Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Duh...................
It went private. Maybe you didn't get your check because they lost your address. LOLOLOL!
ACEL is listed as a private company. What does that mean? Have the minor shareholders like myself been effectively eliminated and pushed aside? Appreciate response from anyone in the know/ Rg
ACEL: SEC Admin Law Judge Initial Decision to revoke ACEL's Registration.
http://www.sec.gov/alj/aljdec/2013/id488ce.pdf
Updates on ACEL? Tried selling shares and wouldn't go through, anyone have any news on what is going on?
ACEL SEC Suspension:
http://www.sec.gov/litigation/suspensions/2013/34-68726.pdf
ORDER:
http://www.sec.gov/litigation/suspensions/2013/34-68726-o.pdf
Admin Proceeding:
http://www.sec.gov/litigation/admin/2013/34-68727.pdf
ACEL .0149 515kvs61k_vol10dayavg Up 148% http://stockcharts.com/c-sc/sc?s=ACEL&p=d&b=7&g=0&id=p17970548896
ACEL may come off bottom with this 8-K filing @ 5:30pm on 12/15/2011...
Item 1.01 Entry into Material and Definitive Agreement
Entry into a Memorandum of Understanding
On November 23, 2011, Tamir Biotechnology, Inc (formerly Alfacell Corporation) hereafter (Tamir) entered into a Memorandum of Understanding (MOU), with US Pharmacia hereafter (USPI and/or affiliates), pursuant to which USPI, will be granted the exclusive marketing, sales, and distribution rights of ONCONASE® for use in Human Papillioma Virus (HPV) in the European Territory upon completion of full License payment. Territory includes Western and Eastern Europe. USPI shall pay Tamir the sum of $1,000,000.00 (one million dollars) in installments provided Tamir achieves certain milestones. Under the MOU, on December 12, 2011, Tamir received an initial payment of $405,000.00 (four hundred five thousand dollars) and Tamir shall provide USPI with Proof of Concept (POC) demonstrating successful formulation drug formulation of ONCONASE® which will be used to treat genital and skin warts caused by the HPV and in vivo penetration of Onconase into the skin. Tamir shall provide this POC by March 31, 2011. If successful, USPI shall pay Tamir balance of $595,000.00 in installments once other pre-clinical milestones have been met.
The terms of the License Agreement shall be agreed upon by the parties as outlined in the MOU. Tamir will also be entitled to receive milestone payments based on the achievement of certain regulatory approvals and certain sales goals. In addition, Tamir will receive a royalty on net sales as well as a transfer price for product sold by Tamir to USPI.
Tamir will be responsible for making regulatory filings with and seeking marketing approval of ONCONASE® in the Territory and manufacturing and supplying ONCONASE® to USPI. USPI will be responsible for all commercial activities and related costs in the Territory.
Link > ACEL 8-K
ACEL L2 screenshot:
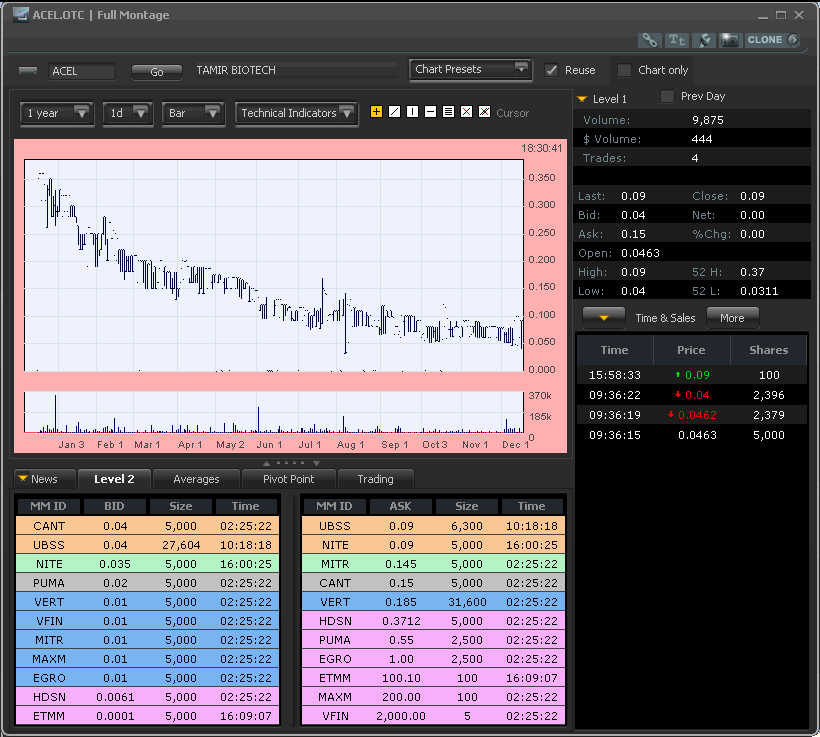
I dont know. I liked the chart more. No position.
Might look to get back in.
Noticed it dropped around .35 the other day
The chart is bearish and there will probably more some more downside
Looking for .16 - .23 entry
JMO
GL
When do you expect new relevant news on the company. And in which phase are they at the moment?
Tamir Biotechnology Filed Resale Registration Statement as Required by the Investor Rights Agreement in a Previously Closed Private Placement
SOMERSET, N.J., May 3, 2010 (GLOBE NEWSWIRE) -- Tamir Biotechnology, Inc. (formerly known as Alfacell Corporation) (OTCQB:ACEL) filed a resale registration statement on Form S-1 on April 30, 2010 with the Securities and Exchange Commission to register all the shares of common stock issuable upon conversion of the notes and exercise of the warrants that were previously sold by the company in a $3.25 million private placement closed in October 2009. As previously announced by the company, according to an Investor Rights Agreement with the investors in the private placement, the company is required to file a resale registration statement to cover the shares underlying the notes and warrants sold by the company in the private placement. Such registration rights of investors are customary in similar private placement transactions, and the filing of the registration statement does not necessarily indicate whether any investor intends to convert the notes, exercise the warrants, or sell the common stock issuable upon such conversion or exercise.
The shares covered by the registration statement include the following: (i) 24,916,667 shares of common stock that will be issued upon conversion of the notes, (b) 21,666,664 shares of common stock that will be issued upon the exercise of the Series A Warrants at $0.15 per share, and (c) 21,666,664 shares of common stock that will be issued upon the exercise of the Series B Warrants at $0.25 per share. The selling stockholders named in the resale registration statement are the investors who participated in the October 2009 private placement, including Charles Muniz, the company's President, Chief Executive Officer Chief Financial Officer, and several other existing investors of the company, including trusts and individuals related to James O. McCash, Europa International Inc. and Unilab LP, an affiliate of US Pharmacia. The investors will not be eligible to sell the shares without any restrictions until the shares are issued upon the conversion of the notes and the exercise of the warrants and until the registration statement is effective or an exemption from the registration requirement is otherwise available.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy any of the securities described herein, nor shall there be any sales of these securities in any jurisdiction in which such an offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction. The securities issued in the private placement have not been effectively registered under the Securities Act, or any state securities laws and were sold in reliance on Section 4(2) of the Securities Act. Accordingly, the securities acquired in the private placement may not be offered or sold in the United States absent registration with the SEC or an applicable exemption from the registration requirements of the Securities Act.
About Tamir Biotechnology, Inc.:
Tamir Biotechnology, Inc. (formerly known as Alfacell Corporation) is the first company to advance a biopharmaceutical product candidate that works in a manner similar to RNA interference (RNAi) through late-stage clinical trials. The product candidate, ONCONASE, is an RNase that overcomes the challenges of targeting RNA for therapeutic purposes while enabling the development of a new class of targeted therapies for cancer and other life-threatening diseases.
For more information regarding the company, including materials from its recent annual shareholders meeting and video messages to shareholders, please visit www.alfacell.com under the heading "Information Center."
