Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Short interest only 19.9%. Anyone understand what’s going on with this PPS?
NEWS: $ZYNE Zynerba Pharmaceuticals Presents Data Supporting FMR1 Methylation Status as a Correlate to Fragile X Syndrome Severity at the Virtual Joint 16th International Child Neurology Congress (ICNC) & 49th Annual Child Neurology Society (CNS) Meeting
DEVON, Pa., Oct. 15, 2020 (GLOBE NEWSWIRE) -- Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, is presenting a poster describing data from the CONNECT-FX ( C...
Got this from ZYNE - Zynerba Pharmaceuticals Presents Data Supporting FMR1 Methylation Status as a Correlate to Fragile X Syndrome Severity at the Virtual Joint 16th International Child Neurology Congress (ICNC) & 49th Annual Child Neurology Society (CNS) Meeting
JUST IN: $ZYNE New Data Describing Statistically Significant Results from the Phase 2 BRIGHT Trial in Patients with Autism Spectrum Disorder (ASD) Presented at the Virtual Joint 16th International Child Neurology Congress (ICNC) & 49th Annual Child Neurology Society (CN
DEVON, Pa., Oct. 15, 2020 (GLOBE NEWSWIRE) -- Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, is presenting a poster describing data from the Phase 2 BRIGHT (...
In case you are interested ZYNE - New Data Describing Statistically Significant Results from the Phase 2 BRIGHT Trial in Patients with Autism Spectrum Disorder (ASD) Presented at the Virtual Joint 16th International Child Neurology Congress (ICNC) & 49th Annual Child Neurology Society (CN
Amazing results resented at the conference today. ZYNE currently halted at +10%!
$zyne $3.41 ^ 0.04 (1.19%)
Volume: 651,327 @10/06/20 5:25:17 PM EDT
“Zynerba is committed to developing Zygel™ CBD gel in certain rare and near-rare conditions, including 22q, for which there is an urgent need for new, innovative therapeutics,” said Armando Anido, Chairman and Chief Executive Officer of Zynerba. “We are pleased that the FDA shares our sense of urgency regarding the development of effective therapeutics in this important patient population. The receipt of this designation represents another important milestone for us, and we look forward to working closely with the FDA to develop Zygel in pediatric and adolescent patients with 22q as expeditiously as possible.”
About Orphan Drug Designation
Under the Orphan Drug Act (ODA), the FDA may grant orphan drug designation to drugs intended to treat rare diseases or conditions that affect fewer than 200,000 individuals in the U.S. The first NDA applicant to receive FDA approval for a particular active moiety to treat a particular disease with FDA orphan drug designation is entitled to various incentives of the ODA, including tax credits for qualified clinical testing, waiver of new drug application (NDA) / biologics license application (BLA) user fees, and eligibility for a seven-year exclusive marketing period for that drug and use upon marketing approval.
ZANERBA is about to get some serious
market attention on two fronts...
Medical,and
Cannabis
https://seekingalpha.com/article/4375033-making-case-for-zynerba-pharmaceuticals
Strong article and makes a great case for why Zynerba will get approval for FMFX and go to $15. It’s a long article but for those who have the time this is a superb read.
NEWS: $ZYNE Zynerba Pharmaceuticals Receives Orphan Drug Designation for Cannabidiol for the Treatment of 22q11.2 Deletion Syndrome
DEVON, Pa., Sept. 17, 2020 (GLOBE NEWSWIRE) -- Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, today announced that the U.S. Food and Drug Administration has ...
In case you are interested ZYNE - Zynerba Pharmaceuticals Receives Orphan Drug Designation for Cannabidiol for the Treatment of 22q11.2 Deletion Syndrome
Love the market response. It’s really about time.
Zynerba Pharmaceuticals Receives Orphan Drug Designation for Cannabidiol for the Treatment of 22q11.2 Deletion Syndrome
September 17 2020 - 01:55PM
GlobeNewswire Inc.
Alert
Print
Share On Facebook
Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, today announced that the U.S. Food and Drug Administration has granted orphan drug designation for cannabidiol (CBD) for use in treating 22q11.2 deletion syndrome (22q). 22q is a rare midline condition featuring physical abnormalities and debilitating neuropsychiatric and behavioral symptoms including anxiety, withdrawn behavior, and social interaction problems.
“Zynerba is committed to developing Zygel™ CBD gel in certain rare and near-rare conditions, including 22q, for which there is an urgent need for new, innovative therapeutics,” said Armando Anido, Chairman and Chief Executive Officer of Zynerba. “We are pleased that the FDA shares our sense of urgency regarding the development of effective therapeutics in this important patient population. The receipt of this designation represents another important milestone for us, and we look forward to working closely with the FDA to develop Zygel in pediatric and adolescent patients with 22q as expeditiously as possible.”
