Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Ok, thanks Vance.
Yep, I'm Sure! There is No RS up for vote.
Hi Vance, are you sure?
I just read a PDF on their website that basically said the want an r/s.
They want to increase the authorized shares.
They want to give the BOD a raise.
I quickly skimmed the article, but those were the sad highlights.
Unfortunately, I can't find the PDF again, but you can probably find it.
I'm not lying and I'm sure there will be more discussion about it.
My experience has been, that reverse splits always pass.
I think I should sell quickly and keep my profit.
Maybe buy back in slowly, after the r/s.
I would rather they announce a partner, no r/s and no BOD raise, for a good increase in share price.
Nope, no mention whatsoever of any rs vote. Why anyone would get that out of the sec filing last Friday I have no idea. Standard business, they are at roughly 88m os with only 100m as, time to raise the bar just in case.
Ok, those are some good thoughts and let's hope there are some good catalysts soon, such as;
A clear win of the lawsuit against Clarus.
A partnership for the sales and marketing of Tlando, three times per day pill.
A good Nash phase II trial result.
Approval of Tlando once per day pill.
A good preterm phase I trial.
Picked up more shares yesterday. I'm surprised there hasn't been any new analyst coverage with the Florida gang overseeing the last offering. I suppose the pending litigation against Clarus is minor cause for concern, but only due to monetary commitments. Regardless of that outcome we're still sitting pretty. TLando will come to market and their pipeline is continuing to advance. All the recent conference exposure has new eyes watching for updates as well. When this heats up, I don't think current prices will be available anymore!
Is a clay trader video post on a stock that has risen recently a reliable signal to sell?
* * $LPCN Video Chart 03-17-2021 * *
Link to Video - click here to watch the technical chart video
Next March at the earliest
When does Tlando tentative FDA approval become full approval?
Do we have to wait until the Clarus exclusivity expires?
When is the court case of Lipocine vs Clarus?
The recent Lipocine conference really bombed the share price, by waiting to market TLando in 2022 and by dealing the fatty liver biopsy results to later in 2021.
When a catalyst comes as we shareholders believe, I expect we'll see new 1yr+ highs. I honestly can't see how this investment can be a risk at this point. They've got cash, they've gotten tentative fda approval, they've got several other promising candidates! The recent offering is not what I consider dilution. If another one comes without a stated reason then I'll cut & run. But as for now, I'm adding every chance I get!
Yeah. I believe it's just the overall market in general. Seems most small-cap bios have taken a beating this week.
The drop in share price to below 1.62 is unfortunate. Let's how what it closes at. Hopefully it won't go down any further.
I've learned to interpret CEO speak:
I will not do a dilutive offering= I will do a dilutive offering, the very next day.
No reverse split= There will be an r/s.
You get the point, CEO's say one thing and then usually do the exact opposite.
The new admin is deflating the stock market.
Current O/S is 88,607,685. Got that number from a Yahoo poster citing O/S values from the offering prospectus and the offering itself. Not bad for where we are at, considering it's still a market cap under 160 million. Company is stronger than ever with several good news press releases at any time. Very safe bet!
I am looking at $10+ by end of year..
Back to $2 and plenty of cash in the coffers! This is Still heading to $3 in the not too distant future and if any of a number of good PR's come out then no holds barred!
looks like a good buying opportunity for LPCN...
Well, the stock price is sure down now. Ya, hopefully just a bump in the road.
Offering just killed off the momentum.. I guess its better to get this crap out now rather than after the outcome of their lawsuits..
Odd they think the stock is under pressure, this was written back in November. We've got good things ahead, this isn't even a bump in the road!
Future looks bright especially if they are favored with the lawsuits.. Still holding 2015 shares will plan to hold long term
Can't blame ya for that, but as steady as it's holding up this morning I'm expecting more is on the horizon!
Lighten up my position into half today now I am riding free shares..
20k views, 117k subscribers.. yeah I'd say that's cause for end of day volume. Still, as we well know Lipocine is undervalued. There are too many good things out and coming out to ignore.
This could be the reason why LPCN is trending up..
Nice day today Vance about time.. up 20% have you heard if there’s news or rumor of any sort?
H.C. Wainwright sticks to their Buy rating for LPCN
https://www.analystratings.com/articles/h-c-wainwright-sticks-to-their-buy-rating-for-lipocine-lpcn/
LPCN: LIPOCINE’S LIFT RESULTS: A COMPARISON WITH PEERS
https://news.yahoo.com/lpcn-lipocine-lift-results-comparison-104300871.html
I agree Vance non whatsoever
There is literally no risk here, except of dilution. And the chances of that are practically non-existent!
