Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Good for you. Keep it up.
Got some today at .132. My gut said wait it'll hits .10 today. Chicken because the high volume said possibly news soon. A thou$ with .10 would get you 10 thou. when this baby blows past one dolla GI! Of fringing well...
The TSX has pushed the deadline for a reversal of the suspension back to 30th august giving CVR Medical a little longer to sort out its relationship with CVR Global.
Don't worry about it, just a mix up with the auditors because for half the reporting year they were trading as a joint venture with CVR global. They MUST resolve before 7th August to prevent downgrading on the TSX . . will be resolved SOON
I am very concerned about the trading freeze in British Columbia announced in May of this year. Has that freeze been listed? There has been no update as to when audited results will be announced. Even more worrying is that it seems the delay in reporting has caused some major US to stop providing accurate data on their platforms. Charles Schwab volume data has been listed at at same number since the announcement. If major trading platforms are not accurate when the news of FDA approval is announced, i am afraid this stock will “ bloom in the dark” and the true value of this wonderful product will not be realized for patient investors. I urge all to contact the companie’s investor relations to rectify this problem Immediately.
Quiet. Any longs hiding in the shadows? Here's what I can best guess piece together. Always remember, if there is a major problem by law they must report to stock holders. The restructuring made the financials more complicated. The FDA approval is on track but most likely had additional questions. IMO they don't want to post financial glitches with FDA positive tract. Both will IMO be posted same time to propel PPS. Been here 2yrs 4mos. The science is solid!
Sorry, crossed another stock symbol with this, disregard.
I've been following for a while with a starter position. Haven't seen any news to account for the last 2 days activity. Did I miss something?
Good point gc2...Good point gc2...Hello hello...
Longs that have been here for yrs like me, put your orders in the high dollar range. When she takes off the market computer averages the ask and it's safer too scope down than chase up. Hello...
Longs that have been here for yrs like me, put your orders in the high dollar range. When she takes off the market computer averages the ask and it's safer too scope down than chase up. Hello...
Got order in at 4.10. Anticipating a launch? Advise high PPS NOW!
Probably better than OB*P. 2016 shot up to $7 PPS 0n a wing and a prayer. Now .0076, but still chugging along in the developmental stage. In contrast, CRRVF is solid in your face science. I suspect CRRVF is not where it should be PPS because there really has been relatively very little news push. Been in 2.4 yrs. Hopefully see FDA approval this month and launch to 2-3 $ PPS. Then on to NASDAQ.
This is finally getting into a good buy range
My guess is there is a minor hiccup re the yearly financials and the restructuring. The science is solid and Cannon is partnered. Expect to see report and FDA approval soon.
Its telling that the board member who resigned had only been with CVR fro a couple of months so it's likely that he just wasn't up to, or couldn't perform the role. Whether or not he was responsible for the late filing is a matter for conjecture; I'm just happy that the size of the BOD has reduced and so has the salary bill, so see the resignation as a positive.
B.S. mm mauling us. That was simply bad management and bad news , just that simple. Board members resigning, why. Maybe the MM ‘ s are mauling them.
MMs mauling us. Don't give them cheap shares. The science re this life saving device is solid. Don't let them kill your future!
...Mostly CIBC CANADIAN IMPERIAL BANK...
No...it's MMs doing the Poki. Verified on another site. Doing the Pokie to scare pi's to give up cheap shares before FDA APPROVAL boom to 1$.
That coupled with balance billing to produce an in office income prior to insurance acceptance. I would wonder if they would want to keep it out of the insurance companies hands. The Insurance companies will dilute the value for the provider so they can pay literally nothing in reimbursement to the physicians that front the money to purchase the new technology. IMHO.
The ease of use of the device, couple with no requirement for special training--therefore lower labor costs--is really making this technology an easy to justify investment for physicians.
Lower priced stocks can have awesome opportunities with big gains as we know. Here is a new Medical device, and this testing machine can be a new innovation since the EKG machine.
* This new CSS device can revolutionize how strokes can be detected and it can save many lives.
