Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
INO Well, we know Inovio's Mr. Everything, Dirty Avtar Dhillon was arrested and charged with stock fraud and obstruction related to two other stocks. Deceiver Dhillon did not break off from Inovio until 2019, very likely the heat on him then. The gov't works a year or two before charging. Kim was Defrauder Dhillon's protegee. Dhillon needed a front man because long before he was in trouble with the gov't, he was reviled in the stock market for his scams.
I don't for a minute believe double-crosser Dhillon was squeaky clean during his incredibly long tenure at Inovio, as Inovio President, CEO, Director and Chairman & highly Paid Consultant to Inovio into 2019.
Who knows if the klieg lights will be shone on Inovio
Blowhard Kim was with associated with Double Dealer Dhillon those many years- that's a very poor association! They say, "you are the company you keep"
MG
Father of The Inovio Scam Expose
INO No US agency will be touching Inovio going forward, in my opinion. It's a poor association. Bureaucrats worry about missteps, appearances.
MG
Father of The Inovio Scam Expose
WHO and MPP announce agreement with NIH for COVID-19 health technologies
12 May 2022 News release Geneva Reading time: 2 min (499 words)
WHO’s COVID-19 Technology Access Pool (C-TAP) and the Medicines Patent Pool (MPP) today finalized a licensing agreement with the United States National Institutes of Health (NIH) for the development of several innovative therapeutics, early-stage vaccines and diagnostic tools for COVID-19.
The licenses, which are transparent, global and non-exclusive, will allow manufacturers from around the world to work with MPP and C-TAP to make these technologies accessible to people living in low- and middle-income countries and help put an end to the pandemic.
The 11 COVID-19 technologies offered under two licences include the stabilized spike protein used in currently available COVID-19 vaccines, research tools for vaccine, therapeutic and diagnostic development as well as early-stage vaccine candidates and diagnostics. The full list of the NIH COVID-19 technologies covered in the agreement is here.
“I welcome the generous contribution NIH has made to C-TAP and its example of solidarity and sharing,” said Dr Tedros Adhanom Ghebreyesus, WHO Director-General. “Whether it’s today’s pandemic or tomorrow’s health emergency, it’s through sharing and empowering lower-income countries to manufacture their own health tools that we can ensure a healthier future for everyone.”
“We are honoured to sign these public health-driven licence agreements with NIH under the auspices of C-TAP with the goal of providing equitable access to life-saving health products for the most vulnerable in the world,” said Charles Gore, MPP Executive Director.
“NIH were the first to share their patents with MPP for an HIV product back in 2010 when we were created, and we are delighted to continue strengthening our partnership. It is clear that MPP’s model works across different health technologies.”
The announcement was made today by the US Government at the second Global COVID-19 Summit, co-hosted by the United States, Belize, Germany, Indonesia and Senegal.
Licensing the NIH technologies to MPP under the auspices of C-TAP will allow greater access to these technologies and hopefully lead to the development of commercial products that can address current and future public health needs. In most circumstances, NIH will not collect royalties on sales of products licensed in 49 countries classified by the United Nations as Least Developed Countries.
Launched in 2020 by the WHO Director-General and the President of Costa Rica, and supported by 43 Member States, C-TAP aims to facilitate timely, equitable and affordable access to COVID-19 health products by boosting their production and supply through open, transparent and non-exclusive licensing agreements. MPP provides the licensing expertise to this initiative and holds the licences.
The 11 technologies include:
WHO and MPP announce agreement with NIH for COVID-19 health technologies
12 May 2022 News release Geneva Reading time: 2 min (499 words)
WHO’s COVID-19 Technology Access Pool (C-TAP) and the Medicines Patent Pool (MPP) today finalized a licensing agreement with the United States National Institutes of Health (NIH) for the development of several innovative therapeutics, early-stage vaccines and diagnostic tools for COVID-19.
The licenses, which are transparent, global and non-exclusive, will allow manufacturers from around the world to work with MPP and C-TAP to make these technologies accessible to people living in low- and middle-income countries and help put an end to the pandemic.
The 11 COVID-19 technologies offered under two licences include the stabilized spike protein used in currently available COVID-19 vaccines, research tools for vaccine, therapeutic and diagnostic development as well as early-stage vaccine candidates and diagnostics. The full list of the NIH COVID-19 technologies covered in the agreement is here.
“I welcome the generous contribution NIH has made to C-TAP and its example of solidarity and sharing,” said Dr Tedros Adhanom Ghebreyesus, WHO Director-General. “Whether it’s today’s pandemic or tomorrow’s health emergency, it’s through sharing and empowering lower-income countries to manufacture their own health tools that we can ensure a healthier future for everyone.”
“We are honoured to sign these public health-driven licence agreements with NIH under the auspices of C-TAP with the goal of providing equitable access to life-saving health products for the most vulnerable in the world,” said Charles Gore, MPP Executive Director.
“NIH were the first to share their patents with MPP for an HIV product back in 2010 when we were created, and we are delighted to continue strengthening our partnership. It is clear that MPP’s model works across different health technologies.”
The announcement was made today by the US Government at the second Global COVID-19 Summit, co-hosted by the United States, Belize, Germany, Indonesia and Senegal.
Licensing the NIH technologies to MPP under the auspices of C-TAP will allow greater access to these technologies and hopefully lead to the development of commercial products that can address current and future public health needs. In most circumstances, NIH will not collect royalties on sales of products licensed in 49 countries classified by the United Nations as Least Developed Countries.
Launched in 2020 by the WHO Director-General and the President of Costa Rica, and supported by 43 Member States, C-TAP aims to facilitate timely, equitable and affordable access to COVID-19 health products by boosting their production and supply through open, transparent and non-exclusive licensing agreements. MPP provides the licensing expertise to this initiative and holds the licences.
The 11 technologies include:
The 11 technologies include:
Prefusion spike proteins (Vaccine Development)
Structure-Based Design of Spike Immunogens (Research Tool for Vaccine Development)
Pseudotyping Plasmid (Research Tool for Vaccine Development)
ACE2 Dimer construct (Research Tool for Drug Development)
Synthetic humanized llama nanobody library and related use (Research Tool for Drug and Diagnostic Development)
Newcastle Disease Virus-Like Particles Displaying Prefusion-Stabilized Spikes (Vaccine Candidate)
Parainfluenza virus 3 based vaccine (Vaccine Candidate)
A VSV-EBOV-Based Vaccine (Vaccine Candidate)
RNASEH-Assisted Detection Assay for RNA (Diagnostic)
Detection of SARS-CoV-2 and other RNA Virus (Diagnostic)
High-Throughput Diagnostic Test (Diagnostic)
Get out now??
INO’s Intro disclaimer>>>
DISCLAIMER The Board Assistants herewith, are not licensed brokers and assume NO responsibility for the actions, investment decisions, and or messages posted on this forum.