CONTACT: Tamir Biotechnology
Charles Muniz, President, CEO and CFO
732-652-4540
looks like insiders need to unload before delisting
or the do a r/v to stay alive?
not a good investment,imo
What do you know about ACEL here? Is this just a technical bounce, or are all the new shares done and some sort of program is underway? If they aren't careful they will get delisted.
either a shake out or insiders know something bad/not so good?
Results not the greatest perhaps and maybe its still 50-50 on approval. Yet, the insider who is resigning next yr has alot of shares to sell (on the news) or?
Anyone know who sold to get it down or was it shorts?
I am in this as well -- the recovery may take a few weeks, but I am sticking it out.
Today's Events - Bouncing back...
This was posted on another message board:
Getting back to the value of this stock
Based on the early morning conference call and webcast - IMO - the drug works; the clinicals will continue; and the partner PRX is positive, and is up for the day.
The weak hands that knew nothing about this drug just ran for the hills - thus putting this stock price on sale for the next few days.
Those that sold after hours at .60 are already probably wishing that they waited.
I didn't wait. I bought more, averaging down substantially. Thanks for today's sale.
IMO, especially at today's prices - this is a big money maker....
It would have been nice to make big $$$ today -- but all is not lost -- just delayed....
Sidransky's other company - CSBR:
LINK:
http://www.championsbiotechnology.com/ne...
Champions Biotechnology and ImClone Systems Establish Agreement for Preclinical Evaluation of Oncology Drugs
Last update: 8:30 a.m. EDT May 7, 2008Print E-mail RSS Disable Live Quotes
ARLINGTON, Va., May 7, 2008 /PRNewswire-FirstCall via COMTEX/ -- Champions Biotechnology, Inc. (CSBR:CSBR
News, chart, profile, more
CSBR, , ) , a company engaged in the development of advanced preclinical platforms and tumor specific data to enhance the value of oncology drugs, today announced that it has established an agreement with ImClone Systems Incorporated for the preclinical evaluation of certain therapeutic antibodies in ImClone's clinical development pipeline. As part of the agreement, ImClone will utilize Champions Biotechnology's Biomerk Tumorgrafts(TM) in the initial preclinical evaluation.
"Studies suggest that our Biomerk Tumorgrafts provide an accelerated and more predictive platform for the evaluation of oncology drugs. The platform also enables biomarker discovery that we expect will benefit us as we facilitate partnerships between therapeutic and diagnostic companies," said Doug Burkett, Ph.D., President of Champions Biotechnology, Inc. "ImClone Systems has an impressive antibody pipeline and we are pleased to be working with them to evaluate such promising oncology therapeutic candidates."
For more information regarding Champions Biotechnology's growing business and recent news, please visit www.championsbiotechnology.com .
About Champions Biotechnology, Inc.
Champions Biotechnology, Inc. is engaged in the development of advanced preclinical platforms and predictive tumor specific data to enhance and accelerate the value of oncology drugs. The Company's Preclinical Platform is a novel approach based upon the implantation of primary human tumors in immune deficient mice followed by propagation of the resulting xenografts (Biomerk Tumorgrafts(TM)) in a manner that preserves the biological characteristics of the original human tumor. The Company believes that these Tumorgrafts closely reflect human cancer biology and their response to drugs is more predictive of clinical outcomes in cancer patients.
Champions Biotechnology leverages its preclinical platform to evaluate drug candidates and to develop a portfolio of novel therapeutic candidates through pre-clinical trials. As drugs progress through this early stage of development, the Company plans to sell, partner or license them to pharmaceutical and/or biotechnology companies, as appropriate. The Company also offers its predictive preclinical platform and tumor specific data to physicians for personalized patient care. In the course of this process, this permits Champions Biotechnology to evaluate oncology drugs for companies by using models that integrate prognostic testing with biomarker discovery.
Champions Biotechnology is dedicated to enhancing preclinical development tools, accelerating development and valuation of oncology drugs, and advancing personalized treatment with a goal to improve the lives of cancer patients globally.
ACEL Announces Retirement of Chief Executive Officer
http://www.snl.com/irweblinkx/news.aspx?IID=4104784
Press Release ALFACELL CORPORATION (NASDAQ - ACEL)
Alfacell Announces Retirement of Chief Executive Officer
Company Release - 04/28/2008 08:30
SOMERSET, N.J., April 28 /PRNewswire-FirstCall/ -- Alfacell Corporation (Nasdaq: ACEL) today announced that Kuslima Shogen, the company's chief executive officer and scientific founder, will retire from the company on or before March 31, 2009. The company also announced that the royalty agreement between Ms. Shogen and Alfacell has been terminated.
The termination of the royalty agreement between Alfacell and Ms. Shogen could reduce payments from the Company to Ms. Shogen by an estimated $20 million over the next 10 years, if the company's lead compound, ONCONASE(R), is granted marketing approval by the U.S. Food and Drug Administration (FDA).
Under the terms of the retirement agreement between the company and Ms. Shogen, Ms. Shogen will continue to receive her current salary for a period of two years after she steps down as chief executive officer. In exchange for the termination of the royalty agreement, Ms. Shogen will receive a cash bonus, an additional cash bonus upon receiving marketing approval by the FDA of ONCONASE for the treatment of malignant mesothelioma, a significant stock option grant that vests upon such FDA approval of ONCONASE, cash payments for the first two years following approval of ONCONASE, and cash payments from Alfacell in the event aggregate annual net sales of ONCONASE exceed $100 million.
"Tina's contributions to Alfacell and the science behind ONCONASE have been immeasurable," said David Sidransky, M.D., Alfacell's chairman. "Her dedication and commitment are unmatched and I know that I speak for everyone at Alfacell in offering congratulations as she moves on to a well-earned retirement."
In addition to the retirement of Ms. Shogen, the company announced that Lawrence A. Kenyon has been named president. Mr. Kenyon joined Alfacell in January 2007 as executive vice president, chief financial officer and corporate secretary. He was promoted to chief operating officer and elected to the board of directors in November 2007. The change in responsibilities is consistent with the efforts of the board of directors to develop a succession plan that allows for a smooth leadership transition upon the potential marketing approval of ONCONASE.
"It is a comfort for me to entrust the further development and potential commercialization of our exciting pipeline to Mr. Kenyon," said Ms. Shogen. "It has been a privilege to be part of Alfacell where I have experienced the rare opportunity to make an important scientific contribution and achieve the realization of my life's work."
"Our focus has begun to shift towards the necessary steps to grow Alfacell and maximize value for our shareholders," added Mr. Kenyon. "The termination of the royalty agreement will allow us to enhance our ability to increase investment in our promising technology platform. We are excited about the future of Alfacell, and I would like to congratulate Tina and commend her for the sound scientific foundation she has built for us."
About ONCONASE(R)
ONCONASE is a first-in-class therapeutic product candidate based on Alfacell's proprietary ribonuclease (RNase) technology. A natural protein isolated from the leopard frog, ONCONASE has been shown in the laboratory and clinic to target cancer cells while sparing normal cells. ONCONASE triggers apoptosis, the natural death of cells, via multiple molecular mechanisms of action.
About Alfacell Corporation
Alfacell Corporation is the first company to advance a biopharmaceutical product candidate that works in a manner similar to RNA interference (RNAi) through late-stage clinical trials. The product candidate, ONCONASE, is an RNase that overcomes the challenges of targeting RNA for therapeutic purposes while enabling the development of a new class of targeted therapies for cancer and other life-threatening diseases. In addition to a Phase IIIb study in malignant mesothelioma, Alfacell is conducting a Phase I/II trial of ONCONASE in non-small cell lung cancer (NSCLC) and other solid tumors. For more information, visit www.alfacell.com.
Safe Harbor
This press release includes statements that may constitute "forward-looking" statements, usually containing the words "believe," "estimate," "project," "expect" or similar expressions. Forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from the forward-looking statements. Factors that would cause or contribute to such differences include, but are not limited to, uncertainties involved in the outcome of the company's ongoing Phase IIIb clinical trial for its lead product, the company's ability to secure necessary approvals from regulatory agencies, uncertainties involving the ability of the company to finance research and development activities and its ongoing operations, potential challenges to or violations of patents, dependence upon third-party vendors, and other risks discussed in the company's periodic filings with the Securities and Exchange Commission. By making these forward-looking statements, the company undertakes no obligation to update these statements for revisions or changes after the date of this release.
