Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Jaguar Health has signed a binding term sheet covering the exclusive license and commercialization agreement for Jaguar’s novel plant-based, FDA-approved prescription drug crofelemer with Turkish specialty pharmaceutical company Gen Ilac Ve Saglik Urunleri Sanayi Ve Ticaret. The Agreement will entail a $2 million investment by GEN in Jaguar stock at a 75% premium to the market price, payment of double-digit royalties to Jaguar on all finished crofelemer products sold in the licensed territory, and transfer pricing terms for crofelemer supplied by Jaguar. As a result of this investment, GEN will own 6.7% of the shares of Jaguar common stock outstanding as of March 18, 2024. The Agreement will cover Jaguar’s FDA-approved indication of crofelemer for HIV-related diarrhea and all potential crofelemer follow-on indications, including cancer therapy-related diarrhea – the subject of Jaguar’s placebo-controlled pivotal Phase 3 OnTarget trial, as well as the rare disease indications short bowel syndrome and microvillus inclusion disease, and any other possible future human indications for crofelemer. The Agreement will allow GEN to manufacture crofelemer finished product and market the drug for all above-referenced indications in Turkey, Belarus, Ukraine, Azerbaijan, Uzbekistan, Kazakhstan, Turkmenistan, Russia, and Georgia following GEN’s receipt of regulatory approval for crofelemer for these indications in these countries
Another great day here for sure.
HKD did crazy squeeze but JAGX at $425 in month????? Sure I’m in lol
Reminds me of ENZC predictions back in the day. But hey if it goes that way we will all be multi millionaires
Jaguar Health Inc Stock (JAGX) Forecast
The Jaguar Health Inc (JAGX) stock price forecast for the next 30 days is generally positive, with an average analyst price target of $477.11, representing a +162623.67% increase from the current price of $0.2932. The highest analyst price target is $532.29, and the lowest is $421.92.
Generally positive. Lol
$1.20-$456 PT!>>>>>>>>>>
If we only had a magic ball… PT all over the place AI and computers are not that smart when searching in this one…
https://coincodex.com/stock/JAGX/price-prediction/
https://walletinvestor.com/stock-forecast/jagx-stock-prediction
https://www.marketwatch.com/investing/stock/jagx/analystestimates
https://www.macroaxis.com/valuation/JAGX/Jaguar-Animal-Health
https://stockscan.io/stocks/JAGX/forecast
https://foreverycast.info/forecast/JAGX/
https://stockscan.io
ANALYST 12 MONTH PRICE TARGET AVG. $477.11!!!!
Home › NASDAQ: JAGX › Forecast
Biotechnology
JAGX
0.2932
price up icon30.31%
0.0682
after-market AFTER HOURS: 0.3237 0.0305 +10.40%
Jaguar Health Inc (JAGX)
Date: 2024-05-02 16:00 O: 0.2200 H: 0.3210 L: 0.2200 C: 0.2932 Vol: 178.5M
Jaguar Health Inc Stock (JAGX) Forecast
The Jaguar Health Inc (JAGX) stock price forecast for the next 30 days is generally positive, with an average analyst price target of $477.11, representing a +162623.67% increase from the current price of $0.2932. The highest analyst price target is $532.29, and the lowest is $421.92.
Disclaimer: The information on this page is not intended to be financial advice. It is for general information only and should not be used to make any investment decisions. Please consult with a professional financial advisor before making any investment decisions.
12-Month JAGX Price Target
Average 612.82 (+208909.54% Upside)
Past 12 Months12-Month Forecast
Is Jaguar Health Inc (JAGX) A Buy Now?
