Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
~ how nice :) great job as always, captain (^^)/
btw, please check out lbsr.... went up big today (i don't know why, though).... i used to follow... but i'm out now...darn ![]()
~ nice call as ususal, captain :)
Kei!
Up 6.5% today to a new 52-week high !
DB bank recommendation they say.
~ nice call as ususal, captain :)
Elan Provides an Update on ELND005 (Scyllo-inositol)
finance.yahoo.com/news/Elan-Provides-Update-ELND005-bw-2161394995.html?x=0
DUBLIN--(BUSINESS WIRE)-- Elan Corporation, plc (NYSE: ELN - News) today provides an update on ELND005 (Scyllo-inositol).
Elan has entered into a manufacturing agreement for the supply of the active pharmaceutical ingredient for ELND005 with Lonza Group AG. This agreement is fundamental to ensuring that a high quality supply of ELND005 will be available to support the advancement of the program.
During the 4th Conference Clinical Trials on Alzheimer’s Disease (CTAD), held in November 2011, ELND005 was featured during four oral presentations and on two posters where new analyses were presented from the Phase II Alzheimer’s disease study (AD201, Salloway et al. Neurology 2011) by globally recognized Alzheimer’s disease experts. The presentations focused on treatment effects at earlier stages of the disease, using validated “composite” cognitive endpoints. These results support the general direction of the field for earlier intervention.
In addition, data on ELND005’s role in reducing the emergence of neuropsychiatric symptoms in Alzheimer’s patients was highlighted. ELND005 may have applications in additional psychiatric indications such as bipolar. Elan’s goal is to initiate a proof of concept, Phase II study in bipolar disorder post completion of discussions with therapeutic experts and regulators.
For earlier stages of Alzheimer’s disease, experts and regulators continue to evolve their understanding of the disease from a biological, clinical and biomarker perspective. Elan will continue to seek advice as it advances the ELND005 program in Alzheimer’s disease.
Further updates will be provided as appropriate.
Happy Thanksgiving Folks !
Have a safe one !
I don't believe we'll get out of this silly movement in the Markets until our European friends get their act together with their debt problems and we get our act together by voting every incumbent politician out of office in the next election.
Watched this and about half way through they talk about Elan. Quite interesting and thought those who are interested in the company would like to be aware. Don't imagine it will do anything price wise to the stock at present. Compelling story as it relates to ELN though.
Today’s BIIB story on BG-12 is old news (#msg-62320268); however, there will be new news on BG-12 before the end of 2011 when data are reported for the second BG-12 phase-3 study (the one called COMFIRM). Regards, Dew
Biogen Idec appears to have good news with BG-12 MS Drug.
http://www.bloomberg.com/news/2011-10-21/biogen-idec-multiple-sclerosis-drug-
enter BIIB in search window for news
Thanks for posting it ' blackcat '
This is our news as well
http://by168w.bay168.mail.live.com/default.aspx?wa=wsignin1.0&rru=inbox#fid=1&fav=1&n=235728646&rru=inbox&mid=ac83bcf3-e8f8-11e0-897f-00237de41626&fv=1
Transition Therapeutics Announces Publication of ELND005 Phase 2 Clinical Study Data in Alzheimer's Disease
TORONTO, Sept. 27, 2011 (GLOBE NEWSWIRE) -- Transition Therapeutics Inc. ("Transition" or the "Company") (TSX:TTH) (Nasdaq:TTHI) announced that Phase 2 clinical study data of ELND005 in mild to moderate Alzheimer's disease has been published today in the peer-reviewed journal, Neurology. The Neurology article is entitled "A Phase 2 randomized trial of ELND005, scyllo-inositol, in mild-moderate Alzheimer's disease". In addition, the embargo on the ELND005 Phase 2 data previously presented at the International Conference on Alzheimer's Disease (ICAD) in July 2011 has been lifted and the data can be viewed on our website www.transitiontherapeutics.com. Below Transition has summarized the combined data from the article and the presentations.
Welcome. And - we do, Germany up 6,15 so far. ;)
Thanks for posting this 'ThumsUp'.
I have been out of pocket for the last two weeks ( road trip ) and appreciate you sharing the info.
Hopefully we will see some continuing upward movement in the PPS.
