Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Did anyone notice more sells than buys today of INO is going up ? That looks very good.
yes...saw that...could be huge
4:10AM Inovio Pharma moves closer to 'universal' influenza vaccine by demonstrating protective immune responses against multiple H3N2 and Type B strains in animal studies (INO) 0.57 : Co announces that its synthetic vaccines for influenza Type A H3N2 and Type B achieved protective antibody responses in immunized animals against multiple unmatched strains. Inovio previously reported that its H5N1 synthetic vaccine achieved hemagglutination inhibition titers against six unmatched strains of this influenza subtype in a Phase I human study. The animals immunized with the SynCon H3N2 vaccine developed HI titers exceeding the 1:40 level. Additional animal testing of the remaining few H3N2 clades will continue in 2012 and will include a new strain, H3N2v, which was selected this month by the CDC as a pandemic vaccine target. Similarly, in the study of Inovio's SynCon Type B vaccine, investigators tested blood samples from immunized mice for immune responses against multiple, unmatched strains of Type B influenza. All the animals immunized with the SynCon? Type B vaccine developed HI titers exceeding the 1:40 level against all of the strains of Type B tested, including those circulating and consequently a part of the vaccine formulation in 2001-02, 2008-09, and 2011-12. Type B influenza mutates more slowly than Type A, but enough to preclude lasting immunity.
Inovio Pharmaceuticals and Keryx Biopharmaceuticals Benefit From Strengthening Biotech Sector
The Paragon Report Provides Equity Research on Inovio Pharmaceuticals & Keryx Biopharmaceuticals
MarketwirePress Release: Paragon Financial Limited – 2 hours 30 minutes ago
NEW YORK, NY--(Marketwire -01/25/12)- The biotechnology industry has performed well in the early stages of 2012. The iShares NASDAQ Biotechnology Index is presently up nearly eight percent month-on-month as the industry continues to recover nicely from the global economic crisis. According to a recent report from Global Industry Analysts, Inc. (GIA) the improving economic conditions have led to an increase in funding for the biotechnology industry. The Paragon Report examines investing opportunities in the Biotechnology Industry and provides equity research on Inovio Pharmaceuticals, Inc. (AMEX: INO - News) and Keryx Biopharmaceuticals Inc. (NASDAQ: KERX - News). Access to the full company reports can be found at:
www.paragonreport.com/INO
www.paragonreport.com/KERX
GIA predicts the Global Biotechnology Market to Surpass US$320 Billion by 2015, as increased availability of funding for R&D, expanding use of biotechnology in agriculture and medical sciences, and favorable government initiatives are expected to drive growth in the biotechnology industry.
GIA argues that developing markets, especially India and China, will emerge as major agricultural and industrial biotechnology markets owing to rapid increase in population and income levels. "The two countries are emerging as key markets for biotech drugs due to their rich talent pool and low cost of investment," GIA stated in a press release.
The Paragon Report provides investors with an excellent first step in their due diligence by providing daily trading ideas, and consolidating the public information available on them. For more investment research on the biotechnology industry register with us free at www.paragonreport.com and get exclusive access to our numerous stock reports and industry newsletters.
Inovio Biomedical Corporation is focused on developing multiple DNA-based immunotherapies. The Company develops applications of electroporation using brief, controlled electrical pulses to increase cellular uptake of a useful biopharmaceutical. Inovio is advancing its universal influenza vaccine strategy with a second Phase I influenza vaccine study.
Keryx Biopharmaceuticals, Inc., together with its subsidiaries, focuses on the acquisition, development, and commercialization of pharmaceutical products for the treatment cancer and renal disease.
The Paragon Report has not been compensated by any of the above-mentioned publicly traded companies. Paragon Report is compensated by other third party organizations for advertising services. We act as an independent research portal and are aware that all investment entails inherent risks. Please view the full disclaimer at http://www.paragonreport.com/disclaimer
Pick a GREAT company then All it takes is PATIENCE in todays market to receive the rewards. ![]() MBD
MBD
Round Two, Amigo
Between INO and ACTC....it was a good day.
