Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
A partnership announcement to develop a drug would do that.
After reading through the report, my thoughts are that Ore is in much better shape now to have PPS upward pressure now than in the past couple of years. At current prices it is worth the risk to purchase & hold for the coming year. ORXE could have one of those PR's that does spike the stock into the multiple dollar range in the coming months.
surf
loss of 11 cents, at least the trend has dramatically improved in that area...
Ore Pharmaceutical Holdings Inc. Reports Results for Third Quarter 2009
Press Release
Source: Ore Pharmaceutical Holdings Inc.
On 7:30 am EST, Friday November 13, 2009
Companies:Ore Pharmaceutical Holdings Inc.
GAITHERSBURG, Md.--(BUSINESS WIRE)--Ore Pharmaceutical Holdings Inc. (NASDAQ:ORXE - News) today reported financial results and provided an operational update for the third quarter ended September 30, 2009.
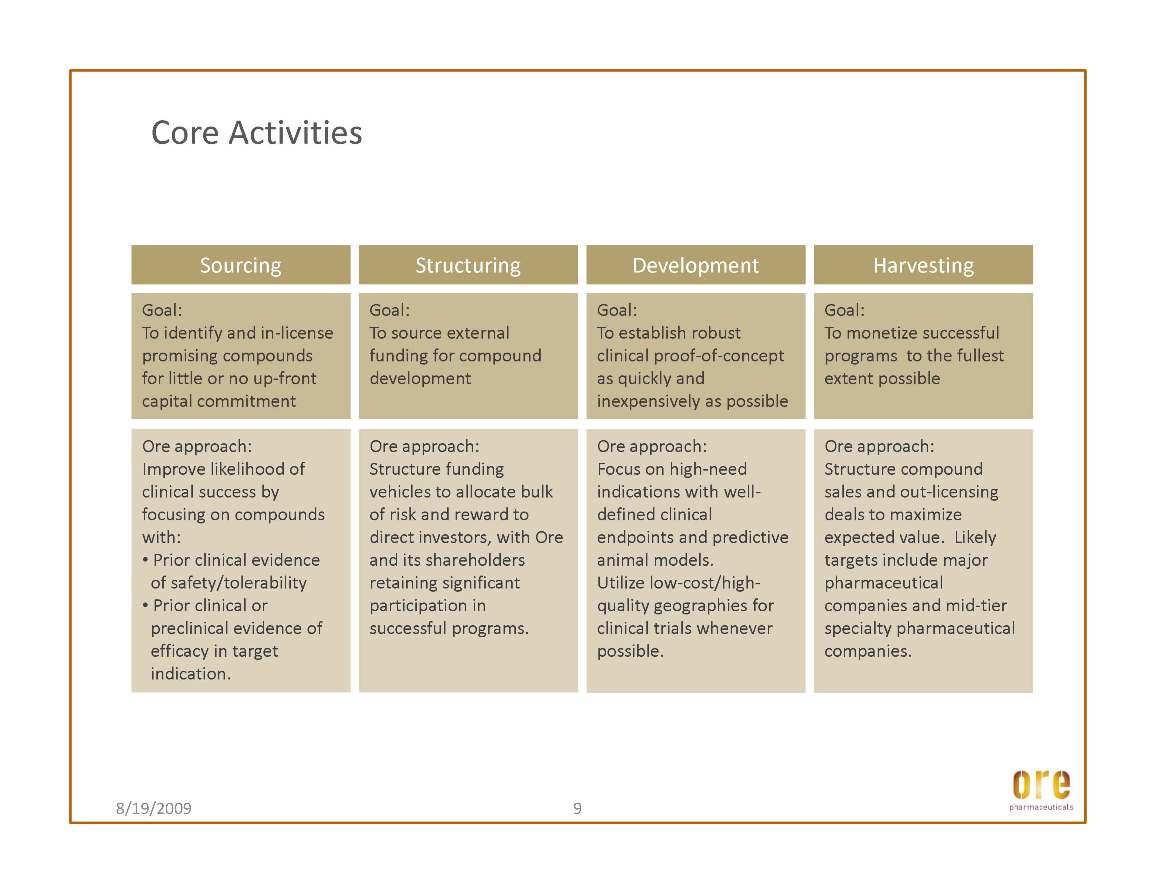
During the third quarter of 2009, Ore Pharmaceutical Holdings Inc. (“Ore Holdings” or “Ore”) completed its transition into a pharmaceutical asset management company. The Company’s strategy is focused on acquiring interests in pharmaceutical assets whose value, Ore believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through sale or out-licensing transactions. Ore anticipates that it will fund these programs through designing, raising and investing alternative financing vehicles as a regular part of its ongoing activities as a company. Progress during the third quarter and since includes:
Continued tight control of cash and expenses. Due to ongoing cost reductions, the collection of a note receivable and sale of an equity investment in the third quarter, the Company’s cash and marketable securities balance at the end of the third quarter was $7 million, which is $2.3 million higher than at the end of the second quarter. As a result of these aggressive cost controls and cash generation efforts, the Company now anticipates that it has sufficient financial resources to be able to fund operations into the first quarter of 2011;
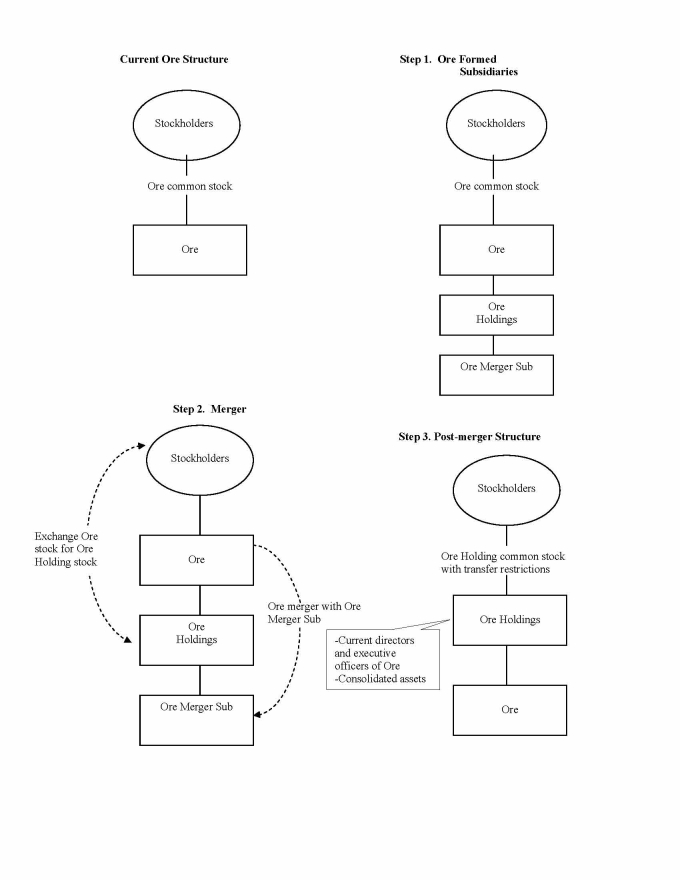
Received shareholder approval in October for, and completed, a reorganization whereby Ore Pharmaceuticals Inc. became a wholly owned subsidiary of a new company, Ore Holdings, with Ore Holdings becoming the new publicly traded, NASDAQ listed company with the symbol “ORXE”. This reorganization was undertaken primarily in order to better protect for shareholders the value of Ore’s approximately $324 million in gross net operating loss carryforwards;
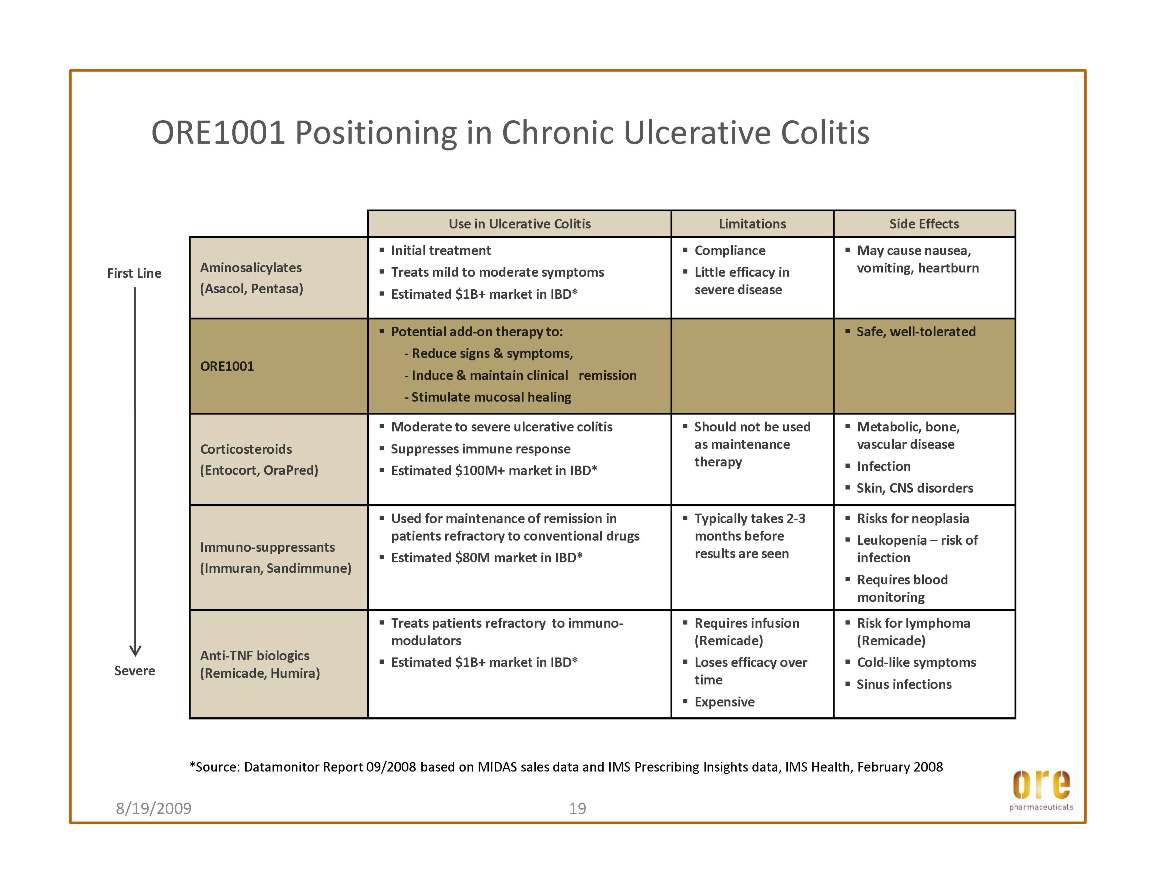
Continued on track to initiate a Phase Ib/IIa trial for ORE1001 in ulcerative colitis in the fourth quarter of 2009; and
Established a research agreement with the Armed Forces Radiobiology Research Institute and the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. to evaluate ORE1001 as a potential countermeasure agent in treating radiation injury.
