Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
This is really good !
http://cvrmed.com/press-release/cvr-medical-completes-tuv-testing-2-2/
This is really good !
http://cvrmed.com/press-release/cvr-medical-completes-tuv-testing-2-2/
Is this company also operating under (CYHHZ) ticker?
See some our bothered by financing. Takes money to make money and in this case CRRVF wouldn't be unless their confident FDA approval. Chill!
NASDAQ is a given. Patience shall make one millions. After $1 pps trading on margin also.
- Canidain Health can take 30 days for approval. I assume once and if obtaining approval CRV Medical will go to market. I’m assuming they arranging to set up sales and distribution now.
- I think the stock has potential for $5 to $10 a share, it just depends on when. If this device catch’s on the sky is the limit. The market makers need to get out of way ! (The criminals in my eyes) The short selling and the ghost market is not helping. The shorts will get caught soon come FDA submission !
- I agree Gc2
Go CVR Medical ??
I'm thinking 5-10 dollar stock in 2 yrs. Gut guess only due to impact of the device world wide.
I'm curious about PPS in the coming months. How much involvement might there be by the MM's? Is the current price likely the bottom, or once they take it to market will it be pushed down and to what extent? I don't have level 2 so hoping someone else here does. Also, long term what are your thoughts $5 stock, $10, $50...???
That is great news! I read on their website that once they submit to FDA, they plan to commence sales in Canada. So they could begin going to market next month potentially.
CVR Medical is planning to submit with the FDA by the end of the year ( 4th quarter ). The filing is expected to be a “De Novo” filing. Usually it’s a 90-120 day process for the approval process. ( If Approved )
thanks for the response. based off your last post I assumed it was some big conference that would have had some media coverage/press and I was unsuccessful in googling. I look for devices like this, currently a shareholder in another medical device company that has some revolutionary tech in treating burns and they were just granted FDA approval. They filed a PMA with the FDA that took about 1 year to be approved. Based off my research I agree with you about the product/technology being a game changer, still think it is to early for entry based off experience elsewhere. I will continue to monitor this forum it seems like posts have slowed down, keep up the posts! BOL Longs!
All thought this might be helpful with insight on De Novo.
https://www.fda.gov/medicaldevices/deviceregulationandguidance/howtomarketyourdevice/premarketsubmissions/ucm462775.htm
CRRVF $$$$
De Nova (of new) for a brand new device class is still issued a timeframe for review set at 120 FDA days. And who knows what "FDA days" means. I hope you are right. I am just being cautious with my expectations. Q1 rollout would be tremendous.
CVR has stated they are filing De Novo for FDA approval. My understanding this greatly expedites FDA approval.
I should clarify. I think Q1 2019 is goal for rollout, but they will not start full force manufacturing until FDA approval is granted and they are able to fully start marketing. All IMO of course.
Yes, 1st quarter target. Don’t forget there is hope for a Canadian approval, Canada is a big market.
I am still thinking Q3 2019 because FDA approval is long. But I think they will move quick to enter the market once the approval is done.
From what I've read, I am expecting product roll out 1Q 2019. Any thoughts???
DENVER, NC / November 9th, 2018 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces submission of the Carotid Stenotic Scan (CSS) to TUV Rheinland for electrical safety and EMC testing. TUV Rheinland is one of the leading providers of product safety certifications worldwide, covering an assortment of items such as medical devices, home appliances, audio/video products, medical products, textiles and telecommunication equipment. This testing is required for the CSS’s subsequent De Novo submission to the FDA for US market clearance and will show that the device is compliant with all IEC 60601-1 requirements. IEC 60601 is a series of technical standards for the safety and essential performance of medical electrical equipment, published by the International Electrotechnical Commission. First published in 1977 and regularly updated and restructured, IEC 60601-1 is widely accepted as the benchmark for electronic medical equipment. Additional information about TUV Rheinland can be found here (https://www.tuv.com/usa/en/).