Accumulation, WOW, great day!!!
this could be our breakout alert. lets keep it over .46 for the close.
Thank you for INO summary. Its electroporation delivery of DNA antigen is a novel idea and very interesting. But, I like to know if the company ever published a theory or fact about how the injected antigens traveled sequentially through each step in immune activating chain. Starting point is intramuscular injection enhanced by electroporation of limited area of muscle. Most of DNA injected is supposed to enter electroporated local muscle cells. This much is what I know and understand but what happens next? Is the muscle the place where antibody generated, or can the muscle cell migrate to some place to incite T-cell and antibody reaction? Or, does it excite local mechanism such as through dendric cell?
enough consolidating between >40 and .43....need to get over the 50 dma at .46 and hold it,.lots of gains between there and the 200 dma at .66
Lots of people buying today, but Market Makers are holding it down , maybe they are waiting for a big sell or maybe they are shorting , INO is a company that I would be hesitant to short to much of an up side here.
what's the deal with the high volume today. Looks like there was a large sale at the open followed by a peak in buying...
welcome...good to see a familiar name....
Bought some today future looks bright for INO.
nice find...thanks for posting
Universal Flu Vaccine Could Be Available by 2013
'Holy Grail' of Flu Vaccines by Next Year
By JASON KOEBLER
January 13, 2012
Annual flu shots might soon become a thing of the past, and threats such as avian and swine flu might disappear with them as a vaccine touted as the "holy grail" of flu treatment could be ready for human trials next year.
That's earlier than the National Institutes of Health estimated in 2010, when they said a universal vaccine could be five years off. By targeting the parts of the virus that rarely mutate, researchers believe they can develop a vaccine similar to the mumps or measles shot—people will be vaccinated as children and then receive boosters later.
That differs from the current '60s-era technology, according to Joseph Kim, head of Inovio Pharmaceuticals, which is working on the universal vaccine. Each year, the seasonal flu vaccinetargets three or four strains that researchers believe will be the most common that year. Previous seasons' vaccines have no effect on future strains of the virus, because it mutates quickly. The seasonal vaccine also offers no protection against outbreaks, such as 2009's H1N1 swine flu. A universal vaccine would offer protection against all forms of the virus.
"It's like putting up a tent over your immune system that protects against rapidly mutating viruses," Kim says. At least two other companies are working on a similar vaccine. In late 2010, Inovio earned a $3.1 million grant from the National Institutes of Health to work on the vaccine.
"It's a completely different paradigm than how [the vaccines] are made seasonably every year," Kim says.
Kim says early research has been promising. Flu strains fall into different "buckets," he says. All H1N1 strains share similar characteristics, as do all H5N1 strains, including the the Asian bird flu strain that has killed more than 60 percent of the 500 or so people it has infected over the past decade.
Kim says Inovio has already made and completed successful human tests for vaccines that protect against all H1N1 and H5N1 flu strains.
In late 2011, two research groups created a strain of H5N1 bird flu that could be passed from human to human, leading the World Health Organization to issue a statement that said they were "deeply concerned about the potential negative consequences" that publishing their research could cause. Some news outlets have called the new strain "engineered doomsday" and wondered whether terrorist organizations could create and distribute a similar virus. Kim says not to worry.
"I am very certain our vaccine can already neutralize that newly made virus," he says. "We're trying to get our hands on it."
Inovio is working on vaccines that'll protect against other strains, such as H3N2, which is seen in a newly-emerged swine flu virus. Those vaccines will be combined with the already-developed H1N1 and H5N1 vaccines to be delivered in one shot by the 2013 flu season. Researchers are taking a similar approach to HIV vaccine development, but working on the flu might be easier.
