Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
MYMX ask .0022 6.47million Shares traded
Still hanging on here and trying to remain hopeful for a decent outcome however I am concerned there doesn't seem to be much of an underlying speculative interest in MYMX. I would be surprised if the purposed buyout details were a tightly guarded secret. Maybe they are but it seems someone always knows what's likely to happen and they act accordingly nudging the price up or down.
That said, even though there hasn't been a steady stream of accumulation, there hasn't been a real sell off either. Since any selling at all will produce disastrous results like today's beat down it's hard to judge. Maybe it's just retail guy throwing in the towel or a margin call. Volume is climbing now so maybe there will be a confirmation of sorts.
We'll see if anyone rushes in to buy down here,Cheers.
MYMX ask .0037 OneMillionNineHundredThousandShares It would appear trust has been broken and the sell off has begun.
A polyvalent virosomal influenza vaccine induces broad cellular and humoral immunity in pigs
16 August 2023.
Brazil.
https://virologyj.biomedcentral.com/articles/10.1186/s12985-023-02153-5
Thank you Staypositive1.
Did you see MYMX's consortium partner received The King's Award at Buckingham Palace?
What happened to that small percentage of a percentage arrangement with Upperton Pharma?
https://upperton.com/upperton-pharma-solutions-founder-heads-to-buckingham-palace-for-kings-award-honour/
Qubit dephasing Dr. Thomas Staehelin?
Speen Holding AG could parlay MYMX's delivery system to Swiss Rockets.
Hope Gotthard rail tunnel gets rebuilt quickly and glad no one was injured.
According to what I'm reading at the end of the paragraph they only have 5 days to file.
The subject annual report, semi-annual report, transition report on Form 10-K, Form 20-F, Form 11-K, Form N-CEN or Form N-CSR, or
portion thereof, will be filed on or before the fifteenth calendar day following the prescribed due date; or the subject quarterly report or
transition report on Form 10-Q or subject distribution report on Form 10-D, or portion thereof, will be filed on or before the fifth calendar day
following the prescribed due date;
The NT has been filed.
Surprising to me, Mr. Kempers writes: "[...] the Registrant will file its Form 10-Q no later than the fifteenth calendar day following the prescribed due date." Was there a recent change in obligations in the US, since if I remember correctly only 10-K's NT forms lead to a grace period of 15 days and 10-Q's only to 5 days?
I think the deadline was yesterday. If the 10-Q does not come before market open and no NT is published they should be in delay.
I would think they would want to take advantage soon. The market has a lot of penny annie stocks which are popping after the sp500 stocks are starting to peter out. A little like the 1999 penny stock runs but not as many as then when the internet stocks were the big thing. Just my opinion but I think the market about to make a reversal sometime in the next 6 to 9 months big time reversal.... Pennies stocks will push as the big boys look for fresh meat... MYMX should want to make a move soon if it really has some kind of pending offer..
Back below a cent. Is something coming soon I hope?
VTGN spike 14.00 MYMX Staehelin Combo Next spike wishful thinking
I saw this filing last night. They said in the filing it would take several months for the wind down to be completed. If that's the case, is it going to hold up the sale of the company. I'm not sure if they can sell until this is completed. They had two companies for 7 months looking into the value of MYMX. When they put themselves up for sale through the bid process you would think they would have everything in order. We might find out more details in the next filing in a couple weeks. I'm hoping that's not the case. The one good thing is they wouldn't be going through the trouble of getting the patent or patents back if they were going to fold the company as a whole. If anyone knows if the sale could still go through prior to this wind down being completed, I welcome your insight. Thanks
8K: "BH has a remaining patent, which the Company expects to acquire to partially compensate for the approximate Euro 6 million accumulated intercompany loans from Mymetics Corporation to BH."
BH actually has two remaining patents on record:
1) Antigen-complexes Bestewil BV. It expires on November 19, 2023.
2) Virosome-like particles Bestewil BV. It expires on December 2, 2023.
Were Toon Stegmann and Jan Wilschut being compensated through these accumulated intercompany loans?
I guess Martin O. Pestalozzi (Ulrich Burkhard) and the offshore trust members are okay with this move.
Thomas Staehelin's favorite biotech is Serbia bound.
July 26, 2023.
Swiss Rockets AG to open an innovation and cancer research centre on BIO4 campus
https://www.serbianmonitor.com/en/swiss-rockets-ag-to-open-an-innovation-and-cancer-research-centre-on-bio4-campus/
"The project has received $500 million in private funding from the family of Dr. Thomas Staehelin and Monique Staehelin."
https://www.forbes.com/sites/moorinsights/2023/07/17/strategic-partnership-between-ionq-and-quantumbasel-could-transform-switzerland-into-a-european-quantum-powerhouse/?sh=1618261845f7
July 17, 2023.
You're welcome Castaways.
