Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$PGLC / $T.PGLC Pershing Gold outlines how it will build value
ICNB is the biggest scam to hit IHUB since Sponge Tech. It is our duty to destroy ICNB and bring justice to the remaining shareholders.
ICNB
BUYER BEWARE
$MCGI Nurses Lounge Announces They Are On Target to Reach Their Goal of 10,000 Job Postings By End of Second Quarter MCGI
http://www.medcareersgroup.com/
$RGNP Security Details
Share Structure
Market Value1 $7,009,529 a/o Mar 17, 2017
Authorized Shares 150,000,000 a/o Mar 01, 2017
Outstanding Shares 43,809,554 a/o Mar 01, 2017
-Restricted Not Available
-Unrestricted Not Available
Held at DTC Not Available
Float 4,085,000 a/o Jan 20, 2017
Par Value Not Available
$AFOM upgraded from Pink to OTCQB!
https://www.otcmarkets.com/stock/AFOM/quote
Giggles N' Hugs $GIGL, a multiple award winning unique restaurant franchise, that has received over 300 franchising offers so far in the US and globally. With revenues of almost $1 million per quarter, growing quarter over quarter, year over year, a tiny float of 10 million shares, featured in major news publications like The Wall Street Journal and an amazing star studded Hollywood customer base.
Giggles N' Hugs (GIGL) HIGHLIGHTS:
- Revenues of almost $1 million per quarter growing at double digit rates.
- Assets of over $1.3 million.
- Over 300 franchising offers from as far away as Australia, Germany, England, Dubai, Russia, Singapore and many others. Giggles N’ Hugs (GIGL) are about to launch their massive franchising program. As company founder Joey Parsi recently stated "We're in a very enviable position in the restaurant world."
- IN DEMAND: The U.S.’s top three mall operators (Westfield Group, Simon Properties, and General Growth Properties) so badly want Giggles N’ Hugs (GIGL) as tenants in their malls across the country they are offering to pay the majority of Giggles N’ Hugs (GIGL) per restaurant construction cost; this incentive ensures a higher ROI and quicker payback period. They know based upon the success of the current operations that Giggles N’ Hugs (GIGL) will bring huge foot traffic to their malls benefiting everyone.
- Giggles N’ Hugs (GIGL) is completely undervalued, respected analyst company Redchip applied a 10.5x EV/EBITDA multiple to the 4Q16E run rate EBITDA projection of $764,735 ($3.06 million over a full-year). Dividing this by the 4Q16E shares outstanding projection of 46.5 million gives a target price of $0.70. This is based upon current numbers, with the coming continued expansion across the nation and globally this share price is going to rocket into the dollars (trading now in the sub-penny range you can see how ridiculously undervalued this stock is, now is the time to buy!)
-Brand recognition: Giggles N’ Hugs (GIGL) has been featured in numerous popular media outlets, attesting to the company's growing brand recognition. This press coverage includes New York Post, People, Forbes Magazine, Wall Street Journal, LA Times, UK Daily Mail, London Times, Australia Heral Sun Bloomberg Business Week, examiner.com, Entrepreneur, The Herald Sun, OK!, US Weekly and more.
-The vision for Giggles N’ Hugs (GIGL) far surpasses just a restaurant rollout. Management has also set its sights on branded merchandising. The Giggles N’ Hugs (GIGL) brand will include merchandise such as food products, high-quality children’s apparel, drink ware, coloring books, and children’s vitamin water. Management plans to sell the merchandise in-store as well as at top-tier retail outlets such as Nordstrom, Bloomingdales, and Whole Foods.
An example of this could be Giggles N’ Hugs (GIGL) kids clothing line, which is slated to include branded kids shirts, hats, pants, bibs, and other apparel.
Many of the Company’s celebrity clientele are frequently photographed, and any pictures of their children wearing Giggles N’ Hugs (GIGL) apparel could provide powerful and free marketing for Giggles N’ Hugs. Typically, top celebrities get paid at least tens of thousands of dollars to wear a particular brand’s clothing.
-Giggles N’ Hugs (GIGL) is structured to benefit from multiple revenue drivers;
Admission fee for play area access
Food and beverages
Birthday parties and private rentals
Child drop-off service
Beer and wine
Branded in-store and retail outlet merchandising via licensing and merchandising model
U.S. and international franchising
- star-studded list of customers and their children including Sandra Bullock, Heidi Klum, Jessica Alba, Halle Berry, Jennifer Garner and Ben Affleck, Denis Quaid, Mark Whalberg, Adam Sandler and Dustin Hoffman.
- Giggles N’ Hugs (GIGL) was rated among the best family and kid-friendly restaurants by CitySearch, GoCityKids, and Nickelodeon, Giggles N' Hugs (GIGL) has been voted the No. 1 family restaurant, No. 1 birthday party place, and the No. 1 indoor play space in all of Los Angeles.
- Giggles N’ Hugs (GIGL) has an O/S count of 44.1 million and a tiny float of just 10 million shares. (This means the share price could rise very quickly with such a small amount of shares available )
- Giggles N’ Hugs (GIGL) has a very powerful management team, just look at these amazing people and what they bring to Giggles N’ Hugs (GIGL):
Joey Parsi, Founder & Director
Joey Parsi had a long and successful career as an investment manager and entrepreneur. He had a 20+ year career working on Wall Street for Lehman Brothers, Sutro and Company, and Prudential Securities. Parsi previously served as an investment advisor for Lehman Brothers; senior vice president at Sutro and Company, where he managed and oversaw millions of dollars for individual and institutional investors specializing in IPOs and technology equities; and worked at Prudential Securities where he oversaw client assets in a number of investments.
In 1998, Parsi opened Barron Chase and was able to expand the company to more than 30 employees. In total, Parsi and his team raised more than $30 million in funding for nine separate companies, many of which are now publicly traded on the NASDAQ markets. After liquidating the business in 2001 he joined TD Waterhouse, where he personally managed more than $350 million in assets for clients, and oversaw more than $1 billion in assets in his region. Parsi then went on to found Giggles N’ Hugs (GIGL).
Giggles N' Hugs President John Kaufman was the COO at California Pizza Kitchen when the founders had just two locations. Joined by Giggles N' Hugs' CFO Phillip Gay, who at the time was CFO of California Kitchen, Kaufman grew the company from two to more than 100 locations – at which time it was bought by Pepsi Co.
Kaufman was recruited as president of Koo Koo Roo Chicken, one of the fastest growing fast-casual concepts on the west coast, while Gay joined Wolfgang Puck Restaurants group as CFO, eventually becoming the CEO.
Michelle Steinberg has an impressive history of successfully assisting numerous A-list celebrities and major corporate brands with product launches and various other ventures. Her experience includes launching several renowned restaurant brands, including TAO restaurants, the Palazzo Hotel, House of Blues and Planet Hollywood and All Star Cafe.
MIchelle Steinberg has also handled product launches for many celebrity brands, including, Elton John, Nicole Richie, Jennifer Hudson as well as many others and currently works with corporate clients across the hospitality, lifestyle, fashion and beauty spaces, including iconic brands such as Volkswagen, TAO Group, The Los Angeles Lakers, Forevermark, a De Beers Company, Crocs, Kohl's, Bruno Magli and Ben Sherman among others. Leveraging her astute capabilities and significant industry relationships, Steinberg will likewise introduce the Giggles N’ Hugs brand to a targeted audience of investors, media professionals, manufacturers, retailers, advertisers, designers, distributers and consumers.
In addition to strengthening Giggles N’ Hugs’ overall brand strategy, Steinberg will assist with the market launch of new restaurant locations, services and products at a grassroots level in several major markets nationwide, including New York, Miami, Chicago and Los Angeles.
About Giggles N'Hugs (GIGL)
Los Angeles-based Giggles N' Hugs (GIGL) is a first-of-its-kind, award-winning family restaurant and play space that combines organic gourmet food made with fresh and local ingredients, with the play elements for children in a 2500-square-foot play space in the middle of the restaurant. The concept is similar to Chuck E. Cheese, but offers a unique healthier, high-end version for health conscious parents and families. Parents eat and relax while the kids have an incredible time playing in the custom-made play area with giant climbers, dragons, castles, pirate ships slides and swings and a multitude of other toys.
Giggles N' Hugs (GIGL) also features nightly entertainment such as magic shows, concerts, puppet shows and face painting, and hugely popular party packages for families.
Top restaurant trends align with Giggles N' Hugs (GIGL) business plan. According to the National Restaurant Association, the top four restaurant trends are: 1. Locally-sourced meats and seafood 2. Locally-grown produce 3. Environmental sustainability 4. Healthy kids’ meals.
According to NPD CREST research, kids are eating more healthful food options than ever before, while other studies show that children aged five years or younger are most likely to influence restaurant visits.
$PMCB RSI Review on Shares of Pharmacyte Biotech Inc. (PMCB) PMCB
http://yankeeanalysts.com/2016/12/19/rsi-review-on-shares-of-pharmacyte-biotech-inc-pmcb/
Go $PMCB
$BDCI Check Out DD At http://pennybuster.com/
$PMCB Cell-in-a-Box
What it does
The Cell-in-a-Box® technology, developed by Austrianova, is a means to protect, isolate, store and transport living cells.