"Unlike other diseases, we have 50 plus years of diagnostics on the flu," Kim says. "There are lots of toolkits that let us know if our approach will work or not. ... Our goal is to have a vaccine strategy that can protect us from all mutations."
http://www.usnews.com/news/articles/2012/01/13/universal-flu-vaccine-could-be-available-by-2013
INO RESEARCH / DD SUMMARY!!!
by The $tockfather

Inovio Pharmaceuticals, Inc., formerly Inovio Biomedical Corporation, is engaged in the discovery, development, and delivery of vaccines, called deoxyribonucleic acid (DNA) vaccines, focused on cancers and infectious diseases. The Company's SynCon technology enables the design of universal DNA-based vaccines capable of providing cross-protection against new, unmatched strains of pathogens, such as influenza. Its electroporation DNA delivery technology uses brief, controlled electrical pulses to increase cellular DNA vaccine uptake. Inovio's clinical programs include human papillomavirus (HPV)/cervical cancer (therapeutic), avian influenza (preventative), hepatitis C virus (HCV) and human immunodeficiency virus (HIV) vaccines. It is advancing preclinical research for a universal seasonal/pandemic influenza vaccine. The Company's HIV vaccines consist of candidates for HIV prevention as well as therapy or treatment.
Management
J. Joseph Kim, Ph.D.
President, Chief Executive Officer, Director
Mark L. Bagarazzi
Chief Medical Officer
Peter Kies
Chief Financial Officer
Niranjan Y. Sardesai, Ph.D.
Chief Operating Officer
Revenue 10.52m
Net income -16.07m
Founded 1983
Employees 41
Average volume 528.10k
Shares outstanding 127.27m
Free float 114.81m
P/E (TTM) --
Market cap 55.07m USD
EPS (TTM) -0.1386 USD
Short Interest 568,281
Total assets 56
Total liabilities 9.56
FINANCIALS
http://markets.ft.com/research/Markets/Tearsheets/Financials?s=INO:ASQ&subview=BalanceSheet
SEC Filings
http://ir.inovio.com/secfilings
PRESS RELEASES
http://ir.inovio.com/index.php?year=2011&s=43
INSIDER TRADING
http://www.nasdaq.com/symbol/ino/insider-trades/sells
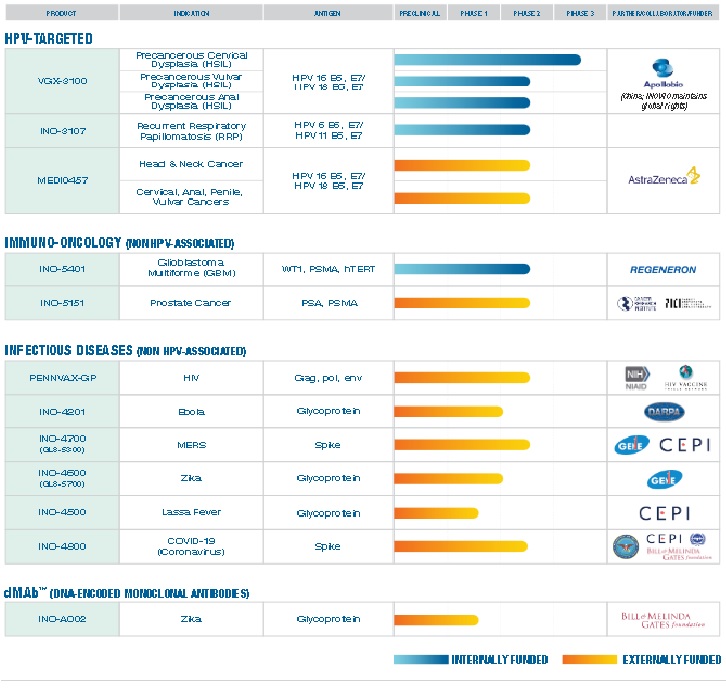
Inovio Product Pipeline
Inovio is focused on the discovery, development and delivery of an important new generation of vaccines, called DNA vaccines. The ability of DNA vaccines to induce not only a strong antibody response but also a strong T-cell response creates the potential to prevent and treat cancers and chronic infectious diseases. Furthermore, the capabilities of genomic engineering enables the design of optimized vaccines that may protect against a broad spectrum of evolving strains of a pathogen, such as influenza, that is not specifically matched to the genetic make-up of the vaccine.
