Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Merrimack Reports Ipsen Announcement of Approval By The US FDA of Onivyde® (Irinotecan Liposome Injection) Plus 5 Fluorouracil/Leucovorin and Oxaliplatin (NALIRIFOX) as a First-Line Treatment for Metastatic Pancreatic Ductal Adenocarcinoma (mPDAC) (2/13/24)
FDA Approval Triggers $225 Million Milestone Payment from Ipsen to Merrimack
Merrimack Expects to Hold Special Meeting of Stockholders to Approve Plan of Dissolution in May 2024
Following Approval of Plan of Dissolution, Merrimack Expects to Issue a Liquidating Dividend Currently Estimated to be Between $14.65 and $15.35 per Share in the Late Spring or Early Summer
CAMBRIDGE, Mass.--(BUSINESS WIRE)--Merrimack Pharmaceuticals, Inc. (Nasdaq: MACK) (“Merrimack” or the “Company”) reported that Ipsen, SA (“Ipsen”) issued a press release today announcing the U.S. Food and Drug Administration has approved the supplemental new drug application for Onivyde® (irinotecan liposome injection) plus 5 fluorouracil/leucovorin and oxaliplatin (NALIRIFOX) as a first-line treatment for people living with metastatic pancreatic ductal adenocarcinoma (mPDAC).
Under the terms of the 2017 Asset Purchase Agreement between Ipsen and Merrimack (the “Ipsen Agreement”), previously approved by Merrimack’s stockholders, this U.S. Food and Drug Administration approval triggers a $225 million milestone payment from Ipsen to Merrimack which is due on or before March 29, 2024.
“Today’s announcement is the culmination of nearly seven years of clinical trials and regulatory approval efforts by Ipsen following its 2017 purchase of the Onivyde technology from Merrimack. The Company has received several payments from the Ipsen Agreement including: a $575 million payment in 2017, a $5.7 Million working capital adjustment payment later that year, and a $5 million milestone payment in 2019” said Gary Crocker, Chairman of Merrimack’s Board of Directors. “The Board of Merrimack plans to hold a Special Meeting of Stockholders to approve a plan for a corporate dissolution and followed by a distribution of the proceeds to our stockholders from this new $225 million milestone payment, plus residual cash, net of corporate taxes and interest charges accruing under IRS installment sale rules, and other expenses.”
Merrimack’s Board of Directors has evaluated the likelihood of receiving additional milestone payments under the Ipsen Agreement and from the 2019 Agreement with Elevation Oncology (the “Elevation Agreement”) and has concluded that it is unlikely that any additional milestone payments from either agreement will become payable. Based on this analysis, Merrimack expects to hold a special meeting of stockholders by May of 2024 to seek approval for the adoption of a Plan of Dissolution including issuance of a liquidating dividend after the special meeting of stockholders. The Plan will include establishment of a liquidating trust for the benefit of stockholders in the unlikely event that Merrimack might receive any future milestone payments from Ipsen or Elevation Technology.
Stockholders subject to taxation in the United States who receive a liquidating distribution generally will recognize a capital gain or loss equal to the amount received for their shares over their adjusted basis in such shares if shares are held in a taxable account. The tax consequences of the plan of dissolution may vary depending upon the particular circumstances of the stockholder. We recommend that each stockholder consult their tax advisor regarding the U.S. federal income tax consequences of the Plan of Dissolution as well as the state, local and non-U.S. tax consequences.
Based on the information currently available, Merrimack management estimates that the aggregate amount of the liquidating dividend payable to stockholders will be in the range of between approximately $14.65 and $15.35 per share. These estimates are based on, among other things, the receipt of the $225M milestone from Ipsen, Merrimack’s current and projected balance of cash, cash equivalents and marketable securities, estimated federal and state taxes payable for 2024, interest payable to the federal government pursuant to Internal Revenue Code Section 453A and state governments pursuant to similar state tax provisions due on the receipt of the Ipsen milestone payment, proceeds from the expected exercise of stock options, and amounts that will need to be retained by the Company to fund close down expenses and the liquidating trust. The estimated liquidating dividend represents Company management’s estimate of the amount to be distributed to stockholders during the liquidation but does not represent the minimum or maximum distribution amount. The total actual amount of distributions, if any, could be higher or lower, or, although highly unlikely, there may not be any liquidating distributions at all. It is possible that any initial distribution could be followed in the future by further distributions if it is determined that any reserved amounts no longer need to be held by the Company. The Company cannot predict with certainty the amount of liquidating distributions to be paid to Merrimack’s stockholders.
