Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
QuantRx Biomedical Signs Letter of Intent to Acquire Global Cancer Diagnostics
Combination Would Form an Integrated CLIA-certified Laboratory Company with Multiple Commercial Products
TUALATIN, Ore.--(BUSINESS WIRE)-- QuantRx® Biomedical Corporation ("QuantRx" or the "Company") (OTCBB: QTXB) and Global Cancer Diagnostics Inc. (“Global”), a privately held laboratory, jointly announce that they have signed a letter of intent to combine. Global, a CLIA-certified diagnostics laboratory based in Tempe, Arizona, has developed a low cost, highly accurate over-the-counter lung cancer screening test marketed under the name “The Lung Cancer Test™”. Global’s proprietary technology combines low reagent and labor costs to provide an affordable self-pay solution. “The Lung Cancer Test™” serves as a screening tool providing risk assessment, and is being sold as a laboratory developed test. Global’s management brings deep industry knowledge and expertise in the areas of medical laboratory operations and CLIA laboratory licensing, as well as the marketing of blood tests.
Dr. Shalom Hirschman, Chairman & CEO of QuantRx, stated, “The combination of our intellectual property and proprietary PadKit diagnostic products for female and fetal health with Global’s CLIA laboratory will enable the integrated company to market a multitude of non-invasive diagnostic tests in addition to The Lung Cancer Test™, beginning with endometrial and cervical cancer screens, non-invasive HPV infection screening, fetal fibronectin to monitor pregnancy, and fetal gender determination. I believe the combination of commercial operations with a certified CLIA laboratory will prove invaluable in our plan to launch a series of important patent protected molecular diagnostics tests over the next 12 – 24 months.”
William Gartner, CEO of Global Diagnostics Inc., said, “The Lung Cancer Test™ has demonstrated clinical accuracy at the level of a spiral CT scan with fewer false positives. With nearly 43 million smokers and 50 million former smokers in the United States alone, we are very excited about the opportunity to provide millions of people with a reliable, accurate and affordable solution for annual lung cancer screening. Our enthusiasm is supported and validated by several large drug store chains, many of which have expressed significant interest in the product’s potential especially in light of the importance of early detection and its profound impact on prognoses and survival rates. Moreover, the combination of QuantRx and Global provides an ideal platform to develop and launch numerous additional proprietary and non-invasive diagnostic tests to establish the new company as leading provider of unique and non-invasive diagnostic solutions dedicated to advancing patient outcomes.”
Nice to be able to see behind the curtain huh?......et z
Haven't seen anything , but wouldn't doubt it......will only reenter here on any significant dips.
Glta
QTXB
I hear this may get pumped today anyone have any thoughts?
Yesterdays alert work out great for me after holding .02's for months.
Glta new buyers.
QTXB
Potent dose of that today! $$$$
Excellent chart layout great day here.
DDAmanda Chart on QTXB:
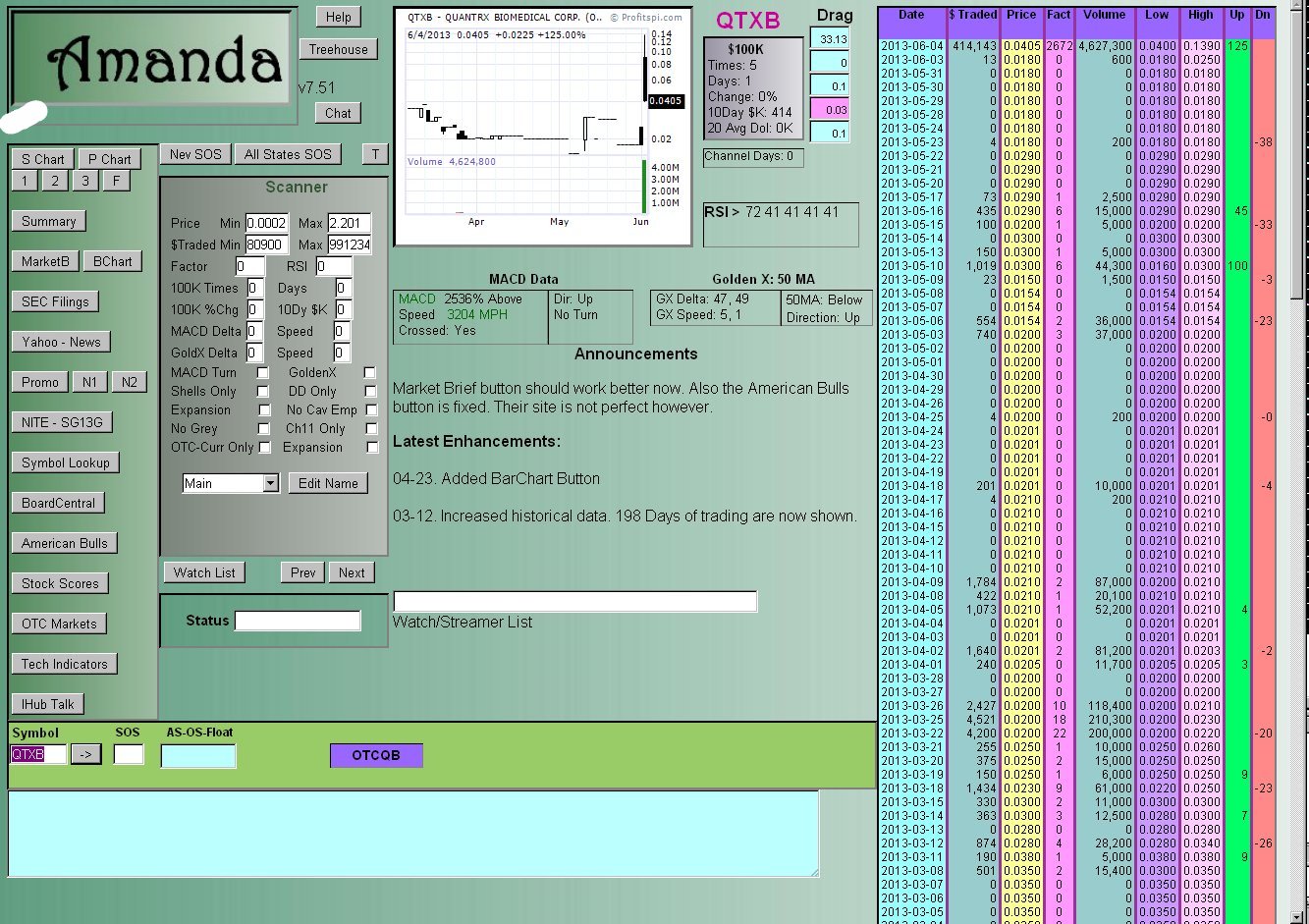
What is DDAmanda?