Why it's unique
The Cell-in-a-Box® technology is based on a natural cotton derived polymer and is extremely well tolerated by the body allowing long term implantation and biological molecule secretion.
Why it works
The Cell-in-a-Box® technology builds a porous capsule round the cells, allowing free diffusion of nutrients in, and waste products out, and also allowing any biological molecules synthesized by the cells to escape.
What it can be used for
In healthcare, it facilitates the safe implantation of cells, holding them isolated from the immune system and allowing long term production of therapeutic molecules in patients.
In biotechnology, the Cell-in-a-Box® technology protects cells in bioreactors and fermenters, while at the same time simplifying purification of bioproducts produced from the cells and reducing production cost.
In logistics, it allows cells to be frozen, stored for long periods and easily shipped round the world
The technology also forms the basis of a powerful research tool.
What its made of
The company’s proprietary cell packaging material, Gel8, used for the Cell-in-a-Box® technology, is derived from a naturally occurring material and is thus harmless to the environment, biologically inert and has been shown to be safe in humans.
$AEXE Aim commissioned Gustavson Associates (of Colorado, USA) to visit the site, and to recommend plans for commencing mining projects on the property. The visit occurred at the end of 2013, and the report thereof outlined a four-phased approach for obtaining an “Indicated & Measured” classification for the deposits, and for compiling a Preliminary Economic Assessment. Aim now has a 36-month operations plan that accounts for the steps recommended by Gustavson, while enabling the company to immediately commence the business of selling coal sourced from artisan miners on and around its property.
AEXE
https://twitter.com/AEXEQB
$PMCB Obama signs bill to fund 'Moonshot' research to cure cancer.
Hopefully PMCB will get a big funding from the government direct for our upcoming clinical trials or have the government push the FDA agency to speed up and grant us the Fast Track Approval
https://www.engadget.com/2016/12/13/obama-signs-bill-to-fund-moonshot-research-to-cure-cancer/
$TXHD Education is always moving forward, and so is technology. Textmunication allows you to combine the two in order to meet your schools needs.
Instant Campus Security Alerts.
General notifications and informational updates.
Increase Bookstore traffic and student store sales.
Create polls and get instant feedback from your student body.
Drive revenue to sports and other programs with our Mobile Donation Program.
Create contests that engage students and build school spirit.
Student Enrollment Reminders and deadline reminers.
$BDCI This cool video shows how bottled #water is a very small and very efficient water user.
$BDCI With its local partner BDC will establish a warehouse on the main street for consumer-facing vendors to purchase goods right near the port where they set up shop daily. We want to provide vendors with products that are locally made. The government wants these initiatives performed however; capitalization has been an issue to local entrepreneurs. BDC sees this as a great opportunity to provide local jobs and have a light-manufacturing company established rather quickly.
http://bahamasdevelopmentcorp.com/subs/
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=126983719
$PMCB Technical Buzz on PharmaCyte Biotech, Inc. (OTCPK:PMCB)

http://wslnews.com/technical-buzz-on-pharmacyte-biotech-inc-otcpkpmcb/31920/
$PMCB The Gloves Come Off For Pharmacyte Biotech Inc (OTCMKTS:PMCB)

http://www.microcapdaily.com/the-gloves-come-off-for-pharmacyte-biotech-inc-otcmktspmcb/117205/
$PMCB Artificial Organs: 3 Disruptive Medical Devices Making Waves

http://investingnews.com/daily/life-science-investing/medical-device-investing/artificial-organs-3-disruptive-medical-devices-headed-for-market/
$BDCI Twitter Page. https://twitter.com/BahamasDevCorp
$PMCB Functional Analysis of Melligen Cells
Project Member(s):
Simpson, A.
Funding or Partner Organisation:
Pharmacyte Biotech Inc
Start year:
2016
Summary:
Conduct of a research project to gather data on the Melligen cells prior to submission to the University of Chicago Micro-fluidic based beta cell function test.
Pharmacyte, Pharmacyte Biotech, Nuvilex, Melligen, Diabetes, Simpson
Regenerative Medicine (incl. Stem Cells and Tissue Engineering), Diabetes
I guess we will see the PR from PMCB when they submit to University of Chicago. We haven't receive the news on this one yet. Can't wait to hear about it.
http://cfsites1.uts.edu.au/find/projects/detail.cfm?ProjectId=PRO16-1086&_ga=1.12384317.801912675.1473799254
Go $PMCB.
$PMCB Engineering a cure for diabetes
Having successfully reversed type 1 diabetes in mice, Professor Ann Simpson and her team from the Centre for Health Technologies are collaborating with biotechnology company PharmaCyte Biotech to deliver a solution for humans.

A US patent has been approved for what could be the first functional cure for type 1 diabetes?—?by combining cells engineered to produce insulin with technology that will protect these cells from the immune system.
The cells are called ‘Melligen cells’, and they can produce, store and release insulin in response to human blood sugar levels?—?as the pancreas would normally do in a healthy person.
Professor Ann Simpson and her team developed the cells, and have successfully used them to reverse the disease in immunocompromised mice.
However, they faced a challenge in transplanting the Melligen cells into humans: the body’s immune response normally destroys foreign tissue, so the cells would be attacked on entry.
To address the problem, Simpson is collaborating with US clinical stage biotechnology company PharmaCyte Biotech, who has developed a product called Cell-in-a-Box® that can encapsulate the Melligen cells, protecting them from the immune system.
In May 2016, they patented the combination.
“My team and I are extremely pleased that the US patent for the Melligen cells has been granted,” says Simpson.
“This takes us a step closer to releasing diabetics from the need to inject insulin daily and, more importantly, protecting them from the debilitating complications of the disease such as blindness, kidney failure and cardiovascular problems.”
The aim of Cell-in-a-Box is to remove the immune system issue by protecting the Melligen cells with a protective cocoon thereby preventing them being attacked by immune-system cells.
“This is a culmination of many years’ work by our group and we look forward to working with PharmaCyte’s Diabetes Consortium to utilise the Cell-in–a-Box technology to encapsulate the cells for preclinical trials aimed at curing diabetes,” says Simpson.
Cell-in-a-Box is able to stay in the body for at least two years without damage, meaning that combining the Melligen with this technology could produce a long-term solution for type 1 diabetes sufferers.
Preclinical studies of the Melligen cells are already underway by PharmaCyte’s International Diabetes Consortium, with the goal of entering into a clinical trial.
“If we are successful in our efforts, it will bring to fruition the many years of research that have been conducted by Professor Simpson and her colleagues at UTS in developing these remarkable cells,” says PharmaCyte’s CEO, Kenneth L Waggoner.
“For the millions of people worldwide who suffer from a disease of epidemic proportions, our treatment could relieve them of the onerous daily requirements for insulin administration and dietary restrictions and offer a life free from the very serious and even life-threatening complications associated with diabetes.”
Interview with the CEO of $TBEV
High Performance Beverage Company
$PMCB PharmaCyte’s CEO Discusses Development of its Pancreatic Cancer Therapy

LAGUNA HILLS, Calif., Sept. 15, 2016 (GLOBE NEWSWIRE) -- PharmaCyte Biotech, Inc. (OTCQB:PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, today addressed shareholders on the development of PharmaCyte's therapy for advanced pancreatic cancer.
The Chief Executive Officer of PharmaCyte, Kenneth L. Waggoner commented, "We understand that shareholders are frustrated with our current share price; however, the share price doesn't reflect where PharmaCyte is today. In what has been a relatively short time for a biotech company, we have dramatically moved our Cell-in-a-Box® technology forward along the path toward the clinic, we have surrounded our technology with some of the best minds in the industry and we have put together a team that is working diligently every single day to get PharmaCyte in front of the U.S. Food and Drug Administration (FDA) so that we can clear the final hurdles that will allow us to begin our clinical trial in advanced pancreatic cancer. There are numerous moving parts on many fronts. Just because our shareholders aren't seeing news on a weekly basis, this does not mean there isn't a flurry of work being done every single day.
"It is important for our shareholders to realize that PharmaCyte's pancreatic cancer therapy is classified by drug regulatory authorities as a ‘biologic' rather than a single molecule drug or a New Chemical Entity (NCE). This is because our therapy involves the use of living, genetically altered human cells. The overall development process is much more involved and more complex for a biologic than an NCE. Nevertheless, PharmaCyte's therapy is moving forward at a realistic pace. Our partner Austrianova will be carrying out an engineering manufacturing ‘run' in the near future of the production of the capsules that will be used with low dose ifosfamide as our pancreatic cancer therapy. If successful, not only will this run define the specifications of the capsules that will be use to encapsulate the living cells, it will also set the stage for cGMP production of the capsules for our planned clinical trial.
"An example of the different environment that we're working in is that biologic products - particularly encapsulated living cell products like PharmaCyte's and unlike most pharmaceutical products which can be simply ‘end-sterilized' and delivered as sterile products - must be kept alive from the moment the cells are thawed from the frozen research cell bank and then encapsulated until they are implanted in a patient. At all times during these operations our product must be kept free of contamination by bacteria, yeast and fungi. Thus our biologic product is more akin to a live vaccine or to a stem cell therapy. The cell culture medium used to maintain the living cells within the capsules is also an excellent growth medium for all of those previously mentioned undesirable contaminants. Contaminant and other testing of our research cell bank is underway at ViruSure, one of PharmaCyte's contractors, while Austrianova continues to make steady progress in order to manufacture cGMP clinical trial material that we will need to begin the trial.
