Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
HALB (GlobeNewswire, 5-26-22)): "Halberd Corp. (HALB, OTCMKTS) today demonstrated that their Patented "Extracorporeal Process" can quickly & easily REMOVE all disease-causing substances from HUMAN FECES (Excrement) --- just like from Human BLOOD, SPINAL FLUIDS, URINE, & WATER, inter alia. Then, when said FECES is RETURNED to the patients' INTESTINES, they remain completely FREE of all GASTROINTESTINAL infections, cancers, & otherwise DISEASES for the remainder of their natural lives." (Quoted in relevant part.)

"Hey, last time I saw a SCAM COMPANY like this one, it was in a SIDESHOW at a local Ringling Brothers, Barnum & Bailey, CIRCUS! Right next to the Fat Lady, the Dog-Faced Boy, and the Wild Man From BORNEO!!"
HALB: The only "RANTS", Sir, going on here are from the (scamming) HALB Company itself! And, there are NO defects whatsoever in any of my repeated AUTOMOBILE INDUSTRY metaphors, allegory plays, etc. E.g., the World's Auto Industry can fix, repair, resurrect, etc., FOREVER, any frickin' (dead) vehicle the great JAY LENO wants to COLLECT! However, can the HALB-like, Worldwide, so-called "MEDICAL SCIENCE" Industry extend JAY LENO's life span by even ONE DAY??? NO, it can NOT do that!!


In response to your rant :
“within the ORGANS with which said bodily FLUIDS interact!!!!”
Ironically, with your own words you have inadvertently shown that you fully agree with the HALB technology. Obviously, molecules in the blood : glucose, sodium , etc. interact with the body’s organs. This interaction then subsequently changes the pathology of the organs.
Simple examples :
Very high glucose in the blood , will cause diabetic ketoacidosis-destroying the kidneys and brain. Obviously, lowering the glucose in the bloodstream -whether it is done intracorporeally or extracorporeally will ameliorate the condition and save the organs.
Almost all anti-cancer therapy is based around controlling cancer related molecules (VEGF, Tumor growth factor, etc. ) which then effects the cancer pathology in the effected organs.
Alzheimer’s Disease : in all medical articles is based around the fact that there is an excess of p-Tau, beta amyloid, inflammatory cytokines, which then subsequently causes the pathology in the CNS parenchyma.
You wrote ;
If you believe HALB's crap about the Human BODY, then I can FIX your blown-out truck's TRANSY, ENGINE, & RADIATOR to boot by simply CHANGING or FILTERING the FLUIDS for same!!!!!!!
This is a ridiculous and completely wrong analogy . A truck’s engine, radiator, etc. is made out of steel, and does not have a biologic interaction with the fluids. Obviously human organs are not made out of steel, and have a direct relationship interacting with the body fluids-blood and CSF. No physician on earth will argue that the body’s tissue do not intimately interact with the blood and CSF..
Very well stated, Sir! Indeed, said "Company" very likely operates out of a MAIL-RECEIVING box at a local UPS Store. I have PASTED below just some of HALB's impressive-sounding verbiage --- but NONE of which makes ANY truly "scientific" sense whatsoever.
"Halberd Corporation is a biotechnology company and with its partners (Dr. Qiang Shawn Chen and his team at Arizona State University; Dr. William Sturrus and his team at Youngstown State University) is developing an extra-corporeal (out of the body) treatment against PTSD, CTE, Alzheimer's Disease and other virusses and diseases. With the use of antibodies the disease antigens is targeted. Using radiofrequency / laser technology the "antibody-metallic moiety-antigens complex" is destroyed/eradicated." (From the TOP of this Board.)
In FACT, NONE of the (sample) afflictions noted above can be FIXED in any fashion at all from OUTSIDE of the body. In ALL such diseases, the physical/chemical problems reside within the respective bodily ORGANS themselves. Especially within the CORRUPTED/DEFECTIVE genomes within the CELLS of such organs.
THE ULTIMATE "BOTTOM LINE": Any time you hear on the news, for example, that a 'celebrity' has DIED of this or that Human malady, in TRUTH they have died of the GROSS IGNORANCE of the Human Doctors, Nurses, & Technicians who could not fix what killed them. And more importantly, when YOU YOURSELF, Sir, appear before your OWN "Family Physician" --- and she/he tells you that you have contracted this or that disease, cancer, or whatever --- remember what I tell you today, right here in this post: In TRUTH, you have merely 'CONTRACTED' THEIR reprobate IGNORANCE!!! You see, Sir, NO Human Being have ever DIED of this or that "disease", because NO SUCH DISEASES exist!!! The ultimate disease WE ALL eventually die of is the Medical IGNORANCE of Mankind itself. (End of debate.)
I recommend that you read the works of G.W.F. HEGEL --- the greatest philosopher & otherwise THINKER of all Human history. To Hegel, Mankind ITSELF would one day become "God". HOW?? Vis-a-vis the attainment of "ABSOLUTE KNOWLEDGE", across the entire spectrum of Human inquiry.
