Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
No point to give up now. My understanding is still waiting on financing, unfortunately there’s no set time. I still believe they’ll Get it done. I’ve been patient for several years now so I’m hanging on, but that’s just me.
When are we going to hear about these positive things?
Maybe things might start happening sooner rather than later
Boy you sure won't get rich selling $600 worth of stock here and there. And keep doing this for a couple of years. Maybe things might start happening sooner rather than later
Yep, par for the course, another filing saying that great things are imminent. What do they have now, $2k in cash and total assets of $21k? Versus liabilities - most of which is salaries accrued to themselves of >$3m. And their amazing tech still hasn't been discovered by industry.....And the 10+ year old tech is still really current, despite no valid research or clinical studies, only the old one that was crooked as hell according to the FDA who shut it down. But don't worry, awesome things are on the horizon. I see that two staff have lent the company some $, $70k odd, and accrued salaries went up >$500k. Wonder if that is their risk/reward payoff. Maybe they have found some sucker to fund something, but it will come to nothing in the end. Amazing how long McCoy has been able to keep this gravy train rolling for though.
From 10Q filed today.
No new shares issued/sold for years.
https://www.sec.gov/Archives/edgar/data/1412659/000166225218000083/rgin10q.htm
From 10Q filed today.
Overview
We have been working for the past year on a significant debt financing which we hope to consummate within the next quarter. As previously noted, such financings are difficult to complete and cannot be relied upon. As a result, we have also been in ongoing discussions with several additional financing sources, including a foreign government related to a potential joint venture and licensing opportunity. Although it has taken much longer than anticipated, we remain strong in our expectation of success, and our focus continues to be to secure financing that will minimize shareholder dilution as much as possible.
Upon obtaining such funding, our operating plan involves the following:
We intend to finalize our IND application with the FDA in order to begin clinical trials. This will involve several steps, including the performance of certain additional independent testing as requested by the FDA, as well as updating NovaDerm’s Orphan Status with the Agency. We have already had discussions with a well-respected University to conduct these additional independent tests. All necessary materials for the tests will be produced by the same group that manufactured the material for the previous research study. A Clinical Research Organization (CRO) familiar with the NovaDerm product will assist in the independent testing as well as our preparation of the IND. We estimate it will take about six months to complete this process once financing is obtained at a cost of approximately $1.5 million for related testing, engineering runs and special equipment.
Upon completion of the IND, we plan to begin our clinical trial. The company has already held preliminary discussions with the FDA related to the protocol to be employed, as well as, the number of patients to be included in the trial. Phase 1 of the trial will cover 10 patients. Clinical site selections will be limited to the 150 burn centers in the U.S. Our chosen CRO will assist in the management of the trial and the materials will be produced by the same group that manufactured the material for the independent tests. The initial trial will begin with a Data Safety Monitoring Board (DSMB) review of the safety of the process as shown by the first three subjects (once they have reached 6 months post-transplant). We estimate it will take 8 - 12 months to complete this trial and direct costs will be approximately $2.0 million.
Management is considering various options on the ultimate commercial manufacturing facilities of NovaDerm. No final decision has been made regarding this facility at this time.
https://www.sec.gov/Archives/edgar/data/1412659/000166225218000083/rgin10q.htm
Exactly. What a scumbag.
Nearing the end of the month. Randy has bills to pay on every shareholders back.
Watch for the 10000 share drops. $ 600 here $700 there.
A leech or say a maggot doesn't even touch this guys credibility. Pure scumbaggerry
Volume is so low....looks like nobody is buying or selling.
Keep calm and don't sell message number 80 from Nicole/Randall.
Does “positive things” mean funding is happening very very soon?
Very positive things are happening, keep calm
Any updates or insight from anybody?
If you dig around enough you can find Omniview, Broadsmoore as Reginicin shareholders. But seems like they are careful to keep them under the 5% disclosure thresh hold. They are Discala associated entities. Kyleen Kane is another that pops up. As far as the science goes, it looked OK 10 years ago but pretty dubious now it seems.