• We do NOT recommend that anyone buy or sell any securities posted herewith. Any trade entered into risks the possibility of losing the funds invested.
No it didn’t hit ten cents today. We’re living in the present not the far away past.
INO Get out now, in my opinion.
It'll go under a dollar and then they will do a R/S to get in compliance.
I haven't had any position in INO in about seven years or more.
I recollect having no losses on INO. Certainly nothing of consequence.
I just couldn't stand to see this scam going on.
The pumpers who surrounded this stock are evil, as bad as the Inovio. The company could never have continued without their abetting.
MG
INO Here's a typical warning from me, dated December, 2021
MiamiGent
Friday, December 17, 2021 10:36:48 PM
Post# 39683 of 40210
INO The stun guy is discarded technology. Inovio picked it up at the equivalent of a bio yard sale price. Inovio has milked it for pumping PR's these many years.
Folks, stay away from Inovio. It's a failed company, masterminded by penny stock promoter, Avtar Dhillon, currently awaiting trial for stock fraud and obstruction of Justice.
MG
Father of The Inovio Scam Expose
INO I have been consistently railing against INO since July of 2013 !!
A scam !!!
https://investorshub.advfn.com/boards/boardposters.aspx?boardid=13093
INO Inovio hit .10 today (ten cents), when two most recent R/S are accounted for.
What does ten cents buy today? One of three things- a pencil, a lollipop or a share of Inovio (INO)
Nothing at the Dollar Store.
Stay away from this Pump & Dump. Bag holders are legion. Conceivably as many INO investors have died waiting 43 years for Inovio to produce one, single approved drug, vaccine or therapy as there are now dead Russian invaders. Both ugly stains on humankind!
MG
Father of The Inovio Scam Expose
Inovio's longtime CEO is out as Covid vaccine laggard throws in the towel on PhIII, pivots to booster
Stepping down to make room for Jacqueline Shea — who first joined Inovio as COO in 2019 — Kim leaves behind a legacy of boisterous claims and promises about DNA vaccines over decades and a habit of capitalizing on pandemics but no approved product to show for it.
Shea will be overseeing a change in direction as Inovio, a laggard in the Covid-19 vaccine race, shutters a Phase III trial in favor of a booster strategy.
Not sure why favoring "Booster" if they are giving up on Covid vaccine all together, what is the point ??
https://endpts.com/inovios-longtime-ceo-is-out-as-covid-vaccine-laggard-throws-in-the-towel-on-phiii-pivots-to-booster/
9:20a ET 5/11/2022 - MarketWatch
Inovio Pharmaceuticals stock price target cut to $1 from $3 at Stifel Nicolaus
Mentioned: INO
(END) Dow Jones Newswires
May 11, 2022 09:20 ET (13:20 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
Well, that was an interesting conference call.
Thank MG for the reminder. Quarterly excuse call.
……
https://ir.inovio.com/investors-and-media/default.aspx
INO Inovio hit .1650 today (sixteen and a half cents), when two most recent R/S are accounted for.
Stay away from this Pump & Dump!
Hundreds and hundreds of INO investors have died waiting 43 years for Inovio to produce a single approved drug.
MG
Father of The Inovio Scam Expose
Information for new readers/investors which should do their own DD and find out this company
- has an impressive list of partners https://www.inovio.com/about-inovio/partnerships/overview/
- has an impressive leadership team https://www.inovio.com/about-inovio/leadership/
- has an impressive board of directors https://www.inovio.com/about-inovio/board-of-directors/
Be aware of the false rumors
- Avtar Dhillon never was INO's founder https://www.wsj.com/market-data/quotes/INO/company-people/executive-profile/77702278
- Avtar Dhillon is not a company consultant nor has he any ties with the company since several years.
- INO is trading on the Nasdaq and is not a penny stock since more than 7 years
- INO 4800 has not FDA failed phase two as they rteceived clearance for phase three https://ir.inovio.com/news-releases/default.aspx
TT the exposer of false statements.
False information Avtar Dhillon never was INO’s founder
Inovio!-Wow!- https://www.linkedin.com/posts/jpeocbrnd_jpeocbrnd-cbrn-biotechnology-activity-6925813173111410690-Nf-G
Meet the CELLECTRA 3PSP. This handheld, battery-operated device opens cells with electrical pulses to better deliver DNA vaccines. It can deliver 100 doses with a single battery charge and is designed for easy transport. Our JPL CBRND Enabling Biotechnologies team works with INOVIO Pharmaceuticals, Inc., the creator of the CELLECTRA 3PSP, to support the large-scale manufacturing of this tool.
CBRN warfighters often serve in environments without electricity or medical centers. The CELLECTRA 3PSP is designed to function reliably in these environments and can be stockpiled without maintenance, ensuring that our warfighters can get the vaccines they need anywhere.
#JPEOCBRND #CBRN #Biotechnology #vaccines
Denmark becomes the first country to halt its Covid vaccination program
………
https://www.cnbc.com/amp/2022/04/28/denmark-the-first-country-to-halt-its-covid-vaccination-program.html
DNA-Based Vaccines
DNA vaccine, yet another type of nucleic acid vaccine, is made from DNA encoding antigen protein. Unlike traditional vaccines, DNA vaccines are designed to inject a specific naked DNA code of the pathogen directly into the human body (219, 238). Once inside, it can be transcribed into mRNA in the nucleus and translated into antigen in the cytoplasm, thereby inducing the body to produce an immune response (239). Thus, vaccinated individuals obtain corresponding immune protection and disease prevention capabilities (240). Interestingly, its rapid technological development may create a whole new generation of immunologic tools. Multiple DNA vaccines are currently under development, including malaria, influenza, rotavirus, HIV, and so on. Furthermore, many of them have already entered CTs (241–243). DNA vaccination offers more potential advantages, compared to traditional vaccines, including stimulation of the B and T cell immune responses, stabilization of the vaccine, avoidance of any infectious agents, and ease of large scale production (244). More importantly, a large-scale COVID-19 vaccination is under rapid execution around the world, and the “stability” that determines whether a vaccine is convenient for transportation, and storage is a major indicator of vaccine feasibility (245). Compared to RNA vaccines, DNA vaccines can be stored for a longer period of time at the same refrigeration temperature, or even for a longer time at room temperature (246). Once successfully developed and put into use, this will likely bring great convenience to the mass production of vaccines. However, DNA vaccines usually exhibit low immunogenicity, and must be inoculated through delivery devices (such as, electric perforators) to be effective, which also limits their use.
