Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
On the daily, the chart looks really good and (almost) ready to run. I think 0.25 was the bottom and it's only up from here.
Here we go come on need a PR and go break the .50 mark!!!
Which reasons do you have to assume it's bad? I can give you several reasons why I believe it will be good
Because if you do a little bit of DD on the company's history and its main ingredient used (crofelemer, already approved and used on humans -HIV- and dogs -cancer-, for the preventive treatment of diarrhea with no side effects for long term use), you would assume there's little chance for the study not being successful. But, of course, this is not 100% certain, so i recommend doing your own DD.
Some serious 'indecision' going on, with a touch of bias to the downside since the open...
So will whoever is absorbing whats being sold at 26 keep doing it, and for how long..
Hmmm
Or maybe it will be bad results. How you know it’s going to be good?!!!
100% Agreeee thank you so much! Best luck to you!!!
feel that posters pain...I wouldn't put anything past any ticker these days....It's just when someone speaks in absolutes ("for sure")on anything,It "definitely" lol gives me pause.
Next week, we are a week before ASCO congress. Napo pharmaceuticals is exhibiting there with a booth.
If they release the phase 3 results next week, NAPO is allowed to talk about it at the congress. They can not present an abstract (since they were too late for submission), but you are allowed to talk about it. Which would be VERY convenient on a congress like ASCO.
If you want to miss phase 3 data.. be our guest
My dog has diarrhea
Was todays activity a STOP LOSS ORDER take out??
that move that dropped the share price sharply down to 25 cents or so reminded me of a manuever by the market to clear out
STOP LOSS orders just sitting there. Volume has been way down, and I imagine there are shareholders who set up stop loss orders
to protect against a falling share price. The market makers can 'see' these orders, know how many shares are 'ripe' for the picking
and todays 'hit' has me thinking a lot of folks looked at their accounts and realized they no longer had their shares.
Sad part is, the share price is dropped thru the stop losses, bottoms out and then the trades are executed. And once they are cleaned out,
a bounce off the lows takes place.
Unfortunately, this is happening at a time when the momentum has been dampened and who knows when the next PR bump will occur and if
anyone is watching/waiting for it, with the ultimate question being, how big a bump might it get.
And someone just posted there could be shares available down in the 22 cents/sh range. If buyers are scarce?? its possible.. cause that move today
did not help things..
He should be able to get 22s tomorrow or Friday
REPLYING to: I want to buy it again so bad but worry about RS
They have until sometime in the 3rd qtr to be back above a $...
Unless they need to raise cash (r/s and then do an ATM or secondary)???
I think the bigger reason to be hesitant after todays action is that the share price might take a deeper leg down..
Its been on one heck of a run since 10 cents or less.. It could use a good consolidation/basing action.
Going to be interesting to see how serious the share holders are (how tight they hold their shares)
RS coming for sure they been dumping for a while and already been through 2 RS can’t take another one. Thank you and good luck!
They won't do r/s before announcing PIII results. And even after, it depends on the share price (above $1 or not) and their plans (they might still want to achieve $5+ with r/s), all before august (deadline for them to have the stock above $1 for ten consecutive trading days).
Personally, i'm more afraid of a sell the news type of event, rather than r/s.
I want to buy it again so bad but worry about RS
Bought a new round of shares on the current lows..
30 cents seems to be achievable based on recent activity. And if they can replicate that number, I will be happy with the purchase
Note: I don't discount the possibility that this could drop sub 26 cents if enough holders get restless and buyers don't step up.
Call it your standard share price CONSOLIDATION PHASE after a very nice sustained run up from sub 10 cents..
But if it does, can't do anything about it. Just have to hope the companys 'expectations' for something positive to come out of the PH 3
is fulfilled..
Disclaimer: own some, could have been more patient. And with time, will find out if I should have been (smile)
Good luck.
Was a thinking I was done trying to trade/flip shares.. But its chart is shaping up indicating
if there isn't any positive PR 'imminently'?? Some selling pressure is to be expected..
Got it trying to hold above its Moving Average line (around 27 cents), but if it sees 26 cents?? The MA
line will now be an overhead resistance it needs to claw back thru.. And a MA line that will
confirm rolling over lower and help 'push' the share price lower if buyers don't step up.
Disclaimer: own none, looking for another round for the 'imminent' PH 3 results... Going to be curious how high they
will be able to run up the share price on the news..
