Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
30,000,000.00 Class Action Settlement | Deadline INO Stock News
………
https://stocktwits.com/Lookingdown/message/498183010
Nucleic Acid Vaccines against SARS-CoV-2
https://www.mdpi.com/2076-393X/10/11/1849
“ 2. The Research and Development Process of Nucleic Acid Vaccines
...
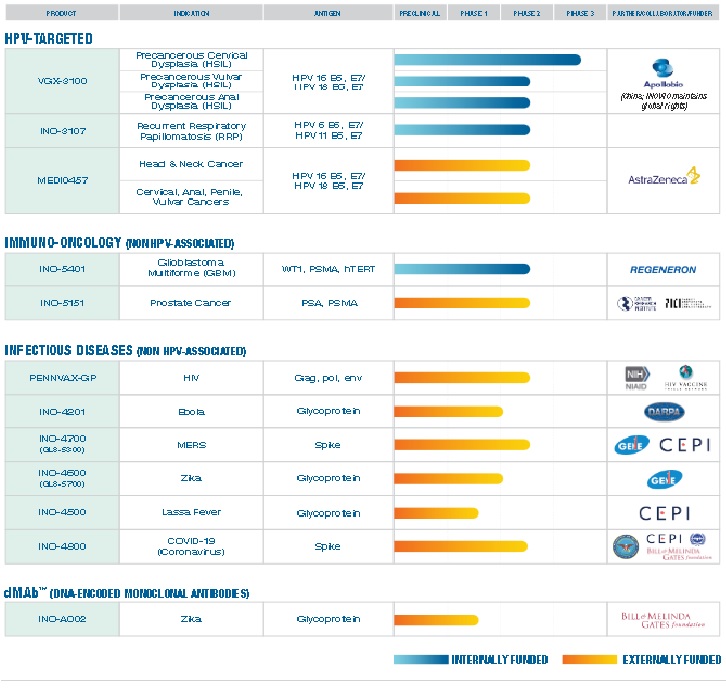
Additionally, the vaccine life cycle includes production, supply, available on the market, and post-marketing research in the real world. Currently, the ZyCoV-D vaccine developed by Cadila in Ahmedabad, Gujarat, India is the first DNA vaccine for people to be approved anywhere in the world [28]. INO-4800, developed by Inovio (the leading global development corporation of DNA vaccines), is the first DNA vaccine to advance to clinical trials and is currently undergoing phase three clinical trials, having the prospect of being commercially available within one year.
"
https://www.reddit.com/r/Inovio/comments/z5aiyf/nucleic_acid_vaccines_against_sarscov2/?utm_source=share&utm_medium=ios_app&utm_name=iossmf
INO I don't know if you mean down 49% AMA, or at 49% but I will point out for the benefit of young viewers that the ten largest institutional holders of INO stock are Index Funds. By their prospectuses, they have to buy the universe of listed stocks, even stinkers like Inovio.
It is no sign of institutional approval or support of this scam.
Mr Inovio everything, penny stock promoter Avtar Dhillon (an Inovio Founder, Inovio President, Inovio CEO, Inovio Director and Inovio Chairman and Paid Consultant until 2019, when he became too hot) was handcuffed and taken out of his LA home on multiple Stock Fraud and Obstruction of Justice charges and is awaiting trial in Boston related to other penny stocks he was deeply involved.
Inovio has not originated a single approved drug, therapy or vaccine in its 44 years of scamming investors.
The number of bagholders of INO stock are legion, and many have died waiting to recover their losses here.
Stay far away from this stock unless you are the nimblest of penny stock players. The drops have been 60% and 70% in one day.
You are the mark. Scammers want you in. They will post official sounding but meaningless stuff to lure you in.
There are a ton of good low priced bio stocks to play for big gains. You don't need to stop here. You don't want to stop here.
The value of an Inovio share today is thirteen cents, when the two most recent Reverse Splits are accounted for.
Garbage.
MG
No position in INO in about 8 years.
Was not burnt by INO
Over 50,000 stock posts of integrity across multiple stock forums- all as Miami Gent
INO Institutional Shares 123,576,898 - 49.53% (ex 13D/G)
“announced that it has discontinued its internally funded efforts to develop INO-4800 as a COVID-19 heterologous booster vaccine. The decision follows INOVIO's comprehensive review of its portfolio, market conditions, and global demand for COVID-19 vaccines.”