- HIGH GROWTH MARKETS & SIGNIFICANT ADVANTAGES OF THE CSS- Stroke and Carotid Artery disease is the 5th Leading Cause of Death, with 795,000 Stroke Incidents Yearly, and 140,232 Deaths Yearly. $11B Opportunity.
- The CSS machine is in the middle of De Novo application with the FDA.
- As of now clinical trials are being performed in Mayo Clinic, Cleveland Clinic, Henry Ford Hospital, Thomas Jefferson University and more. More TBA
- Game changing technology - 2 minute detection; Mobile Devices, 10-15 minute train for health professionals to preform test
- “Cost" of strokes expected to TRIPLE by 2030 from 2010 $71.6 Billion cost globally(2010)
- Carotid Stenotic Scan(CSS) - groundbreaking tool assessing arterial health in a way unavailable to patient/provider now
- Manufacturing Agreement signed with Canon Virginia Inc(subsidiary of Canon USA - also a Strategic Partner)
- Currently in Clinical Trials and submitted in January 2019 to FDA for US Market Approval
* This is a great investment opportunity and it could have huge gains. CVR Medical has huge potential and can possibly change how we look at detecting stroke, check it out.
Symbols: CVRRF and CVM ........... Website: CVRMedical.com
https://www.gurufocus.com/news/832105/5-biotech-stocks-to-keep-an-eye-on-in-2019
Anyone want to have fun with predictions? Been doing this for sometime and my gut says upon FDA approval will pop approaching 2$ pls and then will sell off approaching 1$ until revs commence. 2$ may be conservative!!
CRRVF INVESTMENT HIGHLIGHTS
*Strokes - leading cause of disability globally; 5th leading cause of death(US); Asymptomatic
*Game changing technology - 2 minute detection; Mobile Devices
*"Cost" of strokes expected to TRIPLE by 2030 from 2010 $71.6 Billion cost globally(2010)
*Carotid Stenotic Scan(CSS) - groundbreaking tool assessing arterial health in a way unavailable to patient/provider now
*Manufacturing Agreement signed with Canon Virginia Inc(subsidiary of Canon USA - also a Strategic Partner)
*Currently in Clinical Trials and submitted in January 2019 to FDA for US Market Approval
Patiently waiting here..Once approved its off to the races and who knows how high it can go..
Large daily volumes equals with soon FDA approval a pop over 1 dolla GI!
Canon to Showcase CVR Medical's Carotid Stenotic Scan (CSS) at Medical Design & Manufacturing West (MD&M)
DENVER, NC / February 5th, 2019 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR”) a Canadian listed and US based healthcare company in the medical device sector announces it will have its upcoming “Carotid Stenotic Scan (CSS)” device showcased by its manufacturing and strategic partner Canon Virginia, Inc. (CVI) a wholly owned subsidiary of Canon USA at this week’s Medical Design & Manufacturing West (MD&M) in Anaheim, CA. MD&M West is known as the largest and most comprehensive yearly medical technology and manufacturing expo and conference in North America. CVR has partnered with CVI for the manufacturing, supply chain management, logistics, white glove delivery to end user, telephone tech support, and on-site service for the CSS; providing key advantages when CVR scales to meet potential global demand.
From February 5-7, over 19,000 members of the global medical care and manufacturing community will gather at MD&M West to learn about and exhibit the latest in cutting-edge advancements. With Canon’s presence as a recognized leader in imaging technology, CVR will have the opportunity to gain invaluable exposure and credibility within the industry. Canon’s showcase of the CSS will highlight the unique, customized manufacturing of the device, as well as logistics and product support needs. The manufacturing partnership between CVR Medical and Canon, announced in 2017, established the production means through which the CSS will be launched.
CVR Chief Executive Officer Peter Bakema states, “We are excited to have the CSS displayed at MD&D West with our incredible partners at Canon. Consumers across the world resonate with their brand as one that creates user-friendly, reliable, high-quality products. Combined with our industry-disrupting, noninvasive diagnostic technology, we expect to stand out on this stage and continue building momentum toward the CSS market launch.”
