NIST researchers envision uses for the new light-emitting nanowires in chip-sized “laboratories” and other miniature devices for specialized purposes. Though large-scale applications are a possibility, that seems pretty remote at the present, leaving LEDs in the lead for now in terms of providing an energy saving solution to lighting needs. LEDs are already lending themselves to large scale applications in street lighting and parking garages, for example, and new research is yielding more powerful LEDs, as well as new technologies for boosting the efficiency and lifespan of LED fixtures.

Breakthroughs Are Slow but Steady in Quantum Computers
Michio Kaku on September 29, 2010
One day, quantum computers may replace the standard silicon chips found in all computers around the world. In fact, by 2020 to 2025, transistors will be so small and will generate so much heat that standard silicon technology (and Moore's law, which states that computer power doubles every 18 months) may eventually collapse. Silicon Valley could become a new Rust Belt in 10 to 15 years, with profound implications for the world economy. Who will want to upgrade their computers and buy new products if computers are just as fast today as they were last year?
Hence, there is intense interest in alternatives to silicon transistors, such as molecular computers, optical computers, DNA computers, protein computers, carbon nano computers, quantum dot computers, and quantum computers. The most ambitious are quantum computers, which compute on individual atoms.
In some sense, they are the ultimate computer. In fact, quantum computers in principle can crack difficult codes that may take a standard digital computer centuries to solve. Quantum computers, however, are notoriously difficult to build, because individual atoms have to vibrate in perfect unison (i.e. they have to be coherent). The slightest vibration, interference, impurity, etc. can destroy this delicate coherence and ruin the calculation. Because of the decoherence problem, so far the world's record for a quantum computer calculation is just 3 x 5 = 15.
But progress is being made every day in quantum computers. Researchers at an IBM research facility (Almaden Research Center) in Zurich, Switzerland have had a busy week thus far. Researchers there made two discoveries they say represent a major leap in the development of femtotechnology. If you haven't heard of the field of study known as femtotechnology; It's generally a term that is used by some futurists to refer to structuring of matter on a femtometer scale (dealing with elements that are less than 1/1000th of a nanometer in scale). Their first announcement stated that the researchers had successfully measured how long a single atom can store information. Just three days later, the researchers announced that they had successfully snapped a picture of a single atom by trapping a single atom inside a tractor beam.
(Technical Note: Using a modified version of a Scanning Tunneling Microscope (STM), a technology that was originally devised in 1981; scientists have confirmed that it's now possible to observe things on a scale never before seen. Using a brand new technique called the "pump-probe" has allowed them to detect how the orientation of an atom's magnetic movement evolves in time. One of the researchers; Sebastian Loth states "When objects shrink down to the nanoscale, physical effects tend to speed up, To explore the fast processes in individual nanostructures, scientists need a tool that can keep up in speed." We can now see things in real-time and on the atomic scale.
The new alternating voltage pump-probe technique has allowed them for the first time to both record and visualize the magnetism and dynamic processes of individual atoms. Not only have they improved the system of visualization but they are also doing this at speeds 1 million times faster than we were previously able to do. This breakthrough allows for a major increase in that of image resolution and now offers the ability for us to examine exactly how long these individual atoms can retain information. Previously, scientists were always able to observe this effect in a time-slice of around one second which has of course limited our ability to take our research methods to the next level. The new IBM technique has decreased that very time-slice to one nanosecond -- A substantial improvement that IBM believes will soon be widely employed by their research colleagues around the globe. This of course means that as more researchers start employing the technique, more advancements will follow.)
Advancing technologies and game changers like these are going to open the doors for all sorts of new avenues while significantly improving previous ones. IBM is already claiming that its new technology will lay the groundwork for quantum computing researchers by giving them a new way to explore the feasibility of an entire new approach to quantum computer through atomic spins on surfaces. This new technology could also give rise to various applications in data storage as we will now be able to determine whether single atoms can hold data. Other fields of research and new engineering improvements that would benefit from this are things like storage-class memory systems and even photovoltaics or solar cells.
So when might we find quantum computer on our desktops? Unfortunately, no one knows. Although stunning breakthroughs have been made in manipulating individual atoms, there is still the stubborn decoherence problem, especially if we want to lash thousands to millions of atoms together into a single computing device.
So it may take many more miracles and breakthroughs, spanning decades, to make quantum computers come to life. But when this happens, it will change world history.
The perfect nanocube: Precise control of size, shape, and composition

(Nanowerk News) With growing interest in using nanoparticles for everything from antibacterial socks to medical imaging to electronic devices, the need to understand the environmental, health and safety risks of these particles also grows. Researchers at the National Institute of Standards and Technology (NIST) have developed a simple process for producing nanocrystals that will enable studies of certain physical and chemical properties that affect how nanoparticles interact with the world around them.
Because nanoparticles behave differently from bulk samples of the same material, new tests to understand how they affect biological systems must be developed. Toxicologists determine the hazards posed by nanoparticles by introducing them to a biological system and monitoring the effects, but they currently lack a set of control particles whose size, shape and composition have been carefully produced and characterized.
In a recent paper published in Angewandte Chemie ("Monodisperse gold-copper bimetallic nanocubes: facile one-step synthesis with controllable size and composition"), NIST scientists describe a one-step process that allows them to control the size, shape and composition of gold-copper alloy nanocrystals to create perfect-edged nanocubes as small as 3.4 nanometers—just half the thickness of a cell wall and on the same size range as DNA.
The researchers combined and heated gold and copper precursors with other chemicals to produce highly crystalline, homogeneous, perfect nanocubes with abundant yield. To study the formation process, they removed samples at 1 hour, 1.5 hours, 5 hours, and 24 hours and found that just five hours were needed to produce perfectly cubic nanoparticles of uniform size. By adjusting the ratios of the chemicals in the original solution and the reaction time, they were able to precisely control the size, shape and composition of the nanocubes. This process is unique in allowing control of the ratio of copper to gold atoms within the nanocube to either 3:1 or 1:3.
"It's a simple process, and to the best of our knowledge is the first to use synthetic chemistry, or 'bottom up' technology, to produce gold-containing nanocubes below 5 nanometers. Anything less than 10 nanometers has been extremely challenging due to the mobile behavior of the gold atoms," says NIST physicist Angela R. Hight Walker, who wrote the paper with Yonglin Liu, a guest researcher at NIST.
The NIST-developed process for creating such nanocubes will allow toxicologists to systematically alter one of the nanocubes' characteristics and observe how the change affects the biological response, if at all.
This synthesis and the resulting high-quality nanocubes may have other applications in areas such as solar energy, says Liu. "Typically, we cannot make big batches of high-quality samples for testing; now we can."
The perfect-edged nanocubes are unique from other nanocubes in the literature, says Hight Walker. The sharp edges, as opposed to truncated or rounded edges, will enable different, more reactive chemistry that could be beneficial in applications such as catalysis—in which the nanocubes would be used to initiate or enhance a chemical reaction.
The process has received a provisional patent.
Cheap nano crystals promise huge storage boost
3D memory chips built from silicon
Rice University announced today that scientists there have created the first two-terminal memory chips that use only silicon, extending the limits of miniaturisation subject to Moore's Law. The new technology places multiple layers of memory capacity on the same chip, creating what is referred to as a 3D memory architecture.
According to a Rice University spokesman, the new memory technology will improve scalability by an order of magnitude compared to NAND flash technology available today. "The fact that they can do this in 3D makes makes it highly scalable," he said. "We've got memory that's made out of dirt-cheap material and it works."
In 2008, researchers at the university showed how electrical currents could repeatedly break and reconnect 10-nanometer strips of graphite, which could potentially boost flash memory capacity by many times. The Rice researchers said then that the new technology could withstand radiation and temperatures of 200 degrees Celsius that would cause solid-state disk memory to disintegrate.
At the time, the research team acknowledged that they weren't sure why their discovery worked so well. With the latest finding, the research team, including professors James Tour, Douglas Natelson and Lin Zhong, proved the circuit doesn't need the carbon to function, only silicon. During the project, Jun Yao, a graduate student in Tour's lab, was able to confirm the hypothesis when he sandwiched an insulating layer of silicon oxide between semiconducting sheets of polycrystalline silicon that served as the top and bottom electrodes, Rice said.
