Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
KK,
Thanks for replying to my post earlier.
Also, what do you mean by the following quote:
This is where the risk is. Cubist may choose not to go after 10% which means these shares will be difficult to trade in Nasdaq.
I don't think they need to wait for the 90% to pay cash to folks who tender within offer period as it will be illegal. However they need 90% to exercise so called top-off option to buy remaining shares either privately or from open market. This is where the risk is. Cubist may choose not to go after 10% which means these shares will be difficult to trade in Nasdaq.
So for folks who tender now and get their cash, might turn out to be lucky if the merger fails as once the offer is consummated, Cubist cannot take the money back.
Good example. Were there any lawsuits ? This is what likely to happen for Trius; however for the merger to be fully effective, the lawsuits filed so far need to be thrown out in the court and I am not sure how long it will take.
So for folks who tender now and get their cash, might turn out to be lucky if the merger fails as once the offer is consummated, Cubist cannot take the money back.
When BMY announced its TO for MEDX @ 16/share a few years back,the original expiration period was extended some days, and finally BMY got the necessary 90% of shares for a so-called short form merger. A few of us did not tender our shares by the new expiration date. A very few days after TO expiration, our shares disappeared from our accounts to be replaced by $16/share.
If you miss or decline the first tender offer, you may get another chance with either an extension or second offer. However if Cubist gets their majority on their first offer and the merger goes thru, you will be automatically bought out and you have no choice. However any second offer is at discretion of Cubist.
Due to the non disclosure agreement,no one really knows what actually transpired behind the scenes.
I'm not crying foul but the only two parties that were privy to the entirity of the contractual agreements from A to B was TSRX and CBST.We can only go on hearsay,as we weren't privy to any conversations or deals presented.
I'm wondering if the other 3 companies signed a NDA, since they're only represented by letter in the company filings. What's the secret after the fact?
I would like to know who they were.
Take Care and God Bless....God Bless Us All.
Truth4Once
Tender Offer
I am wondering what the negative ramifications would be if I chose NOT to accept the tender offer for the buyout of Trius by Cubist...for instance if I chose to participate in a class action suit.
For example, if I didn't accept the tender and the buyout did ultimately go through, am I simply throwing away my investment and not getting any return? Or would I still be paid the $13.50 per share along with the maximum of a $2 CVR, even though I didn't accept the tender offer?
My Schwab acct people told me that I would still get the payout at the terms agreed upon ($13.50 + $2 CVR), even if I declined tender offer and the buyout did go through.
My Scottrade acct people told me that they weren't provided w/that info and I would have to contact Trius IR.
Any thoughts??
Thanks
I am with you on not surrendering yet. For me it will really help if I can even push it out to 2014 for tax deferral. But it looks like Trius did not get competitive bids at higher price around the time Cubist proposed the merger. Could they have waited ? Sure but no guarantee and Cubist is still a better fit than any other big pharma.
Wikipedia:
Institutional investors are organizations which pool large sums of money and invest those sums in securities, real property and other investment assets. They can also include operating companies which decide to invest their profits to some degree in these types of assets.
It says investors not institutions ?
How do you know institutions are not tendering ? The. Sec dock filed clearly shows due deligence was followed. So likely courts will dismiss. Market always knows. If there were any merit, stock would have been hit ( back to 11:30 support) the risk is if majority is tendered, the minority has a small risk of not being included and if they do get included it could be higher or lower. Scare tactics yes but improbable ? No
It appears that the offering will go ahead as planned and if Trius doesn't accumulate a majority of the shares,as explained in their SEC filing,many things can happen.
At this point in time,i'm more inclined to join the class-action suit,as it was explained to me that all the suits will merge into one and they will proceed from there.No commitment either way yet but there is still time.
The fact that not only retail but also corporations are opposed to the deal leads me to believe that they won't get majority approval.
It makes me wonder if that was another reason they purchased the additional 5 million shares for themselves.........Speculation,of course.
To be continued;definitely.
Take Care and God Bless....God Bless Us All.
Truth4Once
Exactly my thoughts. Please let us know what you find out. I will be talking to Corporate Affairs department at TDAmeritrade on Monday as well and will post what they have to say.
I will thoroughly go over this:
Click Here For Contents Of Offer
They may realize that the average shareholder may be intimidated by the wording and simply surrender.
After reviewing said documents and taking notes,I find it best to contact several of the law firms and will do so this coming Monday.
My feeling is that they won't get the majority they seek and if another deal for a higher price is tendered,those who already surrendered may not be able to partake in the increased pps offering.
I'm speculating but will definitely give it a look-see.
Either way,i'm still holding.
Take Care and God Bless....God Bless Us All.
Truth4Once
The offer letter has Q&A style that gives more clarity. The question related 'If I decide not to tender , how will Offer affect my shares ?' is bit confusing, though. Looks they are saying, if offer is consummated but merger does not go thru, shares that were not tendered might not be eligible for public trading due to being reduced to minority or insignificant quantity.
What's interesting is the last few lines for this question, where it says they can buy at any price higher or lower the remaining shares that were not tendered after the offer is consummated and thru the exercise of top-up option.
So definitely there is a risk of not tendering the shares as part of offer.
Don't want to kill the goose that's in the process of laying the golden egg ![]()
I also received the offering notice.
Contacted my broker and asked to speak to the Corporate Actions Department.
They confirmed the notice and next month's deadline date of the 11th.
CBST can raise the offer at any time but most likely not until around or after said deadline.
If they don't receive a total of 90% majority,then it has to go to a shareholder vote.
Too many lawsuits=less than 90% surrender of stock.
I believe Cubist has both a plan B and C(possibly D and E),in the event that the initial proposal doesn't go their way.
The closer they get to the deadline,the more interesting things will get.
Take Care and God Bless....God Bless Us All.
Truth4Once
Got notice from my broker yesterday for tendering in my shares (voluntarily) by September 11th. I am trying to see how much time I can borrow if I choose to appraise under Delaware law purely for tax implications.
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION Filed August 15,2013
WASHINGTON, D.C. 20549
Click For Filing
SCHEDULE TO
TENDER OFFER STATEMENT
UNDER SECTION 14(d)(1) OR SECTION 13(e)(1) OF THE SECURITIES EXCHANGE ACT OF 1934
(Amendment No. 2)
TRIUS THERAPEUTICS, INC.
(Name of Subject Company)
BRGO CORPORATION
(Name of Filing Persons (Offeror))
a wholly owned subsidiary of
CUBIST PHARMACEUTICALS, INC.
(Name of Filing Persons (Parent of Offeror))
COMMON STOCK, $0.0001 PAR VALUE PER SHARE
(Title of Class of Securities)
89685K100
(CUSIP Number of Class of Securities)
Thomas J. DesRosier
Senior Vice President, Chief Legal Officer, General Counsel and Secretary
Cubist Pharmaceuticals, Inc.
65 Hayden Avenue
Lexington, MA 02421
(781) 860-8660
(Name, address and telephone number of person authorized
to receive notices and communications on behalf of filing persons)
with copies to:
Paul M. Kinsella
Christopher D. Comeau
Ropes & Gray LLP
800 Boylston Street
Boston, Massachusetts 02199
Telephone: (617) 951-7000
CALCULATION OF FILING FEE
Transaction Valuation*
Amount of Filing Fee**
$ 820,187,779
$ 111,874
* Estimated solely for purposes of calculating amount of filing fee in accordance with Rule 0-11 under the Securities Exchange Act of 1934 (as amended). The transaction value was calculated by adding (1) the product of (a) $15.50, which is the sum of (i) the closing cash payment of $13.50 per share and (ii) $2.00 per share, which is the maximum amount payable with respect to the contingent value rights and (b) 48,353,557, which is the sum of (i) 48,268,557 shares of common stock, par value $0.0001 per share (the “Shares”), of Trius Therapeutics, Inc. (“Trius”) issued and outstanding on August 1, 2013 and (ii) 85,000 Shares issuable with respect to Trius’ employee stock purchase plan estimated as of August 1, 2013, (2) the product of (a) the difference between (x) $15.50 and (y) an exercise price of $8.84 (the weighted-average exercise price of the outstanding warrants) and (b) 1,565,134 shares issuable pursuant to outstanding warrants as of July 29, 2013, and (3) the product of (a) the difference between (x) $15.50 and (y) an exercise price of $4.56 (the weighted-average exercise price of the outstanding options), and (b) 5,510,407 shares issuable pursuant to outstanding options as of July 29, 2013.
** The amount of the filing fee calculated in accordance with Rule 0-11 of the Securities Exchange Act of 1934, as amended, and Fee Advisory #1 for fiscal year 2013, issued August 31, 2012 by the Securities and Exchange Commission, equals $136.40 per million of the value of the transaction.
x Check the box if any part of the fee is offset as provided by Rule 0-11(a)(2) and identify the filing with which the offsetting fee was previously paid. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing.
Amount Previously Paid:
$111,874
Form or Registration No.:
Schedule TO
Filing Party:
Cubist Pharmaceuticals, Inc.
Date Filed:
August 13, 2013
o Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer.
Check the appropriate boxes below to designate any transactions to which the statement relates:
x third-party tender offer subject to Rule 14d-1.
o issuer tender offer subject to Rule 13e-4.
o going-private transaction subject to Rule 13e-3.
o amendment to Schedule 13D under Rule 13d-2.
Check the following box if the filing is a final amendment reporting the results of the tender offer: o
This Amendment No. 2 (this “Amendment”) amends and supplements the Tender Offer Statement on Schedule TO filed with the Securities and Exchange Commission (the “SEC”) on August 13, 2013, as amended (the “Schedule TO”). The Schedule TO relates to the tender offer by BRGO Corporation, a Delaware corporation (“Purchaser”) and a wholly owned direct subsidiary of Cubist Pharmaceuticals, Inc., a Delaware Corporation (“Parent” or “Cubist”) for all of the outstanding shares of common stock, per value $0.0001 per share (“Shares”), of Trius Therapeutics, Inc., a Delaware corporation (“Trius”), at a price of $13.50 per Share, without interest thereon and less any applicable withholding taxes plus one contingent value right per Share, which represents the contractual right to receive up to $2.00 per Share, upon the terms and conditions set forth in the Offer to Purchase dated August 13, 2013, attached hereto as Exhibit (a)(1)(A) (which, together with the supplement described below and any subsequent amendments and supplements thereto, collectively constitute the “Offer to Purchase”), and in the related Letter of Transmittal (the “Letter of Transmittal”), a copy of which is attached hereto as Exhibit (a)(1)(B), which collectively constitute the “Offer.” This Amendment is being filed on behalf of the Purchaser and Parent.
All information set forth in the Offer to Purchase and the related Letter of Transmittal is incorporated by reference in answer to Items 1 through 9 and Item 11 in the Schedule TO, except those items to which information is specifically provided herein. Capitalized terms used but not otherwise defined herein shall have the meanings ascribed to such terms in the Offer to Purchase.
Amendment to the Offer to Purchase
Item 11. ADDITIONAL INFORMATION
The Offer to Purchase and Item 11 of the Schedule TO, to the extent such Item incorporates by reference the information contained in the Offer to Purchase, are hereby amended and supplemented by adding the following under Section 15 “Legal Matters” of the Offer to Purchase under the heading “Securityholder Litigation”:
“ On August 14, 2013, a lawsuit was filed in the Court of Chancery of the State of Delaware against Trius, and each member of Trius’ board of directors, Cubist and us. The action was brought by Norman Shifrin and Irmgard Kaufhold, who claim to be stockholders of Trius, on their own behalf, and seeks certification as a class action on behalf of all of Trius’ stockholders. The complaint alleges that the defendants breached their fiduciary duties, and/or aided and abetted the breach of fiduciary duties, owed to Trius’ stockholders in connection with the Offer and the Merger.The complaint seeks injunctive relief enjoining the Offer and the Merger, or, in the event the Offer or the Merger has been consummated prior to the court’s entry of final judgment, rescinding the Offer and the Merger. The complaint also seeks an accounting for all damages and an award of costs, including a reasonable allowance for attorneys’ and experts’ fees and expenses.”
2
SIGNATURES
After due inquiry and to the best of their knowledge and belief, each of the undersigned certifies that the information set forth in this statement is true, complete and correct.
Dated: August 15, 2013
BRGO CORPORATION
By:
/s/ THOMAS J. DESROSIER
Name:
Thomas J. DesRosier
Title:
Secretary
CUBIST PHARMACEUTICALS, INC.
By:
/s/ THOMAS J. DESROSIER
Name:
Thomas J. DesRosier
Title:
Senior Vice President, Chief Legal Officer, General Counsel and Secretary
3
EXHIBIT INDEX
Exhibit No.
Description
(a)(1)(A)
Offer to Purchase, dated August 13, 2013.*
(a)(1)(B)
Form of Letter of Transmittal.*
(a)(1)(C)
Form of Notice of Guaranteed Delivery.*
(a)(1)(D)
Form of Letter to Brokers, Dealers, Banks, Trust Companies and Other Nominees.*
(a)(1)(E)
Form of Letter to Clients for Use by Brokers, Dealers, Banks, Trust Companies and Other Nominees.*
(a)(5)(A)
Joint Press Release issued by Cubist Pharmaceuticals, Inc. and Trius Therapeutics, Inc., dated July 30, 2013 (incorporated in the Schedule TO by reference to Exhibit 99.1 to the Current Report on Form 8-K filed by Cubist Pharmaceuticals, Inc. on July 30, 2013).*
(a)(5)(B)
Investor Presentation Slideshow, dated July 30, 2013 (incorporated in the Schedule TO by reference to Exhibit 99.1 to the Current Report on Form 8-K filed by Cubist Pharmaceuticals, Inc. on August 1, 2013).*
(a)(5)(C)
Transcript of July 30, 2013, Investor Presentation (incorporated in the Schedule TO by reference to Exhibit 99.1 of the Schedule TO-C filed by Cubist Pharmaceuticals, Inc. on August 1, 2013).*
(a)(5)(D)
Form of Communication with infection disease experts (incorporated in the Schedule TO by reference to Exhibit 99.1 of the Schedule TO-C filed by Cubist Pharmaceuticals, Inc. on August 2, 2013).*
(a)(5)(E)
Summary Advertisement published in The Wall Street Journal on August 13, 2013.*
(a)(5)(F)
Press Release issued by Cubist Pharmaceuticals, Inc., dated August 13, 2013.*
(d)(1)
Agreement and Plan of Merger, dated as of July 30, 2013, by and between Cubist Pharmaceuticals, Inc., BRGO Corporation and Trius Therapeutics, Inc. (incorporated in the Schedule TO by reference to Exhibit 2.1 to the Current Report on Form 8-K filed by Cubist Pharmaceuticals, Inc. on August 1, 2013).*
(d)(2)
Form of Contingent Value Rights Agreement, by and between Cubist Pharmaceuticals, Inc. and Broadridge Corporate Issuer Solutions, Inc. (incorporated in the Schedule TO by reference to Exhibit 10.1 to the Current Report on Form 8-K filed by Cubist Pharmaceuticals, Inc. on August 1, 2013).*
(d)(3)
Tender and Voting Agreement, dated as of July 30, 2013, by and between Cubist Pharmaceuticals, Inc., BRGO Corporation and the executive officers, directors and certain stockholders affiliated with the directors of Trius Therapeutics, Inc. (incorporated in the Schedule TO by reference to Exhibit 10.2 to the Current Report on Form 8-K filed by Cubist Pharmaceuticals, Inc. on August 1, 2013).*
(d)(4)
Mutual Non-Disclosure Agreement, dated as of July 24, 2012 (the ‘‘Confidentiality Agreement’’), by and between Cubist Pharmaceuticals, Inc. and Trius Therapeutics, Inc.*
(d)(5)
Amendment to Confidentiality Agreement, dated as of July 1, 2013, by and between Cubist Pharmaceuticals, Inc. and Trius Therapeutics, Inc.*
* Previously filed with the Tender Offer Statement on Schedule TO filed with the SEC on August 13, 2013.
4
UNITED STATES SECURITIES AND EXCHANGE COMMISSION[t][/t]
Washington, D.C. 20549
Click For Filing
SCHEDULE 14D-9
(Rule 14d-101)
Solicitation/Recommendation Statement Under Section 14(d)(4)
of the Securities Exchange Act of 1934
(Amendment No. 1)
TRIUS THERAPEUTICS, INC.
(Name of Subject Company)
TRIUS THERAPEUTICS, INC.
(Name of Person Filing Statement)
Common Stock, $0.0001 par value per share
(Title of Class of Securities)
89685K100
(CUSIP Number of Class of Securities)
Jeffrey Stein, Ph.D.
President and Chief Executive Officer
Trius Therapeutics, Inc.
6310 Nancy Ridge Drive, Suite 105
San Diego, California 92121
(858) 452-0370
(Name, Address and Telephone Number of Person Authorized to Receive Notices and
Communications on Behalf of the Person Filing Statement)
With copies to:
Barbara L. Borden, Esq.
Charles J. Bair, Esq.
Cooley LLP
4401 Eastgate Mall
San Diego, CA 92121
(858) 550-6000
¨ Check the box if the filing relates solely to preliminary communications made before the commencement of a tender offer.
This Amendment No. 1 to Schedule 14D-9 amends and supplements the Schedule 14D-9 previously filed by Trius Therapeutics, Inc., a Delaware corporation (“Trius” or the “Company”), with the Securities and Exchange Commission on August 13, 2013, relating to the offer by Cubist Pharmaceuticals, Inc., a Delaware corporation (“Parent” or “Cubist”) and BRGO Corporation, a Delaware corporation and a wholly owned subsidiary of Parent (“Purchaser”), to purchase all the issued and outstanding shares of Trius’ common stock, $0.0001 par value per share (the “Shares”), for a purchase price of (i) $13.50 per Share in cash, without interest, plus (ii) one non-transferrable contingent value right for each Share, which represents the contractual right to receive up to $2.00 per Share upon the achievement of certain milestones as set forth in a Contingent Value Rights Agreement, subject to any required withholding of taxes, if any, upon the terms and conditions set forth in the Offer to Purchase dated August 13, 2013, and in the related Letter of Transmittal, each of which may be amended or supplemented from time to time.
Item 8. Additional Information.
Item 8 of the Schedule 14D-9 is hereby amended and supplemented by deleting the first paragraph under the heading “ Legal Proceedings ” on page 44 of the Schedule 14D-9 and replacing it with the following paragraph:
On August 1, 2013, a putative class-action lawsuit challenging the Merger, captioned Bemis v. Trius Therapeutics, Inc. , Case No. 37-2013-00060593-CU-SL-STL, was filed in the Superior Court of the State of California, County of San Diego (the “Bemis Case”). On August 6, 2013, a putative class-action lawsuit challenging the Merger, captioned Phillip Hurst v. Trius Therapeutics, Inc. , Case No. 37-2013-00061332-CU-SL-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “Hurst Case”). On August 7, 2013, a putative class-action lawsuit challenging the Merger, captioned Collins v. Trius Therapeutics, Inc. , Case No. 37-2013-00061612-CU-SL-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “Collins Case”). On August 7, 2013, a putative class-action lawsuit challenging the Merger, captioned Frazzano v. Trius Therapeutics, Inc. , Case No. 37-2013-00061751-CU-BT-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “Frazzano Case”). On August 9, 2013, a putative class-action lawsuit challenging the Merger, captioned Cast v. Trius Therapeutics, Inc. , Case No. 37-2013-00062038-CU-BT-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “Cast Case”). On August 9, 2013, a putative class-action lawsuit challenging the Merger, captioned Greenwald v. Trius Therapeutics, Inc. , Case No. 37-2013-00062069-CU-SL-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “Greenwald Case”). On August 9, 2013, a putative class-action lawsuit challenging the Merger, captioned Beidler v. Trius Therapeutics, Inc. , Case No. 8794-, was filed in the Court of Chancery for the State of Delaware (the “Beidler Case”). On August 12, 2013, a putative class-action lawsuit challenging the Merger, captioned McPherson v. Trius Therapeutics, Inc. , Case No. 37-2013-00062130-CU-SL-CTL, was filed in the Superior Court of the State of California, County of San Diego (the “McPherson Case”). On August 13, 2013, a putative class-action lawsuit challenging the Merger, captioned Weeks v. Trius Therapeutics, Inc. , Case No. 8799-, was filed in the Court of Chancery for the State of Delaware (the “Weeks Case”). On August 14, 2013, a putative class-action lawsuit challenging the Merger, captioned Shifrin v. Trius Therapeutics, Inc., Case No. 8806-, was filed in the Court of Chancery for the State of Delaware (the “Shifrin Case” and together with the Bemis Case, the Hurst Case, the Collins Case, the Frazzano Case, the Cast Case, the Greenwald Case, the Beidler Case, the McPherson Case and the Weeks Case, collectively, the “Stockholder Litigations”).
2
SIGNATURE
After due inquiry and to the best of my knowledge and belief, I certify that the information set forth in this statement is true, complete and correct.
TRIUS THERAPEUTICS, INC.
By:
/s/ JEFFREY STEIN
Name:
Jeffrey Stein
Title:
President and Chief Executive Officer
Dated: August 14, 2013
3
SHAREHOLDER ALERT: Law Firm Brower Piven Announces That a Class Action Lawsuit Has Been Filed in Connection With the Trius Therapeutics, Inc. Proposed Buyout Alleging That Trius's Directors Have Breached Their Fiduciary Duties to Shareholders 8/14/13 3:50pm
STEVENSON, Md., Aug. 14, 2013 (GLOBE NEWSWIRE) -- The securities litigation firm of Brower Piven, A Professional Corporation, announces that a class action lawsuit has been filed in the Delaware Chancery Court on behalf of all stockholders of Trius Therapeutics, Inc. ("Trius" or the "Company") (Nasdaq:TSRX) common stock.