Inovio's novel SynCon® DNA vaccine construct technology and electroporation DNA delivery technology have enabled the development of an active pipeline of proprietary preclinical and clinical DNA vaccines, as shown below. Multiple partners and collaborators are also using Inovio's electroporation technology under license to facilitate their DNA vaccine research. These programs are described in the following sections under the categories of cancers and infectious diseases.
Inovio has conducted early research to assess the potential utility of electroporation-delivered DNA vaccines for a range of diseases. These programs help to characterize the potential applicability and importance of DNA vaccines and may be the focus of additional future R&D effort. Various collaborators are also funding and conducting research using Inovio's electroporation-based DNA delivery systems in conjunction with experimental vaccines for different diseases.
Inovio has a non-vaccine, small molecule drug under development that is focused on inflammatory diseases including rheumatoid arthritis (RA), Type 1 diabetes (T1D), uveitis, and colitis.
The company owns a majority interest in VGX Animal Health, Inc., which markets LifeTide™ animal growth hormone for swine. LifeTide™ is one of only four DNA-based treatments approved for use in animals and is the only DNA-based agent delivered using electroporation that has been granted marketing approval (Australia).

Inovio Clinical Trials: Cancer
Inovio Pharmaceuticals or its partners/collaborators are conducting the following ongoing clinical trials for the treatment of cancers. Patients are being recruited for these trials, with details available by following the links below.
Indications: Cervical dysplasia / cancer caused by human papillomavirus (HPV)
Treatment: VGX-3100
Study title: Phase II Placebo Controlled Study of VGX-3100, (HPV16 E6/E7, HPV18 E6/E7 DNA Vaccine) Delivered IM Followed by Electroporation with CELLECTRA®-5P for the Treatment of Biopsy-proven CIN 2/3 or CIN 3 with Documented HPV 16 or 18
Trial Stage: Phase II
http://www.clinicaltrials.gov/ct2/show/NCT01304524?term=NCT01304524&rank=1
Indications: Acute and chronic myeloid leukemia (Partner/Sponsor: University of Southampton)
Treatment: DNA vaccine encoding for Wilms' tumor gene 1 (WT1)
Study title: WT1 Immunity via DNA Fusion Gene Vaccination in Haematological Malignancies by Intramuscular Injection Followed by Intramuscular Electroporation (WIN)
Trial Stage: Phase II
http://clinicaltrials.gov/ct2/show/NCT01334060?term=leukemia+southampton&rank=1
Indications: Breast, lung, prostate & other cancers
Treatment: V934/V935
Study title: A Phase I Investigation of the Safety, Tolerability and Immunogenicity of V934/V935 hTERT Vaccination in Cancer Patients With Selected Solid Tumors
Trial Stage: Phase I
http://clinicaltrials.gov/ct2/show/NCT00753415?term=v934&rank=1
Inovio Product Pipeline: Infectious Disease DNA Vaccines
DNA vaccine technology has created the potential for a new generation of infectious disease vaccines with not only preventive but therapeutic capabilities. Inovio has a proprietary DNA vaccine candidate for HIV in Phase I clinical studies, an avian influenza candidate awaiting IND approval, and multiple R&D programs for different infectious diseases. Many of these programs are being conducted and/or funded in collaboration with respected research organizations.
License partners and other collaborators are also using Inovio's electroporation DNA delivery technology to facilitate their vaccine research.
Multiple clinical studies are progressing and are described in the following sections.
