Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
woodstock...... 259,000 ALDA bought today- now .035- Maybe you should have emailed him earlier... :)
This is in answer to another email that I sent today concerning a question that I forgot to ask in my earlier email. This was from his gmail account.
Hello #####.
Due to the disclosure rules for public companies, we cannot provide specific information that is not already disclosed. It was particularly difficult for us to see our sales of the hand sanitizer drop from nearly a $1 million in one quarter to virtually zero particularly when everyone who uses the product likes it so much better than competing products. This experience is like a death in the family and it has been very hard on me personally and financially. We are working on arrangements for ALDA that will bring the most benefit to shareholders in these terrible economic times. As I commented in my earlier e-mail, I have been through tough times before but I have never seen such a simultaneous collapse of sales of a product line and the stock market.
As for Seavan, we still have an agreement in place but due to the market, it is obviously on hold.
Regards,
Terry Owen
woodstock...... Thanks for posting that..... I believe Dr Owen, in that these are the most difficult times(especialy) for small cap stocks- Nobody has any money to throw at these companies- If thses is any cash, it is on the sidelines- I haven't posted much on my board for awhile, because there is not much to post- Every chart I look at is a disaster- Indivuduals, businesses, investors, banks, are holding(tightly) onto their cash- everyone sees more bad times ahead, before it all turns around- (hopefully, starting Nov, 2012, here in the US)- One effect of these harsh economic times, is the increase in scams, rip-offs, pump and dumps to the max-
I actually think that ALDA can still have success with their products- but not for another couple years at least, if they can hang on-
I will not post my email to Dr Owen suffice to say that it was rather harsh; but below was his answer, and I can only say that it was good of him to reply.
Your e-mail below was passed on to me. I have responded to both e-mail addresses provided.
We can only report news as it happens, such as the recent announcement about the new NPN for hydrocortisone ointment.
As disclosed in recent quarterly reports, the market for hand sanitizers, which had been our main source of revenue during the 2010 fiscal year, dropped unexpectedly and precipitously during 2011. Other producers have faced the same problem.
The global financial turmoil has affected small cap companies particularly severely, even those in the gold sector. This has led to lower share prices and a shortage of capital.
The company has developed a significant portfolio of intellectual property and we are working very hard on pulling the company through these difficult times and are seeking to regain value for shareholders from this asset.
Having been in public companies since 1987, I can comment that the these are the most difficult times that I have seen despite having survived many previous downturns.
Regards,
Terry Owen
--
Full contact info:
Terrance G. Owen, Ph.D., M.B.A.
President & CEO
ALDA Pharmaceuticals Corp.
(APH:TSX-V, OTCBB:APCSF)
170 - 4320 Viking Way
Richmond, BC V6V 2L4
604-521-8300 Ext. 207 Phone
604-521-8322 Fax
1-866-521-ALDA (2532)
owen.terrance@gmail.com
www.aldacorp.com
Thanks for that. Let's hope that maybe something good will happen sooner than later, because I think hope is all that's left.
The shareholders, I believe own part of the intellectual property..... their press releases always stated that ALDA received the patents..... Unfortunately, all things considered, the market is valuing the shares at .02 right now....
Surf, it wouldn't surprise me in the least if they sell off all the intellectual property leaving shareholders in the cold. Any thoughts...
All I can say is I wish I never heard of this POS. Not having the luxury of knowing the real story we can only spectulate, but if it ever comes out I'd say it's been a liteny of mistakes, combined with incopetence, arrogance, and dishonesty by this bush league management. I've been saying this all along, as you well know, but somehow I held on to a flicker of hope that somehow things would eventually turn around.
I can only therefore feel at this point that as Alda Pharma slowly sinks into the abyss, these %^&* will for once think of the shareholders, who have bourne the brunt of Alda management's inability, or maybe even intent to do the right thing, and pursue a vigoris effort to find a party with the where-with-all to get the company on it's feet someday.
If in fact all the intellectual property does exist that we've been led to believe, then with a good shot of $ and a talented marketing team, this should not be an impossible task.
At this point, you would need to get a psychic's opinion, to read TO's mind on this..... without financing, seems like a "done" deal..... That being said, the products and patents have some value, if anyone is interested.....
Surf, any thoughts? I guess it's all over. What a shame.
Surf, for Alda I agree it's the end, but it ain't well. Aed sorry to say, but it should be no surprise that I do have hard feelings towards the CEO.
Thanks, I wrote to DB.
Well, I can accept that "modification"........ I also have always liked "all's well that ends well"..... I have no ill will towards the ceo.....