Oscillators
Name Value Action
RSI(14) 81.39 Sell
STOCH(9,6) 63.66 Neutral
STOCHRSI(14) 100.00 Sell
MACD(12,26) 0.04 Buy
ADX(14) 58.32 Buy
William %R -12.59 Sell
CCI(14) 227.27 Buy
Buy: 3Sell: 3Neutral: 1
Summary: NEUTRAL
Moving Averages
Period Simple Exponential
MA10
0.20
Buy
0.21
Buy
MA20
0.16
Buy
0.17
Buy
MA50
0.11
Buy
0.14
Buy
MA100
0.12
Buy
0.17
Buy
MA200
0.25
Buy
0.52
Sell
Buy: 9Sell: 1Neutral: 0
Summary: BUY
According to our latest analysis, JAGX could be considered a Strong Buy, with 18 technical analysis indicators signaling 12 Buy signals, 4 signaling Sell signals and 1 Neutral signals. This might be a good time to open fresh positions on JAGX, as trading bullish markets is always a lot easier
RSI (Relative Strength Index): The RSI(14) value of 81.39 indicates that JAGX is overbought. This suggests that JAGX price has experienced a significant increase over a specified lookback period, potentially leading to a correction.
STOCH (Stochastic Oscillator): The STOCH value of 63.66 indicates that JAGX is neither overbought nor oversold. It indicates a balanced market sentiment without any extreme buying or selling pressure.
STOCHRSI (Stochastic Relative Strength Index): The STOCHRSI value of 100.00 indicates that JAGX is oversold. This suggests that JAGX price is at or near the highest level relative to its recent price history.
ADX (Average Directional Index): The ADX value of 58.32 suggests an extremely strong trend in the market. Such high values are relatively rare and indicate a very robust directional movement.
CCI (Commodity Channel Index): A CCI(14) value of 227.27 indicates that JAGX is overbought. This means that the price has moved significantly higher than its average, indicating a potential selling opportunity.
Long-term JAGX price forecast for 2025, 2030, 2035, 2040, 2045 and 2050
Based on our analysis about Jaguar Health Inc financial reports and earnings history, Jaguar Health Inc (JAGX) stock could reach $924.1 by 2030, $626.5 by 2040 and $2,070 by 2050. See the projected annual prices until 2050 of the Jaguar Health Inc stock below:
Jaguar Health Inc (JAGX) is expected to reach an average price of $180.0 in 2035, with a high prediction of $205.7 and a low estimate of $180.0. This indicates an +61288.36% change from the last recorded price of $0.2932.
Jaguar Health Inc (JAGX) stock is projected to chart a bullish course in 2040, with an average price target of $597.7, representing an +203770.78% change from its current level. The forecast ranges from a conservative $597.8 to a sky-high $626.5.
Our analysts predict Jaguar Health Inc (JAGX) to change +412371.20% by 2045, soaring from $1,211 to an average price of $1,209, potentially reaching $1,241. While $1,211 is the low estimate, the potential upside is significant.
Jaguar Health Inc (JAGX) stock is expected to climb by 2050, reaching an average of $2,013, a +686319.76% change from its current level. However, a wide range of estimates exists, with high and low targets of $2,070 and $2,030, respectively, highlighting the market's uncertainty.
Jaguar Health Inc Stock (JAGX) Year by Year Forecast
Jaguar Health Inc Stock (JAGX) Price Forecast for 2024
Our forecast for JAGX stock price in 2024 now indicates an average price target of $401.9 with a high forecast of $803.7 and a low forecast of $0.0512. The average prediction reflects an increase of +136966.54% from the last recorded price of $0.2932.
Month Average Low High Change from today's price
June, 2024 $569.8 $421.9 $532.3 +194233.79%
July, 2024 $777.2 $609.3 $759.2 +264982.83%
August, 2024 $810.9 $785.6 $776.4 +276459.29%
September, 2024 $772.5 $782.3 $796.4 +263379.13%
October, 2024 $805.2 $788.6 $803.7 +274527.22%
November, 2024 $796.3 $799.6 $800.3 +271474.34%
December, 2024 $795.2 $795.9 $793.6 +271107.35%
Jaguar Health Inc Stock (JAGX) Price Forecast for 2025
Jaguar Health Inc Stock (JAGX) is expected to reach an average price of $720.9 in 2025, with a high prediction of $788.9 and a low estimate of $652.9. This indicates an +245766.08% rise from the last recorded price of $0.2932.