Merger with Alkermes completed !
http://www.businesswire.com/news/home/20110916005889/en/Alkermes-plc-Elan-Corporation-plc-Announce-Completion" rel="nofollow" target="_blank" >http://www.businesswire.com/news/home/20110916005889/en/Alkermes-plc-Elan-Corporation-plc-Announce-Completion
DUBLIN--(BUSINESS WIRE)--Alkermes plc (NASDAQ: ALKS) and Elan Corporation, plc (NYSE: ELN) (“Elan”) today announced the completion of the merger between Alkermes, Inc. and Elan Drug Technologies (EDT), the drug formulation and manufacturing business unit of Elan, following the approval of the merger by Alkermes, Inc. shareholders on Sept. 8, 2011. The businesses were combined under a newly-formed company, Alkermes plc, which is incorporated in Ireland and headquartered in Dublin. Alkermes plc will be listed on the Nasdaq stock exchange under the ticker symbol “ALKS.”
“Through this transaction, we have strategically combined two highly innovative companies with proprietary technologies and important commercial products, creating a strong platform for accelerating future growth and increasing shareholder value.”
“We are very excited about the creation of Alkermes plc – a unique, global, diversified company and a leader in CNS medications,” stated Richard Pops, Chief Executive Officer of Alkermes plc. “Through this transaction, we have strategically combined two highly innovative companies with proprietary technologies and important commercial products, creating a strong platform for accelerating future growth and increasing shareholder value.”
Under the terms of the business combination agreement, Elan receives $500 million in cash and 31.9 million ordinary shares of Alkermes plc, representing approximately 25% of Alkermes plc. Based on the closing share price of Alkermes, Inc. on Thursday, Sept. 15, 2011 of $16.52, this represents a total transaction value of approximately $1.0 billion.
Comment by George Scangos ( CEO / Biogen Idec ) during Biogen Idec's Earnings Report today, quote,
" During the last 12 months, our improved focus and reinvigoration of the organization has driven strong product performance and allowed our late stage pipeline to mature. Francesco and Doug will talk about these in greater depth.
But to highlight, after tremendous work by many people in the organization, we received a new label for TYSABRI in the EU to include JC Virus Antibody Status as an additional risk factor for PML. Physicians and Patients can now consider JCV Antibody Status, as well as prior immunosuppressant use and treatment duration when assessing the risk of PML for individual patients.
Along with the commercial availability of the JCV Assay in Europe, the MS community now has more tools to make an informed treatment decision, which we all believe will unlock the value of TYSABRI. Concurrently, the European Commission renewed the 5- year marketing authorization for TYSABRI.
The impressive growth of the TYSABRI business over this past year and the strong demand we've seen this quarter is a testament to the efficacy of TYSABRI, as well as the excellent planning and execution."
.....of course then he goes on to mention AVONEX
Should turn out to be a most interesting Quarter for Elan.
they also went into some detailed numbers.....some highlights...
the run rate for Tysabri is now $ 1.5 Billion in annualized in-market sales.
Q2 was very strong ....revenue @ $ 389 million, an increase of 31 % vs. the same quarter last year......
ended the quarter with 61,500 patients an increase of 2,400 over last quarter,......reached milestone with TYSABRI..... 1 million infusions since relaunch in 2006
Biogen IDEC's Earnings Report is scheduled for July 26th.
Should be able to get a preliminary view of Elan's progress.
BI's earnings are expected to be up by the ANALysts prognostications, as they have been in the last 4 Quarters.
ELN is holding up well and not getting too seasick with the daily rollercoaster rides in the Markets...... Nice and Steady upward trend.
Unique brain PK properties of 3D6 and bapineuzumab depend on cerebral amyloid load in PDAPP transgenic mice
P4-406
Background: Passive immunization with anti-Aß antibodies leads to the reduction of AD-like neuropathology in transgenic mice. Previously we showed that anti Aß antibodies enter the brain and bind to amyloid plaques.
Methods: Now using 125I-labeled 3D6, the mouse form of the phase 3 clinical candidate bapineuzumab, we further characterized the specificity and the pharmacokinetics of this antibody in the brain and serum.
Results: The results of our studies demonstrate that 125I-labeled anti-Aß antibodies (3D6 and bapineuzumab) delivered intravenously accumulated over time in the brain. This accumulation was particularly evident in regions rich in amyloid plaques and was correlated with the overall level of plaque burden. The binding of 3D6 to brain amyloid plaques did not saturate at the doses tested since cold 3D6 dosed as high as 30 mg/kg did not compete with the binding of the 125I-labeled 3D6. Following one intravenous injection, serum levels of 125I-labeled 3D6 decreased exponentially over time while levels in the hippocampus increased to reach the maximal concentrations by day 14 (brain Tmax) and remained stable up to 27 days. Based on mean PK profile, the brain half-life (T1/2) of 3D6 was estimated to be 54 days whereas the serum T1/2 was 6 days.