No, I'm afraid nothing positive to offer about the corp or its executive. And I've modified the old adage "if you can't say anything nice, don't say anything at all" to "say as little as possible, without misleading."
Grace4........ do you feel positive for the future of ALDA?.... Just a gut feeling, but i feel like you know something......
Grace4..... At least that would bring about some free advertising!.....
The Vancouver Sun's David Baines focuses on white collar crime, especially stock fraud, and seems most likely. Also,Ian Mulgrew focuses on legal affairs more generally; individual's prepetrating fruads of various sorts have been focus of stories (eg, Fri's article on "golddigger" who faked a marriage to a vulnerable, aged millionaire).
I'd love to... anybody in particular you could recommend. It would be great to expose exactly what's been going on the last few years, and market conditions got nothing to do with it, Terry!
You may want to pass it on to an investigative newspaper reporter ... there are a couple who have experience exploring such things
Surf, you don't know how sorry I am to say this, but I guess I was right all along about this crook. We just witnessed another obvious example of blatant insider trading. Too bad even with a case like this it's almost impossible to prove. Even at .035 he keeps trying to squeeze every last cent out of Alda before it's game over. New product... give me a break.
drum............... Agreed
Surf, look at the pps now. Right back to where it started. Same old same old with Alda. So sad, but unfortunately I'm convinced nothing's going to change as long as TO's in charge. 4 years of watching now... it's as clear as the nose on your face. He uses these NRs to try and influence a short lived selling opportunity for himself and his associates I'm sure. By the looks of things I'd say the golden goose is getting very tired. I'll guess that the reason we haven't heard any news for so long is that finances are low. But that's what they get for not developing the fundamentals when times were better. Love to see a real marketer take over. If we are to believe that in fact the intellectual property they have is true, then the door is wide open.
could be...... he was buying some at .03..... so, he could have sold some for a penny profit.... I still have a bunch of this stock, so it was nice to hear something..... I give it a very low probability of success at this point, but, will be interesting to see if they can sell any of the stuff... and, it was mostly buying today, so someone around it was accumulating...
Surf, wonder who the dumper was. I could give you 3 guesses, but the 1st 2 don't count. TO & co. playing the same old games I'm afraid.
somebody pretty quick to dump....oh well, at least some activity
Today's News: VANCOUVER, BRITISH COLUMBIA--(Marketwire - Sept. 8, 2011) - ALDA Pharmaceuticals Corp. (TSX VENTURE:APH - News; OTCQB:APCSF) (the "Company" or "ALDA") announces that Natural Product Number ("NPN") 80026033 has been issued for a 0.5% hydrocortisone (0.5% HC) ointment. The Company is authorized to sell this product in Canada for temporary relief of minor skin irritations, rashes, itching and redness due to eczema, insect bites, poison ivy and other irritating plants; contact dermatitis caused by soaps, detergents, cosmetics, jewellery and other materials that irritate the skin; seborrheic dermatitis and psoriasis.
Typically, skin conditions of these types (which affect over 75% of Canadian adults at one time or another) begin as an inflammatory condition, but are often worsened or prolonged by secondary infection with bacteria or fungi (such as the fungi that causes athlete's foot). The combination of an effective anti-microbial product (such as the T36® formulation) with an anti-inflammatory agent (such as 0.5% hydrocortisone) would address this common medical condition more effectively and make treatment recommendations more straightforward for the health care provider and much easier to carry out for the patient. This innovative combination of an anti-microbial and anti-inflammatory therapy (avoiding the need to apply separate products to treat a single condition) represents an important step forward in the non-prescription therapeutic market. It is also consistent with the current approach favoured by a number of health providers encouraging Canadians to take charge in a positive way of their own care. Obtaining registration of a hydrocortisone ointment is a first step for the Company in bringing this approach to market.
Over 800,000 shares bought in 10 minutes- .03-.035- ALDA lives?
from Stockhouse- No confirmation if any of it is true(except for the "selling")......
"Hello
I am new to this board. But definitely i would say this is an extended shake out. I have heard that over seas investors are involved and that the company is working with a new investment firm. I think they have the pp
lined up but are trying to shakeout scared investors for all they are worth. Also some people are selling to raise cash to buy the pp- this has been going on for a while. Mr Terry and cohorts bought in at 10 cents in January and he and his buddies certainly are not ready/planning to lose all this money. This is just all planned. Once the pp is announced (God knows when) we should move up and more news will come forth. I am in between 5 cent to 10 cents. If I could I would be buying more. Terry is too well connected me believes. "
woodstock....... I don't know..... It's a glimmer of hope anyway.... I aleady have plenty from before, so I'm good!.