It was next to impossible to find anybody talking about this up until today.
Now people can't shut up. lots of eyes on this. possible gapper tomorrow?
On stocktwits a lot of people are saying possibly $10 in the next few months after the global distribution of the product.
$8.09+ PT???
Are you saying the PRICE TARGET is $8??
Do you have someone who is saying this??
Just asking cause on Yahoo at the moment, current target price is 60 cents..
Thanks in advance
Too soon?? lol
Bullish lol I can't say that enough
Just put a limit order in at $2.00 shorties not taking my shares!
With the right news
Thanks for the heads up
And making money;)
lol he loves his BullFlags…
.46 and .52 levels we break that watch out!
For shorting most likely.
I bet… been sidelined on this one but uptrend looks killer!
I'm so convicted it's not even funny, this is about to be THE bio stock of the summer!
not me, its been along time coming
Claytrader on YouTube has JAGX for tomorrow watch list that’s cool!
We all gonna shit like the patients if it hits $8+ lol
MAY 6TH, we are going to RIP. #BILLIONS
its 1am in abu dhabi and those rich mfrs just bought it up after todays poster session i almost willing to put money on it!!!!!
Look for yourself! https://www.elite-pedgi.com/agenda
DAY 1 | THURSDAY | 2 MAY 2024
13:30 - 15:30 Pre-Conference Workshop: Pediatric Cholestasis in the Era of IBAT-inhibitors
By Invitation Only
14:00 Onsite Registration
15:50 Welcome Note
16:00 - 18:00 Session 1: State of the Art Lectures
18:00 - 18:30 Poster Session, Coffee Break & Exhibits
18:30 - 21:00 Session 2: Keynote Lectures
21:00 - 21:30 Abstract Oral Presentations
21:30 - 22:30 Dinner
its 1am now...
pumped all day
any takers on my hypothesis??
all these rich BILLIONAIRES about to here PRAVIN speak...... OMFG lets GOOOOOOOOOOOOOOOOOOO!!!!!!!!!!!
for you brains bears
https://www.elite-pedgi.com/
The 10th Annual Elite Ped-Gi Congress 2024: Home
https://www.elite-pedgi.com/speakers
peep
Dr. Pravin Chaturvedi
Chief Scientific Officer
Napo Pharmaceuticals,
California, USA
;)
LETS NOT FORGET, we filed a shelf offering the other day to use from time to time. As we climb the ladder in $1's you will see sophisticated sells after mega pumps. That is what its used for. $IBRX filed a shelf not too long ago and there marketcap has now went from 1B to 6B and climbing. They anticipated there share price and took action early just like $JAGX did. See yall @ $1
$JAGX Yes GREAT catalysts here.
You to bud! Been busy on the job. JAGX > some solid catalysts taking us higher.
It's been a while since I've seen you—great to have you here, bro!
Study Overview
Brief Summary
A 24-week, (two 12-week stages), randomized, placebo-controlled, double-blind study to evaluate the safety and efficacy of crofelemer in providing prophylaxis of diarrhea in adult patients with solid tumors treated with targeted cancer therapy-containing treatment regimens. Diarrhea grading will be done according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0.
Patients will be randomized 1:1 to placebo or crofelemer and will be stratified by the type of targeted cancer therapy and the tumor type. Placebo and/or crofelemer will be dispensed at Visit 1/Day 1 with the concurrent start of the targeted cancer therapy regimen. The initial Stage I double-blind placebo-controlled primary treatment phase will occur over a 12-week period to accommodate approximately 3 cycle chemotherapy cancer treatment dosing-cycles. The Primary and Secondary Endpoints will be analyzed after the last patient last visit (LPLV) of Stage I.