About Merrimack
Merrimack Pharmaceuticals, Inc. is a biopharmaceutical company based in Cambridge, Massachusetts that is entitled to receive up to $225 million in contingent milestone payments related to its sale of Onivyde to Ipsen S.A. in April 2017. This milestone payment is payable by Ipsen on or before March 29, 2024 as Ipsen has received FDA approval on February 13, 2024 for the use of Onivyde® plus 5 fluorouracil/leucovorin and oxaliplatin (NALIRIFOX regimen) as a treatment of first line metastatic pancreatic ductal adenocarcinoma (mPDAC).
Ipsen has not publicly communicated any current clinical trials which could lead to future milestone payments under the Ipsen Agreement. Elevation Oncology previously announced that it has paused activities under the program from which future milestone payments might be issued to Merrimack under the Elevation Agreement. As a result, Merrimack is not currently expecting to receive these additional potential milestone payments. Neither Ipsen nor Elevation Oncology share any non-public information regarding these programs with Merrimack.
Merrimack Reports Second Quarter 2023 Financial Results (8/03/23)
Cambridge, MA, August 3, 2023 /Business Wire/ – Merrimack Pharmaceuticals, Inc. (Nasdaq: MACK) [(“Merrimack” or the “Company”)] today announced its second quarter 2023 financial results for the period ended June 30, 2023.
“We were excited to see the announcement from Ipsen that the U.S. Food and Drug Administration (FDA) had accepted its supplemental new drug application (sNDA) Onivyde® (irinotecan liposome injection) plus 5 fluorouracil/leucovorin and oxaliplatin (NALIRIFOX regimen) as a potential first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC) and that the FDA had provided a Prescription Drug User Fee Act goal date of 13 February 2024 for review of the application” said Gary Crocker, Chairman of Merrimack’s Board of Directors. “We will continue to monitor the progress of this program which, if approved, would entitle Merrimack to a $225 million milestone payment from Ipsen.”
Second Quarter 2023 Financial Results
Merrimack reported a net loss of $391 thousand for the second quarter ended June 30, 2023, or $0.03 per basic and diluted share on a fully diluted basis, compared to a net loss of $478 thousand, or $0.04 per basic and diluted share on a fully diluted basis, for the same period in 2022.
Interest income in the second quarter ended June 30, 2023, was $178 thousand compared to $8 thousand for the same period in 2022.
General and administrative expenses for the second quarter ended June 30, 2023, were $569 thousand, compared to $486 thousand for the same period in 2022.
As of June 30, 2023, Merrimack had cash, cash equivalents and short term investments of $19.0 million, compared to $19.4 million as of December 31, 2022.
As of June 30, 2023, Merrimack had 14.3 million shares of common stock outstanding.
Updates on Programs Underlying Potential Milestone Payments
Ipsen
Metastatic Pancreatic Ductal Adenocarcinoma
- In November 2022, Ipsen announced the Phase III NAPOLI 3 trial of Onivyde (irinotecan liposome injection) plus 5-fluorouracil/leucovorin and oxaliplatin (the “NALIRIFOX regimen”) met its primary endpoint demonstrating clinically meaningful and statistically significant improvement in overall survival compared to nab-paclitaxel plus gemcitabine in 770 previously untreated patients with mPDAC and key secondary efficacy outcome of progression-free survival (PFS) also showed significant improvement over the comparator arm. Ipsen also announced that the safety profile of Onivyde in the NAPOLI 3 trial was consistent with those observed in the previous phase I/II mPDAC study.
- In January 2023, Ipsen presented clinical trial results at the 2023 American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium.
