Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Website new owner Medisun http://www.medisun.hk/index.php?route=common/home
8k out
Entry into a Material Definitive Agreement.
On April 18, 2017, Accurexa Inc. (the “Company”) entered into a license agreement under which Medisun Holdings Limited (“Medisun”) granted a non-exclusive licence (the “Licence”) (including access to Medisun’s clinical network facilities in the Greater China) to use Medisun’s NK (Natural Killer) cell technology to Accurexa for 10 years (the “Term”) in the United States and the Greater China (People’s Republic of China, Hong Kong, the Macau Special Administrative Region and Taiwan). Medisun has developed its NK cell technology for the treatment of cancer. The NK cell technology is currently clinically used to treat cancer patients at Medisun’s network clinical facilities. The Licence includes Medisun’s provision of relevant technical advice and support from time to time required by Accurexa during the Term. In exchange for the License, Accurexa issued 10,000,000 shares of the Company’s common stock to Medisun. These shares were issued pursuant to Regulation D under the Securities Act of 1933, as amended, are exempt from registration by reason of Section 4(2) of the Securities Act of 1933, as amended (the “Act”), and bear an appropriate restrictive legend.
Item 5.02
Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers.
Resignations
On April 18, 2017, Bryan Lee, PhD resigned as President & Chief Executive Officer, and Director of the Company’s Board, effective immediately. Mr. Lee’s resignation does not arise from any disagreement on any matter relating to the Company’s operations, policies or practices, or regarding the general direction of the Company.
On April 18, 2017, Mr. Stefan Moll resigned as Chief Financial Officer, effective immediately. Mr. Moll’s resignation does not arise from any disagreement on any matter relating to the Company’s operations, policies or practices, or regarding the general direction of the Company.
On April 18, 2017, Mr. Oliver Jackson resigned as Director of the Company’s Board, effective immediately. Mr. Jackson’s resignation does not arise from any disagreement on any matter relating to the Company’s operations, policies or practices, or regarding the general direction of the Company.
Appointments
On April 18, 2017, the Board of Directors of the Company appointed Ms. Sophia Yaqi Sun as President & Chief Executive Officer, Chief Financial Officer, and Director of the Company’s Board, effective immediately.
Ms. Sun (age 35) is the Senior Vice President of Medisun Holdings Limited (“Medisun”, together with its subsidiaries and its affiliated companies, collectively, the “Medisun Group”), and the Vice President of National Investments Fund Limited (“NIF”). The Medisun Group is a group of companies principally engaged in medical and healthcare related businesses, and is committed to stem cell regenerative and precision medicine industry investments and the commercialization of regenerative and precision medicine technologies. NIF is a company incorporated in the Cayman Islands, the shares of which are listed on the Main Board of The Stock Exchange of Hong Kong Limited. Prior to joining Medisun and NIF, Ms. Sun has worked for Ernest & Young LLP in Beijing and San Francisco, leading audit teams in financial auditing, bad debt evaluation, merger acquisition due diligence for commercial banks. Ms. Sun has obtained her Bachelor’s degree at University of International Business and Economics in the People’s Republic of China, and her Master of Business Administration degree at the University of Illinois.
On April 18, 2017, the Board of Directors of the Company appointed Ms. Lisha Huang as Director of the Company’s Board, effective immediately.
Ms. Huang (age 57) has been a Director of Medisun Holdings Limited and Medisun Group companies since 2014. Prior, Ms. Huang was an Excutive Director of New Sakai Hong Kong Limited, a company incorporated in Hong Kong.
Looks like maybe some more downside. Wait & see.
This company has some possibilities but the notes are numerous and one toxic. Does anyone know the current state of the warrants/notes?
"The Holder has the right to convert the Note, in whole or in part, into shares of common stock of the Company (“Common Stock”) at a "Variable Conversion Price" of 50% multiplied by the Market Price (representing a discount rate of 50%)."
