Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Lol told ya!our own government stealing from us
Shorts going to drop a bomb rite out the gate to get everyone that bought premarket to hit stop loss
Don't worry they will short it till we close in red like usual
Insiders are loading now. Hhhmmmm, could very positive news be on the horizon here
.25 coming then the big RS, thats what happens when scum an thieves run a company! Lmao
Maybe some good science coming here folks. $$$
Yep been awhile my friend
Long time no see.
Is it a buy now? I lost on this crap on the fda rejection for the vaccine approval.
Dumped, in an out! Quick money. This company is on pins an needles, only good for flippn
We loaded up this little bio could pop to 3.50/4.00, patience an sit back on them
After hours great news yesterday!!!
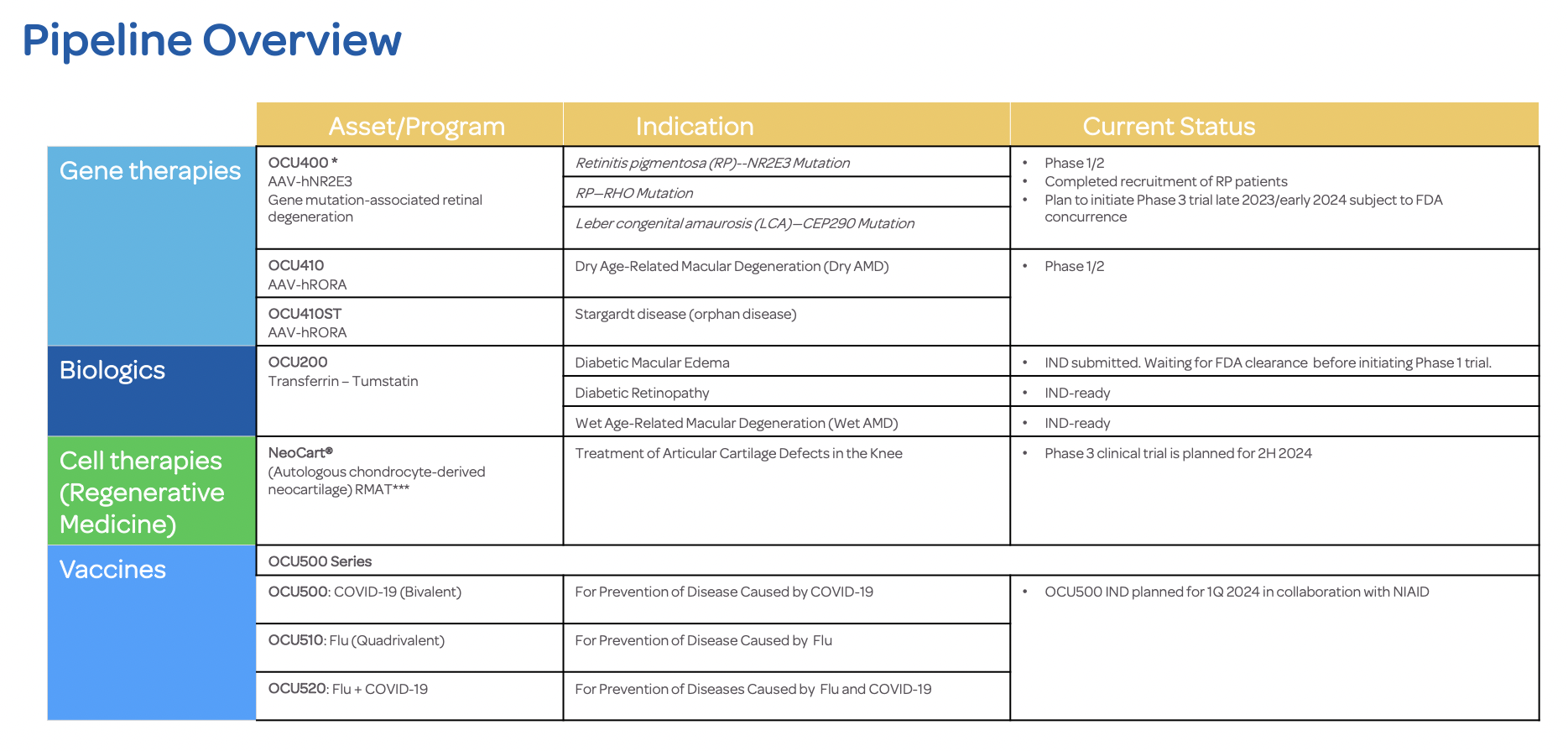
Ocugen Announces Completion of Dosing in OCU400 Phase 1/2 Cohort 2
Source: GlobeNewswire Inc.?
Ocugen, Inc. (Ocugen or the Company) (NASDAQ: OCGN), a biotechnology company focused on discovering, developing, and commercializing novel gene and cell therapies and vaccines, today announced that the Independent Data and Safety Monitoring Board (DSMB) for the OCU400 Phase 1/2 clinical trial completed a review of safety data for subjects enrolled in Cohort 2 and recommends proceeding to enroll subjects in Cohort 3. The Company expects to complete Cohort 3 enrollment in Q4 2022 as planned.
“I’m very pleased with the progress of the clinical trial,” said David Birch, PhD, Director, Retina Foundation of the Southwest, primary investigator of the study. “Currently, patients with inherited retinal degeneration have nothing to address their condition in the long-term. It is imperative to keep working toward a solution for these patients who currently have no hope.”
OCU400 clinical trial is a Phase 1/2 Study to Assess the Safety and Efficacy of OCU400 for Retinitis Pigmentosa Associated with NR2E3 (Nuclear Receptor Subfamily 2 Group E Member 3) and RHO (Rhodopsin) Mutations. OCU400 is part of Ocugen’s Modifier Gene Therapy Platform—targeting inherited retinal disease and dry age-related macular degeneration.