Tony Robinson, CVR Medical Chief Operating Officer commented, “It feels good to have the CSS in the hands of TUV and progressing through one of the critical final steps necessary for our FDA submission. With the upcoming transition from IEC 60601-1 3rd Edition to 4th edition, a change which the CSS was already designed in compliance with, the queue to get devices into TUV substantially lengthened, exceeding what we originally allocated for an intended October FDA submission. This is pushing our timeline further back, but I have complete confidence in the engineers at CVR and the quality of the product they designed. With their focus on developing a product that is first and foremost safe, I believe that we will progress smoothly through the TUV testing schedule without delay. With the majority of our De Novo documentation finalized and current clinical trial numbers exceeding the requirements anticipated by our industry-leading regulatory consultants, we are still targeting an end of Q4 submission. However, this is contingent on external forces and is out of CVR’s control, though all key stakeholders are working tirelessly to expedite the process.
CVR also announces the intention to submit to Health Canada for a Canadian Medical Device License (MDL), which would provide approval for sales of the CSS into Canada, forthwith upon submitting to the FDA for market clearance. Obtaining the ability to distribute the CSS into Canada would open up one of the largest global economies, with diagnostic imaging representing 21% of their $6.7 billion medical device market.
Peter Bakema, CEO and Board Chairman, elaborates on the decision to launch into Canada in the near term, “It only makes sense that we continue our momentum with submitting to the FDA, leveraging both relational and geographic advantages. With the submission requirements mirroring most of the EU Technical dossier we will be able to advance the pace of global availability.”
About CVR Medical
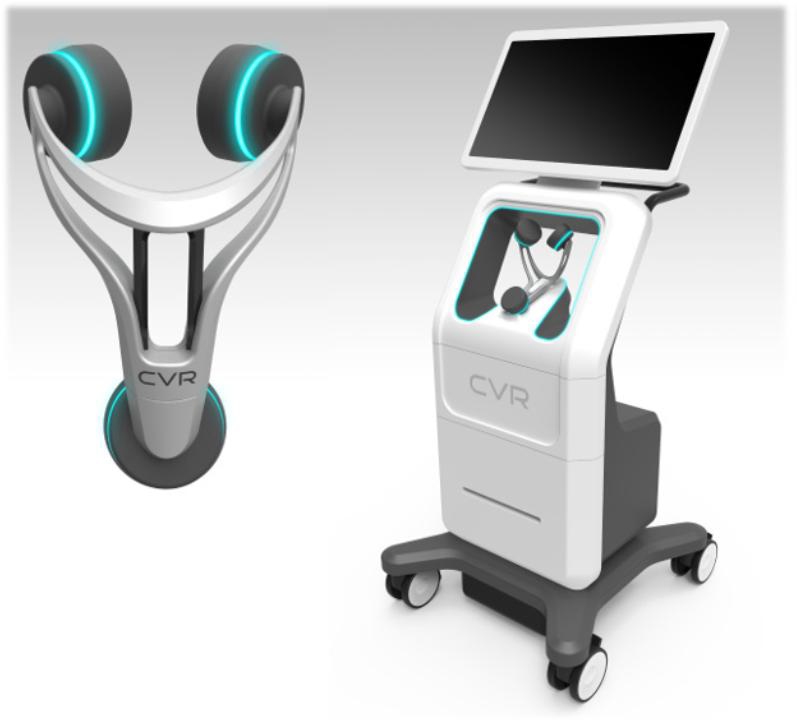
CVR Medical Corp. is a healthcare company that operates in the medical device industry focused on the commercialization of its disruptive, proprietary Carotid Stenotic Scan (CSS). The CSS device is a diagnostic tool that encompasses subsonic, infrasonic, and low frequency sound wave analysis technology. The CSS is a patented device designed to detect and measure carotid arterial stenosis which significantly increases the risk of stroke. CVR is currently in pivotal clinical trials in preparation for its planned submission to the FDA. CVR is led by an experienced and proven team of professionals with extensive healthcare, medical device, international expansion, regulatory and sales experience. CVR Medical trades on the TSX Venture Exchange under the symbol CVM. Additional information regarding the Company can be found in our recent filings with the SEDAR as well as the information maintained on our website at www.cvrmed.com
ON BEHALF OF THE BOARD:
(signed) "Peter Bakema"
CEO, President & Chairman
For further information contact:
Peter Bakema, CEO, President and Chairman
Email: info@cvrmed.com
or
Marc S. Lubow.