Why YES, I do. But I can make money trading a stock like this one, without aiding in the BS put forth by it's leaders.
It's just another version of Friday Night Poker.
Without knowing as much about medicine, I too have been calling out Mr. Hartless for never quite getting any of this to market. But damn if it doesn't sound good....to some.
Not because I know the science, but because it's just a repeat of the techniques he used at BIEI for years proceeding his move to this ticker. (Because it quit working there, so needed a new gig.)
If any of their ideas were going to really benefit mankind, they would be able to market them at some point. But again, they never quite get there.
Hey, HALB Company! For your NEXT PR, why don't you claim that you have successfully removed & FILTERED/CLEANSED all of the WATER from a Human patient!! And, when you simply PUT-IT-ALL-BACK-INTO the patient, he/she was SANITIZED of all physical, mental, & even emotional disorders known to Mankind!!! RIGHT????

so you are angel from God, maybe God self.
Indeed, Sir, such Diabetes shamanism is PRIMITIVE beyond imagination --- and fixes NOTHING!!! Again, my MOTOR VEHICLE ANALOGIES are PERFECT in all respects. Can your car's RADIATOR --- full of bullet holes from a drive-by gang banger --- be REPAIRED by "EXTRA-CORPOREAL FILTERING" of the radiator FLUID??? And then pouring it back into the radiator??????

Why you are so nice to tell us?
The CURE for every single affliction which besets MANKIND can only be accomplished vis-a-vis the TOTAL reconstruction of the Human GENOME. Companies like HALB are conspicuously FRAUDULENT. If anyone here can NOT see that, well, then I need not post anything more here.
take a look at yourself. If HALB technique work well now, it is on nasdaq. Need development, so on the pink.
WRONG! Such does NOT change any aspect of any such diseased Human Organs --- any more then merely changing or filtering the OIL of a broken truck ENGINE can "fix" that engine. NOTHING that I have posted here "contradicts" the logic, science, nor TRUTH of which I speak.
WRONG! The only extant potential "CURE" for Type-1 (Insulin Dependent) DIABETES is NOT "Dialysis" but, rather, the injection of (Donor) Human PANCREAS CELLS --- at least ONE-MILLION of them --- directly just beneath the surface of such patients LIVERS (of all organs). In SOME such experimental treatments for Type-1 Diabetes, miraculous "cures" have occured; whereby a 'surrogate' Pancreas of sorts commences to grow right upon the surface of said Livers. And, they secrete life-saving INSULIN directly into the patients blood stream.
But on the other hand, do NOT listen to ANYTHING this scamming HALB Company PR's, for they are the DEVIL!
The HALB Company Fraud Revealed: Anyone who believes the conspicuous Medical NONSENSE from this Patently-SCAMMING (pun intended) HALB Company is in desperate need of a BASIC remedial course in HUMAN PHYSIOLOGY & DISEASE ETYMOLOGY from your local Elementary School. Human DISEASES do NOT emerge from the Blood, Spinal Fluid, Urine, nor Water within the Human body! Humans, of course, can CONTRACT many infectious/contagious maladies vis-à-vis said fluids. But the substantive diseases THEMSELVES, cancers, etc., begin & reside WITHIN the internal ORGANS with which said bodily FLUIDS interact. Even so-called "Cancer of the Blood" (Leukemia) resides in the BONE MARROW, SPLEEN, & DNA of the children & adults who succumb to said malady. Indeed, today's ONLY effective "therapeutic" vs. LEUKEMIA is Human BONE MARROR Transplants --- and NOT primitive Blood Transfusions, nor "Extracorporeal" FILTERING of same. (I have reported this SCAM company to the SEC, OTC, & FBI as well.)
So you do not like dollar?
lol, BTW, that was a sincere question. ( Thinking the Dr. handle was real?)
may HALb work for abnormal brain?
You wrote:
“within the ORGANS with which said bodily FLUIDS interact!!!!”
Ironically, you have inadvertently shown that you fully agree with the HALB technology. Obviously, molecules in the blood : glucose, sodium , etc. interact with the body’s organs. This interaction then subsequently changes the pathology of the organs.
Simple examples :
Very high glucose , will cause diabetic ketoacidosis-destroying the kidneys and brain.
Almost all anti-cancer therapy is based around controlling cancer related molecules (VEGF, Tumor growth factor, etc. ) which then effects the cancer pathology.
Alzheimer’s Disease : in all medical articles is based around the fact that there is an excess of p-Tau, beta amyloid, inflammatory cytokines, which then subsequently causes the pathology in the CNS parenchyma.
You wrote ;
If you believe HALB's crap about the Human BODY, then I can FIX your blown-out truck's TRANSY, ENGINE, & RADIATOR to boot by simply CHANGING or FILTERING the FLUIDS for same!!!!!!!
This is a ridiculous and completely wrong analogy . A truck’s engine, radiator, etc. is made out of steel, and does not have a biologic interaction with the fluids. No physician on earth will argue that the body’s tissue do not intimately interact with the blood and CSF.
