Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Good Catch Accipiterr !!! Thanks That should bring the price up some when announced.
Also today we closed above 1.0 !! that marks 10 consecutive days of closing with a bid price greater than 1.0 this will remove the deficiency label from the ONCY ticker on NASDAQ and will bring us to good standing with NASDAQ.
another good day.
Totally agree !! Good points. The Investors buying at .90 are doing it because they know ONCY will be going up. They understand Huge gains are coming. Totally disagree with those who say the company is only worth .90. That is ridiculous. The Investors negotiated the price in order to make lot's of money.
NEW news 1.25 million of equity gained by institutional investors buying at 0.90 This corroborates that this company is something truly worth investing in.
Yes I agree. solid investment. short term investors will miss out on the real profit margins by selling before buyout or major partnership. This is the kind of stock to just put in the sock drawer for a while and aim for collecting long term gains with.
Most big pharma buy out targets are those who are in the late phase 2 and early phase 3 stage of the development.
Looking forward to seeing where things go with this stock, im definitly not worried about this one.
Holding long. GLTA
There will be good news coming from a Major that will work or buy ONCY.
Starting to top out and pull back from 1.40 area it seems.
Not regretting taking the 200% profit (from .43 Limit or less suggestion) within 3 weeks as posted a few days ago since 1.30+ area as it was developing.
$ONCY is an interesting biotech with a lot of news catalyst potential, but my protocols on these types of stocks from many years of experience have taught me that trading these short term is the best consistent strategy, rather than 'investing' longer term as many common retail sheep like to do.
These stocks are not good investments most of the time due to their inherent risks such as dilutive tendencies, etc.
I believe as a rule it's always best to trade them quick & move on to the next stock by taking advantage of short term circumstances. These types of stocks overall are not trustworthy for longer term investments, but serve as good shot term trade mechanisms.
Most people who apply a long term 'investment' concept on these non-profitable junk stocks under 1.00 (for example) tend to lose a lot of money---- that's a statistical fact
Was a nice ride for my private followers from low .43 area after being suggested in early October. Approx 200% gain into the 1.30's today.
Happy to take profits here!!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=151983902
Always take profits with discipline!!
Never become emotional with these stocks!!
--------------------------------------------------------------------------
$ONCY ~ One hell of a nice move so far the last couple of days!! 1.06 for a high today (10/29/19)....
https://c.stockcharts.com/c-sc/sc?s=ONCY&p=D&yr=0&mn=5&dy=15&i=p19945227782&r=1570537545947
Been riding this since Oct 8 from the low .40's with my private e-mail followers, but looking to start take profits soon as this is watched with vigilance
The Open Window of opportunity study midterm results to be announced by end of year along with guidance for initiation of pivotal phase 3 registration study that will probably be initiated mid 2020 with cash equity associated with phase 3 initiation milestone. Then my best guess is a six months to a year for study completion and another 3-6 months for approval.
The current cash balance of about 12-14 million might be enough cash to keep things running until the cash payment associated with phase 3 initiation. But part of me thinks the company might have one more equity raising event between now and next july.
Given the late stage nature and the broad use of the REOLYSIN treatment and the intellectual property of the company patents, I also think this is undervalued. The recent clinical trial sponsored in part by pfizer may corroborate this.
Part of me expects to see another licensing agreement for some major countries that will give the company upfront cash and milestone equity payments. This could allow the company to forgo anymore dilutionary capital raising events,and if it is done correctly will allow the company enough equity to transition into a commercial biotech company selling treatments starting by as early as first half 2021 and maybe as late as about EOY 2021 or early 2022.
Therefore I am investing at these levels due to the undervalued nature of the stock and with the idea that in the next year there will be plenty of things to move this stock up one of them being a high probability of a licensing agreement, partnership, or buyout, with a major pharmaceutical company. Looking to hold for at least 1 year.
The science looks amazing, Imo way undervalued even at the $3 price target !
ONCYF Reverse Split 1 for 9.5 effective May 25, 2018
http://otce.finra.org/DailyList
Anyone left in here???