Portfolio Update
During the third quarter, the Company continued to advance its lead drug candidate, ORE1001. Ore is currently developing ORE1001 under its incubation program, in which the Company may conduct initial development on certain drug assets in order to determine the best investment pathway forward. Pathways may include monetizing the asset through partnership or out-licensing, or funding further development through one of the alternative financing vehicles that are central to Ore’s business strategy.
Ore is in the process of initiating a Phase Ib/IIa clinical trial for ORE1001 in ulcerative colitis – one of the two main disorders comprising Inflammatory Bowel Disease (“IBD”). This trial is designed as a randomized, double blind, placebo controlled trial of 50 patients with ulcerative colitis and is intended to assess the safety, tolerability and therapeutic activity of ORE1001 when administered by mouth as capsules. Ore selected ulcerative colitis as the initial indication for ORE1001 on the basis of multiple preclinical studies, although Ore believes that ORE1001 may also have efficacy in Crohn’s disease, the other major category of IBD. Ore currently anticipates that this trial will be completed in the third quarter of 2010.
In addition, during the third quarter Ore entered into a research agreement with the Armed Forces Radiobiology Research Institute (AFRRI) and the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. for ORE1001. Under the terms of this agreement, AFRRI will assess the utility of ORE1001 in preventing or treating adverse effects from exposure to ionizing radiation as part of their screening project funded by a National Institute of Allergy and Infectious Diseases (NIAID)-AFRRI interagency agreement.
The AFRRI agreement provides Ore with a means to evaluate ORE1001 as a potential countermeasure agent in treating radiation injury. ORE1001 has demonstrated beneficial effects in several animal models of gastrointestinal disease, including a model of radiation injury. Severe gastrointestinal injury can occur after exposure to ionizing radiation, such as from a nuclear accident or attack. AFRRI will perform testing in their established program investigating radiation effects and will share the data with Ore.
In traditional drug development progression, a drug candidate undergoes extensive efficacy testing in clinical trials. A drug intended as a radiation countermeasure, however, must rely solely on animal data as evidence for efficacy. Therefore, as long as the safety profile of the drug can be established, the path to approval of the drug can be greatly accelerated relative to the traditional route that requires demonstration of efficacy in humans. Ore believes that ORE1001, a small molecule inhibitor of the ACE2 enzyme, has characteristics that could facilitate rapid development, including an efficient manufacturing route, oral administration via capsules, and good tolerability results in both preclinical toxicology studies and Phase 1 clinical trials.
Under the AFRRI agreement, Ore will retain all commercial rights to ORE1001. Pending results of the collaboration with AFRRI, Ore may choose to examine the utility of ORE1001 as a supportive treatment in connection with radiation therapy for certain cancers in addition to its ongoing incubation for IBD.
AFRRI, a part of the Uniformed Services University of the Health Sciences and located at the National Navy Medical Center, is a leader in studying the short and long-term effects of ionizing radiation injury. AFRRI has been actively involved in developing pharmaceutical agents that can be used to protect or mitigate injury resulting from exposure to different types of radiation.
The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. provides innovative services to meet the unique needs of military medicine.
Liquidity
As of September 30, 2009, the Company had $7.0 million in cash, cash equivalents and marketable securities available-for-sale, compared to $10.8 million as of December 31, 2008. The Company currently anticipates that its existing cash, cash equivalents and marketable securities available-for-sale, combined with anticipated payments on its remaining note receivable, will be sufficient to fund its operations into the first quarter of 2011.
Results of Operations
The Company recorded no revenue for the third quarter of 2009.
When compared to the prior year, third quarter 2009 Research and Development expenses decreased $1.8 million primarily as a result of lower employee and facility-related costs due to significant workforce reductions.
Compared to the prior year, Selling, General and Administrative expenses decreased $1.6 million primarily as a result of lower employee costs due to significant workforce reductions and reduced professional fees relating to strategic planning.
For the third quarter of 2009, the Company’s net loss was $0.6 million, or $0.11 per share, compared to a net loss of $7.4 million, or $1.36 per share, for the third quarter of 2008.
For the first nine months of 2009, the Company’s net loss was $6.3 million, or $1.15 per share, compared to a net loss of $19.1 million, or $3.33 per share, for the first nine months of 2008.
About Ore Pharmaceutical Holdings Inc.
Ore Pharmaceutical Holdings Inc. (the “Company”) is a pharmaceutical asset management company. The Company acquires interests in pharmaceutical assets whose value, it believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through sales or out-licensing transactions. The Company currently is focusing on developing and monetizing its current portfolio, which includes four clinical-stage compounds in-licensed from major pharmaceutical companies. The four compounds in the Company’s development portfolio are: ORE1001, its lead compound, ORE10002, ORE5002 (tiapamil) and ORE5007 (romazarit).
Because the reorganization whereby Ore Pharmaceuticals Inc. became a wholly owned subsidiary of Ore Pharmaceutical Holdings Inc. occurred subsequent to the quarterly reporting period ended September 30, 2009, the financial results included herein consist of the accounts of Ore Pharmaceuticals Inc.
Safe Harbor Statement
This press release contains “forward-looking statements,” as such term is used in the Securities Exchange Act of 1934, as amended. Such forward-looking statements include our ability to identify strategies for making our business successful and the impact of such strategies on our business and financial performance and on shareholder value. Forward-looking statements typically include the words “expect,” “anticipate,” “believe,” “estimate,” “intend,” “may,” “will,” and similar expressions as they relate to Ore Pharmaceutical Holdings Inc. or its management. Forward-looking statements are based on our current expectations and assumptions, which are subject to risks and uncertainties. They are not guarantees of our future performance or results. Our actual performance and results could differ materially from what we project in forward-looking statements for a variety of reasons and circumstances, including particularly risks and uncertainties that may affect the Company’s operations, financial condition and financial results and that are discussed in detail in our Annual Report on Form 10-K and our other subsequent filings with the Securities and Exchange Commission. They include, but are not limited to: whether the compounds we develop will be commercially viable; the timing of initiation of, and receipt of results from, our Phase Ib/IIa clinical trial of ORE1001 in ulcerative colitis; the pathway for development of ORE1001 as a potential countermeasure agent in treating radiation injury; whether we will be able to begin to generate sufficient new revenue from licensing or other transactions early enough to support our operations and continuing compound development; whether there will be valid claims for indemnification from the buyers of our Genomics Assets; whether there will be claims from the landlords of the leased properties we have assigned, the buyer of our Preclinical Division or the assignee of our Cambridge facility lease, that we would be required to pay as guarantors of such leases; whether we will be able to collect amounts due under the terms of the promissory note from the buyer of our molecular diagnostic business; whether we will be able to manage our existing cash adequately and whether we will have access to financing on sufficiently favorable terms to maintain our businesses and effect our strategies; whether we will be able to maintain our NASDAQ listing; whether we will be able to attract and retain qualified personnel for our business; and potential negative effects on our operations and financial results from workforce reductions and the transformation of our business. Ore Pharmaceutical Holdings Inc. undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Ore Pharmaceuticals Inc.
Consolidated Condensed Statements of Operations
(in thousands, except per share amounts)
(unaudited)
Three Months Ended Nine Months Ended
September 30, September 30,
2009 2008 2009 2008
Services revenue $ - $ 200 $ 25 $ 1,950
Expenses:
Research and development 501 2,342 2,080 8,547
Selling, general and administrative 986 2,632 5,292 10,302
Total expenses 1,487 4,974 7,372 18,849
Loss from operations (1,487 ) (4,774 ) (7,347 ) (16,899 )
Interest (income), net (335 ) (146 ) (502 ) (648 )
(Income)/loss on equity investments (558 ) 2,964 (558 ) 2,964
Gain on sale of DioGenix Inc. - (146 ) - (146 )
Net loss $ (594 ) $ (7,446 ) $ (6,287 ) $ (19,069 )
Basic and diluted net loss per share $ (0.11 ) $ (1.36 ) $ (1.15 ) $ (3.33 )
Shares used in computing basic and diluted net loss per share
5,474 5,477 5,474 5,719
Ore Pharmaceuticals Inc.
Consolidated Condensed Balance Sheets
(in thousands, except share data)
September 30, December 31,
2009 2008
ASSETS (unaudited)
Current assets:
Cash and cash equivalents $ 6,957 $ 10,784
Marketable securities available-for-sale 68 -
Prepaid expenses 229 200
Notes receivable, net 790 3,252
Other current assets 37 70
Total current assets 8,081 14,306
Property and equipment, net 36 483
Other intangibles, net 694 573
Notes receivable, net - 338
Other assets 25 -
Total assets $ 8,836 $ 15,700
LIABILITIES AND STOCKHOLDERS' EQUITY
Current liabilities:
Accounts payable $ 758 $ 623
Accrued compensation and employee benefits 144 1,185
Other accrued expenses 1,434 1,267
Current portion of long-term debt 450 477
Total current liabilities 2,786 3,552
Deferred rent 24 -
Total liabilities 2,810 3,552
Commitments and contingencies - -
Stockholders' equity:
Preferred stock, $.01 par value; 10,000,000 shares authorized; and no shares issued and outstanding as of September 30, 2009 and December 31, 2008
- -
Common stock, $.01 par value; 60,000,000 shares authorized; 5,473,519 and 5,483,519 shares issued and outstanding as of September 30, 2009 and December 31, 2008, respectively
55 55
Additional paid-in capital 385,019 384,922
Accumulated other comprehensive income 68 -
Accumulated deficit (379,116 ) (372,829 )
Total stockholders' equity 6,026 12,148
Total liabilities and stockholders' equity $ 8,836 $ 15,700
Contact:
Ore Pharmaceutical Holdings Inc.