"It goes without saying that great care has to be taken at every step to ensure the maintenance of aseptic conditions; this is one reason for carrying the production out in an ‘isolator' that prevents actual contact between production staff and the product being manufactured. A living cell product also brings with it new regulatory challenges. PharmaCyte has been steadily working through these challenges with a number of regulatory advisors both in the United States and in the United Kingdom. These exciting new "Advanced Medicinal Products" are the future of treatment for serious diseases, and in many ways pioneering companies, like PharmaCyte, are working with the regulators to lay the ground work for tomorrow's regulatory framework."
About PharmaCyte Biotech
PharmaCyte Biotech a clinical stage biotechnology company developing therapies for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as "Cell-in-a-Box®." This technology will be used as a platform upon which therapies for several types of cancer and diabetes are being developed. PharmaCyte's therapy for cancer involves encapsulating genetically engineered human cells that convert an inactive chemotherapy drug into its active or "cancer-killing" form. These encapsulated cells are implanted as close to the patient's cancerous tumor as possible. Once implanted, a chemotherapy drug that is normally activated in the liver (ifosfamide) is given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been implanted. When the ifosfamide comes in contact with the encapsulated cells they act as an artificial liver and activate the chemotherapy drug at the source of the cancer. This "targeted chemotherapy" has proven effective and safe to use in past clinical trials and results in no side effects.
In addition to developing a novel therapy for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and insulin-dependent Type 2 diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology. Once the encapsulated cells are implanted in a diabetic patient they will function as an "artificial pancreas" for purposes of insulin production.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte Biotech and its future events and results that involve inherent risks and uncertainties. The words "anticipate", "believe", "estimate", "expect", "intend", "plan" and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte's ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte's intellectual property and PharmaCyte's continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: Info@PharmaCyte.com
Read more: http://www.nasdaq.com/press-release/pharmacytes-ceo-discusses-development-of-its-pancreatic-cancer-therapy-20160915-00572#ixzz4KKciXLpt
$PTLF Technical Chart http://www.barchart.com/charts/stocks/PTLF&style=technical #dog #cats #doglover #doglovers #meds #lfl #dogsofinsta #dogsofinstaworld #cutedogs #pawsome
$PMCB Promising Diabetes Treatment Using Human Liver Cells May Be on the Horizon
BY KATIE MCCALLUM IN NEWS.

A collaboration between Prof. Ann Simpson of the University of Technology Sydney (UTS) in Australia and a U.S. clinical stage biotechnology company, PharmaCyte Biotech, shows promise for a unique alternative therapy for a generation of type 1 diabetes patients.
Utilizing human liver cells engineered to supply the body with insulin, the therapy could eliminate the daily requirements for insulin management and prevent life threatening complications associated with diabetes.
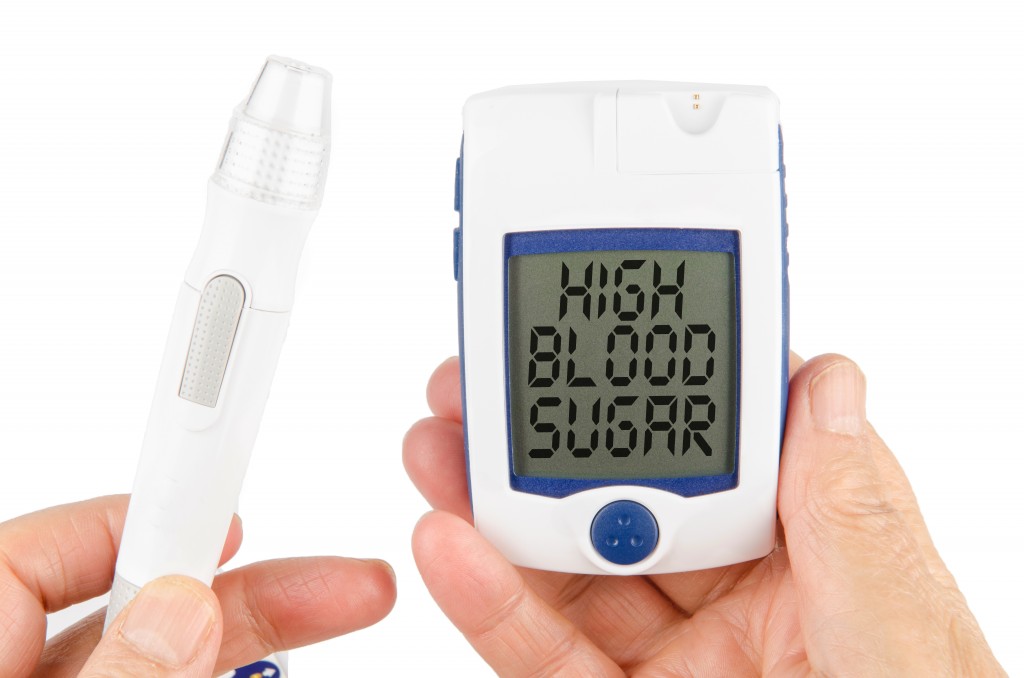
In 2012 it was estimated that diabetes affects 29.1 million people worldwide. The Juvenile Diabetes Research Foundation estimates that 3 million Americans alone suffer from the autoimmune disease type 1 diabetes, for which there is currently no cure. The current treatment of type 1 diabetes requires frequent insulin injections and dietary restrictions. More seriously, type 1 diabetes is a lifelong autoimmune disease that can ultimately result in eye and kidney diseases, nerve impairment, and cardiovascular complications.
Simpson and her colleagues developed insulin-producing, artificially engineered “Melligen” cells that produce, store, and release insulin in amounts relevant to the blood sugar levels of individual patients. A patent was recently obtained for Melligen cells through the U.S. Patent Office, and the cell line is now in the process of being commercialized for human preclinical trials by PharmaCyte.
PharmaCyte’s Cell-in-a-Box technology will be used to prevent the inflammatory immune response that commonly happens upon transplantation of foreign tissue into the body. Cell-in-a-Box is a signature technology of PharmaCyte and serves to encapsulate live cells in a robust manner for an extended time to aid in successful transplantation. PharmaCyte hopes to apply this technology to Melligen cells to ensure their success in the human body.
Using the insulin-producing Melligen cells developed at UTS, along with PharmaCyte’s advanced biological delivery technology Cell-in-a-Box, researchers hope to commercialize a groundbreaking therapy as they work toward a possible cure for type 1 diabetes. Melligen cells could also be used in cases of advanced type 2 diabetes, where current medications are no longer effective.
“For the millions of people worldwide who suffer from a disease of epidemic proportions, our treatment could relieve them of the onerous daily requirements for insulin administration and dietary restrictions and offer a life free from the very serious and even life-threatening complications associated with diabetes,” said PharmaCyte CEO Kenneth L. Waggoner.

Katie McCallum is a PhD candidate at University of Texas Health Science Center at Houston in the Microbiology and Molecular Genetics program. While at the bench, her research is primarily focused on the basic biology governing host-microbial interactions. Katie's research interests extend far beyond the scope of medical microbiology. Her exposure to the enormity of collaborative effort and innovative research that exists in the Texas Medical Center has fostered her interest in science communication and science outreach.
TSXV: $SIL Current Work Program
In 2016, the plans are to access and evaluate most of the estimated 11.5 kilometres of historic workings at Las Chispas. Initial rehabilitation will take several months to complete and will consist of cleaning and securing all underground areas, removing historic backfill and stockpiling for possible future processing, building a ladder system to access multiple levels, detailed mapping and sampling of accessed workings, and implementation of overall safety protocols.
$TRXC Great Move On After Hours News! Conference Call This Week Also.
TransEnterix, Inc.
TransEnterix, Inc. Announces First Sale of ALF-X? Surgical Robotic System
RESEARCH TRIANGLE PARK, N.C. --(BUSINESS WIRE)-- TransEnterix, Inc. (NYSE MKT: TRXC), a medical device company that is pioneering the use of robotics to improve minimally invasive surgery, today announced the first global sale of its ALF-X? Surgical Robotic System to Humanitas Hospital in Milan, Italy .
"We are pleased to announce the first global sale of our ALF-X System," said Todd M. Pope , President and CEO of TransEnterix . "Humanitas is an outstanding academic hospital with a respected multi-specialty surgical program. The ALF-X enables an enhanced surgical program through the use of advanced robotic technology with responsible procedural economics."
"The initiation of sales of ALF-X represents an important milestone towards realizing our vision to transform surgical robotics," continued Mr. Pope . "We have made substantial progress building out our global commercial, distribution and service capabilities throughout 2016."
The ALF-X System is CE Marked and is indicated for use in general surgery, gynecology, urology and thoracic surgery. We are actively preparing a submission for U.S. FDA Clearance for the ALF-X System.
About Humanitas
Humanitas is a highly specialized research and teaching hospital partnered with Humanitas University Medical School . Humanitas University trains globally-oriented healthcare professionals through state-of-the-art interactive teaching methods and close integration with their clinical and research community. Humanitas believes that it is essential to unite organizational efficiency and clinical quality. For this reason, it was the first Italian general hospital and one of the few in Europe , to be certified for quality by the International Joint Commission .