I'm no Doctor, but doesn't dialysis just do the work that the kidneys can't do on their own? (Failing Kidneys)
It's not curing anything...is it?
HALB (GlobeNewswire): "Halberd Corp. (HALB, OTCMKTS) today demonstrated that their Patented "Extracorporeal Process" can quickly & easily REMOVE all disease-causing substances from HUMAN URINE --- just like from Human BLOOD & SPINAL FLUIDS. Then, when said URINE is RETURNED to the patients' BLADDERS, they remain completely FREE of all BLADDER and/or URINARY TRACT infections, cancers, & otherwise DISEASES for the remainder of their natural lives." (Quoted in relevant part.)

"Hey, last time I saw a SCAM COMPANY like this one, it was in a SIDESHOW at a local Ringling Brothers, Barnum & Bailey, CIRCUS! Right next to the Fat Lady, the Dog-Faced Boy, and the Wild Man From BORNEO!!"
//////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////
$HALB Halberd Successfully Eliminates Excess Levels of Inflammatory CytokinesPress Release | 05/24/2022
JACKSON CENTER, PA / ACCESSWIRE / May 24, 2022 / Halberd Corporation (OTC PINK:HALB) researchers have demonstrated a protocol which allows for the precise level of elimination of a specific targeted cytokine from a homogeneous mixture of two different cytokines in a fluid. Several adverse health conditions are caused by excess quantities of particular components in blood or cerebral spinal fluid (CSF), such as proteins, cytokines, amino acids, etc. Elimination of excessive quantities of these elements is key to minimizing damage, curtailing progressive damage, and even restoring health.
Halberd's researchers successfully reduced the level of Interleukin-4 (IL-4) in a mixture of IL-4 and Interleukin-6 (IL-6) in vitro to a predetermined level without significantly reducing other non-targeted (collateral) cytokines. This was accomplished through determining the precise level of IL-4 desired and calculating the number of antibodies needed to be chemically bonded to plastic beads to remove the predetermined excess IL-4. The fluid mixture was then flowed through a chromatography column filled with these treated beads. A 75% reduction of IL-4 from the mixture was set as the target, with the actual results being an 80% reduction - within the margin of acceptability. This was accomplished in a total time of approximately five minutes using gravity flow through the column. If determined to be necessary, a more precise control and reduced treatment time in practice can be accomplished through the use of a pump to speed the flow through the column.
Dr. Mitchell S. Felder, Halberd's Chief Technology Officer and a board-certified attending neurologist stated, "This is a spectacular accomplishment in that it allows for the fine-tuning of disease-causing proteins/target elements, which has never been accomplished previously. Using this technique, doctors will be able to precisely control the specific concentration of disease-causing target elements in blood or CSF. This could revolutionize not only medicine, but psychiatry as well, in that a psychiatrist would be able to exactly replicate the CSF picogram/ml levels of inflammatory cytokines to a desired level and quickly eliminate severe clinical depression and/or PTSD. Note that this was accomplished in minutes! Antidepressants take from weeks to months to achieve similar clinical results and often with harmful side-effects which are completely avoided with Halberd's methodology.
"This also opens the door to a quantum leap forward in cancer treatment. Chemotherapy is like trying to fix a pocket watch with a hammer. This technique, in which the precise picogram/ml level of a multitude of cancer-causing and/or related molecules (VEGF, TNF-alpha, CTLA-4, etc.), are for the first time able to be precisely controlled in order to replicate the exact, precise concentration needed to cure a cancer patient."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, added, "Halberd's patented technology has been demonstrated to be effective in selectively eliminating target elements from a homogeneous mixture of elements. This tool can be universally applied to a variety of disease conditions from PTSD/CTE, suicide ideation, neurodegenerative maladies, to blood-borne illnesses. Halberd's on-going research program will focus on elimination of pathogens from blood serum, followed by animal testing on our path to obtain FDA certification."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC PINK:HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed 20 related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our readers that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time the statements are made. These statements may address issues that involve significant risks, uncertainties and associated estimates made by management. Actual results could differ materially from current projections or implied results. Halberd Corporation undertakes no obligation to revise these statements following the date of this news release.
SOURCE: Halberd Corporation
View source version on accesswire.com:
https://www.accesswire.com/702442/Halberd-Successfully-Eliminates-Excess-Levels-of-Inflammatory-Cytokines
$HALB Halberd Successfully Eliminates Excess Levels of Inflammatory Cytokines
https://www.newsfilecorp.com/release/125039
Jackson Center, Pennsylvania--(Newsfile Corp. - May 24, 2022) - Halberd Corporation (OTC Pink: HALB) researchers have demonstrated a protocol which allows for the precise level of elimination of a specific targeted cytokine from a homogeneous mixture of two different cytokines in a fluid. Several adverse health conditions are caused by excess quantities of particular components in blood or cerebral spinal fluid (CSF), such as proteins, cytokines, amino acids, etc. Elimination of excessive quantities of these elements is key to minimizing damage, curtailing progressive damage, and even restoring health.