Due to the COVID-19 pandemic outbreak, numerous DNA vaccine candidates entered CTs. According to the information from ICTRP, three of them entered phase III CTs, namely INO-4800, AG0302-COVID19, and, ZyCoV-D. INO-4800, developed by INOVIO Pharmaceuticals, is a DNA vaccine candidate for the prevention of the new coronavirus (243, 247). After the new coronavirus gene sequence was released, INOVIO employed a proprietary DNA drug platform to quickly design the INO-4800 vaccine (247). Pre-CT revealed that INO-4800 strongly induces SARS-CoV-2 specific antibody and T cell responses in mice and guinea pigs, hence, it was quickly approved for CTs (248). Based on the phase I CT results, INO-4800 is immunogenic in all subjects, and effectively produces humoral immune and/or cellular responses (145). Currently, the INO-4800 vaccine is simultaneously in both phase II/III CTs, and its safety and effectiveness reports are worthy of attention. AG0301-COVID19, a DNA vaccine developed by Osaka University/AnGes/Takara Bio, expresses the full-length S protein upon host cell entry. In a pre-CT, AG0301-COVID19, with an aluminium-containing adjuvant, strongly stimulated neutralizing antibody production, and enhanced T cell responses in rats, with no toxic response to body organs. Yet another COVID-19 DNA vaccine candidate is AG0301-COVID19, also developed by the Osaka University/AnGes/Takara Bio. The safety and effectiveness of the AG0301-COVID19 vaccine was examined in phase I/II CTs, but the results are not yet reported (243). It is worth noting that the phase III CTs are in progress at the present time. Recently, India urgently authorized and approved the listing of a DNA COVID-19 vaccine ZyCov-D that consists of a DNA plasmid vector harboring the S protein genetic code. A pre-CT study revealed that ZyCov-D elicits marked antibody and Th-1 responses, as demonstrated by augmented IFN-c expression (249). In a phase I CT, the ZyCov-D vaccine, which is administered by pressing a needle-free device on the skin, was reported to be safe, well tolerated, and immunogenic (142). The device creates a tiny stream of high-pressure liquid that pierces the skin surface, thus causing less pain for the recipient. ZycoV-D is currently conducting a large-scale phase III CT in India, involving tens of thousands of subjects, but the data is not public yet. However, due to its high efficiency and safety, India urgently authorized the application of ZycoV-D. Thus far, this is not only the first DNA vaccine for COVID-19, but also the first DNA vaccine in the world. Till date, no DNA vaccine completed phase III trials or received approval.
https://www.frontiersin.org/articles/10.3389/fimmu.2022.843928/full
Fauci says U.S. is transitioning out of 'pandemic phase'
……..
https://www.nbcnews.com/news/amp/rcna26236
INO It is very important for investors, especially newbees, to made aware of what a scam INO has been these many years. And that Inovio's Mr Everything, Avtar Dhillon, according to government charges, is a huge scammer.
I feel very validated after years of being one of the few speaking out against Inovio.
INO has caused so much financial heartache for often desperate people, lured in by Inovio's slick PR machine, that Inovio should have been delisted years ago, in my opinion.
MG
Father of The Inovio Scam Expose
NOWHERE in this article does it mention any reference to Avtar Dhillion or Inovio Pharmaceutical Co. Just because you didn't score $$ on this stock you've decided to continually bash it. MOVE ON !!
Sounds like Kim - raising funds, selling shares…
……..
https://emerald.life/emerald_leadership/avtar-dhillon/
INO News story about Avtar Dhillon,
Dhillon was an Inovio (INO) Founder as well as Inovio President, Inovio CEO, Inovio Director and Inovio Chairman & highly Paid Consultant to 2019.
https://biv.com/article/2021/11/time-ticking-vancouver-man-charged-1-billion-international-stock-fraud-scheme
INO, the company which claimed to be uniquely positioned to solve an array of world pandemics, closed today at the low of day, $2.82.
$2.82 equates to eighteen cents (.18) a share, when the most recent two reverse split are accounted for.
Inovio (INO) has never originated an approved product in its forty-three (43) corporate years of fleecing Newbees of their hard earned money with slick, misleading PRs, hundreds and hundreds and hundreds of them. No reputable pharma acts that way. Jim Cramer described Inovio as "publicity rich."
A small consolation for investors who were duped is the knowledge that one of Inovio's founders, Inovio President, Inovio CEO, Inovio Director and Inovio Chairman & highly Paid Consultant to 2019, Avtar Dhillon, was arrested in August 2021 by a joint force, charged with multiple counts of Stock Fraud, Conspiracy and Obstruction of Justice related to other stocks he was also pumping.
Stay far away from INO, perhaps the longest running stock scam in the stock market.
MG
Father of The Inovio Scam Expose
Years Railing Against This Scam
https://cannabislifenetwork.com/vancouver-dr-avtar-singh-dhillon-billion-dollar-fraud-case/
INOVIO to Report First Quarter 2022 Financial Results on May 10, 2022
This ought to be another big disappointment.
This company really sucks. With all this company is supposedly doing and the share price sits in the $2-3 range. Garbage
Lakers, everything / news as been very quite from Inovio. Are you hearing of any new developments or anything of interest? Thanks in advance.
Any update on this?
Japan officially approves Novavax COVID-19 vaccine
……….
https://www3.nhk.or.jp/nhkworld/en/news/20220419_19/amp.html
………
Just a memory: https://www.reuters.com/article/idCNL4N2IJ37E
INO My message to the SEC- "Delist this scurrilous pump & dump! "
INO Broke under $3.00,. Now $2.98, LOD.
Pump and Dump.
All were amply warned by moi.
MG
Father of The Inovio Scam Expose
INO Inovio is hit .1875 (eighteen & three quarters cents) today, when two most recent R/S are accounted for.
Stay away from this Pump & Dump. Bag holders are legion. INO investors have died waiting 43 years for Inovio to produce one, single approved drug, vaccine or therapy.
And there are plenty of sore, disgruntled holders still alive. You don't want to be one of them.
MG
Father of The Inovio Scam Expose
False information
INO Oh, he most certainly was one of Inovio's founders. You could also say notorious Dhillon was an architect of Inovio. You could even go so far as to say he is the Father of 43 Year Scam Inovio, just as you may call me the Father of The Inovio Scam Expose.
I'll repeat my post in case you stumbled over the wording. I said Dirty Dhillon was a Founder.
All along your DD has been porous on Inovio and is now, too.
INO And there's the dump. Adding bagholders, already legion in number.
Inovio (INO) is a 43 year, pump & dump that has not originated a single approved product in all its years..
Avtar Dhillon, an Inovio founder, as well as its past President, CEO, Director, and Chairman & highly Paid Consultant to 2019 is currently charged with Stock Fraud as well as Obstruction of Justice.
Only the unscrupulous play this P&D penny stock*.
MG
Father of The Inovio Scam Expose
* when the most recent two Reverse Splits are factored for, INO is presently valued at at .20/SH (20 cents a share). It's a penny stock.
Avtar Dhillon never was Inovio’s founder. Telling this is spreading false information.
UPenn Researchers Enrolling BRCA1/2 Carriers into Inovio hTERT Cancer Vaccine study
Apr 11, 2022 | Caroline Hopkins
NEW ORLEANS — Researchers at the University of Pennsylvania in collaboration with the drugmaker Inovio have launched a Phase I clinical trial assessing whether a cancer vaccine, dubbed INO 5401, can prevent high-risk breast cancer patients with BRCA1/2 germline mutations from relapsing and healthy individuals with these mutations from developing cancer at all.