Yet another ORXE chart, this time with with some dead Italian's SMAs
From AMTD: They still won't front shares even on a 1-for-1 re-org. That's almost criminal.
And p.s.: It's not just a name change.
Date: 10/21/09 11:16 AM
Thank you for your inquiry. The security ORE PHARMACEUTICALS INC (ORXE) is pending completion of a mandatory name to ORE PHARMACEUTICAL HOLDING INC (ORXE) effective on 10/21/2009. Although it is just a name change, they are issuing a new CUSIP, which requires a 1 for 1 exchange of the shares. The anticipated completion date for this name change has net yet been indicated by the depository. Once the new shares have been received through the depository, TD AMERITRADE will update accounts accordingly with no action required on your part. Please feel free to status for updates as needed.
Sincerely,
Shakes The Clown
Apex Reorganization and Safekeeping, TD AMERITRADE
Division of TD AMERITRADE, Inc.
Re-org approved, just like they wanted...
Ore Pharmaceuticals Announces Shareholder Approval of All Annual Meeting Proposals & Reorganization into a Holding Company
Ore Pharmaceuticals Inc. (Nasdaq: ORXE), announced today the results of the voting for four proposals put forth at its annual shareholders’ meeting and the consummation of its corporate reorganization.
In addition to two routine items, Ore shareholders approved two additional proposals that Ore management believes are critical to its business strategy.
The first proposal was for a reorganization under which the current company, Ore Pharmaceuticals Inc., would become a wholly owned subsidiary of a new company, Ore Pharmaceutical Holdings Inc. This action was proposed in order to better protect for shareholders the long term value of Ore’s approximately $324 million in net operating loss tax carry forwards (“NOL’s”) by establishing transfer limitations designed to prevent an inadvertent change of control that could result in the substantial loss of those NOLs. The proposal was approved and the reorganization has been completed.
The second proposal was for a new Omnibus Equity Incentive Plan that will give Ore the flexibility to attract and retain the high quality personnel required to implement its strategy of becoming a pharmaceutical asset management company.
The proposals approved by shareholders were:
The reorganization of Ore Pharmaceuticals. The reorganization having been completed,, the current Ore is now a subsidiary of a new company, Ore Pharmaceutical Holdings Inc. (“Ore Holdings”) effective immediately. Ore Holdings will begin to trade on the Nasdaq Capital Market as of October 21, 2009 under the symbol “ORXE”, the same as previously used for Ore. Shareholders’ ownership will automatically be converted to ownership in Ore Holdings without requiring any further action by shareholders.
The re-election of G. Anthony Gorry, Ph.D. to the Ore Holdings Board of Directors.
The adoption of the 2009 Omnibus Equity Incentive Plan; and
The ratification of the selection of Ernst & Young LLP as Ore Holdings’ auditors for 2009.
Mark J. Gabrielson, President & CEO of Ore Holdings said, “We are grateful for the support from our shareholders for the important matters we proposed at this annual meeting. We continue to move ahead aggressively with the execution of our business strategy in order to create value for our shareholders. Management believes that our new corporate structure and our new name better reflect and support our business model. These shareholder approvals will help us achieve our goal of transforming Ore into a pharmaceutical asset management company. I look forward to providing you with further updates as we make progress.”
Ore Pharmaceutical Holdings Overview
Ore Pharmaceutical Holdings Inc. (the “Company”) is a pharmaceutical asset management company. The Company acquires interests in pharmaceutical assets whose value, it believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through a sale or out-licensing. Initially, the Company will focus on developing and monetizing its current portfolio, which includes four clinical-stage compounds in-licensed from major pharmaceutical companies. The Company’s four compounds in its development portfolio are: ORE1001, its lead compound, ORE10002, ORE5002 (tiapamil) and ORE5007 (romazarit).....
Movin' on up...
I noticed level II on ORXE was moving up for the meeting next week. We may get another one of those spikes to .80+ in the next few days......
surf
Most volume seems to be at the ask again lately
The next couple of weeks could be a make or break in the near term for ORXE stock with all the possible news events coming out. The chart is holding at a point where it is going to test the $1 area or fall apart, we will see soon enough.
surf
The Company also announced that it currently plans to hold its 2009 Annual Meeting of Stockholders on Tuesday, October 20, 2009 at 11:00 a.m. EDT. As provided in the registration statement filed August 14th with the SEC, the Company plans to submit for shareholder approval, among other matters, a proposal to change Ore’s structure to a holding company, in order to better protect Ore’s approximately $324 million in tax loss carryforwards.
ORXE net operating loss preservation plan... this was in the news today and I wonder if this is a big trend?
I am not very well versed in these types of accounting maneuvers. But the headline caught my attention.
Ford Adopts Tax Benefit Preservation Plan
Last update: 9/11/2009 10:39:00 AM
DEARBORN, Mich., Sept 11, 2009 /PRNewswire-FirstCall via COMTEX/ -- Ford Motor Company (F) today announced that its Board of Directors has adopted a tax benefit preservation plan designed to preserve substantial tax assets.
Through year-end 2008, Ford had tax attributes, including net operating losses, capital losses and tax credit carryforwards, that would offset approximately $19 billion of taxable income. Ford can utilize the tax attributes in certain circumstances to offset taxable income and reduce its federal income tax liability.
Ford's ability to use the tax attributes would be substantially limited if there were an "ownership change" as defined under Section 382 of the Internal Revenue Code and Internal Revenue Service rules.
In general, an ownership change would occur if Ford's "5-percent shareholders," as defined under Section 382, collectively increase their ownership in Ford by more than 50 percentage points over a rolling three-year period.
Five-percent shareholders do not include certain institutional holders, such as mutual fund companies, that hold Ford stock on behalf of several individual mutual funds where no single fund owns 5 percent or more of Ford stock.
The plan is similar to tax benefit preservation plans adopted by many other public companies with significant tax attributes.
As part of the plan, the Ford Board of Directors declared a dividend of one preferred share purchase right for each outstanding share of its common stock and Class B stock. The preferred share purchase rights will be distributed to stockholders of record as of Sept. 25, 2009, but would only be activated if triggered by the plan.
Effective today, if any person or group acquires 4.99 percent or more of the outstanding shares of common stock (subject to certain exceptions), there would be a triggering event under the plan resulting in significant dilution in the ownership interest of such person or group in Ford stock.
"This plan is designed to protect shareholder value and safeguard valuable tax attributes by reducing the likelihood of an unintended 'ownership change' through actions involving Ford common stock," said Lewis Booth, Ford's chief financial officer.
Ford's Board of Directors has the discretion to exempt any acquisition of common stock from the provisions of the tax benefit preservation plan. The plan may be terminated by the Board at any time prior to the preferred share purchase rights being triggered.
The preferred share purchase rights will expire upon:
-- The close of business on Sept. 11, 2012 (unless that date is advanced or extended by the Board);
-- The time at which these rights are redeemed or exchanged under the plan;
-- The final adjournment of Ford's 2010 annual meeting of shareholders if shareholder approval of the plan has not been received prior to that time;
-- The repeal of Section 382 or any successor statute, if Ford's Board of Directors determines that the plan is no longer necessary for the preservation of tax attributes; or
-- The beginning of a taxable year of the company to which the Board determines that no tax attributes may be carried forward.
The issuance of the preferred share purchase rights will not affect Ford's reported earnings per share and is not taxable to Ford or its stockholders. Additional information regarding the tax benefit preservation plan will be contained in a Form 8-K and in a Registration Statement on Form 8-A that Ford is filing with the Securities and Exchange Commission.
In addition, Ford stockholders of record as of Sept. 25, 2009 will be mailed a detailed summary of the plan.
About Ford Motor Company Ford Motor Company, a global automotive industry leader based in Dearborn, Mich., manufactures or distributes automobiles across six continents. With about 201,000 employees and about 90 plants worldwide, the company's brands include Ford, Lincoln, Mercury and Volvo. The company provides financial services through Ford Motor Credit Company. For more information regarding Ford's products, please visit . SOURCE Ford Motor Company Copyright (C) 2009 PR Newswire. All rights reserved
ORXE is one of the few biotech stocks that I follow that hasn't had a big move this year. May be time for this dog to hunt............
surf
looks like some folks like ths stock today
Ore Pharmaceuticals Announces Upcoming Publication of Research Study on ORE1001
Press Release
Source: Ore Pharmaceuticals Inc.
On Wednesday September 9, 2009, 8:00 am EDT
Companies:Ore Pharmaceuticals Inc.
GAITHERSBURG, Md.--(BUSINESS WIRE)--Ore Pharmaceuticals Inc. (Nasdaq:ORXE - News), announced today the publication of an article in the online version of the journal Inflammation Research titled, “Effects of the ACE2 inhibitor GL1001 on acute dextran sodium sulfate-induced colitis in mice.”