……….
https://www.prnewswire.com/news-releases/inovio-provides-update-on-covid-19-heterologous-booster-vaccine-candidate-ino-4800-301661270.html
“Although INO-4500 and INO-4700 were well-tolerated by participants in our clinical trials and generated immune responses, the two-dose regimen did not meet CEPI's selection criteria for further development. “
…….
https://ir.inovio.com/news-releases/news-releases-details/2022/INOVIO-Provides-an-Update-on-Lassa-Fever-and-MERS-Programs/default.aspx
dana-farber center for dna damage and repair....segue crispr; RAD51
The CDDR has developed a RAD51 immunohistochemistry assay for RAD51. Tumor cells with proficient homologous recombination (HR) have RAD51 foci.; Organoids
The CDDR generates patient tumor-derived organoid cultures which can be used for biomarker analysis and for precision medicine. The organoids can be analyzed for their sensitivity to novel DNA Repair inhibitors and drug combinations.;
Biomarkers for Drug Response
The CDDR identifies new biomarkers and establishes immunohistochemistry assays to analyze tumor tissue taken from cancer patients before and after drug treatment.;
Computational Approaches
The CDDR uses computational analysis of public and CDDR experimental databases to identify biomarkers in cancers and to assess cancer risk and survival.; etc.
Safety, Immunogenicity, and Efficacy of INO-4800 for COVID-19 in Adults at High Risk of SARS-CoV-2 Exposure
https://ichgcp.net/clinical-trials-registry/NCT04642638
"
Overall Status
Terminated
Start Date
2020-11-30
Completion Date
2022-09-13
Primary Completion Date
2022-09-13
Phase
Phase 2/Phase 3
Study Type
Interventional
Primary Outcome
Measure Time Frame
Phase 2: Change From Baseline in Antigen-specific Cellular Immune Response Measured by Interferon-gamma (IFN-?) Enzyme-linked Immunospot (ELISpot) Assay Baseline up to Day 393
Phase 2: Change From Baseline in Neutralizing Antibody Response Measured by a Pseudovirus-based Neutralization Assay Baseline up to Day 393
Phase 3: Percentage of Participants, (SARS-CoV-2 seronegative at baseline), With Virologically-confirmed COVID-19 Disease From 14 days after completion of the 2-dose regimen up to 12 months post-dose 2 (i.e. Day 42 up to Day 393)
Secondary Outcome
Measure Time Frame
Phase 2 and 3: Percentage of Participants With Solicited and Unsolicited Injection Site Reactions From time of consent up to 28 days post-dose 2 (up to Day 56)
Phase 2 and 3: Percentage of Participants With Solicited and Unsolicited Systemic Adverse Events (AEs) From time of consent up to 28 days post-dose 2 (up to Day 56)
Phase 2 and 3: Percentage of Participants With Serious Adverse Events (SAEs) Baseline up to Day 393
Phase 2 and 3: Percentage of Participants With Adverse Events of Special Interest (AESIs) Baseline up to Day 393
Phase 3: Percentage of Participants With Death From All Causes Baseline up to Day 393
Phase 3: Percentage of Participants, (SARS-CoV-2 seronegative at baseline), With Non-Severe COVID-19 Disease From 14 days after completion of the 2-dose regimen up to 12 months post-dose 2 (i.e. Day 42 up to Day 393)
Phase 3: Percentage of Participants, (SARS-CoV-2 seronegative at baseline), With Severe COVID-19 Disease From 14 days after completion of the 2-dose regimen up to 12 months post-dose 2 (i.e. Day 42 up to Day 393)
Phase 3: Percentage of Participants, (SARS-CoV-2 seronegative at baseline), With Death From COVID-19 Disease From 14 days after completion of the 2-dose regimen up to 12 months post-dose 2 (i.e. Day 42 up to Day 393)
Phase 3: Percentage of Participants, (SARS-CoV-2 seropositive at baseline), With Virologically-Confirmed SARS-CoV-2 COVID-19 Disease From 14 days after completion of the 2-dose regimen up to 12 months post-dose 2 (i.e. Day 42 up to Day 393)
Phase 3: Change From Baseline in Antigen-specific Cellular Immune Response Measured by IFN-gamma ELISpot Assay Baseline up to Day 393
Phase 3: Change From Baseline in Neutralizing Antibody Response Measured by a Pseudovirus-based Neutralization Assay Baseline up to Day 393
Enrollment
1308
"
xray8 posted ino/celletra4500 data from 10/25
but gavi article notes inovio /not confidential 10/22:https://www.gavi.org/vaccineswork/cepi-ramps-vaccine-research-new-study-warns-lassa-fever-set-expand-its-reach-across
"INO" looking good !!
https://www.researchgate.net/publication/341518402_Immunogenicity_of_a_DNA_vaccine_candidate_for_COVID-19
http://news.foodmate.net/wap/index.php?moduleid=21&itemid=641724
The author of this research report will be at this conference; Wang, Bin and is chairman of company that has contract with INO for distribution in China
Novel Vaccine R&D Summit 2022
1. Introduction of the meeting
After the 21st century, with the development of genomics, human beings began to develop new vaccines, new vaccines mainly include genetic engineering subunit vaccines, recombinant vaccines, synthetic peptide vaccines, genetic engineering vector vaccines, nucleic acid vaccines and anti-unique antibody vaccines, etc., since the outbreak of the new crown epidemic, the research and development of nucleic acid vaccines has been promoted.