- In June 2023, Ipsen announced that the U.S. Food and Drug Administration (FDA) had accepted its supplemental new drug application (sNDA) Onivyde® (irinotecan liposome injection) plus 5 fluorouracil/leucovorin and oxaliplatin (NALIRIFOX regimen) as a potential first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC) and that the FDA had provided a Prescription Drug User Fee Act goal date of 13 February 2024 for review of the application.
Small Cell Lung Cancer
- In August 2022, Ipsen announced that the Phase III RESILIENT trial did not meet its primary endpoint of overall survival compared to topotecan. The trial is evaluating Onivyde versus topotecan in patients with small cell lung cancer, who have progressed on or after platinum-based first-line therapy treatment. In the announcement, Ipsen indicated that detailed results from the RESILIENT trial would be presented at an upcoming medical conference. The analysis concluded that the primary endpoint overall survival was not met in patients treated with Onivyde versus topotecan. However, a doubling of the secondary endpoint of objective response rate in favor of Onivyde was observed. In the August 2022 announcement, Ipsen reported that the clinical study results would be communicated with the regulatory agency. Ipsen indicated that while the results from the analysis of the RESILIENT trial have not demonstrated an overall survival benefit with Onivyde in patients in second-line small cell lung cancer, Ipsen intends to analyze the data further before decisions regarding next steps are made.
- To date, there have been no further announcements by Ipsen regarding these matters and it remains unclear as to whether Ipsen will continue to seek approval for the use of Onivyde in the small cell lung cancer application. If Ipsen elects not to proceed with seeking regulatory approval, or if regulatory approval is not obtained, Merrimack would not be entitled to the $150 million milestone payment tied to FDA approval of Onivyde for treatment of small cell lung cancer.
Elevation Oncology
- In January 2023, Elevation announced it is pausing further investment in the clinical development of seribantumab and intends to pursue further development only in collaboration with a partner. If Elevation elects not to proceed with seeking regulatory approval, or if regulatory approval is not obtained, Merrimack would not be entitled to the $54.5 million in additional potential development, regulatory approval and commercial-based milestone payments.
About Merrimack
Merrimack Pharmaceuticals, Inc. is a biopharmaceutical company based in Cambridge, Massachusetts that is entitled to receive up to $450.0 million in contingent milestone payments related to its sale of ONIVYDE® to Ipsen S.A. in April 2017. These milestone payments would be payable by Ipsen upon approval by the U.S. Food and Drug Administration (“FDA”) of ONIVYDE for certain additional clinical indications. ONIVYDE® is already approved by the FDA in combination with fluorouracil (5-FU) and leucovorin (LV) for the treatment of patients with metastatic adenocarcinoma of the pancreas after disease progression following gemcitabine-based therapy. This existing approval is unrelated to any future potential milestone payments. Merrimack’s agreement with Ipsen does not require Ipsen to provide Merrimack with any information on the progress of ONIVYDE clinical trials that is not publicly available. Merrimack is also entitled to receive up to $54.5 million in contingent milestone payments related to its sale of anti-HER3 programs to Elevation Oncology (formerly 14ner Oncology, Inc.) in July 2019.
Closed $12.80 aftermarket. up 3% and just under HOD of $12.82.
Holding perfect.
Very little profit taking. She could take off again. :)
If I am awake, I begin looking at 1 AM PST but defiantly by 4 AM PST.
Hey buddy! Totally agree, I will be up at 5 am tomorrow looking for another. Really best to look early for these runners!
I had to leave a bit early today but yes what a premarket then AM run especially. Even the dips were great to add as we added another dollar after that. Definitely the best play today.
Not a bad day's pay! GLTY
Not a bad day's pay! GLTY
Not a bad day's pay! GLTY
Yeah know his theory’s… indeed very interesting. But German. Germans and Austrians are not to far away of the way they think… good once as bad once… who ever is good or bad? You have to decide haha have a good one
THANKS!! (Austria?? In passing, G.W.F. HEGEL was BEST philosopher in the History of Mankind!! Identical theory as Darwin's!! Only difference, the CONTENT thereof!!)
