Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$RNXT RenovoRX “Unfortunately, #bileductcancer today has a poor prognosis for patients. Despite early diagnosis, the 5-yr survival rate is approx. 24%.” - Paula Novelli, MD FSIR, CouGar National Principal Investigator. Learn about RenovoRx's pivotal Ph. III trial: https://bit.ly/4asSZjH $RNXT
“Unfortunately, #bileductcancer today has a poor prognosis for patients. Despite early diagnosis, the 5-yr survival rate is approx. 24%.” - Paula Novelli, MD FSIR, CouGar National Principal Investigator. Learn about RenovoRx's pivotal Ph. III trial: https://t.co/415aZnLVk7 $RNXT pic.twitter.com/pEMHFTo0Iq
— RenovoRx (@RenovoRx) December 22, 2023
BEING SOLD OFF TODAY.. DOWN 13% WHERE ARE THE PAID PUMPERS? GOING BACK UNDER A DOLLAR... FOR GOOD
$RNXT RenovoRx Engaged Oklahoma University (OU) Health as First Clinical Site in Preparation for the Pivotal Phase III CouGar Clinical Trial in Bile Duct Cancer - #NEWS https://www.businesswire.com/news/home/20231221037143/en/RenovoRx-Engages-Oklahoma-University-OU-Health-as-First-Clinical-Site-in-Preparation-for-the-Pivotal-Phase-III-CouGar-Clinical-Trial-in-Bile-Duct-Cancer?utm_campaign=shareaholic&utm_medium=twitter&utm_source=socialnetwork
WHERE ARE ALL THE PAID PUMPERS? NO BS SPEWING TODAY? THIS IS GOING TO HAVE A REVERSE SPLIT OR GO TO ZERO ON THE PINKSHEETS... BOTH ARE BAD..LOL
$RNXT is UP 25.00% on Amazing new Patent News! RenovoRx Holds a Strong Intellectual Property ? Portfolio with 9 Issued Patents for its Proprietary Trans-Arterial Micro-Perfusion (TAMP™) Therapy Platform and Delivery System https://t.co/TDqAoUmVtl
SO IF ALL THIS HUGE NEWS YOU FEW KEEP SPLASHING OVER AND OVER ON ALL THE BOARDS IS SO GREAT...
WHY IS THE STOCK .70'S ??? PLEASE ANSWER THIS QUESTION.
$RNXT More Updates: The pivotal Phase III CouGar study will be the second clinical trial evaluating RenovoGem™, a combination drug-device product, in a difficult-to-access solid tumor cancer. Learn more: https://bit.ly/4asSZjH
#BileDuctCancer
The pivotal Phase III CouGar study will be the second clinical trial evaluating RenovoGem™, a combination drug-device product, in a difficult-to-access solid tumor cancer. Learn more: https://t.co/415aZnLVk7 $RNXT #BileDuctCancer pic.twitter.com/846Lnj84jc
— RenovoRx (@RenovoRx) December 21, 2023
The pivotal Phase III CouGar study is a randomized study designed to evaluate the safety and efficacy of unresectable, locally advanced eCCA using intravenous cisplatin, gemcitabine, and durvalumab (all forms of chemotherapy) versus Trans-Arterial Micro-Perfusion via RenovoGem (delivery system and gemcitabine) with intravenous durvalumab. Oklahoma University (OU) Health is the first clinical site for the CouGar study.
$RNXT
"We are pleased to engage our first clinical site for our second pivotal clinical trial. The CouGar Trial marks an important milestone for RenovoRx as we expand our clinical pipeline for RenovoGem into bile duct cancer, a difficult-to-treat solid tumor cancer," said Leesa Gentry, Senior Vice President of Clinical Operations at RenovoRx. "I’m extremely proud of our team and our collaborators for their tremendous work reinforcing our mission to improve patient lives by delivering innovative therapies that can potentially change the current paradigm of cancer care."
$RNXT
Wow, this is great News from the company!
$RNXT
LOL...DOES NO ONE SEE THE HUGE NEWS? ONLY THE INSIDERS AND PAID PUMPERS... WHO KEEP POSTING IT OVER AND OVER.. ASK .78 .77 .76 NOW .73 ......NO TAKERS... LOL SCAM!!
lol... huge news today? Not for a stock scam from India. Should end the day down about .05 or so. All this is .... stock selling scam that no one is falling for that they say is thinly traded..lol.. 4 or 5 new paid pumper bots. Obviously paid promotion on vauge news... over and over.