I agree with you there.. They will be Featured on FOX Business Network's "NEW TO THE STREET" on 10/23... This should be great exposure for ACXA !
https://finance.yahoo.com/news/streets-october-line-broadcast-fox-182437825.html
Check out this nice Collaboration between DMPI and ACXA
$ACXA ~ DelMar Pharmaceuticals (Nasdaq - DMPI) and Accurexa to Collaborate
In the Development of a Novel Combination Chemotherapy for the Local Treatment of Brain Cancer
http://ir.delmarpharma.com/press-releases/detail/830/delmar-pharmaceuticals-and-accurexa-to-collaborate-in-the
Company Presentation ~ Accurexa, Inc.
Presented by: Daniel Lim, M.D., Ph.D., Assistant Professor of Neurological Surgery, University of California, San Francisco. Accurexa is developing a medical stereotactic device, which the Company believes has the potential to significantly improve the delivery of stem cells into the human brain and enhance the efficacy of a wide range of stem cell therapeutics.
I agree my friend with volume this one can go up easily!
$ACXA
It looks like ACXA can come alive here once a little volume develops. Chart looks nice
The Company is focused on developing novel neurological therapies to be directly delivered into specific regions of the brain. It is developing its ACX-31 program to deliver temozolomide as an adjunct to BCNU, locally to brain tumor sites. The Company is collaborating in the development of its ACX-31 program with Prof. Henry Brem who built one of the largest brain tumor research and treatment centers in the world at Johns Hopkins University. Prof. Robert Langer, who is the David H. Koch Institute Professor at MIT and the most cited engineer in history, is also advising the Company in the development of its ACX-31 program. Oral temozolomide is a generic, FDA approved, first-line chemotherapy drug that is indicated for the treatment of adult patients with newly diagnosed glioblastoma multiforme concomitantly with radiotherapy and then as maintenance treatment. Before oral temozolomide became generic, it generated US sales of approximately $420 million and global sales of approximately $910 million under its brand name Temodar(R) in 2012. Additional information about the Company may be found on its website, www.accurexa.com.
$ACXA
Take a look at the Key Stats for $ACXA: https://ycharts.com/companies/ACXA
Volatility is increasing here on ACXA. Let's see a green finish to the day!
About ACXA's partner DelMar Pharmaceuticals, Inc. (Nasdaq - DMPI)
DelMar Pharmaceuticals was founded to develop and commercialize proven cancer therapies in new orphan drug indications where patients are failing or have become intolerable to modern targeted or biologic treatments. http://ir.delmarpharma.com/press-releases/detail/830/delmar-pharmaceuticals-and-accurexa-to-collaborate-in-the
$1.00 just hit for 100 shares! Above the ask........interesting!
$ACXA
good to see some action here. I am looking forward to the next ACXA press release
+5.88 at the .90 HOD and asking for a new HOD at $1.00
This one is super thin on the Ask! ~ Any sizable buying could send this one flying ![]()
http://www.barchart.com/quotes/stocks/ACXA
I am looking forward to it too! Nice gains this morning! We could hit a $1 easily here...very thin!
$ACXA
just got done doing some deep DD on ACXA. I am looking forward to the Fox interview!
Have you seen this interview with the CEO & President of ACXA?
You can learn a lot about their BranchPoint neurological therapy product here!
The Company is focused on developing novel neurological therapies to be directly delivered into specific regions of the brain. It is developing its ACX-31 program to deliver temozolomide as an adjunct to BCNU, locally to brain tumor sites. The Company is collaborating in the development of its ACX-31 program with Prof. Henry Brem who built one of the largest brain tumor research and treatment centers in the world at Johns Hopkins University. Prof. Robert Langer, who is the David H. Koch Institute Professor at MIT and the most cited engineer in history, is also advising the Company in the development of its ACX-31 program. Oral temozolomide is a generic, FDA approved, first-line chemotherapy drug that is indicated for the treatment of adult patients with newly diagnosed glioblastoma multiforme concomitantly with radiotherapy and then as maintenance treatment. Before oral temozolomide became generic, it generated US sales of approximately $420 million and global sales of approximately $910 million under its brand name Temodar(R) in 2012. Additional information about the Company may be found on its website, www.accurexa.com.