I bowed out yesterday after holding when I bought at .83 a couple of years back. I took a small loss but rs is never good. Good luck.
Just as I suspected >>
Unfortunately, Most Co's get the Price up via Reverse Split ...
It wouldn't .. Unfortunately, Most Co's get the Price up via Reverse Split ... >
Let's hope that doesn't happen here .. !
Why would listing on NASDAQ be possibly bad??
Thanks!
To whoever setup this message board, THANKS!!!
Started investing in ONCY 2 years ago and this board was very helpful getting up to speed on the stock.
Expecting a buyout by Merck.
( ONCY ) ~ Sep 11, 2017 (7:00am EDT) >
To view this release online and get more information about Oncolytics Biotech® Inc. visit: http://oncolyticsbiotech.mediaroom.com/2017-09-11-Oncolytics-Biotech-R-Announces-the-Presentation-of-REOLYSIN-R-Clinical-Data-at-ESMO-2017-Congress
Oncolytics Biotech® Announces the Presentation of REOLYSIN® Clinical Data at ESMO 2017 Congress
Additional randomized phase 2 data in metastatic breast cancer demonstrates doubling of overall survival in Hormone Receptor Positive (ER+PR+)/HER2- patients
Largest immuno-oncolytic virus safety database reported to-date
demonstrates REOLYSIN® is safe and well-tolerated
CALGARY and SAN DIEGO, Sept. 11, 2017 /CNW/ - Oncolytics Biotech® Inc. (TSX: ONC) (OTCQX: ONCYF) (Oncolytics or the Company), a biotech company developing REOLYSIN®, a first-in-class, systemically delivered immuno-oncolytic virus that activates the innate and adaptive immune systems, today announced that two poster presentations focused on REOLYSIN were presented on Sunday, September 10 at the European Society for Medical Oncology (ESMO) 2017 Congress.
•1193P, Pooled data analysis of the safety and tolerability of intravenous Pelareorep in combination with chemotherapy in 500 + cancer patients, Dr. Andres Gutierrez, Oncolytics Biotech
"Our poster on pooled data outlines the largest safety database available to date for an oncolytic virus and demonstrates that REOLYSIN is safe and well tolerated when administered in combination with chemotherapy," said Dr. Matt Coffey, President and CEO of Oncolytics Biotech.
"Regarding the metastatic breast c
Cancer data presented within this pooled safety data, further analysis of our phase two study highlights a doubling of overall survival benefit for patients with hormone receptor double-positive, HER2-negative breast cancer – a major genetic subgroup – when treated with REOLYSIN/paclitaxel combination treatment versus paclitaxel alone.
The poster demonstrates a strong safety profile and statistically significant efficacy as we continue to advance towards regulatory approval for REOLYSIN in this indication."
In the IND 213 randomized phase 2 study in metastatic breast cancer, conducted by the Canadian Cancer Trials Group, HR+ (ER+/PR+) / HER2- patients (n=47) demonstrated that the test arm of REOLYSIN/paclitaxel more than doubled median overall survival (OS) from 10.8 months on the control arm (paclitaxel alone) to 21.8 months.
The hazard ratio was 0.36 and p-value was 0.003. The ITT (intent-to-treat) group (n=74, all genetic subtypes) improved median OS from 10.4 months on the control arm to 17.4 months on the test arm.
The topline ITT data was initially announced at the American Association for Cancer Research in April 2017, continues to mature and we are hopeful that complete OS data is made available in the future.
Highlights of the pooled safety data study include:
•Adverse events reported most frequently by REOLYSIN-treated patients were reversible Grade 1 and 2 events, including fever, chills, fatigue and the gastrointestinal-related AEs of nausea, vomiting, diarrhea.
•REOLYSIN did not modify or increase chemotherapy-induced Grade 3 or 4 treatment-emergent adverse events (TEAEs).
•Certain serious TEAEs were more common in the REOLYSIN-treated arms, however the incidence of serious AEs due to febrile neutropenia and/or infection was similar in each group.