Vice President Capital Markets, Director / Investor Relations
marclubow@cvrmed.com
This press release contains forward-looking information that involves various risks and uncertainties regarding future events related to the Joint Venture. Such statements are subject to risks and uncertainties that may cause actual results, performance or developments to differ materially from those contained in the statements and are not guarantees of future performance of the Company. No assurance can be given that any of the events anticipated by the forward-looking statements will occur or, if they do occur, what benefits the Company will obtain from them. These forward-looking statements reflect management's current views and are based on certain expectations, estimates and assumptions which may prove to be incorrect. A number of risks and uncertainties could cause our actual results to differ materially from those expressed or implied by the forward-looking statements, including: (1) a downturn in general economic conditions in North America and internationally, (2) the inherent uncertainties and speculative nature associated with commercialization of technology and the practice of medicine, (3) a change in health regulations, (4) any number of events or causes which may delay or cease commercialization and development of the Joint Venture, (5) the risk that the Company or the Joint Venture does not execute its business plan, (6) inability to retain key employees, (7) inability to finance operations and growth, and (8) other factors beyond the Company's control. These forward-looking statements are made as of the date of this news release and, except as required by law, the Company assumes no obligation to update these forward-looking statements, or to update the reasons why actual results differed from those projected in the forward-looking statements.
THE TSX VENTURE EXCHANGE INC. HAS NEITHER APPROVED NOR DISAPPROVED THE CONTENTS OF THIS PRESS RELEASE. NEITHER THE TSX VENTURE EXCHANGE NOR ITS REGULATION SERVICES PROVIDER (AS THAT TERM IS DEFINED IN THE POLICIES OF THE TSX VENTURE EXCHANGE) ACCEPTS RESPONSIBILITY FOR THE ADEQUACY OR ACCURACY OF THIS RELEASE.
Looks like everything is on track ! And a Canadian approval would be a good boost.
https://mailchi.mp/4b06fe56a644/cvr-medical-provides-update?e=7e3ff08634
Dubey - I don’t have premium service to answer your question. And I’m here to post my opinions and if I can help anyone I will.
Back on May 9, 2018, the CEO stated it would be 60-90 day extension to FDA submission of the De Novo process. However, once again we seem to be sitting here as investors with this CEO continuing to set expectations and fail to meet them.
I get that this process is long and difficult, and I am a patient person. However, in my experiences setting expectations with investors and the market over and over again, and missing those dates, or failing to update them continues to scare me.
Does anyone in this forum have a different perspective? It just seems that the communication is way to "positive" and fails to meet.
Sorry dubey - spell ck
Hello dunes,
I went to a private presentation given by CVR. I meet some of the board members, and I know Peter from a previous meeting. I’m part of small group investment group that has done a private placement with CVR Medical. I believe in the company and the people in Management. This device can change the industry and save lives for the future.
do you mind expanding on the conference you attended. who put it on? was it exclusive to CVR or was it a large medical device conference? Did not see any press releases or news about it, just curious. TY
Yep. WORLD wide. Ca ching! I envision a $10 pps less the 2yrs. Have 45 thou shares currently. To make coin and help humanity at the same time? Sweet!
Good post. Got to love the potential. This will be as common as a blood pressure check on your annual exam.