Yao applied a charge to the electrodes, which created a conductive pathway by stripping oxygen atoms from the silicon oxide, forming a chain of nanometer-sized silicon crystals. Once formed, the chain can be repeatedly broken and reconnected by applying a pulse of varying voltage, the University said.
"It is more than 5 times denser than 20 nanometer flash... without 3D stacking," Zhong said. "I would argue the nanowire-based solution is much more amenable to vertical stacking, which makes the technology very scalable as process technology improves. The density can be further doubled or tripled with two or three layers."
Unlike NAND flash memory, which is controlled by three terminals or wires, the new silicon memory requires two terminals, making it more viable for three-dimensional or stacked silicon arrays, multiplying a chip's capacity. But like flash memory, chips made with silicon consume virtually no power while keeping data intact.
The nanocrystal wires are as small as 5 nanometers wide. A nanometer is one billionth of a meter. "The beauty of it is its simplicity," said Tour, a professor of mechanical engineering and materials science and computer science, in a statement.
Because the layers of silicon-oxide memory are not required to hold a charge, they can also be stacked one atop another, he added. "I've been told by industry that if you're not in the 3D memory business in four years, you're not going to be in the memory business. This is perfectly suited for that," Tour said. "Manufacturers feel they can get pathways down to 10 nanometers. Flash memory is going to hit a brick wall at about 20 nanometers," Tour said. "But how do we get beyond that? Well, our technique is perfectly suited for sub-10-nanometer circuits."
PrivaTran, a tech design company, is already testing a silicon-oxide chip with 1,000 memory elements built in collaboration with Tour's lab. "We're real excited about where the data is going here," said PrivaTran CEO Glenn Mortland, who is using the technology in several projects supported by the Army Research Office, National Science Foundation, Air Force Office of Scientific Research, and the Navy Space and Naval Warfare Systems Command Small Business Innovation Research (SBIR) and Small Business Technology Transfer programs.
"Our original customer funding was geared toward more high-density memories," Mortland said. "That's where most of the paying customers see this going. I think, along the way, there will be side applications in various nonvolatile configurations."
NuPGA, a company formed last year through collaborative patents with Rice University, is also demonstrating how silicon oxide also works in reprogrammable gate arrays. NuPGA's devices will assist in the design of computer circuitry based on vertical arrays of silicon oxide embedded in "vias," the holes in integrated circuits that connect layers of circuitry.
Such rewritable gate arrays could drastically cut the cost of designing complex electronic devices, according to Tour.
===============================================================
Will the next generation memory chips be made of silicon oxide, not carbon?
There’s a reason why you can store what seems like a gazillion songs and pictures on your smart phone. The gadget has an incredible memory in its inner core.
![]()
But there’s a limit to the development of electronic memory. Computer experts worry that we might reach that limit in five years.
It turns out, a chip made from silicon dioxide can store more information than a flash drive. As a bonus, this method does not require any carbon.
Rice University chemists discovered that silicon dioxide can actually store information in the breaks in its crystal structure. Eventually, the researchers hope the nanocrystals can be used to create 3-D storage devices, which would severely cut the cost of electronics.
It wasn’t easy for graduate student Jun Yao to prove that building a silicon oxide circuit was even possible. When Yao was looking for a graphite replacement, nothing could properly charge the circuit.
However, Yao got rid of carbon and instead, used silicon oxide next to silicon terminals. The student found that the silicon oxide nanowire performed just as well as the graphite device. The silicon oxide nanowire only had two terminals, which didn’t require it to hold a charge.
“I’ve been told by industry that if you’re not in the 3-D memory business in four years, you’re not going to be in the memory business. This is perfectly suited for that,” Rice professor James Tour said in a statement.
Flash memory is expected to hit a wall when it gets down to the 20 nanometer scale.
That’s why the Rice researchers’ technique looks promising — it’s a proof of concept that it can be used to build 10 nanometer circuits.
And the best part is that the chemistry is rather simple.
A Texas based company called RivaTran is working with the lab on a number of projects. According to a statement, the projects are supported by the:
* Army Research Office
* National Science Foundation
* Air Force Office of Scientific Research
* Navy Space and Naval Warfare Systems Command
* Small Business Innovation Research (SBIR)
* Small Business Technology Transfer program
Whether or not this discovery can break the memory barrier and help computer chips continue to shrink remains to be seen.
However, not everyone is convinced silicon oxide circuits are the way to go. The New York Times reports that I.B.M and Intel are looking at phase-change memory.
Nanosys Opens Korean Facility to Support Recent Expansion in the Asian Market
Firm Taps Renowned Materials and Semiconductor Expert to Lead New Facility for the Continued Development of Architected Materials for Asian Electronics Manufacturers
PALO ALTO, Calif., Sep 02, 2010 (BUSINESS WIRE) -- Nanosys, Inc., an advanced materials architect, today announced the opening of a new subsidiary, Nanosys Korea, Inc. with facilities in Gyeonggi-do, Korea. The company named former LG Electronics vice president Dr. Jong-Uk Bu as president of Nanosys Korea, Inc., which will support its growing Korean and Asian customer base.
This year, Nanosys commercialized its quantum dot technology with the QuantumRail(TM), a process-ready component that improves LED backlit display color gamut and efficiency, and has announced partnerships with LG Innotek and Samsung Electronics. The company is currently working with major battery manufacturers to improve lithium-ion battery capacity using its SiNANOde(TM), a silicon composite material that can increase battery cell capacity by up to 40 percent. SiNANOde(TM) --enhanced batteries will be available to consumers in 2011.
"Samsung Electronics and LG Innotek have demonstrated the visionary nature of the Korean electronics industry by adopting new architected material solutions that deliver exponential performance results," said Jason Hartlove, CEO of Nanosys. "Dr. Bu will lead Nanosys Korea's efforts to meet our customers' needs for architected materials and to expand our presence in Asia."
Dr. Bu, who will lead the facility located in the provincial capital city of Suwon, brings a breadth of knowledge and experience in the display and microelectronics fields. From 1984 to March, 2006, he worked on the development of silicon micro-machining and MEMS sensors as a research fellow and vice president of the LG Electronics Institute of Technology in Seoul, Korea. He holds a Ph.D. degree in Metallurgical Engineering from Korea University, worked as a research fellow at the University of Michigan and has taught at the Korea Advanced Institute of Science and Technology and the Korean Polytechnic University in Ansan.
Nanoelectronic gemstone
DIAMONDS, gemstones have always amazed us with their colors. Now in the era of nanotechnology, it's the Quantum Dot (QD) that is dazzling our eyes. QDs are semiconductor nanostructures, 25 billionths of a meter in diameter, which can confine electrons in three dimensions and emit light when exposed to ultraviolet radiation or X-ray. About 50 thousandth the width of a human hair, these nano-scale gems are built by confining electrons in some insulating material(s) around a central, well-conducting region. Although they can be composed of hundreds of atoms, quantum dots in many ways behave almost as if they were single gigantic atoms. This enables the study of quantum mechanical effects on a length scale that is around 100-500 times larger than the pure atomic scale.
QDs display unique optical and electrical properties that are different from bulk material. Excited QDs emit light, this emission depends not only on the material from which the QD is made, but also on the dot's size. QDs can therefore be “tuned” during production to emit any color of light desired. The smaller the dot, the closer it is to the blue color, and the larger the dot, the closer to the red end of the spectrum. QDs can even be tuned beyond visible light.
These glittering nanostructures are now in action at different technological war-fronts. QD lasers can be operated at wavelengths previously not possible using semiconductor laser technology. QD light-emitting diodes can be incorporated into a new generation of applications such as flat-panel TV screens, digital cameras, mobile phones, personal gaming equipments.
QD technology is one of the most promising candidates for use in solid-state quantum computation. With several entangled QDs, quantum bits (qubit) are realizable. QDs as a source of pairs of entangled photons can play a major role in the development of quantum information technologies. QD detectors can be used in medical and environmental sensing, optical communications, thermal imaging, night vision cameras, and missile tracking and recognition. In modern biological analysis, organic dyes are being replaced by QDs. QDs have been tested in most biotechnological applications that use the fluorescence, including DNA recognition, cell animal biology, biological motion tracking.
Research on new generation of 31% to 42% efficient Photovoltaic devices using QDs may be the next big breakthrough in our energy crisis. With so many potentials, QD is a technology for everyone to keep an eye on.