Mizuho Securities Sticks to Their Buy Rating for Ocugen (OCGN)
Source: TipRanks
Mizuho Securities analyst Uy Ear maintained a Buy rating on Ocugen (OCGN - Research Report) today and set a price target of $5.00. The company's shares closed yesterday at $1.64.Ear covers the Healthcare sector, focusing on stocks such as Arcutis Biotherapeutics, ACADIA Pharmaceuticals, and Ocugen. According to TipRanks, Ear has an average return of -10.4% and a 27.27% success rate on recommended stocks. The word on The Street in general, suggests a Strong Buy analyst consensus rating for Ocugen with a $6.33 average price target, which is a 285.98% upside from current levels.
https://www.tipranks.com/news/blurbs/mizuho-securities-sticks-to-their-buy-rating-for-ocugen-ocgn?utm_source=advfn.com&utm_medium=referral
Bbv152 looks promising folks. Just never know
$30 options open for January
OCGN
Been loading the 2.30’s an flipping the 2.80’s, good wealth on these lines
If the shorts pound this down to the 2 area i would expect a huge volume spike to 2.50’s again, lets c where they take it. The flip has been the 2.50’s to the 2.80’s on a large level but with the recent 8k an news, it’s probable new trading ranges develope since nothing real positive came out here
It still could I’m just stating facts about their recent press release on available funds. These types of stocks are very sketchy/risky. So, I’m both ways on this one.
What happened to your multi bagger
Then what? Let the ATM begin an dilution. We see this as a 1.25/1.50 pps stock without any real positive news on fda approvals or new science trials
“The Company believes that its current cash and cash equivalents balance will enable it to fund its operations into the second quarter of 2023.”
https://ir.ocugen.com/news-releases/news-release-details/ocugen-provides-business-update-second-quarter-2022-financial
OCGN
Hmm. If they're announcing the earnings for Q2 early tomorrow, maybe that means it's good news.
Possible multi bagger here is what could happen
17.65 52 week high, on some positive news this rips folks
We are here an adding, $4 pps is a safe an reasonable target imo
Quad witching for options.
Ocugen, Inc. Announces Issuance of U.S. Patent for Treating Retinal Degenerative Diseases Using ...
MALVERN, Pa., June 13, 2022 (GLOBE NEWSWIRE) -- Ocugen, Inc. (NASDAQ:OCGN), a clinical-stage biopharmaceutical company focused on discovering, developing, and commercializing novel gene and cell therapies, biologicals, and vaccines, today announced that on June 7, 2022, the United States Patent and Trademark Office (“USPTO”) issued U.S. Patent No. 11,351,225, which is directed to methods for preventing or treating an ocular disease or disorder associated with a retinal degenerative disease.
U.S. Patent No. 11,351,225 (the “‘225 Patent”) covers the use of a nuclear hormone receptor gene, such as NR2E3, RORA, NUPR1, and NR2C1, in treating retinal degenerative diseases as well as reducing the risk of developing such diseases. Additional issued claims pertain to using a nuclear hormone receptor gene to treat retinitis pigmentosa, age-related macular degeneration, and inherited retinal degenerative diseases. The ‘225 Patent contains 18 claims and expires in March 2034.
“We are pleased to have been granted this new U.S. patent through our exclusive license agreement with The Schepens Eye Research Institute, an affiliate of Harvard Medical School. We believe this patent significantly validates our modifier gene therapy platform developed by Dr. Neena Haider and augments our growing global patent portfolio,” commented Dr. Shankar Musunuri, Chairman, CEO, and Co-Founder of Ocugen.
This newly allowed patent is exclusive to Ocugen and is the latest U.S. patent issued in connection with Ocugen’s gene therapy program for treating retinal degenerative diseases.
About Ocugen, Inc.
Ocugen, Inc. is a biotechnology company focused on discovering, developing, and commercializing novel gene and cell therapies, biologicals and vaccines that improve health and offer hope for people and global communities. We are making an impact through courageous innovation, taking science in new directions in service of patients. Our breakthrough modifier gene therapy platform has the potential to treat multiple diseases with one drug and we are advancing research in other therapeutic areas to offer new options for people with unmet medical needs. Discover more at www.ocugen.com and follow us on Twitter and LinkedIn.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995, which are subject to risks and uncertainties. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from our current expectations. These and other risks and uncertainties are more fully described in our periodic filings with the Securities and Exchange Commission (“SEC”), including the risk factors described in the section entitled “Risk Factors” in the quarterly and annual reports that we file with the SEC. Any forward-looking statements that we make in this press release speak only as of the date of this press release. Except as required by law, we assume no obligation to update forward-looking statements contained in this press release whether as a result of new information, future events or otherwise, after the date of this press release.
Contact
Tiberend Strategic Advisors, Inc.
Jonathan Nugent / Daniel Kontoh-Boateng (Investors)
How much is ocugen paying for postage? I have been getting a piece in the mail every day for two weeks regarding the annual meeting. Today it looks like I’m getting three.
News this morning. Also a related 8k, link here:
FDA hold lifted
https://last10k.com/sec-filings/ocgn/0001104659-22-063664.htm?utm_source=stocktwits&utm_medium=forum&utm_campaign=8K&utm_term=ocgn
GO,GO,GO OCGN NOW IN MEXICO!!!HOLDEN THE GOLDEN.
OCGN Definitely NOT a One Trick Pony $$$$
only if you invest now - buy before the NEWS
Been a great swing, flip play here for a couple months.
|
Followers
|
224
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
5736
|
|
Created
|
11/13/15
|
Type
|
Free
|
| Moderators sgunderbarth | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