Partners
Merck

In May 2004, Merck & Co. licensed Inovio's electroporation-based DNA delivery technology to use in conjunction with proprietary DNA vaccine candidates. The agreement granted Merck worldwide non-exclusive rights to use Inovio's electroporation technology for intramuscular delivery of certain proprietary DNA vaccines. The deal involves upfront and milestone payments, development fees, royalties, and a supply agreement.
Merck files first Investigational New Drug application, triggering a $2M milestone payment.
Merck's first clinical program.
Merck's files IND for a second DNA vaccine, triggering another $2M milestone payment.
Merck's second clinical program.
University of Southampton

The University of Southampton (UK) is advancing an investigator-sponsored clinical study using Inovio's technology in conjunction with a DNA-based vaccine against prostate cancer.
Southampton's clinical program.
Significant antibody response in humans reported at international vaccine conference.
Significant T-cell response in humans reported in scientific paper published in Nature Reviews Cancer.
Tripep

Tripep AS is advancing a Phase I/II clinical study for a hepatic C virus DNA vaccine using Inovio's DNA delivery technology.
Tripep clinical study.
Tripep announces pre-clinical results for hepatic C virus DNA vaccine.
HIV Vaccine Trials Network (HVTN)

The HIV Vaccine Trials Network (HVTN) is an international collaboration of scientists and educators searching for an effective and safe HIV vaccine. The HVTN's mission is to facilitate the process of testing preventive vaccines against HIV/AIDS. Our organization conducts all phases of clinical trials, from evaluating experimental vaccines for safety and the ability to stimulate immune responses, to testing vaccine efficacy.
HVTN is currently funding and conducting clinical trials of Inovio's PENNVAX™-B HIV DNA vaccine candidate.
The University of Pennsylvania

Inovio (through VGX Pharmaceuticals, with which it merged on June 1, 2009) entered into an original research agreement with the University of Pennsylvania on December 22, 2005. It entered into a subsequent license for exclusive worldwide rights to develop a number of DNA plasmids and constructs with the potential to treat and/or prevent HIV, HCV, HPV and influenza, and an amended agreement adding novel adjuvants to enhance the effectiveness of DNA vaccines. The underlying technology was developed in the laboratory of Professor David B. Weiner at the University of Pennsylvania. Professor Weiner is a pioneer in the field of DNA vaccines.
The University of Pennsylvania is conducting a clinical trial of Inovio's PENNVAX™-B HIV DNA vaccine, without electroporation, in a therapeutic setting.
The National Institute of Allergy and Infectious Diseases

The National Institute of Allergy and Infectious Diseases of the National Institutes of Health is supporting HIV DNA vaccine research being conducted by Inovio. The NIAID contract, announced October 1, 2008, provides $23.5 million of funding over seven years, including a base period and follow-on option years, to Inovio's PENNVAX™-G vaccine candidate targeting HIV clades A, C, and D, and its novel intradermal electroporation (ID-EP) technology.
PATH Malaria Vaccine Initiative (MVI)

The PATH Malaria Vaccine Initiative (MVI) is a global program of the international nonprofit organization PATH. MVI was established in 1999 through a grant from the Bill & Melinda Gates Foundation.
PATH MVI is assessing Inovio's SynCon® DNA vaccine development platform with respect to malaria.

USAMRIID, located at Fort Detrick, Maryland, is the lead medical research laboratory for the U.S. Biological Defense Research Program, and plays a key role in national defense and infectious disease research. The Institute's mission is to conduct basic and applied research on biological threats resulting in medical solutions (such as vaccines, drugs and diagnostics) to protect the warfighter. USAMRIID is conducting research using Inovio's electroporation technology.
US Army demonstrates protection from possible biological warfare agent in pre-clinical research.
Inovio receives third appropriation of US Dept. of Defense funding for US Army research.