Oncolytics recently attended an End-of-Phase 2 meeting with the United States Food and Drug Administration (FDA), which addressed registration pathways for REOLYSIN for the treatment of metastatic breast cancer, the indication for which the FDA has granted Fast Track designation.
The Company will announce the outcome of this meeting after receiving the formal minutes from the agency and expects to announce the outcome of any filings coming as a result of this meeting before the end of the year.
•523P, Mechanism of Pelareorep (Pel)-mediated cell death in a Phase I study in combination with irinotecan/ fluorouracil/ leucovorin/ bevacizumab (FOLFIRI/B) in patients with KRAS mutant metastatic colorectal cancer (mCRC), Dr. Sanjay Goel, Montefiore Medical Center, NY
Poster 523, presented by Dr. Sanjay Goel of Montefiore Medical Center in New York, characterizes the mechanism of pelareorep-mediated cell death. The phase 1 trial studied the combination of REOLYSIN with irinotecan/ fluorouracil/ leucovorin/ bevacizumab (FOLFIRI/B) in patients with KRAS mutant metastatic colorectal cancer.
The study found that REOLYSIN is safe and well tolerated in combination with FOLFIRI/B, and suggests that REOLYSIN compromises cancer cell integrity via a novel mechanism of viral-mediated cytotoxicity.
About REOLYSIN
REOLYSIN® is a non-pathogenic, proprietary isolate of the unmodified reovirus: a first-in-class systemically delivered immuno-oncolytic virus for the treatment of solid tumors and hematological malignancies. The compound induces selective tumor lysis and promotes an inflamed tumor phenotype through innate and adaptive immune responses to treat a variety of cancers.
About Oncolytics Biotech Inc.
Oncolytics is a biotechnology company developing REOLYSIN, a systemically delivered immuno-oncolytic virus. The compound induces selective tumor lysis and promotes an inflamed tumor phenotype through innate and adaptive immune responses to treat a variety of cancers. Oncolytics' clinical development program emphasizes three pillars: chemotherapy combinations to trigger selective tumor lysis; immuno-therapy combinations to produce adaptive immune responses; and immune modulator (IMiD) combinations to facilitate innate immune responses. Oncolytics is currently planning its first registration study in metastatic breast cancer, as well as studies in combination with checkpoint inhibitors as well as targeted and IMiD therapies in solid and hematological malignancies. For further information about Oncolytics, please visit: www.oncolyticsbiotech.com.
This press release contains forward-looking statements, within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements, including the Company's belief as to the potential of REOLYSIN® as a cancer therapeutic; the Company's expectations as to the success of its research and development programs in 2017 and beyond, the Company's planned operations, the value of the additional patents and intellectual property; the Company's expectations related to the applications of the patented technology; the Company's expectations as to adequacy of its existing capital resources; the design, timing, success of planned clinical trial programs; and other statements related to anticipated developments in the Company's business and technologies involve known and unknown risks and uncertainties, which could cause the Company's actual results to differ materially from those in the forward-looking statements. Such risks and uncertainties include, among others, the availability of funds and resources to pursue research and development projects, the efficacy of REOLYSIN as a cancer treatment, the success and timely completion of clinical studies and trials, the Company's ability to successfully commercialize REOLYSIN, uncertainties related to the research and development of pharmaceuticals, uncertainties related to the regulatory process and general changes to the economic environment. Investors should consult the Company's quarterly and annual filings with the Canadian and U.S. securities commissions for additional information on risks and uncertainties relating to the forward-looking statements. Investors are cautioned against placing undue reliance on forward-looking statements. The Company does not undertake to update these forward-looking statements, except as required by applicable laws.
SOURCE Oncolytics Biotech Inc.
For further information: Investor Relations & Company Contact: Michael Moore, Vice President, Investor Relations & Corporate Communications, 858-886-7813, mmoore@oncolytics.ca; Media Contact: Mark Corbae, Canale Communications, (619) 849-5375, mark@canalecomm.com
Click Here for a complete listing of Oncolytics press releases.