About Orphan Drug Designation
Under the Orphan Drug Act (ODA), the FDA may grant orphan drug designation to drugs intended to treat rare diseases or conditions that affect fewer than 200,000 individuals in the U.S. The first NDA applicant to receive FDA approval for a particular active moiety to treat a particular disease with FDA orphan drug designation is entitled to various incentives of the ODA, including tax credits for qualified clinical testing, waiver of new drug application (NDA) / biologics license application (BLA) user fees, and eligibility for a seven-year exclusive marketing period for that drug and use upon marketing approval.
About 22q11.2 Deletion Syndrome (22q)
As the second most common chromosomal disorder after Down syndrome, 22q is caused by a small missing piece of the 22nd chromosome. The deletion occurs near the middle of the chromosome at a location designated q11.2. It is considered a mid-line condition, with physical symptoms including characteristic palate abnormalities, heart defects, immune dysfunction, and esophageal / GI issues, as well as debilitating neuropsychiatric and behavioral challenges. Anxiety is among the most common neuropsychiatric symptoms of 22q and researchers have found that for children with 22q, anxiety is linked to poorer adaptive behaviors such as self-care and communication skills that affect daily life. Children with 22q also experience withdrawn behavior, ADHD, cognitive impairment, and autism spectrum disorder that affect communication and social interaction. Later in life, they are at an increased risk of developing mental illnesses such as schizophrenia. It is estimated that 22q occurs in between one in 3,000 and one in 6,000 live births, suggesting that there are approximately 81,000 people living with 22q in the U.S.
About Zynerba Pharmaceuticals, Inc.
Zynerba Pharmaceuticals is the leader in pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders. We ar
I think your right!!
Just a reminder...The next FMFX response from FDA will be virtual,not written.
Zynerba has been notified that the U.S. Food and Drug Administration (FDA) will meet with the Company via teleconference in the fourth quarter of 2020 to discuss the pivotal CONNECT-FX data and the regulatory path forward in patients with FXS and a fully methylated FMR1 gene (FMet). The Company also expects to disclose the outcome of the meeting in the fourth quarter of this year.
As I said ... (one step ahead)
The Company is in the process of finalizing its evaluation of which epileptic syndromes it may pursue with Zygel.
KEY WORD..."FINALIZING"
The meeting with the FDA will be an important milestone for patients and their families who live with the debilitating behavioral impact of Fragile X,” said Armando Anido, Chairman and Chief Executive Officer of Zynerba. “Our ongoing evaluation of the pivotal CONNECT-FX data continues to clarify the impact that Zygel achieved in the most severely impacted children and adolescents with FXS, as well as the excellent tolerability profile. We look forward to discussing the pivotal data and the regulatory path for potential approval in FMet patients with the FDA in the fourth quarter of this year.”
Stockfun,maybe we get a little feed back on DEES at this afternoons presentation .
My guess would be Zanerba team is all ready a step ahead with this.
Agreed dhbuzz. Very much agreed.
Concerning yesterdays updates.
This link will help in understanding what the FDA is asking with the DEES path forward.
.https://www.epilepsy.com/learn/types-epilepsy-syndromes
*************************
With that thinking in mind,I think Zanerba has done just that with FX phase lll trial in as they had positive results with a specific group,FMFX
This gives me more reason to feel NDA from FDA on Zygel for FMFX will happen.
*************************
Congrats on the latest patent.
Maybe something like this in Zanerbas future one day?
https://ih.advfn.com/stock-market/NASDAQ/immunomedics-IMMU/stock-news/83250738/immunomedics-shares-more-than-double-after-gilead
I'm sure it will be recorded Stockfun.
LOL,a Zanerba tease...
Lol!! Enticing me to buy now aren’t you??? I wish I could be in the virtual call. Hoping I can still listen to it later in the day via Zynerba website. Will it be recorded and still be available on the site?
Wouldn't it be a nice Mon. if FDA gave their blessing tomarrow on FMFX.Now that would sure give our CEO something to add to the presentation.
$zyne $3.55 ^ 0.04 (1.14%)
Volume: 257,991 @09/11/20 5:00:31 PM EDT
I think its better than 50\50 simply because of how Zanerba went about the trial.A very smart move on their part IMO,giving them some pie instead of none perhaps with the FMFX group.
Looking forward to the days ahead,and news on several fronts.