Almost tempted to buy some more.
That's good news for ALDA- apparantly still alive!.... First US patent(if granted), for therapeutic use of T36 compound... I think this is significant news....
ALDA Enters National Phase with New Patent Application
Vancouver, BC - ALDA Pharmaceuticals Corp. (APH:TSX-V, APCSF:OTCQB) (the “Company” or “ALDA”) has filed its patent application, “Antiseptic Compositions for the Treatment of Infections”, with the US Patent and Trademark Office (Serial #12/933,358). The application, first submitted to the Canadian Intellectual Property office for review in March, 2008 under the Patent Cooperation Treaty, seeks protection for the composition and preparation of T36® formulations that also contain steroids, anesthetics or analgesics for use on topical infections and, in particular, inflamed infections. Typically, infections with associated inflammation are treated with separate antiseptic and anti-inflammatory preparations. The new T36® formulations combine these properties into a single treatment, making the prescription process easier for the physician and the application easier for the patient. Submissions to the European and Canadian patent offices are also planned. This patent is the third in a series of patents providing protection for the T36® technology that have been issued or are pending.
The first patent, providing protection for the composition and production methods for ALDA's T36® formulation until August 20, 2022, is:
· issued in Australia (#2002322916), the US (#7,338,927) and China (#ZL02829642.7),
· allowed and pending in Canada (Patent Application #2,495,938) and
· pending in the EU (Patent Application #02754054.1-2113).
The second patent, providing protection for the use of T36® as a component of a personal lubricant, in a method of preventing or reducing the transmission of a sexually transmitted diseases including Herpes, Chlamydia and HIV and for use in sanitizers and cleansers in creams, ointments and wipes until August 20, 2022, is
· issued in Australia (#2007237333) and the US (#7,560,422),
· allowed and is pending in Canada (Patent Application #2,495,938) and
· pending in China (Chinese Divisional Patent Application #200710142798.3) and the EU (Patent Application #02754054.1-2113).
According to Dr. Terrance Owen, President & CEO, “Intellectual property protection is one of the key assets of emerging pharmaceutical companies. We are committed to building the Company’s patent portfolio so that we have the protection required in the main international markets that we are targeting with the therapeutic products derived from the T36® technology”.
I don't think T.O. is a highly structured kind of guy.
Ron.
NR- Pretty vague about the relationship.... Who gets the profits, if any?..... Is this a "rent to buy" relationship?... Will ALDA's potential profits go to Seavan for purchase?.... At this point it doesn't really matter much to remaining shareholders, so why not detail the "relationship".....
Finally, it's the best thing they ever did, I hope???
ALDA Enters Into Joint Venture with Seavan
Vancouver, BC - ALDA Pharmaceuticals Corp. (APH:TSX-V) (the “Company” or “ALDA”) announces that a joint venture agreement has been established with Seavan Health & Beauty Partnership (“Seavan”). Under the terms of the agreement, Seavan will manufacture ALDA’s products and ALDA’s personnel will undertake the marketing and sales of Seavan’s products and services in addition to those of ALDA. The acquisition of substantially all of the assets and undertakings of Seavan by ALDA is still in progress.
Dr. Terrance Owen, President & CEO states, “During our discussions with Seavan, it became clear that it would be beneficial for us to work together now because the two companies are very complimentary and can benefit from each other’s strengths. This joint venture will allow us to combine our resources, fine-tune our delivery of both ALDA’s and Seavan’s products and services and grow both businesses as we work on the acquisition. The ultimate goals are for ALDA to manufacture as well as distribute T36® products, acquire expertise in R&D, quality assurance and regulatory matters and re-focus on the therapeutic applications of the T36® technology”.
About Seavan Health & Beauty Partnership (www.seavanlabs.com)
Seavan, located in Vancouver, manufactures pharmaceutical, nutritional and personal care products under licenses from Health Canada and the FDA. Examples of products manufactured at Seavan include glucosamine and magnesium supplements, sun screens, topical delivery systems, pain relievers, body washes, hair care products and ALDA’s T36® products.
The only thing new there(I think), is the Canadian patent allows for therapeutic use of the product?.... the onlt value to this pr, would be garner interest in an upcoming PP?......imo At what point is it impossible to revive a comatose patient?
ALDA news today:
VANCOUVER, BRITISH COLUMBIA--(Marketwire - April 6, 2011) - ALDA Pharmaceuticals Corp. (TSX VENTURE:APH - News; OTCQB:APCSF) ("ALDA") has been notified that the Canadian Intellectual Property Office ("CIPO") has accepted ALDA's patent application that describes the composition, production methods and certain therapeutic uses for ALDA's T36® formulation.