About TransEnterix
TransEnterix is a medical device company that is pioneering the use of robotics to improve minimally invasive surgery by addressing the clinical and economic challenges associated with current laparoscopic and robotic options. The company is focused on the commercialization of the ALF-X Surgical Robotic System, a multi-port robotic system that brings the advantages of robotic surgery to patients while enabling surgeons with innovative technology such as haptic feedback and eye sensing camera control. The company is also developing the SurgiBot™ System, a single-port, robotically enhanced laparoscopic surgical platform. The ALF-X Surgical System has been granted a CE Mark but is not available for sale in the US. For more information, visit the TransEnterix website at www.transenterix.com.
Forward Looking Statements
This press release includes statements relating the ALF-X® System and SurgiBot™ System and our current regulatory and commercialization plans for these products. These statements and other statements regarding our future plans and goals constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, and are intended to qualify for the safe harbor from liability established by the Private Securities Litigation Reform Act of 1995. Such statements are subject to risks and uncertainties that are often difficult to predict, are beyond our control and which may cause results to differ materially from expectations. For a discussion of the risks and uncertainties associated with TransEnterix's business, please review our filings with the Securities and Exchange Commission (SEC), including our Annual Report on Form 10-K filed on March 3, 2016 , our other filings we make with the SEC . You are cautioned not to place undue reliance on these forward looking statements, which are based on our expectations as of the date of this press release and speak only as of the origination date of this press release. We undertake no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise.
View source version on businesswire.com: http://www.businesswire.com/news/home/20160801006226/en/
For TransEnterix, Inc.
Investor Contact:
Mark Klausner , 443-213-0501
invest@transenterix.com
or
Media Contact:
Mohan Nathan , 919-765-8400
media@transenterix.com
Source: TransEnterix, Inc.
You are subscribed to TransEnterix, Inc. Investor Relations' e-mail alerts as pennystockprophecy@gmail.com.
To update your e-mail and alert preferences, please click here.
To unsubscribe, please click here.
TransEnterix, Inc.
635 Davis Drive Suite 300, Morrisville, NC 27713
Service provided by Nasdaq
635 Davis Drive
Suite 300
Morrisville, NC 27560
United States
919-765-8400
http://www.transenterix.com
$PMCB SCN's Wendy Gillette Interviews CEO Kenneth Waggoner of PharmaCyte Biotech, Inc. (OTCQB:PMCB)
$RNVA ALERT http://www.rennovahealth.com/about/overview
$PMCB Valuing PharmaCyte Biotech's New Treatment Option for Pancreatic Cancer

BALTIMORE, MD / ACCESSWIRE / March 9, 2016 / The current gold standard of care for patients with advanced, inoperable pancreatic cancer is the combination therapy of Abraxane® + gemcitabine, which was approved by the U.S. Food and Drug Administration (FDA) in late 2013. Another treatment, which is not used as widely, is FOLFIRINOX, a four-drug combination therapy. These treatments offer varying degrees of efficacy but what happens to the patients when they no longer respond to these therapies? With limited options currently available, PharmaCyte Biotech (PMCB) is set to launch a new, mid-stage clinical trial in 2016 to specifically address what is clearly an unmet medical need and potentially increase the lifespan prospects for these late stage pancreatic cancer sufferers.
Positive results from this landmark trial would move PharmaCyte toward the front of the pack of cancer treatment providers, substantially enhance the value of the Company and attract the attention of major pharmaceutical companies. After all, a successful trial could be the trigger to change the way advanced inoperable pancreatic cancer is treated. It would finally give patients, who no longer respond to the premier combination therapy, a viable option. PharmaCyte's pancreatic cancer treatment (Cell-in-a-Box®+ low-dose ifosfamide chemotherapy) seeks to satisfy this critical unmet medical need by acting as a consolidation (and post- Abraxane® + gemcitabine or FOLFIRINOX) therapy.
Huge Addressable Market
A recent report issued by Research and Markets suggests that the size of the pancreatic cancer therapeutic markets in just the major developed countries will approach $3 billion in the next 5 years. However, the near term addressable market for PharmaCyte's therapy in the U.S. alone is substantial. A typical current price tag for pancreatic cancer therapy for the roughly 43,000 patients diagnosed with the disease each year ranges anywhere from $50,000 - $75,000. More than 60% of this patient group, or roughly 25,000 patients, will no longer respond to the premier combination therapy (Abraxane® + gemcitabine or FOLFIRINOX) and require a new, or consolidation therapy. Therefore, should PharmaCyte ultimately receive FDA approval for its treatment, the Company's initial target market size would range from $1.25 billion to $1.8 billion, depending upon pricing for the Company's therapy. Clearly, there is tremendous underlying value in PharmaCyte and its novel therapy.
The Trial
PharmaCyte has designed a Phase 2b clinical trial that will include a hard-stop at 6 months to evaluate the data. The primary endpoints of the trial will essentially mirror some of the primary endpoints of other treatments that have received FDA approval, such as Abraxane®. These include progression-free survival (PFS) and the side effects experienced from the combination treatment that occurs in the patients. PFS is the time that elapses from the first day of treatment until the disease gets worse and will be measured and determined at 6 and 12 months. The occurrence of any side effects will be monitored throughout the trial.
The trial design also includes a series of measurable secondary endpoints. These include:
- Onset of pain and need for pain medications
- Whether inoperable tumors become operable as a result of treatment;
- Change in tumor size; and
- Patient overall quality of life during the treatment.
The Company's consolidation therapy approach is poised to emerge as a win-win for the Company and patients alike. This positioning, along with an innovative trial design, enable PharmaCyte to launch and complete this clinical trial at a substantially reduced cost and shorter time period relative to typical studies since it will be conducted with fewer patients and in key sites in the U.S. and abroad. In addition, by measuring pain as one of the secondary endpoints, the Company eliminates the need to launch costly separate trials or studies to track the unbearable and untreatable pain that is associated with pancreatic cancer in 20-25% of patients. Finally, because the design of the trial allows for the release of interim data relatively quickly, it only enhances the value of the therapy to the Company and prospective patients.
Impact of Potential Approval
Positioning its innovative treatment as the next or last stage therapy for the difficult patient treatment group may be the fastest route to PharmaCyte's FDA approval, as there is no truly effective therapy for patients at this stage that can materially extend survival rates and improve their quality of life. Measurements will also be taken to determine if PharmaCyte's treatment can convert an inoperable tumor to an operable one. If tumor shrinkage data proves encouraging, it could have a materially favorable impact on a trial's outcome, let alone the quality of life and lifespan of pancreatic cancer patients, and the overall valuation of the Company among cancer treatment leaders.
About Goldman Small Cap Research: Founded in 2009 by former Piper Jaffray analyst and mutual fund manager Rob Goldman, Goldman Small Cap Research produces sponsored and non-sponsored small cap and microcap stock research reports, articles, stock market blogs, and popular investment newsletters. Goldman Small Cap Research is not in any way affiliated with Goldman Sachs & Co.
This press release reflects our most recently published sponsored research article on PharmaCyte Biotech, Inc. The information used and statements of fact made have been obtained from sources considered reliable but we neither guarantee nor represent the completeness or accuracy. Goldman Small Cap Research relied solely upon information derived from PharmaCyte Biotech Inc. ("the Company") authorized press releases or legal disclosures made in its filings with the U.S. Securities and Exchange Commission http://www.sec.gov.
Separate from the factual content of our articles about the Company, we may from time to time include our own opinions about the Company, its business, markets and opportunities. Any opinions we may offer about the Company are solely our own, and are made in reliance upon our rights under the First Amendment to the U.S. Constitution, and are provided solely for the general opinionated discussion of our readers. Our opinions should not be considered to be complete, precise, accurate, or current investment advice. Statements herein may contain forward-looking statements and are subject to significant risks and uncertainties affecting results.
A Goldman Small Cap Research report, update, newsletter, or article is not intended as an offering, recommendation, or a solicitation of an offer to buy or sell the securities mentioned or discussed is to be used for informational purposes only. Please read all associated full disclosures, disclaimers, and analyst background on our website before investing. Neither Goldman Small Cap Research nor its parent is a registered investment adviser or broker-dealer with FINRA or any other agency. To download our research, view our disclosures, or for more information, visit www.goldmanresearch.com. In 2015, Goldman Small Cap Research was compensated by a third party (not PharmaCyte or anyone affiliated with PharmaCyte) in the amount of $22,000 for investment research services that includes the publication of research reports, updates, and a series of articles.
Goldman Small Cap Research
Rob Goldman, Analyst
410-609-7100
rob@goldmanresearch.com
SOURCE: Goldman Small Cap Research
$GRCU Headlines http://www.barchart.com/news/stocks/GRCU
$PMCB Austrianova’s former Chief Scientist, Lilli Brandtner, features in an article about using Austrianova’s cell encapsulation technology to treat diabetes. Dr Brandtner is working together with Austrianova, PharmaCyte Inc. and other partners as part of PharmaCyte’s International Diabetes Consortium

http://themavorarlberg.at/made-in-vorarlberg/diabetes-heilen
Translation -
Cure Diabetes !