That’s how it is… not to sure about the mall thing, as I’m from Austria haha having some friends here now. Have a nice day buddy
EXACTLY!! Simply launch your buy, then go shopping at Westfield Mall; at worst, you only lose a little, but can score a bundle!
Yeah I do like the tool of a sl… just never fell in love with a stock :)
Very SMART way to do it, Dude! (That stop-loss.)
Hoping for a squeeze… but as I did say before, worried about market and Algos are taking it … let’s see. I have a stop 5% below my entry. But looking good
MACK: 1K shares, or $1K bucks worth, Bro??? (This is the ONLY act in town today, as we all know.)
One man show here haha just bought 1K for a gamble. But news seem to be sweet. But scared that the over all market will take quiet a dive
MACK: I feels like old John-The-Baptist!!! Screaming to the World, but nobody listening!!! GEEEEEEZUS J. KRISTOOOOO!!! Bottom line, Peeps: You can pray to GOD for a Pancreatic Cancer CURE; or, see what MACK Company has up-their-sleeves!!

(LIVE vid., above, of MY 'Personal Financial Planner & Stocks Prognosticator'.)
MACK: The ONLY stock holding STRONG on this DISMAL Market's day!!! BIG BUYS coming in, minute-to-minute!! This Company is ATTACKING Metastatic Pancreatic Cancer, Peeps!!! That beast of a disease that took-out STEVE JOBS!! This ain't NO kids-stuff "Metaverse" crap; nor "NFTs" silliness; nor soaring in price because they just got SUED, or INVESTIGATED, or merely taking a couple months off simply to PONDER their next move!!! This Firm is trying to keep YOU & ME ALIVE!!!!
MACK: Look out, old 'MACKY' is back!!! "When that Shark bites, with those teeth babe, scarlet rivers begin to spread!!" (Well, I hope the PRICE here don't drop to 'scarlet' colors!!! I HATE when THAT happens!!!)

"Now listen-up, all California SURFERS!! When that SHARK bites, with those TEETH, babe!! You better be headin' to the SHORE like you had an Evenrude 400 Horsepower outboard strapped to your ASS!!!"
MACK: Maybe the late Steve Jobs wishes he had THIS drug, right Subs???
MACK: Rollin' like a 'Mack Truck'!!! WHAT?????
MACK Looking like a runaway freight train
Nice action today! I wonder if there is news out from Ipsen that might indicate there is a payment coming soon to MACK, and thus us shareholders?
Merrimack sold its oncology portfolio to Ipsen in April 2017 for $575M in upfront cash. MACK is eligible to receive up to an additional $450M triggered by the approval of additional indications for Onivyde in the U.S., a bullish prospect considering its current market cap of ~$77M.
https://seekingalpha.com/news/3640924-merrimack-pharma-jumps-42-on-bullish-timeline-for-potential-onivyde-milestone-payment
Alright so we aren’t looking at any exponential gains until 2022
Can you post that full link? When are they expected to receive potential cash?
If you receive that amount of money, you will explode like $ KDAK, $ 46 in 3 days
Thank you. That is basically the info I had found. Just wondering if a potential dividend payment to is shareholders is imminent or if we will still be waiting another year or so for one.
Did u see the options on this ... insane 3,100% closed up on 1
$MACK:Merrimack sold its oncology portfolio to Ipsen in April 2017 for
$575M
in upfront cash. MACK is eligible to receive up to an additional
$450M
triggered by the approval of additional indications for Onivyde in the U.S., a bullish prospect considering its current market cap of ~
$77M
seekingalpha.com/news/36409...
Merrimack Pharmaceuticals, Inc. (NASDAQ:MACK) was the recipient of some unusual options trading activity on Wednesday. Stock investors purchased 8,239 call options on the company. This is an increase of 5,115% compared to the average daily volume of 158 call options.