In addition, the Company has two patents issued in the both the US and Australia, one issued in China and one pending in both China and the EU. An additional patent application, seeking protection for the T36® formula in combination with anesthetics, analgesics and anti-inflammatories for the treatment of a wide range of topical infections has been filed in the US and the EU.
Dr. Terrance Owen, President & CEO, states, "Notification that the patent will now be issued in Canada provides further evidence that our intellectual property is very robust. We are gratified to see rapid allowance of the patent applications in a number of jurisdictions. Clearly, ALDA's T36® formulation is unique and is a significant asset to the company."
Surf, this is from SH. Any thoughts? Not very encouraging...
http://www.stockhouse.com/Bullboards/MessageDetail.aspx?s=APH&t=LIST&m=29481237&l=0&pd=2&r=0
China... right, what ever happened to that. Maybe I'm just naively idealistic, but they had the goods, but no ability to market. The Olympic opportunity was totally squandered. I can't help thinking they lacked the intention. TO's constant excuse of market conditions, give me a break. If they are so interested in the therapeutic applications, why were'nt they at the HIV/AIDS conference in South Africa last year. They could have been part of the Candian delagation even. And the big slap in the face with that was there was a vaginal lubricant concept presented by another company... unbelievable!
drum.............well, as that poster from stockhouse said, selling the sanitizer was never the main goal, but a means(by selling enough of it), to finance ongoing development of the therapeutic products..... But, the stuff didn't sell as planned...... Maybe they shuld have gone right for the therapeutics.... who knows... I do think the sanitizer had a rough road- too much competition in that arena... They were supposed to manufacture and sell in China- there was never any follow up info on that- 1st bad sign...
Surf, I really sorry to see that all my ranting against TO, was pretty well true. I have no faith in anything this guy says. He squandered so many opportunities, the Olympics last year being the big l. If he couldn't get it going with that, how can we seriously expect anything to be any different now,
I really can't understand the problem, other than incompetence, or simply a lack of intention. It's so too bad. I can only imagine where this company could be today with a commited and skillful management. So much potential down the drain.
Copnsidering the latest, whatever is going on if anything, I expect it to go nowhere.
When will aph be sub .01, that's the only question I have.
Like the last post says, "strike 3 or home run".............
I assume that poster knows whats coming- it fits with recent history- PP's at ever declining prices.... They don't need deep pockets anymore- just a bunch of shallow ones....
from stockhouse:
Expect a PP at $.06
So, lets break it down:
40,000,000 shares at $.06 = $2,400,000 (Perhaps in 2 tranches)
$1,000,000 for the facility
$400,000 to get it fixed up plus merge plus market
$500,000 to start new product testing
$500,000 payment to Olympic Committe installment
Plus 3,000,000 shares of course.
At least there is no need to worry about previous warrant dillution, this is so far off the money it is ridiculous.
Seavan building will be a must completion for APH as its do or die time to get revenues going, pay off bills, build product line and start the long long overdue testing procedures.
Besides, with a real mnfg building on the books, easier to get line of credit or debt financing instead of constant PPs. NOT TO MENTION LOOKS BETTER FOR TAKEOVER WHICH WE ALL WANT AND NEED!
SHBP is hard to track down but looks like they had a satellite office in seattle back in 2005 with product lines including SPA Luna, UltraCare Labs and Vivian Woodard Cosmetics in the production cue. Ultracare Labs even today appears to have the same address as Seavan - HMMM. They had a President until 2008 but looks like one of the partners took over to date.
So looks like there is product being produced. We should learn more as time goes on.
Not happy about dilution but looks like a necessity now. If this falls through, what are we left with?? TO's record of blaming everyone else aint too impressive as validated per the many rants and raves of fellow posters - me included.
This is definately going to be strike three or a home run scenario playing out. How much longer can we afford PPs just to pay salaries and option bonuses without pushing forward with the new holy grail product lines.
The Sanitizer business was to provide revenues for this, facts are it hasn't worked thus we REALLY need this manufacturing plant and related product revenues coming in to faster track our future product lines.
I hope it's a share swap rather than another pp.
Ron.
from Scott: (I asked him about financing for the manufacturing deal) .............................................