The author is a freelance science enthusiast and student of electrical engineering in BUET.
What do a California solar company poised to build the world's largest nanotechnology manufacturing plant and the builder of next-generation, advanced energy storage devices have in common?
Answer: They both chose Oregon as the place where they will produce their 21st century technologies.
Catching the next wave of business development
I recently attended events celebrating the decision of both of these companies to invest hundreds of millions of dollars here in Oregon.
California solar manufacturer Solexant Corp.’s commitment to build the world's largest nanotechnology manufacturing facility in Gresham means Oregon has now caught the next wave of the renewable energy revolution — thin-film solar — sweeping the globe. And it comes after Oregon has already established itself as the leading site for solar manufacturing in the U.S. with such firms as SolarWorld, Sanyo Solar and PV Powered.
Solexant, a developer of third-generation, ultrathin-film solar technology, said the plant will be its first commercial-scale nanocrystal manufacturing facility. Upon completion, the 100-megawatt plant will be Oregon's first thin-film solar manufacturing plant and the largest nanotechnology manufacturing facility in the world.
The factory will initially be housed in an existing 150,000-square-foot facility with plans to construct a new facility in 2011. It will employ as many as 200 Oregonians, and the company plans to build additional 100-megawatt lines in Oregon after the first line becomes operational.
Based in San Jose, Calif., Solexant produces the first ultrathin-film solar cell that incorporates high-performance, inorganic nanocrystals on flexible substrates. The technology platform — originally developed at Lawrence Berkeley National Lab — can utilize currently available and emerging high-efficiency materials to dramatically reduce manufacturing costs.
The second announcement came earlier this month in Albany. After receiving a stimulus grant of $21.3 million from the U.S. Department of Energy, Oregon Freeze Dry, in partnership with Seattle-based EnerG2, broke ground on a manufacturing facility that will produce material for the next generation of ultracapacitors (an advanced energy storage device similar to a battery).
The facility is in Albany's Enterprise Zone so state incentives were instrumental in helping the participants move forward with partnership agreements necessary for the project. Business Oregon, the state's economic development department, partnered with the local economic development agency (Albany-Millersburg Economic Development Corporation) to provide community support. In addition, we will continue to work to integrate this project with the Oregon Nanoscience and Microtechnology Institute's research initiatives.
Construction is expected to sustain at least 50 jobs through the commissioning of the plant. The plant is expected to create at least 35 permanent jobs for local workers as operators, technicians, engineers and managers. The plant will be completed on an 18-month fast track and produce electrode carbon for use in ultracapacitors for electric vehicles. The total project cost will be approximately $29 million.
The plant will be the first facility in the world dedicated to the commercial scale production of nanostructured, synthetic, high-performance carbon electrode material. Oregon Freeze Dry, the world leader in contract freeze-drying, has been an EnerG2 R&D manufacturing partner since 2006.
These first-of-their-kind commitments by Solexant and EnerG2 came upon the heels of Facebook's recent decision to not only build its first company-owned data center near Prineville, but immediately double the size of the facility. Facebook now plans to build a 160,000-square-foot expansion that will be connected to the first phase development of the data center.
These three investments in Oregon’s future reflect my efforts over the last eight years as Governor to help re-engineer the state’s economy to compete and win in the 21st century’s global economy. Through incentives and help from local partners, such as those in Gresham and Albany, we continue to attract the kind of companies that will employ thousands of Oregonians for decades to come.
Nanostructured Photocatalytic Materials Enable Capturing Solar Energy and Simultaneously Powering Water Purification
An interview of Associate Prof Darren Delai SUN, Nanyang Technological University, Singapore
Both Water and Energy are indispensable for our lives., however, the demand for drinking water and energy has been increasing dramatically with the explosive population growth and ongoing rapid industrialization in the past decades. Water treatment and clean energy technologies attract intensive attention from both academia and industry. A Singapore research group led by Dr Darren Delai SUN, associate professor of School of Civil and Environmental Engineering, Nanyang Technological University (NTU) has developed innovative materials and technologies in the area of TiO2-based nanostructures for clean water and energy (hydrogen and solar energy) production applications. In mid July 2010, NanoGlobe team had the privilege to interview Dr SUN and learned about the R&D activities and achievements of his research group.
Dr Sun completed his PhD in Chemical Engineering at the University of New South Wales, Australia in 1992 and currently holds the associate professor position in NTU. He is also a Chair for International Water Associate (IWA) Specialist Group on Chemical Industries and Editor for Water Science and Technology. He is the author or co-author of more than 100 scientific publications in the peer-reviewed journals such as Small, Nano Letters, Advanced Functional Materials, Environmental Science and Technology and so on. He is also a co-editor of 2 books and co-author of a chapter in 3 books. Dr SUN and his group won the Prestigious Engineering Achievement Award 2008 from Institute of Engineering Singapore and the Nanyang Award 2009 for Innovation & Research. As a chemist, he has always devoted in the fundamental research for physical and chemical properties of TiO2, ZnO, SnO2 and other nano oxides especially for membrane material applications. At the same time, his engineering background enables him to focus on materials application of water and wastewater treatment, water reclamation and heavy metals stabilization. TiO2 nanomaterials have promising applications in both areas of photovoltaics and photocatalysis, such as TiO2 nanocrystalline electrode for dye sensitized solar cells (DSSC) and TiO2 nanomaterials photocatalytic water splitting. Instead of using dopants to modify the TiO2 properties, Dr. SUN and his colleagues control the TiO2 material properties by controlled synthesis of different nanostructures. For example, they use the electrospinning technique to make fibers about 100 nm wide consisting of two thinner threads fused together. One of these two threads is made of TiO2 and the other of SnO2 which enhances the photocatalytic activity of TiO2. The twin fiber also has larger surface area on which photocatalytic reaction can occur more efficiently than other composite structures. In addition, they also succeeded in growing ZnO nanocrystals directly onto the surface of porous TiO2 nanotube arrays for increasing the quantum efficiency and recyclability of the TiO2 photocatalysts.
In order to realize the production of clean water with almost zero cost, Dr SUN and his colleagues solved the problems - sludge, nitrogen, membrane fouling and energy - step by step. At first, they successfully eliminate sludge and nitrogen by adding the prolonged sludge retention time aerobic membrane bioreactor (MBR) into the wastewater treatment system. However, the cost reduction for water purification is still limited by a membrane fouling problem which is caused by deposition of the foulants on membrane surface such as nature organic matters (NOMs) and bacteria etc., since the consequence of membrane fouling is the reduction of permeate production or need of high driving force to maintain a required rate of permeate production. TiO2 nanomaterials are the most popular photocatalysts which can effectively oxide the organic pollutants in the wastewater. Dr SUN's group developed the nonwoven TiO2 nanofiber/tube/wire micro and ultrafiltration membrane (see Figure 1) using their proprietary process in the electronspinning or alkaline hydrothermal technology. This robust and free standing TiO2 nanofiber membrane has the concurrent filtration and photocatalytic oxidization functions in the presence of UV or visible light irradiation. This design has the following advantages: a high surface area of nanofiber structures allow enhanced adsorption rate of various trace organics and bacteria, while the full surface exposure to UV or visible lights accelerates photocatalytic oxidation and effectively overcome the membrane fouling problem. Not only filtration efficiency is enhanced, but the lifetime span of filtration membrane is also prolonged. As a result, the cost of water treatment can be dramatically reduced by integrating this membrane into the water treatment system. In addition, its unique flexible property enables this self-generation membrane to be formed into various membrane modules for larger commercial application, e.g. roll to roll production.
Not only taking advantage of the photocatalytic property of TiO2 nanofiber membrane for wastewater reclamation applications, Dr Sun's group has been also investigating the photovoltaic property of their membranes for the application in DSSCs. Using their flexible TiO2 nanofiber/tube/wire membrane as the electrode, the low-cost flexible DSSCs with conversion efficiency of about 5% can be fabricated . Combining the photocatalytic and photovoltaic properties of TiO2 nanofiber/tube/wire membrane, Dr Sun's group designed a dye sensitized TiO2 nanostructure-based sandwich structure to smartly realize the concurrent production of clean water and electricity. Furthermore, a nano composited low pressure RO membrane with multifunctional properties for the concurrent desalination and production of electricity has been recently developed. "Our dream is to produce drinking water with almost zero cost, and we believe this dream is coming true," concluded Dr Sun.