Visit Canon at MD&M West, booth #3901, to learn more about the CSS and discuss future opportunities.
About CVR Medical
CVR Medical Corp. is a healthcare company that operates in the medical device industry focused on the commercialization of its disruptive, proprietary Carotid Stenotic Scan (CSS). The CSS device is a diagnostic tool that encompasses subsonic, infrasonic, and low frequency sound wave analysis technology. The CSS is a patented device designed to detect and measure carotid arterial stenosis. CVR is currently in clinical trials at several research institutions and has submitted its CSS device to the FDA as a De Novo application. CVR is led by an experienced and proven team of professionals with extensive healthcare, medical device, international expansion, regulatory and sales experience. CVR Medical trades on the TSX Venture Exchange under the symbol CVM. Additional information regarding the Company can be found in our recent filings with the SEDAR as well as the information maintained on our website at www.cvrmed.com
ON BEHALF OF THE BOARD:
(signed) "Peter Bakema"
CEO, President & Director
For further information contact:
Peter Bakema, CEO, President and Director
Email: info@cvrmed.com
or
Marc S. Lubow.
Executive Vice President
(904) 923 - 4037
marclubow@cvrmed.com
CVR Medical Mourns The Passing of Ron Birch, Director
DENVER, NC / January 14th, 2019 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces that Ron Birch, Director passed away January 9th, 2019. Mr. Birch has been a member of the Board of Directors since the inception of CVR Medical in 2016.
Peter Bakema, Chairman and CEO commented “It is with heartfelt sadness that we announce the loss of not only a great business man, but also a wonderful human being. Ron’s years of public sector experience and leadership were instrumental in the advancement of this project. We are grateful not only for his business acumen, but more importantly for the example he set of how to treat people with respect and dignity in all his dealings. The board has lost a great man and our deepest sympathies go out to his family.”
Yup..This last two PRs made me add shares..
With the hard work that Peter Bekema and his crew are putting in we are witnessing this company is coming into fruition..
Prestigious Mayo Clinic Approves CVR Medical’s CSS Device For IRB Clinical Trial
Company Begins Pre-Market Activities With Clinical Foot Print Expansion
DENVER, NC / January 7th, 2019 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces Internal Review Board (IRB) approval by Mayo Clinic’s Gonda Vascular Center sanctioning CVR to conduct clinical trials using the Carotid Stenotic Scan (CSS) device. The famed academic medical center located in Rochester, Minnesota will become the latest distinguished source for gathering results-based and user experience data on the CSS device. Thanila A. Macedo, M.D., Associate Professor and a Radiologist at Mayo Clinic Rochester will lead the clinical trial as Primary Investigator. Dr. Macedo’s interest in noninvasive diagnostics aligns with CVR’s goal of delivering a novel device that will alter the way carotid artery stenosis is examined and treated.
The Mayo Clinic is recognized for high-quality patient care more often than any other academic medical center in the nation. U.S. News & World Report ranked Mayo Clinic's campus in Rochester, Minn. the best hospital in the nation in its 2018-2019 rankings. Mayo Clinic has ranked at or near the top of "Best Hospitals Honor Roll" hospitals throughout the history of U.S. News and World Report's best hospital rankings.
“We’re thrilled to begin working with the vascular medicine specialists at the Mayo Clinic,” states CVR Chairman & CEO Peter Bakema. “It means the world to us to be associated with this caliber of hospital and its professionals that are on the forefront of advancing medicine. Our goal is to use this technology to benefit everyone: patients, their families, doctors, clinics and entire hospital systems. Beyond the invaluable insight gained through trials, the partnerships we continue to grow with the nation’s most reputable healthcare centers strengthens the CVR Medical brand, making that vision more and more a reality.”
Having just announced the De Novo Submission of the CSS to the FDA, CVR Medical has begun additional pre-market activities which will include the further expansion of our clinical foot print. CVR further identified multiple additional research institutions who have expressed announce interest and intend to launch clinical trials throughout 1H 2019.