National Cancer Institute

Inovio established an agreement with the National Cancer Institute to assess novel HIV constructs in non-human primates, cytokine genes as vaccine adjuvants (immune system stimulants), and possibly anticancer therapies delivered using Inovio's electroporation-mediated DNA delivery technology.
Inovio milestones
1996: First data in small animals using electroporation and gene therapy
2004: First industry license agreement of electroporation technology with a Big Pharma (Merck) relating to a DNA vaccine
2004: First-in-man clinical study using a DNA-based immunotherapy delivered with electroporation (with partner, Moffitt Cancer Center)
2007: First human data from a DNA-based immunotherapy delivered using electroporation (with partner, Moffitt Cancer Center)
2008: First DNA-based product delivered using electroporation approved for use in an animal (growth hormone replacement hormone (GHRH) for swine).
Analyst Coverage
Firm Brean Murray
Analyst Jonathan Aschoff, PhD
Phone 212-702-6652
Firm Cowen & Company
Analyst Edward Nash
Phone 646-562-1385
Firm JMP
Analyst Charles Duncan, PhD
Phone 212-906-3510
Firm Noble Financial
Analyst Nathan Cali
Phone 561-994-5723
Firm Rodman & Renshaw
Analyst Ren Benjamin, Ph.D.
Phone 212-430-1743
Firm ROTH Capital Partners
Analyst David S. Moskowitz, RPh.
Phone 646-358-1906
Industry Biotechnology & Drugs
Location
Inovio Pharmaceuticals Inc
11494 Sorrento Valley Road
San Diego92121
United States
USA
PATIENCE pays off BIGTIME here imo! Good Luck to all..MBD
Insider Buying 1/5/2012, Me too...GL
Resistance @ .435 and .44..if it busts thru .45 it's going to .52's
Following the chatter. INO Board & mods marked.
AGREED!! Have been adding all along. GREAT BUYS out in the market place! Adding to all my LONG positions! MBD
I agree. It's unfortunate some can't see through the foggy glass. This is a great business strategy on behalf of INO, as well a great buying opportunity for investors! Thank you for your post.
Nano,
I wrote to Investor Relations complaining about the new stock financing, and, sure enough got the answer that they weren't doing it for financing purposes, but in order to attract a few new institutional investors.
You will note that Austin & Greenhouse is selling out their entire position (see post by Surf1944). The company simply believes that they need institutional holders and wanted to bring on new ones to replace Austin & Greenhouse.
Austin & Greenhouse has a reputation for selling out at low prices (probably for tax losses for their clients), so having 5 new institutional investors with more conventional strategies (i.e. to make money) is a far cry better situation than they had before.
Therefore, this market reaction appears to me to be a buying opportunity.
Rip
Note: Company did NOT say that Austin & Greenhouse has a habit of selling low. I picked that up from several other sources on the internet.
Funds buying in.
What kind of radical change? Sorry,but I can't even guess what you are implying.
Rip
Could portend a radical change of some considerable import!
What happened to cause this SP decline?
It seems to me to be pretty simple. In their Nov. 7 presentation they indicated that they had $31 Million in cash, enough to fund their operations through mid-2013.
Then, 3 weeks later they announce a financing of $3.5 Million in the middle of a down market! No wonder people started running for the exits.
I have no idea what the story is here. It could be as simple as a way for them to get more institutions on board as shareholders. Institutions may have been wary of buying stock on the market because they may have been concerned about driving the stock price too high before they were able to fill their positions.
However, to state in November that they have plenty of money to last them through mid-2013 and then immediately turn around and do a new stock issuance is definitely a contradiction between what they say and what they do. The market reaction may pass, but I certainly would like an official explanation of this apparent contradiction.
I have no clue at all.
Inovio Pharma announces $3.5 mln underwritten financing (INO) 0.52 : Co announced that it has priced an underwritten public offering of 6,737,247 shares of its common stock and warrants to purchase up to 5,052,935 additional shares of its common stock purchased by five institutional investors. The shares of common stock and warrants are being offered in units, consisting of one share of common stock and 0.75 of a warrant to purchase one share of common stock, at a price of $0.5195 per unit.
yes but it didn't happen
are you sure?