Oncolytics Biotech® Inc. Opens U.S. Based Office in San Diego
CALGARY and SAN DIEGO, June 27, 2017 /CNW/ - Oncolytics Biotech® Inc. (TSX: ONC) (OTCQX: ONCYF) (Oncolytics or the Company) today announced that it has opened an office in San Diego, California. The office will support investor relations, business development and clinical operations functions.
"Expanding our operations into this leading biotech hub within one of the largest biopharmaceutical markets in the world, allows us to aggressively pursue our business development efforts and leverage the talent and expertise this region offers," said Dr. Matt Coffey, President and CEO of Oncolytics Biotech.
"Not only does it bring us closer to a pool of talent that will help us with preparation and execution of our planned phase three registration study in metastatic breast cancer, it also provides immediate proximity to our manufacturing group located in Carlsbad, California, which is responsible for our late-stage clinical lead agent REOLYSIN."
About Oncolytics Biotech Inc.
Oncolytics is a biotechnology company developing REOLYSIN, an immuno-oncology viral agent, as a potential treatment for a variety of tumor types. The compound induces selective tumor lysis and promotes an inflamed tumor phenotype through innate and adaptive immune responses to treat a variety of cancers. Oncolytics' clinical development program emphasizes three pillars: chemotherapy combinations to trigger selective tumor lysis; immuno-therapy combinations to produce adaptive immune responses; and immune modulator (IMiD) combinations to facilitate innate immune responses. Oncolytics is currently planning its first registration study in breast cancer, as well as studies in combination with checkpoint inhibitors and IMID/targeted therapies in solid and hematological malignancies. For further information about Oncolytics, please visit: www.oncolyticsbiotech.com.
This press release contains forward-looking statements, within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements, including the Company's belief as to the potential of REOLYSIN® as a cancer therapeutic; the Company's expectations as to the success of its research and development programs in 2017 and beyond, the Company's planned operations, the value of the additional patents and intellectual property; the Company's expectations related to the applications of the patented technology; the Company's expectations as to adequacy of its existing capital resources; the design, timing, success of planned clinical trial programs; and other statements related to anticipated developments in the Company's business and technologies involve known and unknown risks and uncertainties, which could cause the Company's actual results to differ materially from those in the forward-looking statements. Such risks and uncertainties include, among others, the availability of funds and resources to pursue research and development projects, the efficacy of REOLYSIN as a cancer treatment, the success and timely completion of clinical studies and trials, the Company's ability to successfully commercialize REOLYSIN, uncertainties related to the research and development of pharmaceuticals, uncertainties related to the regulatory process and general changes to the economic environment. Investors should consult the Company's quarterly and annual filings with the Canadian and U.S. securities commissions for additional information on risks and uncertainties relating to the forward-looking statements. Investors are cautioned against placing undue reliance on forward-looking statements. The Company does not undertake to update these forward-looking statements, except as required by applicable laws.
SOURCE Oncolytics Biotech Inc.
For further information: Oncolytics Investor & Media Contact: Oncolytics Biotech Inc., Michael Moore, Vice President, Investor Relations & Corporate Communications, 858-886-7813, mmoore@oncolytics.ca
Click Here for a complete listing of Oncolytics press releases.
$ONCYF offering closed and ASCO presentation this weekend
Usually, when it comes to pending news, short sellers will come in to get people to sell so they can gobble up those shares. This stock, like many others, have their ups and downs because of this manipulation.
Exactly what bad news?? Why could the price be going down like this?
What bad news?
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
Report of Foreign Private Issuer
Pursuant to Rule 13a-16 or 15d-16
of the Securities Exchange Act of 1934
For the month of May 2017
Commission File Number 000-31062
Oncolytics Biotech Inc.
(Translation of registrant’s name into English)
Suite 210, 1167 Kensington Crescent NW
Calgary, Alberta, Canada T2N 1X7
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F þ Form 40-F ?