Media Contact: Investor Contact:
David Schull or Wendy LauTimothy EngelRusso PartnersRusso Partners
212-845-4271 212-845-4242
David.Schull@russopartnersllc.comTimothy.Engel@russopartnersllc.comWendy.Lau@russopartnersllc.com
SOURCE Alfacell Corporation
ACEL Team Receives Prestigious Reward
The Landon Foundation-AACR INNOVATOR Award for International Collaboration in Cancer Research at the American Association for Cancer Research (AACR) Annual Meeting in San Diego.
We are pleased to inform you that Michele Carbone, M.D., Ph.D., Alfacell’s chairman of the thoracic cancer advisory board (TCAB) and a world-renowned mesothelioma researcher at the University of Hawaii’s Cancer Research Center of Hawaii, Harvey I. Pass, M.D., a member of Alfacell’s TCAB and a professor of surgery and chief of the division of thoracic surgery and thoracic oncology at NYU School of Medicine and Comprehensive Cancer Center, and their team, were presented The Landon Foundation-AACR INNOVATOR Award for International Collaboration in Cancer Research at the American Association for Cancer Research (AACR) Annual Meeting in San Diego.
The team of international collaborators has discovered a unique mesothelioma epidemic in three Turkish villages and has demonstrated that it is caused by a genetic predisposition to mineral fiber carcinogenesis, a gene-environment interaction. These findings and additional research may be applied to understand other geographic areas and communities worldwide to help prevent this deadly form of cancer and help find new life-saving treatments.
The AACR INNOVATOR grant (two-year $100,000 grant) will be applied to additional research for their study.
We, here at Alfacell, congratulate Drs. Carbone and Pass, and their team on receiving this prestigious award and look forward to their continued support with research on the ONCONASE® (ranpirnase) program and mesothelioma.
Please access the news release by going to: http://www.aacr.org/home/public--media/news-releases.aspx?d=1027
Lawrence A. Kenyon
COO and CFO
Alfacell Corporation
Press Release: Onconase by ACEL could be a radiation sensitizer
Company Release - 04/14/2008 08:30
link:
http://www.snl.com/irweblinkx/news.aspx?IID=4104784
SOMERSET, N.J., April 14 /PRNewswire-FirstCall/ -- Alfacell Corporation (Nasdaq: ACEL) today announced that Dr. Intae Lee with the University of Pennsylvania has reported that Alfacell's ONCONASE (ranpirnase) could be a promising radiation sensitizer for lung cancer treatment. Dr. Lee presented the pre-clinical in vivo and in vitro data in a poster at the 2008 American Association of Cancer Research (AACR) Annual Meeting being held April 12 - 16 in San Diego.
In the poster titled "The inhibition of radiation repair by ranpirnase +/- I-buthionine sulfoximide on lung cancer," Dr. Lee provided pre-clinical evidence that ONCONASE +/- I-buthionine sulfoximide significantly increased the radiation-induced growth delay of lung tumors in vivo without increases in skin reaction, compared to radiation alone. Additionally, in vivo and in vitro data presented indicated that ONCONASE significantly increased apoptosis (programmed cell death) in several human non-small cell lung cancer (NSCLC) cell lines (A549, NCI-H1975 and HOP-62). Dr. Lee and his team of researchers also showed that the radiation repair mechanisms known as sub-lethal damage repair (SLDR) and potentially lethal damage repair (PLDR), which lead to radiation resistance in tumors, were significantly inhibited by ONCONASE in vitro.
"Our research suggests potential utility of ONCONASE as a radiotherapy enhancer for the treatment of NSCLC patients," said Dr. Lee. "It is important to overcome PLDR as it can ultimately lead to radiation resistance. One of our key findings is the inhibiting impact of ONCONASE on the PLDR mechanism, which is a pre-requisite to developing a radiotherapy enhancer."
Kuslima Shogen, Alfacell's chief executive officer, added: "This work by Dr. Lee and his team provides further evidence of the potential for ONCONASE to mitigate the resistance that often develops in, and confounds the treatment of NSCLC."
About ONCONASE(R)
ONCONASE is a first-in-class product candidate based on Alfacell's proprietary ribonuclease (RNase) technology. A natural protein isolated from the leopard frog, ONCONASE has been shown in the laboratory and clinic to target cancer cells while sparing normal cells. ONCONASE triggers apoptosis, the natural death of cells, via multiple molecular mechanisms of action.
About Alfacell Corporation
Alfacell Corporation is the first company to advance a biopharmaceutical product candidate that works in a manner similar to RNA interference (RNAi) through late-stage clinical trials. The product candidate, ONCONASE, is an RNase that overcomes the challenges of targeting RNA for therapeutic purposes while enabling the development of a new class of targeted therapies for cancer and other life-threatening diseases. In addition to an ongoing Phase IIIb study in malignant mesothelioma, Alfacell is conducting a Phase I/II trial of ONCONASE in non-small cell lung cancer (NSCLC) and other solid tumors. For more information, visit www.alfacell.com.
Safe Harbor
This press release includes statements that may constitute "forward-looking" statements, usually containing the words "believe," "estimate," "project," "expect" or similar expressions. Forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from the forward-looking statements. Factors that would cause or contribute to such differences include, but are not limited to, uncertainties involved in transitioning from concept to product, uncertainties involving the ability of the company to finance research and development activities, potential challenges to or violations of patents, uncertainties regarding the outcome of clinical trials, the company's ability to secure necessary approvals from regulatory agencies, dependence upon third-party vendors, and other risks discussed in the company's periodic filings with the Securities and Exchange Commission. By making these forward-looking statements, the company undertakes no obligation to update these statements for revisions or changes after the date of this release.
Media Contact: Investor Contact:
David Schull or Wendy LauTimothy EngelRusso PartnersRusso Partners
212-845-4271 212-845-4242
David.Schull@russopartnersllc.comTimothy.Engel@russopartnersllc.comWendy.Lau@russopartnersllc.com
SOURCE Alfacell Corporation
shorts will cover ,big pop coming ,undervalued plus some.
worth 7-10 bucks minimal.
Cover Story is out on ACEL's Onconase:
THIS IS THE DRUG DISCOVERY & DEVELOPMENT MAGAZINE COVER STORY - JUST OUT
Here is the link:
http://www.dddmag.com/Article-Conquering-Cancer-Resistance.aspx
ACEL Press Release
link:
http://www.snl.com/irweblinkx/file.aspx?IID=4104784&FID=5853286
Press Release ALFACELL CORPORATION (NASDAQ - ACEL)
Alfacell Updates Status of ONCONASE(R) Phase III Clinical Program
Company Release - 04/02/2008 08:30
SOMERSET, N.J., April 2, 2008 /PRNewswire-FirstCall/ -- Alfacell Corporation (Nasdaq: ACEL) today announced that it has confirmed that 316 evaluable events (patient deaths) have occurred in the confirmatory Phase IIIb clinical trial of its lead compound, ONCONASE (ranpirnase), for the treatment of patients with unresectable malignant mesothelioma (UMM). In accordance with the statistical plan for the trial, the company has begun the process necessary to conduct the formal statistical analyses required to complete the final sections of the ONCONASE rolling New Drug Application (NDA).
The trial was designed to show a statistically significant improvement in overall survival for UMM patients who were treated with a combination of ONCONASE and doxorubicin as compared to UMM patients who were treated with doxorubicin as a single agent. Enrollment in the ONCONASE Phase IIIb clinical trial closed on Sept. 30, 2007. A total of 428 patients were enrolled in the trial.