Halberd's researchers successfully reduced the level of Interleukin-4 (IL-4) in a mixture of IL-4 and Interleukin-6 (IL-6) in vitro to a predetermined level without significantly reducing other non-targeted (collateral) cytokines. This was accomplished through determining the precise level of IL-4 desired and calculating the number of antibodies needed to be chemically bonded to plastic beads to remove the predetermined excess IL-4. The fluid mixture was then flowed through a chromatography column filled with these treated beads. A 75% reduction of IL-4 from the mixture was set as the target, with the actual results being an 80% reduction - within the margin of acceptability. This was accomplished in a total time of approximately five minutes using gravity flow through the column. If determined to be necessary, a more precise control and reduced treatment time in practice can be accomplished through the use of a pump to speed the flow through the column.
Dr. Mitchell S. Felder, Halberd's Chief Technology Officer and a board-certified attending neurologist stated, "This is a spectacular accomplishment in that it allows for the fine-tuning of disease-causing proteins/target elements, which has never been accomplished previously. Using this technique, doctors will be able to precisely control the specific concentration of disease-causing target elements in blood or CSF. This could revolutionize not only medicine, but psychiatry as well, in that a psychiatrist would be able to exactly replicate the CSF picogram/ml levels of inflammatory cytokines to a desired level and quickly eliminate severe clinical depression and/or PTSD. Note that this was accomplished in minutes! Antidepressants take from weeks to months to achieve similar clinical results and often with harmful side-effects which are completely avoided with Halberd's methodology.
"This also opens the door to a quantum leap forward in cancer treatment. Chemotherapy is like trying to fix a pocket watch with a hammer. This technique, in which the precise picogram/ml level of a multitude of cancer-causing and/or related molecules (VEGF, TNF-alpha, CTLA-4, etc.), are for the first time able to be precisely controlled in order to replicate the exact, precise concentration needed to cure a cancer patient."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, added, "Halberd's patented technology has been demonstrated to be effective in selectively eliminating target elements from a homogeneous mixture of elements. This tool can be universally applied to a variety of disease conditions from PTSD/CTE, suicide ideation, neurodegenerative maladies, to blood-borne illnesses. Halberd's on-going research program will focus on elimination of pathogens from blood serum, followed by animal testing on our path to obtain FDA certification."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
Halberd Successfully Eliminates Excess Levels of Inflammatory Cytokines
Press Release | 05/24/2022
Jackson Center, Pennsylvania--(Newsfile Corp. - May 24, 2022) - Halberd Corporation (OTC Pink: HALB) researchers have demonstrated a protocol which allows for the precise level of elimination of a specific targeted cytokine from a homogeneous mixture of two different cytokines in a fluid. Several adverse health conditions are caused by excess quantities of particular components in blood or cerebral spinal fluid (CSF), such as proteins, cytokines, amino acids, etc. Elimination of excessive quantities of these elements is key to minimizing damage, curtailing progressive damage, and even restoring health.
Halberd's researchers successfully reduced the level of Interleukin-4 (IL-4) in a mixture of IL-4 and Interleukin-6 (IL-6) in vitro to a predetermined level without significantly reducing other non-targeted (collateral) cytokines. This was accomplished through determining the precise level of IL-4 desired and calculating the number of antibodies needed to be chemically bonded to plastic beads to remove the predetermined excess IL-4. The fluid mixture was then flowed through a chromatography column filled with these treated beads. A 75% reduction of IL-4 from the mixture was set as the target, with the actual results being an 80% reduction - within the margin of acceptability. This was accomplished in a total time of approximately five minutes using gravity flow through the column. If determined to be necessary, a more precise control and reduced treatment time in practice can be accomplished through the use of a pump to speed the flow through the column.
Dr. Mitchell S. Felder, Halberd's Chief Technology Officer and a board-certified attending neurologist stated, "This is a spectacular accomplishment in that it allows for the fine-tuning of disease-causing proteins/target elements, which has never been accomplished previously. Using this technique, doctors will be able to precisely control the specific concentration of disease-causing target elements in blood or CSF. This could revolutionize not only medicine, but psychiatry as well, in that a psychiatrist would be able to exactly replicate the CSF picogram/ml levels of inflammatory cytokines to a desired level and quickly eliminate severe clinical depression and/or PTSD. Note that this was accomplished in minutes! Antidepressants take from weeks to months to achieve similar clinical results and often with harmful side-effects which are completely avoided with Halberd's methodology.
"This also opens the door to a quantum leap forward in cancer treatment. Chemotherapy is like trying to fix a pocket watch with a hammer. This technique, in which the precise picogram/ml level of a multitude of cancer-causing and/or related molecules (VEGF, TNF-alpha, CTLA-4, etc.), are for the first time able to be precisely controlled in order to replicate the exact, precise concentration needed to cure a cancer patient."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, added, "Halberd's patented technology has been demonstrated to be effective in selectively eliminating target elements from a homogeneous mixture of elements. This tool can be universally applied to a variety of disease conditions from PTSD/CTE, suicide ideation, neurodegenerative maladies, to blood-borne illnesses. Halberd's on-going research program will focus on elimination of pathogens from blood serum, followed by animal testing on our path to obtain FDA certification."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC Pink: HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed 20 related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our readers that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time the statements are made. These statements may address issues that involve significant risks, uncertainties and associated estimates made by management. Actual results could differ materially from current projections or implied results. Halberd Corporation undertakes no obligation to revise these statements following the date of this news release.
