Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Ok ok we see you ..but going to hold $3s this time or another fake out. Support been at $2.60s past few weeks. Looking better
Mpox has spread to at least 14 African countries, including Morocco, which has confirmed its first case in the current outbreak. (Source: BBC Africa)
The Democratic Republic of Congo (DRC) is the epicenter of the outbreak, with over 20,000 cases reported and more than 500 deaths. (Source: WHO)
Neighboring countries, such as Burundi, Kenya, Rwanda, and Uganda, have also reported cases. (Source: WHO)
Vaccination Efforts
The World Health Organization (WHO) recommends use of MVA-BN or LC16 vaccines, or the ACAM2000 vaccine when others are not available. (Source: WHO)
Only people at risk of exposure to mpox should be considered for vaccination, according to WHO. (Source: WHO)
Transmission and Prevention
Mpox is primarily spread through close person-to-person contact with someone who is infected, including through sex. (Source: Nature)
Cleaning and disinfecting surfaces or objects and washing hands after touching contaminated surfaces or objects can help prevent transmission. (Source: WHO)
Avoiding unprotected contact with wild animals, especially those that are sick or dead, and cooking food thoroughly can reduce the risk of getting mpox from animals. (Source: WHO)
Public Health Emergency
The WHO declared the mpox outbreak a Public Health Emergency of International Concern (PHEIC) on August 19, 2024, citing the rapid spread of a new, more transmissible strain of the virus. (Source: WHO)
Research and Development
Scientists are racing to answer questions about the outbreak, including whether vaccines will curb the spread and how the virus is transmitted. (Source: Nature)
Looking good bottom about in for hedgies
Watch for 2.52 - .2.92 too hold for flip. 2.08- 2.20 next zone underneath if doesn’t
Hedgies determine new downtrend. New entry as we see $2.80s for another flip
Ok hedgies confirming new upward trend perhaps give it a few hours still
Flip the $3.06 at 3.30s and wait for hedgies to determine direction
Tummies yummy $3.06 bought and then if happens to test next level down add again.
Hedgies want to take it as low as possible before the next big run into Covid vaccine season. Luv you Gary Gensler
Good sign 3.06 hedgies covering shorts now volume picking up 600,000. Almost time to add some more goodies
3.19 zone being tested if holds a few days start adding some again
price of $5.125 per share for expected gross proceeds of $5 million.
4:39 AM EDT, 08/29/2024 (MT Newswires) -- GeoVax Labs (GOVX) said late Wednesday it has agreed to sell 975,610 common shares in a registered direct offering to an institutional investor at a price of $5.125 per share for expected gross proceeds of $5 million.
The company will also issue warrants to purchase up to 975,610 common shares to the investor in a concurrent private placement, it said. The warrants will be exercisable at a price of $5 per share.
Net proceeds will be used for working capital and general corporate purposes, the company said.
The offering is expected to close around Friday, sub
Test 3.19- 3.55 zone wait to confirm before pulling trigger $2.52- 2.92 is the other zone. The hedgies that have most of the shares will determine that as they move this one as they please
Covid flue season blooming still holding a few here but inclined to add some more ..
GeoVax Labs, Inc. is a clinical-stage biotechnology company developing novel vaccines for many of the world’s most threatening infectious diseases and therapies for solid tumor cancers. The company’s lead clinical program is GEO-CM04S1, a next-generation COVID-19 vaccine for which GeoVax was recently awarded a BARDA-funded contract to sponsor a 10,000-participant Phase 2b clinical trial to evaluate the efficacy of GEO-CM04S1 versus an approved COVID-19 vaccine. In addition, GEO-CM04S1 is currently in three Phase 2 clinical trials, being evaluated as (1) a primary vaccine for immunocompromised patients such as those suffering from hematologic cancers and other patient populations for whom the current authorized COVID-19 vaccines are insufficient, (2) a booster vaccine in patients with chronic lymphocytic leukemia (CLL) and (3) a more robust, durable COVID-19 booster among healthy patients who previously received the mRNA vaccines. In oncology the lead clinical program is evaluating a novel oncolytic solid tumor gene-directed therapy, Gedeptin®, in a multicenter Phase 1/2 clinical trial for advanced head and neck cancers. GeoVax has a strong IP portfolio in support of its technologies and product candidates, holding worldwide rights for its technologies and products. The Company has a leadership team who have driven significant value creation across multiple life science companies over the past several decades. For more information about the current status of our clinical trials and other updates, visit our website: www.geovax.com.