After completing the Stage I double-blind, placebo-controlled primary treatment phase, the subjects will have the option to remain on their assigned treatment arm and reconsented to enter into the Stage II extension phase. Reconsent will be required to enter into Stage II. For subjects who do not reconsent, visit 5 will be the last study visit.
Show less
Detailed Description
A randomized, placebo controlled, double blind study to evaluate the safety and efficacy of crofelemer in providing prophylaxis of diarrhea in adult patients with solid tumors receiving targeted cancer therapy containing regimens. Diarrhea grading will be done according to the National Cancer Institute's Common Terminology Criteria for Adverse Events (CTCAE Ver. 5.0).
Randomization will be at a 1:1 ratio with subjects randomized either to crofelemer 125 mg delayed-release tablets or matching placebo tablets administered orally twice daily with or without food. Randomization will be stratified by the type of targeted cancer therapy and by tumor type. Placebo and crofelemer treatment will be initiated concomitantly with the administration of targeted cancer therapy-containing regimens.
The Stage I double-blind placebo-controlled primary treatment phase will be the first 12-week period to accommodate targeted cancer therapy with approximately three (3) cycle chemotherapy regimens (if needed) over the inclusive 12-week period after initiation of crofelemer or placebo treatment in Stage I.
After completing the Stage I treatment phase (12 weeks), and after the LPLV of the primary Stage I treatment phase, the primary and secondary endpoints will be analyzed. The subjects will have the option to remain on their assigned treatment arm and reconsented to enter into the Stage II extension phase. Reconsent will be required to enter into Stage II. For subjects who do not reconsent, Visit 5 will be the last study visit. Subjects who enter into the Stage II extension phase will continue on their originally assigned study treatment commenced at the beginning of Stage I.
Show more
Official Title
