Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
ARYC! @Arrayit
www.arrayit.com
Unbelievably low market cap of $720K! Trading at .15 x earnings!
Earnings of over $1 million in first 2 months of 2016.
Located in the heart of Silicon Valley near Apple, Yahoo, AMD, Juniper, MBZ, LinkedIn, Facebook and many other leaders of the tech industry.
Earnings of $2.3 million last year and on track for even higher this year
Trading at unprecedented low of .0007!!!!!!!!
No other sub penny company has the potential of ARYC
Check out their tweets @Arrayit or go to their Facebook page.
A couple updates from this past week @Arrayit:
Arrayit quotes $179,430 microarrays to biomedical leader Progenie Molecular Valencia España http://www.progenie-molecular.com
Arrayit quotes $51,680 microarrays to top leader De La Salle University Manila Philippines http://www.dlsu.edu.ph
Arrayit quotes $102,240 microarray platform to venture think tank Leading Technology Group http://www.leadingtechnology.co
Arrayit quotes $38,485 microarray platform to top Korean distributor Daemyung Science Seoul http://www.dm4you.com
Arrayit Corporation ?@arrayit
Arrayit completes conference call on huge DNA testing opportunity in massive market in India http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit reports DNA testing inquiry from top agency in India. Market penetration of 0.1% would produce $48M revenues http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit Corporation ?@arrayit
Arrayit quotes $235,000 microarray instruments to top lab University Ottawa Ontario Canada http://www.uottawa.ca
Arrayit reports microarray sale to advanced technology leaders Waki Company Tokyo Japan http://waki-bg.jp
Arrayit reports microarray sale to automated molecular testing leader AutoGenomics Vista CA http://www.autogenomics.com
Arrayit reports microarray sale to life sciences European Biotech Network Dolembreux Belgium http://www.euro-bio-net.com
Arrayit reports microarray sale to top medical researcher at $13.4B endowed MIT Cambridge MA http://web.mit.edu
Arrayit expands marketing on $100K+ TissueMax™ Automated and Personal Tissue Microarrayers
Arrayit reports $400,000 microarray instruments inquiry from top European Union distributor http://arrayit.com/Products/Microarrayers/Microarray_Printers/microarray_printers.html …
Arrayit invited to appear on Business Television BTV hosted by broadcast partner Bloomberg http://www.b-tv.com
Arrayit $17.7MM OvaDx® spin-off and 6,000 diagnostic test pipeline possibility emphasizes the $17.7MM x 6,000 = $106B ARYC market potential.
Arrayit reports $9K chemical sale in specialty trading subsidiary TeleChem International Inc http://www.telecheminternational.com
Arrayit reports $100K microarray inquiry Al Rayyan International Science Center Amman Jordan https://www.linkedin.com/in/nader-ghanayem-3b1306a2?authType=NAME_SEARCH&authToken=ofog&locale=en_US&srchid=2270647621456852532077&srchindex=1&srchtotal=2&trk=vsrp_people_res_photo&trkInfo=VSRPsearchId%3A2270647621456852532077%2CVSRPtargetId%3A365047477%2CVSRPcmpt%3Aprimary%2CVSRPnm%3Atrue%2CauthType%3ANAME_SEARCH …
Most honest post on the JNSH Board. Read this: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122808872
JNSH
BUYER BEWARE
LOL. The JNSH MODS are deleting their own posts so as not to make a connection to the ones I am using on this Board to warn newbie investors about their share selling scam. Too funny. Everyone knows already. Let the scam be known.
JNSH
BUYER BEWARE
LOL:"HOLD FOR A FEW YEARS!!!" NO ONE HOLDS A PINKY FOR A FEW YEARS. THIS IS NOT NASDAQ OR NYSE. THIS IS THE OTC. MORE PROOF OF THE PUMP AND DUMP: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122808927
BUYER BEWARE
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122813613
Yeah the games will begin all over again as in pumping and dumping on unsuspecting, novice IHUB traders: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122813613
BUYER BEWARE OF THIS SHARE SELLING SCAM.