Tell folks management 'could' be your friend when things are going well for the company.
But when things are tough, its the company's 'survival' and 'best interests' that come first. And as a shareholder??
We should expect that or you may get 'nothing' instead of 'something'..
And its generally ugly for shareholders if you are owners of shares before something is being produced and revenues have
started flowing.. Ugly in terms of wall street demand for the shares may not be there, possible ATMs, secondaries and best of all??
Reverse splits..
And now it's time to finally reward shareholders !
Have been back in for a while. Just had to leave the board it was a bit nuts here for a while. Still not a fan of Lisa but I would say she has gotten the job done for the company but has had little or no regard for shareholders in the way she accomplished it
Yeah but they were too late to submit for ASCO. The titles of the abstract were allready released. Nothing about ONTARGET. So NO , they won't present at ASCO. Even not an abstract. Although I agree that they will release the results prior to the ASCO conference, to give NAPO something to talk about.
But don't pin May 23 in your agenda, because they won't be in it
P3 IMMINENT Then dollar land $$$
Thank you for the insight, i didn't know that. That would explain it...
And if, in their view, "imminent" doesn't mean "next 2-3 days from now", then i also think it will be closer to the ASCO conference, so end of may/1st days of june. Maybe even as early as 23th of may, because that's the date abstracts are presented/uploaded on the ASCO site, if i've read/understood this correctly.
If you read the filing you will see that the 16 million shares were set at a price of .18 which means they have a cap on how much they can be sold for... multiply 16 million time ,18 and what ever that figure is that is all that can be converted... that comes to $2,880,000 so if the sell at .26 or .50 or .99 once they have sold enough shares to rach $2.8 million they can not sell anymore... and Jaguar would prefer that all conversions take place prior to the Phase III so there is not a sell off that could drive the price back to a dollar or lower...
For me I think she said imminent because they are getting closer to the June conference and the seller will be done by then... Then when the conference is here they will release the Phase III results..... thats just my thinking but I have not seen anything that would tell me I am wrong.
Or maybe Gen Ilac preferred to do this before PIII, to not negatively impact the stock afterwards, since it needs to get to $1 till august, hopefully without r/s? idk... I wish Jaguar won't drag this for weeks, idk how much it can hold current levels without drops of positive news coming every 3 days or so.
Maybe 0.26 will be a strong support now and SMA200 (currently around 0.24) won't be touched again. Fingers crossed.
Hmm... i've forgot about that, lol. Probably because i thought at the time that someone with over 16 mil of shares to sell would wait for the PIII results, especially since they (Gen Ilac) have a partnership with Jaguar, so they should know better about the upcoming results.
Did you read the May 1st filing. That’s why it dropped and rises then drops and rises. As soon as all of that is cleaned up it will move up and keep going up. IMO
Imminent is just a tad longer than a Few Weeks…. If you get my drift LOL LOL LOL

Look at the chart, 1 minute timeframe, regular hours (not AM/PM). Find me another one minute red candle,with a >10-14% drop, in recent days/weeks. Not a drop lasting several minutes, just 1 minute red candle with that big of a move. You won't find one.
Maybe it's nothing, but i've found the move strange.
Are you new? it was a stop loss raid like they have done before.
That instant drop from 0.31 to 0.27 is weird. I wonder why was that... I know it can move like that, but rarely in an instant and only on some news. Did they posted anything new, that the market didn't like it? I can't find any...
What are you expecting with 'imminent'? what kind of movement do you forecast? I think the stock is underperforming
Imminent...any day now
I am wondering the same thing and holding
$JAGX now BOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOm
They need a dollar for ten days on the BID before Mid August or they will RS... so if its not a dollar by Aug first expect the RS to happen in a week from Aug 1
: can't forget they 'need' a $ before the end of the 3rd qtr or a r/split comes into play..
.. it is now and JAGX moving to GREEEEEEEEEEEEEEEEEEEEEEEEEN soon , looks very good now
imo
Is there a place to watch their webcast recording? I'm assuming it's over now...
Results are imminent, so in good case scenario, +500% - +1500% , who knows, let us dream. Happened before, with much less attention on some stocks. JAGX is being watched by lots of people.
Institutional holdings are finally showing an increase. Blackrock as biggest example.
had an order in to sell when the price went over 28.40.. SOBs wouldn't take the shares...
Expecting a 'hit' on the open and then to see how long that lasts, how deep it goes.. EXPECTING..