Conclusions: In summary, we demonstrated that both 3D6 and bapineuzumab cross the blood brain barrier, that the CNS clearance is markedly slower than the serum clearance, and that this slower CNS clearance correlates with binding to amyloid in a transgenic model of Alzheimer’s disease.
Frederique Bard, Janssen Alzheimer Immunotherapy Research and Development
Gene Kinney, Janssen Alzheimer Immunotherapy R&D
Michael Fox, Elan Pharmaceuticals
Stuart Friedrich, Wyeth Biotech
Ted Yednock, Janssen Alzheimer Immunotherapy R&D
http://icad.sclivelearningcenter.com/index.aspx
Newt Gingrich thinks he can revive his debilitated campaign by talking about Alzheimer’s.
http://www.washingtonpost.com/politics/newt-gingrich-bets-on-alzheimers-as-key-to-a-2012-comeback/2011/06/29/AGR0JawH_story.html
Have a Great & Safe 4th of July Holiday !
Closing PPS of Elan as of :
12/31/2010 - $ 5.73
6/3/2011 - $ 9.59
7/1/2011 - $11.86
that appears to be roughly a 106.98 % increase for the first half of 2011 and a 23.67 % increase this last month.
Welcome back to the Living ELN !
Nice and steady without much fanfare the PPS is inching upward.....interesting comments yesterday at Seeking Alpha....see link below
http://seekingalpha.com/article/276834-pharma-don-t-count-elan-out
although a visit by my 82 year old Step Mother-in-Law last week might have caused me to lose my focus
Thanks for the correction 'bc'.
I must have been asleep at the switch , although a visit by my 82 year old Step Mother-in-Law last week might have caused me to lose my focus
:)
Well, actually they did it last week.
UBS raises Elan Corp to buy
//June 9 (Reuters) - Elan Corp Plc: * UBS raises Elan Corp to buy from neutral For a summary of rating actions and price target changes on FTSEurofirst 300 companies:
Link: http://classic.cnbc.com/id/43335717
UBS upgraded Elan to ' Buy " this morning and raised their target price to $ 12.78
Holding up well today in what is another mindless sea of red trading today pretty much across the board
Im getting slammed except for ELN
Looks like Elan is holding up well after hitting the double digits, considering the rest of the market is mostly a sea of red.
Hopefully we'll finish in the green today......green.....like in dollars and cents .....not Leprechauns.
Give it time fellas !!! $$$
JNJ’s Investor Day webcast two weeks ago evidently spurred some newfound bullishness about Bapineuzumab.
You also HAK41....looking good!!!
Have a great and safe Memorial Day weekend !
Maybe we can break the 10's next week !
Yes, indeed !
Suspect ELN should have a very decent year for the remainder of 2011.
Time for a run !!!
AGM tomorrow pre-mkt.
$9.24 $9.51 next resistance levels
9's it is ! .....with nice volume and a 7.73% increase for the day.
That's not a bad day for Elan.
:)
Pressure build-up should bust thru 9's this week.
Once we break the 9's were clear to 12
:)
Thanks, I'll try to catch you at happy hour tomorrow
Soon you will be rich :]
sorry gem57, no longer have PM capability, gave up premium subscription back in March, since I no longer post much.
Have Norxx Bxx Resxx also..........am up in Greenville.
GM gem57
I do believe you are on target with that one. Slow and steady is fine by me with ELN.
Noticed you're over at Siexxa Gxxd also....am holding a fistful, waiting for that one to break open.
Have about a dozen penny dragons ( mostly Junior Miners ) for the fun of it, but they have been dragging their feet for the last year.
Where in G are you located ....am up the road a piece from you, one state over...
The 960 Mill Deal should propel us foward.
It held up well in the last few days.
Am also looking forward to being back in the double digits.
Setting up nicely for a run up to $12.50
You're right, I had forgotten about the daily pattern back then.
I was fortunate enough to accumulate a fistful of shares between about $4 and $9 , never traded them, just held them at the time until they ran into the high teens and then into the twenties,
Cashed out then, missing the highs, when they ran into the thirties, but held on to a small core position. If this pattern continues, it could be time to accumulate some more, especially since it looks like they might just get their financial house in order this year.
Those years ago, it traded quite predictably. Many, if not most, mornings it gapped down at the open, stayed red for an hour or two, and then meandered to green for the rest of the day. Those of us who made swing trades around our core had good days. Day traders too, although I rarely participated in that.
Another slightly green day ( third one in a row )
Up about 32% since 5/10/2010, and up about 46 % from the close of $ 5.73 on 12/31/2010........
Reminds of the days years ago, when Elan was beaten down mercilessly, and started on its road to recovery.