Nanosys Raises $25 Million, Unveils Three-Pronged Deal with Samsung
Palo Alto, CA-based Nanosys, the nanotechnology startup that has struggled to regain altitude after a high-flying debut in 2001, is switching on the afterburners this week.
To finance a move to a larger facility where it will have more space to manufacture its nano-engineeered materials for lighting and digital displays, the company is about to close a $25 million Series E venture round, with $5 million more potentially to follow by October 1, CEO Jason Hartlove told Xconomy on Monday.
Samsung, the South Korean electronics giant, is supplying $15 million of the new equity investment through its affiliate Samsung Venture Investment Corporation. The rest is from the company’s existing investors, and the $5 million second tranche is reserved for new investors. [Correction: In an earlier version of this paragraph, the second tranche amount was listed as $10 million.]
At the same time, Nanosys is announcing two other agreements with Samsung. There’s a multi-million-dollar licensing deal that will give Samsung access to Nanosys technology that could help it manufacture more efficent thin-film solar panels. And Samsung will also fund work at Nanosys to develop new quantum-dot crystals—the core of the startup’s technology for lighting enhancement—that don’t contain cadmium, a toxic element whose use is restricted in Europe and other regions.
The deals are critical ones for Nanosys, which wandered for years without a commercial product and brought in Hartlove as a turnaround CEO in 2008 (see this July 21 profile). The agreements give the startup more resources to expand into the two markets—displays and batteries—where Hartlove believes it can most quickly commercialize its own work, while at the same time allowing it to cash in on unexploited parts of its patent portfolio, particularly in solar technology. “This is a strategically important deal for us,” Hartlove says.
Gearing up to move to new quarters has been one of Hartlove’s highest priorities. The company has won a contract to supply the quantum dot phosphors inside the “QuantumRail,” a component that increases the brightness and efficiency of LED backlights for mobile device displays, to Korea’s LG Innotek, and is pursuing additional customers. That means it now has to make the phosphors in industrial quantities. But in its current location—tucked into a Palo Alto office park with burgeoning Web and software companies such as Facebook a stone’s throw away—the startup is “basically out of capacity, really footprint-limited and fire code limited,” says Hartlove.
The company will use the growth capital round to open a new facility in a larger industrial park. “It’s here, still, within the Bay Area,” Hartlove says. “We haven’t announced a site yet, but that’s what the new capital is for, the capacity expansion to meet our 2011 revenue goals.”
The next problem is an environmental one. Manufacturers of smartphones and notebook computers can use the QuantumRail technology to tune the frequencies emitted by the LED backlights in their liquid crystal displays, resulting in a far richer range of colors. But there’s a downside—the quantum dots, which are actually nanocrystals made of cadmium selenide. Cadmium is a controlled compound under the European Union’s Restriction of Hazardous Substances (ROHS) directive, and to sell the QuantumRail to manufacturers who market hardware in Europe and other regions that follow the ROHS limits, Nanosys will ultimately need to eliminate cadmium from its chemistry.
Hartlove says Nanosys has a way to do this. Essentially, it will get the same efficiencies out of future quantum dot crystals by creating graded layers of indium phosphide and zinc sulfide. But that’s a tricky process that hasn’t been done economically on a nanometer scale, and Samsung has agreed to help fund the necessary R&D.
Finally, Samsung is paying Nanosys for a license to nano-engineered structures that the company investigated with the goal of making photovoltaic panels more efficient at converting sunlight into electricity. When Hartlove joined the company in 2008 and reviewed its intellectual property portfolio, he chose not to pursue the solar technology for commercialization within Nanosys, because “it didn’t make sense to us to jump in when a number of other companies were already in product,” he says. But Nanosys did form a wholly owned division called QD Soleil around the solar patents, with the goal of either selling the division or licensing its technology. That’s what Samsung is doing now.
“Samsung has made an announcement that they are going to spend $20 billion to go into the renewable energy and green technology fields, and they have a very aggressive plan in solar,” says Hartlove. “They are looking to invest heavily in that area to try to catch up quickly, and by licensing some technology from us, they believe they can do that. It’s mutually beneficial in that it gives them access to good nanoscale technologies in terms of wavelength conversion and efficiency improvements that they are going to be able to put into their products as they bring solar panels to market. And for us it’s a way to see the technology actually get deployed in a field we were not planning to enter ourselves.”
As I recounted in my July profile of Nanosys, Hartlove spent part of his own career in South Korea, turning around a struggling specialty semiconductor company called MagnaChip and orienting it toward more mainstream customers. He says the relationships he formed in Korea laid the foundation for the agreements announced today.
“I have known Samsung senior management people since about 2003,” Hartlove says. “I have been dealing with a lot of the same folks now, through this process. Knowing the Samsung company culture and business culture and how they are approaching these markets has been very helpful in securing this deal.”
Dye-sensitised solar cells
A low-cost solar cell that imitates photosynthesis opens up new applications for photovoltaics.
Solar power works; if it didn’t, we’d all be dead. The sun provides the power for every plant on Earth and therefore underpins the food chain. But man’s attempts to copy photosynthesis, the process by which plants harness solar energy, haven’t always been so successful.
Successive generations of solar cells have been developed since the 1950s, with the promise of solar power always seemingly just about to make a breakthrough. Now, silicon-based solar cells compete with a newer generation of plastic photovoltaics, and with interest in renewable power at its highest level for years, solar power may yet make it into the big time.
One of the most interesting entrants into the photovoltaic field, however, is a cell that copies very closely the mechanisms of photosynthesis. Based on cheap materials and manufacturable through simple processes, it is set to make a big impact into the most neglected of global energy markets, the developing world.
The dye-sensitised solar cell is also known as the Grätzel cell, after its inventor, Prof Michael Grätzel of the Ecole Polytechnique of Lausanne in Switzerland. The invention, which won Grätzel this year’s Millennium Technology Prize, has recently hit the market for the first time, courtesy of Cardiff-based company G24 Innovations.
In a conventional silicon-based photovoltaic cell, sunlight strikes the crystalline silicon surface and displaces an electron, which can then diffuse through the structure of the semiconductor, creating a current. The silicon is ’doped’ with elements to increase its conductivity, easing the path of the electron. However, the efficiency is limited, and this is partly because the silicon meets the role of both energy harvesting and electron transport.
This isn’t the case in photosynthesis. Chlorophyll, the pigment that makes leaves green, acts as the energy harvester; it absorbs the light and generates a free electron. Other mechanisms within the plant’s cells then handle the movement of the electron, carrying it into the processes where it reacts with carbon dioxide and water to make glucose and oxygen.
In the early 1970s, concerns about the price of oil triggered a wave of research into alternative energy sources, and Grätzel became interested in photosynthesis. ’At the time I was educated, the detail on how photosynthesis worked was not well established,’ he said. ’I was interested by the way that plants use their molecules to generate charges and separate those charges.’
Grätzel cells copy this process, using three essential ingredients: a conductive electrolyte solution, in which is dispersed nanocrystals of the white pigment titanium dioxide (TiO2) coated with an organic dye. The dye takes the place of the chlorophyll – in early versions, chlorophyll itself was used, but electrons couldn’t diffuse through it – and absorbs sunlight, which knocks electrons free from the organic molecular structure of the highly coloured material. These are knocked into the TiO2, which has semiconducting properties, and carries the charge through the electrolyte to an external circuit. Electrons flowing back into the cell through the other side of the circuit replace those that are displaced from the dye molecules.
Grätzel’s first cells were perfected in the 1980s, as a result of work with new nanoscale forms of TiO2 created at Lausanne. ’This was a fundamental study, driven by our curiosity – no one had done it before,’ he said. The research came to fruition in 1988, using a porous film coated with TiO2 crystals, creating a very high surface roughness to absorb sunlight.
’I asked my PhD student Hans Desilvestro how the experiment had gone,’ Grätzel remembered. ’He did not seem too enthusiastic initially, adding that he had only measured a few milliampères current. I knew it was a lot; other researchers had only measured micro or nano-amps.’
The initial cells achieved efficiencies of around seven per cent – a thousand times better than original versions from the 1970s – and currently reach around 12 per cent.