And its setting up that way from the looks of it.. see what happens.
Anyone else see that 1.3m bid currently at .2827
During the webcast (which is ongoing right now) she just really emphasized 2 times (!) the word IMMINENT... She literally said, we used the word 'forthcoming' for a while, but now it is really IMMINENT
nice good $JAGX NEWS - green coming now WEEEEEEEEEEEEEEEEEEEEE
|
Followers
|
151
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
6877
|
|
Created
|
12/01/15
|
Type
|
Free
|
| Moderators ProfitChaser oldstocks DTGoody crudeoil24 SmellMyFinger | |||
Jaguar Health, Inc. $JAGX
Bringing Plant-based Medicines to Life™
Pharmaceutical Manufacturing
San Francisco, California
Jaguar Health’s organizational framework is designed to enable our product pipeline to reach the world’s interconnected marketplace, allowing for development and commercialization opportunities worldwide.
Products:

Mytesi Crofelemer 125mg delayed release tablets
Mytesi is FDA-approved for symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy
Market size of the FDA HIV Mytesi Crofelemer could be $25-$30 Million in the USA
Mytesi has a novel mechanism of action that modulates chloride secretion in the gastrointestinal tract, reducing excessive chloride and accompanying water flow and relieving watery diarrhea.
Mytesi is not indicated for the treatment of infectious diarrhea. In clinical studies, the most common adverse reactions occurring at a rate greater than placebo were upper respiratory tract infection (6%), bronchitis (4%), cough (4%), flatulence (3%), and increased bilirubin (3%).
Canalevia-CA1 Crofelemer delayed release tablets
Distributors listed:
https://www.canalevia.com/distributor-information
CANALEVIA-CA1 is conditionally approved for the treatment of chemotherapy-induced diarrhea (CID) in dogs.
Conditionally approved by FDA pending a full demonstration of effectiveness under application number 141-552.
CAUTION: Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. It is a violation of Federal Law to use this product other than as directed in the labeling.

Lytham Partners Investor Select Conference February 1st, 2024
https://wsw.com/webcast/lytham10/jagx/2084640
Pipeline: https://jaguar.health/pipeline/
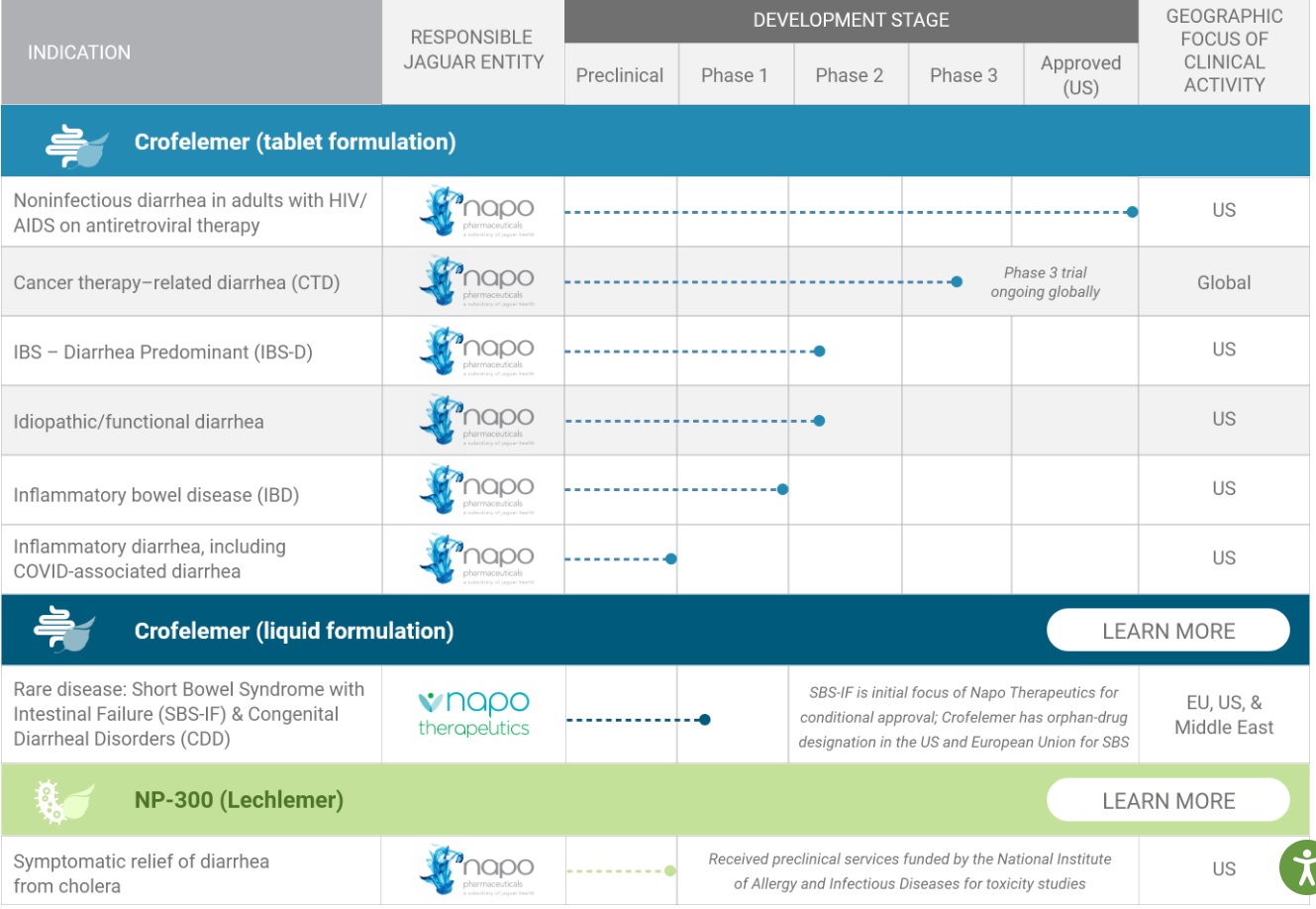
How to calculate the value of drugs and biotech companies
https://www.baybridgebio.com/drug_valuation.html
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, and bowel incontinence.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study, a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study , a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan and/or rare diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar's Entheogen Therapeutics Initiative is focused on developing novel prescription medicines derived from plants for mental health indications.
