Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Here is a great web site for FX info.
https://www.fraxa.org/fragile-x-news-updates/
LOL,Stockfun,I hear you,I had to vent a tad today.
I feel for the FDA at at this point,and the load that's on them.
Not being able to meet in person with companies is slowing things down for everyone.My gut feeling keeps me here though,and if that instinct pays off...
Solid support around 3.50. Wish I had some cash to buy more!!
Lol!! Your telling me. Every time I think it can go lower it does just that![]() It’s clear that investor sentiment is low on this stock, but it really is a buy. People just not trusting since they surprised us with the FX news last time. I think this will surprise us to the (+ ) on the upcoming FDA meeting news and then a rush to buy will occur.
It’s clear that investor sentiment is low on this stock, but it really is a buy. People just not trusting since they surprised us with the FX news last time. I think this will surprise us to the (+ ) on the upcoming FDA meeting news and then a rush to buy will occur.
The next news from FDA on Zygel and ASD subject will be on a path forward for the next trial.Phase ll results looked excellent.I suspect FDA will ask for a phase lll on ASD.
The next FX news could bring a green light from FDA for NDA on FMFX
To say these next four months are important for Zanerba,would rank in the top five understatements of the year.
Here's to success to all involved!
If we can get approval for ASD and Fully Methylated FX, this stock will be golden again. I’m hoping we get word back on FDA talks re: one or both in Sept/Oct. The RSI on this stock for 5 day, 1month, 3 month and 1 yr all suggest that we are at oversold prices. We are at a level of support @$3.65- $3.70 so I’m seriously thinking about buying because these are steal prices. Just hate to get surprise bad news again. That FX took me for a loop. Really wasn’t expecting that one ![]()
Yes sir. This is where I’ve always thought Zynerba’s biggest opportunity for success is. Parents need something safe and effective to help these children. I’m hoping for good news soon.
Some facts on ASD.
Autism Cases on the Rise; Reason for Increase a Mystery
Scientists are scouring genetic and evironmental data to find a cause for the rise in autism.
By Kathleen Doheny
FROM THE WEBMD ARCHIVES
The number of children diagnosed with autism or related disorders has grown at what many call an alarming rate. In the 1970s and 1980s, about one out of every 2,000 children had autism.
Today, the CDC estimates that one in 150 8-year-olds in the U.S. has an autism spectrum disorder, or ASD. This expanded definition refers not only to autism but also to a collection of brain development disorders such as Asperger's syndrome and a condition known as pervasive developmental disorder -- not otherwise specified (PDD-NOS). Though all the disorders share some symptoms, they are different in other ways, including the timeline of symptoms and the severity, according to the CDC.
The apparent rise in cases triggers two burning questions for parents, physicians, and scientists:
Is autism truly on the rise, or do the new statistics simply reflect the growing awareness of the condition, the expanded definition, and other factors?
If autism is on the rise, as most experts believe, what is causing the increase?
Autism: A True Increase or Semantics?
The jump in autism cases has spawned not only alarm but also debate about whether the number of children with autism could have increased that much in a relatively brief time.
"There's a lot of controversy about that," says Jeff Milunsky, MD, director of clinical genetics and associate director of the Center for Human Genetics at Boston University.
Two researchers who tracked the rate of autism in children born in the same area of England from 1992 to 1995 and then from 1996 to 1998 found that the rates were comparable, and concluded that the incidence of autism was stable. The study was published in the American Journal of Psychiatry in 2005.
But, Milunsky says, several studies have documented an increase in the U.S.
In a recent report in the journal Archives of Disease in Childhood, Milunsky and his colleagues point to several studies finding an increase in autism rates. In 2003, for instance, a large study conducted in Atlanta found that one in 166 to one in 250 children had autism, according to a report published in the Journal of the American Medical Association.
SUGGESTED
Another study conducted by the CDC in 14 states found an overall prevalence of one in 152, which Milunsky and others say is the generally accepted figure today.
Other experts say autism is on the increase but that factors other than more children being diagnosed play a role. Some of the increase in reported cases is because of "diagnostic substitution," says Paul Shattuck, PhD, assistant professor of social work at Washington University in St. Louis and an autism researcher.