Susan Domchek, executive director of the Basser Center for BRCA at the University of Pennsylvania, presented the trial details during the American Association for Cancer Research's annual meeting on Saturday. The study is enrolling participants to two groups: Cohort A includes 16 patients with BRCA1 or BRCA2 mutations who have had a prior diagnosis of invasive breast or ovarian cancer, pancreatic cancer, or prostate cancer and have completed adjuvant therapy with no clinical evidence of disease. Cohort B will include 28 healthy participants carrying BRCA1 or BRCA2 mutations.
The immuno-interception strategy that Domchek and her team are evaluating in the trial involves Plymouth Meeting, Pennsylvania-based Inovio's INO 5401, a DNA plasmid encoding hTERT, WT-1, and PSMA, alone or together with INO-9102, which encodes the gene for IL-12.
Following the vaccine, participants are given a small electric charge via an electroporation device called the Cellectra 2000 to increase the amount of vaccine taken up by muscle. Both cohort A and cohort B are separated into two treatment arms: one arm will receive INO 5401 followed by electroporation and one arm will received INO 5401 and INO 9102 together followed by electroporation.
Positive data from both the metastatic breast cancer setting and from a Phase I clinical trial of the strategy for solid tumor patients at a high risk of relapse offered the rationale for the Basser Center study to assess the feasibility for non-surgical prevention.
That earlier study, dubbed TRT-001, was published recently in the Journal for Immunotherapy of Cancer. Among 93 patients with breast, ovarian, and pancreatic cancers, among others, an earlier iteration of the Inovio vaccine, INO-1400, was not only immunogenic, but also safe. Immune responses were observed across all tumor types, and the vaccine increased a CD8-positive phenotype associated with improved overall survival in pancreatic cancer patients. Patients experienced very few side effects, save local injection site reactions, which was a key consideration for moving the vaccine into trials of healthy individuals.
At Basser, Domchek and colleagues have already completed enrolling cohort A part one and are actively enrolling cohort B. "Soon, we will be vaccinating healthy BRCA1 and BRCA 2 carriers," she said, adding that there "seems to be great interest from healthy carriers who want to come on this study." Cancer vaccines typically have not been tested in early-stage or adjuvant trials, let alone intervention trials for germline mutation carriers who are at high risk of developing cancer, but who do not actually have a diagnosis.
Clinical trials evaluating drugs as a means to prevent or intercept, rather than treat, cancers are challenging and lengthy. Because the endpoints for these trials are often related to whether a patient goes on to develop malignancies far down the line, the trials can take decades or more. For this reason, prevention trials often lack adequate funding and drugmaker support. But researchers like those in Domchek's lab are increasingly determined to tackle them.
The Inovio-UPenn vaccination study is designed to evaluate the cancer vaccine's safety and dose-limiting toxicities as well as antigen-specific immune response over a period of two years, though it will take much longer to show the intervention's ability to prevent cancer development.
"We've considered cancer interception for years, but now I would argue that we actually have the tools to translate it," Domchek said during a plenary session at the meeting on Saturday. "There are multiple viable strategies for non-surgical prevention in BRCA1 or BRCA2 carriers that are being tested or have a good opportunity to be tested."
Because BRCA1 and BRCA2 carriers are at high-risk for a number of the most common malignancies, including breast, ovarian, pancreatic, and prostate cancer, proving the success of an interception strategy could not only have sweeping implications, but also be generalizable to future efforts in other high-risk populations and cancer types, Domchek suggested.
Moving PARPi into the prevention space
Other interventional possibilities could be on the horizon, too, when it comes to breast cancer prevention. For example, Merck and AstraZeneca's PARP inhibitor Lynparza (olaparib) has demonstrated efficacy in the adjuvant treatment setting for early-stage, high-risk breast cancer patients harboring BRCA1 or BRCA2 mutations. The drug recently secured US Food and Drug Administration approval for the adjuvant treatment of HER2-negative, high-risk BRCA1/2-positive breast cancer, based on an event-free survival benefit seen in the large, Phase III randomized OlympiA trial.
As Domchek suggested during her presentation, the fact that the PARP inhibitor has been effective for preventing recurrence following initial treatment for this high-risk, early-stage patient population raises the possibility that Lynparza might be an alternative to prophylactic surgery for healthy BRCA1/2 carriers.
As is the case across much of drug development, Merck and AstraZeneca began by evaluating Lynparza in the metastatic disease setting and then gradually moved the agent into earlier treatment lines from there. "We've gone from metastatic to adjuvant," Domchek said of PARP inhibitors. "Can we go to prevention? I don't know."
She pointed to several early "clues" that the strategy may be worth pursuing. For example, patients in the OlympiA trial who received Lynparza were less likely to develop second primary cancers than patients in the control arm. Indeed, 19 patients receiving Lynparza developed second primary cancers versus 32 patients who received a placebo. The numbers are not statistically significant, and it will take more time to see if the trend continues, but the signal is encouraging, Domchek said.
Within cancer prevention trials it is critical to balance potential benefit against the risk of toxicities. The fact that Lynparza in OlympiA did not appear to cause secondary cancers such as myelodysplastic syndromes or acute myeloid leukemia, as some other treatments do, and had a relatively manageable toxicity profile, is also encouraging for the potential of PARP inhibitors to be used for prevention.
"Compared to placebo [in OlympiA], there was no overall effect on quality of life with olaparib," Domchek added. "There are some differences with nausea and fatigue, but [managing that] is really a question of what should a schedule look like and what is considered tolerable for patients."
"We need to answer many of these questions," she continued. "There are a lot of people thinking about these issues and how we can move forward."
While drugmakers have not announced any concrete plans to evaluate Lynparza as a cancer interception strategy for healthy BRCA1/2 carriers, Domchek seemed to suggest that such a trial does not seem out of the realm of possibilities, especially in light of the high cost and quality-of-life implications of prophylactic mastectomies, oophorectomies, and other such invasive procedures too often presented as the sole option for high-risk BRCA1/2 carriers.
"I admit that large-scale interception studies are difficult, even in high-risk patients," she acknowledged. "They are large, they take a long time … but that does not mean we shouldn't do them. At the end of the day, this is what we want to do: intercept cancers and change people's lives."
https://www.precisiononcologynews.com/cancer/upenn-researchers-enrolling-brca12-carriers-inovio-htert-cancer-vaccine-study?utm_campaign=Precision+Oncology+News+Welcome+Email&utm_medium=email&CSAuthResp=1649718133700%3A0%3A2514068%3A0%3A24%3Asuccess%3A40EC667EE29FABF7195C9F6B1DFF7A91&_ga=2.8889842.658874540.1649717985-1590207770.1649717985&adobe_mc=MCMID%3D58018332632319430302833232159497549527%7CMCORGID%3D138FFF2554E6E7220A4C98C6%2540AdobeOrg%7CTS%3D1649718003&utm_source=Sailthru&CSAuthReq=1#.YlSzeiVHYlQ
What ever happened to general Gus Perna and Inovio?