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ?
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ?
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
Indicate by check mark whether by furnishing the information contained in this Form, the registrant is also thereby furnishing the information to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934.
Yes ? No þ
If “Yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b): 82 -
EXHIBIT
NUMBER
DESCRIPTION
99.1 News Release Dated May 23, 2017 - Oncolytics Biotech® Announces Proposed Public Offering
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Oncolytics Biotech Inc.
(Registrant)
Date: May 23, 2017 By: /s/ Kirk Look
Kirk Look
Chief Financial Officer
Click on the above link. It explains all.
$2 according to Zack's Research
http://scr.zacks.com/News/Press-Releases/Press-Release-Details/2017/ONCYF-FDA-Grants-Fast-Track-Authority/default.aspx
A partnership could boost it to that level, but I'm hoping for a
buyout.
Price target?
Realistically...what would be a price target for Oncolytics? Do you forsee a partnership or a buyout? I think the future is bright for Oncolytics.
I agree. I have been in this for a long time.
Yes, I would like for it to go north, but these short sellers play their game with these stocks (especially biotech/pharma co's) and it will be in their court unless some crazy news comes out. I'm hoping for a major partnership or a total buyout.
Time to go North.
More to come from this company. IMHO
Updates from the Annual General Meeting
I was wondering if anyone could provide updates from the annual general meeting held yesterday for shareholders. I do believe that this company is headed in the right direction with the leadership of Matt Coffey. Curious to see the potential of Reolysin and its effect on various cancers.
But I see it right track and work hard
Did you listen to the conference, and if so, are basing your figure on what you heard?
Is that your prediction for close tomorrow or runup next week?
|
Followers
|
73
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1874
|
|
Created
|
10/09/01
|
Type
|
Free
|
| Moderators | |||


See the official company website for the most current information. www.oncolyticsbiotech.com
CURRENT SUMMARY - March 9th 2014
OVERVIEW

 Dr Kevin Harrington - UK
Dr Kevin Harrington - UK  , Dr. James A Bonner - US
, Dr. James A Bonner - US
Publication of data for the PH I/II Ovarian cancer trial also sponsored by the NCI and conducted by Principal Investigator Dr. David Cohn 
COMPANY PROGRESS
The ubiquitous reovirus

Reovirus, an acronym for Respiratory Enteric Orphan virus, is generally believed to inhabit the respiratory and bowel systems in humans. Reovirus is found naturally in sewage and water supplies. By age 12, half of all children show evidence of reovirus exposure and by adulthood, most people have been exposed. However, the disease is non-pathogenic, meaning there are typically no symptoms from infections. The link to its cancer-killing ability was established after the reovirus was discovered to reproduce well in various cancer cell lines.

Using improved microscope technology, a team including Purdue's Timothy S. Baker and a colleague at Harvard has determined the structure of a reovirus (short for "respiratory enteric orphan" virus) down to the 7.6-angstrom scale, better than twice the 18-angstrom resolution previously available. http://www.purdue.edu/UNS/html4ever/031215.Baker.reovirus.html
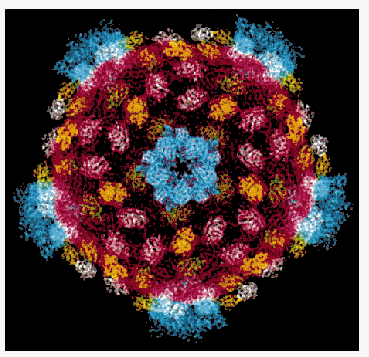
http://news.chess.cornell.edu/archive/highlights/rvrscr.htm
Synchrotron radiation is the only tool available for the determination of very large molecular structures at high resolution such as the reovirus core. One of the largest structures solved to-date has been reported from work carried out at MacCHESS by Karin Reinish in the Harrison group at Harvard. The reovirus core is a macromolecular assembly with a molecular mass of 52 million. The core synthesizes, modifies, and exports viral messenger RNA. The core contains five of the eight proteins that make up a complete virion and is about 700 Angstroms in diameter. They crystallize in a centered cubic space group with unit cell dimensions of 1255 Angstrom with crystal growth requiring 9 to 12 months. Using the CHESS F1 facility, one of the two Biosafety Level 2 facilities in the US, scientists have been able to "see" into the three-dimensional structure of the core using the tools of x-ray crystallography.