A Phase 3 Multicenter, Randomized, Double-blind Placebo-controlled Trial Evaluating Crofelemer for the Prophylaxis of Diarrhea in Adult Patients With Solid Tumors Receiving Targeted-cancer Therapies With or Without Standard Chemotherapy
Conditions
Cancer Therapy-Related Diarrhea
Chemotherapy-related Diarrhea
Adult Solid Tumor
Prophylaxis of Diarrhea
Show 2 more conditions
Intervention / Treatment
Drug: Crofelemer 125 MG [Mytesi]
Drug: Placebo
Other Study ID Numbers
NP 303-102
Study Start (Actual)
2020-10-07
Primary Completion (Estimated)
2024-04
Study Completion (Estimated)
2024-06
Enrollment (Estimated)
256
Study Type
Interventional
Phase
Phase 3
Awesome day. +48.36 moving up higher $$$
Beautiful day! JAGXZEE 🎲 🎲 🎲 🎲 🎲 LOL 🤣
$JAGX
|
Followers
|
151
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
6877
|
|
Created
|
12/01/15
|
Type
|
Free
|
| Moderators ProfitChaser oldstocks DTGoody crudeoil24 SmellMyFinger | |||
Jaguar Health, Inc. $JAGX
Bringing Plant-based Medicines to Life™
Pharmaceutical Manufacturing
San Francisco, California
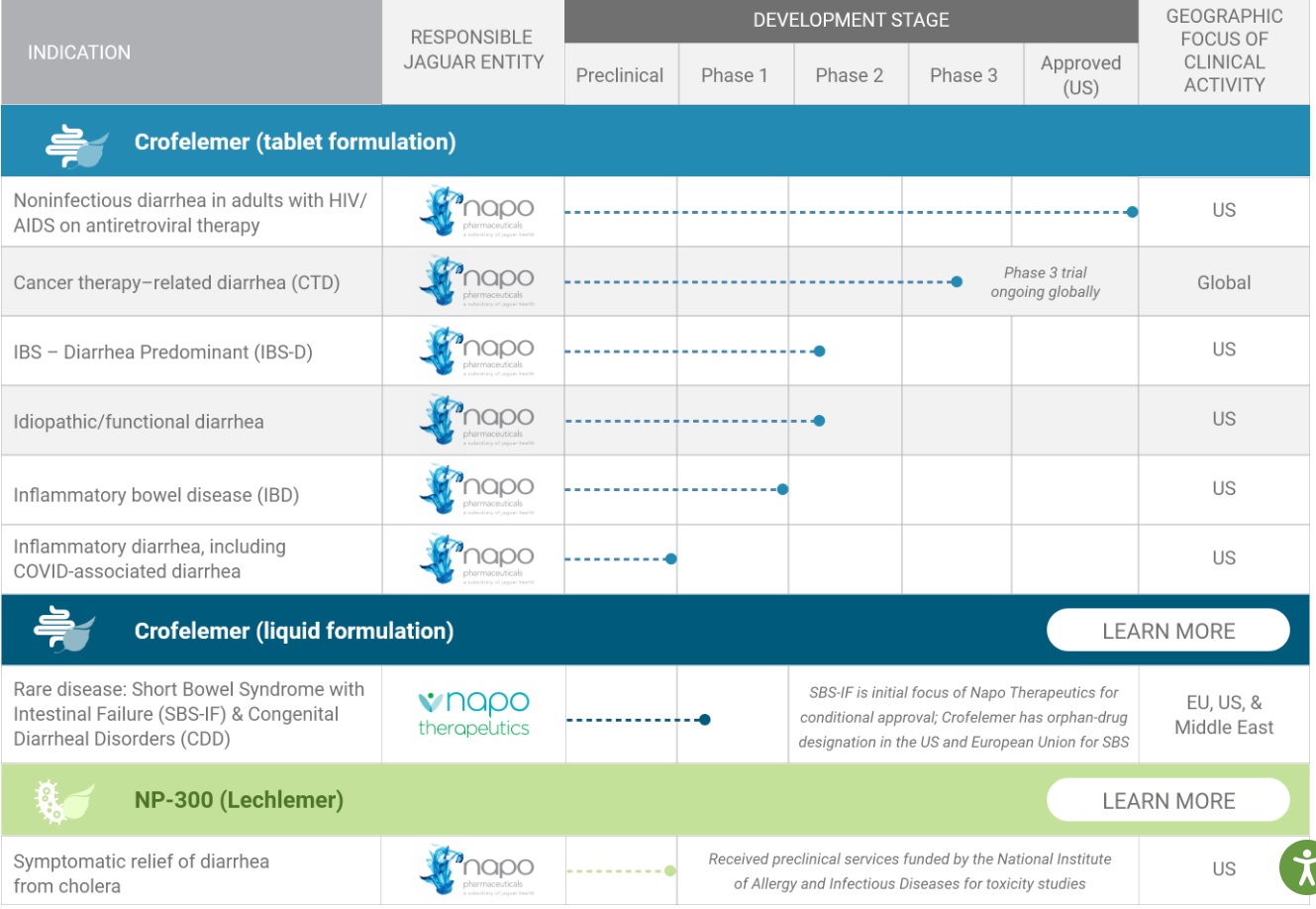
Jaguar Health’s organizational framework is designed to enable our product pipeline to reach the world’s interconnected marketplace, allowing for development and commercialization opportunities worldwide.
Products:

Mytesi Crofelemer 125mg delayed release tablets
Mytesi is FDA-approved for symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy
Market size of the FDA HIV Mytesi Crofelemer could be $25-$30 Million in the USA
Mytesi has a novel mechanism of action that modulates chloride secretion in the gastrointestinal tract, reducing excessive chloride and accompanying water flow and relieving watery diarrhea.
Mytesi is not indicated for the treatment of infectious diarrhea. In clinical studies, the most common adverse reactions occurring at a rate greater than placebo were upper respiratory tract infection (6%), bronchitis (4%), cough (4%), flatulence (3%), and increased bilirubin (3%).
Canalevia-CA1 Crofelemer delayed release tablets
Distributors listed:
https://www.canalevia.com/distributor-information
CANALEVIA-CA1 is conditionally approved for the treatment of chemotherapy-induced diarrhea (CID) in dogs.