$ACXA
This one looks like an up and coming stock. With the interview coming mid month I would imagine the interest increasing for ACXA
DelMar Pharmaceuticals (Nasdaq - DMPI) and Accurexa to Collaborate
In the Development of a Novel Combination Chemotherapy for the Local Treatment of Brain Cancer
http://ir.delmarpharma.com/press-releases/detail/830/delmar-pharmaceuticals-and-accurexa-to-collaborate-in-the
Dont miss this interview with ACXA guys.. "NEW TO THE STREET's" Anchor, Ken Evseroff, kicks off the show interviewing George Yu, CEO of Accurexa, Inc. ( $ACXA ). http://finance.yahoo.com/news/streets-october-line-broadcast-fox-182437825.html
Barchart.com upgraded the daily chart to a 80% Strong BUY after the close!
ON Radar for a Blue Sky Breakout ![]() http://www.barchart.com/quotes/stocks/ACXA
http://www.barchart.com/quotes/stocks/ACXA
This interview is going to be huge..want to be in before the interview I am thinking ! "NEW TO THE STREET's" October Line-Up Broadcast on FOX Business -- Accurexa (ACXA) Oakridge Global Energy Solutions (OGES), Fundamental Applications Corp. (FUAPF), International Western Petroleum (INWP) and PetProducts.com Corp.
http://www.otcmarkets.com/stock/ACXA/news/-quot-NEW-TO-THE-STREET-s-quot--October-Line-Up-Broadcast-on-FOX-Business----Accurexa--ACXA--Oakridge-Global-Energy-Solutions--OGES---Fundamental-Applications-Corp---FUAPF---International-Western-Petroleum--INWP--and-PetProducts-com-Corp-?id=141801&b=y
$ACXA
hey guys good morning. This is looking like an ice OTCQB here with ACXA
56% Strong Buy rating on the daily chart from Barchart.com today!
http://www.barchart.com/quotes/stocks/ACXA
looking very interesting here. I just did some DD on ACXA.. they are to be featured in a Fox business interview
Check out the information on this link, great information:
Trader's Cheat Sheet: http://www.barchart.com/cheatsheet.php?sym=ACXA
That it is my friend I am glad to see that!
http://www.barchart.com/opinions/stocks/ACXA
ACXA slowly been going up
http://stockcharts.com/h-sc/ui?s=ACXA
"NEW TO THE STREET's" October Line-Up Broadcast on FOX Business -- Accurexa (ACXA) Oakridge Global Energy Solutions (OGES), Fundamental Applications Corp. (FUAPF), International Western Petroleum (INWP) and PetProducts.com Corp.
October 3, 2016
NEW YORK, NY--(Marketwired - October 03, 2016) - "NEW TO THE STREET" announces its Sunday, October 23, 2016, at 1:30 PM show. Broadcasting on FOX Business Network, "NEW TO THE STREET" interviews Accurexa, Inc. (ACXA), Oakridge Global Energy Solutions, Inc. (OGES), Fundamental Application Corp. (FUAPF), International Western Petroleum (INWP) and Pet products.Com, Corp.
"NEW TO THE STREET's" Anchor, Ken Evseroff, kicks off the show interviewing George Yu, CEO of Accurexa, Inc. (ACXA). George will give a brief overview of the company and then talk about the Corporation's recent partnership with DelMar Pharmaceuticals, and how together Accurexa's technology and DelMar's drug can be a winning combination for patients.
George Yu states, "We are excited to be featured on Fox Business, and to describe how our ACXA focuses on delivering new therapies directly to the brain with novel technologies such as implantable, biodegradable wafers. Our technologies not only enabled the development of our ACX-31 brain cancer program and our collaboration with DelMar Pharmaceuticals, but also the development of stem cell therapies that directly delivered for the treatment of Parkinson's Disease
Hmm glad to hear that in a way, I was actually thinking possibly considering entry at around 40 or less, if a promoter is touting it it apparently is time to start watching this closer in case it pops a little
There is a stock promoter making phone calls about this trying to get people to take positions no higher than 0.75/share - got a call about it a couple weeks ago. The company is called investor market preview, I got on their call list recently - I have had 5 tips from them in the past year and all of them have been bad tips just a heads up.