LOL: It's a sound investment if you received free shares, pumped and dumped it and are now riding free shares again.
JNSH
BUYER BEWARE
Total scam.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=122813613
JNSH. DD is overwhelming. LMAO. So is the pump. .0081 to .0035 in 5 trading sessions. Dirty group frontloaded and dumped. Slow bleed.
BUYER BEWARE
ONCI ripping today :D AMMX loaded a few on that news :)
$PMCB PharmaCyte’s Novel Diabetes Treatment Approach Could Prove Superior to Other Methods Under Development
Forecasts for the number of people that will be diagnosed with diabetes in the coming years are staggering. While many treatments exist to treat some of the effects of diabetes on patients afflicted with the disease, a “holy grail” to truly treat the disease has yet to be developed. Clearly, an out of the box approach is required. If future test results affirm the data derived from recent studies, the PharmaCyte Biotech (OTCQB – PMCB - $0.07 – Spec Buy) Cell-in-a-Box® diabetes treatment platform could emerge as the treatment of choice for this disease.
Dozens of studies and trials to treat Type 1 and Type 2 diabetes are ongoing, given that the size of the global diabetes market for therapeutic devices and drugs is expected to reach US $114.3 billion by 2018, according to a report by Transparency Market Research. However, since results have been mixed for even the largest and most successful companies, industry participants are now exploring new ideas and new approaches that have demonstrated efficacy in early studies, which could serve as an indirect or direct benefit to PharmaCyte.
For example, one of the top two players in diabetes treatment, Novo Nordisk (NYSE – NVO), announced that it is collaborating with IBM (NYSE – IBM) to combine IBM’s cognitive computing capabilities with diabetes research by collecting and analyzing real-time data from patients using Novo Nordisk treatments and devices. The hope is that this venture leads to improved solutions for diabetes management. Novo Nordisk also submitted a new type of fast-acting mealtime insulin to the FDA for approval, a departure from its primary offerings. Meanwhile, Novo Nordisk competitor Eli Lilly (NYSE – LLY), which has had six diabetes treatments approved since 2014 halted development of a promising diabetes treatment under development. If a company with 6 approved drugs in a treatment category is still working on the problem, and another leader is moving outside of its comfort zone in an effort to develop the most effective therapy available, it is an indication that the time is now for PharmaCyte.
The Company has the exclusive worldwide rights to use Melligen cells to treat diabetes. Melligen cells are genetically engineered from human liver cells and have been shown to secrete insulin in response to the concentrations of glucose (blood sugar) in their environment. A recent article published in scientific journal Molecular Therapy noted that when Melligen cells were transplanted into diabetic mice whose immune systems were essentially not functioning, the blood glucose levels of the mice became normal.
This observation illustrates that Melligen cells can reverse the diabetic condition. PharmaCyte plans to encapsulate a human cell line that has been genetically modified to produce, store and release insulin in response to blood glucose levels in their surroundings. Therefore, the Melligen cell line, when combined with Cell-in-a-Box® encapsulation, could ultimately become a treatment that has clear advantages over current therapies used for Type 1 diabetes and Type 2 insulin-dependent diabetes and could potentially replace them. As a result, we look for these shares to move toward the $0.20 level in early 2016.
$AEXFF Aminex PLC chief: First gas production in Tanzania a step towards continuous growth
$AEXFF Aminex PLC, an independent oil and gas company premium-listed on the London Stock Exchange and primary-listed on the Irish Stock Exchange with activities focused in Tanzania, announces that all the
resolutions put to shareholders at the Annual General Meeting of the Company held earlier today were
duly passed. Details of the numbers of shares in respect of which valid proxy appointments were made in
advance of the AGM are available on the Company's website, www.aminex-plc.com
http://www.aminex-plc.com/
Thank you Pete. We feel this stock has ETSH potential.
This is a really good alert that you got here my Friend.
Pete,
I have learned that they have a minimum of $86 million in total oil assets, (conservative estimate) and that a big announcement / filing is coming next week either Monday / Tuesday.