GL my friend.
Agree with you on the ASD. Also in the PPS if FDA does not give okay on FMFX. Curious what your thoughts are on the odd of an okay by FDA for FMFX. 50/50? Or better than??
Trial results info on this are also in our near future.
https://en.wikipedia.org/wiki/DiGeorge_syndrome
If FDA does not give NDA ok with FMFX I can see it dropping more.
RED ZONE ...This is a VERY big call by the (reffs)FDA.It creates a starting mark ,and opens some doors not only in the U.S. but also over seas IMO.for Zanerba and the FMFX population.
Going forward IMO FDA will ask for a phase lll on ASD with a larger number of people in the trial.
I dont expect any big news on anything with the 2 presentations this week other than all is still on track.
We are certainly in buying territory now. I may be averaging down this week. Trying to beat any announcements. I don’t think Zynerba can have any further appreciable drop below $3.50.
It is always good to learn new things.
https://www.fraxa.org/a-tribute-to-doris-buffett-philanthropist/
I think it is good that Zanerba has kept the float so low.
The dilution monster has this far been kept in its cage.I think that if FDA gives Zygel for FMFX NDA that Zanerba will move quickly to secure an overseas pharma partner for foreign market sales,I can only assume that Zanerba has this in mind ,but realizing the future potential of Zygel with FMFX and then ASD my guess would be is that Zanerba most likely has this plan in place with someone now...waiting for FDA green light...
Here is a great web site for FX info.
https://www.fraxa.org/fragile-x-news-updates/
LOL,Stockfun,I hear you,I had to vent a tad today.
I feel for the FDA at at this point,and the load that's on them.
Not being able to meet in person with companies is slowing things down for everyone.My gut feeling keeps me here though,and if that instinct pays off...
Solid support around 3.50. Wish I had some cash to buy more!!
Lol!! Your telling me. Every time I think it can go lower it does just that![]() It’s clear that investor sentiment is low on this stock, but it really is a buy. People just not trusting since they surprised us with the FX news last time. I think this will surprise us to the (+ ) on the upcoming FDA meeting news and then a rush to buy will occur.
It’s clear that investor sentiment is low on this stock, but it really is a buy. People just not trusting since they surprised us with the FX news last time. I think this will surprise us to the (+ ) on the upcoming FDA meeting news and then a rush to buy will occur.
The next news from FDA on Zygel and ASD subject will be on a path forward for the next trial.Phase ll results looked excellent.I suspect FDA will ask for a phase lll on ASD.
The next FX news could bring a green light from FDA for NDA on FMFX
To say these next four months are important for Zanerba,would rank in the top five understatements of the year.
Here's to success to all involved!