OK, that Discala. I get the need money thing but looking for movement. The science looks valid. Staying in but would sure like to see signs of life.
Discala was/is a crook and has been indicted. Broadsmore group does not own any stock. These two have been out of the picture for awhile. We are in waiting mode for financing. Has it taken awhile heck yes, but can only lose 7 cents. FB shares dropped more than that!!!!
Discala, Broadsmoore or Omniview?
Well today was a nice change. 600K plus traded and +16%. Any thoughts on why and what it means?
It's got scam written all over it in capital letters. Look at the trail of promises on this board promising great things just over the horizon. Never happens. Look at their own accounts and SEC returns. They have no money. There are 50+ companies chasing this space, most of them way way ahead of these clowns. They actually have products for example, Reginicin has a long dead brand and research that is >10 years old and was discredited by FDA etc. The claims extend to owning things they don't - like Hikurangi in NZ, never had any ownership, proved by public records. Do a search on Discala indictment. Do some DD on the names. You will easily find associations and links to Reginicin. Discala is alleged to have arranged a massive $300m pump and dump scheme. Is this outfit involved? Time will tell, but many of the actors are the same. Is there a prospect of these shares jumping in price? Yep, not long after hell freezes over.
Thanks Huntsman
I believe the financing is real, but I do not know a time frame.
Got a tip from a friend. Can you answers my question please?
Out of curiosity, since you said you were new to the stock, what brought it to your attention?
New to this BD and stock. Any insight to this coming financing? Or is it just typical msg bd pumping?
Just checking in to see if this had resolved.
These things do not take time. The sooner you understand that the better off you'll be.
If your foolish enough to continue on with a pipe dream from 2004 . Good luck.
The entire operation is a scam with NO intent on closing any deal or producing any product whatsoever.
Hikurangi has no farms and is sitting idle. The relationship with NZ is solid.
Yes, if they have found another plant in NZ that will be interesting. My information is that they are talking with people, but their reputation is preceeding them by a bit, and no deals have been signed. Also that they have no farms at all contracted. The Hikurangi ones are in legal dispute I believe, but this may be rumour. I find it hard to acept the "it takes time" comment as that has been what they have said for over five years - always it is just around the corner.
OK...thx...need/want to see some positive outcome here ASAP.
This is not dead. Unfortunately things sometimes take longer than we think they should, sometimes a lot longer. Everyone is working hard and there is light, but no definitive time table.
Thanks Huntsman. Any thoughts/insight on when we will movement here? Need to see signs of life soon.
RGIN is no longer associated with Hikurangi, however they do have farms and a processing plant.
Nidan - similar theme, new information. Back then, the McCoy company in NZ was claiming to own and/or operate the facility in Hikurangi. That page still shows up on their website, but is now blank. Public information you can see shows it is owned by the people McCoy fell out with. But their website still implies they have farms and facilities in NZ. My information is that they don't. So why would they claim that? My point is that this seems to just be a trail of false claims, and the pattern continues.
Isn't this an identical post to one you made many months ago? What exactly is your tie/interest in RGIN? Do you own shares? Are you trying to talk us off the stock? Why?
Get out while you can.
More rumours are going around and none of them are good.
This link is to a website in New Zealand. The claim that they own the Hikurangi facility has been removed, probably for legal reasons as public information shows this is owned by the people McCoy fell out with.
https://www.newzealandlifesciences.com/new-facilities.html
But they are still implying that they have facilities and farms in New Zealand. The rumour I've heard is that they have neither, what they do have is a trail of highly annoyed people, who wish they had never heard of McCoy.
Is it legal to make false claims in New Zealand?
Sadly NOT soon enough as some shareholders have declared bankruptcy just to keep their house.
Thank You Randy we believe in you???
LOL
Any info on that 154,627 buy at .06 today? Almost a $10,000 buy!?!
Been out of this for a while, got out around .10 but always thought it had potential.
Good luck to everyone.
Good luck Nidan, unfortunately if history is any guide, more likely to be some kind of "Discala" manipulation going down.
Well, we'll see I guess. Looking/hoping for good news after much wait.
I am keeping the faith so far.