I hope life is good my friend.
Enjoy your weekend.
Thanks yet again for your timely updates, Phosphene. Rapid turnaround on patent application, top researchers working with MYMX, critical need vaccine development. And this patent approval comes AFTER management put this company on the market…!
Recombinant HIV compositions and uses thereof in animal models
Patent number: 11684666
Abstract: Provided here are certain recombinant HIV compositions and animal models to evaluate prophylactic and therapeutic antiviral compositions.
Type: Grant
Filed: November 7, 2017
Date of Patent: June 27, 2023
Assignee: University of Louisiana at Lafayette
Inventors: Ruth M. Ruprecht, Samir K. Lakhashe
https://patents.justia.com/inventor/ruth-m-ruprecht
I think it's going to be more than 10 cents. I read in one of the filings that the 786k potential issuable shares related to the convertible loans had an average conversion price of 11 cents. I would think they would want to make something for all their hard work. I'm thinking above 11 cents. Just a matter of how much more.
Insiders are holding a lot of shares here. Hopefully we profit with them.
Thoughts.. Yes a sell but a large amount of shares like that in my years of watching usually that means it was planned. Somebody collected those shares and will not be departing with them for awhile. The fact that MYMX kept the stock in play by keeping the the price at .01 or above is also no lucky play. Timing is well to planned. Stock was at .006 range for a long time and then all of a sudden it stays in the .01 to .02 range.. Several high and low amount of shares trading of late at both ends of the spectrum.
Wish full thinking but I think something is getting closer. Good or Bad of course I am hoping for good been here to long and hoping The Powers that Be in MYMX land are remembering the little guys who put so much faith in them and held this long.. The stockholder.. I would be happy with anything above .10 a share.
Sme buys but a lot of sold. One block of 448k sold at .0091
Wow almost 900k shares today. Is that someone offloading or someone collecting?
Appears someone doesn't want the stock price to rise just yet.. or someone is collecting shares any and all in anticipation of a big reveal...I like that thought pattern.. Big Reveal
Volume last 2 days Impressive. Any thoughts
I view that as a definite win for shareholders. We remain part of any deal that MYMX makes. They clearly are negotiating to receive value for decades of leadership, research, patents and collaborations in virosome development and its application to vaccines for major illnesses. We wait, and support their efforts with our loyalty.
Yesterday was the 10th day in a row that MYMX traded above .01. At least MYMX won't be delisted now. A small win.
They are paying massive amounts for soccer and golf stars, maybe they’d also like to pay for some best in class virosome IP.
Toon Stegmann. May 24, 2023.
Enhanced HPV16 E6/E7+ tumor eradication via induction of tumor-specific T cells by therapeutic vaccination with virosomes presenting synthetic long peptides.
https://link.springer.com/article/10.1007/s00262-023-03462-y
"These experiments demonstrate that virosomes with Amplivant-SLP and also virosomes with SLP coupled to lipid are suitable for use in therapeutic cancer vaccines, the latter if they also contain the adjuvants QS-21 and 3D-PHAD."
Virosome: An engineered virus for vaccine delivery
May 2023.
Saudi Pharmaceutical Journal
https://www.sciencedirect.com/science/article/pii/S1319016423000749?via%3Dihub
"Hence, due to increasing attention on alum-free antigenic immunization products virosomes are gaining significant consideration."
"The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this Research work through the project number ISP22-16."
Crossing fingers for a big reveal!
I have to agree, But I do like the fact that he hasn't tried to push the stock and the company to sell shares. My hope is of course all this silence has a cause. I would rather have the silence until the big reveal. Gets rid of a lot of baggage and people unwilling to hold and wait. Anytime you have a penny stock and your holding it your risk is large for a big win and large for a loss. Here is to Kempers plan for a big win for himself, the stockholders and the MYMX team... Maybe I'm putting to much faith in Kempers and MYMX team but time will tell.
Mr. Kempers keeps us informed. A bit.
Some time ago - I didn't notice when exactly - he added to his Linkedin profile "commercial CFO". And in the last few days he added "Exits".
Since I am not a native speaker I am wondering if "Exits" is linguistic closer related to "exitus" or "(monetary) ex(c)itement". Just kidding. But shouldn't it mean to put such words in one's Linkedin profile that one is extraordinarily good in such things...
At least that shows he is still alive.
Lol you are correct!!
Rum makes us numb....and right now with Mymetics it is a good thing.
Thanks castaways, I better stop with rum lol
See Phosphene’s message from yesterday; the published article is cited in it’s entirety at the end (just click on it)
Can someone post the publication? Tia
Great timing of this publication - thanks for sharing! I have wondered about MYMX’ silence regarding their COVID vaccine efforts…this affirms progress in another strand of that research.