Alfacell has licensed the U.S. commercial rights for ONCONASE to Strativa, the branded product division of Par Pharmaceuticals, Inc. Strategic marketing and distribution agreements for ONCONASE have been secured with BL&H Co. Ltd. for Korea, Taiwan and Hong Kong, USP Pharma Spolka Z.O.O., an affiliate of US Pharmacia, for Eastern Europe, and GENESIS Pharma, S.A. for Southeastern Europe.
ONCONASE has been granted fast track status and orphan-drug designation for the treatment of malignant mesothelioma by the U.S. Food and Drug Administration (FDA). Additionally, ONCONASE has been granted orphan-drug designation in the European Union and Australia.
About ONCONASE(R)
ONCONASE is a first-in-class product candidate based on Alfacell's proprietary ribonuclease (RNase) technology. A natural protein isolated from the leopard frog, ONCONASE has been shown in the laboratory and clinic to target cancer cells while sparing normal cells. ONCONASE triggers apoptosis, the natural death of cells, via multiple molecular mechanisms of action.
About Alfacell Corporation
Alfacell Corporation is the first company to advance a biopharmaceutical product candidate that works in a manner similar to RNA interference (RNAi) through late-stage clinical trials. The product candidate, ONCONASE, is an RNase that overcomes the challenges of targeting RNA for therapeutic purposes while enabling the development of a new class of targeted therapies for cancer and other life-threatening diseases. In addition to the Phase IIIb study in malignant mesothelioma, Alfacell is conducting a Phase I/II trial of ONCONASE in non-small cell lung cancer (NSCLC) and other solid tumors. For more information, visit www.alfacell.com.
Safe Harbor
This press release includes statements that may constitute "forward-looking" statements, usually containing the words "believe," "estimate," "project," "expect" or similar expressions. Forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from the forward-looking statements. Factors that would cause or contribute to such differences include, but are not limited to, uncertainties involved in transitioning from concept to product, uncertainties involving the ability of the company to finance research and development activities, potential challenges to or violations of patents, uncertainties regarding the outcome of clinical trials, the company's ability to secure necessary approvals from regulatory agencies, dependence upon third-party vendors, and other risks discussed in the company's periodic filings with the Securities and Exchange Commission. By making these forward-looking statements, the company undertakes no obligation to update these statements for revisions or changes after the date of this release.
Media Contact: Investor Contact:
David Schull or Wendy LauTimothy EngelRusso PartnersRusso Partners
212-845-4271 212-845-4242
David.Schull@russopartnersllc.comTimothy.Engel@russopartnersllc.comWendy.Lau@russopartnersllc.com
SOURCE Alfacell Corporation
Contact: Media, David Schull, David.Schull@russopartnersllc.com, or Wendy Lau, Wendy.Lau@russopartnersllc.com, +1-212-845-4271; or Investors, Timothy Engel, +1-212-845-4242, Timothy.Engel@russopartnersllc.com, all of Russo Partners
Click here for Printer-Friendly Version
ACEL NEWS is out this morning!
ACEL completes its P3B FDA trial for ACEL's cancer drug ONCONASE!
The 7 year FDA final phase III B trial is now complete!
From this point on events will now move very fast to submitting the NDA and waiting for "fast-track" and "orphan drug" statuses FDA approval.
Things should get very exciting from here now.
ACEL News
Alfacell Updates Status of ONCONASE(R) Phase III Clinical Program
Apr 2, 2008 8:30:00 AM
SOMERSET, N.J., April 2, 2008 /PRNewswire-FirstCall/ -- Alfacell Corporation (Nasdaq: ACEL) today announced that it has confirmed that 316 evaluable events (patient deaths) have occurred in the confirmatory Phase IIIb clinical trial of its lead compound, ONCONASE (ranpirnase), for the treatment of patients with unresectable malignant mesothelioma (UMM). In accordance with the statistical plan for the trial, the company has begun the process necessary to conduct the formal statistical analyses required to complete the final sections of the ONCONASE rolling New Drug Application (NDA).
The trial was designed to show a statistically significant improvement in overall survival for UMM patients who were treated with a combination of ONCONASE and doxorubicin as compared to UMM patients who were treated with doxorubicin as a single agent. Enrollment in the ONCONASE Phase IIIb clinical trial closed on Sept. 30, 2007. A total of 428 patients were enrolled in the trial.
Alfacell has licensed the U.S. commercial rights for ONCONASE to Strativa, the branded product division of Par Pharmaceuticals, Inc. Strategic marketing and distribution agreements for ONCONASE have been secured with BL&H Co. Ltd. for Korea, Taiwan and Hong Kong, USP Pharma Spolka Z.O.O., an affiliate of US Pharmacia, for Eastern Europe, and GENESIS Pharma, S.A. for Southeastern Europe.
ONCONASE has been granted fast track status and orphan-drug designation for the treatment of malignant mesothelioma by the U.S. Food and Drug Administration (FDA). Additionally, ONCONASE has been granted orphan-drug designation in the European Union and Australia.
About ONCONASE(R)
ONCONASE is a first-in-class product candidate based on Alfacell's proprietary ribonuclease (RNase) technology. A natural protein isolated from the leopard frog, ONCONASE has been shown in the laboratory and clinic to target cancer cells while sparing normal cells. ONCONASE triggers apoptosis, the natural death of cells, via multiple molecular mechanisms of action.
About Alfacell Corporation
Alfacell Corporation is the first company to advance a biopharmaceutical product candidate that works in a manner similar to RNA interference (RNAi) through late-stage clinical trials. The product candidate, ONCONASE, is an RNase that overcomes the challenges of targeting RNA for therapeutic purposes while enabling the development of a new class of targeted therapies for cancer and other life-threatening diseases. In addition to the Phase IIIb study in malignant mesothelioma, Alfacell is conducting a Phase I/II trial of ONCONASE in non-small cell lung cancer (NSCLC) and other solid tumors. For more information, visit www.alfacell.com.
ACEL Short interest 2,289,700 as of this morning
link:
http://www.shortsqueeze.com/index.php?symbol=acel&submit=Enter
Alfacell Corp. $ 2.30
ACEL -0.15
Short Interest (Shares Short) 2,289,700
Days To Cover (Short Interest Ratio) 20.1
Short Percent of Float 5.86 %
Short Interest - Prior 2,300,200
Short % Increase / Decrease -0.46 %
Short Squeeze Ranking™ 76
% From 52-Wk High ($ 3.74 ) -62.61 %
% From 52-Wk Low ($ 1.32 ) 42.61 %
% From 200-Day MA ($ 2.00 ) 13.04 %
% From 50-Day MA ($ 2.23 ) 3.04 %
Price % Change (52-Week) 64.30 %
Shares Float 39,070,000
Total Shares Outstanding 47,066,880
% Owned by Insiders 22.96 %
% Owned by Institutions 16.40 %
Market Cap. 108,253,824
Trading Volume - Today 132,620
Trading Volume - Average 113,900
Trading Volume - Today vs. Average 116.44 %
Earnings Per Share -0.19
PE Ratio
Record Date 2008-MarB
Sector Healthcare
Industry Biotechnology
Data Provided Without Warranty
Mission Statement
The mission of ShortSqueeze.com TM is to provide short interest stock market data and services, so our members will be better informed of short selling in the market, track shorts in stocks and gain from the advantages that can be achieved from this valuable market data.
Sponsor
Compare discount rates from top companies
It's fast and it's free!
Save up to 43% now...
Enter Zip Code:
Congratulations skyyy...you've been calling this one right on target all along. Your insights on the short attack, when ACEL was over-sold and your TA throughout has been great. I'm following you on this one all the way to a new 52-week high ($3.74). I'm back in buying more...IMO it's poised to take off!
ACEL($2.45) looks like it is ready to breakout and run.
It had a range of $2.19 to $2.45 today and closed at the HOD($2.45).
The shorts have started to slowly cover. Any move above $2.68 should trigger a run to $3.74 quickly ... IMO.
Nice to be long here.
The cover story about ACEL's cancer drug "Onconase" in DRUG DISCOVERY Magazine ... is due to be released on this friday April 28. That strong exposure should help to accelerate the move.