Huge news thanks! $RNXT aRenovoRx Engages Oklahoma University (OU) Health as First Clinical Site in Preparation for the Pivotal Phase III CouGar Clinical Trial in Bile Duct Cancer https://finance.yahoo.com/news/renovorx-engages-oklahoma-university-ou-133000405.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$RNXT News: RenovoRx Engages Oklahoma University (OU) Health as First Clinical Site in Preparation for the Pivotal Phase III CouGar Clinical Trial in Bile Duct Cancer
This study will be the Second Clinical Trial Evaluating RenovoGem™, a Combination Drug-Device Product, in a Difficult-to-Access Solid Tumor Cancer
December 21, 2023 08:30 AM Eastern Standard Time
LOS ALTOS, Calif.--(BUSINESS WIRE)--RenovoRx, Inc. (“RenovoRx” or the “Company”) (Nasdaq: RNXT), a clinical-stage biopharmaceutical company developing novel precision oncology therapies based on a local drug-delivery platform, today announced the expansion of their clinical development pipeline in preparation for the commencement of a second Phase III trial. The CouGar Trial will evaluate RenovoGem in bile duct cancer, specifically unresectable locally advanced extrahepatic cholangiocarcinoma (eCCA).
“Unfortunately, bile duct cancer today has a poor prognosis for patients. Despite early diagnosis, the five-year survival rate is approximately 24%”
“We are pleased to engage our first clinical site for our second pivotal clinical trial. The CouGar Trial marks an important milestone for RenovoRx as we expand our clinical pipeline for RenovoGem into bile duct cancer, a difficult-to-treat solid tumor cancer,” said Leesa Gentry, Senior Vice President of Clinical Operations at RenovoRx. “I’m extremely proud of our team and our collaborators for their tremendous work reinforcing our mission to improve patient lives by delivering innovative therapies that can potentially change the current paradigm of cancer care.”
The pivotal Phase III CouGar study is a randomized study designed to evaluate the safety and efficacy of unresectable, locally advanced eCCA using intravenous cisplatin, gemcitabine, and durvalumab (all forms of chemotherapy) versus Trans-Arterial Micro-Perfusion via RenovoGem (delivery system and gemcitabine) with intravenous durvalumab. Oklahoma University (OU) Health is the first clinical site for the CouGar study.
“Unfortunately, bile duct cancer today has a poor prognosis for patients. Despite early diagnosis, the five-year survival rate is approximately 24%,” said Paula Novelli, MD FSIR, CouGar National Principal Investigator, and Associate Professor of Radiology and Director of Interventional Radiology Research at the University of Pittsburgh Medical Center. “There are currently limited treatment options for patients diagnosed with this aggressive cancer, which only prolongs life by a few months. The CouGar study brings hope for a new treatment option. RenovoGem has the potential to meet the urgent clinical need for a safe and effective therapy in this important patient population.”
Dr. David Geller, Director, UPMC Liver Cancer Center, Richard L. Simmons Professor of Surgery, University of Pittsburgh School of Medicine is the CouGar study’s Co-Investigator.
“The current standard of care for bile duct cancer is systemic (intravenous) chemotherapy, which has poor uptake and delivery since there are few blood vessels feeding this type of tumor,” said Dr. Hassan Hatoum, Principal Investigator, Associate Professor and Hematologist and Oncologist at Oklahoma University Health Stephenson Cancer Center – Gastrointestinal Cancer Clinic. “RenovoRx’s clinical trial is evaluating targeted, trans-arterial delivery of chemotherapy that is locally directed to the tumor site. Our team looks forward to participating in the CouGar study to bring this novel therapy to patients.”
RenovoGem received FDA Orphan Drug Designation for pancreatic cancer and bile duct cancer which provides 7 years of market exclusivity upon NDA approval.
About RenovoRx, Inc.
RenovoRx is a clinical-stage biopharmaceutical company developing proprietary targeted combination therapies for high unmet medical need with a goal to improve therapeutic outcomes for cancer patients undergoing treatment. The Company’s proprietary Trans-Arterial Micro-Perfusion (TAMPTM) therapy platform is designed to ensure precise therapeutic delivery to directly target the tumor while potentially minimizing a therapy’s toxicities versus systemic (intravenous (IV) therapy). RenovoRx’s unique approach to targeted treatment offers the potential for increased safety, tolerance, and improved efficacy. Our Phase III lead product candidate, RenovoGemTM, a novel oncology drug-device combination product, is being investigated under a US IND that is regulated by FDA 21 CFR 312 pathway. RenovoGem is currently being evaluated for the treatment of locally advanced pancreatic cancer (LAPC) by the Center for Drug Evaluation and Research (the drug division of FDA.)
RenovoRx is committed to transforming the lives of patients by delivering innovative solutions to change the current paradigm of cancer care. RenovoGem is currently under investigation for TAMP therapeutic delivery of gemcitabine and has not been approved for commercial sale.