The reovirus core particle shows the subunits in different colors. There are 120 copies of the part in red that forms the shell and that packages the RNA. This part defines the symmetry and size of the particle. Other subunits, shown in yellow, green and white stabilize the shell. The blue parts form turret-like structures around the fivefold axes that exports mature mRNA into the cytoplasm of the infected cell.
THE PRODUCT

Mechanism of Action

The reovirus, or Respiratory Enteric Orphan virus, has been demonstrated to replicate specifically in tumor cells that have a constitutively activated Ras pathway. Activating mutations of Ras and mutations along the Ras pathway occur in approximately two-thirds of all tumors. Tumors bearing an activated Ras pathway are deficient in their ability to activate an anti-viral response mediated by the host cellular protein, PKR. Since PKR is responsible for preventing reovirus replication, tumor cells that lack the activity of PKR are susceptible to reovirus infection and eventual cell death. As normal cells do not possess Ras activation, these cells are able to thwart reovirus infection by the activity of PKR. In a tumor cell with an activated Ras pathway, the reovirus is able to freely replicate and kill the host tumor cell. Progeny virus particles are then able to infect and kill surrounding cancer cells. This cycle of infection, replication and cell death is believed to be repeated until there are no longer any tumor cells carrying an activated Ras pathway available.
The Company's technologies are based on discoveries made in the 1990s in the Department of Microbiology and Infectious Diseases at the University of Calgary. The potential products are being developed using the naturally occurring reovirus for treatment of cancers in humans.
The KRAS Opportunity
In mid-2009, the U.S. FDA approved revisions to labeling of the epidermal growth factor receptor (EGFR) class of antibodies, indicating that colorectal patients who have KRAS mutations in their tumours do not respond to EGFR-inhibiting antibodies and that the use of this class of pharmaceuticals is not recommended for these patients.
REOLYSIN, Oncolytics' proprietary isolate of the reovirus, preferentially replicates in cancer cells that have an activated RAS pathway. Approximately two-thirds of all cancers have an activated RAS pathway, including most metastatic disease. A large number of mutations, including mutations in EGFR, Her2 or KRAS along the RAS pathway lead to RAS pathway activation. A significant clinical opportunity for REOLYSIN is in the treatment of patients with metastatic cancers who have a mutated KRAS gene and are unlikely to respond to treatment with anit-EGFR monoclonal antibodies.
Current Clinical Trials Roadmap
Company published list by Trial Number http://www.oncolyticsbiotech.com/clinical.html
List of US based clinical trials on the official ClinicalTrials.gov website...Search Term: Reolysin
List of EU based clinical trials on the official EU .... https://www.clinicaltrialsregister.eu/ctr-search/search?query=reovirus
Key Published Scientific Data
Links collected here on the company website
Oncolytic Reovirus Effectively Targets Breast Cancer Stem Cells by Patrick Lee's lab published March 17, 2009 in Mol Therapy
DAILY and WEEKLY Stock Charts
| Trading Data | Share Structure | ||
| | |||
| Exchange Symbol: | TSX: ONC | Estimated As at Jun, 2012 | |
| NASDAQ: ONCY | Outstanding: | 76.6 million | |
| ***Cash On Hand Reported to be approx CAD $31.25 Mil on June 30th, 2012 including the bought deal of CAD $18+Mil conversion announced in Feb 2012. Monthly Burn Rate has increased to $3.1 Mil - From Q2 2012 6mo Income Statement. Current and Past Financials can be found by clicking here! | |||
- All numbers are stated in Canadian Dollars
History
Curing Cancer? Patrick Lee's Path to the Reovirus Treatment
http://cogsci.uwaterloo.ca/Articles/Lee.htm
by Paul Thagard from the Philosophy Department of Waterloo in 2002
Dalhousie virologist Patrick Lee:
http://bhcri.ca/dr-patrick-lee