According to the complaint, the proposed buyout provides for Trius shareholders to receive $13.50 per share in cash for each share of Trius common stock they own in addition to a Contingent Value Right ("CVR") entitling the holder to receive an additional one-time cash payment of up to $2.00 for each share they own if certain commercial sales milestones are achieved. The lawsuit alleges that the members of Trius's Board of Directors have breached their fiduciary duties by agreeing to the proposed buyout for an unfair price and by agreeing to lock up the proposed buyout with deal protection provisions that deter the most likely bidders from making a successful competing offer for the Company.
If you currently own common stock of Trius and would like to learn more about the claims being asserted and your rights, you may email or call Brower Piven, who will, without obligation or cost to you, attempt to answer your questions. You may contact Brower Piven by email at hoffman@browerpiven.com, by calling (410) 415-6616, or at Brower Piven, A Professional Corporation, 1925 Old Valley Road, Stevenson, Maryland 21153. Attorneys at Brower Piven have combined experience litigating securities and other class action cases of over 60 years.
CONTACT: Brower Piven, A Professional Corporation
Stevenson, Maryland
Charles J. Piven, (410) 415-6616
hoffman@browerpiven.com
Take Care and God Bless....God Bless Us All.
Truth4Once
Yep. The near term catalyst of NDA filing was not taken into account for considering the price floor. May be CBST was smart knowing the price will spike in the hot biotech stock market.
Regardless, with the tender offer unchanged from original PR release, I doubt cubist will re-negotiate now. We have to wait for next 20 days to see the outcome.
Yep. The near term catalyst of NDA filing was not taken into account for considering the price floor. May be CBST was smart knowing the price will spike in the hot biotech stock market.
Regardless, with the tender offer unchanged from original PR release, I doubt cubist will re-negotiate now. We have to wait for next 20 days to see the outcome.
Well, the tender offer filing allows appraisal for anyone who may not agree with the terms.
Hence,the plethora of investigations and proposed lawsuit/s on behalf of not only retail but corporate investors.
It could be argued:With the anticipation of the NDA filing,we may have already surpassed the 13.60-13.70's range we are forced by the market to remain at.
Take Care and God Bless....God Bless Us All.
Truth4Once
Well, the tender offer filing allows appraisal for anyone who may not agree with the terms.
CBST announced the commencement of its tender offer for all outstanding shares of the common stock of Trius Therapeutics(TSRX) for $13.50/share in cash, plus one Contingent Value Right, entitling the holder to receive an additional cash payment of up to $2/share they tender if certain sales milestones are achieved.
Our Board of Directors also considered a variety of risks and other potentially negative factors of the Merger Agreement and the Transactions, including the following:
•
the fact that Trius' stockholders will not participate in any potential future benefit from obtaining regulatory approval for and commercializing tedizolid phosphate or from value created through its preclinical programs other than up to $2.00 per Share under the CVR Agreement;
•
the effect of the announcement and pendency of the Merger Agreement and the Offer on Trius' operations, employees, collaboration partners, suppliers and its ability to retain employees;
•
the fact that our executive officers and members of our Board of Directors may be deemed to have interests in the execution and delivery of the Merger Agreement and all of the Transactions, including the Offer and the Merger, that may be different from or in addition to those of our stockholders, generally;
•
the fact that, if the Offer and the other Transactions are not consummated in a timely manner or at all:
•
the trading price of Shares may be adversely affected;
•
Trius will have incurred significant transaction and opportunity costs attempting to consummate the Transactions;
•
Trius may have lost potential collaboration partners and employees after announcement of the Offer;
•
Trius' business may be subject to significant disruption;
•
Trius' directors, officers and other employees will have expended considerable time and effort to consummate the Transactions;
•
the $23.3 million termination fee payable to Purchaser upon the occurrence of certain events, including the potential effect of such termination fee to deter other potential acquirers from publicly making a competing offer for Trius that might be more advantageous to Trius' stockholders, and the impact of the termination fee on Trius' ability to engage in certain other transactions for twelve months from the date of the Merger Agreement is terminated in certain circumstances;
•
the fact that the gain realized by Trius' stockholders as a result of the Offer and the Merger generally will be taxable to the stockholders for U.S. federal income tax purposes; and
•
the restrictions in the Merger Agreement on the conduct of Trius' business prior to the consummation of the Merger, which may delay or prevent Trius from undertaking business or other opportunities that may arise prior to the consummation of the Offer or the Merger.
4:00PM Trius Therapeutics: Cubist Pharmaceuticals commences tender offer for all outstanding shares of Trius Therapeutics for $13.50 per share plus one contingent value right (TSRX) 13.73 +0.03 : CBST announced the commencement of its tender offer for all outstanding shares of the common stock of Trius Therapeutics(TSRX) for $13.50/share in cash, plus one Contingent Value Right, entitling the holder to receive an additional cash payment of up to $2/share they tender if certain sales milestones are achieved. The tender offer is being made by BRGO Corporation, a wholly-owned subsidiary of Cubist, and is dated 7/30/13.
Seems a typo on "plus one Contingent value right". Sounds like total of 3 CVR ?? Am I misreading the briefing.com news ?
PACER SYSTEM RESULTS for CUBIST PHARMACEUTICALS:
All Court Types Party Search
Tue Aug 13 17:57:01 2013
11 records found
Client:
Search: All Court Types Party Search Name Cubist pharmaceuticals All Courts Page: 1
Civil Results
Party Name Court Case NOS Date Filed Date Closed
1 Cubist Pharmaceuticals Inc. (cd) dedce 1:2012-cv-00367 830 03/21/2012
2 Cubist Pharmaceuticals Inc. (cd) dedce 1:2012-cv-00859 830 07/09/2012
3 Cubist Pharmaceuticals Inc. (cd) dedce 1:2012-cv-01142 830 09/17/2012
4 Cubist Pharmaceuticals Inc. (pla) dedce 1:2009-cv-00189 830 03/23/2009 04/11/2011
5 Cubist Pharmaceuticals Inc. (pla) dedce 1:2012-cv-00367 830 03/21/2012
6 Cubist Pharmaceuticals Inc. (pla) dedce 1:2012-cv-00859 830 07/09/2012
7 Cubist Pharmaceuticals Inc. (pla) dedce 1:2012-cv-01142 830 09/17/2012
8 Cubist Pharmaceuticals, Inc. (cd) madce 1:2004-cv-11313 190 06/14/2004 05/20/2005
9 Cubist Pharmaceuticals, Inc. (dft) madce 1:2011-cv-11519 442 08/29/2011 09/06/2012
10 Cubist Pharmaceuticals, Inc. (pla) madce 1:2004-cv-11313 190 06/14/2004 05/20/2005
11 CUBIST PHARMACEUTICALS, INC. (dft) paedce 2:2011-cv-07045 850 11/10/2011 10/09/2012
Take Care and God Bless....God Bless Us All.
Truth4Once
Initial Tender Offer agreement filed today..gives more details around page 14
http://investor.triusrx.com/secfiling.cfm?filingid=1047469-13-8399
SHAREHOLDER ALERT: Law Office of Brodsky & Smith, LLC Announces Investigation of Trius Therapeutics, Inc. - TSRX
Newswire August 06, 2013 12:45 PM
BALA CYNWYD, Pa., Aug. 6, 2013 /PRNewswire/ -- Law office of Brodsky & Smith, LLC announces that it is investigating potential claims against the Board of Directors of Trius Therapeutics, Inc. ("Trius" or the "Company") (Nasdaq- TSRX-News) relating to the proposed acquisition by Cubist Pharmaceuticals, Inc.
Click here to learn more about the investigation http://brodsky-smith.com/624-tsrx-trius-therapeutics-inc.html, or call: 877-534-2590. There is no cost or obligation to you.
Under the terms of the transaction, Trius shareholders will receive only $13.50 in cash and a Contingent Value Right for an additional $2.00 in cash contingent on certain commercial sales milestones being met for each share of Trius stock they own. The investigation concerns possible breaches of fiduciary duty and other violations of state law by the Board of Directors of Trius for not acting in the Company's shareholders' best interests in connection with the sale process. The transaction may undervalue the Company as Trius stock traded at $14.10 per share on July 31, 2013 and an analyst has set an $18.00 per share price target on Trius.
If you own shares of Trius common stock and wish to discuss the legal ramifications of the proposed transaction, or have any questions, you may e-mail or call the law office of Brodsky & Smith, LLC who will, without obligation or cost to you, attempt to answer your questions. You may contact Jason L. Brodsky, Esquire or Evan J. Smith, Esquire at Brodsky & Smith, LLC, Two Bala Plaza, Suite 602, Bala Cynwyd, PA 19004, by e-mail at investorrelations@brodsky-smith.com, by visiting http://brodsky-smith.com/624-tsrx-trius-therapeutics-inc.html, or calling toll free 877-LEGAL-90.
Brodsky & Smith, LLC is a litigation law firm with extensive expertise representing shareholders throughout the nation in securities and case action lawsuits. The attorneys at Brodsky & Smith have been appointed by numerous courts throughout the country to serve as lead counsel in class actions and successfully recovered millions of dollars for our clients and shareholders. Attorney advertising. Prior results do not guarantee a similar outcome.
SOURCE Brodsky & Smith, LLC
Take Care and God Bless....God Bless Us All.
Truth4Once
I continue to be long with 58k shares. At minimum I will hold until the BO date for $2 CVR payout down the road as free ride. Holding my shares for couple of more months is not bad for this reason. In case the BO date gets delayed, all the better for long term capital gains as I have still 6 months to go before 1 year period is over for LTCG holding.
In short, not worried a bit ![]()
For Those Who Are Still Long TSRX:
In this time of uncertainty,I applaud your efforts in standing up for what's fair.
Investigations have begun and a lawsuit to halt is in progress.
Know that i'll be here,right along each and every shareholder for the duration.
Always remember that there's strength in numbers;united we stand.
Take Care and God Bless....God Bless Us All.
Truth4Once
Trius Therapeutics, Inc. (TSRX) Investor Lawsuit to Halt Takeover Announced by Shareholders Foundation
Aug 5, 2013 09:35:02 (ET)
SAN DIEGO, Aug 05, 2013 (GLOBE NEWSWIRE via COMTEX) -- The Shareholders Foundation, Inc. announces that an investor who currently holds Trius Therapeutics, Inc. (TSRX) shares filed a lawsuit to halt the proposed takeover of Trius Therapeutics, Inc. by Cubist Pharmaceuticals, Inc.
Investors who purchased shares of Trius Therapeutics, Inc. (TSRX) prior to July 30, 2013 and currently hold any of those TSRX shares, have certain options and should contact the Shareholders Foundation, Inc. at mail@shareholdersfoundation.com or call +1 (858) 779-1554.
On July 30, 2013, Cubist Pharmaceuticals, Inc. (CBST) and Trius Therapeutics, Inc. (TSRX) announced that they have signed an agreement under which Cubist Pharmaceuticals will acquire all outstanding shares of Trius Therapeutics for $13.50 per share in cash or approximately $707 million on a fully diluted basis. In addition to the upfront cash payment, each Trius Therapeutics stockholder will receive one Contingent Value Right (CVR), entitling the holder to receive an additional cash payment of up to $2.00 for each share they own if certain commercial sales milestones are achieved.
However, the plaintiff alleges that the defendants breached their fiduciary duties by agreeing to sell the company at an unfair price via an unfair process. The plaintiff claims that the offer is too low and undervalues the company. Indeed, at least one analyst has set the high target price for TSRX shares at $20.00 per share.
Furthermore, Trius Therapeutics' performance improved lately. In fact, Trius Therapeutics, Inc. reported that its annual Total Revenue rose from $5.02 million in 2009 to $27.189 million in 2012. Shares of Trius Therapeutics, Inc. grew from $3.184 per share in November 2010 to $12.40 on July 23, 2013.
Those who currently are investors in Trius Therapeutics, Inc. (TSRX) shares and purchased any of those TSRX shares prior to the announcement have certain options and should contact the Shareholders Foundation.
The Shareholders Foundation, Inc. is a professional portfolio legal monitoring and settlement claim filing service, which does research related to shareholder issues and informs investors of securities class actions, settlements, judgments, and other legal related news to the stock/financial market. The Shareholders Foundation, Inc. is not a law firm. The information is provided as a public service. It is not intended as legal advice and should not be relied upon.
CONTACT: Shareholders Foundation, Inc.
Trevor Allen
+1 (858) 779-1554
mail@shareholdersfoundation.com
3111 Camino Del Rio North
Suite 423
San Diego, CA 92108
Take Care and God Bless....God Bless Us All.
Truth4Once
I stuck the funds from here in another partner/acquisition play. That stock has quickly become either overheated or very close to a deal (no way to know for sure).
I am pretty sure I'm not coming back to this...at least not now.
I got out recently. Not because of any reason other than I have more money to make elsewhere.
Just need to put my funds to use.
That choice is yours and only yours.
$TSRX
so....should I get back in and hold, or be happy with the status quo? Wonder how long I have to decide.
No. They want Trius ;)
This does NOT justify that weak offer!
Strategic Initiatives, Executive Transitions, and Strengthened Portfolios to Increase Shareholder Value - Research Report
8/5/13
Do you think it could end up with no deal at all?
We should see further development(s)/commentary from Trius and Cubist regarding the evident negative response we have given them.
It's obvious this deal will not "go through" based upon the overwhelming amount of negative response to this proposed buyout.
I think they'll just have to come back with a better offer.
That's all there is to it.
$TSRX
I agree with you as to the difference of a zero(maybe poster misread calculation).
Now don't get me wrong....whether or not they did is of little concern to me at this point only because of the investigations filed on behalf of certain shareholders(corporations included).
It's unfortunate but every action that was executed will be scrutinized with a fine toothed comb.
This means that every move will have to be answered for,as I am sure that many questions were and/or will be raised.
A loftier bid may cause said investigations to disappear.
Believe me,both Trius and Cubist have already spoken to their respective counsel,advising them of the best way to proceed.
Next week could be an interesting one.
Take Care and God Bless....God Bless Us All.
Truth4Once
Yep..I am aware of this..however 5 million vs 50 million ? Huge difference..
I do agree with you that there is a 'smoke' if not fire yet. Regardless of timing, though, if BOD approves 5 million shares we have to see if they actually got any. It's one thing to raise the OS as a shelf but another thing allocating them as an option or otherwise private placements. I have not see the later part yet.
Until they are actually issued, the total OS is still ~47 million.
UNITED STATES (Facts highlighted in red)
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(D)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of Earliest Event Reported): May 21, 2013
TRIUS THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
Delaware 001-34828 20-1320630
(State of incorporation
or organization)
(Commission
file number)
(I.R.S. Employer
Identification Number)
6310 Nancy Ridge Drive, Suite 105
San Diego, CA
92121
(Address of principal executive offices) (Zip code)
Registrant’s telephone number, including area code: (858) 452-0370
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
¨ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
¨ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
¨ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
¨ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Item 5.02 Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers.
On May 21, 2013, our 2010 Equity Incentive Plan (the “2010 Plan”) was amended to, among other things, increase the aggregate number of shares of common stock authorized for issuance under the 2010 Plan by 5,100,000 shares.
The foregoing description of the amendment to the 2010 Plan does not purport to be complete and is qualified in its entirety by reference to the 2010 Plan, as amended, attached to this Current Report on Form 8-K as Exhibit 99.1.
Item 5.07 Submission of Matters to a Vote of Security Holders.
Our 2013 Annual Meeting of Stockholders was held on May 21, 2013. We had 47,869,291 shares of common stock outstanding and entitled to vote as of April 5, 2013, the record date for the Annual Meeting. At the Annual Meeting, 41,266,312 shares of common stock were present in person or represented by proxy for the five proposals specified below.
At the Annual Meeting, stockholders:
(1) elected Karin Eastham, Seth H. Z. Fischer, Theodore R. Schroeder and Jeffrey Stein, Ph.D. as Class III directors to hold office until the 2016 Annual Meeting of Stockholders;
(2) approved, on an advisory basis, the compensation of our named executive officers, as disclosed in the proxy statement for the Annual Meeting;
(3) indicated, on an advisory basis, that the preferred frequency of stockholder advisory votes on the compensation of our named executive officers is three years;
(4) approved an amendment to the 2010 Plan to, among other things, increase the aggregate number of shares of common stock authorized for issuance under the 2010 Plan by 5,100,000 shares; and
(5) ratified the selection of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2013.
The following sets forth detailed information regarding the final results of the voting for the Annual Meeting:
Proposal 1. Election of Directors
Name
Votes For Votes Withheld Broker Non-Votes
Karin Eastham
29,193,883 225,950 11,846,479
Seth H. Z. Fischer
29,213,769 206,064 11,846,479
Theodore R. Schroeder
28,951,526 468,307 11,846,479
Jeffrey Stein, Ph.D.
29,199,475 220,358 11,846,479
Proposal 2. Advisory Vote on Executive Compensation
Votes For Votes Against Abstentions Broker Non-Votes
27,391,616 1,782,410 245,807 11,846,479
Proposal 3. Advisory Vote on the Frequency of an Advisory Vote on Executive Compensation
1 Year 2 Years 3 Years Abstentions Broker Non-Votes
12,252,893 234,489 16,867,247 65,204 11,846,479
Consistent with the preference of our stockholders indicated by the voting results for Proposal 3, we have decided to include a stockholder advisory vote on the compensation of our named executive officers every three years until the next required vote on the frequency of such advisory votes.
Proposal 4. Approval of Amendment to 2010 Plan
Votes For Votes Against Abstentions Broker Non-Votes
26,578,276 2,564,781 276,776 11,846,479
Proposal 5. Ratification of the selection of Independent Registered Public Accounting Firm
Votes For Votes Against Abstentions Broker Non-Votes
40,806,124 163,968 296,220 —
Item 8.01 Other Events.
On May 21, 2013, our Amended and Restated 2010 Non-Employee Directors’ Stock Option Plan (the “2010 Directors’ Plan”) was amended to, among other things, eliminate the “evergreen” provision providing for automatic annual increases in the number of shares of common stock available for issuance under the 2010 Directors’ Plan after the increase in shares that takes effect on January 1, 2015.
The foregoing description of the amendment to the 2010 Directors’ Plan does not purport to be complete and is qualified in its entirety by reference to the 2010 Directors’ Plan, as amended, attached to this Current Report on Form 8-K as Exhibit 99.2.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
Exhibit
No.
Description
99.1 2010 Equity Incentive Plan, as amended.
99.2 Amended and Restated 2010 Non-Employee Directors’ Stock Option Plan, as amended.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
Trius Therapeutics, Inc.
Dated: May 24, 2013 By:
/s/ John P. Schmid
Name: John P. Schmid
Title: Chief Financial Officer
INDEX TO EXHIBITS
Exhibit
No.
Description
99.1 2010 Equity Incentive Plan, as amended.
99.2 Amended and Restated 2010 Non-Employee Directors’ Stock Option Plan, as amended.
Exhibit 99.1
T RIUS T HERAPEUTICS , I NC .
2010 E QUITY I NCENTIVE P LAN
A DOPTED BY THE B OARD OF D IRECTORS : F EBRUARY 9, 2010
A PPROVED BY THE S TOCKHOLDERS : F EBRUARY 9, 2010
A MENDED BY THE B OARD OF D IRECTORS : M ARCH 7, 2013
A PPROVED BY THE S TOCKHOLDERS : M AY 21, 2013
T ERMINATION D ATE : F EBRUARY 8, 2020
1. G ENERAL .
(a) Successor to and Continuation of Prior Plan. The Plan is intended as the successor to and continuation of the Trius Therapeutics, Inc. Amended and Restated 2006 Equity Incentive Plan (the “ Prior Plan ”). Following the Effective Date, no additional stock awards shall be granted under the Prior Plan. Any shares remaining available for issuance pursuant to the exercise of options or issuance or settlement of stock awards under the Prior Plan as of the Effective Date (the “Prior Plan’s Available Reserve” ) shall become available for issuance pursuant to Stock Awards granted hereunder. From and after the Effective Date, all outstanding stock awards granted under the Prior Plan shall remain subject to the terms of the Prior Plan; provided, however , any shares underlying outstanding stock awards granted under the Prior Plan that expire or terminate for any reason prior to exercise or settlement or are forfeited because of the failure to meet a contingency or condition required to vest such shares (the “ Returning Shares ”) shall become available for issuance pursuant to Awards granted hereunder. All Awards granted on or after the Effective Date of this Plan shall be subject to the terms of this Plan.
(b) Eligible Award Recipients. The persons eligible to receive Awards are Employees, Directors and Consultants.
(c) Available Awards. The Plan provides for the grant of the following Awards: (i) Incentive Stock Options, (ii) Nonstatutory Stock Options, (iii) Stock Appreciation Rights (iv) Restricted Stock Awards, (v) Restricted Stock Unit Awards, (vi) Performance Stock Awards, (vii) Performance Cash Awards, and (viii) Other Stock Awards.
(d) Purpose. The Company, by means of the Plan, seeks to secure and retain the services of the group of persons eligible to receive Awards as set forth in Section 1(b), to provide incentives for such persons to exert maximum efforts for the success of the Company and any Affiliate and to provide a means by which such eligible recipients may be given an opportunity to benefit from increases in value of the Common Stock through the granting of Awards.
2. A DMINISTRATION .
(a) Administration by Board. The Board shall administer the Plan unless and until the Board delegates administration of the Plan to a Committee or Committees, as provided in Section 2(c).
1.
(b) Powers of Board. The Board shall have the power, subject to, and within the limitations of, the express provisions of the Plan:
(i) To determine from time to time (A) which of the persons eligible under the Plan shall be granted Awards; (B) when and how each Award shall be granted; (C) what type or combination of types of Award shall be granted; (D) the provisions of each Award granted (which need not be identical), including the time or times when a person shall be permitted to receive cash or Common Stock pursuant to a Stock Award; (E) the number of shares of Common Stock with respect to which a Stock Award shall be granted to each such person; and (F) the Fair Market Value applicable to a Stock Award.