ACXA up 20 % @ .75 light volume 52 week high of 1.64
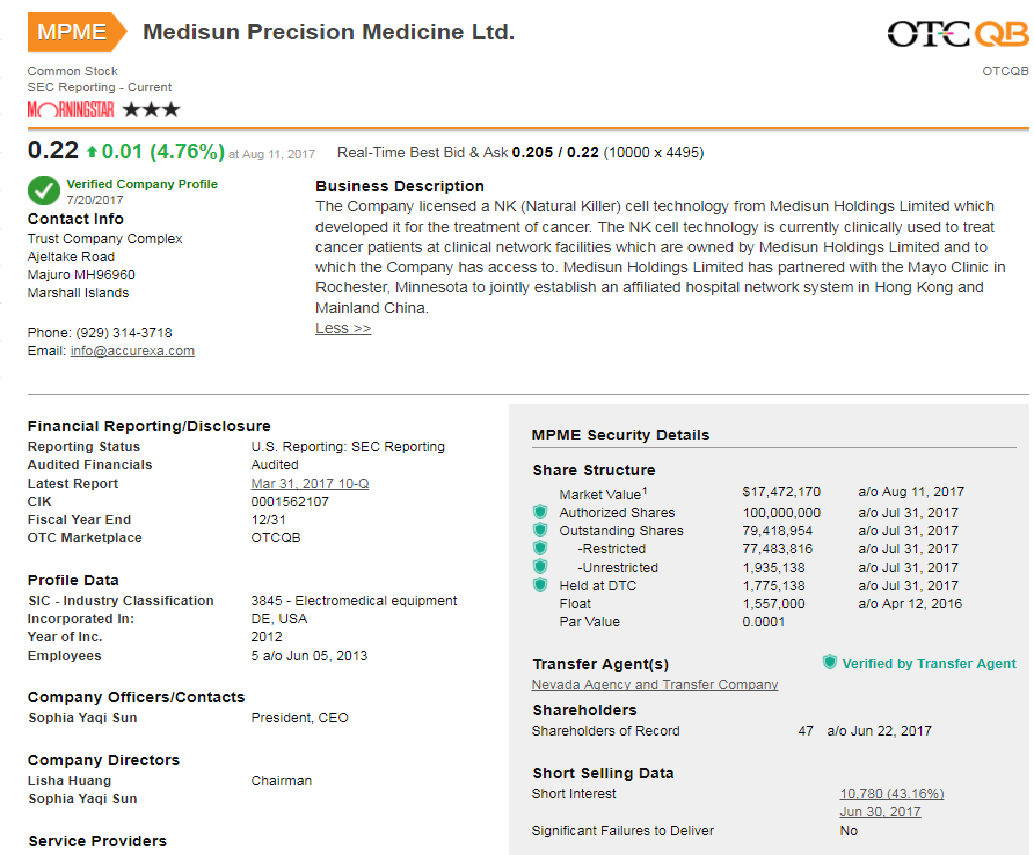
ACXA Security Details
Share Structure
Market Value1 $6,380,667 a/o Sep 23, 2016
Authorized Shares 20,000,000 a/o Aug 12, 2016
Outstanding Shares 9,180,816 a/o Aug 12, 2016
-Restricted Not Available
-Unrestricted Not Available
Held at DTC Not Available
Float 1,557,000 a/o Apr 12, 2016
<<The convertible preferred stock is convertible into shares of common stock of the Company at a conversion price of $1.25 per share.>>
in the 10Q. Does this mean a one to one conversion? The par value is .0001 per share but after trying to understand what that means, I still don't.
<<On June 22, 2015, we closed a private placement of 2,250 shares of the Company's convertible preferred stock for gross proceeds to the Company of $2,250,000 and net proceeds of $1,993,500.>>
How does the .0001 relate to 2,250 and $2,250,000 or does it?