About CVR Medical
CVR Medical Corp. is a healthcare company that operates in the medical device industry focused on the commercialization of its disruptive, proprietary Carotid Stenotic Scan (CSS). The CSS device is a diagnostic tool that encompasses subsonic, infrasonic, and low frequency sound wave analysis technology. The CSS is a patented device designed to detect and measure carotid arterial stenosis. CVR is currently in pivotal clinical trials in preparation for its planned submission to the FDA. CVR is led by an experienced and proven team of professionals with extensive healthcare, medical device, international expansion, regulatory and sales experience. CVR Medical trades on the TSX Venture Exchange under the symbol CVM. Additional information regarding the Company can be found in our recent filings with the SEDAR as well as the information maintained on our website at www.cvrmed.com
ON BEHALF OF THE BOARD:
(signed) "Peter Bakema"
CEO, President & Director
For further information contact:
Peter Bakema, CEO, President and Director
Email: info@cvrmed.com
or
Marc S. Lubow.
Executive Vice President
904-923-4037
marclubow@cvrmed.com
This press release contains forward-looking information that involves various risks and uncertainties regarding future events related to the Joint Venture. Such statements are subject to risks and uncertainties that may cause actual results, performance or developments to differ materially from those contained in the statements and are not guarantees of future performance of the Company. No assurance can be given that any of the events anticipated by the forward-looking statements will occur or, if they do occur, what benefits the Company will obtain from them. These forward-looking statements reflect management's current views and are based on certain expectations, estimates and assumptions which may prove to be incorrect. A number of risks and uncertainties could cause our actual results to differ materially from those expressed or implied by the forward-looking statements, including: (1) a downturn in general economic conditions in North America and internationally, (2) the inherent uncertainties and speculative nature associated with commercialization of technology and the practice of medicine, (3) a change in health regulations, (4) any number of events or causes which may delay or cease commercialization and development of the Joint Venture, (5) the risk that the Company or the Joint Venture does not execute its business plan, (6) inability to retain key employees, (7) inability to finance operations and growth, and (8) other factors beyond the Company's control. These forward-looking statements are made as of the date of this news release and, except as required by law, the Company assumes no obligation to update these forward-looking statements, or to update the reasons why actual results differed from those projected in the forward-looking statements.
CVR Medical Completes FDA Submission for Highly Anticipated CSS Device
DENVER, NC / January 7th, 2019 / CVR Medical Corp. (CVM.V) (TSXV: CVM) (OTCQB: CRRVF) ("CVR Medical”) a Canadian listed and US based healthcare company in the medical device sector announces it has completed a Food and Drug Administration De Novo Submission for their “Carotid Stenotic Scan (CSS)” device. A De Novo Submission allows for a company to receive a “not substantially equivalent” risk-based evaluation for any legally marketed device for which there is no prior, comparable classification. As a novel device that uses patented sound wave analysis technology to detect and measure carotid arterial stenosis, the CSS fits within the criteria of this FDA filing. Due to US government shutdown, the submission will be reviewed by the FDA upon resumption of government activity.
Peter Bakema, Chairman and CEO of CVR Medical, states, “Today’s FDA submission marks the culmination of CVR’s many years of product development, design, trials and organizational preparation. It is an important and necessary step for any new device entering the market, and one that is worth the wait to be executed correctly. I am proud of our staff and partners around the world for their contributions in getting us to this stage, and look forward to keeping shareholders, as well as the greater public, up to date on the process as it moves ahead. We at CVR will continue to work during the submission phase to ensure that upon its success, we are ready to maintain momentum with the market release of the CSS.”