(ii) To construe and interpret the Plan and Awards granted under it, and to establish, amend and revoke rules and regulations for its administration. The Board, in the exercise of this power, may correct any defect, omission or inconsistency in the Plan or in any Stock Award Agreement or in the written terms of a Performance Cash Award, in a manner and to the extent it shall deem necessary or expedient to make the Plan or Award fully effective.
(iii) To settle all controversies regarding the Plan and Awards granted under it.
(iv) To accelerate the time at which an Award may first be exercised or the time during which an Award or any part thereof will vest in accordance with the Plan, notwithstanding the provisions in the Award stating the time at which it may first be exercised or the time during which it will vest.
(v) To suspend or terminate the Plan at any time. Suspension or termination of the Plan shall not impair rights and obligations under any Award granted while the Plan is in effect except with the written consent of the affected Participant.
(vi) To amend the Plan in any respect the Board deems necessary or advisable, including, without limitation, by adopting amendments relating to Incentive Stock Options and certain nonqualified deferred compensation under Section 409A of the Code and/or to bring the Plan or Awards granted under the Plan into compliance therewith, subject to the limitations, if any, of applicable law. However, except as provided in Section 9(a) relating to Capitalization Adjustments, to the extent required by applicable law or listing requirements, stockholder approval shall be required for any amendment of the Plan that either (A) materially increases the number of shares of Common Stock available for issuance under the Plan, (B) materially expands the class of individuals eligible to receive Awards under the Plan, (C) materially increases the benefits accruing to Participants under the Plan or materially reduces the price at which shares of Common Stock may be issued or purchased under the Plan, (D) materially extends the term of the Plan, or (E) expands the types of Awards available for issuance under the Plan. Except as provided above, rights under any Award granted before amendment of the Plan shall not be impaired by any amendment of the Plan unless (1) the Company requests the consent of the affected Participant, and (2) such Participant consents in writing.
(vii) To submit any amendment to the Plan for stockholder approval, including, but not limited to, amendments to the Plan intended to satisfy the requirements of (A) Section 162(m) of the Code regarding the exclusion of performance-based compensation from the limit on corporate deductibility of compensation paid to Covered Employees, (B) Section 422 of the Code regarding “incentive stock options” or (C) Rule 16b-3.
2.
(viii) To approve forms of Award Agreements for use under the Plan and to amend the terms of any one or more Awards, including, but not limited to, amendments to provide terms more favorable to the Participant than previously provided in the Award Agreement, subject to any specified limits in the Plan that are not subject to Board discretion; provided however , that except with respect to amendments that disqualify or impair the status of an Incentive Stock Option, a Participant’s rights under any Award shall not be impaired by any such amendment unless (A) the Company requests the consent of the affected Participant, and (B) such Participant consents in writing. Notwithstanding the foregoing, subject to the limitations of applicable law, if any, the Board may amend the terms of any one or more Awards without the affected Participant’s consent if necessary to maintain the qualified status of the Award as an Incentive Stock Option or to bring the Award into compliance with Section 409A of the Code.
(ix) Generally, to exercise such powers and to perform such acts as the Board deems necessary or expedient to promote the best interests of the Company and that are not in conflict with the provisions of the Plan or Awards.
(x) To adopt such procedures and sub-plans as are necessary or appropriate to permit participation in the Plan by Employees, Directors or Consultants who are foreign nationals or employed outside the United States.
(c) Delegation to Committee.
(i) General. The Board may delegate some or all of the administration of the Plan to a Committee or Committees. If administration of the Plan is delegated to a Committee, the Committee shall have, in connection with the administration of the Plan, the powers theretofore possessed by the Board that have been delegated to the Committee, including the power to delegate to a subcommittee of the Committee any of the administrative powers the Committee is authorized to exercise (and references in this Plan to the Board shall thereafter be to the Committee or subcommittee), subject, however, to such resolutions, not inconsistent with the provisions of the Plan, as may be adopted from time to time by the Board. The Board may retain the authority to concurrently administer the Plan with the Committee and may, at any time, revest in the Board some or all of the powers previously delegated.
(ii) Section 162(m) and Rule 16b-3 Compliance. The Committee may consist solely of two or more Outside Directors, in accordance with Section 162(m) of the Code, or solely of two or more Non-Employee Directors, in accordance with Rule 16b-3.
(d) Effect of Board’s Decision. All determinations, interpretations and constructions made by the Board in good faith shall not be subject to review by any person and shall be final, binding and conclusive on all persons.
(e) Cancellation and Re-Grant of Stock Awards . Neither the Board nor any Committee shall have the authority to: (i) reduce the exercise price of any outstanding Options or Stock Appreciation Rights under the Plan, or (ii) cancel any outstanding Options or Stock
3.
Appreciation Rights that have an exercise price or strike price greater than the current Fair Market Value of the Common Stock in exchange for cash or other Stock Awards under the Plan, unless the stockholders of the Company have approved such an action within twelve (12) months prior to such an event.
3. S HARES S UBJECT TO THE P LAN .
(a) Share Reserve. Subject to Section 9(a) relating to Capitalization Adjustments, the aggregate number of shares of Common Stock of the Company that may be issued pursuant to Stock Awards after the Effective Date shall not exceed 10,827,745 shares, which number is the sum of (i) the number of shares subject to the Prior Plan’s Available Reserve, plus (ii) an additional 2,400,000 new shares approved by the stockholders on February 9, 2010, plus (iii) an aggregate of 2,309,459 new shares added pursuant to automatic increases that occurred on January 1, 2011, January 1, 2012 and January 1, 2013, plus (iv) an additional 5,100,000 new shares approved by the stockholders on May 21, 2013, plus (v) the Returning Shares, if any, as such shares become available from time to time. For clarity, the limitation in this Section 3(a) is a limitation in the number of shares of Common Stock that may be issued pursuant to the Plan. Accordingly, this Section 3(a) does not limit the granting of Stock Awards except as provided in Section 7(a). Shares may be issued in connection with a merger or acquisition as permitted by, as applicable, NASDAQ Listing Rule 5635(c), NYSE Listed Company Manual Section 303A.08, AMEX Company Guide Section 711 or other applicable stock exchange rules, and such issuance shall not reduce the number of shares available for issuance under the Plan. Furthermore, if a Stock Award or any portion thereof (i) expires or otherwise terminates without all of the shares covered by such Stock Award having been issued or (ii) is settled in cash ( i.e. , the Participant receives cash rather than stock), such expiration, termination or settlement shall not reduce (or otherwise offset) the number of shares Common Stock that may be available for issuance under the Plan.
(b) Subject to subsection 3(c), the number of shares available for issuance under the Plan shall be reduced by: (i) one (1) share for each share of stock issued pursuant to (A) an Option granted under Section 5, or (B) a Stock Appreciation Right granted under Section 5 with respect to which the strike price is at least one hundred percent (100%) of the Fair Market Value of the underlying Common Stock on the date of grant; and (ii) on or after March 15, 2013, 1.5 shares for each share of Common Stock issued pursuant to a Restricted Stock Award, Restricted Stock Unit Award, Performance Stock Award or Other Stock Award.
(c) Reversion of Shares to the Share Reserve.
(i) Shares Available For Subsequent Issuance. If any shares of common stock issued pursuant to a Stock Award are forfeited back to the Company because of the failure to meet a contingency or condition required to vest such shares in the Participant, then the shares that are forfeited shall revert to and again become available for issuance under the Plan. Notwithstanding the provisions of this Section 3(c), on and after March 15, 2013, to the extent (i) there is issued a share of Common Stock pursuant to a Stock Award under the Plan (other than an Option or Stock Appreciation Right), and (ii) there are any Returning Shares granted under the Prior Plan pursuant to an award other than an option or stock appreciation right, and such share of Common Stock becomes available for issuance under the Plan pursuant to Section 1(a), Section 3(a) or this Section 3(c), then the number of shares of Common Stock available for issuance under the Plan shall increase by 1.5 shares for each such share.
4.
(ii) Shares Not Available For Subsequent Issuance. If any shares subject to a Stock Award are not delivered to a Participant because the Stock Award is exercised through a reduction of shares subject to the Stock Award ( i.e ., “net exercised”), the number of shares that are not delivered to the Participant shall not remain available for issuance under the Plan. Also, any shares reacquired by the Company pursuant to Section 8(g) or as consideration for the exercise of an Option shall not again become available for issuance under the Plan.
(d) Incentive Stock Option Limit. Notwithstanding anything to the contrary in this Section 3 and, subject to the provisions of Section 9(a) relating to Capitalization Adjustments, the aggregate maximum number of shares of Common Stock that may be issued pursuant to the exercise of Incentive Stock Options shall be 21,655,490 shares of Common Stock.
(e) Source of Shares. The stock issuable under the Plan shall be shares of authorized but unissued or reacquired Common Stock, including shares repurchased by the Company on the open market or otherwise.
4. E LIGIBILITY .
(a) Eligibility for Specific Stock Awards . Incentive Stock Options may be granted only to employees of the Company or a “parent corporation” or “subsidiary corporation” thereof (as such terms are defined in Sections 424(e) and (f) of the Code). Stock Awards other than Incentive Stock Options may be granted to Employees, Directors and Consultants; provided, however , Nonstatutory Stock Options and SARs may not be granted to Employees, Directors and Consultants who are providing Continuous Service only to any “parent” of the Company, as such term is defined in Rule 405, unless the stock underlying such Stock Awards is treated as “service recipient stock” under Section 409A of the Code because the Stock Awards are granted pursuant to a corporate transaction (such as a spin off transaction) or unless such Stock Awards comply with the distribution requirements of Section 409A of the Code.
(b) Ten Percent Stockholders. A Ten Percent Stockholder shall not be granted an Incentive Stock Option unless the exercise price of such Option is at least one hundred ten percent (110%) of the Fair Market Value on the date of grant and the Option is not exercisable after the expiration of five (5) years from the date of grant.
(c) Section 162(m) Limitation on Annual Grants . Subject to the provisions of Section 9(a) relating to Capitalization Adjustments, at such time as the Company may be subject to the applicable provisions of Section 162(m) of the Code, no Participant shall be eligible to be granted during any calendar year Options, Stock Appreciation Rights and Other Stock Awards whose value is determined by reference to an increase over an exercise or strike price of at least one hundred percent (100%) of the Fair Market Value on the date the Stock Award is granted covering more than 800,000 shares of Common Stock.
5.
5. P ROVISIONS R ELATING TO O PTIONS AND S TOCK A PPRECIATION R IGHTS .
Each Option or SAR shall be in such form and shall contain such terms and conditions as the Board shall deem appropriate. All Options shall be separately designated Incentive Stock Options or Nonstatutory Stock Options at the time of grant, and, if certificates are issued, a separate certificate or certificates shall be issued for shares of Common Stock purchased on exercise of each type of Option. If an Option is not specifically designated as an Incentive Stock Option, then the Option shall be a Nonstatutory Stock Option. The provisions of separate Options or SARs need not be identical; provided, however , that each Option Agreement or Stock Appreciation Right Agreement shall conform to (through incorporation of provisions hereof by reference in the applicable Award Agreement or otherwise) the substance of each of the following provisions:
(a) Term. Subject to the provisions of Section 4(b) regarding Ten Percent Stockholders, no Option or SAR shall be exercisable after the expiration of ten (10) years from the date of its grant or such shorter period specified in the Award Agreement.
(b) Exercise Price. Subject to the provisions of Section 4(b) regarding Ten Percent Stockholders, the exercise price (or strike price) of each Option or SAR shall be not less than one hundred percent (100%) of the Fair Market Value of the Common Stock subject to the Option or SAR on the date the Option or SAR is granted. Notwithstanding the foregoing, an Option or SAR may be granted with an exercise price (or strike price) lower than one hundred percent (100%) of the Fair Market Value of the Common Stock subject to the Option or SAR if such Option or SAR is granted pursuant to an assumption of or substitution for another option or stock appreciation right pursuant to a Corporate Transaction and in a manner consistent with the provisions of Sections 409A and, if applicable, 424(a) of the Code. Each SAR will be denominated in shares of Common Stock equivalents.
(c) Purchase Price for Options. The purchase price of Common Stock acquired pursuant to the exercise of an Option shall be paid, to the extent permitted by applicable law and as determined by the Board in its sole discretion, by any combination of the methods of payment set forth below. The Board shall have the authority to grant Options that do not permit all of the following methods of payment (or otherwise restrict the ability to use certain methods) and to grant Options that require the consent of the Company to utilize a particular method of payment. The permitted methods of payment are as follows:
(i) by cash, check, bank draft or money order payable to the Company;
(ii) pursuant to a program developed under Regulation T as promulgated by the Federal Reserve Board that, prior to the issuance of the stock subject to the Option, results in either the receipt of cash (or check) by the Company or the receipt of irrevocable instructions to pay the aggregate exercise price to the Company from the sales proceeds;
(iii) by delivery to the Company (either by actual delivery or attestation) of shares of Common Stock;
(iv) if the option is a Nonstatutory Stock Option, by a “net exercise” arrangement pursuant to which the Company will reduce the number of shares of Common Stock
6.
issuable upon exercise by the largest whole number of shares with a Fair Market Value that does not exceed the aggregate exercise price; provided, however , that the Company shall accept a cash or other payment from the Participant to the extent of any remaining balance of the aggregate exercise price not satisfied by such reduction in the number of whole shares to be issued; provided, further, that shares of Common Stock will no longer be subject to an Option and will not be exercisable thereafter to the extent that (A) shares issuable upon exercise are reduced to pay the exercise price pursuant to the “net exercise,” (B) shares are delivered to the Participant as a result of such exercise, and (C) shares are withheld to satisfy tax withholding obligations; or
(v) in any other form of legal consideration that may be acceptable to the Board.
(d) Exercise and Payment of a SAR. To exercise any outstanding Stock Appreciation Right, the Participant must provide written notice of exercise to the Company in compliance with the provisions of the Stock Appreciation Right Agreement evidencing such Stock Appreciation Right. The appreciation distribution payable on the exercise of a Stock Appreciation Right will be not greater than an amount equal to the excess of (A) the aggregate Fair Market Value (on the date of the exercise of the Stock Appreciation Right) of a number of shares of Common Stock equal to the number of Common Stock equivalents in which the Participant is vested under such Stock Appreciation Right, and with respect to which the Participant is exercising the Stock Appreciation Right on such date, over (B) the strike price that will be determined by the Board at the time of grant of the Stock Appreciation Right. The appreciation distribution in respect to a Stock Appreciation Right may be paid in Common Stock, in cash, in any combination of the two or in any other form of consideration, as determined by the Board and contained in the Stock Appreciation Right Agreement evidencing such Stock Appreciation Right.
(e) Transferability of Options and SARs. The Board may, in its sole discretion, impose such limitations on the transferability of Options and SARs as the Board shall determine. In the absence of such a determination by the Board to the contrary, the following restrictions on the transferability of Options and SARs shall apply:
(i) Restrictions on Transfer. An Option or SAR shall not be transferable except by will or by the laws of descent and distribution and shall be exercisable during the lifetime of the Participant only by the Participant; provided, however , that the Board may, in its sole discretion, permit transfer of the Option or SAR in a manner that is not prohibited by applicable tax and securities laws upon the Participant’s request. Except as explicitly provided herein, neither an Option nor a SAR may be transferred for consideration.
(ii) Domestic Relations Orders. Notwithstanding the foregoing, an Option or SAR may be transferred pursuant to a domestic relations order; provided, however , that if an Option is an Incentive Stock Option, such Option may be deemed to be a Nonstatutory Stock Option as a result of such transfer.
(iii) Beneficiary Designation. Notwithstanding the foregoing, the Participant may, by delivering written notice to the Company, in a form provided by or otherwise satisfactory to the Company and any broker designated by the Company to effect Option
7.
exercises, designate a third party who, in the event of the death of the Participant, shall thereafter be entitled to exercise the Option or SAR and receive the Common Stock or other consideration resulting from such exercise. In the absence of such a designation, the executor or administrator of the Participant’s estate shall be entitled to exercise the Option or SAR and receive the Common Stock or other consideration resulting from such exercise.
(f) Vesting Generally. The total number of shares of Common Stock subject to an Option or SAR may vest and therefore become exercisable in periodic installments that may or may not be equal. The Option or SAR may be subject to such other terms and conditions on the time or times when it may or may not be exercised (which may be based on the satisfaction of Performance Goals or other criteria) as the Board may deem appropriate. The vesting provisions of individual Options or SARs may vary. The provisions of this Section 5(f) are subject to any Option or SAR provisions governing the minimum number of shares of Common Stock as to which an Option or SAR may be exercised.
(g) Termination of Continuous Service. Except as otherwise provided in the applicable Award Agreement or other agreement between the Participant and the Company, if a Participant’s Continuous Service terminates (other than for Cause or upon the Participant’s death or Disability), the Participant may exercise his or her Option or SAR (to the extent that the Participant was entitled to exercise such Award as of the date of termination of Continuous Service) but only within such period of time ending on the earlier of (i) the date three (3) months following the termination of the Participant’s Continuous Service (or such longer or shorter period specified in the applicable Award Agreement), or (ii) the expiration of the term of the Option or SAR as set forth in the Award Agreement. If, after termination of Continuous Service, the Participant does not exercise his or her Option or SAR within the time specified herein or in the Award Agreement (as applicable), the Option or SAR shall terminate.
(h) Extension of Termination Date. If the exercise of an Option or SAR following the termination of the Participant’s Continuous Service (other than for Cause or upon the Participant’s death or Disability) would be prohibited at any time solely because the issuance of shares of Common Stock would violate the registration requirements under the Securities Act, then the Option or SAR shall terminate on the earlier of (i) the expiration of a total period of three (3) months (that need not be consecutive) after the termination of the Participant’s Continuous Service during which the exercise of the Option or SAR would not be in violation of such registration requirements, or (ii) the expiration of the term of the Option or SAR as set forth in the applicable Award Agreement. In addition, unless otherwise provided in a Participant’s Award Agreement, if the sale of any Common Stock received upon exercise of an Option or SAR following the termination of the Participant’s Continuous Service (other than for Cause) would violate the Company’s insider trading policy, then the Option or SAR shall terminate on the earlier of (i) the expiration of a period equal to the applicable post-termination exercise period after the termination of the Participant’s Continuous Service during which the exercise of the Option or SAR would not be in violation of the Company’s insider trading policy, or (ii) the expiration of the term of the Option or SAR as set forth in the applicable Award Agreement.
(i) Disability of Participant. Except as otherwise provided in the applicable Award Agreement or other agreement between the Participant and the Company, if a Participant’s Continuous Service terminates as a result of the Participant’s Disability, the Participant may
8.
exercise his or her Option or SAR (to the extent that the Participant was entitled to exercise such Option or SAR as of the date of termination of Continuous Service), but only within such period of time ending on the earlier of (i) the date twelve (12) months following such termination of Continuous Service (or such longer or shorter period specified in the Award Agreement), or (ii) the expiration of the term of the Option or SAR as set forth in the Award Agreement. If, after termination of Continuous Service, the Participant does not exercise his or her Option or SAR within the time specified herein or in the Award Agreement (as applicable), the Option or SAR (as applicable) shall terminate.
(j) Death of Participant. Except as otherwise provided in the applicable Award Agreement or other agreement between the Participant and the Company, if (i) a Participant’s Continuous Service terminates as a result of the Participant’s death, or (ii) the Participant dies within the period (if any) specified in the Award Agreement after the termination of the Participant’s Continuous Service for a reason other than death, then the Option or SAR may be exercised (to the extent the Participant was entitled to exercise such Option or SAR as of the date of death) by the Participant’s estate, by a person who acquired the right to exercise the Option or SAR by bequest or inheritance or by a person designated to exercise the Option or SAR upon the Participant’s death, but only within the period ending on the earlier of (i) the date eighteen (18) months following the date of death (or such longer or shorter period specified in the Award Agreement), or (ii) the expiration of the term of such Option or SAR as set forth in the Award Agreement. If, after the Participant’s death, the Option or SAR is not exercised within the time specified herein or in the Award Agreement (as applicable), the Option or SAR shall terminate.
(k) Termination for Cause. Except as explicitly provided otherwise in a Participant’s Award Agreement, if a Participant’s Continuous Service is terminated for Cause, the Option or SAR shall terminate upon the date of such Participant’s termination of Continuous Service, and the Participant shall be prohibited from exercising his or her Option or SAR from and after the time of such termination of Continuous Service.
(l) Non-Exempt Employees . No Option or SAR granted to an Employee who is a non-exempt employee for purposes of the Fair Labor Standards Act of 1938, as amended, shall be first exercisable for any shares of Common Stock until at least six months following the date of grant of the Option or SAR. Notwithstanding the foregoing, consistent with the provisions of the Worker Economic Opportunity Act, (i) in the event of the Participant’s death or Disability, (ii) upon a Corporate Transaction in which such Option or SAR is not assumed, continued, or substituted, (iii) upon a Change in Control, or (iv) upon the Participant’s retirement (as such term may be defined in the Participant’s Award Agreement or in another applicable agreement or in accordance with the Company’s then current employment policies and guidelines), any such vested Options and SARs may be exercised earlier than six months following the date of grant. The foregoing provision is intended to operate so that any income derived by a non-exempt employee in connection with the exercise or vesting of an Option or SAR will be exempt from his or her regular rate of pay.