In related news, Director Noah G. Levy bought 69,087 shares of the stock in a transaction dated Tuesday, November 24th. The stock was purchased at an average cost of $3.65 per share, with a total value of $252,167.55. The transaction was disclosed in a legal filing with the Securities & Exchange Commission, which is available at the SEC website. 21.60% of the stock is currently owned by insiders.Several institutional investors and hedge funds have recently modified their holdings of MACK. Private Advisor Group LLC purchased a new stake in Merrimack Pharmaceuticals in the second quarter worth about $25,000. LVW Advisors LLC purchased a new stake in Merrimack Pharmaceuticals in the third quarter worth about $80,000. Bronte Capital Management Pty Ltd. purchased a new stake in Merrimack Pharmaceuticals in the third quarter worth about $2,052,000. Nantahala Capital Management LLC lifted its stake in Merrimack Pharmaceuticals by 0.6% in the third quarter. Nantahala Capital Management LLC now owns 751,943 shares of the biopharmaceutical company’s stock worth $3,008,000 after acquiring an additional 4,308 shares during the period. Finally, Western Standard LLC lifted its stake in Merrimack Pharmaceuticals by 3.1% in the second quarter. Western Standard LLC now owns 869,354 shares of the biopharmaceutical company’s stock worth $2,895,000 after acquiring an additional 26,441 shares during the period. 45.83% of the stock is owned by hedge funds and other institutional investors.
Shares of MACK traded up $2.41 during trading hours on Wednesday, reaching $6.49. 77,082,195 shares of the company traded hands, compared to its average volume of 370,655. Merrimack Pharmaceuticals has a 12-month low of $1.49 and a 12-month high of $7.10. The stock’s fifty day moving average price is $3.83 and its 200 day moving average price is $3.66.
https://www.americanbankingnews.com/2020/12/02/merrimack-pharmaceuticals-sees-unusually-high-options-volume-nasdaqmack.html
* * $MACK Video Chart 12-02-2020 * *
Link to Video - click here to watch the technical chart video
Unless I found the wrong one, all I gathered was that someone bought a lot of shares???
Cool, thank you for the info! Will have to check it out. Filed by Merrimack or Ipsen?
SEC filing 13D > out an hour ago!
Anybody have an idea regarding the price movement today? I glanced at the Ipsen press release, but didn’t see anything that indicated a payment to Merrimack would be imminent. I definitely could have missed something in there though.