A number of different scenarios, and the odds of all of them being favorable to Lipocine are high. Hopefully we'll know more next week!
Hey Vance,
I believe Clarus and LIPO have a virtual meeting this Friday, do you expect anything to come of that? I've read that a summary of judgment was requested. Any idea what that could mean/lead to?
Good info Vance..
Thanks Vance. Hope you're right !
Short sellers have been taunting active LPCN forums for awhile now. They ate up the tentative approval, mistaken web page fiasco. Clarus has much to lose with the upcoming litigation, therefore they likely have a hand in the cookie jar so to speak. Plain English, Clarus shorting hoping LPCN gets strapped for cash & dilutes. Causing further shorting & big profits for those doing it.
Problem is, per the conference call yesterday a TLando distribution partner is imminent and today's news is extremely positive. Non-dilutive funds are not going to be a problem. LPCN is about to be upgraded and shorts will have to cover.
With the news we should be inching in to $2.. Oh well hopefully this spring will give us the return we deserved
Could you explain please? TIA!
Shorts gotta cover & Clarus likely in the mix. These prices are literally a gift!
What in the world is keeping this price down? Shouldn't we be flying today?
Positive Phase 2 Results just out!
https://ir.lipocine.com/
Definitely gearing up for an increase in share price soon. Should reach a higher base on any news of TLANDO partnership, Clarus trial, or 1144 phase 2 results. Any other pipeline news is also potential catalysts for increases in share price. This stock is a winner investment right now, just a matter of time before it pays off!
Patel mentioned in the webcast this morning that P2 NASH TLD was coming. Could be AH today or tomorrow. By EOW for sure. Seems like the run up.
Something is brewing its up 13%.. GO LPCN!!
|
Followers
|
50
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
785
|
|
Created
|
08/29/14
|
Type
|
Free
|
| Moderators | |||

675 Arapeen Drive
Suite 202
Salt Lake City, Utah 84108
Our telephone number is (801) 994-7383
Lipocine became a public company through a reverse merger in July 2013. Immediately after the reverse merger the Company raised $38 million in a private placement to institutional investors at $6.00 per share.
Lipocine pipeline products are based on its proprietary solubilization technology for effective oral delivery of water insoluble drugs to improve patient compliance.
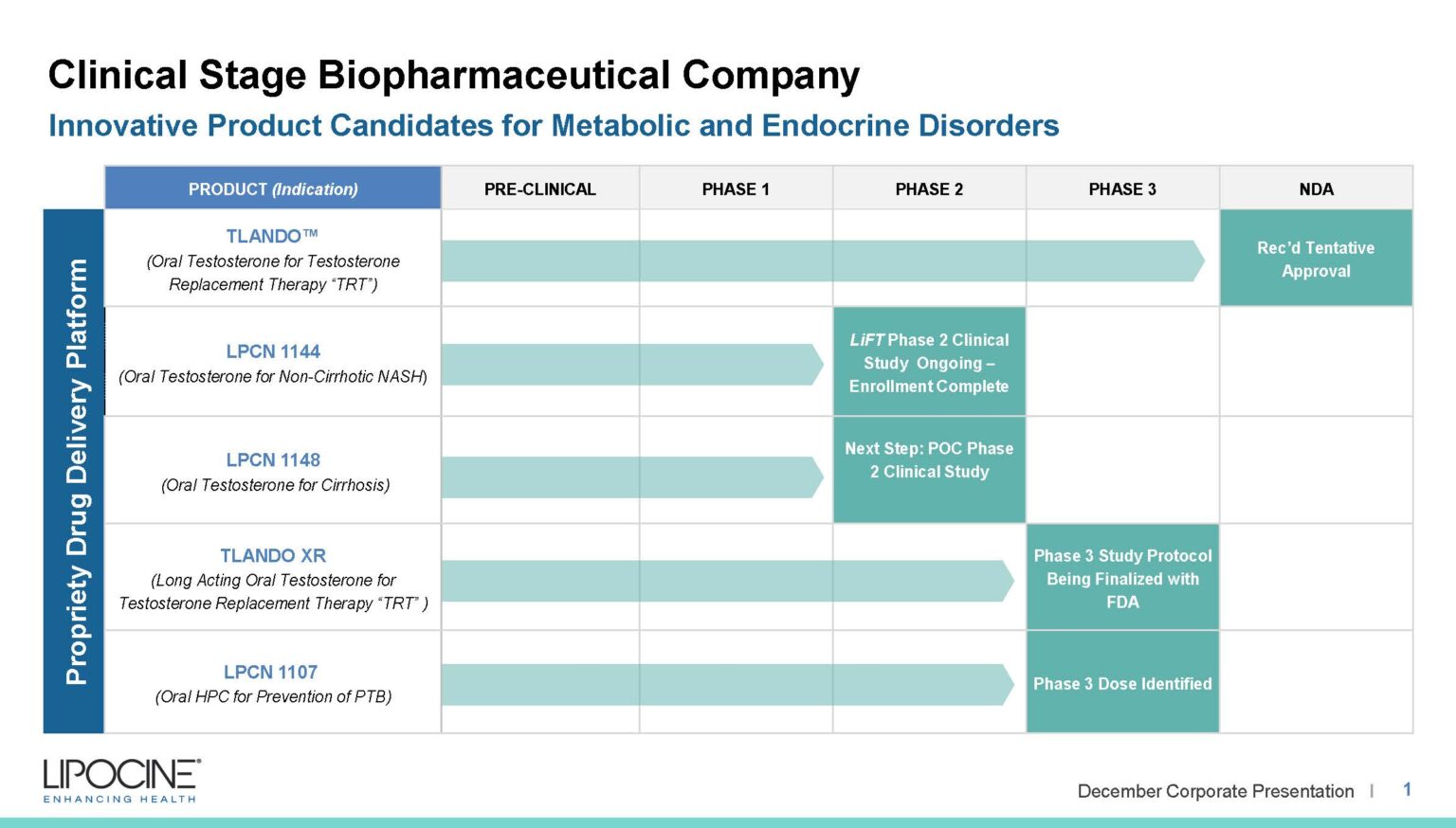