"A kid labeled autistic today could have been labeled mentally retarded 10 years ago in the same school system," Shattuck says. It wasn't until 1992 that schools began to include autism as a special education classification.
Today, children diagnosed as having autism spectrum disorder are often more mildly affected than the classic "Rain Man" stereotype some people associate with the disorder, Shattuck says. After autism was first identified in 1943, some of the first studies found most of the children intellectually disabled. "Today the minority of kids [with ASD are intellectually disabled],'' Shattuck tells WebMD.
The debate about whether the reported increase in autism is affected by factors such as more awareness misses the point, says Isaac Pessah, PhD, a professor of toxicology, director of the Center for Children's Environmental Health Sciences, and a member of the MIND Institute at the University of California Davis. Rather than argue about whether the increase is because of some children being reclassified or other factors, he says, "We need to understand why it's one in 150."
Focusing on the actual numbers -- rather than the debate -- is wise, says Craig Newschaffer, PhD, chairman and professor of the department of epidemiology and biostatistics at Drexel University School of Public Health in Philadelphia. "We thought autism was a very rare occurrence, and it's clear that it's not."
Getting to the Causes of Autism
Getting to the cause -- or, more accurately, causes -- of autism will be more difficult than unraveling the causes of cancer, says Gary Goldstein, MD, president and CEO of Kennedy Krieger Institute in Baltimore, a facility that helps children with autism and other developmental disorders.
SUGGESTED
A reminder...there is this news also in the pipe line.
27 May 20
Zynerba Pharmaceuticals Announces Positive Top Line Results from Exploratory Open Label Phase 2 BRIGHT Trial of Zygel in Autism Spectrum Disorder
– THE BRIGHT TRIAL ACHIEVED STATISTICALLY SIGNIFICANT AND CLINICALLY MEANINGFUL IMPROVEMENTS FROM BASELINE IN ALL SUBSCALES OF THE ABERRANT BEHAVIOR CHECKLIST –
– SAFETY DATA REINFORCE EXCELLENT TOLERABILITY PROFILE OF ZYGEL –
– ZYNERBA TO HOST CONFERENCE CALL AND WEBCAST TODAY, MAY 27, 2020 AT 8:30 AM ET –
Devon, PA, May 27, 2020 — Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, today announced positive top line results from the exploratory, open label Phase 2 BRIGHT (An Open-Label Tolerability and Efficacy Study of ZYN002 Administered as a Transdermal Gel to Children and Adolescents with Autism Spectrum Disorder) trial.
The trial was designed to assess the safety, tolerability and efficacy of Zygel™ in pediatric and adolescent patients with autism spectrum disorder (ASD). Zygel was administered to patients with moderate-to-severe symptoms of ASD as add-on therapy to their standard of care utilizing a variety of efficacy assessments. Key findings from the trial disclosed today include:
All five subscales of the Aberrant Behavior Checklist – Community (ABC-C) as well as the Parent Rated Anxiety Scale – Autism Spectrum Disorder (PRAS-ASD) showed both statistically significant and clinically meaningful improvements at 14 weeks of treatment from baseline;
The results observed in other efficacy outcome measures, including Clinical Global Impressions – Improvement scale (CGI-I), support the subscale results observed in the ABC-C;
Zygel was well tolerated in this trial with no serious or severe adverse events reported.
“We are very encouraged by the compelling top line results of the BRIGHT trial and we expect to meet with the FDA to discuss the clinical pathway for developing Zygel for the treatment of behavioral symptoms of ASD in the second half of this year,” said Armando Anido, Zynerba’s Chairman and Chief Executive Officer. “Our goal is to develop Zygel for patients suffering from debilitating neuropsychiatric disorders including ASD, Fragile X syndrome, 22q and DEE. I want to thank the patients, families, physicians, clinical staff, and the Zynerba team for their support of this key study in ASD.”
Study Design
The 14-week exploratory, open label Phase 2 BRIGHT trial enrolled 37 patients with ASD at a single clinical site. The trial was designed to evaluate the safety, tolerability and efficacy of Zygel in children and adolescents ages three to 17 with ASD as confirmed by DSM-5 diagnostic criteria. Enrolled patients received weight-based doses of 250 mg or 500 mg daily of Zygel.