In order to explore and learn the technical route of new vaccines, build a communication platform for researchers and research institutions, deepen academic exchanges and industrialization cooperation, promote the research and development process of new vaccines, and benefit human health, the 2022 New Vaccine R&D Summit will be held in Shanghai on November 12-13 (Saturday-Sunday). The main contents include: global vaccine strategy, policy and market trends, epidemic prevention and control and immunization, disease prevention and new vaccine research and development, vaccine process development and quality, vaccine research and development of new technologies, clinical research, registration application, etc. The organizing committee will invite famous international and domestic vaccine experts, entrepreneurs, academic experts from scientific research institutes and heads of disease control departments to give wonderful reports.
Second, the main topics
1. Global vaccine strategies, policies and market trends
2. Disease prevention and new vaccine research and development (new crown vaccine, influenza vaccine, monkeypox vaccine, shingles vaccine, cancer vaccine, rabies vaccine, hypertension vaccine, tuberculosis vaccine)
3. Nucleic acid vaccine special session (mRNA vaccine, DNA vaccine, breakthrough in delivery technology, nucleic acid vaccine process and quality)
4. Vaccine process development and quality control (vaccine adjuvants, production processes, digital factories, vaccine quality control)
5. New technologies for vaccine research and development (DC vaccines, recombinant vaccines, nanoparticle vaccines, nasal spray vaccines)
6. Future vaccine innovation strategy and R&D outlook (R&D, production, investment, market)
3. The guest of the report has been identified
Chang Lingqian, professor of Beihang School of Biological and Medical Engineering, founder of Zaiyue Biotechnology
Chen Yixin is an associate professor at Xiamen University Academy of Sciences/School of Public Health
Dai Lianpan is a researcher at the Institute of Microbiology, Chinese Academy of Sciences, and the leader of the Innovative Vaccines and Immunology Research Group
Gong Wenping, deputy director and associate researcher of the Institute of Tuberculosis Medicine Department of the PLA General Hospital
Hu Yeqin, Vice President of R&D of Chengdu Meikekang Biotechnology Co., Ltd
Jiang Chunlai, Deputy General Manager of Changchun Baike Biotechnology Co., Ltd
Jing Lulin, Deputy Director of the Vaccine Clinical Research Center of Sichuan Provincial Center for Disease Control and Prevention
Li Qihan is the director of the Institute of Medical Biology, Chinese Academy of Medical Sciences
Shaowei Li is a professor at the School of Life Sciences, Xiamen University
Li Xiuling, General Manager of Shanghai Institute of Biological Products Co., Ltd
Liao Yuhua, Chief Scientist of Wuhan Huajiyuan Biotechnology, Professor of Huazhong University of Science and Technology Union Hospital
Liu Mi, Professor of the Department of Pharmacy, School of Pharmacy, Soochow University, Chairman of Suzhou Ersheng Biotechnology
Liu Yong, Founder and General Manager of Jiangsu Ruike Biotechnology Co., Ltd
Lu Jiahai is a professor at the Sun Yat-sen Research Institute, School of Public Health, Sun Yat-sen University
Shi Li, Chairman/CEO of Yidao Biotech
Sun Bingbing is a professor at Dalian University of Technology
Wang Xuanyi is a researcher at the Institute of Biomedical Sciences, Fudan University
Wang Bin, Distinguished Professor of Fudan University, Chairman of Ai Di Weixin
Wang Xiangxi is the deputy director of the Key Laboratory of Infection and Immunology, Institute of Biophysics, Chinese Academy of Sciences
Wei Jiangbo, Director of the R&D Department of Sinopharm Zhongsheng Biotechnology Research Institute
Wu Ke, CEO of Wuhan Bowo Biotechnology Co., Ltd
Xiaojun Xia is a researcher at Sun Yat-sen University Cancer Center/State Key Laboratory of Oncology, South China
Xu Jianqing is a professor at Shanghai Public Health Clinical Center
Xu Yan, Vice President of R&D of Wuhan Binhui Biotechnology Co., Ltd
Yan Huimin, Center for Vaccine and Immunology, Public Health Clinical Center, Fudan University
Daniel Zhang, Associate Professor, Jilin University
Zhang Yinchuan, Deputy General Manager of Jiangsu Kunli Biotechnology
Tao Zhu, Chief Scientific Officer of CanSino Biologics AG
Mark Newman is CSO of GeoVax Corporation
4. Contact person
Teacher Hu 18153592317 (same number as WeChat)
https://seekingalpha.com/article/4548533-inovio-pharmaceuticals-catalysts-on-horizon
Looks like some catalysts are coming in the next 8-16 weeks. Good for trading. GBM could be a sleeper.