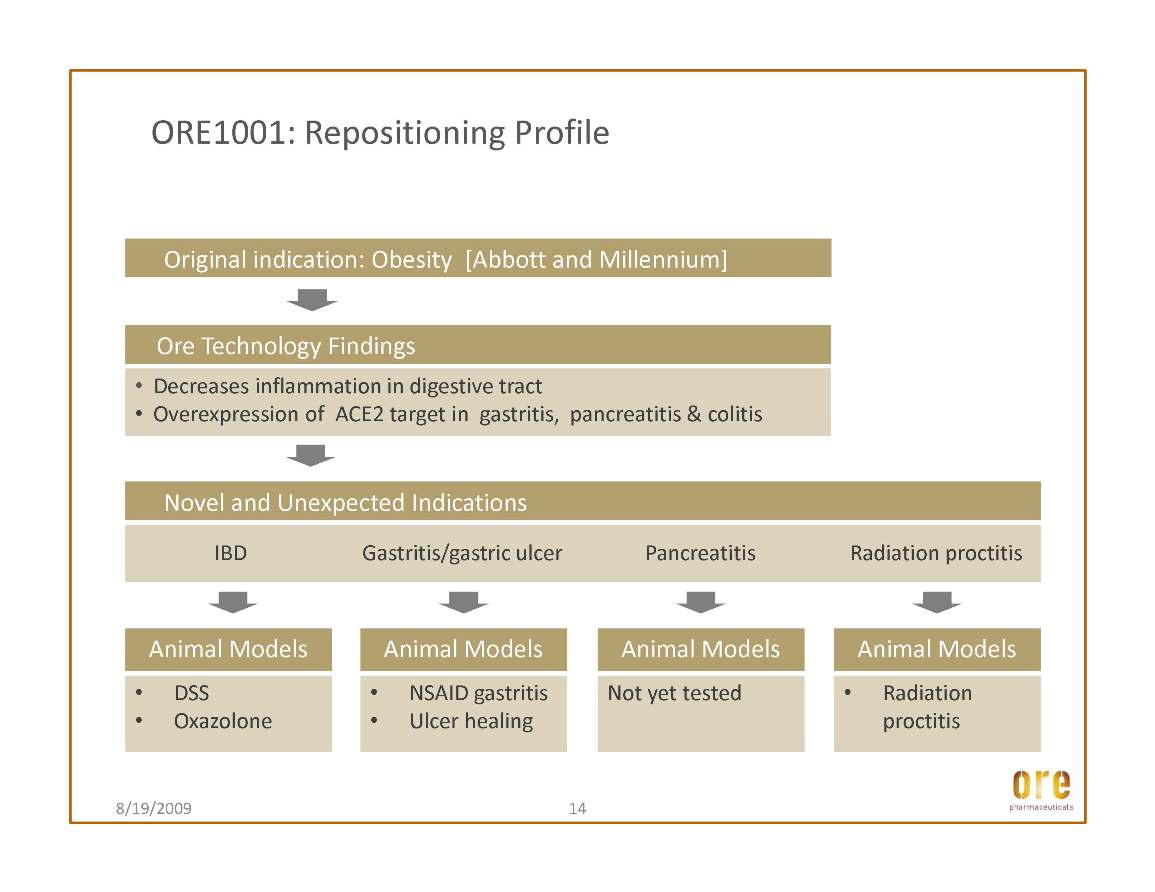
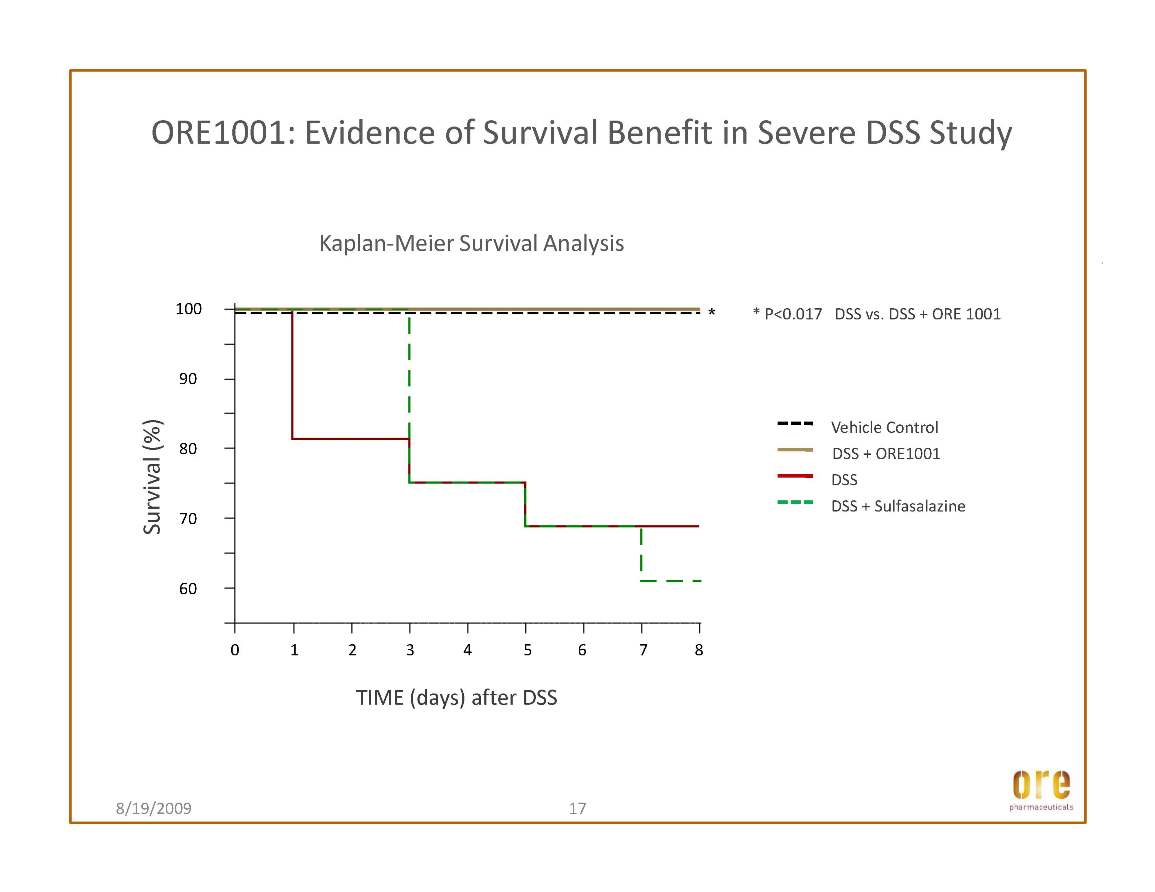
This article discussed the efficacy of Ore’s lead drug candidate, ORE1001 (formerly GL1001), in the dextran sodium sulfate animal screening model for inflammatory bowel disease drugs. The results show that treatment with ORE1001 displayed efficacy on par with that of the oral standard, sulphasalazine. ORE1001 improved common measures of the extent of damage, such as histopathology, in a dose-related and statistically significant manner. Moreover, ORE1001 markedly decreased tissue myeloperoxidase activity, a well-known marker of inflammation. The findings, when considered along with other studies of ORE1001, support further development of the compound in gastrointestinal inflammatory conditions. ORE1001 has progressed through multiple dose clinical phase I testing in the U.S. and is on track to commence a Phase Ib/IIa trial in ulcerative colitis, one of the two main disorders that comprise inflammatory bowel disease (IBD), in the second half of 2009.
It is estimated that up to one million Americans are affected by IBD. With typical onset in childhood or early adulthood, these disorders cause many decades of pain and suffering and result in significant lost productivity, in addition to the direct costs of medical and surgical care. The burden on the U.S. healthcare system alone is significant; IBD is associated with health care costs estimated at more than $1.7 billion. Ore believes that ORE1001, if approved, could represent a significant enhancement to current therapies for treating this debilitating disease.
The print article is expected to be published in an upcoming issue of Inflammation Research. The full text article is currently available online at: http://www.springer.com/birkhauser/biosciences/journal/11.
Ore Pharmaceuticals Overview
Ore Pharmaceuticals Inc. (the “Company”) is a pharmaceutical asset management company. The Company acquires interests in pharmaceutical assets whose value, it believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through a sale or out-licensing. Initially, the Company will focus on developing and monetizing its current portfolio, which includes four clinical-stage compounds in-licensed from major pharmaceutical companies. The Company’s four compounds in its development portfolio are: ORE1001, its lead compound, ORE10002, ORE5002 (tiapamil) and ORE5007 (romazarit).
Rodman & Renshaw Conference
Hot Biotech Events for Next Week: BioBuzz
09/04/09 - 07:50 AM EDT See All 2 Comments
Adam Feuerstein
BOSTON, Mass. (TheStreet) -- Labor Day is finally upon us. Summer is over. Here are some biotech events to watch for next week:
Investor conference season kicks off in earnest, with three smaller banks holding healthcare conferences simultaneously. Rodman & Renshaw and Robert W. Baird will be gathering in investors in New York, while Thomas Weisel Partners occupies Boston. All three confabs begin on Sept. 9.
Small-cap biotech and drug stocks will be the main focus of all three events, so look for presentations from the likes of Geron, Cell Therapeutics, Genta and Antigenics among many others.
The Rodman conference wins for most star power, with a guest talk by former Fed chief Alan Greenspan and an opening night gala dinner on Ellis Island featuring a performance by Diana Ross. Now we know where Rodman is spending the commission dollars it's made recently from all those biotech fund-raising deals.
On the clinical data front next week, I'd expect Osiris Therapeutics to release the data from its two pivotal studies of Prochymal in patients with graft-versus-host disease.
Vivus should be close to announcing phase III data on its obesity drug soon (perhaps not next week); same goes for Chelsea Therapeutics and its phase III drug droxidopa for orthostatic hypotension.
Next week's medical conference calendar includes the start on Sept. 11 of the American Society for Bone and Mineral Research (ASBMR) annual meeting.
http://www.thestreet.com/_yahoo/story/10594671/1/hot-biotech-events-for-next-week-biobuzz.html
The overview that you posted should be the presentation that the CEO is going to cover. If the stock doesn't start a move after the meeting, we still have a dog here. My guess is that it will start to breakout in the coming weeks, it is currently setting at a support line that it has been above since the reversal in April/May. I would hope that the stock breaks .75 before the end of this month, then starts the move to $1.25, the high made on the last big volume surge from Nov 2008.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=40708133
http://www.orepharma.com/drug-development-partnering-opportunities
surf
maybe something good will come from that
Ore Pharmaceuticals Announces Upcoming Conference Presentation
Press Release
Source: Ore Pharmaceuticals Inc.
On Tuesday September 1, 2009, 4:29 pm EDT
Companies:Ore Pharmaceuticals Inc.
GAITHERSBURG, Md.--(BUSINESS WIRE)--Ore Pharmaceuticals Inc. (Nasdaq:ORXE - News), announced today that Mark J. Gabrielson, President and CEO, will present at the Rodman & Renshaw 11th Annual Healthcare Conference, to be held on September 9-11, 2009 at the New York Palace Hotel in New York. Mr. Gabrielson will provide an overview of the company and a strategic and operational update on September 11, 2009 at 3:40 p.m. Eastern Time.