Patient Disposition and Baseline Demographics
Thirty-seven (37) patients were enrolled in the trial and are included in the safety analysis. One patient was lost to follow up with no post-treatment efficacy evaluation and, as a result, thirty-six (36) patients are included in the efficacy analyses. Twenty-eight (28) patients completed the 14-week trial. The discontinuation rate is consistent with other trials in ASD.
The mean age of patients enrolled in the trial was 9.2 years and thirty-four (92%) of the patients were male. Patients weighed between 15 and 108 kilograms (mean=41.6; median=30.2). The mean time to diagnosis in this population was 5.4 years. Using the Autism Diagnostic Observation Schedule (ADOS-2), 94% of enrolled patients had moderate-to-severe symptoms of ASD. The mean baseline ABC-C Irritability subscale score of 30.3 further supports the severity of the enrolled patient population. Ninety-two percent (92%) of patients entered the trial with the use of at least one underlying medication. Sixty-five percent (65%) of patients were on at least one psychotropic medication, for example, anti-depressants, anxiolytics and antipsychotics.
Top-line Efficacy Results
The trial evaluated multiple efficacy assessments, including the ABC-C, PRAS-ASD, Autism Parenting Stress Index, Autism Impact Measure (AIM), and Clinical Global Impression – Severity (CGI-S) and Improvement (CGI-I). The ABC-C irritability subscale was used as the basis for approval for the two atypical antipsychotics indicated for ASD.
Results from each of the subscales of the ABC-C after 14 weeks of treatment with Zygel are as follows:
ABC-C Subscale Baseline
(n=36) Week 14
(n=28) Mean %
improvement p Value
Irritability 30.3 18.2 39.1% <0.0001*
Inappropriate Speech 7.4 5.2 42.5% 0.0002*
Stereotypy 12.3 7.9 39.1% <0.0001*
Social withdrawal 25.1 16.5 36.4% <0.0001*
Hyperactivity 37.0 23.9 35.6% <0.0001*
*Statistically significant
The results of other efficacy assessments reinforce the results demonstrated in the ABC-C. For example, patients on Zygel experienced a mean improvement of 46% at week 14 from a baseline score of 40.8 as measured by the PRAS-ASD (p<0.0001) and 57% of patients were assessed as “very much improved” or “much improved” at week 14 as measured by the CGI-I.
“I am very impressed with the improvements my patients made over the 14-week treatment period while receiving Zygel; the reduction in irritability, communication deficits, and repetitive movements were especially noteworthy since some of these are core autistic behaviors,” said Helen Heussler, FRACP, Associate Professor at Children’s Health Queensland, Medical Director Child Development and principal investigator in the BRIGHT trial. “The magnitude of effect on autistic behaviors in this trial is significant, including hyperactivity and stereotypy, which are among the most difficult behaviors to improve with therapeutic intervention. The results of this study strongly suggest the potential of this drug as an important treatment for ASD and I look forward to participating in future clinical studies with Zygel.”
Safety and Tolerability
Zygel was very well tolerated, and the safety profile was consistent with previously released data from other Zygel clinical trials. Less than half (49%) of the patients experienced any adverse event (whether unrelated or related to study drug), all of which were mild (75%) or moderate (25%). Only 14% of patients experienced an adverse event that was deemed to be treatment-related, all of which were application site-related; most were mild and transient. There were no severe or serious adverse events reported during the study. Eighteen (18) patients who completed the BRIGHT trial have rolled into the open label extension.
Conference call information
Zynerba management will host a live conference call and webcast today at 8:30 am Eastern Time to discuss the results of this clinical trial. The call can be accessed by dialing (866) 573-0180 (U.S. and Canada) or (430) 775-1345 (international) and referencing conference ID 6196218. To access the live webcast or the replay, visit the investor page of the Company’s website at http://ir.zynerba.com/. The webcast will be recorded and available on the Company’s website for 30 days.