If we can get approval for ASD and Fully Methylated FX, this stock will be golden again. I’m hoping we get word back on FDA talks re: one or both in Sept/Oct. The RSI on this stock for 5 day, 1month, 3 month and 1 yr all suggest that we are at oversold prices. We are at a level of support @$3.65- $3.70 so I’m seriously thinking about buying because these are steal prices. Just hate to get surprise bad news again. That FX took me for a loop. Really wasn’t expecting that one ![]()
|
Followers
|
91
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2676
|
|
Created
|
08/06/15
|
Type
|
Free
|
| Moderators | |||

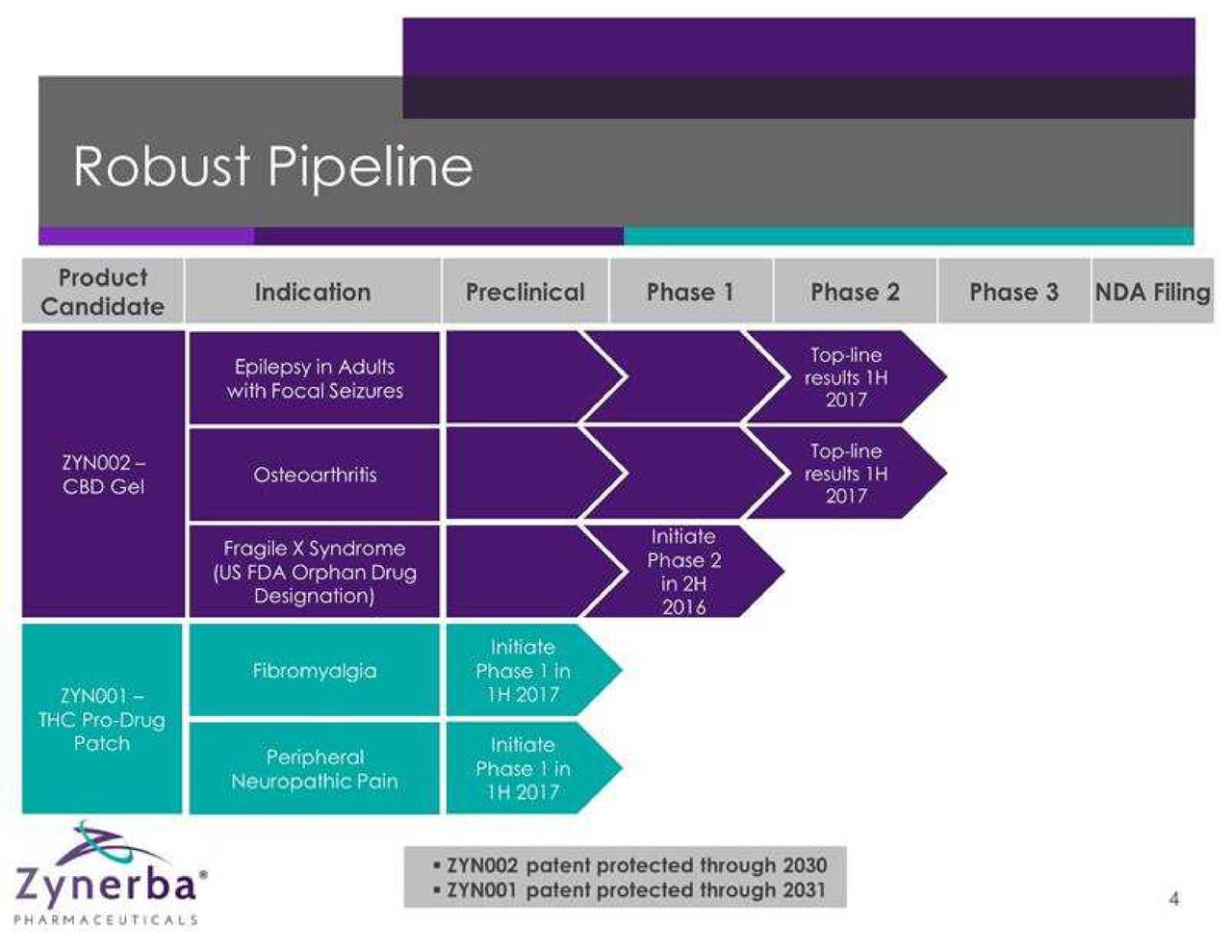
Zynerba Pharmaceuticals, Inc. is a specialty pharmaceutical company focused on developing and commercializing synthetic cannabinoid therapeutics formulated for transdermal delivery. The Company is evaluating approximately two product candidates, ZYN002 and ZYN001, in over five indications. The Company intends to study ZYN002 in patients with refractory epilepsy, osteoarthritis and Fragile X syndrome. The Company's ZYN002 is synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. ZYN002 is being developed as a clear that is designed to provide controlled drug delivery with once- or twice-daily dosing. ZYN001 is a pro-drug of tetrahydrocannabinol (THC) that enables transdermal delivery through a patch. The Company intends to test the ZYN001 patch for application to the arm, back and thigh. The Company intends to study ZYN001 in patients with fibromyalgia and peripheral neuropathic pain.
| Corporate Profile |
Zynerba (NASDAQ: ZYNE) is pioneering the development of patent-protected, next-generation synthetic cannabinoid therapeutics formulated for transdermal delivery. Its two lead product candidates in development include ZYN002 and ZYN001, which are being evaluated in five indications. ZYN002 is the first and only synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. In June 2016, the company initiated the STAR 1 Phase 2 clinical trial in refractory epilepsy patients and in August 2016, initiated the STOP Phase 2 clinical trial in patients with osteoarthritis of the knee. A Phase 2 clinical trial in patients with Fragile X syndrome will be initiated in the second half of 2016.
ZYN001, a prodrug of THC that enables transdermal delivery through the skin and circulatory system via a patch, is in preclinical development. A Phase 1 clinical trial is planned in the first half of 2017.
In August 2015, Zynerba completed an initial public offering, raising net proceeds of $42.1 million. As of June 30, 2016, cash and cash equivalents totaled $32.1 million, which is projected to fund five Phase 2 clinical trials through 2017.
| EPS (TTM) | 9/30/2016 | -2.47 |
|---|---|
| P/E Ratio | 9/30/2016 | -- |
| Market Cap | Micro Cap | 146M |
| Shares Outstanding | 9.95M |
| Float | 7.2M |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