Surfaces with antiviral properties
Watching viruses "burst”
June 15, 2023
Using a novel analytical method, Empa researchers in collaboration with Mymetics have tracked viruses as they pass through face masks and compared their inactivation on the filter layers of different mask types. The new method should now accelerate the development of surfaces that can specifically eliminate viruses [1].
https://analyticalscience.wiley.com/do/10.1002/was.0019000125/
As per Section 4.1 of the OTCQB Standards, the
company will be granted a cure period of 90 calendar days during which the closing bid price for the Company’s common stock must be $0.01 or greater
for ten consecutive trading days in order to continue trading on the OTCQB marketplace. If this requirement is not met by August 10, 2023, the company
will be removed from the OTCQB marketplace.
In addition, the Company has been informed that in the event that the Company’s closing bid price falls below $0.001 at any time for five consecutive
trading days, the Company will be immediately removed from OTCQB.
As per Section 4.1 of the OTCQB Standards, the
company will be granted a cure period of 90 calendar days during which the closing bid price for the Company’s common stock must be $0.01 or greater
for ten consecutive trading days in order to continue trading on the OTCQB marketplace. If this requirement is not met by August 10, 2023, the company
will be removed from the OTCQB marketplace.
In addition, the Company has been informed that in the event that the Company’s closing bid price falls below $0.001 at any time for five consecutive
trading days, the Company will be immediately removed from OTCQB.
Ha! I wish that were the case. But for now they need to keep above a cent for I think 30 days.
A great story would be mymx rises from 2 cents to 20 dollars a share!!
|
Followers
|
85
|
Posters
|
|
|
Posts (Today)
|
1
|
Posts (Total)
|
27660
|
|
Created
|
09/25/02
|
Type
|
Free
|
| Moderators | |||

http://www.mymetics.com/
COMPANY OVERVIEW
https://www.mymetics.com/files/8116/2081/0445/May21-Mymetics_Overview.pdf
Mymetics Corporation is US registered biotechnology company with its main offices in Switzerland and the Netherlands.
Focused on developing next generation preventative vaccines for infectious diseases.
Mymetics core technology and expertise are in the use of virosomes,
lipid-based carriers containing functional fusion viral proteins in combination with rationally designed antigens and membrane proteins.
Objective: "Build small / medium size innovative R&D virosome vaccine company with strong
partnerships, Phase II – III clinical vaccine pipeline and have optionality for M&A or sale."
Current Share Structure:
Outstanding Shares: 303.7 million (as of 01/18/2019 unchanged since 2014)
Floating Shares: 79.8 million* (a/o 01/18/19)
(https://www.otcmarkets.com/stock/MYMX/profile)
Rationale and Impact of MACIVIVA
With few exceptions, commercialized vaccines are generally delivered by injection through the intramuscular or subcutaneous route.
Vaccines contain immunogens classically found within a large variety of biological compounds such as peptides, proteins, glycoproteins and sometimes carbohydrates and lipids.
These immunogens may trigger the immune system for producing antibodies and/or cytotoxic T cells for preventing the pathogen transmission or blocking and/or slowing down the disease progression.
However, these vaccines generally exist as liquid formulation that are inherently prone to physical and/or chemical modifications. The cold chain storage is still fundamental for preserving the
bioactivity of most liquid and freeze-dried vaccines. For reconstituted freeze dried vaccines, they harbor important instability and must be used within hours and kept refrigerated. Vaccine degradation generally takes place
during shipment and/or storage of liquid or lyophilized products, which may affect the immunological properties of the immunogens, with unwanted immune responses or insufficient immune protection.
There is growing evidence that solid dosage formats (e.g. powder form) for vaccines may offer several advantages over the liquid formulations, such as the prevention of molecular motion and shear-induced degradation,
and slowing down modifications and degradation reactions involving water and oxygen radicals, resulting in improved stability, enhanced shelf-life of vaccines and greatly simplified logistics.
MYMETICS BV
Expertise: R&D on virosome formulations
Project responsability: Investigating and compiling the results about the physical and biochemical properties of the virosome-based vaccines obtained by spray-drying and lyophilization.
MYMETICS SA
Expertise : Non-GMP and GMP virosome production, clinical development
Project responsability: Excipient selection for liquid virosomes, supervising the non-GMP and GMP manufacturing of the liquid virosomes and development of analytical methods.
UPPERTON LIMITED
Expertise: Non-GMP and GMP Spray drying
Project responsability: Identification of excipients and experimental conditions suitable for virosome spray drying, production of non-GMP and GMP powder forms for nasal and oral delivery.
CATALENT U.K. SWINDON ZYDIS LIMITED
Expertise: Zydis technology for fast-dissolving tablet, world leader in drug formulation and distribution
Project responsability: Identification of excipients and experimental conditions suitable for virosome lyophylization, according to the Zydis technology, non-GMP and GMP tablets for sublingual delivery.