About CVR Medical
CVR Medical Corp. is a healthcare company that operates in the medical device industry focused on the commercialization of its disruptive, proprietary Carotid Stenotic Scan (CSS). The CSS device is a diagnostic tool that encompasses subsonic, infrasonic, and low frequency sound wave analysis technology. The CSS is a patented device designed to detect and measure carotid arterial stenosis. CVR is currently in pivotal clinical trials in preparation for its planned submission to the FDA. CVR is led by an experienced and proven team of professionals with extensive healthcare, medical device, international expansion, regulatory and sales experience. CVR Medical trades on the TSX Venture Exchange under the symbol CVM. Additional information regarding the Company can be found in our recent filings with the SEDAR as well as the information maintained on our website at www.cvrmed.com
ON BEHALF OF THE BOARD:
(signed) "Peter Bakema"
CEO, President & Director
For further information contact:
Peter Bakema, CEO, President and Director
Email: info@cvrmed.com
or
Marc S. Lubow.
Executive Vice President
marclubow@cvrmed.com
(904) 923 4037
This press release contains forward-looking information that involves various risks and uncertainties regarding future events related to the Joint Venture. Such statements are subject to risks and uncertainties that may cause actual results, performance or developments to differ materially from those contained in the statements and are not guarantees of future performance of the Company. No assurance can be given that any of the events anticipated by the forward-looking statements will occur or, if they do occur, what benefits the Company will obtain from them. These forward-looking statements reflect management's current views and are based on certain expectations, estimates and assumptions which may prove to be incorrect. A number of risks and uncertainties could cause our actual results to differ materially from those expressed or implied by the forward-looking statements, including: (1) a downturn in general economic conditions in North America and internationally, (2) the inherent uncertainties and speculative nature associated with commercialization of technology and the practice of medicine, (3) a change in health regulations, (4) any number of events or causes which may delay or cease commercialization and development of the Joint Venture, (5) the risk that the Company or the Joint Venture does not execute its business plan, (6) inability to retain key employees, (7) inability to finance operations and growth, and (8) other factors beyond the Company's control. These forward-looking statements are made as of the date of this news release and, except as required by law, the Company assumes no obligation to update these forward-looking statements, or to update the reasons why actual results differed from those projected in the forward-looking statements.
This is really good !
http://cvrmed.com/press-release/cvr-medical-completes-tuv-testing-2-2/
This is really good !
http://cvrmed.com/press-release/cvr-medical-completes-tuv-testing-2-2/
|
Followers
|
17
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
486
|
|
Created
|
03/16/17
|
Type
|
Free
|
| Moderators | |||









Michael Rhodes
Vice President of Quality Systems
•20 Years of Quality Know-how
•Former Vice President of Quality for HSBC & Motorola
•Experience in multiple markets including healthcare, defense, public safety, retail, energy, and manufacturin

Dr. W. Douglas Weaver
CVR Global Board of Directors / Scientific Advisory Board
•Past President of the American College of Cardiology
•Former Vice President and System Medical Director of Heart and Vascular Services at Henry Ford Health System