For more information, visit www.renovorx.com. Follow RenovoRx on Facebook, LinkedIn, and Twitter.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, and Section 21E of the Securities Exchange Act of 1934, including but not limited to statements regarding our clinical trials and studies, including anticipated timing, statements regarding the potential of RenovoCath®, RenovoGemTM or TAMPTM or regarding our ongoing TIGeR-PaC Phase III clinical trial study in LAPC, statements regarding the potential for our product candidates to treat or provide clinically meaningful outcomes for certain medical conditions or diseases, and our preliminary financial results, cash position and related ability to continue as a going concern. Statements that are not purely historical are forward-looking statements. The forward-looking statements contained herein are based upon our current expectations and beliefs regarding future events, many of which, by their nature, are inherently uncertain, outside of our control and involve assumptions that may never materialize or may prove to be incorrect. These may include estimates, projections and statements relating to our research and development plans, clinical trials, therapy platform, business plans, objectives and expected operating results, which are based on current expectations and assumptions that are subject to known and unknown risks and uncertainties that may cause actual results to differ materially from those expressed or implied by these forward-looking statements. These statements may be identified using words such as “may,” “expects,” “plans,” “aims,” “anticipates,” “believes,” “forecasts,” “estimates,” “intends,” and “potential,” or the negative of these terms or other comparable terminology regarding RenovoRx’s expectations strategy, plans or intentions, although not all forward-looking statements contain these words. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, that could cause actual events to differ materially from those projected or indicated by such statements, including, among other things: the timing of the initiation, progress and potential results of our preclinical studies, clinical trials and our research programs; the possibility that interim results may not be predictive of the outcome of our clinical trial, which may not demonstrate sufficient safety and efficacy to support regulatory approval of our product candidate, or the regulatory authority may disagree with our interpretation of the data; research and clinical development plans and timelines, and the regulatory process for our product candidates; future potential regulatory milestones for our product candidates, including those related to current and planned clinical studies; our ability to use and expand our therapy platform to build a pipeline of product candidates; our ability to advance product candidates into, and successfully complete, clinical trials; the timing or likelihood of regulatory filings and approvals; our estimates of the number of patients who suffer from the diseases we are targeting and the number of patients that may enroll in our clinical trials; the commercialization potential of our product candidates, if approved; our ability and the potential to successfully manufacture and supply our product candidates for clinical trials and for commercial use, if approved; future strategic arrangements and/or collaborations and the potential benefits of such arrangements; our estimates regarding expenses, future revenue, capital requirements and needs for additional financing and our ability to obtain additional capital; the sufficiency of our existing cash and cash equivalents to fund our future operating expenses and capital expenditure requirements; our ability to retain the continued service of our key personnel and to identify, hire and retain additional qualified personnel; the implementation of our strategic plans for our business and product candidates; the scope of protection we are able to establish and maintain for intellectual property rights, including our therapy platform, product candidates and research programs; our ability to contract with third-party suppliers and manufacturers and their ability to perform adequately; the pricing, coverage and reimbursement of our product candidates, if approved; developments relating to our competitors and our industry, including competing product candidates and therapies; negative impacts of the ongoing COVID-19 pandemic on our operations; our ability to comply with the continued listing standards of Nasdaq Stock Market LLC (“Nasdaq”) or the continued listing of our securities on Nasdaq; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled “Risk Factors” in documents that we file from time to time with the Securities and Exchange Commission.
Forward-looking statements included herein are made as of the date hereof, and RenovoRx does not undertake any obligation to update publicly such forward-looking statements to reflect subsequent events or circumstances, except as required by law.
Contacts
Investor Contact:
KCSA Strategic Communications
Valter Pinto or Jack Perkins
T:212-896-1254
renovorx@kcsa.com
Media Contact:
Kimberly Ha
KKH Advisors
917-291-5744
kimberly.ha@kkhadvisors.com
https://www.businesswire.com/news/home/20231221037143/en/RenovoRx-Engages-Oklahoma-University-OU-Health-as-First-Clinical-Site-in-Preparation-for-the-Pivotal-Phase-III-CouGar-Clinical-Trial-in-Bile-Duct-Cancer?utm_campaign=shareaholic&utm_medium=twitter&utm_source=socialnetwork
.72 thin on the ask here $RNXT Research Supports RenovoRx’s Lead Oncology Product Candidate, RenovoGem™, and its Novel Therapy Platform, TAMP™, for the Treatment of Locally Advanced Pancreatic Cancer
LOS ALTOS, Calif., December 19, 2023--(BUSINESS WIRE)--RenovoRx, Inc. (Nasdaq: RNXT), a clinical-stage biopharmaceutical company developing novel precision oncology therapies based on a local drug-delivery platform, today announced the acceptance of a clinical data abstract for the 2024 Society of Interventional Radiology (SIR) Annual Scientific Meeting, held March 23-28 in Salt Lake City, Utah. The abstract will highlight the Company’s lead product candidate, RenovoGem, and proprietary Trans-Arterial Micro-Perfusion (TAMP) therapy platform for the treatment of Locally Advanced Pancreatic Cancer (LAPC), a difficult-to-access, solid tumor cancer.