CHIMERA BIOTEC GMBH
Expertise: Ultra sensitive immunoassays development and bioanalysis based on Imperacer® (Immuno-PCR) technology.
Project responsability: Immunogenicity study in animals with spray-dried and lyophilized virosomes. Imperacer® immunoassay development and evaluation of the vaccine-induced antibody response.
BACHEM AG
Expertise: R&D, non-GMP and GMP manufacturing of API, world supplier
Project responsability: Process Development and manufacture of peptide P1, GMP-grade, including development and validation of analytical methods.

Vaccines are poorly accessible in developing countries
Vaccines require cold-chain storage and are often delivered by injection, which is undesirable, less safe and more expensive to administer.
Developing thermostable solid form vaccines through non-invasive routes may represent a long-term global solution to the vaccination challenge (Amorij, 2008).
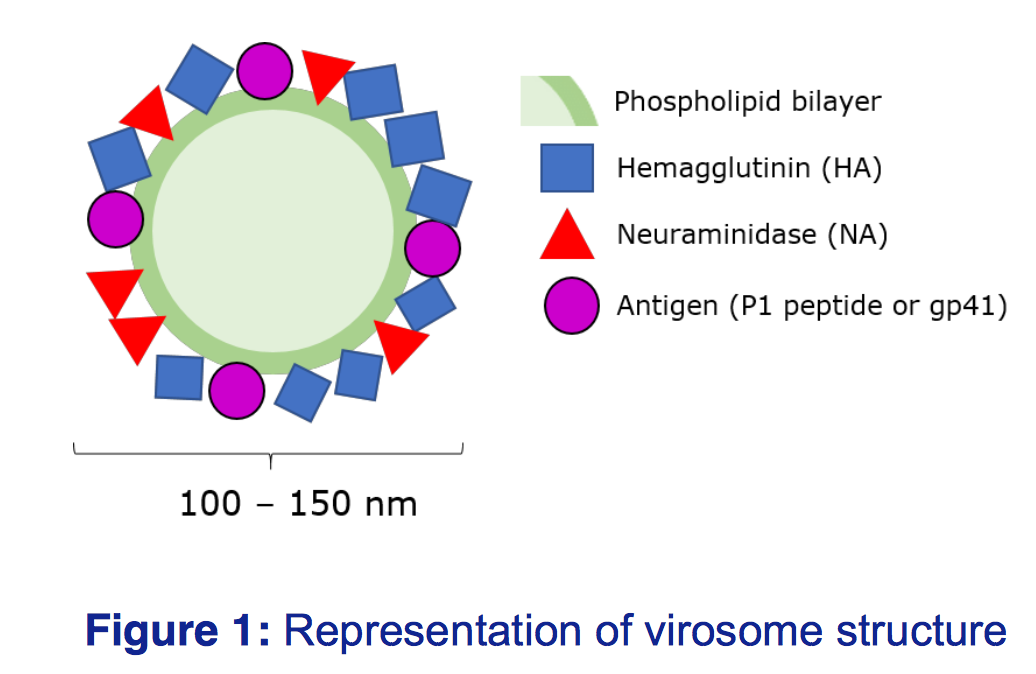
Virosomes are an efficient vaccine delivery system
Virosomes are spherical, unilamellar lipid-based carriers, intercalated with functional glycoproteins to reflect the natural virus, however the lack of viral RNA means there is no risk of infection
(Figure 1). Virosomes can be tagged with different antigens and adjuvants, meaning they can be tailored to target different viruses, and offer increased immunogenicity over inactivated viruses.
Currently, virosomal influenza vaccines are only available in liquid form (Amorij, 2008).
Spray drying can produce dry powders for a range of dosage forms, including inhaled or nasal drug delivery.
A dry powder is formed when a liquid feed solution or suspension is atomised using a spray nozzle, and rapidly dried using hot air. However, while the drying process is gentle due to evaporative cooling,
there is still the potential to stress and inactivate vaccine components. It has been found that subunit and live-attenuated vaccines (and other delicate molecules such as proteins)
can be protected during processing b by incorporating them in an amorphous sugar matrix, which also offers longer term stability during storage (Kanojia, 2016).
A method has been developed to produce a powder form of virosome based influenza vaccine using spray-drying.
Formulations have been optimised for oral and nasal delivery.



Virosomal technology is approved by the FDA for use in humans, and has a high safety profile
Virosomes are biodegradable, biocompatible, and non-toxic12
No disease-transmission risk
No autoimmunogenity or anaphylaxis10
Broadly applicable with almost all important drugs (anticancer drugs, proteins, peptides, nucleic acids, antibiotics, fungicides)
Enables drug delivery into the cytoplasm of target cell
Promotes fusion activity in the endolysosomal pathway
Protects drugs against degradation
Intellectual Property
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