Ah, just more negative comments with no actual fact to support your claim of a R/S. BLAH,BLAH.
Pursuing hypothetical lines of questions are a waste of time.
Inovio (INO) is a long running scam.
No one should come near this stock.
MG
Father of The Inovio Scam Expose
R/S ? speculation or fact? An answer is expected based on your post.
Rinse and repeat. Lol. Their will be a reverse split coming and no approved drugs ever - IMO. Just a wasteland of money losing believers.
INO Haven't you heard the saying, "Those who forget history are condemned to repeat it?"
Anyway, I hope you have a great weekend and can put your INO 11 cents a share value* out of yoiur mind for awhile.
* When two most recent R/s's are accounted for
Not interested in past situations regarding "INO" . Your comment suggested possible future R/S. I said provide facts to your latest statement.
Facts? Facts are Inovio has had multiple R/S's. The two most recent were both "one for four". So you can divide today's price by those two splits. Let me do the math for you. At tonight's close of 1.64, that puts today's real INO share price at ten cents, when the two most recent R/S's are accounted for. That is just since 2,000. This scam stock has been around a lot longer than that- 44 years.
You could ask Mr Inovio everything, Avtar Dhillon (INO Founder, INO President, INO CEO, INO Director and INO Chairman and INO Paid Consultant to 2019) how many Reverse Splits there have been but he's prolly busy preparing for his trial for stock fraud and obstruction of justice.
My opinion, a R/S will not happen.
They will have to, must be compliant to rules
Ino is a pos, only good for shorting. Wait for the rs an then short the sh.. out of it!
Old news or not this company is a POS
"ALERT", ignore negative misinformation given about "INO" !!
ALERT: INO IS A PUMP & DUMP
Please see my thread for details
Stay away- no approved product originated in its 43 years of fleecing the investing public
Long time Founder, President, CEO, Director, Chairman and Paid Consultant to Inovio, Avtar Dhillon, currently facing trial for other penny stock scams he was involved in.
This stock is about traders getting in on the drops and then posting to make the stock sound legit. You are the mark. They need your money to come in, to further the pump.... and then the rug pull.
Are you that nimble?
So many good stocks to make $$ on. Avoid this junk.
MG
No position whatsoever in about 8 years
Was not burnt by INO
Just HATE scams
.
Yes, Inovio (INO) faded crooked Dhillon in 2019 because he had become such a lightning rod for shareholder wrath.
Crooked Dhillon is in deep chit now with the government for stock fraud on a couple of penny stocks other than Inovio.
Inovio was one of many Dhillon penny stocks babies. INO is valued at 12 cents now when the two most recent reverse splits are accounted for.
INO is a 43 year pump & dump with no product originated and approved in that time.
Inovio (INO) should shut down and return what money is left to shareholders
Inovio is a disgrace of a company.
MG
Father of The Inovio Scam Expose
Avtar Dhillon and Inovio are old news. He has no ties anymore with the company since almost 3 years.
(from discussion of BBBY, pumps & dumps, bad stock market behavior, etc)
People do these things without thinking it through.
Those thinking about conducting a pump and dump, never give a thought to it segueing into a pump & jump.
Mr Inovio (INO) everything, Avtar Dhillon, is now under indictment for stock fraud in other stocks he was also involved with. He has been living a lavish lifestyle in Los Angeles. The possibility of wearing an orange one piece for the balance of his good years may have him feeling a little jumpy.
INO Nobody is interested in tainted by close association to notorious Avtar Dhillon Inovio.
And no product originated and approved in its 43 years further sullies this "company"
Inovio is not on big pharma's radar. It is not even spoken of.
Inovio should have shut down years ago.
MG
Father of The Inovio Scam Expose
Mergers and acquisitions could be a "tailwind" into the end of the year, especially after recent announced deals by Pfizer Inc. (NYSE:PFE), according to a Jefferies analyst.