Conditionally approved by FDA pending a full demonstration of effectiveness under application number 141-552.
CAUTION: Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. It is a violation of Federal Law to use this product other than as directed in the labeling.

Lytham Partners Investor Select Conference February 1st, 2024
https://wsw.com/webcast/lytham10/jagx/2084640
Pipeline: https://jaguar.health/pipeline/
How to calculate the value of drugs and biotech companies
https://www.baybridgebio.com/drug_valuation.html
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, and bowel incontinence.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study, a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study , a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan and/or rare diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar's Entheogen Therapeutics Initiative is focused on developing novel prescription medicines derived from plants for mental health indications.
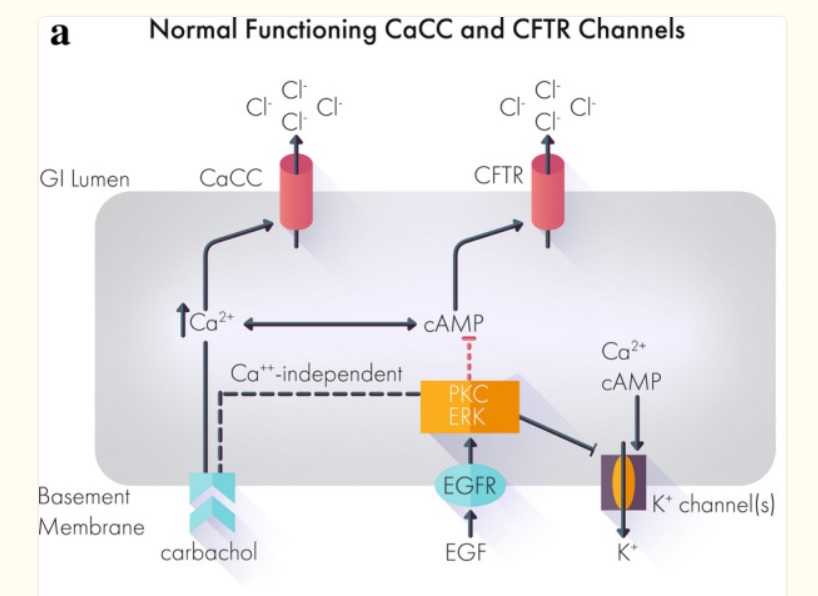
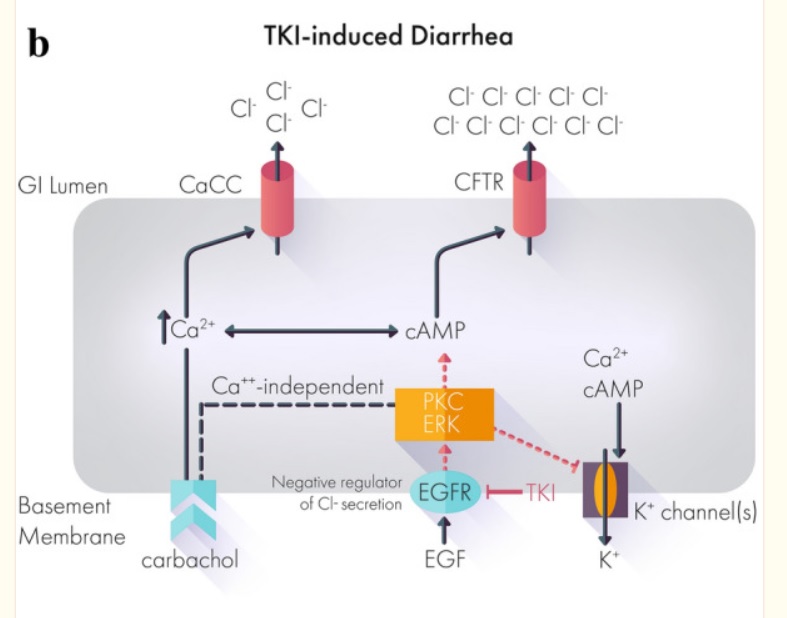
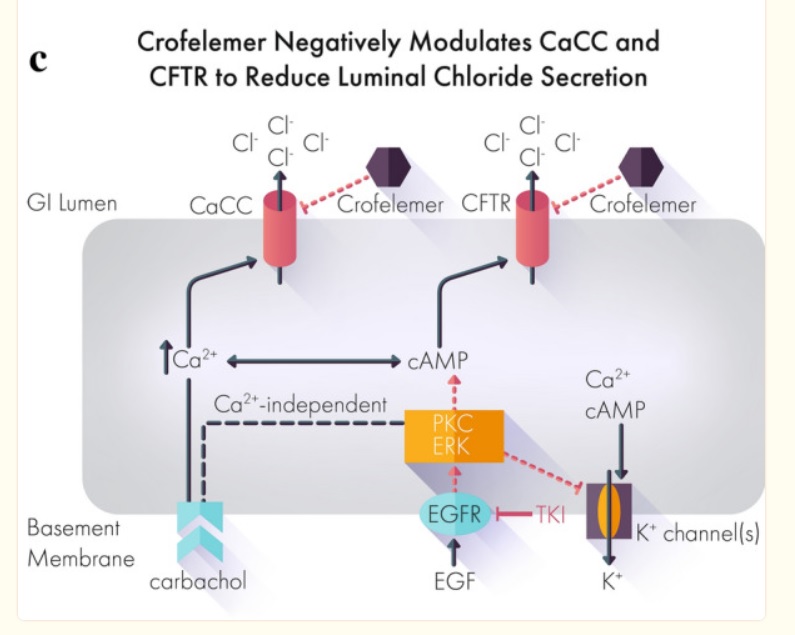
A Case Report: Improved Control of Tyrosine Kinase Inhibitor-Induced Diarrhea
Despite the efficacy of tyrosine kinase inhibitors (TKIs) across multiple cancers, side effects including treatment-related diarrhea can impede a patient’s ability to reach therapeutic doses or stay on therapy.
Below, we present the case of a 72-year-old patient with metastatic papillary renal cell carcinoma recurrent despite nephrectomy.
Over the course of treatment, the patient received multiple different tyrosine kinase inhibitors with varying efficacy.
Treatment with the TKI cabozantinib after failure of two prior TKIs resulted in a clinical response with shrinkage of his nodal metastatic disease.
However, the severe treatment-related diarrhea refractory to conventional management required both dose holds and dose reductions of cabozantinib.