About Autism Spectrum Disorder (ASD)
Autism Spectrum Disorder is a developmental disorder that affects communication and behavior in approximately one million pediatric and adolescent patients between the ages of five and 17 in the U.S. It refers to a range of conditions characterized by anxiety, repetitive patterns of behavior, impairments in social communication including verbal and non-verbal communication, and deficits in developing and maintaining relationships. Although autism can be diagnosed at any age, it is said to be a “developmental disorder” because symptoms generally appear in the first two years of life. Research suggests that genes can act together with influences from the environment to affect development in ways that lead to ASD. Newer studies suggest that ASD is linked to disruption in the endocannabinoid system.
About Zynerba Pharmaceuticals, Inc.
Zynerba Pharmaceuticals is the leader in pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders. We are committed to improving the lives of patients and their families living with severe, chronic health conditions including Fragile X syndrome, autism spectrum disorder, 22q11.2 deletion syndrome, and a heterogeneous group of rare and ultra-rare epilepsies known as developmental and epileptic encephalopathies. Learn more at www.zynerba.com and follow us on Twitter at @ZynerbaPharma.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from the Company’s current expectations. Management’s expectations and, therefore, any forward-looking statements in this press release could also be affected by risks and uncertainties relating to a number of other factors, including the following: the Company’s cash and cash equivalents may not be sufficient to support its operating plan for as long as anticipated; the Company’s ability to obtain additional funding to support its clinical development programs; the results, cost and timing of the Company’s clinical development programs, including any delays to such clinical trials relating to enrollment or site initiation; clinical results for the Company’s product candidates may not be replicated or continue to occur in additional trials and may not otherwise support further development in a specified indication or at all; actions or advice of the U.S. Food and Drug Administration and foreign regulatory agencies may affect the design, initiation, timing, continuation and/or progress of clinical trials or result in the need for additional clinical trials; the Company’s ability to obtain and maintain regulatory approval for its product candidates, and the labeling under any such approval; the Company’s reliance on third parties to assist in conducting pre-clinical and clinical trials for its product candidates; delays, interruptions or failures in the manufacture and supply of the Company’s product candidates the Company’s ability to commercialize its product candidates; the size and growth potential of the markets for the Company’s product candidates, and the Company’s ability to service those markets; the Company’s ability to develop sales and marketing capabilities, whether alone or with potential future collaborators; the rate and degree of market acceptance of the Company’s product candidates; the Company’s expectations regarding its ability to obtain and adequately maintain sufficient intellectual property protection for its product candidates; the timing and outcome of current and future legal proceedings; and the extent to which health epidemics and other outbreaks of communicable diseases, including COVID-19, could disrupt our operations or adversely affect our business and financial conditions. This list is not exhaustive and these and other risks are described in the Company’s periodic reports, including the annual report on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K, filed with or furnished to the Securities and Exchange Commission and available at www.sec.gov. Any forward-looking statements that the Company makes in this press release speak only as of the date of this press release. The Company assumes no obligation to update forward-looking statements whether as a result of new information, future events or otherwise, after the date of this press release.
Zynerba Contact
William Roberts, Vice President, Investor Relations and Corporate Communications
Zynerba Pharmaceuticals
484.581.7489
robertsw@zynerba.com
Media contact
Molly Devlin
Evoke KYNE
215.928.2199
Molly.Devlin@evokegroup.com
This site uses cookies to give you a better browsing experience. By continuing to use this website, you accept our use of these cookies. For more information, see our Privacy Notice.
CLOSE
Privacy Policy
Terms of Use
Contact Us
© 2020 Zynerba Pharmaceuticals, Inc. All rights reserved
Well I thought we would have heard by mid to late Aug.So much for my feeling...Anyway.....Guess every day that goes by brings us a day closer to news.
CLAWS OUT, AND HANGING ON...
Same here my friend. Same here. You think we will here anything soon regarding FDA talks?
Hey STOCKFUN ,Im just waiting.Been away from the computer for a while,still holding all shares.
Price is attractive down here but really hard to put more into it until I see a little more from company.
Hey dhbuzz. Curious about your thoughts on that update done by the CEO a few days ago. Market did not respond well. I didn’t here CEO say much we didn’t already know. Stock was upgraded by Cannacord Genuity but market did not respond favorably. Any way, any thoughts??