Ore Pharmaceuticals Overview
Ore Pharmaceuticals Inc. (the “Company”) is a pharmaceutical asset management company. The Company acquires interests in pharmaceutical assets whose value, it believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through a sale or out-licensing. Initially, the Company will focus on developing and monetizing its current portfolio, which includes four clinical-stage compounds in-licensed from major pharmaceutical companies. The Company’s four compounds in its development portfolio are: ORE1001, its lead compound, ORE10002, ORE5002 (tiapamil) and ORE5007 (romazarit).
active early today.
54 cents. usual market making thievery here. lol
print 100 here, 200 there
Time & Sales
Price Size Exch Time
0.54 100 NYE 15:48:24
0.5799 1739 NDD 14:34:27
0.58 100 NGS 14:30:51
0.58 998 NGS 14:27:14
0.58 108 NDD 14:27:11
0.58 100 NDD 14:11:03
0.58 100 NDD 14:11:03
0.5801 1731 NDD 13:45:11
0.54 200 NDD 13:31:19
0.6111 300 NDD 10:49:46
0.62 500 NDD 10:12:14
If the CEO wants to make the big bucks, he'll get this little boat moving in the right direction. My guess would be that the updated overview you posted will be the main part of a roadshow in the coming months to get the word out on what ORE is now involved in.
http://secfilings.nasdaq.com/filingFrameset.asp?FileName=0001209191%2D09%2D021562%2Etxt&FilePath=%5C2009%5C04%5C15%5C&CoName=GENE+LOGIC+INC&FormType=4&RcvdDate=4%2F15%2F2009&pdf=
http://www.sec.gov/Archives/edgar/data/1043914/000115752309006091/a6033249ex99_1.htm
I'm really curious about the option grants and their trigger prices from earlier this year...
#msg-37289622
First insider purchase in nearly two years, the CEO bought 2k shares. What is he thinking? Not worth the paper work..........
http://www.form4oracle.com/company/ore-pharmaceuticals-inc-orxe?id=967
Sounds interesting.
After reading the overview, I am closer now to believing that the company may finally have a plan to get the company/stock going in the right direction. I understand the concept of what they want to do & within the coming months we may get a real run in the stock. Many biotech/Pharma companies would rather outsource some of their product trials processes/work to someone that can provide that expertise & helps those firms better manage their need to size up or down their own staffs. LGND is another biotech firm that went in this direction.
Ligand Pharmaceuticals
Ligand Pharmaceuticals is a royalty-driven biotechnology company with strong research and drug discovery capabilities. We have numerous valuable corporate partnerships, a broad product pipeline targeting large markets, proven scientific credentials in combination with partners and a strong balance sheet.
I also added the overview to the sticky's post section.
surf
Introduction
As of December 31, 2008, we had accumulated net operating losses totaling approximately $324 million (includes an estimate of our 2008 tax loss of approximately $19 million) that we can carry forward as potential tax deductions until they expire between 2009 and 2028. We have also incurred capital losses of less than $1 million that we can carry forward as potential tax deductions (only against capital gains) until they expire between 2011 and 2012. In this proxy statement/ prospectus, we refer to these net operating losses and capital losses together as NOLs.
NOLs can benefit us by offsetting federal taxable income dollar-for-dollar by the amount of the NOLs, thereby reducing or eliminating our U.S. federal corporate tax (other than alternative minimum tax) on such income. Assuming we can fully use our NOLs to reduce our U.S. federal corporate tax, we expect to be subject to the alternative minimum tax, which would result in 10% of our alternative minimum taxable income being subject to the 20% alternative minimum tax. The maximum federal corporate tax rate is currently 35%.
The benefit of the NOLs to us can be reduced or eliminated under section 382 of the Internal Revenue Code of 1986, as amended (the "Code"), if we experience an “ownership change,” as defined in section 382 of the Code and described in more detail below. An ownership change can occur through one or more acquisitions of our stock, whether occurring contemporaneously or pursuant to a single plan, by which stockholders or groups of stockholders, each of whom owns or is deemed to own directly or indirectly at least 5% of our stock, increase their ownership of our stock by more than 50 percentage points within a three-year period. We currently do not have the ability to restrict transactions that could result in an ownership change.
Our Board of Directors believes the best interests of Ore and its stockholders will be served by adopting provisions that are designed to restrict direct and indirect transfers of our stock if such transfers will affect the percentage of stock that is treated as owned by a 5% stockholder. In this proxy statement/ prospectus, we refer to these provisions as the “transfer restrictions.”
As of December 31, 2008, we do not believe that we have experienced an ownership change, but calculating whether an ownership change has occurred is subject to inherent uncertainty. This uncertainty results from the complexity and ambiguity of the section 382 provisions, as well as limitations on the knowledge that any publicly traded company can have about the ownership of and transactions in its securities.
The transfer restrictions will be included as Article VIII in the certificate of incorporation of Ore Holdings.
Limitations on use of NOLs
The benefit of the NOLs to us can be reduced or eliminated under section 382 of the Code if we experience an “ownership change,” as defined in section 382. Generally, an ownership change can occur through one or more acquisitions, whether occurring contemporaneously or pursuant to a single plan, by which one or more stockholders, each of whom owns or is deemed to own directly or indirectly 5% or more in value of a corporation’s stock, increase their aggregate percentage ownership by more than 50 percentage points over the lowest percentage of stock owned by such stockholders (with the lowest percentage measured separately for each stockholder) at any time during the preceding three-year period. The amount of the increase in the percentage of stock ownership of each 5% stockholder is computed separately, and each such increase is then added together with any other such increase to determine whether an ownership change has occurred. For this purpose, all holders who own less than 5% of a corporation’s stock are generally treated together as one 5% stockholder (although in some circumstances these smaller holders may be counted as two or more separate stockholders, with each being a “public group” and a separate 5% stockholder, for purposes of section 382 of the Code). Transactions in the public markets among stockholders owning less than 5% of the equity securities generally do not affect the calculation of an ownership change (but can if a corporation has more than one public group). In addition, certain constructive ownership rules, which generally attribute ownership of stock owned by estates, trusts, corporations, partnerships or other entities to the ultimate indirect individual owner thereof, or to related individuals, are applied in determining the level of stock ownership of a particular stockholder. Special rules, described below, can result in the treatment of options (including warrants) or other similar interests as having been exercised if such treatment would result in an ownership change. All percentage determinations are based on the fair market value of a corporation’s stock.
For example, if a single investor acquired 50.1% of our stock in a three-year period, a change of ownership would occur. Similarly, if ten persons, none of whom owned our stock, each acquired slightly over 5% of our stock within a three-year period (so that such persons owned, in the aggregate more than 50%), an ownership change would occur.
If we were to experience an ownership change, then the amount of taxable income in any year (or portion of a year) subsequent to the ownership change that could be offset by NOLs from periods prior to such ownership change could not exceed the product obtained by multiplying (i) the aggregate value of our stock immediately prior to the ownership change (with certain adjustments) by (ii) the then applicable federal long-term tax exempt rate (this resulting product is referred to as the section 382 limitation). If we experience an ownership change for tax purposes, the section 382 limitation would be reduced to zero in the event we were deemed to fail to continue the business enterprise that we engaged in before the ownership change for the two-year period following the ownership change. Any portion of the annual section 382 limitation amount not utilized in any year may be carried forward and increase the available section 382 limitation amount for the succeeding tax year. Thus, an ownership change could significantly reduce or eliminate the annual utilization of our NOLs and cause a substantial portion or all of such NOLs to expire prior to their use.
Trying to perserve NOLs by using a holding company setup?
http://www.sec.gov/Archives/edgar/data/1043914/000115752309006007/a6028740.htm
Is this due to some tax law change?
New roadshow presentation filing
http://www.sec.gov/Archives/edgar/data/1043914/000115752309006091/a6033249ex99_1.htm



















Second Quarter 2009 Financial Results Reported
Press Release
Source: Ore Pharmaceuticals Inc.
On Monday August 17, 2009, 7:30 am EDT
Companies:Ore Pharmaceuticals Inc.
Topics:Health Care SectorIPOs & Offerings
GAITHERSBURG, Md.--(BUSINESS WIRE)--Ore Pharmaceuticals Inc. (NASDAQ:ORXE - News) today reported financial results for the second quarter ended June 30, 2009.
Highlights
During the second quarter of 2009, Ore Pharmaceuticals continued its transformation into a pharmaceutical asset management company. The Company’s strategy is focused on acquiring interests in pharmaceutical assets whose value, Ore believes, it can significantly enhance through targeted development, with the goal of then monetizing these assets through a sale or out-licensing. Steps taken during the second quarter and since in advancing towards that goal include:
Further reduced cash usage to $2.4 million in the second quarter. The Company expects cash usage to continue to decline on a quarterly basis during the remainder of 2009 and currently anticipates that it has sufficient financial resources to be able to fund operations through late 2010;
Hired Benjamin L. Palleiko as CFO, bringing onboard valuable industry experience, strategic financial insight and capital markets expertise;
Appointed James W. Fordyce to the Board of Directors, providing Ore with the benefits of his lengthy experience as an investor focused on the healthcare sector;
Acquired rights to ORE10002 from H. Lundbeck A/S, further increasing the asset portfolio at very little cost;
Continued on track to initiate a Phase Ib/IIa trial for ORE1001 in ulcerative colitis in the second half of 2009; and
Filed on Friday, August 14th with the SEC a registration statement announcing our 2009 Annual Meeting of Shareholders and pursuant to which the Company intends to propose, among other matters, a change of Ore’s corporate structure in order to better protect Ore’s approximately $324 million in tax loss carryforwards for shareholders.
Mark J. Gabrielson, Ore’s President and CEO stated, “The second quarter was a period of tremendous activity at Ore. We largely completed our cost cutting efforts, placing the Company on firmer financial ground, continued to advance our asset portfolio, and made some important enhancements to our management and board. I believe that all of these moves place us in a much stronger position as we continue to implement our strategy of transitioning Ore into a pharmaceutical asset management company.”
Liquidity
As of June 30, 2009, the Company had $4.6 million in cash and cash equivalents, compared to $10.8 million as of December 31, 2008. The cash usage for the second quarter of 2009 was $2.4 million. The operating cash usage for the third quarter of 2009 is currently expected to be lower than that of the second quarter. The Company also expects its operating cash usage for the fourth quarter of 2009 to be significantly lower than in the third quarter. The Company currently anticipates that its existing cash and cash equivalents balance, combined with anticipated payments on notes receivable, will be sufficient to fund its operations through late 2010.