Although not as efficient as silicon cells, whose efficiencies are generally around 15 per cent, Grätzel cells are cheaper. TiO2 is a readily available material, used widely in white paint and sunscreens; organic dyes are also inexpensive. Grätzel is fond of demonstrating the system in schools, asking pupils to make their own cells using blackberry juice as the organic dye.
Moreover, the cells can be made using a simple roll-to-roll process, using machinery similar to that used in printing, coating and packaging – a system that is being used by G24 Innovations in Wales.
The sheets of dye-sensitised cells rolling off their production line are cut up to be stitched into the company’s first commercial product, a range of backpacks with integral solar panels to charge mobile phones and MP3 players.
This technology and application have a wider significance, however. ’This really comes into its own in Africa,’ Grätzel explained. ’More than 90 per cent of internet access there is via mobile phone, but the electricity grid is poor so people can’t easily charge their phones.’ Much of this mobile phone use is to check prices for agricultural products, so that smallholders can locate the best market to maximise their profits, Grätzel said.
G24 Innovations is planning a portable phone charger that would be given out free, with the cost spread across call and data download charges, to allow users to charge easily and regularly.
Further applications could be extended to solar power for water treatment, making it possible for households to operate their own small purification units without the need for an electricity connection or a diesel generator; similarly, the units could power fridges, both for domestic use and to keep pharmaceuticals and blood supplies cool in medical clinics. The low cost, light weight and flexibility of the printed panels makes them ideal for such applications, Grätzel said.
Other applications include coatings for windows, as the dye component of the cell could be made transparent, absorbing only the near-IR and ultraviolet component of the solar radiation. This is a particularly interesting application, Grätzel believes, as the cells can produce electricity in lower light conditions, unlike conventional silicon cells that require bright sunlight to work effectively. Pilkington’s North American operation is working with a US-based Grätzel cell provider, Dyesol, to produce electricity-generating glass panels for building-integrated photovoltaics.
Another potential spin-off is currently in Grätzel’s laboratories at Lausanne, using nanocrystalline iron-oxide films working in conjunction with Grätzel cells to generate hydrogen from water.
Meanwhile, work continues to further refine the Grätzel cell. Solid versions are being developed, using a solid inorganic matrix to replace the electrolyte solution; other work is focusing on improving the efficiency of the dye or its sensitivity to specific portions of the solar spectrum.
Kicking around solar cell materials
August 15, 2010
Sheffield's David Lidzey working with Athene Donald of the University of Cambridge and experts from Cardiff University and Nick Terrill at the Diamond Light Source, the UK's synchrotron facility in Didcot, have investigated the structure of solar cell materials to help them improve photovoltaic efficiencies. The research into understanding the structure of plastics used in new-types of low-cost solar cell based on fullerenes could improve their efficiency significantly.
Photovoltaic semiconductor devices have the potential to generate low-cost renewable-electricity simply by absorbing and converting the energy of sunlight. Current silicon-based devices are expensive and fragile so researchers have been looking for alternative organic-based materials with the right optical and electronic properties. A photovoltaic film made from plastic would be far less expensive than silicon as well as being flexible and easier to fabricate to fit particular devices, roof profiles, or other areas where renewable energy might be needed.
The development of plastic photovoltaic devices will inevitably rely on being able to control morphology of thin organic semiconductor films at the nanoscale where charge-generation and charge-extraction occur. Specifically, the researcher say, "The power conversion efficiency in a conjugated polymer-functionalized fullerene bulk heterojunction organic photovoltaic (OPV) device [turns out to be] dependent both on the electronic properties of the constituent materials and on the nanoscale morphology of the active semiconductor layer thin-film."
Earlier research has investigated thin film structure after casting the materials from solution. But, members of the Sheffield-led collaboration wanted to understand the dynamic processes that occur as the semiconductor thin-film blend is first being deposited. This, they hoped, would give them insights into the structure and morphology of the film in real-time as the solution dried. Such dynamic information would allow them to uncover the basic mechanisms of film formation and so provide new clues to the design and production of improved materials.
"This is the first time we have unravelled the nanostructure evolution from random, long chains into densely packed nanocrystals," says Sheffield team member and post-doctorate researcher Tao Wang. "The results will direct us to better control our device fabrication process for high efficient large-area plastics solar cells."
The data obtained using the very bright synchrotron X-rays generated by Diamond to carry out grazing incidence X-ray scattering (GI-XS) combined with the equally powerful technique of spectroscopic ellipsometry (carried out at Sheffield) has given the team just such information. They investigated molecular self-organization in thin film formation from a solution of a polymer blend of poly(3-hexylthiophene) (P3HT) and the all-carbon material fullerene in the form of [6,6]-phenyl C61-butyric acid methyl ester (PCBM). Both are required in their photovoltaic material for electrical charge generation.
The data showed that the drying process and consequent crystallisation take place in three main stages, with the second stage of rapid polymer crystallisation being the most important. "We illustrate the evolution of the extinction coefficient from a solution to a solid, semi-crystalline state," the team explains. "We show that once the solvent fraction in the film falls below 50%, the P3HT undergoes rapid crystallization via heterogeneous nucleation; a process that is complete in seconds." A higher degree of crystallinity is associated with more rapid electrical charge-conduction and therefore more efficient device operation. As such, the team adds that the mechanistic insights into film formation revealed by their study demonstrate how the deposition processes might be optimized for large-area organic photovoltaic manufacture. The team is now working on manipulating the crystallisation of various polymers.
As the University of Sheffield launches Project Sunshine, aimed at bringing together scientists from various disciplines to tackle the challenge of solar energy, Lidzey enthuses about his team's work: "This has been a very exciting experiment for us - we have used Diamond Light Source to carry out some important science on a technologically important class of materials," he says." This has allowed us to understand a process that has so far remained unexplored in these materials. This information will quickly feed into our solar-cell research programme."
"Operational devices based on this material system have been around for some time," Lidzay told SpectroscopyNOW. "However, lots of groups are trying to understand how the structures that self-assemble in the active layers of devices confer efficient (or otherwise) operation. Once we understand this better, we can apply knowledge to the design and processing of new photovoltaic materials that chemists are currently synthesizing to make even more efficient devices."
Nanosys Raises $25 million for Thin Solar Panel Development
Nanosys, the Palo-Alto advanced materials architect, announced it raised $25 million in Series E funding this week, as well as plans to form an alliance with Samsung in the development of thin solar panels, memory products and display technologies.
Samsung and the nanotechnology provider will co-develop commercial applications for nano-architected materials for thin film solar and electronics markets, working together to create more efficient and better cost effective models for solar, display, semiconductor, LED and memory markets. As part of the deal, Samsung will invest $15 million of that Series E funding through its Samsung Venture Investment branch.
“We believe that working closely with Nanosys will help us to develop exciting new products for our customers that will address needs in the world’s largest markets across a number of technologies,” said Seungho Ahn, senior vice president of the Intellectual Property Center at Samsung Electronics. “We believe that working closely with Nanosys will help us to develop exciting new products for our customers that will address needs in the world’s largest markets across a number of technologies.”
Connected to the Samsung deal, another $10 million will be invested into Nanosys from existing backers that include Arch Venture Partners, El Dorado Ventures, Polaris Venture Capital and Venrock.
Since 2001, Nanosys has managed to raise around $94 million. This latest cash investment serves as a nice tip from Samsung, a South Korean electronics giant that had $116.8 billion in consolidated sales in 2009. As the solar and electronic display industries evolve in technology, Nanosys is posed to become a major player. Earlier this year, the company announced an agreement with LG Innotek, the electronics component manufacturer, for the use of Nanosys' quantum dot phosphors in displays, which the company claims is the first “real world” commercial application of quantum dots in electronics.
“Samsung is a clear leader in the electronics and solar sectors,” said Jason Hartlove, CEO of Nanosys. “Not only does Samsung bring deep knowledge of solar, memory and display technologies and markets, they bring tremendous experience in scaling up new technologies, which will help us accelerate the development, production and market introduction of devices enabled by our nano-architected materials.”
A new window dressing for solar power
For centuries windows have helped light our indoor worlds. But this week, scientists from a Norwegian company and a British university have come together to try to make windows bring electric light to us as well.
EnSol AS and University of Leicester are developing a thin-film-solar coating that might allow windows (and other building facades) to generate solar power. Their metal nano-particles will sit in a clear composite matrix, which is then applied to window glass.