Sporty,
I learned about a corrupt group on IHUB this week. And I intend to exploit them. Only on this Board can you trust everyone. Have a great weekend.
that's what ive been saying guys :D go AMMX
Thank you...I believe its far from done...
Blessings, my friend!
TRADE ALERT: AEXFF
Hearing of a company announcement forthcoming Monday, possibly about the commencement of oil drilling. Lending credence to this rumor is the substantive volume which is a predictive indicator of price volatility. The equity has traded over 1 million shares today and is up significantly. This move so far today is nothing in comparison to what we feel is coming next week.
Could the announcement involve this?
“After drilling the Ntorya-2 appraisal well, Aminex plans to apply for a licence to fast-track development of the Ntorya field so as to monetise gas discoveries through the new gas infrastructure. Aminex is also in discussions with the TPDC to identify opportunities for an early production system from Ntorya-1 and Ntorya-2,” says Jay Bhattacherjee Chief Executive.
Check out CLRB
40m AS
838k OS
Crazy...have a blessed weekend
Take a look at these news releases, have you ever seen a penny stock trading in this range this profitable?
http://www.ammx.net/Press_Releases
ammx superb news
$PMCB PharmaCyte Biotech Appoints Former Johnson & Johnson Executive as Senior Business Development Advisor

SILVER SPRING, Md., May 03, 2016 (GLOBE NEWSWIRE) -- PharmaCyte Biotech, Inc. (OTCQB:PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, today announced that it has appointed Dr. Sanjay Batra as its Senior Business Development Advisor.
Dr. Sanjay Batra brings to PharmaCyte more than 20 years of global healthcare and entrepreneurial experience in start-ups, biotech and large pharma. Dr. Batra spent 10 years at Johnson & Johnson, culminating as Vice President, R&D Pharmaceuticals, Asia-Pacific and Japan. He is highly skilled in partnering with diverse stakeholders, such pharmaceutical companies, the investment community, academia and key opinion leaders. In addition, Dr. Batra has been involved in over 80 clinical trials in all phases of development and commercialization.
PharmaCyte’s Chief Executive Officer, Kenneth L. Waggoner, said of Dr. Batra’s appointment, “We are pleased that Dr. Batra has agreed to join our team, as we now have a cGMP-compliant facility and are ready to enter into the next phase of our life-cycle as a biotech company. We believe that his experience at Johnson & Johnson and his global network will prove invaluable to PharmaCyte. His expertise in scientific, medical and business areas, both domestically and internationally, will be a key asset as we move PharmaCyte forward towards the successful development of our novel platform technology to treat pancreatic cancer and diabetes.”
Dr. Batra said, “I am very excited to be part of the PharmaCyte team at this pivotal time. The technology holds tremendous promise to help physicians and their patients in areas of significant unmet medical need. Mr. Waggoner has built a very solid organization, and I look forward to making contributions with pace and rigor to advance our product pipeline towards commercialization.”
Most recently, Dr. Batra served as President and CEO of Aesthetic Factors, an emerging company providing autologous, point-of-care therapies in Regenerative Medicine. In this role, Dr. Batra led the commercial growth of their platelet-rich plasma and autologous fat products and was instrumental in establishing the company as the science driven leader. In 2013, Dr. Batra founded VIAS Partners with the aspiration to take his diverse experiences and global network to partner with entrepreneurs and companies to advance their business concepts.
Dr. Batra obtained his Ph.D. in medical physiology from the University of Ottawa, Canada, and completed his post-doctoral training with world-renowned scientists in Japan and Switzerland. He is a Fellow of the American College of Cardiology and an Adjunct Professor at the Wake Forest Institute for Regenerative Medicine. Dr. Batra has published over 50 papers and 70 abstracts, and has made more than 100 invited scientific presentations.