Results of Operations
The Company recorded less than $0.1 million of revenue for the second quarter of 2009.
When compared to the prior year, second quarter 2009 Research and Development expenses decreased $2.7 million primarily as a result of lower employee and facility-related costs due to significant workforce reductions and lower third-party costs associated with the further development of Ore’s lead compound, ORE1001.
Compared to the prior year, Selling, General and Administrative expenses decreased $0.5 million primarily as a result of lower employee costs due to significant workforce reductions, partially offset by facility-related lease abandonment costs associated with the Company substantially vacating its Gaithersburg, Maryland facility.
For the second quarter of 2009, the Company’s net loss was $2.5 million, or $0.47 per share, compared to a net loss of $4.7 million, or $0.86 per share, for the second quarter of 2008.
For the first six months of 2009, the Company’s net loss was $5.7 million, or $1.04 per share, compared to a net loss of $11.6 million, or $1.99 per share, for the first six months of 2008.
2009 Annual Meeting of Stockholders and Proposal to Further Protect Tax Loss Carryforwards
The Company also announced that it currently plans to hold its 2009 Annual Meeting of Stockholders on Tuesday, October 20, 2009 at 11:00 a.m. EDT. As provided in the registration statement filed August 14th with the SEC, the Company plans to submit for shareholder approval, among other matters, a proposal to change Ore’s structure to a holding company, in order to better protect Ore’s approximately $324 million in tax loss carryforwards.
The record date for determining the stockholders entitled to receive notice of, and to vote at, the 2009 Annual Meeting is expected to be the close of business on September 4, 2009. In order to be considered timely and to be included in our proxy materials for the 2009 Annual Meeting, a stockholder proposal must be received by our Corporate Secretary no later than August 20, 2009.
Stockholders also have the right to make stockholder proposals at the meeting by following specified procedures. For a stockholder proposal for the 2009 Annual Meeting that is not intended to be included in our proxy statement, the stockholder must (1) provide the information required by our Bylaws and (2) give timely notice to our Corporate Secretary at 610 Professional Drive, Suite 101, Gaithersburg, Maryland 20879, Attention: Benjamin L. Palleiko. To be timely, the notice must have been received by our Corporate Secretary not earlier than the close of business on June 22, 2009, and not later than August 24, 2009.
Form 10-Q for ORE PHARMACEUTICALS INC.
14-Aug-2009
Quarterly Report
Note 2 - Liquidity and management's plans
Since inception, the Company has incurred, and continues to incur, significant losses from operations. At June 30, 2009, the Company had $4,650 in cash and cash equivalents. The Company has realigned its corporate resources and as a result significantly reduced its workforce from 71 employees on December 31, 2007 to 7 employees as of June 30, 2009. In addition, the Company assigned its Cambridge, Massachusetts lease and leased new space at a lower cost. The Company believes that its existing cash and cash equivalents, continuing cash savings resulting from its ongoing realignment and cash conservation efforts and proceeds from the collection of its outstanding notes receivable, will be sufficient to allow the Company to operate through late 2010, including the costs of initiating and completing the Phase Ib/IIa clinical trial for ORE1001, which is expected to be completed in the third quarter of 2010. However, there can be no assurance that the Company will be successful in its continuing realignment and cash conservation efforts, the collection of its outstanding notes receivable or, if necessary, attracting additional financing to allow the Company to complete the clinical trial. Furthermore, there is no assurance if the Company completes its Phase Ib/IIa clinical trial, that the results will be satisfactory or will enable the Company to successfully outlicense its compound. If the Company is not successful in achieving its objectives, it might be necessary to liquidate the Company in late 2010. The balance sheet at June 30, 2009 does not include any adjustments relating to recoverability and classification of recorded asset amounts or the amounts and classifications of liabilities that might be necessary in the event of such liquidation.
Note 3 - Stock-based compensation
At June 30, 2009, the Company has the following stock-based compensation plans:
the 1997 Equity Incentive Plan (the "Stock Plan") and the 1997 Non-Employee Directors' Stock Option Plan (the "Directors' Plan").
The Company recorded stock-based compensation expense of $28 and $70 for the three months ended June 30, 2009 and 2008, respectively, and $56 and $229 for the six months ended June 30, 2009 and 2008, respectively.
Stock Option Awards
The Company determined the fair value of each option grant on the date of grant
using the Black-Scholes option pricing model for the indicated periods, with the
following assumptions:
Three Months Ended Six Months Ended
June 30, June 30,
2009 2008 2009 2008
Weighted average fair value of grants $0.34 $0.96 $0.28 $0.97
Expected volatility 79% 65% 79% 63%
Risk-free interest rate 1.38% 2.53 % to 2.57% 1.31 % to 1.38% 2.53 % to 3.04%
Expected lives 5 years 3 years 5 years 3 years
Dividend rate 0% 0% 0% 0%
The following is a summary of option activity for the six months ended June 30, 2009:
Per Share
Weighted- Aggregate
Number of Average Intrinsic
Shares Exercise Price Value
Outstanding at January 1, 2009 683,847 $ 24.10
Options granted 786,300 $ 0.44
Options exercised - $ -
Options cancelled (353,020 ) $ 19.40
Outstanding at June 30, 2009 1,117,127 $ 8.93 $ 103
Exercisable at June 30, 2009 348,898 $ 27.17 $ 8
8.
--------------------------------------------------------------------------------
The aggregate intrinsic value in the table above represents the total intrinsic value (the excess of the Company's closing stock price on the last trading day of June 2009 over the exercise price, multiplied by the number of in-the-money options) that would have been received by the option holders had all option holders exercised their options on June 30, 2009. This amount is subject to change based on changes to the fair market value of the Company's Common Stock.
Restricted Stock Awards
The Committee had previously approved grants for shares of restricted stock
under the Stock Plan subject to certain performance- or time-based vesting
conditions which, if not met, would result in forfeiture of the shares and the
reversal of any previously recognized related stock-based compensation expense.
The following is a summary of restricted stock awards activity for the six
months ended June 30, 2009:
Per Share
Weighted-
Average
Number of Grant-Date
Shares Fair Value
Outstanding at January 1, 2009 10,000 $ 6.15
Restricted stock granted - $ -
Restricted stock vested - $ -
Restricted stock forfeited - $ -
Outstanding at June 30, 2009 10,000 $ 6.15
Performance-based non-vested restricted stock awards are recognized as compensation expense over the expected vesting period based on the fair value at the date of grant and the number of shares ultimately expected to vest. The Company's outstanding restricted stock award at June 30, 2009 was forfeited in the third quarter of 2009. In 2008, the Company had reversed the previously recognized related stock-based compensation expense for this restricted stock award since the Company believed that the achievement of the performance milestones were not probable.
As of June 30, 2009, $220 of total unrecognized compensation cost related to stock options is expected to be recognized over a weighted-average period of 1.9 years. This estimate does not include the impact of other possible stock-based awards that may be made during future periods.
Note 4 - Loan and grant agreements with the State of Maryland
During the second quarter of 2009, the Company received a notice requiring repayment of all amounts potentially due under a loan and a grant agreement with the State of Maryland that total $710 at June 30, 2009. The Company has recorded the amounts due under the loan and grant agreement within current portion of long-term debt and other accrued expenses. The Company is in discussions with the State of Maryland concerning the terms of potential repayment of this amount.
Note 5 - Ocimum Biosolutions Inc. promissory note and related agreements
In connection with the sale of the Company's Genomics business pursuant to a certain Asset Purchase Agreement with Ocimum Biosolutions Limited, as parent, and Ocimum Biosolutions Inc., a Delaware corporation ("Ocimum"), as Purchaser, which was completed on December 14, 2007, and previously reported, the Company had received as partial payment for the sales price, a $3,000 secured promissory note from Ocimum and Ocimum Biosolutions Limited, guaranteed by Coramandel Prestcrete Private Limited, a company incorporated in the Republic of India. The promissory note, secured by a security agreement between Ocimum and the Company, was due and payable on June 15, 2009.
In June 2009, the Company entered into a superseding $3,000 secured promissory note ("Note") with Ocimum and its affiliate, Ocimum Biosolutions India Limited, a company incorporated in the Republic of India, which is secured pursuant to a superseding security agreement ("Security Agreement") with Ocimum and repayment of which is guaranteed by a guaranty agreement with Coramandel Infrastructure Private Limited, a company incorporated in the Republic of India, in favor of the Company. The superseding agreements are effective as of June 15, 2009, and the original promissory note, security agreement and related guarantee agreement have been cancelled and terminated.
In connection with the sale of the Genomics business, the Company had assigned its related real estate lease, but had remained primarily liable through January 2011 in the event Ocimum failed to perform its obligations under the lease. An escrow account served partially to secure Ocimum's performance. In July 2009, the landlord agreed to release the Company from liability under the lease. The escrow agreement between Ocimum and the Company was terminated and $500 from the escrow amount was paid to the Company and applied to the outstanding principal of the Note.
9.
--------------------------------------------------------------------------------
Under the Note, Ocimum was required to pay at least fifty percent (50%) of the unpaid principal to the Company on or before August 1, 2009. To date, the Company has received payments of $1,500 (including the $500 discussed above), and the remaining balance is required to be paid on or before September 15, 2009. The Note bears interest at the rate of 15% per annum and includes an adjustment to the unpaid principal of four percent (4%) on July 15, 2009, five percent (5%) on August 15, 2009 and eight percent (8%) on September 15, 2009. The Security Agreement secures the Note with Ocimum collateral that includes, but is not limited to, the assets, properties and rights of Ocimum, its cash accounts and its receivables.
Note 6 - Lease abandonment
In the second quarter of 2009, the Company vacated substantially all of its Gaithersburg, Maryland facility and recorded a non-cash accelerated lease expense and write-down of leasehold improvements and other related assets of $749, which is included in Selling, General and Administrative expenses in the Company's Consolidated Condensed Statements of Operations.