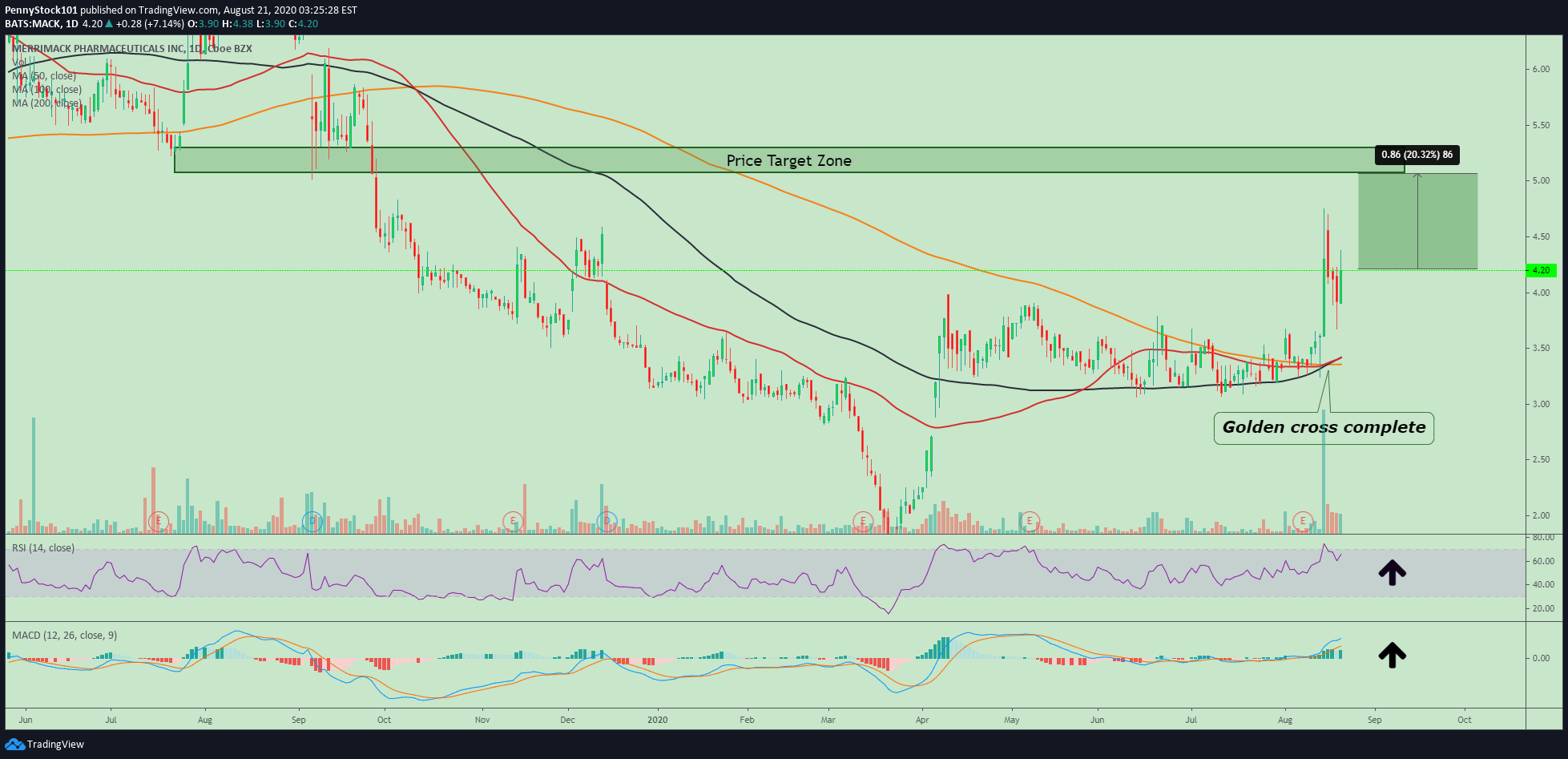
$MACK | #Merrimack #Pharmaceuticals #GoldenCross
Merrimack Pharmaceuticals , Inc. is a clinical stage biopharmaceutical company, which engages in discovering, developing, and commercializing medicines consisting of novel therapeutics paired with diagnostics for the treatment of cancer. Its pipeline includes MM-131, MM-141, and MM-310. The company was founded by Anthony J. Sinskey, Gavin MacBeath, and Ulrik B. Nielsen in 1993 and is headquartered in Cambridge, MA.
News: $MACK Merrimack Pharmaceuticals Adopts Section 382 Net Operating Loss Rights Plan
Merrimack Pharmaceuticals, Inc. (Nasdaq: MACK) (the “ Company ” or “ Merrimack ”), announced today that its Board of Directors (the “ Board ”) has adopted a Section 382 net operating loss rights plan (the “ Plan ”) and declared a dividen...
Find out more MACK - Merrimack Pharmaceuticals Adopts Section 382 Net Operating Loss Rights Plan
|
Followers
|
51
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
894
|
|
Created
|
03/29/12
|
Type
|
Free
|
| Moderators | |||

Robert J. Mulroy has served as our President and Chief Executive Officer and a member of our board of directors since May 1999. Prior to joining us, Mr. Mulroy worked as a management consultant in the pharmaceutical and healthcare industries. Mr. Mulroy has also worked as a consultant in the field of international development and has served as an advisor to multiple start-up companies in the biotechnology industry. Mr. Mulroy holds a master’s degree in public and private management from Yale University and a BA from Stanford University.
Yasir B. Al-Wakeel, BM BCh, has served as our Chief Financial Officer and Head of Corporate Development since August 2015. Dr. Al-Wakeel previously served in various capacities at Credit Suisse, an investment bank, from January 2008 to June 2015. While at Credit Suisse, Dr. Al-Wakeel was most recently a Director of Healthcare Investment Banking focused on biotechnology and, prior to that role, he was an Equity Research Analyst covering the biotechnology and specialty pharmaceuticals sectors. Before joining Credit Suisse, Dr. Al-Wakeel was a practicing physician, holding both clinical and academic medical posts. Dr. Al-Wakeel holds a BM BCh (Doctor of Medicine) from Oxford University, an MA in theology from Cambridge University and a BA from Cambridge University.