With this ambitious target is currently employs a team of researchers from around the world in a unique project . It is headed by the Vorarlberg scientist Dr. Eva Maria ( Lilli ) Brandtner from the molecular biology laboratory VIVIT in Dornbirn . Together they tried to protect insulin-producing cells from an immune attack to transplant - thereby curing type 1 diabetes would be possible to even have no intact insulin producing cells more and therefore repeatedly have to rely on insulin injections daily . Brandtner are in the " Long Night of Research " on April 22, an exclusive insight into the EU-funded project .
$AUY 4.99
Profile Get Profile for:
GO
Yamana Gold, Inc.
Royal Bank Plaza
Suite 2200
Toronto, ON M5J 2J3
Canada - Map
Phone: 416-815-0220
Fax: 416-815-0021
Website: http://www.yamana.com
Details
Index Membership: N/A
Sector: Basic Materials
Industry: Gold
Full Time Employees: N/A
Business Summary
Yamana Gold Inc. engages in the exploration and development of precious metal properties in Brazil, Argentina, Chile, Mexico, and Canada. It explores for gold, silver, and copper. It primarily holds interests in the Chapada, El Peñón, Gualcamayo, Mercedes, Canadian Malartic, Minera Florida, and Jacobina properties. The company was formerly known as Yamana Resources Inc. and changed its name to Yamana Gold Inc. in July 2003. Yamana Gold Inc. was founded in 1980 and is based in Toronto, Canada.
$PMCB PharmaCyte’s Novel Diabetes Treatment Approach Could Prove Superior to Other Methods Under Development
Forecasts for the number of people that will be diagnosed with diabetes in the coming years are staggering. While many treatments exist to treat some of the effects of diabetes on patients afflicted with the disease, a “holy grail” to truly treat the disease has yet to be developed. Clearly, an out of the box approach is required. If future test results affirm the data derived from recent studies, the PharmaCyte Biotech (OTCQB – PMCB - $0.07 – Spec Buy) Cell-in-a-Box® diabetes treatment platform could emerge as the treatment of choice for this disease.
Dozens of studies and trials to treat Type 1 and Type 2 diabetes are ongoing, given that the size of the global diabetes market for therapeutic devices and drugs is expected to reach US $114.3 billion by 2018, according to a report by Transparency Market Research. However, since results have been mixed for even the largest and most successful companies, industry participants are now exploring new ideas and new approaches that have demonstrated efficacy in early studies, which could serve as an indirect or direct benefit to PharmaCyte.
For example, one of the top two players in diabetes treatment, Novo Nordisk (NYSE – NVO), announced that it is collaborating with IBM (NYSE – IBM) to combine IBM’s cognitive computing capabilities with diabetes research by collecting and analyzing real-time data from patients using Novo Nordisk treatments and devices. The hope is that this venture leads to improved solutions for diabetes management. Novo Nordisk also submitted a new type of fast-acting mealtime insulin to the FDA for approval, a departure from its primary offerings. Meanwhile, Novo Nordisk competitor Eli Lilly (NYSE – LLY), which has had six diabetes treatments approved since 2014 halted development of a promising diabetes treatment under development. If a company with 6 approved drugs in a treatment category is still working on the problem, and another leader is moving outside of its comfort zone in an effort to develop the most effective therapy available, it is an indication that the time is now for PharmaCyte.
The Company has the exclusive worldwide rights to use Melligen cells to treat diabetes. Melligen cells are genetically engineered from human liver cells and have been shown to secrete insulin in response to the concentrations of glucose (blood sugar) in their environment. A recent article published in scientific journal Molecular Therapy noted that when Melligen cells were transplanted into diabetic mice whose immune systems were essentially not functioning, the blood glucose levels of the mice became normal.
This observation illustrates that Melligen cells can reverse the diabetic condition. PharmaCyte plans to encapsulate a human cell line that has been genetically modified to produce, store and release insulin in response to blood glucose levels in their surroundings. Therefore, the Melligen cell line, when combined with Cell-in-a-Box® encapsulation, could ultimately become a treatment that has clear advantages over current therapies used for Type 1 diabetes and Type 2 insulin-dependent diabetes and could potentially replace them. As a result, we look for these shares to move toward the $0.20 level in early 2016.
$COYN 1.30 Bounce Off 52 Wk Low. O/S 8.80M
Float 6.70M
Insiders 42.96%
http://www.copsync.com/
$OPGN 1.58 If They Figure Out The Super Bug The Gains Here Could Be Enormous. Merck & Co. venture arm backs superbug fighter OpGen in $10.3 million funding round.
http://www.bizjournals.com/washington/news/2016/06/01/merck-co-venture-arm-backs-superbug-fighter-opgen.html?ana=twt
OpGen Inc. (NASDAQ: OPGN) raised $10.4 million in a recent funding round led by the Merck Global Health Innovation Fund, the venture capital arm of pharmaceutical giant Merck & Co.
The Gaithersburg biotech combats infectious disease, including so-called superbugs, with its molecular diagnostics and bioinformatics tools. The raise will support sales and marketing, as well as ongoing research and development, of the company's multidrug resistant organism platform called Acuitas Lighthouse.
Evan Jones, CEO of OpGen and seen here in 2007. The company recently raised $10.3 million to support its Acuitas Lighthouse product.
JOE BRIER
In a Securities and Exchange Commission filing earlier this week, the Merck fund reported it now has a 37.5 percent stake in OpGen. That's up from a previously reported stake of 11.8 percent.
Sabby Management LLC also led the round. OpGen management and certain directors also participated.
OpGen's rapid DNA testing technology can quickly identify drug-resistant bacteria, or bugs with genes that could potentially lead them to become resistant.The rapid testing allows hospitals or public health experts to identify and more quickly respond to patients who are found to be infected.
It's an increasingly necessary practice in response to growing global concerns about the threat of antibiotic resistance. Last month, a Pennsylvania woman was discovered to have an E. coli bacteria resistant to the antibiotic known as a "last resort" drug, although it responded to other drugs. That finding has raised fears of future infections that are untreatable.
A recent study conducted by OpGen of hospitals around the District found a 5.1 percent prevalence rate of another antibiotic-resistant bacteria known as carbapenem-resistant Enterobacteriaceae, or CRE. CRE is particularly alarming because it spreads easily and is particularly lethal. There has not been a significant CRE incident reported in the District, although the National Institutes of Health Clinical Center in Bethesda battled a deadly CRE outbreak in 2011.
OpGen issued an aggregate of about 6.7 million shares of common stock, 2.3 shares of preferred stock and warrants to purchase 6.8 million shares of common stock. The financing consisted of units sold at a price of $1.14 per unit, with each consisting of either one share of common stock or one share of non-voting, convertible preferred stock, and one warrant to acquire 0.75 of one share of common stock.
The private placement will be closed in two transactions with the second closing is expected to occur later this month, executives said.
OpGen to Present at the 6th Annual LD Micro Invitational Conference
GAITHERSBURG, MD / ACCESSWIRE / June 2, 2016 / OpGen, Inc. (OPGN), a precision medicine company using molecular diagnostics and bioinformatics to combat infectious disease, announced today that Kevin Krenitsky, president, will present at the 6th Annual LD Micro Invitational conference on June 7, 2016 at 2:30 p.m. Pacific Time. The conference is being held from June 7- 9 at the Luxe Sunset Boulevard Hotel in Los Angeles.
A webcast of the presentation will be available in the investor relations section of the Company's website, www.opgen.com, and will be archived for 90 days following the presentation.
About OpGen
OpGen, Inc. is developing and deploying precision medicine tools to combat infectious disease in global healthcare settings, helping physicians improve patient outcomes by providing more rapid information about life-threatening infections and decreasing the spread of infections caused by multidrug-resistant microorganisms. OpGen offers a full portfolio of in vitro diagnostic products and clinical laboratory services that employ state-of-the-art molecular diagnostics and bioinformatics. Its QuickFISH® products are a suite of FDA-cleared and CE-marked diagnostics used to rapidly detect pathogens in positive blood cultures. Clinical laboratory services utilize the Acuitas® products, including the MDRO Gene Test, the Resistome Test, microbial Whole Genome Sequence Analysis and Acuitas Lighthouse™ bioinformatics system designed to detect, type, track and trend antibiotic resistant organisms in real-time. Learn more at http://www.opgen.com and follow OpGen on Twitter and LinkedIn.
About LD Micro
LD Micro was founded in 2006 with the sole purpose of being an independent resource in the microcap space. What started out as a newsletter highlighting unique companies has transformed into an event platform hosting several influential conferences annually (Invitational, Summit, and Main Event).
In 2015, LDM launched the first pure microcap index (the LDMi) to exclusively provide intraday information on the entire sector. LD will continue to provide valuable tools for the benefit of everyone in the small and microcap universe. For those interested in attending, please contact David Scher at david@ldmicro.com or visit http://www.ldmicro.com for more information.