6. P ROVISIONS OF S TOCK A WARDS OTHER THAN O PTIONS AND SAR S .
(a) Restricted Stock Awards. Each Restricted Stock Award Agreement shall be in such form and shall contain such terms and conditions as the Board shall deem appropriate. To
9.
the extent consistent with the Company’s Bylaws, at the Board’s election, shares of Common Stock may be (x) held in book entry form subject to the Company’s instructions until any restrictions relating to the Restricted Stock Award lapse; or (y) evidenced by a certificate, which certificate shall be held in such form and manner as determined by the Board. The terms and conditions of Restricted Stock Award Agreements may change from time to time, and the terms and conditions of separate Restricted Stock Award Agreements need not be identical; provided, however , that each Restricted Stock Award Agreement shall conform to (through incorporation of the provisions hereof by reference in the agreement or otherwise) the substance of each of the following provisions:
(i) Consideration. A Restricted Stock Award may be awarded in consideration for (A) cash, check, bank draft or money order payable to the Company, (B) past services to the Company or an Affiliate, or (C) any other form of legal consideration (including future services) that may be acceptable to the Board, in its sole discretion, and permissible under applicable law.
(ii) Vesting. Shares of Common Stock awarded under the Restricted Stock Award Agreement may be subject to forfeiture to the Company in accordance with a vesting schedule to be determined by the Board.
(iii) Termination of Participant’s Continuous Service. If a Participant’s Continuous Service terminates, the Company may receive through a forfeiture condition or a repurchase right any or all of the shares of Common Stock held by the Participant that have not vested as of the date of termination of Continuous Service under the terms of the Restricted Stock Award Agreement.
(iv) Transferability. Rights to acquire shares of Common Stock under the Restricted Stock Award Agreement shall be transferable by the Participant only upon such terms and conditions as are set forth in the Restricted Stock Award Agreement, as the Board shall determine in its sole discretion, so long as Common Stock awarded under the Restricted Stock Award Agreement remains subject to the terms of the Restricted Stock Award Agreement.
(v) Dividends. A Restricted Stock Award Agreement may provide that any dividends paid on Restricted Stock will be subject to the same vesting and forfeiture restrictions as apply to the shares subject to the Restricted Stock Award to which they relate.
(b) Restricted Stock Unit Awards . Each Restricted Stock Unit Award Agreement shall be in such form and shall contain such terms and conditions as the Board shall deem appropriate. The terms and conditions of Restricted Stock Unit Award Agreements may change from time to time, and the terms and conditions of separate Restricted Stock Unit Award Agreements need not be identical; provided, however , that each Restricted Stock Unit Award Agreement shall conform to (through incorporation of the provisions hereof by reference in the Agreement or otherwise) the substance of each of the following provisions:
(i) Consideration. At the time of grant of a Restricted Stock Unit Award, the Board will determine the consideration, if any, to be paid by the Participant upon delivery of each share of Common Stock subject to the Restricted Stock Unit Award. The consideration to
10.
be paid (if any) by the Participant for each share of Common Stock subject to a Restricted Stock Unit Award may be paid in any form of legal consideration that may be acceptable to the Board, in its sole discretion, and permissible under applicable law.
(ii) Vesting. At the time of the grant of a Restricted Stock Unit Award, the Board may impose such restrictions on or conditions to the vesting of the Restricted Stock Unit Award as it, in its sole discretion, deems appropriate.
(iii) Payment . A Restricted Stock Unit Award may be settled by the delivery of shares of Common Stock, their cash equivalent, any combination thereof or in any other form of consideration, as determined by the Board and contained in the Restricted Stock Unit Award Agreement.
(iv) Additional Restrictions. At the time of the grant of a Restricted Stock Unit Award, the Board, as it deems appropriate, may impose such restrictions or conditions that delay the delivery of the shares of Common Stock (or their cash equivalent) subject to a Restricted Stock Unit Award to a time after the vesting of such Restricted Stock Unit Award.
(v) Dividend Equivalents. Dividend equivalents may be credited in respect of shares of Common Stock covered by a Restricted Stock Unit Award, as determined by the Board and contained in the Restricted Stock Unit Award Agreement. At the sole discretion of the Board, such dividend equivalents may be converted into additional shares of Common Stock covered by the Restricted Stock Unit Award in such manner as determined by the Board. Any additional shares covered by the Restricted Stock Unit Award credited by reason of such dividend equivalents will be subject to all of the same terms and conditions of the underlying Restricted Stock Unit Award Agreement to which they relate.
(vi) Termination of Participant’s Continuous Service. Except as otherwise provided in the applicable Restricted Stock Unit Award Agreement, such portion of the Restricted Stock Unit Award that has not vested will be forfeited upon the Participant’s termination of Continuous Service.
(c) Performance Awards.
(i) Performance Stock Awards . A Performance Stock Award is a Stock Award that may vest or may be exercised contingent upon the attainment during a Performance Period of certain Performance Goals. A Performance Stock Award may, but need not, require the completion of a specified period of Continuous Service. The length of any Performance Period, the Performance Goals to be achieved during the Performance Period, and the measure of whether and to what degree such Performance Goals have been attained shall be conclusively determined by the Committee, in its sole discretion. The maximum number of shares covered by an Award that may be granted to any Participant in a calendar year attributable to Stock Awards described in this Section 6(c)(i) (whether the grant, vesting or exercise is contingent upon the attainment during a Performance Period of the Performance Goals) shall not exceed 800,000 shares of Common Stock. The Board may provide for or, subject to such terms and conditions as the Board may specify, may permit a Participant to elect for, the payment of any Performance Stock Award to be deferred to a specified date or event. In addition, to the extent permitted by applicable law and the applicable Award Agreement, the Board may determine that cash may be used in payment of Performance Stock Awards.
11.
(ii) Performance Cash Awards . A Performance Cash Award is a cash award that may be paid contingent upon the attainment during a Performance Period of certain Performance Goals. A Performance Cash Award may also require the completion of a specified period of Continuous Service. At the time of grant of a Performance Cash Award, the length of any Performance Period, the Performance Goals to be achieved during the Performance Period, and the measure of whether and to what degree such Performance Goals have been attained shall be conclusively determined by the Committee, in its sole discretion. In any calendar year, the Committee may not grant a Performance Cash Award that has a maximum value that may be paid to any Participant in excess of $1,000,000. The Board may provide for or, subject to such terms and conditions as the Board may specify, may permit a Participant to elect for, the payment of any Performance Cash Award to be deferred to a specified date or event. The Committee may specify the form of payment of Performance Cash Awards, which may be cash or other property, or may provide for a Participant to have the option for his or her Performance Cash Award, or such portion thereof as the Board may specify, to be paid in whole or in part in cash or other property.
(iii) Section 162(m) Compliance . Unless otherwise permitted in compliance with the requirements of Section 162(m) of the Code with respect to an Award intended to qualify as “performance-based compensation” thereunder, the Committee shall establish the Performance Goals applicable to, and the formula for calculating the amount payable under, the Award no later than the earlier of (a) the date ninety (90) days after the commencement of the applicable Performance Period, or (b) the date on which twenty-five (25%) of the Performance Period has elapsed, and in any event at a time when the achievement of the applicable Performance Goals remains substantially uncertain. Prior to the payment of any compensation under an Award intended to qualify as “performance-based compensation” under Section 162(m) of the Code, the Committee shall certify the extent to which any Performance Goals and any other material terms under such Award have been satisfied (other than in cases where such relate solely to the increase in the value of the Common Stock). Notwithstanding satisfaction of any completion of any Performance Goals, to the extent specified at the time of grant of an Award to “covered employees” within the meaning of Section 162(m) of the Code, the number of Shares, Options, cash or other benefits granted, issued, retainable and/or vested under an Award on account of satisfaction of such Performance Goals may be reduced by the Committee on the basis of such further considerations as the Committee, in its sole discretion, shall determine.
(d) Other Stock Awards . Other forms of Stock Awards valued in whole or in part by reference to, or otherwise based on, Common Stock, including the appreciation in value thereof (e.g., options or stock rights with an exercise price or strike price less than 100% of the Fair Market Value of the Common Stock at the time of grant) may be granted either alone or in addition to Stock Awards provided for under Section 5 and the preceding provisions of this Section 6. Subject to the provisions of the Plan, the Board shall have sole and complete authority to determine the persons to whom and the time or times at which such Other Stock Awards will be granted, the number of shares of Common Stock (or the cash equivalent thereof) to be granted pursuant to such Other Stock Awards and all other terms and conditions of such Other Stock Awards.
12.
7. C OVENANTS OF THE C OMPANY .
(a) Availability of Shares. During the terms of the Stock Awards, the Company shall keep available at all times the number of shares of Common Stock reasonably required to satisfy such Stock Awards.
(b) Securities Law Compliance. The Company shall seek to obtain from each regulatory commission or agency having jurisdiction over the Plan such authority as may be required to grant Stock Awards and to issue and sell shares of Common Stock upon exercise of the Stock Awards; provided, however , that this undertaking shall not require the Company to register under the Securities Act the Plan, any Stock Award or any Common Stock issued or issuable pursuant to any such Stock Award. If, after reasonable efforts, the Company is unable to obtain from any such regulatory commission or agency the authority that counsel for the Company deems necessary for the lawful issuance and sale of Common Stock under the Plan, the Company shall be relieved from any liability for failure to issue and sell Common Stock upon exercise of such Stock Awards unless and until such authority is obtained. A Participant shall not be eligible for the grant of a Stock Award or the subsequent issuance of Common Stock pursuant to the Stock Award if such grant or issuance would be in violation of any applicable securities law.
(c) No Obligation to Notify or Minimize Taxes. The Company shall have no duty or obligation to any Participant to advise such holder as to the time or manner of exercising such Stock Award. Furthermore, the Company shall have no duty or obligation to warn or otherwise advise such holder of a pending termination or expiration of a Stock Award or a possible period in which the Stock Award may not be exercised. The Company has no duty or obligation to minimize the tax consequences of a Stock Award to the holder of such Stock Award.
8. M ISCELLANEOUS .
(a) Use of Proceeds from Sales of Common Stock. Proceeds from the sale of shares of Common Stock pursuant to Stock Awards shall constitute general funds of the Company.
(b) Corporate Action Constituting Grant of Stock Awards. Corporate action constituting a grant by the Company of a Stock Award to any Participant shall be deemed completed as of the date of such corporate action, unless otherwise determined by the Board, regardless of when the instrument, certificate, or letter evidencing the Stock Award is communicated to, or actually received or accepted by, the Participant.
(c) Stockholder Rights. No Participant shall be deemed to be the holder of, or to have any of the rights of a holder with respect to, any shares of Common Stock subject to such Stock Award unless and until (i) such Participant has satisfied all requirements for exercise of the Stock Award pursuant to its terms, if applicable, and (ii) the issuance of the Common Stock subject to such Stock Award has been entered into the books and records of the Company.
(d) No Employment or Other Service Rights. Nothing in the Plan, any Stock Award Agreement or any other instrument executed thereunder or in connection with any Award granted pursuant thereto shall confer upon any Participant any right to continue to serve the
13.
Company or an Affiliate in the capacity in effect at the time the Stock Award was granted or shall affect the right of the Company or an Affiliate to terminate (i) the employment of an Employee with or without notice and with or without cause, (ii) the service of a Consultant pursuant to the terms of such Consultant’s agreement with the Company or an Affiliate, or (iii) the service of a Director pursuant to the Bylaws of the Company or an Affiliate, and any applicable provisions of the corporate law of the state in which the Company or the Affiliate is incorporated, as the case may be.
(e) Incentive Stock Option $100,000 Limitation. To the extent that the aggregate Fair Market Value (determined at the time of grant) of Common Stock with respect to which Incentive Stock Options are exercisable for the first time by any Optionholder during any calendar year (under all plans of the Company and any Affiliates) exceeds one hundred thousand dollars ($100,000), the Options or portions thereof that exceed such limit (according to the order in which they were granted) shall be treated as Nonstatutory Stock Options, notwithstanding any contrary provision of the applicable Option Agreement(s).
(f) Investment Assurances. The Company may require a Participant, as a condition of exercising or acquiring Common Stock under any Stock Award, (i) to give written assurances satisfactory to the Company as to the Participant’s knowledge and experience in financial and business matters and/or to employ a purchaser representative reasonably satisfactory to the Company who is knowledgeable and experienced in financial and business matters and that he or she is capable of evaluating, alone or together with the purchaser representative, the merits and risks of exercising the Stock Award; and (ii) to give written assurances satisfactory to the Company stating that the Participant is acquiring Common Stock subject to the Stock Award for the Participant’s own account and not with any present intention of selling or otherwise distributing the Common Stock. The foregoing requirements, and any assurances given pursuant to such requirements, shall be inoperative if (A) the issuance of the shares upon the exercise or acquisition of Common Stock under the Stock Award has been registered under a then currently effective registration statement under the Securities Act, or (B) as to any particular requirement, a determination is made by counsel for the Company that such requirement need not be met in the circumstances under the then applicable securities laws. The Company may, upon advice of counsel to the Company, place legends on stock certificates issued under the Plan as such counsel deems necessary or appropriate in order to comply with applicable securities laws, including, but not limited to, legends restricting the transfer of the Common Stock.
(g) Withholding Obligations. Unless prohibited by the terms of a Stock Award Agreement, the Company may, in its sole discretion, satisfy any federal, state or local tax withholding obligation relating to an Award by any of the following means or by a combination of such means: (i) causing the Participant to tender a cash payment; (ii) withholding shares of Common Stock from the shares of Common Stock issued or otherwise issuable to the Participant in connection with the Award; provided, however, that no shares of Common Stock are withheld with a value exceeding the minimum amount of tax required to be withheld by law (or such lesser amount as may be necessary to avoid classification of the Stock Award as a liability for financial accounting purposes); (iii) withholding cash from an Award settled in cash; (iv) withholding payment from any amounts otherwise payable to the Participant; or (v) by such other method as may be set forth in the Award Agreement.
14.
(h) Electronic Delivery . Any reference herein to a “written” agreement or document shall include any agreement or document delivered electronically or posted on the Company’s intranet.
(i) Deferrals. To the extent permitted by applicable law, the Board, in its sole discretion, may determine that the delivery of Common Stock or the payment of cash, upon the exercise, vesting or settlement of all or a portion of any Award may be deferred and may establish programs and procedures for deferral elections to be made by Participants. Deferrals by Participants will be made in accordance with Section 409A of the Code. Consistent with Section 409A of the Code, the Board may provide for distributions while a Participant is still an employee or otherwise providing services to the Company. The Board is authorized to make deferrals of Awards and determine when, and in what annual percentages, Participants may receive payments, including lump sum payments, following the Participant’s termination of Continuous Service, and implement such other terms and conditions consistent with the provisions of the Plan and in accordance with applicable law.
(j) Compliance with Section 409A. To the extent that the Board determines that any Award granted hereunder is subject to Section 409A of the Code, the Award Agreement evidencing such Award shall incorporate the terms and conditions necessary to avoid the consequences specified in Section 409A(a)(1) of the Code. To the extent applicable, the Plan and Award Agreements shall be interpreted in accordance with Section 409A of the Code. Notwithstanding anything to the contrary in this Plan (and unless the Award Agreement specifically provides otherwise), if the Shares are publicly traded and a Participant holding an Award that constitutes “deferred compensation” under Section 409A of the Code is a “specified employee” for purposes of Section 409A of the Code, no distribution or payment of any amount shall be made upon a “separation from service” before a date that is six (6) months following the date of such Participant’s “separation from service” (as defined in Section 409A of the Code without regard to alternative definitions thereunder) or, if earlier, the date of the Participant’s death.
9. A DJUSTMENTS UPON C HANGES IN C OMMON S TOCK ; O THER C ORPORATE E VENTS .
(a) Capitalization Adjustments . In the event of a Capitalization Adjustment, the Board shall appropriately and proportionately adjust: (i) the class(es) and maximum number of securities subject to the Plan pursuant to Section 3(a), (ii) the class(es) and maximum number of securities that may be issued pursuant to the exercise of Incentive Stock Options pursuant to Section 3(d), (iii) the class(es) and maximum number of securities that may be awarded to any person pursuant to Sections 4(c) and 6(c)(i), and (iv) the class(es) and number of securities and price per share of stock subject to outstanding Stock Awards. The Board shall make such adjustments, and its determination shall be final, binding and conclusive.
(b) Dissolution or Liquidation . Except as otherwise provided in the Stock Award Agreement, in the event of a dissolution or liquidation of the Company, all outstanding Stock Awards (other than Stock Awards consisting of vested and outstanding shares of Common Stock not subject to a forfeiture condition or the Company’s right of repurchase) shall terminate immediately prior to the completion of such dissolution or liquidation, and the shares of Common Stock subject to the Company’s repurchase rights or subject to a forfeiture condition
15.
may be repurchased or reacquired by the Company notwithstanding the fact that the holder of such Stock Award is providing Continuous Service, provided, however , that the Board may, in its sole discretion, cause some or all Stock Awards to become fully vested, exercisable and/or no longer subject to repurchase or forfeiture (to the extent such Stock Awards have not previously expired or terminated) before the dissolution or liquidation is completed but contingent on its completion.
(c) Corporate Transaction. The following provisions shall apply to Stock Awards in the event of a Corporate Transaction unless otherwise provided in the instrument evidencing the Stock Award or any other written agreement between the Company or any Affiliate and the holder of the Stock Award or unless otherwise expressly provided by the Board at the time of grant of a Stock Award. In the event of a Corporate Transaction, then, notwithstanding any other provision of the Plan, the Board shall take one or more of the following actions with respect to Stock Awards, contingent upon the closing or completion of the Corporate Transaction:
(i) arrange for the surviving corporation or acquiring corporation (or the surviving or acquiring corporation’s parent company) to assume or continue the Stock Award or to substitute a similar stock award for the Stock Award (including, but not limited to, an award to acquire the same consideration paid to the stockholders of the Company pursuant to the Corporate Transaction);
(ii) arrange for the assignment of any reacquisition or repurchase rights held by the Company in respect of Common Stock issued pursuant to the Stock Award to the surviving corporation or acquiring corporation (or the surviving or acquiring corporation’s parent company);
(iii) accelerate the vesting of the Stock Award (and, if applicable, the time at which the Stock Award may be exercised) to a date prior to the effective time of such Corporate Transaction as the Board shall determine (or, if the Board shall not determine such a date, to the date that is five (5) days prior to the effective date of the Corporate Transaction), with such Stock Award terminating if not exercised (if applicable) at or prior to the effective time of the Corporate Transaction;
(iv) arrange for the lapse of any reacquisition or repurchase rights held by the Company with respect to the Stock Award;
(v) cancel or arrange for the cancellation of the Stock Award, to the extent not vested or not exercised prior to the effective time of the Corporate Transaction, in exchange for such cash consideration, if any, as the Board, in its sole discretion, may consider appropriate; and
(vi) make a payment, in such form as may be determined by the Board equal to the excess, if any, of (A) the value of the property the Participant would have received upon the exercise of the Stock Award, over (B) any exercise price payable by such holder in connection with such exercise.
The Board need not take the same action or actions with respect to all Stock Awards or portions thereof or with respect to all Participants.
16.
(d) Change in Control. A Stock Award may be subject to additional acceleration of vesting and exercisability upon or after a Change in Control as may be provided in the Stock Award Agreement for such Stock Award or as may be provided in any other written agreement between the Company or any Affiliate and the Participant, but in the absence of such provision, no such acceleration shall occur.
10. T ERMINATION OR S USPENSION OF THE P LAN .
(a) Plan Term. The Board may suspend or terminate the Plan at any time. Unless terminated sooner by the Board, the Plan shall automatically terminate on the day before the tenth (10th) anniversary of the earlier of (i) the date the Plan is adopted by the Board, or (ii) the date the Plan is approved by the stockholders of the Company. No Awards may be granted under the Plan while the Plan is suspended or after it is terminated.
(b) No Impairment of Rights. Suspension or termination of the Plan shall not impair rights and obligations under any Award granted while the Plan is in effect except with the written consent of the affected Participant.
11. E FFECTIVE D ATE OF P LAN .
This Plan shall become effective on the Effective Date, but no Award shall be exercised (or in the case of a Restricted Stock Award, Restricted Stock Unit Award, or Other Stock Award shall be granted) unless and until the Plan has been approved by the stockholders of the Company, which approval shall be within twelve months before or after the date the Plan is adopted by the Board.
12. C HOICE OF L AW .
The law of the state of California shall govern all questions concerning the construction, validity and interpretation of this Plan, without regard to that state’s conflict of laws rules.
13. D EFINITIONS . As used in the Plan, the following definitions shall apply to the capitalized terms indicated below:
(a) “ Affiliate ” means, at the time of determination, any “parent” or “subsidiary” of the Company as such terms are defined in Rule 405 of the Securities Act. The Board shall have the authority to determine the time or times at which “parent” or “subsidiary” status is determined within the foregoing definition.
(b) “ Award ” means a Stock Award or a Performance Cash Award.
(c) “ Award Agreement ” means a written agreement between the Company and a Participant evidencing the terms and conditions of an Award.
(d) “ Board ” means the Board of Directors of the Company.
(e) “ Capitalization Adjustment ” means any change that is made in, or other events that occur with respect to, the Common Stock subject to the Plan or subject to any Stock Award
17.
after the Effective Date without the receipt of consideration by the Company through merger, consolidation, reorganization, recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, stock split, liquidating dividend, combination of shares, exchange of shares, change in corporate structure or any similar equity restructuring transaction, as that term is used in Statement of Financial Accounting Standards No. 123 (revised). Notwithstanding the foregoing, the conversion of any convertible securities of the Company shall not be treated as a Capitalization Adjustment.
(f) “Cause” shall have the meaning ascribed to such term in any written agreement between the Participant and the Company defining such term and, in the absence of such agreement, such term shall mean with respect to a Participant, the occurrence of any of the following events, if such event results in a demonstrably harmful impact on the Company’s business or reputation, or that of any of its Subsidiaries: (i) such Participant’s commission of any felony or any crime involving fraud, dishonesty or moral turpitude under the laws of the United States or any state thereof; (ii) such Participant’s attempted commission of, or participation in, a fraud or act of dishonesty against the Company; (iii) such Participant’s intentional, material violation of any contract or agreement between the Participant and the Company or of any statutory duty owed to the Company; (iv) such Participant’s unauthorized use or disclosure of the Company’s confidential information or trade secrets; or (v) such Participant’s gross misconduct. The determination that a termination of the Participant’s Continuous Service is either for Cause or without Cause shall be made by the Company in its sole discretion. Any determination by the Company that the Continuous Service of a Participant was terminated by reason of dismissal without Cause for the purposes of outstanding Awards held by such Participant shall have no effect upon any determination of the rights or obligations of the Company or such Participant for any other purpose.