Peter N. Laivins has served as our Head of Development since May 2014. Mr. Laivins served as our Vice President of Clinical Development from October 2011 to May 2014. Previously, Mr. Laivins ran an independent consulting practice specializing in oncology and neuroscience from January 2010 to October 2011. Prior to that, Mr. Laivins served as Vice President of Strategic Marketing at Elan Corporation, plc, a biopharmaceutical company, from 2006 to January 2010. Mr. Laivins began his career at Pfizer Canada, joined Pfizer International in New York in 1995 and held positions of increasing responsibility at Pfizer Inc., culminating as Group Leader of Pfizer US Oncology Marketing until 2006. Mr. Laivins holds an MBA and a BS from McGill University.
Gavin MacBeath, PhD, is a founder of Merrimack and Head of Translational Medicine. Dr. MacBeath leads translational research and drives our innovative diagnostic programs, helping to ensure we get the right drug to the right patient at the right time. He is also a founder of Chestnut Pharmaceuticals, serves on the Scientific Advisory Board of Aushon Biosciences and is a Lecturer and Principal Investigator at Harvard Medical School. He joined Merrimack in 2009 after serving on the faculty in the Department of Chemistry and Chemical Biology at Harvard University for eight years where he focused on developing and applying protein microarray technology to facilitate basic research in the area of systems biology. Dr. MacBeath holds a Bachelors of Science from University of Manitoba, Canada, and a PhD in Macromolecular and Cellular Structure and Chemistry from The Scripps Research Institute in La Jolla, California.
William M. McClements has served as our Head of Corporate Operations since September 2011. Previously, Mr. McClements served as Chief Human Resources Officer of Integreon Managed Solutions, Inc., a global research and business services company, from May 2010 to September 2011. Prior to that, Mr. McClements served as Chief Operating Officer and a partner at Monitor Group, a global strategic advisory firm, where he worked from 1987 to May 2010. From September 2009 to March 2010, Mr. McClements also served as Acting President of Be the Change, Inc., a nonprofit focused on creating national issue-based campaigns. Mr. McClements holds an MBA from Harvard Business School and a BA from Williams College.
Birgit M. Schoeberl, PhD, has served as our Head of Discovery since January 2015. Dr. Schoeberl served as our Senior Vice President of Research from January 2014 to January 2015, as our Vice President of Research from October 2009 to January 2014 and in various other research roles since she joined us in 2003. Prior to joining us, Dr. Schoeberl was a post-doctoral fellow at the Massachusetts Institute of Technology, where she built some of the earliest computational models of signaling networks. Dr. Schoeberl holds a PhD in systems biology from the Max Planck Institute for Dynamics of Complex Technical Systems in Germany and an MS in chemical engineering from the University of Karlsruhe in Germany.
Edward J. Stewart has served as our Head of Commercial since December 2011. Mr. Stewart served as our Senior Vice President of Business Development from March 2009 to December 2011 and in various other business development roles since he joined us in 2001. Mr. Stewart began his career at KPMG Peat Marwick LLP in the life sciences strategy consulting group. Mr. Stewart holds an MBA from the Johnson Graduate School of Management at Cornell University and a BS from Bates College.
William A. Sullivan has served as our Head of Finance and Accounting since August 2015 and our Treasurer since February 2010. Mr. Sullivan served as our Chief Financial Officer from May 2011 to August 2015, as our Vice President of Finance from February 2010 to May 2011, and as our Controller from November 2007 to February 2010. Previously, Mr. Sullivan served as Corporate Controller of Vette Corp., a thermal management solutions company, from 2004 to 2007. Mr. Sullivan began his career at Arthur Andersen LLP, where he obtained his certified public accountant license. Mr. Sullivan holds an MBA and an MS in accounting from Northeastern University’s Graduate School of Professional Accounting and a BA from Williams College.
One Kendall Square
Building 700, Suite B7201
Cambridge, MA 02139
Phone: 617-441-1000
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