Contacts:
OpGen
Michael Farmer
Director, Marketing
(240) 813-1284
mfarmer@opgen.com
InvestorRelations@opgen.com
Investor Relations
LHA
Kim Sutton Golodetz
(212) 838-3777
kgolodetz@lhai.com
or
Bruce Voss
(310) 691-7100
bvoss@lhai.com
OpGen media
MacDougall Biomedical Communications
Cammy Duong
781-591-3443
cduong@macbiocom.com
wshaw14' welcome to 'Metals Creek Resources Corp. (TSXV:MEK)

Hole TOG-15-39 Section 1512W - 11.45g/t Au over 1.53m & Hole TOG-15-41
Section 1475W - 1.47g/t Au over 17.83m and
3.93g/tAu over 2.8m (MEK PR February 11, 2016)
480 meters step out west of Thomas-Ogden Gold Horizon -
5.06g/tAu over 2.6m (MEK PR March 4, 2016)
http://www.metalscreek.com/article/ogden-315.asp
Additional drilling planned for 2016
MEK potential = 30 million oz Gold and MEK with closer location in
pipeline fault zone to
Hollinger Goldcorp (19.4M),
Dome Mine (13.3M),
McIntyre (10.8M) and longer
8KM of Destor Fault old Gold Mines property has bigger Potential of
more Gold -

http://www.metalscreek.com/article/ogden-315.asp#
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122195868
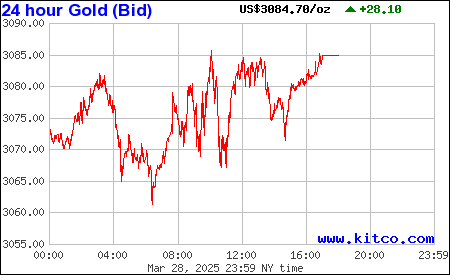
http://www.kitconet.com/images/live/au0001wb.gif
- God Bless -
$PMCB Stonegate Capital Partners Publishes Research Report on PharmaCyte Biotech

SILVER SPRING, Md., Feb. 10, 2016 -- PharmaCyte Biotech, Inc. (OTCQB:PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, announced today that Stonegate Capital Partners (Stonegate), a privately held corporate advisory firm based in Dallas, Texas, with offices in New York and Boston, issued a Research Report featuring PharmaCyte Biotech. The report can be viewed at http://stonegateinc.com/reports/Pharma_Feb_2016.pdf
PharmaCyte Biotech’s Chief Executive Officer, Kenneth L. Waggoner, commented on the report saying, “In our opinion the Research Report released by Stonegate Capital Partners and authored by Senior Research Analyst Laura S. Engel is comprehensive and captures the essence of what we are trying to accomplish at PharmaCyte. Particular emphasis is placed on the two major programs underway that employ our proprietary Cell-in-a-Box® live-cell encapsulation technology to tackle two of the most difficult to treat diseases in the world - cancer and diabetes.”
Stonegate’s Senior Research Analyst, Laura S. Engel, authored the Research Report, which focuses on PharmaCyte’s clinical development of a variety of potential therapies across several areas of disease utilizing its novel live cell encapsulation platform, Cell-in-a-Box®.
Stonegate’s Senior Research Analyst writes that PharmaCyte offers a significant opportunity for investors looking to participate in this development. “We believe that PMCB has a solid pathway towards commercialization with the approach outlined for its Phase 2b study for pancreatic cancer patients set to begin in the Q2-Q3 of calendar year 2016. Additionally, management will be actively moving other potential treatments along; these treatments are related to malignant ascites fluid accumulation in the abdomens of patients with abdominal cancers and Type 1 and Type 2 diabetes. Positive news flow should yield investor returns as the Company accomplishes its milestones that have been set for 2016.”
Senior Research Analyst Laura S. Engel started a career in public accounting following graduation from the University of Virginia, McIntire School of Business, with a B.S. in Accounting. After several years with Arthur Andersen, Engel transitioned in industry where she worked for several publicly traded companies, managing both domestic and international accounting operations. Engel subsequently transitioned into the finance realm, joining a Financial Services consulting team with KPMG Consulting focusing on systems implementations for several Wall Street banks. She later joined a smaller wealth management group, servicing a book of clientele through investment management and financial planning. Since 2003, Engel has teamed with Stonegate and worked in several areas of the business.
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company focused on developing and preparing to commercialize treatments for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®.” This unique and patented technology will be used as a platform upon which treatments for several types of cancer and diabetes are being developed. PharmaCyte’s treatment for cancer involves encapsulating genetically modified live cells that convert an inactive chemotherapy drug (ifosfamide) into its active or “cancer-killing” form. These encapsulated live cells are placed as close to a cancerous tumor as possible. Once implanted in a patient, a chemotherapy drug which needs to be activated in the body (prodrug) is then given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been placed. When ifosfamide, which is normally activated in the liver, comes in contact with the encapsulated live cells, activation of the chemotherapy drug takes place at the source of the cancer without any side effects from the chemotherapy. This “targeted chemotherapy” has proven remarkably effective and safe to use in past clinical trials.
In addition to developing a novel treatment for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and Type 2 insulin-dependent diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte Biotech and its future events and results that involve inherent risks and uncertainties. The words "anticipate," "believe," "estimate," "expect," "intend," "plan" and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte's ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte’s intellectual property and PharmaCyte’s continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: info@PharmaCyte.com
http://www.econotimes.com/Stonegate-Capital-Partners-Publishes-Research-Report-on-PharmaCyte-Biotech-158732
$AMRS Filing With Bill and Melinda Gates Foundation.
http://ih.advfn.com/p.php?pid=nmona&article=71476165
As previously reported, on April 8, 2016, Amyris, Inc. (the “ Company ”) and the Bill & Melinda Gates Foundation (the “ Gates Foundation ”) entered into (i) a Securities Purchase Agreement (the “ Purchase Agreement ”), pursuant to which the Company agreed to sell and issue 4,385,964 shares of its common stock (the “ Shares ”) to the Gates Foundation at a purchase price per share equal to $1.14 (the “ Gates Foundation Investment ”) and (ii) a Charitable Purposes Letter Agreement (the “ Letter Agreement ”), pursuant to which the Company agreed to use the proceeds from the Gates Foundation Investment to develop a yeast strain that produces artemisinic acid and/or amorphadiene at a low cost and to supply such artemisinic acid and amorphadiene to companies qualified to convert artemisinic acid and amorphadiene to artemisinin for inclusion in artemisinin combination therapies used to treat malaria. The entry into the Purchase Agreement and the Letter Agreement was previously reported in the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on April 11, 2016, which is incorporated herein by reference.
On May 10, 2016, the Company and the Gates Foundation closed the Gates Foundation Investment, resulting in the issuance of 4,385,964 Shares to the Gates Foundation for proceeds to the Company of approximately $5.0 million.
$PTX .53 News..
Pernix Therapeutics Overcomes Challenge to Treximet® ‘183 Patent
Pernix Therapeutics Holdings, Inc.
1 hour ago
GlobeNewswire
????
MORRISTOWN, N.J., May 09, 2016 (GLOBE NEWSWIRE) -- Pernix Therapeutics Holdings, Inc. (PTX), a specialty pharmaceutical company, today announced that on May 6, 2016, the Patent Trial and Appeal Board (PTAB) of the United States Patent and Trademark Office (USPTO) denied a petition for inter partes review (IPR) filed by Graybar Pharmaceuticals, LLC (now Gray Square Pharmaceuticals, LLC) against Pozen, Inc. This petition, which was filed on November 12, 2015, sought review of certain claims of U.S. Patent No. 7,332,183 (’183 patent), which expires 2026. The ‘183 patent is licensed by Pernix and is one of several FDA Orange Book listed patents covering Treximet®, manufactured and sold by Pernix Therapeutics, (sumatriptan/naproxen sodium), a prescription medication for acute migraine attacks, with or without aura.
The PTAB Ruled: “After considering the Petition and Preliminary Response, we determine that Petitioner has not established a reasonable likelihood of prevailing with respect to any of the challenged claims of the ’183 patent. See 35 U.S.C. § 314(a). Accordingly, we deny the Petition, and do not institute inter partes review.”
“We are pleased with the PTAB’s decision to deny Gray Square’s petition,” said Doug Drysdale, Chairman and CEO of Pernix. “We continue to be confident in the validity of the ’183 patent and the other FDA Orange Book listed patents covering Treximet as well as all of our brands.”
The ‘183 patent has resulted in the denial of at least three generic companies gaining market entry until 2026. The validity of the ‘183 patent has been previously up-held by the Federal Circuit and now the PTAB; further strengthening this patent and reinforcing limited generic entry beyond 2018 and until the ‘183 patent expires in 2026. As a result, Pernix expects to retain a meaningful market share with its own authorized generic version of Treximet, which it intends to launch via its Macoven subsidiary, in 2018.
About Pernix Therapeutics
Pernix Therapeutics is a specialty pharmaceutical business with a focus on acquiring, developing and commercializing prescription drugs primarily for the U.S. market. The Company targets underserved therapeutic areas such as CNS, including neurology and pain management, and has an interest in expanding into additional specialty segments. The Company promotes its branded products to physicians through its Pernix sales force and markets its generic portfolio through its wholly owned subsidiaries, Macoven Pharmaceuticals, LLC and Cypress Pharmaceutical, Inc.