"We are pleased to present research supporting our TAMP therapy platform at the upcoming SIR 2024 conference, the largest professional gathering of interventional radiologists in North America," said Shaun Bagai, CEO of RenovoRx. "This research adds to the continued progress of our pivotal Phase III TIGeR-PaC study in LAPC. We remain steadfast in our commitment to deliver transformative therapies to potentially extend and improve the quality of life of cancer patients."
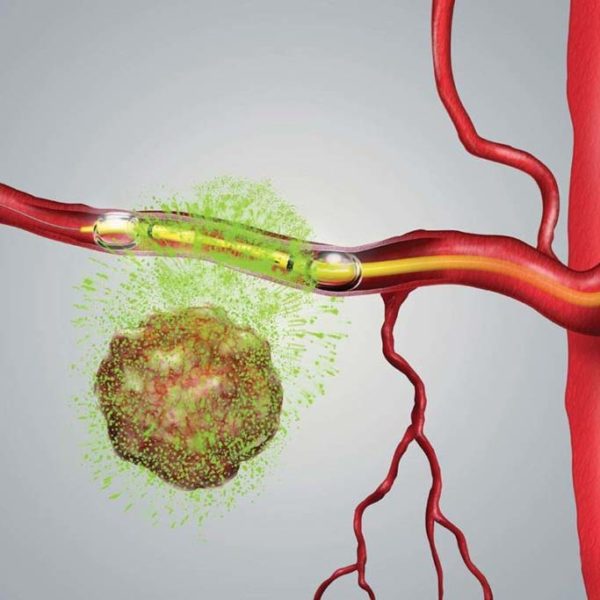
$RNXT RenovoRx TAMP Therapy platform delivery system in health care. #FDA
$RNXT RenovoRx holds a strong IP portfolio with 9 issued patents for its TAMP therapy platform and delivery system. https://bit.ly/3uUlo1Y
$RNXT is a Biopharma Company that is at Phase 3 for Cancer Treatments; 9 Patents; Collaboration with Imugene: RenovoRx
https://apnews.com/press-release/ein-presswire-newsmatics/medical-research-clinical-trials-cancer-c5e2cf2e84632b2b5156b9659aa6136f
Are you looking forward to when they announce the reverse split to stay on the Nasdaq? Does that effect free shares? Please advise
Looking forward to hearing more from the company on further developments...
Research Supports RenovoRx’s Lead Oncology Product Candidate, RenovoGem™, and its Novel Therapy Platform, TAMP™, for the Treatment of Locally Advanced Pancreatic Cancer
$RNXT
HAHAHAHAHAHA.... 17 STOCKS ON MY SCREEN.. ONE DOWN.. 10% LOL.. THE ONLY ONE BEING PAID PUMPED...
SOMEONE NEEDED SOME CHRISTMAS $
CROOKS
LOL... big news.. March 23-28 this will be a micro penny or have had a huge reverse split to stay selling shares on the Nasdaq.. either way you are screwing investors by pumping this stock scam from India. stop it
so funny... some idiot bought INSIDERS shares for .90 because of these idiot pumpers. Right back to .75x.77 at the open because there are no new suckers and only insiders selling with the desperate help of paid pumpers. Going to zero
$RNXT .86 +7.90% Up Pre-market on News! https://finance.yahoo.com/news/renovorx-announces-clinical-data-abstract-133000856.html
"We are pleased to present research supporting our TAMP therapy platform at the upcoming SIR 2024 conference, the largest professional gathering of interventional radiologists in North America," said Shaun Bagai, CEO of RenovoRx. "This research adds to the continued progress of our pivotal Phase III TIGeR-PaC study in LAPC. We remain steadfast in our commitment to deliver transformative therapies to potentially extend and improve the quality of life of cancer patients."