Presentation- This past weekend I went to a CVR Medical presentation for the Carotid Stenotic Scan device. I was amazed at the potential this machine has for the future. The CSS device creates a new medical market that can saves lives and creates new revenue for doctors. This is an inexpensive first line defense for unknown future stroke victims. The CSS device is easily used by personnel in a doctor’s offices as I experienced. The numbers work for medical personnel to show a profit for doctors and health professionals very quickly. Take a look at management and the medical professionals involved with CVR Medical. They have many years of experience and are very talented in their field. I was very impressed on how everything is coming together for FDA submission.
I had the test performed on me, it took only a few minutes. Then I had a Doppler sonogram test to correlate the two tests. The two tests were conclusive to each other and had similar results. It is amazing how the CSS device will revolutionize the health services industry for strokes ! Stay the course and keep believing !
I'm with you nugnug. Same exact average and everything.
Looks like someone dumped 25K shares at .24 and a few small buys walked it up. It is such a low volume and low float, any buying or selling can be volatile for movement. Lets hope the MMs keep the ask at .27 tomorrow.
Any gauge of purchase interest by doctors or networks? Any medical associations endorsing the concept?
IMO, this is a long-play, no-brainer for a 10 to 20 bagger in 12-24 months. But this isn't a sexy stock since there are not any swings to play and not enough volume to flip. It could still all fall apart and I lose my investment, but the deck is being stacked for success. I am perfectly happy holding and adding when I can. My average is .27 and I am fine being red on this one. We all still have plenty of time to accumulate shares before the uphill climb.
Kanter coming on is and end game strategy to get this over the goal line. Great News!
In My Opinion of course... GLTA
Mr. Kanter. Venture capitalist dealing in billions. History Share holder value increase in billions. I think I may be hyperventilating!
Leading Medical Device / Life Science / Healthcare Veteran Joel Kanter Joins Board of Directors of CVR Medical
DENVER, NC / October 15th, 2018 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces that Mr. Joel Kanter has joined the Board of Directors. Mr. Kanter has served as President of Windy City, Inc. a privately held investment firm since July 1986. Mr. Kanter, is a veteran healthcare venture capitalist whose family office has financially backed numerous medical device, life science and healthcare companies throughout his 35-year career. Mr. Kanter has helped create billions in shareholder value, most notably; I-Flow which in 2009 was acquired by Kimberly-Clark for $324M, Clarisonic which in 2011 was acquired by L’Oréal for $525M, Prolor BioSciences which was acquired by OPKO Health in 2013 for USD $480M, Encore Medical which was acquired in 2006 by Blackstone for $860M, GranCare which in 1999 completed a $1.8B merger with Living Centers of America led by Apollo Management and Walnut Financial where he was Chief Executive Officer which was acquired in a transaction with Tower Hill Capital which resulted in an increase of $400M in shareholder value in 1999.
Mr. Kanter currently serves on the Board of Directors of two public companies including Magna-Labs, Inc. and MEDITE Cancer Diagnostics, Inc. and also serves on the Boards of several private concerns including Fibralign Corporation, First Wave Technologies, Orpheus Biosciences, Inc., Primal Therapies, Inc., and Serpin Pharma.
Mr. Kanter is also a current Trustee Emeritus and past President of the Board of Trustees of The Langley School in McLean, Virginia, a former Trustee at the Georgetown Day School in Washington, D.C., and of the Union Institute & University, the Country’s first Online University. He is also the current Board Chair of the Black Student Fund, and serves on the Kennedy Center’s National Committee on the Performing Arts.
Peter Bakema, Chairman and CEO of CVR Medical, states, “We are thrilled to welcome Joel to our Board of Directors and to have his rare breadth of experience and knowledge going forward. His track record of investment and acquisition success speaks to a visionary quality that we strive for at CVR. His involvement in moving our Carotid Stenotic Scan (CSS) device to market is another confident step towards providing a solution for a major sector of the healthcare industry beset with high costs and access barriers.”

It seems to me that a lot of short positions are being liquidated on the past good news from the critical trial.