Related Quotes
PTX
0.53
-7.75%
Pernix Therapeutics Holdings, …? Watchlist
0.53-0.0445(7.75%)
NASDAQFri, May 6, 2016 4:00 PM EDT
8:05 am Pernix Therapeutics announces that the USPTO denied a petition for inter partes review filed by Graybar Pharmaceuticals against Pozen Briefing.com 1 hr 17 mins ago
Is Pernix Therapeutics in a Race to Zero? 24/7 Wall St. q 3 days ago
More
To learn more about Pernix Therapeutics, visit http://www.pernixtx.com.
About TREXIMET®
TREXIMET was first approved by the U.S. Food and Drug Administration (FDA) in April 2008 for the acute treatment of migraine attacks, with or without aura, in adults. The product is formulated with POZEN’s patented technology of combining a triptan with a non-steroidal anti-inflammatory drug (NSAID) and GlaxoSmithKline’s (GSK) RT Technology™. TREXIMET has been shown to provide superior sustained pain relief compared to placebo and to both of the single mechanism of action components. In clinical trials, TREXIMET provided a significantly greater percentage of adult patients with migraine pain relief at two hours compared to sumatriptan 85 mg or naproxen sodium 500 mg alone. In addition, TREXIMET provided more patients sustained migraine pain relief from two to 24 hours compared to the individual components.
Indication
Prescription TREXIMET® is used to treat acute migraine headaches with or without aura in patients 12 years of age and older.
TREXIMET® is not used to treat other types of headaches such as hemiplegic or basilar migraines. TREXIMET® is not used to prevent or decrease the number of migraine headaches you have. It is not known if TREXIMET® is safe and effective to treat cluster headaches.
Important Safety Information
TREXIMET® may increase your chance of a heart attack or stroke that can lead to death. Your chance of a heart attack or stroke increases with longer use of NSAID medicines or if you have heart disease or risk factors for heart disease.
Serious allergic or skin reactions, or stomach and intestine problems such as bleeding and ulcers, can occur without warning and may cause death. Risk of stomach and intestinal problems increases in the elderly.
Do not take TREXIMET® if you have heart problems, history of heart problems, or have ever had heart bypass surgery; had a stroke, TIAs, or problems with your blood circulation; hemiplegic migraines or basilar migraines; narrowing of blood vessels to your legs and arms, stomach, or kidneys; uncontrolled blood pressure; an allergy to aspirin, NSAIDs, sumatriptan or any of the ingredients in TREXIMET®; taken any medicines in the last 24 hours that are triptans or contain ergotamine; taken an MAOI antidepressant within the last 2 weeks; during third trimester of pregnancy; or liver problems. TREXIMET® should never be used if you have ever had a heart surgery called a coronary artery bypass graft (CABG).
Before you take TREXIMET®, tell your healthcare provider about all of your medical conditions including if you have risk factors for heart disease like high blood pressure, high cholesterol, diabetes, smoking, obesity, and heart problems or a family history of heart problems or stroke; kidney problems; liver problems; history of epilepsy or seizures; are pregnant, think you might be pregnant, or are trying to become pregnant; are breastfeeding or plan to breastfeed. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Serotonin syndrome is a rare but serious problem that can happen in people using TREXIMET®, especially if used with antidepressants called SSRIs or SNRIs.
The most common side effects of TREXIMET® include: dizziness; feeling weak, drowsy, or tired; pain, discomfort, or stiffness in your neck, throat, jaw, or chest; nausea; tingling or numbness in your fingers or toes; heartburn; dry mouth; feeling hot; heartbeat problems; and muscle tightness.
For more information, please see the complete Prescribing Information, including BOXED WARNINGS, and the Medication Guide at http://www.treximet.com.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
Contact:
CONTACT
Investor Relations
Sanjay Patel, 800-793-2145 ext. 1009
Chief Financial Officer
spatel@pernixtx.com
Matthew P. Duffy, 212-915-0685
LifeSci Advisors, LLC
matthew@lifesciadvisors.com
$BSSP this #2/ important milestone in our goal to develop and commercialize our immune-oncology based IPF treatment as a new therapy for difficult-to-treat tumor indications on a scalable international level initially in Mexico."
this #2 from http://www.barchart.com/headlines/story/1412302/reve-acquires-exclusive-license-for-canteck-s-ipf-irreversable-pepsin-fraction-specific-to-cancer-indication-only-for-mexico
Reve Acquires Exclusive License for Canteck's IPF Irreversable Pepsin Fraction Specific to Cancer Indication Only for Mexico
ACCESSWIRE via CMTX - 1 hr 53 mins ago
Embarks on Shift to Immunotherapeutic IPF Platform Technology for Development, Manufacture and Commercialization
SAN CLEMENTE CA / ACCESSWIRE / April 25, 2016 / Reve Technologies, Inc. (OTCPK:BSSP) (the "Company" or "Reve") a development stage technology company is pleased to announce entered into a Purchase and Sale Agreement (the "Purchase Agreement") with Canteck Pharma, Inc.,
a Delaware corporation ("Canteck" or "CKPH") and certain majority shareholders of CKPH.
Canteck has developed and patented technology
(US 8,067,531 B2) known as Irreversible Pepsin Fraction ("IPF").
IPF is a therapeutic platform technology that can be used to
facilitate a broad range of applications. It is free from neurological,
gastrointestinal and hematological side effects. IPF has not shown to be subject to viral resistance and is cost effective.
The Purchase Agreement sets forth the plan through which the Company acquires One Hundred Percent (100%) ownership Interest in Canteck Pharma, Inc. for its current and planned operations in Mexico through the exclusive Sub Licensing Agreement for Irreversible Pepsin Fraction specific to the Cancer indication only;
As part of the Agreement with Canteck, the Company, and majority shareholders of both Canteck and Reve, approved a change of control
by appointing Mrs. Diana Zhabilov, who has extensive medial based qualifications and certifications as Chairman and President,
(director) and Mr. Harry Zhabilov Jr. BSc, McS Science as Chief Scientist of the Company.
Mr. Zhabilov is responsible for development of the Patented Technology and various applications including the platform for the Irreversible Pepsin Fraction specific to the Cancer indication.
Mr. Dennis Alexander continues as Chief Executive Offer and Director, and Ms. Joanne M. Sylvanus continues as Chief Financial Officer, Secretary and Director.
It was further agreed that Valentine Dimitrov is to be appointed to
the Board of Directors after the closing of the Purchase Agreement as
a fifth Director.
The initial deployment for sales of the specific IPF for Cancer indication product and treatment programs to Mexico are now in
process with four hospitals positioned and in process of licensing requirements which have been submitted.
Canteck is positioned to oversee the development, manufacturing and commercialization processes for our lead product to be the Irreversible Pepsin Fraction (IPF) specific to the Cancer indication, with options for other indications.
"Our transition to the corporate platform for development and deployment of targeted therapeutic immunotherapeutic technology represents an important event in our transition and growth," said Dennis Alexander, CEO of the Company. "
Further, the Company welcomes our current and future development with Canteck Pharma, Inc., and working together with our new Board of Directors and Management including Mr. and Mrs. Zhabilov,
as the Company looks forward to bringing these novel technological advancements to the forefront initially to Mexico to assist treatment and solutions for specific Cancer treatment and to become available
in a cost effective manner to assist the severity of medical needs
going unmet."
"The licensing agreement with Reve Technologies, Inc. is very much
an important milestone in our goal to develop and commercialize our immune-oncology based IPF treatment as a new therapy for difficult-to-treat tumor indications on a scalable international level initially in Mexico."
Said Mr. Harry Zhabilov, Jr. BSc, McS and newly appointed Chief Sciencist of the Company.
"We are delighted to have a committed partner and management team and with our missions aligned and look forward to working together to obtain approvals for our IPF Irreversible Pepsin Fraction specific to the Cancer indication, and to bring these much-needed therapeutic
options as soon as possible for delivery and availability to Mexico."
About Canteck Pharma, Inc. IPF for Cancer treatment
Immunotherapy has the potential to provide an alternative and/or complementary treatment for most types of cancer. The advantage of immunotherapy over radiation and chemotherapy is that it can act specifically against the tumor without causing normal tissue damage.
Current data indicates that immune protection against all cancer requires the generation of a potent cellular immune response against
a unique tumor antigen expressed by the malignant cell.
As a consequence successful immune protection first requires a unique antigen expressed in the tumor cells (tumor specific antigen) and second, an induction of a potent T-cell immune response, targeted to the tumor antigen.
Unfortunately the immune system by itself can't recognize specific
tumor antigens and reject them; however recent advances have revealed that certain proteins binding with specific tumor antigens can be recognized by the immune system, this is what IPF does.
IPF proteins attach to tumor antigens, creating superantigens (Sags), which increases the number of antibodies against the malignant cells
and induces a potent T-cell immune response targeted to the tumor antigen. For a stronger immune response, IPF may be paired with different kinds of adjuvants such as IL-2, IL-6, IL-12 or other cytokines. Another form of immunotherapy can also provide active immunization, which allows for amplification of the immune response.
In addition, vaccines can generate a memory immune response.
Recent advances have revealed that any cellular protein (expressed in virally infected cells or cancer cells) can be recognized by the immune system if those proteins are presented to the immune system in a form that results in an activation rather than ignorance or tolerance to the antigen.
In addition, T-cells rather than B-cells are usually responsible for this recognition.