The Chart Is Ready
I expect we shall see the move we've all been waiting all these weeks for any day now, maybe today.
The downtrending top pennant line on the Weekly MACD has been broken and this is very significant.
fringe
skyyy....I think the move on ACEL is simply the shorts getting out of the way. This stock has wanted to go higher for weeks and has been under a fierce highly organized short attack, which seems to be over. There must be around 2.5 million short shares out there. As the stock moves higher covering the shorts will fuel a strong rally IMO...
fringe...ACEL($2.44) was up .15 today on growing volume of 271,000+ shares.
How is ACEL's chart looking to you now?
What is your TA read here now?
Many thanks in advance.
Post Marked -e-
COVER STORY - ACEL'S ONCONASE
APRIL ISSUE OF DRUG DISCOVERY & DEVELOPMENT
LINK TO SUBSCRIBE TO THE ARTICLE:
http://www.dddmag.com/DDD-upcoming-issues.aspx?menuid=54
April, 2008 Issue
This issue is free to all that register to receive it. This issue will be out shortly.
The April issue features ACEL's Onconase as a Front Page Cover Story.
Brief description of how ACEL's Onconase works
link:
http://www.mesotheliomasos.com/treatmentOnconase.php
Onconase (ranpirnase)
Currently in clinical trials, Onconase, manufactured by Alfacell Corporation, offers hope to patients with unresectable malignant mesothelioma. Developed from the eggs of the leopard frog, Onconase is a ribonuclease protein that is designed to enhance the anti-cancer effects of traditional chemotherapy, allowing lower doses to be effective and causing fewer adverse side effects. Though Onconase works slowly, its lower toxicity levels mean no loss of hair, anemia, or nausea usually associated with standard chemo drugs.
Technically speaking, Onconase slows down cancer cell growth by decaying RNA. Without certain RNA strands, cancer cells cannot produce certain essential proteins and, therefore, cannot replicate. This serves to slow the growth of the tumor.
While Phase III trials are currently underway, treating approximately 300 patients that have been stricken with inoperable malignant mesothelioma, the results of Phase II trials, released in 2002, demonstrated that “the tumors either shrank or stopped growing in 41 of the 81 patients that were assessable for tumor response.” The median survival of the patients who entered the trial without symptoms from the cancer was 18.5 months.
Onconase is also one of the first embryonic stem cell products to reach the final stages of testing. Doctors hope to soon be using Onconase in place of Doxorubicin, a chemotherapy drug with crippling side effects that have a serious effect on the patient’s quality of life.
Looking ahead on ACEL....When ACEL gets FDA approval, which IMO should be within a year, it should run to at least $40. Given contracts already in place they will have earnings of $2/share...at a PE of 30 the stock is between $40 and $60......To illustrate a recent comparison, in August, 2006 ACOR's stock price was $2. Two months later the stock price was $22. It rose to a high of roughly $27 in May, 2007 and is currently trading over $17.
What moved the stock price so dramatically from $2 to over $20 in the fall of 2006? On September 26, 2006 ACOR issued a press release reporting positive results of the Phase III trial of its drug candidate ... Fampridine-SR ... to improve walking capabilities in patients with MS. The PR news of positive Phase III trial results for this drug literally moved the stock price of ACOR from $2 to $20 in two months.
Unlike ACEL ... ACOR does not have "fast track" or " orphan drug " status with the FDA. ACOR is now conducting the Phase IIIB trial of the drug for any cardiovascular effects. This won't be completed until mid 2008. The FDA is not expected to rule on any NDA until sometime in 2009. Yet ACOR's stock rose from $2 to $20 in two months after the press release of positive Phase III results in September of 2006 ... and is still trading over $17 today. Barron's estimates that annual sales of the drug could approach $500 million/year within 5 years of any FDA approval. Barron's rates ACOR as a BUY at the current price ... as it moves toward a possible FDA decision in the next 2 years. The market for ONCONASE is substantially more lucrative and FDA approval for ACEL could be forthcoming much sooner.
The relevance of this summary above to ACEL is very important IMO in properly understanding ACEL's " Share Price Potential " from this point on. If ACOR"s stock can soar from $2 to $20 simply on the reporting and publicizing of positive Phase III trial results ... what can ACEL's stock price achieve if it properly announces and publicizes positive Phase IIIB trial results for ONCANASE?
A massive panic "Short Squeeze" is in the cards for ACEL($2.32)!!!!!
The latest short interest figures for ACEL as of 2-29-08 is a record setting 2.3 million shares short (short interest)!!!!!
The next leg up in this stock will trigger a massive panic "short squeeze" that should run this stock up to $5+ extremely fast.
During last April, 2007 ACEL ran from $1.30 to $3.74 in only 3 weeks!!!!! The initial move up triggered a "short squeeze" that ran the stock to $3.74!
There were 2.1 million short shares at the beginning of that run. At the end of the 3 week run ... there were only 250,000 shares short!!!!!
ACEL is now on the verge of a dramatic move up from here. I expect the stock to be 200% to 300% higher within 3 to 6 weeks from right now.
Check out the chart and the TA read on ACEL right now. It is a major strong buy. Tradingday.com is now rating ACEL as an 80% strong Technical analysis BUY!!!!1
Add the coming massive "short squeeze" panic coming to the picture ... and ACEL is on the verge of a dynamic move up in price.
Do you have any yet?
This Patient is taking ACEL's Onconase
Onconase clinical trial patient has input
Here is the link:
http://www.mesothel.com/profiles/douglas.htm
Canadian Boilermaker Travels to Bremerton, Washington
"Thanks to Roger Worthington's Website"
An Update -- 01/10/08
--------------------------------------------------------------------------------
July 28, 2001
Over the past few years I have used your web-site to gather information on the different trials and patient profiles. Here's my situation that I'd like to share with all meso-patients!!
First of all my trade is that of a construction boilermaker, working on industrial furnaces in pulp mills, oil refineries, all of which were insulated with asbestos at one time
In the fall of 1998 I was working on a project, building penstock and scroll casing, while I was there, I started to feel sluggish climbing stairs and staging, it would take me up to five minutes to get my breath back? I was 5ft 9 inches tall 210lbs, in fairly good shape as far as boilermakers go. So I went to my physician, who in turn sent me to get my lung x-rayed. The x-ray showed pleural infusion. The specialist taped my lung and removed 3 ½ liters of fluid.
Something was definitely wrong inside there. A bronchoscopy proved negative which was good news, next was on February 22, 1999 another specialist did a thoroscopy, where they go between your ribs and into your pleural to take biopsies, test results showed Mesothelioma, a form of cancer related to asbestos exposure. What the oncologist was to say next really floored the family, he said there's no Known cure for malignant mesothelioma "go home, do your paperwork you've got 3 to 6 months to live."
WOW! Talk about getting hit by a Mack Truck. After getting out of the hospital it takes awhile to figure everything out. Which avenues to go, and believe me there weren't too many. Choices included Chemotherapy, Radiation, or Radical surgery. I hadn't really decided but the third option was definitely out. As news of my health got out friends started to call wishing the best but soon a call from a firefighter we knew asked if we ever heard of Essiac Tea, What a history this herbal tea has, anyway I started on my tea then a friend phoned , saying I'm using this herbal extract called Oncolyn, he was using it for the prostate cancer he had. I did some calling around and soon I to was using the Oncolyn,I started 6 tablets a day for the first week then down to 3 a day . I went back to where I purchased the Oncolyn and told him there's something happening with my lung, he said Its the Oncolyn , at this point all I knew something was cooking?
Further to my research the immune "system" kept coming up. Low and behold another friend's wife was selling Usana products, vitamin supplements, okay, the reason these cancer "s take off is your immune system does not have the firepower to fight them, so I added vitamin essentials and pro -c, I also found out you have to have de-toxifiers, grape seed extract, garlic, vitamin E with selenium, milk thistle, and Quercetin C, All these play a huge role in your fight.