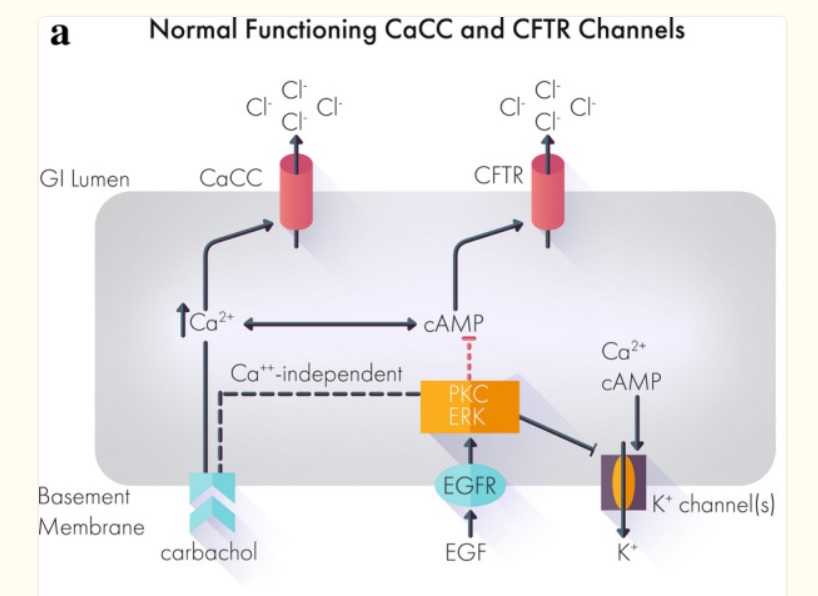
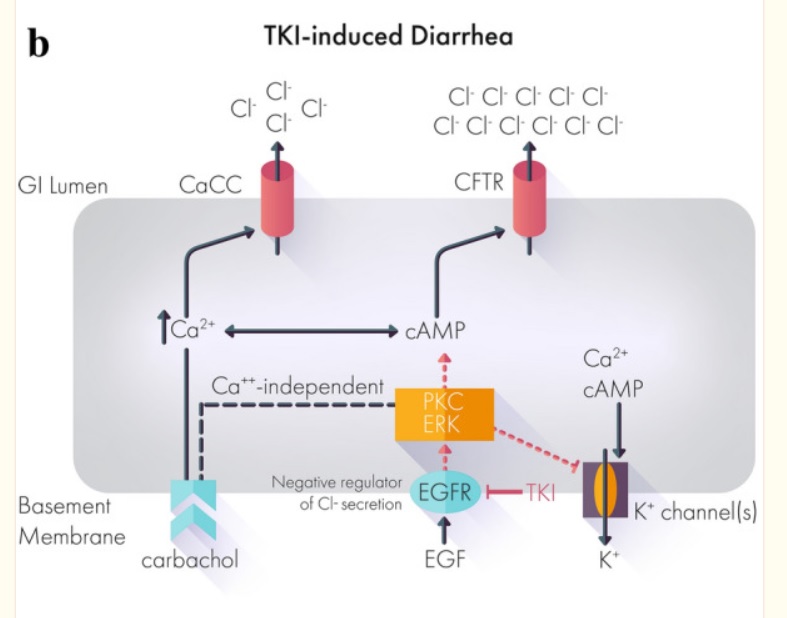
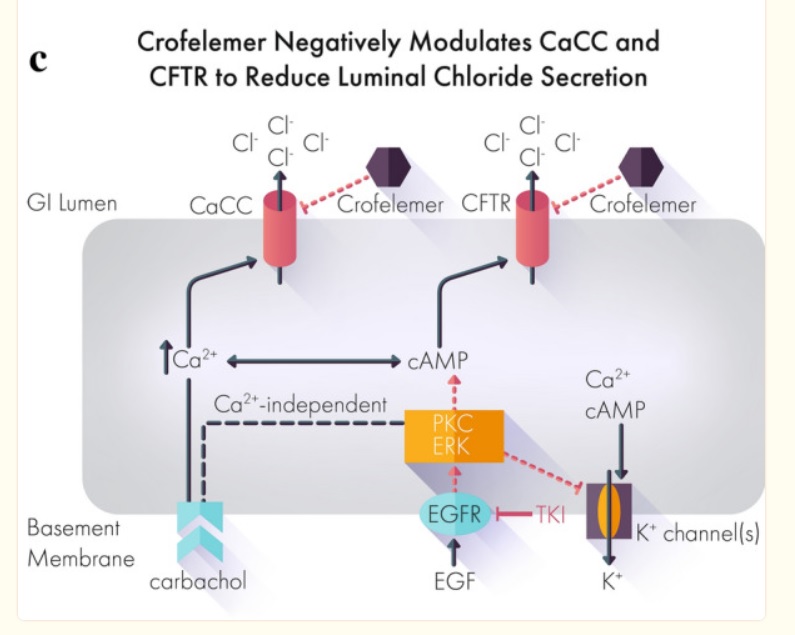
A Case Report: Improved Control of Tyrosine Kinase Inhibitor-Induced Diarrhea
Despite the efficacy of tyrosine kinase inhibitors (TKIs) across multiple cancers, side effects including treatment-related diarrhea can impede a patient’s ability to reach therapeutic doses or stay on therapy.
Below, we present the case of a 72-year-old patient with metastatic papillary renal cell carcinoma recurrent despite nephrectomy.
Over the course of treatment, the patient received multiple different tyrosine kinase inhibitors with varying efficacy.
Treatment with the TKI cabozantinib after failure of two prior TKIs resulted in a clinical response with shrinkage of his nodal metastatic disease.
However, the severe treatment-related diarrhea refractory to conventional management required both dose holds and dose reductions of cabozantinib.
Off-label administration of crofelemer, a novel FDA-approved antidiarrheal agent, successfully controlled the treatment-related diarrhea and allowed resumption and partial dose increase of cabozantinib.
This case suggests that crofelemer could be a viable therapeutic strategy to address TKI-induced diarrhea.