Corporate Logo
who sell? who buy? MMs?
Insider info. Perhaps a researcher on the project.
$HALB Halberd Engages Professional Firm to Proceed with FDA FilingsPress Release | 05/18/2022
JACKSON CENTER, PA / ACCESSWIRE / May 18, 2022 / Halberd Corporation (OTC PINK:HALB) has engaged the services of mdi Consultants, Inc. (mdi) to assist Halberd in navigating the U.S. Food and Drug Administration's (FDA) complex application and certification processes. mdi will guide Halberd on the FDA application and testing protocol and assist in filing the appropriate documents and disclosures with the FDA in each stage of the process. After numerous discussions, Halberd is confident that mdi is the right choice to effectively and efficiently facilitate this significant milestone in Halberd's monumental and groundbreaking, historic journey.
Alan Schwartz, Executive Vice-President of mdi Consultants, Inc., commented, "We look forward to assisting Halberd's new technology through the FDA review process. mdi has 44 years of regulatory experience and has successfully guided over 4,000 devices/products through the FDA review process."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "Halberd recognizes that it does not have the expertise required to efficiently and effectively navigate the intracacies of the FDA's application process for new drugs and devices. The expertise that mdi brings to the table will fill that gap and speed us along in our quest to get FDA certification for our groundbreaking patented and patent-pending devices, antibodies and associated drugs."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About mdi Consultants Inc.
mdi Consultants has been providing FDA regulatory consulting services since 1978. They have successfully assisted in obtaining over 4,000 510(k) approvals for their clients on all types of medical devices. mdi has worked with companies worldwide.
About Halberd Corporation.
Halberd Corporation (OTC PINK:HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed 20 related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our readers that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time the statements are made. These statements may address issues that involve significant risks, uncertainties and associated estimates made by management. Actual results could differ materially from current projections or implied results. Halberd Corporation undertakes no obligation to revise these statements following the date of this news release.
Run around $0.06 on Tuesday
Must be a PR coming Monday.
someone buy over 2million just before close
$HALB Halberd Engages Professional Firm to Proceed with FDA FilingsPress Release | 05/18/2022
Jackson Center, Pennsylvania--(Newsfile Corp. - May 18, 2022) - Halberd Corporation (OTC Pink: HALB) has engaged the services of mdi Consultants, Inc. (mdi) to assist Halberd in navigating the U.S. Food and Drug Administration's (FDA) complex application and certification processes. mdi will guide Halberd on the FDA application and testing protocol and assist in filing the appropriate documents and disclosures with the FDA in each stage of the process. After numerous discussions, Halberd is confident that mdi is the right choice to effectively and efficiently facilitate this significant milestone in Halberd's monumental and groundbreaking, historic journey.
Alan Schwartz, Executive Vice-President of mdi Consultants, Inc., commented, "We look forward to assisting Halberd's new technology through the FDA review process. mdi has 44 years of regulatory experience and has successfully guided over 4,000 devices/products through the FDA review process."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "Halberd recognizes that it does not have the expertise required to efficiently and effectively navigate the intracacies of the FDA's application process for new drugs and devices. The expertise that mdi brings to the table will fill that gap and speed us along in our quest to get FDA certification for our groundbreaking patented and patent-pending devices, antibodies and associated drugs."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About mdi Consultants Inc.
mdi Consultants has been providing FDA regulatory consulting services since 1978. They have successfully assisted in obtaining over 4,000 510(k) approvals for their clients on all types of medical devices. mdi has worked with companies worldwide.
About Halberd Corporation.
Halberd Corporation (OTC Pink: HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed 20 related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our readers that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time the statements are made. These statements may address issues that involve significant risks, uncertainties and associated estimates made by management. Actual results could differ materially from current projections or implied results. Halberd Corporation undertakes no obligation to revise these statements following the date of this news release.
Corporate Logo
Let the truth set us free............ :)
$HALB Halberd Engages Professional Firm to Proceed with FDA Filings
https://www.accesswire.com/701668/halberd-engages-professional-firm-to-proceed-with-fda-filings
JACKSON CENTER, PA / ACCESSWIRE / May 18, 2022 / Halberd Corporation (OTC PINK:HALB) has engaged the services of mdi Consultants, Inc. (mdi) to assist Halberd in navigating the U.S. Food and Drug Administration's (FDA) complex application and certification processes. mdi will guide Halberd on the FDA application and testing protocol and assist in filing the appropriate documents and disclosures with the FDA in each stage of the process. After numerous discussions, Halberd is confident that mdi is the right choice to effectively and efficiently facilitate this significant milestone in Halberd's monumental and groundbreaking, historic journey.