(g) “ Change in Control ” means the occurrence, in a single transaction or in a series of related transactions, of any one or more of the following events:
(i) any Exchange Act Person becomes the Owner, directly or indirectly, of securities of the Company representing more than fifty percent (50%) of the combined voting power of the Company’s then outstanding securities other than by virtue of a merger, consolidation or similar transaction. Notwithstanding the foregoing, a Change in Control shall not be deemed to occur (A) on account of the acquisition of securities of the Company directly from the Company, (B) on account of the acquisition of securities of the Company by an investor, any affiliate thereof or any other Exchange Act Person that acquires the Company’s securities in a transaction or series of related transactions the primary purpose of which is to obtain financing for the Company through the issuance of equity securities, or (C) solely because the level of Ownership held by any Exchange Act Person (the “ Subject Person ” ) exceeds the designated percentage threshold of the outstanding voting securities as a result of a repurchase or other acquisition of voting securities by the Company reducing the number of shares outstanding, provided that if a Change in Control would occur (but for the operation of this sentence) as a result of the acquisition of voting securities by the Company, and after such share acquisition, the Subject Person becomes the Owner of any additional voting securities that, assuming the repurchase or other acquisition had not occurred, increases the percentage of the then outstanding voting securities Owned by the Subject Person over the designated percentage threshold, then a Change in Control shall be deemed to occur;
18.
(ii) there is consummated a merger, consolidation or similar transaction involving (directly or indirectly) the Company and, immediately after the consummation of such merger, consolidation or similar transaction, the stockholders of the Company immediately prior thereto do not Own, directly or indirectly, either (A) outstanding voting securities representing more than fifty percent (50%) of the combined outstanding voting power of the surviving Entity in such merger, consolidation or similar transaction or (B) more than fifty percent (50%) of the combined outstanding voting power of the parent of the surviving Entity in such merger, consolidation or similar transaction, in each case in substantially the same proportions as their Ownership of the outstanding voting securities of the Company immediately prior to such transaction;
(iii) the stockholders of the Company approve or the Board approves a plan of complete dissolution or liquidation of the Company, or a complete dissolution or liquidation of the Company shall otherwise occur, except for a liquidation into a parent corporation;
(iv) there is consummated a sale, lease, exclusive license or other disposition of all or substantially all of the consolidated assets of the Company and its Subsidiaries, other than a sale, lease, license or other disposition of all or substantially all of the consolidated assets of the Company and its Subsidiaries to an Entity, more than fifty percent (50%) of the combined voting power of the voting securities of which are Owned by stockholders of the Company in substantially the same proportions as their Ownership of the outstanding voting securities of the Company immediately prior to such sale, lease, license or other disposition; or
(v) individuals who, on the date the Plan is adopted by the Board, are members of the Board (the “ Incumbent Board ”) cease for any reason to constitute at least a majority of the members of the Board; provided, however , that if the appointment or election (or nomination for election) of any new Board member was approved or recommended by a majority vote of the members of the Incumbent Board then still in office, such new member shall, for purposes of this Plan, be considered as a member of the Incumbent Board.
Notwithstanding the foregoing or any other provision of this Plan, (A) the term Change in Control shall not include a sale of assets, merger or other transaction effected exclusively for the purpose of changing the domicile of the Company, and (B) the definition of Change in Control (or any analogous term) in an individual written agreement between the Company or any Affiliate and the Participant shall supersede the foregoing definition with respect to Awards subject to such agreement; provided, however , that if no definition of Change in Control or any analogous term is set forth in such an individual written agreement, the foregoing definition shall apply.
(h) “ Code ” means the Internal Revenue Code of 1986, as amended, including any applicable regulations and guidance thereunder.
(i) “ Committee ” means a committee of one or more Directors to whom authority has been delegated by the Board in accordance with Section 2(c).
(j) “ Common Stock ” means the common stock of the Company.
(k) “ Company ” means Trius Therapeutics, Inc., a Delaware corporation.
19.
(l) “ Consultant ” means any person, including an advisor, who is (i) engaged by the Company or an Affiliate to render consulting or advisory services and is compensated for such services, or (ii) serving as a member of the board of directors of an Affiliate and is compensated for such services. However, service solely as a Director, or payment of a fee for such service, shall not cause a Director to be considered a “Consultant” for purposes of the Plan. Notwithstanding the foregoing, a person is treated as a Consultant under this Plan only if a Form S-8 Registration Statement under the Securities Act is available to register either the offer or the sale of the Company’s securities to such person.
(m) “ Continuous Service ” means that the Participant’s service with the Company or an Affiliate, whether as an Employee, Director or Consultant, is not interrupted or terminated. A change in the capacity in which the Participant renders service to the Company or an Affiliate as an Employee, Consultant or Director or a change in the entity for which the Participant renders such service, provided that there is no interruption or termination of the Participant’s service with the Company or an Affiliate, shall not terminate a Participant’s Continuous Service; provided, however, if the Entity for which a Participant is rendering services ceases to qualify as an Affiliate, as determined by the Board, in its sole discretion, such Participant’s Continuous Service shall be considered to have terminated on the date such Entity ceases to qualify as an Affiliate. To the extent permitted by law, the Board or the chief executive officer of the Company, in that party’s sole discretion, may determine whether Continuous Service shall be considered interrupted in the case of (i) any leave of absence approved by the Board or Chief Executive Officer, including sick leave, military leave or any other personal leave, or (ii) transfers between the Company, an Affiliate, or their successors. Notwithstanding the foregoing, a leave of absence shall be treated as Continuous Service for purposes of vesting in a Stock Award only to such extent as may be provided in the Company’s leave of absence policy, in the written terms of any leave of absence agreement or policy applicable to the Participant, or as otherwise required by law.
(n) “ Corporate Transaction ” means the occurrence, in a single transaction or in a series of related transactions, of any one or more of the following events:
(i) the consummation of a sale or other disposition of all or substantially all, as determined by the Board, in its sole discretion, of the consolidated assets of the Company and its Subsidiaries;
(ii) the consummation of a sale or other disposition of at least ninety percent (90%) of the outstanding securities of the Company;
(iii) the consummation of a merger, consolidation or similar transaction following which the Company is not the surviving corporation; or
(iv) the consummation of a merger, consolidation or similar transaction following which the Company is the surviving corporation but the shares of Common Stock outstanding immediately preceding the merger, consolidation or similar transaction are converted or exchanged by virtue of the merger, consolidation or similar transaction into other property, whether in the form of securities, cash or otherwise.
20.
(o) “ Covered Employee ” shall have the meaning provided in Section 162(m)(3) of the Code.
(p) “ Director ” means a member of the Board.
(q) “ Disability ” means, with respect to a Participant, the inability of such Participant to engage in any substantial gainful activity by reason of any medically determinable physical or mental impairment which can be expected to result in death or which has lasted or can be expected to last for a continuous period of not less than twelve (12) months, as provided in Sections 22(e)(3) and 409A(a)(2)(c)(i) of the Code, and shall be determined by the Board on the basis of such medical evidence as the Board deems warranted under the circumstances.
(r) “ Effective Date ” means the effective date of this Plan document, which is the date of the underwriting agreement between the Company and the underwriter(s) managing the initial public offering of the Common Stock, pursuant to which the Common Stock is priced for the initial public offering.
(s) “ Employee ” means any person employed by the Company or an Affiliate. However, service solely as a Director, or payment of a fee for such services, shall not cause a Director to be considered an “Employee” for purposes of the Plan.
(t) “ Entity ” means a corporation, partnership, limited liability company or other entity.
(u) “ Exchange Act ” means the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
(v) “ Exchange Act Person ” means any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act), except that “Exchange Act Person” shall not include (i) the Company or any Subsidiary of the Company, (ii) any employee benefit plan of the Company or any Subsidiary of the Company or any trustee or other fiduciary holding securities under an employee benefit plan of the Company or any Subsidiary of the Company, (iii) an underwriter temporarily holding securities pursuant to a registered public offering of such securities, (iv) an Entity Owned, directly or indirectly, by the stockholders of the Company in substantially the same proportions as their Ownership of stock of the Company; or (v) any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act) that, as of the Effective Date, is the Owner, directly or indirectly, of securities of the Company representing more than fifty percent (50%) of the combined voting power of the Company’s then outstanding securities.
(w) “ Fair Market Value ” means, as of any date, the value of the Common Stock determined as follows:
(i) If the Common Stock is listed on any established stock exchange or traded on any established market, the Fair Market Value of a share of Common Stock shall be the closing sales price for such stock as quoted on such exchange or market (or the exchange or market with the greatest volume of trading in the Common Stock) on the date of determination, as reported in a source the Board deems reliable.
21.
(ii) Unless otherwise provided by the Board, if there is no closing sales price for the Common Stock on the date of determination, then the Fair Market Value shall be the closing selling price on the last preceding date for which such quotation exists.
(iii) In the absence of such markets for the Common Stock, the Fair Market Value shall be determined by the Board in good faith and in a manner that complies with Sections 409A and 422 of the Code.
(x) “ Incentive Stock Option ” means an option granted pursuant to Section 5 of the Plan that is intended to be, and qualifies as, an “incentive stock option” within the meaning of Section 422 of the Code.
(y) “ Non-Employee Director ” means a Director who either (i) is not a current employee or officer of the Company or an Affiliate, does not receive compensation, either directly or indirectly, from the Company or an Affiliate for services rendered as a consultant or in any capacity other than as a Director (except for an amount as to which disclosure would not be required under Item 404(a) of Regulation S-K promulgated pursuant to the Securities Act (“ Regulation S-K ”)), does not possess an interest in any other transaction for which disclosure would be required under Item 404(a) of Regulation S-K, and is not engaged in a business relationship for which disclosure would be required pursuant to Item 404(b) of Regulation S-K; or (ii) is otherwise considered a “non-employee director” for purposes of Rule 16b-3.
(z) “ Nonstatutory Stock Option ” means any option granted pursuant to Section 5 of the Plan that does not qualify as an Incentive Stock Option.
(aa) “ Officer ” means a person who is an officer of the Company within the meaning of Section 16 of the Exchange Act.
(bb) “ Option ” means an Incentive Stock Option or a Nonstatutory Stock Option to purchase shares of Common Stock granted pursuant to the Plan.
(cc) “ Option Agreement ” means a written agreement between the Company and an Optionholder evidencing the terms and conditions of an Option grant. Each Option Agreement shall be subject to the terms and conditions of the Plan.
(dd) “ Optionholder ” means a person to whom an Option is granted pursuant to the Plan or, if applicable, such other person who holds an outstanding Option.
(ee) “ Other Stock Award ” means an award based in whole or in part by reference to the Common Stock which is granted pursuant to the terms and conditions of Section 6(d).
(ff) “ Other Stock Award Agreement ” means a written agreement between the Company and a holder of an Other Stock Award evidencing the terms and conditions of an Other Stock Award grant. Each Other Stock Award Agreement shall be subject to the terms and conditions of the Plan.
22.
(gg) “ Outside Director ” means a Director who either (i) is not a current employee of the Company or an “affiliated corporation” (within the meaning of Treasury Regulations promulgated under Section 162(m) of the Code), is not a former employee of the Company or an “affiliated corporation” who receives compensation for prior services (other than benefits under a tax-qualified retirement plan) during the taxable year, has not been an officer of the Company or an “affiliated corporation,” and does not receive remuneration from the Company or an “affiliated corporation,” either directly or indirectly, in any capacity other than as a Director, or (ii) is otherwise considered an “outside director” for purposes of Section 162(m) of the Code.
(hh) “ Own, ” “ Owned, ” “ Owner, ” “ Ownership ” A person or Entity shall be deemed to “Own,” to have “Owned,” to be the “Owner” of, or to have acquired “Ownership” of securities if such person or Entity, directly or indirectly, through any contract, arrangement, understanding, relationship or otherwise, has or shares voting power, which includes the power to vote or to direct the voting, with respect to such securities.
(ii) “ Participant ” means a person to whom an Award is granted pursuant to the Plan or, if applicable, such other person who holds an outstanding Stock Award.
(jj) “ Performance Cash Award ” means an award of cash granted pursuant to the terms and conditions of Section 6(c)(ii).
(kk) “ Performance Criteria ” means the one or more criteria that the Board shall select for purposes of establishing the Performance Goals for a Performance Period. The Performance Criteria that shall be used to establish such Performance Goals may be based on any one of, or combination of, the following as determined by the Board: (i) earnings (including earnings per share and net earnings); (ii) earnings before interest, taxes and depreciation; (iii) earnings before interest, taxes, depreciation and amortization; (iv) total stockholder return; (v) return on equity or average stockholder’s equity; (vi) return on assets, investment, or capital employed; (vii) stock price; (viii) margin (including gross margin); (ix) income (before or after taxes); (x) operating income; (xi) operating income after taxes; (xii) pre-tax profit; (xiii) operating cash flow; (xiv) sales or revenue targets; (xv) increases in revenue or product revenue; (xvi) expenses and cost reduction goals; (xvii) improvement in or attainment of working capital levels; (xiii) economic value added (or an equivalent metric); (xix) market share; (xx) cash flow; (xxi) cash flow per share; (xxii) share price performance; (xxiii) debt reduction; (xxiv) implementation or completion of projects or processes; (xxv) customer satisfaction; (xxvi) stockholders’ equity; (xxvii) capital expenditures; (xxiii) debt levels; (xxix) operating profit or net operating profit; (xxx) workforce diversity; (xxxi) growth of net income or operating income; (xxxii) billings; and (xxxiii) to the extent that an Award is not intended to comply with Section 162(m) of the Code, other measures of performance selected by the Board.
(ll) “ Performance Goals ” means, for a Performance Period, the one or more goals established by the Board for the Performance Period based upon the Performance Criteria. Performance Goals may be based on a Company-wide basis, with respect to one or more business units, divisions, Affiliates, or business segments, and in either absolute terms or relative to the performance of one or more comparable companies or the performance of one or more relevant indices. Unless specified otherwise by the Board (i) in the Award Agreement at the time the Award is granted or (ii) in such other document setting forth the Performance Goals at
23.
the time the Performance Goals are established, the Board shall appropriately make adjustments in the method of calculating the attainment of Performance Goals for a Performance Period as follows: (1) to exclude restructuring and/or other nonrecurring charges; (2) to exclude exchange rate effects, as applicable, for non-U.S. dollar denominated Performance Goals; (3) to exclude the effects of changes to generally accepted accounting principles; (4) to exclude the effects of any statutory adjustments to corporate tax rates; and (5) to exclude the effects of any “extraordinary items” as determined under generally accepted accounting principles. In addition, the Board retains the discretion to reduce or eliminate the compensation or economic benefit due upon attainment of Performance Goals and to define the manner of calculating the Performance Criteria it selects to use for such Performance Period. Partial achievement of the specified criteria may result in the payment or vesting corresponding to the degree of achievement as specified in the Stock Award Agreement or the written terms of a Performance Cash Award.
(mm) “ Performance Period ” means the period of time selected by the Board over which the attainment of one or more Performance Goals will be measured for the purpose of determining a Participant’s right to and the payment of a Stock Award or a Performance Cash Award. Performance Periods may be of varying and overlapping duration, at the sole discretion of the Board.
(nn) “ Performance Stock Award ” means a Stock Award granted under the terms and conditions of Section 6(c)(i).
(oo) “ Plan ” means this Trius Therapeutics, Inc. 2010 Equity Incentive Plan.
(pp) “ Restricted Stock Award ” means an award of shares of Common Stock which is granted pursuant to the terms and conditions of Section 6(a).
(qq) “ Restricted Stock Award Agreement ” means a written agreement between the Company and a holder of a Restricted Stock Award evidencing the terms and conditions of a Restricted Stock Award grant. Each Restricted Stock Award Agreement shall be subject to the terms and conditions of the Plan.
(rr) “ Restricted Stock Unit Award ” means a right to receive shares of Common Stock which is granted pursuant to the terms and conditions of Section 6(b).
(ss) “ Restricted Stock Unit Award Agreement ” means a written agreement between the Company and a holder of a Restricted Stock Unit Award evidencing the terms and conditions of a Restricted Stock Unit Award grant. Each Restricted Stock Unit Award Agreement shall be subject to the terms and conditions of the Plan.
(tt) “ Rule 16b-3 ” means Rule 16b-3 promulgated under the Exchange Act or any successor to Rule 16b-3, as in effect from time to time.
(uu) “ Securities Act ” means the Securities Act of 1933, as amended.
(vv) “ Stock Appreciation Right ” or “ SAR ” means a right to receive the appreciation on Common Stock that is granted pursuant to the terms and conditions of Section 5.
24.
(ww) “ Stock Appreciation Right Agreement ” means a written agreement between the Company and a holder of a Stock Appreciation Right evidencing the terms and conditions of a Stock Appreciation Right grant. Each Stock Appreciation Right Agreement shall be subject to the terms and conditions of the Plan.
(xx) “ Stock Award ” means any right to receive Common Stock granted under the Plan, including an Incentive Stock Option, a Nonstatutory Stock Option, a Restricted Stock Award, a Restricted Stock Unit Award, a Stock Appreciation Right, a Performance Stock Award or any Other Stock Award.
(yy) “ Stock Award Agreement ” means a written agreement between the Company and a Participant evidencing the terms and conditions of a Stock Award grant. Each Stock Award Agreement shall be subject to the terms and conditions of the Plan.
(zz) “ Subsidiary ” means, with respect to the Company, (i) any corporation of which more than fifty percent (50%) of the outstanding capital stock having ordinary voting power to elect a majority of the board of directors of such corporation (irrespective of whether, at the time, stock of any other class or classes of such corporation shall have or might have voting power by reason of the happening of any contingency) is at the time, directly or indirectly, Owned by the Company, and (ii) any partnership, limited liability company or other entity in which the Company has a direct or indirect interest (whether in the form of voting or participation in profits or capital contribution) of more than fifty percent (50%).
(aaa) “ Ten Percent Stockholder ” means a person who Owns (or is deemed to Own pursuant to Section 424(d) of the Code) stock possessing more than ten percent (10%) of the total combined voting power of all classes of stock of the Company or any Affiliate.
25.
Exhibit 99.2
T RIUS T HERAPEUTICS , I NC .
A MENDED AND R ESTATED 2010 N ON -E MPLOYEE D IRECTORS ’
S TOCK O PTION P LAN
A MENDED AND R ESTATED ON A UGUST 11, 2011
A MENDED BY THE B OARD OF D IRECTORS : M ARCH 7, 2013
A MENDED BY THE B OARD OF D IRECTORS : M AY 21, 2013
1. G ENERAL .
(a) Eligible Option Recipients. The persons eligible to receive Options are the Non-Employee Directors of the Company.
(b) Purpose. The Company, by means of the Plan, seeks to retain the services of its Non-Employee Directors, to secure and retain the services of new Non-Employee Directors and to provide incentives for such persons to exert maximum efforts for the success of the Company and any Affiliate by giving them an opportunity to benefit from increases in value of the Common Stock through the automatic grant of Nonstatutory Stock Options.
2. A DMINISTRATION .
(a) Administration by Board. The Board shall administer the Plan. The Board may not delegate administration of the Plan.
(b) Powers of Board. The Board shall have the power, subject to, and within the limitations of, the express provisions of the Plan:
(i) To determine the provisions of each Option to the extent not specified in the Plan.
(ii) To construe and interpret the Plan and Options granted under it, and to establish, amend and revoke rules and regulations for its administration. The Board, in the exercise of this power, may correct any defect, omission or inconsistency in the Plan or in any Option Agreement, in a manner and to the extent it shall deem necessary or expedient to make the Plan fully effective.
(iii) To amend the Plan or an Option as provided in Section 10.
(iv) To terminate or suspend the Plan as provided in Section 11.
(v) Generally, to exercise such powers and to perform such acts as the Board deems necessary or expedient to promote the best interests of the Company and that are not in conflict with the provisions of the Plan.
(c) Effect of Board’s Decision. All determinations, interpretations and constructions made by the Board in good faith shall not be subject to review by any person and shall be final, binding and conclusive on all persons.
1.
3. S HARES S UBJECT TO THE P LAN .
(a) Share Reserve. Subject to Section 9(a) relating to Capitalization Adjustments, the aggregate number of shares of Common Stock of the Company that may be issued pursuant to Options after the Effective Date shall not exceed 300,000 shares, plus an annual increase to be added on January 1st of each year for a period of four years commencing on January 1, 2011 and ending on (and including) January 1, 2015, in an amount equal to the lesser of (i) the aggregate number of shares of Common Stock subject to Options granted pursuant to Section 5 of Plan during the immediately preceding calendar year or (ii) 150,000 shares as determined by the Board. Notwithstanding the foregoing, the Board may act prior to the first day of any calendar year, to provide that there shall be no increase in the share reserve for such calendar year or that the increase in the share reserve for such calendar year shall be a lesser number of shares of Common Stock than would otherwise occur pursuant to the preceding sentence. For clarity, the limitation in this Section 3(a) is a limitation in the number of shares of Common Stock that may be issued pursuant to the Plan. Accordingly, this Section 3(a) does not limit the granting of Options except as provided in Section 7(a). Shares may be issued in connection with a merger or acquisition as permitted by, as applicable, NASDAQ Listing Rule 5635(c), NYSE Listed Company Manual Section 303A.08, AMEX Company Guide Section 711 or other applicable stock exchange rules, and such issuance shall not reduce the number of shares available for issuance under the Plan. Furthermore, if an Option or any portion thereof (i) expires or otherwise terminates without all of the shares covered by such Option having been issued or (ii) is settled in cash ( i.e. , the Optionholder receives cash rather than stock), such expiration, termination or settlement shall not reduce (or otherwise offset) the number of shares Common Stock that may be available for issuance under the Plan.