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=68606693
someone said
INO will be the stock of the year in 2012
I think .48 will hit by eod 20% from your alert!
nothing concrete yet.. it will take at list one year to develop something.. they need to dilute for financing
Not sure, just IMO, I seen a bottom went in but ended up as a daytrade, since I can't find any reason it was selling so much yesterday, hit my scanners and bought in early, the New$$ helped the volume, thought the New$$ would take it back up??
expecting another big dump here..
INO is a good intraday trade today I alerted earlier, not sure who sold, not sure if this is a long term play.
WHY DO YOU THINK (INO .40) flopped on Funding New$??
Placing INO back on the watch list this morning:
Inovio Pharmaceuticals Announces $3.5 Million Underwritten Financing
PR NewswirePress Release: Inovio Pharmaceuticals, Inc. – 45 minutes ago
BLUE BELL, Pa. , Dec. 1, 2011 /PRNewswire/ -- Inovio Pharmaceuticals, Inc. (NYSE Amex: INO), which is advancing synthetic vaccines to fight cancers and infectious diseases, announced today that it has priced an underwritten public offering of 6,737,247 shares of its common stock and warrants to purchase up to 5,052,935 additional shares of its common stock purchased by five institutional investors. The shares of common stock and warrants are being offered in units, consisting of one share of common stock and 0.75 of a warrant to purchase one share of common stock, at a price of $0.5195 per unit. Inovio also granted the underwriter a 45-day option to purchase at the public offering price up to an aggregate of 1,010,587 additional units to cover overallotments, if any. The warrants have a term of five years and an exercise price of $0.65 per share. Inovio may call these warrants if the closing bid price of the common stock has been at least $1.30 over 20 trading days and certain other conditions are met.
The gross proceeds of the offering are expected to be $3.5 million . Net proceeds, after deducting the underwriter's discounts and other estimated offering expenses payable by Inovio, and assuming no exercise of the over-allotment option, are expected to be approximately $3.2 million . Brean Murray , Carret & Co. is acting as the sole bookrunner of this offering. Inovio intends to use proceeds from the offering for general corporate purposes, including clinical trial expenses and research and development expenses. The offering is expected to close on or about December 6, 2011 , subject to the satisfaction of customary closing conditions.
The securities described above are being offered by Inovio pursuant to a registration statement previously filed and declared effective by the Securities and Exchange Commission, or the SEC. A prospectus supplement related to the offering will be filed with the SEC. Copies of the prospectus supplement and accompanying base prospectus relating to this offering may be obtained at the SEC's website at www.sec.gov or from Brean Murray , Carret & Co., LLC by e-mail to syndicate@bmur.com or Inovio by mail at 1787 Sentry Parkway West, Building 18, Suite 400, Blue Bell, Pennsylvania 19422. This announcement is neither an offer to sell nor a solicitation of an offer to buy any of our common stock. No offer, solicitation or sale will be made in any jurisdiction in which such offer, solicitation or sale is unlawful.
(WO2011137221) ORAL MUCOSAL ELECTROPORATION DEVICE AND USE THEREOF
Pub. No.:
WO/2011/137221
International Application No.:
PCT/US2011/034277
Publication Date:
03.11.2011
International Filing Date:
28.04.2011
Applicants:
INOVIO PHARMACEUTICALS, INC. [US/US]; 1787 Sentry Parkway West Building 18, Suite 400 Blue Bell, Pennsylvania 19422 (US) (For All Designated States Except US).
KEMMERRER, Stephen V. [US/US]; (US) (For US Only).
MCCOY, Jay [US/US]; (US) (For US Only).
BRODERICK, Kate [GB/US]; (US) (For US Only)
Inventors:
KEMMERRER, Stephen V.; (US).