CVR Medical Announces Proposed Financing
VANCOUVER, BC / August 23rd, 2018 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (FRANKFURT: B3BN) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces that it intends to complete a non-brokered private placement financing (the "Financing") of units (each, a "Unit") for gross proceeds of up to C$2,000,000 with a price per Unit of C$0.30 or such other price determined by CVR Medical management in compliance with TSX Venture Exchange (the "Exchange") pricing regulations.
Each Unit will consist of one common share (each, "Share") and one common share purchase warrant (each, a "Warrant"), with each Warrant exercisable to acquire one additional Share (each, a "Warrant Share") at a price of C$0.36 per Warrant Share for a period of five (5) years following the closing date of the Financing ("Closing Date").
The Warrants will be subject to an acceleration right (the "Warrant Acceleration Right") if on any ten (10) consecutive trading days, beginning on the date that is four (4) months and one (1) day following the Closing Date, the daily closing price of the Shares on the Exchange is at or greater than C$0.50. If the Company exercises its Warrant Acceleration Right, the new expiry date of the Warrants will be the 30th day following the date on which such notice is given by the Company.
CVR Medical intends to pay finder’s fees of up to 6% in cash and 6% in finder's warrants in connection with the Financing. Proceeds from the Financing are expected to be used for ongoing working capital requirements relating to the development and commercialization of the proprietary subsonic, infrasonic, and low frequency sound wave analysis technology and diagnostic device designed to detect and measure carotid arterial disease, known as the CSS Device.
Completion of the Financing is subject to Exchange acceptance and all securities issued pursuant to the Financing will be subject to a hold period of four months as required under applicable securities legislation.
The securities referred to herein will not be or have not been registered under the United States Securities Act of 1933, as amended, and may not be offered or sold in the United States absent registration or an applicable exemption from registration requirements.
All interested accredited investors who wish to participate may contact the Company directly at marclubow@cvrmed.com.
About CVR Medical
CVR Medical Corp. is a healthcare company that operates in the medical device industry focused on the commercialization of its disruptive, proprietary CSS Device. The CSS device is a diagnostic tool that encompasses subsonic, infrasonic, and low frequency sound wave analysis technology. The CSS Device is a patented device designed to detect and measure carotid arterial stenosis. CVR is currently in pivotal clinical trials in preparation for its planned submission to the FDA. CVR is led by an experienced and proven team of professionals with extensive healthcare, medical device, international expansion, regulatory and sales experience. CVR Medical trades on the TSX Venture Exchange under the symbol CVM. Additional information regarding the Company can be found in our recent filings with the SEDAR as well as the information maintained on our website at www.cvrmed.com
ON BEHALF OF THE BOARD:
(signed) "Peter Bakema"
CEO, President & Director
For further information contact:
Peter Bakema, CEO, President and Director
Email: info@cvrmed.com
or
Marc S. Lubow.
Vice President Capital Markets, Investor Relations
904-923-4037
marclubow@cvrmed.com
This press release contains forward-looking information that involves various risks and uncertainties regarding future events related to the Joint Venture. Such statements are subject to risks and uncertainties that may cause actual results, performance or developments to differ materially from those contained in the statements and are not guarantees of future performance of the Company. No assurance can be given that any of the events anticipated by the forward-looking statements will occur or, if they do occur, what benefits the Company will obtain from them. These forward-looking statements reflect management's current views and are based on certain expectations, estimates and assumptions which may prove to be incorrect. A number of risks and uncertainties could cause our actual results to differ materially from those expressed or implied by the forward-looking statements, including: (1) a downturn in general economic conditions in North America and internationally, (2) the inherent uncertainties and speculative nature associated with commercialization of technology and the practice of medicine, (3) a change in health regulations, (4) any number of events or causes which may delay or cease commercialization and development of the Joint Venture, (5) the risk that the Company or the Joint Venture does not execute its business plan, (6) inability to retain key employees, (7) inability to finance operations and growth, and (8) other factors beyond the Company's control. These forward-looking statements are made as of the date of this news release and, except as required by law, the Company assumes no obligation to update these forward-looking statements, or to update the reasons why actual results differed from those projected in the forward-looking statements.