(b) Reversion of Shares to the Share Reserve.
(i) Shares Available For Subsequent Issuance. If any shares of Common Stock issued pursuant to an Option are forfeited back to the Company because of the failure to meet a contingency or condition required to vest such shares in the Optionholder, then the shares that are forfeited shall revert to and again become available for issuance under the Plan.
(ii) Shares Not Available For Subsequent Issuance. If any shares of Common Stock issued pursuant to an Option are not delivered to an Optionholder because the Option is exercised through a reduction of shares subject to the Option ( i.e ., “net exercised”), the number of shares that are not delivered to the Optionholder shall not remain available for issuance under the Plan. Also, any shares reacquired by the Company pursuant to Section 8(e) or as consideration for the exercise of an Option shall not again become available for issuance under the Plan.
(c) Source of Shares. The stock issuable under the Plan shall be shares of authorized but unissued or reacquired Common Stock, including shares repurchased by the Company on the open market or otherwise.
2.
4. E LIGIBILITY .
The Options shall automatically be granted under the Plan as set forth in Section 5 to all Non-Employee Directors who meet the specified criteria.
5. N ON -D ISCRETIONARY G RANTS .
(a) Initial Grants. Without any further action of the Board, each person who after the IPO Date is elected or appointed for the first time to be a Non-Employee Director automatically shall, upon the date of his or her initial election or appointment to be a Non-Employee Director, be granted an Option (the “ Initial Grant ”) to purchase 30,000 shares of Common Stock on the terms and conditions set forth herein.
(b) Annual Grants. Without any further action of the Board, on the date of each Annual Meeting, commencing with the first Annual Meeting following the IPO Date, each person who is then a Non-Employee Director automatically shall be granted an Option (the “ Annual Grant ”) to purchase 15,000 shares of Common Stock on the terms and conditions set forth herein; provided, however, that the number of shares subject to such Annual Grant shall be reduced on a pro rata basis for each full month that the recipient thereof did not serve as a member of the Board during the 12 month period prior to the date of grant.
(c) Chairperson Grants. In addition to the Initial Grant, without any further action of the Board, each person who after the IPO Date is elected or appointed for the first time to be the chairperson of the Board automatically shall, upon the date of his or her initial election or appointment to such position, be granted an Option (the “ Initial Chairperson Grant ”) to purchase 12,000 shares of Common Stock on the terms and conditions set forth herein. Further, in addition to the Annual Grants, without any further action of the Board, on the date of each Annual Meeting, commencing with the first Annual Meeting following the IPO Date, each person who is then the chairperson of the Board automatically shall be granted an Option (the “ Annual Chairperson Grant ”) to purchase 6,000 shares of Common Stock on the terms and conditions set forth herein; provided, however, that the number of shares subject to such Annual Chairperson Grant shall be reduced on a pro rata basis for each full month that the recipient thereof did not serve as chairperson of the Board during the 12 month period prior to the date of grant.
6. O PTION P ROVISIONS .
Each Option shall be in such form and shall contain such terms and conditions as required by the Plan. Each Option shall contain such additional terms and conditions, not inconsistent with the Plan, as the Board shall deem appropriate. Each Option shall include (through incorporation of provisions hereof by reference in the Option or otherwise) the substance of each of the following provisions:
(a) Term. No Option shall be exercisable after the expiration of ten (10) years from the date it was granted.
3.
(b) Exercise Price. The exercise price of each Option shall be one hundred percent (100%) of the Fair Market Value of the Common Stock subject to the Option on the date the Option is granted.
(c) Purchase Price. The purchase price of Common Stock acquired pursuant to the exercise of an Option shall be paid, to the extent permitted by applicable law, by any combination of the following methods of payment:
(i) by cash, check, bank draft or money order payable to the Company;
(ii) to the extent permitted by law, pursuant to a program developed under Regulation T as promulgated by the Federal Reserve Board that, prior to the issuance of the stock subject to the Option, results in either the receipt of cash (or check) by the Company or the receipt of irrevocable instructions to pay the aggregate exercise price to the Company from the sales proceeds;
(iii) by delivery to the Company (either by actual delivery or attestation) of shares of Common Stock; or
(iv) by a “net exercise” arrangement pursuant to which the Company will reduce the number of shares of Common Stock issuable upon exercise by the largest whole number of shares with a Fair Market Value that does not exceed the aggregate exercise price; provided, however , that the Company shall accept a cash or other payment from the Optionholder to the extent of any remaining balance of the aggregate exercise price not satisfied by such reduction in the number of whole shares to be issued; provided, further, that shares of Common Stock will no longer be subject to an Option and will not be exercisable thereafter to the extent that (A) shares issuable upon exercise are reduced to pay the exercise price pursuant to the “net exercise,” (B) shares are delivered to the Optionholder as a result of such exercise, and (C) shares are withheld to satisfy tax withholding obligations.
(d) Transferability. An Option shall not be transferable except by will or by the laws of descent and distribution and to such further extent as permitted by the Rule as to Use of Form S-8 specified in the General Instructions of the Form S-8 Registration Statement under the Securities Act, and shall be exercisable during the lifetime of the Optionholder only by the Optionholder. Notwithstanding the foregoing, the Optionholder may, by delivering written notice to the Company, in a form satisfactory to the Company, designate a third party who, in the event of the death of the Optionholder, shall thereafter be entitled to exercise the Option.
(e) Vesting Generally. Options shall vest as follows:
(i) Initial Grant and Initial Chairperson Grant . The Initial Grant and the Initial Chairperson Grant shall vest in a series of thirty-six (36) successive equal monthly installments during the Optionholder’s Continuous Service over the three (3)-year period measured from the date of grant.
(ii) Annual Grant and Annual Chairperson Grant . The Annual Grant and Annual Chairperson Grant shall vest in a series of twelve (12) successive equal monthly installments during the Optionholder’s Continuous Service over the one (1)-year period measured from the date of grant.
4.
(f) Termination of Continuous Service. In the event that an Optionholder’s Continuous Service terminates (other than upon the Optionholder’s death or Disability), the Optionholder may exercise his or her Option but only within such period of time ending on the earlier of (i) the date twelve (12) months following the termination of the Optionholder’s Continuous Service (or such longer or shorter period specified in the applicable Option Agreement, which period shall not be less than 30 days), or (ii) the expiration of the term of the Option as set forth in the Option Agreement. If, after termination of Continuous Service, the Optionholder does not exercise his or her Option within the time specified herein or in the Option Agreement (as applicable), the Option shall terminate.
(g) Extension of Termination Date. In the event that the exercise of an Option following the termination of the Optionholder’s Continuous Service (other than upon the Optionholder’s death or Disability) would be prohibited at any time solely because the issuance of shares of Common Stock would violate the registration requirements under the Securities Act, then the Option shall terminate on the earlier of (i) the expiration of a total period of twelve (12) months (that need not be consecutive) after the termination of the Optionholder’s Continuous Service during which the exercise of the Option would not be in violation of such registration requirements, or (ii) the expiration of the term of the Option as set forth in the applicable Option Agreement. In addition, unless otherwise provided in an Optionholder’s Option Agreement, if the sale of any Common Stock received upon exercise of an Option following the termination of the Optionholder’s Continuous Service would violate the Company’s insider trading policy, then the Option shall terminate on the earlier of (i) the expiration of a period equal to the applicable post-termination exercise period after the termination of the Optionholder’s Continuous Service during which the exercise of the Option would not be in violation of the Company’s insider trading policy, or (ii) the expiration of the term of the Option as set forth in the applicable Option Agreement.
(h) Disability of Optionholder. In the event that an Optionholder’s Continuous Service terminates as a result of the Optionholder’s Disability, the Optionholder may exercise his or her Option (to the extent that the Optionholder was entitled to exercise such Option as of the date of termination of Continuous Service), but only within such period of time ending on the earlier of (i) the date twelve (12) months following such termination of Continuous Service or (ii) the expiration of the term of the Option as set forth in the Option Agreement. If, after termination of Continuous Service, the Optionholder does not exercise his or her Option within the time specified herein or in the Option Agreement (as applicable), the Option shall terminate.
(i) Death of Optionholder. In the event that (i) an Optionholder’s Continuous Service terminates as a result of the Optionholder’s death, or (ii) the Optionholder dies within the twelve (12) month period after the termination of the Optionholder’s Continuous Service for a reason other than death, then the Option may be exercised (to the extent the Optionholder was entitled to exercise such Option as of the date of death) by the Optionholder’s estate, by a person who acquired the right to exercise the Option by bequest or inheritance or by a person designated to exercise the Option upon the Optionholder’s death, but only within the period ending on the earlier of (i) the date eighteen (18) months following the date of death, or (ii) the expiration of the term of such Option as set forth in the Option Agreement. If, after the Optionholder’s death, the Option is not exercised within the time specified herein, the Option shall terminate.
5.
7. C OVENANTS OF THE C OMPANY .
(a) Availability of Shares. During the terms of the Options, the Company shall keep available at all times the number of shares of Common Stock reasonably required to satisfy such Options.
(b) Securities Law Compliance. The Company shall seek to obtain from each regulatory commission or agency having jurisdiction over the Plan such authority as may be required to grant Options and to issue and sell shares of Common Stock upon exercise of the Options; provided, however , that this undertaking shall not require the Company to register under the Securities Act the Plan, any Option or any Common Stock issued or issuable pursuant to any such Option. If, after reasonable efforts, the Company is unable to obtain from any such regulatory commission or agency the authority that counsel for the Company deems necessary for the lawful issuance and sale of Common Stock under the Plan, the Company shall be relieved from any liability for failure to issue and sell Common Stock upon exercise of such Options unless and until such authority is obtained. A Optionholder shall not be eligible for the grant of an Option or the subsequent issuance of Common Stock pursuant to the Option if such grant or issuance would be in violation of any applicable securities law.
(c) No Obligation to Notify or Minimize Taxes. The Company shall have no duty or obligation to any Optionholder to advise such holder as to the time or manner of exercising such Option. Furthermore, the Company shall have no duty or obligation to warn or otherwise advise such holder of a pending termination or expiration of an Option or a possible period in which the Option may not be exercised. The Company has no duty or obligation to minimize the tax consequences of an Option to the holder of such Option.
8. M ISCELLANEOUS .
(a) Use of Proceeds from Sales of Common Stock. Proceeds from the sale of shares of Common Stock pursuant to Options shall constitute general funds of the Company.
(b) Stockholder Rights. No Optionholder shall be deemed to be the holder of, or to have any of the rights of a holder with respect to, any shares of Common Stock subject to such Option unless and until (i) such Optionholder has satisfied all requirements for exercise of the Option pursuant to its terms, if applicable, and (ii) the issuance of the Common Stock subject to such Option has been entered into the books and records of the Company.
(c) No Service Rights. Nothing in the Plan, any instrument executed, or Option granted pursuant thereto shall confer upon any Optionholder any right to continue to serve the Company or an Affiliate as a Non-Employee Director or shall affect the right of the Company or an Affiliate to terminate the service of a Director pursuant to the Bylaws of the Company or an Affiliate, and any applicable provisions of the corporate law of the state in which the Company or the Affiliate is incorporated, as the case may be.
6.
(d) Investment Assurances. The Company may require an Optionholder, as a condition of exercising or acquiring Common Stock under any Option, (i) to give written assurances satisfactory to the Company as to the Optionholder’s knowledge and experience in financial and business matters and/or to employ a purchaser representative reasonably satisfactory to the Company who is knowledgeable and experienced in financial and business matters and that he or she is capable of evaluating, alone or together with the purchaser representative, the merits and risks of exercising the Option; and (ii) to give written assurances satisfactory to the Company stating that the Optionholder is acquiring Common Stock subject to the Option for the Optionholder’s own account and not with any present intention of selling or otherwise distributing the Common Stock. The foregoing requirements, and any assurances given pursuant to such requirements, shall be inoperative if (A) the issuance of the shares upon the exercise or acquisition of Common Stock under the Option has been registered under a then currently effective registration statement under the Securities Act, or (B) as to any particular requirement, a determination is made by counsel for the Company that such requirement need not be met in the circumstances under the then applicable securities laws. The Company may, upon advice of counsel to the Company, place legends on stock certificates issued under the Plan as such counsel deems necessary or appropriate in order to comply with applicable securities laws, including, but not limited to, legends restricting the transfer of the Common Stock.
(e) Withholding Obligations. The Optionholder may satisfy any federal, state or local tax withholding obligation relating to the exercise or acquisition of Common Stock under an Option by any of the following means (in addition to the Company’s right to withhold from any compensation paid to the Optionholder by the Company) or by a combination of such means: (i) tendering a cash payment; (ii) authorizing the Company to withhold shares from the shares of Common Stock issued or otherwise issuable to the Optionholder as a result of the exercise or acquisition of Common Stock under the Option; provided, however , that no shares of Common Stock are withheld with a value exceeding the minimum amount of tax required to be withheld by law (or such lower amount as may be necessary to avoid classification of the Option as a liability for financial accounting purposes); (iii) authorizing the Company to withhold payment from any amounts otherwise payable to the Optionholder; or (iv) by such other method as may be set forth in the Option Agreement.
(f) Electronic Delivery . Any reference herein to a “written” agreement or document shall include any agreement or document delivered electronically or posted on the Company’s intranet.
9. A DJUSTMENTS UPON C HANGES IN C OMMON S TOCK ; O THER C ORPORATE E VENTS .
(a) Capitalization Adjustments . In the event of a Capitalization Adjustment, the Board shall appropriately and proportionately adjust: (i) the class(es) and maximum number of securities subject to the Plan pursuant to Section 3(a), (ii) the class(es) and maximum number of securities by which the share reserve is to increase automatically each year pursuant to Section 3(a), (iii) the class(es) and number of securities for which the nondiscretionary grants of Options are made pursuant to Section 5, and (iv) the class(es) and number of securities and price per share of stock subject to outstanding Options. The Board shall make such adjustments, and its determination shall be final, binding and conclusive.
7.
(b) Dissolution or Liquidation . In the event of a dissolution or liquidation of the Company, all outstanding Options shall terminate immediately prior to the completion of such dissolution or liquidation.
(c) Corporate Transaction. In the event of a Corporate Transaction, then, notwithstanding any other provision of the Plan, the Board shall take one or more of the following actions with respect to Options, contingent upon the closing or completion of the Corporate Transaction:
(i) arrange for the surviving corporation or acquiring corporation (or the surviving or acquiring corporation’s parent company) to assume or continue the Option or to substitute a similar Option for the Option (including, but not limited to, an Option to acquire the same consideration paid to the stockholders of the Company pursuant to the Corporate Transaction);
(ii) arrange for the assignment of any reacquisition or repurchase rights held by the Company in respect of Common Stock issued pursuant to the Option to the surviving corporation or acquiring corporation (or the surviving or acquiring corporation’s parent company);
(iii) accelerate the vesting of the Option (and, if applicable, the time at which the Option may be exercised) to a date prior to the effective time of such Corporate Transaction as the Board shall determine (or, if the Board shall not determine such a date, to the date that is five (5) days prior to the effective date of the Corporate Transaction), with such Option terminating if not exercised (if applicable) at or prior to the effective time of the Corporate Transaction;
(iv) arrange for the lapse of any reacquisition or repurchase rights held by the Company with respect to the Option;
(v) cancel or arrange for the cancellation of the Option, to the extent not vested or not exercised prior to the effective time of the Corporate Transaction, in exchange for such cash consideration, if any, as the Board, in its sole discretion, may consider appropriate; and
(vi) make a payment, in such form as may be determined by the Board equal to the excess, if any, of (A) the value of the property the Optionholder would have received upon the exercise of the Option, over (B) any exercise price payable by such holder in connection with such exercise.
The Board need not take the same action or actions with respect to all Options or portions thereof or with respect to all Optionholders.
(d) Change in Control. A Option may be subject to additional acceleration of vesting and exercisability upon or after a Change in Control as may be provided in the Option Agreement for such Option or as may be provided in any other written agreement between the Company or any Affiliate and the Optionholder, but in the absence of such provision, no such acceleration shall occur.
8.
10. A MENDMENT OF THE P LAN AND O PTIONS .
(a) Amendment of Plan . Subject to the limitations, if any, of applicable law, the Board, at any time and from time to time, may amend the Plan. However, except as provided in Section 9(a) relating to Capitalization Adjustments, no amendment shall be effective unless approved by the stockholders of the Company to the extent stockholder approval is necessary to satisfy applicable law.
(b) Stockholder Approval . The Board, in its sole discretion, may submit any other amendment to the Plan for stockholder approval.
(c) No Impairment of Rights . Rights under any Option granted before amendment of the Plan shall not be impaired by any amendment of the Plan unless (i) the Company requests the consent of the affected Optionholder, and (ii) such Optionholder consents in writing.
(d) Amendment of Options . The Board, at any time and from time to time, may amend the terms of any one or more Options; provided, however, that the rights under any Option shall not be impaired by any such amendment unless (i) the Company requests the consent of the Optionholder, and (ii) the Optionholder consents in writing.
11. T ERMINATION OR S USPENSION OF THE P LAN
(a) Plan Term . The Board may suspend or terminate the Plan at any time. No Options may be granted under the Plan while the Plan is suspended or after it is terminated.
(b) No Impairment of Rights . Suspension or termination of the Plan shall not impair rights and obligations under any Option granted while the Plan is in effect except with the written consent of the Optionholder.
12. E FFECTIVE D ATE OF P LAN .
The Plan shall become effective on the IPO Date, but no Option shall be exercised unless and until the Plan has been approved by the stockholders of the Company, which approval shall be within twelve (12) months before or after the date the Plan is adopted by the Board.
13. C HOICE OF L AW .
The law of the state of California shall govern all questions concerning the construction, validity and interpretation of this Plan, without regard to that state’s conflict of laws rules.
14. D EFINITIONS . As used in the Plan, the following definitions shall apply to the capitalized terms indicated below:
(a) “ Affiliate ” means, at the time of determination, any “parent” or “subsidiary” of the Company as such terms are defined in Rule 405 of the Securities Act. The Board shall have the authority to determine the time or times at which “parent” or “subsidiary” status is determined within the foregoing definition.
9.
(b) “ Annual Chairperson Grant ” means an Option granted annually to the chairperson of the Board who meet the specified criteria pursuant to Section 5(c).
(c) “ Annual Grant ” means an Option granted annually to all Non-Employee Directors who meet the specified criteria pursuant to Section 5(b).
(d) “ Annual Meeting ” means the first annual meeting of the stockholders of the Company held each calendar year at which the Directors are selected.
(e) “ Board ” means the Board of Directors of the Company.
(f) “ Capitalization Adjustment ” means any change that is made in, or other events that occur with respect to, the Common Stock subject to the Plan or subject to any Option after the Effective Date without the receipt of consideration by the Company through merger, consolidation, reorganization, recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, stock split, liquidating dividend, combination of shares, exchange of shares, change in corporate structure or any similar equity restructuring transaction, as that term is used in Statement of Financial Accounting Standards No. 123 (revised). Notwithstanding the foregoing, the conversion of any convertible securities of the Company shall not be treated as a Capitalization Adjustment.
(g) “ Change in Control ” means the occurrence, in a single transaction or in a series of related transactions, of any one or more of the following events:
(i) any Exchange Act Person becomes the Owner, directly or indirectly, of securities of the Company representing more than fifty percent (50%) of the combined voting power of the Company’s then outstanding securities other than by virtue of a merger, consolidation or similar transaction. Notwithstanding the foregoing, a Change in Control shall not be deemed to occur (A) on account of the acquisition of securities of the Company directly from the Company, (B) on account of the acquisition of securities of the Company by an investor, any affiliate thereof or any other Exchange Act Person that acquires the Company’s securities in a transaction or series of related transactions the primary purpose of which is to obtain financing for the Company through the issuance of equity securities, or (C) solely because the level of Ownership held by any Exchange Act Person (the “ Subject Person ”) exceeds the designated percentage threshold of the outstanding voting securities as a result of a repurchase or other acquisition of voting securities by the Company reducing the number of shares outstanding, provided that if a Change in Control would occur (but for the operation of this sentence) as a result of the acquisition of voting securities by the Company, and after such share acquisition, the Subject Person becomes the Owner of any additional voting securities that, assuming the repurchase or other acquisition had not occurred, increases the percentage of the then outstanding voting securities Owned by the Subject Person over the designated percentage threshold, then a Change in Control shall be deemed to occur;
(ii) there is consummated a merger, consolidation or similar transaction involving (directly or indirectly) the Company and, immediately after the consummation of such merger, consolidation or similar transaction, the stockholders of the Company immediately prior thereto do not Own, directly or indirectly, either (A) outstanding voting securities representing
10.
more than fifty percent (50%) of the combined outstanding voting power of the surviving Entity in such merger, consolidation or similar transaction or (B) more than fifty percent (50%) of the combined outstanding voting power of the parent of the surviving Entity in such merger, consolidation or similar transaction, in each case in substantially the same proportions as their Ownership of the outstanding voting securities of the Company immediately prior to such transaction;
(iii) the stockholders of the Company approve or the Board approves a plan of complete dissolution or liquidation of the Company, or a complete dissolution or liquidation of the Company shall otherwise occur, except for a liquidation into a parent corporation;
(iv) there is consummated a sale, lease, exclusive license or other disposition of all or substantially all of the consolidated assets of the Company and its Subsidiaries, other than a sale, lease, license or other disposition of all or substantially all of the consolidated assets of the Company and its Subsidiaries to an Entity, more than fifty percent (50%) of the combined voting power of the voting securities of which are Owned by stockholders of the Company in substantially the same proportions as their Ownership of the outstanding voting securities of the Company immediately prior to such sale, lease, license or other disposition; or
(v) individuals who, on the date the Plan is adopted by the Board, are members of the Board (the “ Incumbent Board ”) cease for any reason to constitute at least a majority of the members of the Board; provided, however , that if the appointment or election (or nomination for election) of any new Board member was approved or recommended by a majority vote of the members of the Incumbent Board then still in office, such new member shall, for purposes of this Plan, be considered as a member of the Incumbent Board.