I appreciate any clarification.
The Chances of Surviving Brain Cancer are Minimal.
Just came across a great article on Dr. Henry Brem (from 2005), Director of the Department of Neurosurgery, and Neurosurgeon-in-Chief at The Johns Hopkins University.
Accurexa (ACXA) recently announced it had entered into an exclusive license agreement which allows the company to collaborate with Prof. Brem's outstanding team at Johns Hopkins University - with regards to a composition for treating an individual with a solid tumor comprising a combination of BCNU and temozolomide.
BCNU (carmustine) is a chemotherapy drug that is contained in Gliadel(R), a biodegradable polymer that is implanted locally into the resection cavity after surgical removal of a brain tumor and is indicated for the treatment of newly diagnosed and recurrent GBM.
Temozolomide is a now a generic, approved chemotherapy drug that is indicated for the treatment of glioblastoma multiforme (GBM). Before temozolomide became generic, it generated US sales of $420 million and global sales of $910 million under its brand name Temodar(R) in 2012.
The first glimpse I had of Dr. Henry Brem has stuck with me.
It was in 1987, well before he'd become head of neurosurgery at Johns Hopkins. As a media specialist for Hopkins, I was wedged into a corner of an elevator, watching a man I presumed was Brem on his way to a press conference. He was talking about how he had been placing therapeutic wafers of some sort into the brains of tumor patients. I remember less about the science than Brem's demeanor. Though his voice was soft, it was excited and genuine. He radiated an optimism that left us mesmerized as the doors opened. An E.F. Hutton moment. We'd have bought stock in his technique that afternoon, had it existed.
FULL ARTICLE:
http://biotechstockreview.ning.com/profiles/blogs/the-chances-of-surviving-brain-cancer-are-minimal
How long it takes the FDA to "approve" a 510(k) submission.
Emergo conducted a review of public data on medical devices cleared by the US Food and Drug Administration between January 1, 2010 and December 31, 2014.
The 15,000+ device clearances examined went through the FDA’s Premarket Notification program, more commonly known as the 510(k). This process applies to nearly all Class 2 devices, and a relatively small percentage of Class 1 and 3 devices.
For purposes of analysis we separated the devices into four basic categories:
Traditional 510(k) submissions reviewed by internal FDA staff (72% of all)
Traditional 510(k) submissions reviewed by Third Party Reviewers authorized by the FDA (6% of all)
Special 510(k) submissions (20% of all)
Abbreviated 510(k) submissions (5% of all)
FULL STUDY RESULTS:
http://biotechstockreview.ning.com/profiles/blogs/how-long-it-takes-the-fda-to-approve-a-510-k-submission-1
Dr Henry Brem
Henry Brem has developed new tools and techniques that have changed the field of neurosurgery. Brem carried out the pivotal clinical study that introduced navigational imaging into the neurosurgical suite. His work led to the FDA's approval of the first image guidance computer system for intraoperative localization of tumors. Furthermore, he has changed the surgical armamentarium against brain tumors by inventing and developing Gliadel® wafers to intraoperatively deliver chemotherapy to brain tumors. His work has shown that surgeons can accurately deliver potent therapies directly at the tumor site.
At Hopkins, he has built one of the largest brain tumor research and treatment centers in the world. He reinstituted the Hunterian Neurosurgery Laboratory (originally founded by Cushing) and has trained numerous researchers who have revolutionized the fields of intraoperative imaging, angiogenesis, immunotherapy, and controlled release polymers for drug delivery to the brain.
http://www.hopkinsmedicine.org/neurology_neurosurgery/experts/profiles/team_member_profile/EF48C2ECB225F29CCA8C801AAEB2BE26/Henry_Brem
Not familiar with timelines for FDA approval, maybe the company has an idea when, call them.
when do you expect the FDA approval?
when do you expect the FDA approval?
|
Followers
|
9
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1210
|
|
Created
|
06/27/14
|
Type
|
Free
|
| Moderators | |||


| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