STT Radio Show on DDAmanda: http://ih.advfn.com/videos/educational/stt-radio-covers-dd-amanda-our-favorite-trading-tool-a-must-have-for-traders_2MmO4m5Kz08
z
bad mid day alert/pump
I almost dove in head 1st!
It didn t heal anything today !
loool
MK
-----Original Message-----
From: The Stock Psycho <PennyPsycho@PennyStockAlerts.com>
To: common_cents
Sent: Tue, Jun 4, 2013 3:20 pm
Subject: *** QTXB Set to Heal What Ails You ***
Worst pump/alert ever, whoever bought the open
500% over close , deserves it
unreal amateurs
MK
lots of shares bought above .10 would expect to see another bounce here
I own 50,000 shares if anyone is interested.
just watching for now:)
Any action here ?
QuantRx Biomedical Corporation Provides Corporate Update
Last update: 7/14/2011 12:58:00 PM
PORTLAND, Ore., July 14, 2011 /PRNewswire via COMTEX/ -- QuantRx® Biomedical Corporation (otcqb:QTXB), an emerging leader in the development and commercialization of innovative diagnostic products based on its patented technology platforms, is pleased to announce that following the successful resolution and full settlement of all matters related to the QN Joint Venture as disclosed in a Form 8-K filed with the SEC on July 8, 2011, the Company is currently implementing a go to market strategy for its market ready over-the-counter (OTC) healthcare products.
Further, the Company plans to initiate a clinical study to confirm an innovative, and potentially practice changing, approach to fetal genomic diagnostics using its patented PadKit technology. These clinical investigations will be done in collaboration with GMSBIOTECH, with initial data anticipated to be generated prior to year end.
Dr. Shalom Hirschman, Chairman and CEO, stated, "The company is developing strategic plans to bring products to market as quickly as possible. Our OTC pads for daily feminine hygiene and intergluteal pads for the temporary relief of hemorrhoids will be the focus of the initial commercialization effort."
Further, Dr, William Fleming, Chief Scientific Officer, stated, "We are committed to building shareholder value by capitalizing on our innovative patented diagnostic technologies, particularly our genomic-based diagnostic technology targeting the monitoring of recurring cancers and serious fetal genomic disease and health conditions."
About QuantRx Biomedical
QuantRx Biomedical Corporation (otcqb:QTXB) is focused on the development and commercialization of innovative over-the-counter and genomic products for advanced diagnosis of serious disease and health conditions. With synergistic expertise in the discovery of diagnostic platforms leveraging a vast portfolio of intellectual property, QuantRx's mission is to introduce products for use by its medical laboratory and consumers that deliver more accurate, reliable, and cost-effective diagnoses which result in improved patient care and a reduction in overall healthcare costs. The Company's technology portfolio, with more than a dozen patents, patents pending and licensed patents, includes: PAD technology for over-the-counter applications, and the diagnosis and treatment of women's health concerns and other medical needs, and significant investments in genome-based diagnostic chips for the laboratory and healthcare professional markets, addressing significant unmet medical needs by providing clinicians with important tools for early discovery and assessment.