Notwithstanding the foregoing or any other provision of this Plan, (A) the term Change in Control shall not include a sale of assets, merger or other transaction effected exclusively for the purpose of changing the domicile of the Company, and (B) the definition of Change in Control (or any analogous term) in an individual written agreement between the Company or any Affiliate and the Optionholder shall supersede the foregoing definition with respect to Options subject to such agreement; provided, however , that if no definition of Change in Control or any analogous term is set forth in such an individual written agreement, the foregoing definition shall apply.
In the event that a Change in Control affects any Option that is deferred, then “Change in Control” shall conform to the definition of Change of Control under Section 409A of the Code, as amended, and the Treasury Department or Internal Revenue Service Regulations or Guidance issued thereunder.
(h) “ Code ” means the Internal Revenue Code of 1986, as amended, including any applicable regulations and guidance thereunder.
(i) “ Common Stock ” means the common stock of the Company.
(j) “ Company ” means Trius Therapeutics, Inc., a Delaware corporation.
11.
(k) “ Consultant ” means any person, including an advisor, who is (i) engaged by the Company or an Affiliate to render consulting or advisory services and is compensated for such services, or (ii) serving as a member of the board of directors of an Affiliate and is compensated for such services. However, service solely as a Director, or payment of a fee for such service, shall not cause a Director to be considered a “Consultant” for purposes of the Plan. Notwithstanding the foregoing, a person is treated as a Consultant under this Plan only if a Form S-8 Registration Statement under the Securities Act is available to register either the offer or the sale of the Company’s securities to such person.
(l) “ Continuous Service ” means that the Optionholder’s service with the Company or an Affiliate, whether as an Employee, Director or Consultant, is not interrupted or terminated. A change in the capacity in which the Optionholder renders service to the Company or an Affiliate as an Employee, Consultant or Director or a change in the entity for which the Optionholder renders such service, provided that there is no interruption or termination of the Optionholder’s service with the Company or an Affiliate, shall not terminate an Optionholder’s Continuous Service; provided, however, if the Entity for which an Optionholder is rendering services ceases to qualify as an Affiliate, as determined by the Board, in its sole discretion, such Optionholder’s Continuous Service shall be considered to have terminated on the date such Entity ceases to qualify as an Affiliate. To the extent permitted by law, the Board, in that party’s sole discretion, may determine whether Continuous Service shall be considered interrupted in the case of (i) any leave of absence approved by the Board, including sick leave, military leave or any other personal leave, or (ii) transfers between the Company, an Affiliate, or their successors. Notwithstanding the foregoing, a leave of absence shall be treated as Continuous Service for purposes of vesting in an Option only to such extent as may be provided in the Company’s leave of absence policy, in the written terms of any leave of absence agreement or policy applicable to the Optionholder, or as otherwise required by law.
(m) “ Corporate Transaction ” means the occurrence, in a single transaction or in a series of related transactions, of any one or more of the following events:
(i) the consummation of a sale or other disposition of all or substantially all, as determined by the Board, in its sole discretion, of the consolidated assets of the Company and its Subsidiaries;
(ii) the consummation of a sale or other disposition of at least ninety percent (90%) of the outstanding securities of the Company;
(iii) the consummation of a merger, consolidation or similar transaction following which the Company is not the surviving corporation; or
(iv) the consummation of a merger, consolidation or similar transaction following which the Company is the surviving corporation but the shares of Common Stock outstanding immediately preceding the merger, consolidation or similar transaction are converted or exchanged by virtue of the merger, consolidation or similar transaction into other property, whether in the form of securities, cash or otherwise.
(n) “ Director ” means a member of the Board.
12.
(o) “ Disability ” means, with respect to a Optionholder, the inability of such Optionholder to engage in any substantial gainful activity by reason of any medically determinable physical or mental impairment which can be expected to result in death or which has lasted or can be expected to last for a continuous period of not less than twelve (12) months, as provided in Sections 22(e)(3) and 409A(a)(2)(c)(i) of the Code.
(p) “ Effective Date ” means the effective date of this Plan document, as set forth in Section 12.
(q) “ Employee ” means any person employed by the Company or an Affiliate. However, service solely as a Director, or payment of a fee for such services, shall not cause a Director to be considered an “Employee” for purposes of the Plan.
(r) “ Entity ” means a corporation, partnership, limited liability company or other entity.
(s) “ Exchange Act ” means the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
(t) “ Exchange Act Person ” means any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act), except that “Exchange Act Person” shall not include (i) the Company or any Subsidiary of the Company, (ii) any employee benefit plan of the Company or any Subsidiary of the Company or any trustee or other fiduciary holding securities under an employee benefit plan of the Company or any Subsidiary of the Company, (iii) an underwriter temporarily holding securities pursuant to a registered public offering of such securities, (iv) an Entity Owned, directly or indirectly, by the stockholders of the Company in substantially the same proportions as their Ownership of stock of the Company; or (v) any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act) that, as of the Effective Date, is the Owner, directly or indirectly, of securities of the Company representing more than fifty percent (50%) of the combined voting power of the Company’s then outstanding securities.
(u) “ Fair Market Value ” means, as of any date, the value of the Common Stock determined as follows:
(i) If the Common Stock is listed on any established stock exchange or traded on any established market, the Fair Market Value of a share of Common Stock shall be the closing sales price for such stock as quoted on such exchange or market (or the exchange or market with the greatest volume of trading in the Common Stock) on the date of determination, as reported in a source the Board deems reliable.
(ii) Unless otherwise provided by the Board, if there is no closing sales price for the Common Stock on the date of determination, then the Fair Market Value shall be the closing selling price on the last preceding date for which such quotation exists.
(iii) In the absence of such markets for the Common Stock, the Fair Market Value shall be determined by the Board in good faith and in a manner that complies with Sections 409A and 422 of the Code.
13.
(v) “ Initial Chairperson Grant ” means an Option granted to a Non-Employee Director who meets the specified criteria pursuant to Section 5(c).
(w) “ Initial Grant ” means an Option granted to a Non-Employee Director who meets the specified criteria pursuant to Section 5(a).
(x) “ IPO Date ” means the date of the underwriting agreement between the Company and the underwriter(s) managing the initial public offering of the Common Stock, pursuant to which the Common Stock is priced for the initial public offering.
(y) “ Non-Employee Director ” means a Director who is not an Employee.
(z) “ Nonstatutory Stock Option ” means an Option not intended to qualify as an incentive stock option within the meaning of Section 422 of the Code and the regulations promulgated thereunder.
(aa) “ Officer ” means a person who is an officer of the Company within the meaning of Section 16 of the Exchange Act.
(bb) “ Option ” means a Nonstatutory Stock Option to purchase shares of Common Stock granted pursuant to the Plan.
(cc) “ Option Agreement ” means a written agreement between the Company and an Optionholder evidencing the terms and conditions of an individual Option grant. Each Option Agreement shall be subject to the terms and conditions of the Plan.
(dd) “ Optionholder ” means a person to whom an Option is granted pursuant to the Plan or, if applicable, such other person who holds an outstanding Option.
(ee) “ Own, ” “ Owned, ” “ Owner, ” “ Ownership ” A person or Entity shall be deemed to “Own,” to have “Owned,” to be the “Owner” of, or to have acquired “Ownership” of securities if such person or Entity, directly or indirectly, through any contract, arrangement, understanding, relationship or otherwise, has or shares voting power, which includes the power to vote or to direct the voting, with respect to such securities.
(ff) “ Plan ” means this Trius Therapeutics, Inc. Amended and Restated 2010 Non-Employee Directors’ Stock Option Plan.
(gg) “ Rule 16b-3 ” means Rule 16b-3 promulgated under the Exchange Act or any successor to Rule 16b-3, as in effect from time to time.
(hh) “ Securities Act ” means the Securities Act of 1933, as amended.
(ii) “ Subsidiary ” means, with respect to the Company, (i) any corporation of which more than fifty percent (50%) of the outstanding capital stock having ordinary voting power to elect a majority of the board of directors of such corporation (irrespective of whether, at the time, stock of any other class or classes of such corporation shall have or might have voting power by reason of the happening of any contingency) is at the time, directly or indirectly, Owned by the
14.
Company, and (ii) any partnership, limited liability company or other entity in which the Company has a direct or indirect interest (whether in the form of voting or participation in profits or capital contribution) of more than fifty percent (50%).
--------------------------------------------------------------------------
Trius Therapeutics: The Next Big Small-Cap Biotech Mover?
Article
Jun 4 2013, 07:55 | 49 comments | about: TSRX (Trius Therapeutics, Inc.), includes: ACAD, ARNA, CBST, SRPT
Disclosure: I am long TSRX. (More...)
Normally, I write about biotech companies with near-term catalysts I feel offer traders good long based trade opportunities heading into these catalysts. Every so often, I find a small cap biotech where I feel a speculation investment is truly warranted. This year, a few small cap companies have realized strong and steady stock appreciation long after their respective catalysts events have come and gone.
Trius Therapeutics, Inc. (TSRX) is one such company I strongly believe is a great speculation investment which could see strong stock appreciation after its positive catalyst has come and gone. Also, I feel it's likely the company will be acquired soon based on some factors I will mention in this article, notwithstanding the strong buzz I have been hearing in this regard.
Company Treatment Platform:
Methicillin-resistant Staphyloccus aureus
Strong antibiotics have become of increasing importance as bacteria mutate to become resistant to current treatments. Even though an antibiotic can kill a majority of the bacteria and rid a patient of symptoms, stronger and more resilient bacteria survive. Over time, that bacterium replicates and becomes the majority of that population. To combat a new strain, scientists are continually developing newer and stronger antibiotics.
Methicillin-resistant Staphyloccus aureus (MRSA) is one pathogen that has become a prevalent problem because it is responsible for several difficult to treat infections. MRSA is any strain of Staphylococcus aureus that has developed, through the process of natural selection, resistance to beta-lactam antibiotics, which include penicillin. MRSA is especially troublesome in hospitals, prisons and nursing homes, where patients with open wounds, invasive devices and weakened immune systems are at greater risk of infection than the general public.
MRSA is on the rise, with infections doubling in just the last 5 years. Chicago scientists say the new estimate might even be low-balling the disease's pervasiveness because the database they use tends to under-report instances of MRSA if patients were hospitalized for some other ailment. When the researchers went back to correct for the statistical inaccuracy, they discovered that the insurance claims had missed between one-third to one-half of actual MRSA cases as recorded by the hospitals' own records.
The continual need of a newer and stronger form of antibiotic has sparked several biotechnology companies to develop drugs to combat MRSA, and Trius might be on to one that could be a multi-billion dollar solution.
Oxazolidinones
Trius largely works with a class of compounds called oxazolidinones, which are mainly used as antimicrobials. Oxazolidinones can be used as an antibacterial by working as a protein synthesis inhibitor that can target an early step involving the binding of N-formylmethionyl-tRNA to the ribosome. Some of the most important oxazolidinones are the last generation of antibiotics used against gram-positive pathogens, including super-bugs such as MRSA. These antibiotics are considered as a choice of last resort where every other antibiotic therapy has failed.
Examples of antibiotic oxazolidinones include:
Linezolid - Is available for intravenous administration and also has the advantage of having excellent oral bioavailability.
Posizolid - Appears to have excellent, targeted bactericidal activity against all common gram-positive bacteria, regardless of resistance to other classes of antibiotics.
Tedizolid - Is in phase-II clinical trials.
Radezolid (RX-1741) - Has completed some phase-II clinical trials.
Cycloserine - Is a second line drug against tuberculosis.
The market to treat MRSA with oxazolidinones is a very large one, with some products bringing in billions of dollars in revenue. As strains mutate and science advances, several companies are in the market to capture a share of the profits. Trius is in development with its top product tedizolid, which has the potential to generate huge profits if eventually approved.
Company Pipeline:
Tedizolid
Tedizolid (TR-701), formerly Torezolid phosphate, is an IV and orally administered second-generation oxazolidinone for the treatment of serious gram-positive infections, including methicillin-resistant Staphylococcus aureus (MRSA). As a second-generation oxazolidinone, Tedizolid phosphate is chemically differentiated from the first generation of clinically developed oxazolidinones and is designed for improved potency, resistance, and spectrum of activity. There is only one approved first generation oxazolidinone, linezolid, which is currently the leading branded antibiotic for serious gram-positive infections. Pfizer markets linezolid under the brand name Zyvox and reported worldwide sales of $1.35 billion in 2012.
Currently, vancomycin, daptomycin and linezolid (Zyvox) are the leading drugs used for the treatment of infections caused by gram-positive bacteria, most of which are hospital-acquired infections. Tedizolid has been studied and evaluated for the treatment of serious gram-positive infections, including those caused by MRSA.
Tedilozid is being developed to treat multiple clinical indications including ABSSSI and other important indications involving infections of the lungs and blood, such as commonly acquired bacterial pneumonia (CABP), hospital and ventilator acquired pneumonia (HAP and VAP).
Tedizolid vs. Linezolid
Zyvox (linezolid) treats the same indications as tedizolid and has found a niche treating Vancomycin resistant enterococci (VRE). Additionally, off-label use of linezolid is becoming more popular also. Since it was approved in 2000, linezolid has proven to be extremely successful due to its safety profile and general bacterial resistance. However, positive results from the ESTABLISH-2 Phase III clinical trial shows that tedizolid shows significant advantages over linezolid. The company provides a very accurate and thorough description of the advantages of tedizolid over linezolid:
Greater Potency.
The potency of tedizolid is four to eight times greater than linezolid against linezolid-susceptible strains and up to 16 times greater than linezolid against linezolid-resistant strains. The greater potency of tedizolid phosphate should enable a shorter course of treatment as compared to linezolid. We believe that this enhanced potency may result in improved clinical outcomes, significant savings for hospitals and payor organizations, faster eradication of the pathogen and earlier discharge from the hospital.
Shorter Dosing Regimen and More Convenient, Once Daily Dosing.
Tedizolid Phosphate is administered once daily for six days for the treatment of cSSSI (now termed ABSSSI), as compared to twice daily for 10 to 14 days for linezolid. We believe this shorter and once daily dosing regimen will contribute to improved patient compliance, decrease the risk of drug induced adverse events and limit the emergence of resistance.
Bactericidal Activity In Vivo.
Tedizolid Phosphate (TR-701), unlike linezolid, concentrates to a high extent inside certain white blood cells, which engulf pathogenic bacteria and concentrate at the site of infection. This feature of Tedizolid phosphate (TR-701) contributes to its in vivo bactericidal activity, or killing of pathogenic bacteria in the body, which is thought to yield a higher degree of efficacy and faster eradication of the pathogenic bacteria than is achieved with bacteriostatic antibiotics, which are antibiotics that arrest the growth of bacteria.
Activity Against Key Gram-Positive Drug-Resistant Strains
and Select Atypical and Gram-Negative Bacteria.
Tedizolid phosphate is active against all clinically relevant gram-positive bacteria tested to date, including organisms resistant to linezolid and other antibiotics. Tedizolid phosphate is also active against strains of the gram-negative bacterium Legionella and strains of the atypical bacterium Chlamydia, and thus may have utility in treating lower respiratory tract infections involving these bacteria.
Low Intrinsic Frequency of Resistance.
The frequency at which MRSA evolves resistance to Tedizolid phosphate is 16 times lower than the frequency at which it evolves resistance against linezolid. We believe that this may enable wider use of Tedizolid phosphate and limit the emergence of resistance.
Favorable and Predictable Pharmacokinetics
There is little patient-to-patient variability in the concentration of tedizolid phosphate in blood, as compared to linezolid. As a result, we expect that tedizolid phosphate will have more predictable drug exposure which may lead to a more uniform efficacy and safety profile across different patients when compared to linezolid.
Fewer Drug-Drug Interactions.
Unlike linezolid, tedizolid phosphate has not been shown to inhibit the monoamine oxidase system which mediates the metabolism of tyramine, SSRI's and vasoconstrictors.
Improved Safety Profile for Longer Term Dosing.
The results of our comparative 21-day Phase 1 clinical trial show that a 200 mg daily dose of tedizolid phosphate had less impact on hematological parameters indicative of myelosuppression than the labeled dose of Zyvox (600 mg twice daily). Based upon the results of this clinical trial, we believe that Tedizolid phosphate may offer a safer alternative to linezolid for infections requiring longer term dosing, such as bacteremia.
With these advantages, doctors should be more inclined to prescribe tedizolid over linezolid. Since Zyvox (linezolid) brings in over $1 billion in revenue each year, it is clear that developing a better drug would be extremely profitable. With evidence building that some strains of bacteria are becoming resistant to Zyvox, there is always room for a newer and more effective antibiotic. As a newly developed antibiotic, tedizolid may be a better pick by doctors over its competitor as bacterial resistance becomes even more prevalent.
From an economic standpoint, tedizolid provides additional advantages over linezolid. Given that it costs about $115 per pill and a 10-day course consists of taking a 600mg pill taken twice daily, linezolid would cost about $2,300. Contrastingly, tedizolid only has to be taken once a day for a 6-day course. This conservatively puts tedizolid at $700 for one course. The cost difference is a huge advantage over current treatments and given similar drug profiles, tedizolid seems to be the better choice.
If eventually approved by the Federal Drug Administration (FDA), analysts have speculated that tedizolid could reach peak sales of up to $900 million. With a current market cap of only $367 million, Trius Therapeutics is grossly undervalued. With positive Phase III data in hand, I expect shares to increase in value as investors begin to realize the potential of this company.
We have seen a few small-cap biotechs with potential blockbuster treatments move a lot higher and attract heavy institutional support this year after their positive catalyst events have occurred.
Sarepta Therapeutics (SRPT) traded under $3 a year ago as it worked through a Phase II trial for eteplirsen, indicated for the treatment of Duchenne Muscular Dystrophy (([DMD)). Since the company reported promising data late last year for its drug eteplirsen, its stock price has been on fire. In one trading session alone, the stock rallied from around $14 a share to a parabolic price of $45.
Sarepta now trades at over $35 a share ahead of the company expecting to hear from the FDA on accelerated approval for eteplirsen soon. I believe the FDA will grant accelerated approval for many reasons, one being strong political pressure from advocates of the drug like Jenn McNary, mother of a child who suffers from DMD.
Arena Pharma (ARNA), after Belviq received a positive FDA advisory committee recommendation last year, rallied to over $10 a share, before falling back to under $6. At that time, the company engaged in a public offering at $5.50 a share, and the stock rallied back to over $10 before receiving marketing approval from the FDA. Afterwards, the stock sold off a bit, but has rebounded to near $9.
Belviq is intended for weight management, including weight loss and maintenance of weight loss. There are currently limited pharmaceutical treatment options to help patients lose weight, so the drug is seen as somewhat of an unmet need. Belviq became the first FDA drug approved to treat obesity in over a decade. Arena's public offering was a big success as the company attracted a lot of institutional interest then and in the last year. I believe Arena will be a full fledged successful pharma -- in 5 years or so.
Acadia (ACAD) began 2013 trading around $4.60 a share. Just in the last few months, the stock has nearly tripled on both positive data for its lead drug pimavanserin, indicated for the treatment of Parkinson's disease psychosis (PDP), and allowance of an expediated New Drug Application (NDA) filing from the FDA.
PDP is an unmet need with a potentially huge market, and the company deserves its current speculation valuation. ACADIA is another company I strongly feel will likely be acquired in the next year or so.
I feel once institutions like Baker Brothers LLC (who have a substantial stake in ACADIA) fully study Trius and its enormous speculation value, I am confident they, along with other institutions will take a substantial stake in the company, which should further drive Trius stock considerably higher.
Buyout Speculation:
With studies showing a better safety profile and patient compliance than current drugs on the market, Tedizolid is primed to not only get approved by the FDA, but also potentially grab a majority market share. However, Trius may never get to bring its drugs to market because there is a solid chance big pharmas are interested in acquiring its assets. Recently, investors have speculated that Trius will be bought out because the company just announced a notice of allowance of U.S. patent application related to tedizolid phosphate combination with daptomycin.
Daptomycin is marketed as Cubicin by Cubist Pharmaceuticals (CBST) and should pull in near $1 billion in 2013. Cubist has turned Cubicin into a blockbuster antibiotic and its continued revenue stream is integral to the company's success. The allowance of this patent makes Trius an even more prime target for a buyout for the several following reasons.
Reduce Resistant Bacteria
Adaptation and evolution are two of the most troubling problems when it comes to developing antibiotics. For example, if scientists are able to develop a drug that kills 99% of bacteria, that 1% will survive and become the genetically most prevalent form. How long this takes depends on several factors including how rapidly the bacteria can adapt and replicate. In order to kill stronger or different strains of bacteria, scientists are always creating newer and more powerful drugs.
The prevalence of these mutant strains has become an increasing problem for daptomycin. Although daptomycin has been a successful drug in treating patients in the gram-positive bacteria market, it is slowly losing its effectiveness as the bacteria are becoming more resistant.
Dr. Ralph Corey, Professor of Medicine and Infectious Diseases at Duke University, stated:
MRSA bacteremia is a potentially fatal disease with limited treatment options. As a clinician who treats bacteremia patients, I am concerned by the growing resistance to one of our last effective treatment options, daptomycin, and welcome this news that tedizolid phosphate may prevent daptomycin resistance when used in a combination therapy.