$RNXT
$RNXT News: RenovoRx Announces Clinical Data Abstract at the 2024 Society of Interventional Radiology Annual Scientific Meeting
Research Supports RenovoRx’s Lead Oncology Product Candidate, RenovoGem™, and its Novel Therapy Platform, TAMP™, for the Treatment of Locally Advanced Pancreatic Cancer
December 19, 2023
LOS ALTOS, Calif.--(BUSINESS WIRE)-- RenovoRx, Inc. (Nasdaq: RNXT), a clinical-stage biopharmaceutical company developing novel precision oncology therapies based on a local drug-delivery platform, today announced the acceptance of a clinical data abstract for the 2024 Society of Interventional Radiology (SIR) Annual Scientific Meeting, held March 23-28 in Salt Lake City, Utah. The abstract will highlight the Company’s lead product candidate, RenovoGem, and proprietary Trans-Arterial Micro-Perfusion (TAMP) therapy platform for the treatment of Locally Advanced Pancreatic Cancer (LAPC), a difficult-to-access, solid tumor cancer.
“We are pleased to present research supporting our TAMP therapy platform at the upcoming SIR 2024 conference, the largest professional gathering of interventional radiologists in North America,” said Shaun Bagai, CEO of RenovoRx. “This research adds to the continued progress of our pivotal Phase III TIGeR-PaC study in LAPC. We remain steadfast in our commitment to deliver transformative therapies to potentially extend and improve the quality of life of cancer patients.”
Presentation Details: Title: Mesenteric Venous Thrombosis as a Predictor of Target Artery Thrombosis and Tran-Arterial Micro Perfusion Treatment Completion Among Patients with Locally Advanced Pancreatic Cancer
Authors: Michel Accad, MD, et al.
Abstract ID: 1632169
For more information about the conference, visit https://www.sirmeeting.org.
About RenovoRx, Inc.
RenovoRx is a clinical-stage biopharmaceutical company developing proprietary targeted combination therapies for high unmet medical need with a goal to improve therapeutic outcomes for cancer patients undergoing treatment. The Company’s proprietary Trans-Arterial Micro-Perfusion (TAMP™) therapy platform is designed to ensure precise therapeutic delivery to directly target the tumor while potentially minimizing a therapy’s toxicities versus systemic (intravenous (IV) therapy). RenovoRx’s unique approach to targeted treatment offers the potential for increased safety, tolerance, and improved efficacy. Our Phase III lead product candidate, RenovoGem™, a novel oncology drug-device combination product, is being investigated under a US IND that is regulated by FDA 21 CFR 312 pathway. RenovoGem is currently being evaluated for the treatment of locally advanced pancreatic cancer (LAPC) by the Center for Drug Evaluation and Research (the drug division of FDA.)
RenovoRx is committed to transforming the lives of patients by delivering innovative solutions to change the current paradigm of cancer care. RenovoGem is currently under investigation for TAMP therapeutic delivery of gemcitabine and has not been approved for commercial sale.
For more information, visit www.renovorx.com. Follow RenovoRx on Facebook, LinkedIn, and Twitter.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, and Section 21E of the Securities Exchange Act of 1934, including but not limited to statements regarding our clinical trials and studies, including anticipated timing, statements regarding the potential of RenovoCath®, RenovoGem™ or TAMP™ or regarding our ongoing TIGeR-PaC Phase III clinical trial study in LAPC, statements regarding the potential for our product candidates to treat or provide clinically meaningful outcomes for certain medical conditions or diseases, and our preliminary financial results, cash position and related ability to continue as a going concern. Statements that are not purely historical are forward-looking statements. The forward-looking statements contained herein are based upon our current expectations and beliefs regarding future events, many of which, by their nature, are inherently uncertain, outside of our control and involve assumptions that may never materialize or may prove to be incorrect. These may include estimates, projections and statements relating to our research and development plans, clinical trials, therapy platform, business plans, objectives and expected operating results, which are based on current expectations and assumptions that are subject to known and unknown risks and uncertainties that may cause actual results to differ materially from those expressed or implied by these forward-looking statements. These statements may be identified using words such as “may,” “expects,” “plans,” “aims,” “anticipates,” “believes,” “forecasts,” “estimates,” “intends,” and “potential,” or the negative of these terms or other comparable terminology regarding RenovoRx’s expectations strategy, plans or intentions, although not all forward-looking statements contain these words. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, that could cause actual events to differ materially from those projected or indicated by such statements, including, among other things: the timing of the initiation, progress and potential results of our preclinical studies, clinical trials and our research programs; the possibility that interim results may not be predictive of the outcome of our clinical trial, which may not demonstrate sufficient safety and efficacy to support regulatory approval of our product candidate, or the regulatory authority may disagree with our interpretation of the data; research and clinical development plans and timelines, and the regulatory process for our product candidates; future potential regulatory milestones for our product candidates, including those related to current and planned clinical studies; our ability to use and expand our therapy platform to build a pipeline of product candidates; our ability to advance product candidates into, and successfully complete, clinical trials; the timing or likelihood of regulatory filings and approvals; our estimates of the number of patients who suffer from the diseases we are targeting and the number of patients that may enroll in our clinical trials; the commercialization potential of our product candidates, if approved; our ability and the potential to successfully manufacture and supply our product candidates for clinical trials and for commercial use, if approved; future strategic arrangements and/or collaborations and the potential benefits of such arrangements; our estimates regarding expenses, future revenue, capital requirements and needs for additional financing and our ability to obtain additional capital; the sufficiency of our existing cash and cash equivalents to fund our future operating expenses and capital expenditure requirements; our ability to retain the continued service of our key personnel and to identify, hire and retain additional qualified personnel; the implementation of our strategic plans for our business and product candidates; the scope of protection we are able to establish and maintain for intellectual property rights, including our therapy platform, product candidates and research programs; our ability to contract with third-party suppliers and manufacturers and their ability to perform adequately; the pricing, coverage and reimbursement of our product candidates, if approved; developments relating to our competitors and our industry, including competing product candidates and therapies; negative impacts of the ongoing COVID-19 pandemic on our operations; our ability to comply with the continued listing standards of Nasdaq Stock Market LLC (“Nasdaq”) or the continued listing of our securities on Nasdaq; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled “Risk Factors” in documents that we file from time to time with the Securities and Exchange Commission.
Forward-looking statements included herein are made as of the date hereof, and RenovoRx does not undertake any obligation to update publicly such forward-looking statements to reflect subsequent events or circumstances, except as required by law.
https://cts.businesswire.com/ct/CT?id=bwnews&sty=20231219375297r1&sid=acqr8&distro=nx&lang=en
View source version on businesswire.com: https://www.businesswire.com/news/home/20231219375297/en/
Investor Contact:
KCSA Strategic Communications
Valter Pinto or Jack Perkins
T:212-896-1254
renovorx@kcsa.com
Media Contact:
Kimberly Ha
KKH Advisors
917-291-5744
kimberly.ha@kkhadvisors.com
Source: RenovoRx, Inc.
$RNXT RenovoRx today announced the acceptance of a clinical data abstract for the 2024 Society of Interventional Radiology (SIR) Annual Scientific Meeting, held March 23-28 in Salt Lake City, Utah. The abstract will highlight the Company’s lead product candidate, RenovoGem™, and proprietary Trans-Arterial Micro-Perfusion (TAMP) therapy platform for the treatment of Locally Advanced Pancreatic Cancer (LAPC), a difficult-to-access, solid tumor cancer. For details: https://bit.ly/3NAWc74
#SIR24SLC #PancreaticCancer #LocallyAdvancedPancreaticCancer #LAPC https://www.linkedin.com/posts/renovorx_sir24slc-pancreaticcancer-locallyadvancedpancreaticcancer-activity-7142871521638699008-S4DW?utm_source=share&utm_medium=member_desktop
Expecting updates on these clinical programs.
"I look forward to contributing to the advancement of the Company’s novel clinical programs that have the potential to revolutionize the treatment of challenging cancers."
$RNXT
$RNXT RenovoRx holds a strong IP portfolio with 9 issued patents for its TAMP therapy platform and delivery system. https://bit.ly/3uUlo1Y
RNXT looks like all the right pieces are falling into place for this company to take a significant position in the vital high tech biotech field with special emphasis in oncology advancements. The recent news of Michel Ducreux, M.D., Ph.D. joining the company's advisory board was another very positive development.
RenovoRx Expands Scientific Advisory Board with Appointment of Michel Ducreux, M.D., Ph.D.
https://finance.yahoo.com/news/renovorx-expands-scientific-advisory-board-133500185.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr
RenovoRx, Inc. ("RenovoRx" or the "Company") (Nasdaq: RNXT), a clinical-stage biopharmaceutical company developing targeted combination therapies, today announced the appointment of Michel Ducreux, M.D., Ph.D. to the Company’s Scientific Advisory Board (SAB). Dr. Ducreux is the Head of the Gastrointestinal Oncology Unit and Gastrointestinal Oncology Tumor Board at Gustave Roussy, Professor of Oncology at Paris-Saclay University in France, and Vice-Chair of ESMO GI.