It is important to point out that when we discuss vaccines for cancer
we are referring to treatment rather than prevention, because the antigens expressed by tumor cells (which are the immunogens recognized by the immune system) are not yet known. Attaching known proteins will increase the number of antibodies to fight against them.
This mechanism of action will give us an exact answer
(known antigens we have to make known for immune system).
In contrast we can use vaccines to prevent infectious diseases because the antigens expressed the causative agent
- fraction and/or its proteins that can attach,
serve as the immunogen are already known.
About Reve Technologies, Inc.
The Company was incorporated on May 11, 2010 (Date of Inception) under the laws of the State of Nevada, as Bassline Productions, Inc.
On March 21, 2014 the Company amended its articles of incorporation
and changed its name to Reve Technologies, Inc. and investing to
develop and market emerging hardware, mobile and web applications later establishing a new Capital Purchase Division.
The Company is now a transitioning forward with the acquisition of an Exclusive License Agreement for Patented Technology for Irreversible Pepsin Fraction (IPF) specific to the Cancer indication only,
for Mexico with privately held immune-oncology and Therapeutics company.
Through the terms for the Company's Exclusive Sub Licensing Agreement with Canteck Pharma, Inc. we will focus on the development, manufacture and commercialize our lead product Irreversible Pepsin Fraction (IPF) specific to the Cancer indication only,
for Mexico, with options for other indications.
This release contains statements that constitute forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These statements appear in a number of places in this release and include all statements that are not statements of historical fact regarding the intent, belief or current expectations of Reve Technologies, Inc., its directors or its officers with respect to, among other things: (i) financing plans; (ii) trends affecting its financial condition or results of operations; (iii) growth strategy and operating strategy. The words "may," "would," "will," "expect," "estimate," "can," "believe," "potential" and similar expressions and variations thereof are intended to identify forward-looking statements. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties, many of which are beyond Reve Technologies, Inc.'s ability to control, and that actual results may differ materially from those projected in the forward-looking statements as a result of various factors. More information about the potential factors that could affect the business and financial results is and will be included in Reve Technologies, Inc.'s filings with the Securities and Exchange Commission.
Public Relations and Shareholder Information
Name: Dennis Alexander
Phone: (602) 326-7371
Email: revestechnology1@gmail.com
SOURCE: Reve Technologies, Inc.
http://www.accesswire.com/img.ashx?id=439165
Copyright 2016 ACCESSWIRE
$TRXC 1.95 Watching 2 See If It Gets a Technical Bounce Here Or Possibly More Downside. It Should Be Pretty Volatile Here 4 Awhile.
Shares of TransEnterix Inc. NYSEMKT: $TRXC went into free fall on Thursday morning following a devastating judgment by the U.S. Food and Drug Administration (FDA). The company announced after the markets closed on Wednesday that the FDA notified TransEnterix that it had determined that the SurgiBot System does not meet the criteria for substantial equivalence based on the data and information submitted.
Over the course of the week, the stock was down 64%. Shares of TransEnterix fell to $1.94 to close Friday. The consensus analyst price target is $4.04, and the 52-week trading range is $1.28 to $6.10.
http://247wallst.com/investing/2016/04/24/4-companies-that-have-destroyed-shareholders/
$NXTD The deal also de-risks the story by a fair amount. The product still has to be delivered in volume, but so far the progress has been good and is on schedule. We acknowledge that there is always a possibility that things will slip, but it's far from an "invention needed" type of situation.
At least one additional deal is highly probable in 2016, and two or more are possible. That would put NXT-ID in a very comparable position to LoopPay and other technology properties that have been acquired. Based on prior deals, that would suggest something on the order of $150 to $250M versus the current $50M market value. $NXTD #wocket #wocketwallet

http://stockpumpers.blogspot.com/2016/04/soundview-technology-group-issues-nxt.html
$LEGX and SelectaDNA/CSI Protect Push Boundaries in the Use of DNA
https://finance.yahoo.com/news/legacyxchange-selectadna-csi-protect-push-120000919.html
|
Followers
|
53
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1933
|
|
Created
|
12/26/11
|
Type
|
Free
|
| Moderators | |||
Why Penny Stocks?
There really is only one reason to invest in penny stocks, and that reason is penny stocks offer the best chance of Enormous Profits in the stock market! Stocks that trade on the NYSE and NASDAQ just do not offer the same opportunities to book large profits, as penny stocks do. The key is for individual investors to take the initiative to learn how to Trade, Invest, and Prosper from the Plentiful Profit Opportunities that penny stocks offer! A 100% Gain (or a double) is considered a Home Run when investing in NYSE and NASDAQ stocks.
While 100% is a nice gain, only penny stocks offer on the opportunity to make 1,000% Plus Grand Slam Gains (ten bagger), which is why so many people invest in penny stocks! Penny Stock Prophecy takes the guessing game out of penny stock trading and finds the penny stocks that have the potential to make Big Gains
Many people believe in scouring through penny stocks in hope of finding the next Google or Microsoft...
But this is unlikely to ever pay off
Although there are many companies that have allot of potential trading penny stocks it is a common misconception that all the Large companies like Microsoft and Google started Started out as penny stocks. Most of these companies started out trading at the $25-$30 mark.
What are Penny Stocks?
The definition of a penny stock varies According to the Securities & Exchange Commission (SEC) any stock under $5 is a penny stock others set the cut-off point at $3, while some consider only stocks trading at less than $1 are penny stocks or the any stock trading on the Pink Sheets or OTCBB to be a penny stock.
What are the Risks
Penny stocks are incredible risky to trade in, although just a small increase of a few cents can potentially triple your investment, a few cents in the opposite direction could just as well cripple you financially. That's why its important to know your risk tolerance before you invest, just take a look at some of the risks when trading in penny stocks
The Lack of Information Revealed to the Public
Because companies on the Pink Sheets don't have to register with the Securities & Exchange Commission (SEC) enough company history is not always available to the public as is needed to make an informed decision when purchasing stock unlike on the NYSE and the NASDAQ exchanges. To make a correct decision enough reputable information needs to be available to the trader as possible.
Lack of Trading History
Not knowing the trading history only multiplies the difficulties to picking the correct stock.
Lack of Liquidity
Liquidity is the The degree to which an asset.
or security can be bought or sold in the market without affecting the asset's price. Penny stocks are known for there erratic movements these can either make or break an investment.
Biased recommendations
Some companies will pay for people to make fake recommendations, Look to see if the author of the recommendation is being paid for making this recommendation as this is a giveaway of a bad pick and make sure that any press releases aren't authored by people looking solely to influence the price of a stock.
These are just some of the potential risks investors face when looking for profitable penny stocks.
But of course with all these risks come great benefits too. A well chosen and painfully analyzed stock can increase dramatically within minutes of the market opening taking you with it.
There are also allot of new companies who have just started trading as penny stocks, some of these companies are working extremely hard to make their way up to the more reputable NASDAQ and NYSE and buying in these stocks can make you very nice profit as long as you make sure your covered for all possible eventuality and you have got all the information you can about the stock.
MicrocapMarkets NASDAQ <$5 - http://www.microcapmarkets.com/data_main_nav.jsp?market=NASDAQ
MicrocapMarkets OTCBB - http://www.microcapmarkets.com/data_main_nav.jsp?market=OTCBB
Mining - http://www.miningmx.com/
MSN Money Central - http://www.moneycentral.msn.com/investor/home.asp
Multicollinearity - www.stockcharts.com/help/doku.php
Mutual Fund Facts About Individual Stocks - http://www.mffais.com/
Naked Shorting - www.businessjive.com/
NASDAQ Stock Market - http://www.nasdaq.com/
NASDAQ/NYSE/AMEX Stock Info - www.secfilings.com/
Natural Resources News & Commentary - http://www.resourceinvestor.com/Pages/default.aspx
New York Stock Exchange - http://www.nyse.com/home.html
OTCBB Daily List - http://www.otcbb.com/dailylist/
Patterns - http://thepatternsite.com/
Pinksheets Stock Info - www.otcmarkets.com/pink/index.jsp
Platform - http://www.quotetracker.com/
Precious Metals - http://www.kitco.com/
Press Releases - http://www.prnewswire.com/news-releases/
REG SHO List - www.regsho.com/tools/short_list.php
Sites to help you find the penny stocks.
http://www.stockhouse.com/mostactives/index.asp?type=winners&exchange=OTCBB http://www.allstocks.com/pennystocks/
http://www.smallcapcenter.com/
http://www.smallcapcenter.com/tools_technicalSearch.asp?page=ANALYTICSSEARCH_IN.ASP
http://knobias.10kwizard.com/main.php http://www.otcbb.com/dynamic/marketstats.htm
E-book on penny stocks. http://pennytraders.netfirms.com/
Here is where you get the highest percent gainers from Pink Sheets. http://www.pinksheets.com/marketactivity/trade_stats.jsp
Here is a great place to learn how to set up scans. http://yepher.com/~yepher/stockfetcher/command.html
Stock Screener: http://screener.finance.yahoo.com/stocks.html
Another Stock Screener (paid subscription required): http://www.stockfetcher.com/
Minute to minute trades http://data.island.com/ds/tools/timeandsales/index.jsp?rows=100&width=555&links=false&ht

| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