Alan Schwartz, Executive Vice-President of mdi Consultants, Inc., commented, "We look forward to assisting Halberd's new technology through the FDA review process. mdi has 44 years of regulatory experience and has successfully guided over 4,000 devices/products through the FDA review process."
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "Halberd recognizes that it does not have the expertise required to efficiently and effectively navigate the intracacies of the FDA's application process for new drugs and devices. The expertise that mdi brings to the table will fill that gap and speed us along in our quest to get FDA certification for our groundbreaking patented and patent-pending devices, antibodies and associated drugs."
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
$HALB Halberd Submits Audited Financials; Files Form-10 as First Step to Up-list to OTCQBPress Release | 05/12/2022
JACKSON CENTER, PA / ACCESSWIRE / May 12, 2022 / Halberd Corporation (OTC PINK:HALB) submitted its fully audited financials through its fiscal second quarter and Form-10 to the SEC as its initial step to up-list from OTC-PINK to OTCQB on the OTC Markets exchange. Once Halberd's shares are qualified under the Securities Exchange Act of 1934 (the "34 Act"), Halberd will become a fully reporting issuer pursuant to Section 12(g) of the '34 Act. The OTCQB index is intended for development/venture stage companies and requires the Company to submit audited financials which adhere to GAAP standards and the company must be current with its SEC filings/reports -- Form 10-K Annual (audited), Form 10-Q Quarterly (unaudited), and the occasional Form 8-K .
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "We see this as a major step forward for the corporation. Having fully audited filings lends credibility to the company as well as confidence to our investors. This is another example of Halberd's team meeting its stated goals. We have been very open in our statement of quarterly goals vs. our accomplishments, and intend to continue this practice going forward.
"Halberd's technical accomplishments, in our opinion, are unparalleled, and we believe, could revolutionize the medical industry because our patented process operates extracorporeally (outside the body). This medical procedure can be applied safely and without any of the harmful side-effects associated with drugs injected or ingested. Halberd's patented and patent-pending technology also has the advantage to address neurological diseases by extracting the pathophysiologic basis (cause) of the disease from cerebral spinal fluid, or from blood in the case of blood-borne diseases. We have been very active and transparent in reporting our scientific and business-related achievements to two separate audiences: #1) Our stockholders and #2) the scientific community consisting primarily of larger pharmaceutical firms."
Hartman added, "Halberd continues to advance its technology via initiation of laboratory testing on blood, animal testing, which is currently in the planning stage, followed by human clinical trials, and ultimately, via direct application to the FDA for certification. If Halberd Corporation is awarded a pending government contract, it would expedite the program timeline significantly. The entire process of testing and certification could be accelerated via partnering with larger organizations who have to answer two basic strategic questions:
What if we do partner with Halberd Corporation? and,
What if we don't and our competitors do?"
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC PINK:HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed twenty related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our readers that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time the statements are made. These statements may address issues that involve significant risks, uncertainties and associated , estimates made by management. Actual results could differ materially from current projections or implied results. Halberd Corporation undertakes no obligation to revise these statements following the date of this news release.
SOURCE: Halberd Corporation
View source version on accesswire.com:
https://www.accesswire.com/701057/Halberd-Submits-Audited-Financials-Files-Form-10-as-First-Step-to-Up-list-to-OTCQB
What we're waiting for: * the step-by-step scenario to convert the IP into a commercial product
What we're waiting for: * partner news
What we're waiting for: * scientific article for publication by Dr. Chen (ASU)
What we're waiting for: * government contract
Halberd Submits Audited Financials; Files Form-10 as First Step to Up-List to OTCQB
Press Release | 05/12/2022
Jackson Center, Pennsylvania--(Newsfile Corp. - May 12, 2022) - Halberd Corporation (OTC Pink: HALB) submitted its fully audited financials through its fiscal second quarter and Form-10 to the SEC as its initial step to up-list from OTC-PINK to OTCQB on the OTC Markets exchange. Once Halberd's shares are qualified under the Securities Exchange Act of 1934 (the "34 Act"), Halberd will become a fully reporting issuer pursuant to Section 12(g) of the '34 Act. The OTCQB index is intended for development/venture stage companies and requires the Company to submit audited financials which adhere to GAAP standards and the company must be current with its SEC filings/reports -- Form 10-K Annual (audited), Form 10-Q Quarterly (unaudited), and the occasional Form 8-K .
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "We see this as a major step forward for the corporation. Having fully audited filings lends credibility to the company as well as confidence to our investors. This is another example of Halberd's team meeting its stated goals. We have been very open in our statement of quarterly goals vs. our accomplishments, and intend to continue this practice going forward.