Has anyone had discussion with the company directly regarding their feelings on FDA approval for fully methylated FX patients. Seems that their favorable “pre-planned” additional arm studies in this group would be in their favor. If it were something they had added later on as the trial was being conducted I’d say FDA frowns. But “pre-planned” was really a smart move on their part.
Cannabinoid Manufacturing:
R. Mannion. Cannabinoid Manufacturing & Product Quality.
U.S. Food and Drug Administration’s Scientific Data and Information about Products Containing Cannabis or Cannabis-Derived Compounds Public Hearing. Silver Spring, MD. May 31, 2019.
Zynerba presentation from FDA’s Cannabis or Cannabis-Derived Compounds public hearing
https://zynerba.com/wp-content/uploads/2019/05/Mannion.Ray_.Zynerba_Pharmaceuticals.pdf
...Zynerba believes that its manufacturing process will allow it to reproducibly manufacture its transdermal CBD gel to meet the stringent global regulatory agencies’ standards while ensuring consistent and efficient supply – without the costs and logistical requirements of growing and harvesting Cannabis....
Zygel™ (ZYN002 CBD Gel) is the first and only pharmaceutically-manufactured CBD, a non-euphoric cannabinoid, formulated as a patent-protected permeation-enhanced gel for transdermal delivery through the skin and into the circulatory system. Zygel is being developed for patients suffering from neuropsychiatric conditions including Fragile X syndrome (FXS), Autism Spectrum Disorder (ASD) in pediatric patients, 22q, and certain rare refractory epilepsies called DEEs. The U.S. Food and Drug Administration has granted Orphan Drug designation to Zynerba for the use of CBD for treatment of patients with FXS.
80 W. Lancaster Avenue, Suite 300
Devon, PA 19333
484.581.7505
https://zynerba.com/our-focus/why-pharmaceutical-manufacturing/
Leveraging manufacturing technology to CBD
Zynerba utilizes an established pharmaceutical manufacturing process to replicate the structure and function of the CBD found in the Cannabis plant. Lot consistency, reproducibility, purity and supply are required elements of any pharmaceutical manufacturing process. Zynerba believes that its manufacturing process will allow it to reproducibly manufacture its transdermal CBD gel to meet the stringent global regulatory agencies’ standards while ensuring consistent and efficient supply – without the costs and logistical requirements of growing and harvesting Cannabis.
Our commitment to patients drives our research efforts into pharmaceutically manufactured CBD – the same structure and function, without plants
https://zynerba.com/our-focus/why-pharmaceutical-manufacturing/
Standing by to put launch team in place...
SHOUT OUT ! TO LUMPY hope all is good.
Hang on longs...
Nice to see the 3.90 mark reached this morning.4's will be reached again soon IMO.Looking forward to todays update and Q&A.
Resetting this chess board.
If anyone can tell me at this point why zygel will not get a green light from FDA for NDA status for (“full methylation”)concerning FXS group I'm all ears.
Yes sir... I agree 100%
Stockfun this is the point to focus on...
A pre-planned ad hoc analysis of the most severely impacted patients in the trial, as defined by patients having at least 90% methylation (“full methylation”) of the impacted FMR1 gene, demonstrated that patients receiving Zygel achieved statistical significance in the primary endpoint of improvement at 12 weeks of treatment in the Social Avoidance subscale of the ABC-CFXS compared to placebo (p=0.020). This group comprised 80% of the patients enrolled in the CONNECT-FX study
If FDA comes back with an all clear for this group (FULL METHYLATION) for NDA.....................Lots of things change...........
Short answer....yes.To buy out.
I can only guess as to when we get FDA feedback on last FX trial results.
I'm thinking mid Aug. at the latest.
$zyne $3.48 v -0.11 (-3.06%)
Volume: 908,936 @07/17/20 7:59:26 PM EDT
Do you think Zynerba is vulnerable to a buy out or take over with how the PPS has been depressed? And any contact with company to know when meetings with FDA will take place??