•Over 330 publications, most of which are related to drug and device discovery and methods toward improving the care provided to patients with cardiovascular disease

| Shares on Issue: | 73,832,151 |
| Options: | 4,050,000 |
| Warrants ($0.40): | 6,382,949 (Expires September 15, 2018) |
| Warrants ($0.70): | 2,197,591 (Expires August 22, 2018) |
| Warrants ($0.70): | 3,261,120 (Expires October 19, 2018)) |
| Warrants ($0.70): | 830,000 (Expires October 22, 2018) |
| Warrants ($0.70): | 444,600 (Expires February 26, 2019) |
| Warrants ($0.70): | 408,286 (Expires March 6, 2019) |
| Warrants ($0.70): | 107,500 (Expires March 16, 2019) |
| Warrants ($0.70): | 712,823 (Expires March 21, 2019) |
| Warrants ($0.70): | 700,000 (Expires May 02, 2019) |
| Warrants ($0.70): | 855,193 (Expires June 18, 2019) |
| Warrants ($1.50): | 2,518,600 (Expires April 6, 2020) |
| Trading Symbols: | TSX.V: CVM FRANKFURT: B3BN OTCQB: CRRVF |
| CUSIP #: | 126632108 |
| ISIN #: | CA1266321084 |
| Transfer Agents: | AST Trust Company (Canada)1600 – 1066 West Hastings St. American Stock Transfer & Trust Company, LLC6201 15th Avenue |
| IR Contact: | Brisco Capital Partners Corp.Scott Koyich, President |
CVR Intellectual Property Summary & Legal Comment
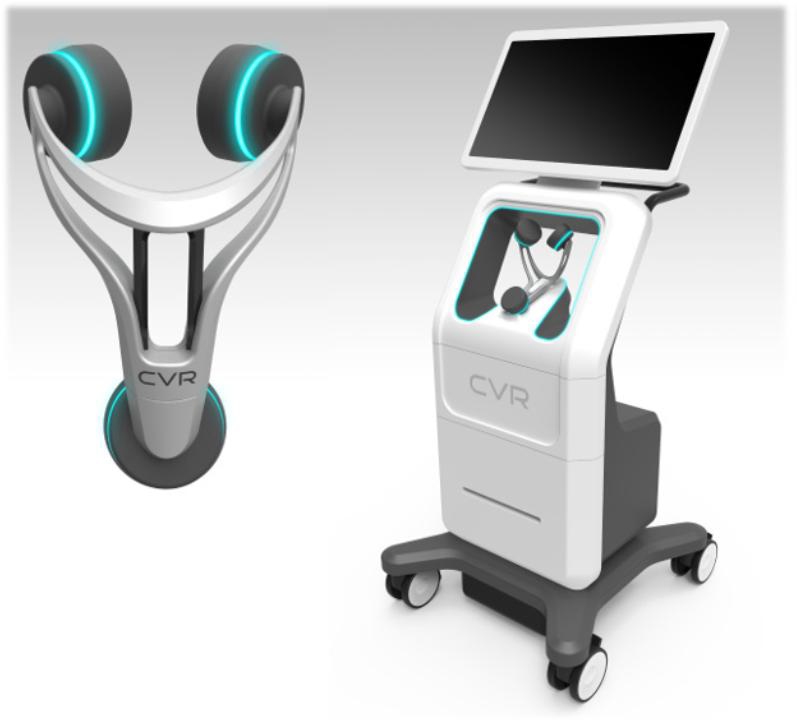
CVR has instituted a comprehensive IP policy to cover several aspects of the CSS device as it prepares to go to market. The IP includes 1 issued US patent, US 9,101,274. An European Counterpart is pending at the moment that parallels the allowed US case. A continuation-in-part, Application No. 14/803,389 is also pending, with a priority date extending back to June 24, 2010.
In June of 2015, CVR file two applications covering the Yoke, utilized for holding the sensors and positioning on a patient, and methods of quantifying and detecting sounds in the carotid artery. Both applications were converted into PCT applications in June of 2016 and now await conversion into the US and other jurisdictions at the end of 2017. These applications have been assigned to a joint venture, but an exclusive license remains with CVR.
In June of 2016, CVR filed five additional provisional patent applications covering several aspects of the CSS device, including additional methods for detecting blockage in a fluid flow vessel, disposable sensing elements (think of the razor/razor blade model), methods for generating clean data for analysis, methods for detecting sounds and determining stenosis, methods for determining appropriate location for detecting fluid flow in the body, and methods for detecting and ablating stenosis.
In December of 2016, CVR filed an additional provisional application directed towards sensing pods that do not need a yoke, in order to position the sensing pods for determining stenosis.
Finally, in January of 2017, CVR filed two design patents, one directed towards the device cart itself, and the second to a version of the Yoke for positioning sensors on a patient.
A summary of these are included in the attached Excel file. These filings are highly confidential and not to be disseminated without authorization by CVR.
CVR continues to develop and expand its IP portfolio as development continues. It is expected that additional applications will be filed to cover final versions of any product that will go to market. Such protection will include patent, design patent, trademark, trade dress, and copyright, where applicable.
CVR also expects that the several pending provisional applications will be converted into PCT applications and that the pending PCT applications (now assigned to a joint venture), will also be converted in due course.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