Dr. Ducreux was trained in medicine, gastroenterology, and gastrointestinal tract oncology at the University of Paris Sud. Dr. Ducreux earned his master’s degree in biological sciences and PhD in health sciences. He has held previous positions as assistant physician and professor of oncology at the Gastrointestinal Oncology Unit of Gustave Roussy and Paul Brousse Hospital in Villejuif, France. He was a Medical Affairs Director at Gustave Roussy from January 2011 to December 2019. He is the former Chair of the European Organisation for Research and Treatment of Cancer (EORTC) Gastrointestinal Tract Cancer Group and the former Chair of the Gastrointestinal Group of the French Federation of Anticancer Centers (FNCLCC). He is a co-editor for gastrointestinal oncology of the European Journal of Cancer.
"I am excited to be working with my distinguished colleagues on RenovoRx’s Scientific Advisory Board," stated Dr. Ducreux. "I look forward to contributing to the advancement of the Company’s novel clinical programs that have the potential to revolutionize the treatment of challenging cancers."
Dr. Ducreux added, "Having previously worked on intra-arterial and intra-tumoral delivery, what excites me most about RenovoRx’s published data is its platform delivery mechanism. The trans-arterial micro-perfusion platform appears to unlock best-in-class improvements to increase local therapeutic tissue concentration with deep tissue penetration to overcome dense tumor microenvironments while avoiding dose-limiting systemic toxicities beyond conventional intra-tumoral and traditional intra-arterial delivery. This platform has the potential to extend across a variety of high unmet needs beyond targeted delivery of gemcitabine and pancreatic tumors."
lol... of course they do... still going to zero.. or will be a huge reverse split to stay on Nasdaq... either way all investors will be wiped out.. except the insider scammers.
Very nice News this week...
RenovoRx believes that these methods and apparatuses via the TAMP therapy platform may provide a novel and important pathway for the targeted delivery of therapeutic classes across DNA/RNA-altering modalities, cell therapy, oncolytic viruses, bi-specific antibodies, and monoclonal antibodies for the treatment of a variety of clinical indications. The Company anticipates that the patent application will be published in May of 2024.
$RNXT
I'M VERY CURIOUS HOW MUCH YOU NEW GUYS ARE PAID TO POST. IT'S SO OBVIOUS WHAT YOUR DOING.. SAME SHIT ON MULTIPLE BOARDS... SAME POSTS... WERE YOU GIVEN SHARES OR PAID SO THEY CAN DUMP THEIRS? FOR WE KNOW THIS IS INSIDERS POSTING... OR SOME OF THE CHINESE EMPLOYESS ON LUNCH BREAK AT THE RESTARAUNT.... CURIOUS
$RNXT - New Patent Would Expand IP Coverage for TAMP to Deliver Additional Therapeutic Classes Across DNA/RNA Altering Modalities, Cell Therapy, and Antibody-Based Therapies
https://finance.yahoo.com/news/renovorx-files-international-patent-novel-133100235.html
$RNXT RenovoRx Holds a Strong Intellectual Property (IP) Portfolio with 9 Issued Patents for its Proprietary Trans-Arterial Micro-Perfusion (TAMP™) Therapy Platform and Delivery System https://finance.yahoo.com/news/renovorx-files-international-patent-novel-133100235.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
lol.. will you stop it.. paid pumping is a crime. This is a pos scam from India. Google the address of their office.. it is a Chinese Restaurant ... try to call.
It's now trading 7x over its 10-day average volume as lunchtime continues... UP almost 26%, a nice continuation of volume and price action on yesterday's News!
$RNXT
It's trading nearly 2x within the first 30 minutes of the day... UP almost 11%! Looking good.
$RNXT
It's now trading 17x over its 10-day average volume in Power Hour... UP almost 23% on today's News! Looking good.
$RNXT
$RNXT +18.60% nice buys going, super thin to .80's!
$RNXT Bullish Move on NEWS .77 +21.26% HOD .87 on NEWS!
It's trading 11x over its 10-day average volume within the first hour... UP almost 28% on today's News!
$RNXT
$RNXT RenovoRx Files New International Patent for Novel Targeted Combination Drug-Delivery Oncology Therapy Platform
https://www.businesswire.com/news/home/20231213583366/en/
RenovoRx Holds a Strong Intellectual Property (IP) Portfolio with 9 Issued Patents for its Proprietary Trans-Arterial Micro-Perfusion (TAMP™) Therapy Platform and Delivery System
New Patent Would Expand IP Coverage for TAMP to Deliver Additional Therapeutic Classes Across DNA/RNA Altering Modalities, Cell Therapy, and Antibody-Based Therapies
LOS ALTOS, Calif.--(BUSINESS WIRE)--RenovoRx, Inc. (Nasdaq: RNXT), a clinical-stage biopharmaceutical company developing novel precision oncology therapies based on a local drug-delivery platform, announced today that the Company has filed an international patent application under the Patent Cooperation Treaty (PCT) for its novel Trans-Arterial Micro-Perfusion (TAMP) therapy platform. The Company already holds a strong intellectual property portfolio with 9 issued patents and 9 pending patents for its proprietary TAMP platform and delivery system in the US, EU, and Asia. The patent portfolio covers two main areas, mechanical and biological.