Considering the positive contributions they have made to many MS sufferers , they are about due for some positive breaks themselves.
|
Followers
|
36
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
890
|
|
Created
|
08/09/05
|
Type
|
Free
|
| Moderators | |||
 __________________________________
__________________________________
5/9/2011 Alkermes merges with Elan's Technology Unit - http://www.businesswire.com/news/home/20110508005086/en/Elan-Corporation-PLC-UK-Regulatory-Announcement-Alkermes
Elan Corp PLC ........http://www.elan.com........Real Time quotes: http://moneycentral.msn.com/investor/quotes/pprtq.asp?Symbol=ELN
Transcript - Elan Q2'07 Earnings Call: http://seekingalpha.com/article/42570-elan-q2-2007-earnings-call-transcript?source=feed
Transcript - Elan Q3'07 Earnings Call: http://seekingalpha.com/article/51390-elan-q3-2007-earnings-call-transcript
Notes - Elan @ JPM, January '08: http://investorshub.advfn.com/boards/read_msg.asp?message_id=25943778
Replay - Elan Q3'07 Oct '08 Earnings Call : http://www.elan.com/investorrelations/events/webcast_details.asp?eventID=1986779
Slides - Elan @ Credit Suisse/London Nov'07 Earnings Call: http://www.csfb.com/conferences/pharma2007/pdf/elan.pdf ___________________________________________________________________________________________________________
DDT & NANO-CRYSTAL
Elan announces J&J NDA filing Oct'07, and lists Nano-Crystal Technology products: http://www.elan.com/news/full.asp?ID=1068768
Acorda (& Elan) drug aids walking in MS patients: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=29731491
___________________________________________________________________________________________________________
TYSABRI in MS
Tysabri Demonstrates Improvement in Cognitive Function: http://www.elan.com/News/full.asp?ID=910099
Tysabri Demonstrate Significant Improvements in QoL: http://www.elan.com/News/full.asp?ID=840112
Tysabri Shows Reduction in Steroid Use/Hospitalizations: http://www.elan.com/News/full.asp?ID=913012
Tysabri Reduces Vision Loss: http://tinyurl.com/366f8w
Tysabri Increases Proportion of Patients with Disease-Free Status: http://www.elan.com/news/full.asp?ID=1061738
Tysabri MS FDA Panel Hearing, March '06: http://www.fda.gov/ohrms/dockets/ac/cder06.html#PeripheralCentralNervousSystem
Tysabri in MS update (May '07), Safety & sustained Efficacy 3 years out: http://www.elan.com/News/full.asp?ID=995005
Tysabri backed by NICE for Reimbursment in UK: http://www.elan.com/News/full.asp?ID=1022211
Tysabri - Accelerating Weekly Uptake: http://investorshub.advfn.com/boards/read_msg.asp?message_id=25870407
Tysabri MS net gain/wk: 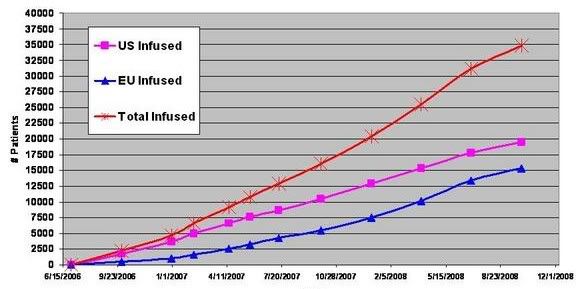
___________________________________________________________________________________________________________
TYSABRI in CROHN's
FDA & Elan's briefing materials, Tysabri Hearing July '07: http://www.fda.gov/ohrms/dockets/ac/07/briefing/2007-4313b1-00-index.htm
FDA & Elan's & Public's SLIDES, Crohn's Tysabri Hearing: http://www.fda.gov/ohrms/dockets/ac/07/slides/2007-4313s1-00-index.htm
Summary Minutes & Voting, Crohn's Tysabri Hearing: http://www.fda.gov/ohrms/dockets/ac/07/minutes/2007-4313m1-final.pdf
FDA Advisory Committee Recommends Approval of Tysabri in Crohn's: http://www.elan.com/News/full.asp?ID=1034453
FDA Approves Tysabri in Crohn's: http://www.elan.com/news/full.asp?ID=1096281
----------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Tysabri Reduces Hospitalization in Crohn's: http://investorshub.advfn.com/boards/read_msg.asp?message_id=29492160
Study suggest Crohn's demand for Tysabri: http://investorshub.advfn.com/boards/read_msg.asp?message_id=23853928
Expert Commentary - http://investorshub.advfn.com/boards/read_msg.aspx?message_id=33432160
Remission Rates - http://investorshub.advfn.com/boards/read_msg.aspx?message_id=32785546