The Fauch knows how to party!
Maybe the evil bastard will drop dead .
Buy 6.30s sell $7.30s was todays lesson dip time coming again.. Dr. Fauci Somehow Contracts Ultra-Rare West Nile Virus On Heels Of COVID-19 Infection
Good, keep giving that evil idiot all my vaccines and boosters.
He should double up
Catch the dips play next.. Dr. Fauci has COVID again for the third time and has been vaccinated and boosted six times
GOVX...............................https://stockcharts.com/h-sc/ui?s=GOVX&p=W&b=5&g=0&id=p86431144783
sale of 1,700,000 shares of the Company’s common stock (or common stock equivalents) at a price of $5.00 per share in a registered direct offering priced above-the-market under Nasdaq rules.
In addition, in a concurrent private placement, the Company will issue to the investor warrants to purchase up to 1,700,000 shares of common stock. The warrants have an exercise price of $5.00 per share, will be exercisable immediately following the date of issuance and will have a term of five years from the date of issuance.
Next stop $15 then $20…By Amanda Alvarado
Published: Aug. 21, 2024 at 8:02 AM EDT
(Gray News) - COVID-19 cases are rising and KP.3.1.1 is the predominant variant in the United States, according to the Centers for Disease Control and Prevention.
The variant accounted for between 31% and 43% of the cases in the two-week period ending on Aug. 17.
KP.3.1.1 is currently the only major variant increasing in proportion nationally, according to the CDC.
Emergency room visits diagnosed as COVID-19 started increasing in May in the West and are now the highest in the South and Southeast. The wastewater viral activity level for COVID-19 has been rising since mid-May and is currently very high across most parts of the country, according to the CDC.
- Kamala Harris issues vaccine mandate for campaign workers..Good news for vaccine makers pump em up.
Still have $1.80 shares left from a couple weeks back holding for the Gold longer term rest been cashed out
GOVX....................................https://stockcharts.com/h-sc/ui?s=GOVX&p=W&b=5&g=0&id=p86431144783
Damn… nice play guys. Missed this one. I bought my first stock today in months!
Been terrible year
Huge winner today,warrents was even better because of big spread, easy 15-19% wins
and back out before the dump, but it tried.
i grabbed some on the dip today. ![]()
Can we see 20$+ this week?
Yes we can 💲💲💲
Cash out some more…Shares of mpox vaccine maker Bavarian Nordic saw an increase this week after the World Health Organization (WHO) declared a public health emergency and as the company’s CEO announced it could provide 10 million doses of the vaccine.
“We have inventory and we have the capabilities. What we’re missing are the orders,” CEO Paul Chaplin told Bloomberg this week.
Merry Christmas $$$$$$$$$$))))))))
So, what is the catalyst for this latest rise?
Mpox is threatening to go global
Scientist in labNathan Posner/Getty Images
A more dangerous version of the virus formerly known as monkeypox spread outside of Africa for the first time this week, leading European officials to raise the Continent’s risk level from low to moderate.
ICYMI: The World Health Organization declared mpox a global health emergency on Wednesday, about two years after pulling the same alarm on a different variant that infected almost 100,000 people worldwide and 32,000+ in the US, according to the New York Times.
The new version is deadlier and spreading. On Thursday, Sweden reported that a traveler who returned from Africa contracted the variant, and yesterday, Pakistan reported its first case. In Africa, infections have sprung up in at least a dozen countries, in addition to the Democratic Republic of Congo, where 15,600 people caught the virus and 537 died from it this year, per the WHO.
The virus appears to spread through skin or fluid contact with another infected person or their items.
Vax news: Danish biotech firm Bavarian Nordic—the only company with an FDA-approved mpox vaccine—submitted data to EU regulators yesterday for approval to use its shot on teens, who account for more than 70% of the cases in Africa, Bavarian’s CEO told CNBC. The vax-maker says it has 300,000 doses ready to ship out and could deliver 10 million by the end of 2025. Its stock soared 50% this week.—ML
Indeed. But hope to reload soon. Great to make good money in this bear 🐻 market.
Hagwe
Excellent $8 definitely hardest resistance. Unloaded another batch but still holding some for the $20 could be a while dependent on Congo mpox
Sold at $8 from $2 nice money for the weekend 💰😍
Gltya... hope to see you all at $20 soon.
Next runner will be....