The new international patent application is filed under the Patent Cooperation Treaty on methods and apparatuses that may be used to deliver one or more therapeutic agents through the vaso vasorum (small blood vessels that supply the walls of larger arteries or veins) to a target tissue. RenovoRx believes that these methods and apparatuses via the TAMP therapy platform may provide a novel and important pathway for the targeted delivery of therapeutic classes across DNA/RNA-altering modalities, cell therapy, oncolytic viruses, bi-specific antibodies, and monoclonal antibodies for the treatment of a variety of clinical indications. The Company anticipates that the patent application will be published in May of 2024.
"This patent application further expands the value of TAMP into the delivery of larger therapeutic assets, as a unique platform in oncology, beyond our current Phase III clinical asset," said Shaun Bagai, CEO, RenovoRx. "Once approved, it will further extend our IP coverage, opening up new market potential, expanding the upside value proposition of TAMP commercially. We look forward to providing further highlights and updates related to this patent, as well as our progress in our Phase III TIGeR-PaC clinical program."
TAMP is being investigated in the Company’s Phase III TIGeR-PaC clinical trial, an ongoing randomized multi-center study. The study is evaluating trans-arterial delivery of an FDA-approved chemotherapy, gemcitabine, to treat Locally Advanced Pancreatic Cancer (LAPC) following stereotactic body radiation therapy (SBRT). The study is comparing treatment of gemcitabine with TAMP versus systemic IV administration of gemcitabine and nab-paclitaxel.
The first of two interim analyses was completed in March 2023, and the Data Monitoring Committee (DMC) recommended a continuation of the study. The study is prespecified to provide a primary endpoint of a 6-month Overall Survival benefit and secondary endpoints including reduced side effects versus standard of care.
$RNXT .84 +32.00% on NEWS! New Patent Would Expand IP Coverage for TAMP to Deliver Additional Therapeutic Classes Across DNA/RNA Altering Modalities, Cell Therapy, and Antibody-Based Therapies https://www.businesswire.com/news/home/20231213583366/en/RenovoRx-Files-New-International-Patent-for-Novel-Targeted-Combination-Drug-Delivery-Oncology-Therapy-Platform
|
Followers
|
6
|
Posters
|
|
|
Posts (Today)
|
1
|
Posts (Total)
|
197
|
|
Created
|
08/31/21
|
Type
|
Free
|
| Moderators | |||
RenovoRx is a clinical-stage biopharmaceutical company developing proprietary targeted combination therapies for high unmet medical need with a goal to improve therapeutic outcomes for cancer patients undergoing treatment. We are committed to developing transformative therapies for improved quality of life and extended life in cancer patients.
Developing Innovative Combination Therapies for Targeted Treatment of Difficult-to-Access Cancers
The Phase III TIGeR-PaC clinical trial is an ongoing randomized multi-center study using RenovoRx’s innovative therapy platform, Trans-Arterial Micro-Perfusion (TAMP™), to evaluate RenovoRx’s first product candidate, RenovoGem™. The study is evaluating trans-arterial delivery, a form of intra-arterial administration, of an FDA-approved chemotherapy, gemcitabine, to treat LAPC following stereotactic body radiation therapy (SBRT). The study is comparing treatment of gemcitabine with TAMP versus systemic IV administration of gemcitabine and nab-paclitaxel.
RenovoRx’s proprietary Trans-Arterial Micro-Perfusion (TAMP) therapy platform is designed to ensure precise therapeutic delivery to directly target the tumor while potentially minimizing a therapy’s toxicities versus systemic (intravenous (IV) therapy). RenovoRx’s unique approach to targeted treatment offers the potential for increased safety, tolerance, and improved efficacy. Our Phase III lead product candidate, RenovoGem, a novel oncology drug-device combination product, is being investigated under a US IND that is regulated by FDA 21 CFR 312 pathway. RenovoGem is currently being evaluated for the treatment of locally advanced pancreatic cancer (LAPC) by the Center for Drug Evaluation and Research (the drug division of FDA.)
RenovoGem will also be evaluated as a potential therapy in bile duct cancer, with a Phase III clinical trial expected to begin in late 2023 with other potential pipeline indication opportunities to follow including non-small cell lung cancer, uterine tumors, glioblastoma, and sarcoma. RenovoGem received FDA Orphan Drug Designation for pancreatic cancer and bile duct cancer which provides 7 years of market exclusivity upon NDA approval.



Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