THE TSX VENTURE EXCHANGE INC. HAS NEITHER APPROVED NOR DISAPPROVED THE CONTENTS OF THIS PRESS RELEASE. NEITHER THE TSX VENTURE EXCHANGE NOR ITS REGULATION SERVICES PROVIDER (AS THAT TERM IS DEFINED IN THE POLICIES OF THE TSX VENTURE EXCHANGE) ACCEPTS RESPONSIBILITY FOR THE ADEQUACY OR ACCURACY OF THIS RELEASE.
NOT FOR DISTRIBUTION TO UNITED STATES NEWSWIRE SERVICES OR FOR DISSEMINATION IN THE UNITED STATES
FDA submission target Oct. 2018! Tell a friend!
CVR MEDICAL REPORTS POSITIVE INTERIM RESULTS FROM ONGOING PIVOTAL TRIAL WITH CSS DEVICE
CVR MEDICAL REPORTS POSITIVE INTERIM RESULTS FROM ONGOING PIVOTAL TRIAL WITH CSS DEVICE
New PR
CVR Medical Enlists JD Lymon Group to Provide Reimbursement and Study Design Support
VANCOUVER, BC / August 14th, 2018 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (FRANKFURT: B3BN) (OTCQB: CRRVF) (“CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector has retained the services of Minneapolis-based medical device consulting firm, JD Lymon Group (www.jdlymon.com) to provide ongoing reimbursement guidance and to help support the company’s FDA submission of the Carotid Stenotic Scan (CSS) device for US market clearance by providing clinical trial design support.
With more than 70 combined years of medical device and pharmaceutical experience across the firm’s partners, JD Lymon focuses on accelerating market access through multidisciplinary strategies that address the complex interrelationship between policy, evidence, and practice in order to optimize the market position of emerging therapies. Specifically, JD Lymon will be guiding CVR through the complex and ever evolving reimbursement landscape. This will be done by designing the clinical trials in support of FDA submission, payer advocacy, and overall medical reimbursement coding needs.
CVR Medical Chief Operating Officer Tony Robinson stated, “As we prepare our FDA submission and eventual market release for the CSS device, the JD Lymon team will apply its insights and experience to help map out and articulate a reimbursement pathway to government and commercial payors.
Chris Lyle, Partner at JD Lymon, states, “We are honored to be selected by CVR to support their market access and study design needs. We understand the reimbursement challenges faced by small companies and emerging technologies. We look forward to a great collaboration and advancing the CSS System.”
About CVR Medical
It's hard to say. It might have just been someone wanting to play another runner and knowing he can come back here 6 months from now and probably get right back in between .25 and .30. Someone could have died and his children liquidated his accounts and bounced. Who is to say really. I am not that worried about it. This stock will bounce from .25-.30 for a while i bet.
If someone did get word of bad trials and dumped, why risk insider trading and felony charges for a few hundred grand. I wouldn't.
On the other hand, if it was a long position that was stopped out or the owner sold because they might have heard something about the trials, then that puts a different spin on it. Granted, in either scenario we wouldn't know if anything was learned of the trials. It could just be profit taking on the short side or just fear on the long.
I'm trying to find out if it was a reversal of a short position from someone that knows, or heard, something about the trials. If something is coming down the pike that is positive from the trials, a large short position would want to liquidate and then go long. Is there anywhere to tell if a short position was liquidated?
I don't know exactly. but it was quite a few. The biggest single sell i remember seeing was 119000 shares. It was lots of smaller 5000-35000 sells.
Like i mentioned, it really looked like a stop loss was triggered or someone just dumped at market and drove the price down. I managed to grab 5000K at .21 but that is all I had funds for.