"our financing plans should become more evident in the next week or so - can't tell you much more at his time" sy
from the PR:
The Term Sheet is non-binding and contains a list of key terms and conditions precedent for the establishment of commercial arrangements between the parties. Definitive agreements will be negotiated and prepared after further due diligence, anticipated to be completed by March 11, 2011,
ALDA's financials "ouch".....
http://secfilings.com/searchresultswide.aspx?link=2&filingid=7772017
No updates from ADLA- I still can't imaging how they will get $1mil for the manufacturing facility(although, it sounds like a good idea, as that facility is already manufacturing their own products).... maybe a rich uncle died? anyway, from Agoracom:
It's about time that TO put out a letter to shareholders. Although it sounds like there is still a breath of life in the co. I still think that Alda is still on life support. I am glad that Alda is moving forward and at least working on something. It remains to be seen how purchasing this manufacturing and distribution entity will help Alda move it's products. From what I have seen, their existing products have not been flying off the shelves. I was actually quite surprise to see that Shoppers has restocked their T36 hand sanitizer and spray disinefectant, but now in only travel sizes.
What irks me is how TO asserts that they were too busy during 2010 with the Olympics to focus on other strategic initiatives. That's just poppycock. Unless Terry and Peter were driving personally to the few venues that had dispensers, Alda was sadly absent from a lot of the games. In addition, the games only lasted into March. What happened during the other 3 quarters of 2010?
Let's hope 2011 will be different and look ahead instead of the past blunders.
drum......... cool...... if your selling makes it spike, I'll buy you a drink......
I still can't believe how they blew the Olympic opportunity. I lost a lot on this company as I'm sure I've made quite clear to you guys over the years. Just about to try & cash in what I got left and put it into something else, finally. I sell and watch it spike.
|
Followers
|
26
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
4094
|
|
Created
|
08/07/04
|
Type
|
Free
|
| Moderators | |||
INVESTMENT SUMMARY
- Received a Drug Establishment License ("DEL") and approvals from Health Canada for 30 of its 49 prescription generics products; already has manufacturing approved, labeling and packing secured, and is establishing a sales force 1Q15 salesforc
- Simple business model: Vanc sources drugs that have already obtained FDA equivalent approvals through affiliated companies in China and India, in exchange for manufacturing rights when the drugs are approved by Health Canada
- Potential to scale quickly, comparable to another Canadian company, Paladin Labs, that got bought out for $1.6 billion in 2013 by Endo Health (NASDAQ:ENDP)
- CEO, Arun Nayyar has an extensive track record in the industry - specifically generics - having held executive positions with pharma companies in Latin America, Asia, and Canada and played an instrumental role opening up new markets abroad
- Pre-revenue, albeit with all of the components in place to immediately impact the generics market in Western Canada, and unlock shareholder value through a number of visible, value-unlocking events throughout 2015
Between 2010 and August 2013, brand-name drugs with sales totaling $6 billion a year in Canada lost their patent protection - opening the door to far cheaper generic copies, according to the IMS Brogan market-research company.
Compounding the so-called "patent cliff," a growing list of insurance companies that manage private, workplace drug plans have recently made substituting generics for brands a mandatory policy - a step that most government plans took years ago. The result: more then two thirds of prescriptions in Canada are now filled with generics, while some brand manufacturers lack new drugs in the pipeline to take up the slack.
COMPANY OVERVIEW
Vanc Pharmaceuticals Inc. ("Vancpharma") (OTCQB: NUVPF) is a Canadian company (TSX-V: NPH) focused on providing Canadian health care professionals and consumers with high quality, affordable generics and over-the-counter ("OTC") healthcare products. They are the first Canadian generics company in Western Canada.
GENERICS PORTFOLIO
The company's currently approved in-licensed generics portfolio consists of 30 molecules, comprising of 67 dosage forms across various therapeutic categories: including both chronic (long-term) therapy and acute (short-term) therapy. Management estimates that the aggregate annual Canadian sales of its 30 approved products represents "a $1-billion market opportunity". Furthermore, the company plans to launch "with additional products and we will provide further updates in the coming months," said Arun.
The generics division of the company was only launched last Spring, since then the new management team has reached a number of significant milestones including:
On April 15th, 2014 Vancpharma announced that it had signed Cross Referencing Agreements ("CRA") "for prescription generic products for Canadian markets. These agreements are with three large pharmaceutical companies and cover 48 prescription generic products. The suppliers will handle manufacturing, and Vancpharma will market and sell these new product lines under its own label.
On November 18th, 2014 the company received approval via a Notice of Compliance ("NOC") from Health Canada for 22 generic molecules, comprising 51 dosage forms.
On December 10th, 2014 the company announced that it had been issued a drug establishment licence ("DEL") (licence No. 102220-A) by Health Canada. "The issuance of a drug establishment licence, along with the approval of our partner's GMP manufacturing site, is a key step towards the commercialization of our generic drug portfolio," said Arun Nayyar, CEO. The licence allows Vancpharma to import pharmaceutical products and distribute them within Canada.