This release may contain forward-looking statements within the meaning of the federal securities laws. Such forward-looking statements reflect, among other things, management's current expectations, management's current plans and strategies, and anticipated financial results, all of which are subject to known and unknown risks, uncertainties and factors that may cause our actual results to differ materially from those expressed or implied by these forward-looking statements. Many of these risks are beyond our ability to control or predict including; general economic conditions, the Company's need for additional funds, the early state of the products the Company is developing, uncertainties relating to clinical trials and regulatory reviews, competition and dependence on collaborative partners, the Company's ability to avoid infringement of the patent rights of others, and the Company's ability to obtain adequate patent protection and to enforce these rights. Because of these risks, uncertainties and assumptions, you should not place undue reliance on these forward-looking statements. Furthermore, forward-looking statements speak only as of the date they are made. QuantRx does not undertake any obligation to update or review any such forward-looking information, whether as a result of new information, future events or otherwise.
CONTACT:Dr. William FlemingChief Scientific OfficerQuantRx Biomedical Corporationwfleming@quantrx.comTel. 305-740-4465
SOURCE QuantRx Biomedical
Copyright (C) 2011 PR Newswire. All rights reserved
QuantRx Biomedical Settles Suit: Launches New PAD-Based Genomic Diagnostic Products
Last update: 7/14/2011 7:00:00 AM
PORTLAND, Ore., July 14, 2011 /PRNewswire via COMTEX/ -- QuantRx® Biomedical Corporation (QTXB), an emerging leader in the development and commercialization of innovative diagnostic products based on its patented technology platforms for the worldwide healthcare industry, today announced that it has settled the lawsuit with NURX, its prior Joint Venture partner, with prejudice, and will be focusing its operations to expand its over-the-counter products and laboratory diagnostics to better serve its growing market demands. The settlement allows NURX to become QN Diagnostics, complete the development of the technology that was the basis of the QND Joint Venture, and provides QuantRx shareholders with 12 million shares of the new entity while voiding 4 million warrants in QuantRx.
The operational changes will facilitate the transition by QuantRx from the Joint Venture-based developmental stage company to an enterprise focused on the marketing and manufacturing of innovative over-the-counter (OTC) healthcare products and expanding its genomic-based diagnostic technology targeting the monitoring of recurring cancers and serious fetal genomic disease and health conditions to the market. The company's OTC pads for daily feminine hygiene and intergluteal pads for the temporary relief of hemorrhoids are being readied for market launch. Of special significance, the company is initiating a clinical study to confirm its innovative, and potentially practice changing, approach to fetal genomic diagnostics using its patented PadKit technology.
"Now that the settlement with our former joint venture partner has been successfully concluded, we will focus our efforts on bringing our products to the market as quickly as possible," said Shalom Hirschman, MD, QuantRx Chairman and CEO. "As we now move to manufacturing and distribution of our unique products, and establish our brand names, we will need to focus our initial efforts on our market-ready products and validating our key research assumptions. We look to our OTC pads to provide our initial revenue base that will allow the company to pursue its creative diagnostic technologies."
The imminent entry into clinical investigations to develop our novel approach to fetal genomic diagnostics is especially exciting," stated Dr. William Fleming, QuantRx's President & Chief Scientific Officer. "The refocusing of our efforts will enable us to rapidly enter the market and build shareholder value back into QuantRx with revenue and exciting new technology."
About QuantRx Biomedical ()
QuantRx Biomedical Corporation (QTXB) is focused on the development and commercialization of innovative over-the-counter and genomic products for advanced diagnosis of serious disease and health conditions. With synergistic expertise in the discovery of diagnostic platforms leveraging a vast portfolio of intellectual property, QuantRx's mission is to introduce products for use by its medical laboratory and consumers that deliver more accurate, reliable, and cost-effective diagnoses which result in improved patient care and a reduction in overall healthcare costs.
The QuantRx strategy targets significant market opportunities estimated to be in excess of $5 billion worldwide. The Company's technology portfolio, with more than a dozen patents, patents pending and licensed patents, includes: PAD technology for over-the-counter applications, and the diagnosis and treatment of women's health concerns and other medical needs, and significant investments in genome-based diagnostic chips for the laboratory and healthcare professional markets, addressing significant unmet medical needs by providing clinicians with important tools for early discovery and assessment.