Somebody knows something, maybe its the Arabs funding Randys endeavors.
NO IND funding has been announced,
NO FDA trial approvals,
None of the stuff Nicole was hawking since November has come to fruition,
Maybe Randy is in Korea with Kim Jong UN getting in on the funds the US are providing to keep the west coast from needing scorched skin replacement?
Any thoughts on the significance of + trades over last couple of days?
Applevision, doesn't the SEC require markets to be real? If there is a bid and offer quoted, legally, should it not actually transact?
Just a scam, pure and simple IMO. Nicole/McCoys periodic "big money coming" posts have been going on for years, but nothing ever happens. Nothing good for shareholders other than McCoy anyway.
Trading barely 10k in revenue isn't gonna cut it.
Wouldn't it be great to get some good news for once.
Ya no takers at bid or under. Stuck with this worthless holding.
|
Followers
|
71
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2931
|
|
Created
|
08/06/10
|
Type
|
Free
|
| Moderators | |||
Regenicin, Inc.
10 High Court
Little Falls, NJ 07424
**updated and correct phone number** (973) 557-8914
they filed this on aug 22 2016- http://www.otcmarkets.com/edgar/GetFilingHtml?FilingID=11558959
| Name: | Randall McCoy |
| Title: | Principal Executive Officer and Director |
| Date: | August 22, 2016 |
| Name: | John J. Weber |
| Title: | Principal Financial Officer and Director |
| Date: | August 22, 2016 |


LITTLE FALLS, N.J., June 12, 2012 /PRNewswire/ -- Regenicin, Inc. (OTC Bulletin Board: RGIN) a biotechnology company specializing in the development of and commercialization of regenerative cell therapies to restore the health of damaged tissues and organs, announced today that the Food and Drug Administration (FDA) has granted Orphan Status approval for the PermaDerm® product, the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers, that is indicated for catastrophic burn patients. The FDA had previously designated PermaDerm® to be a biological/drug (permanent skin replacement) not a medical device (temporary skin replacement). PermaDerm® is the only skin replacement technology to receive this Biological/drug designation.
COMPANY HIGHLIGHTS:
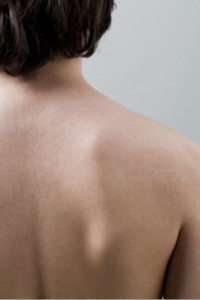
PermaDerm™ is being developed to be the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers. A small harvested section of the patient's own skin can be grown to graft an area one hundred times its size in as little as thirty days.
These living, self-to-self skin graft tissue are intended to form permanent skin tissue that will not be rejected by the immune system of the patient, a critical possibility in porcine or cadaver skin grafts used today.
The technology has been clinically tested in over 150 pediatric, catastrophic burn patients. Currently Regenicin is working with its contract manufacturer to prepare for pre-market approval of PermaDerm™ from the FDA.
PermaDerm™ is being designed to save lives, reduce healthcare costs by decreasing the patient's stay in the Critical Care Unit and reduce the need for additional surgeries. An insurance company procedural code has been approved for reimbursement of costs to hospitals. The American Medical Association has assigned CPT (Current Procedural Terminology) code for cultured skin substitutes under the dermal substitute category which enables insurance companies to process and hospitals to be reimbursed for cultured skin substitutes once approved by the FDA.
According to the American Burn Association, there are currently over 2,000 cases annually involving burns covering over 50% of the patient's total body surface area.
PermaDerm™ is being developed for the treatment of catastrophic, full-thickness burns covering over 50% of the total body surface area.
PermaDerm™ has been used in clinical trials on pediatric patients with catastrophic burns covering from 50 to 90+% of their body.
PermaDerm™ is being developed to require less donor skin from the patient's own body, thus needing fewer surgeries to harvest skin. The result is expected to be less scarring, with fewer infections and faster healing.