I actually meant the day before yesterday...you know, the day of the big drop of 800K shares, how many trades were made that day?
|
Followers
|
17
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
486
|
|
Created
|
03/16/17
|
Type
|
Free
|
| Moderators | |||









Michael Rhodes
Vice President of Quality Systems
•20 Years of Quality Know-how
•Former Vice President of Quality for HSBC & Motorola
•Experience in multiple markets including healthcare, defense, public safety, retail, energy, and manufacturin

Dr. W. Douglas Weaver
CVR Global Board of Directors / Scientific Advisory Board
•Past President of the American College of Cardiology
•Former Vice President and System Medical Director of Heart and Vascular Services at Henry Ford Health System
•Over 330 publications, most of which are related to drug and device discovery and methods toward improving the care provided to patients with cardiovascular disease

| Shares on Issue: | 73,832,151 |
| Options: | 4,050,000 |
| Warrants ($0.40): | 6,382,949 (Expires September 15, 2018) |
| Warrants ($0.70): | 2,197,591 (Expires August 22, 2018) |
| Warrants ($0.70): | 3,261,120 (Expires October 19, 2018)) |
| Warrants ($0.70): | 830,000 (Expires October 22, 2018) |
| Warrants ($0.70): | 444,600 (Expires February 26, 2019) |
| Warrants ($0.70): | 408,286 (Expires March 6, 2019) |
| Warrants ($0.70): | 107,500 (Expires March 16, 2019) |
| Warrants ($0.70): | 712,823 (Expires March 21, 2019) |
| Warrants ($0.70): | 700,000 (Expires May 02, 2019) |
| Warrants ($0.70): | 855,193 (Expires June 18, 2019) |
| Warrants ($1.50): | 2,518,600 (Expires April 6, 2020) |
| Trading Symbols: | TSX.V: CVM FRANKFURT: B3BN OTCQB: CRRVF |
| CUSIP #: | 126632108 |
| ISIN #: | CA1266321084 |
| Transfer Agents: | AST Trust Company (Canada)1600 – 1066 West Hastings St. American Stock Transfer & Trust Company, LLC6201 15th Avenue |
| IR Contact: | Brisco Capital Partners Corp.Scott Koyich, President |
CVR Intellectual Property Summary & Legal Comment
CVR has instituted a comprehensive IP policy to cover several aspects of the CSS device as it prepares to go to market. The IP includes 1 issued US patent, US 9,101,274. An European Counterpart is pending at the moment that parallels the allowed US case. A continuation-in-part, Application No. 14/803,389 is also pending, with a priority date extending back to June 24, 2010.
In June of 2015, CVR file two applications covering the Yoke, utilized for holding the sensors and positioning on a patient, and methods of quantifying and detecting sounds in the carotid artery. Both applications were converted into PCT applications in June of 2016 and now await conversion into the US and other jurisdictions at the end of 2017. These applications have been assigned to a joint venture, but an exclusive license remains with CVR.
In June of 2016, CVR filed five additional provisional patent applications covering several aspects of the CSS device, including additional methods for detecting blockage in a fluid flow vessel, disposable sensing elements (think of the razor/razor blade model), methods for generating clean data for analysis, methods for detecting sounds and determining stenosis, methods for determining appropriate location for detecting fluid flow in the body, and methods for detecting and ablating stenosis.
In December of 2016, CVR filed an additional provisional application directed towards sensing pods that do not need a yoke, in order to position the sensing pods for determining stenosis.
Finally, in January of 2017, CVR filed two design patents, one directed towards the device cart itself, and the second to a version of the Yoke for positioning sensors on a patient.
A summary of these are included in the attached Excel file. These filings are highly confidential and not to be disseminated without authorization by CVR.
CVR continues to develop and expand its IP portfolio as development continues. It is expected that additional applications will be filed to cover final versions of any product that will go to market. Such protection will include patent, design patent, trademark, trade dress, and copyright, where applicable.
CVR also expects that the several pending provisional applications will be converted into PCT applications and that the pending PCT applications (now assigned to a joint venture), will also be converted in due course.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