M&A could pick up, particularly in to January, when the JPMorgan Healthcare conference takes place, which tends to be a deal catalyst, Jefferies analyst Michael Yee said on Friday.
"People start to get excited into January," Yee explained.
Last month Pfizer (PFE) agreed to buy Global Blood Therapeutics (GBT), in a deal that valued he sickle cell disease drug maker at $5.4 billion and in May the pharmaceutical giant agreed to acquire Biohaven Pharmaceutical (BHVN) for $11.6 billion in cash. And in April Pfizer agreed to purchase closely held ReViral for up to $525M.
Yee highlighted the recent rally in SPDR S&P Biotech exchange-traded fund XBI (NYSEARCA:XBI), which is 25% off its bottom.
"I think it's a really important data point that we've had a bunch of positive clinical data sets, a bunch of financings, a bunch of good news and I do think we are headed higher into the end of the year and into 2023," Jefferies said in a CNBC interview.
"You have had a bunch of deals in the past two months, Pfizer being particularly aggressive in the space, deploying a lot of their Covid money across the space, got everybody excited," Yee added.
Yee highlighted that even with Inflation Reduction Act and the Medicare negotiation and drug pricing concern that has always been a concern the sector, the biotech group moved higher over the last two months.
"We think people digested that, we think people are comfortable with that," Yee said. "You look at where stocks and valuations are basically five to six year lows on valuation. People are willing to forgo that, particularly in this macro, recessionary environment, there's not a lot of concern about that."
Last month a report said that J&J (JNJ), Merck (MRK) and Regeneron (REGN) were the most exposed to Medicare drug price negotiation.
"With M&A, drug pricing behind us, I think you can start to dip your toes back into here," Yee said. "I like the pullback here by the way, a 10% pullback is healthy and I think we move higher off of this correction," Yee added.
Also see a recent RBC Capital Markets report found the negotiation would impact revenue for pharmaceutical and biotech companies, but it wouldn't be that significant.
Should I laugh now or later INO$ HA HA HAHAHA
Lol WOOHOOOOOO INO$
BEWARE ,Incorrect negative info on "INO is being posted on this site . Do your own research and evaluation.
"INO" going up
"INO looking good
No urgent update "INO" looking good
"INO" looking good
"INO" looking good !
URGENT UPDATE from the Father of The Inovio Scam Expose:
INO IS A 43 YEAR SCAM!
NO APPROVED PRODUCT ORIGINATED IN 43 YEARS
LONG TIME INOVIO FOUNDER, INOVIO PRESIDENT, INOVIO CEO, INOVIO DIRECTOR AND INOVIO CHAIRMAN AND INOVIO PAID CONSULTANT UNTIL 2019, AVTAR DHILLON, CURRENTLY FACING STOCK FRAUD CHARGES IN BOSTON BY A JOINT GOVERNMENT TASK FORCE
SEE MY ST POSTS HERE TO LEARN MORE: https://investorshub.advfn.com/boards/boardposters.aspx?boardid=13093
CLICK ON "1701" ACROSS FROM MY MONIKER, MIAMIGENT.
NEWBEES GET SCREWED ON THESE INO PUMPS & DUMPS.
YOU HAVE SELF REPECT, KNOW HOW TO TRADE STOCKS OF REAL COMPANIES, NOT ROLL IN THE MUD OF A SCAM.
MG
$2.44 weeeee Monkeypox spreading INO$ CNBC showed five companies doing Monkeypox INO$ was one of them. No there not doing nothing with Monkeypox lmao hahahaha
Very likely
Guarantee they’re working on something with monkeypox WOOHOOOOOO INO$
OK INO is going up for nothing lmao
"Thank you for your inquiry. Unfortunately, we are not currently conducting any clinical trial for monkeypox.
We encourage you to refer to www.clinicaltrials.gov for recruiting trials for your indication of interest.
https://clinicaltrials.gov/ct2/results?cond=Monkey+Pox&Search=Apply&recrs=b&recrs=a&age_v=&gndr=&type=&rslt=
Sincerely,
Clinical Trials Team at Inovio"
Up pretty good in premarket Somethings going on Definitely monkeypox
|
Followers
|
631
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
40500
|
|
Created
|
07/25/08
|
Type
|
Free
|
| Moderators | |||







DISCLAIMER The Board Assistants herewith, are not licensed brokers and assume NO responsibility for the actions, investment decisions, and or messages posted on this forum.
• We do NOT recommend that anyone buy or sell any securities posted herewith. Any trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.
Whatch out for fake comments by manipulators
https://www.griproom.com/fun/how-to-spot-fake-comments-on-stock-boards
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
