Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Do you own shares of ocugen?
Ya think so, lol. We are not the ones on the losing end here pal
Scam stocks never hold value, an this turd is one of them
Oh come on, realize the reality of this stock, lets be real. This ceo has done nothing and never has accomplished anything, last company he ran went bankrupt, do your DD, they have no drug phases anywhere near FDA status for approval!!
Looks like their conferences were a total failure as expected, no good news just bad as usual with this ceo, scamming thief lol
Loving this stock, another round of massive shorting to .50 or lower, anyone long here is absolutely crazy, its junk and worthless lmao
Hahahahaha
Here we are brainless, a few pennies on the green side an u think this turd is a winner, back down she goes soon enough
Where’s that tard harry crumb lol
Good afternoon, Ocuhub.
OCGN
It is a loser, i or we are not my friend but you will be in due time after the huge RS coming
Anybody can post pal, so u get lost. My opinion yur opinion. Ocgn is junk
Agreed. He always complains
Dude, if you think it’s a scam, then don’t invest and leave the board. Nobody really cares what you think.
There was a R/S on 9/30/19 (1 for 50). I will wait for the aftermath to settle down after the next R/S before getting in here.
It’ll come crashing down soon enough, this turd is tarnished an no revenue, ceo an board are just taking money, look up his track record. All failures!
NEWS
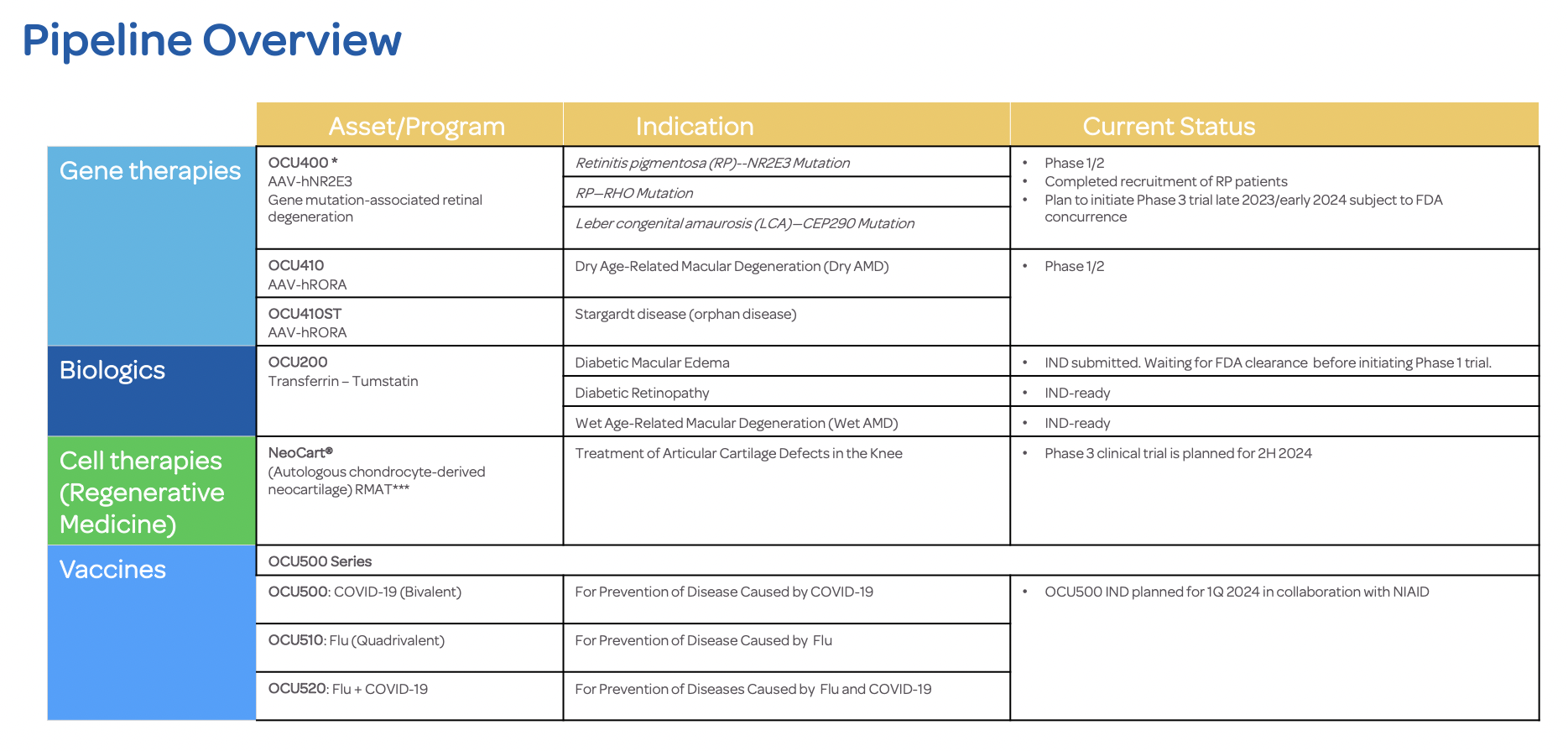
Ocugen Gains FDA Alignment on Key Aspects of OCU400—Modifier Gene Therapy—Pivotal Phase 3 Study Design
MALVERN, Pa., Dec. 21, 2023 (GLOBE NEWSWIRE) -- Ocugen, Inc. (Ocugen or the Company) (NASDAQ: OCGN), a biotechnology company focused on discovering, developing, and commercializing novel gene and cell therapies and vaccines, today announced that the Company received alignment from FDA on key aspects of the Phase 3 clinical trial design to assess the safety and efficacy of OCU400 in patients with RHO and other gene mutations associated with Retinitis Pigmentosa (RP).