This news was particularly important for the company and shareholders because: it positioned Vancpharma to become one of the only 40 companies in Canada that are licensed to manufacture current and future drugs at its GMP facility; allows Vancpharma to import from other manufacturers across the world and faces less barriers to entry; allows for importing both generic and non-generic drugs thus allowing the company to compete with other companies in branded drugs as well as their core generics business; and adds major clout when negotiating for exclusivity rights across Canada. Previously, manufacturers wouldn't commit their exclusively to Vancpharma as they were unsure if it could make good on importing and selling their products.
On December 15th, 2014 the company received an NOC from Health Canada for 7 additional generic molecules, comprising 15 dosage forms.
Lastly, on January 14th, 2015 Vancpharma placed inventory purchase orders for 30 generic molecules and expects to deliver these products 2Q 2015. "We are excited to take this important step towards commercialization and look forward to launching sales in Q2 2015. These 30 molecules represent best-selling generics in the Canadian market and our aim to provide Canadians with quality and cost-effective products is well served by them," said Arun. "Our initial marketing and outreach activities with select pharmacy customers in Western Canada have been positive and we look forward to working with our partners."
Figure 1: Generics Portfolio

Source: Press Release/Company Website
*Note: I'm aware that's only 29, however the 30th hasn't been updated on the website although it's been mentioned as approved (new approvals could come at any time)
Here are the next steps with the generics portfolio:
Figure 2: Generics Through Global Partnership
For those wondering what Vancpharma's margins are, the company has not made that public yet. As you can imagine, they don't want to expose their margins for specific drugs too early. Investors will be able to see them in the financial statements at a later date. However, generics are quite lucrative. Vancpharma will be taking on the risk of inventory, licensing, approvals, sales and marketing, whereas the manufacturer takes and fills an order from Vancpharma as necessary and collects payment. Accordingly, the reward follows the risk and in this case, Vancpharma would pay a fixed price to the manufacturer depending on the size of the manufacturing run. If you look at the financials of another generics company, Biosyent (TSX-V: RX) you will see margins typically ~70%-75%.
The margins generated from strategic cross-reference partnerships, while very enticing, pale in comparison to the financial opportunities presented by exclusive or co-development partnerships which management has indicated they want to pursue in the future.
OTC PRODUCTS
The OTC Products Division is focused on the marketing and sales of novel and proprietary healthcare products and consists of four (4) such products, all of which now have a Natural Product Number.
Figure 3: Vancpharma Pharmaceuticals OTC Products
It can't and won't be the cash cow for Vancpharma like its generics portfolio - the Canadian market for these OTC products is only ~$60-70 million - but it should actually start generating revenue sooner. Manufacturing should commence in January, with the first sales hopefully starting to come sometime during March.
Looking ahead at the future pipeline of OTC products, they include nutraceuticals, vitamin supplements, and skin care products.
MANAGEMENT
The team has extensive experience and expertise that spans across various functions such as research, development, manufacturing, and marketing of generics and OTC health care products in the global pharmaceuticals industry. This understanding of industry best practices and strong insight allows the company to identify emerging trends in medicine and the marketplace.
The secret to being able to license so many drugs within such a short period of time, less than a year after being restructured is CEO, Arun Nayyar.
Arun only joined the company November 25th, 2013, but came with an extensive track record in the industry - specifically generics - having held executive positions with pharmaceutical companies in Latin America, Asia, and Canada. He has been instrumental in opening up new markets abroad, and domestically his accomplishments include Director, Business Development and International Sales for Shoppers Drug Mart ("SDM"), and consulting for Sanis Healthcare, George Weston Ltd. (Loblaws group), and SDM. To have a more extensive look at Arun's resume and job history, you can view his LinkedIn here.
Some information that you won't find on LinkedIn is that he's an owner of a few Shoppers Drug stores in the Vancpharmaouver area, and also has deep-ties, and solid connections in India. This helps to secure licensing of the generics in exchange for manufacturing rights when Health Canada approves the products.
The newest hire, replacing Jamie Lewin as director and CFO and announced December 3rd, 2014 was Aman Parmar. "I look forward to working with the team at Vancpharma Pharmaceuticals and am impressed by how far they have progressed with limited capital," said Mr. Parmar. "Capital efficiency and creating shareholder value will be my primary focus at Vancpharma." Since joining Vancpharma, Aman has purchased 135,000 shares on the open market ranging from 19 to 24 cents - clearly indicative that he believes the company is undervalued at these levels.