“the maker of the Medigen vaccine looked towards the results of the World Health Organization’s Solidarity Trial Vaccines clinical study—a test to evaluate the effectiveness of vaccines around the world. The results are scheduled to be published in early April.” 4/6/22
That should include INO-4800 as well.
psuvanguard.com/medigen-vaccine-controversial-or-helpful/
“Medigen is now waiting for the results of the WHO’s Solidarity Trial Vaccines clinical study, which evaluates the effectiveness of vaccines from around the world.
If the trial proceeds as scheduled, the results are expected by the end of next month or in early April, Medigen chief executive officer Charles Chen said”
taipeitimes.com/News/front/archives/2022/02/16/2003773205
INO And there's the dump. Adding bagholders, already legion in number.
Inovio (INO) is a 43 year, pump & dump that has not originated a single approved product in all its years..
Avtar Dhillon, an Inovio founder, as well as its past President, CEO, Director, and Chairman & highly Paid Consultant to 2019 is currently charged with Stock Fraud as well as Obstruction of Justice.
Only the unscrupulous play this P&D penny stock*.
MG
Father of The Inovio Scam Expose
* when the most recent two Reverse Splits are factored for, INO is presently valued at at .22/SH (22 cents a share).
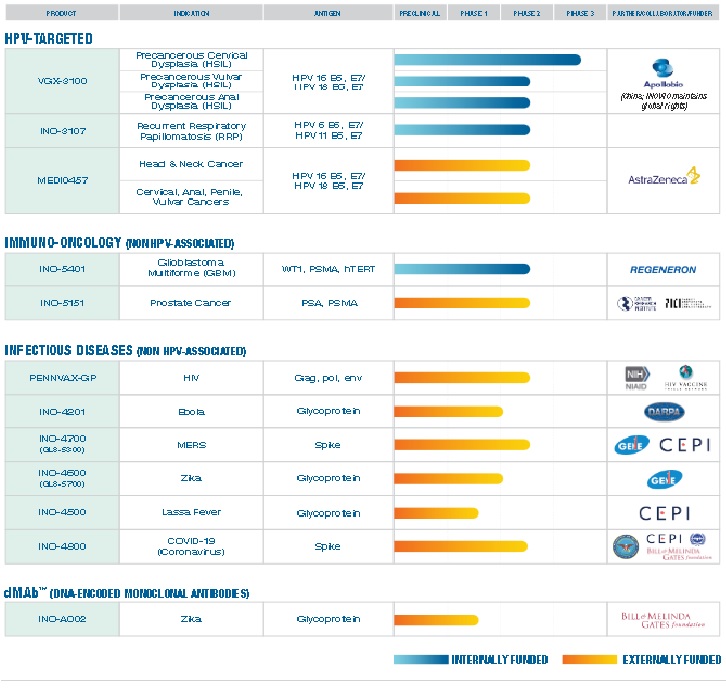
UCSF Brain Tumor Center: This clinical trial will evaluate the safety and efficacy of INO-5401, INO-9012, and cemiplimab, alongside radiation therapy and temozolomide (TMZ), in treating patients with newly diagnosed glioblastoma.
INO-5401 and INO-9012 are comprised of DNA molecules, or plasmids, and are injected intramuscularly and then delivered using electroporation – a technique using electric pulses to push the DNA plasmids into cells. The cells that are electroporated will begin producing specific molecules based on the instructions in the DNA plasmid. For example, cells electroporated with INO-5401 will begin producing antigens that are common in many cancer cells (including WT1, PSMA, and hTERT). Production of these antigens helps activate T-cells to better recognize cancer cells. INO-5401 is therefore considered a T-cell activating immunotherapy.
INO-9012 is another immunotherapeutic agent, a DNA plasmid encoding the human interleukin-12 (IL-12) gene. Cells electroporated with INO-9012 will begin producing IL-12, a protein that promotes the activation of natural killer cells, among other immune-stimulating effects.
Cemiplimab is an antibody-based immunotherapy, administered intravenously, that also targets the immune system and activates it to stop cancer growth and/or kill cancer cells. More specifically, cemiplimab works by binding and inhibiting PD-1, a protein that normally protects the body from attacking itself. Normally, PD-1 works by detecting a molecular signal (called PDL-1) made by various cells across the body. Some cancer cells take advantage of this protection mechanism making the protective signal themselves. Blocking PD-1 with cemiplimab allows the immune system to activate its T-cells and recognize these tumors as cells to be attacked.
Eligible participants will be separated into two cohorts, depending on MGMT promoter methylation status. Both cohorts will receive the INO-5401, INO-9012, and cemiplimab at the same doses and schedule, in addition to radiation therapy and TMZ, if clinically indicated:
Experimental Cohort A (Unmethylated MGMT Promoter): INO-5401 + INO-9012 + Cemiplimab + RT + TMZ
Experimental Cohort B (Methylated MGMT Promoter): INO-5401 + INO-9012 + Cemiplimab + RT + TMZ
Actual Study Start Date :
May 31, 2018
Estimated Primary Completion Date :
June 30, 2022
Estimated Study Completion Date :
June 30, 2022
21 study locations
United States, California
City of Hope
Duarte, California, United States, 91010
Stanford University, School of Medicine
Palo Alto, California, United States, 94304
University of California, San Francisco
San Francisco, California, United States, 94143
United States, Florida
University of Miami - Sylvester Comprehensive Cancer Center
Miami, Florida, United States, 33136
Moffitt Cancer Center
Tampa, Florida, United States, 33612
United States, Georgia
Emory University School of Medicine
Atlanta, Georgia, United States, 30322
United States, Massachusetts
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02215
United States, Michigan
Henry Ford Health System
Detroit, Michigan, United States, 48202
United States, New Jersey
Rutgers University - Cancer Institute of New Jersey
New Brunswick, New Jersey, United States, 08901
United States, New York
New York University Langone Medical Center; Perlmutter Cancer Center
New York, New York, United States, 10016
Icahn School of Medicine at Mount Sinai
New York, New York, United States, 10029
Columbia University Medical Center The Neurological Institute of New York
New York, New York, United States, 10032
New York-Presbyterian Hospital/Weill Cornell Medical Center
New York, New York, United States, 10065
United States, North Carolina
University of North Carolina School of Medicine
Chapel Hill, North Carolina, United States, 27599
United States, Oklahoma
Stephenson Cancer Center
Oklahoma City, Oklahoma, United States, 73104
United States, Oregon
Oregon Health & Science University
Portland, Oregon, United States, 97239
United States, Pennsylvania
University of Pennsylvania Health System: Penn Medicine
Philadelphia, Pennsylvania, United States, 19104
UPMC Cancer Center Neuro-Oncology; UPMC Cancer Pavilion
Pittsburgh, Pennsylvania, United States, 15232
United States, Texas
Texas Oncology
Austin, Texas, United States, 78705
Baylor College of Medicine
Houston, Texas, United States, 77030
United States, Utah
Huntsman Cancer Institute
Salt Lake City, Utah, United States, 84112
Monday, April 04, 2022, at 18:53- Colombia Conducts Trials to Determine Effectiveness of New COVID-19 Vaccine. The World Health Organization (WHO) has been working with a group of experts in order to select from all the vaccines that are in phases of research I and II, those that have the greatest results in efficacy and safety. "The objective has been to be able to carry out a trial that allows them to be compared with each other and to have immunizers that are effective towards variants of the virus that are now and that may appear later," Carlos Álvarez, national coordinator of studies on COVID-19 in Colombia, said on previous occasions. The Ministry of Health and Social Protection (MinSalud) has announced that it is accompanying the study in eight cities in the country. Many of the current vaccines are found with patents that do not allow their adequate distribution in the world. It should be remembered that, the WHO has been developing a vaccine to bring it to the world in an equitable way. In this way, once we are all vaccinated, this pandemic can be terminated. Likewise, the Clinical Study 'Vaccine Solidarity' is being evaluated in the eight cities of the country evaluating through volunteers, how safe the new vaccines against COVID-19 are, with the accompaniment of MinSalud. "This new vaccine is safe. It has been verified with most of the participants we have had in Bogotá. Of 80 people, some have had a headache or malaise that resolves spontaneously. We have had no serious or severe adverse effects. And no one has had to be taken to the emergency room," said Freddy Carreño, a researcher at the Solidarity Study.