Note 7 - Subsequent events
In May 2009, the Company was notified by Nasdaq that it no longer met the requirement for continued listing on the Nasdaq Global Market, due to the fact that its stockholders' equity fell below the minimum of $10,000. In response, the Company applied for, and Nasdaq approved, a transfer of the Company's listing from the Nasdaq Global Market to the Nasdaq Capital Market. This transfer occurred on August 3, 2009. The Company's Common Stock is currently trading below the $1.00 per share price required by Nasdaq for continued listing.
The Company evaluated all events or transactions that occurred after June 30, 2009 and through August 14, 2009, the date these financial statements were issued for recognition and disclosure.
10.
--------------------------------------------------------------------------------
Item 2. Management's Discussion and Analysis of Financial Condition
and Results of Operations
This Quarterly Report on Form 10-Q ("Form 10-Q") contains forward-looking statements regarding future events and the future results of Ore Pharmaceuticals Inc. ("Ore Pharmaceuticals") that are based on current expectations, estimates, forecasts and projections about the industries in which Ore Pharmaceuticals operates and the beliefs and assumptions of the management of Ore Pharmaceuticals. Words such as "expects," "anticipates," "targets," "goals," "projects," "intends," "plans," "believes," "seeks," "estimates," variations of such words, and similar expressions are intended to identify such forward-looking statements. These forward-looking statements are only predictions and are subject to risks, uncertainties and assumptions. Therefore, actual results may differ materially and adversely from those expressed in any forward-looking statements. Factors that might cause or contribute to such differences include those discussed in the Company's Annual Report on Form 10-K for the year ended December 31, 2008 under the section entitled "Risk Factors" and in our subsequent filings with the United States Securities and Exchange Commission ("SEC"). Ore Pharmaceuticals undertakes no obligation to revise or update publicly any forward-looking statements to reflect any change in management's expectations with regard thereto or any change in events, conditions, or circumstances on which any such statements are based.
Unless the context otherwise requires, references in this Form 10-Q to "Ore Pharmaceuticals," "DioGenix," the "Company," "we," "us," and "our" refer to Ore Pharmaceuticals Inc. and its formerly wholly owned subsidiary, DioGenix Inc.
Overview
Ore Pharmaceuticals Inc. is a pharmaceutical asset management company. We acquire interests in pharmaceutical assets whose value, we believe, we can significantly enhance through targeted development, with the goal of then monetizing these assets through a sale or out-licensing. In order to fund these activities, we intend to source third-party financing using alternative investment vehicles designed to align the investment profile of each program with the interests of its direct investors, as well as Ore shareholders.
We are establishing a business model under which Ore will earn program management advisory fees, as well as retain substantial economic interests in successful programs. Our goal is to create an investment-driven, investor returns-focused organization with the appropriate corporate structure and skill sets to execute on our strategy. To this end, we have assembled a management team with three key areas of expertise: clinical development, public- and private-market healthcare finance and pharmaceutical business development.
Initially, we will focus on developing and monetizing our current portfolio, which includes four clinical-stage compounds in-licensed from major pharmaceutical companies. Each of these compounds has been successfully tested for tolerability in human clinical trials. We are evaluating our lead compound, ORE1001, as a potential treatment for Inflammatory Bowel Disease (IDB). IDB is a severe gastrointestinal condition that is estimated to affect as many as one million patients in the United States alone. We expect to initiate a Phase Ib/IIa clinical trial in patients with ulcerative colitis - one of the two main disorders comprising IBD - in the second half of 2009.
In May 2009, we were notified by Nasdaq that we no longer met the requirement for continued listing on the Nasdaq Global Market, due to the fact that our stockholders' equity fell below the minimum of $10 million. In response, we applied for, and Nasdaq approved, a transfer of our listing from the Nasdaq Global Market to the Nasdaq Capital Market effective August 3, 2009.
We have incurred net losses in each year since our inception, including losses of $22.5 million in 2008 and $34.7 million in 2007. At June 30, 2009, we had an accumulated deficit of $378.5 million. Our losses have resulted principally from costs incurred by our ongoing business, as well as businesses we have sold. We expect to incur additional losses in the future.
Results of Operations
Three Months Ended June 30, 2009 and 2008
Revenue. We had less than $0.1 million of revenue for the three months ended June 30, 2009 compared to $1.0 million for the three months ended June 30, 2008. During the three months ended June 30, 2008, our revenue resulted primarily from a licensing agreement for certain technology unrelated to our pharmaceutical asset management business.
Research and Development Expense. Research and development expenses, which now consist almost entirely of costs associated with the further development of ORE1001, decreased to $0.6 million for the three months ended June 30, 2009 from $3.4 million for the same period in 2008. The decrease is primarily a result of lower employee and facility-related costs due to our significant workforce reductions and lower third-party costs for the further development of our lead compound, ORE1001. For 2009, we expect a significant decrease in research and development expenses over 2008, primarily as a result of workforce reductions.
Selling, General and Administrative Expense. Selling, general and administrative expenses, which now consist primarily of accounting, legal, human resources and other general corporate expenses, decreased to $2.0 million for the three months ended June 30, 2009 from $2.5 million for the same period in 2008 primarily as a result of lower employee costs due to our significant workforce reductions, partially offset by $0.7 million of facility-related lease abandonment costs (see Note 6). For 2009, we expect a significant decrease in selling, general and administrative expenses over 2008, primarily as a result of workforce reductions.
11.
--------------------------------------------------------------------------------
Net Interest Income. Net interest income decreased to $0.1 million for the three months ended June 30, 2009 from $0.2 million for the same period in 2008, due to the decline in the balance of our cash and cash equivalents and marketable securities available-for-sale and a decrease in our rates of return on investments.
Six Months Ended June 30, 2009 and 2008
Revenue. We had less than $0.1 million of revenue for the six months ended June 30, 2009 compared to $1.8 million for the six months ended June 30, 2008. During the six months ended June 30, 2008, our revenue resulted primarily from a licensing agreement for certain technology unrelated to our pharmaceutical asset management business.
Research and Development Expense. Research and development expenses, which now consist almost entirely of costs associated with the further development of ORE1001, decreased to $1.6 million for the six months ended June 30, 2009 from $6.2 million for the same period in 2008. The decrease is primarily a result of lower employee and facility-related costs due to our significant workforce reductions.
Selling, General and Administrative Expense. Selling, general and administrative expenses, which now consist primarily of accounting, legal, human resources and other general corporate expenses, decreased to $4.3 million for the six months ended June 30, 2009 from $7.7 million for the same period in 2008 primarily as a result of lower employee costs due to our significant workforce reductions, reduced professional fees relating to strategic planning and the absence of $0.4 million of expense related to the purchase of shares from a former director that occurred in 2008, partially offset by $0.4 million in net facility-related lease abandonment costs in 2009.
Net Interest Income. Net interest income decreased to $0.2 million for the six months ended June 30, 2009 from $0.5 million for the same period in 2008, due to the decline in the balance of our cash and cash equivalents and marketable securities available-for-sale and a decrease in our rates of return on investments.
Liquidity and Capital Resources
Historically, we have financed our operations through the issuance and sale of equity securities, payments from customers and sales of parts of our business and assets from time to time. As of June 30, 2009, we had approximately $4.6 million in cash and cash equivalents, compared to $10.8 million as of December 31, 2008.
Net cash used in operating activities decreased to a negative $6.1 million for the six months ended June 30, 2009 from a negative $12.5 million for the same period in 2008, primarily due to our reduced net loss for the six months ended June 30, 2009. Based on current expectations of cash usage and collection of outstanding notes, we presently anticipate that we will have sufficient cash to operate through late 2010, including the costs of initiating and completing the Phase Ib/IIa clinical trial for ORE1001, which is expected to begin in the second half 2009 and to be completed in the third quarter of 2010. We currently expect our operating cash usage for the third quarter of 2009 to be lower than that of the second quarter. We also expect our operating cash usage for the fourth quarter of 2009 to be significantly lower than in the third quarter.
For the six months ended June 30, 2009, our investing activities were not significant.
In connection with the 2008 sale of DioGenix Inc. to Nerveda, Inc., the balance of the purchase price is due pursuant to a $0.8 million interest bearing promissory note, with receipt of two principal payments of $0.4 million plus interest due December 2009 and June 2010, subject to acceleration in certain events.
In 2008, we assigned our lease in Cambridge, Massachusetts, but remain liable under the lease in the event of the assignee's default. The lease expires in August 2013 and at June 30, 2009, the total remaining amounts due under the lease for the balance of the term is $4.7 million.
In connection with the 2007 sale of our Genomics business to Ocimum, the balance of the sales price is now due pursuant to a $3 million interest bearing promissory note of which 50% was due August 1, 2009, and the remaining balance in September 2009. To date, we have received payments from Ocimum of $1.5 million under the note.
In connection with the 2006 sale of our Preclinical Division to Bridge Pharmaceuticals, Inc. ("Bridge"), less than $0.1 million of the sales price remains in escrow pending resolution between the parties. We continue to guarantee two leases now held by Bridge. The leases expire in February 2011 and December 2013 and at June 30, 2009, the total remaining amounts due under the leases for the balance of the terms is $0.9 million and $3.2 million, respectively.
Our financing activities for the six months ended June 30, 2008 consisted of the purchase of shares from a former director for $3.0 million.
In the second quarter of 2009, we received a notice requiring repayment of all amounts potentially due under a loan and a grant agreement with the State of Maryland that total $0.7 million. We have recorded the amounts due under the loan and grant agreement within current portion of long-term debt and other accrued expenses. We are in discussions with the State of Maryland concerning the terms of potential repayment of this amount.
12.
--------------------------------------------------------------------------------
We believe that existing cash and cash equivalents, the anticipated receipt of approximately $2.3 million remaining principal relating to the promissory notes from Ocimum and Nerveda and our ongoing realignment and cash conservations efforts, will enable us to support our operations through late 2010, including the costs of initiating and completing the Phase Ib/IIa clinical trial for ORE1001, which is expected to be completed in the third quarter of 2010. However, there can be no assurance that we will be successful in our continuing realignment and cash conservation efforts, the full collection of our outstanding notes receivable or, if necessary, attracting additional financing to allow us to complete the clinical trial. Furthermore, there is no assurance if we complete our clinical trial, that the results will be satisfactory or will enable us to successfully out-license our compound. If we are not successful in achieving our objectives, it might be necessary to liquidate the Company in late 2010. We currently expect long-term support of our operations to come from possible future financings and payments from commercial arrangements from our pipeline of drug candidates. These estimates are forward-looking statements that involve risks and uncertainties. Our actual future capital requirements and the adequacy of our available funds will depend on those factors discussed above and in the Company's Annual Report on Form 10-K for the year ended December 31, 2008 under the section entitled "Risk Factors" and in our subsequent filings with the SEC.