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company developing and preparing to commercialize treatments for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®.” This technology will be used as a platform upon which treatments for several types of cancer and diabetes are being developed. PharmaCyte’s treatment for cancer involves encapsulating genetically modified live cells that convert an inactive chemotherapy drug into its active or “cancer-killing” form. These encapsulated live cells are placed as close to a cancerous tumor as possible. Once implanted in a patient, a chemotherapy drug which needs to be activated in the body (ifosfamide) is then given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been placed. When the ifosfamide, which is normally activated in the liver, comes in contact with the encapsulated live cells, activation of the chemotherapy drug takes place at the source of the cancer without any side effects from the chemotherapy. This “targeted chemotherapy” has proven remarkably effective and safe to use in past clinical trials.
In addition to developing a novel treatment for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and Type 2 insulin-dependent diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte Biotech and its future events and results that involve inherent risks and uncertainties. The words "anticipate", "believe", "estimate", "expect", "intend", "plan" and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte's ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte’s intellectual property and PharmaCyte’s continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: Info@PharmaCyte.com
AMMX News May 16, 2016
AmeraMex International Reports Revenue of $3.2 Million with Net Income of $1.2 Million for the First Quarter of 2016
CHICO, CA – May 16, 2016 – AmeraMex International, Inc. (OTC:AMMX), a provider of heavy equipment for logistics companies (stevedoring/shipping), infrastructure construction, logging and mining companies, today reported financial results for its first quarter ended March 31, 2016.
Highlights for the First Quarter Ended March 31, 2016
· Revenue for the first quarter was approximately $3.2 million versus revenue of $.936 for the comparable quarter.
· Gross profit for the first quarter was approximately $2.7 million—a significant increase when compared to gross profit of $.549 million for the quarter ended March 31, 2015.
· Gross profit margin, as a percentage of revenue, was 83 percent, versus a gross profit margin of 59 percent for the comparable 2015 quarter.
· The company net profit for the quarter of $1.2 million (after taxes) compared to net profit of $.020 million for the first quarter ended March 31, 2015.
· First quarter balance sheet continues to improve when compared to the 2015 year-end balance sheet.
· AmeraMex International received $3.7 million in sales and rental contracts during the first quarter and to date in the second quarter, the company has reported approximately $1.8 million in sales.
AmeraMex CEO Lee Hamre commented, “Progress has been made with various projects in our African pipeline but there hasn’t been any significant breakthroughs since our March 25 conference call. When there are, we will happily share the information via a public news release.
“We are gearing up for the beginning of our SEC audit and are pleased to ‘get the show on the road’ to become a fully reporting company. I am also happy to report that we expect to sign a dealership agreement with a U.S.-based equipment manufacturer. This dealership agreement will increase the depth of our product line and provide a more cost-effect solution for our potential customers in Africa. We are pushing to make this happen within the next three to four weeks, added Hamre.”
Guys I know SCNP is at an ask of $8.50 but it is about to break out.
As you know I am partial to Golden Retrievers.
AMMX IS NEW PICK GUYS :D
HUGE news 2 days ago and according to ceo, they have great things headed our way :) http://www.ammx.net/
ha me too, thanks
I absolutely love that picture.
Now at .0039
$URBF
.002 looks to be bottom with 22.98 RSI
$URBF
__URBF kaBOOMage .0032 X .0039
$GCEI ALERT> I may have over read this.
In case you guys missed it.
The term "named executive officer" refers to our principal executive officer, our two most highly compensated executive officers other than the principal executive officer who were serving as executive officers at the end of 2015 and two additional individuals for whom disclosure would have been provided but for the fact that the individuals were not serving as executive officers of the Company at the end of 2015.
2015 annual report filed april 15,2016 (page 27 Top of page.) (above)
http://www.otcmarkets.com/financialReportViewer?symbol=GCEI&id=153010
http://www.marketwired.com/press-release/global-clean-energy-inc-year-end-update-otc-pink-gcei-1979572.htm (below)
The management team will be comprised of top executives from the 2 companies.