Randall McCoy
Chief Executive Officer
Randall McCoy has more than 37 years of experience in the healthcare industry. His experience includes executive positions and scientific research and development. His previous employment includes George Washington University, Temple University Medical School and RCA/SRI International (Director of Life Sciences and Electronic Displays). Mr. McCoy has assisted both small and major pharmaceutical/device companies address FDA issues. He has also helped over 225 foreign and domestic companies introduce their FDA regulated drug and medical device products into the US and World market. Mr. McCoy currently has over 30 Patents US and International.
Changed to AMBS
Dr. Joseph Rubinfeld Ph.D
Director at CytRX; Initial Co-Founder of Amgen, Inc.
Dr. Joseph Rubinfeld co-founded SuperGen, Inc. in 1991 and has served as its Chief Executive Officer and President, Chief Scientific Officer and as a director. He has been a director of CytRX since July 2002. Dr. Rubinfeld is also a founder of JJ Pharma. Dr. Rubinfeld was one of the four initial founders of Amgen, Inc. in 1980 and served as a Vice President and its Chief of Operations until 1983. From 1987 until 1990, Dr. Rubinfeld was a Senior Director at Cetus Corporation and from 1968 to 1980, Dr. Rubinfeld was employed at Bristol-Myers Company. Dr. Rubinfeld received a B.S. degree in chemistry from C.C.N.Y. and an M.A. and Ph.D in chemistry from Columbia University.
John Weber
Regenicin™'s Interim CFO
John Weber most recently served as Executive Vice President of Fujifilm USA, the highest ranked American corporate officer, from 2006 until his departure in 2009. His responsibilities included overseeing all corporate activity with the exception of R&D. During that time Fujifilm was ranked as the fastest growing medical imaging company, consistently ranking #1 or #2 in customer satisfaction. From 1998 through 2006 he served as Senior Vice President, Operations at Fujifilm USA where he spearheaded the transition of the company from a film distributor to a digital medical informatics company. From 1986 until 1998 he served as CFO where he helped to profitably manage Fuji's growth from an employee base of 75 to over 1,000. Prior to his distinguished career at Fujifilm USA, Mr. Weber served as the CFO for the confectionary and drinks division of Cadbury Schweppes Limited for three years and as Corporate Controller for an additional five years.
Dr. Craig Eagle
Pfizer Oncology
Dr. Craig Eagle joined Pfizer Australia in 2001 as part of the medical group. In Australia, his role involved leading and participating in scientific research, regulatory and pricing & re-imbursement negotiations for compounds in therapeutic areas including oncology, anti-infectives, respiratory, arthritis and pain management. In 2003, Pfizer relocated Dr. Eagle to the United States where he was appointed as the worldwide lead for development of Celecoxib in oncology to oversee the global research program. Since that time he has had increasing responsibility for overseeing the global research plans and teams for Irinotecan and Dalteparin. In 2007, he became head of Medical Affairs and Outcomes Research for Pfizer, including the US oncology business. Dr. Eagle has led, or been directly involved with, teams that resulted in eight new products or indications. As part of his current role at Pfizer, he has led the integration of the Pfizer/Wyeth oncology businesses and portfolio.
The Broadsmoore Group, LLC 9,223,770(8)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
PDA Associates LLC 7,770,000(9)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
Officers, directors and 5 percent shareholders collectively 54,508,099(10) 59.74%
* Less than 1%
(1) Unless otherwise indicated, each person or entity named in the table has sole voting power and investment power (or shares that power with that person's spouse) with respect to all shares of common stock listed as owned by that person or entity.
(2) A total of 83,807,964 shares of the Company's common stock are considered to be outstanding pursuant to Rule 13d-3(d)(1) under the Securities Exchange Act of 1934.
(3) Includes 32,821,641 shares of common stock held in his name and options to purchase 885,672 shares of common stock.
(4) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(5) Includes 1,050,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock
(6) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(7) Includes 33,971,641 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(8) Includes 5,706,270 shares of common stock held in its name, warrants to purchase 167,500 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(9) Includes 7,400,000 shares of common stock held in its name and warrants to purchase 370,000 shares of common stock,
(10) Includes 47,077,911 shares of common stock, warrants to purchase 537,500 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock
http://ih.advfn.com/p.php?pid=nmona&article=5144097
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