"Halberd's technical accomplishments, in our opinion, are unparalleled, and we believe, could revolutionize the medical industry because our patented process operates extracorporeally (outside the body). This medical procedure can be applied safely and without any of the harmful side-effects associated with drugs injected or ingested. Halberd's patented and patent-pending technology also has the advantage to address neurological diseases by extracting the pathophysiologic basis (cause) of the disease from cerebral spinal fluid, or from blood in the case of blood-borne diseases. We have been very active and transparent in reporting our scientific and business-related achievements to two separate audiences: #1) Our stockholders and #2) the scientific community consisting primarily of larger pharmaceutical firms."
Hartman added, "Halberd continues to advance its technology via initiation of laboratory testing on blood, animal testing, which is currently in the planning stage, followed by human clinical trials, and ultimately, via direct application to the FDA for certification. If Halberd Corporation is awarded a pending government contract, it would expedite the program timeline significantly. The entire process of testing and certification could be accelerated via partnering with larger organizations who have to answer two basic strategic questions:
What if we do partner with Halberd Corporation? and,
What if we don't and our competitors do?"
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC Pink: HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed twenty related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
https://www.otcmarkets.com/stock/HALB/news/Halberd-Submits-Audited-Financials-Files-Form-10-as-First-Step-to-Up-List-to-OTCQB?id=356750
$HALB Halberd Submits Audited Financials; Files Form-10 as First Step to Up-List to OTCQB
https://www.newsfilecorp.com/release/123710
Jackson Center, Pennsylvania--(Newsfile Corp. - May 12, 2022) - Halberd Corporation (OTC Pink: HALB) submitted its fully audited financials through its fiscal second quarter and Form-10 to the SEC as its initial step to up-list from OTC-PINK to OTCQB on the OTC Markets exchange. Once Halberd's shares are qualified under the Securities Exchange Act of 1934 (the "34 Act"), Halberd will become a fully reporting issuer pursuant to Section 12(g) of the '34 Act. The OTCQB index is intended for development/venture stage companies and requires the Company to submit audited financials which adhere to GAAP standards and the company must be current with its SEC filings/reports -- Form 10-K Annual (audited), Form 10-Q Quarterly (unaudited), and the occasional Form 8-K .
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "We see this as a major step forward for the corporation. Having fully audited filings lends credibility to the company as well as confidence to our investors. This is another example of Halberd's team meeting its stated goals. We have been very open in our statement of quarterly goals vs. our accomplishments, and intend to continue this practice going forward.
"Halberd's technical accomplishments, in our opinion, are unparalleled, and we believe, could revolutionize the medical industry because our patented process operates extracorporeally (outside the body). This medical procedure can be applied safely and without any of the harmful side-effects associated with drugs injected or ingested. Halberd's patented and patent-pending technology also has the advantage to address neurological diseases by extracting the pathophysiologic basis (cause) of the disease from cerebral spinal fluid, or from blood in the case of blood-borne diseases. We have been very active and transparent in reporting our scientific and business-related achievements to two separate audiences: #1) Our stockholders and #2) the scientific community consisting primarily of larger pharmaceutical firms."
Hartman added, "Halberd continues to advance its technology via initiation of laboratory testing on blood, animal testing, which is currently in the planning stage, followed by human clinical trials, and ultimately, via direct application to the FDA for certification. If Halberd Corporation is awarded a pending government contract, it would expedite the program timeline significantly. The entire process of testing and certification could be accelerated via partnering with larger organizations who have to answer two basic strategic questions:
What if we do partner with Halberd Corporation? and,
What if we don't and our competitors do?"
Halberd Submits Audited Financials; Files Form-10 as First Step to Up-List to OTCQB
Press Release | 05/12/2022
Jackson Center, Pennsylvania--(Newsfile Corp. - May 12, 2022) - Halberd Corporation (OTC Pink: HALB) submitted its fully audited financials through its fiscal second quarter and Form-10 to the SEC as its initial step to up-list from OTC-PINK to OTCQB on the OTC Markets exchange. Once Halberd's shares are qualified under the Securities Exchange Act of 1934 (the "34 Act"), Halberd will become a fully reporting issuer pursuant to Section 12(g) of the '34 Act. The OTCQB index is intended for development/venture stage companies and requires the Company to submit audited financials which adhere to GAAP standards and the company must be current with its SEC filings/reports -- Form 10-K Annual (audited), Form 10-Q Quarterly (unaudited), and the occasional Form 8-K .
William A. Hartman, Chairman, President & CEO of Halberd Corporation, stated, "We see this as a major step forward for the corporation. Having fully audited filings lends credibility to the company as well as confidence to our investors. This is another example of Halberd's team meeting its stated goals. We have been very open in our statement of quarterly goals vs. our accomplishments, and intend to continue this practice going forward.
"Halberd's technical accomplishments, in our opinion, are unparalleled, and we believe, could revolutionize the medical industry because our patented process operates extracorporeally (outside the body). This medical procedure can be applied safely and without any of the harmful side-effects associated with drugs injected or ingested. Halberd's patented and patent-pending technology also has the advantage to address neurological diseases by extracting the pathophysiologic basis (cause) of the disease from cerebral spinal fluid, or from blood in the case of blood-borne diseases. We have been very active and transparent in reporting our scientific and business-related achievements to two separate audiences: #1) Our stockholders and #2) the scientific community consisting primarily of larger pharmaceutical firms."