We are a clinical-stage biopharmaceutical company focused on metabolic and endocrine disorders. We improve compliance, absorption, and more with our proprietary drug delivery technologies.
LPCN 1144, an oral prodrug of bioidentical testosterone, is being developed as a treatment for pre-cirrhotic non-alcoholic steatohepatitis ("NASH") and is currently being studied in a Phase 2 paired biopsy NASH confirmed clinical study. Liver imaging results from the Phase 2 clinical study are expected by the end of 2020 with biopsy results expected by the end of the second quarter of 2021.
TLANDO™ is an oral testosterone replacement therapy product candidate containing Testosterone Undecanoate (TU) that is designed to help restore normal testosterone levels in males for conditions associated with a deficiency or absence of endogenous testosterone.
TLANDO™ is designed to overcome many of the issues related to TRT products on the U.S. market. TLANDO is being studied for both Primary and Secondary hypogonadism and is targeting the established chronic US TRT market.
TLANDO XR is a next-generation, novel ester prodrug of testosterone which uses the patent protected Lip'ral technology to enhance solubility and improve systemic absorption. Lipocine completed a Phase 2b dose finding study in hypogonadal men in 2016. The primary objectives of the Phase 2b clinical study were to determine the starting Phase 3 dose of TLANDO XR along with the safety and tolerability of TLANDO XR and its metabolites following oral administration of single and multiple doses in hypogonadal men. The Phase 2b clinical trial was a randomized, open label, two-period, multi-dose PK study. Results suggested that the primary objectives were met, including identifying the dose expected to be tested in the planned Phase 3 study. Good dose-response relationship was observed over the tested dose range. Additionally, the target Phase 3 dose met primary and secondary end points. TLANDO XR was well tolerated with no drug-related severe or serious adverse events reported in the Phase 2b study.
Cirrhosis is an end-stage non-alcoholic fatty liver disease (NAFLD) for which there is no FDA approved drug treatment. During 2015, approximately 1.3M NASH patients had cirrhosis (fibrosis grade 4). NASH cirrhosis patients typically experience increased morbidity and symptoms of hypogonadism such as alteration of hair distribution, anemia, sexual dysfunction, testicular atrophy, muscle wasting, fatigue, osteoporosis, and gynecomastia.