Given the fact that the company is ramping up its business, they've already started putting together a sales and marketing team, it wouldn't surprise me if another executive was brought in to help.
You can read more about the entire Vancpharma team by clicking this link (note, this page needs to be updated with Jamie/Aman).
FINANCIALS
Although Vancpharma is a pre-revenue company, I see a multiple number of visible, value-unlocking events through 2015 that can meaningfully impact stock performance.
Figure 4: Summary of Quarterly Results
The company closed an oversubscribed, non-brokered private placement for gross proceeds of $1,141,000 by issuing 7,607,332 units at $0.15/unit on December 11th, 2014. "This round of funding enables us to move our portfolio of generic drugs and OTC products into commercialization," said Arun Nayyar. "Specifically we will be acquiring generic drug inventory and building our sales team to target pharmacy customers."
The company is financed for the time being, but may have to do another round in March depending on how the roll-outs are going, and for general working capital purposes. If so, I'm sure that it would be strategic - I would think at least >$0.20, comprised primarily by sophisticated retailers and brokers, and it would not be a raise of much more than $1 million. Management only wants to raise whatever money they believe is necessary right now because they know as soon as the company starts generating revenue that its valuation has easily be many multiples of where it sits today.
SHARE STRUCTURE
Shares outstanding: 44,374,407
Stock Options: 3,975,000
Warrants: 16,212,252
Fully Diluted: 64,561,659
Major shareholders of the company, and percentage owned include:
It's a tight share structure, the 'effective' float is ~9 million shares - much less than what's been traded the past few months - 42,798,548 shares at an average price of $0.194:
Figure 5: House Positions
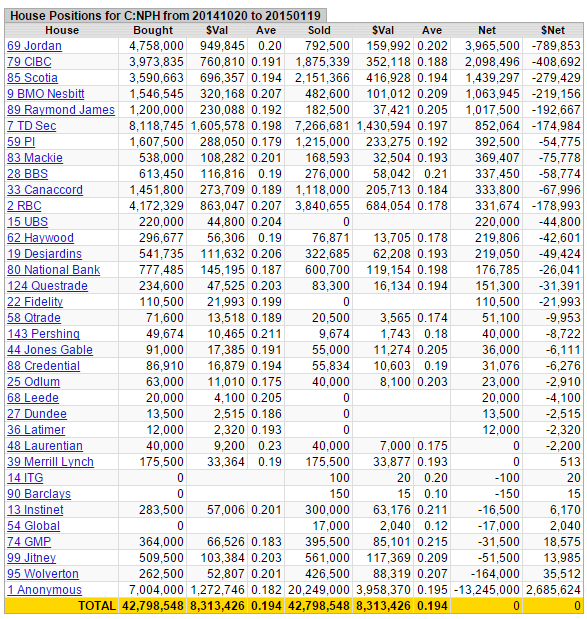
Source: Stockwatch
Figure 6: Management Insider Filings
REVENUE RAMP/VALUATION
It's a little early for me to try and assign a valuation, or forecast revenue and earnings for the company, however I anticipate doing so before year-end. The company hasn't informed investors of the specific margins and obviously we don't know the adoption rate because we don't know how good the sales team will be. Take a look at this article though, "The Top 11 Fastest-Growing Generis Companies", when compared some of the other generics companies, Vancpharma looks very undervalued based solely off of its standing drug portfolio.
The two best examples that come to my mind are: Paladin Labs and BioSyent (OTCPK:BIOYF).
Former pharma sales rep., and Cantech Letter contributor Hogan Mullally wrote that Paladin "became the poster child for Canadian specialty pharma. They built a remarkably successful business by acquiring the Canadian rights to a wide variety of drugs. These drugs were either too small for medium/big pharma, or were developed by a company without a Canadian commercial footprint, whatever the reason, Paladin was able to amass an eclectic and diverse portfolio of prescription and OTC drugs for the Canadian market. Through their "sum of the parts" strategy, Paladin grew to over $200 million in annual sales and in 2013 was acquired by Endo Pharmaceuticals for approximately $1.6 billion.