This was expected Peter uterus
|
Followers
|
267
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
19565
|
|
Created
|
12/11/01
|
Type
|
Free
|
| Moderators | |||
July 26 (Reuters) - The Biden administration estimates it may need nearly $7 billion to deal with the monkeypox outbreak in the United States, the Washington Post reported on Tuesday.
The funding estimate was part of a memo marked to President Joe Biden outlining a series of options, as opposed to a formal funding request, the report said. (https://wapo.st/3oxXimZ)
The White House and the U.S. Department of Health and Human Services did not immediately respond to requests for comment.
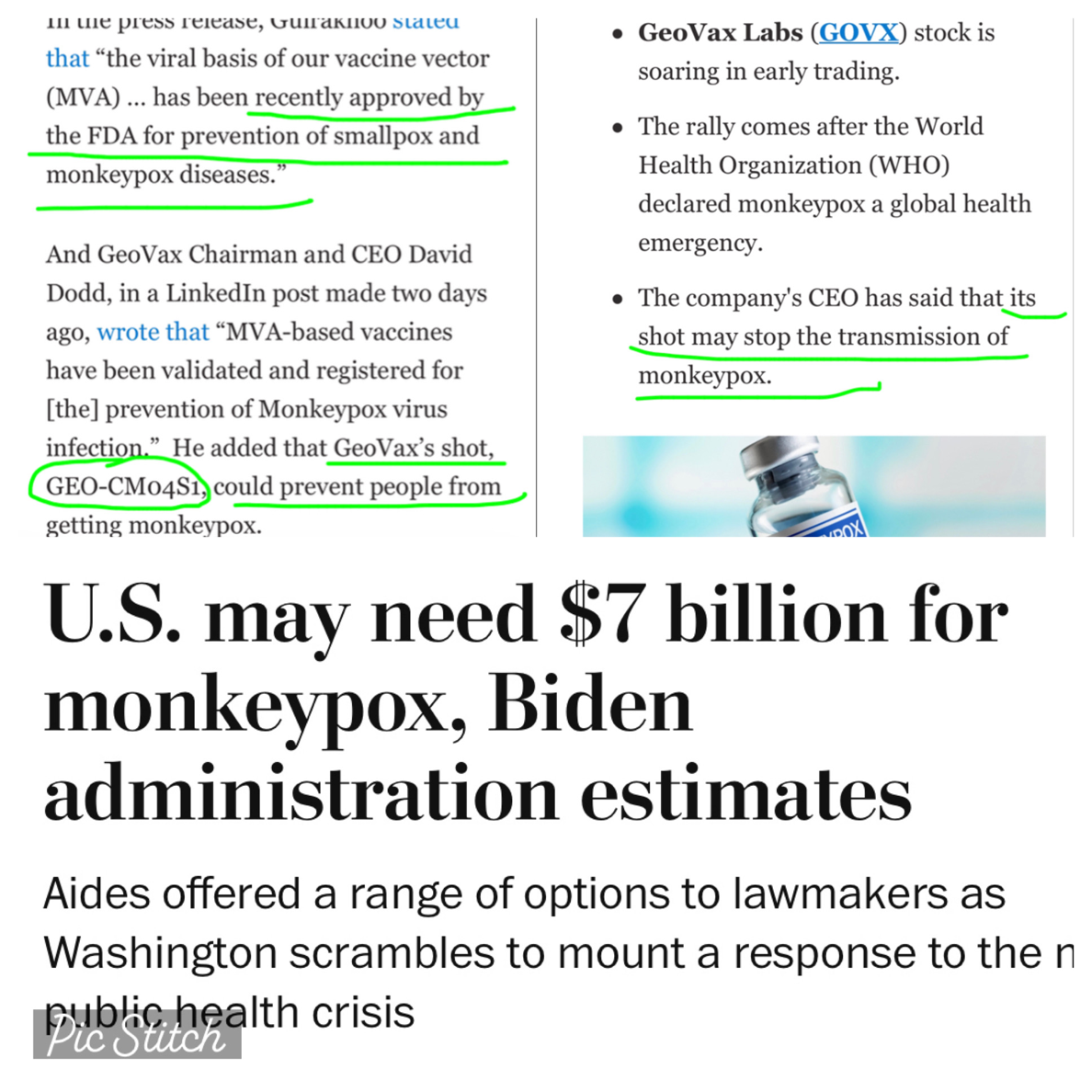
CREDIT MONEYMADE FOR DD AND ALERTING GROUP BEFORE THE CLOSE. WEEEEEEEE $1.70https://investorshub.advfn.com/boards/read_msg.aspx?message_id=169502961
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