-----------------------------------------------------------------On track to close PGM Acquistion in Early June 2016.(below)
http://www.altenergymag.com/news/2016/05/02/global-clean-energy-on-tract-to-close-pgm-acquisition-by-early-june-2016---first-quarter-update-/23523/
If this is related to the PGM Acquisition we are close to finalizing it imho.
-----------------------------------------------------------------
STOCK IS EXTREMELY UNDERVALUED IMHO!
Go GREEN with Global Clean Energy a Enviromentally Friendly Waste 2 Fuel & PGM (Platinum-Group-Metals) Auto Catalysts Recovery Company.
http://www.globalcleanenergy.net/
glta
$AMRS QB low float bottom stock!! CHART setting for bounce >>
SPORTYNORTY is in AMMX and PYHH
Looking for HUGE moves on both heading into next week.
CGRA Q1 Out!!!!
$CGRA fins out.
$3.5 million cash on hand
$17 million in assets
https://www.otciq.com/otciq/ajax/showFinancialReportById.pdf?id=155553
SIGO when this gets PRd it's going to run. AS reduced, RM in works......
b]SIGO two more filings out AH. RM with A.W Realty; 500M AS
ARTICLE THREE. (OPERATIONS) On May 18, 2016, the Company acquired AW Realty, a Colorado Corporation. The Company
will operate through this wholly owned subsidiary with the intended business purpose of acquiring and flipping houses in California.
http://www.sos.state.co.us/biz/ViewImage.do?fileId=20161343260&masterFileId=20051365777
AS reduced from 1B to 500M; NO RS mentioned
The corporation shall have authority to issue an aggregate of FIVE HUNDRED AND THIRTY MILLION (530,000,000) shares of
stock, par value ONE MILL ($0.0001) per share divided into two (2) classes of stock as follows:
a) Non-Assessable Common Stock: Five Million (500,000,000) shares of Common stock, Par Value One Mill ($0.0001) per
share, and b) Preferred Stock: Thirty Million (30,000,000) shares of Preferred stock, Par Value One Mill ($0.0001) per share.
SIGO Merger taking place, first Registered Agent changes after hours Tuesday night, last night they filed acquiring a new company. There is 10-15m in the float
Colorado Secretary of State
Date and Time: 05/18/2016 05:39 PM