Throughout this difficult time I used visualization and visualized little angels pouring acid over my tumors, There's a lot of literature out there, and one book I found Uplifting was "Love, Medicine, & Miracles", a book by Dr. Bernie Siegal. A must read. This book reveals how people cope with there health problems the above mentioned , prayer etc. All these things play an important role in your path to healing.
At this point I'm totally Convinced the course I was on was working for me. My EX-wife and still great friend, was surfing the net and low and behold found www.mesothel.com, Thanks to Roger Worthington's website Linda was able to access information on mesothelioma and clinical trails that were taking place across the U.S. Here in Canada the resources are not available for this type of research, but a Onconase trail was taking place in Bremerton Washington. The information stated that this light form of chemotherapy killed meso cells in the lab, this study was a phase II trail so this treatment was available for a while. Anyway I was accepted into this Onconase trail on April 23, 1999. Nine weeks later on June 27, 1999 my first cat-scan revealed no tumors at all, Gone! Some scaring of calcified plaque but no tumors.
What an amazing turn of events, one cannot begin to express the emotions that goes through you, ALL I can say is thanks To many, many friends and this web-site for its information. So many of us need this kind of information because without it, it’s hard to know where to start. It's now July 28, 2001 and last Tuesday's cat-scan revealed a stable diagnosis, no change.
So there's my story, and I'm sure there are many, many more out there, so in closing to those out there, keep fighting the fight, keep a positive attitude even though at times you wish other things, give your body the necessary firepower so that your immune system operates at peak efficiency.
Good health to all.
Paul Douglas
*** POSTED JULY 31, 2001 ***
--------------------------------------------------------------------------------
An Update -- 03/05/02
It's only been 8 months, but I just wanted to up-date my condition to every-one out there who uses the Web-site.
It's been 8 months and 5 CT scans later and everything is stable as far as my meso is concerned. It's been a remarkable 3 years since I was diagnosed, and I hope to be giving you up-dates like this for years to come, I still take my regiment of vitamins, still on the Onconase trial, and still take my oncolyn. I know there are thousand's of us out there so if you need moral support or someone to relate to, you can reach me at: shrnedd@telus.net.
Best of health to all.
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 10/02/02
It's been awhile but the good news is I've just had my 20th C-t scan and alls well , my condition is stable, no tumors just the same calcified plaque where the tumors used to be. The past 4 or 5 months I've had the opportunity threw your web-site and my E-mail to talk to some very fine people who's love ones have been diagnosed with this terrible disease an been able to assist them in their fight which is all of our fight to lead them where they may find some successes, their questions are many and I think when you can give direction and hope it sure makes them feel better and ultimately the same for me. I wish the very best to all meso victims and their families, this is a fight we can't lose. I can still be reached a my E-mail address and thanks again for your web-site, I'm on it almost every other day.
Best of health to all,
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 02/26/03
This past Saturday, February 22, 03 was my 4th anniversary of sorts!! Not with-standing a few bumps on the road along the way, medical opinions out of the spectrum things are going really well, I feel good, not without the odd aches and pain one has to endure!! which in turn gets the mind process out of kilter, it's never to far away those thoughts tend to creep in once and awhile!!
When I was first diagnosed in 1999 I said to my wife I guess I'm "toast" I had forgotten I'd said that as she reminded me. Well, were still kicking up a storm so to speak!! Backin the first few days after getting out of hospital, I was gathering information as to how to begin the battle I was facing, I said to myself I've got to stop this disease in it's tracks, there's still a few loose ends to tie up here, so I set a few goals 1 being my daughter's graduation, this June it's happening not only do I get to see this I've also watched my daughter grow into a beautiful, caring and loving young adult, 2nd I said to myself what about my 50th birthday, that's down the road but definitely within reach!! It's funny sometimes, people seem to have forgotten about that day 4 years ago but that will never be lost in my mind, the fight continues, every day one must do the things that have helped me sustain my health, my vitamin regiment, the herbal tea, oncolyn and the clinical trail of onconase, which I owe to this Web-site, that test trial was the last piece of the puzzle that seemingly put me over the top??
Every day is a new day, what lies ahead only God knows I must say to all meso victims never give up hope, enjoy your families life is so precious, it feels good to wake up and greet the day and string those days into weeks, months and years as I've been so fortunate to do!! It's crispy cold today, fresh snow on the mountains. I am going to take Eddie to the dyke and enjoy the fresh air!! I can still be reached at my E-mail address for those of you who want to speak to someone that has and still fighting this terrible disease, and thank's again to Roger and all the people who help put together the valuable information this web-site provides!!
Good health to all.
Paul
--------------------------------------------------------------------------------
An Update -- 09/02/03
Another 6 months has passed and every-thing is fine, my last ct-scan was clearer then the previous scan go figure eh!!! As you know there are peaks and valley's with this disease. In April a scan revealed plurel infusion, so I thought here we go, on to my next iron in the fire, It looked like I was off the onconase trial but not to give up on that I went to my oncologist here in Vancouver, 60 cc's of fluid and no conclusive tumor growth visible on the ct-scan wasn't enough to be excluded from the trail. So both doctor's chatted on the phone and Bingo back on the onconase trial, I was prescribed Zithromax and then wait till the next scan to see if there indeed any growth as luck and good faith would have it no tumor growth the infusion had cleared up and every-thing is back to normal!! It seems every 2 years I run into a funk of sorts.
Last week at the clinic in Bremerton, there was three of us meso patient's sitting side by each, I was on the onconase Matt had had his lung resected, and Vern was on the Alimta- cisplatin. Needless to say we had a great conversation as to the pro's and con's of the different treatments we were using!!! So in closing I like to wish all meso people the very best and never give up hope, every-one likes a good fight now an then an this one we have to win. Roger and the rest of the team, keep up the great work your doing many kudo's to you guy's. I can still be reached at my E-mail address if any needs support in their fight?
Thanks again, Best of health to all.
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 02/16/04
Great News!! February 10th was my 29th C-T scan and all is well. The doctor's have noticed a small amount of pleural thickening at the base of the lung but no tumor growth so no fluid.
I can't believe it's been 5 years since I was diagnosed with meso, and that fateful statement, "You've got 3 to 6 months to live". Well, with the hope and prayers of a lot of friends and this website, we have proven good things do happen all the time!! Miracles might be a little under-statement but some have said that!!
Believe me there is a lot time and dedication to help keeping your immune system running at peak efficiency to fight against those cancerous cells that want to grow out of control. I still maintain my Vitamin regiment, along with Oncolyn, Essiac tea, while also managing to stay on the Onconase trail in Bremerton Washington thanks to this Web-site. Over the past few years there have been occasions where it has been touch and go, but we have managed to weather those storms. In maintaining my health I've found the goals I set a few short years ago are coming to fruition, my 50th birthday is coming this October and now I'm making some new goals to strive for, but not to far away!!
As you and everyone who uses this Website are aware, mesothelioma is such a sneaky disease, and there are different types which do not react favorably to the forms of chemo we throw at it. So when one has some success the key is stay the course and find what works for the individual patient, and keep with it. This website enables people to aid in their struggles to get that information and put them on the right track.
One has to thank Roger Worthington and all the people associated with him for the tireless work these individuals do to help us and further be able to help ourselves in the tragic disease that invades our families lives. Once again Kudo's to all the people who devote their time and energy to this disease!! I look forward to the month's ahead where all these new grants that have been made start to show a positive result to further help all those afflicted with disease, for those who wish to talk to someone who is also fighting this unending battle, my E-mail is shrnedd@telus.net
Good health to all
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 09/09/04
Yesterday, Sept., 8th I had my 31st Ct-scan and low and behold no change, I don't know how long I can keep this batting average at 1000, it's a challenge but I'm up to it. Come October I'll be reaching another goal that I set for myself, the big FIFTY, my sister and brother are planning a birthday bash, family and friends should be a good one!! Sometimes I sit back and wonder why I have been so fortunate while others seem to be not so lucky, as I've said before if you have some success stay with it, the rewards are priceless, to wake up every day an knowing the hand I was dealt and to be able achieve the goals I set is amazing!!