The developers are aiming for the films to have an active sun ray capturing area of more than 16 square centimeters. The hope is the photovoltaic cells could reach 20 percent efficiency.
Chris Binns, professor of nanoscale physics at University of Leicester, says in a statement:
Obviously some light has to be absorbed in order to generate power but the windows would just have a slight tinting (though a transmission of only 8-10% is common place for windows in the “sun belt” areas of the world). Conversely the structural material of the building can also be coated with a higher degree of absorption. This could be side panels of the building itself, or even in the form of “clip-together” solar roof tiles.
According to researchers, the metal nanocrystals, shown to the right under an electron microscope, behave differently than silicon-based solar cells.
After demonstrating their new solar cell concept, EnSol AS is looking to the Condensed Matter Physics group at University of Leicester to design, produce and test prototypes. Construction of a thin film deposition facility in Norway is also in the works.
Should the partnership’s results prove successful, industrial scale production of the solar cells might be achievable through spray-on applications.
Nano drives improve displays
Display technology is currently realizing the benefits of nanotechnology in lighting support for the displays and the display construction itself.
One of the new display technologies is the Mirasol display from Qualcomm. This MEMS (microelectromechanical-system)-on-glass device targets low-power, daylight-readable color displays for portable-system applications.
Most LCD devices operating at low power, such as with mobile phones and tablet PCs, have issues with color representation. In varying light, the color accuracy of the display changes, altering the viewer’s perception of the image. The Mirasol display attempts to overcome these issues.
The display is a front-reflective display rather than a traditional backlit display. The properties of nanoscale materials combine with advanced MEMS-processing techniques, allowing the display to mimic naturally occurring phenomena. The display works by creating a color from an interference pattern on the reflected light that hits the top of the display. This process is the same one that makes a butterfly’s wing shimmer and display different colors.
The display uses red, green, and blue subpixels to create a single pixel measuring less than 1 micron. Each of the colored subpixels comprises cells, which form an array of devices. An applied voltage then switches these devices to “collapse” the MEMS device and turn off the reflective element (Figure 1).
The Mirasol display is just one example of the use of nanotechnology in a diverse range of applications. The area of nanomaterials—compounds, elements, and polymers that behave differently or exhibit new and unique properties at the nanoscale—is bringing many new support applications to electronics.
In the case of products from Cima NanoTech, the key nanomaterial is nanocrystalline silver. With its high conductive characteristics, silver has been in use as a standard material for more than 100 years. Cima formulates the silver and silver/copper into nanoparticle form and places these particles in suspensions and emulsions that designers can apply to various surfaces. Using self-assembly techniques, these nanoparticles form microscopic conductive networks that are transparent but that have properties similar to those of traditional opaque materials. Designers can apply the nanoparticle layer as a transparent film with an inexpensive, wet-rolling process in a large format, so it can become a low-cost, effective EMI (electromagnetic-interference) filter for plasma and other large displays. Designers can also pattern the material to form large-format resistive-touch-sensor overlays for LCD screens. In a new and growing application, the silver-and-copper particle solution can find use in conductive inks. Designers are able to print these inks on a surface using time-proven ink-jet technology.
This ability to print a conductor is drastically changing the RF- and PCB (printed-circuit-board)-prototyping flows by allowing, for example, the creation of one-time unique antenna designs that designers print and test on paper. Designers can similarly print a few fine-pitch prototypes on paper or mylar to test a design. The technique currently works in single-layer-system applications. However, multilayer-system applications are in progress.
Quantum dots are nanoparticles that have tunable optical characteristics. Companies including Nanosys and Zymera have turned these properties into commercial products. Nanosys has a series of quantum-dot lighting techniques that color-correct the wavelength of backlit LEDs in cell phones. Zymera is using the dots as bioluminescent particle tags for drugs to track the absorption and location of certain medications in the treatment of cancer, for example.
The Next Gen PV Cell Is Smaller than Small
A nanocrystal cell could see all high-rise windows double up as photovoltaic panels.
A surge in innovative design to capture sunlight could see the cost of photovoltaics (PVs) plummet as their applications soar.
The inventor of a nanocrystal cell that could see “the glass of all high-rises in New York” become electricity-generating panels has been awarded the €800,000 Millennium Technology prize. Professor Michael Grätzel of the Lausanne Federal Technology Institute and creator of the dye-sensitised, ultra-efficient Grätzel cell, took inspiration from natural photosynthesis to develop films in which the PV particles “are so small, they don’t scatter light”. His microscopic suntraps are capable of collecting light from all angles, and could be incorporated into window panes, assigning heavy roof-top panels to the past.
Small is beautiful for researchers at Sandia National Laboratories, New Mexico. By producing PV cells no bigger than glitter, they claim to have opened up the door to electricity-generating coatings for almost any shape or surface. The tiny cells are made from crystalline silicon and can be produced cheaply using techniques common to today’s electronics industry.
Researcher Murat Okanda claims that the micro-cells match the efficiency of current PVs, with the added bonus of “a significant reduction in manufacturing and installation cost compared with (standard) techniques”.
Glitter-sized or paper-thin? Scientists at the Solar Frontiers Research Center at the Massachusetts Institute of Technology say they have managed to print solar cells on sheets of paper. Carbon-based dyes act as semiconductors that can convert sunlight into electricity. As yet, the paper cells are far less efficient than current PV cells, but their relatively low cost could make them an attractive option.
Penn collaboration leads to simpler method for building varieties of nanocrystal superlattices
July 25, 2010
The study demonstrates a new and spontaneous way to grow long-range-ordered BNSL membranes with rigorous control of nanocrystal size, shape and concentration by combining two types of nanocrystals and assembling them during a drying stage at the surface of a liquid under normal conditions.
The method overcomes several limitations of the existing assembly strategies and produces large, free-standing membranes that can be transferred to any desired substrate such as silicon wafers, glass slides and plastic substrates, allowing the nanocryatalline films to be introduced at any stage in the device fabrication process.
The team demonstrated the potential for integrating these novel materials by growing millimeter-scale superlattice membranes containing iron oxide nanocrystals of two different sizes and incorporating the membranes into magnetoresistive devices. Measurements showed that the magnetoresistance of the resulting device was dependent on the structure of the BNSL and therefore controllable.
The physical properties intrinsic in these nanocrystals -- nanometer sized crystalline building blocks — offer a modern twist on the studies of interfacial assembly that reach as far back as Penn founder Benjamin Franklin and his studies of oil spreading on water in the 1770s.
Single and multi-component nanocrystal films are already under intense investigation by researchers as enablers of novel optical technologies that range from low-cost solar cells, light-emitting diodes and photo detectors and also in electronic systems that include field-effect transistors and solid-state thermoelectric coolers and generators and magnetic technologies that include magnetic recording materials and magnetic sensors and even as tailored electrocatalytic and photocatalytic films.
Co-assembly of two types of nanocrytals into BNSLs provides a low-cost, modular route to program the self assembly of materials with a precisely controlled combinations of properties. Advances in these complex interfacial assemblies and improvements in the transfer of single-component nanocrystal membranes in the past few years have heightened anticipation that this control could be extended to much more complex systems.
This Penn study establishes a route to free-standing large-area BNSLs membranes with the added ability to laminate them on any arbitrary substrate.
Fundamentally, growing BNSLs on a liquid surface will shed light on the mechanisms of multi-component nanocrystal assembly, which are critical to new concepts in self-assembly based nanomanufacturing, said Christopher B. Murray, the Richard Perry University Professor of Chemistry and Material Science and Engineering at Penn.
The research, funded by the U.S. Army Research Office and a National Science Foundation Materials Research Science and Engineering Centers Award, is published in this weeks Nature.
Existing strategies for growing BNSLs involve a more complex process of evaporating a two-nanocrystal solution on a solid substrate under carefully regulated temperature and pressure that influence BNSL formation. The method suffers from several limitations, most notably a limited choice of substrate, nucleation of irregular micrometer-sized, isolated islands of BNSLs on the substrates and an inability to transfer them once formed.
Given the fact that this novel assembly strategy is general for different nanocrystal combinations, we anticipate that membranes of quasicrystalline BNSLs and ternary nanocrystal superlattices will also be grown by this method, greatly expanding the systems that can be explored Murray said. Our dream is to program the organization of materials on all lengths scales for nanometers to millimeters combining the desirable physical properties multiple nanoscale systems. Fundamentally we are focused on identifying, understanding and optimizing new synergistic interactions in nanomaterials and in exploiting these emergent properties in new devices and systems.