Recently Issued Accounting Pronouncements
In June 2009, the Financial Accounting Standards Board ("FASB") issued SFAS No. 168, "The FASB Accounting Standards Codification and the Hierarchy of Generally Accepted Accounting Principles, a replacement of FASB Statement No. 162" ("SFAS 168"), which establishes the FASB Accounting Standards Codification as the source of authoritative accounting principles recognized by the FASB to be applied in the preparation of financial statements in conformity with generally accepted accounting principles ("GAAP"). SFAS 168 explicitly recognizes rules and interpretive releases of the SEC under federal securities laws as authoritative GAAP for SEC registrants. SFAS 168 is effective for financial statements issued for interim and annual reporting periods ending after September 15, 2009 (the quarter ending September 30, 2009 for the Company) and will not have an impact on our final position or results of operations.
In May 2009, the FASB issued SFAS No. 165, "Subsequent Events," ("SFAS 165"), which establishes general standards of accounting for and disclosure of events that occur after the balance sheet date, but before financial statements are issued or are available to be issued. SFAS 165 is effective for financial statements issued for interim and annual reporting periods ending after June 15, 2009 (the quarter ending June 30, 2009 for the Company). The adoption of SFAS 165 did not have an impact on our financial position or results of operations.
In April 2009, the FASB issued FSP No. FAS 107-1 and Accounting Principles Board ("APB") Opinion No. 28-1, "Interim Disclosures about Fair Value of Financial Instruments" ("FSP FAS 107-1" and "APB 28-1"). FSP FAS 107-1 and APB 28-1 amend SFAS No. 107, "Disclosures about Fair Value of Financial Instruments," to require disclosures, in interim reporting periods and in financial statements for annual reporting periods, regarding the fair value of all financial instruments for which it is practicable to estimate that value, whether recognized or not on the company's balance sheet. FSP FAS 107-1 and APB 28-1 also amend FASB APB Opinion No. 28, "Interim Financial Reporting," to require entities to disclose the methods and significant assumptions used to estimate the fair value of financial instruments and describe changes in methods and significant assumptions, in both interim and annual financial statements. FSP FAS 107-1 and APB 28-1 are effective for interim reporting periods ending after June 15, 2009 (the quarter ending June 30, 2009 for the Company). While the adoption of FSP FAS 107-1 and APB 28-1 impacts our disclosures, it does not have an impact on our financial position or results of operations.
In April 2009, the FASB issued FSP SFAS 115-2 and SFAS 124-2, "Recognition and Presentation of Other-Than-Temporary Impairments," which modify the recognition requirements for other-than-temporary impairments of debt securities and enhances existing disclosures with respect to other-than-temporary impairments of debt and equity securities. FSP SFAS 115-2 and SFAS 124-2 are effective for interim and annual reporting periods ending after June 15, 2009 (the quarter ending June 30, 2009 for the Company). The adoption of FSP SFAS 115-2 and SFAS 124-2 had no impact on our financial position or results of operations.
In February 2008, the FASB issued a one-year deferral for non-financial assets and liabilities to comply with SFAS No. 157, "Fair Value Measurements" . . .
This is securities manipulation. And is illegal. There is NO 57 cent trade today.
Time & Sales
Price Size Exch Time
08/13 << zero volume
0.5846 500 NDD 08/12
0.57 100 NDD 08/12
0.57 200 NYE 08/12
0.61 400 NDD 08/12
0.5748 300 NDD 08/12
0.57 300 NDD 08/12
0.57 300 NDD 08/12
0.57 100 NDD 08/12
0.57 100 NDD 08/12
0.57 200 NDD 08/12
0.57 100 NDD 08/12
0.58 100 NDD 08/12
0.61 1438 NDD 08/12
0.6096 100 NDD 08/12
0.57 100 CBW 08/12
0.62 338 NGS 08/12
ORXE shows downtick before open zero volume today
ORXE Bid: 0.59 Ask: 0.5901 Last: 0.57 ($): -0.0146
Volume: zero
ORXE is now Nasdaq Capital Market
NASDAQ offers three market tiers:
I) NASDAQ Global Select Market – This market tier has the highest listing standards of any other market in the world as measured by financial and liquidity requirements, along with new corporate governance standards.
II) NASDAQ Global Market - formerly known as the NASDAQ National Market (NNM)
III) NASDAQ Capital Market – formerly known as the NASDAQ Small Cap Market (SCM) prior to September 2005.
2 new people recently...
James W. Fordyce Named to Ore Pharmaceuticals Board of Directors
Press Release
Source: Ore Pharmaceuticals Inc.
On Monday August 3, 2009, 5:33 pm EDT
Companies:Ore Pharmaceuticals Inc.
GAITHERSBURG, Md.--(BUSINESS WIRE)--Ore Pharmaceuticals Inc. (Nasdaq: ORXE - News), announced today the appointment of James W. Fordyce to its Board of Directors, as of July 31, 2009.
Mark J. Gabrielson, CEO and President of Ore, said, “Jim Fordyce is a wonderful addition to our Board. He has expertise in corporate development, public finance, health sciences venture capital and acquisitions. His investment and management track records have been highly successful over a long period of time. On top of all that, Jim’s dedication to the support and development of medical research through his life-long commitment to the Albert and Mary Lasker Foundation, Inc. shows that he holds progress in human health as a personal priority. We at Ore are delighted that he’s joining us.”
Mr. Fordyce is currently Managing Partner of MEDNA Partners LLC, a private advisory firm. From 1981 to 2004, he was a general partner of Prince Ventures LP, a venture capital management firm focused on investments in medicine and the life sciences. His early investments include Genentech, Applied Biosystems, Centocor and Regeneron Pharmaceuticals amongst other noteworthy health sciences companies. From 1998 to 2004, Mr. Fordyce also served as the Managing Member of Fordyce & Gabrielson LLC, a private investment management firm. He is currently a member of the Board of Directors of Dyax Corp. and several privately held companies. He is Chairman Emeritus of the Board of Directors of the Albert and Mary Lasker Foundation. Mr. Fordyce is a graduate of the University of Pennsylvania, Magdalen College, Oxford University and holds an MBA from Harvard Business School.
Mr. Fordyce said, “I am pleased to be joining the Board of Ore Pharmaceuticals as the company begins its exciting transformation into a pharmaceutical asset management company. I have great respect for Mark Gabrielson and the management team he has put in place, and I look forward to working with them and my fellow board members to build company value.”
Ameritrade shows a different price right before the open than the previous night's close -- this happens very often and there are ZERO trades for the day, ZERO trades in premarket.
Only reason I know if because I have shares, and the value of my account drops before the open -- it's been ORXE every time I have observed this.
I'm not sure. Perhaps because a higher bid was taken off the table over night or because a higher ask was taken off the table over night.
Why does ORXE tick lower before the open on zero volume?
This has happened about a dozen times in the past few weeks.
On July 15, 2009, the Company was notified that Nasdaq approved the Company's application to list its Common Stock on The Nasdaq Capital Market.
The Company's securities will be transferred from The Nasdaq Global Market to the Nasdaq Capital Market at the opening of business on August 3, 2009.
http://ih.advfn.com/p.php?pid=news&cb=1248201803&symbol=N%5EORXE&old_symbol=N%5EORXE&from_month=1&from_day=1&from_year=2001&order=desc&to_month=12&to_day=30&to_year=2011&selsrc[0]=edgar&email=Enter+Email+Here&elists=1%7C3%7C7&sec=1
Time & Sales
Price Size Exch Time
0.55 100 CBW 14:36:39
0.5596 1000 NDD 14:35:35
0.54 1355 NDD 14:23:53
0.605 939 NGS 14:23:53
0.61 100 NGS 14:23:52
0.64 139 NGS 13:19:47
0.64 521 NGS 13:19:47
0.65 200 NDD 09:47:28
0.65 100 NYE 09:47:28
0.65 100 NGS 09:47:28
surf -- is this selling, or somebody switching sides?
While running some small cap biotech scans last night, I found what must be the only stock in the sector that hasn't had any increase in volume & any positive PPS movement. May be time for ORXE the aka, "POS" biotech stock that it is to join the game.
Look at conversion prices on these stock options lol
http://ih.advfn.com/p.php?pid=nmona&cb=1240590417&article=37300167&symbol=N%5EORXE
any news?
check the local papers?
or someone buying w/o limit order...only 4600 shares traded
The only news event that can bring this "bow wow" up is partnering news, they have been trying to close some deals:
http://www.orepharma.com/drug-development-partnering-opportunities
|
Followers
|
6
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
400
|
|
Created
|
09/01/05
|
Type
|
Free
|
| Moderators | |||
http://finance.yahoo.com/q/ks?s=ORXE.PK
http://www.orepharma.com/InvestorRelations/SECFilings.aspx
http://www.nasdaq.com/asp/quotes_sec.asp?symbol=orxe&selected=orxe
http://www.form4oracle.com/company/ore-pharmaceutical-holdings-inc-orxe/company-transactions?id=967
Ore Pharmaceuticals is a drug development and indication discovery company. We utilize proprietary capabilities in integrative pharmacology to identify new uses for drugs and drug candidates. Our goal is to enrich pharmaceutical pipelines by supplying the industry's demand for development-stage drug compounds.
We do this in two ways: we identify new uses for stalled or failed drug compounds provided by our pharmaceutical partners. And we also identify new uses for drug compounds acquired from the public domain.
We use our Indication-Seeking Program and Drug Development Pipeline to quickly and efficiently reposition high-quality compounds and get them back into the pipeline so they can provide a rapid return on their initial investment.
How are we doing so far? Ore Pharmaceuticals has initiated the evaluation of more than 100 compounds provided by our pharmaceutical company partners. And we are ready to out-license the first drug from our own development pipeline: GL1001, a late-stage compound originally developed for obesity which we have identified and validated for inflammatory bowel disease (IBD) and other gastrointestinal disorders.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