I think the way Zanerba went about this last trial was the right way.It proved that the full blown FX group using zygel met the requirements.IMO this should give the FDA ample reason to give Zanerba NDA green light for this group.Then Zanerba can work with FDA on any future FX trials with other groups.WE HAVE A WINNER HERE FOR THOSE WHO SUFFER THE MOST WITH FX
I also think the last trial results for the population with full blown FX,will play a major role in negotiations with a foreign pharma co.
Took a while to straighten out with ihub,i have to use on screen key board now.PITA................I've added some shares with this pull back,and feel good with where Zanerba is positioned.IMO FDA news early to mid Aug onFX.Also watching out for DEES news.Good luck friend and all longs.SHOUT OUT TO LUMPY!
So glad to here your thoughts on ZYNE. As far as the windows 10. I feel you! Has a similar problem myself. Nightmare![]()
Thanks Stockfun.To make a long story short,I down loaded windows 10 and the nightmare began.As far as ZYNE,I feel strongly that Zygel will get the green light from FDA for full blown Fx Syndrome NDA.I think we will get news before end of Aug.
curious about that myself. signed up just to try to find out.
So glad to see you back. Hope you’ve been okay. You disappeared before the announcements and the plummet and we all got worried. People wondering whether you were just a paid pumper. Any who.... I’ve been here telling others this stock is not dead. And these are buying opportunities. What are your thoughts about the path forward??
WOW''''IM STILL HERE AND STILL BUYING
Really?? It doesn’t matter how much OTC CBD is out there to buy. If it doesn’t have FDA approval and have proven indications for a disease state then it will never replace pharmaceutical grade, clinically tested and proven medicine. For example. The otc world has tons of Omega 3 fish oil that people buy everyday. It’s probably a billion dollar otc market. Yet and still Amarin still has the prescription (Vascepa) as there lead product doing gang busters in sales because Physicians write this product for its proven indications and FDA approval. ZYNE will survive. Still a strong buy. To $8 with next news cycle!!
News: $ZYNE Face the Facts: The CBD Hype Train Has Derailed
For years, there wasn't a hotter investment opportunity on Wall Street than marijuana. With tens of billions of dollars in sales being conducted in the black market, it seemed only logical that North America's legal cannabis stocks would benefit after Canada became the first industrialized cou...