Trius found that when a low dose of tedizolid phosphate is combined with daptomycin, the amount of bacteria resistant to daptomycin is significantly reduced. This is significant because it would increase the efficacy of daptomycin and extends its life as an antibiotic. In the same press release, it stated:
As detailed in the patent application, tedizolid phosphate, at concentrations substantially below its current therapeutic dose to treat skin infections, unexpectedly prevents the formation of daptomycin resistant mutants of Staphylococcus aureus. Tedizolid appears to be unique in this mutant prevention activity given that seven other medicines tested, including linezolid, the only other drug of the same class as tedizolid, failed to show the same mutant prevention activity.
The allowance of this patent is of extreme importance to both Trius and Cubist. From a scientific standpoint, Cubist would be interested in the combination of tedizolid and daptomycin since the combination of the two would solve the problem daptomycin has with resistant bacteria. This would extend the effectiveness of daptomycin and keep that revenue stream steady for Cubist. Also, as previously mentioned, analysts have speculated that tedizolid could reach peak sales of up to $900 million - how much more in sales can be realized with the combo tedizolid phosphate/daptomycin that was just issued a patent allowance? With a market cap of $360M, Trius is ridiculously undervalued and under speculated in my strongest opinion.
Extend Exclusivity
The allowance of this patent could also provide Cubist with an opportunity to extend the exclusivity period of Cubicin. Until 2018, no company can create any generic forms of Cubicin and Cubist has been able to bring in large revenues because of that. After 2018, generic drug companies could create a cheaper alternative and grab some market share away from Cubicin. Obviously, it would be in the best interest of Cubist to extend the period of exclusivity for as long as they can do so, notwithstanding the potential for potentially significant additional revenue from tedizolid phosphate combination with daptomycin.
The combination of tedizolid and daptomycin would be a way for Cubist to extend and realize revenue from a patent exclusivity period by an additional five years. With guidance this year of near $1 billion in revenues for Cubicin, exclusivity from a more effective combo solution could easily realize $5 to $8 billion in revenues before any generic potentially comes to market. Long-term Cubist shareholders should encourage and support a buyout or partnership with Trius because it should provide a huge revenue stream for several additional years.
Broaden Profile
With a buyout, Cubist would gain the rights to tedizolid, along with the newly patented combo drug, plus the other drugs in the Trius pipeline. As stated previously, Trius reported positive Phase III data for tedzolid, meeting all primary endpoints when compared to Pfizer's Zyvox. However, tedizolid also appeared to have a better safety profile and dosing schedule. Cubist also could complete Trius' scheduled Phase III program for the treatment of pneumonia in the second half of 2013. In the pipeline, Trius also has an array of products ranging from preclinical to Phase III trials that would add additional value to Cubist.
Additionally, in the latest 8k from Trius, we read the following:
Item 5.02 Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers.
On May 21, 2013, our 2010 Equity Incentive Plan (the "2010 Plan") was amended to, among other things, increase the aggregate number of shares of common stock authorized for issuance under the 2010 Plan by 5,100,000 shares.
I saw similar activity with Obagi Medical Products before it was acquired recently. When considering the fundamental reasons why Cubist might acquire Trius, it makes sense that management would want "extra shares" to be awarded themselves upon an acquisition. This is an indicator to me that a buyout may be in short order for the company.
Much has been made lately about a few insider sells last month. I would point out that these sells were "automatic sales" under rule 10b5-1, which in the case of Trius insiders, they were on schedule to sell the shares regardless of the price action or any other factor. I'm always a little surprised just how many traders and investors are unaware of what automatic sales are.
Additional leverage from the Gain Act:
Realizing the ever-growing problem of treatment resistant infections like MRSA, in October 2012, Congress passed and President Obama signed into law the GAIN Act legislation (Generating Antibiotics Incentives Now Act).
The legislation encourages companies like Trius to develop new antibiotics, allowing them to streamline certain regulatory processes for commercialization. Perhaps the most important aspect of the GAIN Act for Trius is that it offers companies 5 years of market exclusively, which would give Trius additional leverage if it chooses to partner tedizolid, (and even more leverage with its newly patented combo drug) and/or sell the company.
Financial and Share Structure
Balance Sheet
Total Cash (mrq):
83.65M
Total Cash Per Share (mrq):
1.75
Total Debt (mrq):
0.00
Total Debt/Equity (mrq):
N/A
Current Ratio (mrq):
5.05
Book Value Per Share (mrq):
1.51
Trius is burning roughly $12.5M a quarter, with roughly $84M in cash. The company has nearly 2 years of cash reserve to fund operations.
Share Statistics
Avg Vol (3 month):
899,153
Avg Vol (10 day):
848,043
Shares Outstanding:
*47.87M
Float:
37.23M
% Held by Insiders:
29.99%
% Held by Institutions:
37.10%
Shares Short (as of May 15, 2013):
2.33M
Short Ratio (as of May 15, 2013):
2.80
Short % of Float (as of May 15, 2013):
6.20%
*52.97M shares potentially outstanding assumed when factoring in management's recent 8K to add 5.1M shares to the authorized shares for compensation under its equity incentive plan (assuming what I speculate to be a clear sign the company will be acquired soon).
Above, we see good insider ownership, and plenty of room for new institutions to come aboard. I also like the decent size short interest here. Too much short interest sometimes is indicative of possible negative issues with a company, but too little short interest usually means it takes a ton of actual investors to significantly move a stock price upwards.
Conclusion:
Trius is a small-cap biotech with a developing potentially huge story, with a transitional market cap of $360M. According to recent media reports, Cubist reportedly made an unsolicited $1 billion bid for another company in the segment, that was not accepted. With the timing of the patent grant along with Trius upping its authorized share count for the purpose of incentive rewards, it's likely in my opinion that the company will be acquired soon by Cubist for at least double its current price.
As mentioned above, we have seen a few small-cap biotechs lately move a lot higher and attract heavy institutional support after their positive catalyst events have occurred -- Acadia, Sarepta and Arena are but a few examples. I am confident Trius will do the same and move into the double digit stock price range very soon. At the very least, Cubist could partner with Trius, and any such deal would be a lucrative one for Trius. However, Cubist would be better served buying Trius straight out for the reasons I have mentioned here in this article.
Additionally, there has been a lot of news about multi-drug-resistant organisms (MDROs), such as MRSA, vancomycin-resistant enterococci (VRE) and Carbapenem-resistant enterobacteriaceae (CRC).
NBC ran a story in February this year which stated that there has been a sharp jump in the number of rare but potentially deadly types of super-bug bacterias resistant to nearly all last-resort antibiotics. As mentioned, Congress took action passing The Gain Act, to incentivize companies to develop new treatments to combat this problem, as new strains become more resistant to current treatments on the market.
Considering all these factors, my one-year price target opinion for Trius is $20 a share, which would roughly equate to a $1B market cap - or what was reportedly offered by Cubist for another company in the segment. As already stated, I believe the stock will cross over the $10 mark soon.
I believe the 8K speaks for itself.
[color=red]Res Ipsa Loquitur[/color
Take Care and God Bless....God Bless Us All.
Truth4Once
Total BS.The OS of the company is around 48 Mill and directors got 50 Million shares.? ..ROFLMAO
From the Yahoo message board:
annieg007 • Aug 1, 2013 9:59 PM
I'm with you Chim, I think the Feds are really gonna look at these Directors, something about 50 million shares as compensation immediately prior to Selling Shareholders Up the River, not to include any backside secrets by Cubist.
Seems the market already feels TSRX is work more than $13.50 ;)
Come on now Cubist, make us a fair offer!
$TSRX
I'd hold to see how this entire situation plays out.
Give me the cash equivalent of the voucher now.
Either Cubist believes in Trius or not;enough with the balancing act,I say.
Life is unpredictable....eg:my mother reached retirement age,filed for social security and passed away before she could collect any benefits.
There's truth to the saying that the only absolutes are death and taxes.
Of course,any decision you make should be based upon your specific financial situation and goals going forward.
Take Care and God Bless....God Bless Us All.
Truth4Once
Trius Patent Intrigues Investors, And Perhaps Cubist Pharmaceuticals
May 16 2013, 09:57 | 7 comments | about: TSRX (Trius Therapeutics, Inc.), includes: CBST
Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. (More...)
By Jason Napodano, CFA
On May 13, 2013, Trius Therapeutics (TSRX) announced that it had received a notice of allowance from the USPTO for a patent application relating to the use of its tedizolid phosphate antibiotic in combination with Cubist Pharmaceuticals' (CBST) antibiotic Cubicin (daptomycin). The application claims the use of sub-therapeutic doses of tedizolid for preventing bacterial resistance development when daptomycin is used long term (we assume over 28 days). In this article, we explore the potential utility of this combination, project what the clinical development and regulatory path might look like, and hypothesize the extent to which this new intellectual property will lead to genuine commercial exclusivity. We conclude with a look at what the commercial strategy for such a combination might look like, and provide an estimate of the potential value of this combination to Trius. TSRX is up 30% since PropThink called weakness in the stock "undue" just two weeks ago.
The Increasing Need for Alternatives to Vancomycin
Vancomycin, daptomycin, and linezolid (Zyvox) are the leading drugs for the treatment of infections caused by gram-(+) bacteria, especially those acquired in the hospital. The most common and virulent of these gram-(+) organisms is Staphylococcus aureus. Variants that have become resistant to the antibiotic methicillin are known as methicillin-resistant Staphylococcus aureus, or MRSA. This name understates the danger of these strains, as most MRSA are highly resistant to all but a handful of antibiotics. MRSA and other gram-(+) pathogens are particularly common causes of acute bacterial skin and skin structure infections (ABSSSI), pneumonia, septicemia (bloodstream infections), osteomyelitis (bone and joint infections), and endocarditis (infections of the heart). Generic vancomycin is the most commonly used antibacterial agent for hospital acquired gram-(+) infections, but rising resistance rates are eroding its share of total treatment days.
Source: Cubist Pharmaceuticals.
Tedizolid to Fill Much Needed Void
Trius' tedizolid recently completed two Phase III clinical trials, ESTABLISH-1 and ESTABLISH-2, for the treatment of acute bacterial skin and skin structure infections (ABSSSI), and is in Phase I for the treatment of hospital acquired pneumonia. We predict it will become the second antibiotic of the oxazolidinone structural class to reach the market, sometime around the middle of 2014. The prototype of this class, Pfizer's (PFE) Zyvox (linezolid), was approved in April 2000, and achieved $1.35 billion in worldwide sales in 2012. Zyvox is approved for the treatment of gram-(+) infections, including hospital and community acquired pneumonia (HAP and CAP), skin and skin structure infections, and any infection caused by vancomycin resistant enterococci (VRE). Its commercial success has arisen in large part due to its excellent activity against a broad spectrum of gram-(+) pathogens (including MRSA), its high barrier to resistance development, and its availability in both oral and IV formulations.
That being said, we believe tedizolid exhibits several advantages over Zyvox, including a bactericidal mechanism versus bacteriostatic, once daily versus twice daily dosing, and a shorter course of therapy at only six days vs. the 10 to 14 days recommended with Zyvox. Another key potential driver of tedizolid is that the drug demonstrates activity against most linezolid-resistant strains. Finally, tedizolid lacks monoamine oxidase activity (an important source of drug-drug and drug food interactions for Zyvox) and apparently has reduced propensity to cause myelosuppression, a side effect that has limited the use of Zyvox for applications requiring long-term therapy.
Cubicin Is Widely Used for Infections Requiring Long-Term Therapy
Cubicin (daptomycin) is a lipopeptide antibiotic used in the treatment of serious infections caused by gram-(+) positive infections, especially SSTIs, bacteremia, endocarditis, and osteomyelitis caused by methicillin-resistant Staphylococcus aureus (MRSA). It is available as an IV formulation only, but is used in preference to Zyvox in many situations because it is bactericidal rather than bacteriostatic, and because of Zyvox's tendency to cause myelosupression as noted above.
Cubicin worldwide revenues totaled $860 million in 2012. Data from the European Cubicin Outcomes Registry and Experience (EUCORE) dataset shows that the major use of Cubicin is in SSTI, accounting for 44% of treatment days, of which 33% were complex SSTIs. Bacteremia followed at 22%, followed by endocarditis at 12% and osteomyelitis at 6%. Standard treatment of osteomyelitis involves at least four weeks treatment. For example, data from the daptomycin registry concluded that the average duration of treatment of osteomyelitis was 35 days and 37 days for infective endocarditis. Treatment for endocarditis is similar.
A U.S. study of patients treated with daptomycin for more than 14 days found that ABSSSI were slightly more common among this group than osteomyelitis, so we assume that about 6% of Cubicin treatment days correspond to long-term treatment for ABSSSI. Thus it appears that at least 24% of Cubicin treatment days are as part of a long-term therapeutic regimen. These treatment days corresponded to at least $206 million in revenue in 2012.
Subtherapeutic Administration of Tedizolid May Prevent Daptomycin Resistance
U.S. Patent Application 13/655,704, "Therapeutic Combination of Daptomycin and Protein Synthesis Inhibitor Antibiotic, and Methods of Use" describes the use of subtherapeutic doses of tedizolid to inhibit resistance development in bacteria treated with therapeutic concentrations of daptomycin. Tedizolid appears to be unique in this mutant prevention activity given that seven other antibiotics tested, including the oxazolidinone linezolid, failed to show the same mutant preventing activity. As described in the application, an MRSA strain was repeatedly cultured in the presence of daptomycin, with the most resistant bacteria used each time to start the next culture. After 30 repetitions, the bacteria were able to grow in the presence of 32 ug/mL daptomycin, compared to only 0.25 ug/mL at the beginning of the experiment.
Similar results were obtained when the culture medium contained both daptomycin and a subtherapeutic concentration of vancomycin, clindamycin, tetracycline, or tiamulin. The best results were obtained with tedizolid, which at one quarter the therapeutic concentration, prevented the minimum inhibitory concentration from rising above 2 ug/mL. A second Trius oxazolidinone and a two drug combination of trimethoprim/sulfamethoxazole gave the next best results.
Regulatory and Commercial Strategy
These findings and the notice of allowance from the USPTO provide Trius with 20 years market exclusivity for combinations of daptomycin with subtherapeutic amounts of tedizolid. From a development and regulatory point of view, this combination is very similar to the combination of a beta lactam antibiotic plus a beta lactamase inhibitor, such as ceftazidime/avibactam. Phase III trials of these combinations are designed like any other antibiotic trial, with demonstration of non-inferiority to a currently accepted standard of care as the primary endpoint. Demonstration of superiority to monotherapy is not required. While a clinical demonstration of superiority of daptomycin/tedizolid to daptomycin monotherapy would be useful, it is likely to be difficult to achieve in practice. We believe that most physicians faced with a patient who will receive extended daptomycin therapy would likely be attracted based on a demonstration of clinical efficacy plus an improved in vitro resistance profile.
The commercial strategy will likely reflect the expiration of Cubist's patent exclusivity in 2017 (composition of matter) or 2019 (dosing), which is about the earliest that a daptomycin/tedizolid combination product could reach the market. The most likely strategy for Trius would be to develop an IV co-formulation of Cubicin with tedizolid in the appropriate proportions (perhaps a once daily dose around 75 to 100 mg). If approved, we believe the U.S. FDA would recognize daptomycin/tedizolid as a "New Combination," and thus provide market exclusivity to Trius.
While this product would likely face some competition from "home brew" combinations of tedizolid with generic daptomycin, we believe the threat of triple damages for willful patent infringement and the extensive paper trail created by the reimbursement process will tend to minimize this risk. Assuming that vancomycin market share continues to fall due to rising resistance and equal market shares for generic daptomycin and daptomycin tedizolid combination therapy among patients requiring long-term treatment, we estimate that sales could easily run in the $350 million per year range.
Conclusion
We have been and continue to be big fans of Trius Therapeutics and tedizolid. We believe the commercial opportunity for Trius' tedizolid monotherapy is significant, at roughly $750 million worldwide. We see the story as mostly de-risked, given the positive and consistent results from the two Phase III studies in skin infections. We believe tedizolid can gain meaningful market share after commercialization based on superior dosing, safety, tolerability, and potentially improved efficacy in a growing number of vancomycin and linezolid-resistant strains. Upside comes from adding bacteremia and pneumonia to the label.
Trius owns unencumbered U.S. and EU rights to tedizolid. The company has partnered with Bayer Healthcare for the commercialization of tedizolid in the Asia/Pacific market. We believe the company is seeking a European commercial partner and in talks to close a deal hopefully soon. As of now, we suspect that Trius would like to keep U.S. rights to tedizolid themselves, and follow the model that Cubist has successfully developed with Cubicin. Keep in mind though that Cubicin will lose exclusivity in 2017. However, the news that Trius has now obtained a patent that could extend the market exclusivity of daptomycin beyond 2017 certainly should raise eyeballs from Cubist management. We believe investors should pay up to $12 per share for Trius -- Cubist could pay a whole lot more. See PropThink's latest report on Cubist, released early this week, here.
Additional disclosure: PropThink is a team of editors, analysts, and writers. This article was written by Jason Napodano, CFA. We did not receive compensation for this article, and we have no business relationship with any company whose stock is mentioned in this article. Use of PropThink’s research is at your own risk. You should do your own research and due diligence before making any investment decision with respect to securities covered herein. You should assume that as of the publication date of any report or letter, PropThink, LLC and persons or entities with whom it has relationships (collectively referred to as "PropThink") has a position in all stocks (and/or options of the stock) covered herein that is consistent with the position set forth in our research report. Following publication of any report or letter, PropThink intends to continue transacting in the securities covered herein, and we may be long, short, or neutral at any time hereafter regardless of our initial recommendation. To the best of our knowledge and belief, all information contained herein is accurate and reliable, and has been obtained from public sources we believe to be accurate and reliable, and not from company insiders or persons who have a relationship with company insiders. Our full disclaimer is available at www.propthink.com/disclaimer.
-------------------------------------------------------------------------
Cubist could pay a whole lot more.
4 Top Small Cap Biotech Acquisition Targets- (Trius Excerpt Only)
May 31 2013, 08:00 | 12 comments by: StockMatusow | includes: ACAD, ACRX, ATRS, CBST, TSRX
Disclosure: I am long ATRS, TSRX. (More...)
Today, I list small cap companies I believe are top acquisition targets for large pharma. I combine speculation, rumors and buzz I am hearing, and company financial position as a few factors for consideration. Buy-outs are hard to predict in general, and in many ways, holding stock in a company that receives a tender offer is similar to holding a winning lottery ticket. The companies mentioned in this article are worthy speculation investments, regardless of whether one or all of them are acquired.
Trius Therapeutics (TSRX) is a biopharmaceutical company focused on the discovery, development, and commercialization of innovative antibiotics for serious infections. Trius was one of my catalyst trade picks in March and I believe that it is time to revisit this company. Trius just announced a notice of allowance of U.S. patent application related to tedizolid phosphate combination with daptomycin. When a low dose of tedizolid phosphate is combined with daptomycin, the amount of bacteria resistant to daptomycin is significantly reduced. In the company's press release it stated:
As detailed in the patent application, tedizolid phosphate, at concentrations substantially below its current therapeutic dose to treat skin infections, unexpectedly prevents the formation of daptomycin resistant mutants of Staphylococcus aureus. Tedizolid appears to be unique in this mutant prevention activity given that seven other medicines tested, including linezolid, the only other drug of the same class as tedizolid, failed to show the same mutant prevention activity.
The company is also expecting to file an NDA in the second half of 2013 for oral torezolid phosphate for acute bacterial skin and skin structure infections. Tedizolid is on track for approval in mid-2014. MSRA has become a hot topic serious issue as of late, so we expect TSRX to be a big runner in the months ahead.
In addition, it has been speculated that Trius is a buyout candidate. As detailed in an article by Prop Think's Jason Napodano, Cubist Pharmaceuticals (CBST) might be interested in acquiring Trius because it would solve it's problem with daptomycin resistance and also extend the market exclusivity of daptomycin to beyond 2017. I agree with Jason when he says, "We believe investors should pay up to $12 per share for Trius -- Cubist could pay a whole lot more." With sales of daptomycin expected to reach about $1 Billion for Cubist Pharmaceuticals in 2013, it would make sense that they would want to extend their exclusivity period and increase its efficacy.
Trius could represent an opportunity for investors to capitalize on a potential buyout or partnership deal.
Buy out price target opinion: $15-$17
Take Care and God Bless....God Bless Us All.
Truth4Once
|
Followers
|
16
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
909
|
|
Created
|
09/13/10
|
Type
|
Free
|
| Moderators | |||

http://www.triusrx.com/
http://finance.yahoo.com/q/ks?s=TSRX+Key+Statistics
Trius Therapeutics, Inc., a biopharmaceutical company, focuses on the discovery, development, and commercialization of antibiotics for serious, life-threatening infections in the United States. Its products under development include Torezolid Phosphate, which has completed phase 2 clinical trials for the treatment of serious gram-positive bacterial infections, including those caused by methicillin-resistant Staphylococcus aureus. The company is also developing antibiotics for gram-negative infections using its proprietary discovery platform under a contract funded by the National Institutes of Health. It has a cooperative research and development agreement with Lawrence Livermore National Security LLC for the research and development of gram-negative biodefense pathogens; and a license agreement with Dong-A Pharmaceutical Co., Ltd. to develop and sell Torezolid Phosphate outside of Korea. The company was formerly known as Rx3 Pharmaceuticals, Inc. and changed its name to Trius Therapeutics, Inc. in February 2007. Trius Therapeutics, Inc. was founded in 2004 and is headquartered in San Diego, California.
TSRX SHARE STRUCTURE INFORMATION CURRENT
Shares Outstanding:
*47.87M
Float:
37.23M
% Held by Insiders:
~29.00%
% Held by Institutions:
~69.00%
WHY TRIUS? THIS IS WHY:
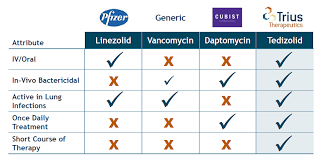
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