BioSyent is in between Vancpharma and Paladin, as it has already amassed an impressive portfolio by searching the globe to in-license or acquire innovative pharmaceutical products for the Canadian market. It too focuses on products that are too small for medium/big pharma, and have a competitive angle that can be exploited by a modest sales force. BioSyent's business model is structured to minimize risk, and to produce high growth. For the four years ended December 31, 2013 the company experienced a CAGR of 67% while consistently growing profits:
Figure 7: BioSyent Financials at a Glance
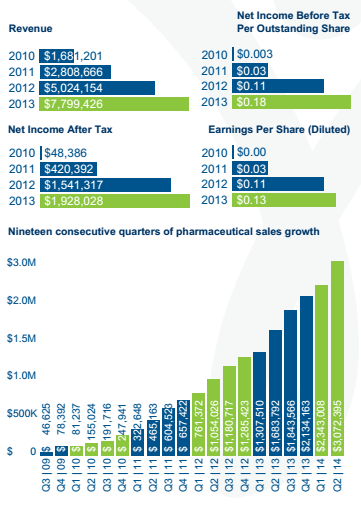
Source: Fact Sheet
There is an exit strategy for the company and shareholders - take-out target for an M&A transaction. It might take getting into a few hundred pharmacies, but the precedent has already been set. The initial adoption rate risk is reduced from the outset because between members of the management team, they own a little more than a dozen pharmacies. Not to mention now that Health Canada has granted the company a Drug Establish License it can pursue additional revenue opportunities and further de-risked the investment.
RISKS
Vancpharma is still considered an early stage company, as such there are a number of risks associated with making an investment at this point in time, including but not limited to:
(1) The company just hired a new CFO, and has a management team has dozens of years of business experience, most dealing with generic pharmaceuticals.
(2) The company plans to manufacture its products at four certified GMP pharmaceuticals factories in Canada, India and China. These U.S. FDA approved plants are capable of manufacturing a wide range of Generic Pharmaceuticals and OTC Health care products at these facilities, under the VANCPHARM label.
(3) Like I mentioned before, management controls a number of pharmacies which will de-risk the rollout process and immediately start generating revenue form the company's generics portfolio.
(4) Arun has built deep, extensive relationships in India and China, and has a lot of ideas to add more products to the portfolio (not to mention the future potential to co-development or development exclusively).
(5) The company sources its products from big products that are already approved by either the U.S. FDA or UPHRA, and also approved by Health Canada. As long as the company is in compliance with Health Canada guidelines then the ANDS application gets expedited, updated and faster tracked. The company hasn't had so much of a hiccup yet, because of the experienced team in place filing all of the paperwork.
(6) The company hasn't had an issue raising money despite the poor market for companies trading on the TSX Venture. The last PP was oversubscribed, and the shares were spread around to strong, strategic hands. Presuming management continues to achieve its milestones, I anticipate lots of eagerness for the next round.
CONCLUSION
Vancpharma set forth some ambitious goals, but has already accomplished so much in such a short amount of time that I really have large aspirations for it (and shareholders).
The company has a tried and tested business model which is simple to understand. Vancpharma enters into Cross-Referencing agreements with affiliated companies whom source products from China and India which are already approved, in exchange for manufacturing rights when the drugs are approved by Health Canada. This is very economical as there is a minimal cross-referencing fee paid to Health Canada, and a very large market opportunity (>$1 billion for current generics portfolio), with margins typically ~70%-75%.
Source: StockCharts.com
Taking a look at recent trading, there has been some healthy consolidation after the initial big run-up, ~33% off its 52-week high. MACD and RSI indicators are now in 'oversold' territory, and the stock is approaching its 200-day moving average. Up until a few months ago this stock traded 'by appointment only'. Since the middle of October, 42,798,548 shares at an average price of $0.194 have traded.
Now that the company has received 30 of its 49 Health Canada approvals (with the rest to be submitted shortly), there is a bit of work that needs to be done in order advance it from being a purely speculative growth biotech with Health Canada approvals, to one that's one being noticed by institutions and an American audience.
In the meantime, investors have two options: (1) buy now, taking the risk knowing that the company may need to finance further, but is extremely close to generating revenue; or (2) sit on the sidelines and wait until the second quarter financials are released, and make a decision then.
The latter is much less risky, however if management successfully executes on its business plan and stays on track with milestones, then I'm sure its share price will reflect it this, and it will be at a significant premium.
Bottom line, I think that there is a very compelling opportunity to invest right now given: (1) the recent pullback in share price; (2) the significance of forthcoming news releases to act as catalysts to unlock shareholder value; (3) management's achievements to date and their track record to deliver results; (4) market opportunity (>$1 billion on current portfolio) and attractive valuations of sector peers; and (5) lack of competition in the Western Canadian generics market and the opportunity to generate imminent revenue.
(For additional liquidity, NUVPF. trades in Canada on the TSX Venture as "NPH". 3-month average volume 637,531 shares/day.)
Please feel free to comment below or send me an inbox message if you have any questions or comments about this article.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