Got this from ZYNE - Face the Facts: The CBD Hype Train Has Derailed
Yep. Been trading some and buying back. Same idiots making market at AFH, AIKI are here too. So, we play them the same way....
|
Followers
|
91
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2676
|
|
Created
|
08/06/15
|
Type
|
Free
|
| Moderators | |||

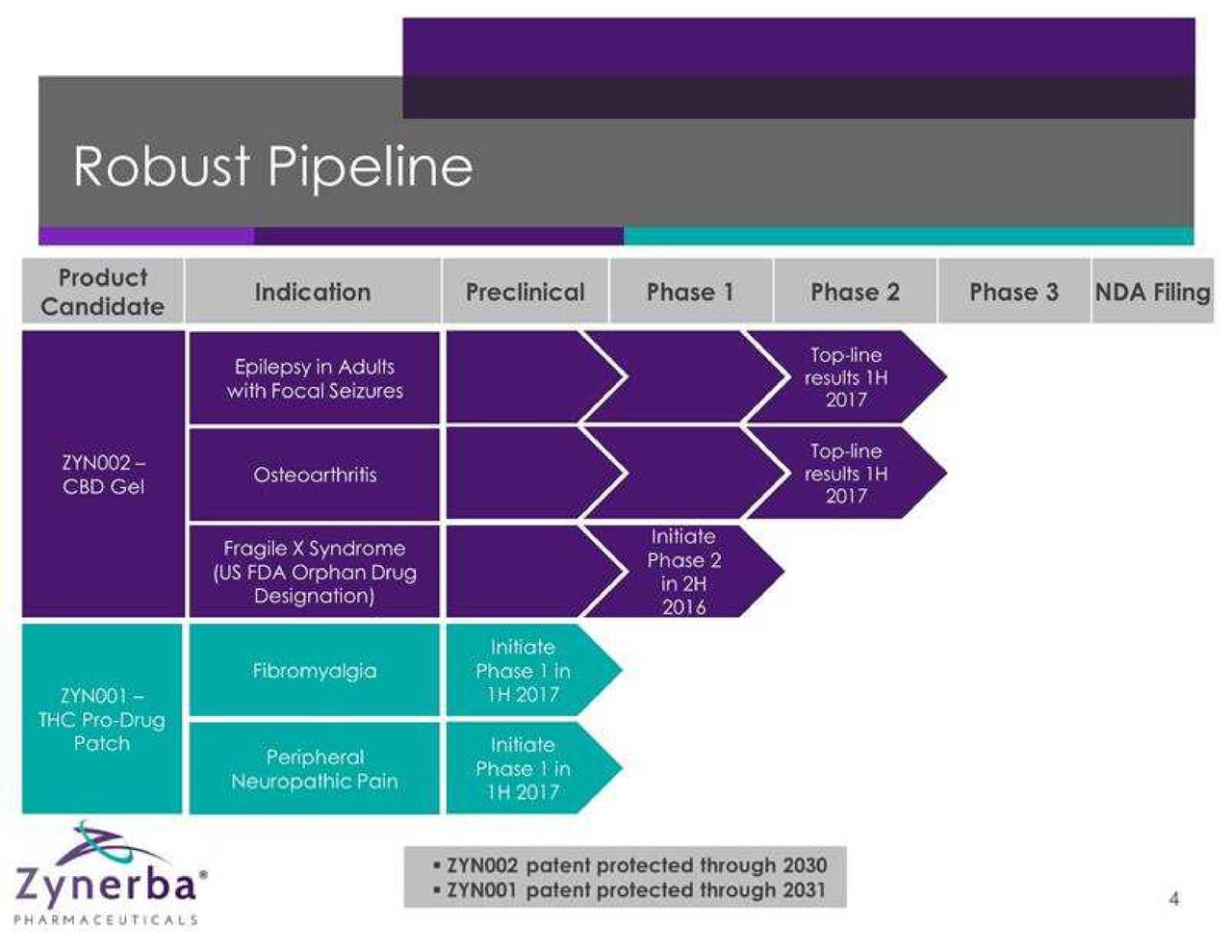
Zynerba Pharmaceuticals, Inc. is a specialty pharmaceutical company focused on developing and commercializing synthetic cannabinoid therapeutics formulated for transdermal delivery. The Company is evaluating approximately two product candidates, ZYN002 and ZYN001, in over five indications. The Company intends to study ZYN002 in patients with refractory epilepsy, osteoarthritis and Fragile X syndrome. The Company's ZYN002 is synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. ZYN002 is being developed as a clear that is designed to provide controlled drug delivery with once- or twice-daily dosing. ZYN001 is a pro-drug of tetrahydrocannabinol (THC) that enables transdermal delivery through a patch. The Company intends to test the ZYN001 patch for application to the arm, back and thigh. The Company intends to study ZYN001 in patients with fibromyalgia and peripheral neuropathic pain.
| Corporate Profile |
Zynerba (NASDAQ: ZYNE) is pioneering the development of patent-protected, next-generation synthetic cannabinoid therapeutics formulated for transdermal delivery. Its two lead product candidates in development include ZYN002 and ZYN001, which are being evaluated in five indications. ZYN002 is the first and only synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. In June 2016, the company initiated the STAR 1 Phase 2 clinical trial in refractory epilepsy patients and in August 2016, initiated the STOP Phase 2 clinical trial in patients with osteoarthritis of the knee. A Phase 2 clinical trial in patients with Fragile X syndrome will be initiated in the second half of 2016.
ZYN001, a prodrug of THC that enables transdermal delivery through the skin and circulatory system via a patch, is in preclinical development. A Phase 1 clinical trial is planned in the first half of 2017.
In August 2015, Zynerba completed an initial public offering, raising net proceeds of $42.1 million. As of June 30, 2016, cash and cash equivalents totaled $32.1 million, which is projected to fund five Phase 2 clinical trials through 2017.
| EPS (TTM) | 9/30/2016 | -2.47 |
|---|---|
| P/E Ratio | 9/30/2016 | -- |
| Market Cap | Micro Cap | 146M |
| Shares Outstanding | 9.95M |
| Float | 7.2M |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