Off-label administration of crofelemer, a novel FDA-approved antidiarrheal agent, successfully controlled the treatment-related diarrhea and allowed resumption and partial dose increase of cabozantinib.
This case suggests that crofelemer could be a viable therapeutic strategy to address TKI-induced diarrhea.
9.8 million people
receive chemotherapy worldwide per year
Wilson B, Jacob S, Yap M, et al. Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. Lancet Oncol. 2019;20(6):769-780. doi:10.1016/S1470-2045(19)30163-9
Chemotherapy-induced
diarrhea occurs in
50%-80%
of treated patients
Stein A, Voigt W, Jordan K. Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management. Ther Adv Med Oncol. 2010;2(1):51-63. doi:10.1177/1758834009355164
Patients with cancer therapy–
related diarrhea (CTD) were
40% more likely to
discontinue chemotherapy or
targeted cancer therapy than
patients without CTD
Okhuysen PC, Schwartzberg LS, Roeland E, et al. The impact of cancer-related diarrhea on changes in cancer therapy patterns. J Clin Oncol. 2021;39(15):12111. doi:10.1200/JCO.2021.39.15_suppl.12111
Research & Development
In October 2020, Napo Pharmaceuticals initiated a pivotal Phase 3 clinical trial
This Phase 3 study is evaluating crofelemer for prophylaxis of diarrhea in adult cancer patients receiving targeted therapy
The principal investigator and co-investigators identified are from MD Anderson Cancer Center
Jaguar Health https://jaguar.health/
Napo Pharmaceuticals https://napopharma.com/
Napo Therapeutics https://napotherapeutics.com/en/
https://www.magdalenabiosciences.com/
Magdalena is a joint venture formed by Jaguar Health and Filament Health. The company holds an exclusive license to Jaguar's proprietary library of approximately 2,300 highly characterized medicinal plants and 3,500 plant extracts for designated therapeutic indications.