| The waterfall graph above shows the percentage of tumour regression on a per patient basis at the lesion injected with REOLYSIN®, measured against baseline. |
| *Bar graph is ordered by best response at injected lesion, starting from tumour regression of 100% as shown by first two bars on far left of graph. Results do not reflect the order in which patients were treated, or the dosage received. http://www.oncolyticsbiotech.com/English/clinical-trials/clinical-trials-news/clinical-trials-details/2013/REO-001/ |
Selected Highlights
Since January 1, 2014, selected highlights announced by the Company include:
Clinical Program
Completion of patient enrollment in an ongoing, NCIC Clinical Trials Group sponsored randomized Phase II study of REOLYSIN® in patients with advanced or metastatic colorectal cancer (IND 210). The Company awaits preliminary data from this study;
Reporting completion of enrollment and interim overall and KRAS-mutated patient data from an NCI-sponsored randomized Phase II study of REOLYSIN® in combination with carboplatin and paclitaxel in patients with recurrent or metastatic pancreatic cancer (NCI-8601). The Company awaits final data from this study, which will be available once all remaining patients have progressed;
Completion of patient enrollment in an ongoing, NCI-sponsored randomized Phase II study of REOLYSIN® in combination with paclitaxel in patients with persistent or recurrent ovarian, fallopian tube or primary peritoneal cancer (GOG-186H). The Company awaits preliminary data from this study;
Reporting final data from the Company's randomized, double-blinded clinical study examining REOLYSIN® in combination with carboplatin and paclitaxel in patients with second-line, platinum-refractory, taxane-naïve head and neck cancers;
Presentation by the Company's collaborators of preliminary clinical data demonstrating that intravenously delivered REOLYSIN® can cross the blood brain barrier to access tumours in the brains of humans;
Regulatory
Application for Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) for REOLYSIN® in the treatment of ovarian and pancreatic cancers. The Company was granted designations in ovarian, fallopian tube, primary peritoneal and pancreatic cancer subsequent to year end;
Application to the European Medicines Agency for Orphan Designation for REOLYSIN® in the treatment of pancreatic and ovarian cancers;
Subsequent to year end, the Company applied to the U.S. FDA for a fifth Orphan Drug Designation for high grade gliomas in paediatric patients;
Basic Research
Presentation of a poster entitled "Combination Therapy with Reovirus and PD-1 Blockade Effectively Establishes Tumour Control Via Innate and Adaptive Immune Responses" by the Company's research collaborators, Vile et al., at the AACR Tumor Immunology and Immunotherapy Conference;
A series of presentations made by the Company's research collaborators at the 8th Annual International Conference on Oncolytic Virus Therapeutics held in Oxford, UK, covering:
Preclinical research examining the synergies associated with treatment in animal models with GM-CSF prior to administering REOLYSIN®;
Preclinical research focused on identifying biomarkers predictive of sensitivity/resistance to reovirus in head and neck cancer cell lines; and
Preclinical research into the treatment of hepatocellular carcinoma associated with infection by Hepatitis B and Hepatitis C;
Governance
The nomination and election of Ms. Linda Hohol and Ms. Angela Holtham to the Company's Board of Directors;
Financial
Entry into and subsequent amendments to a share purchase agreement with Lincoln Park Capital Fund, LLC;
Entry into a $20 million "At-the-Market" equity distribution agreement with Canaccord Genuity Inc.; and
At December 31, 2014 the Company reported $16.2 million in cash, cash equivalents and short-term investments. At March 13, 2015, the Company had approximately $27.5 million in cash, cash equivalents and short-term investments.
http://ih.advfn.com/p.php?pid=nmona&article=65886481

| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