###
This interdisciplinary study was conducted by Murray and Angang Dong in the Department of Chemistry in the School of Arts and Sciences and the Department of Materials Science and Engineering in the School of Engineering and Applied Science, Jun Chen of Materials Science and Engineering and Patrick M. Vora and James M. Kikkawa of the Department of Physics and Astronomy in SAS.
Two dimensional nanostructures emerge through self-organisation
An international team of researchers has succeeded in producing nanocrystals that build conductive two-dimensional nanostructures trough self-organisation
Nanocrystals are tiny particles that are not greater than 100 nanometres. Due to their crystalline structure, nanocrystals have a special property profile, which makes them highly interesting for the development of new materials and innovative technologies. The use requires a detailed fundamental research. An international team of researchers has now achieved pioneering results. For the first time, researchers have succeeded in producing nanocrystals that merge through self-organisation to form flat crystalline nanostructures that are highly conductive.
Conductive nanostructures extend the fields of application of nanocrystals Electrical components are produced on the basis of nanocrystals for quite some time now. However, these components are barely conductive, due to gaps between the nanoparticles that hinder the flow of free electrons. Flat nanostructures in contrast allow a considerably improved flow of electrons. Due to their conductivity, they provide a broad range of applications, such as inflexible electronic circuits, solar cells and photo sensors.
Organic molecules call nanocrystals to order
The nanocrystals are small lead sulphide particles that have a relatively simple structure. The driving forces behind the orderly arrangement of particles are organic molecules. These molecules (oleic acid), that are located on the surface of the nanocrystals, have a stabilising effect. When the organic molecules start to crystallise among themselves, the process of self-organisation begins. This causes the nanocrystals to form a crystalline and coherent structure. Instead of composing any shape, the nanocrystals cluster and form a well-structured surface.
Learning from nature: the key function of colloid research
A large number of research projects worldwide deals with the central question how matter organises itself in the nature. The better the comprehension of these processes the greater the opportunity to learn from the nature when it comes to the development of artificial systems. Following the example of nature, complex macromolecular systems with tailor-made properties and functionalities can be produced. The centre of interest are the colloids. Those are tiny particles that disperse in another (solid, liquid or gaseous) medium. Colloid research is of central significance insofar, as it provides new insights into the self-organisation of matter and the construction of complex systems. At the University of Bayreuth, the macromolecule and colloid research is one of the main focuses of both, teaching and research. This profile is strengthened by the research findings presented.
Publication:
Constanze Schliehe, Beatriz H. Juarez, Marie Pelletier, Sebastian Jander, Denis Greshniykh, Mona Nagel, Andreas Meyer, Stephan Förster, Andreas Kornowski, Christian Klinke, Horst Weller, "Ultra-thin PbS sheets by two-dimensional oriented attachment"
in: Science (2010), Vol. 329. no. 5991, pp. 550 - 553
We need more small stuff !
Hey, freegrass, how are you doing?
You must be busy not to be posting!
Best to you!
Puravida19
Nanotech is a part of TechConnect going on
now in Anaheim until June 24. Anyone in L.A.
might want to check it out.
Puravida19
In 2011 a global commodities exchange will go live, linking up buyers and investors with suppliers of nano-engineered materials, to help unlock the commercial potential of this far-reaching field of technology.
Trading exchange for nanomaterials to launch in 2011

The Integrated Nano-Science and Commodity Exchange (INSCX) will provide a 24-hour trading platform for nanomaterials, akin to exchanges for commodities such as oil, metals and grains.
The exchange could help suppliers of nanomaterials - for example carbon nanotubes, quantum dots and nanoceramics - find buyers in the form of companies developing products that exploit the properties of various advanced materials. Developers work across applications as diverse as LCD and OLED displays, new types of solar cells, optical films, conductive inks, medical diagnostic devices, barrier packaging and engine lubricants.
Buyers and investors would benefit from being able to buy nanomaterials, on an open and transparent platform, which have been subject to the AssuredNano Safety, Health and Environment (SHE) accreditation scheme. The exchange will provide them with the option to buy from more than one source.
INSCX's CEO is Charles McGovern, a qualified stockbroker and professional risk trader across securities and commodity markets for US and international investment houses. McGovern announced the exchange initiative at this year's NanoMaterials2010 conference on 8-10 June, in London. NanoCentral, which founded the event as part of its strategy to aid the commercial application of nanomaterials, supports the proposed platform.
Nanomaterials maturing
Presentations at the event came from the likes of David Baxter, the lead researcher in emerging risks for the global insurer Lloyds, Cientifica's Tim Harper, who has also founded a specialist VC firm, and Matthias Hölling, who manages technology transfer activity within ETH Zurich. The conference tackled some of the key challenges and risks facing suppliers of nanomaterials and developers of products exploiting them.
Discussions and presentations focused on alternative financing sources, with instructive examples of failed nanotechnology start-ups; an emphasis on developing products and solutions based on nanomaterials to meet real societal needs; demands for the long-term, addressing how to equip start-ups with the right management team that can take new technology from the realms of science and guide it towards commercial opportunities; media perceptions of nano and other emerging technologies; and managing risk from an insurer's perspective.
Hi Databoss.
Nanotech is the future, and a lot of amazing things are going on in nanoworld. Feel free to ad your input, and welcome aboard!
carbon nanotubes facilitates quantum leap in desalination technology
Revolutionary New Solution for Semiconductor, Nano Materials

Schematic of hybrid core-shell growth process
University of Maryland researchers have created a completely new way to produce high quality semiconductor materials critical for advanced microelectronics and nanotechnology. Published in the March 26 issue of Science, their research is a fundamental step forward in nanomaterials science that could lead to significant advances in computer chips, photovoltaic cells, biomarkers and other applications, according to the authors and other experts.
"This is a major, major advance that shows it is possible to do something that was impossible to do before," said Massachusetts Institute of Technology Associate Professor Francesco Stellacci, whose own work focuses on discovery of new properties in nanoscale materials and the development of new nanofabrication schemes. "This research actually shows that it's possible at the nanoscale for two materials to happily coexist at their interface, two materials that would not coexist otherwise," explained Stellacci, who was not involved in the study.
Led by Min Ouyang, an assistant professor in the department of physics and the Maryland NanoCenter, the University of Maryland team has created a process that uses chemical thermodynamics to produce, in solution, a broad range of different combination materials, each with a shell of structurally perfect mono-crystal semiconductor around a metal core.
Ouyang and fellow researchers Jiatao Zhang, Yun Tang and Kwan Lee, say their method offers a host of benefits over the existing process, known as epitaxy, used to create single crystal semiconductors and related devices. The biggest advantage of their non-epitaxial process may be that it avoids two key constraints of epitaxy -- a limit on deposition semiconductor layer thickness and a rigid requirement for "lattice matching."
The constraints of the epitaxial method restrict the materials that can be formed with it. For example, authors Ouyang, Zhang, Tang and Lee note that attempts to use epitaxy to achieve the kind of hybrid core-shell nanostructures they demonstrate in their article have been unsuccessful.
"Our process should allow creation of materials that yield highly integrated multi-functional microelectronic components; better, more efficient materials for photovoltaic cells; and new biomarkers," said Ouyang, who noted his team is in the process of applying for a patent. "We envision for example that we can use this method to create new types of photovoltaic cells that are ten times more efficient in converting sunlight to electricity than current cells.
"Our method doesn't require a clean room facility and the materials don't have to be formed in a vacuum the ways those made by conventional epitaxy do," Ouyang said. "Thus it also would be much simpler and cheaper for companies to mass produce materials with our process."
Epitaxy is one of the cornerstones of contemporary semiconductor industry and nanotechnology. It has been considered the most affordable method of high quality crystal growth for many semiconductor materials including silicon-germanium, gallium nitride, gallium arsenide, indium phosphide and graphene.
A Quantum Leap
The new method also can be used to design and fabricate artificial quantum structures that help scientists understand and manipulate the basic physics of quantum information processing at the nanoscale, said Ouyang, noting that he and his team have a separate paper on the quantum science applications of this method that they expect to be published in the near future.