Hartman added, "Halberd continues to advance its technology via initiation of laboratory testing on blood, animal testing, which is currently in the planning stage, followed by human clinical trials, and ultimately, via direct application to the FDA for certification. If Halberd Corporation is awarded a pending government contract, it would expedite the program timeline significantly. The entire process of testing and certification could be accelerated via partnering with larger organizations who have to answer two basic strategic questions:
What if we do partner with Halberd Corporation? and,
What if we don't and our competitors do?"
To get the latest news on Halberd's exciting developments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC Pink: HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed twenty related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are "forward-looking statements" (as defined in the Private Securities Litigation Reform Act of 1995). The Company' cautions our
(responsible mod: Bill Moynihan/EHRjunkie)
Halberd Corp. (OTC PINK:HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements.
Halberd holds the exclusive rights to 4 granted U.S. Patents, no. 9,216,386 no. 8,758,287 and no. 11,186,629, no. 8,865,733
Halberd Corporation is a biotechnology company and with its partners (Dr. Qiang Shawn Chen and his team at Arizona State University; Dr. William Sturrus and his team at Youngstown State University; Dr. Richard Carr and his team at Mississippi State University), is developing an extra-corporeal (out of the body) treatment against PTSD, CTE, Alzheimer's Disease and other virusses and diseases. With the use of antibodies the disease antigens is targeted. Using radiofrequency / laser technology the "antibody-metallic moiety-antigens complex" is destroyed/eradicated. All based on 4 granted patents and 22 pending provisional/non-provisional (some PCT) patent applications.
2024: In close cooperation with partner (revenue sharing agreement in place) AthenaGTX, Halberd Corp is working on contracts from the military, to develop treatments to tackle Traumatic Brain Injury/TBI (patent pending nasal spray) and Post Traumatic Stress Syndrome/PTSD (patented Low-Dose-Naltrexone product).
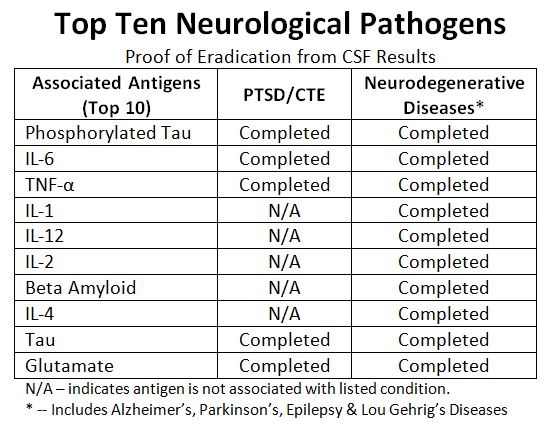
JACKSON CENTER, PA / ACCESSWIRE / March 14, 2023 / Halberd Corporation (OTC PINK:HALB) is scheduled to meet with Center of Disease Control (CDC) representatives to discuss Halberd's incredible success in eradicating multiple strains of antibiotic resistant (AR) E. coli bacteria and Candida auris samples previously provided by the CDC.
JACKSON CENTER, PA / ACCESSWIRE / September 6, 2023 / Halberd Corporation's (OTC PINK:HALB) research at Mississippi State University (MSU) has shown significant reduction in two key markers of traumatic brain injury (TBI) in early testing of Halberd's patent-pending nasal spray to mitigate the effects of TBI. Anecdotal data in preliminary testing reveals a 20% reduction in Neuron Specific Enolase and a 50% reduction in the levels of Glial Fibrillary Acid Protein compared to the control group.
JACKSON CENTER, PA / ACCESSWIRE / September 20, 2023 / Halberd Corporation (OTC PINK:HALB) and Athena GTX, Inc, of Johnston, Iowa commenced collaboration on critical development of submissions for government contracts seeking proposals for the detection and treatment of traumatic brain injury (TBI). The initial focus will be on accelerating and fine-tuning Halberd's proprietary nasal spray for suppressing the effects of traumatic brain injury (TBI).
JACKSON CENTER, PA / ACCESSWIRE / December 13, 2023 / Halberd Corporation (OTC PINK:HALB) and CrodenHealth.com of Canada agree to produce, market, sell and distribute Halberd's patented low dose naltrexone combo product(s) worldwide. The agreement also allows CrodenHealth.com right of first refusal regarding similar rights to future products in Halberd's development pipeline.
JACKSON CENTER, PA / ACCESSWIRE / January 10, 2024 / Halberd Corporation (OTC-PINK:HALB) is thrilled to declare the formalization of a profitable revenue-sharing accord with Athena GTX, Inc., of Johnston, Iowa, (www.athenagtx.com). Athena GTX is a private enterprise specializing in providing medical care solutions tailored for first responders in both civilian and government sectors.
Halberd's / Athena's treatment for PTSD is called WatchDawg
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