I am in touch with a lot of people who use this web-site and they tell me what an inspiration I am to them and their families who have been afflicted with this disease, truly it makes me feel good and also makes me want to do what I can to help others who have my not know which way to turn!! The road blocks are there both medically and financially and it's no easy task in a life or death situation.
It also gives me great pleasure to know that Roger and every who works for him spend their time and resources to further help us find a way to eliminate mesothelioma as a life ending disease, it's not the way it was supposed to be and it looks like the politicians are warming to the idea that we have a disaster happening right in front of their eyes, I think the sight of the Twin Towers collapsing and all the asbestos floating over Down town Manhattan has woken a lot of senators up as to the magnitude of the problem asbestos will eventually pose, it's about time!!
Any how keep up the tireless work you guys and gals do for every one of us that uses the Web-site and for the families that are affected by this terrible disease!!
Thanks again, I can still be reached at my web-site for those who wish to communicate with someone who has meso, I think it's easier to relate to??
Good health too all!!
Paul Douglas
shrnedd@telus.ne
--------------------------------------------------------------------------------
An Update -- 01/17/05
Hoping every-one had a good a Holiday season at MARF. Since my last up-date a few things have changed, remember a few posts ago I had mentioned what a sneaky disease this meso is, well low and behold About 6 months ago I could feel this lump on the outside of my rib-cage, I didn't think to much about it as the c-t scans were saying it was stable!! After getting 4 or 5 opinions I decided on my own free will to get a biopsy and see what this thing was?? After 4 cancellations I finally went under the knife Dec. 30th., the surgeon asked me if he should biopsy it or cut it right out!! We've waited this long so take it all out. 4 days later my suspicions were confirmed, it was meso!! Little did I know what a fire storm of sorts this would turn in-to!! Everyone started freaking out, meanwhile here I was wondering where their thought process had disappeared??
Ever since I was diagnosed way back in Feb., 99 I had envisioned a plan of attack if any thing should develop along the way, well almost 6 years later here we are. Taking the "bull by the horns" so to speak, let's not jump to conclusions here, there is a progression thing in place so why don't we follow it?? After everyone had time to reflect a little bit things did not appear to be that difficult; another ct-scan showed little or no progression in lung itself, so that was good news.
So now were on to the second phase of the Onconase trial which adrimiacin is introduced as a second agent, I'm told I should tolerate it very well, I'll lose my hair but since I'm going bald any way, maybe I'll get an earring and they can call me Paul "Mr. Clean" Douglas, hopefully I'll garner a few laughs along the way!!
In the meantime I feel great a little set back like that did not cause to much concern to me it' s gone with a little radiation to clean up any cells that might be around, it's a done deal. I see no reason not to be giving more positive updates in the future and looking forward to some good news in the research that is progressing as we speak. Saying that I like to say to everyone who uses this website stay positive, life is good, Now if I can get my golf game to come around life will be real good!!
I still can be reached at shrnedd@telus.net and wishing every one a healthy new year!!
Thanks again and good health to all.
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 06/13/05
It's been a while and just to let you know everything is real good.
Back on my previous post in January I mentioned that I was going onto the second faze of the Oncanese clinical trail, that being Adriamician with Ocanese, but low an behold a Mugascan and an Echogram showed that my heart might not able to tolerate the Adriamiacin as the valve that pumps the blood, it was working at about 50 per-cent so my oncologist didn't feel good about those results so we were taken off the trial all together. I was off treatment for 2 months still taking my Oncolyn, Essiac Tea and my vitamin regiment, as time went by I inquired about treatment from my oncolgist here in N. Vancouver B.C. and he said without a tumor marker to gauge the progress if any the Alimta Cisplatin was making and the toxic effects it has that we would have wait for tumor progression before any treatments would be administered!!
From a Doctor's stand point I guess that was the best way to proceed? But being pro-active I called Joe Johnson in Bremerton Washington for a second opinion?? It was his thought that we should go ahead with 4 regiments of the Alimta-Cisplatin and see if there was any change, our ct-scan showed it was stable from the previous ct so it was a hunch on his part and amazing enough there was a change after 2 treatments and significant change after 4 treatments. I always had this pocket of fluid in the upper lung area since day 1 but the treatment had cleared a lot of the fluid up!! So there you have it his hunch paid dividends that we didn't expect, the results needless to say were eventful. Through the treatments with the help of steroids and anti-nausea pills I never endured the sickness that comes with this treatment, a little tired for week after but nothing to stop my daily activities nor did it effect my appetite, so that was a bonus!!
As of June 7th I have been off any treatments and looking forward to a great summer and our trip to New York later this month, we;ll go back in September for a ct-scan and hopefully there will be nothing to report and move to 3 month check-ups.
Till my next up-date I hope and pray for all those affected families that have to endure this terrible disease that good things do happen and never give up hope, If anyone wishes to communicate with me via E-mail it is still the same and look forward to helping anyone who has trouble getting through the hoops that the medical system seems to put us through, more turmoil then we are already!! So in closing again thanks to Marf and every one else for the hard work that will eventually lead us to a cure.
Good health to all,
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 09/30/05
Just to let every-one know my Sept 18th ct-scan was clear, it's quite amazing because I've been off treatment for over 3 and 1/2 months. I went 2 months without doing any thing before but this the longest so far. My oncologist was very ecstatic as was my family, the nurses and all my friends and relatives. I guess the good lord is watching out for me and my mother must be kicking him in the pants saying watch out for this guy!! It never fails before a ct-scan there are all these aches and pains, my wife says your imaging them , but in my mind they are there?? Once the scan is done I wait for Dr. Cook's assessment and low an behold your clear all the symptoms slowly fade away!! The mind truly is a deceptive organ. Thanks for the invitation to the 2nd annual symposium, I don't know if I'll make it, the New York trip was fantastic, the people there are so friendly, always willing to help the lost tourist!! Yankee Stadium is a baseball fans dream, the Yanks lost both games but who cares I was there I got pictures I drank the 8 dollar and 50 cent beer what more could you ask for?? We enjoyed the shows, I'd go back in a heart beat but all this put a hit in the pocket book, but don't count me out just yet, it's Oct. 1st and there's still time. I'll close for now and hoping to see some results on the on going research to this disease yield some dividends in the near future!!
The best of health to all.
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 01/04/06
Happy New Year to you and every one at MARF (www.marf.org). In past year things have gone really well for me health wise, as I mentioned in my last up-date, I've been able to put 3 ct-scans together with a stable diagnosis, I was in a dilemma whether to get a scan before Christmas or wait till the New Year, my oncologist said I wouldn't leave to long, there I was thinking, good news bad news. SO Nov. 29th we did a scan and low and behold stable again. Once more a little pleural thickening, but other wise no change, so it made for a very Merry Christmas. I go back in 3 months for another scan, as we continue keep the mesothelioma at bay!! This February will be 7 years since the doctor famous words, "you've got 3 to 6 months go home and do your paper work" We kind of blew that statement out of the water, one can only hope for continued successes till more treatment options are available to people who have been afflicted with this killer disease. Also hoping to see some results on the on going research projects that have been trying to aid us in our fight!! In closing, wishing every one the very best in 06, I can't believe how fast 05 went. I can still be reached at my E-mail address Shrnedd@telus.net for those who desire some one to converse with. Good Health to All, Paul Douglas
--------------------------------------------------------------------------------
An Update -- 06/11/06
It's been over 5 months so I thought I'd touch base and let every-one know things are still pretty good health wise. My last ct-scan was stable so that's always great news, it keeps me away from the Alimta-cisplatin chemo which is a good thing!! June 8th was 1 year since I last had any treatment, I guess the chemo did it's job but you never know when it will sneak up on you?? I have a ct-scan on July 11th so we're a month away and I'm feeling pretty good right now, let's keep our fingers and toes crossed so our luck keeps on going.