This work was supported by the Office of Naval Research, the National Science Foundation (NSF) and the Beckman Foundation. Facility support was from Maryland Nanocenter and its Nanoscale Imaging, Spectroscopy and Properties Laboratory, which is supported in part by the NSF as a Materials Research Science and Engineering Centers shared experiment facility.
More information: "Nonepitaxial Growth of Hybrid Core-Shell Nanostructures with Large Lattice Mismatches," Jiatao Zhang, Yun Tang, Kwan Lee, Min Ouyang*, Science, March 26, 2010. http://www.sciencemag.org/
Provided by University of Maryland
Paper supercapacitor could power future paper electronics

Photos at left and center show the paper supercapacitor printed on Xerox paper. The right photo shows the paper supercapacitor printed on newspaper. Image credit: Liangbing Hu, et al. ©2010 AIP.
All those paper transistors and paper displays that scientists have been designing can now be powered by an onboard power source, thanks to the development of a new paper supercapacitor. Designed by researchers at Stanford University, the paper supercapacitor is made by simply printing carbon nanotubes onto a treated piece of paper. The researchers hope that the integrated design could lead to the development of low-cost, disposable paper electronics.
In the paper supercapacitor, all the necessary components are integrated onto a single sheet of paper in the form of single walled carbon nanotubes (SWNTs). High-speed printing could be used to print the SWNTs directly onto a piece of paper - anything from Xerox paper to newspaper and even grocery ads will work. At first, the researchers found that the SWNTs were so small that they penetrated the paper through micron-sized pores, which would cause the device to short-circuit. To solve this problem, the researchers first coated both sides of the paper with polyvinylidene fluoride (PVDF), which blocked the pores but still allowed for electrolytes to be transported through the paper. As such, the treated paper could function as an electrolyte membrane and separator without short-circuiting.
“The key design is that SWNTs stick well on paper and do not penetrate through paper completely to avoid shorting,” Yi Cui of Stanford University told PhysOrg.com.
Once the SWNTs were printed onto the treated paper, they experienced strong bonding forces similar to those experienced when writing with a pen or pencil on paper. Even when rubbed or subjected to tape, the SWNTs remained attached to the paper. After printing SWNTs on both sides of single sheets of paper, electrolyte was loaded to form a supercapacitor. The SWNTs served as both the electrodes and current collectors in the supercapacitor, which had a capacitance of about 3 F/g. The device also showed an excellent cycling stability, with very little loss of capacitance after 2500 cycles. The researchers say the same concept could be extended to make batteries, as well.
The fully integrated supercapacitor is based on an earlier version that the researchers made, in which nanomaterials were coated separately onto different anode and cathode substrates and then assembled together with a separator. The advantage of the new integrated structure is that it allows for high-speed printing, which greatly reduces fabrication costs and brings disposable, flexible, and lightweight paper electronics closer to reality. Cui said that, in the future, the researchers plan “to use this new design for real applications.”
More information: Liangbing Hu, Hui Wu, and Yi Cui. “Printed energy storage devices by integration of electrodes and separators into single sheets of paper.” Applied Physics Letters 96, 183502 (2010).
Nanoshell structures: Self-assembly method yields materials with unique optical properties

Heptamers containing seven nanoshells have unique optical properties. Credit: Rice University
Scientists from four U.S. universities have created a way to use Rice University's light-activated nanoshells as building blocks for 2-D and 3-D structures that could find use in chemical sensors, nanolasers and bizarre light-absorbing metamaterials. Much as a child might use Lego blocks to build 3-D models of complex buildings or vehicles, the scientists are using the new chemical self-assembly method to build complex structures that can trap, store and bend light.
The research appears in this week's issue of the journal Science.
"We used the method to make a seven-nanoshell structure that creates a particular type of interference pattern called a Fano resonance," said study co-author Peter Nordlander, professor of physics and astronomy at Rice. "These resonances arise from peculiar light wave interference effects, and they occur only in man-made materials. Because these heptamers are self-assembled, they are relatively easy to make, so this could have significant commercial implications."
Because of the unique nature of Fano resonances, the new materials can trap light, store energy and bend light in bizarre ways that no natural material can. Nordlander said the new materials are ideally suited for making ultrasensitive biological and chemical sensors. He said they may also be useful in nanolasers and potentially in integrated photonic circuits that run off of light rather than electricity.
The research team was led by Harvard University applied physicist Federico Capasso and also included nanoshell inventor Naomi Halas, Rice's Stanley C. Moore Professor in Electrical and Computer Engineering and professor of physics, chemistry and biomedical engineering.
Nordlander, the world's leading theorist on nanoparticle plasmonics, had predicted in 2008 that a heptamer of nanoshells would produce Fano resonances. That paper spurred Capasso's efforts to fabricate the structure, Nordlander said.
The new self-assembly method developed by Capasso's team was also used to make magnetic three-nanoshell "trimers." The optical properties of these are described in the Science paper, which also discusses how the self-assembly method could be used to build even more complex 3-D structures.
Nanoshells, the building blocks that were used in the new study, are about 20 times smaller than red blood cells. In form, they resemble malted milk balls, but they are coated with gold instead of chocolate, and their center is a sphere of glass. By varying the size of the glass center and the thickness of the gold shell, Halas can create nanoshells that interact with specific wavelengths of light.
"Nanoshells were already among the most versatile of all plasmonic nanoparticles, and this new self-assembly method for complex 2-D and 3-D structures simply adds to that," said Halas, who has helped develop a number of biological applications for nanoshells, including diagnostic applications and a minimally invasive procedure for treating cancer.
Solexant's Nanocrystal Solar Technology Lands $41.5M Series C Round
“Solexant’s innovative printed nanocrystal technology platform has the potential to deliver the lowest cost per watt in the solar industry”
Berkeley Researchers Light Up White OLEDs

Biwu Ma, a staff scientist with the Molecular Foundry, was part of a research team that found a new way to process white OLEDs for solid state lighting. (Photo by Roy Kaltschmidt, Berkeley Lab Public Affairs)
Light-emitting diodes, which employ semiconductors to produce artificial light, could reduce electricity consumption and lighten the impact of greenhouse gas emissions. However, moving this technology beyond traffic signals and laser pointers to illumination for office buildings and homes -- the single largest use of electricity -- requires materials that emit bright, white light cheaply and efficiently. White light is the mix of all the colors, or wavelengths, in the visible spectrum.
Organic light-emitting diodes (OLEDs), based on organic and/or polymer semiconductor materials, are promising candidates for general lighting applications, as they can cover large-area displays or panels using low-cost processing techniques. Indeed, single-color OLED displays are already available commercially. A mix of red-, green- and blue-emitting materials can be used to generate white light, but these bands of color often interact with one another, degrading device performance and reducing color quality.
Using polymer nanoparticles to house light-emitting ‘inks’, scientists at the Molecular Foundry, a U.S. Department of Energy nanoscience center located at Berkeley Lab, and the University of California, Berkeley, have made a thin film OLED using iridium-based guest molecules to emit various colors of visible light. The polymer nanoparticle surrounding a guest light-emitter serves as a ‘do not disturb’ sign, isolating guest molecules from one another. Each guest can then emit light without pesky interactions with neighboring nanoparticles, resulting in white luminescence.
“This simple and bright approach to achieving nanoscale site isolation of phosphors opens a new door for facile processing of white OLEDs for solid state lighting,” said Biwu Ma, a staff scientist with the Molecular Foundry’s Organic Nanostructures Facility who contributed to this study. With this proof-of-concept device under their belts, Ma and his colleagues plan to vary the ratio of each color nanoparticle in the OLED to enhance efficiency and brightness. White light from OLEDs can be adjusted from cooler to warmer whites, making these materials easy to use in office or home environments. Buildings account for more than 40 percent of carbon emissions in the United States, so replacing even a fraction of conventional lighting with OLEDs could result in a significant reduction in electricity use.
A paper reporting this research titled, “Site isolation of emitters within cross-linked polymer nanoparticles for white electroluminescence,” appears in the journal Nano Letters.
|
Followers
|
2
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
82
|
|
Created
|
05/30/10
|
Type
|
Free
|
| Moderators | |||
Nanotechnology - Wikipedia, the free encyclopedia
Nanotechnology basics, news, and general information
Nanotechnology news from physorg
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