___________________________________________________________________________________________________________
TYSABRI in MULTIPLE MYELOMA & CLL
Phase I / II clinical trial in MM: http://clinicaltrials.gov/ct2/show/NCT00675428
TYSABRI in Chronic Lymphocytic Leukemia
pre-clinical paper: http://www.ncbi.nlm.nih.gov/pubmed/18518761
___________________________________________________________________________________________________________
ALZHEIMER's ____________________________ __________________________
__________________________
What we know and what we don’t know about AD: http://www.alzforum.org/res/for/journal/centennial/whatweknow/whatweknow.asp
Mild Cognitive Impairment - leading to Alzheimer's: http://investorshub.advfn.com/boards/read_msg.asp?message_id=27669636
Discussion Panel - "Relative Optimism": http://investorshub.advfn.com/boards/read_msg.asp?message_id=25942353
"The Amyloid Hypothesis" - Paper plus Q&A plus Debate: http://www.alzforum.org/res/adh/cur/knowntheamyloidcascade.asp
Apoe4 gene prevalence: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=30134888
Earlier Alzheimer's Results in halted AN_1792 vaccine candidate:
"Antibodies against Beta-Amyloid Slow Cognitive Decline in Alzheimer’s Disease" : http://www.tixx.com/alzheimers_2003.pdf
Selection / AN_1792 autopsy cases : http://www.elan.com/Images/FINAL%20Slide%20%232%20Autopsy%20Case%20report_tcm3-6289.pdf
Polysorbate explanation/T-cell response :http://www.ncbi.nlm.nih.gov/pubmed/18322388?ordinalpos=13&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_RVDocSum
Elan-Wyeth Symposium: Immunotherapy for AD & PD, including Long-Term Follow-Up of AN1792 Patients:
http://www.elan.com/InvestorRelations/events/ElanWyethSymposium_ADPD.asp
Current Elan Alzheimer Trials:
aab-001 Phase I in Japan : http://www.clinicaltrials.gov/ct/show/NCT00397891
aab-001 Phase IIa (n=30) : http://clinicaltrials.gov/ct2/show/NCT00174525
aab-001 Phase II (n=240) : http://www.clinicaltrials.gov/ct/show/NCT00112073
aab-001 subcutaneous Phase II (n=120) : http://clinicaltrials.gov/ct2/show/NCT00663026
acc-001 Phase II (n=228) : http://www.clinicaltrials.gov/ct/show/NCT00498602
acc-001 Phase II (n=56) : http://www.clinicaltrials.gov/ct/show/NCT00479557
oral ELND005 Phase II (n=340) : http://www.clinicaltrials.gov/ct2/show/NCT00568776
aab-001 Phase III, Apo-E4 carriers (n=800) : http://clinicaltrials.gov/ct2/show/NCT00575055
aab-001 Phase III, Apo-E4 non-carriers (n=1,250) : http://clinicaltrials.gov/ct2/show/NCT00574132
aab-001 extension study (Phase II, n=220) : http://clinicaltrials.gov/ct2/show/NCT00606476
gamma secretase pIII (ELY) : http://www.clinicaltrialsplus.com/dms/index.jsp?RQ_mode=view&RQ_releaseid=399821&RQ_selectedDb=cp
Analyst view by NCB (Ireland) of aab-001 potential (Dec '06 report): http://tinyurl.com/2z4cwm
Elan to Initiate Phase III Clinical Trial of aab-001 in Alzheimer's: http://www.elan.com/News/full.asp?ID=1004531
Design of aab-001 Phase III trials; see also q2'07 CC above: http://investorshub.advfn.com/boards/read_msg.asp?message_id=24067889
Targeted AD genotypes in aab-001 Phase III program: http://investorshub.advfn.com/boards/read_msg.asp?message_id=24098790
Elan & Transition Dose 1st Patient in Phase 2 Study of ELND005: http://www.elan.com/news/full.asp?ID=1089758
Elan & Wyeth Dose 1st Patient in Phase 3 Program for Bapineuzumab (aab-001): http://www.elan.com/news/full.asp?ID=1089760
Adas_COG & NTB Tests / aab-001 Trials: http://investorshub.advfn.com/boards/read_msg.asp?message_id=27669389
Top-line Results from Phase 2 aab-001 Trials: http://www.elan.com/news/full.asp?ID=1166655
Elan & TTHI finish Enrolling Phase 2 ELND005: http://www.elan.com/news/full.asp?ID=1213863
 ___
___ ___
___
..................................................................no ordinary mice, but élan knock-out mice..................................................................
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