12/15/21 Inovio vaccine arrives in Veracruz, with immunity in 24 hours
from the 1st dose
The biological Inovio arrives in Veracruz as a high-tech and effective alternative against Covid-19, for this reason, Training for Comprehensive Consulting for the Clinical Research Center (FAICIC) will have 250 antigens to apply to the same number of 18-year-olds in forward that they have never been vaccinated against the coronavirus, nor are they pregnant or currently breastfeeding; as well as those who suffer from chronic diseases such as: diabetes, hypertension, cancer, HIV, obesity, allergies, among others.
The general director of the Faicic Clinical Research Center, Sharzy Molina Guízar, explained that it is a very safe antigen, which has the characteristic that it is not injected, but through a shot in the arm that will impact at the cellular level, through a electrode, since it has a very different molecular composition that makes it different from those already known, because it travels at the cellular level, causing immunity in 24 hours, for longer and with greater efficiency.
“Previous vaccines were given through an injection in the arm; this also goes on the arm, but it is done by means of a device that is like a small pistol, which what it does is that here in the arm, in the muscle, a shot is fired that does not cut, that does not open the skin, it does not hurt, it is painless and this allows it to travel to the cellular level and how is it that we achieve immunity in 24 hours? We achieve it because it travels so fast in the cells that what it does is reproduce, they cause an excitation to the cells that makes one infect the other very quickly and, then, we can go from one to 10, from 10 to 100 , from 100 to a thousand and not only do we cause immunity for a longer time, but also, much more effectively”.
He explained that Inovio decided to do it this way, since the existing vaccines that are applied in the injected arm, their route at the muscular level generated immunity from 28 to more than 40 days, depending on the organism, due to its composition and its journey in the body, which also caused other allergic reactions or altered some parts of the body, while Inovio causes immediate immunity.
Likewise, Molina Guízar recognized that, based on studies carried out, Inovio has proven high efficacy against the Omicron variant, although investigations continue, so it is added to the list of qualities of this antigen.
“This vaccine, so far, is showing an efficacy of 95.5% in the first application that is given at 24 hours. This vaccine consists of two doses (two applications): on day zero, which is the day the vaccine is applied, and on day 28”, stated the interviewee.
The general director of Faicic admitted that there are side effects similar to those of injected vaccines, such as: headache, nausea, dizziness, weakness, fever, among others, which are treatable and will be treated by the medical staff of the center of research.
In this sense, he recognized that there is still a large amount of population not vaccinated against this disease for various reasons, such as: fear, resistance, lack of information, among others.
“After two years, we already have a lot of experience, we have seen how to improve it and we invite the population that was afraid to come because now we have a very safe vaccine; It is also aimed at those people who, for example, are allergic, who have chronic-degenerative diseases”.
Sharzy Molina highlighted that they will be working every day for the Inovio application, including holidays such as December 25 and January 1, 2022, for which she invited citizens with these characteristics to sign up for this application, since It will serve the population of Veracruz and foreigners, where in the case of the latter, they will be supported with the transfer, therefore, they require social leaders who support with this process the population of marginalized areas whose expenses are assumed by Faicic.
He added that in addition to the support for the transfer to the Faicic headquarters, patients will be provided with: medical, nutritional, psychological consultation, laboratory studies and for a year and a half they will remain in contact with them, with open consultation for any symptom or subsequent condition that they may present.
He recalled that Faicic is a research center that seeks to improve people's health and lives, through studies and in coordination with recognized pharmaceutical companies.
https://www-eldictamen-mx.translate.goog/noticias-de-veracruz/boca-ver/llega-a-veracruz-vacuna-inovio-con-inmunidad-en-24-horas-desde-la-1a-dosis/?_x_tr_sl=auto&_x_tr_tl=en&_x_tr_hl=en
4/4/22 Prime-boost vaccination regimens with INO-4800 and INO-4802 augment and broaden immune responses against SARS-CoV-2 in nonhuman primates
Inovio Pharmaceuticals, Plymouth Meeting, PA, 19462, USA
Vaccine and Immunotherapy Center, The Wistar Institute, Philadelphia, PA, 19104, USA
Received 2 November 2021, Revised 2 March 2022, Accepted 23 March 2022, Available online 4 April 2022.
Abstract
The enhanced transmissibility and immune evasion associated with emerging SARS-CoV-2 variants demands the development of next-generation vaccines capable of inducing superior protection amid a shifting pandemic landscape. Since a portion of the global population harbors some level of immunity from vaccines based on the original Wuhan-Hu-1 SARS-CoV-2 sequence or natural infection, an important question going forward is whether this immunity can be boosted by next-generation vaccines that target emerging variants while simultaneously maintaining long-term protection against existing strains. Here, we evaluated the immunogenicity of INO-4800, our synthetic DNA vaccine candidate for COVID-19 currently in clinical evaluation, and INO-4802, a next-generation DNA vaccine designed to broadly target emerging SARS-CoV-2 variants, as booster vaccines in nonhuman primates. Rhesus macaques primed over one year prior with the first-generation INO-4800 vaccine were boosted with either INO-4800 or INO-4802 in homologous or heterologous prime-boost regimens. Both boosting schedules led to an expansion of T cells and antibody responses which were characterized by improved neutralizing and ACE2 blocking activity across wild-type SARS-CoV-2 as well as multiple variants of concern. These data illustrate the durability of immunity following vaccination with INO-4800 and additionally support the use of either INO-4800 or INO-4802 in prime-boost regimens.
https://www.sciencedirect.com/science/article/pii/S0264410X22003681
(Repost) Summary by @StockHound Posted on November 01, 2021.