ID Number: 20051365777
$PMCB ~ PharmaCyte Biotech, Inc. DD

FDA Grants Orphan Drug Designation to Pharmacyte Biotech for Pancreatic Cancer Treatment

http://www.accessdata.fda.gov/scripts/opdlisting/oopd/OOPD_Results_2.cfm?Index_Number=457014
PharmaCyte Biotech Obtains Orphan Drug Designation in Europe for Its Pancreatic Cancer Treatment

http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/orphans/2009/11/human_orphan_000395.jsp&mid=WC0b01ac058001d12b
$PMCB PharmaCyte Biotech - Diabetes Treatment Program


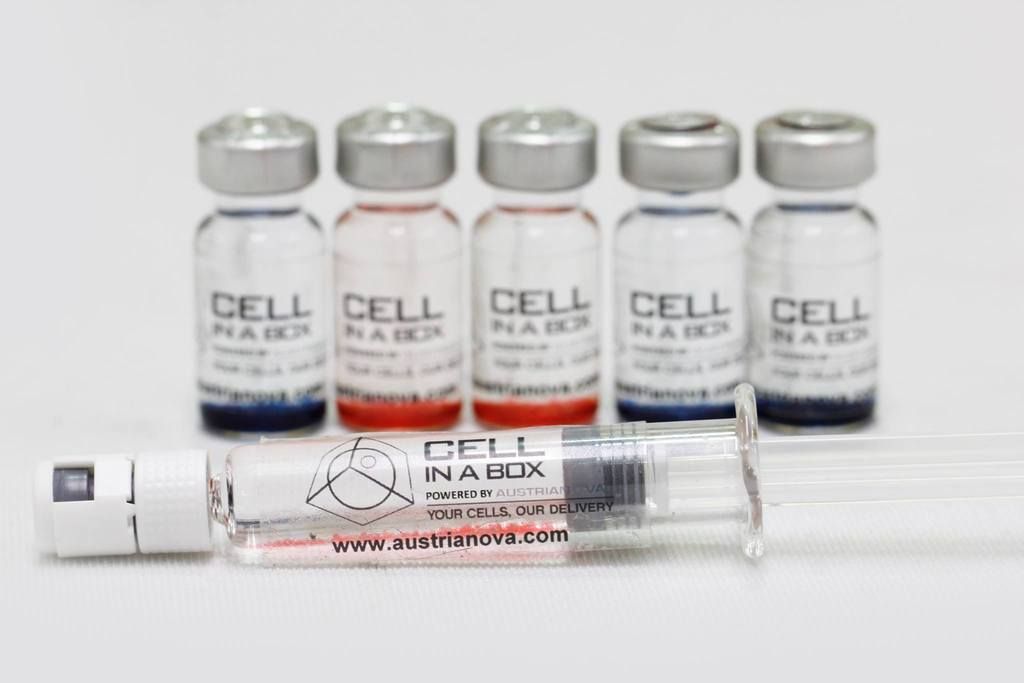
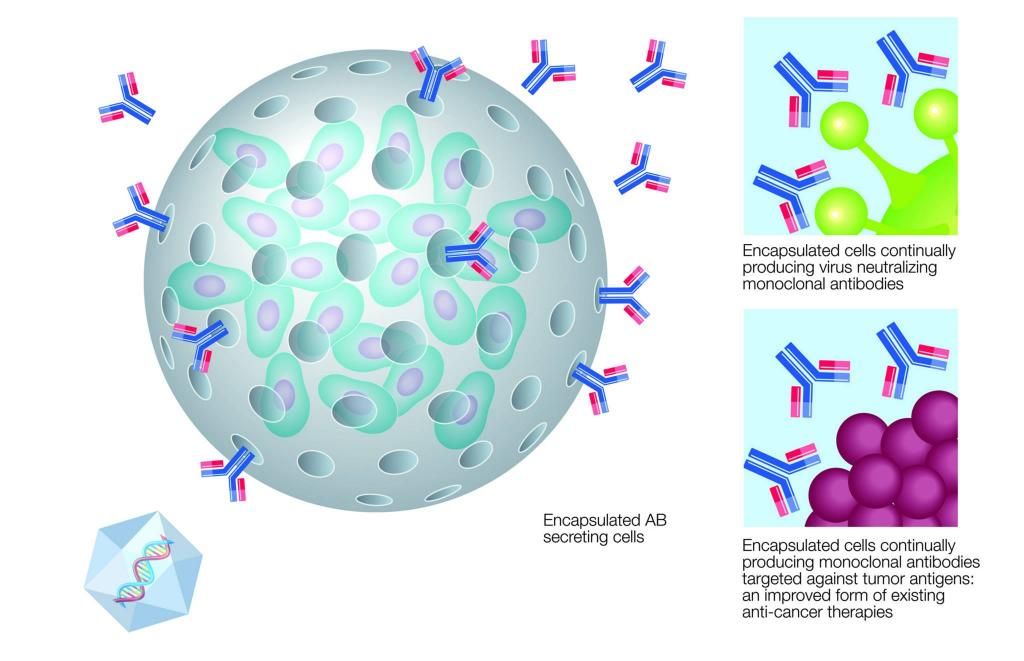

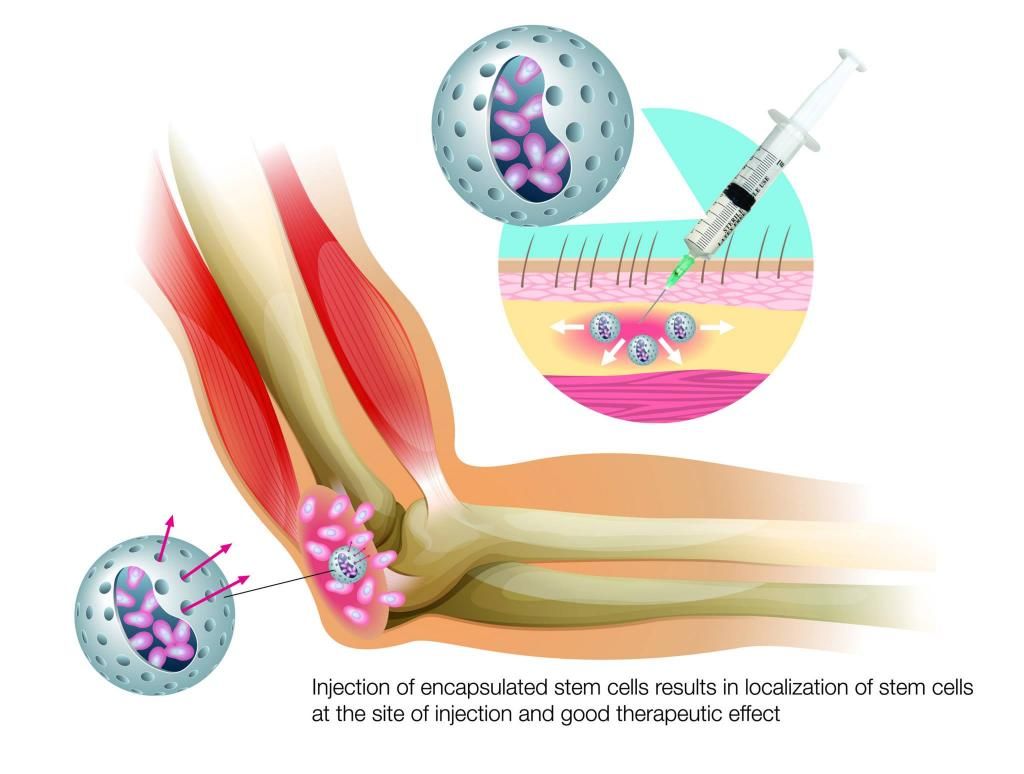
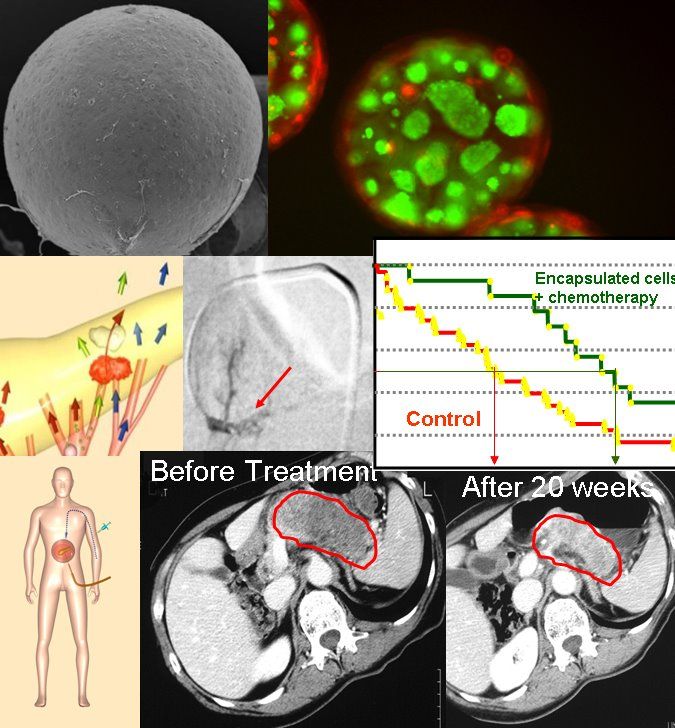
Nice ![]()
Go $PMCB
PMCB GOT MY APPROVAL
Thank you ![]()
Go $PMCB
The market has not caught on as to how big this news really is. Load and hold until everybody else figures it out! :)
|
Followers
|
363
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
14043
|
|
Created
|
04/20/14
|
Type
|
Free
|
| Moderators sharky OptimusPrime | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