Testosterone levels fall progressively with increased chronic liver disease severity. Low testosterone levels, reported in up to 90% of male cirrhotic patients, is known to increase adverse outcomes and is a predictor of mortality in these patients with increased risk of major infections, transplantation rates, increased risk of for hepatic decompensation, worsening of sarcopenia, and higher Child-Pugh score grade and MELD score. This could also include the severity of portal hypertension and ascites grade.
Our team is currently formulating plans to conduct a proof-of-concept study in male subjects with cirrhosis through consultations with the FDA and key opinion leaders to evaluate the therapeutic potential of LPCN 1148 for the treatment of cirrhosis.
LPCN 1107 is an oral product candidate of 17-alpha-hydroxyprogesterone caproate (HPC) under development for the indication of prevention of recurrent preterm birth. LPCN 1107 has the potential to become the first oral HPC product for the prevention of preterm birth in women with a prior history of at least one preterm birth. Potential benefits of our oral product candidate relative to current injectable products include the elimination of pain and site reactions associated with weekly injections, elimination of weekly doctor visits or visits from the nurse, and elimination of interference/disruption of personal, family or professional activities associated with weekly visits.
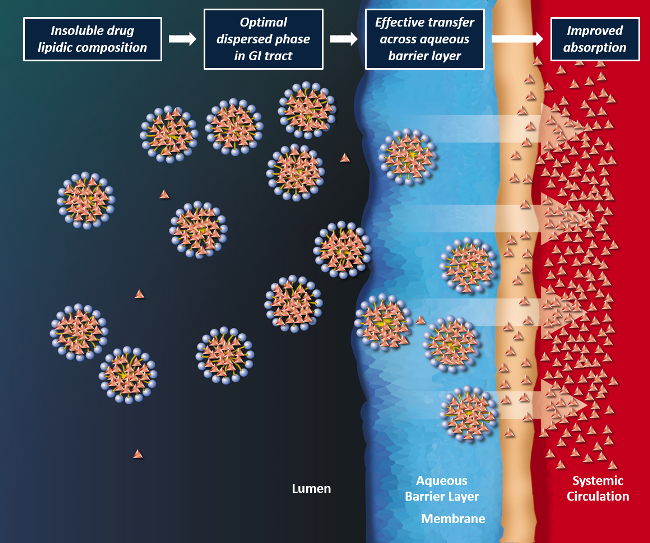
Lip'ral is a patented technology based on lipidic compositions which form an optimal dispersed phase in the gastrointestinal environment for improved absorption of the insoluble drug. Lip'ral presents insoluble drugs efficiently to the intestinal absorption site, thus bringing the absorption process under formulation control and making the product robust to physiological variables such as dilution, pH and food effects.
Non-alcoholic fatty liver disease ("NAFLD") is a reversible condition wherein large vacuoles of triglyceride fat accumulate in liver cells via the process of steatosis.
NASH is a more advanced state of NAFLD and can progress to a cirrhotic liver and eventually hepatocellular carcinoma or liver cancer. Twenty to thirty percent of the U.S. population is estimated to suffer from NAFLD and fifteen to twenty percent of this group progress to NASH, which is a substantially large population that lacks effective therapy. NAFLD/NASH is becoming more common due to its strong correlation with obesity and metabolic syndrome, including components of metabolic syndrome such as diabetes, cardiovascular disease and high blood pressure. In men, especially with comorbidities associated with NAFLD/NASH, testosterone deficiency has been associated with an increased accumulation of visceral adipose tissue and insulin resistance, which factors contributing to NAFLD/NASH.
Preclinical and clinical studies in the literature have shown the prevalence of testosterone deficiency across the NAFLD/NASH histological spectrum wherein low testosterone was independently associated with NAFLD/NASH with an inverse relationship between testosterone and NAFLD/NASH.
Post hoc analyses of existing clinical trial in subjects with comorbidities typically associated with NASH indicate that oral testosterone therapy significantly and consistently reduces elevated levels of key serum biomarkers (liver function enzymes and serum triglyceride) generally associated with NAFLD/NASH.
NAFLD prevalence in general population is estimated to be 20-30% in the Western world (Masarone et al, Rev Recent Clin Trials, 2014)
By 2020, prevalence of NAFLD cirrhosis is set to overtake hepatitis B and hepatitis C related cirrhosis (Starley et al, Hepatol, 2010)
The NASH market could peak at $30-40 billion by 2025 (Deutsche Bank industry report, “NASH – the next big global epidemic in 10 years?”, 2014)
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