“This news brings us even closer to fulfilling our mission to bring our first-in-class, gene-agnostic therapies to market and provide access to patients globally,” said Dr. Shankar Musunuri, Chairman, Chief Executive Officer, and Co-Founder of Ocugen. “We look forward to beginning the Phase 3 clinical trial, which we plan to initiate in early 2024.”
During a multidisciplinary meeting with FDA, based on preliminary results from an ongoing Phase 1/2 study, Ocugen received alignment on key aspects of the Phase 3 study design—including the study endpoint, patient enrollment strategy, and study duration of one year. The Phase 3 clinical trial will enroll a broader group of RP patients, including patients with the most common RHO gene mutation, based on OCU400’s potentially gene-agnostic mechanism of action.
With orphan drug and RMAT designations in place for OCU400, FDA’s alignment on key aspects of the Phase 3 study design positions Ocugen to confidently move forward in pursuing product development and licensure for OCU400.
Currently there are approximately 110,000 patients in the United States with RP and 1.6 million patients globally. Of these patients, more than 10% have the RHO genetic mutation. Advancing OCU400 to Phase 3 clinical development will be an important step toward addressing unmet needs in the RP patient community.
About Ocugen, Inc.
Ocugen, Inc. is a biotechnology company focused on discovering, developing, and commercializing novel gene and cell therapies and vaccines that improve health and offer hope for patients across the globe. We are making an impact on patient’s lives through courageous innovation—forging new scientific paths that harness our unique intellectual and human capital. Our breakthrough modifier gene therapy platform has the potential to treat multiple retinal diseases with a single product, and we are advancing research in infectious diseases to support public health and orthopedic diseases to address unmet medical needs. Discover more at www.ocugen.com and follow us on X and LinkedIn.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding qualitative assessments of available data, potential therapeutic and clinical benefits of our product candidates, expectations for clinical trial timing and results, anticipated timing of clinical trial updates and expectations for timing and outcome of regulatory interactions, which are subject to risks and uncertainties. We may, in some cases, use ter
Total junk. 💩 Managed by 🤡.
https://www.marketscreener.com/quote/stock/OCUGEN-INC-19157135/news/OCUGEN-INC-Results-of-Operations-and-Financial-Condition-Notice-of-Delisting-or-Failure-to-Sati-43760568/
130 days to delisting. At $.37 and staying there all year. Can anyone think this is going to get to $1 by May?
“Project NextGen will also support 10 to 15 phase 1 trials and 6 to 8 phase 2 trials at the NIAID, Beigel said. Companies will provide their vaccine candidates, while the NIAID will cover the costs of the trials.”
“Ocugen is the other company that has publicized its vaccine’s selection for NIAID trials. Its mucosal vaccine uses a chimp adenovirus to deliver the SARS-CoV-2 spike protein. Although the vaccine hasn’t yet been tested in humans in the US, it has completed phase 1, 2, and 3 trials in India, where, in the form of nose drops, it was authorized more than a year ago as a primary vaccine as well as a mix-and-match booster.”
https://jamanetwork.com/journals/jama/fullarticle/2812830
OCGN
OCGN Noblecon replay https://www.youtube.com/embed/6ik0xs3Z9Us
Gm ocuhub. Noble conference 2pm et
OCGN
Wait for the bad news coming soon, lmao
.05? I don’t think so
OCGN
Nothing but junk, lets get it to .05 before the massive RS! $$$$$!
|
Followers
|
226
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
5674
|
|
Created
|
11/13/15
|
Type
|
Free
|
| Moderators sgunderbarth | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