"In short, compared to our competitors, 4802 as a booster is displaying amazing numbers. No wonder why the WHO has chosen as 1 of 2 vaccines in the Solidarity trial.":
Here's my take on the article from BioRxiv on the INO-4800 and INO-4802 boosters.
Inovio originally conducted a Pre-Clinical study on Mice and Guinea Pigs to advance to P1 human trials. They also did a study on Non-Human Primates Rhesus Macaques monkeys. These monkeys elicit similar immune responses as humans do. During this study they found the following antibody responses:
• GMT Binding Antibodies = 258,032 @ 6 weeks (Convalesent (Covid Recovered patients) had a GMT BA = 142,140)
• GMT Neutralizing Antibodies = 175 @ 6 weeks (Convalesent had an approx. GMT NA = 150)
These numbers were also seen in the Phase 2 clinical trials using the 2 mg dose. It's noted that the Binding Antibody measurement had changed at this point, unfortunately it will be hard to compare to other vaccines providers at this point. I'm concluding that since the neutralizing antibodies were similar between the monkey study and P2, I can assume that Binding Antibodies of 258,032 from the monkey study should be close to the Binding Antibodies of P2 of 2210. INO 4800 P2 results:
• GMT Binding Antibodies = 2210
• GMT Neutralizing Antibodies = 150
Neutralizing Antibodies of a Convalesent patient was nearly 150 and Binding Antibodies of 142,140. Comparing to mRNA vaccines at this time, their GMT NA was about 343-900 (MRNA). MRNA also produced GMT Binding Antibodies of 782,719 for the 100ug dose.
Coming back to the most recent booster article, here are the numbers at weeks 43-64, essentially 1 year since the 2nd dose on these monkeys.
4800:
• GMT Binding Antibodies: Wild Card=3077, Beta=2338, Gamma=2077, Delta=21,044
• GMT Neutralizing Antibodies: Wild Card=2286, Beta=1199, Gamma=1596, Delta=785
4802:
• GMT Binding Antibodies: Wild Card=6285, Beta=6285, Gamma=6285, Delta=6285
• GMT Neutralizing Antibodies: Wild Card=3712, Beta=1452, Gamma=4389, Delta=1434
It is controversial which antibody is more important as both have an important role within the immune system and response to a pathogen. Neutralizing antibodies defend the cell and can result in lifelong immunity to the target infection. Binding Antibodies bind to the pathogen and trigger the immune system to attack it with white blood cells.
What this article is telling me, boosting the INO-4800 as a primary vaccine with either INO-4800 or INO-4802 as a booster 1 year later shows significant increases in Neutralizing Antibodies especially with INO-4802 and increase in Binding Antibodies well past what Convalescent patients have displayed. The significantly higher number Neutralizing antibodies providing an advantage to long term protection. It's worth noting that mRNA vaccine booster will also increase their Neutralizing Antibodies to an approximate GMT of 2000 or 800 for Delta. Though these numbers are similar to INO, it is good in the fact that we can achieve the same and slightly better results of a leading EUA vaccinev while providing a superior safety profile, thermo stability and shelf life.
This also doesn't include INO advantage with the production and sustainment of T, B, CD4 and CD8 cells.
In short, compared to our competitors, 4802 as a booster is displaying amazing numbers. No wonder why the WHO has chosen as 1 of 2 vaccines in the Solidarity trial.
World Vaccine Congress Washington 2022, WASHINGTON DC, Apr 21 09:40
Inovio’s DNA COVID vaccine update
COVID & Beyond - M4, Independence Salon D
Joseph Kim,Chief Executive Officer, Inovio Pharmaceuticals
Apr 18 14:00
ANTIBODIES FOR INFECTIOUS DISEASES 2pm - 5pm - M4, Liberty Salon O
ANTIBODIES FOR INFECTIOUS DISEASES - Liberty Salon O
Mark Esser,Vice President, Early Vaccines and Immune Therapies, AstraZeneca
Flonza Isa,Associate Medical Director, Regeneron Pharmaceuticals
Alina Baum,Director, Infectious Diseases, Regeneron Pharmaceuticals
Apr 20 09:00
Chair’s opening remarks
Emerging and Infectious Diseases - M4, Liberty Salon M
David Weiner,VP, Director of Vaccine & Immunology Center, The Wistar Institute
Apr 21 11:30
GT-EPIC platform to target advanced hepatocellular cancer in a patient specific manner
Cancer and Immunotherapy - M4, Liberty Salon I-K
Niranjan Sardesai,President & Chief Executive Officer, Founder, Geneos Therapeutics
Apr 20 17:10
Panel: Advances in our understanding of the key parameters of tumor epitope immunogenicity
Cancer and Immunotherapy - M4, Liberty Salon I-K
Niranjan Sardesai,President & Chief Executive Officer, Founder, Geneos Therapeutics
Apr 20 17:10
Panel: What are the risks to self-spreading viruses?
Veterinary - M4, Supreme Court Room
Stanley Plotkin,Emeritus Professor, University of Pennsylvania School Of Medicine
3/28/22 Geneos Therapeutics, a clinical stage biotherapeutics company spun out of Inovio Pharmaceuticals, raised $17 million in a private stock sale.
The 6-year-old Plymouth Meeting company's Series A2 financing was led by Flerie Invest, a European venture capital firm based in Stockholm and London that specializes in backing life sciences companies tackling major medical challenges. Existing Series A investors Santé Ventures, Korea Investment Partners Global Bio Fund, and Inovio (NASDAQ: INO) also participated. Geneos closed its initial $12 million Series A round in March 2021.
Geneos plans to use the bulk of the proceeds to expand the clinical trials for its lead new drug candidate GNOS-PV02 from 24 to 36 patients. The personalized cancer vaccine is under development as a treatment for patients with advanced hepatocellular carcinoma, a type of liver cancer.
The experimental treatment is a tumor-specific DNA plasmid-based product that is designed and manufactured for each patient based on their unique tumor mutations. The mutations are identified by sequencing each patient’s tumor. The company's GT-EPIC platform targets unique neoantigens or abnormal mutations produced by cancer cells.
The investment money will also be used to expand the company's pipeline into a new, undisclosed cancer indication. The remaining funds will be used for general company operations.
In February 2021, Geneos completed a second closing of the Series A-1 preferred stock financing, in which the Company did not participate. Following this transaction, the Company held approximately 35% of the outstanding equity, on an as-converted to common stock basis.