9.8 million people
receive chemotherapy worldwide per year
Wilson B, Jacob S, Yap M, et al. Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. Lancet Oncol. 2019;20(6):769-780. doi:10.1016/S1470-2045(19)30163-9
Chemotherapy-induced
diarrhea occurs in
50%-80%
of treated patients
Stein A, Voigt W, Jordan K. Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management. Ther Adv Med Oncol. 2010;2(1):51-63. doi:10.1177/1758834009355164
Patients with cancer therapy–
related diarrhea (CTD) were
40% more likely to
discontinue chemotherapy or
targeted cancer therapy than
patients without CTD
Okhuysen PC, Schwartzberg LS, Roeland E, et al. The impact of cancer-related diarrhea on changes in cancer therapy patterns. J Clin Oncol. 2021;39(15):12111. doi:10.1200/JCO.2021.39.15_suppl.12111
Research & Development
In October 2020, Napo Pharmaceuticals initiated a pivotal Phase 3 clinical trial
This Phase 3 study is evaluating crofelemer for prophylaxis of diarrhea in adult cancer patients receiving targeted therapy
The principal investigator and co-investigators identified are from MD Anderson Cancer Center
Jaguar Health https://jaguar.health/
Napo Pharmaceuticals https://napopharma.com/
Napo Therapeutics https://napotherapeutics.com/en/
https://www.magdalenabiosciences.com/
Magdalena is a joint venture formed by Jaguar Health and Filament Health. The company holds an exclusive license to Jaguar's proprietary library of approximately 2,300 highly characterized medicinal plants and 3,500 plant extracts for designated therapeutic indications.
Visit Jaguar on LinkedIn:
https://www.linkedin.com/company/jaguar-health/
Visit Jaguar on Instagram: https://www.instagram.com/jaguarhealthcommunity/
Visit Jaguar on X: https://twitter.com/Jaguar_Health
Phone: 415-371-8300
Contact: hello@jaguar.health
Press Releases: https://jaguarhealth.gcs-web.com/news-releases
Most if not all works by mechanism chloride ions into the gut then water comes and diarrhea out. Crofelemer normalizes abnormal ion flow targeting only locally in the gut.
Jaguar has been putting the expense into making sure Crofelemer drug supply is ready to get into this opportunity to treat cancer related chemo patients since pharmacy’s already can get FDA approved, all ready in the in the supply chain in the pharmacy as Crofelemer for AIDs patients.
Research has shown it costs 3x as much to treat a cancer patient with diarrhea.
It would be better to treat the cancer patient for diarrhea with Crofelemer pending OnTarget Study and FDA approval.
Since Crofelemer is FDA approved to treat dogs for Cancer Chemo related Diarrhea it has been said:
If your going to have cancer and cancer related Diarrhea, it’s better to be a dog than a human right now.
The companies that manufacture the cancer agents/drugs sort of point to the availability of Crofelemer because it allows them to keep their patients on their therapies thereby improving their penetration and their revenues opportunity.
3rd party market research put Short Bowel Syndrome at $5-$12 Billion opportunity because these patience are so ill, high mortality, high morbidity (the state of being unhealth for a particular disease or situation), so high of expense to manage through life.
That opportunity can be reached in small trials around the world to show benefits to be able to show global development.
Product already approved, already fully in the supply chain in the pharmacy.
Potential Market of Mytesi (crofelemer)
Jaguar and Napo $JAGX if the Phase 3 OnTarget Study has good results.
The Phase 3 OnTarget Study results are due to be out soon in this quarter.
The Phase 3 OnTarget study is testing whether the company's plant-based prescription drug crofelemer can prevent or substantially reduce diarrhea and other symptoms of CIOB when any one of the selected 24 different targeted therapies that are associated with diarrhea in patients is initiated.
Each year, more than 1 million cancer patients in the United States receive chemotherapy or radiation. Globally 9.8 million require chemotherapy.
Diarrhea is a common side effect of targeted therapies. Up to 95% of patients on tyrosine kinase inhibitors (TKIs) suffer from diarrhea, depending on the TKI used.
Any drug used to treat cancer (including tyrosine kinase inhibitors or TKIs) can be considered chemo, but here chemo is used to mean treatment with conventional cytotoxic (cell-killing) drugs that mainly kill cells that are growing and dividing rapidly. Chemo was once one of the main treatments for CML.
The cost for Mytesi oral delayed release tablet 125 mg is around $2,600 for a supply of 60 tablets. This amount is the one month supply as testing in the OnTarget Study.
That is a 24 week study = 5.6 month supply needed = $14,560.00 for treatment.
Potential Market of Mytesi (crofelemer)
100,000 patients a year = $1,456,000,000
250,000 patients a year = $3,640,000,000
500,000 patients a year = $7,280,000,000
1,000,000 patients a year = $14,560,000,000 (assuming worldwide)
5,000,000 patients a year = $72,800,000,000 (assuming worldwide)
The estimate of patients depends on how many quit taking chemo, the onTarget Study, FDA approval and or quit taking Mytesi (crofelemer).
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