Visit Jaguar on LinkedIn:
https://www.linkedin.com/company/jaguar-health/
Visit Jaguar on Instagram: https://www.instagram.com/jaguarhealthcommunity/
Visit Jaguar on X: https://twitter.com/Jaguar_Health
Phone: 415-371-8300
Contact: hello@jaguar.health
Press Releases: https://jaguarhealth.gcs-web.com/news-releases
Most if not all works by mechanism chloride ions into the gut then water comes and diarrhea out. Crofelemer normalizes abnormal ion flow targeting only locally in the gut.
Jaguar has been putting the expense into making sure Crofelemer drug supply is ready to get into this opportunity to treat cancer related chemo patients since pharmacy’s already can get FDA approved, all ready in the in the supply chain in the pharmacy as Crofelemer for AIDs patients.
Research has shown it costs 3x as much to treat a cancer patient with diarrhea.
It would be better to treat the cancer patient for diarrhea with Crofelemer pending OnTarget Study and FDA approval.
Since Crofelemer is FDA approved to treat dogs for Cancer Chemo related Diarrhea it has been said:
If your going to have cancer and cancer related Diarrhea, it’s better to be a dog than a human right now.
The companies that manufacture the cancer agents/drugs sort of point to the availability of Crofelemer because it allows them to keep their patients on their therapies thereby improving their penetration and their revenues opportunity.
3rd party market research put Short Bowel Syndrome at $5-$12 Billion opportunity because these patience are so ill, high mortality, high morbidity (the state of being unhealth for a particular disease or situation), so high of expense to manage through life.
That opportunity can be reached in small trials around the world to show benefits to be able to show global development.
Product already approved, already fully in the supply chain in the pharmacy.
Potential Market of Mytesi (crofelemer)
Jaguar and Napo $JAGX if the Phase 3 OnTarget Study has good results.
The Phase 3 OnTarget Study results are due to be out soon in this quarter.
The Phase 3 OnTarget study is testing whether the company's plant-based prescription drug crofelemer can prevent or substantially reduce diarrhea and other symptoms of CIOB when any one of the selected 24 different targeted therapies that are associated with diarrhea in patients is initiated.
Each year, more than 1 million cancer patients in the United States receive chemotherapy or radiation. Globally 9.8 million require chemotherapy.
Diarrhea is a common side effect of targeted therapies. Up to 95% of patients on tyrosine kinase inhibitors (TKIs) suffer from diarrhea, depending on the TKI used.
Any drug used to treat cancer (including tyrosine kinase inhibitors or TKIs) can be considered chemo, but here chemo is used to mean treatment with conventional cytotoxic (cell-killing) drugs that mainly kill cells that are growing and dividing rapidly. Chemo was once one of the main treatments for CML.
The cost for Mytesi oral delayed release tablet 125 mg is around $2,600 for a supply of 60 tablets. This amount is the one month supply as testing in the OnTarget Study.
That is a 24 week study = 5.6 month supply needed = $14,560.00 for treatment.
Potential Market of Mytesi (crofelemer)
100,000 patients a year = $1,456,000,000
250,000 patients a year = $3,640,000,000
500,000 patients a year = $7,280,000,000
1,000,000 patients a year = $14,560,000,000 (assuming worldwide)
5,000,000 patients a year = $72,800,000,000 (assuming worldwide)
The estimate of patients depends on how many quit taking chemo, the onTarget Study, FDA approval and or quit taking Mytesi (crofelemer).
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