Sharon and I just got back from Spain and Portugal, 3 weeks in the sunshine, 10 days in Spain and 8 days in the Algarves of southern Portugal. A lot of history in Spain dating back to the 11th century, truly some amazing places to see. In Portugal we spent one night in Tavira then went on to Salema, a quiet little fishing village, 6 days laying on the beach. What a great place to recharge your batteries, r&r at it's best, the seafood was fabulous, beer and wine aplenty, the whole trip there was no rain at all, but all good things come to an end, we arrived back in Vancouver, you guessed it RAIN!!
Looking at the web-site it seems we're making some progress but lets hope more resources are available for research by way of that 50 million dollar Dept of Defense budget hassle aside. We're all hoping there soon will be good news coming down the pipeline!! I still maintain my vita-min regiment with the Oncolyn, I've added ip6 to it, I watched a program where this oncologist insists his cancer patients take it, so what the heck it can only do good.
I'll leave you for now, I'll give another up-date in December, wishing good health to all.
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 01/17/07
Happy New Year! I think 06 went faster than 05. Just would like to let everyone who uses the web-site an up-date on my health!! It's been 7 months and there is always something that creeps up on you?? Remember back Dec. 05 I had an operation to remove a tumor growing outside of my rib-cage well you guessed it, it reappeared but not in the same place it slide back up to where I had the Thoracoscopy 4 ribs higher!! Talk about being a sneaky desease, so we had radiation treatments and after 10 shots the tumor was gone!! I was really pleased with the out come to be sure. This past week I had a ct-scan and wouldn't you know it the scan showed no changes except maybe a little pleural thickening at base of the lung, after 8 years this didn't come as a big surprise I still feel good and there's no crackling in the lung so every thing is o.k. till we get another ct-scan in April. Because I've been so fortunate and maybe a little lucky so far, I got talking with Joe Johnson in Bremerton Wash. With the positive scan wouldn't it be a good idea to be pro-active and get 4 treatments of Alimta-cisplatin just to be sure?? He agreed that would be a great idea, the thought being you don't have to wait for a reoccurance and keep a lid on it before it does come back!! 20 months since I last had Alimta-cisplatin all the accumalative effects would have passed on as time had gone by, so there's where we stand to-day, looking forward to a great year in 07, got our 2nd annual golf tournament in Whistler B.C. this summer, and where planning a trip to Mexico In April we've got a lot of snow this year, even though I like the clear an cold weather I'm lacking the Vitamin D that comes with sunshine!! Hoping to see more treatment options come down the pipeline with all the research going on!! Wishing every-one a healthy 2007!!
Paul Douglas
--------------------------------------------------------------------------------
An Update -- 07/27/07
It's been a little over 6 months since my last correspondence so I'll bring every-one up to date.
Let me tell you it's not without any excitement what occurred down at the clinic in June, you know how I've always tried to be pro-active in fighting this disease well it almost cost me!! The first treatment of Alimta-Cisplatin went great, the second one didn't, I was sitting there and I started to itch, I looked over to the nurse and no sooner then about 15 seconds I was turning beet red, I started to lose my breath all I could do is little gasps of air. I was having an allergic reaction to the Cisplatin, before my eyes 4 nurses were on to me, they gave me a shot of effrigen, I think that was what it was called anyway things started to get better, this shot boosts your heart-rate, one nurse took my pulse and it was 184 over 114 and after a short time it started to go back to a more normal pressure, the thing about this emergency is I couldn't do anything, I just sat there and let the nurses do their thing. My daughter was with me that trip but she had gone to Sub-way to get lunch, although she's just completed her 2nd year of nursing I was happy that she didn't have to see her Dad in that state!!
So needless to say we stopped treatment for now and will have a ct-scan in September to see where were at. Thinking back both Dr. Johnson and I thought it was a great idea because back in the fall I had radiation to get rid of the tumor that had came back on my ribs we figured if there were any cells left we could blast them and be done with it. They say a cat has 9 lives and if I were a cat I think I used a few up that day!! We all have been waiting for news on the research that is on going, I sure hope and pray the scientists are closing in on some treatments that will help all us meso patients continue to at least live a some what normal life. The stress that is put on the family's is a lot to bare, in my case it's not really there but on the people I have met with this disease and what they have to go through is incredible. Like I said I'm looking forward to news coming down the pipe-line so in closing keep up the tireless work Roger and the staff plus the people at M.A.R.F. in trying to eliminate meso as a life ending disease!! Thanks again!!
Good health to all, Paul Douglas
--------------------------------------------------------------------------------
An Update 01/10/08
Happy New Year to you and every-one at M.A.R.F.
I just had a ct-scan Jan.8th and everything was stable. As we were reading the CTt-scan Dr Leen looked over at me an said "your not on any treatment right now are you'" no I'm not. This is truly amazing, what ever your doing keep it up. I remember a stretch where I went 18 months or so where I had no treatment so maybe I'm on another run of sorts!!
Anyhow I'll take and run, if I can stay away from the chemo all the better. I purchased a book by Bill Henderson, " Cancer Free" your guide to Gentle, Non-Toxic Healing" I would recommend that cancer patients read this book. It has some very good directives that might interest a lot of us, there's a chapter where there's 10 things you should take!! Low and behold I'm taking 6 of them, so there you have it. What works for some might not work for others!! but if you don't try how do you know?? Is it just me or is time flying right by??
Feb. 22nd will be 9 years since I was diagnosed with Meso, what a ride it's been, I've set a few more goals to reach for and hopefully I'll get there. Let's hope this year brings us more treatment options so we can live a some what normal life!! I've talked to a lot a families out there and it's really tough on the family structure. So in closing I wish the best of health to every-one at M.A.R.F. and all those who use and read this web-site. Have a great 08 and I'll give everyone an update in 6 months!! Bye for now, I still can be reached at my web-site Paul Douglas
....tradingday.com is a comprehensive, reliable source with an extremely strong track record of accuracy. In this case, however, I think they're off by 20%...ACEL is a 100% super strong buy IMO. The triple play is stronger than ever: great chart on a breakout; short squeeze getting tighter; huge news coming soon...add to this that the sellers have dried up.
igot ... tradingday.com just issued an 80% strong buy rating on ACEL($2.38) this evening ... after today's breakout.
Looks like you called it right. Your target of $5 on this current move may be conservative.
Nice to be long this one. It will be fun to watch.
Imagine what it would do in a good market!
Congrats to you and all longs.
skyyy...what I'm seeing with ACEL is the last of the sellers getting out of the way. As I previously stated it was clear that the selling was drying up. If this continues, the short squeeze will begin coming into play and we'll see a steady climb to $3.
What makes this so exciting, IMO, is that it appears that this upward trend is now perfectly timed for the upcoming huge phase III news. My only previous concern was that the imminent huge news would hit before the stock got traction and the bounce on the news would have been somewhere between $2.90 and $3.50. This was prior to learning from the board's CC on Friday that there have only been an additional two "evaluable events" since the late January update of 311. (Which is also a great indication that patients using ONCONASE are living much longer)
I thought they would have hit the magic 316 by now and that the news would have come out thus giving us a move to a high of $3.50. It now has time to build. This breakout combined with a delay in the huge news and fueled by the short squeeze easily puts ACEL over $4, IMO, on the announcement that the company is proceeding with the NDA. I don't think this is the least bit overly optimistic...I wouldn't be surprised to see $4.50 maybe it touches $5. Sorry for the long response, but you asked.....
SKYYY - World News National Article
ABC WORLD NEWS NATIONAL
6:30 pm eastern std time today, ABC World News aired a piece on a possible cancer treatment from a frog in China that appears to have a similar effect as ACEL's Onconase.
Here is a link to the article:
http://abcnews.go.com/Health/story?id=4421562&page=1
igotbigbulls ... great call.
Looks like a breakout now.
What are you seeing from here?
ACEL Chart Is Ready
Tomorrow should see a big volume surge, and a really fine candle in response. I look forward to doing some channel work on the basis of that forthcoming break.
Please link back for the chart.
fringe
|
Followers
|
9
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
159
|
|
Created
|
10/22/05
|
Type
|
Free
|
| Moderators | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