This release may contain forward-looking statements within the meaning of the federal securities laws. Such forward-looking statements reflect, among other things, management's current expectations, management's current plans and strategies, and anticipated financial results, all of which are subject to known and unknown risks, uncertainties and factors that may cause our actual results to differ materially from those expressed or implied by these forward-looking statements. Many of these risks are beyond our ability to control or predict including; general economic conditions, the Company's need for additional funds, the early state of the products the Company is developing, uncertainties relating to clinical trials and regulatory reviews, competition and dependence on collaborative partners, the Company's ability to avoid infringement of the patent rights of others, and the Company's ability to obtain adequate patent protection and to enforce these rights. Because of these risks, uncertainties and assumptions, you should not place undue reliance on these forward-looking statements. Furthermore, forward-looking statements speak only as of the date they are made. QuantRx does not undertake any obligation to update or review any such forward-looking information, whether as a result of new information, future events or otherwise.
CONTACTS:Dr. William FlemingPresident and CSOQuantRx Biomedical Corporationwfleming@quantrx.com
Investor Relations:Ann CarterExecutive Vice President, Marketingacarter@quantrx.com
SOURCE QuantRx Biomedical
Copyright (C) 2011 PR Newswire. All rights reserved
Yea ive been away, but starting tomorrow ill be adding to my position.
major motion over here last few days.
QTXB WEEEEEEEEEEEEEEEEEEEEEEEEE!
About to make a big move here according to the lineup on L2!
Looks like someone liked it! ![]()
Strong close, with a .06x.12 spread at the EOD.
nice! about time! lemme read through it.
does this company have a pulse??
no phone, or web site.....
anyone know if still around?
wow so sorry. totally didn't realize. was so used to people responding to my posts on VSHC. havent checked in on QTXB in a while...but seems like it could be getting active all over again.
abe, I'm talking about QTXB.....I hope you're doing well with the virtual
yep. here you go... really good one too! http://finance.yahoo.com/news/Visual-Healthcare-Opens-iw-587514663.html?x=0&.v=1
thanks so much, didn't see the PR, any chance you can send the link?
website still fully up last I saw. They just issued a PR today as well. contact info should be in there...
is this company still in business?.....phone & fax lines disconnected....web site down too
Care to share?
All I can add to this is a blurb at the bottom of an news letter I received this weekend and that to hang in there and things are taking longer than expected, yada yada yada....
Not to sound negative, because I have not sold my shares...Am hanging here to see what comes to fruition.If it does pass, then we are off and running...
Take care all.......
well it's rallied several times lately...from .07 to .11. Now seems like good buys, yea?
Yes,l that's true - can't argue that point unless we hear differently - but it sure is a disappointment...If it comes, the news must be good for it to push it back up this much....But I'll still wait - see what happens...
Take care
I mean its December 9th...which marks the 6 month date since their submission.
Lots of volume today..something is up?
still awaiting the FDA approval. The DD stands strong...you just can't rely on squirym investors. This is a great flipper in the meantime...can't wait to hear from the FDA.
|
Followers
|
21
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
129
|
|
Created
|
09/13/08
|
Type
|
Free
|
| Moderators | |||
CURRENT PROSPECTS as of 11-4-2010: On June 9th, 2010, “QuantRx Biomedical Submitted A 510(k) Application To FDA For Thyroid Point-of-Care Testing System Following Successful Completion Of Studies”. Biomedical websites across the internet put a six month estimate on 510(k) decisions.
There is usually a 6 month window on these 510k approvals for devices. Putting our expected hearing date to be December 9th, 2010.
A key point to note is that every single submission of theirs for a 510k has been approved.
About QuantRx Biomedical (www.quantrx.com)
QuantRx Biomedical Corporation (OTCBB:QTXB) is a broad-based diagnostics company focused on the development and commercialization of innovative diagnostic products based on its patented technology platforms for the worldwide healthcare industry. With synergistic expertise in the discovery of diagnostic platforms and the commercialization of products for use by healthcare professionals and consumers, QuantRx is focused on providing more accurate, reliable, and faster diagnoses that result in improved patient care.
The QuantRx strategy targets significant market opportunities estimated to be in excess of $5 billion worldwide. The Company's technology portfolio, with more than three dozen patents, patents pending and licensed patents, includes: (1) RapidSense(R) point-of-care testing products based on QuantRx core intellectual property related to lateral flow techniques for the consumer and healthcare professional markets; (2) genome-based diagnostic chips for the laboratory and healthcare professional markets; (3) molecular imaging agents for positron emission tomography (PET) and fluorescence imaging, with initial application in cardiovascular disease, addressing significant unmet medical needs by providing clinicians with important tools for early discovery and assessment; and (4) PAD technology for diagnosis and treatment of women's health concerns and other medical needs.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