MCCOY, Jay; (US).
BRODERICK, Kate; (US)
Agent: KIM, Thomas S.; Inovio Pharmaceuticals, Inc. 1787 Sentry Parkway West Building 18, Suite 400 Blue Bell, Pennsylvania 19422 (US)
Priority Data:
61/328,868 28.04.2010 US
Title (EN) ORAL MUCOSAL ELECTROPORATION DEVICE AND USE THEREOF
(FR) DISPOSITIF D'ÉLECTROPORATION À USAGE ORAL, APPLIQUÉ À UNE MUQUEUSE, ET SON UTILISATION
Abstract:
(EN)The present invention relates to oral electroporation (EP) devices that are able to generate an electroporation causing electrical field at the mucosal layer, and preferably in a tolerable manner. Further, it includes the generation of a protective immune response, cellular and/or humoral, using the oral EP device along with a genetic construct that encodes an immunogenic sequence.
Summary of the Invention
There are provided electroporation devices capable of electroporating cells of a mucosal membrane of a mammal. Such devices include an electrode microneedle plate, a counter electrode plate, a main housing and an energy source. The main housing is in physical communication with said microneedle plate and counter electrode plate, wherein the main house is in fluid communication with a syringe capable of storing a pharmaceutical formulation for delivery. The energy source is in electrical communication with the microneedle plate and capable of generating an electric potential and delivering the electric potential to the cells through the microneedle plate.
http://is.gd/RrHVLX
See Fig. 17 for cheek electroporation device.
http://www.wipo.int/patentscope/search/docservicepdf_pct/id00000015503208
Inovio Pharma announced earlier Synthetic Avian flu vaccine demonstrates inhibition of multiple H5N1 strains in Phase I trial (INO) 0.57 : Co announced that a single intradermal electroporation boost of its SynCon avian influenza vaccine generated HAI titers against six different, unmatched strains of H5N1 - a distinct new clinical achievement on the global research community's path to develop universal influenza vaccines. This single synthetic vaccine generated a four-fold or greater rise in hemagglutination inhibition titers in 50% of boosted subjects in its Phase I clinical study. In the intramuscular part of the study, VGX-3400X generated antigen-specific antibody and cytotoxic T-lymphocyte responses against all three antigens that the vaccine was encoded to produce. High levels of binding antibodies were observed in 27 of 28 evaluated subjects. After two vaccinations, 13 of 18 vaccinated subjects from the first two cohorts developed strong CTL responses to at least one of the vaccine components. The patients boosted with the minimally invasive ID vaccination have been given a second ID booster vaccination. Additional interim data is expected in 1Q 2012.
Q looks VERY good and above what i expected!![]() Blue skies ahead for INO! What i really liked from the Q was this! Have to take a flight haven't had time to read further.
Blue skies ahead for INO! What i really liked from the Q was this! Have to take a flight haven't had time to read further.
Capital Resources As of September 30, 2011, cash and cash equivalents plus short-term investments in certificates of deposit were $31.1 million, compared with $21.8 million as of December 31, 2010. This change primarily resulted from the January 2011 financing, from which the Company received net proceeds of approximately $23.0 million, partially offset by expenditures related to our research and development and general and administrative activities. Based on management's projections and analysis, the Company believes that its cash and cash equivalents are sufficient to meet its planned working capital requirements through the second quarter of 2013.
|
Followers
|
629
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
40503
|
|
Created
|
07/25/08
|
Type
|
Free
|
| Moderators | |||







DISCLAIMER The Board Assistants herewith, are not licensed brokers and assume NO responsibility for the actions, investment decisions, and or messages posted on this forum.
• We do NOT recommend that anyone buy or sell any securities posted herewith. Any trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.
Whatch out for fake comments by manipulators
https://www.griproom.com/fun/how-to-spot-fake-comments-on-stock-boards
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |