Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
You willbe green soon
Making progress, I'm averaged in at 9.49 trying to climb back up.
Get birth control
Hello Piper Sandler. My price target far exceeds yours.
Where did you see that?
VXRT$ $20 TARGET PRICE WOOHOOOOOO
Vaxart Inc (VXRT)
6.44 ? 0.22 (3.54%)
Volume: 2,204,318 @05/26/21 12:39:43 PM EDT
Bid Ask Day's Range
6.43 6.44 6.19 - 6.47
VXRT Detailed Quote
* * $VXRT Video Chart 05-21-2021 * *
Link to Video - click here to watch the technical chart video
Very quiet over here although we’ve seen a nice rise
Heart was in the right place, but dangerous move. You can count on BP to make unadulterated product.
* * $VXRT Video Chart 05-10-2021 * *
Link to Video - click here to watch the technical chart video
Hey conix, Good to see you here. What does the Chart say? TIA
* * $VXRT Video Chart 05-07-2021 * *
Link to Video - click here to watch the technical chart video
Shares of COVID vaccine makers plummet after US backs IP waiver
By Will Feuer
May 6, 2021 |
Shares of Moderna and other makers of the COVID-19 vaccines plummeted after the US backed a plan to waive intellectual property protections for the life-saving products.
Moderna stock fell more than 6 percent Wednesday after the announcement and extended those losses a further 2 percent in premarket trading on Thursday. Pfizer shares were down more than 3 percent in that same period and shares of its vaccine partner BioNTech were down more than 7 percent.
US Trade Representative Katherine Tai announced Wednesday that the US will advocate for the “extraordinary measures,” which would potentially enable companies in developing countries to manufacture their own versions of the vaccines, increasing the supply globally.
The proposal is working its way through the World Trade Organization, and there’s no certainty it will become policy.
A healthcare worker administers the Moderna Covid-19 vaccine.
Stocks dropped after the US announced it would waive intellectual protections for the COVID-19 vaccines.
Bloomberg via Getty Images
“The Administration’s aim is to get as many safe and effective vaccines to as many people as fast as possible,” Tai said in a statement. “The Administration believes strongly in intellectual property protections, but in service of ending this pandemic, supports the waiver of those protections for COVID-19 vaccines.”
Proponents of suspending the protections, such as the World Health Organization, say doing so will save lives and end the pandemic more quickly. Weekly new cases of COVID-19 remain at record highs globally as the virus devastates many poor and developing countries.
US Trade Representative Katherine Tai
Trade Representative Katherine Tai said the decision to waive protections would potentially enable companies in developing countries to manufacture their own versions of the vaccines.
But pharmaceutical companies and industry representatives oppose the policy move, saying it won’t immediately boost the global supply of doses, because there are more obstacles than just IP rights. They say vaccine manufacturing is complex and waiving IP protections globally could cause new issues.
The technological know-how behind the vaccines is also seen as proprietary by the industry and sharing any of it could hurt a company’s competitive advantage.
Pfizer CEO Albert Bourla told the Wall Street Journal that waiving IP protections would discourage biotech companies from developing products for the next pandemic.
The surprise move by the US comes as the outbreaks in other countries have become increasingly dire.
India and South Africa were among the first countries to push for the waiver, which was initially opposed by the US, the European Union and other wealthy governments. But pressure from global health advocates has mounted on the US to commit to the policy move.
But it remains to be seen if US backing will be enough for the policy to be implemented globally. The WTO will need to achieve consensus among its 164 member nations for the proposal to be adopted.
Quarterly report they have a lot of cash
* * $VXRT Video Chart 05-06-2021 * *
Link to Video - click here to watch the technical chart video
Good summary on the advantages of an oral vaccine. Thanks for posting
Vaxart Has 3 Advantages Over Pfizer and Moderna
Its coronavirus vaccine candidate may stand out, if upcoming trials are successful.
Adria Cimino
(TMFAdriaCimino)
May 6, 2021 at 6:00AM
Author Bio
Things were looking gloomy for Vaxart (NASDAQ:VXRT) a few months ago. The clinical-stage biotech company reported that its COVID-19 vaccine candidate didn't produce neutralizing antibodies in a phase 1 trial. These antibodies are considered key since their role is to fight infection. As a result, Vaxart's shares tumbled 58% in one trading session.
But all is not lost. This week, the company shared details of its studies and its development plans. Vaxart's investigational vaccine works in a different manner than those of market leaders Pfizer (NYSE:PFE) and Moderna (NASDAQ:MRNA). And it may even offer some advantages. Let's look at three of them.
A young woman takes a pill with a glass of water.
IMAGE SOURCE: GETTY IMAGES.
1. It's a pill
Here's why Vaxart has intrigued investors from the start. The company is developing an oral coronavirus vaccine. It's a tablet to be taken with a glass of water -- as simple as taking a common pain reliever.
It's clear that if such a candidate is successful, it could win over many individuals -- even some who have refused vaccination so far. A Quadrant Strategies poll commissioned by Vaxart confirmed that: About a third of those who refuse vaccination would give it a go if they could choose a pill, the poll showed.
A pill also would be a game-changer for countries and healthcare systems. It's stable at room temperature so it doesn't require refrigerated transport or storage. This makes it easier to deliver and stock the vaccine anywhere in the world.
2. Higher T cell responses
The Pfizer and Moderna vaccines stimulate the body to produce neutralizing antibodies against the coronavirus' spike protein. The spike is used to infect. These antibodies later will protect the vaccinated individual in case of contact with the virus.
Vaxart's candidate doesn't work this way. Instead, it results in strong production of killer T cells. These cells don't completely prevent infection, but they can quickly remove virus from the body. That's because their role is to kill infected cells. And here, Vaxart may have an edge over the market leaders.
Vaxart studied T cell responses in nine volunteers vaccinated with the Pfizer or Moderna vaccines. It found the responses were lower than those produced by its investigational vaccine in the phase 1 trial. Scientists say T cells may play an important role in coronavirus immunity. For example, studies have detected T cells reactive to severe acute respiratory syndrome in former patients 17 years post-infection.
So there is evidence T cells could provide lasting immunity. Of course, Vaxart's later-stage trials must confirm the ability of these cells to quickly clear infection, before illness takes hold.
3. Targeting strains
The Pfizer and Moderna vaccines have demonstrated ability to handle new strains so far. That's because mutations to the spike protein have been limited. But if mutations increase, effectiveness could decrease. Both companies are working on boosters to handle the potential problem.
Vaxart may not have to worry about new strains. Its candidate doesn't target the spike protein only. Instead, it targets both the spike protein and the nucleocapsid, or "N" protein -- that protein is involved in viral replication. Targeting both broadens the vaccine's ability to protect, especially if the spike mutates.
"The strength of T-cell responses against both S and N proteins, which we targeted, leads us to believe that VXA-CoV2-1 offers a promising solution to variants," said Dr. Sean Tucker, Vaxart chief scientific officer.
Now what's the plan?
Vaxart aims to advance its coronavirus vaccine candidate into a phase 2 trial by the middle of this year. The company also will test two candidates targeting the spike protein only and focusing on specific variants. Those will enter phase 1/2 studies in the third quarter. And Vaxart also plans on testing its candidates as boosters in individuals who already have been vaccinated with an authorized vaccine. That should happen in the second half.
If one of Vaxart's candidates is successful, the biotech company clearly will be a latecomer to the coronavirus vaccine market. That means it has to stand out if it hopes to gain market share. The advantages highlighted above show Vaxart may have what it takes to carve out a spot in this competitive market in the future.
Still, risk is high -- at least until we see efficacy data from the phase 2 trial or the booster trials. Vaxart's potential product works differently than more-advanced rivals. So we can't look to others for clues about whether its candidate might work. All of this means Vaxart is definitely a stock to watch, but only the most aggressive investors should consider buying it right now.
Moderma made 1.2 billion on there vaccines today
* * $VXRT Video Chart 05-05-2021 * *
Link to Video - click here to watch the technical chart video
Actually, they do know. Now the whole world knows after today’s World Vaccine Congress. Talk about clueless.
Vaxart recovers day after shares take post-earnings drubbing
May 04, 2021
By: Jonathan M Block, SA News Editor
Vaxart (NASDAQ:VXRT) shares are up more than 9.3% in late afternoon trading, a day after the stock dropped 26% due to disappointing Q1 earnings and a delay in a phase 2 trial of its oral COVID-19 vaccine candidate.
However, shares rose more than 20% after-hours yesterday as the company reported promising phase 1 results on the candidate in COVID-19 variants.
B. Riley analyst Mayank Mamtani today reiterated his buy rating and $13 price target.
He wrote that he is encouraged by the company's build-up of its manufacturing capabilities, as well as having some production tasks completed by partners.
In July with AMA we get pay coding for cms and they will know how much device will get paid by medicare
Actually, they do know. Now the whole world knows after today’s World Vaccine Congress. Talk about clueless.
* * $VXRT Video Chart 05-04-2021 * *
Link to Video - click here to watch the technical chart video
Based on what vaxart believes, this vaccine is going to end the pandemic. The type of immune response they are seeing if similar to cov-1 would last for 17 years. This will destroy Moderna and I couldn’t be happier. VAXART can apply this technology to multiple diseases.
New COVID-19 vaccines could be a pill or a nasal spray: Report
Updated : May 04, 2021 01:23:57 IST
The newer vaccines are from the US government labs and companies including Sanofi , Altimmune and Gritstone Oncology
There are 277 COVID-19 vaccines under development across the globe of which 93 have entered the human testing phase
The next generation of COVID-19 vaccines could be launched as a pill or a nasal spray and be easier to store and transport than the current handful of vaccine shots at work to contain the spread of the pandemic world-wide, the Wall Street Journal reported.
The newer vaccines are from the US government labs and companies including Sanofi , Altimmune and Gritstone Oncology with the potential to offer better immune responses and be more effective against novel viral variants, possibly helping fight the future pandemics, the report mentioned.
The US has currently authorised vaccines from Pfizer Inc and its partner BioNTech SE as well as Moderna and Johnson & Johnson. These vaccines need to be transported and stored at a low temperature and are administered in two dose.
The next generation vaccines could ‘constitute some improvement’ over the limitations mentioned above and make vaccination efforts in rural areas easier, the report said, citing Gregory Poland, professor and vaccine researcher at the Mayo Clinic in Rocheste.
“You will see second-generation, third-generation vaccines,” Professor Poland said, according to the WSJ report.
As of now, there are 277 COVID-19 vaccines under development across the globe, of which 93 have entered the human testing phase, the report said citing the World Health Organization.
Most of the vaccines under testing are injected but there are two oral formulations and seven nasal-spray formulations mentioned the WSJ report.
Many of the next-generation vaccines are in the early-to-middle phase of human testing which means they may not become available until this year end or early 2022.
Sounding a note of caution the report added that there is no guarantee the vaccines will pass testing and some of the companies developing them, such as Altimmune and Gritstone, have never launched vaccine to market.
Vaxart starts enrollment in early-stage norovirus boosting regimen study
May 04, 2021 9:31 AM ETVaxart, Inc. (VXRT)
By: Aakash Babu, SA News Editor
Vaxart (VXRT +16.3%) announces that it has enrolled the first subject in a Phase 1b boosting regimen trial of its norovirus vaccine candidate.
The Phase 1b study is designed to enroll 30 subjects aged 18 to 55 years old, with endpoints being safety and immunogenicity.
Subjects will be randomized into 3 cohorts: Cohort 1 will receive the vaccine candidate on day 1 and week 4; Cohort 2 will receive the vaccine candidate on day 1 and week 8; Cohort 3 will receive the vaccine candidate on day 1 and week 12.
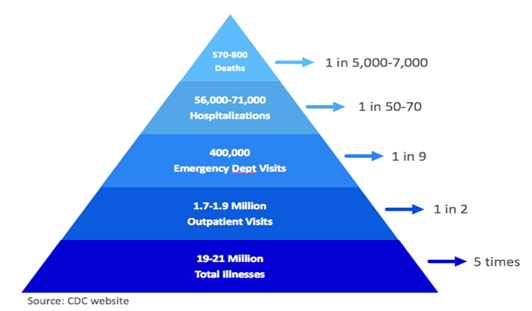
Norovirus is a potentially fatal illness that affects around 20M Americans annually.
The booster regimen trial is the second of four Vaxart norovirus trials that are ongoing or are planned for 2021.
The company is also scheduled to initiate an age escalation trial in subjects over 65 years old and plans to launch a Phase 2 challenge study later this year.
New data obtained from Vaxart's Phase I COVID-19 trial suggests that its oral coronavirus vaccine, VXA-CoV2-1, triggers mucosal immunity, includes both S and N SARS-Cov-2 proteins, and has broad cross-coronavirus activity, the company had said on Monday.
So far, so good...
Short squeeze potential will keep VXRT interesting. Wait until Reddit guys kick in. (If)
Missed the dip in the 7.90 range yesterday. Good luck longs.
like the short interest here.
$PDSB
Yes it is and could be available by the end of the year
I don’t think you read the data correctly. Based on what vaxart believes, this vaccine is going to end the pandemic. The type of immune response they are seeing if similar to cov-1 would last for 17 years. This will destroy Moderna and I couldn’t be happier. VAXART can apply this technology to multiple diseases.
$VXRT favorably mentioned in tonight’s Wall Street Journal article: https://www.wsj.com/articles/next-generation-of-covid-19-vaccines-could-be-pill-or-spray-11620034381?st=j1w115wsk7b6g30&reflink=article_imessage_share
It sounds like VXRT's vaccine might also have some effectiveness in protecting from some cold viruses. I say that because a PR posted at Briefing.com at 5:10 PM ET (see also the one on Yahoo) said "The Vaxart vaccine candidate elicited a T cell response against SARS-Cov-2, as well as showed cross-reactivity against diverse endemic coronaviruses such as 229E, NL63, HKU1, and OC43[.] Vaxart's vaccine candidate triggered specific IgA antibodies in the mucosa. These mucosal IgA responses appeared to be cross-reactive against other coronaviruses such as SARS-CoV-1, MERS, and the endemic common cold viruses 229E, NL63, HKU1, and OC43."
I was surprised by the move up during after hours since the stock trended down during the first hour of the webinar (and stayed down during the rest of the regular session of trading). I was thinking that if great data had been reported during the webinar, then the stock should have moved up considerably during that time.
Guess what we’re not freed from covid the comming years. Although some big ones were first there’s still a lot to gain for everybody.
The Pfizer pill is a therapeutic.
That’s ok. As seen with the COVID vaccine injection with a world population of 7.8 Billion there is room for more than one player. And this will be needed annually for many years to come.
Pfizer is working on a pill too
$VXRT Pill >>> $PFE injection + $MRNA injection IMO.
Wry impressive: “We measured the T-cell responses in 9 volunteers vaccinated with the mRNA vaccines under EUAs and compared them in the same assays at the same timepoints to T-cell responses induced in subjects participating in our clinical trial (N=26 paired samples, or N=7 in the sub-study). Our vaccine appeared to produce CD8+ T-cell responses of a higher magnitude,” said Dr. Sean Tucker, chief scientific officer at Vaxart. “CD8+ T-cells produced by our vaccine can recognize and are capable of destroying cells infected with the virus, including in the mucosa. If you are effective at killing the virus-infected cells in the upper respiratory tract, you may be more likely to recover quicker or have a very mild course of infection,”
Good News!!! Stock headed back UP!!!
https://finance.yahoo.com/news/data-vaxart-oral-covid-19-210600367.html
* * $VXRT Video Chart 05-03-2021 * *
Link to Video - click here to watch the technical chart video
Need more Corrections.Pretty Dismal Q Revs.eom
insiders prolly knew the run was imminent, they could have waited but hey prolly wanted to reduce the loss to shareholders by killing the momentum now rather than when it was at $20 to 30....oh well sold at 10.70
|
Followers
|
186
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
6197
|
|
Created
|
01/12/05
|
Type
|
Free
|
| Moderators | |||

The Vaxart tablet vaccine is composed of three basic elements:
The vector: the Ad5 delivery vehicle that carries the DNA “payload” to the gut
The antigen: the pathogen protein designed to trigger the targeted immune response
The adjuvant: a “booster” molecule that stimulates and adds to the immune response
By using the same vector, but with different antigens, Vaxart has designed a modular, scalable and standardized approach to vaccine development.
With this system, we can support the rapid development of numerous vaccines against established targets, as well as against new and emerging pathogens.
We can achieve important synergies – we can use the same manufacturing processes for all of our vaccines.
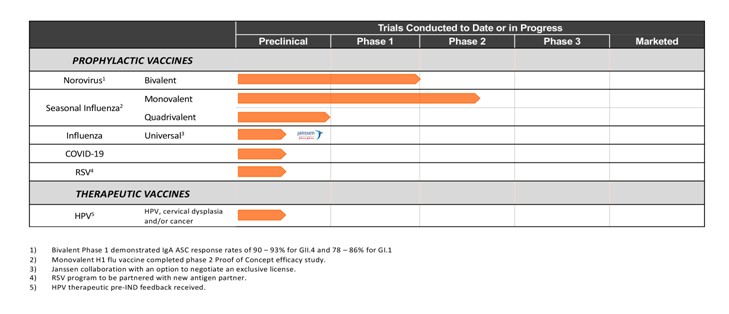
Norovirus
Three Phase 1 studies of Vaxart’s norovirus tablet vaccine indicated it is well tolerated and generated systemic and local immune responses that are both robust and persistent. The most recent Bivalent Phase 1b study demonstrated subjects obtained robust responses to both the mucosal and the systemic immune system.
Influenza
Influenza is a worldwide infectious disease, with symptoms that range from mild to life-threatening and sometimes resulting in death. Serious illness occurs not only in susceptible populations, such as older adults, but also in the general population when unique strains of influenza arise.
Respiratory Syncytial Virus (RSV)
Respiratory syncytial virus (RSV) is increasingly recognized as an important cause of respiratory infection. While RSV infections can occur at any age, the elderly and those immunocompromised and living with underlying cardiopulmonary disease are particularly vulnerable.
Human Papillomavirus (HPV)
HPV is a group of more than 100 related DNA viruses that have the ability to infect the skin or mucous membranes. HPV is the most common sexually transmitted virus in the U.S. and infection is very prevalent following the onset of sexual activity. Vaxart is targeting HPV infection that causes cervical dysplasia and cancer due to HPV 16 and 18.
Vaxart’s COVID-19 Vaccine Selected
for the U.S. Government’s Operation Warp Speed
June 26, 2020 at 8:00 AM EDT
OWS to Test First Oral COVID-19 Vaccine in Non-Human Primates
SOUTH SAN FRANCISCO, Calif., June 26, 2020 (GLOBE NEWSWIRE) -- Vaxart, Inc., a clinical-stage biotechnology company developing oral vaccines that are administered by tablet rather than by injection, today announced that its oral COVID-19 vaccine has been selected to participate in a non-human primate (NHP) challenge study, organized and funded by Operation Warp Speed, a new national program aiming to provide substantial quantities of safe, effective vaccine for Americans by January 2021.
The study is designed to demonstrate the efficacy of Vaxart’s oral COVID-19 vaccine candidate.
“We are very pleased to be one of the few companies selected by Operation Warp Speed, and that ours is the only oral vaccine being evaluated. SARS-CoV-2, the coronavirus that causes COVID-19, is primarily transmitted by viral particles that enter through the mucosa - nose, mouth or eyes - strongly suggesting that mucosal immunity could serve as the first line of defense,” said Andrei Floroiu, Chief Executive Officer of Vaxart Inc. “In addition, our vaccine is a room temperature-stable tablet, an enormous logistical advantage in large vaccination campaigns.”
SOUTH SAN FRANCISCO, Calif., Aug. 10, 2020 (GLOBE NEWSWIRE) -- Vaxart, Inc., a clinical-stage biotechnology company developing oral recombinant vaccines that are administered by tablet rather than by injection, today announced that its COVID-19 Investigational New Drug (IND) application has been filed with the US Food and Drug Administration (FDA).
“We are very excited to reach this important milestone in advancing our oral COVID-19 vaccine candidate towards the clinic,” said Andrei Floroiu, chief executive officer of Vaxart. “We expect our upcoming Phase I study to generate data that will further differentiate our oral vaccine from injectable vaccines by substantiating the importance of activating both systemic and mucosal immunity in protecting against COVID-19. We believe that this mechanistic benefit combined with the significant advantages of oral administration to the patient while eliminating the need for cold chain distribution, could make our COVID-19 vaccine an ideal candidate for successful mass vaccination campaigns globally.”
“Filing the IND is the first major step of many we are taking to advance our oral vaccine in the prevention of COVID-19,” said Sean Tucker, Ph.D., chief scientific officer of Vaxart. “We are excited to be moving this project toward clinical trials, and potentially demonstrating similarly potent mucosal and systemic immune responses like we have seen with our other vaccine candidates using the same oral tablet vaccine platform.”

Emergent BioSolutions Signs Development and Manufacturing Agreement
with Vaxart for their Experimental Oral Vaccine Candidate for Coronavirus Disease
GAITHERSBURG, Md., March 18, 2020 (GLOBE NEWSWIRE) -- Emergent BioSolutions Inc. (NYSE:EBS) announced today that it has entered into an agreement with Vaxart, Inc. (Nasdaq: VXRT), a clinical-stage biotechnology company, whereby Emergent has agreed to utilize its molecule-to-market contract development and manufacturing (CDMO) services to develop and manufacture Vaxart’s experimental oral vaccine candidate for coronavirus disease (COVID-19). Development services will begin immediately, and upon Vaxart’s election, Emergent agrees to produce clinical material expected to enable Vaxart to initiate a Phase 1 clinical study anticipated early in the second half of 2020. Vaxart’s oral recombinant vaccine candidate is based on its proprietary VAAST™ platform.
VXRT Investor Relations
F.D.A. great read KindredBio Partners with Vaxart for COVID-19 Oral Vaccine
October 9, 2020
https://www.fdanews.com/articles/199454-kindredbio-partners-with-vaxart-for-covid-19-oral-vaccine

$VXRT THEN 1. Oral formulations vs Liquid inoculations
[remember $KIN has partnered wit $VXRT]
https://www.fool.com/investing/2020/08/13/3-reasons-doctors-will-prefer-vaxarts-coronavirus/
1. Oral formulations make for easier storage and transport
When most people think of vaccines, they think of a syringe filled with liquid which is administered by a healthcare worker into the muscles or veins of the patient.
In contrast, Vaxart's technology platform produces oral tablets containing the equivalent active components.
This may seem like a minor distinction,
but it's actually a critical factor that works strongly to Vaxart's advantage when it comes to currying favor with doctors and other providers.
Liquid inoculations typically require refrigeration during manufacturing, shipping, and storage at the point of use.
If they aren't consistently kept at the correct temperature, their efficacy may drop, or they may spoil completely.
This means that traditional liquid vaccines imply a substantial logistical burden on healthcare systems,
as well as their suppliers, as a result of the additional refrigeration hardware and energy expenditures required.
Vaxart's tablets are stable at room temperature, however,
so healthcare systems won't need to spend nearly as much to transport or store its coronavirus prophylactic,
and the risk of spoilage is much low
2. Tablets don't require any consumables to administer
When patients get an inoculation in tablet form, there's no need
for the clinic that distributed it to purchase additional disposable syringes, sterilizing swabs, bandages, or even gloves.
As a bonus, after the patient has taken the vaccine, clinics won't
need to pay for additional biohazard trash removal services to dispose of the used materials.
Thanks to the sum of these savings, healthcare systems won't need to spend as much to administer Vaxart's candidate as they might with a traditional liquid inoculation,
enabling them to vaccinate more patients than they could otherwise.
If these savings seem like they might be trivial, consider that an effective coronavirus prophylactic program would need to be deployed
to about 3 billion people globally,
which could easily create shortages and price spikes for the basic healthcare materials or services required for traditional liquid vaccinations.
3. Less risky self-administration
You've probably taken pills without a doctor or nurse standing at
your side to supervise,
but it's unlikely that you've ever prepared and self-injected a vaccination on your own.
With an oral tablet, Vaxart hopes that patients will have the freedom
to vaccinate themselves in the safety of their homes.
This would be particularly advantageous because it would nullify the risk of patients getting infected in crowded clinics while waiting for their turn to be inoculated.
Vaxart's inoculations win via superior logistics, but oral vaccines aren't for everyone
Vaxart's shelf-stable, syringe-free, and easy-to-deploy prophylactic tablets would thus be popular among clinicians around the world thanks to their lower costs,
and savvy investors should recognize that these advantages could be decisive for future earnings.
However, there is one reason why doctors in underdeveloped regions might not favor Vaxart's candidate.
While the scientific consensus is still forming,
it may be the case that malnutrition makes oral vaccination less effective compared with intramuscular vaccination, especially in children.
Therefore, clinics in areas with low food security might opt for a traditional formulation instead of Vaxart's solution.
Even with this potential disadvantage, I think that the vaccine's logistical strengths -- should it be approved
-- will give the company an enduring advantage over the competition.
Time will tell whether Vaxart's coronavirus candidate can live up
to the necessary medical standards. In the meantime, look for the company's phase 1 clinical trial to start later in the summer.
Alex Carchidi has no position in any of the stocks mentioned. The Motley Fool has no position in any of the stocks mentioned.
The Motley Fool has a disclosure policy.
https://www.otcmarkets.com/stock/VXRT/disclosure
https://www.otcmarkets.com/stock/VXRT/security
https://www.otcmarkets.com/stock/VXRT/profile
https://www.otcmarkets.com/stock/VXRT/quote
https://www.otcmarkets.com/stock/VXRT/overview
PER MGMT DAVE -
| IH Geek [Dave] DISCLAIMER |
01-31-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