The Company continues to exclusively license its SynCon® immunotherapy and CELLECTRA® technology platform to Geneos to be used in the field of personalized, neoantigen-based therapy for cancer. The license agreement provides for potential royalty payments to the Company in the event that Geneos commercializes any products using the licensed technology. The Company is not obligated to use any of its assets to fund the future operations of Geneos.
“I think INO will become the standard of care for glioblastoma patients. If we’ve already extended the lives of methylated glioblastoma patients by AT LEAST 6 months median (that is what we know so far and the study is not yet over).
13,000 people diagnosed with GB each year and I know for a fact Keytruda costs $125,000 per year per patient. So if patients are living AT LEAST 2.5 years that is 32,500 patients being treated per year x $125,000 potential revenue per year (maybe we would even charge more since our treatment is more effective than what is currently being used). That is an extra $4 billion per year in revenue. That also only takes care of USA patients.
$4 billion per year revenue x .20 profit margin x 35 (PE ratio) = over $16 billion to our market cap. That’s a 10x return only for one product.” By JuliekLily5410
Patients can apply for Compassionate Use with FDA to get 5401 GBM treatment immediately.
https://www.terrapinn.com/conference/world-vaccine-congress-washington/agenda.stm?utm_source=email&utm_medium=pardot&utm_campaign=UK_10550_WVC+DC+2022_CONFPROM&utm_term=email
Lakers-W.....Great Posts....Shareholders Appreciate!
INO closed up 5.85% at $3.8 on 4/1. Able to find support at 50-dma. Bullish!
50-Day $3.52
100-Day $4.77
200-Day $6.40 likely tested in 2Q22
2Q22 Catalysts:
? Advaccine’s Homologous and Heterologous pGx9501 booster 2,4,6,8 wks Interim Readout. EUA. Preorder.
? WHO’s STV Interim Readout. Apply EUL. COVAX preorders.
? INNOVATE resumes in Philippines ????, Colombia ????, Brazil ????, Mexico ???? , starts in India ????, Rwanda ????, Tunisia ????, Thailand ????
? RRP 3107 P3 IND approval
? GBM 5401 OS36, median OS MGMT-methylated
? COVID-19 dMAb® P1 starts in May
AZD5396, AZD8076
? Report INO-4500 Lassa P1b data
“We believe this is an important
step for dMAb program. This has to do with announcement we had made in December 2020 regarding dMAbs to treat Covid19. We are working with many collaborators on this project as you know. We are hoping that this can lead to benefits not just for Covid but for our dMAbs program, in general.
Thank you
Investor Relations” 3/25/22
STV Primary outcome measure:
Virologically confirmed COVID-19 disease, through SARS-CoV2 RNA isolation and RRT-PCR amplification in oro-nasopharyngeal specimen, regardless of disease severity, at 14, 180, 365 days after the last dose.
isrctn.com/holding
INNOVATE primary endpoint changes from prevention of virologically confirmed COVID-19 disease to prevention of severe disease due to COVID-19.
Before the amendment, INNOVATE was a duplicate of STV. In the Omicron BA-1 and BA-2 era, all other vaccines have much lower infection prevention efficacy.
3/1 Hartaj Singh: Sanofi and GSK reported a vaccine efficacy of 58%, right? And they ended their trial before Omicron really hit. They’re looking for approval. Their severe disease endpoint looked pretty good. Moderna’s already talking about bivalent vaccines against Omicron.
So, it seems you’re getting proactive in trying to get ahead of what Omicron can do to current vaccines in development or approvals.
How are you thinking if you were to get that protocol amendment? Would you essentially market the vaccine on that disease severity endpoint, hospitalization endpoint, plus the risk benefit profile, the actual product profile of the vaccine?
Dr. Joseph Kim
Yes. Hi Hartaj, absolutely. That’s the view that we have. Obviously, the Omicron has thrown a curveball to all vaccine developers, with reduction in antibody responses from the original ancestral strain targeted vaccine, which, all of the approved vaccines and some of the ones in testing, including INO-4800 is. What’s great about our Omicron data as described by Kate and myself earlier is that our T cell responses, including CD8 killer T cells were fully maintained against Omicron. So, that really leads us to believe that whether we’re targeting the original variants Alpha, Beta, Gamma, Delta, or even Omicron, or even what’s next, right, the stealth Omicron or the next variant, we have full faith that our CD8 T cell responses and our overall T cell responses generated from INO-4800 is going to persist and be maintained.
So, with that in mind, we are taking a proactive step knowing that our probability of success in demonstrating prevention of severe disease with our vaccine against COVID-19 virus is high. And that’s the label that we would look for and all of the other attributes and target profile that we have mentioned earlier. We believe our INO-4800 has a strong position once we get the Phase 3 data, and once we get the emergency and full licensure to demonstrate this -- benefits as a vaccine against SARS-CoV-2.
Hartaj Singh
Yes, great. Joe, I mean, your in vitro data matches up pretty well against some of the commercial and vaccines in approval.
3/15 Hartaj Singh: Any updates on 4802 timeline? Any updates on the S. Korea trial?
JK: That was the 4800 P1. They were completed. And they were P1 trials. So, both P1 trials in S. Korea and China had results similar to our U.S. trial. It was a similarly or identically designed trial. So, we published under U.S. We would also look to publish those results along with our collaborators IVI in S. Korea and in China. We are in P3, so a lot more interest in the later stage program.
For 4802, 4803, we have a pan Covid program that we have published, completed the preclinical trials. We put the pause in 4802 in clinic, just b/c 4800 was so cost effective, both in the animal testing and clinical sample testing compared to 4802. Of course, the Omicron changed - it was a big shift until Omicron. And now we have an Omicron match construct, that’s being tested preclinically.
So, there are many research constructs that we have designed. But our lead horse is 4800. We think we have a vaccine that can provide protection across all different variants in protecting against severe diseases. And we look forward to testing that thesis from our INNOVATE trial but also demonstrate 4800’s ability to serve as a booster.
The as we develop out COVID-19 platform, once we get our first product approved, we would be able to utilize the bridging strategy that many regulators have been deploying to bridge into the new variants as necessary. But our current focus is really in getting 4800 tested and approved through emergency and full approval where appropriate.
“It is my understanding that Advaccine is reengaging the IPO pathway. I also believe they will be able to share data on their boost trial in not too distant future since enrollment had been completed.
Thanks
Investor Relations” 4/2/22
|
Followers
|
629
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
40503
|
|
Created
|
07/25/08
|
Type
|
Free
|
| Moderators | |||







DISCLAIMER The Board Assistants herewith, are not licensed brokers and assume NO responsibility for the actions, investment decisions, and or messages posted on this forum.
• We do NOT recommend that anyone buy or sell any securities posted herewith. Any trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.
Whatch out for fake comments by manipulators
https://www.griproom.com/fun/how-to-spot-fake-comments-on-stock-boards
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
